0001159036false2026Q112/31http://fasb.org/us-gaap/2025#PrepaidExpenseAndOtherAssetsCurrenthttp://fasb.org/us-gaap/2025#PrepaidExpenseAndOtherAssetsNoncurrenthttp://fasb.org/us-gaap/2025#AccruedLiabilitiesCurrenthttp://fasb.org/us-gaap/2025#OtherLiabilitiesNoncurrenthttp://fasb.org/us-gaap/2025#AccruedLiabilitiesCurrenthttp://fasb.org/us-gaap/2025#OtherLiabilitiesNoncurrent0http://www.halozyme.com/20260331#PropertyPlantAndEquipmentAndOperatingLeaseRightOfUseAssetAfterAccumulatedDepreciationAndAmortizationhttp://www.halozyme.com/20260331#PropertyPlantAndEquipmentAndOperatingLeaseRightOfUseAssetAfterAccumulatedDepreciationAndAmortizationhttp://fasb.org/us-gaap/2025#AccruedLiabilitiesCurrenthttp://fasb.org/us-gaap/2025#AccruedLiabilitiesCurrentP7Y0.01146830.01146830.01785170.0129576439xbrli:sharesiso4217:USDiso4217:USDxbrli:shareshalo:productxbrli:purehalo:segmenthalo:trading_dayhalo:businessDay00011590362026-01-012026-03-3100011590362026-04-3000011590362026-03-3100011590362025-12-310001159036us-gaap:RoyaltyMember2026-01-012026-03-310001159036us-gaap:RoyaltyMember2025-01-012025-03-310001159036us-gaap:ProductMember2026-01-012026-03-310001159036us-gaap:ProductMember2025-01-012025-03-310001159036halo:CollaborativeAgreementsMember2026-01-012026-03-310001159036halo:CollaborativeAgreementsMember2025-01-012025-03-3100011590362025-01-012025-03-3100011590362024-12-3100011590362025-03-310001159036us-gaap:CommonStockMember2025-12-310001159036us-gaap:AdditionalPaidInCapitalMember2025-12-310001159036us-gaap:AccumulatedOtherComprehensiveIncomeMember2025-12-310001159036us-gaap:RetainedEarningsMember2025-12-310001159036us-gaap:AdditionalPaidInCapitalMember2026-01-012026-03-310001159036us-gaap:CommonStockMember2026-01-012026-03-310001159036us-gaap:AccumulatedOtherComprehensiveIncomeMember2026-01-012026-03-310001159036us-gaap:RetainedEarningsMember2026-01-012026-03-310001159036us-gaap:CommonStockMember2026-03-310001159036us-gaap:AdditionalPaidInCapitalMember2026-03-310001159036us-gaap:AccumulatedOtherComprehensiveIncomeMember2026-03-310001159036us-gaap:RetainedEarningsMember2026-03-310001159036us-gaap:CommonStockMember2024-12-310001159036us-gaap:AdditionalPaidInCapitalMember2024-12-310001159036us-gaap:AccumulatedOtherComprehensiveIncomeMember2024-12-310001159036us-gaap:RetainedEarningsMember2024-12-310001159036us-gaap:AdditionalPaidInCapitalMember2025-01-012025-03-310001159036us-gaap:CommonStockMember2025-01-012025-03-310001159036us-gaap:RetainedEarningsMember2025-01-012025-03-310001159036us-gaap:AccumulatedOtherComprehensiveIncomeMember2025-01-012025-03-310001159036us-gaap:CommonStockMember2025-03-310001159036us-gaap:AdditionalPaidInCapitalMember2025-03-310001159036us-gaap:AccumulatedOtherComprehensiveIncomeMember2025-03-310001159036us-gaap:RetainedEarningsMember2025-03-310001159036halo:RocheMember2026-01-012026-03-310001159036halo:JanssenMember2026-01-012026-03-310001159036halo:TakedaMember2026-01-012026-03-310001159036halo:BMSMember2026-01-012026-03-310001159036halo:ArgenxMember2026-01-012026-03-310001159036srt:MinimumMember2026-03-310001159036srt:MaximumMember2026-03-310001159036us-gaap:ForeignExchangeForwardMemberus-gaap:CashFlowHedgingMember2026-01-012026-03-310001159036halo:ElektrofiInc.Member2025-11-182025-11-180001159036halo:ElektrofiInc.Member2025-01-012025-12-310001159036halo:ElektrofiInc.Member2025-11-180001159036halo:ElektrofiInc.Member2025-11-192026-03-310001159036halo:ElektrofiInc.Member2026-03-310001159036halo:ElektrofiInc.Member2026-01-012026-03-310001159036halo:ElektrofiInc.Memberhalo:HyperconDevelopedTechnologyPlatformMember2025-11-182025-11-180001159036halo:ElektrofiInc.Memberhalo:HyperconDevelopedTechnologyPlatformMember2025-11-180001159036halo:ElektrofiInc.Memberus-gaap:CustomerRelationshipsMember2025-11-182025-11-180001159036halo:ElektrofiInc.Memberus-gaap:CustomerRelationshipsMember2025-11-180001159036halo:ElektrofiInc.Memberus-gaap:TradeNamesMember2025-11-182025-11-180001159036halo:ElektrofiInc.Memberus-gaap:TradeNamesMember2025-11-180001159036halo:SurfBioIncMember2025-12-222025-12-220001159036us-gaap:USTreasurySecuritiesMember2026-03-310001159036us-gaap:USTreasurySecuritiesMember2025-12-310001159036us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2026-03-310001159036us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel2Member2026-03-310001159036us-gaap:MoneyMarketFundsMember2026-03-310001159036us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2025-12-310001159036us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel2Member2025-12-310001159036us-gaap:MoneyMarketFundsMember2025-12-310001159036us-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel1Member2026-03-310001159036us-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel2Member2026-03-310001159036us-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel1Member2025-12-310001159036us-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel2Member2025-12-310001159036us-gaap:ForeignExchangeForwardMemberus-gaap:FairValueInputsLevel1Member2026-03-310001159036us-gaap:ForeignExchangeForwardMemberus-gaap:FairValueInputsLevel2Member2026-03-310001159036us-gaap:ForeignExchangeForwardMember2026-03-310001159036us-gaap:ForeignExchangeForwardMemberus-gaap:FairValueInputsLevel1Member2025-12-310001159036us-gaap:ForeignExchangeForwardMemberus-gaap:FairValueInputsLevel2Member2025-12-310001159036us-gaap:ForeignExchangeForwardMember2025-12-310001159036us-gaap:FairValueInputsLevel1Member2026-03-310001159036us-gaap:FairValueInputsLevel2Member2026-03-310001159036us-gaap:FairValueInputsLevel1Member2025-12-310001159036us-gaap:FairValueInputsLevel2Member2025-12-310001159036us-gaap:FairValueInputsLevel3Member2026-03-310001159036us-gaap:FairValueInputsLevel3Member2025-12-310001159036halo:ElektrofiAcquisitionMember2025-11-180001159036halo:ProprietaryProductsSalesMember2026-01-012026-03-310001159036halo:ProprietaryProductsSalesMember2025-01-012025-03-310001159036halo:BulkRHuPH20Member2026-01-012026-03-310001159036halo:BulkRHuPH20Member2025-01-012025-03-310001159036halo:DevicePartneredProductsMember2026-01-012026-03-310001159036halo:DevicePartneredProductsMember2025-01-012025-03-310001159036halo:UpfrontFeesMember2026-01-012026-03-310001159036halo:UpfrontFeesMember2025-01-012025-03-310001159036halo:SalesBasedMilestoneMember2026-01-012026-03-310001159036halo:SalesBasedMilestoneMember2025-01-012025-03-310001159036halo:DeviceLicensingAndDevelopmentMember2026-01-012026-03-310001159036halo:DeviceLicensingAndDevelopmentMember2025-01-012025-03-310001159036halo:LicenseFeesAndEventBasedMember2026-01-012026-03-310001159036halo:OthercollaboratorsMember2026-03-310001159036us-gaap:ProductMember2026-03-3100011590362026-04-012026-03-310001159036halo:DevicePartneredProductsMember2026-03-310001159036halo:DevicePartneredProductsMember2025-12-310001159036halo:CollaborativeAgreementsMember2026-03-310001159036halo:CollaborativeAgreementsMember2025-12-310001159036us-gaap:RoyaltyMember2026-03-310001159036us-gaap:RoyaltyMember2025-12-310001159036halo:ProductSalesOtherMember2026-03-310001159036halo:ProductSalesOtherMember2025-12-310001159036halo:OtherReceivableMember2026-03-310001159036halo:OtherReceivableMember2025-12-310001159036halo:ResearchEquipmentMember2026-03-310001159036halo:ResearchEquipmentMember2025-12-310001159036halo:ManufacturingEquipmentMember2026-03-310001159036halo:ManufacturingEquipmentMember2025-12-310001159036halo:ComputerAndOfficeEquipmentMember2026-03-310001159036halo:ComputerAndOfficeEquipmentMember2025-12-310001159036us-gaap:SoftwareAndSoftwareDevelopmentCostsMember2026-03-310001159036us-gaap:SoftwareAndSoftwareDevelopmentCostsMember2025-12-310001159036us-gaap:LeaseholdImprovementsMember2026-03-310001159036us-gaap:LeaseholdImprovementsMember2025-12-310001159036srt:MinimumMember2026-01-012026-03-310001159036srt:MaximumMember2026-01-012026-03-310001159036halo:AutoInjectorTechnologyPlatformMember2026-01-012026-03-310001159036halo:AutoInjectorTechnologyPlatformMember2026-03-310001159036halo:XYOSTEDProprietaryProductMember2026-01-012026-03-310001159036halo:XYOSTEDProprietaryProductMember2026-03-310001159036halo:HyperconDevelopedTechnologyPlatformMember2026-01-012026-03-310001159036halo:HyperconDevelopedTechnologyPlatformMember2026-03-310001159036us-gaap:CustomerRelationshipsMember2026-01-012026-03-310001159036us-gaap:CustomerRelationshipsMember2026-03-310001159036halo:TLANDOProductRightsMember2026-01-012026-03-310001159036halo:TLANDOProductRightsMember2026-03-310001159036halo:AutoInjectorTechnologyPlatformMember2025-01-012025-12-310001159036halo:AutoInjectorTechnologyPlatformMember2025-12-310001159036halo:XYOSTEDProprietaryProductMember2025-01-012025-12-310001159036halo:XYOSTEDProprietaryProductMember2025-12-310001159036halo:HyperconDevelopedTechnologyPlatformMember2025-01-012025-12-310001159036halo:HyperconDevelopedTechnologyPlatformMember2025-12-310001159036us-gaap:CustomerRelationshipsMember2025-01-012025-12-310001159036us-gaap:CustomerRelationshipsMember2025-12-310001159036halo:TLANDOProductRightsMember2025-01-012025-12-310001159036halo:TLANDOProductRightsMember2025-12-310001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:ConvertibleDebtMember2025-11-300001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:ConvertibleDebtMember2025-11-012025-11-300001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:DebtInstrumentRedemptionPeriodOneMemberus-gaap:ConvertibleDebtMember2025-11-012025-11-300001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:DebtInstrumentRedemptionPeriodTwoMemberus-gaap:ConvertibleDebtMember2025-11-012025-11-300001159036halo:A0.875ConvertibleSeniorNotesDue2032Member2025-11-300001159036halo:A0.875ConvertibleSeniorNotesDue2032Member2025-11-012025-11-300001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:ConvertibleDebtMember2025-11-300001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:ConvertibleDebtMember2025-11-012025-11-300001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:DebtInstrumentRedemptionPeriodOneMemberus-gaap:ConvertibleDebtMember2025-11-012025-11-300001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:DebtInstrumentRedemptionPeriodTwoMemberus-gaap:ConvertibleDebtMember2025-11-012025-11-300001159036halo:A0.00ConvertibleSeniorNotesDue2031Member2025-11-300001159036halo:A0.00ConvertibleSeniorNotesDue2031Member2025-11-012025-11-300001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:ConvertibleDebtMember2022-08-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:ConvertibleDebtMember2022-08-012022-08-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:DebtInstrumentRedemptionPeriodOneMemberus-gaap:ConvertibleDebtMember2022-08-012022-08-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:DebtInstrumentRedemptionPeriodTwoMemberus-gaap:ConvertibleDebtMember2022-08-012022-08-310001159036halo:A100ConvertibleSeniorNotesDue2028Member2025-11-012025-11-300001159036halo:A100ConvertibleSeniorNotesDue2028Member2022-08-310001159036halo:A100ConvertibleSeniorNotesDue2028Member2022-08-012022-08-310001159036halo:A100ConvertibleSeniorNotesDue2028Member2022-08-1500011590362022-08-012022-08-310001159036halo:A025ConvertibleSeniorNotesDue2027Memberus-gaap:ConvertibleDebtMember2021-03-310001159036halo:A025ConvertibleSeniorNotesDue2027Member2021-03-012021-03-310001159036halo:A025ConvertibleSeniorNotesDue2027Memberus-gaap:ConvertibleDebtMember2021-03-012021-03-310001159036halo:A025ConvertibleSeniorNotesDue2027Memberus-gaap:DebtInstrumentRedemptionPeriodOneMemberus-gaap:ConvertibleDebtMember2021-03-012021-03-310001159036halo:A025ConvertibleSeniorNotesDue2027Memberus-gaap:DebtInstrumentRedemptionPeriodTwoMemberus-gaap:ConvertibleDebtMember2021-03-012021-03-310001159036halo:A025ConvertibleSeniorNotesDue2027Member2025-11-012025-11-300001159036halo:A0.25ConvertibleSeniorNotesDue2027Memberus-gaap:ConvertibleDebtMember2026-03-310001159036halo:A0.25ConvertibleSeniorNotesDue2027Memberus-gaap:ConvertibleDebtMember2025-12-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:ConvertibleDebtMember2026-03-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:ConvertibleDebtMember2025-12-310001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:ConvertibleDebtMember2026-03-310001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:ConvertibleDebtMember2025-12-310001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:ConvertibleDebtMember2026-03-310001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:ConvertibleDebtMember2025-12-310001159036us-gaap:ConvertibleDebtMember2026-03-310001159036us-gaap:ConvertibleDebtMember2025-12-310001159036halo:A0.25ConvertibleSeniorNotesDue2027Memberus-gaap:ConvertibleDebtMember2026-01-012026-03-310001159036halo:A0.25ConvertibleSeniorNotesDue2027Memberus-gaap:ConvertibleDebtMember2025-01-012025-03-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:ConvertibleDebtMember2026-01-012026-03-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:ConvertibleDebtMember2025-01-012025-03-310001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:ConvertibleDebtMember2026-01-012026-03-310001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:ConvertibleDebtMember2025-01-012025-03-310001159036us-gaap:ConvertibleDebtMember2026-01-012026-03-310001159036us-gaap:ConvertibleDebtMember2025-01-012025-03-310001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:ConvertibleDebtMember2026-01-012026-03-310001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:ConvertibleDebtMember2025-01-012025-03-310001159036halo:A0.25ConvertibleSeniorNotesDue2027Memberus-gaap:ConvertibleDebtMember2025-03-310001159036halo:A100ConvertibleSeniorNotesDue2028Memberus-gaap:ConvertibleDebtMember2025-03-310001159036halo:A0.00ConvertibleSeniorNotesDue2031Memberus-gaap:ConvertibleDebtMember2025-03-310001159036halo:A0.875ConvertibleSeniorNotesDue2032Memberus-gaap:ConvertibleDebtMember2025-03-310001159036us-gaap:RevolvingCreditFacilityMemberhalo:CreditAgreementMember2025-11-300001159036us-gaap:RevolvingCreditFacilityMemberus-gaap:FederalFundsEffectiveSwapRateMemberhalo:CreditAgreementMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberus-gaap:SecuredOvernightFinancingRateSofrMemberhalo:CreditAgreementMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberus-gaap:BaseRateMemberhalo:CreditAgreementMembersrt:MinimumMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberus-gaap:BaseRateMemberhalo:CreditAgreementMembersrt:MaximumMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberus-gaap:SecuredOvernightFinancingRateSofrMemberhalo:CreditAgreementMembersrt:MinimumMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberus-gaap:SecuredOvernightFinancingRateSofrMemberhalo:CreditAgreementMembersrt:MaximumMember2025-11-012025-11-300001159036halo:VariableRateComponentTwoMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:SecuredOvernightFinancingRateSofrMemberhalo:CreditAgreementMembersrt:MinimumMember2025-11-012025-11-300001159036halo:VariableRateComponentTwoMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:SecuredOvernightFinancingRateSofrMemberhalo:CreditAgreementMembersrt:MaximumMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberhalo:CreditAgreementMembersrt:MinimumMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberhalo:CreditAgreementMembersrt:MaximumMember2025-11-012025-11-300001159036us-gaap:RevolvingCreditFacilityMemberhalo:CreditAgreementMember2026-03-310001159036halo:CreditAgreementMemberhalo:TermLoanFacilityMember2026-03-310001159036us-gaap:ResearchAndDevelopmentExpenseMember2026-01-012026-03-310001159036us-gaap:ResearchAndDevelopmentExpenseMember2025-01-012025-03-310001159036us-gaap:SellingGeneralAndAdministrativeExpensesMember2026-01-012026-03-310001159036us-gaap:SellingGeneralAndAdministrativeExpensesMember2025-01-012025-03-310001159036us-gaap:EmployeeStockOptionMember2026-01-012026-03-310001159036us-gaap:EmployeeStockOptionMember2025-01-012025-03-310001159036halo:RSURSAandPRSUawardsMember2026-01-012026-03-310001159036halo:RSURSAandPRSUawardsMember2025-01-012025-03-310001159036srt:MinimumMember2025-01-012025-03-310001159036srt:MaximumMember2025-01-012025-03-310001159036halo:A2021ESPPPlanMemberus-gaap:EmployeeStockMember2021-02-012021-02-280001159036halo:A2021ESPPPlanMemberus-gaap:EmployeeStockMember2021-02-280001159036halo:A2021ESPPPlanMember2026-03-310001159036us-gaap:EmployeeStockOptionMember2026-03-310001159036us-gaap:RestrictedStockUnitsRSUMember2026-03-310001159036us-gaap:RestrictedStockUnitsRSUMember2026-01-012026-03-310001159036us-gaap:PerformanceSharesMember2026-03-310001159036us-gaap:PerformanceSharesMember2026-01-012026-03-310001159036us-gaap:EmployeeStockMember2026-03-310001159036us-gaap:EmployeeStockMember2026-01-012026-03-310001159036halo:A2024CapitalReturnProgramMember2024-02-290001159036halo:A2024CapitalReturnProgramMember2024-01-012024-12-310001159036halo:A2024CapitalReturnProgramTrancheTwoMember2025-05-310001159036halo:A2024CapitalReturnProgramTrancheTwoMember2025-05-012025-06-300001159036halo:A2024CapitalReturnProgramTrancheThreeMember2025-06-300001159036halo:A2024CapitalReturnProgramTrancheThreeMember2025-06-012025-09-300001159036halo:RestrictStockUnitsPerformanceSharesAndESPPMember2026-01-012026-03-310001159036halo:RestrictStockUnitsPerformanceSharesAndESPPMember2025-01-012025-03-310001159036us-gaap:ConvertibleDebtSecuritiesMember2026-01-012026-03-310001159036us-gaap:ConvertibleDebtSecuritiesMember2025-01-012025-03-310001159036halo:MaheshKrishnanMember2026-01-012026-03-310001159036halo:MaheshKrishnanMember2026-03-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-Q
(Mark One)
| | | | | |
☒ | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended March 31, 2026
OR
| | | | | |
☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number 001-32335
_______________________________________
HALOZYME THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
_______________________________________
| | | | | | | | |
| Delaware | | 88-0488686 |
| (State or other jurisdiction of incorporation or organization) | | (I.R.S. Employer Identification No.) |
| |
| 12390 El Camino Real | | 92130 |
| San Diego | | (Zip Code) |
| California | | |
| (Address of principal executive offices) | | |
(858) 794-8889
(Registrant’s telephone number, including area code)
Not Applicable
(Former name, former address and former fiscal year, if changed since last report)
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | | | |
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | | |
| Common Stock, $0.001 par value | HALO | The Nasdaq Stock Market LLC | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes x No ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.:
| | | | | | | | | | | |
| Large accelerated filer | ☒ | Accelerated filer | ☐ |
Non-accelerated filer | ☐ | Smaller reporting company | ☐ |
| | Emerging growth company | ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No x
The number of outstanding shares (in thousands) of the registrant’s common stock, par value $0.001 per share, was 118,613 as of April 30, 2026.
HALOZYME THERAPEUTICS, INC.
TABLE OF CONTENTS
| | | | | | | | | | | | | | |
| | | | Page | |
| | | | |
| |
| Item 1. | | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| Item 2. | | | | |
| Item 3. | | | | |
| Item 4. | | | | |
| |
| |
| Item 1. | | | | |
| Item 1A. | | | | |
| Item 2. | | | | |
| Item 3. | | | | |
| Item 4. | | | | |
| Item 5. | | | | |
| Item 6. | | | | |
| | | | |
| | | | |
PART I — FINANCIAL INFORMATION
Item 1. Financial Statements
HALOZYME THERAPEUTICS, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
(Unaudited)
(In thousands, except per share amounts)
| | | | | | | | | | | | | | |
| | March 31,
2026 | | December 31,
2025 |
| ASSETS | | | | |
| Current assets | | | | |
| Cash and cash equivalents | | $ | 309,749 | | | $ | 133,820 | |
| Marketable securities, available-for-sale | | 8,873 | | | 9,000 | |
| Accounts receivable, net and contract assets | | 457,989 | | | 441,273 | |
| Inventories | | 155,467 | | | 176,475 | |
| Prepaid expenses and other current assets | | 70,958 | | | 64,639 | |
| Total current assets | | 1,003,036 | | | 825,207 | |
| Property and equipment, net | | 82,230 | | | 82,137 | |
| Prepaid expenses and other assets | | 50,915 | | | 53,551 | |
| Goodwill | | 582,323 | | | 580,360 | |
| Intangible assets, net | | 951,955 | | | 981,467 | |
| | | | |
| Restricted cash | | 2,260 | | | 2,601 | |
| Total assets | | $ | 2,672,719 | | | $ | 2,525,323 | |
| | | | |
| LIABILITIES AND STOCKHOLDERS’ EQUITY | | | | |
| Current liabilities | | | | |
| Accounts payable | | $ | 14,098 | | | $ | 20,899 | |
| Accrued expenses | | 140,369 | | | 156,193 | |
| | | | |
| Current portion of long-term debt, net | | 208,743 | | | — | |
| Total current liabilities | | 363,210 | | | 177,092 | |
| | | | |
| Long-term debt, net | | 1,935,896 | | | 2,142,630 | |
| Other long-term liabilities | | 106,448 | | | 113,863 | |
| Deferred tax liabilities, net | | 47,529 | | | 42,924 | |
| | | | |
| Total liabilities | | 2,453,083 | | | 2,476,509 | |
Commitments and contingencies (Note 11) | | | | |
| Stockholders’ equity | | | | |
Preferred stock - $0.001 par value; 20,000 shares authorized; no shares issued and outstanding | | — | | | — | |
Common stock - $0.001 par value; 300,000 shares authorized; 118,523 and 117,782 shares issued and outstanding as of March 31, 2026 and December 31, 2025, respectively | | 118 | | | 118 | |
| Additional paid-in capital | | 27,386 | | | 12,002 | |
Accumulated other comprehensive loss | | (12,703) | | | (18,092) | |
| Retained earnings | | 204,835 | | | 54,786 | |
| Total stockholders’ equity | | 219,636 | | | 48,814 | |
| Total liabilities and stockholders’ equity | | $ | 2,672,719 | | | $ | 2,525,323 | |
See accompanying notes to condensed consolidated financial statements.
HALOZYME THERAPEUTICS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF INCOME
(Unaudited)
(In thousands, except per share amounts)
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
| | | 2026 | | 2025 | | | | |
| Revenues | | | | | | | | |
| Royalties | | $ | 240,681 | | | $ | 168,192 | | | | | |
| Product sales, net | | 130,424 | | | 78,041 | | | | | |
| Revenues under collaborative agreements | | 5,603 | | | 18,628 | | | | | |
| Total revenues | | 376,708 | | | 264,861 | | | | | |
| Operating expenses | | | | | | | | |
| Cost of sales | | 79,238 | | | 48,403 | | | | | |
| Amortization of intangibles | | 29,512 | | | 17,762 | | | | | |
| Research and development | | 25,560 | | | 14,799 | | | | | |
| Selling, general and administrative | | 57,881 | | | 42,362 | | | | | |
| | | | | | | | |
| Total operating expenses | | 192,191 | | | 123,326 | | | | | |
| Operating income | | 184,517 | | | 141,535 | | | | | |
| Other income (expense) | | | | | | | | |
Investment and other income, net | | 1,318 | | | 6,818 | | | | | |
| | | | | | | | |
| | | | | | | | |
| Interest expense | | (5,508) | | | (4,525) | | | | | |
Income before income tax expense | | 180,327 | | | 143,828 | | | | | |
| Income tax expense | | 30,278 | | | 25,733 | | | | | |
| Net income | | $ | 150,049 | | | $ | 118,095 | | | | | |
| | | | | | | | |
| Earnings per share | | | | | | | | |
| Basic | | $ | 1.27 | | | $ | 0.96 | | | | | |
| Diluted | | $ | 1.22 | | | $ | 0.93 | | | | | |
| | | | | | | | |
| Weighted average common shares outstanding | | | | | | | | |
| Basic | | 118,144 | | | 123,215 | | | | | |
| Diluted | | 122,875 | | | 126,644 | | | | | |
See accompanying notes to condensed consolidated financial statements.
HALOZYME THERAPEUTICS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF COMPREHENSIVE INCOME
(Unaudited)
(In thousands)
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
| | 2026 | | 2025 | | | | |
| Net income | | $ | 150,049 | | | $ | 118,095 | | | | | |
Other comprehensive income | | | | | | | | |
Unrealized (loss) gain on marketable securities, net | | (3) | | | 189 | | | | | |
| Foreign currency translation adjustment | | 45 | | | — | | | | | |
| | | | | | | | |
Unrealized gain (loss) on derivative instruments, net | | 4,022 | | | (7,223) | | | | | |
Realized loss (gain) on derivative instruments, net | | 1,325 | | | (208) | | | | | |
| Comprehensive income | | $ | 155,438 | | | $ | 110,853 | | | | | |
See accompanying notes to condensed consolidated financial statements.
HALOZYME THERAPEUTICS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(Unaudited)
(In thousands)
| | | | | | | | | | | | | | |
| | Three Months Ended |
| | March 31, |
| | | 2026 | | 2025 |
| Operating activities | | | | |
| Net income | | $ | 150,049 | | | $ | 118,095 | |
Adjustments to reconcile net income to net cash provided by operating activities: | | | | |
| Share-based compensation | | 16,637 | | | 10,673 | |
| Depreciation and amortization | | 4,221 | | | 2,687 | |
| Amortization of intangible assets | | 29,512 | | | 17,762 | |
| Amortization of debt discount | | 2,245 | | | 1,846 | |
| | | | |
Accretion of premium on marketable securities, net | | (30) | | | (1,709) | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
Lease payments recognized | | 214 | | | 253 | |
| | | | |
| | | | |
| | | | |
| Deferred income taxes | | 3,428 | | | (2,858) | |
| | | | |
Changes in operating assets and liabilities | | | | |
Accounts receivable, net and contract assets | | (19,547) | | | 3,834 | |
| Inventories | | 27,379 | | | (586) | |
| Prepaid expenses and other assets | | (9,108) | | | (4,995) | |
| Accounts payable and accrued expenses | | (24,922) | | | 9,219 | |
| Net cash provided by operating activities | | 180,078 | | | 154,221 | |
| Investing activities | | | | |
| Purchases of marketable securities | | (8,846) | | | (139,639) | |
| Proceeds from sales and maturities of marketable securities | | 9,000 | | | 50,167 | |
Proceeds from working capital adjustments on acquisition | | 710 | | | — | |
| Purchases of property and equipment | | (3,741) | | | (950) | |
| | | | |
Net cash used in investing activities | | (2,877) | | | (90,422) | |
| Financing activities | | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
Taxes paid related to net share settlement, net of proceeds from issuance of common stock under equity incentive plans | | (1,613) | | | (3,321) | |
Net cash used in financing activities | | (1,613) | | | (3,321) | |
Net increase in cash and cash equivalents | | 175,588 | | | 60,478 | |
Cash and cash equivalents at beginning of period | | 136,421 | | | 115,850 | |
Cash and cash equivalents at end of period | | $ | 312,009 | | | $ | 176,328 | |
| | | | |
| Reconciliation of cash, cash equivalents and restricted cash to the condensed consolidated balance sheets | | | | |
| Cash and cash equivalents | | $ | 309,749 | | | $ | 176,328 | |
| Restricted cash | | 2,260 | | | — | |
| Total cash, cash equivalents and restricted cash | | $ | 312,009 | | | $ | 176,328 | |
| | | | |
Supplemental disclosure of non-cash investing and financing activities | | | | |
| Amounts accrued for purchases of property and equipment | | $ | 1,747 | | | $ | 488 | |
| Right-of-use assets obtained in exchange for lease obligation | | $ | 170 | | | $ | 95 | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
See accompanying notes to condensed consolidated financial statements.
HALOZYME THERAPEUTICS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(Unaudited)
(in thousands)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended March 31, 2026 |
| | | Common Stock | | Additional
Paid-In
Capital | | Accumulated
Other
Comprehensive Loss | | Retained Earnings | | Total
Stockholders’
Equity |
| | | Shares | | Amount | |
| BALANCE AS OF DECEMBER 31, 2025 | | 117,782 | | | $ | 118 | | | $ | 12,002 | | | $ | (18,092) | | | $ | 54,786 | | | $ | 48,814 | |
| | | | | | | | | | | | |
| Share-based compensation expense | | — | | | — | | | 16,997 | | | — | | | — | | | 16,997 | |
| | | | | | | | | | | | |
| Issuance of common stock pursuant to exercise of stock options and vesting of restricted stock and performance stock units, net | | 741 | | | — | | | (1,613) | | | — | | | — | | | (1,613) | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
Other comprehensive income | | — | | | — | | | — | | | 5,389 | | | — | | | 5,389 | |
| Net income | | — | | | — | | | — | | | — | | | 150,049 | | | 150,049 | |
| BALANCE AS OF MARCH 31, 2026 | | 118,523 | | | $ | 118 | | | $ | 27,386 | | | $ | (12,703) | | | $ | 204,835 | | | $ | 219,636 | |
| | | | | | | | | | | | |
| | |
| | | | | | | | | | |
| | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | Three Months Ended March 31, 2025 |
| | | Common Stock | | Additional
Paid-In
Capital | | Accumulated
Other
Comprehensive Income (Loss) | | Retained Earnings | | Total
Stockholders’
Equity |
| | | Shares | | Amount | |
| BALANCE AS OF DECEMBER 31, 2024 | | 123,138 | | | $ | 123 | | | $ | — | | | $ | 3,829 | | | $ | 359,869 | | | $ | 363,821 | |
| | | | | | | | | | | | |
| Share-based compensation expense | | — | | | — | | | 10,673 | | | — | | | — | | | 10,673 | |
| | | | | | | | | | | | |
| Issuance of common stock pursuant to exercise of stock options and vesting of restricted stock and performance stock units, net | | 484 | | | — | | | (3,321) | | | — | | | — | | | (3,321) | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| Repurchase of common stock | | (452) | | | — | | | 244 | | | | | — | | | 244 | |
| | | | | | | | | | | | |
Other comprehensive loss | | — | | | — | | | — | | | (7,242) | | | — | | | (7,242) | |
| Net income | | — | | | — | | | — | | | — | | | 118,095 | | | 118,095 | |
| BALANCE AS OF MARCH 31, 2025 | | 123,170 | | | $ | 123 | | | $ | 7,596 | | | $ | (3,413) | | | $ | 477,964 | | | $ | 482,270 | |
| | | | | | | | | | | | |
| | |
| | | | | | | | | | |
| | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
See accompanying notes to condensed consolidated financial statements.
HALOZYME THERAPEUTICS, INC.
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
(Unaudited)
1. Organization and Business
Halozyme Therapeutics, Inc. is a biopharmaceutical company advancing disruptive solutions to improve patient experiences and outcomes for emerging and established therapies.
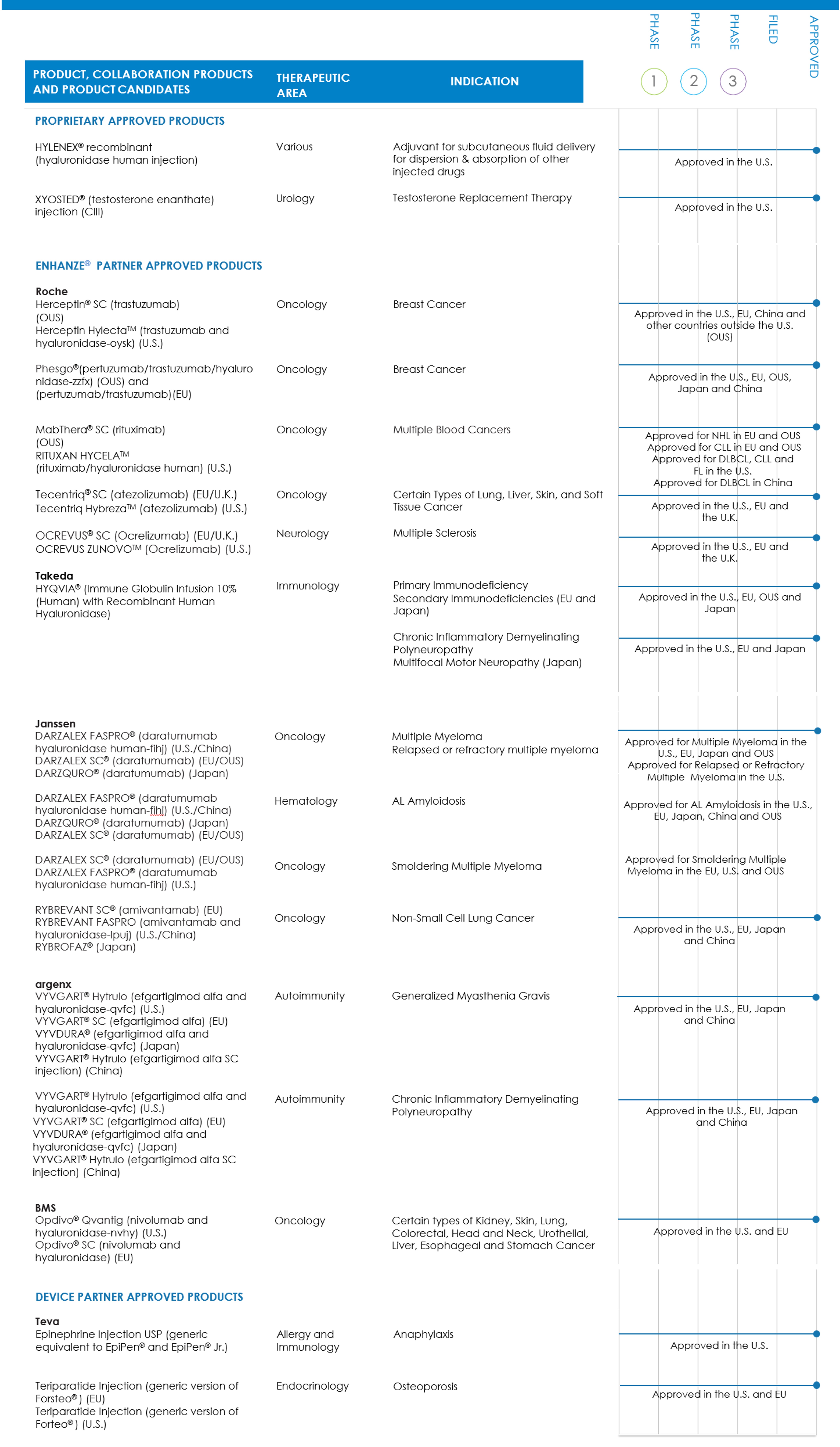
As the innovators of ENHANZE® drug delivery technology (“ENHANZE”) with our proprietary enzyme, rHuPH20, our commercially validated solution is used to facilitate the subcutaneous (“SC”) delivery of injected drugs and fluids, with the goal of improving the patient experience with rapid SC delivery and reduced treatment burden. We license our technology to biopharmaceutical companies to collaboratively develop products that combine ENHANZE with our partners’ proprietary compounds. We are also developing partner products with Hypercon™ drug delivery technology (“Hypercon technology”) and developing Surf Bio’s drug delivery technology to expand the breadth of our drug delivery technology portfolio. Hypercon technology is an innovative microparticle technology that has been demonstrated in non-clinical testing to enable hyperconcentration of drugs and biologics and reduce the injection volume for the same dosage and potentially expanding opportunities for at-home and health care provider administration. The Surf Bio hyperconcentration technology has similarly been demonstrated in non-clinical studies to achieve biologic concentrations of up to 500 mg/mL, for potential delivery in a single auto-injector shot for at-home or in a health care provider’s office use. We also develop, manufacture and commercialize, for ourselves or with our partners, drug-device combination products using our advanced auto-injector technologies that are designed to provide commercial or functional advantages such as improved convenience, reliability and tolerability, and enhanced patient comfort and adherence.
Our ENHANZE partners’ approved products and product candidates are based on rHuPH20, our patented recombinant human hyaluronidase enzyme. rHuPH20 works by breaking down hyaluronan, a naturally occurring carbohydrate that is a major component of the extracellular matrix of the SC space. This temporarily reduces the barrier to bulk fluid flow allowing for improved and more rapid SC delivery of high dose, high volume injectable biologics, such as monoclonal antibodies and other large therapeutic molecules, as well as small molecules and fluids. We refer to the application of rHuPH20 to facilitate the delivery of other drugs or fluids as ENHANZE. We license our ENHANZE technology to form collaborations with biopharmaceutical companies that develop and/or market drugs requiring or benefiting from injection via the SC route of administration. In the development of proprietary intravenous (“IV”) drugs combined with our ENHANZE technology, data have been generated supporting the potential for ENHANZE to reduce patient treatment burden, as a result of shorter duration of SC administration with ENHANZE compared to IV administration. ENHANZE may enable fixed-dose SC dosing compared to weight-based dosing typically required for IV administration, extend the dosing interval for drugs that are already administered subcutaneously and potentially allow for lower rates of infusion-related reactions. ENHANZE may enable more flexible treatment options such as home administration by a healthcare professional or potentially the patient or caregiver. Lastly, certain proprietary drugs co-formulated with ENHANZE have been granted additional exclusivity, extending the patent life of the product beyond the patent expiry of the proprietary IV drug.
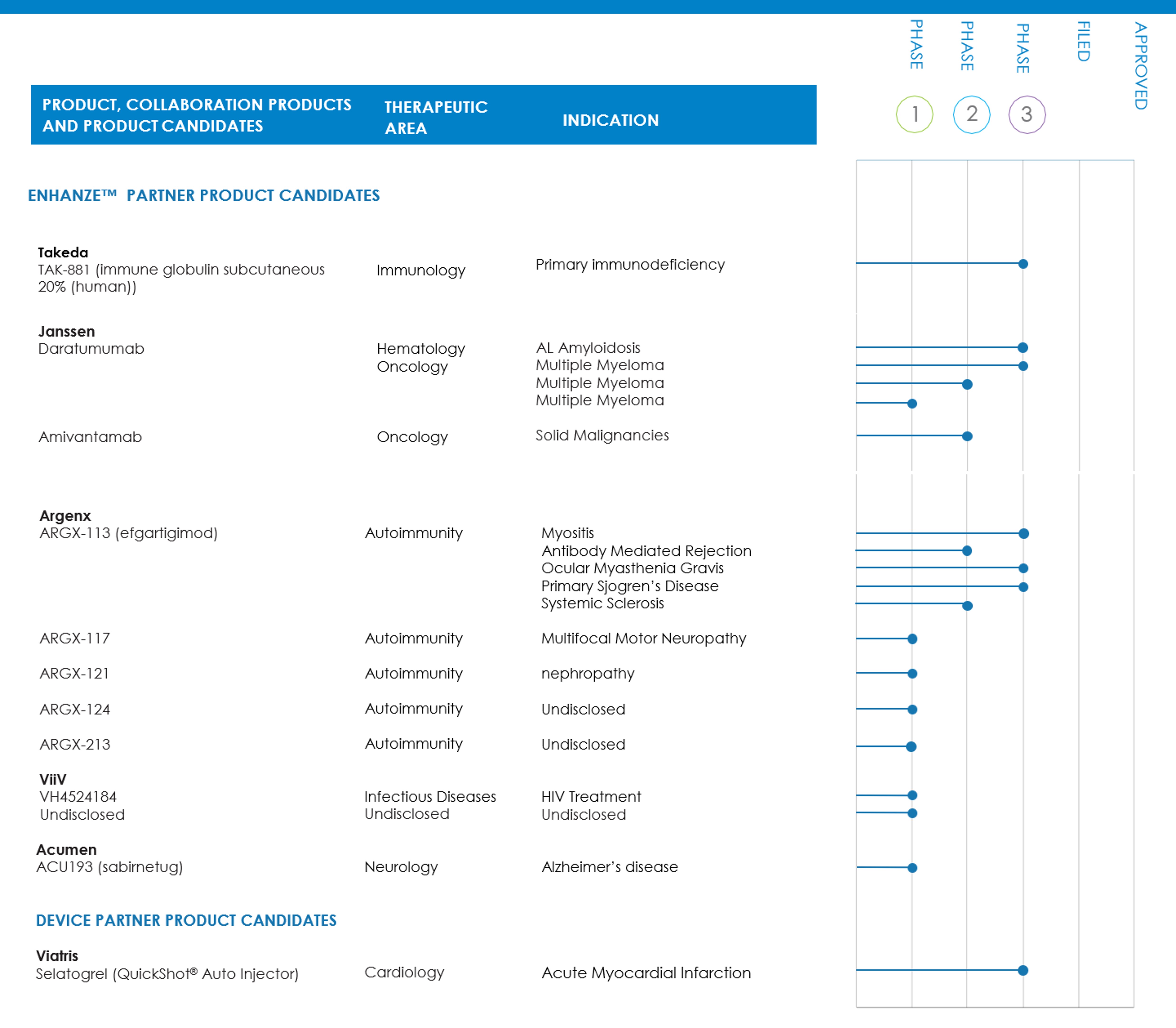
We currently have ENHANZE collaborations and licensing agreements with F. Hoffmann-La Roche, Ltd. and Hoffmann-La Roche, Inc. (“Roche”), Takeda Pharmaceuticals International AG and Baxalta US Inc. (“Takeda”), Pfizer Inc. (“Pfizer”), Janssen Biotech, Inc. (“Janssen”), AbbVie, Inc. (“AbbVie”), Eli Lilly and Company (“Lilly”), Bristol Myers Squibb Company (“BMS”), argenx BVBA (“argenx”), ViiV Healthcare (the global specialist HIV Company majority owned by GlaxoSmithKline) (“ViiV”), Chugai Pharmaceutical Co., Ltd. (“Chugai”), Acumen Pharmaceuticals, Inc. (“Acumen”), Merus N.V. (“Merus”), Skye Bioscience, Inc. (“Skye Bioscience”) and GlaxoSmithKline plc (“GSK”). In addition to receiving upfront licensing fees from our ENHANZE collaborations, we are entitled to receive event and sales-based milestone payments, revenues from the sale of bulk rHuPH20 and royalties from commercial sales of approved partner products co-formulated with ENHANZE. We currently earn royalties from the sales of ten commercial products including sales of five commercial products from the Roche collaboration, two commercial products from the Janssen collaboration and one commercial product from each of the Takeda, argenx and BMS collaborations.
Through our recent acquisition of Elektrofi, Inc. (“Elektrofi”), subsequently renamed Halozyme Hypercon, Inc. (“Hypercon”), we have Hypercon collaboration and license agreements with Janssen, Lilly, argenx, Vertex Pharmaceuticals Incorporated (“Vertex”) and Oruka Therapeutics, Inc. (“Oruka”). In addition to receiving upfront license fees from our Hypercon collaborations, we are entitled to receive event and sales-based milestone payments and royalties from commercial sales for approved partner products co-formulated with Hypercon.
We have commercialized auto-injector products with Teva Pharmaceutical Industries, Ltd. (“Teva”). We have development programs including our auto-injectors with McDermott Laboratories Limited, an affiliate of Viatris Inc. (“Viatris”).
Our commercial portfolio of proprietary products includes Hylenex®, utilizing rHuPH20, and XYOSTED®, utilizing our auto-injector technology.
Except where specifically noted or the context otherwise requires, references to “Halozyme,” “the Company,” “we,” “our,” and “us” in these notes to our condensed consolidated financial statements refer to Halozyme Therapeutics, Inc. and each of its directly and indirectly wholly owned subsidiaries as disclosed in Note 2, Summary of Significant Accounting Policies.
2. Summary of Significant Accounting Policies
Basis of Presentation
The accompanying interim unaudited condensed consolidated financial statements have been prepared for purposes of and in accordance with United States generally accepted accounting principles (“U.S. GAAP”) and with the rules and regulations of the U.S. Securities and Exchange Commission (“SEC”) related to a quarterly report on Form 10-Q. Accordingly, they do not include all of the information and disclosures required by U.S. GAAP for a complete set of financial statements. These condensed consolidated financial statements and notes thereto should be read in conjunction with the audited consolidated financial statements and notes thereto included in our Annual Report on Form 10-K for the year ended December 31, 2025, filed with the SEC on February 17, 2026. The unaudited financial information for the interim periods presented herein reflects all adjustments which, in the opinion of management, are necessary for a fair presentation of the financial condition and results of operations for the periods presented, with such adjustments consisting only of normal recurring adjustments. Operating results for interim periods are not necessarily indicative of the operating results for an entire fiscal year.
The accompanying condensed consolidated financial statements include the accounts of Halozyme Therapeutics, Inc. and our wholly owned subsidiaries, Halozyme, Inc., Antares Pharma, Inc., Antares Pharma, Inc.’s two wholly-owned Swiss subsidiaries, Antares Pharma IPL AG and Antares Pharma GmbH, Halozyme Hypercon, Inc., Halozyme Hypercon, Inc.’s wholly-owned subsidiary, Elektrofi Security Corp., and Halozyme Surf Bio, Inc. All intercompany accounts and transactions have been eliminated.
Use of Estimates
The preparation of condensed consolidated financial statements in conformity with U.S. GAAP requires us to make estimates and assumptions that affect the amounts reported in our condensed consolidated financial statements and accompanying notes. On an ongoing basis, we evaluate our estimates and judgments, which are based on historical and anticipated results and trends and on various other assumptions that we believe to be reasonable under the circumstances. By their nature, estimates are subject to an inherent degree of uncertainty and, as such, actual results may differ from our estimates.
Cash Equivalents and Marketable Securities
Cash equivalents consist of highly liquid investments, readily convertible to cash, which mature within 90 days or less from the date of purchase. As of March 31, 2026, our cash and cash equivalents consisted of money market funds, bank certificate of deposits and demand deposits at commercial banks.
Marketable securities are investments with original maturities of more than 90 days from the date of purchase that are specifically identified to fund current operations. Marketable securities are considered available-for-sale. These investments are classified as current assets, even though the stated maturity date may be one year or more beyond the current balance sheet date which reflects management’s intention to use the proceeds from the sale of these investments to fund our operations, as necessary. Such available-for-sale investments are carried at fair value with unrealized gains and losses recorded in other comprehensive income and included as a separate component of stockholders’ equity. The cost of marketable securities is adjusted for amortization of premiums or accretion of discounts to maturity, and such amortization or accretion is included in investment and other income, net in our condensed consolidated statements of income. We use the specific identification method for calculating realized gains and losses on marketable securities sold. None of the realized gains and losses and declines in value that were judged to be as a result of credit loss on marketable securities, if any, are included in investment and other income, net in our condensed consolidated statements of income.
Restricted Cash
Under the terms of the leases of our facilities and other agreements, we are required to maintain letters of credit as security deposits during the terms of such leases. At March 31, 2026, restricted cash of $2.3 million was pledged as collateral for the letters of credit and other agreements.
Fair Value of Financial Instruments
The authoritative guidance for fair value measurements establishes a three-tier fair value hierarchy, which prioritizes the inputs used in measuring fair value. These tiers include: Level 1, defined as observable inputs such as quoted prices in active markets; Level 2, defined as inputs other than quoted prices in active markets that are either directly or indirectly observable; and Level 3, defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions.
Our financial instruments include cash equivalents, available-for-sale marketable securities, accounts receivable, prepaid expenses and other assets, accounts payable, accrued expenses, long-term debt and contingent liabilities. Fair value estimates of these instruments are made at a specific point in time based on relevant market information. These estimates may be subjective in nature and involve uncertainties and matters of significant judgment and therefore, cannot be determined with precision. The carrying amount of cash equivalents, accounts receivable, prepaid expenses and other assets, accounts payable and accrued expenses are generally considered to be representative of their respective fair values because of the short-term nature of those instruments.
As of March 31, 2026, our available-for-sale marketable securities consisted of United States (“U.S.”) Treasury securities, and were measured at fair value using Level 1. Level 2 financial instruments are valued using market prices on less active markets and proprietary pricing valuation models with observable inputs, including interest rates, yield curves, maturity dates, issue dates, settlement dates, reported trades, broker-dealer quotes, issue spreads, benchmark securities or other market related data. We obtain the fair value of Level 2 investments from our investment manager, who obtains these fair values from a third-party pricing source. We validate the fair values of Level 2 financial instruments provided by our investment manager by comparing these fair values to a third-party pricing source.
Accounts Receivable, net and Contract Assets
Accounts receivable is recorded at the invoiced amount and is non-interest bearing. Accounts receivable is recorded net of estimated prompt pay discounts, distribution fees and chargebacks. Contract assets are recorded when revenue is earned but an invoice has not been issued for payment. Contract assets relate to development milestones deemed probable of receipt for intellectual property licenses granted to partners in prior periods and for goods or services when control has transferred to the customer, and corresponding revenue is recognized but is not yet billable to the customer in accordance with the terms of the contract.
Inventories
Inventories are stated at lower of cost or net realizable value. Cost is determined on a first-in, first-out basis. Net realizable value is the estimated selling price in the ordinary course of business, less reasonably predictable costs of completion, disposal, and transportation. Inventories are reviewed periodically for potential excess, dated or obsolete status. We evaluate the carrying value of inventories on a regular basis, taking into account such factors as historical and anticipated future sales compared to quantities on hand, the price we expect to obtain for products in their respective markets compared with historical cost and the remaining shelf life of goods on hand.
Leases
We have entered into operating leases primarily for real estate and automobiles. These leases have contractual terms which range from three years to twelve years. We determine if an arrangement contains a lease at inception. Right of use (“ROU”) assets and liabilities resulting from operating leases are included in property and equipment, accrued expenses and other long-term liabilities on our condensed consolidated balance sheets. Operating lease ROU assets and liabilities are recognized based on the present value of the future minimum lease payments over the lease term at commencement date. As most of our leases do not provide an implicit rate, we use our incremental borrowing rate based on the information available at commencement date in determining the discount rate to calculate the present value of future payments. The operating lease ROU asset also includes any lease payments made and excludes lease incentives and initial direct costs incurred. Our leases often include options to extend or terminate the lease. These options are included in the lease term when it is reasonably certain that we will exercise that option. Short-term leases with an initial term of 12 months or less are not recorded on our condensed consolidated balance sheet. Lease expense for minimum lease payments is recognized on a straight-line basis over the lease term.
We have lease agreements with lease and non-lease components, which are generally accounted for separately. For certain leases, such as automobiles, we account for the lease and non-lease components as a single lease component.
Property and Equipment, Net
Property and equipment, which includes internal-use software and leasehold improvements, are recorded at cost, less accumulated depreciation and amortization. Equipment and internal-use software are depreciated using the straight-line method over its estimated useful life ranging from three years to ten years and leasehold improvements are amortized using the straight-line method over the estimated useful life of the asset or the lease term, whichever is shorter.
Impairment of Long-Lived Assets
We account for long-lived assets in accordance with authoritative guidance for impairment or disposal of long-lived assets. Long-lived assets are reviewed for events or changes in circumstances, which indicate their carrying value may not be recoverable.
Comprehensive Income
Comprehensive income is defined as the change in equity during the period from transactions and other events and circumstances from non-owner sources.
Convertible Notes
The 2027 Convertible Notes, the 2028 Convertible Notes, the 2031 Convertible Notes and the 2032 Convertible Notes (collectively, the “Convertible Notes”) are accounted for in accordance with authoritative guidance for debt and derivatives. We evaluate all the embedded conversion options contained in the Convertible Notes to determine if there are embedded features that require bifurcation as a derivative as required by U.S. GAAP. Based on our analysis, we account for each of our Convertible Notes as single units of accounting, a liability, because we concluded that the conversion features do not require bifurcation as a derivative under embedded derivative authoritative guidance.
Cash Flow Hedges - Currency Risks
We utilize a cash flow hedging program to mitigate foreign currency exchange risk associated with forecasted royalty revenue denominated in Swiss francs. Under the program, we can hedge these forecasted royalties up to a maximum of four years into the future. We hedge these cash flow exposures to reduce the risk of our earnings and cash flows being adversely affected by fluctuations in exchange rates.
In accordance with the hedge accounting treatment, all hedging relationships are formally documented at the inception of the hedge and are highly effective in offsetting changes to future cash flows on hedged transactions. Both at inception of the hedge and on an ongoing basis, we assess whether the foreign currency forward contracts are highly effective in offsetting changes in cash flows of hedged items on a prospective and retrospective basis. If we determine a (i) foreign currency forward contract is not highly effective as a cash flow hedge, (ii) foreign currency forward contract has ceased to be a highly effective hedge or (iii) forecasted transaction is no longer probable of occurring, we would discontinue hedge accounting treatment prospectively. We measure effectiveness based on the change in fair value of the forward currency forward contract and the fair value of the hypothetical foreign currency forward contract with terms that match the critical terms of the risk being hedged. No portion of our foreign currency forward contracts were excluded from the assessment of hedge effectiveness. As of March 31, 2026, all hedges were determined to be highly effective.
The assets or liabilities associated with our hedging contracts are recorded at fair market value in prepaid expense and other current assets, prepaid expenses and other assets, accrued expenses, or other long-term liabilities, respectively, in our condensed consolidated balance sheets. Gains and losses related to changes in the fair market value of these hedging contracts are recorded as a component of accumulated other comprehensive income (loss) (“AOCI”) within stockholder’s equity in our condensed consolidated balance sheets and reclassified to royalty revenue in our condensed consolidated statements of income in the same period as the recognition of the underlying hedged transaction. In the event the underlying forecasted transaction does not occur, or it becomes probable that it will not occur, within the defined hedge period, we reclassify the gains or losses on the related cash flow hedge from AOCI to royalties revenue in our condensed consolidated statements of income. Settlements from the cash flow hedge are included in operating activities on the condensed consolidated statements of cash flows. Since the fair market value of these hedging contracts is derived from current market rates, the hedging contracts are classified as derivative financial instruments. We do not use derivatives for speculative or trading purposes. As of March 31, 2026, the amount expected to be recognized as a net loss out of AOCI into our condensed consolidated statements of income during the next 12 months is $6.2 million.
Business Combinations
Under the acquisition method of accounting, we allocate the fair value of the total consideration transferred to the tangible and identifiable intangible assets acquired and liabilities assumed based on their estimated fair values on the date of acquisition. These valuations require us to make estimates and assumptions, especially with respect to intangible assets. We record the excess consideration over the aggregate fair value of tangible and intangible assets, net of liabilities assumed, as goodwill. Costs incurred to complete a business combination, such as legal and other professional fees, are expensed as incurred.
If the initial accounting for a business combination is incomplete by the end of a reporting period that falls within the measurement period, we report provisional amounts in our financial statements. During the measurement period, we adjust the provisional amounts recognized at the acquisition date to reflect new information obtained about facts and circumstances that existed as of the acquisition date that, if known, would have affected the measurement of the amounts recognized as of that
date. We record these adjustments to the provisional amounts with a corresponding offset to goodwill. Any adjustments identified after the measurement period are recorded in our condensed consolidated statements of income.
Asset Acquisitions
We evaluate acquisitions of assets and other similar transactions to assess whether the transaction should be accounted for as a business combination or asset acquisition by first applying a test to determine if substantially all of the fair value of the gross assets acquired is concentrated into a single identifiable asset or group of similar identifiable assets. If the test is met, we account for the transaction as an asset acquisition by recognizing net assets based on the consideration paid, which includes transaction costs, on a relative fair value basis. In an asset acquisition where the cost allocated to acquire in-process research and development (“IPR&D”) has no alternative future use, we immediately recognize the cost of the acquired IPR&D on our condensed consolidated statements of income. In an asset acquisition, contingent consideration is not recognized as of the acquisition date but instead is recognized as part of the cost of the assets acquired at the time the consideration is paid.
Goodwill, Intangible Assets and Other Long-Lived Asset
Assets acquired, including intangible assets and IPR&D, and liabilities assumed are measured at fair value as of the acquisition date. Goodwill, which has an indefinite useful life, represents the excess of cost over fair value of the net assets acquired. Intangible assets acquired in a business combination that are used for IPR&D activities are considered indefinite lived until the completion or abandonment of the associated research and development efforts. Upon reaching the end of the relevant research and development project (i.e., upon commercialization), the IPR&D asset is amortized over its estimated useful life. If the relevant research and development project is abandoned, the IPR&D asset is expensed in the period of abandonment.
Goodwill and indefinite-lived intangible assets are not amortized; however, they are reviewed for impairment at least annually during the second quarter, or more frequently if an event occurs indicating the potential for impairment. Goodwill and indefinite-lived intangible assets are considered to be impaired if the carrying value of the reporting unit or indefinite-lived intangible asset exceeds its respective fair value.
We perform our goodwill impairment analysis at the reporting unit level, which is one level below our reporting and operating segment structure. We assigned goodwill to reporting units at the time of acquisition. During the goodwill impairment review, we assess qualitative factors to determine whether it is more likely than not that the fair values of our reporting unit is less than the carrying amount, including goodwill. The qualitative factors include, but are not limited to, macroeconomic conditions, industry and market considerations, and our overall financial performance. If, after assessing the totality of these qualitative factors, we determine that it is not more likely than not that the fair value of our reporting unit is less than the carrying amounts, then no additional assessment is deemed necessary. Otherwise, we proceed to compare the estimated fair value of the reporting unit with the carrying value, including goodwill. If the carrying amount of the reporting unit exceeds the fair value, we record an impairment loss based on the difference. We may elect to bypass the qualitative assessment in a period and proceed to perform the quantitative goodwill impairment test.
During the indefinite-lived intangible asset impairment review, we may elect to start by performing a qualitative assessment. The qualitative factors include, but are not limited to, macroeconomic conditions, industry and market considerations, cost factors, asset-specific commercial and regulatory developments and changes to key personnel or strategy. If the qualitative assessment indicates that it is not more likely than not that the fair value of the indefinite-lived intangible asset exceeds its carrying amount, we compare the estimated fair value of the indefinite-lived intangible asset with its carrying value. Determining fair value requires the exercise of judgment about product pricing, market assumptions, discount rates, and the amount and timing of expected future cash flows. If the carrying value of the indefinite-lived intangible asset exceeds its fair value, an impairment loss is recognized in an amount equal to that excess.
Our identifiable intangible assets with finite useful lives are typically comprised of acquired technologies, customer relationships, and product rights. The cost of identifiable intangible assets with finite lives is generally amortized on a straight-line basis over the assets’ respective estimated useful lives.
We perform regular reviews to determine if any event has occurred that may indicate intangible assets with finite useful lives and other long-lived assets are potentially impaired. If indicators of impairment exist, an impairment test is performed to assess the recoverability of the affected assets by determining whether the carrying amount of such assets exceeds the undiscounted expected future cash flows. If the affected assets are not recoverable, we estimate the fair value of the assets and record an impairment loss if the carrying value of the assets exceeds the fair value. Factors that may indicate potential impairment include a significant decline in our stock price and market capitalization compared to the net book value, significant changes in the ability of a particular asset to generate positive cash flows for our strategic business objectives, and the pattern of utilization of a particular asset.
Revenue Recognition
We generate revenues from payments received (i) as royalties from licensing our ENHANZE technology and other royalty arrangements, (ii) under collaborative agreements and (iii) from sales of our proprietary and partnered products. We recognize revenue when we transfer promised goods or services to customers in an amount that reflects the consideration to which we expect to be entitled in exchange for those goods or services. To determine revenue recognition for contracts with customers, we perform the following five steps: (i) identify the promised goods or services in the contract; (ii) identify the performance obligations in the contract, including whether they are distinct in the context of the contract; (iii) determine the transaction price, including the constraint on variable consideration; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) we satisfy the performance obligations.
ENHANZE and Device Royalties
Under the terms of our ENHANZE collaboration and license agreements, our partners will pay us royalties at an on average mid-single digit percent rate of their sales if products under the collaboration are commercialized. All amounts owed to us are noncancelable after the underlying triggering event occurs, and nonrefundable once paid. Unless terminated earlier in accordance with its terms, collaborations generally continue in effect until the last to expire royalty payment term, as determined on a product by product and country by country basis, with each royalty term starting on the first commercial sale of that product and ending the later of: (i) a specified period or term set forth in the agreement or (ii) expiration of the last to expire of the valid claims of our patents covering rHuPH20 or other specified patents developed under the collaboration which valid claim covers a product developed under the collaboration. In general, when there are no valid claims of a specified patent developed under the collaboration covering the product in a given country, the royalty rate is reduced for those sales in that country upon the expiration of our patents covering rHuPH20. Janssen’s patents covering the SC formulation of DARZALEX (“DARZALEX SC”) do not impact the timing for this royalty reduction. Partners may terminate the agreement prior to expiration for any reason in its entirety or on a target-by-target basis generally upon 90 days prior written notice to us. Upon any such termination, the license granted to partners (in total or with respect to the terminated target, as applicable) will terminate provided; however, that in the event of expiration of the agreement (as opposed to a termination), the on-going licenses granted may become perpetual, non-exclusive and fully paid. Sales-based milestones and royalties are recognized in the period the underlying sales or milestones occur. We do not receive final royalty reports from our ENHANZE partners until after we complete our financial statements for a prior quarter. Therefore, we recognize revenue based on estimates of the royalty earned, which are based on internal estimates and available preliminary reports provided by our partners. We will record adjustments in the following quarter, if necessary, when final royalty reports are received. To date, we have not recorded any material adjustments.
We also earn royalties in connection with our licenses granted under license and development arrangement with our device partner. These royalties are based upon a percentage of commercial sales of partnered products with rates ranging from mid-single digits to low double digits and are tiered based on levels of net sales. These sales-based royalties, for which the license was deemed the predominant element to which the royalties relate, are estimated and recognized in the period in which the partners’ commercial sales occur. The royalties are generally reported and payable to us within 45 to 60 days after the end of the period in which the commercial sales are made. We base our estimates of royalties earned on actual sales information from our partners when available or estimated prescription sales from external sources and estimated net selling price. We will record adjustments in the following quarter, if necessary, when final royalty reports are received. To date, we have not recorded any material adjustments.
Revenue under ENHANZE, Device and Hypercon Technology Collaborative Agreements
ENHANZE Collaboration and License Agreements
Under these agreements, we grant the collaboration partner a worldwide license to develop and commercialize products using our ENHANZE technology to combine our patented rHuPH20 enzyme with their proprietary biologics directed at up to a specified number of targets. Targets are usually licensed on an exclusive, global basis. Targets selected subsequent to inception of the arrangement generally require payment of an additional license fee. The collaboration partner is responsible for all development, manufacturing, clinical, regulatory, sales and marketing costs for any products developed under the agreement. We are responsible for supply of bulk rHuPH20 based on the collaboration partner’s purchase orders, and may also be separately engaged to perform research and development services. While these collaboration agreements are similar in that they originate from the same framework, each one is the result of an arms-length negotiation and thus may vary from one to the other.
We generally collect an upfront license payment from collaboration partners, and are also entitled to receive event-based payments subject to collaboration partners’ achievement of specified development, regulatory and sales-based milestones. In several agreements, collaboration partners pay us annual fees to maintain their exclusive license rights if they are unable to
advance product development to specified stages. We earn separate fees for bulk rHuPH20 supplies and research and development services.
Although these agreements are in form identified as collaborative agreements, we concluded for accounting purposes they represent contracts with customers and are not subject to accounting literature on collaborative arrangements. This is because we grant to partners licenses to our intellectual property and provide supply of bulk rHuPH20 and research and development services which are all outputs of our ongoing activities, in exchange for respective consideration. Under these collaborative agreements, our partners lead development of assets, and we do not share in significant financial risks of their development or commercialization activities. Accordingly, we concluded our collaborative agreements are appropriately accounted for pursuant to U.S. GAAP.
Under all of our ENHANZE collaborative agreements, we have identified licenses to use functional intellectual property as the only performance obligation. The intellectual property underlying the license is our proprietary ENHANZE technology which represents application of rHuPH20 to facilitate delivery of drugs. Each of the licenses grants the partners rights to use our intellectual property as it exists and is identified on the effective date of the license, because there is no ongoing development of the ENHANZE technology required. Therefore, we recognize revenue from licenses at the point when the license becomes effective and the partner has received access to our intellectual property, usually at the inception of the agreement.
When partners can select additional targets to add to the licenses granted, we consider these rights to be options. We evaluate whether such options contain material rights, i.e., have exercise prices that are discounted compared to what we would charge for a similar license to a new partner. The exercise price of these options includes a combination of the target selection fees, event-based milestone payments and royalties. When these amounts in aggregate are not offered at a discount that exceeds discounts available to other customers, we conclude the option does not contain a material right, and we consider grants of additional licensing rights upon option exercises to be separate contracts (target selection contracts).
Generally, we provide indemnification and protection of licensed intellectual property for our customers. These provisions are part of assurance that the licenses meet the agreements’ representations and are not obligations to provide goods or services.
We also fulfill purchase orders for supply of bulk rHuPH20 and perform research and development services pursuant to project authorization forms for our partners, which represent separate contracts. In addition to our licenses, we price our supply of bulk rHuPH20 and research and development services at our regular selling prices, called standalone selling prices (“SSP”). Therefore, our partners do not have material rights to order these items at prices not reflective of SSP. Refer to the discussion below regarding recognition of revenue for these separate contracts.
Transaction price for a contract represents the amount to which we are entitled in exchange for providing goods and services to the customer. Transaction price does not include amounts subject to uncertainties unless it is probable that there will be no significant reversal of revenue when the uncertainty is resolved. Apart from the upfront license payment (or target selection fees in the target selection contracts), all other fees we may earn under our collaborative agreements are subject to significant uncertainties of product development. Achievement of many of the event-based development and regulatory milestones may not be probable until such milestones are actually achieved. This generally relates to milestones such as obtaining marketing authorization approvals. With respect to other development milestones, e.g., dosing of a first patient in a clinical trial, achievement could be considered probable prior to its actual occurrence, based on the progress towards commencement of the trial. In order to evaluate progress towards commencement of a trial, we assess the status of activities leading up to our partner’s initiation of a trial such as feedback received from the applicable regulatory authorities, completion of investigational new drug or equivalent filings, readiness and availability of drug, readiness of study sites and our partner’s commitment of resources to the program. We do not include any amounts subject to uncertainties in the transaction price until it is probable that the amount will not result in a significant reversal of revenue in the future. At the end of each reporting period, we re-evaluate the probability of achievement of such milestones and any related constraint, and if necessary, adjust our estimate of the overall transaction price.
When target exchange rights are held by partners, and the amounts attributed to these rights are not refundable, they are included in the transaction price. However, they are recorded as deferred revenues because we have a potential performance obligation to provide a new target upon an exchange right being exercised. These amounts are recognized in revenue when the right of exchange expires or is exercised.
Because our agreements have one type of performance obligation (licenses) which are typically all transferred at the same time at agreement inception, allocation of transaction price often is not required. However, allocation is required when licenses for some of the individual targets are subject to rights of exchange, because revenue associated with these targets cannot be recognized. When allocation is needed, we perform an allocation of the upfront amount based on relative SSP of licenses for individual targets. We determine license SSP using an income-based valuation approach utilizing risk-adjusted discounted cash flow projections of the estimated return a licensor would receive where applicable, or an alternative valuation method such as indicative value from historical transactions. When amounts subject to uncertainties, such as milestones and royalties, are
included in the transaction price, we attribute them to the specific individual target licenses which generate such milestone or royalty amounts.
We also estimate SSP of bulk rHuPH20 and research and development services, to determine that our partners do not have material rights to order them at discounted prices. For supplies of bulk rHuPH20, because we effectively act as a contract manufacturer to our partners, we estimate and charge SSP based on the typical contract manufacturer margins consistent with all of our partners. We determine SSP of research and development services based on a fully-burdened labor rate. Our rates are comparable to those we observe in other collaborative agreements. We also have a history of charging similar rates to all of our partners.
Upfront amounts allocated to licenses to individual targets are recognized as revenue when the license is transferred to the partner, as discussed above, if the license is not subject to exchange rights, or when the exchange right expires or is exercised. Development milestones and other fees are recognized in revenue when they are included in the transaction price, because by that time, we have already transferred the related license to the partner.
In contracts to provide research and development services, such services represent the only performance obligation. The fees are charged based on hours worked by our employees and the fixed contractual rate per hour, plus third-party pass-through costs, on a monthly basis. We recognize revenues as the related services are performed based on the amounts billed, as the partner consumes the benefit of research and development work simultaneously as we perform these services, and the amounts billed reflect the value of these services to the customer.
Hypercon Technology Collaboration and Licensing Agreements
We have several collaboration and license agreements with collaboration partners who are attempting to formulate Hypercon technology with their existing drugs to develop an additional administrative option. The collaboration partner is responsible for all development, manufacturing, clinical, regulatory, sales and marketing costs for any products developed under the agreement; however, the agreement also includes Hypercon performing certain research and development (“R&D”) services to support the collaboration partner in their efforts. We assessed the nature of the promised goods and services in the contract which includes the license of the Hypercon technology representing a functional license of intellectual property to our customers, R&D services, and manufacturing technology transfer. These promises are not capable of being distinct performance obligations at this time due to the exclusive proprietary know-how and certain regulatory requirements associated with the manufacturing of the product. The collaboration partner simultaneously receives and consumes the benefits of the combined performance obligations as data is generated to support regulatory approval submissions. A significant component of the cost of R&D relates to our clinical trial research consultants, who are assisting with the monitoring of the Hypercon technology to ensure the successful combination of the Hypercon technology with the collaboration partner’s drug.
The agreements include milestone payments, royalties, and an initial upfront payment. This upfront payment was received from the collaboration partner upon inception of the agreement and recorded as deferred revenue to be recognized as we perform certain R&D services to support the collaboration partner in their efforts to successfully combine Hypercon technology with previously developed products. While the services are being provided, we allocate a portion of each deferred payment to the individual drug targets. Revenue is then recognized based upon the expected costs to complete as a percentage of the budget. These agreements are designed in a way that the R&D services are being completed in partnership with the collaboration partner at the approximate costs; therefore, the deferred revenue is being recognized as the work is completed for each partner’s drug. We may incur additional expenses outside of the R&D contract which are invoiced and recognized separately from the upfront payment. Deferred revenue associated with a target that has not been identified or no work has been performed is classified as other long-term liabilities on the condensed consolidated balance sheets. Deferred revenue for expenses expected to be incurred within the next 12 months are classified as accrued expenses on the condensed consolidated balance sheets.
Device License, Development and Supply Arrangements
We have license, development and supply arrangements with pharmaceutical partners, under which we grant a license to our device technology and provide research and development services that often involve multiple performance obligations and highly-customized deliverables. For such arrangements, we identify each of the promised goods and services within the contract and the distinct performance obligations at inception of the contract and allocate consideration to each performance obligation based on relative SSP, which is generally determined based on the expected cost plus mark-up.
If the contract includes an enforceable right to payment for performance completed to date and performance obligations are satisfied over time, we recognize revenue over the development period using either the input or output method depending on which is most appropriate given the nature of the distinct deliverable. For other contracts that do not contain an enforceable right to payment for performance completed to date, revenue is recognized when control of the product is transferred to the customer. Factors that may indicate transfer of control has occurred include the transfer of legal title, transfer of physical
possession, the customer has obtained the significant risks and rewards of ownership of the assets, and we have a present right to payment.
Our payment terms for development contracts may include an upfront payment equal to a percentage of the total contract value with the remaining portion to be billed upon completion and transfer of the individual deliverables or satisfaction of the individual performance obligations. We record a contract liability for cash received in advance of performance, which is presented as deferred revenue within accrued expense and other long-term liabilities in our condensed consolidated balance sheets and recognized as revenue in our condensed consolidated statements of income when the associated performance obligations have been satisfied.
License fees and milestones received in exchange for the grant of a license to our functional intellectual property, such as patented technology and know-how in connection with a partnered development arrangement, are generally recognized at inception of the arrangement, or over the development period depending on the facts and circumstances, as the license is generally not distinct from the non-licensed goods or services to be provided under the contract. Milestone payments that are contingent upon the occurrence of future events are evaluated and recorded at the most likely amount, and to the extent that it is probable that a significant reversal of revenue will not occur when the associated uncertainty is resolved.
Refer to Note 5, Revenue, for further discussion on our collaborative arrangements.
Product Sales, Net
Proprietary Product Sales
Our commercial portfolio of proprietary products includes XYOSTED and Hylenex recombinant which we sell primarily to wholesale pharmaceutical distributors and specialty pharmacies, who sell the products to hospitals, retail chain drug stores and other end-user customers. Sales to wholesalers are made pursuant to purchase orders subject to the terms of a master agreement, and delivery of individual packages of products represents performance obligations under each purchase order. We use contract manufacturers to produce our proprietary products and third-party logistics vendors to process and fulfill orders. We concluded we are the principal in the sales to wholesalers because we control access to services rendered by both vendors and direct their activities. We have no obligations to wholesalers to generate pull-through sales.
Revenue is recognized when control has transferred to the customer, which is typically upon delivery, at the net selling price, which reflects the variable consideration for which reserves and sales allowances are established for estimated returns, wholesale distribution fees, prompt payment discounts, government rebates and chargebacks, plan rebate arrangements and patient discount and support programs. We recognize revenue from product sales and related cost of sales upon product delivery to the wholesaler location. At that time, the wholesalers take control of the product as they take title, bear the risk of loss of ownership, and have an enforceable obligation to pay us. They also have the ability to direct sales of product to their customers on terms and at prices they negotiate. Although wholesalers have product return rights, we do not believe they have a significant incentive to return the product to us.
The determination of certain reserves and sales allowances requires us to make a number of judgments and estimates to reflect our best estimate of the transaction price and the amount of consideration to which we believe we would be ultimately entitled to receive. The expected value is determined based on unit sales data, contractual terms with customers and third-party payers, historical and estimated future percentage of rebates incurred on sales, historical and future insurance plan billings, any new or anticipated changes in programs or regulations that would impact the amount of the actual rebates, customer purchasing patterns, product expiration dates and levels of inventory in the distribution channel. The estimated amounts of credit for product returns, chargebacks, distribution fees, prompt payment discounts, rebates and customer co-pay support programs are included in accrued expenses and accounts receivable, net in our condensed consolidated balance sheets upon recognition of revenue from product sales. We monitor actual product returns, chargebacks, discounts and fees subsequent to the sale. If these amounts differ from our estimates, we make adjustments to these allowances, which are applied to increase or reduce product sales revenue and earnings in the period of adjustment.
Selling prices initially billed to wholesalers are subject to discounts for prompt payment and subsequent chargebacks when wholesalers sell our products at negotiated discounted prices to members of certain group purchasing organizations, pharmacy benefit managers and government programs. We also pay quarterly distribution fees to certain wholesalers for inventory reporting and chargeback processing, and to pharmacy benefit managers and group purchasing organizations as administrative fees for services and for access to their members. We concluded the benefits received in exchange for these fees are not distinct from our sales of our products, and accordingly we apply these amounts to reduce revenues. Wholesalers also have rights to return unsold product nearing or past the expiration date. Because of the shelf life of our products and our lengthy
return period, there may be a significant period of time between when the product is shipped and when we issue credits on returned product.
We estimate the transaction price when we receive each purchase order taking into account the expected reductions of the selling price initially billed to the wholesaler arising from all of the above factors. We have compiled historical experience and data to estimate future returns and chargebacks of our products and the impact of the other discounts and fees we pay. When estimating these adjustments to the transaction price, we reduce it sufficiently to be able to assert that it is probable that there will be no significant reversal of revenue when the ultimate adjustment amounts are known.
Each purchase order contains only one type of product and is usually shipped to the wholesaler in a single shipment. Therefore, allocation of the transaction price to individual packages is not required.
In connection with the orders placed by wholesalers, we incur costs such as commissions to our sales representatives. However, as revenue from product sales is recognized upon delivery to the wholesaler, which occurs shortly after we receive a purchase order, we do not capitalize these commissions and other costs, based on application of the practical expedient allowed within the applicable guidance.
Partnered Product Sales
Bulk rHuPH20
We sell bulk rHuPH20 to partners for use in research and development and, subsequent to receiving marketing approval, we sell it for use in collaboration commercial products. Sales are made pursuant to purchase orders subject to the terms of the collaborative agreement or a supply agreement, and delivery of units of bulk rHuPH20 represent performance obligations under each purchase order. We provide a standard warranty that the product conforms to specifications. We use contract manufacturers to produce bulk rHuPH20 and have concluded we are the principal in the sales to partners. The transaction price for each purchase order of bulk rHuPH20 is fixed based on the cost of production plus a contractual markup, and is not subject to adjustments. Allocation of the transaction price to individual quantities of the product is usually not required because each order contains only one type of product.
We recognize revenue from the sale of bulk rHuPH20 as product sales and related cost of sales upon transfer of title to our partners. At that time, the partners take control of the product, bear the risk of loss of ownership, and have an enforceable obligation to pay us.
Devices
We are party to license, development, supply and distribution arrangements with pharmaceutical partners, under which we produce and are the exclusive supplier of certain products, devices and/or components. We recognize revenue from the sale of certain products, devices and/or components as product sales and related costs of sales at the point in time in which control is transferred to the customer, which is typically upon shipment of the goods to our partner. Sales terms and pricing are governed by the respective supply and distribution agreements, and there is generally no right of return. We provide a standard warranty that the product conforms to specifications. We use contract manufacturers to produce certain products, devices and/or components, and have concluded we are the principal in the sales to partners. Revenue is recognized at the transaction price, which includes the contractual per unit selling price. Allocation of the transaction price to individual quantities of the product is usually not required because each order contains only one type of product.
Cost of Sales
Cost of sales consists primarily of raw materials, third-party manufacturing costs, fill and finish costs, freight costs, internal costs and manufacturing overhead associated with the production of proprietary and partnered products. Cost of sales also consists of the write-down of excess, dated and obsolete inventories and the write-off of inventories that do not meet certain product specifications, if any.
Research and Development Expenses
Research and development expenses include salaries and benefits, allocation of facilities and other overhead expenses, research related manufacturing services, contract services, and other outside expenses related to manufacturing, preclinical and regulatory activities and our partner development platforms. Research and development expenses are charged to operating expenses as incurred when these expenditures relate to our research and development efforts and have no alternative future uses.
We are obligated to make upfront payments upon execution of certain research and development agreements. Advance payments, including nonrefundable amounts, for goods or services that will be used or rendered for future research and development activities are deferred. Such amounts are recognized as expense as the related goods are delivered or the related services are performed or such time when we do not expect the goods to be delivered or services to be performed.
Share-Based Compensation
We record compensation expense associated with stock options, restricted stock units (“RSUs”), performance stock units (“PSUs”) and shares issued under our employee stock purchase plan (“ESPP”) in accordance with the authoritative guidance for share-based compensation. The cost of employee services received in exchange for an award of an equity instrument is measured at the grant date, based on the estimated fair value of the award, and is recognized as expense on a straight-line basis over the requisite service period of the award. Share-based compensation expense for an award with a performance condition is recognized when the achievement of such performance condition is determined to be probable. If the outcome of such performance condition is not determined to be probable or is not met, no compensation expense is recognized and any previously recognized compensation expense is reversed. Forfeitures are recognized as a reduction of share-based compensation expense as they occur.
Income Taxes
We provide for income taxes using the liability method. Under this method, deferred income tax assets and liabilities are determined based on the differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax basis at each reporting period. We measure deferred tax assets and liabilities using enacted tax rates for the year in which the differences are expected to reverse. Significant judgment is required by management to determine our provision for income taxes, our deferred tax assets and liabilities, and any associated valuation allowances recorded against our net deferred tax assets. Deferred tax assets (“DTA”) and other tax benefits are recorded when they are more likely than not to be realized. On a quarterly basis, we assess the need for valuation allowance on our DTAs, weighing all positive and negative evidence, to assess if it is more-likely-than-not that some or all of our DTAs will be realized. We recorded a provision for income tax of $30.3 million with an effective tax rate of 16.8% for the three months ended March 31, 2026. The difference between our effective tax rates and the U.S. federal statutory rate of 21% is primarily due to decreases from share-based compensation windfall tax benefit, Foreign Derived Deduction Eligible Income benefit, and research and development credit generation, partially offset by state income tax and Section 162(m) disallowance.
Segment Information
We generate revenues from payments received (i) as royalties from licensing our ENHANZE technology and other royalty arrangements, (ii) under collaborative agreements with our partners and (iii) from sales of our proprietary and partnered products. There are no intra-entity sales or transfers. We operate our business in one operating segment, which includes all activities related to the research, development and commercialization of our proprietary enzymes, devices, Hypercon technology and the Surf Bio technology. This operating segment also includes revenues and expenses related to (i) research and development and manufacturing activities conducted under our collaborative agreements with third parties, (ii) product sales of proprietary and partnered products and (iii) associated selling, general and administrative expenses.
The chief operating decision-maker (“CODM”), our Chief Executive Officer, reviews the operating results on an aggregate basis and manages the operations as a single operating segment. The CODM assesses the segment’s performance and decides how to allocate resources based on consolidated net income that is reported in our condensed consolidated statements of income. The measure of segment assets is reported on the condensed consolidated balance sheets as total consolidated assets. The significant expense categories regularly provided to the CODM include cost of sales, research and development, amortization of intangibles, and selling, general and administrative expenses. These expense categories are reported as separate line items in our condensed consolidated statements of income.
Adoption and Pending Adoption of Recent Accounting Pronouncements
The following table provides a brief description of recently issued accounting standards, those adopted in the current period and those not yet adopted:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| Standard | | Description | | Effective Date | | Adoption Method | | Effect on the Financial
Statements or Other Significant Matters |
In November 2024, the FASB issued ASU 2024-03, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses. | | The new guidance is intended to enhance expense disclosures by requiring disaggregation of certain expenses included in the consolidated statements of income into specified expense categories in the notes to the consolidated financial statements. | | Annual periods beginning after December 15, 2026 (our 2027 Form 10-K), and interim reporting periods beginning after December 15, 2027 (our Q1 2028 Form 10-Q) - Early adoption is permitted. | | Prospective or Retrospective | | We are currently evaluating the impact of the standard on our consolidated financial statements and related disclosures. |
3. Business Combinations
Elektrofi, Inc.
On November 18, 2025, we acquired all outstanding equity interests of Elektrofi, Inc. according to the terms and conditions of the Agreement and Plan of Merger dated as of September 30, 2025 (the “Elektrofi Merger Agreement”). Elektrofi is a biopharmaceutical company with an innovative microparticle technology that has been demonstrated in non-clinical testing to enable hyperconcentration of drugs and biologics and reduce the injection volume for the same dosage, potentially expanding opportunities for at-home and health care provider administration. We acquired Elektrofi as a part of our strategy to expand our drug delivery technology offerings.
Hypercon technology is an innovative microparticle technology that has been demonstrated in non-clinical testing to enable hyperconcentration of drugs and biologics and reduce the injection volume for the same dosage, potentially expanding opportunities for at-home and health care provider administration.
The total purchase consideration of Elektrofi was $810.4 million. Each share of Elektrofi common stock issued and outstanding was converted into the right to receive $28.80 per share in cash without interest, less any applicable withholding taxes (“Merger Consideration”). Additionally, in connection with the transaction, $56.5 million was paid to Elektrofi option holders for options granted and outstanding as of November 18, 2025 under the Elektrofi 2015 Equity Compensation Plan. Other components of purchase consideration included an estimated fair value of contingent consideration of $23.0 million related to future milestone payments, and cash paid at closing to settle seller transaction costs of $18.4 million paid by us on behalf of Elektrofi.
The acquisition of Elektrofi was funded by cash on hand and rollover equity. We recognized transaction costs of $13.7 million in the year ended December 31, 2025. These costs are reported in selling, general and administrative expenses in our consolidated statements of income. Transaction costs include, but are not limited to, investment banker, advisory, legal, and other professional fees.
Purchase Consideration
The total purchase consideration was comprised of the following (in thousands):
| | | | | | | | |
Cash consideration payments to stockholders and option holders as of November 18, 2025 | | $ | 787,392 | |
| | |
| | |
| Estimated fair value of contingent consideration | | 23,000 | |
Total purchase consideration | | $ | 810,392 | |
Fair Value of Assets Acquired and Liabilities Assumed
The acquisition of Elektrofi has been accounted for using the acquisition method of accounting in accordance with ASC 805, Business Combinations, with Halozyme treated as the accounting acquirer, which requires, among other things, that the assets acquired and liabilities assumed be recognized at their fair value on the acquisition date. Acquisition accounting is dependent upon certain valuations and other studies that have yet to commence or progress to a stage where there is sufficient information for a definitive measurement. The process for estimating the fair values of identifiable intangible assets and certain tangible assets and assumed liabilities requires the use of judgment in determining the appropriate assumptions and estimates.
The table below presents the preliminary estimated fair values of assets acquired and liabilities assumed on the acquisition date based on valuations and management estimates (in thousands). Fair value estimates are based on a complex series of judgments about future events and uncertainties and rely heavily on estimates and assumptions. The judgments used to determine the estimated fair value assigned to each class of assets acquired and liabilities assumed, as well as asset lives, can materially impact our results of operations. We are still finalizing the allocation of the purchase price, therefore, the fair value estimates assigned to intangible assets, goodwill and the related tax impacts of the acquisition, among other items, are subject to change as additional information is received to complete our analysis and certain tax returns are finalized. As a result, the
preliminary estimates may be revised during the measurement period. These differences could change the value of the intangible assets acquired, the contingent liability assumed, and the tax impacts related to the acquisition and could have a material impact on our results of operations and financial position.
| | | | | | | | | | | | | | | | | | | | |
| | Amounts recognized as of acquisition date (as initially reported) | | Measurement period adjustments (3) | | Amounts recognized as of March 31, 2026 (as adjusted) |
Assets acquired | | | | | | |
| Cash and cash equivalents | | $ | 84,427 | | | $ | 710 | | | $ | 85,137 | |
| Accounts receivable, net | | 4,755 | | | (2,831) | | | 1,924 | |
| Prepaid expenses and other current assets | | 879 | | | — | | | 879 | |
| Total current assets | | 90,061 | | | (2,121) | | | 87,940 | |
| Property and equipment, net | | 7,338 | | | — | | | 7,338 | |
| Operating lease right-of-use assets | | 8,441 | | | — | | | 8,441 | |
Goodwill (1) | | 163,539 | | | 1,963 | | | 165,502 | |
| Intangible assets | | 705,000 | | | — | | | 705,000 | |
| Other assets | | 182 | | | — | | | 182 | |
Total assets acquired | | $ | 974,561 | | | $ | (158) | | | $ | 974,403 | |
Liabilities assumed | | | | | | |
| Accounts payable | | $ | (929) | | | $ | — | | | $ | (929) | |
| Accrued expenses and other current liabilities | | (26,803) | | | 158 | | | (26,645) | |
| Deferred revenue, current | | (5,674) | | | — | | | (5,674) | |
| Operating lease liabilities, current | | (3,440) | | | — | | | (3,440) | |
| Other current liabilities | | (51) | | | — | | | (51) | |
| Total current liabilities | | (36,897) | | | 158 | | | (36,739) | |
| Deferred revenue, net of current portion | | (19,974) | | | — | | | (19,974) | |
| Operating lease liabilities, net of current portion | | (4,484) | | | — | | | (4,484) | |
Other liabilities (2) | | (102,816) | | | — | | | (102,816) | |
| Total liabilities assumed | | $ | (164,171) | | | $ | 158 | | | $ | (164,013) | |
| | | | | | |
| Net assets acquired | | 810,392 | | | — | | | 810,392 | |
| Less cash acquired | | (84,427) | | | (710) | | | (85,137) | |
| Total purchase price, net of cash acquired | | $ | 725,965 | | | $ | (710) | | | $ | 725,255 | |
(1) Goodwill is the excess of the consideration transferred over the net assets recognized and represents the expected synergies of the combined operations and the assembled workforce acquired in the acquisition. Goodwill recognized as a result of the acquisition is not expected to be deductible for tax purposes.
(2) Includes $102.8 million of deferred tax liabilities.
(3) The measurement period adjustments have been recorded to reflect facts and circumstances that existed as of the acquisition date.
Identifiable Intangible Assets
The estimated fair value of the Hypercon developed technology platform asset was prepared using the replacement cost method which calculates present-day cost of replacing that asset with a similar asset in a similar condition. The estimated fair value of customer relationship assets were prepared using the multi-period excess earnings method which calculates the present value of the incremental after-tax cash flows attributable solely to each customer relationship. The estimated fair value of the trade name asset was prepared using the relief from royalty method which calculates the value of the trade name based on royalties that would be paid if licensed by a third party. The estimated useful lives are based on forecasted periods of benefit for each intangible asset. Useful lives and preliminary values are presented in the table below.
| | | | | | | | | | | | | | |
| | Amount (in thousands) | | Useful life (years) |
Hypercon developed technology platform | | $ | 230,000 | | | 15 |
Customer relationships | | 470,000 | | | 15 |
Trade name | | 5,000 | | | 15 |
Estimated fair value of intangible assets acquired | | $ | 705,000 | | | |
Surf Bio, Inc.
On December 22, 2025, we acquired all outstanding equity interests of Surf Bio, Inc. according to the terms and conditions of the Agreement and Plan of Merger dated as of December 18, 2025 (the “Surf Bio Merger Agreement”). Surf Bio is a preclinical biopharmaceutical company that is transforming how antibodies and biologics are delivered to patients. We acquired Surf Bio as a part of our strategy to expand our drug delivery technology offerings.
The total purchase consideration of Surf Bio was $305.0 million to selling shareholders, inclusive of $10.2 million in transaction expenses. We acquired $6.4 million in cash and restricted cash, of which $4.3 million was used to fund seller payments and transaction expenses. An incremental $100.0 million in consideration is contingent to the sellers in the form of milestone payments which are dependent on the occurrence of future events.
The acquisition of Surf Bio was not considered a business combination as Surf Bio did not meet the definition of a business under ASC 805-10, Business Combinations. Rather, the asset purchase transaction was accounted for under the authoritative guidance for asset acquisitions within ASC 805-50, whereby the underlying asset was deemed an IPR&D asset with no alternative future use. We allocated the cost of the acquisition, or $294.2 million, among the assets acquired based on the relative fair value of such assets. The $294.2 million consisted of $294.8 million in consideration paid to the sellers, plus $10.2 million of transaction expenses, less $10.8 million in accelerated stock-based compensation expense. The acquired assets’ value is predominately concentrated in the IPR&D asset. In accordance with ASC 805-50, the fair value allocated to the IPR&D asset, or $284.9 million, was recorded in the consolidated statements of income in the year ended December 31, 2025. The fair value of other assets acquired and liabilities assumed were capitalized to the consolidated balance sheet. The $100.0 million in contingent consideration will be recognized in the future when and if milestones are met (i.e., when the contingent consideration is paid or payable) and does not meet the definition of a derivative.
4. Fair Value Measurement
Available-for-sale marketable securities consisted of the following (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | March 31, 2026 |
| | Amortized Cost | | Gross Unrealized Gains | | Gross Unrealized Losses | | Estimated Fair Value |
| | | | | | | | |
| | | | | | | | |
| U.S. treasury securities | | $ | 8,878 | | | $ | — | | | $ | (5) | | | $ | 8,873 | |
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
| Total marketable securities, available-for-sale | | $ | 8,878 | | | $ | — | | | $ | (5) | | | $ | 8,873 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | December 31, 2025 |
| | Amortized Cost | | Gross Unrealized Gains | | Gross Unrealized Losses | | Estimated Fair Value |
| | | | | | | | |
| | | | | | | | |
| U.S. treasury securities | | $ | 9,002 | | | $ | — | | | $ | (2) | | | $ | 9,000 | |
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
| Total marketable securities, available-for-sale | | $ | 9,002 | | | $ | — | | | $ | (2) | | | $ | 9,000 | |
As of March 31, 2026, all available-for-sale marketable securities with a fair market value of $8.9 million were in an immaterial gross unrealized loss position. Based on our review of these marketable securities, we believe none of the unrealized loss is as a result of a credit loss as of March 31, 2026 because we do not intend to sell these securities and it is not more-likely-than-not that we will be required to sell these securities before the recovery of their amortized cost basis.
The estimated fair value of our contractual maturities of available-for-sale debt securities were as follows (in thousands):
| | | | | | | | | | | | | | |
| | March 31, 2026 | | December 31, 2025 |
| | Estimated Fair Value |
| Due within one year | | $ | 8,873 | | | $ | 9,000 | |
Due after one year but within five years | | — | | | — | |
Total estimated fair value of available-for-sale securities | | $ | 8,873 | | | $ | 9,000 | |
The following table summarizes, by major security type, our cash equivalents and available-for-sale marketable securities measured at fair value on a recurring basis and are categorized using the fair value hierarchy (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | March 31, 2026 | | December 31, 2025 |
| | Level 1 | | Level 2 | | Total Estimated Fair Value | | Level 1 | | Level 2 | | Total Estimated Fair Value |
| Assets | | | | | | | | | | | | |
| Cash equivalents | | | | | | | | | | | | |
| Money market funds | | $ | 509 | | | $ | — | | | $ | 509 | | | $ | 175 | | | $ | — | | | $ | 175 | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
Available-for-sale marketable
securities | | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| U.S. treasury securities | | 8,873 | | | — | | | 8,873 | | | 9,000 | | | — | | | 9,000 | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| Derivative instruments | | | | | | | | | | | | |
Currency hedging contracts (1) | | — | | | 740 | | | 740 | | | — | | | — | | | — | |
Total assets measured at fair value | | $ | 9,382 | | | $ | 740 | | | $ | 10,122 | | | $ | 9,175 | | | $ | — | | | $ | 9,175 | |
| | | | | | | | | | | | |
| Liabilities | | | | | | | | | | | | |
| Derivative instruments | | | | | | | | | | | | |
Currency hedging contracts (1) | | $ | — | | | $ | 17,106 | | | $ | 17,106 | | | $ | — | | | $ | 22,891 | | | $ | 22,891 | |
(1) Based on observable market transactions of spot currency rates, forward currency rates or equivalently-termed instruments. Carrying amounts of the financial assets and liabilities are equal to the fair value. As of March 31, 2026, the derivative assets recorded within prepaid expenses and other current assets and prepaid expenses and other assets in our consolidated balance sheets were $0.4 million and $0.3 million, respectively. The derivative liabilities recorded within accrued expenses and other long-term liabilities in our condensed consolidated balance
sheets were $6.6 million and $10.5 million, respectively. As of December 31, 2025, the derivative liabilities recorded within accrued expenses and other long-term liabilities in our condensed consolidated balance sheets were $7.7 million and $15.1 million, respectively.
We had no available-for-sale securities that were classified within Level 3 as of March 31, 2026 and December 31, 2025.
A contingent liability with a preliminary value of $23.0 million was assumed as part of the Elektrofi acquisition related to future milestone payments. The acquisition date fair value of contingent consideration was measured using the income approach, specifically the probability weighted expected return method for the development milestone payments. The fair value of the contingent liability will be remeasured quarterly. Estimates and assumptions used in the valuation include probability of achieving certain milestones, the expected timing of achieving these milestones, and a discount rate. These unobservable inputs represent a Level 3 measurement because they are supported by little or no market activity and reflect our own assumptions in measuring fair value. Changes in the fair value subsequent to the acquisition date will be recognized in our condensed consolidated statements of income.
5. Revenue
Our disaggregated revenues were as follows (in thousands):
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
| | 2026 | | 2025 | | | | |
| Royalties | | $ | 240,681 | | | $ | 168,192 | | | | | |
| | | | | | | | |
| Product sales, net | | | | | | | | |
Proprietary product sales | | 41,873 | | | 36,341 | | | | | |
Bulk rHuPH20 sales | | 73,497 | | | 27,025 | | | | | |
Device partnered product sales | | 15,054 | | | 14,675 | | | | | |
| Total product sales, net | | 130,424 | | | 78,041 | | | | | |
| | | | | | | | |
| Revenues under collaborative agreements | | | | | | | | |
Upfront license and target nomination fees | | — | | | 220 | | | | | |
| | | | | | | | |
Sales-based milestones | | — | | | 15,000 | | | | | |
Device licensing and development revenue | | 5,603 | | | 3,408 | | | | | |
| Total revenues under collaborative agreements | | 5,603 | | | 18,628 | | | | | |
| | | | | | | | |
| Total revenues | | $ | 376,708 | | | $ | 264,861 | | | | | |
During the three months ended March 31, 2026, we recognized revenue related to licenses granted to partners in prior periods in the amount of $240.7 million. This amount represents royalties earned in the current period. We recognized $2.4 million revenue during the three months ended March 31, 2026 that had been included in accrued expense and other long-term liabilities in our condensed consolidated balance sheets as of December 31, 2025.
Accounts receivable, net, other contract assets and deferred revenues (contract liabilities) from contracts with customers, including partners, consisted of the following (in thousands):
| | | | | | | | | | | | | | |
| | March 31, 2026 | | December 31, 2025 |
| Accounts receivable, net | | $ | 457,989 | | | $ | 426,273 | |
| Other contract assets | | — | | | 15,000 | |
| Deferred revenues | | 32,945 | | | 35,482 | |
| | | | |
As of March 31, 2026, the amounts included in the transaction price of our contracts with customers, including collaboration partners, and allocated to goods and services not yet provided were $298.7 million, of which $265.8 million relates to unfulfilled product purchase orders and $32.9 million has been collected and is reported as other long-term liabilities in our condensed consolidated balance sheets. The unfulfilled product purchase orders are estimated to be delivered by the first quarter of 2028. Of the total deferred revenues of $32.9 million, $5.7 million is expected to be used by our customers within the next 12 months.
There were no contract assets related to collaboration agreements as of March 31, 2026. While we may become entitled to receive additional event-based development and regulatory milestones and other fees under our collaborative agreements, which relate to development milestones deemed probable of receipt for intellectual property licenses granted to collaboration partners in prior periods, no amounts were deemed probable.
6. Certain Balance Sheet Items
Accounts receivable, net and contract assets consisted of the following (in thousands):
| | | | | | | | | | | | | | |
| | March 31,
2026 | | December 31,
2025 |
Product sales to partners | | $ | 105,833 | | | $ | 69,832 | |
Revenues under collaborative agreements | | 9,908 | | | 40,020 | |
Royalty payments | | 298,116 | | | 259,298 | |
Other product sales | | 54,388 | | | 65,117 | |
| Other receivable | | — | | | 1,612 | |
| Contract assets | | — | | | 15,000 | |
Total accounts receivable and contract assets | | 468,245 | | | 450,879 | |
| Allowance for distribution fees and discounts | | (10,256) | | | (9,606) | |
Total accounts receivable, net and contract assets | | $ | 457,989 | | | $ | 441,273 | |
Inventories consisted of the following (in thousands):
| | | | | | | | | | | | | | |
| | March 31,
2026 | | December 31,
2025 |
| Raw materials | | $ | 21,902 | | | $ | 21,869 | |
| Work-in-process | | 35,039 | | | 30,920 | |
| Finished goods | | 113,365 | | | 144,454 | |
Total inventories | | 170,306 | | | 197,243 | |
Less long-term portion (1) | | (14,839) | | | (20,768) | |
Total inventories, current | | $ | 155,467 | | | $ | 176,475 | |
(1) Long-term portion of inventories represents inventory expected to remain on hand beyond one year and therefore is included in prepaid expenses and other assets in the condensed consolidated balance sheets.
Prepaid expenses and other assets consisted of the following (in thousands):
| | | | | | | | | | | | | | |
| | March 31,
2026 | | December 31,
2025 |
| Prepaid manufacturing expenses | | $ | 75,285 | | | $ | 71,925 | |
| | | | |
| Other prepaid expenses | | 16,460 | | | 14,563 | |
Long-term inventories | | 14,839 | | | 20,768 | |
Other assets | | 15,289 | | | 10,934 | |
Total prepaid expenses and other assets | | 121,873 | | | 118,190 | |
Less long-term portion | | (50,915) | | | (53,551) | |
Total prepaid expenses and other assets, current | | $ | 70,958 | | | $ | 64,639 | |
Prepaid manufacturing expenses include raw materials, slot reservation fees and other amounts paid to contract manufacturing organizations. Such amounts are reclassified to work-in-process inventory as materials are used or the contract manufacturing organization services are complete.
Property and equipment, net consisted of the following (in thousands):
| | | | | | | | | | | | | | |
| | March 31,
2026 | | December 31,
2025 |
| Research equipment | | $ | 19,195 | | | $ | 18,305 | |
| Manufacturing equipment | | 45,053 | | | 41,863 | |
| Computer and office equipment | | 8,435 | | | 8,034 | |
Internal-use software | | 1,945 | | | 1,878 | |
| Leasehold improvements | | 7,521 | | | 7,521 | |
Subtotal | | 82,149 | | | 77,601 | |
| Accumulated depreciation and amortization | | (35,215) | | | (33,159) | |
Subtotal | | 46,934 | | | 44,442 | |
| Right of use of assets | | 35,296 | | | 37,695 | |
Total property and equipment, net | | $ | 82,230 | | | $ | 82,137 | |
Depreciation and amortization expense was approximately $4.2 million and $2.7 million, inclusive of ROU asset amortization of $2.6 million and $1.5 million for the three months ended March 31, 2026 and 2025, respectively.
Accrued expenses consisted of the following (in thousands):
| | | | | | | | | | | | | | |
| | March 31,
2026 | | December 31,
2025 |
| | | | |
| Accrued compensation and payroll taxes | | $ | 18,135 | | | $ | 28,621 | |
| Accrued outsourced manufacturing expenses | | 9,107 | | | 11,775 | |
Taxes payable | | 48,014 | | | 44,148 | |
| Product returns and sales allowance | | 44,546 | | | 46,594 | |
| Other accrued expenses | | 94,598 | | | 104,349 | |
| Lease liability | | 32,417 | | | 34,569 | |
Total accrued expenses | | 246,817 | | | 270,056 | |
| Less long-term portion | | (106,448) | | | (113,863) | |
Total accrued expenses, current | | $ | 140,369 | | | $ | 156,193 | |
Expense associated with the accretion of the lease liabilities was approximately $0.7 million and $0.5 million for the three months ended March 31, 2026 and 2025, respectively. Total lease expense for the three months ended March 31, 2026 and 2025 was $3.1 million and $2.0 million, respectively.
Cash paid for amounts related to leases for the three months ended March 31, 2026 and 2025 was $2.9 million and $1.8 million, respectively.
7. Goodwill and Intangible Assets, net
Goodwill
A summary of the activity impacting goodwill is presented below (in thousands):
| | | | | | | | |
Balance as of December 31, 2025 | | $ | 580,360 | |
Measurement period adjustment | | 1,963 | |
Balance as of March 31, 2026 | | $ | 582,323 | |
Intangible Assets, net
Our acquired intangible assets are amortized using the straight-line method over their estimated useful lives of seven to fifteen years. The following table shows the cost, accumulated amortization and weighted average useful life in years for our acquired intangible assets as of March 31, 2026 (in thousands).
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Weighted Average Useful Life (in years) | | Gross Carrying Value | | Accumulated Amortization | | Net Carrying Value |
| Auto-injector technology platform | | 7 | | $ | 402,000 | | | $ | 221,378 | | | $ | 180,622 | |
| XYOSTED proprietary product | | 10 | | 136,200 | | | 52,503 | | | 83,697 | |
| Hypercon developed technology platform | | 15 | | 230,000 | | | 5,665 | | | 224,335 | |
Customer relationships (1) | | 15 | | 470,000 | | | 11,576 | | | 458,424 | |
| Trade name | | 15 | | 5,000 | | | 123 | | | 4,877 | |
| Total intangible assets, net | | | | $ | 1,243,200 | | | $ | 291,245 | | | $ | 951,955 | |
(1) A partner is currently evaluating the potential return of a target. Through discussions with the partner, we expect the partner will make a decision by September 2026. If the partner does decide to return the target in the future without nominating an alternative target with similar future cash flows, we would be required to evaluate the impact to our acquired customer relationship intangibles associated with the Elektrofi acquisition, along with other identified intangibles and goodwill of the reporting unit. Such assessment could result in a material interim impairment charge to intangibles and/or goodwill, depending on the facts and circumstances.
The following table shows the cost, accumulated amortization and weighted average useful life in years for our acquired intangible assets as of December 31, 2025 (in thousands).
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Weighted Average Useful Life (in years) | | Gross Carrying Value | | Accumulated Amortization | | Net Carrying Value |
Auto-injector technology platform | | 7 | | $ | 402,000 | | | $ | 207,021 | | | $ | 194,979 | |
| XYOSTED proprietary product | | 10 | | 136,200 | | | 49,098 | | | 87,102 | |
Hypercon developed technology platform | | 15 | | 230,000 | | | 1,831 | | | 228,169 | |
| Customer relationships | | 15 | | 470,000 | | | 3,743 | | | 466,257 | |
Trade name | | 15 | | 5,000 | | | 40 | | | 4,960 | |
Total intangible assets, net | | | | $ | 1,243,200 | | | $ | 261,733 | | | $ | 981,467 | |
Estimated future annual amortization of finite-lived intangible assets is shown in the following table (in thousands). Actual amortization expense to be reported in future periods could differ from these estimates as a result of acquisitions, divestitures, and asset impairments, among other factors.
| | | | | | | | |
| Year | | Amortization Expense |
Remainder of 2026 | | $ | 88,535 | |
2027 | | 118,049 | |
2028 | | 118,049 | |
2029 | | 83,314 | |
2030 | | 60,620 | |
| Thereafter | | 483,388 | |
| Total | | $ | 951,955 | |
8. Long-Term Debt, Net
0.875% Convertible Notes due 2032
In November 2025, we completed the sale of $750.0 million in aggregate principal amount of 0.875% Convertible Senior Notes due 2032 (the “2032 Convertible Notes”). The net proceeds from the issuance of the 2032 Convertible Notes, after deducting the initial purchasers’ fee of $15.0 million, was approximately $735.0 million. We also incurred additional debt issuance costs totaling $0.5 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2032 Convertible Notes pay interest semi-annually in arrears on May 15th and November 15th of each year at an annual rate of 0.875%. The 2032 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2032 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness, and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2032 Convertible Notes have a maturity date of November 15, 2032.
Holders may convert their 2032 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2026, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2032 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, May 15, 2032, until the close of business on the second scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2032 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal, and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2032 Convertible Notes is 11.4683 shares of common stock per $1,000 in principal amount of 2032 Convertible Notes, equivalent to a conversion price of approximately $87.20 per share of our common stock. The conversion rate is subject to adjustment in some events but will not be adjusted for any accrued or unpaid interest.
As of March 31, 2026, we were in compliance with all covenants.
2032 Capped Call Transactions
In connection with the offering of the 2032 Convertible Notes, we entered into capped call transactions with certain counterparties (the “2032 Capped Call Transactions”). The 2032 Capped Call Transactions are expected generally to reduce potential dilution to holders of our common stock upon conversion of the 2032 Convertible Notes or at our election (subject to certain conditions) offset any cash payments we are required to make in excess of the principal amount of such converted 2032 Convertible Notes. The cap price of the 2032 Capped Call Transactions is initially $136.78 per share of common stock, representing a premium of 100% above the last reported sale price of $68.30 per share of common stock on November 5, 2025, and is subject to certain adjustments under the terms of the 2032 Capped Call Transactions. As of March 31, 2026, no 2032 Capped Call Transactions had been exercised.
Pursuant to their terms, the 2032 Capped Call Transactions qualify for classification within stockholders’ equity in our condensed consolidated balance sheets, and their fair value is not remeasured and adjusted as long as they continue to qualify for stockholders’ equity classification. We paid approximately $106.8 million for the 2032 Capped Call Transactions, including applicable transaction costs, which was recorded as a reduction to additional paid-in capital in our condensed consolidated balance sheets. The 2032 Capped Call Transactions are separate transactions entered into by us with certain counterparties, are not part of the terms of the 2032 Convertible Notes, and do not affect any holder’s rights under the 2032 Convertible Notes. Holders of the 2032 Convertible Notes do not have any rights with respect to the 2032 Capped Call Transactions.
0.00% Convertible Notes due 2031
In November 2025, we completed the sale of $750.0 million in aggregate principal amount of 0.00% Convertible Senior Notes due 2031 (the “2031 Convertible Notes”). The net proceeds from the issuance of the 2031 Convertible Notes, after deducting the initial purchasers’ fee of $15.0 million, was approximately $735.0 million. We also incurred additional debt issuance costs totaling $0.5 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2031 Convertible Notes will not bear regular interest and the principal amount of the 2031 Convertible Notes will not accrete. The 2031 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2031 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2031 Convertible Notes have a maturity date of February 15, 2031.
Holders may convert their 2031 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2026, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2031 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, August 15, 2030, until the close of business on the scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2031 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2031 Convertible Notes is 11.4683 shares of common stock per $1,000 in principal amount of 2031 Convertible Notes, equivalent to a conversion price of approximately $87.20 per share of our common stock. The conversion rate is subject to adjustment.
As of March 31, 2026, we were in compliance with all covenants.
2031 Capped Call Transactions
In connection with the offering of the 2031 Convertible Notes, we entered into capped call transactions with certain counterparties (the “2031 Capped Call Transactions”). The 2031 Capped Call Transactions are expected generally to reduce potential dilution to holders of our common stock upon conversion of the 2031 Convertible Notes or at our election (subject to certain conditions) offset any cash payments we are required to make in excess of the principal amount of such converted 2031 Convertible Notes. The cap price of the 2031 Capped Call Transactions is initially $136.78 per share of common stock, representing a premium of 100% above the last reported sale price of $68.30 per share of common stock on November 5, 2025, and is subject to certain adjustments under the terms of the 2031 Capped Call Transactions. As of March 31, 2026, no 2031 Capped Call Transactions had been exercised.
Pursuant to their terms, the 2031 Capped Call Transactions qualify for classification within stockholders’ equity in our condensed consolidated balance sheets, and their fair value is not remeasured and adjusted as long as they continue to qualify for stockholders’ equity classification. We paid approximately $104.0 million for the 2031 Capped Call Transactions, including applicable transaction costs, which was recorded as a reduction to additional paid-in capital in our condensed consolidated balance sheets. The 2031 Capped Call Transactions are separate transactions entered into by us with certain counterparties, are not part of the terms of the 2031 Convertible Notes, and do not affect any holder’s rights under the 2031 Convertible Notes. Holders of the 2031 Convertible Notes do not have any rights with respect to the 2031 Capped Call Transactions.
1.00% Convertible Notes due 2028
In August 2022, we completed the sale of $720.0 million in aggregate principal amount of 1.00% Convertible Senior Notes due 2028 (the “2028 Convertible Notes”). The net proceeds from the issuance of the 2028 Convertible Notes, after deducting the initial purchasers’ fee of $18.0 million, was approximately $702.0 million. We also incurred additional debt issuance costs totaling $1.0 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2028 Convertible Notes pay interest semi-annually in arrears on February 15th and August 15th of each year at an annual rate of 1.00%. The 2028 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2028 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness, and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2028 Convertible Notes have a maturity date of August 15, 2028.
Holders may convert their 2028 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on December 31, 2022, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2028 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, February 15, 2028 until the close of business on the second scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2028 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal, and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2028 Convertible Notes is 17.8517 shares of common stock per $1,000 in principal amount of 2028 Convertible Notes, equivalent to a conversion price of approximately $56.02 per share of our common stock. The conversion rate is subject to adjustment in some events but will not be adjusted for any accrued or unpaid interest.
In connection with the offering of the 2032 Convertible Notes and 2031 Convertible Notes, we used a portion of the net proceeds of the offering to enter into privately negotiated agreements with certain holders of the outstanding 2028 Convertible Notes to repurchase their 2028 Convertible Notes for cash. In connection with the repurchases, we paid approximately $342.9 million in cash, which included a premium, inducement expense and accrued interest.
As of March 31, 2026, we were in compliance with all covenants.
2028 Capped Call Transactions
In connection with the offering of the 2028 Convertible Notes, we entered into capped call transactions with certain counterparties (the “2028 Capped Call Transactions”). The 2028 Capped Call Transactions are expected generally to reduce potential dilution to holders of our common stock upon conversion of the 2028 Convertible Notes or at our election (subject to certain conditions) offset any cash payments we are required to make in excess of the principal amount of such converted 2028 Convertible Notes. The cap price of the 2028 Capped Call Transactions is initially $75.4075 per share of common stock, representing a premium of 75% above the last reported sale price of $43.09 per share of common stock on August 15, 2022, and is subject to certain adjustments under the terms of the 2028 Capped Call Transactions. As of March 31, 2026, no 2028 Capped Call Transactions had been exercised.
Pursuant to their terms, the 2028 Capped Call Transactions qualify for classification within stockholders’ equity in our condensed consolidated balance sheets, and their fair value is not remeasured and adjusted as long as they continue to qualify for stockholders’ equity classification. We paid approximately $69.1 million for the 2028 Capped Call Transactions, including applicable transaction costs, which was recorded as a reduction to additional paid-in capital in our condensed consolidated balance sheets. The 2028 Capped Call Transactions are separate transactions entered into by us with certain counterparties, are not part of the terms of the 2028 Convertible Notes, and do not affect any holder’s rights under the 2028 Convertible Notes. Holders of the 2028 Convertible Notes do not have any rights with respect to the 2028 Capped Call Transactions.
0.25% Convertible Notes due 2027
In March 2021, we completed the sale of $805.0 million in aggregate principal amount of 0.25% Convertible Senior Notes due 2027 (the “2027 Convertible Notes”). The net proceeds from the issuance of the 2027 Convertible Notes, after deducting the initial purchasers’ fee of $20.1 million, was approximately $784.9 million. We also incurred additional debt issuance costs totaling $0.4 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2027 Convertible Notes pay interest semi-annually in arrears on March 1st and September 1st of each year at an annual rate of 0.25%. The 2027 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2027 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2027 Convertible Notes have a maturity date of March 1, 2027.
Holders may convert their 2027 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on June 30, 2021, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2027 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, September 1, 2026 until the close of business on the scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2027 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal, and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2027 Convertible Notes is 12.9576 shares of common stock per $1,000 in principal amount of 2027 Convertible Notes, equivalent to a conversion price of approximately $77.17 per share of our common stock. The conversion rate is subject to adjustment.
In connection with the offering of the 2032 Convertible Notes and 2031 Convertible Notes, we used a portion of the net proceeds of the offering to enter into privately negotiated agreements with certain holders of the outstanding 2027 Convertible Notes to repurchase their 2027 Convertible Notes for cash. In connection with the repurchases, we paid approximately $676.8 million in cash, which included a premium, inducement expense and accrued interest.
As of March 31, 2026, we were in compliance with all covenants.
Net Carrying Amounts of our Convertible Notes
The carrying amount and fair value of our Convertible Notes were as follows (in thousands).
| | | | | | | | | | | | | | |
| | March 31,
2026 | | December 31,
2025 |
| Principal amount | | | | |
| | | | |
| 2027 Convertible Notes | | $ | 209,575 | | | $ | 209,575 | |
| 2028 Convertible Notes | | 469,999 | | | 469,999 | |
| 2031 Convertible Notes | | 750,000 | | | 750,000 | |
| 2032 Convertible Notes | | 750,000 | | | 750,000 | |
Total principal amount | | $ | 2,179,574 | | | $ | 2,179,574 | |
| | | | |
| Unamortized debt discount | | | | |
| | | | |
| 2027 Convertible Notes | | $ | (833) | | | $ | (1,058) | |
| 2028 Convertible Notes | | (5,040) | | | (5,561) | |
2031 Convertible Notes | | (14,378) | | | (15,108) | |
2032 Convertible Notes | | (14,684) | | | (15,217) | |
| Total unamortized debt discount | | $ | (34,935) | | | $ | (36,944) | |
| | | | |
| Carrying amount | | | | |
| | | | |
| 2027 Convertible Notes | | $ | 208,742 | | | $ | 208,517 | |
| 2028 Convertible Notes | | 464,959 | | | 464,438 | |
2031 Convertible Notes | | 735,622 | | | 734,892 | |
2032 Convertible Notes | | 735,316 | | | 734,783 | |
| Total carrying amount | | $ | 2,144,639 | | | $ | 2,142,630 | |
| | | | |
Fair value based on trading levels (Level 2) | | | | |
| | | | |
| 2027 Convertible Notes | | $ | 218,048 | | | $ | 229,736 | |
| 2028 Convertible Notes | | 607,107 | | | 624,690 | |
2031 Convertible Notes | | 733,433 | | | 755,655 | |
2032 Convertible Notes | | 742,245 | | | 764,213 | |
| Total fair value of outstanding notes | | $ | 2,300,833 | | | $ | 2,374,294 | |
| | | | |
| | | | |
| | | | |
| | | | |
| | | | |
The following table summarizes the components of interest expense and the effective interest rates for each of our Convertible Notes (in thousands).
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
| | 2026 | | 2025 | | | | |
| Coupon interest | | | | | | | | |
| | | | | | | | |
| 2027 Convertible Notes | | $ | 131 | | $ | 503 | | | | |
| 2028 Convertible Notes | | 1,175 | | 1,800 | | | | |
| 2032 Convertible Notes | | 1,641 | | — | | | | |
| Total coupon interest | | $ | 2,947 | | $ | 2,303 | | | | |
| | | | | | | | |
| Amortization of debt discount | | | | | | | | |
| | | | | | | | |
| 2027 Convertible Notes | | $ | 226 | | $ | 862 | | | | |
| 2028 Convertible Notes | | 521 | | 787 | | | | |
| 2031 Convertible Notes | | 730 | | — | | | | |
2032 Convertible Notes | | 532 | | — | | | | |
| Total amortization of debt discount | | $ | 2,009 | | $ | 1,649 | | | | |
| | | | | | | | |
| Interest expense | | | | | | | | |
| | | | | | | | |
| 2027 Convertible Notes | | $ | 357 | | $ | 1,365 | | | | |
| 2028 Convertible Notes | | 1,696 | | 2,587 | | | | |
2031 Convertible Notes | | 730 | | — | | | | |
2032 Convertible Notes | | 2,173 | | — | | | | |
| Total interest expense | | $ | 4,956 | | $ | 3,952 | | | | |
| | | | | | | | |
| Effective interest rates | | | | | | | | |
| | | | | | | | |
| 2027 Convertible Notes | | 0.7 | % | | 0.7 | % | | | | |
| 2028 Convertible Notes | | 1.5 | % | | 1.5 | % | | | | |
2031 Convertible Notes | | 0.4 | % | | 0.4 | % | | | | |
2032 Convertible Notes | | 1.2 | % | | 1.2 | % | | | | |
Revolving Credit and Term Loan Facilities
In May 2022, we entered into a credit agreement, which was subsequently amended (i) in August 2022 (the “First Amendment”), (ii) in March 2023 (the “Second Amendment”) and (iii) in November 2025 (the “Third Amendment”) with Bank of America, N.A., as Administrative Agent, Swing Line Lender and an L/C Issuer, and the other lenders and L/C Issuers party thereto (the credit agreement as amended by the First Amendment, the Second Amendment and the Third Amendment, the “2022 Credit Agreement”), evidencing a credit facility (the “2022 Facility”) that provides for a $750 million revolving credit facility (the “Amended Revolving Credit Facility”). The Amended Revolving Credit Facility will mature on the earlier of (a) November 5, 2030 and (b) the Springing Revolver Maturity Date (as defined in the 2022 Credit Agreement), unless the Amended Revolving Credit Facility is extended prior to such date in accordance with the 2022 Credit Agreement.
Borrowings under the Amended Revolving Credit Facility bear interest at a rate equal to an applicable margin plus: (a) the applicable Term SOFR (as defined in the Credit Agreement) rate, or (b) a base rate determined by reference to the highest of (1) the federal funds effective rate plus 0.50%, (2) the Bank of America prime rate, (3) the Term SOFR rate for an interest period of one month plus 1.00%, and (4) 1.00%. The applicable margin for the Amended Revolving Credit Facility ranges, based on our consolidated total net leverage ratio, from 0.25% to 1.25% in the case of base rate loans and from 1.25% to 2.25% in the case of Term SOFR rate loans. In addition to paying interest on the outstanding principal under the Amended Revolving Credit Facility, we will pay (i) a commitment fee in respect of the unutilized commitments thereunder and (ii) customary letter of credit fees and agency fees.
The margin for the 2022 Facility ranges, based on our consolidated total net leverage ratio, from 0.25% to 1.25% in the case of base rate loans and from 1.25% to 2.25% in the case of Term SOFR rate loans. In addition to paying interest on the outstanding principal under the 2022 Facility, we will pay (i) a commitment fee in respect of the unutilized commitments thereunder and (ii) customary letter of credit fees and agency fees. The commitment fees range from 0.15% to 0.35% per annum based on our consolidated net leverage ratio.
As of March 31, 2026, the Revolving Credit Facility was undrawn. We incurred a total of $7.3 million in third-party costs related to the 2022 Credit Agreement which are recorded as debt issuance cost within prepaid expenses and other assets in our condensed consolidated balance sheets. As of March 31, 2026, the unamortized debt issuance cost related to the revolving credit facility was $4.4 million.
9. Stockholders’ Equity
Share-based Compensation
The following table summarizes share-based compensation expense included in our condensed consolidated statements of income related to share-based awards (in thousands):
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
| | | 2026 | | 2025 | | | | |
| Research and development | | $ | 5,044 | | | $ | 3,229 | | | | | |
| Selling, general and administrative | | 11,593 | | | 7,444 | | | | | |
| Total share-based compensation expense | | $ | 16,637 | | | $ | 10,673 | | | | | |
Share-based compensation expense by type of share-based award was as follows (in thousands):
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
| | | 2026 | | 2025 | | | | |
| Stock options | | $ | 5,541 | | | $ | 3,809 | | | | | |
| RSUs, PSUs and ESPP | | 11,096 | | | 6,864 | | | | | |
| Total share-based compensation expense | | $ | 16,637 | | | $ | 10,673 | | | | | |
We granted stock options to purchase approximately 0.3 million and 0.4 million shares of common stock during the three months ended March 31, 2026 and 2025, respectively. The exercise price of stock options granted is equal to the closing price of the common stock on the date of grant. The fair value of each option award is estimated on the date of grant using the Black-Scholes-Merton option pricing model (“Black-Scholes Model”). Expected volatility is based on historical volatility of our common stock. The expected term of options granted is based on analyses of historical employee termination rates and option exercises. The risk-free interest rate is based on the U.S. Treasury yield for a period consistent with the expected term of the option in effect at the time of the grant. The dividend yield assumption is based on the expectation of no future dividend payments. The assumptions used in the Black-Scholes Model were as follows:
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended March 31, | | |
| | | 2026 | | 2025 | | | | |
| Expected volatility | | 29.91 - 41.95% | | 39.68 - 39.68% | | | | |
| Average expected term (in years) | | 4.7 | | 4.8 | | | | |
| Risk-free interest rate | | 3.62 - 3.83% | | 4.34 - 4.34% | | | | |
| Expected dividend yield | | — | | | — | | | | | |
In February 2021, our Board of Directors approved our 2021 ESPP and our stockholders approved the plan in May 2021. The 2021 ESPP enables eligible employees to purchase shares of our common stock at the end of each offering period at a price equal to 85% of the fair market value of the shares on the first business day or the last business day of the offering period, whichever is lower. Share purchases are funded through payroll deduction of at least 1% and up to 15% of an employee’s compensation for each payroll period, and no employee may purchase shares under the 2021 ESPP that exceeds $25,000 worth of our common stock for a calendar year. As of March 31, 2026, 2.5 million shares were available for future purchase. The offering period is generally for a six-month period. Offering periods shall commence on or about the sixteenth day of June and December of each year and end on or about the fifteenth day of the next December and June, respectively, occurring thereafter.
Total unrecognized estimated compensation cost by type of award and the weighted-average remaining requisite service period over which such expense is expected to be recognized was as follows (in thousands, unless otherwise noted):
| | | | | | | | | | | | | | |
| | March 31, 2026 |
| | | Unrecognized
Expense | | Remaining Weighted-Average Recognition Period (in years) |
| Stock options | | $ | 36,478 | | | 2.44 |
| | | | |
| RSUs | | 75,290 | | | 2.83 |
| PSUs | | 28,145 | | | 2.25 |
| ESPP | | 112 | | | 0.16 |
Share Repurchases
In February 2024, our Board of Directors authorized a new capital return program to repurchase up to $750.0 million of our outstanding common stock. During 2024, we repurchased approximately 4.7 million shares for $250.0 million at an average price of $53.95 per share.
In May 2025, we announced the second $250.0 million share repurchase tranche under the $750.0 million approved program from February 2024. The second $250.0 million share repurchase was completed in June 2025, resulting in a total purchase of approximately 4.8 million shares at an average price of $52.09 per share.
In June 2025, we initiated the third $250.0 million share repurchase tranche under the $750.0 million approved program from February 2024. As of September 30, 2025, $92.3 million has been used to repurchase approximately 1.7 million shares at an average price of $52.89 per share.
All shares repurchased under our capital return programs have been retired and have resumed their status of authorized and unissued shares.
10. Earnings per share
Basic earnings per share is computed by dividing net income for the period by the weighted average number of common shares outstanding during the period, without consideration for common stock equivalents. Outstanding stock options, unvested RSUs, unvested PSUs, common shares expected to be issued under our ESPP and the Convertible Notes are considered common stock equivalents and are only included in the calculation of diluted earnings per common share when net income is reported and their effect is dilutive.
Potentially dilutive common shares issuable upon vesting of stock options, RSUs and PSUs are determined using the average share price for each period under the treasury stock method. Potentially dilutive common shares issuable upon conversion of the Convertible Notes are determined using the if-converted method. Since we have committed to settle the principal amount of the Convertible Notes in cash upon conversion only, the number of shares for the conversion spread will be included as a dilutive common stock equivalent.
A reconciliation of the numerators and the denominators of the basic and diluted earnings per share computations is as follows (in thousands, except per share amounts):
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
| | | 2026 | | 2025 | | | | |
| Numerator | | | | | | | | |
| Net income | | $ | 150,049 | | | $ | 118,095 | | | | | |
| Denominator | | | | | | | | |
| Weighted average common shares outstanding for basic earnings per share | | 118,144 | | | 123,215 | | | | | |
| Dilutive potential common stock outstanding | | | | | | | | |
| Stock options | | 1,934 | | | 2,071 | | | | | |
| RSUs, PSUs and ESPP | | 1,074 | | | 900 | | | | | |
| Convertible Notes | | 1,723 | | | 458 | | | | | |
| Weighted average common shares outstanding for diluted earnings per share | | 122,875 | | | 126,644 | | | | | |
| Earnings per share | | | | | | | | |
| Basic | | $ | 1.27 | | | $ | 0.96 | | | | | |
| Diluted | | $ | 1.22 | | | $ | 0.93 | | | | | |
Shares which have been excluded from the calculation of diluted earnings per common share because their effect was anti-dilutive include the following (shares in millions):
| | | | | | | | | | | | | | | | | | |
| | Three Months Ended | |
| | March 31, | | |
| | | 2026 | | 2025 | | | | |
Anti-dilutive securities (1) | | 27.9 | | | 24.1 | | | | | |
(1) The anti-dilutive securities include outstanding stock options, unvested RSUs, unvested PSUs, common shares expected to be issued under our ESPP and Convertible Notes.
11. Commitments and Contingencies
From time to time, we may be involved in disputes, including litigation, relating to claims arising out of operations in the normal course of our business. Any of these claims could subject us to costly legal expenses and, while we generally believe that we have adequate insurance to cover many different types of liabilities, our insurance carriers may deny coverage or our policy limits may be inadequate to fully satisfy any damage awards or settlements. If this were to happen, the payment of any such awards could have a material adverse effect on our condensed consolidated statements of income and balance sheets. Additionally, any such claims, whether or not successful, could damage our reputation and business. We currently are not a party to any legal proceedings, the adverse outcome of which, in our opinion, individually or in the aggregate, would have a material adverse effect on our condensed consolidated statements of income or balance sheets.
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations
As used in this Quarterly Report on Form 10-Q, unless the context suggests otherwise, references to “Halozyme,” “the Company,” “we,” “our,” “ours,” and “us” refer to Halozyme Therapeutics, Inc., its wholly owned subsidiaries, Halozyme, Inc., Antares Pharma, Inc., Antares Pharma, Inc.’s two wholly-owned Swiss subsidiaries, Antares Pharma IPL AG and Antares Pharma GmbH, Halozyme Hypercon, Inc., Halozyme Hypercon, Inc.’s wholly-owned subsidiary, Elektrofi Security Corp., and Halozyme Surf Bio, Inc. References to “Notes” refer to the notes to the condensed consolidated financial statements included herein (refer to Item 1 of Part I of this Quarterly Report on Form 10-Q).
The following information should be read in conjunction with the condensed consolidated financial statements and notes thereto included in Item 1 of this Quarterly Report on Form 10-Q, as well as the audited financial statements and notes thereto and Management’s Discussion and Analysis of Financial Condition and Results of Operations for the year ended December 31, 2025, included in our Annual Report on Form 10-K for the year ended December 31, 2025. Past financial or operating performance is not necessarily a reliable indicator of future performance, and our historical performance should not be used to anticipate results or future period trends.
This Quarterly Report on Form 10-Q contains “forward-looking statements” within the meaning of the “safe harbor” of the Private Securities Litigation Reform Act of 1995, provisions of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. All statements, other than statements of historical fact, included herein, including without limitation those regarding our future product development and regulatory events and goals, product collaborations, our business intentions and financial estimates and anticipated results, are, or may be deemed to be, forward-looking statements. Words such as “expect,” “anticipate,” “intend,” “plan,” “believe,” “seek,” “estimate,” “think,” “may,” “could,” “will,” “would,” “should,” “continue,” “potential,” “likely,” “opportunity,” “project” and similar expressions or variations of such words are intended to identify forward-looking statements, but are not the exclusive means of identifying forward-looking statements in this Quarterly Report on Form 10-Q. Additionally, statements concerning future matters such as the development or regulatory approval of new partner products, enhancements of existing products or technologies, uncertainties in tariffs, trade and pharmaceutical pricing policies, tax legislation, timing and success of the launch of new products by us and our partners, third-party performance under key collaboration agreements, the ability of our bulk drug and device part manufacturers to provide adequate supply for our partners, revenue, expense, cash burn levels and our ability to make timely repayments of debt, anticipated amounts and timing of share repurchases, anticipated profitability and expected trends and other statements regarding our plans and matters that are not historical are forward-looking statements.
Although forward-looking statements in this Quarterly Report on Form 10-Q reflect the good faith judgment of our management, such statements reflect management’s current forecast of certain aspects of our future business and are based on currently available operating, financial and competitive information. Consequently, forward-looking statements are inherently subject to risks, uncertainties and assumptions that could cause actual results and outcomes to differ materially from the results and outcomes discussed in or anticipated by the forward-looking statements. Factors that could cause or contribute to such differences in results and outcomes include without limitation those discussed under the Risk Factors set forth in our most recent Annual Report on Form 10-K referred to below under the section entitled “Risks Factors” and elsewhere in this Quarterly Report on Form 10-Q and our most recent Annual Report on Form 10-K. Readers are urged not to place undue reliance on these forward-looking statements, which speak only as of the date of this Quarterly Report on Form 10-Q. We undertake no obligation to revise or update any forward-looking statements in order to reflect any event or circumstance that may arise after the date of this Quarterly Report on Form 10-Q. Readers are urged to carefully review and consider the various disclosures made in this Quarterly Report on Form 10-Q, which attempt to advise interested parties of the risks and factors that may affect our business, financial condition, results of operations and prospects.
Overview
Halozyme Therapeutics, Inc. is a biopharmaceutical company advancing disruptive solutions to improve patient experiences and outcomes for emerging and established therapies.
As the innovators of ENHANZE® drug delivery technology (“ENHANZE”) with our proprietary enzyme, rHuPH20, our commercially validated solution is used to facilitate the subcutaneous (“SC”) delivery of injected drugs and fluids, with the goal of improving the patient experience with rapid SC delivery and reduced treatment burden. We license our technology to biopharmaceutical companies to collaboratively develop products that combine ENHANZE with our partners’ proprietary compounds. We are also developing partner products with Hypercon™ drug delivery technology (“Hypercon technology”) and developing the Surf Bio drug delivery technology to expand the breadth of our drug delivery technology portfolio. Hypercon technology is an innovative microparticle technology that has been demonstrated in non-clinical testing to enable hyperconcentration of drugs and biologics and reduce the injection volume for the same dosage and potentially expanding
opportunities for at-home and health care provider administration. The Surf Bio hyperconcentration technology has similarly been demonstrated in non-clinical studies to achieve biologic concentrations of up to 500 mg/mL, for potential delivery in a single auto-injector shot for at-home or in a health care provider’s office use. We also develop, manufacture and commercialize, for ourselves or with our partners, drug-device combination products using our advanced auto-injector technologies that are designed to provide commercial or functional advantages such as improved convenience, reliability and tolerability, and enhanced patient comfort and adherence.
Our ENHANZE partners’ approved products and product candidates are based on rHuPH20, our patented recombinant human hyaluronidase enzyme. rHuPH20 works by breaking down hyaluronan, a naturally occurring carbohydrate that is a major component of the extracellular matrix of the SC space. This temporarily reduces the barrier to bulk fluid flow allowing for improved and more rapid SC delivery of high dose, high volume injectable biologics, such as monoclonal antibodies and other large therapeutic molecules, as well as small molecules and fluids. We refer to the application of rHuPH20 to facilitate the delivery of other drugs or fluids as ENHANZE. We license our ENHANZE technology to form collaborations with biopharmaceutical companies that develop and/or market drugs requiring or benefiting from injection via the SC route of administration. In the development of proprietary intravenous (“IV”) drugs combined with our ENHANZE technology, data have been generated supporting the potential for ENHANZE to reduce patient treatment burden, as a result of shorter duration of SC administration with ENHANZE compared to IV administration. ENHANZE may enable fixed-dose SC dosing compared to weight-based dosing typically required for IV administration, extend the dosing interval for drugs that are already administered subcutaneously and potentially allow for lower rates of infusion-related reactions. ENHANZE may enable more flexible treatment options such as home administration by a healthcare professional or potentially the patient or caregiver. Lastly, certain proprietary drugs co-formulated with ENHANZE have been granted additional exclusivity, extending the patent life of the product beyond the patent expiry of the proprietary IV drug.
We currently have ENHANZE collaborations and licensing agreements with F. Hoffmann-La Roche, Ltd. and Hoffmann-La Roche, Inc. (“Roche”), Takeda Pharmaceuticals International AG and Baxalta US Inc. (“Takeda”), Pfizer Inc. (“Pfizer”), Janssen Biotech, Inc. (“Janssen”), AbbVie, Inc. (“AbbVie”), Eli Lilly and Company (“Lilly”), Bristol Myers Squibb Company (“BMS”), argenx BVBA (“argenx”), ViiV Healthcare (the global specialist HIV Company majority owned by GlaxoSmithKline) (“ViiV”), Chugai Pharmaceutical Co., Ltd. (“Chugai”), Acumen Pharmaceuticals, Inc. (“Acumen”), Merus N.V. (“Merus”), Skye Bioscience, Inc. (“Skye Bioscience”) and GlaxoSmithKline plc (“GSK”). In addition to receiving upfront licensing fees from our ENHANZE collaborations, we are entitled to receive event and sales-based milestone payments, revenues from the sale of bulk rHuPH20 and royalties from commercial sales of approved partner products co-formulated with ENHANZE. We currently earn royalties from the sales of ten commercial products including sales of five commercial products from the Roche collaboration, two commercial products from the Janssen collaboration and one commercial product from each of the Takeda, argenx and BMS collaborations.
Through our recent acquisition of Elektrofi, Inc. (“Elektrofi”), subsequently renamed Halozyme Hypercon, Inc. (“Hypercon”), we have Hypercon collaboration and license agreements with Janssen, Lilly, argenx, Vertex Pharmaceuticals Incorporated (“Vertex”) and Oruka Therapeutics, Inc. (“Oruka”). In addition to receiving upfront license fees from our Hypercon collaborations, we are entitled to receive event and sales-based milestone payments and royalties from commercial sales for approved partner products co-formulated with Hypercon. A partner is currently evaluating the potential return of a target. Through discussions with the partner, we expect the partner will make a decision by September 2026. If the partner does decide to return the target in the future without nominating an alternative target with similar future cash flows, we would be required to evaluate the impact to our acquired customer relationship intangibles associated with the Elektrofi acquisition, along with other identified intangibles and goodwill of the reporting unit. Such assessment could result in a material interim impairment charge to intangibles and/or goodwill, depending on the facts and circumstances.
We have commercialized auto-injector products with Teva Pharmaceutical Industries, Ltd. (“Teva”). We have development programs including our auto-injectors with McDermott Laboratories Limited, an affiliate of Viatris Inc. (“Viatris”).
Our commercial portfolio of proprietary products includes Hylenex®, utilizing rHuPH20, and XYOSTED®, utilizing our auto-injector technology.
Our first quarter of 2026 and recent key events are as follows:
Corporate
•In May 2026, we announced a new share repurchase program to repurchase up to $1 billion of our outstanding common stock by December 31, 2028, with an expectation of buying back at least $400 million of shares in 2026.
Partners
•In May 2026, argenx announced U.S. Food and Drug Administration (“FDA”) approval of a supplemental Biologics License Application for VYVGART Hytrulo with ENHANZE for the treatment of adult patients with generalized myasthenia gravis including all serotypes – anti-AChR-Ab positive, anti-MuSK-Ab positive, anti-LRP4-Ab positive, and triple seronegative.
•In May 2026, we and GSK entered into a global collaboration and license agreement for ENHANZE with multiple oncology targets, including the first potential application in antibody-drug conjugates. Under the terms of the agreement, GSK will make an upfront payment and potential future milestone payments and royalties on net sales of products developed with ENHANZE.
•In May 2026, we and Oruka entered into a global exclusive collaboration and license agreement for our Hypercon technology for use with ORKA-001, in development for psoriasis and related inflammatory diseases and one additional target. Under the terms of the agreement, Oruka will make an upfront payment and potential future milestone payments and mid-single digit royalties on net sales of products developed using the Hypercon technology.
•In May 2026, Takeda announced positive topline results from pivotal phase 2/3 clinical trial of TAK-881 in Primary Immunodeficiency Disease.
•In April 2026, we and Vertex entered into a global exclusive collaboration and license agreement that provides Vertex access to Hypercon technology for use in up to three targets. Under the terms of the agreement, Vertex will make a $15 million upfront payment and potential future milestone payments and royalties on net sales of products developed using the Hypercon technology.
•In March 2026, Pfizer nominated a new undisclosed non-exclusive target to be studied with ENHANZE.
•In March 2026, Janssen announced the Committee for Medicinal Products for Human Use of the European Medicines Agency granted approval for self or caregiver administration of DARZALEX (daratumumab) SC formulation for patients living with multiple myeloma from the fifth dose, if determined to be appropriate by their healthcare professional and following proper training, making it the first oncology injectable approved for self-administration in Europe.
•In March 2026, Janssen announced the FDA approved TECVAYLI (teclistamab-cqyv) in combination with DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) for the treatment of adults with relapsed or refractory multiple myeloma who have received at least one prior line of therapy.
•In February 2026, argenx announced positive topline results from the Phase 3 ADAPT oculus trial of VYVGART in ocular myasthenia gravis.
•In January 2026, argenx initiated a Phase 1 study to evaluate ARGX-124 with ENHANZE.
•In January 2026, Janssen announced the FDA approved DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) in combination with bortezomib, lenalidomide and dexamethasone for the treatment of adult patients with newly diagnosed multiple myeloma who are ineligible for autologous stem cell transplant.
Product and Product Candidates
The following table summarizes our marketed proprietary products and product candidates under development and our marketed partnered products and product candidates under development with our partners:
Proprietary Products and Product Candidates
Hylenex Recombinant (hyaluronidase human injection)
We market and sell Hylenex recombinant which is a formulation of rHuPH20 that facilitates SC administration for achieving hydration, increases the dispersion and absorption of other injected drugs and, in SC urography, to improve resorption of radiopaque agents. Hylenex recombinant is currently the number one prescribed branded hyaluronidase.
XYOSTED (testosterone enanthate) Injection
We market and sell our proprietary product XYOSTED for SC administration of testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of endogenous testosterone (primary hypogonadism or hypogonadotropic hypogonadism). XYOSTED is the only FDA-approved SC testosterone enanthate product for once-weekly, at-home self-administration and is approved and marketed in the United States (“U.S.”) in three dosage strengths, 50 mg, 75 mg and 100 mg.
Partnered Products
ENHANZE Collaborations
Roche Collaboration
In December 2006, we and Roche entered into a collaboration and license agreement under which Roche obtained a worldwide license to develop and commercialize product combinations of rHuPH20 and up to twelve Roche target compounds (the “Roche Collaboration”). Under this agreement, Roche initially elected a total of eight targets, two of which are exclusive.
In September 2013, Roche launched a SC formulation of Herceptin (trastuzumab) (Herceptin® SC) in Europe for the treatment of patients with human epidermal growth factor receptor 2-positive breast cancer followed by launches in additional countries. This formulation utilizes our ENHANZE technology and is administered in two to five minutes, compared to 30 to 90 minutes with the standard IV form. Herceptin SC has since received approval in Canada, the U.S. (under the brand name Herceptin Hylecta™) and China.
In June 2020, the FDA approved the fixed-dose combination of Perjeta® (pertuzumab) and Herceptin for SC injection (Phesgo®) utilizing ENHANZE technology for the treatment of patients with human epidermal growth factor receptor 2-positive breast cancer. Phesgo has since received approval in Europe and China. In September 2023, Chugai (a member of the Roche Group) announced that it had obtained regulatory approval for Phesgo from the Ministry of Health, Labour and Welfare in Japan. We receive royalties for Phesgo sales in Japan as part of our licensing agreement with Roche. In April 2025, Roche received a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use recommending an update to the European Union (“EU”) label for Phesgo for human epidermal growth factor receptor 2-positive breast cancer. Administration of Phesgo outside of a clinical setting (such as in a person’s home) by a healthcare professional will be possible, once safely established in a clinical setting.
In June 2014, Roche launched MabThera® SC in Europe for the treatment of patients with common forms of non-Hodgkin lymphoma, followed by launches in additional countries. This formulation utilizes our ENHANZE technology and is administered in approximately five minutes compared to the approximate one and a half to four hour IV infusion. In May 2016, Roche announced that the European Medicines Agency approved MabThera SC to treat patients with chronic lymphocytic leukemia. In June 2017, the FDA approved Genentech’s RITUXAN HYCELA®, a combination of rituximab using ENHANZE technology (approved and marketed under the MabThera SC brand in countries outside the U.S. and Canada), for chronic lymphocytic leukemia and two types of non-Hodgkin lymphoma, follicular lymphoma and diffuse large B-cell lymphoma. In March 2018, Health Canada approved a combination of rituximab and ENHANZE (approved and marketed under the brand name RITUXAN® SC) for patients with chronic lymphocytic leukemia. In April 2024, MabThera SC was approved by the China National Medical Products Administration to treat diffuse large B-cell lymphoma.
In September 2017 and October 2018, we entered into agreements with Roche to develop and commercialize additional exclusive targets using ENHANZE technology. The upfront license payment may be followed by event-based payments subject to Roche’s achievement of specified development, regulatory and sales-based milestones. In addition, Roche will pay royalties to us if products under the collaboration are commercialized.
In August 2023, Roche announced the approval of TECENTRIQ SC with ENHANZE by the Medicines and Healthcare products Regulatory Agency in the United Kingdom (the “UK”). In January 2024, Roche received European Commission marketing authorization for TECENTRIQ SC. In September 2024, Roche announced the FDA approved TECENTRIQ HYBREZA with ENHANZE. TECENTRIQ SC enables SC delivery in approximately seven minutes, compared with 30-60 minutes for IV infusion, and is approved for all adult indications of TECENTRIQ IV.
In June 2024, Roche announced the European Commission granted marketing authorization in the EU for OCREVUS SC as a twice a year ten-minute SC injection for the treatment of relapsing multiple sclerosis and primary progressive multiple sclerosis. In July 2024, Roche announced the Medicines and Healthcare products Regulatory Agency approved OCREVUS SC in the UK. In September 2024, Roche announced the FDA approved OCREVUS ZUNOVO with ENHANZE. In December 2025, Roche nominated a new undisclosed non-exclusive target to be studied using ENHANZE.
Takeda Collaboration
In September 2007, we and Takeda entered into a collaboration and license agreement under which Takeda obtained a worldwide, exclusive license to develop and commercialize product combinations of rHuPH20 with GAMMAGARD LIQUID (HYQVIA®) (the “Takeda Collaboration”). HYQVIA is indicated for the treatment of Primary Immunodeficiency Disorders associated with defects in the immune system.
In May 2013, the European Commission granted Takeda marketing authorization in all EU Member States for the use of HYQVIA as replacement therapy for adult patients with Primary Immunodeficiency and secondary immunodeficiencies. Takeda launched HYQVIA in the first EU country in July 2013 and has continued to launch in additional countries. In May 2016, Takeda announced that HYQVIA received a marketing authorization from the European Commission for a pediatric indication.
In September 2014, HYQVIA was approved by the FDA for treatment of adult patients with Primary Immunodeficiency. HYQVIA is the first SC immune globulin treatment approved for adult Primary Immunodeficiency patients with a dosing regimen requiring only one infusion up to once per month (every three to four weeks) and one injection site per infusion in most patients, to deliver a full therapeutic dose of immune globulin.
In September 2020, Takeda announced the European Medicines Agency approved a label update for HYQVIA broadening its use and making it the first and only facilitated SC immunoglobulin replacement therapy in adults, adolescents and children with an expanded range of secondary immunodeficiencies.
In October 2021, Takeda initiated a Phase 1 single-dose, single-center, open-label, three-arm study to assess the tolerability and safety of immune globulin SC (human), 20% solution with ENHANZE (TAK-881) at various infusion rates in healthy adult subjects. In October 2023, Takeda initiated a Phase 2/3 study to evaluate pharmacokinetic measures, safety, and tolerability of SC administration of TAK-881 in adult and pediatric participants with Primary Immunodeficiency Diseases. In May 2026, Takeda announced positive topline results from pivotal phase 2/3 clinical trial of TAK-881 in Primary Immunodeficiency Disease.
In April 2023, Takeda announced the FDA approved the supplemental Biologics License Application to expand the use of HYQVIA to treat Primary Immunodeficiency in children. In December 2024, Takeda announced the Ministry of Health, Labour and Welfare in Japan approved HYQVIA SC with ENHANZE for patients with agammaglobulinemia or hypogammaglobulinemia disorders characterized by very low or absent levels of antibodies and an increased risk of serious recurring infection caused by Primary Immunodeficiency or secondary immunodeficiencies.
In January 2024, Takeda received FDA and European Commission approval for HYQVIA for the treatment of chronic inflammatory demyelinating polyneuropathy in adults with stable chronic inflammatory demyelinating polyneuropathy. In June 2024, Takeda announced Health Canada approved HYQVIA as replacement therapy for primary humoral immunodeficiency and secondary humoral immunodeficiency in pediatric patients two years of age and older. In March 2025, Takeda announced Health Canada expanded the marketing authorization for HYQVIA to include chronic inflammatory demyelinating polyneuropathy as a maintenance therapy after stabilization with intravenous immunoglobulin to prevent relapse of neuromuscular disability and impairment in adults. In June 2025, Takeda announced the Ministry of Health, Labour and Welfare in Japan approved HYQVIA SC with ENHANZE for treatment of patients with chronic inflammatory demyelinating polyneuropathy and multifocal motor neuropathy.
In December 2025, we and Takeda entered into a new global collaboration and exclusive license agreement which provides Takeda with access to ENHANZE for use with vedolizumab, marketed globally as ENTYVIO®, for the treatment of adults with moderately to severely active Crohns’ disease or ulcerative colitis, which are the two main forms of inflammatory bowel disease.
Pfizer Collaboration
In December 2012, we and Pfizer entered into a collaboration and license agreement, under which Pfizer has the worldwide license to develop and commercialize products combining our rHuPH20 enzyme with Pfizer proprietary biologics in primary care and specialty care indications. Pfizer currently has one non-exclusive target.
In March 2026, Pfizer nominated a new undisclosed non-exclusive target to be studied with ENHANZE.
Janssen Collaboration
In December 2014, we and Janssen entered into a collaboration and license agreement, under which Janssen has the worldwide license to develop and commercialize products combining our rHuPH20 enzyme with Janssen proprietary biologics directed to up to five targets. Targets may be selected on an exclusive basis. Janssen elected CD38 and initiated several Phase 3 studies, Phase 2 studies and Phase 1 studies of DARZALEX® (daratumumab), directed at CD38, using ENHANZE technology in patients with amyloidosis, smoldering myeloma and multiple myeloma.
In May 2020, Janssen launched the commercial sale of DARZALEX FASPRO® (DARZALEX utilizing ENHANZE technology) in the U.S. in four regimens across five indications in multiple myeloma patients, including newly diagnosed, transplant-ineligible patients as well as relapsed or refractory patients. As a fixed-dose formulation, DARZALEX FASPRO can be administered over three to five minutes, significantly less time than DARZALEX IV, which requires multi-hour infusions. In June 2020, Janssen received European marketing authorization and launched the commercial sale of DARZALEX SC utilizing ENHANZE in the EU. Subsequent to these approvals, Janssen received several additional regulatory approvals for additional indications and patient populations in the U.S., EU, Japan and China. Beginning with the U.S., Janssen has marketing authorization for DARZALEX FASPRO in combination with bortezomib, thalidomide, and dexamethasone in newly diagnosed multiple myeloma patients who are eligible for autologous stem cell transplant, in combination with bortezomib, cyclophosphamide and dexamethasone for the treatment of adult patients with newly diagnosed AL amyloidosis, in combination with pomalidomide and dexamethasone for patients with multiple myeloma after first or subsequent relapse, and in combination with Kyprolis® (carfilzomib) and dexamethasone for patients with relapsed or refractory multiple myeloma who have received one to three prior lines of therapy. In the EU, Janssen has marketing authorization for DARZALEX SC in combination with bortezomib, cyclophosphamide and dexamethasone in newly diagnosed adult patients with AL amyloidosis and in combination with pomalidomide and dexamethasone in adult patients with relapsed or refractory multiple myeloma. In Japan, Janssen has marketing authorization for the SC formulation of DARZALEX (known as DARZQURO) for the treatment of multiple myeloma and systemic AL amyloidosis. In China, Janssen has marketing authorization for DARZALEX SC for the treatment of primary light chain amyloidosis, in combination with bortezomib, cyclophosphamide and dexamethasone in newly diagnosed patients.
In July 2024, Janssen announced the FDA approved DARZALEX FASPRO in combination with bortezomib, lenalidomide and dexamethasone for induction and consolidation treatment and with lenalidomide for maintenance treatment of adult patients who are newly diagnosed with multiple myeloma and are eligible for autologous stem cell transplant, with approval also received from the European Commission in October 2024. In April 2025, Janssen received European Commission approval for an indication extension of DARZALEX SC in combination with bortezomib, lenalidomide, and dexamethasone for the treatment of adult patients with newly diagnosed multiple myeloma regardless of transplant eligibility. In July 2025, Janssen announced European Commission approval of a new indication for DARZALEX SC (daratumumab) co-formulated with ENHANZE, as a monotherapy for the treatment of adult patients with smoldering multiple myeloma at high-risk of developing multiple myeloma. In November 2025, Janssen announced the FDA approved DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) co-formulated with ENHANZE, as single treatment of adult patients with high-risk smoldering multiple myeloma. In January 2026, Janssen announced the FDA approved DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) in combination with bortezomib, lenalidomide and dexamethasone for the treatment of adult patients with newly diagnosed multiple myeloma who are ineligible for autologous stem cell transplant. In March 2026, Janssen announced the FDA approved TECVAYLI (teclistamab-cqyv) in combination with DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) for the treatment of adults with relapsed or refractory multiple myeloma who have received at least one prior line of therapy. In March 2026, Janssen announced the Committee for Medicinal Products for Human Use of the European Medicines Agency granted approval for self or caregiver administration of DARZALEX (daratumumab) SC formulation for patients living with multiple myeloma from the fifth dose, if determined to be appropriate by their healthcare professional and following proper training, making it the first oncology injectable approved for self-administration in Europe.
In December 2019, Janssen elected epidermal growth factor receptor and mesenchymal-epithelial transition factor as a bispecific antibody (amivantamab) target on an exclusive basis, which is being studied in solid tumors. In September 2022, following a Phase 1 study, Janssen initiated a Phase 3 study of lazertinib and amivantamab with ENHANZE in patients with epidermal growth factor receptor-mutated advanced or metastatic non-small cell lung cancer (PALOMA-3). In November 2022, Janssen initiated a Phase 2 study of amivantamab with ENHANZE in multiple regimens in patients with advanced or metastatic solid tumors including epidermal growth factor receptor-mutated non-small cell lung cancer (PALOMA-2). The administration time for SC amivantamab was reduced to approximately five minutes from approximately five hours per day for the first IV amivantamab infusion and an average of 2.3 hours for subcutaneous infusions and showed a five-fold reduction in infusion-related reactions. SC amivantamab also demonstrated longer overall survival, progression-free survival and duration of response. In April 2025, Janssen received European Commission marketing authorization of the SC formulation of RYBREVANT (amivantamab) with ENHANZE (“RYBREVANT SC”), in combination with LAZCLUZE (lazertinib), for the first-line treatment of adult patients with advanced non-small cell lung cancer with epidermal growth factor receptor exon 19 deletions or exon 21 L858R substitution mutations. RYBREVANT (amivantamab) is approved as a monotherapy for adult patients with advanced non-small cell lung cancer with activating epidermal growth factor receptor exon 20 insertion mutations after the failure of platinum-based therapy. In December 2025, Janssen announced the FDA approved RYBREVANT FASPRO (amivantamab and hyaluronidase-lpuj) for the treatment of patients with epidermal growth factor receptor-mutated locally advanced or metastatic non-small cell lung cancer. In December 2025, Janssen received approval from the Ministry of Health, Labour and Welfare in Japan for RYBROFAZ (amivantamab) with ENHANZE for the first-line treatment of adult patients with advanced non-small cell lung cancer. In December 2025, Janssen received approval from the National Medical Products Administration in China for RYBREVANT FASPRO for the first-line treatment of adult patients with advanced non-small cell lung cancer.
AbbVie Collaboration
In June 2015, we and AbbVie entered into a collaboration and license agreement, under which AbbVie has the worldwide license to develop and commercialize products combining our rHuPH20 enzyme with AbbVie proprietary biologics. AbbVie currently has the right to select up to nine targets. Targets may be selected on an exclusive basis.
Lilly Collaboration
In December 2015, we and Lilly entered into a collaboration and license agreement, under which Lilly has the worldwide license to develop and commercialize products combining our rHuPH20 enzyme with Lilly proprietary biologics. Lilly currently has the right to select up to three targets. Targets may be selected on an exclusive basis.
BMS Collaboration
In September 2017, we and BMS entered into a collaboration and license agreement, which became effective in November 2017, under which BMS had the worldwide license to develop and commercialize products combining our rHuPH20 enzyme with BMS products directed at up to eleven targets. Targets may be selected on an exclusive basis or non-exclusive basis. BMS has designated multiple immuno-oncology targets including programmed death 1 and has an option to select three additional targets by September 2026. In December 2024, BMS announced the FDA approved Opdivo® Qvantig (nivolumab and hyaluronidase-nvhy) with ENHANZE for SC use in most previously approved adult, solid IV Opdivo (nivolumab) indications. Opdivo Qvantig is the first and only SC administered programmed death 1 inhibitor. In May 2025, BMS received European Commission approval of Opdivo SC, the subcutaneous formulation of Opdivo (nivolumab) developed with ENHANZE, for use across multiple adult solid tumors.
argenx Collaboration
In February 2019, we and argenx entered into an agreement for the right to develop and commercialize one exclusive target, the human neonatal Fc receptor FcRn, which includes argenx’s lead asset efgartigimod (ARGX-113), and an option to select two additional targets using ENHANZE technology. In May 2019, argenx nominated a second target to be studied using ENHANZE technology, a human complement factor C2 associated with the product candidate ARGX-117, which is being developed to treat severe autoimmune diseases in Multifocal Motor Neuropathy. In October 2020, we and argenx entered into an agreement to expand the collaboration relationship, adding three targets for a total of up to six targets under the collaboration. In September 2024, argenx nominated four additional targets under its global collaboration and license agreement that provides them with exclusive access to our ENHANZE drug delivery technology for these targets, for a total of six targets.
In June 2023, argenx received FDA approval under the brand name VYVGART® Hytrulo for the SC injection with ENHANZE for the treatment of generalized myasthenia gravis in adult patients who are anti-acetylcholine receptor antibody positive. In November 2023, argenx received European Commission approval of VYVGART SC for the treatment of generalized myasthenia gravis, which also provides the option for patient self-administration. In January 2024, argenx received Japan approval for VYVDURA® (efgartigimod alfa and hyaluronidase-qvfc) co-formulated with ENHANZE for the treatment of adult patients with generalized myasthenia gravis including options for self-administration. In July 2024, argenx announced the National Medical Products Administration approved the Biologics License Application of efgartigimod alfa SC (efgartigimod SC) for generalized myasthenia gravis patients in China.
In July 2023, argenx reported positive data from the ADHERE study evaluating VYVGART Hytrulo with ENHANZE in adults with chronic inflammatory demyelinating polyneuropathy. In June 2024, argenx announced the FDA approved VYVGART Hytrulo with ENHANZE for the treatment of chronic inflammatory demyelinating polyneuropathy. In November 2024, Zai Lab Limited (argenx commercial partner for China) announced the National Medical Products Administration approval of VYVGART Hytrulo for the treatment of patients with chronic inflammatory demyelinating polyneuropathy. In December 2024, argenx announced the Ministry of Health, Labour and Welfare in Japan approved VYVDURA for the treatment of patients with chronic inflammatory demyelinating polyneuropathy. In June 2025, argenx announced European Commission approval of VYVGART SC with ENHANZE for the treatment of adult patients with progressive or relapsing active chronic inflammatory demyelinating polyneuropathy after prior treatment with corticosteroids or immunoglobulins. VYVGART SC injection is available as a vial or prefilled syringe and can be administered by a patient, caregiver, or healthcare professional.
In April 2025, argenx received FDA approval of VYVGART Hytrulo prefilled syringe for self-injection for the treatment of adult patients with generalized myasthenia gravis who are anti-acetylcholine receptor antibody positive and adult patients with chronic inflammatory demyelinating polyneuropathy. In September 2025, argenx announced the Ministry of Health, Labour and Welfare in Japan approved VYVDURA prefilled syringe for self-injection for the treatment of adult patients with generalized myasthenia gravis and adult patients with chronic inflammatory demyelinating polyneuropathy.
argenx is currently conducting the following studies with the goal of expanding approved indications for efgartigimod with ENHANZE: Phase 2/3 (ALKIVIA) study in active idiopathic inflammatory myopathy (Myositis), Phase 2 (Shamrock) study for kidney transplant recipients with antibody mediated rejection, Phase 3 (ADAPT oculus) study for adult patients with ocular myasthenia gravis, Phase 3 (Unity) study in patients with moderate-to-severe Primary Sjogren’s Disease and Phase 2 (eSScape) study in adults with Systemic Sclerosis. In February 2026, argenx announced positive topline results from the Phase 3 ADAPT oculus trial of VYVGART in ocular myasthenia gravis. In May 2026, argenx announced FDA approval of a supplemental Biologics License Application for VYVGART Hytrulo with ENHANZE for the treatment of adult patients with generalized myasthenia gravis including all serotypes – anti-AChR-Ab positive, anti-MuSK-Ab positive, anti-LRP4-Ab positive and triple seronegative.
In May 2025, argenx initiated a Phase 1 study to evaluate ARGX-213 with ENHANZE.
In the fourth quarter of 2025, the ongoing argenx ARGX-121 Phase 1 program was expanded to include an SC-arm evaluating ARGX-121 with ENHANZE in healthy adults.
In January 2026, argenx initiated a Phase 1 study to evaluate ARGX-124 with ENHANZE.
ViiV Healthcare Collaboration
In June 2021, we and ViiV entered into a global collaboration and license agreement that gives ViiV exclusive access to our ENHANZE technology for four specific small and large molecule targets for the treatment and prevention of HIV. These targets are integrase inhibitors, reverse transcriptase inhibitors limited to nucleoside reverse transcriptase inhibitors and nucleoside reverse transcriptase translocation inhibitors, capsid inhibitors and broadly neutralising monoclonal antibodies, that bind to the gp120 CD4 binding site.
In the third quarter of 2023, ViiV initiated a Phase 1 study with ENHANZE for an undisclosed program. In March 2024, ViiV initiated a Phase 1 study of VH4524184 with ENHANZE to evaluate the safety, tolerability, and pharmacokinetic measures in healthy adults.
In September 2024, we and ViiV expanded the existing global collaboration and license agreement, providing ViiV exclusive access to our ENHANZE drug delivery technology for one additional undisclosed target.
Chugai Collaboration
In March 2022, we and Chugai entered into a global collaboration and license agreement that gives Chugai exclusive access to ENHANZE technology for an undisclosed target. In May 2022, Chugai initiated a Phase 1 study to evaluate the pharmacokinetic measures, pharmacodynamics, and safety of a targeted antibody administered subcutaneously with ENHANZE. Chugai has notified us that they have terminated this program and are evaluating other potential target programs with ENHANZE.
Acumen Collaboration
In November 2023, we and Acumen entered into a global collaboration and non-exclusive license agreement that provides Acumen access to ENHANZE for a single target. Acumen intends to explore the potential use of ENHANZE for ACU193, Acumen’s clinical stage monoclonal antibody candidate to target Amyloid-β Oligomers for the treatment of early Alzheimer’s disease. In May 2024, Acumen initiated a Phase 2 IV study for ACU193. In July 2024, Acumen initiated a Phase 1 study of sabirnetug (ACU193) with ENHANZE to compare the pharmacokinetic measures between SC and IV administrations in healthy volunteers. In March 2025, Acumen announced top-line results from this study that demonstrated weekly SC administration of sabirnetug was well-tolerated with systematic exposure supporting further clinical development.
Merus Collaboration
In November 2025, we and Merus entered into a non-exclusive global collaboration and license agreement that provides Merus access to ENHANZE for the development and potential commercialization of SC formulation of petosemtamab, an epidermal growth factor receptor and leucine-rich repeat-containing G-protein coupled receptor 5 bispecific antibody, for the treatment of head and neck cancer.
Skye Bioscience Collaboration
In December 2025, we and Skye Bioscience entered into a non-exclusive global collaboration and license agreement that provides Skye Bioscience access to ENHANZE for the development and potential commercialization of SC formulation of nimacimab for the treatment of obesity.
GSK Collaboration
In May 2026, we and GSK entered into a global collaboration and license agreement for ENHANZE with multiple oncology targets, including the first potential application in antibody-drug conjugates.
Hypercon Collaborations
argenx Collaboration
In April 2021, we and argenx entered into a collaboration and license agreement that provides argenx the right to develop and commercialize one target directed at the human neonatal fragment crystallizable receptor, including efgartigimod as well as an option to nominate a second target using Hypercon technology.
Lilly Collaboration
In October 2023, we and Lilly entered into a research collaboration and exclusive license agreement that provides Lilly the right to develop and commercialize three initial targets as well as options to nominate up to two additional targets using Hypercon technology.
Janssen Collaboration
In December 2023, we and Janssen entered into a collaboration and exclusive license agreement that provides Janssen with the right to develop and commercialize an initial target as well as options to nominate up to four additional targets using Hypercon technology.
Vertex Collaboration
In April 2026, we and Vertex entered into a global exclusive collaboration and license agreement that provides Vertex access to Hypercon technology for use in up to three targets.
Oruka Collaboration
In May 2026, we and Oruka entered into a global exclusive collaboration and license agreement for our Hypercon technology for use with ORKA-001, in development for psoriasis and related inflammatory diseases and one additional target.
Device and Other Drug Product Collaborations
Teva License, Development and Supply Agreements
In July 2006, we entered into an exclusive license, development and supply agreement with Teva for an epinephrine auto- injector product to be marketed in the U.S. and Canada. We are the exclusive supplier of the device, which we developed, for Teva’s generic Epinephrine Injection USP products, indicated for emergency treatment of severe allergic reactions including those that are life threatening (anaphylaxis) in adults and certain pediatric patients. Teva’s Epinephrine Injection, utilizing our patented VIBEX® injection technology, was approved by the FDA as a generic drug product with an AB rating, meaning that it is therapeutically equivalent to the branded products EpiPen® and EpiPen Jr® and therefore, subject to state law, substitutable at the pharmacy.
In December 2007, we entered into a license, development and supply agreement with Teva under which we developed and supply a disposable pen injector for teriparatide. Under the agreement, we received an upfront payment and development milestones, and are entitled to receive royalties on net product sales by Teva in territories where commercialized. We are the exclusive supplier of the multi-dose pen, which we developed, used in Teva’s generic teriparatide injection product. In 2020, Teva launched Teriparatide Injection, the generic version of Lilly’s branded product Forsteo® featuring our multi-dose pen platform, for commercial sale in several countries outside of the U.S. In November 2023, Teva announced FDA approval of the generic version of Forteo, featuring our multi-dose auto-injector pen platform for the treatment of osteoporosis among certain women and men.
Pfizer Agreement
In August 2018, we entered into a development agreement with Pfizer to jointly develop a combination drug device rescue pen utilizing the QuickShot auto-injector and an undisclosed Pfizer drug. Pfizer has provided the intellectual property rights for further development of the product to us and has retained an option to assist in the marketing, distribution and sale if we complete development of the product and submit for regulatory approval. We are continuing to evaluate the next steps for this program.
Viatris Agreement
In November 2019, we entered into a global development agreement with Idorsia Pharmaceuticals Ltd. (“Idorsia”) which was subsequently assigned to McDermott Laboratories Limited, an affiliate of Viatris, in February 2025 to develop a novel, drug-device product containing selatogrel. A new chemical entity, selatogrel, is being developed for the treatment of a suspected acute myocardial infarction in adult patients with a recent history of acute myocardial infarction.
In August 2021, Idorsia initiated a multi-center, double-blind, randomized, placebo-controlled, parallel-group Phase 3 study to evaluate the efficacy and safety of self-administered SC selatogrel for prevention of all-cause death and treatment of acute myocardial infarction in subjects with a recent history of acute myocardial infarction.
In December 2025, we entered into a commercial license and supply agreement with Viatris under which we license and supply an auto-injector product for self-administered SC selatogrel for the treatment of acute myocardial infarction in adult patients.
Results of Operations
Three Months Ended March 31, 2026 Compared to Three Months Ended March 31, 2025
Royalties – Royalties were as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Increase / (Decrease) |
| | March 31, | |
| | 2026 | | 2025 | | Dollar | | Percentage |
| DARZALEX | | $ | 129,032 | | | $ | 102,195 | | | $ | 26,837 | | | 26 | % |
| VYVGART Hytrulo | | 46,283 | | | 21,090 | | | 25,193 | | | 119 | % |
| Phesgo | | 30,199 | | | 24,076 | | | 6,123 | | | 25 | % |
| Other | | 35,167 | | | 20,831 | | | 14,336 | | | 69 | % |
| Total royalties | | $ | 240,681 | | | $ | 168,192 | | | $ | 72,489 | | | 43 | % |
The increase in royalties was primarily driven by continued sales uptake of ENHANZE partner products that have launched since 2020, predominantly DARZALEX SC by Janssen, VYVGART Hytrulo by argenx, and Phesgo by Roche in all geographies and contributions from other recently launched products. This growth was partially diluted by earlier-launched ENHANZE partner products that are later in their life cycle and experiencing modest price erosion, such as Herceptin and MabThera by Roche.
We expect royalty revenue to grow further as a result of anticipated increasing partner product sales of DARZALEX SC, Phesgo and VYVGART Hytrulo, the largest drivers of our royalty revenues. The total of all other products is also expected to grow, mainly driven by recently launched ENHANZE partner products, TECENTRIQ SC and OCREVUS SC by Roche, RYBREVANT SC by Janssen and Opdivo Qvantig by BMS, partially offset by modest price erosion expected to continue on earlier launched ENHANZE partner products, Herceptin and MabThera.
Product Sales, Net – Product sales, net were as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Increase / (Decrease) |
| | March 31, | |
| | 2026 | | 2025 | | Dollar | | Percentage |
Proprietary product sales | | $ | 41,873 | | | $ | 36,341 | | | $ | 5,532 | | | 15 | % |
Bulk rHuPH20 sales | | 73,497 | | | 27,025 | | | 46,472 | | | 172 | % |
Device partnered product sales | | 15,054 | | | 14,675 | | | 379 | | | 3 | % |
| Total product sales, net | | $ | 130,424 | | | $ | 78,041 | | | $ | 52,383 | | | 67 | % |
The increase in product sales, net was primarily due to increased sales of bulk rHuPH20 driven by partner demand as well as contributions from our proprietary product XYOSTED driven by continued market growth. We expect sales of our proprietary products will grow in future years as we continue to gain market share in the testosterone replacement therapy market. We expect product sales of bulk rHuPH20 and device partnered products to fluctuate in future periods based on the needs of our partners.
Revenues Under Collaborative Agreements – Revenues under collaborative agreements were as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Increase / (Decrease) |
| | March 31, | |
| | 2026 | | 2025 | | Dollar | | Percentage |
| | | | | | | |
Upfront license and target nomination fees | | $ | — | | | $ | 220 | | | $ | (220) | | | (100) | % |
| | | | | | | | |
Sales-based milestones | | — | | | 15,000 | | | (15,000) | | | (100) | % |
Device licensing and development revenue | | 5,603 | | | 3,408 | | | 2,195 | | | 64 | % |
| Total revenues under collaborative agreements | | $ | 5,603 | | | $ | 18,628 | | | $ | (13,025) | | | (70) | % |
The decrease in revenues under collaborative agreements was primarily due to the timing of milestones achieved. Revenue from upfront licenses fees, license fees for the election of additional targets, event-based payments, license maintenance and other license fees vary from period to period based on our ENHANZE collaboration activity. We expect these revenues to continue to fluctuate in future periods based on our partners’ ability to meet various clinical, regulatory and event-based milestones set forth in such agreements and our ability to obtain new collaborative agreements.
Operating expenses – Operating expenses were as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Increase / (Decrease) |
| | March 31, | |
| | 2026 | | 2025 | | Dollar | | Percentage |
| Cost of sales | | $ | 79,238 | | | $ | 48,403 | | | $ | 30,835 | | | 64 | % |
| Amortization of intangibles | | 29,512 | | | 17,762 | | | 11,750 | | | 66 | % |
| Research and development | | 25,560 | | | 14,799 | | | 10,761 | | | 73 | % |
| Selling, general and administrative | | 57,881 | | | 42,362 | | | 15,519 | | | 37 | % |
| Total operating expenses | | $ | 192,191 | | | $ | 123,326 | | | $ | 68,865 | | | 56 | % |
Cost of Sales – Cost of sales consists primarily of raw materials, third-party manufacturing costs, fill and finish costs, freight costs, internal costs and manufacturing overhead associated with the production of our proprietary products, device partnered products and bulk rHuPH20. The increase in cost of sales was primarily due to an increase in bulk rHuPH20 sales.
Amortization of Intangibles – Amortization of intangibles consists primarily of expense associated with the amortization of acquired device technologies and product rights. The increase in amortization of intangibles expense was due to the acquisition of Elektrofi in November 2025.
Research and Development – Research and development expenses consist of external costs, salaries and benefits, and allocation of facilities and other overhead expenses related to research manufacturing, preclinical and regulatory activities related to our collaborations, and our development platforms. The increase in research and development expense was primarily due to the acquisition of Elektrofi and Surf Bio in the fourth quarter of 2025.
Selling, General and Administrative – Selling, general and administrative (“SG&A”) expenses consist primarily of salaries and related costs for personnel in executive, selling and administrative functions, as well as professional fees for legal and accounting, business development, commercial operations support for proprietary products and alliance management, and marketing support for our collaborations. The increase in SG&A expenses was primarily due to an increase in consulting and professional service fees, including litigation costs in connection with a patent infringement litigation case, the acquisition of Elektrofi and Surf Bio, and an increase in compensation expense.
Investment and Other Income, Net – Investment and other income, net was as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Increase / (Decrease) |
| | March 31, | |
| | 2026 | | 2025 | | Dollar | | Percentage |
| Investment and other income, net | | $ | 1,318 | | | $ | 6,818 | | | $ | (5,500) | | | (81) | % |
Investment and other income, net consists primarily of interest income on our cash, cash-equivalent and marketable securities. The decrease in investment and other income, net was primarily due to a decrease in the average invested balance.
Interest Expense – Interest expense was as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Increase / (Decrease) |
| | March 31, | |
| | 2026 | | 2025 | | Dollar | | Percentage |
| Interest expense | | $ | 5,508 | | | $ | 4,525 | | | $ | 983 | | | 22 | % |
Interest expense consists primarily of costs related to our convertible notes and revolving credit facility. The increase in interest expense is due to the issuance of the 2031 and 2032 Convertible Notes in the fourth quarter of 2025.
Income Tax Expense – Income tax expense was as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Increase / (Decrease) |
| | March 31, | |
| | 2026 | | 2025 | | Dollar | | Percentage |
| Income tax expense | | $ | 30,278 | | | $ | 25,733 | | | $ | 4,545 | | | 18 | % |
The increase in income tax expense was primarily due to higher income before income tax expense, an increase in disallowed share-based compensation expense, and an increase in Section 162(m) disallowance, partially offset by an increase in tax benefits associated with share-based compensation windfall, Foreign Derived Deduction Eligible Income benefit, and research and development tax credit generation.
Liquidity and Capital Resources
Overview
Our principal sources of liquidity are our existing cash, cash equivalents and available-for-sale marketable securities. As of March 31, 2026, we had cash, cash equivalents and marketable securities of $318.6 million. We believe that our current cash, cash equivalents and marketable securities will be sufficient to fund our operations for at least the next 12 months. We expect to fund our operations going forward with existing cash resources, anticipated revenues from our existing collaborative agreements and cash that we may raise through future transactions. We may raise cash through any one of the following financing vehicles: (i) new collaborative agreements; (ii) expansions or revisions to existing collaborative relationships; (iii) private financings; (iv) other equity or debt financings; (v) monetizing assets; and/or (vi) the public offering of securities.
We may, in the future, draw on our existing line of credit or offer and sell additional equity, debt securities and warrants to purchase any of such securities, either individually or in units to raise capital for additional working capital, capital expenditures, share repurchases, acquisitions or for other general corporate purposes.
Cash Flows
| | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | |
| | March 31, | | |
(in thousands) | | 2026 | | 2025 | | Change |
| Net cash provided by operating activities | | $ | 180,078 | | | $ | 154,221 | | | $ | 25,857 | |
| Net cash used in investing activities | | (2,877) | | | (90,422) | | | 87,545 | |
| Net cash used in financing activities | | (1,613) | | | (3,321) | | | 1,708 | |
| Net increase in cash and cash equivalents | | $ | 175,588 | | | $ | 60,478 | | | $ | 115,110 | |
Operating Activities
The increase in net cash provided by operations was primarily due to an increase in revenue, partially offset by higher working capital spend.
Investing Activities
The decrease in net cash used in investing activities was primarily due to a net sale of $0.2 million of marketable securities compared to a net purchase of $89.5 million of marketable securities in the prior year, partially offset by an increase in capital spend for property and equipment.
Financing Activities
The decrease in net cash used in financing activities was primarily due to an increase in net proceeds from the issuance of common stock under our equity incentive plan.
Share Repurchases
In February 2024, our Board of Directors authorized a capital return program to repurchase up to $750.0 million of our outstanding common stock. In May 2026, our Board of Directors authorized a new capital return program to repurchase up to $1 billion of our outstanding common stock by December 31, 2028. Refer to Note 9, Stockholders’ Equity, of our condensed consolidated financial statements for additional information regarding our share repurchases.
Long-Term Debt, Net
0.875% Convertible Notes due 2032
In November 2025, we completed the sale of $750.0 million in aggregate principal amount of 0.875% Convertible Senior Notes due 2032 (the “2032 Convertible Notes”). The net proceeds from the issuance of the 2032 Convertible Notes, after deducting the initial purchasers’ fee of $15.0 million, was approximately $735.0 million. We also incurred additional debt issuance costs totaling $0.5 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2032 Convertible Notes pay interest semi-annually in arrears on May 15th and November 15th of each year at an annual rate of 0.875%. The 2032 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2032 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness, and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2032 Convertible Notes have a maturity date of November 15, 2032.
Holders may convert their 2032 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2026, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2032 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, May 15, 2032, until the close of business on the second scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2032 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal, and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2032 Convertible Notes is 11.4683 shares of common stock per $1,000 in principal amount of 2032 Convertible Notes, equivalent to a conversion price of approximately $87.20 per share of our common stock. The conversion rate is subject to adjustment in some events but will not be adjusted for any accrued or unpaid interest.
As of March 31, 2026, we were in compliance with all covenants.
2032 Capped Call Transactions
In connection with the offering of the 2032 Convertible Notes, we entered into capped call transactions with certain counterparties (the “2032 Capped Call Transactions”). The 2032 Capped Call Transactions are expected generally to reduce potential dilution to holders of our common stock upon conversion of the 2032 Convertible Notes or at our election (subject to certain conditions) offset any cash payments we are required to make in excess of the principal amount of such converted 2032 Convertible Notes. The cap price of the 2032 Capped Call Transactions is initially $136.78 per share of common stock, representing a premium of 100% above the last reported sale price of $68.30 per share of common stock on November 5, 2025, and is subject to certain adjustments under the terms of the 2032 Capped Call Transactions. As of March 31, 2026, no 2032 Capped Call Transactions had been exercised.
Pursuant to their terms, the 2032 Capped Call Transactions qualify for classification within stockholders’ equity in our condensed consolidated balance sheets, and their fair value is not remeasured and adjusted as long as they continue to qualify for stockholders’ equity classification. We paid approximately $106.8 million for the 2032 Capped Call Transactions, including applicable transaction costs, which was recorded as a reduction to additional paid-in capital in our condensed consolidated balance sheets. The 2032 Capped Call Transactions are separate transactions entered into by us with certain counterparties, are not part of the terms of the 2032 Convertible Notes, and do not affect any holder’s rights under the 2032 Convertible Notes. Holders of the 2032 Convertible Notes do not have any rights with respect to the 2032 Capped Call Transactions.
0.00% Convertible Notes due 2031
In November 2025, we completed the sale of $750.0 million in aggregate principal amount of 0.00% Convertible Senior Notes due 2031 (the “2031 Convertible Notes”). The net proceeds from the issuance of the 2031 Convertible Notes, after deducting the initial purchasers’ fee of $15.0 million, was approximately $735.0 million. We also incurred additional debt issuance costs totaling $0.5 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2031 Convertible Notes will not bear regular interest and the principal amount of the 2031 Convertible Notes will not accrete. The 2031 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2031 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2031 Convertible Notes have a maturity date of February 15, 2031.
Holders may convert their 2031 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2026, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2031 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, August 15, 2030, until the close of business on the scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2031 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2031 Convertible Notes is 11.4683 shares of common stock per $1,000 in principal amount of 2031 Convertible Notes, equivalent to a conversion price of approximately $87.20 per share of our common stock. The conversion rate is subject to adjustment.
As of March 31, 2026, we were in compliance with all covenants.
2031 Capped Call Transactions
In connection with the offering of the 2031 Convertible Notes, we entered into capped call transactions with certain counterparties (the “2031 Capped Call Transactions”). The 2031 Capped Call Transactions are expected generally to reduce potential dilution to holders of our common stock upon conversion of the 2031 Convertible Notes or at our election (subject to certain conditions) offset any cash payments we are required to make in excess of the principal amount of such converted 2031 Convertible Notes. The cap price of the 2031 Capped Call Transactions is initially $136.78 per share of common stock, representing a premium of 100% above the last reported sale price of $68.30 per share of common stock on November 5, 2025, and is subject to certain adjustments under the terms of the 2031 Capped Call Transactions. As of March 31, 2026, no 2031 Capped Call Transactions had been exercised.
Pursuant to their terms, the 2031 Capped Call Transactions qualify for classification within stockholders’ equity in our condensed consolidated balance sheets, and their fair value is not remeasured and adjusted as long as they continue to qualify for stockholders’ equity classification. We paid approximately $104.0 million for the 2031 Capped Call Transactions, including applicable transaction costs, which was recorded as a reduction to additional paid-in capital in our condensed consolidated balance sheets. The 2031 Capped Call Transactions are separate transactions entered into by us with certain counterparties, are not part of the terms of the 2031 Convertible Notes, and do not affect any holder’s rights under the 2031 Convertible Notes. Holders of the 2031 Convertible Notes do not have any rights with respect to the 2031 Capped Call Transactions.
1.00% Convertible Notes due 2028
In August 2022, we completed the sale of $720.0 million in aggregate principal amount of 1.00% Convertible Senior Notes due 2028 (the “2028 Convertible Notes”). The net proceeds from the issuance of the 2028 Convertible Notes, after deducting the initial purchasers’ fee of $18.0 million, was approximately $702.0 million. We also incurred additional debt issuance costs totaling $1.0 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2028 Convertible Notes pay interest semi-annually in arrears on February 15th and August 15th of each year at an annual rate of 1.00%. The 2028 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2028 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness, and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2028 Convertible Notes have a maturity date of August 15, 2028.
Holders may convert their 2028 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on December 31, 2022, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2028 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, February 15, 2028 until the close of business on the second scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2028 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal, and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2028 Convertible Notes is 17.8517 shares of common stock per $1,000 in principal amount of 2028 Convertible Notes, equivalent to a conversion price of approximately $56.02 per share of our common stock. The conversion rate is subject to adjustment in some events but will not be adjusted for any accrued or unpaid interest.
In connection with the offering of the 2032 Convertible Notes and 2031 Convertible Notes, we used a portion of the net proceeds of the offering to enter into privately negotiated agreements with certain holders of the outstanding 2028 Convertible Notes to repurchase their 2028 Convertible Notes for cash. In connection with the repurchases, we paid approximately $342.9 million in cash, which included a premium, inducement expense and accrued interest.
As of March 31, 2026, we were in compliance with all covenants.
2028 Capped Call Transactions
In connection with the offering of the 2028 Convertible Notes, we entered into capped call transactions with certain counterparties (the “2028 Capped Call Transactions”). The 2028 Capped Call Transactions are expected generally to reduce potential dilution to holders of our common stock upon conversion of the 2028 Convertible Notes or at our election (subject to certain conditions) offset any cash payments we are required to make in excess of the principal amount of such converted 2028 Convertible Notes. The cap price of the 2028 Capped Call Transactions is initially $75.4075 per share of common stock, representing a premium of 75% above the last reported sale price of $43.09 per share of common stock on August 15, 2022, and is subject to certain adjustments under the terms of the 2028 Capped Call Transactions. As of March 31, 2026, no 2028 Capped Call Transactions had been exercised.
Pursuant to their terms, the 2028 Capped Call Transactions qualify for classification within stockholders’ equity in our condensed consolidated balance sheets, and their fair value is not remeasured and adjusted as long as they continue to qualify for stockholders’ equity classification. We paid approximately $69.1 million for the 2028 Capped Call Transactions, including applicable transaction costs, which was recorded as a reduction to additional paid-in capital in our condensed consolidated balance sheets. The 2028 Capped Call Transactions are separate transactions entered into by us with certain counterparties, are not part of the terms of the 2028 Convertible Notes, and do not affect any holder’s rights under the 2028 Convertible Notes. Holders of the 2028 Convertible Notes do not have any rights with respect to the 2028 Capped Call Transactions.
0.25% Convertible Notes due 2027
In March 2021, we completed the sale of $805.0 million in aggregate principal amount of 0.25% Convertible Senior Notes due 2027 (the “2027 Convertible Notes”). The net proceeds from the issuance of the 2027 Convertible Notes, after deducting the initial purchasers’ fee of $20.1 million, was approximately $784.9 million. We also incurred additional debt issuance costs totaling $0.4 million. Debt issuance costs and the initial purchasers’ fee are presented as a debt discount.
The 2027 Convertible Notes pay interest semi-annually in arrears on March 1st and September 1st of each year at an annual rate of 0.25%. The 2027 Convertible Notes are general unsecured obligations and rank senior in right of payment to all indebtedness that is expressly subordinated in right of payment to the 2027 Convertible Notes, rank equally in right of payment with all existing and future liabilities that are not so subordinated, are effectively junior to any secured indebtedness to the extent of the value of the assets securing such indebtedness and are structurally subordinated to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries. The 2027 Convertible Notes have a maturity date of March 1, 2027.
Holders may convert their 2027 Convertible Notes at their option only in the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on June 30, 2021, if the last reported sale price per share of common stock exceeds 130% of the conversion price for each of at least 20 trading days during the 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter; (2) during the five consecutive business days immediately after any five consecutive trading day period (such five consecutive trading day period, the “measurement period”) in which the trading price per $1,000 principal amount of notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day; (3) upon the occurrence of certain corporate events or distributions on our common stock, as described in the indenture for the 2027 Convertible Notes; (4) if we call such notes for redemption; and (5) at any time from, and including, September 1, 2026 until the close of business on the scheduled trading day immediately before the maturity date. As of March 31, 2026, the 2027 Convertible Notes were not convertible.
Upon conversion, we will pay cash for the settlement of principal, and for the premium, if applicable, we will pay cash, deliver shares of common stock or a combination of cash and shares of common stock, at our election. The initial conversion rate for the 2027 Convertible Notes is 12.9576 shares of common stock per $1,000 in principal amount of 2027 Convertible Notes, equivalent to a conversion price of approximately $77.17 per share of our common stock. The conversion rate is subject to adjustment.
In connection with the offering of the 2032 Convertible Notes and 2031 Convertible Notes, we used a portion of the net proceeds of the offering to enter into privately negotiated agreements with certain holders of the outstanding 2027 Convertible Notes to repurchase their 2027 Convertible Notes for cash. In connection with the repurchases, we paid approximately $676.8 million in cash, which included a premium, inducement expense and accrued interest.
As of March 31, 2026, we were in compliance with all covenants.
Revolving Credit and Term Loan Facilities
In May 2022, we entered into a credit agreement, which was subsequently amended (i) in August 2022 (the “First Amendment”), (ii) in March 2023 (the “Second Amendment”) and (iii) in November 2025 (the “Third Amendment”) with Bank of America, N.A., as Administrative Agent, Swing Line Lender and an L/C Issuer, and the other lenders and L/C Issuers party thereto (the credit agreement as amended by the First Amendment, the Second Amendment and the Third Amendment, the “2022 Credit Agreement”), evidencing a credit facility (the “2022 Facility”) that originally provided for (i) a $575 million revolving credit facility (the “Revolving Credit Facility”) and (ii) a $250 million term loan facility (the “Term Facility”). Concurrently, with the entry into the First Amendment, we repaid the entire outstanding Term Facility and repaid all outstanding loans under the Revolving Credit Facility under the 2022 Credit Agreement.
Pursuant to the Third Amendment, we (a) extended the maturity date of the existing revolving credit facility under the 2022 Credit Agreement immediately prior to the effectiveness of the Third Amendment (the “Existing Revolving Credit Facility” and, as amended and upsized as set forth in clause (b) below, the “Amended Revolving Credit Facility”), and (b) incurred additional revolving credit commitments such that the aggregate amount of commitments under the Amended Revolving Credit Facility equal $750 million in total.
Borrowings under the Amended Revolving Credit Facility bear interest at a rate equal to an applicable margin plus: (a) the applicable Term SOFR (as defined in the Credit Agreement) rate, or (b) a base rate determined by reference to the highest of (1) the federal funds effective rate plus 0.50%, (2) the Bank of America prime rate, (3) the Term SOFR rate for an interest period of one month plus 1.00%, and (4) 1.00%. The applicable margin for the Amended Revolving Credit Facility ranges, based on our consolidated total net leverage ratio, from 0.25% to 1.25% in the case of base rate loans and from 1.25% to 2.25% in the case of Term SOFR rate loans. In addition to paying interest on the outstanding principal under the Amended Revolving Credit Facility, we will pay (i) a commitment fee in respect of the unutilized commitments thereunder and (ii) customary letter of credit fees and agency fees.
After giving effect to the Third Amendment, the Amended Revolving Credit Facility will mature on the earlier of (a) November 5, 2030 and (b) the Springing Revolver Maturity Date (as defined in the 2022 Credit Agreement), unless the Amended Revolving Credit Facility is extended prior to such date in accordance with the 2022 Credit Agreement.
The 2022 Credit Agreement contains an expansion feature, which allows us, subject to certain conditions, to establish a term loan facility and/or to increase the aggregate principal amount of the Amended Revolving Credit Facility, provided that certain customary conditions are satisfied, including that our consolidated secured net leverage ratio shall not exceed 3.50 to 1.00 on a pro forma basis.
The terms of the Amended Revolving Credit Facility include certain affirmative and negative covenants as set forth in the Credit Agreement, that, among other things, may restrict our ability to: create liens on assets; incur additional indebtedness; make investments; make acquisitions and other fundamental changes; and sell and dispose of property or assets. The Credit
Agreement also includes financial covenants requiring us to maintain, measured as of the end of each fiscal quarter, a maximum consolidated net leverage ratio of 4.50 to 1.00, and a minimum consolidated interest coverage ratio of 3.00 to 1.00. If we consummate a material acquisition, the consolidated net leverage ratio covenant will be increased by 0.50 to 1.00 (to a level not to exceed 5.00 to 1.00) for a period of three fiscal quarters following such material acquisition, subject to customary conditions.
The 2022 Credit Agreement also contains customary affirmative covenants, representations and warranties and events of default. Except as amended by the Third Amendment, the terms of the 2022 Credit Agreement remain in full force and effect.
As of March 31, 2026, the revolving credit facility was undrawn and we were in compliance with all covenants.
Additional Capital Requirements
Our expected working capital and other capital requirements are described in “Part II, Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our Annual Report on Form 10-K for the year ended December 31, 2025. As of March 31, 2026, there have been no material changes to our expected working capital and other capital requirements described in our Annual Report on Form 10-K for the year ended December 31, 2025.
Critical Accounting Policies and Estimates
The discussion and analysis of our financial condition and results of operations are based on our condensed consolidated financial statements, which have been prepared in accordance with U.S. GAAP. The preparation of our condensed consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses and related disclosure of contingent assets and liabilities.
Our significant accounting policies are described in Part II, Item 8, Note 2, Summary of Significant Accounting Policies, to the consolidated financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2025. The accounting policies and estimates that are most critical to a full understanding and evaluation of our reported financial results are described in Part II, Item 7, Management’s Discussion and Analysis of Financial Condition and Results of Operations of our Annual Report on Form 10-K for the year ended December 31, 2025. There were no material changes to our critical accounting policies or estimates during the three months ended March 31, 2026.
Recent Accounting Pronouncements
Refer to Note 2, Summary of Significant Accounting Policies, of our condensed consolidated financial statements included in Part I, Item 1 for a discussion of recent accounting pronouncements and their effect, if any.
Item 3.Quantitative and Qualitative Disclosures About Market Risk
There have been no material changes in our market risks during the quarter ended March 31, 2026.
As of March 31, 2026, our cash equivalents and marketable securities consisted of investments in money market funds and U.S. Treasury securities. These investments were made in accordance with our investment policy which specifies the categories, allocations, and ratings of securities we may consider for investment. The primary objective of our investment activities is to preserve principal while at the same time maximizing the income we receive without significantly increasing risk. Some of the financial instruments that we invest in could be subject to market risk. This means that a change in prevailing interest rates may cause the value of the instruments to fluctuate. For example, if we purchase a security that was issued with a fixed interest rate and the prevailing interest rate later rises, the value of that security may decline. Based on our current investment portfolio as of March 31, 2026, we do not believe that our results of operations would be materially impacted by an immediate change of 10% in interest rates.
We hedge a portion of foreign currency exchange risk associated with forecasted royalties revenue denominated in Swiss francs to reduce the risk of our earnings and cash flows being adversely affected by fluctuations in exchange rates. These transactions are designated and qualify as cash flow hedges. The cash flow hedges are carried at fair value with mark-to-market gains and losses recorded within AOCI in our condensed consolidated balance sheets and reclassified to royalty revenue in our condensed consolidated statements of income in the same period as the recognition of the underlying hedged transaction. We do not issue derivatives, derivative commodity instruments or other financial instruments for speculative trading purposes.
Further, we do not believe our cash, cash equivalents and marketable securities have significant risk of default or illiquidity. We made this determination based on discussions with our investment advisors and a review of our holdings. While we believe our cash, cash equivalents and marketable securities do not contain excessive risk, we cannot provide absolute assurance that in the future our investments will not be subject to adverse changes in market value. All of our cash equivalents and marketable securities are recorded at fair market value.
Item 4.Controls and Procedures
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our Exchange Act reports is recorded, processed, summarized and reported within the timelines specified in the Securities and Exchange Commission’s rules and forms, and that such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decision regarding required disclosure. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can only provide reasonable assurance of achieving the desired control objectives, and in reaching a reasonable level of assurance, management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
Under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, we conducted an evaluation of our disclosure controls and procedures, as such term is defined under Rule 13a-15(e) promulgated under the Securities Exchange Act of 1934, as amended. Based on this evaluation, our principal executive officer and our principal financial officer concluded that our disclosure controls and procedures were effective as of the end of the period covered by this Quarterly Report on Form 10-Q.
Changes in Internal Control Over Financial Reporting
There have been no significant changes in our internal control over financial reporting that occurred during the quarter ended March 31, 2026 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
PART II – OTHER INFORMATION
Item 1.Legal Proceedings
From time to time, we may be involved in disputes, including litigation, relating to claims arising out of operations in the normal course of our business. Any of these claims could subject us to costly legal expenses and, while we generally believe that we have adequate insurance to cover many different types of liabilities, our insurance carriers may deny coverage or our policy limits may be inadequate to fully satisfy any damage awards or settlements. If this were to happen, the payment of any such awards could have a material adverse effect on our condensed consolidated statements of income and balance sheets. Additionally, any such claims, whether or not successful, could damage our reputation and business. We currently are not a party to any legal proceedings, the adverse outcome of which, in our opinion, individually or in the aggregate, would have a material adverse effect on our condensed consolidated statements of income or balance sheets.
Item 1A.Risk Factors
There have been no material changes to the risk factors set forth under Part I, Item 1A. “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2025.
Item 2.Unregistered Sales of Equity Securities and Use of Proceeds
In February 2024, our Board of Directors authorized a new capital return program to repurchase up to $750.0 million of our outstanding common stock. There were no share repurchases made during the first quarter of 2026. As of March 31, 2026, $157.7 million of outstanding stock is available to be purchased under the program.
In May 2026, our Board of Directors authorized a new capital return program to repurchase up to $1 billion of our outstanding common stock by December 31, 2028.
Item 3.Defaults Upon Senior Securities
Not applicable.
Item 4.Mine Safety Disclosures
Not applicable.
Item 5.Other Information
During the three months ended March 31, 2026, the following director adopted a Rule 10b5-1 trading arrangement (as such terms are defined pursuant to Item 408 of Regulation S-K) as noted in the table below.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Trading Arrangement | | | | |
Name and Title | | Action | | Date | | Rule 10b5-1* | | Non-Rule 10b5-1** | | Total Shares To Be Sold | | Expiration Date |
Mahesh Krishnan | | Adoption | | 3/19/2026 | | X | | | | 7,304 | | 6/1/2027 |
Director |
* Contract, instruction or written plan intended to satisfy the affirmative defense conditions of Rule 10b5-1(c) under the Exchange Act.
** Non-Rule 10b5-1 trading arrangement as defined in Item 408(c) of Regulation S-K under the Exchange Act
Item 6.Exhibits
| | | | | | | | |
| 3.1 | | |
| |
| 3.2 | | |
| | |
| 4.1 | | |
| | |
| 4.2 | | |
| | |
| 4.3 | | |
| | |
| 4.4 | | |
| | |
| 4.5 | | |
| | |
| 4.6 | | |
| | |
| 4.7 | | |
| | |
| 4.8 | | |
| | |
| 31.1 | | |
| |
| 31.2 | | |
| |
| 32 | | |
| |
| 101.INS | | Instance Document - the instance document does not appear in the interactive Data File because its XBRL tags are embedded within the Inline XBRL document (filed herewith) |
| | |
| 101.SCH | | Inline Taxonomy Extension Schema Document (filed herewith) |
| | |
| 101.CAL | | Inline Taxonomy Extension Calculation Linkbase Document (filed herewith) |
| | |
| 101.DEF | | Inline Taxonomy Extension Definition Linkbase Document (filed herewith) |
| | |
| 101.LAB | | Inline Taxonomy Extension Label Linkbase Document (filed herewith) |
| | |
| 101.PRE | | Inline Taxonomy Extension Presentation Linkbase Document (filed herewith) |
| | |
| 104 | | Cover Page Interactive Data File (formatted as inline XBRL and contained in Exhibit 101) (filed herewith) |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| | | | | | | | | | | |
| | | Halozyme Therapeutics, Inc. (Registrant) |
| | |
| | | |
| Dated: | May 11, 2026 | By: | /s/ Helen I. Torley, M.B. Ch.B., M.R.C.P. |
| | | Helen I. Torley, M.B. Ch.B., M.R.C.P.
President and Chief Executive Officer
(Principal Executive Officer) |
| | |
| | |
| | | |
| Dated: | May 11, 2026 | By: | /s/ David Ramsay |
| | | David Ramsay Interim Chief Financial Officer (Principal Financial and Accounting Officer) |
| | |
| | |