
Exhibit 99.2 Q1 2026 Earnings Call & Business Update May 14, 2026

Exhibit 99.2 Q1 2026 Earnings Call & Business Update May 14, 2026

CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTSThis presentation contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995. Statements in this presentation that are not statements of historical fact are forward-looking statements. Words such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “seek,” “could,” “intend,” “target,” “aim,” “project,” “designed to,” “estimate,” “believe,” “predict,” “potential,” or “continue” or the negative of these terms or other similar expressions are intended to identify forward-looking statements, though not all forward-looking statements contain these identifying words. Forward-looking statements include statements concerning, among other things, plans related to the company’s research and development activities, and the timing and potential results thereof; expectations regarding the company’s clinical trial designs, enrollment, event accumulation and progress, regulatory pathway, product profile, potential patient populations, indication, and administration paradigm for VYD2311, including with respect to the company’s REVOLUTION clinical program and the timing of activities, expenditures, and results related thereto; the company’s commercialization plans, strategies, goals, and expectations, including with respect to its preparations for the potential commercial launch of VYD2311, if approved; expectations regarding the COVID landscape and potential advantages of monoclonal antibodies (mAbs); estimates based on arithmetic extrapolation of systemic reactogenicity symptom burden at the population level; the potential net symptomatic benefit of immunization from a very low vaccine efficacy mAb; the company’s plans and expectations with respect to the commercialization of PEMGARDA® (pemivibart); the potential of VYD2311 as a novel mAb candidate that may be able to deliver clinically meaningful titer levels through more patient-friendly means; the company’s plans and expectations with respect to its other product candidates, including VMS063 and VBY329; expectations about the market size and opportunity for the company’s product candidates, as well as its market position; expectations regarding the company’s ‘Antibodies for Any Body’ campaign and partnership with Lindsey Vonn; the company’s business strategies and objectives, and ability to execute on them; the company’s potential to change COVID-19 prevention near-term with a potential best-in-class technology, and major follow-on opportunities; the company’s future prospects; and other statements that are not historical fact. The company may not actually achieve the plans, intentions, or expectations disclosed in the company’s forward-looking statements and you should not place undue reliance on the company’s forward-looking statements. These forward-looking statements involve risks and uncertainties that could cause the company’s actual results to differ materially from the results described in or implied by the forward-looking statements, including, without limitation: uncertainties regarding the company’s expectations, projections, and estimates regarding future costs and expenses, future revenue, capital requirements, and the availability of and the need for additional financing; uncertainties regarding market acceptance, payor coverage, and reimbursement, or future revenue generated by any authorized or approved product; how long the emergency use authorization (EUA) granted by the U.S. Food & Drug Administration (FDA) for PEMGARDA will remain in effect and whether such EUA is revised or revoked by the FDA; the ability to maintain a continued acceptable safety, tolerability, and efficacy profile of any product candidate following regulatory authorization or approval; the success of the company’s in-house sales force, and the company’s ability to maintain and expand sales, marketing, and distribution capabilities to successfully commercialize any authorized or approved product; changes in expected or existing competition; changes in the regulatory environment; the outcome of the company’s engagement with regulators; uncertainties related to the regulatory authorization or approval process, and available development and regulatory pathways; whether or not any preclinical candidate identified by the company is determined to be suitable for clinical development; the timing, progress, and results of the company’s discovery, preclinical, and clinical development activities; clinical trial site activation, enrollment, and event accumulation rates; unexpected safety or efficacy data observed during preclinical studies or clinical trials; the risk that results of nonclinical studies or clinical trials may not be predictive of future results, and interim data are subject to further analysis; the company’s ability to generate the data needed to support a potential Biologics License Application (BLA) submission for VYD2311; potential variability in neutralizing activity of product candidates tested in different assays, such as pseudovirus assays and authentic assays; variability of results in models and methods used to predict activity against SARS-CoV-2 variants; whether the epitopes that pemivibart and VYD2311 target remain structurally intact and the company’s product candidates are able to demonstrate and sustain neutralizing activity against major SARS-CoV-2 variants, particularly in the face of viral evolution; the risk that a lack of awareness of mAb therapies and regulatory scrutiny of mAb therapies to prevent or treat COVID-19 or other infectious diseases may adversely impact the development or commercial success of the company’s product candidates; the company’s reliance on third parties; whether the anticipated benefits of the company’s partnership with Lindsey Vonn are realized; complexities of manufacturing mAb therapies; macroeconomic and political uncertainties; the company’s ability to continue as a going concern; and whether the company has adequate funding to meet future operating expenses and capital expenditure requirements. Other factors that may cause the company’s actual results to differ materially from those expressed or implied in the forward-looking statements in this presentation are described under the heading “Risk Factors” in the company’s Annual Report on Form 10-K for the year ended December 31, 2025, as filed with the Securities and Exchange Commission (SEC), and in the company’s other filings with the SEC, and in its future reports to be filed with the SEC and available at www.sec.gov. Forward-looking statements contained in this press release are made as of this date, and Invivyd undertakes no duty to update such information whether as a result of new information, future events or otherwise, except as required under applicable law.This presentation contains hyperlinks to information that is not deemed to be incorporated by reference in this presentation. All trademarks used in this presentation are the property of their respective owners.

01 Executive Summary02 Clinical and Regulatory03 Virology and Pipelineagenda04 Commercial05 Financial06 Q&A
Executive SummaryRapid continued enrollment of DECLARATION pivotal study post-upsizing (+500 subjects)Expanded government affairs and corporate communications, including recent participation at POLITICO Health Care Summit and launch of ‘Antibodies for Any Body’ Campaign with Lindsey VonnNew preprint describing tolerability of low-dose intramuscular (IM) investigational COVID mAb, adintrevimab, compared to COVID-19 vaccines; LIBERTY clinical trial to evaluate comparative safety between VYD2311 and mRNA COVID-19 vaccineNew in vitro data showed positive neutralization activity of pemivibart and VYD2311 against emerging SARS-CoV-2 variant BA.3.2.2 (“Cicada”)Continued buildout of commercial organization in preparation for VYD2311 launch, if approved

Invivyd: The Big PicturePEMGARDA® ongoing growth in the face of declining vaccine utilization More rapid than expected demand for DECLARATION recruitment Line of sight to changing New analysis showing benefit of highly tolerable mAb vs vaccine COVID-19 prevention near-Broad consumer education term with a potential best-in-on immunology beginning Ongoing pivotal program execution class technology, and major Near-term planned Long COVID study launch follow-on opportunities Early pipeline broadening to include many additional vaccine preventable pathogens

Government Affairs UpdateSubstantial increase in engagement with current administration and advisors Clear demonstrated interest in improvements to public health via novel vaccine-alternatives Plan to continue building awareness with long-term view toward scale and public healthAll trademarks and logos displayed are the property of their respective owners. Their use here is for identification purposes only and does not constitute endorsement or affiliation.

Appreciating monoclonal antibodies appears universal“[COVID mAb] wasn’t just a therapeutic. It just made me better. I “Give them the goddamn monoclonal antibodies when they get sick...”call that a cure.” - Joe Rogan, Podcaster, Nov. 2021– President Donald Trump, Oct. 2020 “It was very disappointing that the administration was only focusing on “mAbs are positioned to play a larger role in future public health responses leaning forward on vaccines and not really choosing to do the same for involving the diagnosis, prevention, and treatment of EIDs, and the lessons learned antibodies. There should have been no hesitation on leaning forward to will most likely apply to infectious diseases in general. If we are to fully realize address the supply chain and the volumes that we knew that we would mAbs’ promise in EIDs, leaders in preparedness and response will have to assign need.” them a high priority in research and development agendas.” – Dr. Rick Bright, Former Director at BARDA,—Dr. Hilary D. Marston, Former CMO at FDA, Dr. Oct. 2020 Anthony Fauci, NIAID Director, Mar. 2018“Florida has administered over 130,000 monoclonal antibody treatments, “I would give myself [REGEN-COV] if I was infected by COVID-19 and I saving thousands of people from being admitted to the hospital. There is no would give it to a loved one” doubt that many lives have been saved as a result of making these – George Yancopoulos, Regeneron Co-founder, treatments widely available,”President & CSO, Oct. 2020 – Ron DeSantis, Florida Governor, Oct. 2021“There’s no question this treatment saves lives when given to “If I was in that position right now, I would be wanting to use these drugs on a appropriate patients.” regular basis to protect me. – Robert Califf, Former FDA Commissioner, – Scott Gottlieb, Former FDA Commissioner, Oct. 2024 Apr. 2021

Lindsey Vonn Educating Americans on Antibody ImmunologyAll trademarks and logos displayed are the property of their respective owners. Their use here is for identification purposes only and does not constitute endorsement or affiliation.

01 Executive Summary02 Clinical and Regulatory03 Virology and Pipelineagenda04 Commercial05 Financial06 Q&A

VYD2311: REVOLUTION Program UpdatesPivotal Efficacy Study mAb & Vaccine Safety Pediatric Study and Immunology Study• Announced modest upsizing based on • Aligned with FDA on Phase 3 trial to • Agreed with FDA on Initial Pediatric conservative, pre-specified analysis examine safety and immunology of Study Plan to support BLA filing• Continued high demand and rapid VYD2311 combined with and versus an • Plan for single study to assess the enrollment in expansion cohort mRNA vaccine immunogenicity & safety of VYD2311 in • IDMC reviews of blinded safety data • Final protocol submitted to FDA children 0 – 11 years, with efficacy support reduced monitoring time, • Operational start-up activities underway extrapolation from DECLARATION reduced visits, and inclusion of pregnant & breastfeeding womenInitiation pending DECLARATION Topline data expected Q3 2026 On track to initiate Q2 2026 success
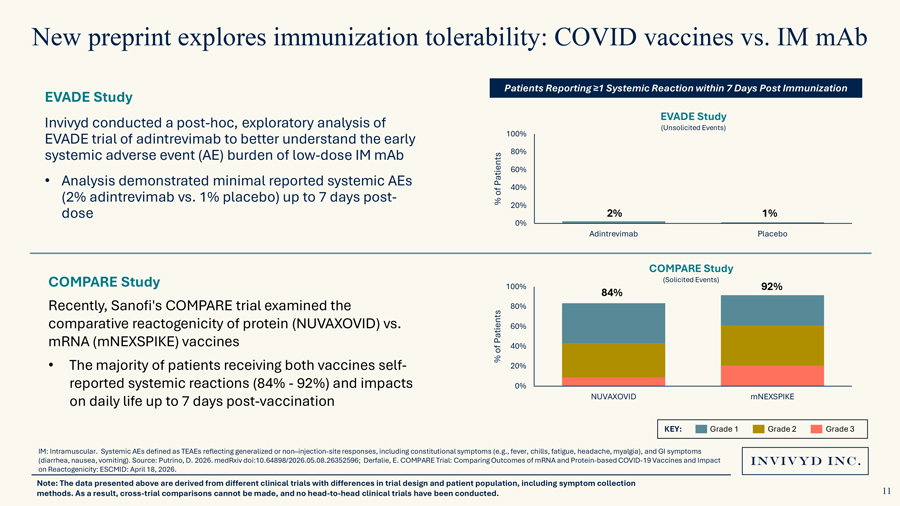
New preprint explores immunization tolerability: COVID vaccines vs. IM mAbPatients Reporting ≥1 Systemic Reaction within 7 Days Post ImmunizationEVADE StudyInvivyd conducted a post-hoc, exploratory analysis of EVADE Study(Unsolicited Events)EVADE trial of adintrevimab to better understand the early 100% systemic adverse event (AE) burden of low-dose IM mAb 80%60%• Analysis demonstrated minimal reported systemic AEs Patients40%(2% adintrevimab vs. 1% placebo) up to 7 days post- of % 20% 2% 1%dose0%Adintrevimab PlaceboCOMPARE StudyCOMPARE Study (Solicited Events)100% 84% 92%Recently, Sanofi’s COMPARE trial examined the 80% comparative reactogenicity of protein (NUVAXOVID) vs. Patients 60% mRNA (mNEXSPIKE) vaccines of 40%% • The majority of patients receiving both vaccines self- 20% reported systemic reactions (84%—92%) and impacts 0% on daily life up to 7 days post-vaccination NUVAXOVID mNEXSPIKEKEY: Grade 1 Grade 2 Grade 3IM: Intramuscular. Systemic AEs defined as TEAEs reflecting generalized or non–injection-site responses, including constitutional symptoms (e.g., fever, chills, fatigue, headache, myalgia), and GI symptoms (diarrhea, nausea, vomiting). Source: Putrino, D. 2026. medRxiv doi:10.64898/2026.05.08.26352596; Derfalie, E. COMPARE Trial: Comparing Outcomes of mRNA and Protein-based COVID-19 Vaccines and Impact on Reactogenicity: ESCMID: April 18, 2026.Note: The data presented above are derived from different clinical trials with differences in trial design and patient population, including symptom collection methods. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted.
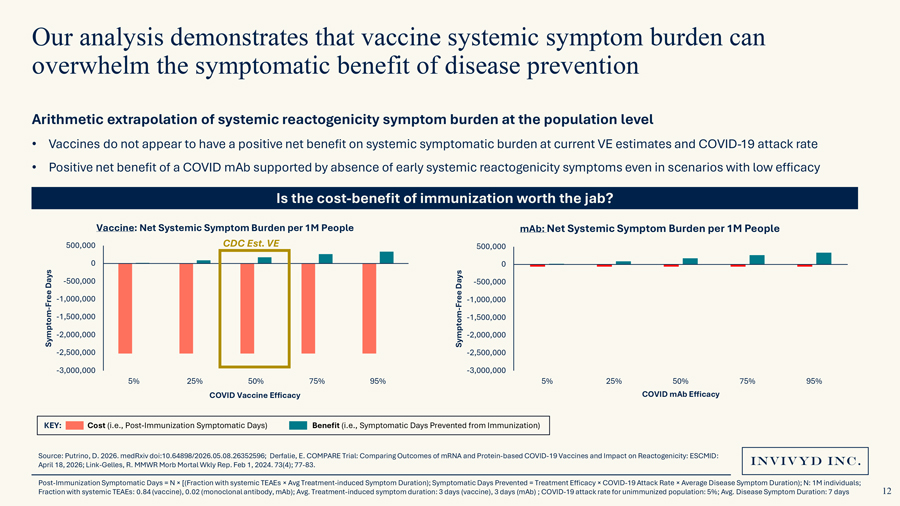
Our analysis demonstrates that vaccine systemic symptom burden can overwhelm the symptomatic benefit of disease preventionArithmetic extrapolation of systemic reactogenicity symptom burden at the population level • Vaccines do not appear to have a positive net benefit on systemic symptomatic burden at current VE estimates and COVID-19 attack rate• Positive net benefit of a COVID mAb supported by absence of early systemic reactogenicity symptoms even in scenarios with low efficacyIs the cost-benefit of immunization worth the jab?Vaccine: Net Systemic Symptom Burden per 1M People mAb: Net Systemic Symptom Burden per 1M People500,000 CDC Est. VE 500,0000 0 Days -500,000 Days -500,000 Free -1,000,000 Free -1,000,000—-1,500,000 -1,500,000 Symptom -2,000,000 Symptom -2,000,000 -2,500,000 -2,500,000 -3,000,000 -3,000,0005% 25% 50% 75% 95% 5% 25% 50% 75% 95%COVID Vaccine Efficacy COVID mAb Efficacy KEY: Cost (i.e., Post-Immunization Symptomatic Days) Benefit (i.e., Symptomatic Days Prevented from Immunization)Source: Putrino, D. 2026. medRxiv doi:10.64898/2026.05.08.26352596; Derfalie, E. COMPARE Trial: Comparing Outcomes of mRNA and Protein-based COVID-19 Vaccines and Impact on Reactogenicity: ESCMID: April 18, 2026; Link-Gelles, R. MMWR Morb Mortal Wkly Rep. Feb 1, 2024. 73(4); 77-83. Post-Immunization Symptomatic Days = N × [(Fraction with systemic TEAEs × Avg Treatment-induced Symptom Duration); Symptomatic Days Prevented = Treatment Efficacy × COVID-19 Attack Rate × Average Disease Symptom Duration); N: 1M individuals; Fraction with systemic TEAEs: 0.84 (vaccine), 0.02 (monoclonal antibody, mAb); Avg. Treatment-induced symptom duration: 3 days (vaccine), 3 days (mAb) ; COVID-19 attack rate for unimmunized population: 5%; Avg. Disease Symptom Duration: 7 days

Key TakeawaysA very low VE (15-20%) monoclonal antibody could improve net systemic symptomatic benefit from immunizationCOVID vaccines generate meaningful symptom burden with uncertain benefit in the contemporary populationSafety and tolerability of immunization is critical for broad uptake in healthy individualsVE: Vaccine Efficacy. Source: Putrino, D. 2026. medRxiv doi:10.64898/2026.05.08.26352596; Derfalie, E. COMPARE Trial: Comparing Outcomes of mRNA and Protein-based COVID-19 Vaccines and Impact on Reactogenicity: ESCMID: April, 18, 2026; NFID. 2024 National Survey. Sept 25, 2024. Accessed May 7, 2026; Gshwend, M. Vaccine; 2026: 72.

01 Executive Summary02 Clinical and Regulatory03 Virology and Pipelineagenda04 Commercial05 Financial06 Q&A
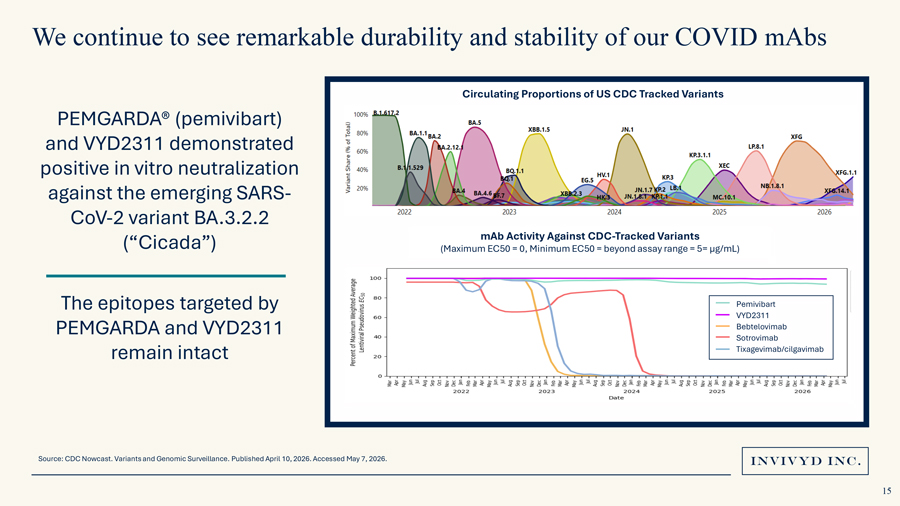
We continue to see remarkable durability and stability of our COVID mAbsCirculating Proportions of US CDC Tracked VariantsPEMGARDA® (pemivibart) and VYD2311 demonstrated positive in vitro neutralization against the emerging SARS-CoV-2 variant BA.3.2.2 (“Cicada”) mAb Activity Against CDC-Tracked Variants(Maximum EC50 = 0, Minimum EC50 = beyond assay range = 5= µg/mL)The epitopes targeted by PemivibartVYD2311PEMGARDA and VYD2311 BebtelovimabSotrovimab remain intact Tixagevimab/cilgavimabSource: CDC Nowcast. Variants and Genomic Surveillance. Published April 10, 2026. Accessed May 7, 2026.

Continue to advance broader pipeline of monoclonal antibodies to combat infectious diseaseMeasles RSV VMS063 VYD329• Announced advancement of VMS063, a novel, potentially best-in- • Selected potential best-in-class mAb for the treatment and prevention of measles class RSV antibody candidate• Demonstrated highly potent and broad in vitro neutralization across • Demonstrated 1.5× potency relevant measles lineages: ~4.2 ng/mL IC50 in authentic virus vs. nirsevimab and 1.2× assays and ~1.4 ng/mL IC50 in pseudovirus assays potency vs. clesrovimab in • Half-life extended to support potential prophylactic and therapeutic vitro use with a single dose • Half-life extension expected to • Presented initial data at TAVI and World Vaccine Congress equal or exceed current SoCTargeting IND readiness Advancing toward IND in late 2026 readiness in 2H 2026Source: Allen, Robert. Title. TAVI; April 2026; Munich, Germany.

01 Executive Summary02 Clinical and Regulatory03 Virology and Pipelineagenda04 Commercial05 Financial06 Q&A

PEMGARDA – Continued Growth$13.7M PEMGARDA Q1 ’26 Established Strong Commercial Foundation 22% YoY Growth Net Product Revenue~1,000 Accounts w/ PEMGARDA Infusion Experience Building from Specialty Infusion Team – Scaling to serve broad market for VYD2311, if approved~1,850 Available Sites Infusing Conferences/ Online backbone being built for broad engagement; ~165 Exhibits Attended VYD2311 requires a greater presence~19,500 GPO Contracted SitesAccess – National Payors & Distribution for broad adoption77% Reordering AccountsAll metrics are Launch to Date (LTD 3/26)

Engaging HCPs via the AI they use most – Building an innovative footprint Early Results EncouragingPEMGARDA HCP queries increased in target cliniciansAI powered platform and medical search engine used by healthcare providers for clinical decision support. 300%in Oncology140%in Infectious Diseaseseeing an increasein new to brand

Engaging HCPs via the AI they use most – Building an innovative footprint Early Results EncouragingPEMGARDA HCP queries increased in target cliniciansAI powered platform and medical search engine used by healthcare providers for clinical decision support. 300%in Oncology140%in Infectious Diseaseseeing an increasein new to brand

01 Executive Summary02 Clinical and Regulatory03 Virology and Pipelineagenda04 Commercial05 Financial06 Q&A
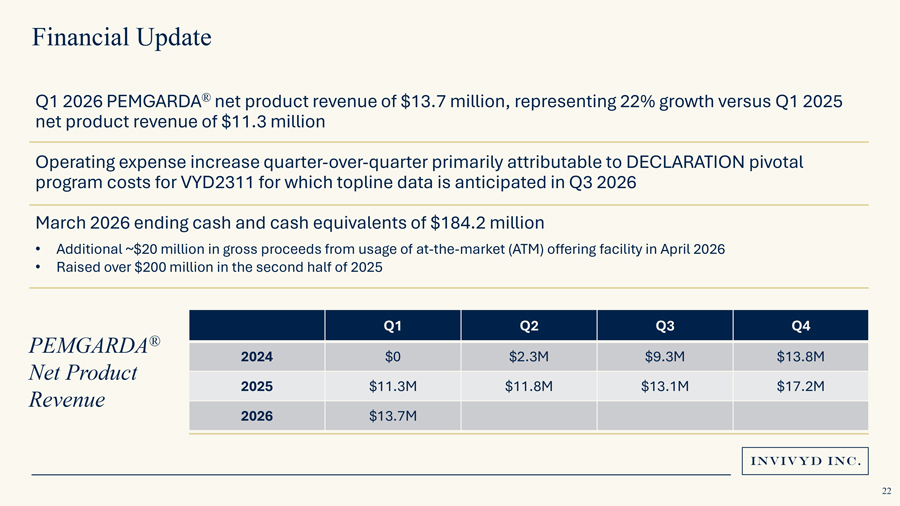
Financial UpdateQ1 2026 PEMGARDA® net product revenue of $13.7 million, representing 22% growth versus Q1 2025 net product revenue of $11.3 million Operating expense increase quarter-over-quarter primarily attributable to DECLARATION pivotal program costs for VYD2311 for which topline data is anticipated in Q3 2026 March 2026 ending cash and cash equivalents of $184.2 million• Additional ~$20 million in gross proceeds from usage of at-the-market (ATM) offering facility in April 2026• Raised over $200 million in the second half of 2025® Q1 Q2 Q3 Q4PEMGARDA 2024 $0 $2.3M $9.3M $13.8MNet Product 2025 $11.3M $11.8M $13.1M $17.2MRevenue2026 $13.7M
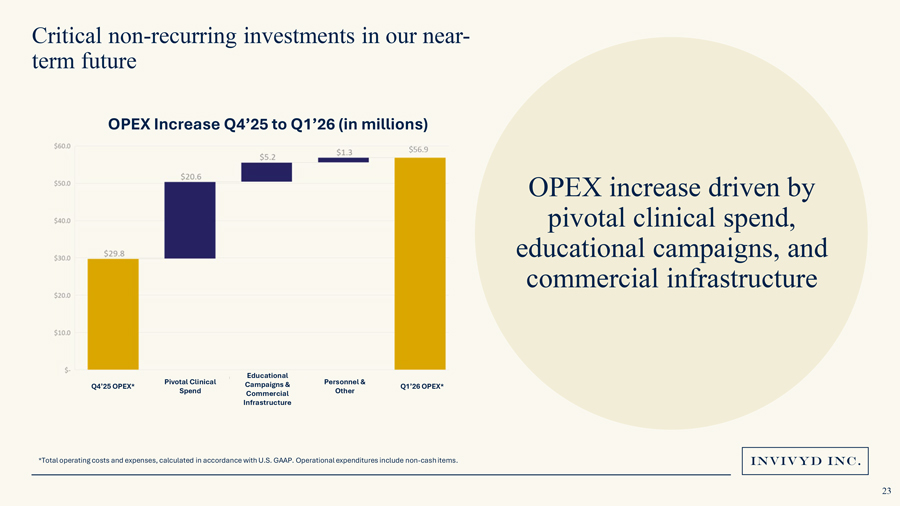
Critical non-recurring investments in our near-term futureOPEX Increase Q4’25 to Q1’26 (in millions)OPEX increase driven by pivotal clinical spend, educational campaigns, and commercial infrastructureEducational Pivotal Clinical Campaigns & Personnel & Q4’25 OPEX* Q1’26 OPEX* Spend Commercial Other Infrastructure*Total operating costs and expenses, calculated in accordance with U.S. GAAP. Operational expenditures include non-cash items.

Q&A
