
Advancing medicines. Solving problems. Improving lives. First Quarter 2026 Earnings Supplemental Materials May 13, 2026

© 2026 Aquestive Therapeutics, Inc. 2 Disclaimer Certain statements in this presentation include “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “believe,” “anticipate,” “plan,” “expect,” “estimate,” “intend,” “may,” “will,” or the negative of those terms, and similar expressions, are intended to identify forward-looking statements. These forward-looking statements include, but are not limited to, statements regarding the advancement and related timing of our product candidate Anaphylm (dibutepinephrine) sublingual film through clinical development and approval by the FDA, including our ability to address the concerns raised by the FDA in the CRL dated January 30, 2026 and the Type A meeting with the FDA, and the timing of our resubmission and FDA review of the NDA; the advancement and related timing of potential international regulatory filings and marketing authorizations for Anaphylm outside of the U.S.; that Anaphylm will be the first and only oral administration of epinephrine, if Anaphylm is approved by the FDA; the advancement, growth and related timing of our AdrenaVerse pipeline epinephrine prodrugs, including AQST-108 (epinephrine) topical gel, through clinical development and the FDA regulatory approval process, including with respect to the design and timing of clinical studies, including those necessary to support the targeted indication of alopecia areata, and potential other treatment indications for AQST-108; market access for Libervant® (diazepam) buccal film for epilepsy patients experiencing acute repetitive seizures (ARS) upon expiration of orphan drug market exclusivity of an approved FDA product of another company; the future commercial opportunity of Anaphylm, Libervant and AQST-108 should these product candidates be approved by the FDA; the potential benefits our product candidates could bring to patients, including with respect to Anaphylm, Libervant and AQST-108, if these product candidates are approved by the FDA, and acceptance by patients, prescribers and payors of our product candidates as an alternative to existing standards of care for the targeted medical indication of these product candidates; that the Company is sufficiently capitalized with sufficient cash in 2026 to perform the necessary clinical work and provide the additional information required to address the concerns of the FDA outlined in the CRL and Type A meeting; our cash requirements, cash funding and cash burn; short-term and longer term liquidity, including access to additional funds if Anaphylm is approved by the FDA, and the ability to fund our business operations and key objectives in 2026 and beyond, including the launch of Anaphylm, if approved by the FDA; our growth and future financial and operating results and financial position, including with respect to our 2026 financial outlook; and business strategies, market opportunities, and other statements that are not historical facts. Such forward-looking statements also include statements regarding anticipated timelines, milestones, and guidance relating to regulatory submissions, clinical studies, regulatory interactions, and potential approvals, which are inherently uncertain and subject to change based on regulatory feedback, protocol alignment, data sufficiency, and other factors outside the Company’s control. These forward-looking statements are based on our current expectations and beliefs and are subject to a number of risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Such risks and uncertainties include, but are not limited to, risks associated with our development work, including any delays or changes to the timing, cost and success of our product development activities and clinical trials and plans for Anaphylm and AQST-108; risk of delays in advancement of the regulatory approval process through the FDA of our product candidates Anaphylm, Libervant and AQST-108, or failure to receive FDA approval at all of any or all of these product candidates; risk of the Company’s ability to generate sufficient clinical and other human factor data, including with respect to our submission of pharmacokinetic and pharmacodynamic (PK/PD) comparability data for FDA approval of Anaphylm; risks associated with our ability to address the FDA’s comments on and identified deficiencies in our NDA, including the concerns raised by the FDA in the CRL and Type A meeting for Anaphylm, and whether the FDA may request further information from us (including additional clinical studies), disagree with our protocols, study designs, and findings or otherwise undertake a lengthy review of the resubmission of our NDA; challenges regarding the following commercial launch of Anaphylm, if approved by the FDA; risk of delays in advancement of the regulatory approval process of our product candidates, including Anaphylm and Libervant, outside of the U.S., or failure to receive approval at all of any or all of these product candidates by such foreign regulatory authorities, including risks that regulatory authorities outside the United States may require different, additional, or more extensive clinical, non-clinical, human factors, pharmacokinetic, or manufacturing data than anticipated, or may not accept data generated for U.S. regulatory purposes; risk of FDA inspections of manufacturing and clinical study sites for any of our product candidates, including Anaphylm, Libervant and AQST- 108; risk of government shutdowns or actions to reduce government workforces on the ability of the FDA to act on a timely basis or at all on the approval of our product candidates, including Anaphylm, Libervant and AQST-108; risks associated with the success of any competing products, including generics; risks and uncertainties inherent in commercializing a new product (including technology risks, financial risks, market risks and implementation risks and regulatory limitations); risk of development of a sales and marketing capability for commercialization of our product candidates, including Anaphylm, Libervant and AQST-108, if these product candidates are approved by the FDA; risks associated with the potential impact on the value of the Company of the sale or outlicensing of our product candidates, including Anaphylm, Libervant and AQST-108; risk of sufficient capital and cash resources, including sufficient access to available debt and equity financing, including under our debt and ATM facilities, and revenues from operations, to satisfy all of our short-term and longer- term liquidity and cash requirements to support our business operations, key initiatives and growth strategy, and other cash needs, at the times and in the amounts needed, and to fund future clinical development and commercial activities for our product candidates, including Anaphylm, Libervant and AQST-108, should these product candidates be approved by the FDA, including risks that assumptions underlying projected cash runway, liquidity, and capital sufficiency may prove incorrect due to changes in operating plans, regulatory requirements, timing or scope of clinical activities, market conditions, or the availability, timing, and terms of financing; risk of the impact of our obligations under the Company's Purchase Agreement and the Royalty Rights Agreement with third parties, each of which agreements requires the Company to make payments to each counterparty thereof, respectively, of a portion of our revenues, on our ability to contribute to the funding of our operations; risk that our manufacturing capabilities will be sufficient to support demand of our product candidates in the U.S. and abroad, including Anaphylm and Libervant, if such product candidates should be approved by the FDA and other regulatory authorities, and our licensed products in the U.S. and abroad; risk of eroding market share for Suboxone® as a sunsetting product, which accounts for a substantial part of our current operating revenue; risk of default of our debt instruments; risks related to the outsourcing of certain sales, marketing and other operational and staff functions to third parties; risk of the rate and degree of market acceptance in the U.S. and abroad of our product candidates, including Anaphylm, Libervant, and AQST-108 should these product candidates be approved by the FDA and other regulatory authorities, and for our licensed products in the U.S. and abroad; risk associated with the size and growth of our product markets and expected related revenues and sales; risk associated with our compliance with all FDA and other governmental and customer requirements for our manufacturing facilities; risks associated with intellectual property rights and infringement claims relating to our products; risk that our patent applications for our product candidates, including for Anaphylm, will not be timely issued, or issued at all, by the U.S. Patent and Trademark Office or, if issued, will be sufficient to provide long-term commercial success of these product candidates; risk of unexpected patent developments; risk of legislation and regulatory actions and changes in laws or regulations affecting our business, including relating to our products and product candidates and product pricing, reimbursement or access therefor; risk of loss of significant customers; risks related to claims and legal proceedings against us including patent infringement, securities, business torts, investigative, product safety or efficacy and antitrust litigation matters; risk of product recalls and withdrawals; risks related to any disruptions in our information technology networks and systems, including the impact of cybersecurity attacks; risk of increased cybersecurity attacks and data accessibility disruptions, including due to remote working arrangements; risk of adverse developments affecting the financial services industry; risks related to inflation and changing interest rates; risks related to the impact of pandemic diseases on our business; risks and uncertainties related to general economic, political (including the Ukraine, Israel and Iraq wars and other acts of war and terrorism), business, industry, regulatory, financial and market conditions and other unusual items; risks related to uncertainty about presidential administration initiatives and their impact on our business, including imposition of government tariffs and other trade restrictions; and other uncertainties affecting the Company including those described in the "Risk Factors" section and in other sections included in the Company’s Annual Report on 10-K, Quarterly Reports on Form 10-Q, and Current Reports on Form 8-K filed with the U.S. Securities and Exchange Commission. Given those uncertainties, you should not place undue reliance on these forward-looking statements, which speak only as of the date made. All subsequent forward-looking statements attributable to the Company or any person acting on its behalf are expressly qualified in their entirety by this cautionary statement. The Company assumes no obligation to update forward-looking statements or outlook or guidance after the date of this press release whether as a result of new information, future events or otherwise, except as may be required by applicable law. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy any of the Company’s securities, nor shall there be any sale of these securities in any state or other jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or other jurisdiction. PharmFilm®, Libervant® and the Aquestive logo are registered trademarks of Aquestive Therapeutics, Inc. The trade name “Anaphylm” for AQST-109 has been conditionally approved by the FDA. Final approval of the Anaphylm proprietary name is conditioned on FDA approval of the product candidate, AQST-109. All other registered trademarks referenced herein are the property of their respective owners.

3 Q1 2026 Earnings Key Messages Anaphylm (dibutepinephrine) sublingual film for severe allergic reactions, including anaphylaxis • Continue to guide to a Q3 2026 resubmission of the Anaphylm New Drug Application (NDA) • Affirms existing clinical data is sufficient for submitting regulatory applications in Canada, European Union, and the United Kingdom • Expanded Medical Affairs team continues to focus on building community awareness AQST-108 (epinephrine) topical gel and the expansion of AdrenaVerse platform • Topline results received from completed dosing in Phase 1 study in patients with androgenic alopecia and healthy normal subjects • Continue to explore other possible dermatological indications for AQST-108 including atopic dermatitis, androgenic alopecia and hypertrophic scars Strengthened balance sheet provides needed cash to support Anaphylm approval and progress Anaphylm ex-U.S. • Extended RTW Strategic Funding Agreement until June 2027 • Secured a $150 million debt facility with Oak Tree Capital Management, a leading life sciences debt provider • Ended Q1 2026 with approximately $110.7 million in cash and cash equivalents

Anaphylm (dibutepinephrine) sublingual film

5 Planned Human Factor (HF) Validation Study Design¹ • 9 distinct user groups with 15 participants each (per FDA guidance) intended to represent the demographics and characteristics of intended Anaphylm population • Adult Patients (split into 3 groups: 2 untrained and 1 familiarized with the Anaphylm product) • Epinephrine Naïve (n=15) • Epinephrine Experienced (n=15) • Familiarized (n=15) • Pediatric Patients (split into 3 groups: 2 untrained and 1 familiarized with the Anaphylm product) • Epinephrine Naïve (n=15) • Epinephrine Experienced (n=15) • Familiarized (n=15) • School Nurses (n=15) • EMTs (n=15) • Passersby (n=15) (epinephrine naïve) 1. Study protocol under review by the FDA.

6 All subjects will be required to receive: • An FDA approved epinephrine injection product • Anaphylm administration by trained study staff • Randomized to periods 1 and 3 Some subjects will also: • Use alternate Anaphylm site (top of tongue) administration consistent with the Complete Response Letter (CRL) • Randomized to periods 1 and 3 • Self-administer Anaphylm using updated product labeling • Using information from the HF validation study • Randomized to periods 1 and 3 Study Size: 48 Subjects Planned Pharmacokinetic (PK) Study Design¹ Period 1 Study Day 1 Randomized Anaphylm Administration HealthCare Provider, Self-administration or Top of Tongue Period 2 Study Day 2 Injectable Epi Administration Period 3 Study Day 15 Randomized Anaphylm Administration HealthCare Provider, Self-administration or Top of Tongue 1. Study protocol under review by the FDA.

7 Expand Awareness of Anaphylm Medical Affairs broadens awareness and enhances familiarity with healthcare providers and the allergy community • Establish relationships, leverage existing reputations, expertise and networks • Hired a dynamic Medical Affairs team to engage the allergy community • Comprehensive medical/scientific publication and presentation strategy to convey mechanism of action and place in therapy through clinical data • Strong, top-down Medical Affairs presence at regional and national meetings • Actively engage the advocacy community

AQST-108 (epinephrine) topical gel
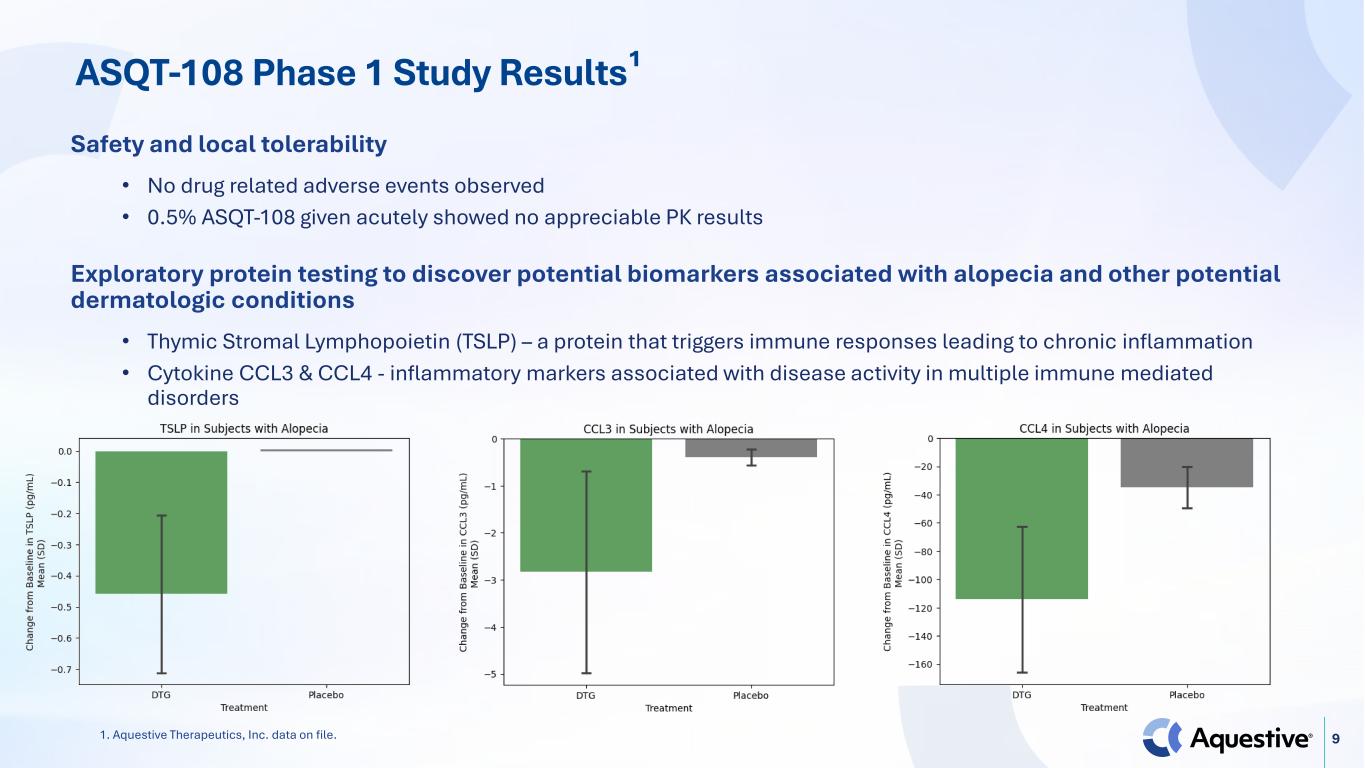
9 Safety and local tolerability • No drug related adverse events observed • 0.5% ASQT-108 given acutely showed no appreciable PK results Exploratory protein testing to discover potential biomarkers associated with alopecia and other potential dermatologic conditions • Thymic Stromal Lymphopoietin (TSLP) – a protein that triggers immune responses leading to chronic inflammation • Cytokine CCL3 & CCL4 - inflammatory markers associated with disease activity in multiple immune mediated disorders ASQT-108 Phase 1 Study Results¹ 1. Aquestive Therapeutics, Inc. data on file.

Key Upcoming Milestones
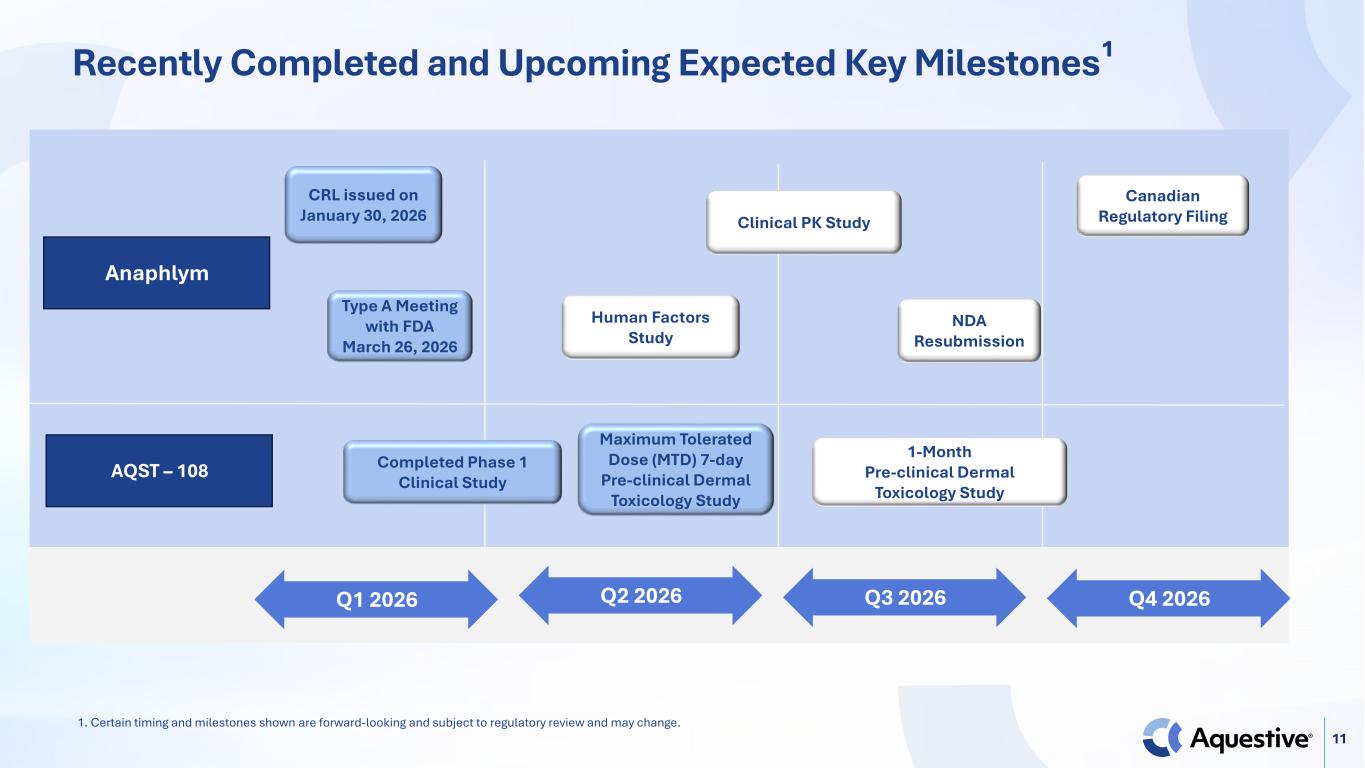
11 Recently Completed and Upcoming Expected Key Milestones¹ AQST – 108 Q4 2026Q1 2026 Q2 2026 Q3 2026 Anaphlym CRL issued on January 30, 2026 Canadian Regulatory Filing Completed Phase 1 Clinical Study Type A Meeting with FDA March 26, 2026 NDA Resubmission Human Factors Study Clinical PK Study Maximum Tolerated Dose (MTD) 7-day Pre-clinical Dermal Toxicology Study 1-Month Pre-clinical Dermal Toxicology Study 1. Certain timing and milestones shown are forward-looking and subject to regulatory review and may change.

First Quarter 2026 Results
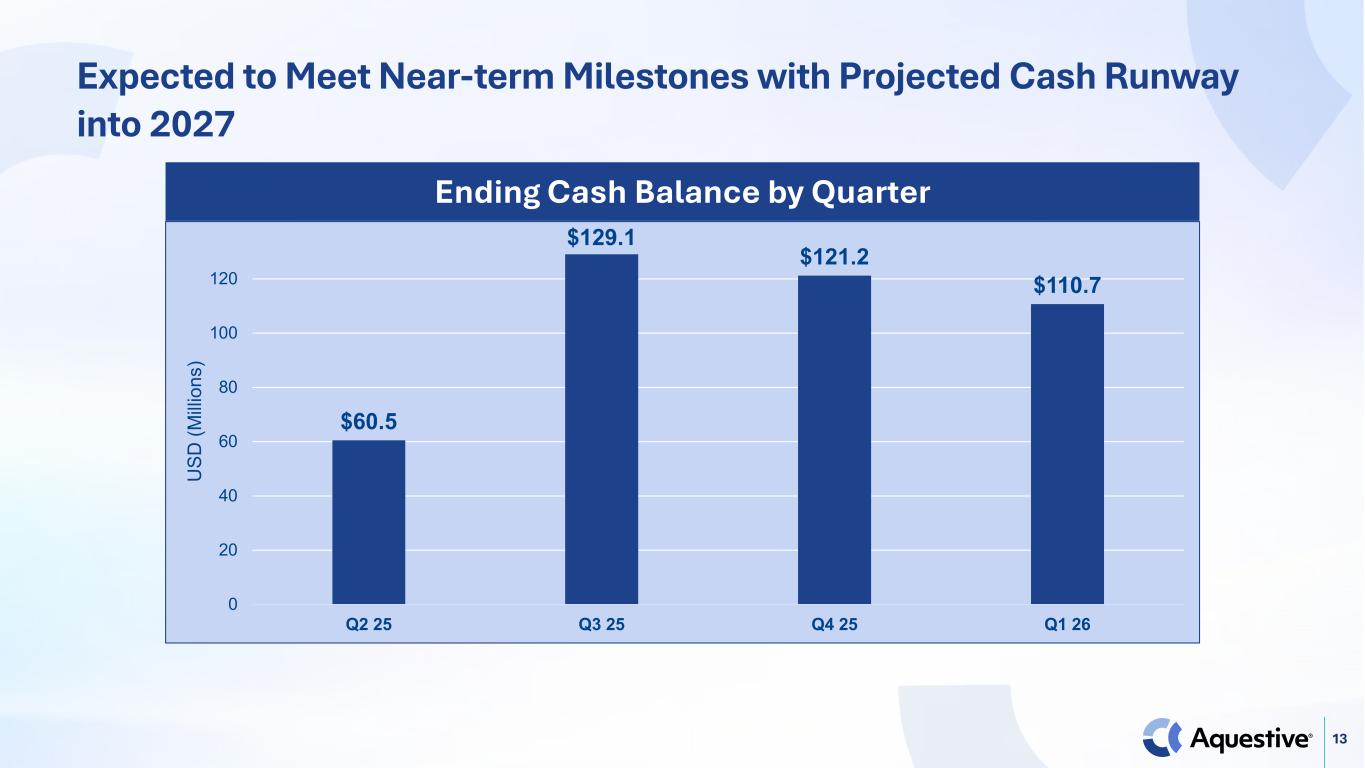
13 Expected to Meet Near-term Milestones with Projected Cash Runway into 2027 $60.5 $129.1 $121.2 $110.7 0 20 40 60 80 100 120 Q2 25 Q3 25 Q4 25 Q1 26 U SD (M illi on s) Ending Cash Balance by Quarter
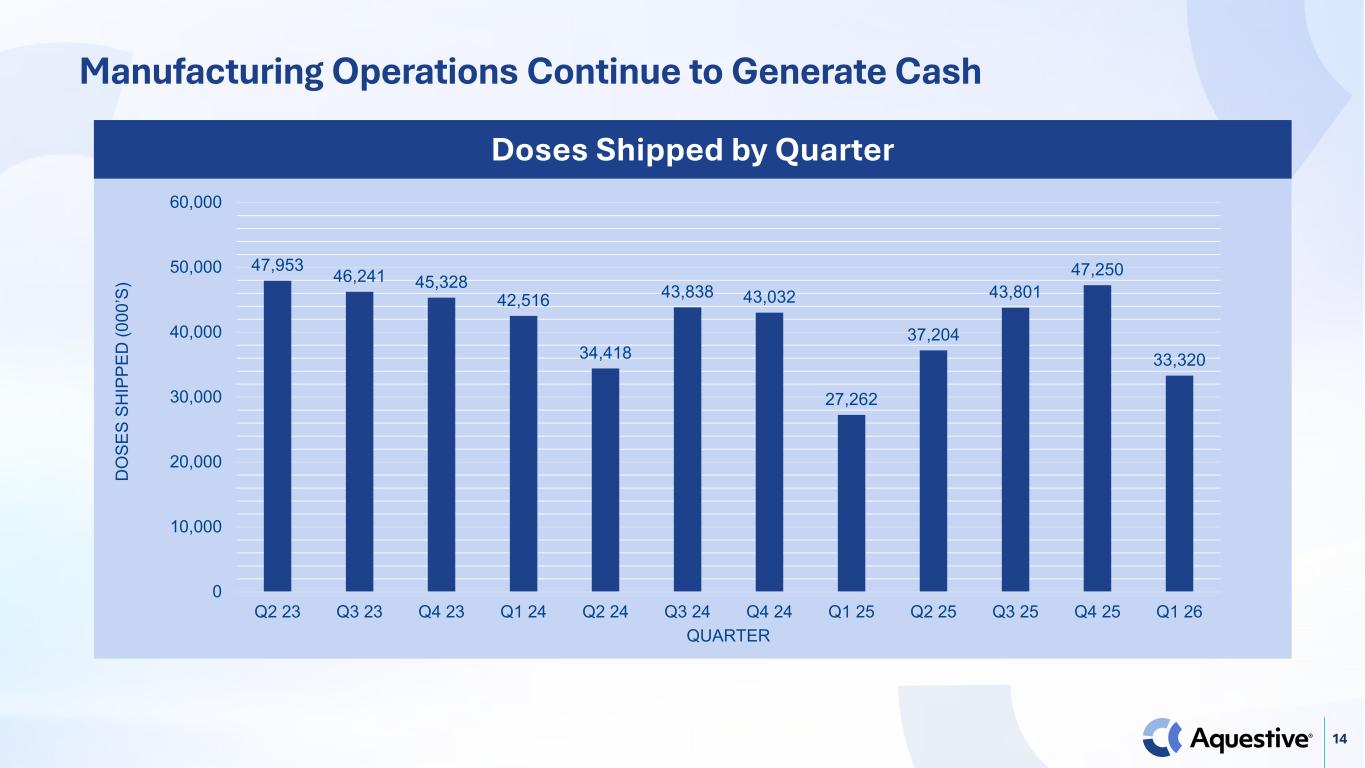
14 47,953 46,241 45,328 42,516 34,418 43,838 43,032 27,262 37,204 43,801 47,250 33,320 0 10,000 20,000 30,000 40,000 50,000 60,000 Q2 23 Q3 23 Q4 23 Q1 24 Q2 24 Q3 24 Q4 24 Q1 25 Q2 25 Q3 25 Q4 25 Q1 26 D O SE S SH IP PE D (0 00 ’S ) QUARTER Doses Shipped by Quarter Manufacturing Operations Continue to Generate Cash

15 2026 Guidance as of May 13, 2026 2026 Outlook • Total revenues of approximately $46-50 million • Non-GAAP adjusted EBITDA loss of approximately $35-30 million

Thank You