Redefining Oncology Treatment with Next-Generation Radiopharmaceuticals

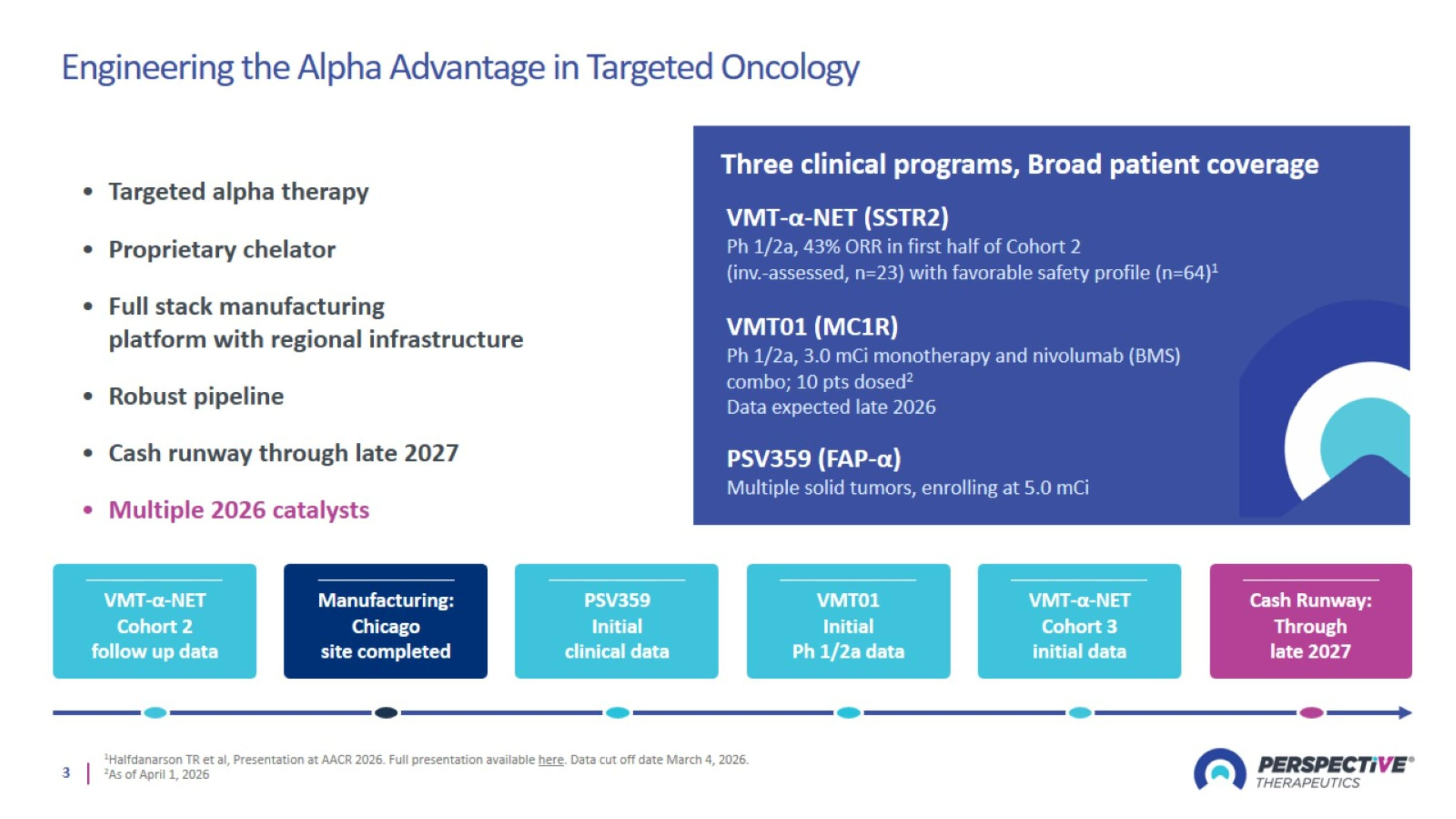
Engineering the Alpha Advantage in Targeted Oncology
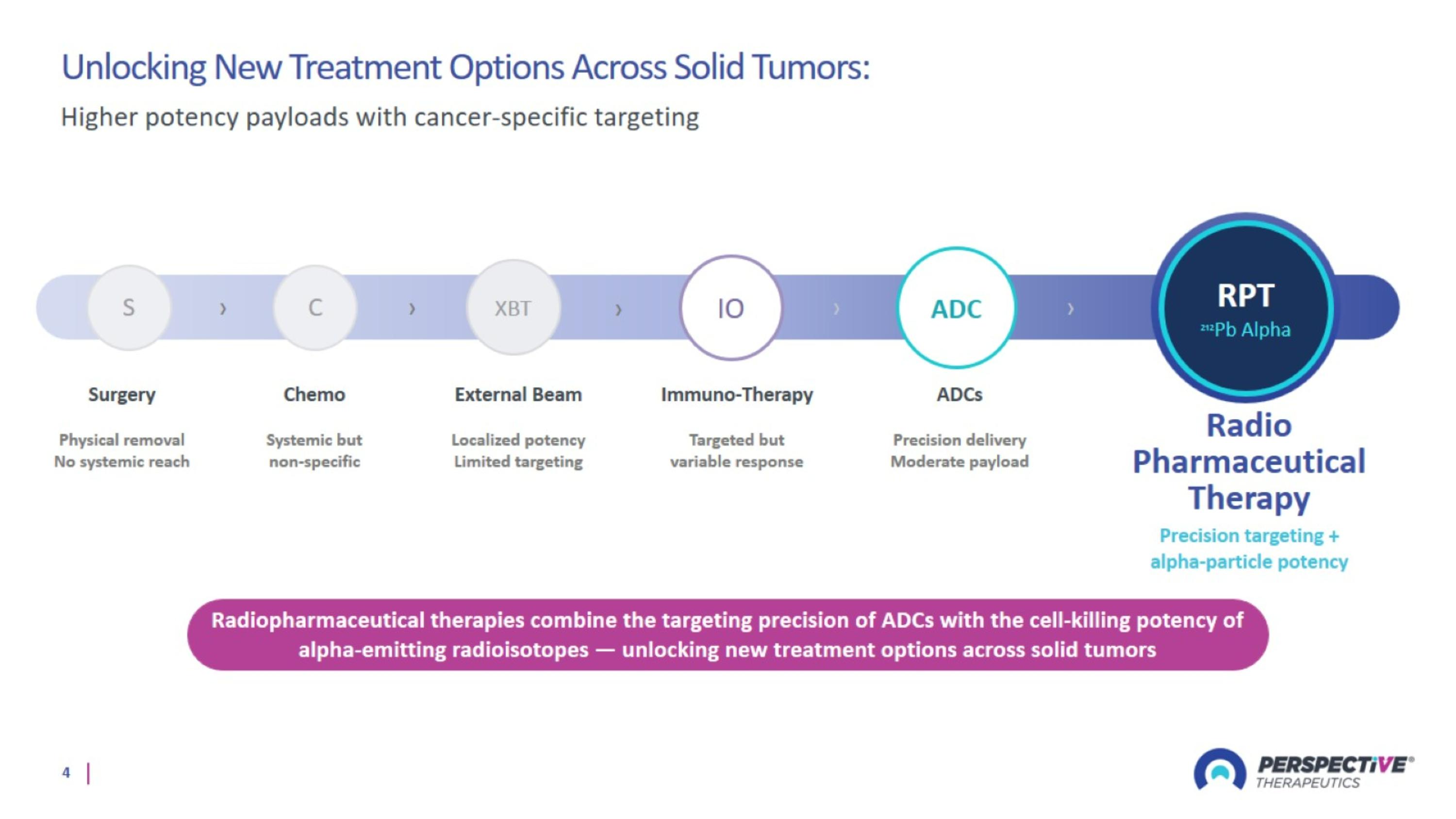
Unlocking New Treatment Options Across Solid Tumors:

Optimizing the Entire System

Rapidly Advancing Best-in-Class Next Generation Radiopharmaceuticals

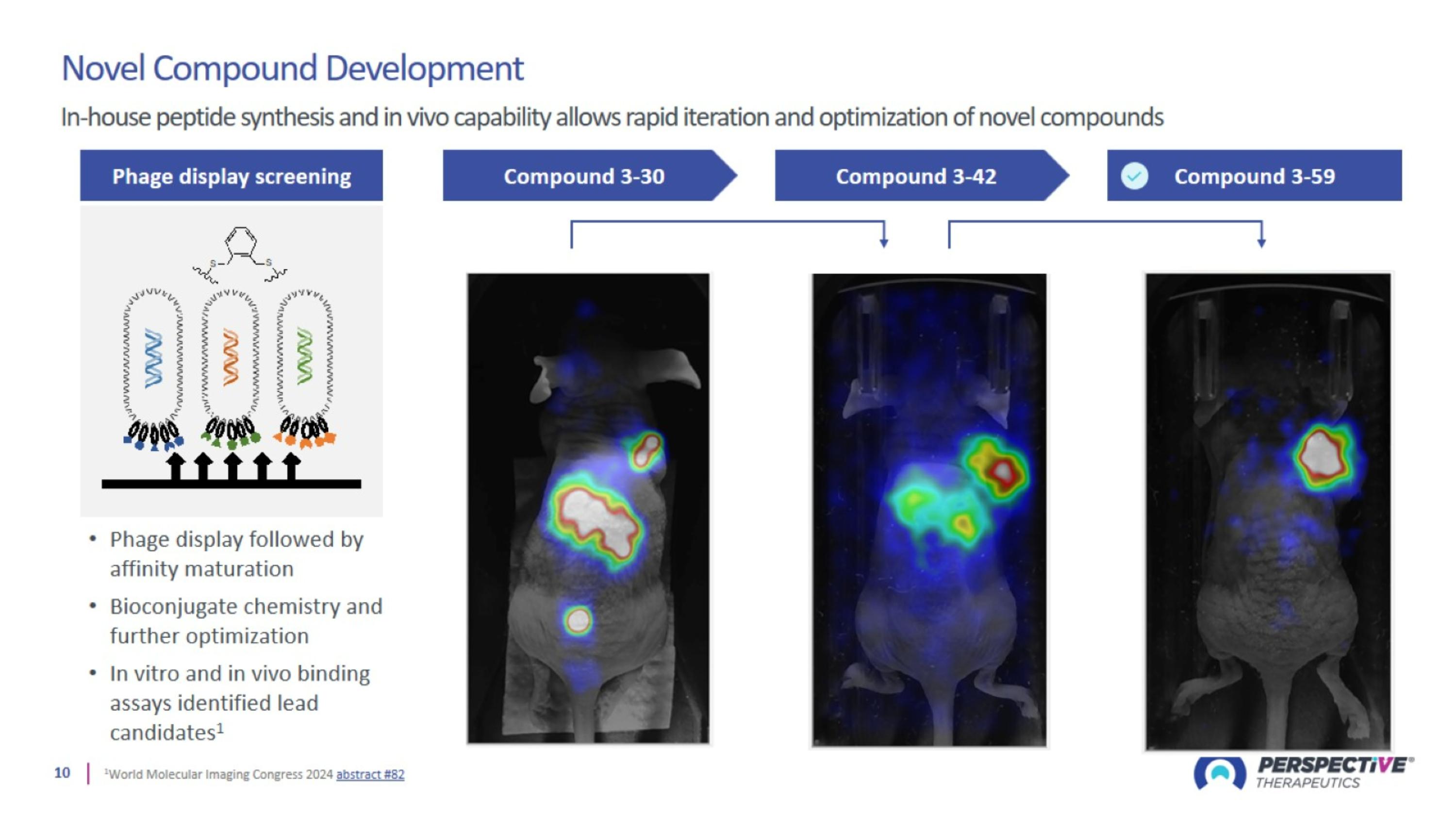
Optimizing Structural Chemistry:

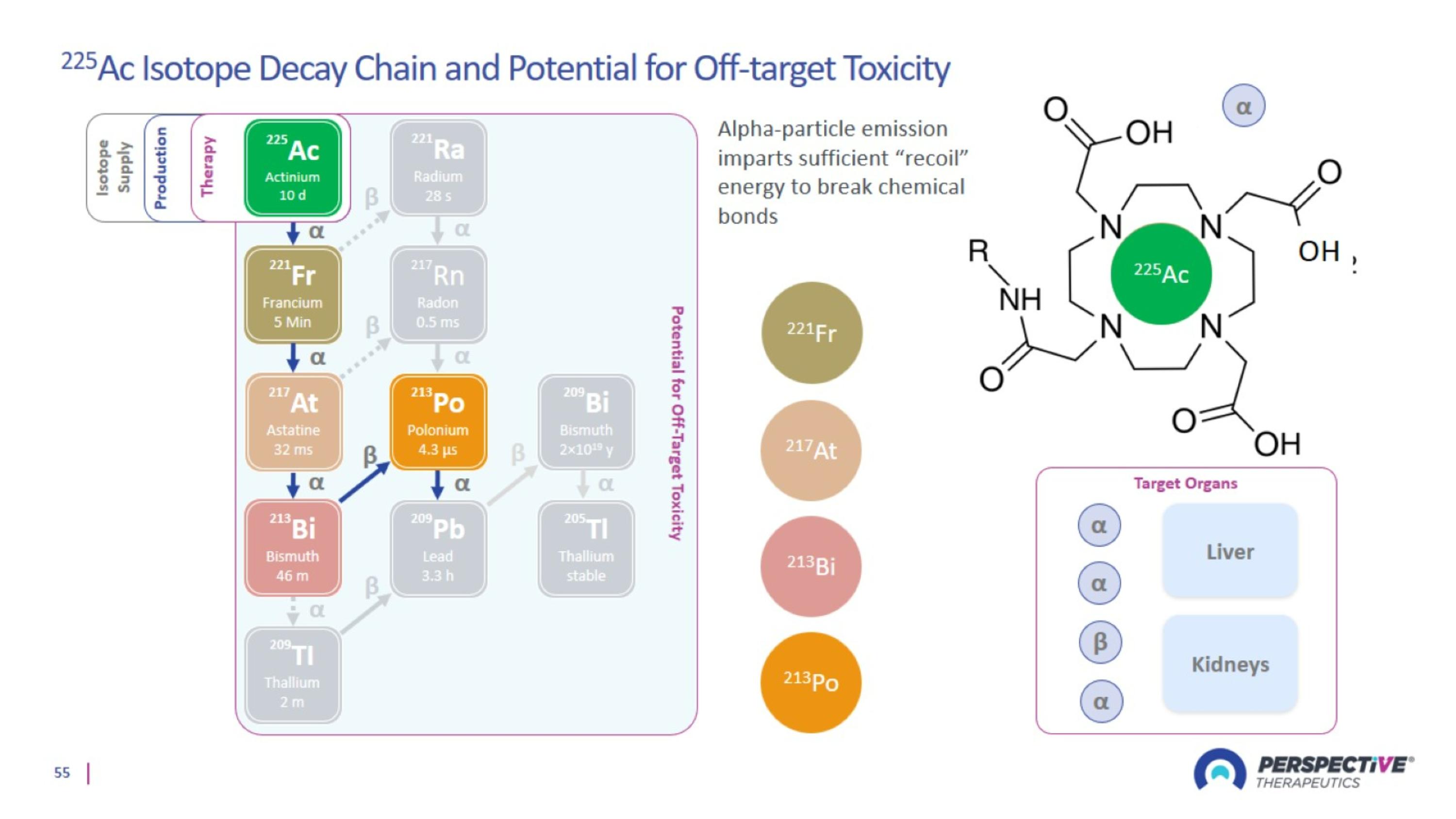
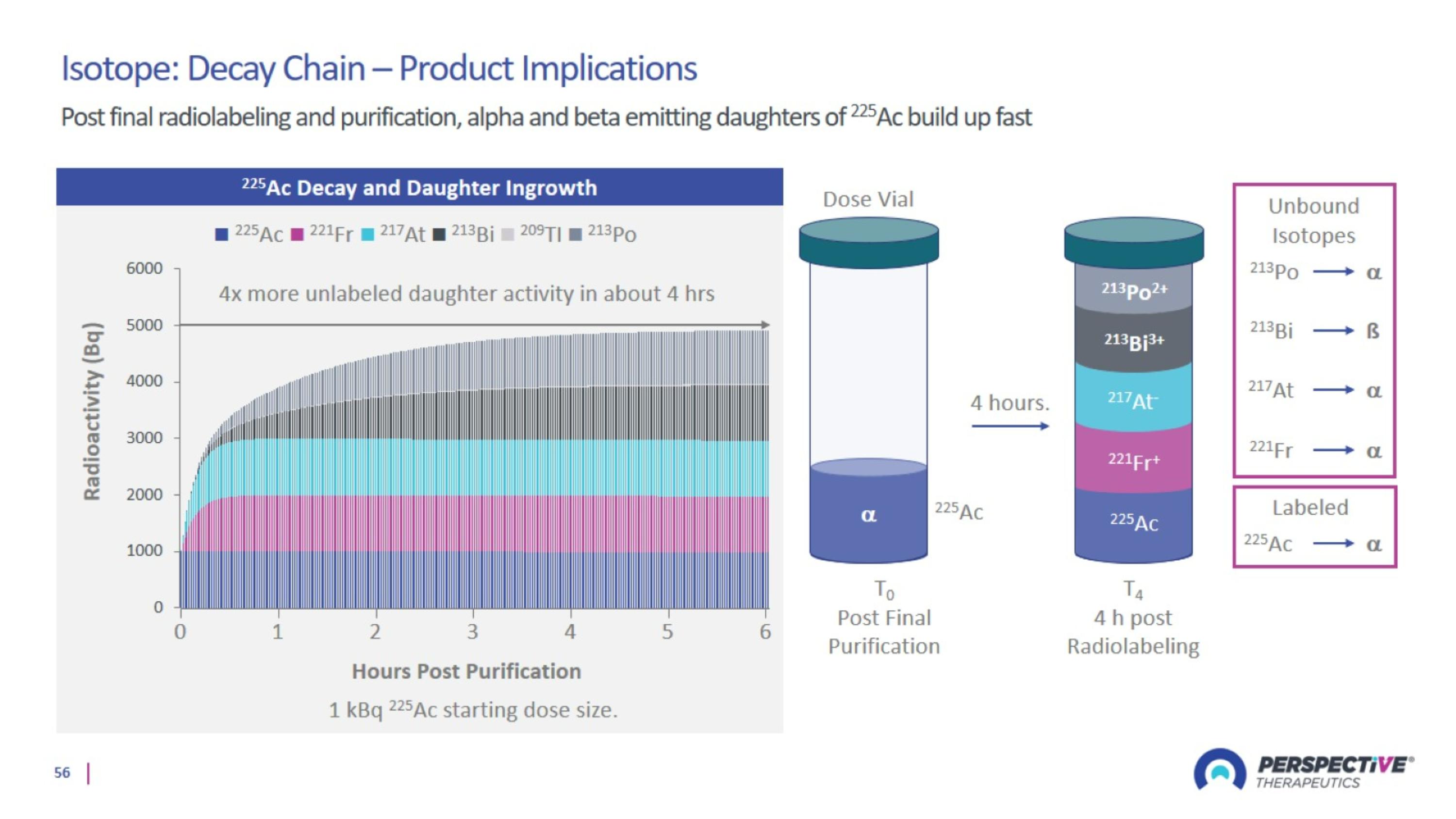
Choosing The Right Isotope
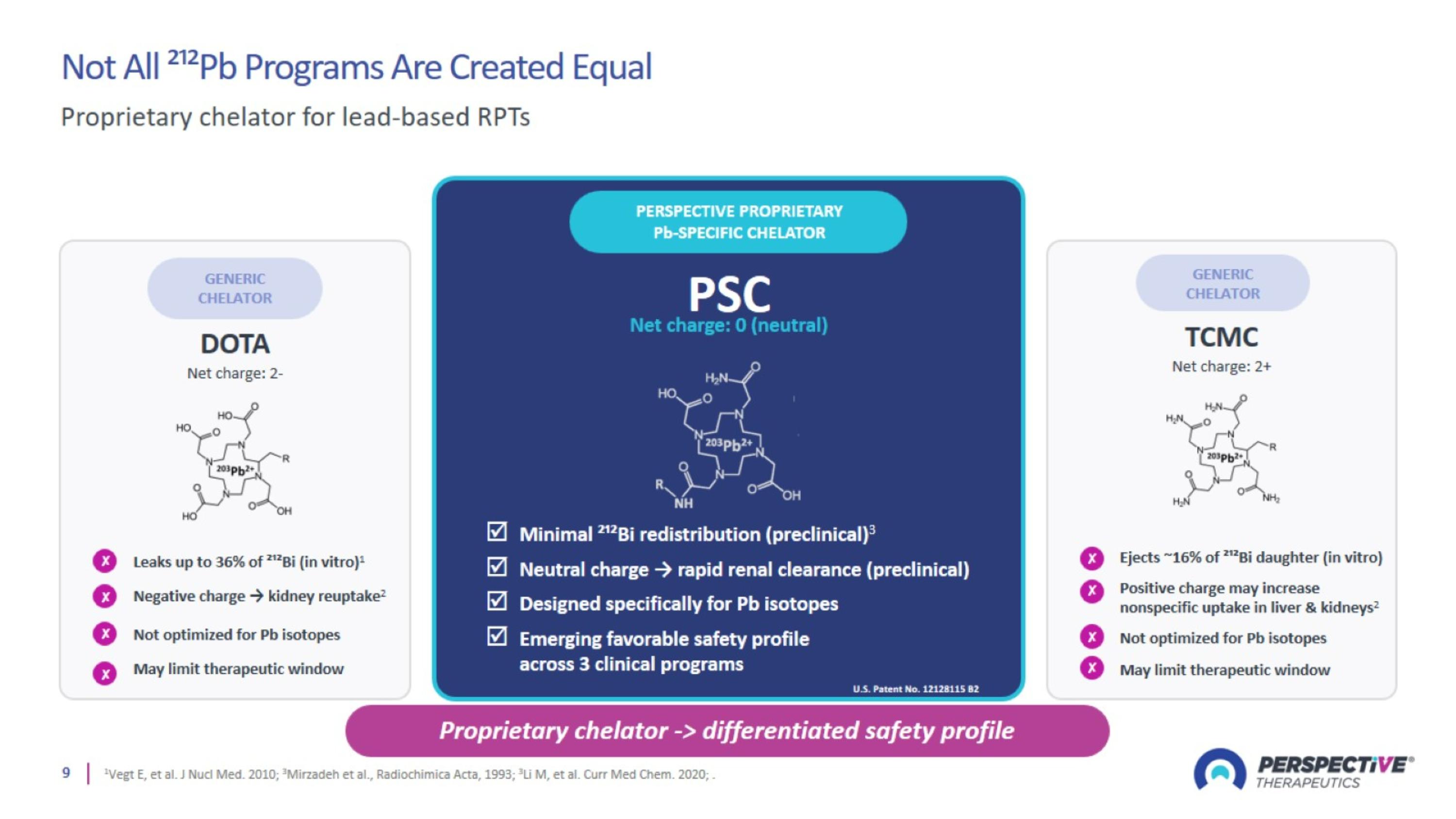
Not All ²¹²Pb Programs Are Created Equal
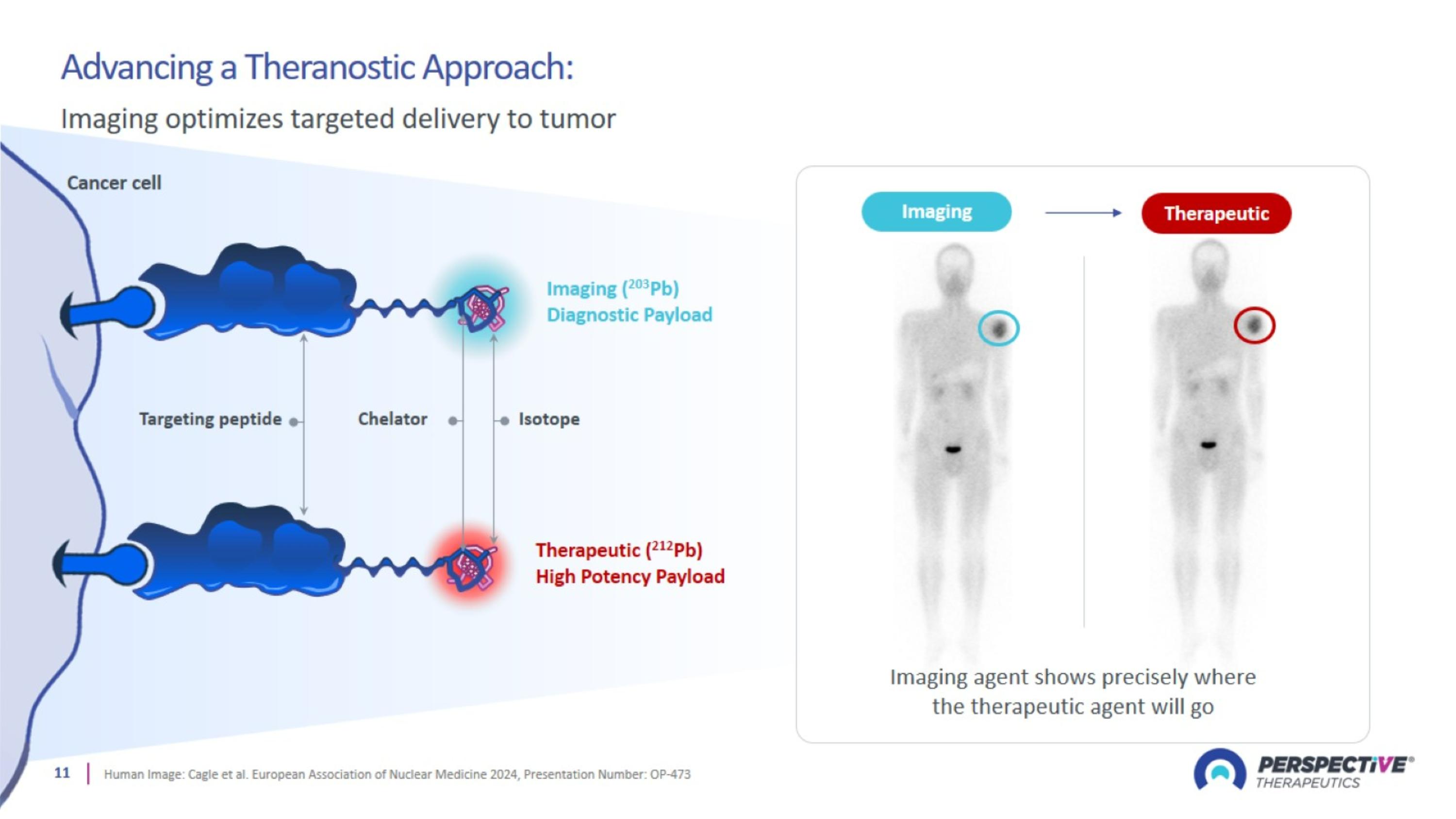
Advancing a Theranostic Approach:
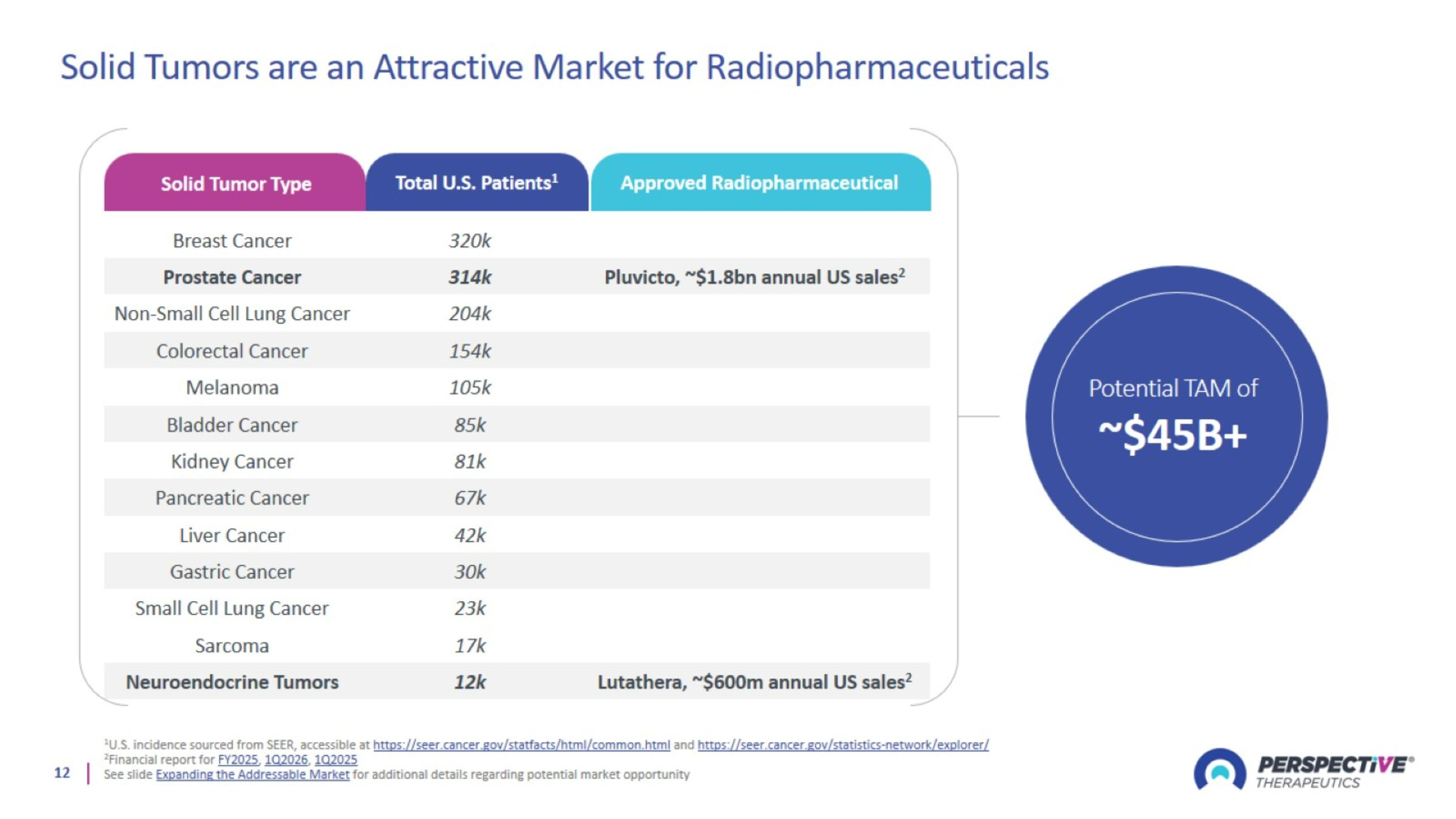
Solid Tumors are an Attractive Market for Radiopharmaceuticals
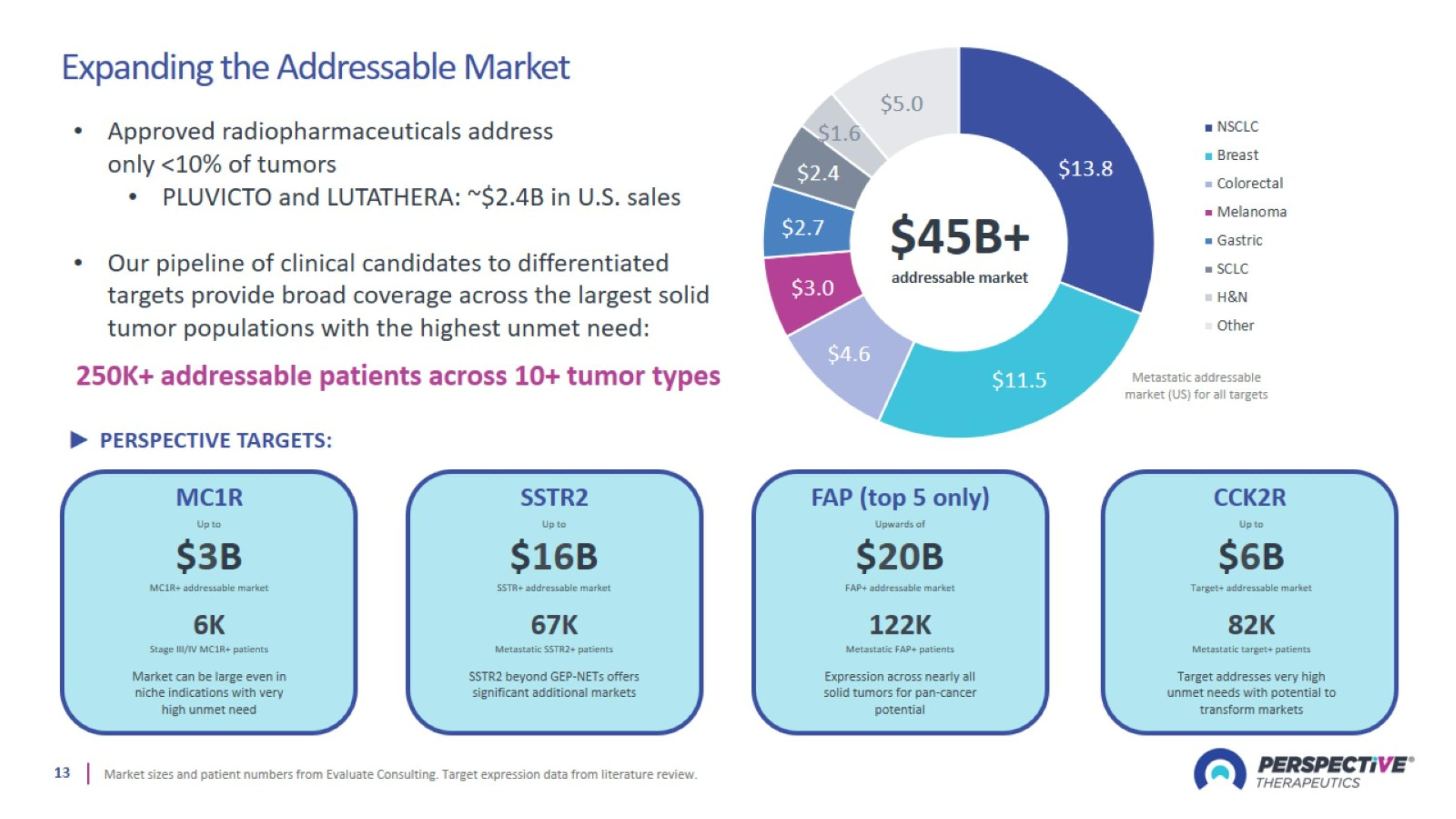
Expanding the Addressable Market
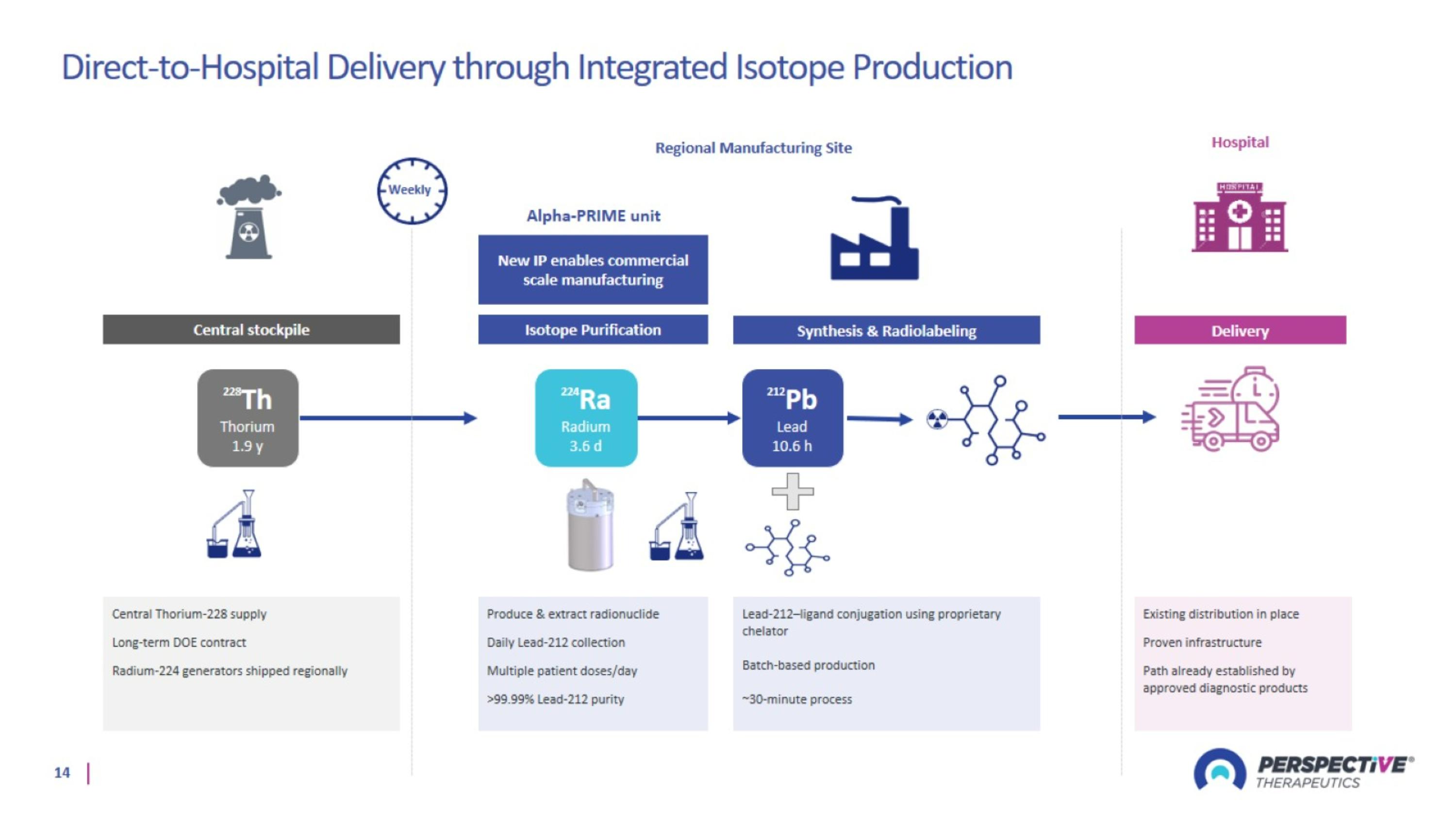
Direct-to-Hospital Delivery through Integrated Isotope Production
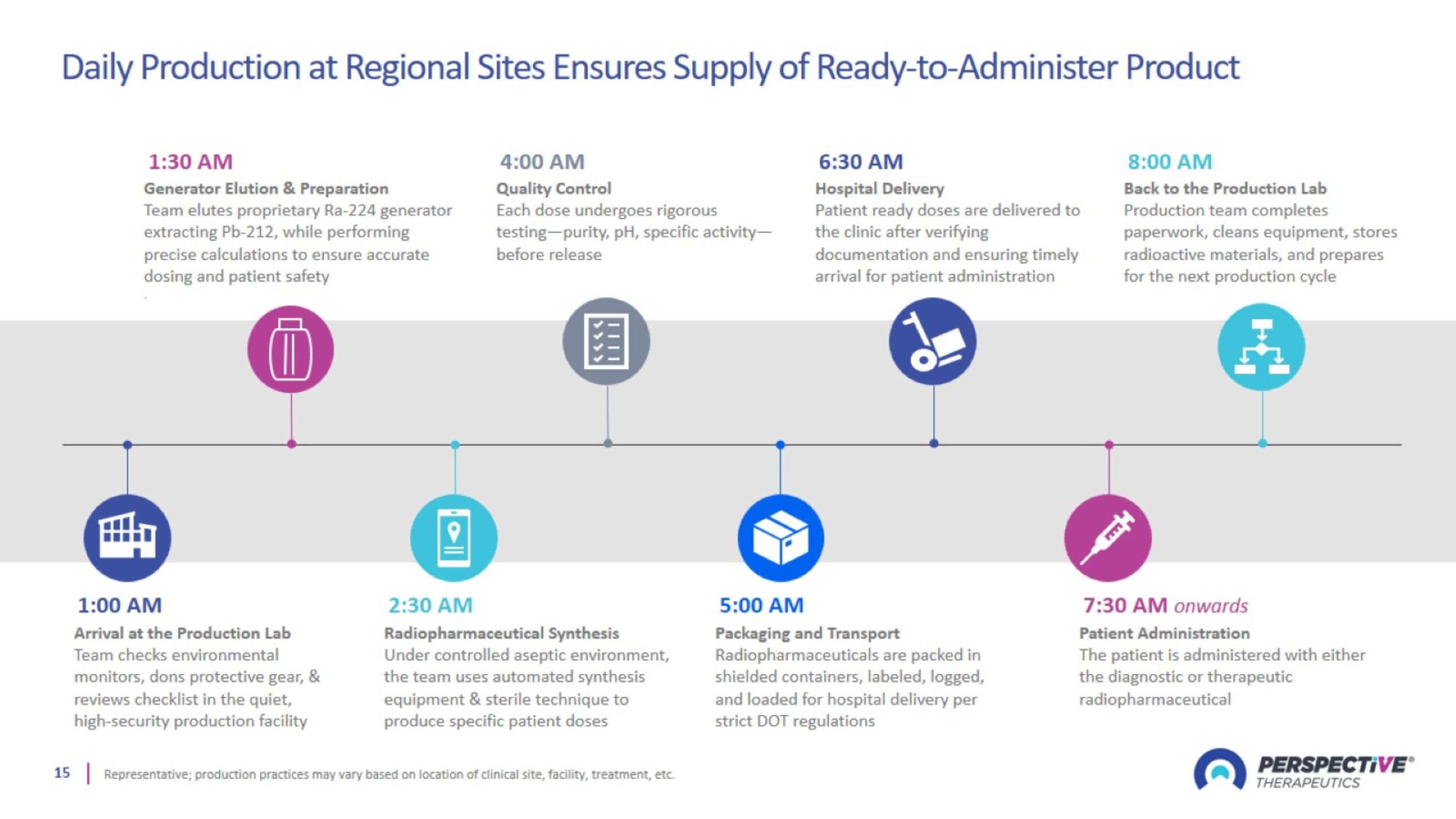
Daily Production at Regional Sites Ensures Supply of Ready-to-Administer Product
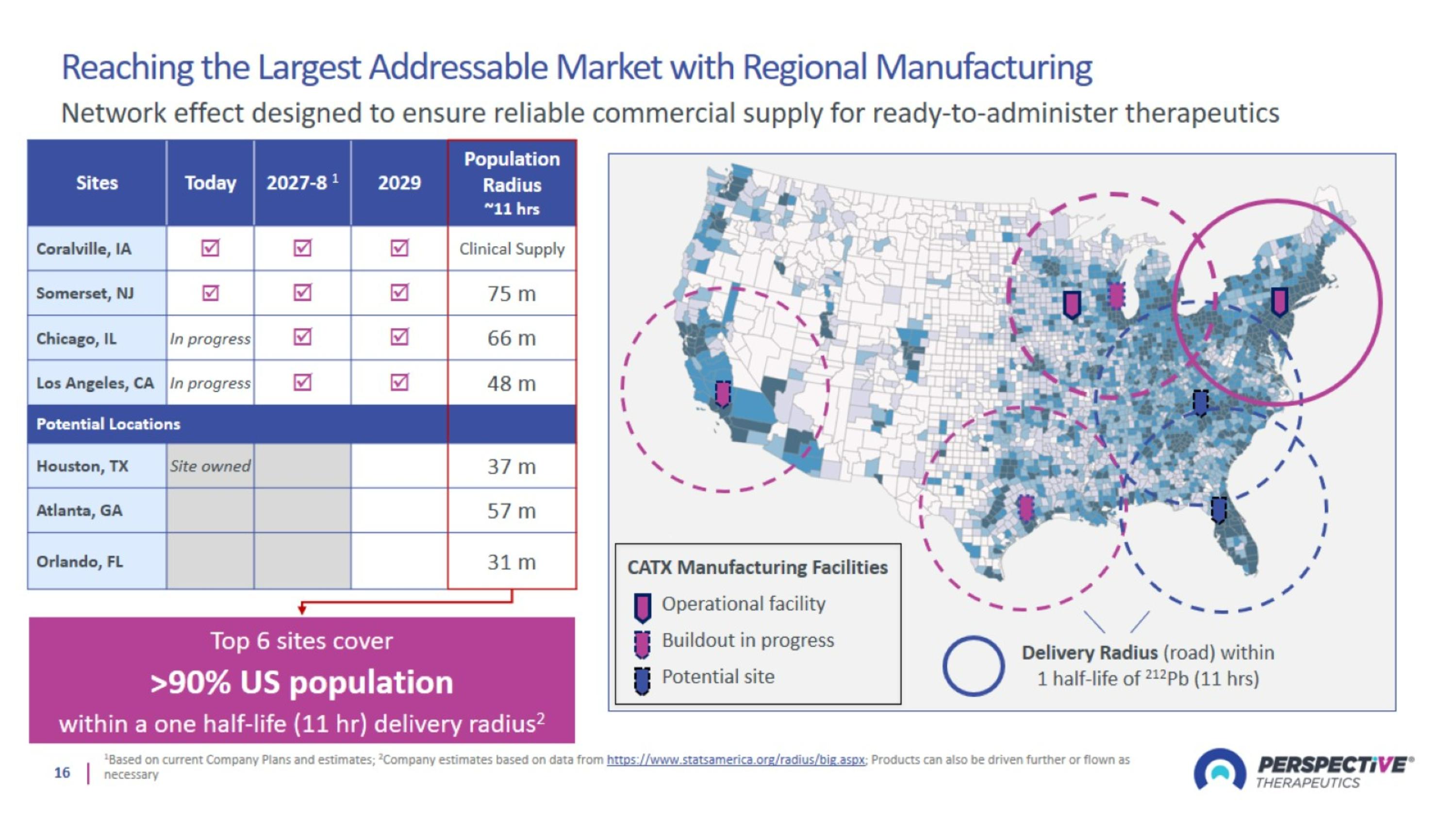
Reaching the Largest Addressable Market with Regional Manufacturing
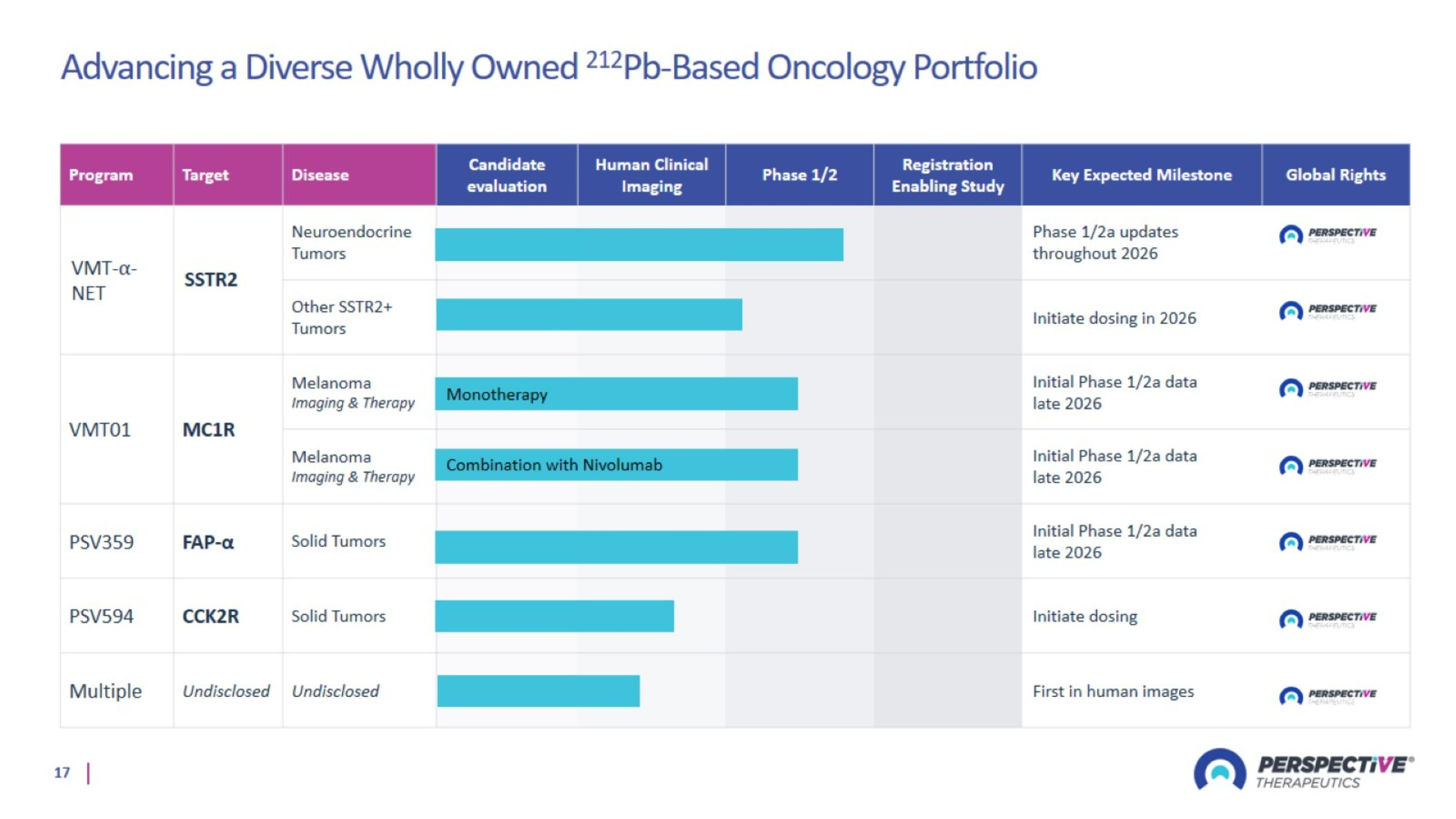
Advancing a Diverse Wholly Owned 212Pb-Based Oncology Portfolio
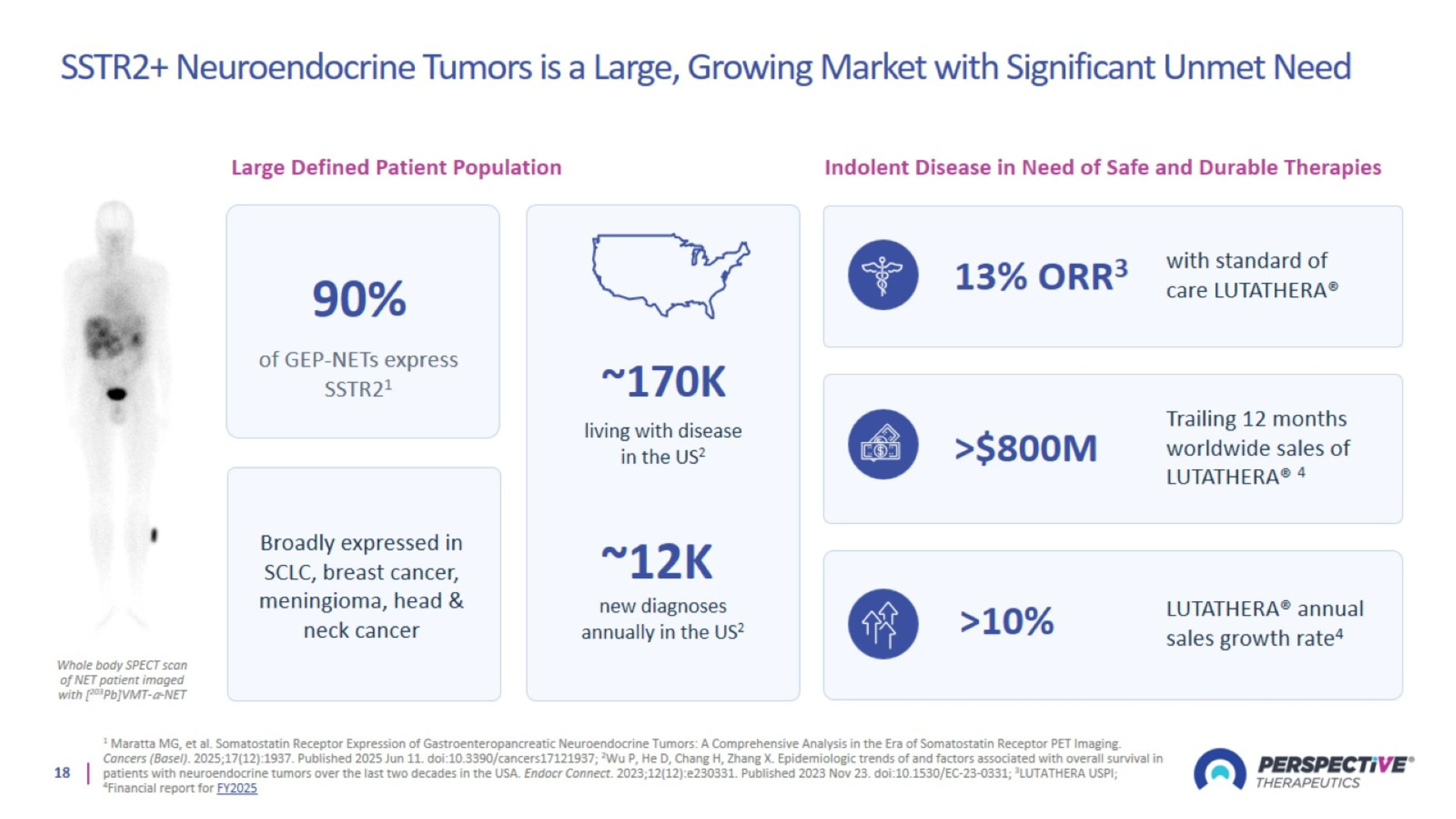
SSTR2+ Neuroendocrine Tumors is a Large, Growing Market with Significant Unmet Need

VMT-⍺-NET: Potential First-in-Class 212Pb-Radioligand Therapy Targeting SSTR2
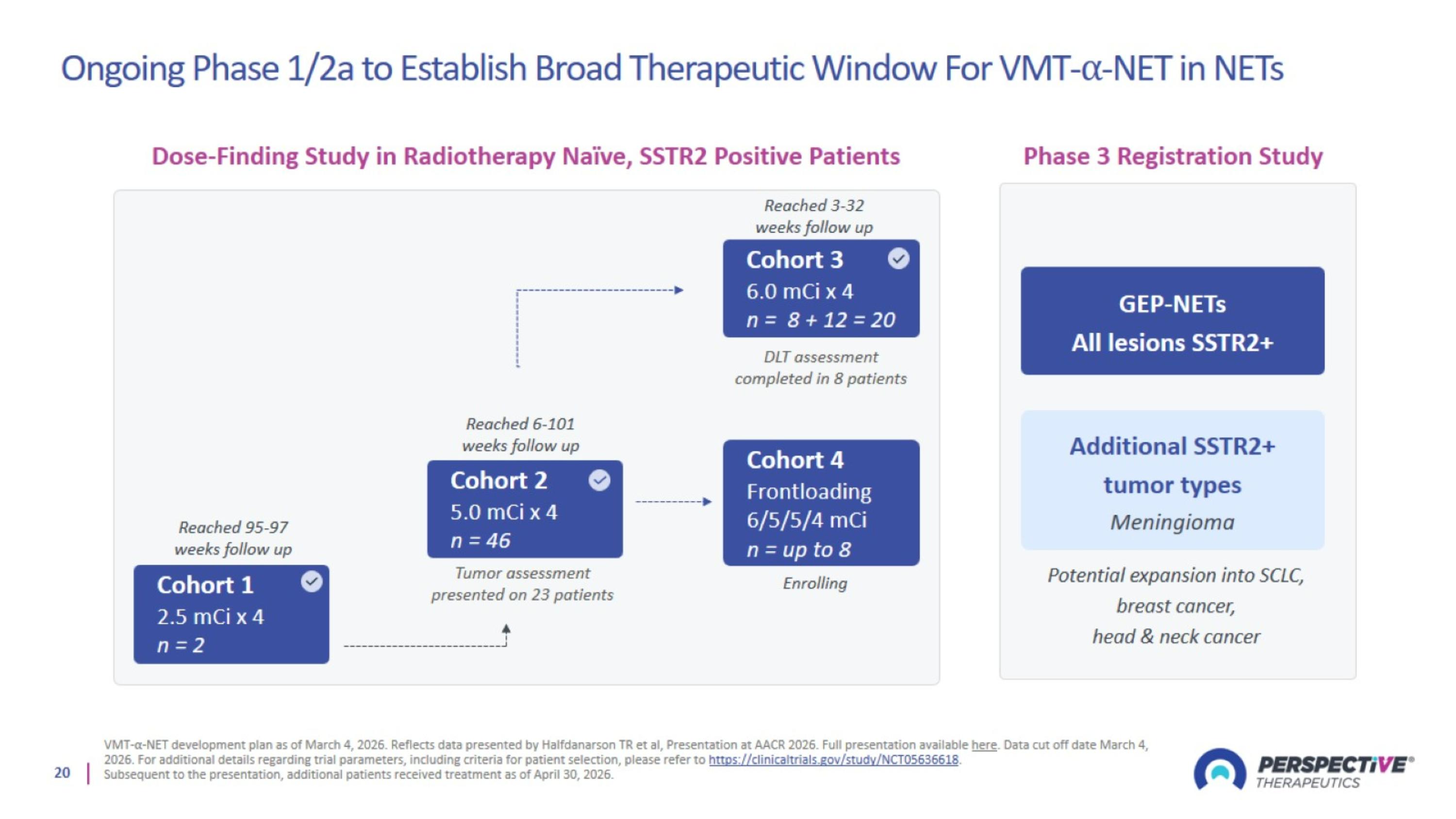
Ongoing Phase 1/2a to Establish Broad Therapeutic Window For VMT-⍺-NET in NETs
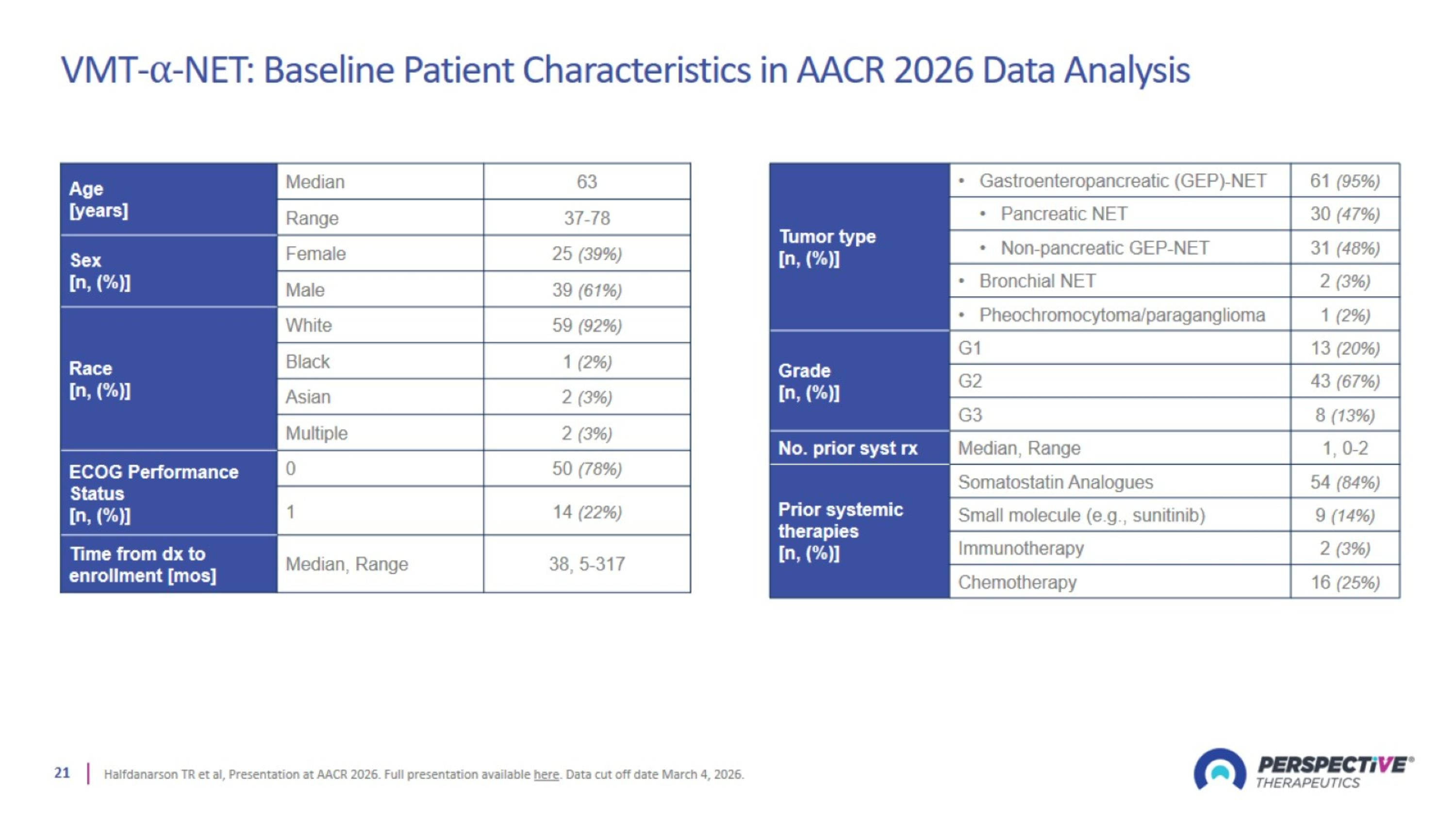
VMT-⍺-NET: Baseline Patient Characteristics in AACR 2026 Data Analysis
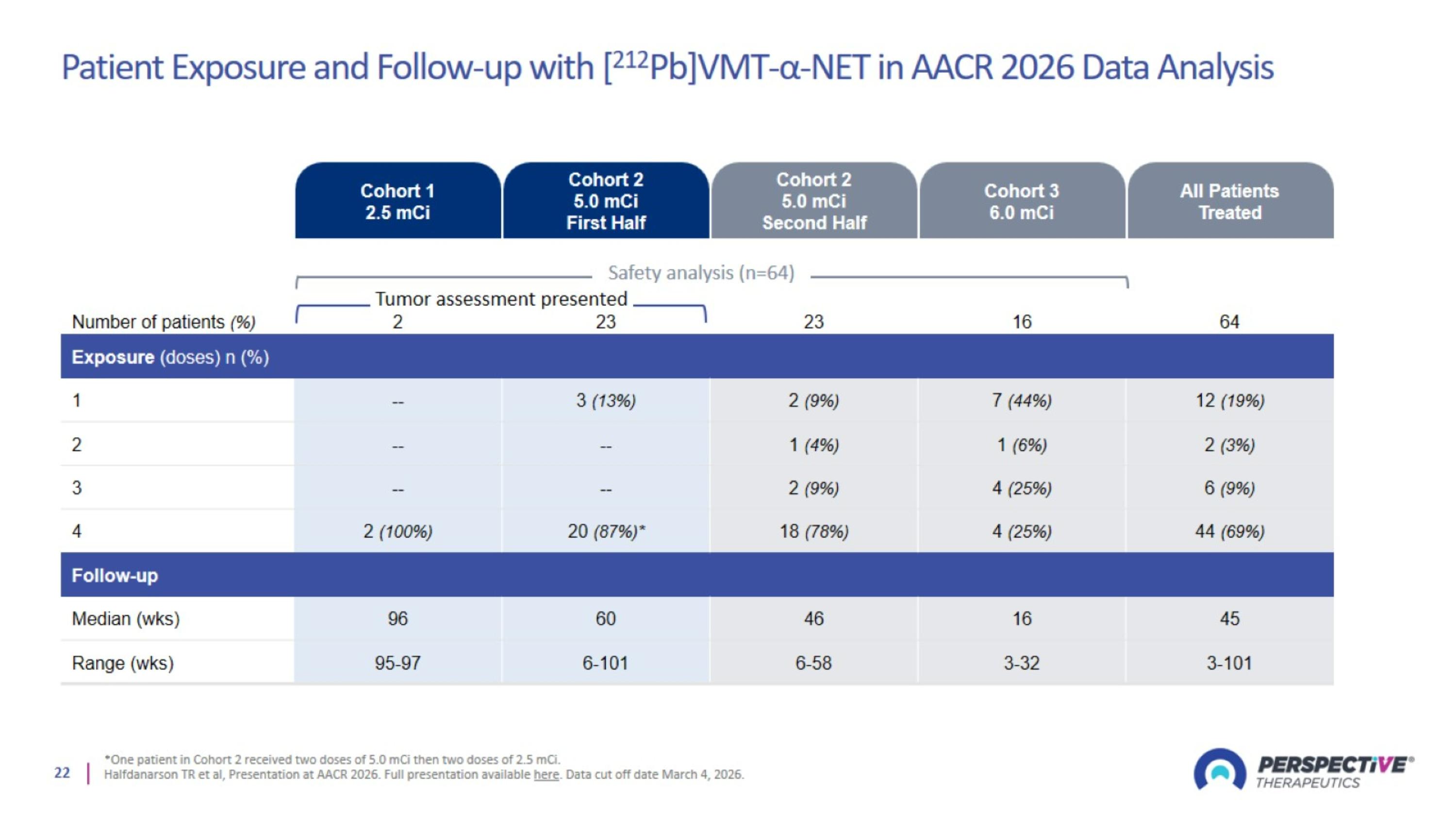
Patient Exposure and Follow-up with [212Pb]VMT-α-NET in AACR 2026 Data Analysis
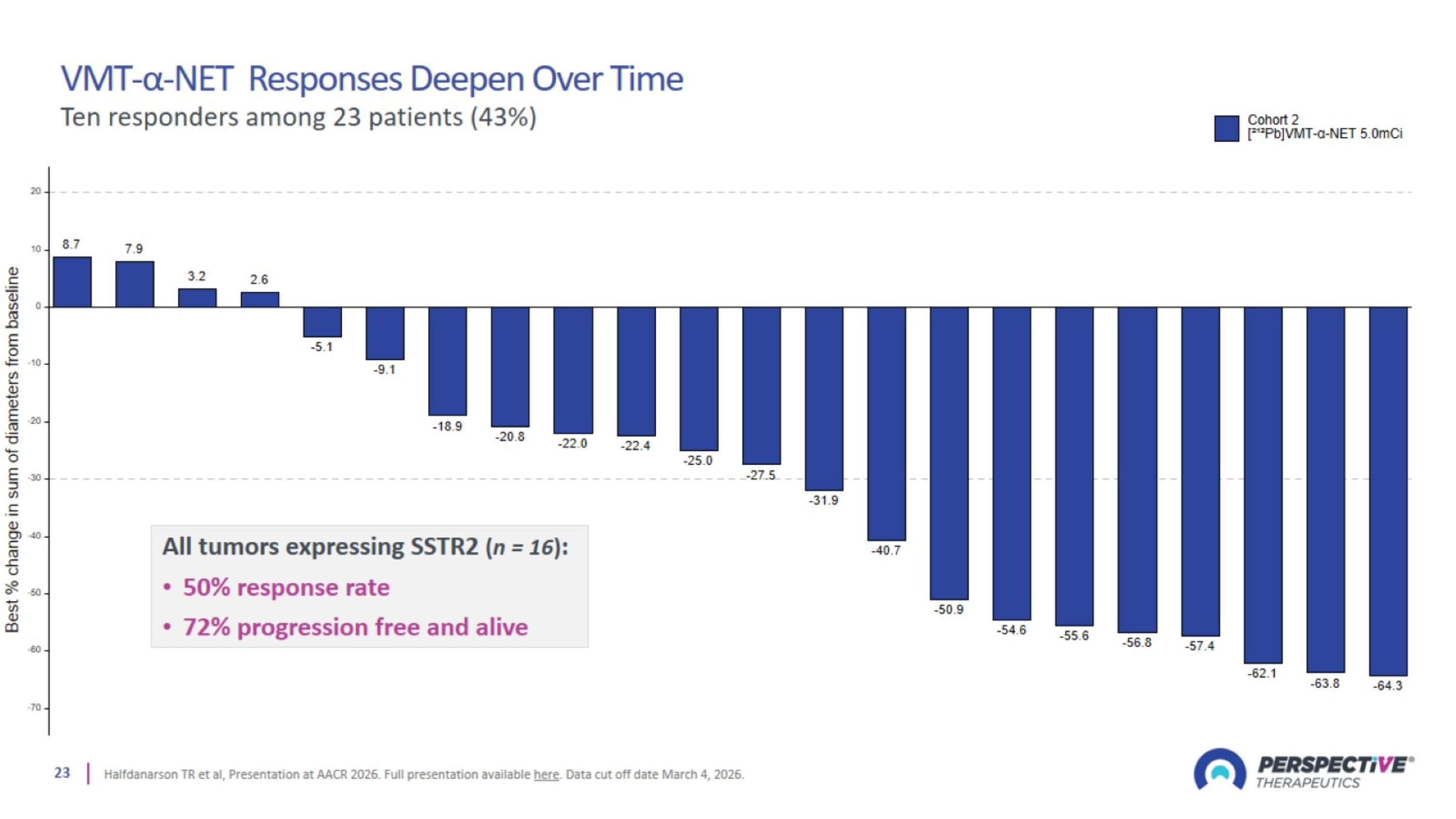
VMT-α-NET Responses Deepen Over Time
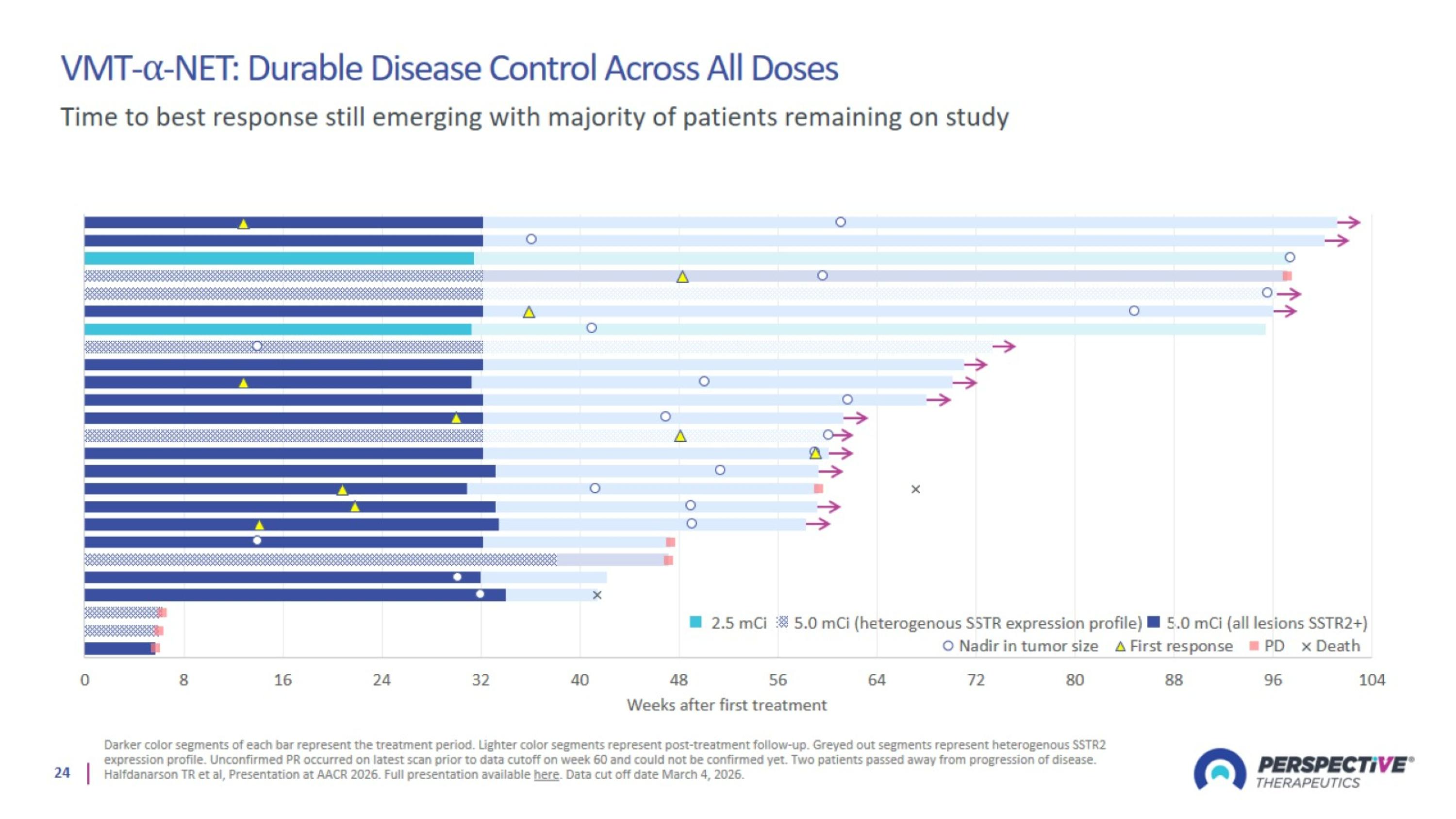
VMT-⍺-NET: Durable Disease Control Across All Doses
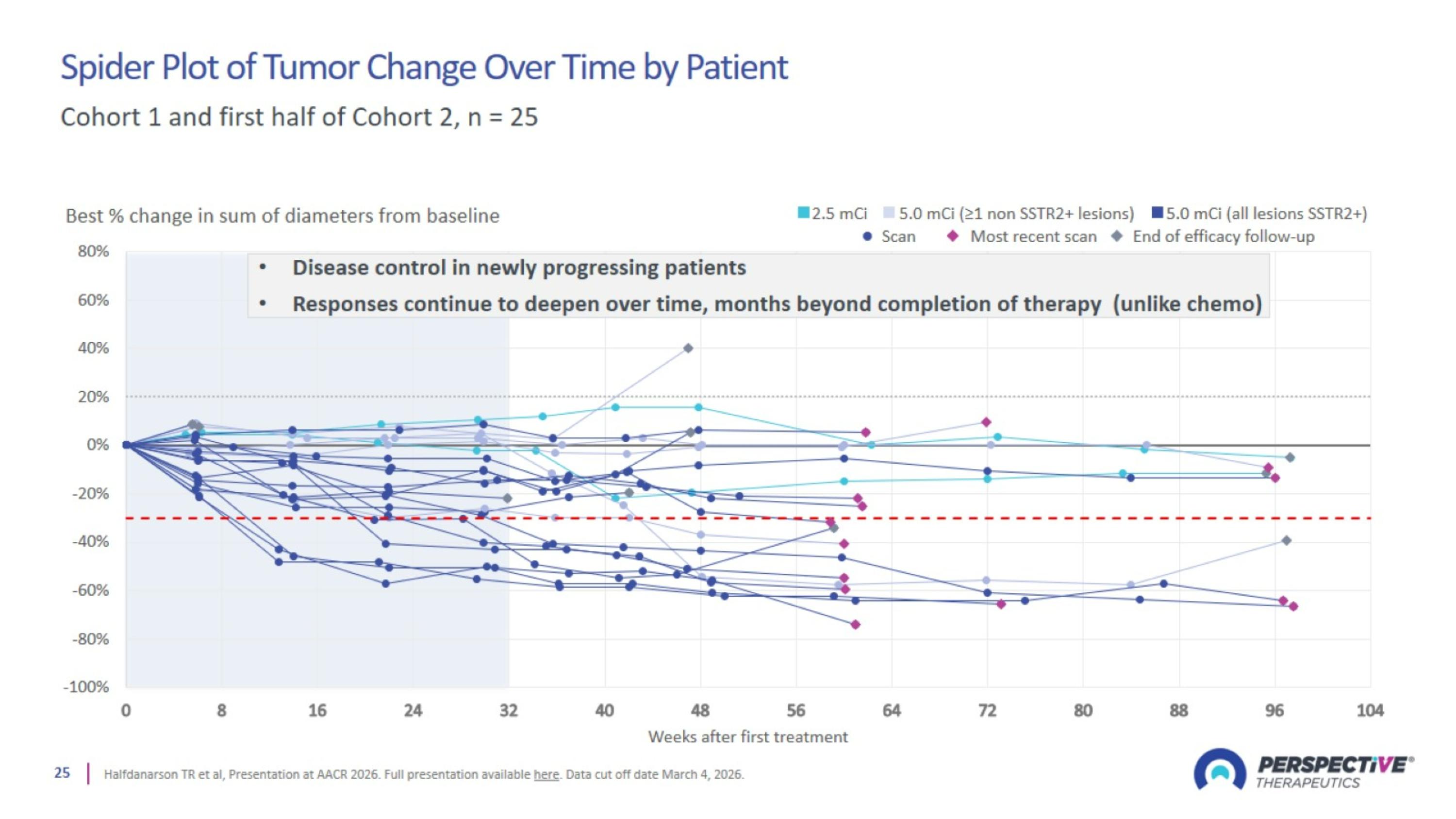
Spider Plot of Tumor Change Over Time by Patient
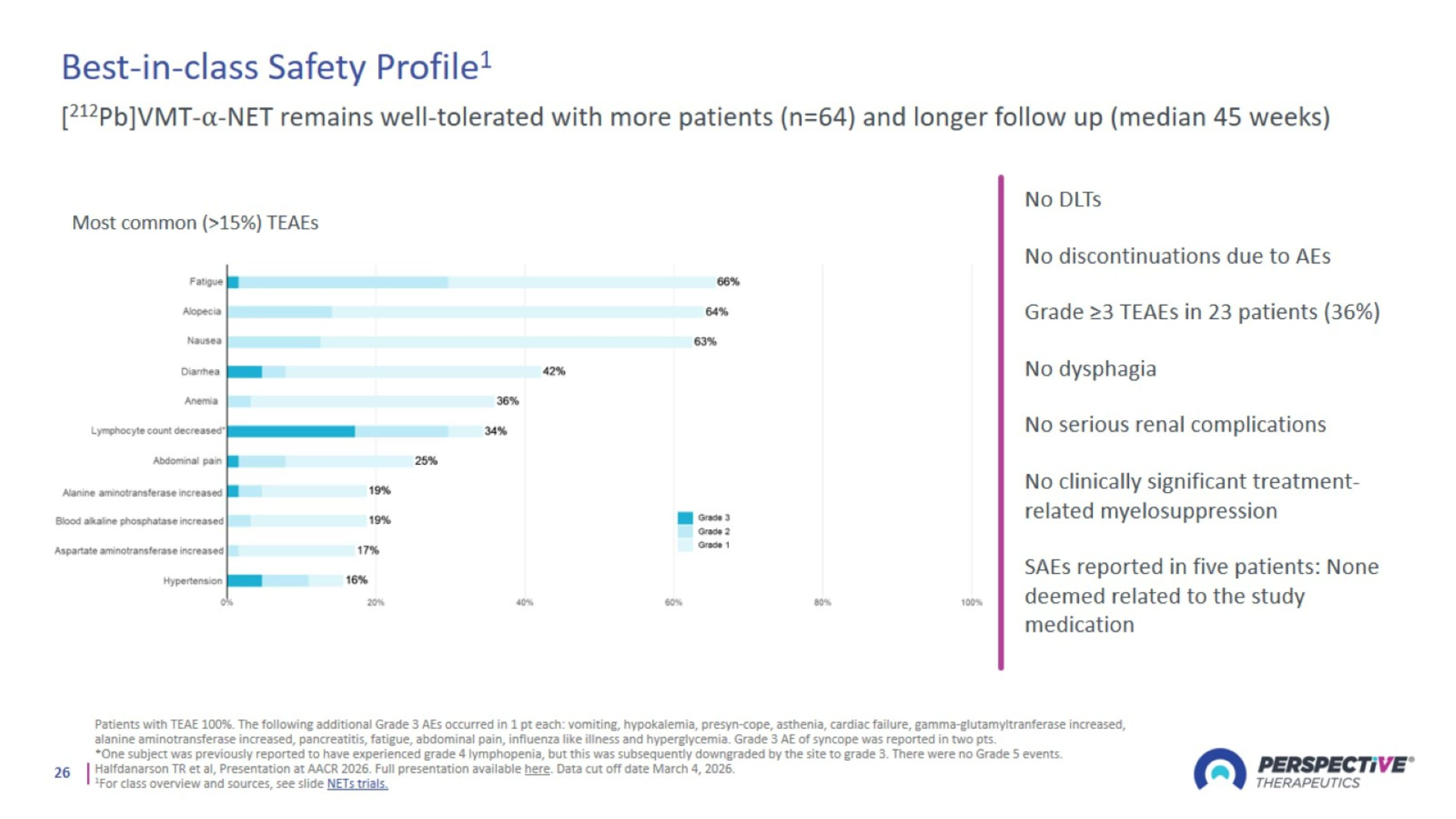
Best-in-class Safety Profile1
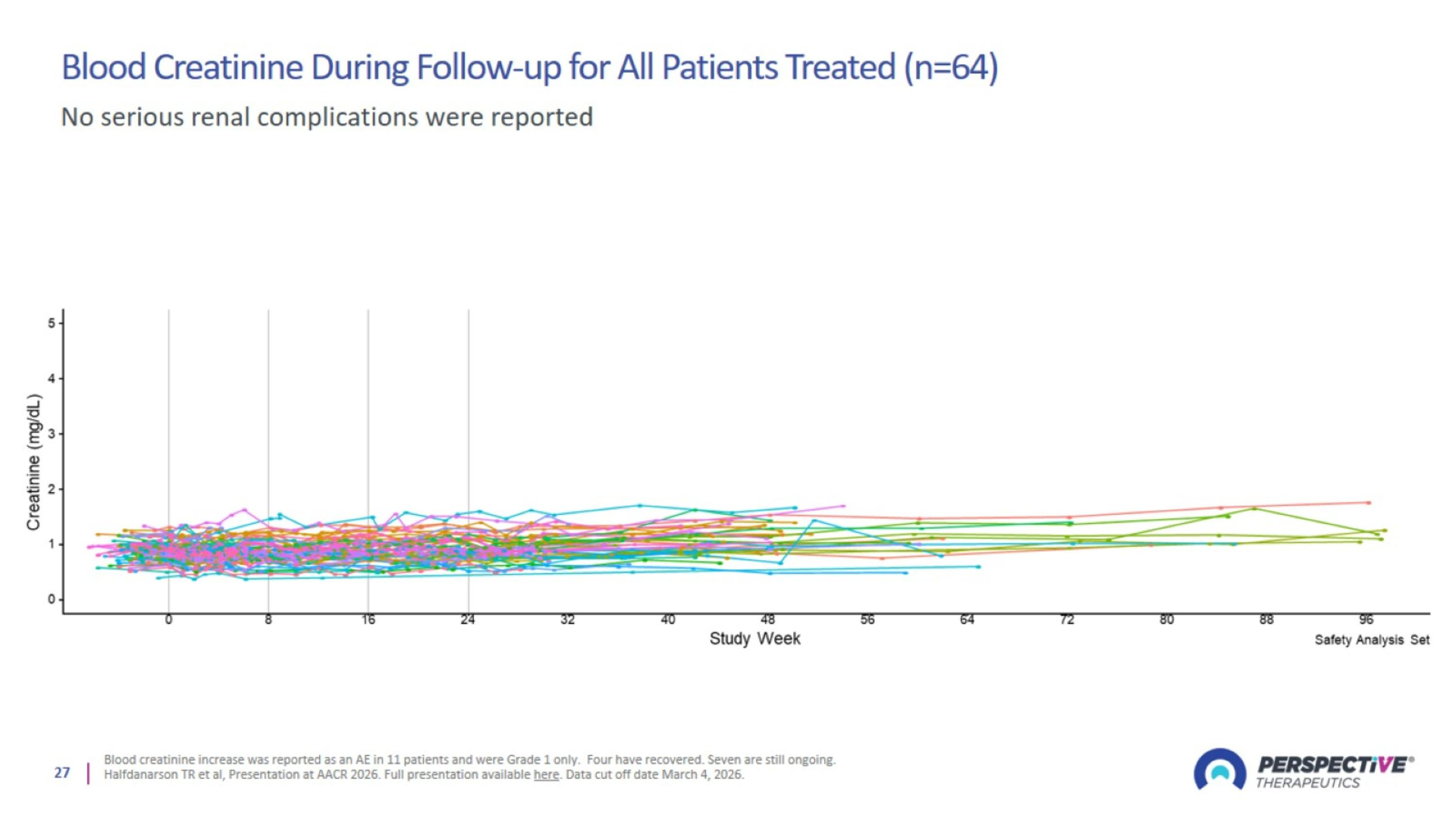
Blood Creatinine During Follow-up for All Patients Treated (n=64)
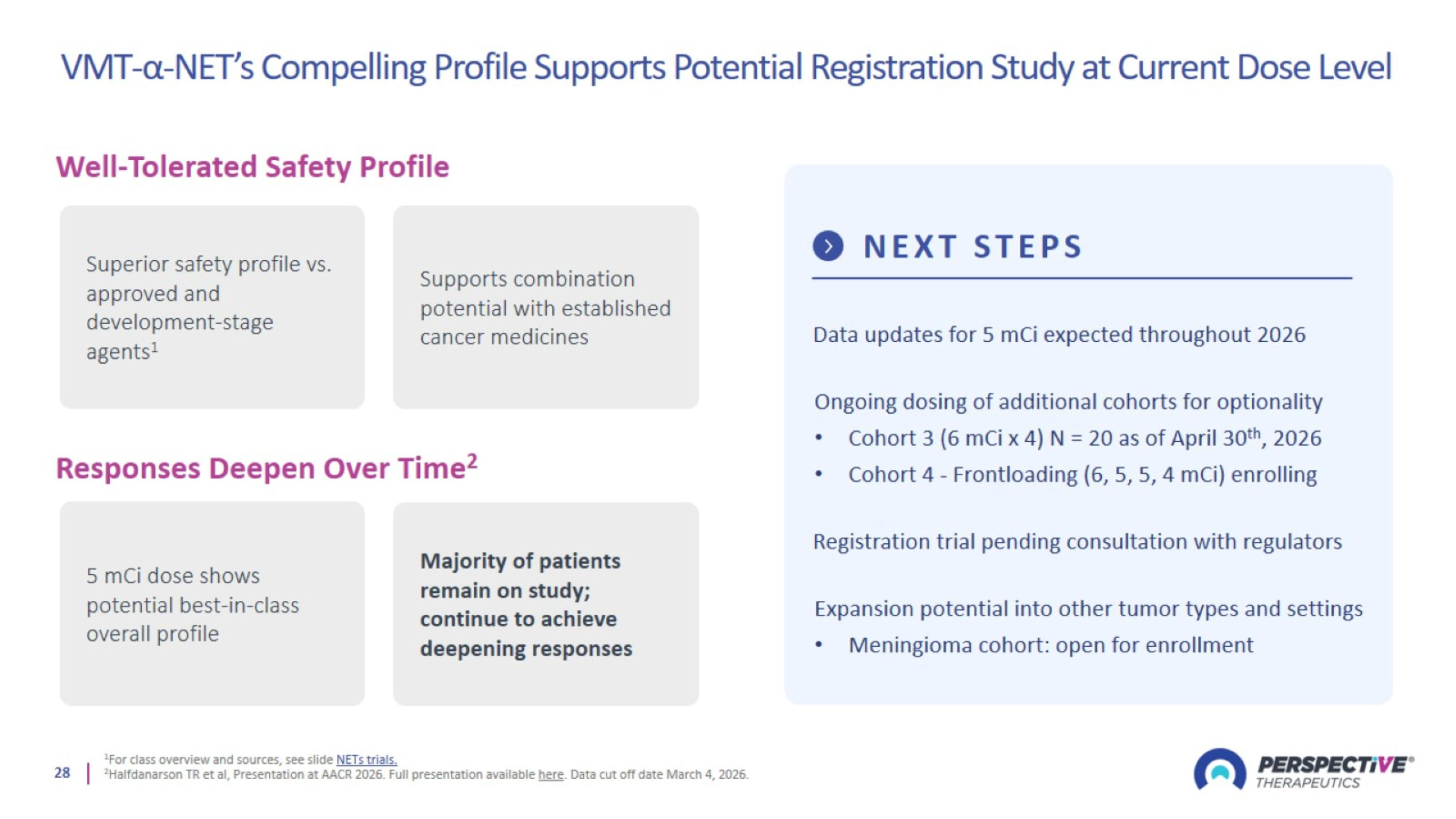
VMT-α-NET’s Compelling Profile Supports Potential Registration Study at Current Dose Level
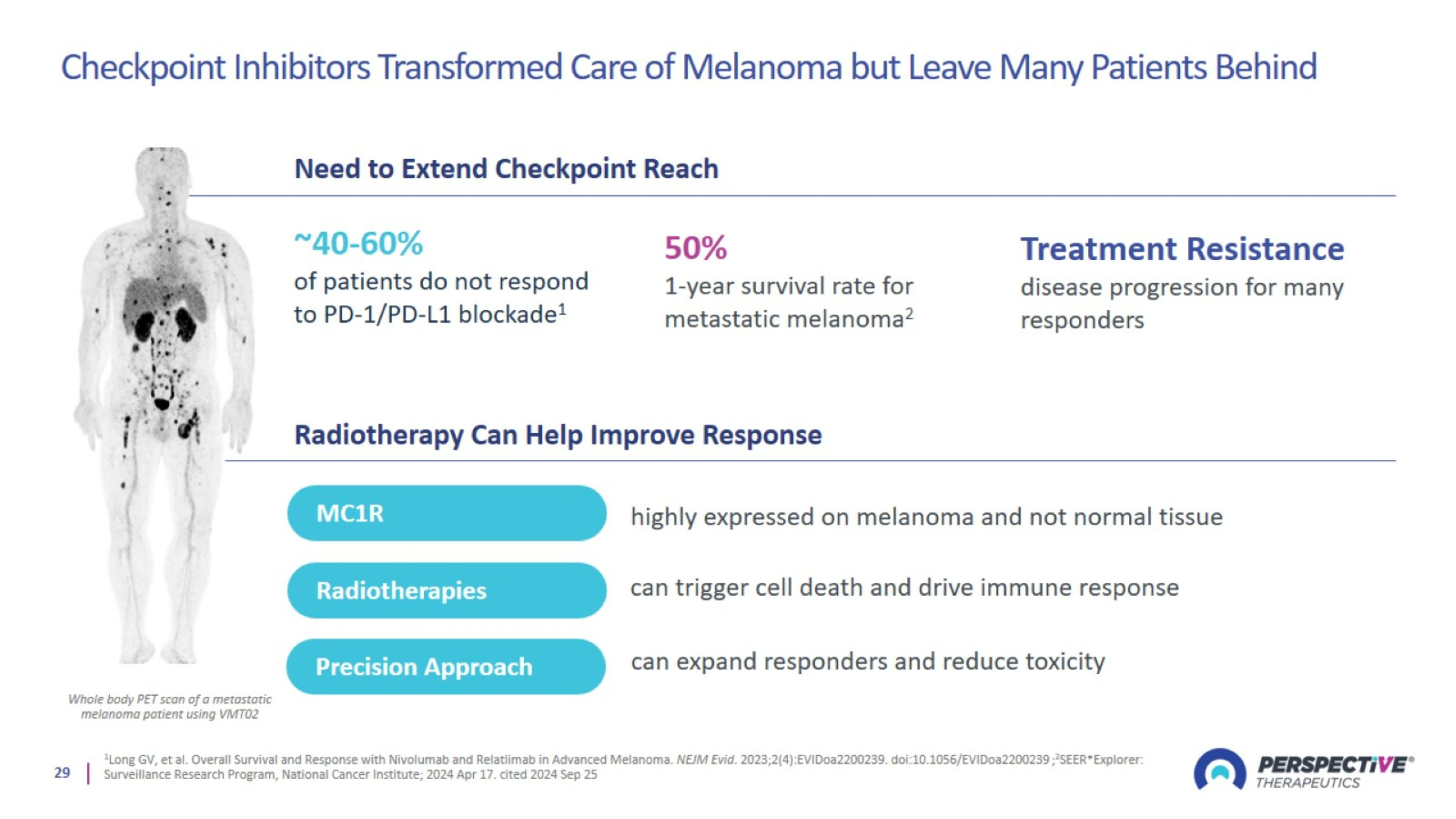
Checkpoint Inhibitors Transformed Care of Melanoma but Leave Many Patients Behind

VMT01: Potential First-in-Class 212Pb Therapy Targeting MC1R for Melanoma
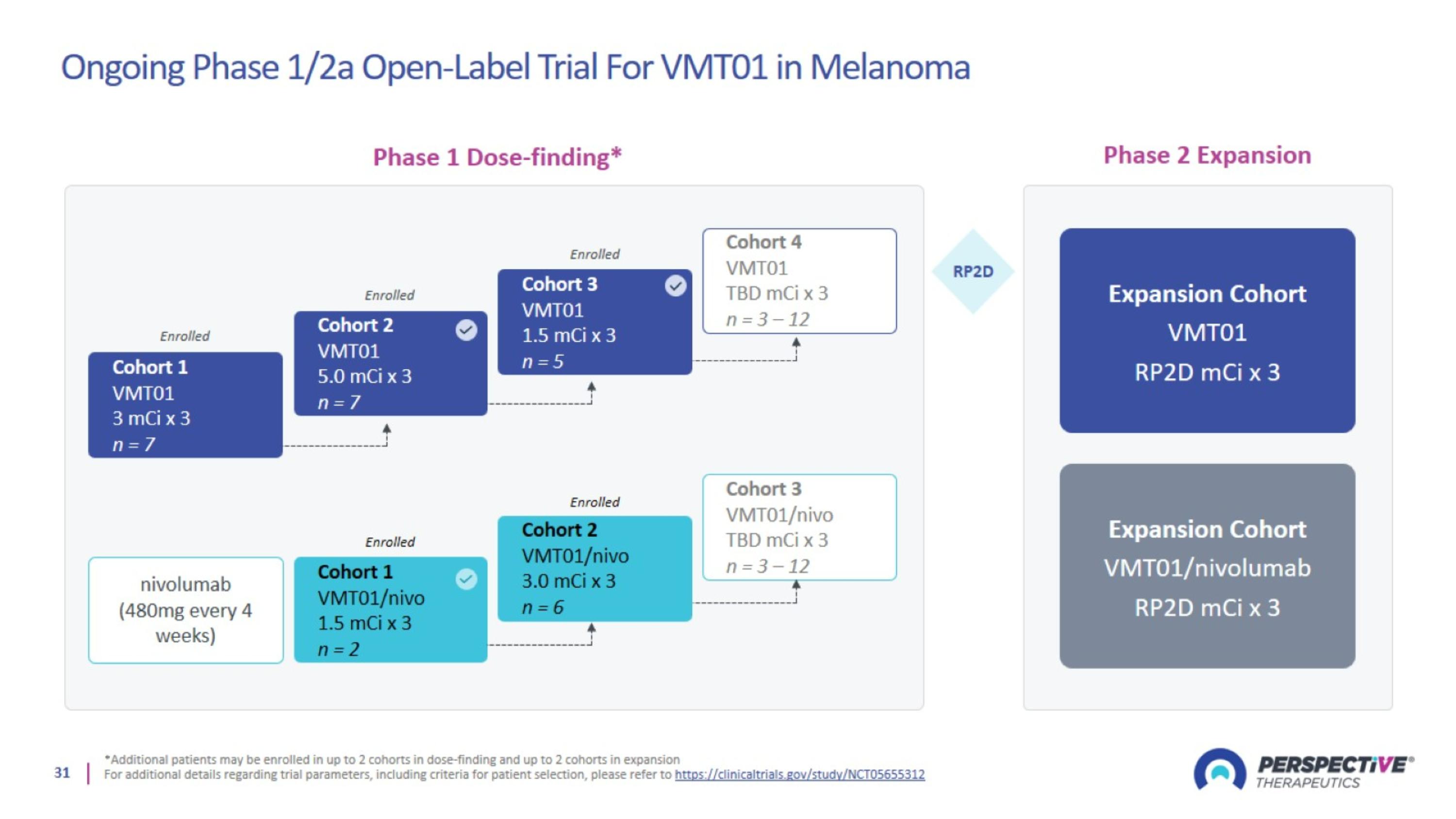
Ongoing Phase 1/2a Open-Label Trial For VMT01 in Melanoma
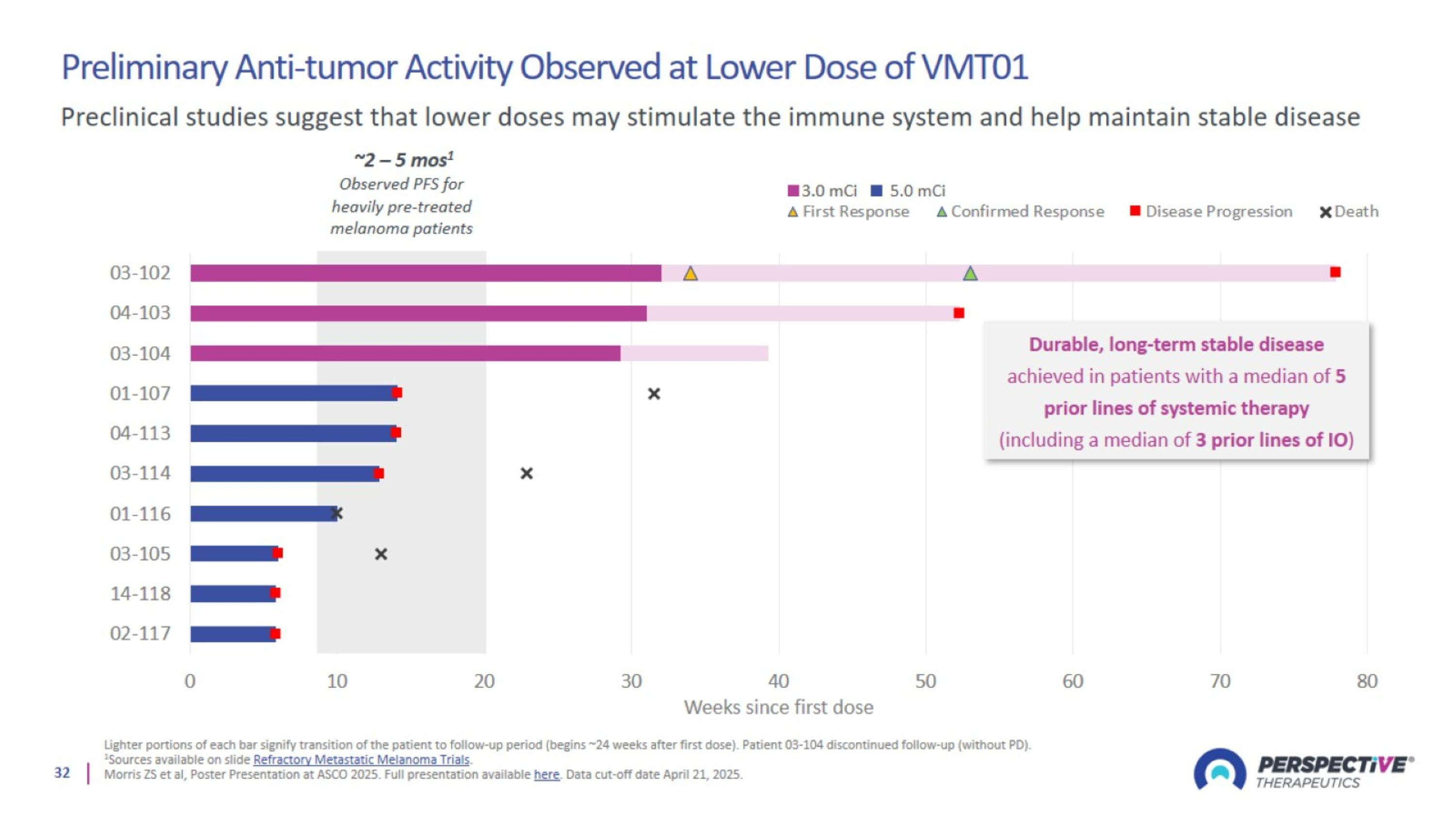
Preliminary Anti-tumor Activity Observed at Lower Dose of VMT01
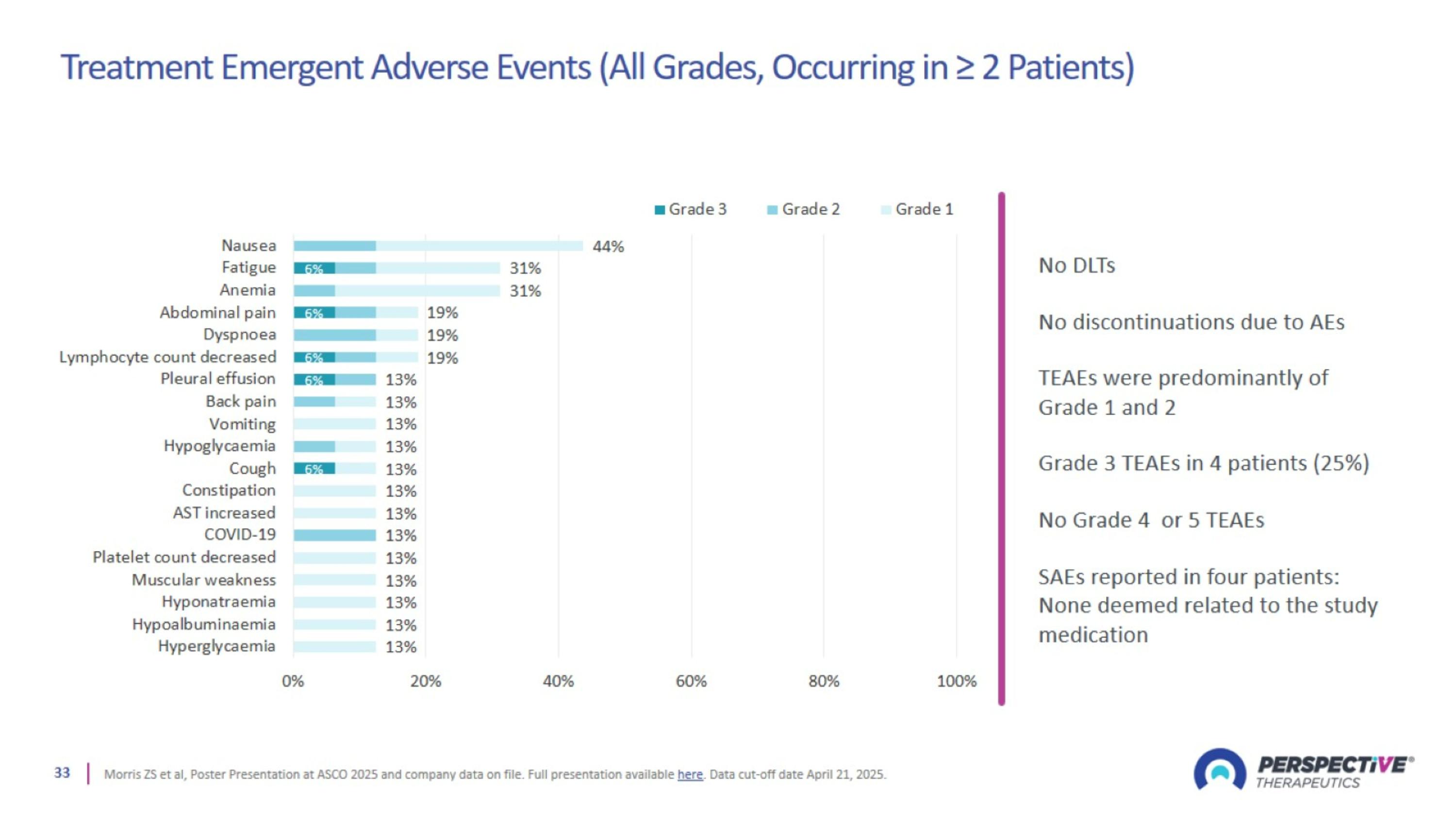
Treatment Emergent Adverse Events (All Grades, Occurring in ≥ 2 Patients)

VMT01 is Well-tolerated with Initial Anti-tumor Activity Supporting Further Development
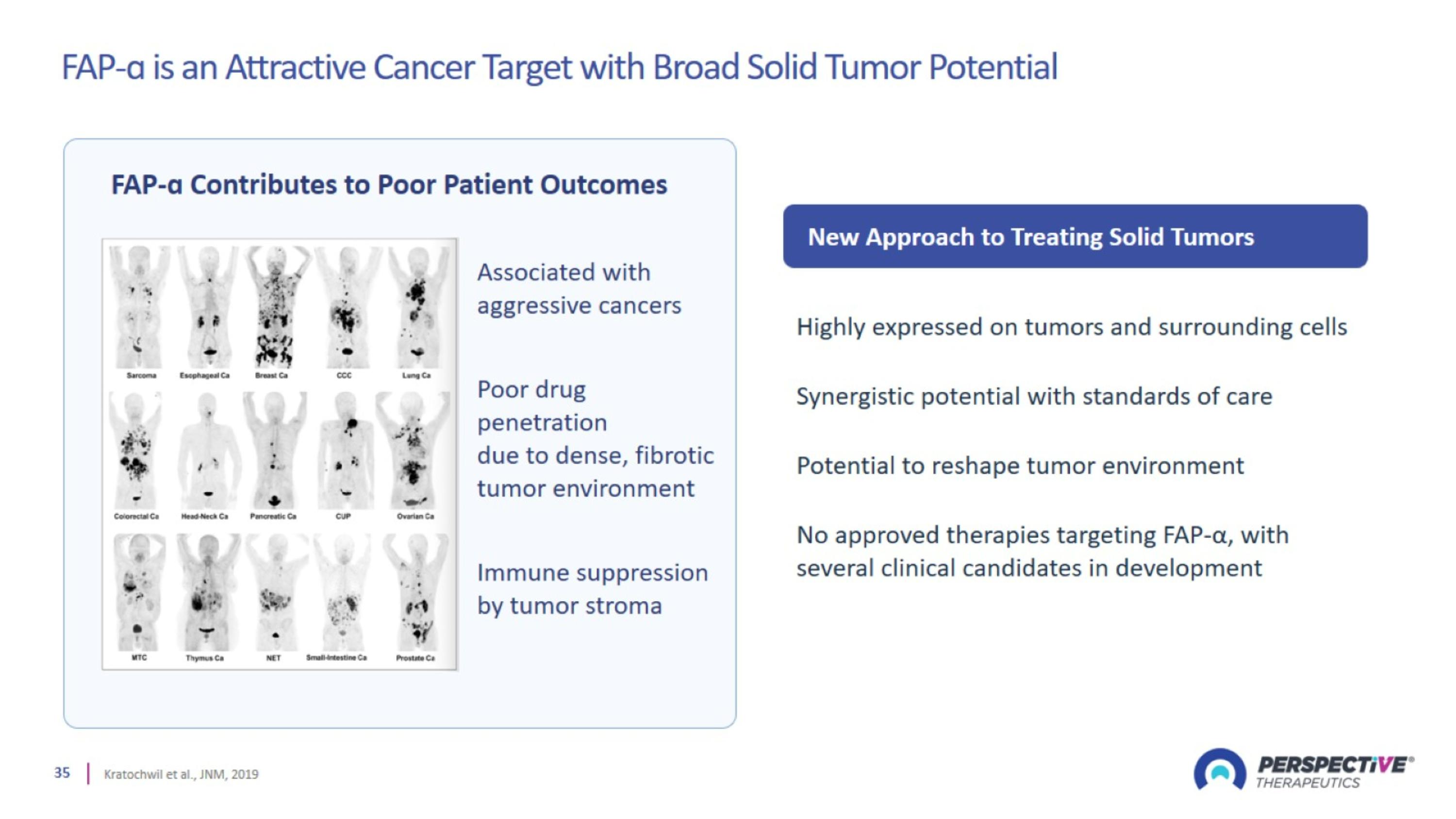
FAP-ɑ is an Attractive Cancer Target with Broad Solid Tumor Potential
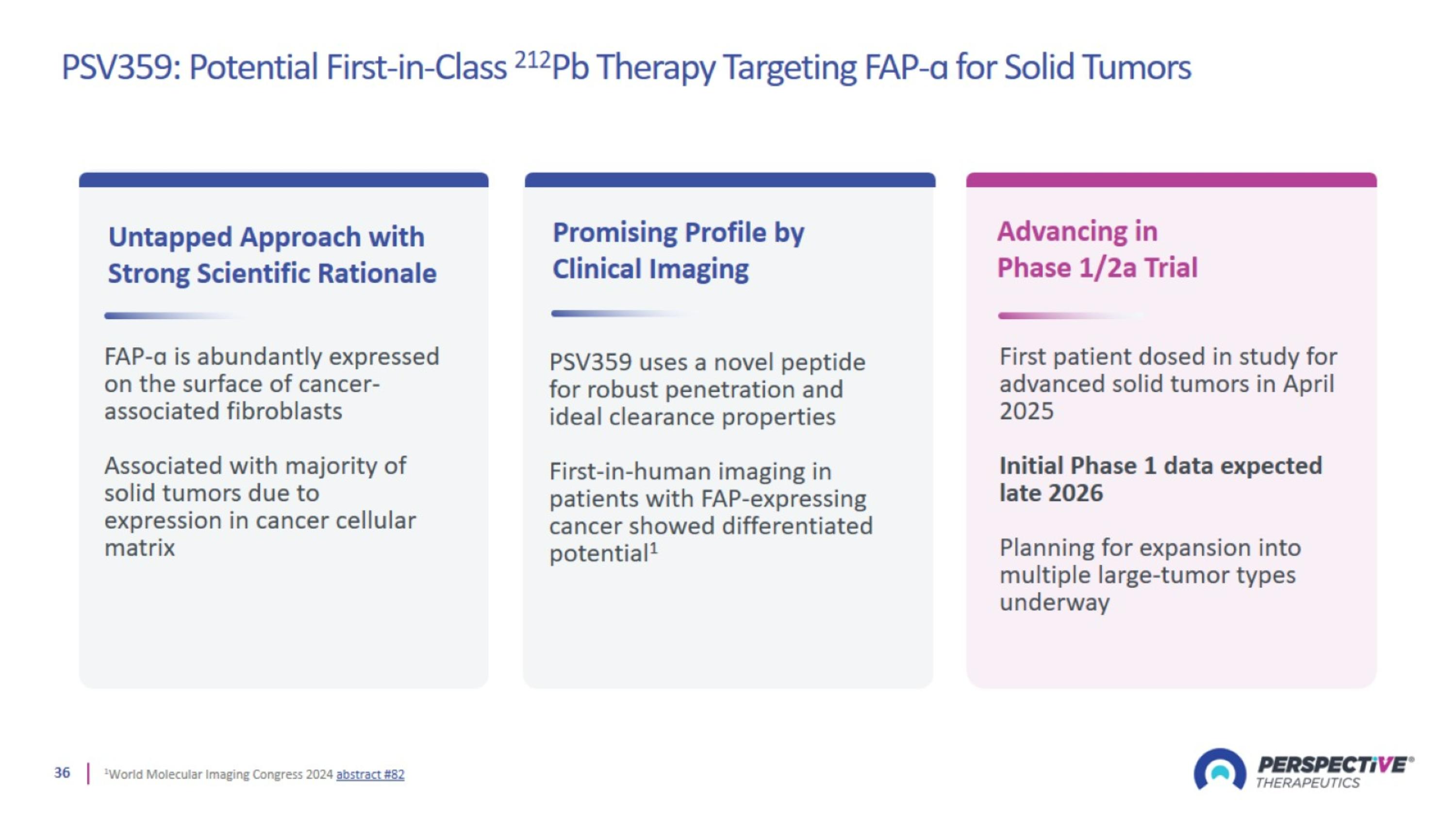
PSV359: Potential First-in-Class 212Pb Therapy Targeting FAP-ɑ for Solid Tumors
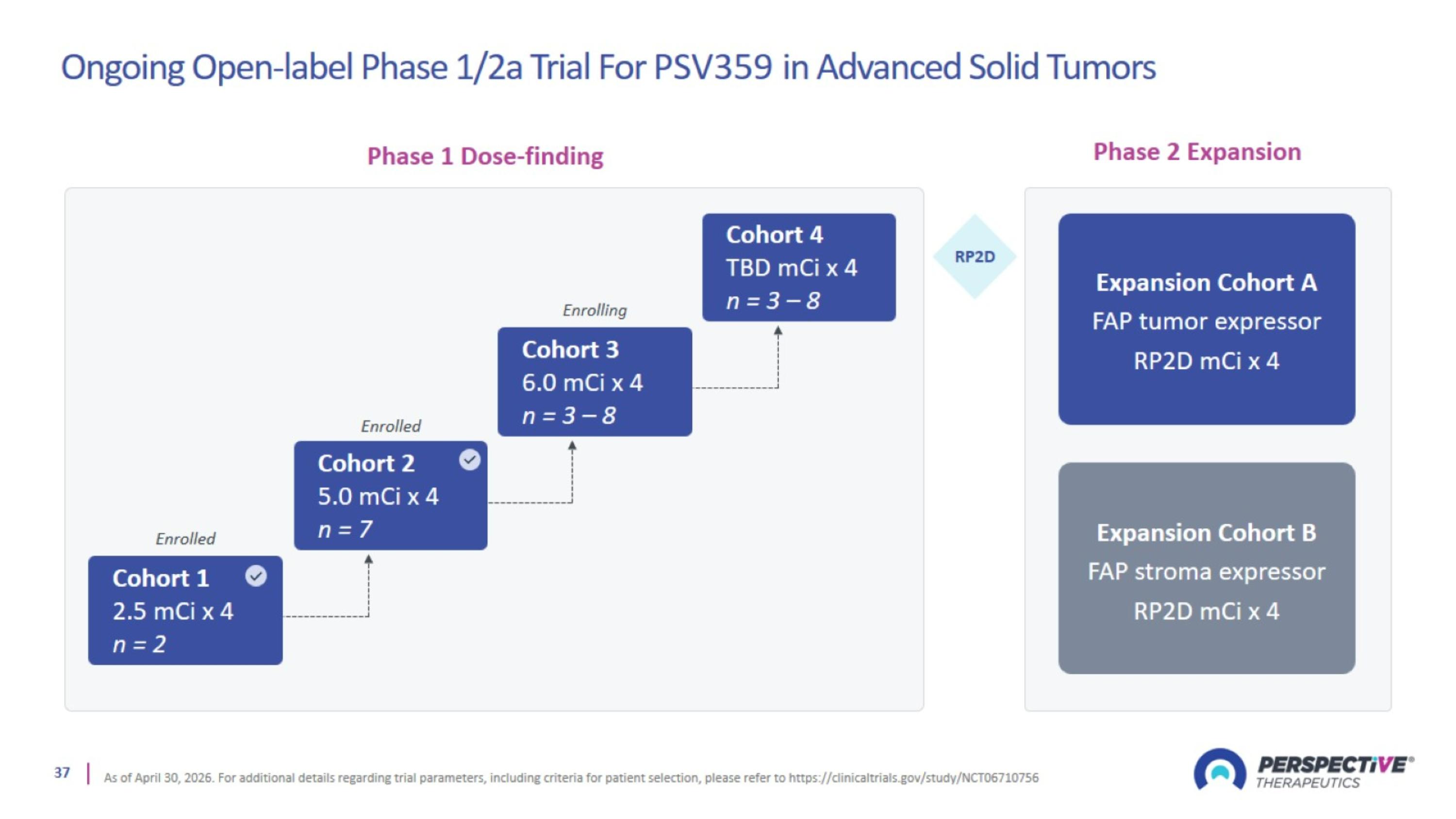
Ongoing Open-label Phase 1/2a Trial For PSV359 in Advanced Solid Tumors
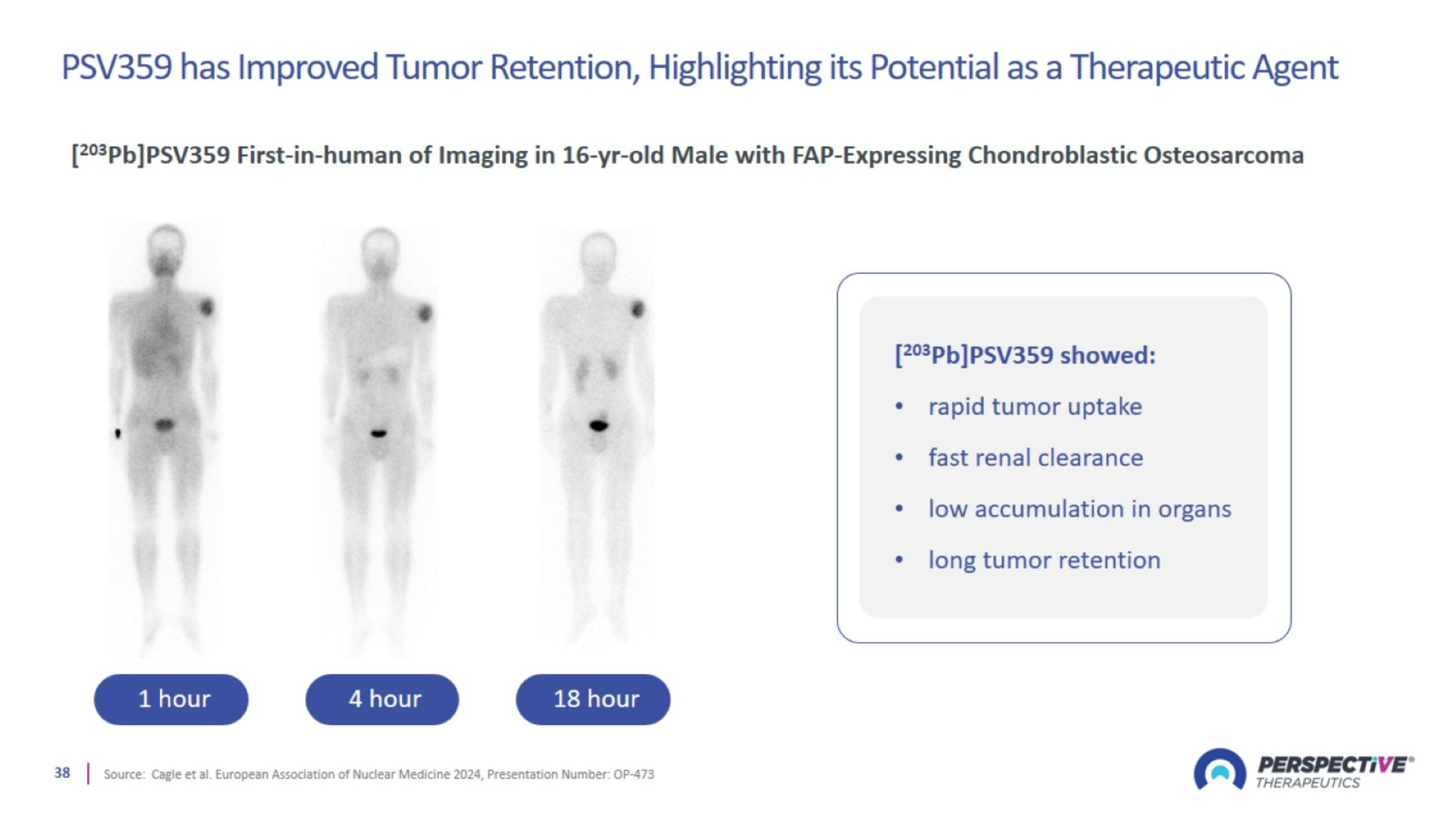
PSV359 has Improved Tumor Retention, Highlighting its Potential as a Therapeutic Agent
Strong IP Portfolio Covering All Aspects of Radiopharmaceutical Value Chain

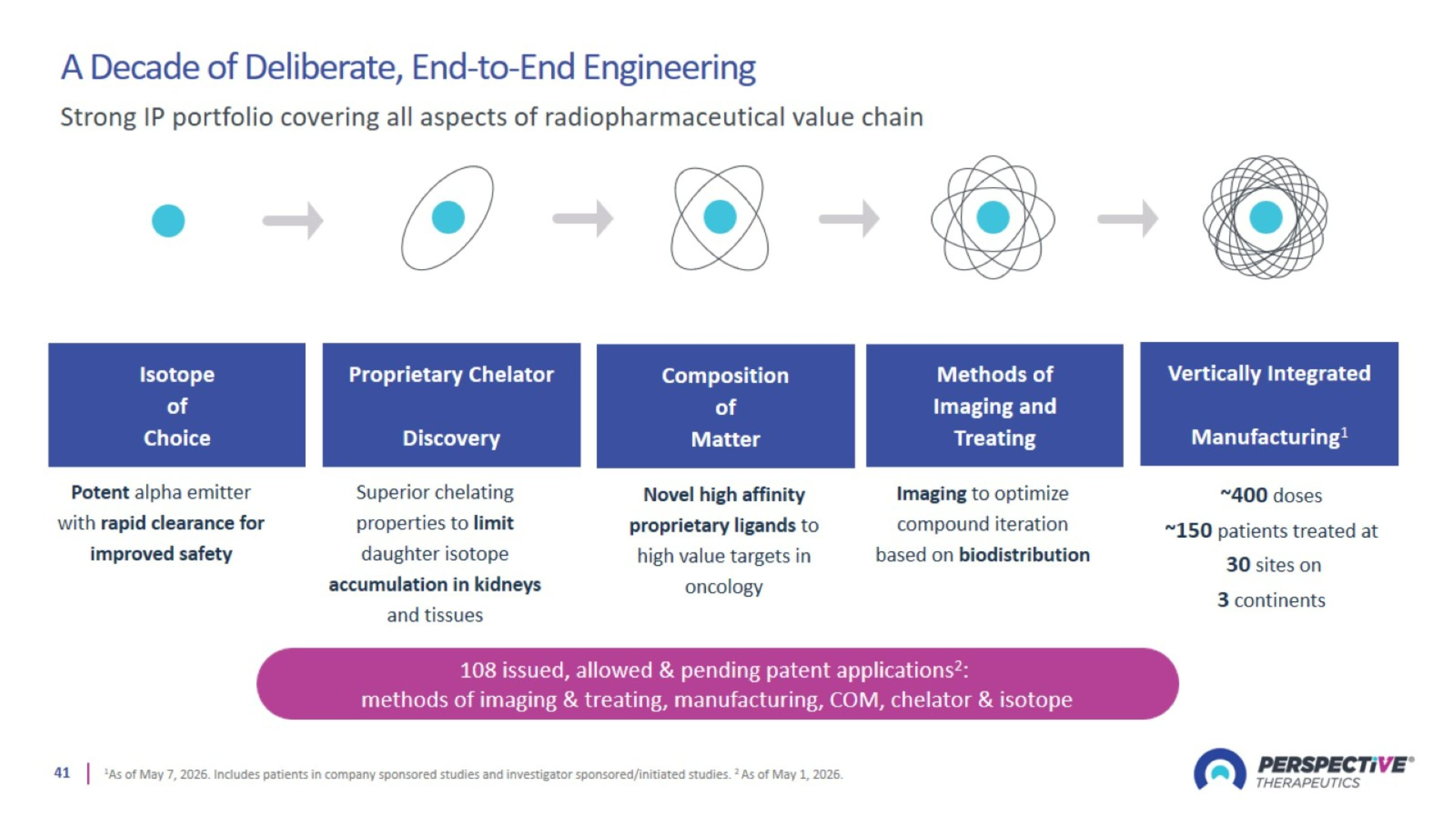
A Decade of Deliberate, End-to-End Engineering
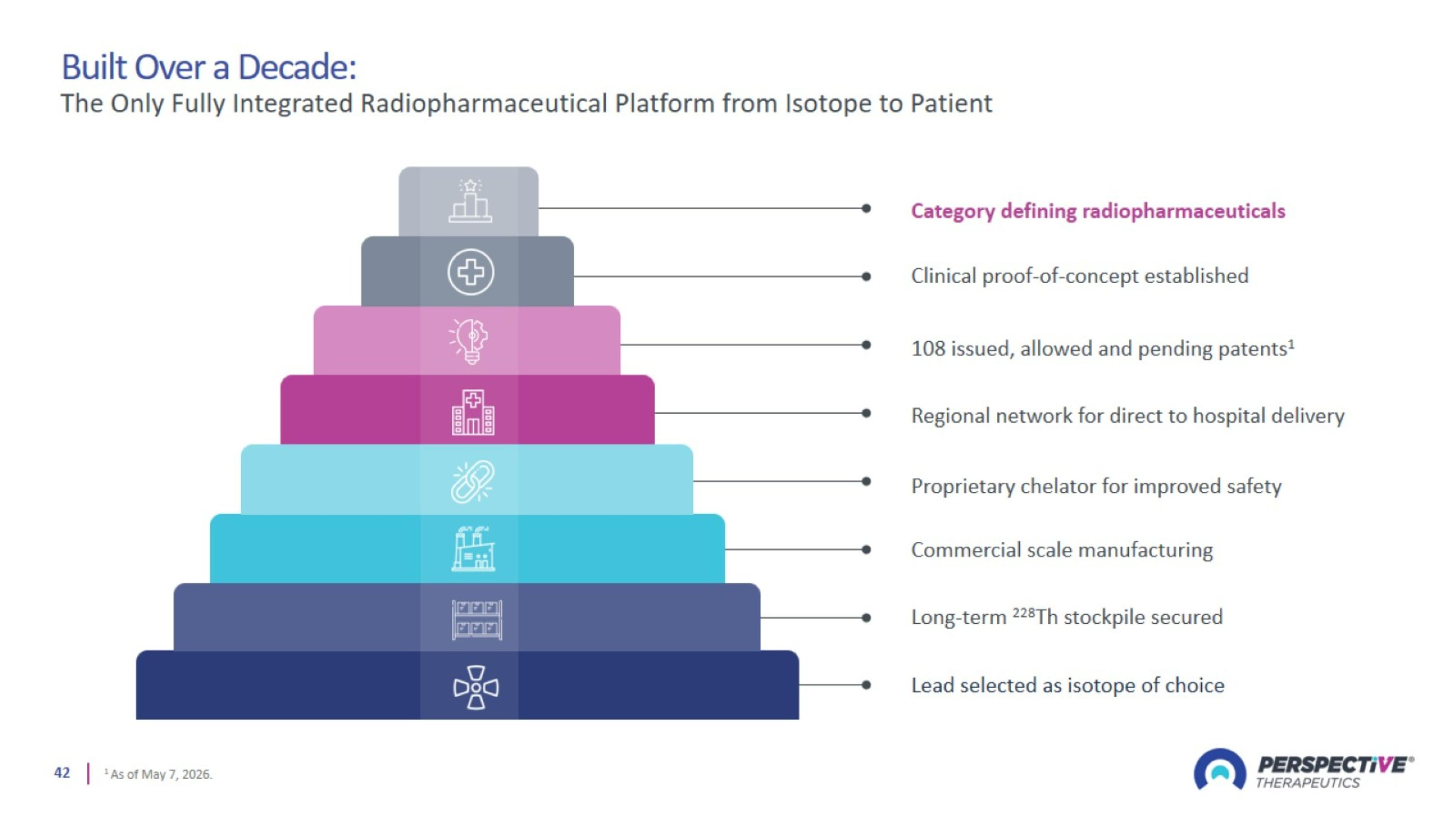
Built Over a Decade:


Abbreviations
APPENDIX

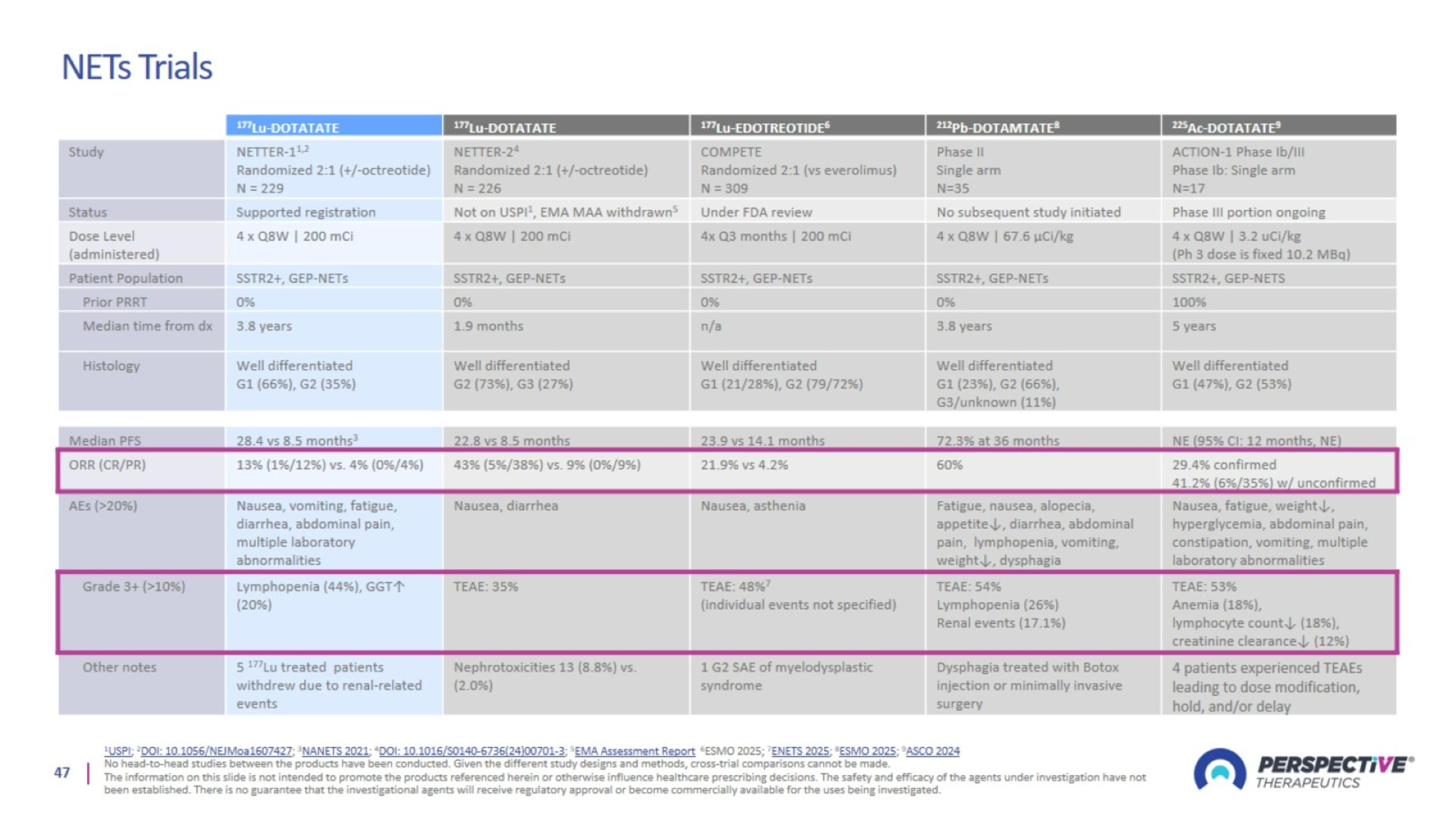
NETs Trials
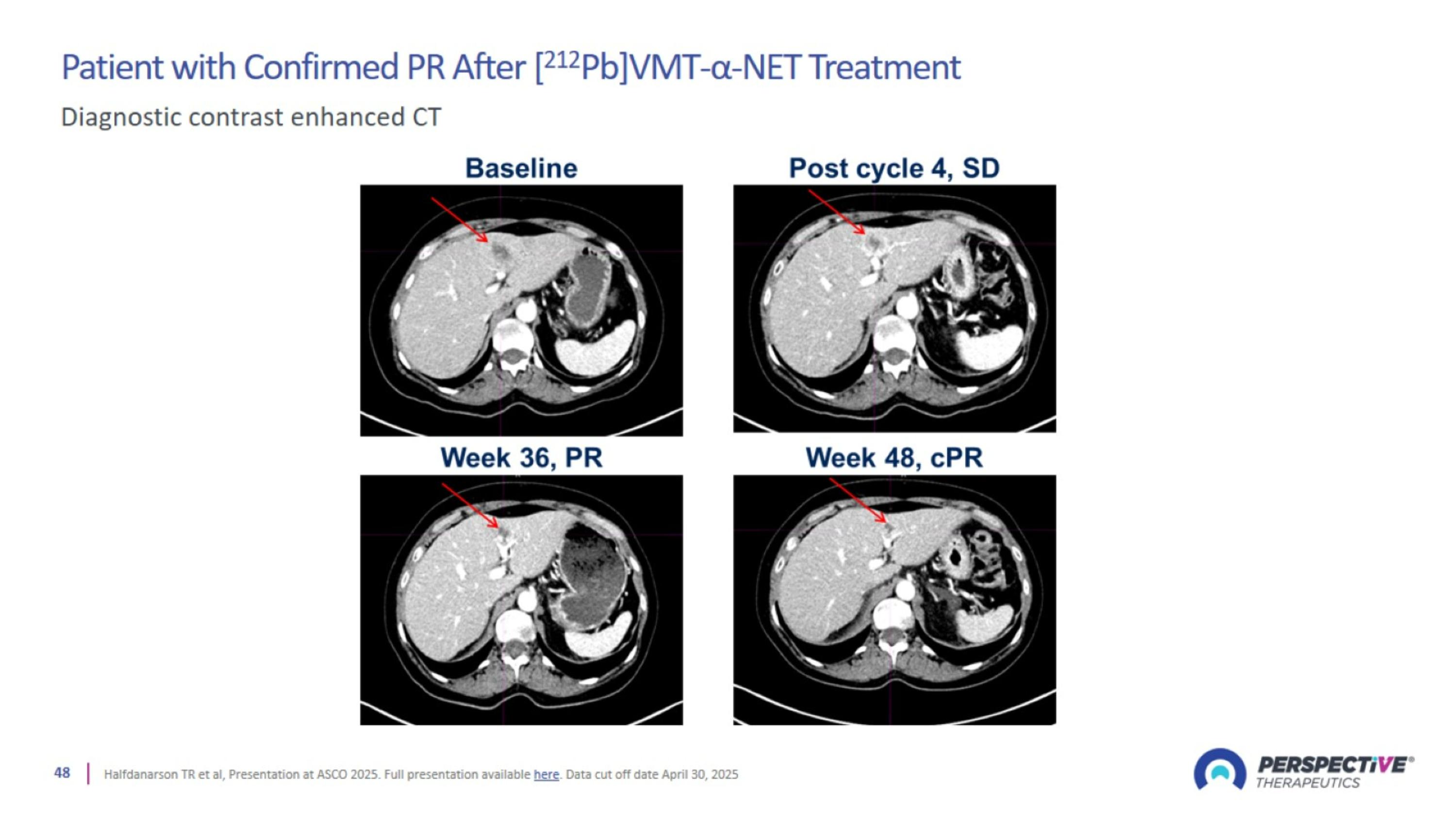
Patient with Confirmed PR After [212Pb]VMT-α-NET Treatment
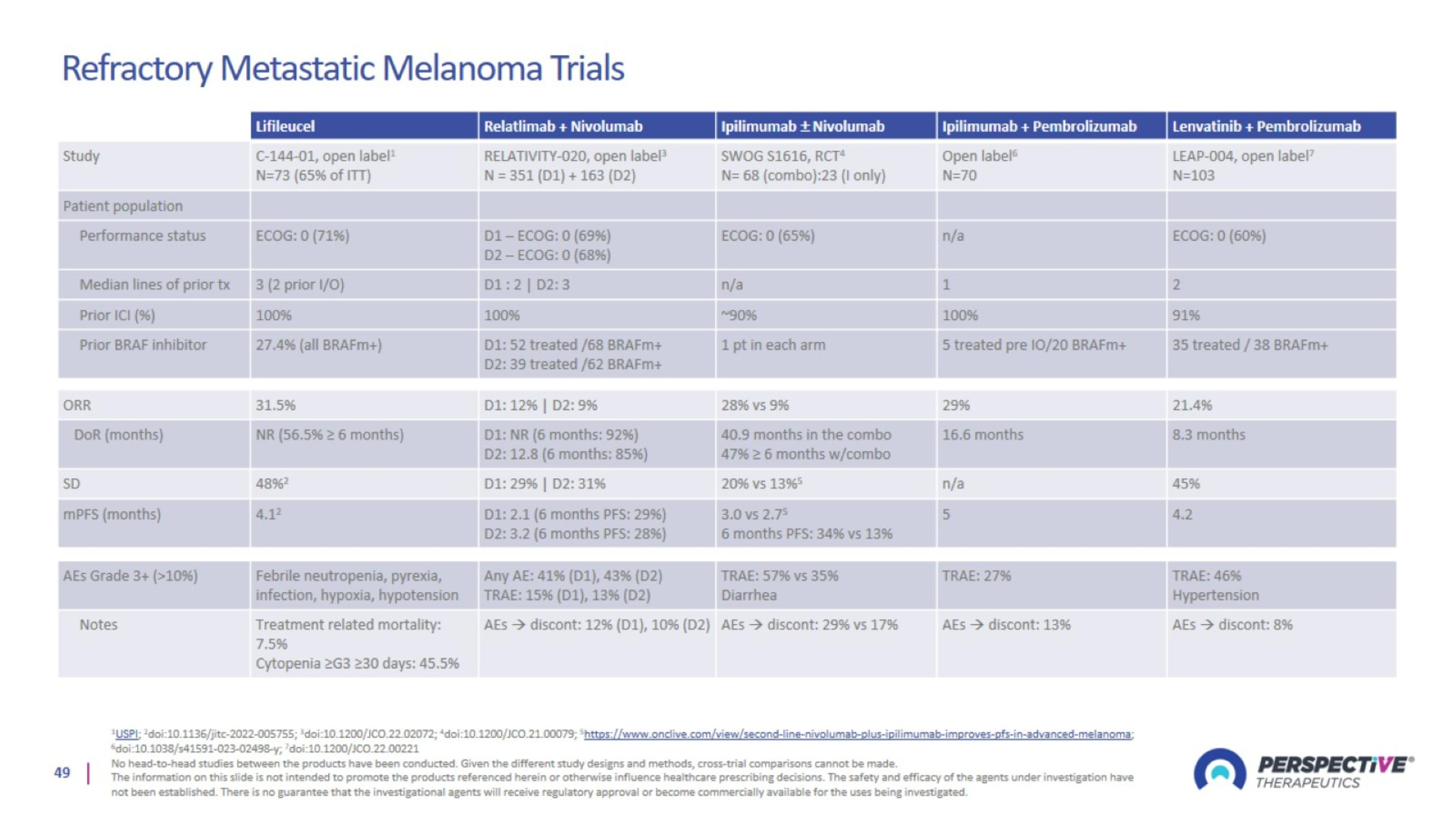
Refractory Metastatic Melanoma Trials
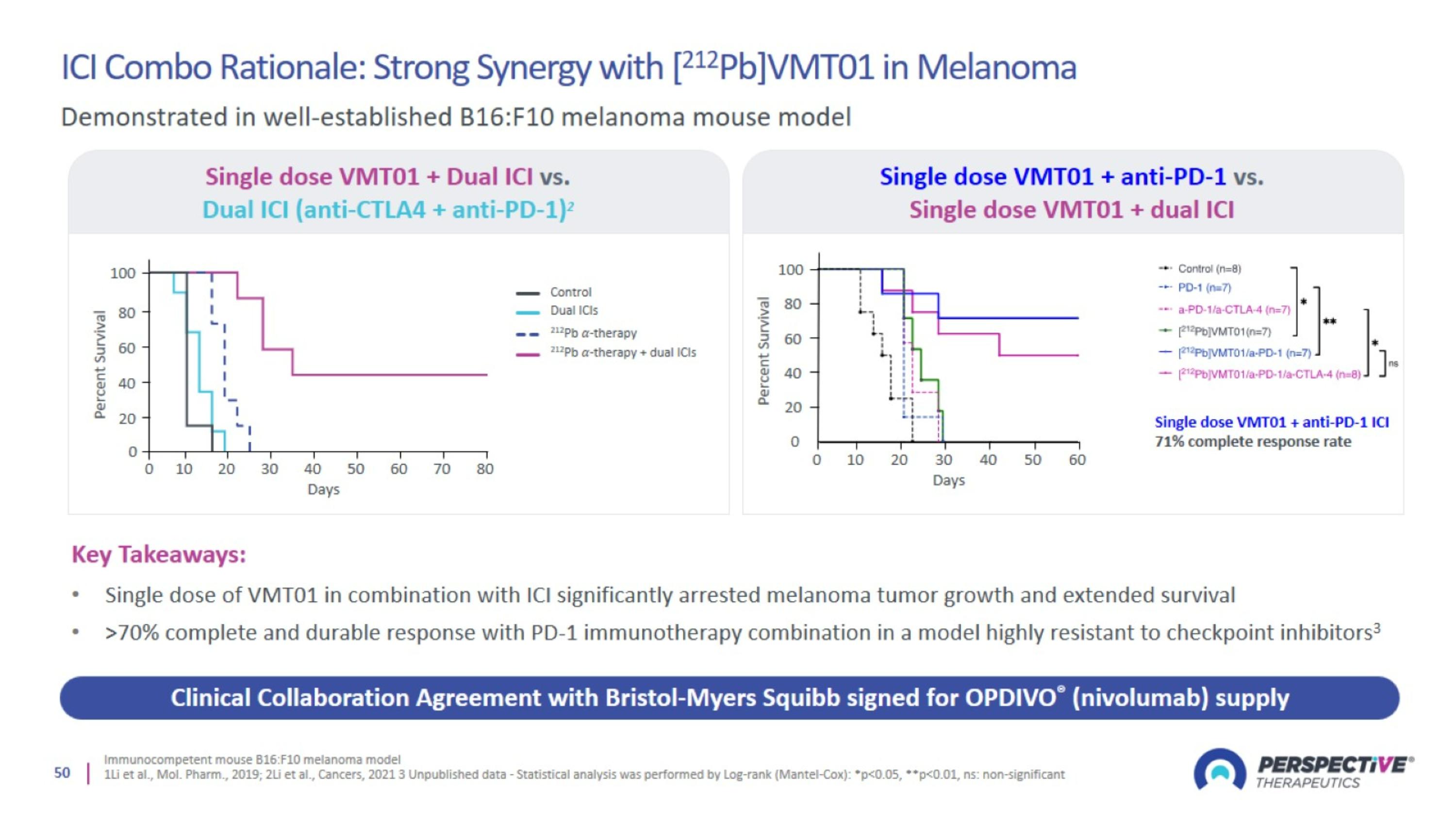
ICI Combo Rationale: Strong Synergy with [212Pb]VMT01 in Melanoma

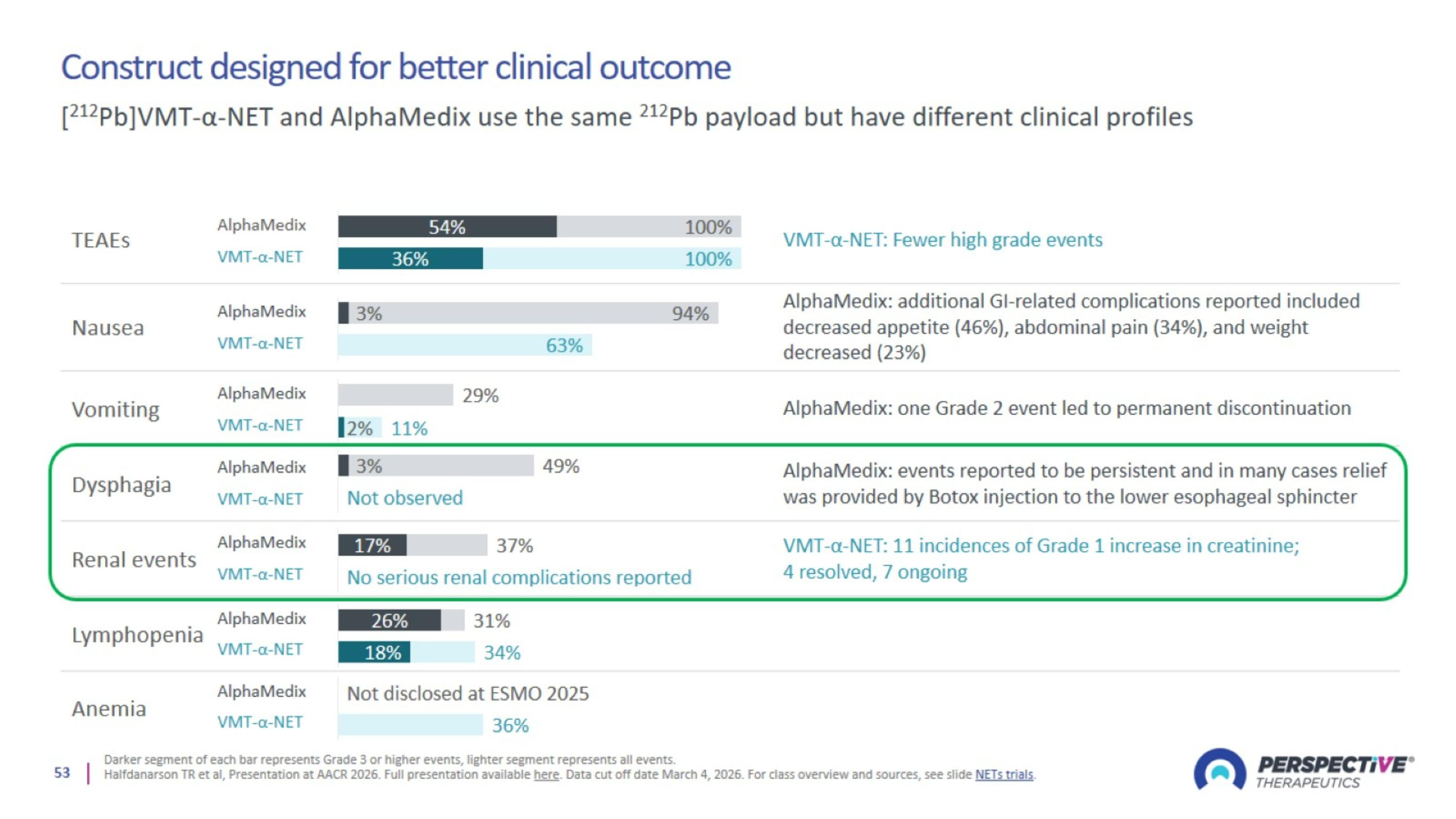
Construct designed for better clinical outcome
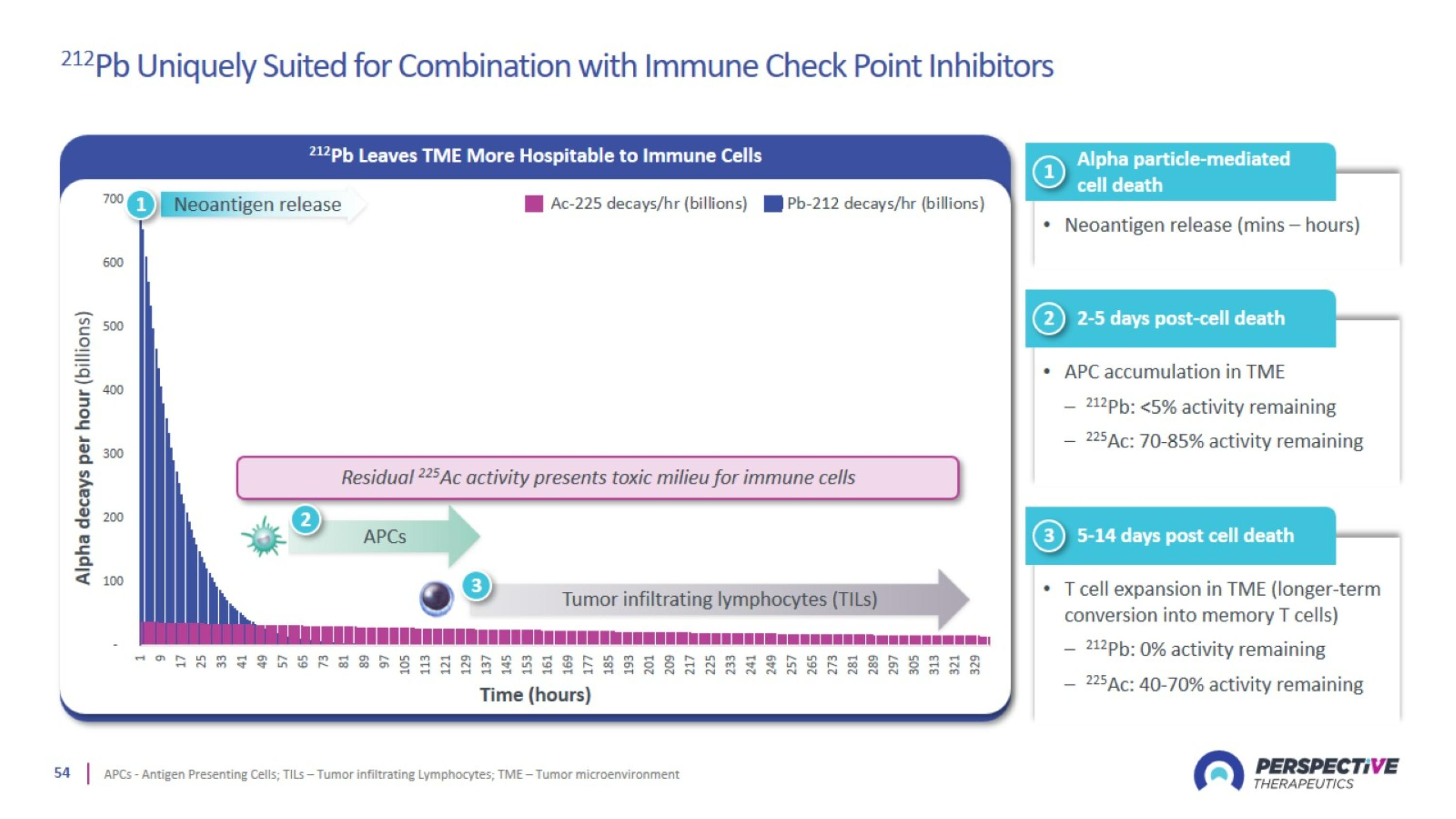
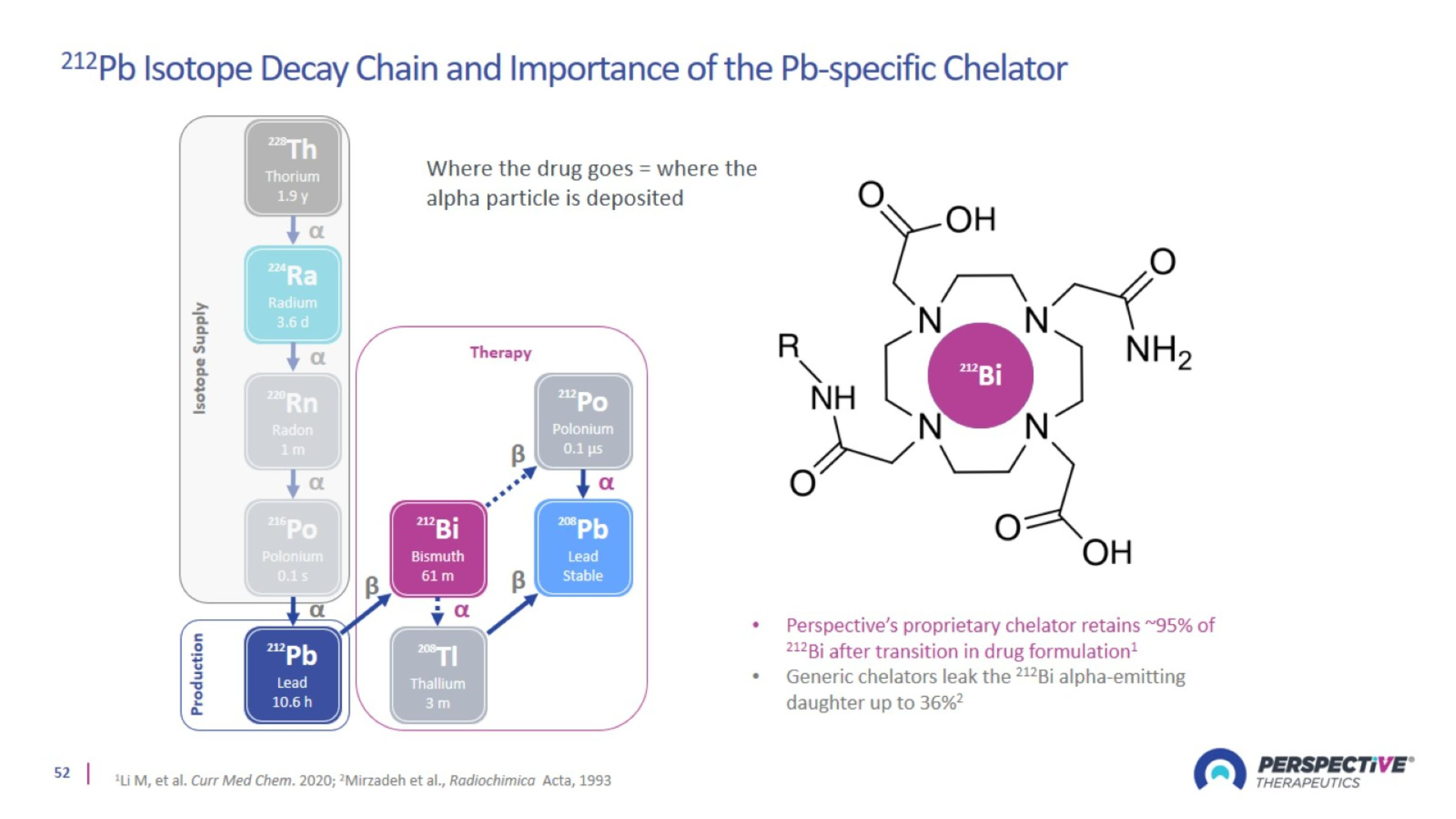
212Pb Uniquely Suited for Combination with Immune Check Point Inhibitors