
Exhibit 99.2

©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED First Quarter 2026 Earnings & Corporate Update Liquidia Corporation May 11, 2026

2 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED Forward - looking statements This presentation may include forward - looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 . All statements contained in this presentation other than statements of historical facts, including statements regarding our future results of operations and financial position, our strategic and financial initiatives, our business strategy and plans and our objectives for future operations, are forward - looking statements . Forward - looking statements, including statements regarding clinical trials, clinical studies and other clinical work (including the funding therefor, anticipated patient enrollment, safety data, study data, trial outcomes, timing or associated costs), regulatory applications and related submission contents and timelines, the timelines or outcomes related to patent litigation with United Therapeutics in the U . S . District Court for the District of Delaware and U . S . District Court for the Middle District of North Carolina, or other litigation between Liquidia and United Therapeutics or others, including rehearings or appeals of decisions in any such proceedings, the issuance of patents by the USPTO and our ability to execute on our strategic or financial initiatives, our estimates regarding future expenses, capital requirements and needs for additional financing, and potential revenue and profitability of YUTREPIA involve significant risks and uncertainties and actual results could differ materially from those expressed or implied herein . Our ability to maintain YUTREPIA’s approval and to continue commercialization of YUTREPIA remain subject to ongoing litigation in which United Therapeutics is seeking injunctive relief, which could block our ability to continue to sell YUTREPIA for one or both of PAH and PH - ILD . The words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “would,” and similar expressions are intended to identify forward - looking statements . We have based these forward - looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy, short - term and long - term business operations and objectives and financial needs . These forward - looking statements are subject to a number of risks discussed in our filings with the SEC, as well as a number of uncertainties and assumptions . Moreover, we operate in a very competitive and rapidly changing environment and our industry has inherent risks . New risks emerge from time to time . It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward - looking statements we may make . In light of these risks, uncertainties and assumptions, the future events discussed in this presentation may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward - looking statements . Nothing in this presentation should be regarded as a representation by any person that these goals will be achieved, and we undertake no duty to update our goals or to update or alter any forward - looking statements, whether as a result of new information, future events or otherwise .

3 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED Use of Non - GAAP Financial Information To supplement our financial results presented in accordance with U . S . Generally Accepted Accounting Principles (GAAP), this presentation includes certain non - GAAP financial measures, such as Adjusted EBITDA . We believe the use of such non - GAAP financial measures provides investors with additional insight into our operational performance . While we compute non - GAAP financial measures using a consistent method from quarter to quarter and year to year, we may consider whether other significant items that arise in the future should be excluded from our non - GAAP financial measures . Adjusted EBITDA is a non - GAAP measure that represents net income for the period before the impact of interest income, interest expense, other income and expense, income taxes, depreciation and amortization, and certain items that impact comparison of the performance of our business either period - over - period or with other businesses . Adjusted EBITDA should not be considered in isolation or as a substitute to net income or any other measure of financial performance calculated and presented in accordance with GAAP . Our calculation of Adjusted EBITDA may not be comparable to similarly titled measures of other companies because other companies may not calculate them in the same manner as we calculate these measures . For a reconciliation of such non - GAAP financial measures to the most directly comparable financial measures prepared in accordance with GAAP, please see the table titled “Reconciliation of Non - GAAP Financial Information” below .

4 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED First quarter 2026 highlights LEADING CATEGORY GROWTH BROADENING THE FRANCHISE SELF - FUNDED INVESTMENT $129.9 net product sales 8 studies $71.2M adj. EBITDA* +44% Q/Q growth in Q12026 from Q4 2025 • >4,500 unique patient prescriptions through 30 - Apr • ~980 prescribers since launch • ~25% growth in physicians prescribing to ≥5 patients since end of February On - going, recruiting or planned clinical studies in 2026 • Recruiting ASCENT Cohort B Tyvaso Transitions (PH - ILD) • Recruiting Phase 3 Re - Spire globally (PH - ILD) • Advancing IPF/PPF, PH - COPD, SSc - Raynaud’s programs 3rd consecutive quarter of profitability • 2.6x increase in adjusted EBITDA* Q/Q • $222.8M ending cash +$32.1M from 2025 year - end *Non - GAAP financial measure. See definition and full reconciliation on slide 10 or in our earnings press release at https://liquidia.com/investors/press - releases .
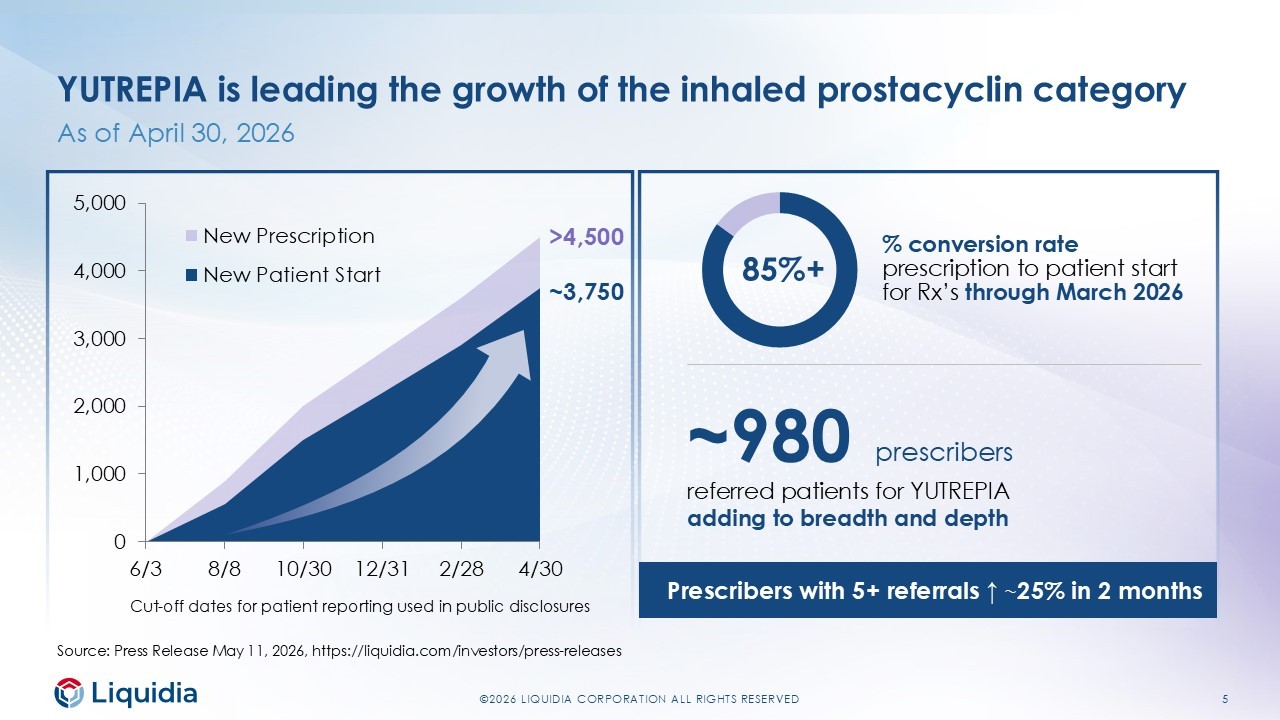
5 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED YUTREPIA is leading the growth of the inhaled prostacyclin category As of April 30, 2026 0 1,000 2,000 3,000 4,000 5,000 6/3 8/8 10/30 12/31 2/28 4/30 New Prescription New Patient Start % conversion rate prescription to patient start for Rx’s through March 2026 85%+ ~3,750 >4,500 Cut - off dates for patient reporting used in public disclosures Source: Press Release May 11, 2026, https://liquidia.com/investors/press - releases ~980 prescribers referred patients for YUTREPIA adding to breadth and depth Prescribers with 5+ referrals ↑ ~ 25% in 2 months
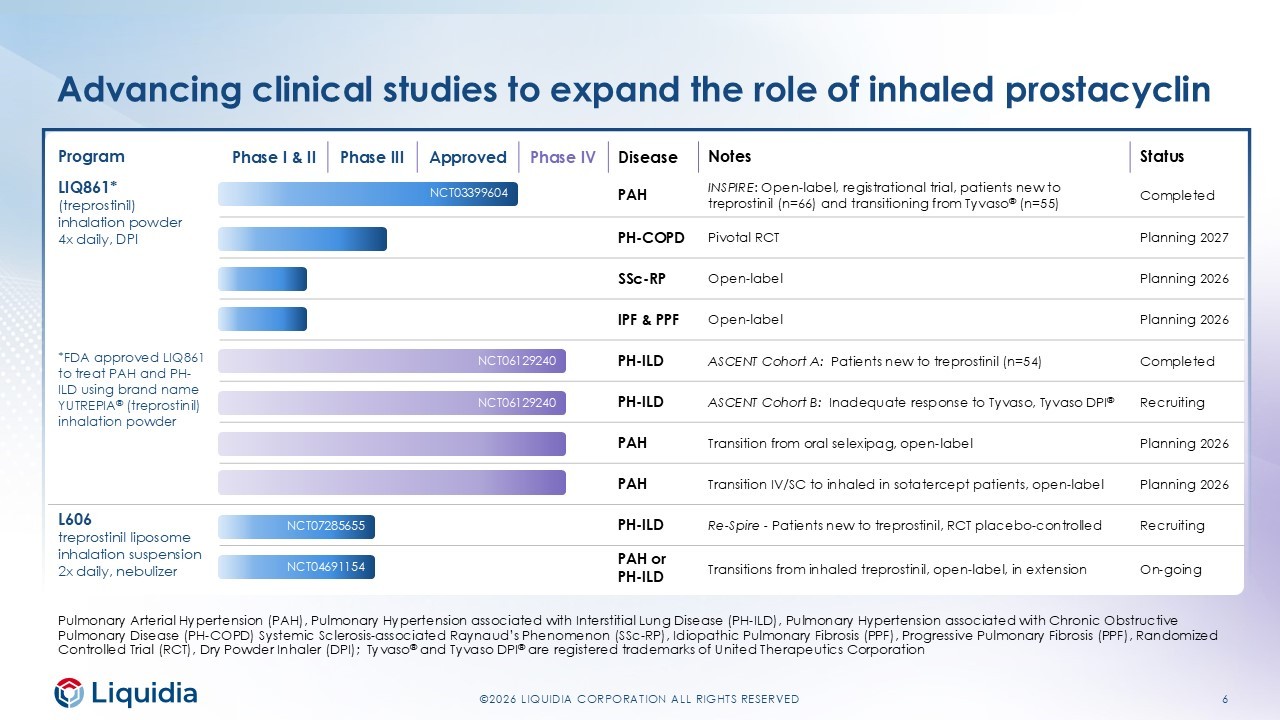
6 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED Status Notes Disease Phase IV Approved Phase III Phase I & II Program Completed INSPIRE : Open - label, r egistrational trial, patients new to treprostinil (n=66) and transitioning from Tyvaso ® (n=55) PAH LIQ861* (treprostinil) inhalation powder 4x daily, DPI *FDA approved LIQ861 to treat PAH and PH - ILD using brand name YUTREPIA ® (treprostinil) inhalation powder Planning 2027 Pivotal RCT PH - COPD Planning 2026 Open - label SSc - RP Planning 2026 Open - label IPF & PPF Completed ASCENT Cohort A: Patients new to treprostinil (n=54) PH - ILD Recruiting ASCENT Cohort B: Inadequate response to Tyvaso, Tyvaso DPI ® PH - ILD Planning 2026 Transition from oral selexipag , open - label PAH Planning 2026 Transition IV/SC to inhaled in sotatercept patients, open - label PAH Recruiting Re - Spire - Patients new to treprostinil, RCT placebo - controlled PH - ILD L606 treprostinil liposome inhalation suspension 2x daily, nebulizer On - going Transitions from inhaled treprostinil, open - label, in extension PAH or PH - ILD NCT03399604 NCT07285655 NCT04691154 NCT06129240 NCT06129240 Advancing clinical studies to expand the role of inhaled prostacyclin Pulmonary Arterial Hypertension (PAH), Pulmonary Hypertension associated with Interstitial Lung Disease (PH - ILD), Pulmonary Hype rtension associated with Chronic Obstructive Pulmonary Disease (PH - COPD) Systemic Sclerosis - associated Raynaud’s Phenomenon ( SSc - RP), Idiopathic Pulmonary Fibrosis (PPF), Progressive Pulmonary Fibrosis (PPF), Randomized Controlled Trial (RCT), Dry Powder Inhaler (DPI); Tyvaso ® and Tyvaso DPI ® are registered trademarks of United Therapeutics Corporation
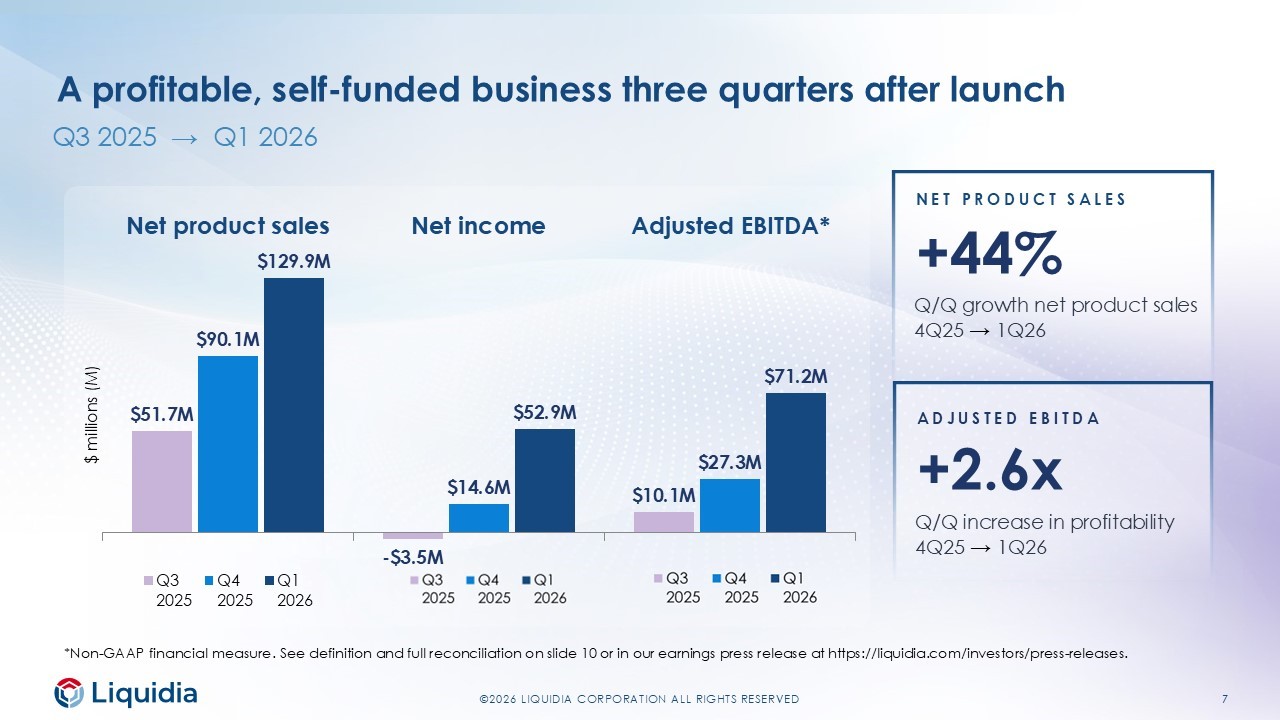
7 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED Q3 2025 → Q1 2026 $51.7M - $3.5M $10.1M $90.1M $14.6M $27.3M $129.9M $52.9M $71.2M Net product sales Net income Adjusted EBITDA* $ millions (M) Q3 2025 Q4 2025 Q1 2026 ADJUSTED EBITDA +2.6x Q/Q increase in profitability 4Q25 → 1Q26 *Non - GAAP financial measure. See definition and full reconciliation on slide 10 or in our earnings press release at https://liquidia.com/investors/press - releases . A profitable, self - funded business three quarters after launch NET PRODUCT SALES +44% Q/Q growth net product sales 4Q25 → 1Q26

8 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED Key financial metrics $222.8M Ending cash and cash equivalents for 1Q 2026 • Increase of $32.1 million in cash during 1Q 2026 • 1Q 2026 was third consecutive quarter of profitability Three Months Ended Select Consolidated Statements of Operations Data 3/31/25 3/31/26 in thousands - 129,881 Product sales, net 3,120 2,984 Service revenue, net $3,120 $132,865 Total revenue - 11,079 Cost of product sales 1,517 773 Cost of service revenue 6,966 12,571 R&D 30,062 46,938 SG&A $38,545 $71,361 Total Costs and Expenses $(35,425) $61,504 Operating Income (Loss)

9 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED Scott Moomaw Chief Commercial Officer Rajeev Saggar Chief Medical Officer Dr. Roger Jeffs Chief Executive Officer Michael Kaseta COO & CFO Russell Schundler General Counsel Q&A session

10 ©2026 LIQUIDIA CORPORATION ALL RIGHTS RESERVED Reconciliation of Net Income to Adjusted EBITDA Three months ended (unaudited, in thousands) Adjusted EBITDA is a non - GAAP measure. See definition and full reconciliation in our earnings press release at https:// liquidia.com /investors/press - releases . Mar 31, 2026 Dec 31, 2025 Sept 30, 2025 $52,862 $14,555 $(3,533) Net income 4,722 5,232 5,300 Interest expense, net 3,920 – – Income tax expense 497 321 476 Depreciation & amortization $62,001 $20,108 $2,243 EBITDA 9,217 7,206 7,899 Stock - based compensation $71,218 $27,314 $10,142 Adjusted EBITDA