
CO PY RI GH T © A BC EL LE RA MAY 11, 2026 Q1 2026 BUSINESS UPDATE

These statements involve risks, uncertainties and other factors that may cause actual results, levels of activity, performance, or achievements to be materially different from the information expressed or implied by these forward-looking statements. These risks, uncertainties, other factors, and definition of our business metrics are described under “Risk Factors,” “Management's Discussion and Analysis of Financial Condition and Results of Operations” and elsewhere in the documents we file with the Securities and Exchange Commission from time to time. We caution you that forward-looking statements are based on a combination of facts and factors currently known by us and our projections of the future, about which we cannot be certain. As a result, the forward-looking statements may not prove to be accurate. The forward-looking statements in this presentation represent our views as of the date hereof. We undertake no obligation to update any forward-looking statements for any reason, except as required by law. DISCLAIMER This presentation contains forward-looking statements, including statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. The forward-looking statements are based on management’s beliefs and assumptions and on information currently available to management. All statements contained in this presentation other than statements of historical fact are forward-looking statements, including statements regarding our ability to develop, commercialize and achieve market acceptance of our current and planned products and services, our research and development efforts, and other matters regarding our business strategies, use of capital, results of operations and financial position, and plans and objectives for future operations. Certain data in this presentation are derived from cross-study comparisons and not based on any head-to-head clinical trials. Cross-study comparisons are inherently limited and may suggest misleading similarities and differences and are presented for informational purposes. In some cases, you can identify forward-looking statements by the words “may,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,” “project,” “potential,” “continue,” “ongoing” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. 2 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 B us in es s U pd at e
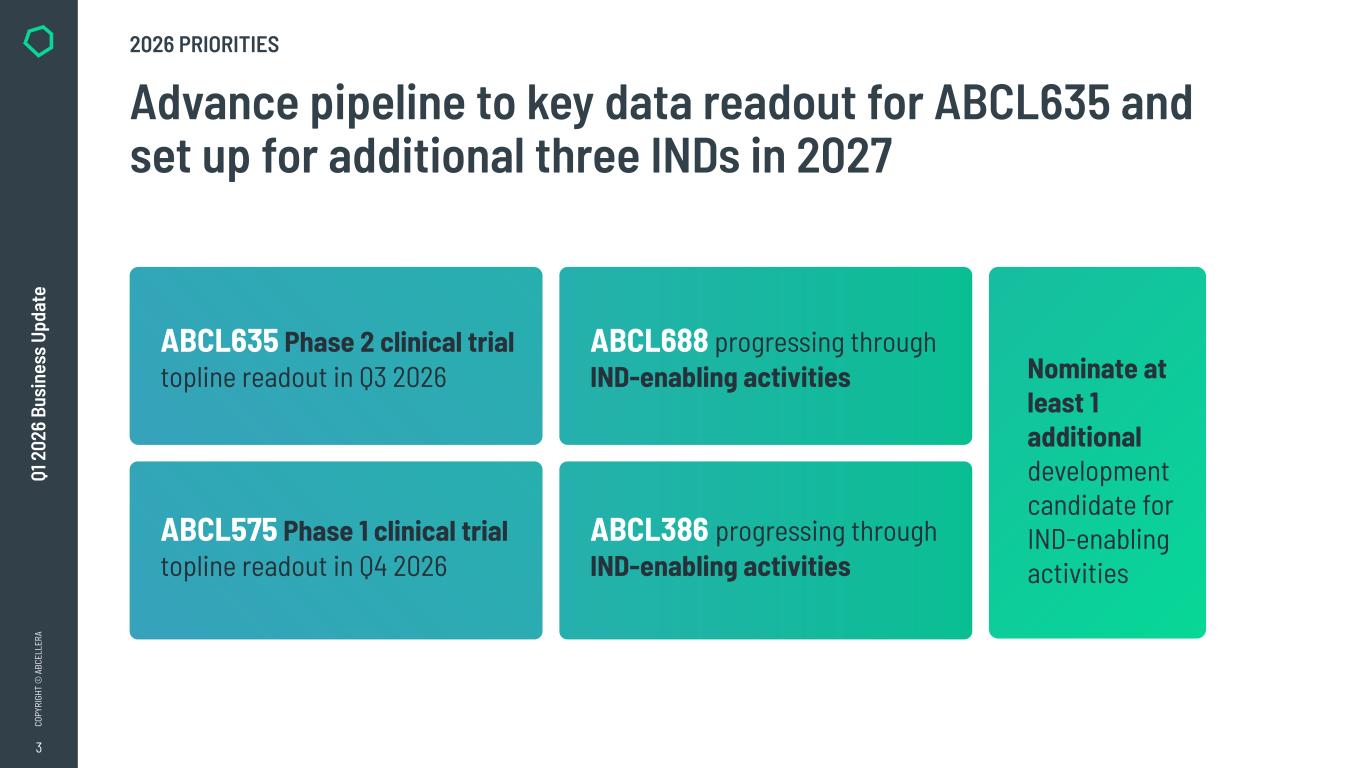
3 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Advance pipeline to key data readout for ABCL635 and set up for additional three INDs in 2027 Nominate at least 1 additional development candidate for IND-enabling activities 2026 PRIORITIES ABCL635 Phase 2 clinical trial topline readout in Q3 2026 ABCL575 Phase 1 clinical trial topline readout in Q4 2026 ABCL688 progressing through IND-enabling activities ABCL386 progressing through IND-enabling activities
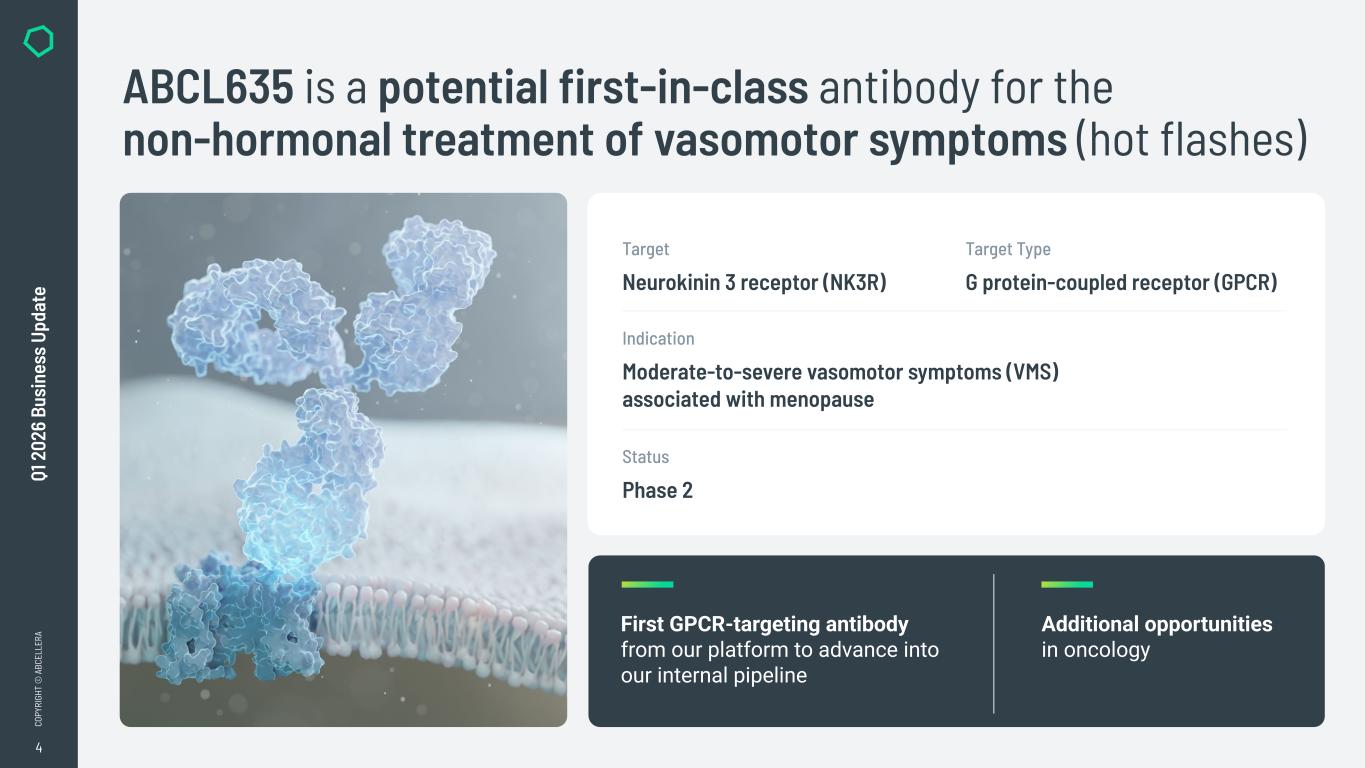
4 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e ABCL635 is a potential first-in-class antibody for the non-hormonal treatment of vasomotor symptoms (hot flashes) First GPCR-targeting antibody from our platform to advance into our internal pipeline Additional opportunities in oncology Target Neurokinin 3 receptor (NK3R) Indication Moderate-to-severe vasomotor symptoms (VMS) associated with menopause Target Type G protein-coupled receptor (GPCR) Status Phase 2

5 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e ABCL635 could help to address a large unmet need for the treatment of VMS associated with menopause Symptoms: Sudden and intense feeling of heat that leads to sweating, chills, and interrupted sleep.2 Investment Thesis ABCL635 aims to provide a treatment option that has: ● Comparable efficacy to approved small molecules ● A differentiated safety profile without the need for liver enzyme monitoring ● Dosing convenience with a once monthly subcutaneous (SC) self-injection Market Opportunity 1M+ women could benefit from safe and effective non-hormonal treatments.3 Clinical Need 4+ years is the median duration for vasomotor symptoms post final menstrual period.1 ~12M women in the US experience moderate-to-severe VMS.3 >6M of which seek treatment.3 20% for whom hormone therapy is unsuitable due to contraindications or other factors.4 through selective antagonism of NK3R with a high-affinity antibody Reduce VMS frequency & severity 1. Avis et al., JAMA Intern Med., 2015. 2. Nappi et al., Menopause, 2021. 3. Management estimate based on US census data, 2023; Todorova et al., Menopause, 2023; Nappi et al., Menopause, 2021; Stute et al., Maturitas, 2022. 4. Management estimate based on Nappi et al., Menopause, 2021; Stute et al., Maturitas, 2022. 5. Management estimate based on Wholesale Acquisition Cost of Veozah and Lynkuet (accessed from Micromedex Red Book) and above sources. Actual market size may differ. $6B+ Total Addressable Market estimated for non-hormonal treatments given small-molecule gross prices of >$5K per patient per year.5
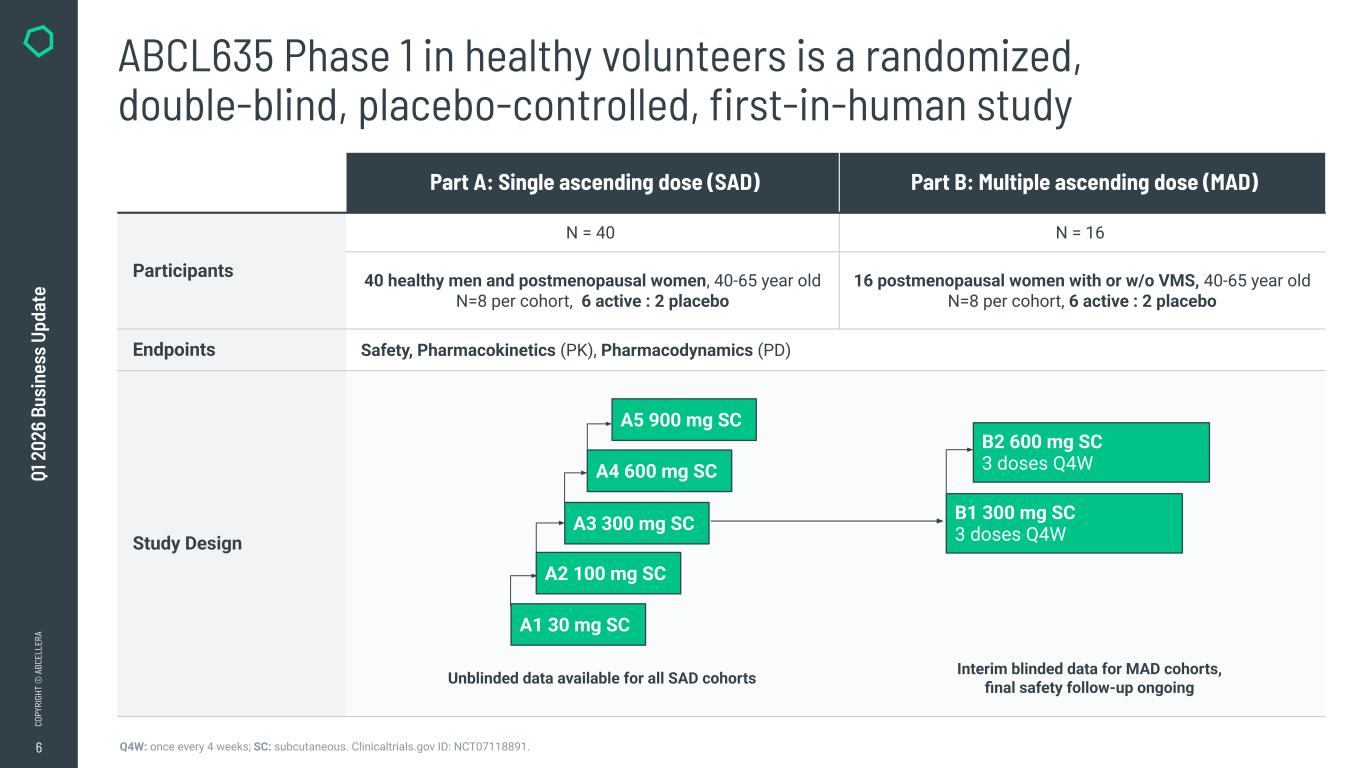
6 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e ABCL635 Phase 1 in healthy volunteers is a randomized, double-blind, placebo-controlled, first-in-human study Part A: Single ascending dose (SAD) Part B: Multiple ascending dose (MAD) Participants N = 40 N = 16 40 healthy men and postmenopausal women, 40-65 year old N=8 per cohort, 6 active : 2 placebo 16 postmenopausal women with or w/o VMS, 40-65 year old N=8 per cohort, 6 active : 2 placebo Endpoints Safety, Pharmacokinetics (PK), Pharmacodynamics (PD) Study Design A1 30 mg SC A2 100 mg SC A3 300 mg SC A5 900 mg SC A4 600 mg SC B1 300 mg SC 3 doses Q4W B2 600 mg SC 3 doses Q4W Q4W: once every 4 weeks; SC: subcutaneous. Clinicaltrials.gov ID: NCT07118891. Unblinded data available for all SAD cohorts Interim blinded data for MAD cohorts, final safety follow-up ongoing
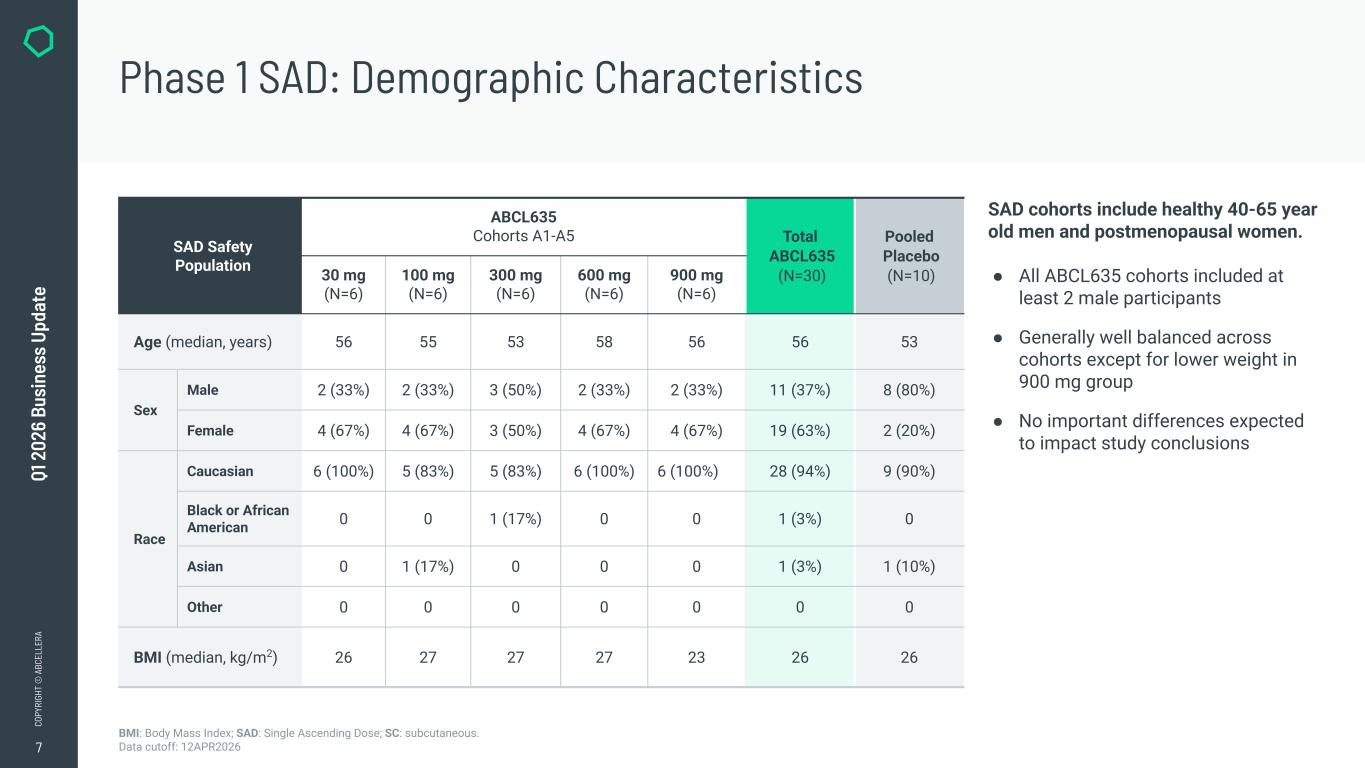
7 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Abbreviations: SAD=Single Ascending Dose, SC = subcutaneous, BMI = Body Mass Index Data extract: 17APR2026; Cut-off Date: 12APR2026 SAD Safety Population ABCL635 Cohorts A1-A5 Total ABCL635 (N=30) Pooled Placebo (N=10)30 mg (N=6) 100 mg (N=6) 300 mg (N=6) 600 mg (N=6) 900 mg (N=6) Age (median, years) 56 55 53 58 56 56 53 Sex Male 2 (33%) 2 (33%) 3 (50%) 2 (33%) 2 (33%) 11 (37%) 8 (80%) Female 4 (67%) 4 (67%) 3 (50%) 4 (67%) 4 (67%) 19 (63%) 2 (20%) Race Caucasian 6 (100%) 5 (83%) 5 (83%) 6 (100%) 6 (100%) 28 (94%) 9 (90%) Black or African American 0 0 1 (17%) 0 0 1 (3%) 0 Asian 0 1 (17%) 0 0 0 1 (3%) 1 (10%) Other 0 0 0 0 0 0 0 BMI (median, kg/m2) 26 27 27 27 23 26 26 BMI: Body Mass Index; SAD: Single Ascending Dose; SC: subcutaneous. Data cutoff: 12APR2026 Phase 1 SAD: Demographic Characteristics SAD cohorts include healthy 40-65 year old men and postmenopausal women. ● All ABCL635 cohorts included at least 2 male participants ● Generally well balanced across cohorts except for lower weight in 900 mg group ● No important differences expected to impact study conclusions
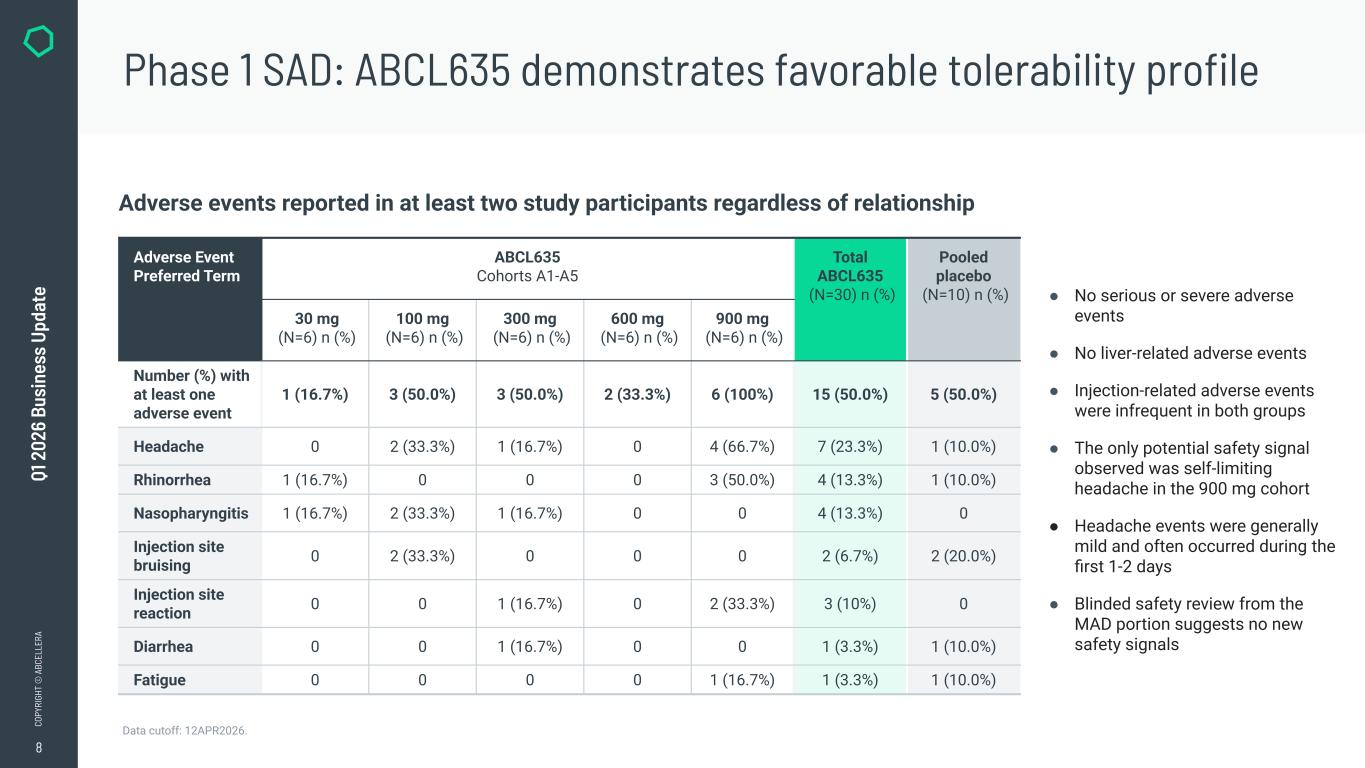
8 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e ● No serious or severe adverse events ● No liver-related adverse events ● Injection-related adverse events were infrequent in both groups ● The only potential safety signal observed was self-limiting headache in the 900 mg cohort ● Headache events were generally mild and often occurred during the first 1-2 days ● Blinded safety review from the MAD portion suggests no new safety signals Data cutoff: 12APR2026. Phase 1 SAD: ABCL635 demonstrates favorable tolerability profile Adverse events reported in at least two study participants regardless of relationship Adverse Event Preferred Term ABCL635 Cohorts A1-A5 Total ABCL635 (N=30) n (%) Pooled placebo (N=10) n (%) 30 mg (N=6) n (%) 100 mg (N=6) n (%) 300 mg (N=6) n (%) 600 mg (N=6) n (%) 900 mg (N=6) n (%) Number (%) with at least one adverse event 1 (16.7%) 3 (50.0%) 3 (50.0%) 2 (33.3%) 6 (100%) 15 (50.0%) 5 (50.0%) Headache 0 2 (33.3%) 1 (16.7%) 0 4 (66.7%) 7 (23.3%) 1 (10.0%) Rhinorrhea 1 (16.7%) 0 0 0 3 (50.0%) 4 (13.3%) 1 (10.0%) Nasopharyngitis 1 (16.7%) 2 (33.3%) 1 (16.7%) 0 0 4 (13.3%) 0 Injection site bruising 0 2 (33.3%) 0 0 0 2 (6.7%) 2 (20.0%) Injection site reaction 0 0 1 (16.7%) 0 2 (33.3%) 3 (10%) 0 Diarrhea 0 0 1 (16.7%) 0 0 1 (3.3%) 1 (10.0%) Fatigue 0 0 0 0 1 (16.7%) 1 (3.3%) 1 (10.0%)
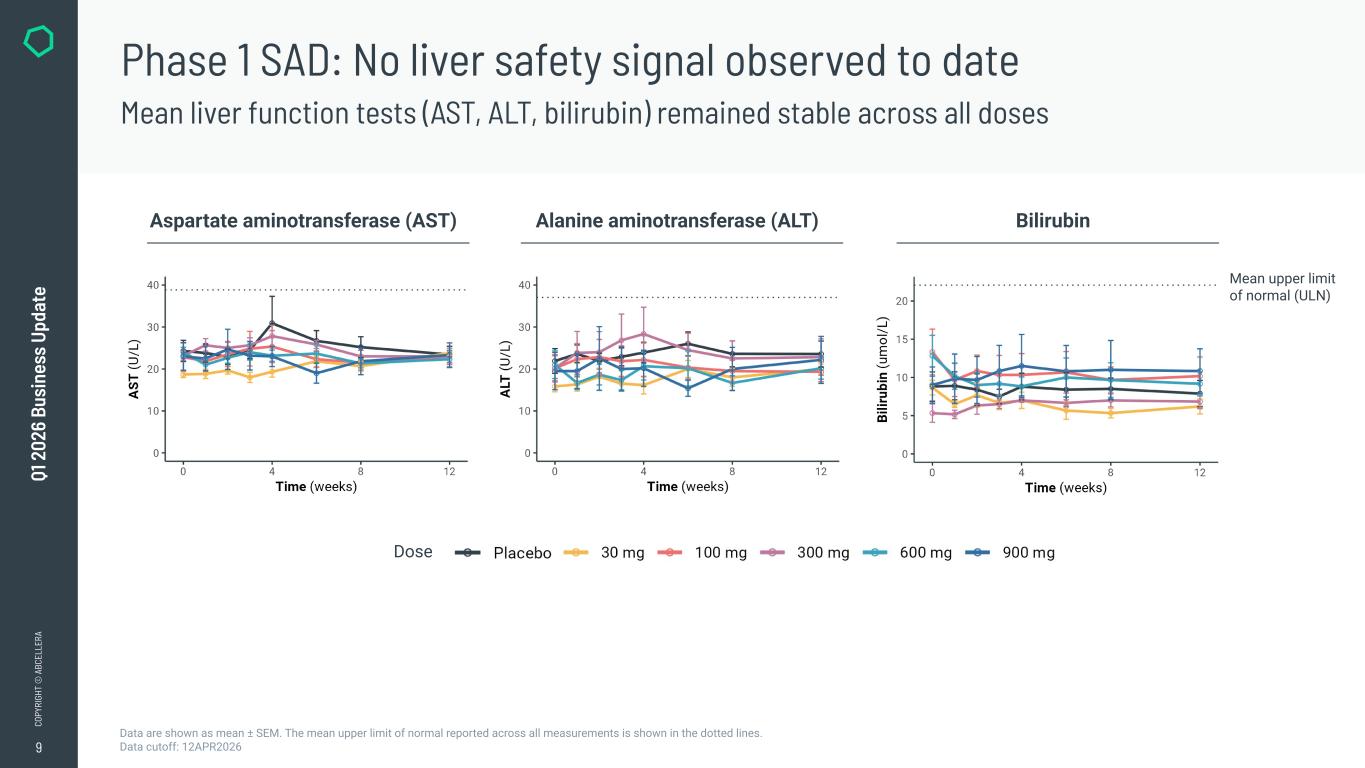
9 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Phase 1 SAD: No liver safety signal observed to date Mean liver function tests (AST, ALT, bilirubin) remained stable across all doses Data are shown as mean ± SEM. The mean upper limit of normal reported across all measurements is shown in the dotted lines. Data cutoff: 12APR2026 Aspartate aminotransferase (AST) Alanine aminotransferase (ALT) Bilirubin Dose Mean upper limit of normal (ULN)
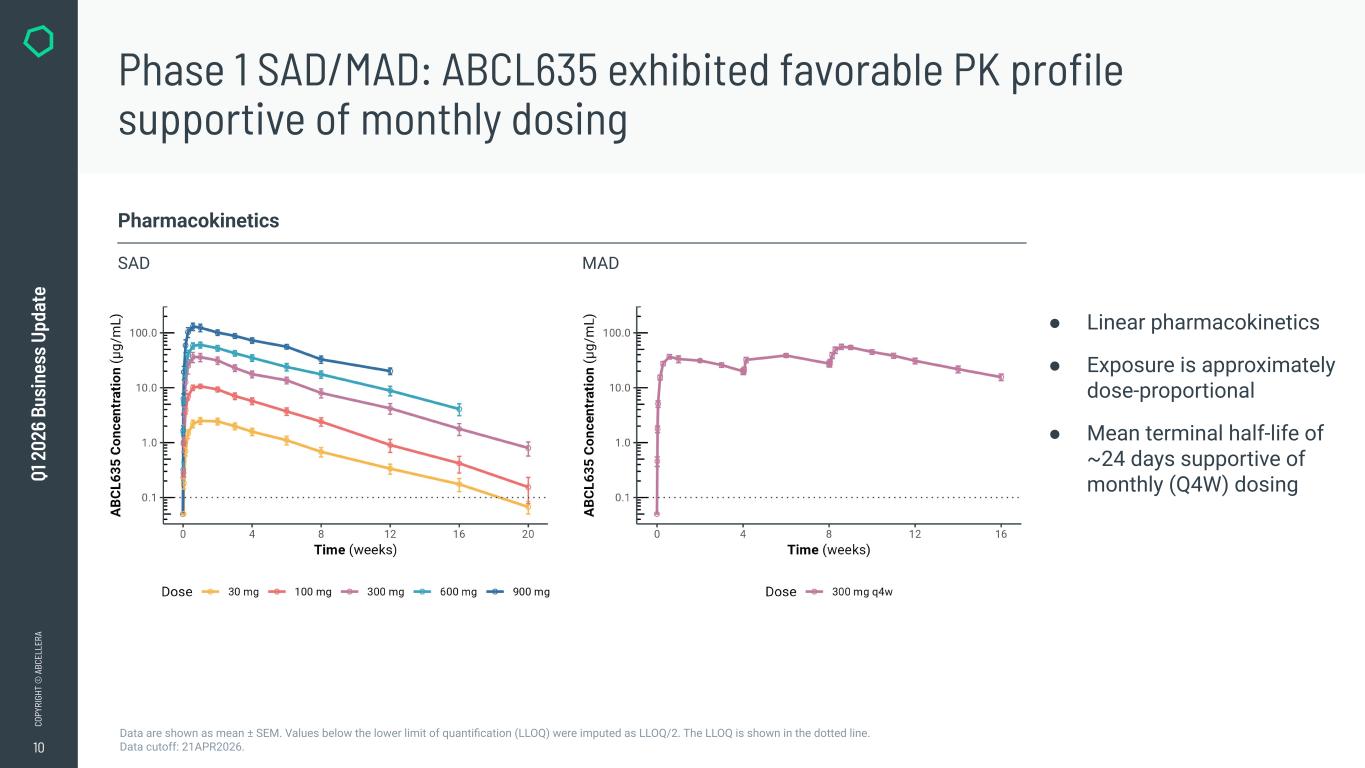
10 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Phase 1 SAD/MAD: ABCL635 exhibited favorable PK profile supportive of monthly dosing Data are shown as mean ± SEM. Values below the lower limit of quantification (LLOQ) were imputed as LLOQ/2. The LLOQ is shown in the dotted line. Data cutoff: 21APR2026. Pharmacokinetics SAD MAD ● Linear pharmacokinetics ● Exposure is approximately dose-proportional ● Mean terminal half-life of ~24 days supportive of monthly (Q4W) dosing
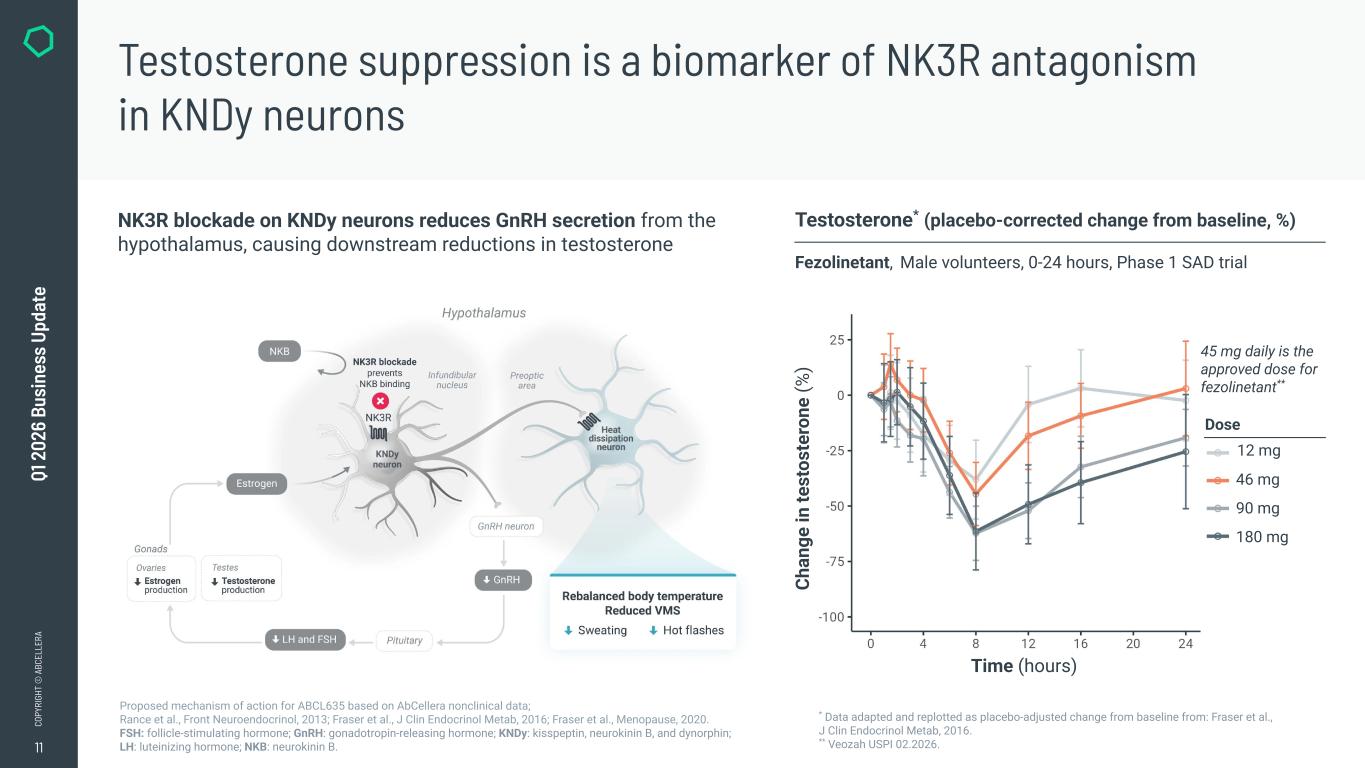
11 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Testosterone suppression is a biomarker of NK3R antagonism in KNDy neurons * Data adapted and replotted as placebo-adjusted change from baseline from: Fraser et al., J Clin Endocrinol Metab, 2016. ** Veozah USPI 02.2026. Testosterone* (placebo-corrected change from baseline, %) Fezolinetant, Male volunteers, 0-24 hours, Phase 1 SAD trial Proposed mechanism of action for ABCL635 based on AbCellera nonclinical data; Rance et al., Front Neuroendocrinol, 2013; Fraser et al., J Clin Endocrinol Metab, 2016; Fraser et al., Menopause, 2020. FSH: follicle-stimulating hormone; GnRH: gonadotropin-releasing hormone; KNDy: kisspeptin, neurokinin B, and dynorphin; LH: luteinizing hormone; NKB: neurokinin B. NK3R blockade on KNDy neurons reduces GnRH secretion from the hypothalamus, causing downstream reductions in testosterone Ch an ge in te st os te ro ne (% ) Time (hours) 45 mg daily is the approved dose for fezolinetant** Dose 12 mg 46 mg 90 mg 180 mg
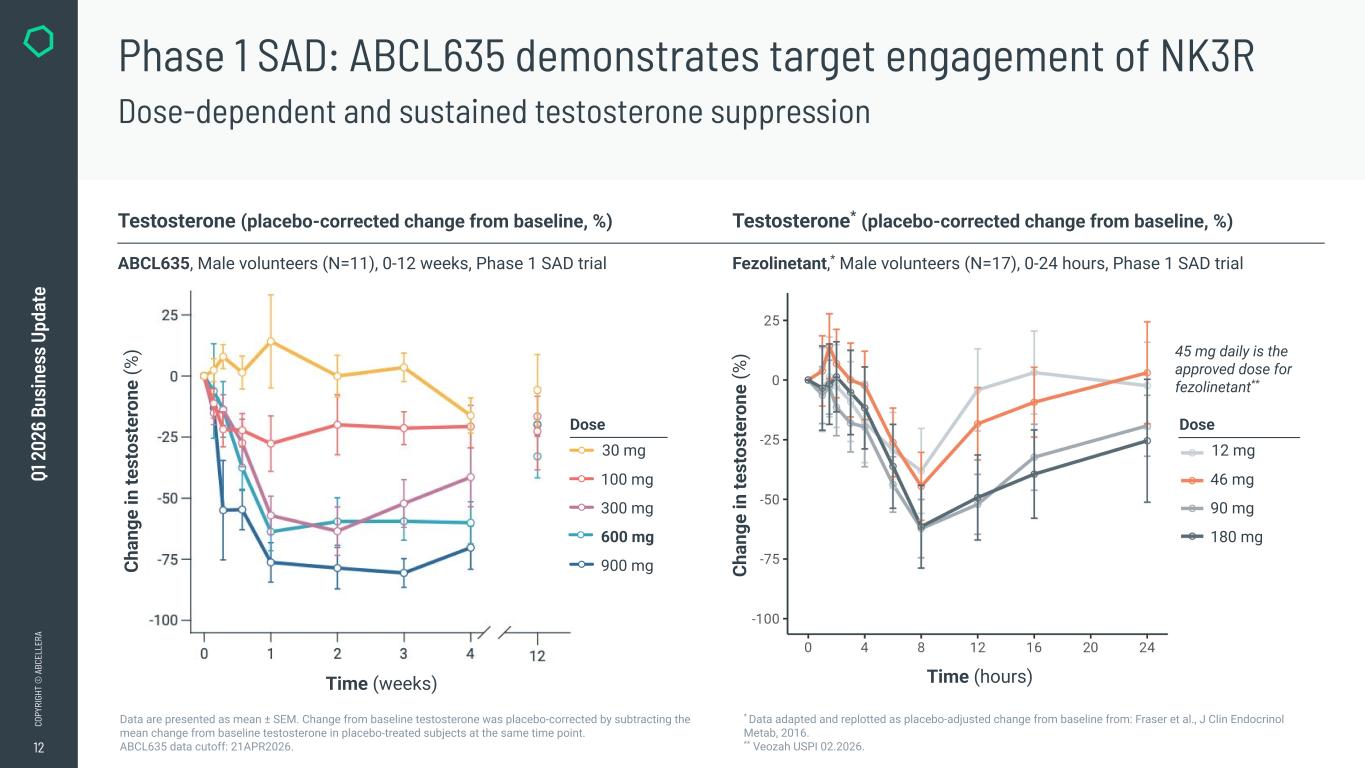
12 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Phase 1 SAD: ABCL635 demonstrates target engagement of NK3R Dose-dependent and sustained testosterone suppression Data are presented as mean ± SEM. Change from baseline testosterone was placebo-corrected by subtracting the mean change from baseline testosterone in placebo-treated subjects at the same time point. ABCL635 data cutoff: 21APR2026. Testosterone (placebo-corrected change from baseline, %) Testosterone* (placebo-corrected change from baseline, %) ABCL635, Male volunteers (N=11), 0-12 weeks, Phase 1 SAD trial Fezolinetant,* Male volunteers (N=17), 0-24 hours, Phase 1 SAD trial * Data adapted and replotted as placebo-adjusted change from baseline from: Fraser et al., J Clin Endocrinol Metab, 2016. ** Veozah USPI 02.2026. Time (weeks) Ch an ge in te st os te ro ne (% ) Time (hours) Ch an ge in te st os te ro ne (% ) 45 mg daily is the approved dose for fezolinetant** Dose 12 mg 46 mg 90 mg 180 mg Dose 30 mg 100 mg 300 mg 600 mg 900 mg
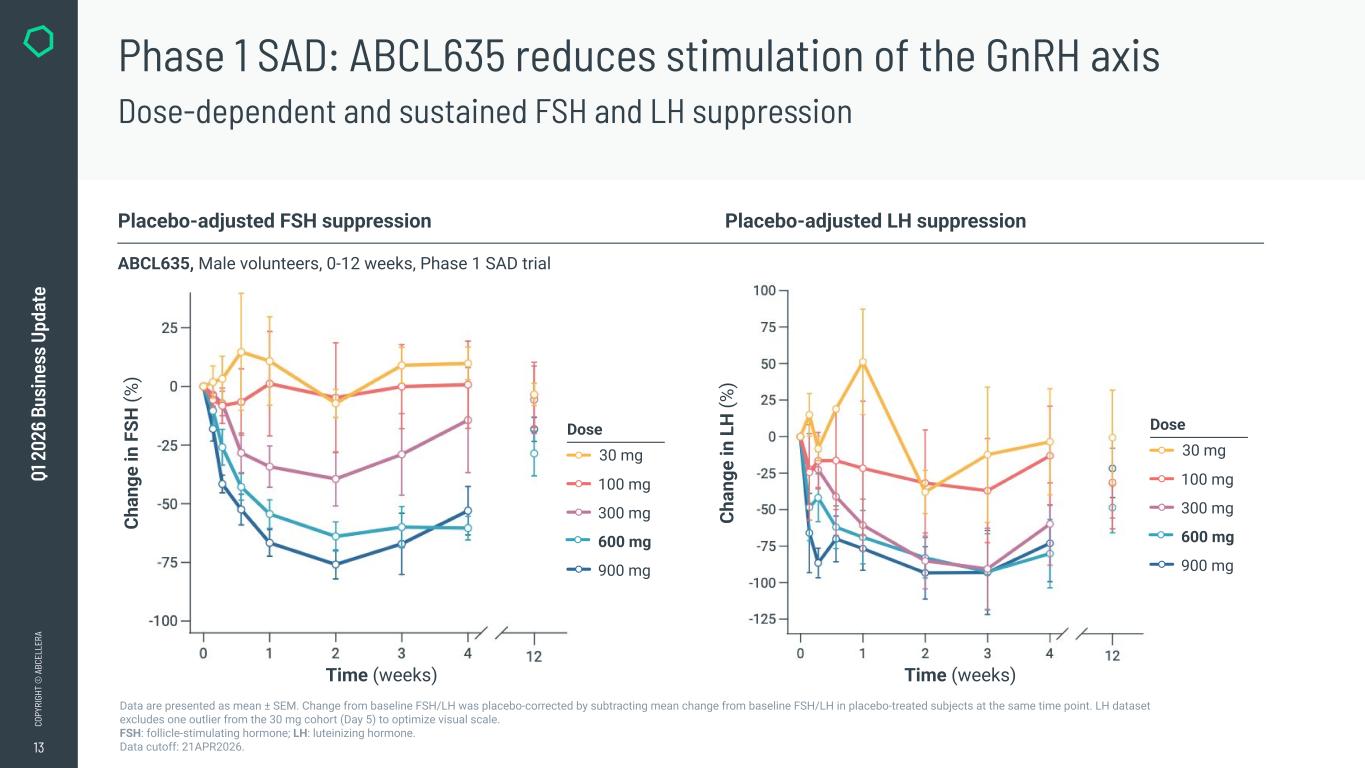
13 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Data are presented as mean ± SEM. Change from baseline FSH/LH was placebo-corrected by subtracting mean change from baseline FSH/LH in placebo-treated subjects at the same time point. LH dataset excludes one outlier from the 30 mg cohort (Day 5) to optimize visual scale. FSH: follicle-stimulating hormone; LH: luteinizing hormone. Data cutoff: 21APR2026. Placebo-adjusted FSH suppression Placebo-adjusted LH suppression ABCL635, Male volunteers, 0-12 weeks, Phase 1 SAD trial Phase 1 SAD: ABCL635 reduces stimulation of the GnRH axis Dose-dependent and sustained FSH and LH suppression Ch an ge in F SH (% ) Ch an ge in L H (% ) Time (weeks) Time (weeks) Dose 30 mg 100 mg 300 mg 600 mg 900 mg Dose 30 mg 100 mg 300 mg 600 mg 900 mg

14 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e ABCL635 Phase 1 data supported advancing to Phase 2 Favorable tolerability profile with no observed liver toxicity PK profile supports once monthly subcutaneous dosing Biomarker data suggests strong and sustained engagement of NK3R Demonstrated suppression of testosterone, LH, and FSH All doses tested generally well tolerated Estimated half-life of ~24 days ABCL635 PHASE 2 DOSE SELECTION A 600 mg SC dose administered once best approximates 300 mg SC at steady state* and is being evaluated in the Phase 2 study SC: subcutaneous. * Based on a comparison of predicted Cmax at steady state of a 300 mg dose once every 4 weeks, vs. the Cmax from a single dose of 600 mg. SAFETY/TOLERABILITY ENDPOINT PHARMACOKINETIC ENDPOINT PHARMACODYNAMIC ENDPOINT

15 CO PY RI GH T © A BC EL LE RA Phase 2 Study Design ● Randomized, double-blind, placebo-controlled, multicenter trial in postmenopausal women with moderate-to-severe VMS, 40-75 years old ● Single dose of ABCL635 600 mg SC or matched placebo ● Open-Label Extension is open to all Phase 2 participants after week 12 Q1 2 02 6 Bu si ne ss U pd at e ABCL635 Phase 2 currently enrolling with topline results expected in Q3 2026 Topline Data in Q3 ● Frequency of VMS at week 4 ● Severity of VMS at week 4 ● Safety and tolerability through week 4 ● Additional study data to follow at future medical conferences SC: subcutaneous. PART C: Proof of Concept SC: subcutaneous OLE: Open Label Extension C1 600 mg SC C2 600 mg SC OLE Placebo-controlled treatment period ABCL635 600 mg SC (N=40) Placebo (N=40) Open-Label Extension 600 mg SC Optional open-label extension (OLE) Sc re en in g N = 80 Key inclusion criteria: Comparable to Phase 3 entry criteria for approved NK3R small molecules R

16 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Q1 Q2 Q3 Q4 Two readouts in 2026 & potential for multiple catalysts in 2027 2026 ABCL635 Menopausal VMS ABCL635 VMS in Oncology ABCL575 Inflammation & Autoimmunity ABCL386 Oncology ABCL688 Autoimmunity New Development Candidate Development candidate selection Phase 1/2 Phase 1 Additionally, 20+ discovery programs in the pipeline anticipated to produce 1-2 development candidates per year IND-enabling IND-enabling 2027 Late stage development of ABCL635 in menopausal VMS Potential Catalysts in 2027 Initiation of Phase 2 studies of ABCL635 in oncology VMS Options for further development or out-licensing of ABCL575 IND submissions and initiation of Phase 1/2 study in patients for ABCL688, ABCL386; and selection of a new development candidate Readout Readout

17 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Q1 2026 FINANCIALS UPDATE

18 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e We continue to maintain a strong liquidity position to execute on our strategy. * As of March 31, 2026 in total available government funding*~$125M in total cash, cash equivalents, & marketable securities*~$530M in available liquidity to execute on our strategy*~$655M
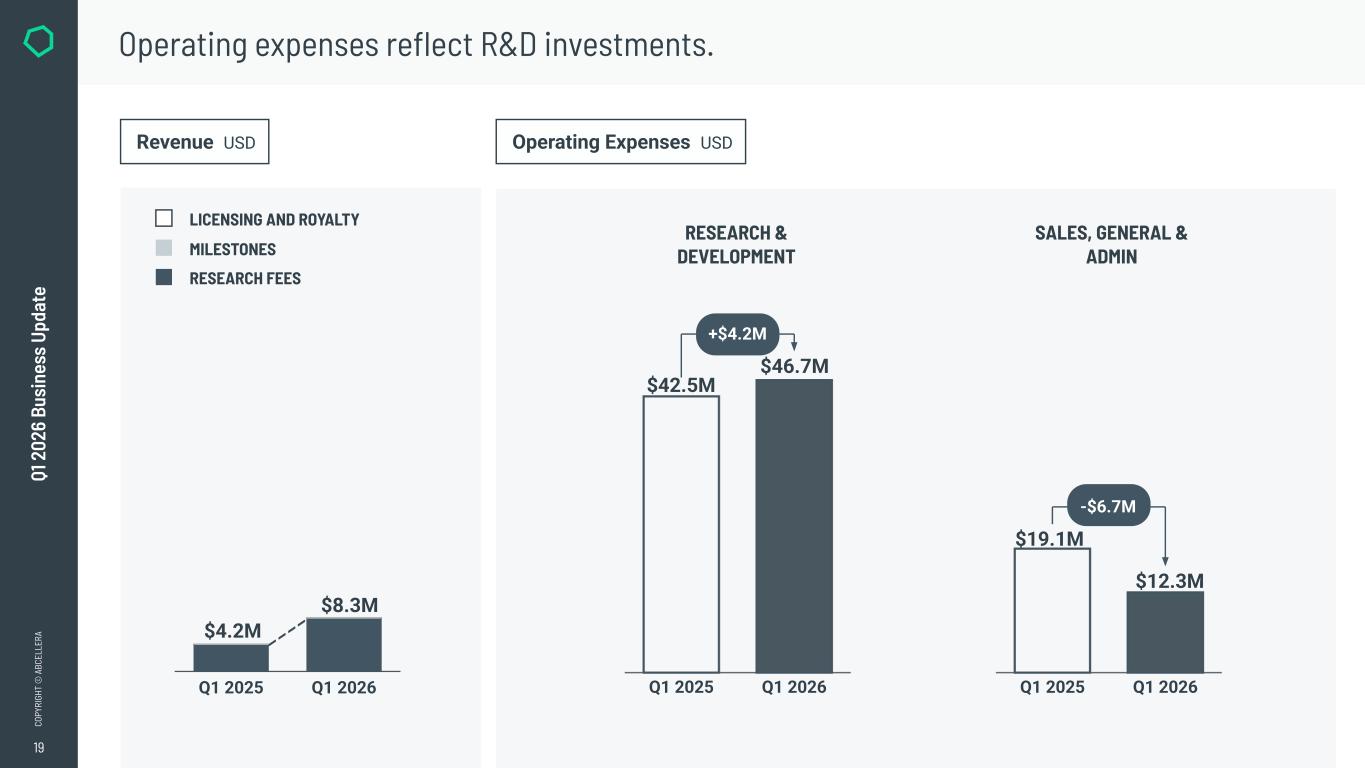
19 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Q1 2025 Q1 2026 $42.5M $46.7M +$4.2M Q1 2025 Q1 2026 $19.1M $12.3M -$6.7M RESEARCH & DEVELOPMENT SALES, GENERAL & ADMIN Operating expenses reflect R&D investments. Operating Expenses USDRevenue USD MILESTONES RESEARCH FEES LICENSING AND ROYALTY Q1 2025 Q1 2026 $4.2M $8.3M
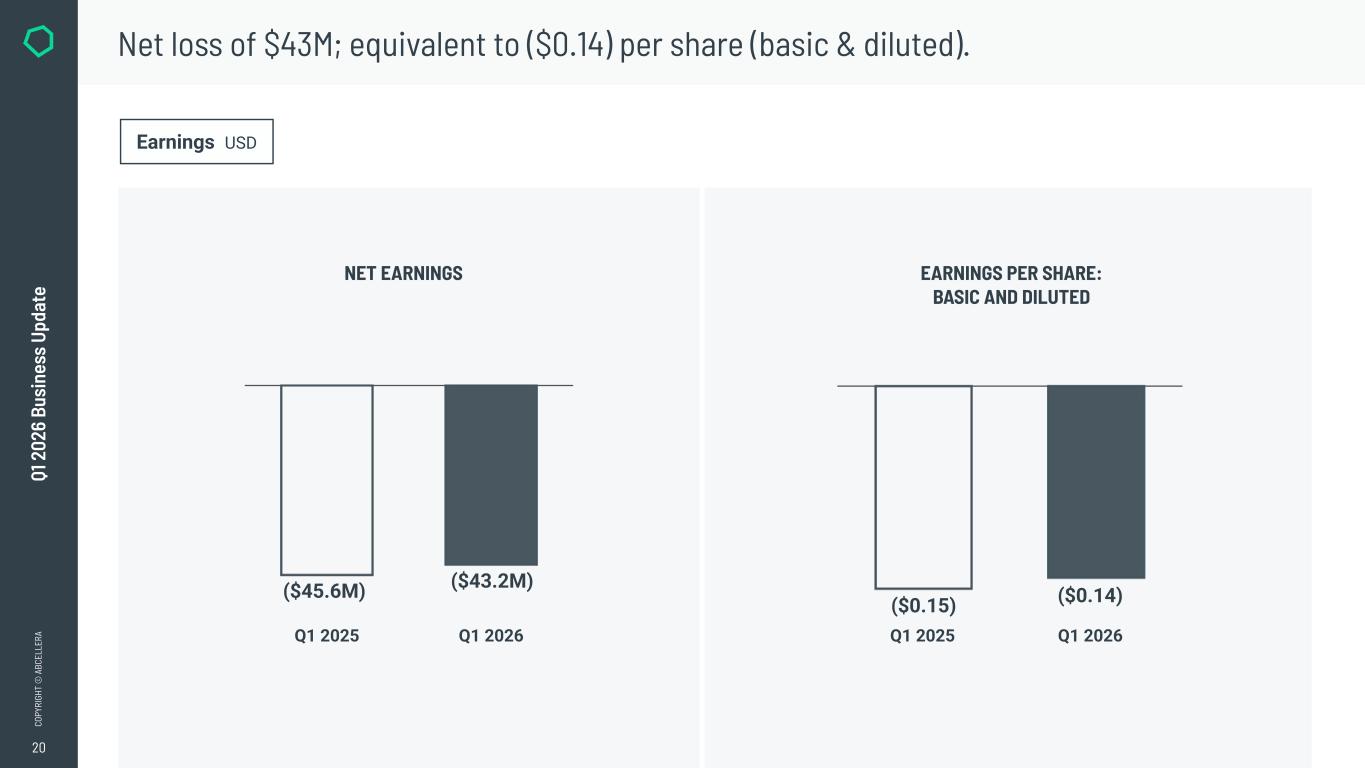
20 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Net loss of $43M; equivalent to ($0.14) per share (basic & diluted). Earnings USD NET EARNINGS EARNINGS PER SHARE: BASIC AND DILUTED Q1 2025 Q1 2026 ($45.6M) ($43.2M) Q1 2025 Q1 2026 ($0.15) ($0.14)
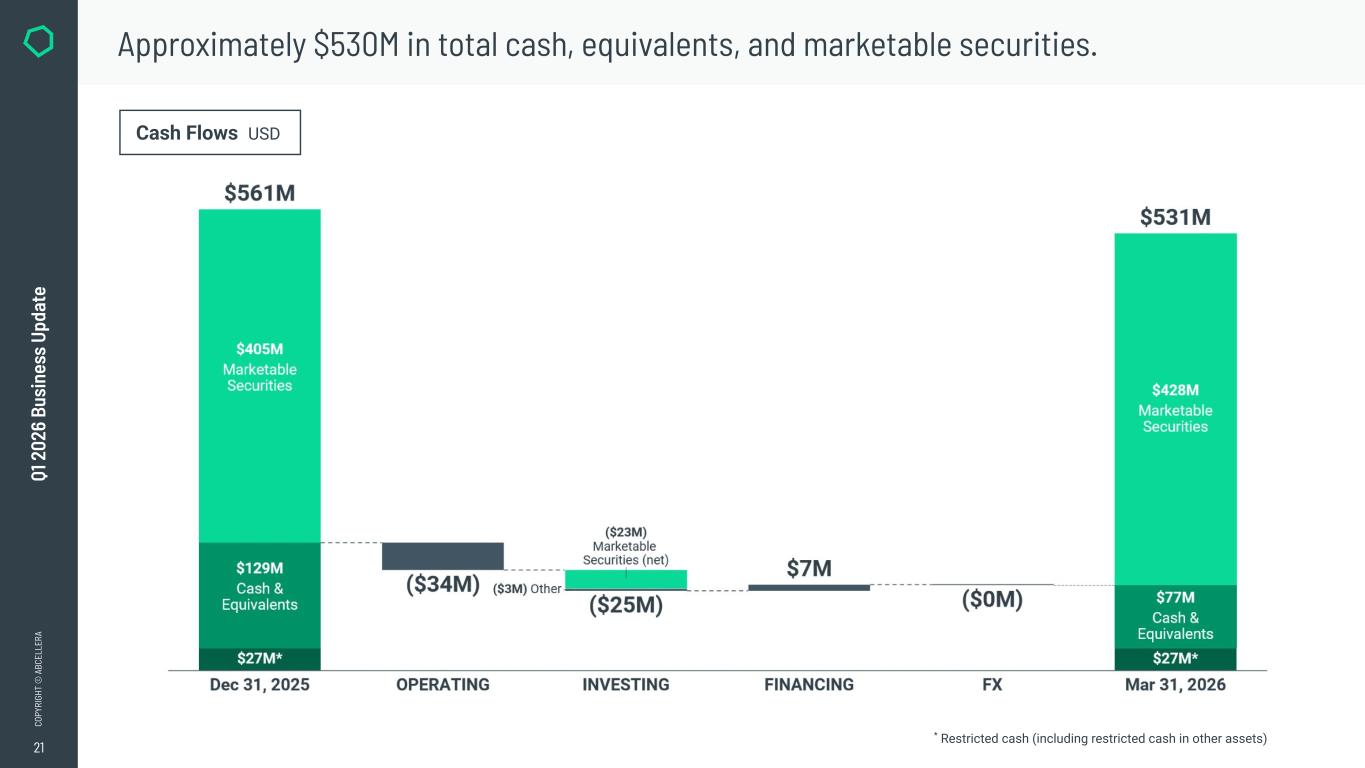
21 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e Approximately $530M in total cash, equivalents, and marketable securities. Cash Flows USD * Restricted cash (including restricted cash in other assets)

22 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e THANK YOU

23 CO PY RI GH T © A BC EL LE RA CO NF ID EN TI AL Q1 2 02 6 Bu si ne ss U pd at e APPENDIX
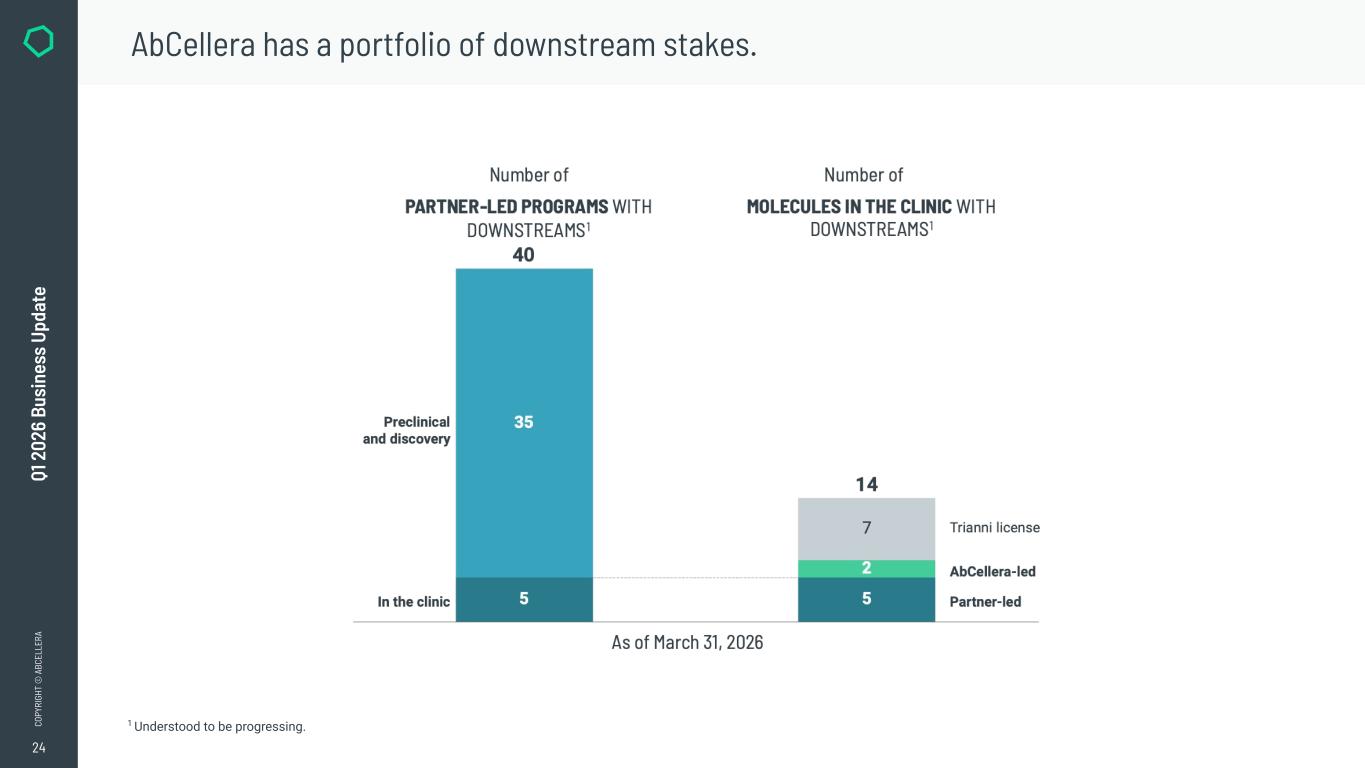
24 CO PY RI GH T © A BC EL LE RA Q1 2 02 6 Bu si ne ss U pd at e AbCellera has a portfolio of downstream stakes. 1 Understood to be progressing.