

Q2 Company Presentation May 2026 Exhibit 99.2

Disclaimer Any statements made in this presentation that are not statements of historical fact, including statements about our beliefs and expectations, are forward-looking statements and should be evaluated as such. Forward-looking statements include information concerning the strategy, initiation, cost, timing, progress, and results of our preclinical studies and clinical trials for our product candidates; our ability to leverage our technology platform to enable more informed drug research and development; estimates of the number of patients with certain diseases and conditions we intend to treat, the number of patients that we plan to enroll in our clinical trials, and the size and nature of the market opportunity for our product candidates, expected milestones and business objectives for 2026 and beyond, including our anticipated presentations at the 2026 annual meeting of the American Society of Clinical Oncology ("ASCO"); and our ability to maintain our current license agreements and collaborations, including our ability to comply with our financial obligations pursuant to the terms of such agreements, and our ability to identify and enter into future license agreements and collaborations. These statements often include words such as "anticipate," "expect," "suggests," "plan," "believe," "intend," "estimates," "targets," "projects," "should," "could," "would," "may," "will," "forecast" and other similar expressions. These forward-looking statements are contained throughout this presentation. We have based these forward-looking statements on our current expectations, plans and assumptions that we have made in light of our experience in the industry, as well as our perceptions of historical trends, current conditions, expected future developments and other factors we believe are appropriate under the circumstances at such time. As you read and consider this presentation, you should understand that these statements are not guarantees of future performance or results. The forward-looking statements are subject to and involve risks, uncertainties and assumptions, and you should not place undue reliance on these forward-looking statements. Although we believe that these forward-looking statements are based on reasonable assumptions at the time they are made, you should be aware that many factors could affect our actual results or results of operations and could cause actual results to differ materially from those expressed in the forward-looking statements. Factors that may materially affect such forward-looking statements include: our limited operating history; our significant net losses incurred since inception and the likelihood of incurring additional losses for the foreseeable future; our need for substantial additional funding; the early stage of development of our product candidates and any future product candidates and the possibility they may fail in development; our dependence on the success of our current product candidates; legal and regulatory risks; intellectual property-related risks; and the other important factors described in the Annual Report on Form 10-K that we have filed with the Securities and Exchange Commission. These cautionary statements should not be construed by you to be exhaustive and are made only as of the date of this presentation. We undertake no obligation to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise, except as required by applicable law. You should, therefore, not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this presentation. Certain information contained in this presentation and statements made orally during this presentation relate to or are based on studies, publications and other data obtained from third-party sources as well as our own internal estimates and research. While we believe these third-party sources to be reliable as of the date of this presentation, we have not independently verified, and make no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third party sources. In addition, estimates involve a number of assumptions and limitations and you are cautioned not to give undue weight to such estimates. This presentation contains references to our trademarks and service marks and to those belonging to other entities. Solely for convenience, trademarks and trade names referred to in this presentation may appear without the® or TM symbols, but such references are not intended to indicate in any way that we will not assert, to the fullest extent under applicable law, our rights or the rights of the applicable licensor to these trademarks and tradenames. We do not intend our use or display of other entities' trade names, trademarks or service marks to imply a relationship with, or endorsement or sponsorship of us by, any other entity.
Advancing breakthrough therapeutics through the purposeful integration of science and engineering © 2026 | 3 Innovative Discovery Machine Learning & Advanced Engineering Experienced Leadership Clinical-Stage Pipeline Eikon is a late-stage clinical biopharmaceutical company

Eikon’s leadership team delivers proven depth and experience to bring novel therapies to market Roger Perlmutter CEO & Board Chair Roy D. Baynes CMO Freddie Bowie CFO Barbara Howes CPO Benjamin Thorner CBO & GC Russ Berman CTO Michael Klobuchar COO Executive Team Board of Directors Kenneth C. Frazier Former CEO, Merck; current Chairman, General Catalyst’s Health Assurance initiatives Leon Chen, Ph. D. Partner, The Column Group; Venture Partner, OrbiMed Advisors Robbie Huffines Former Global Chairman of Investment Banking, JP Morgan Joshua Wolfe Co-Founder & Managing Partner, Lux Capital David W. Meline Former CFO, Moderna; Former CFO, Amgen; Former CFO, 3M
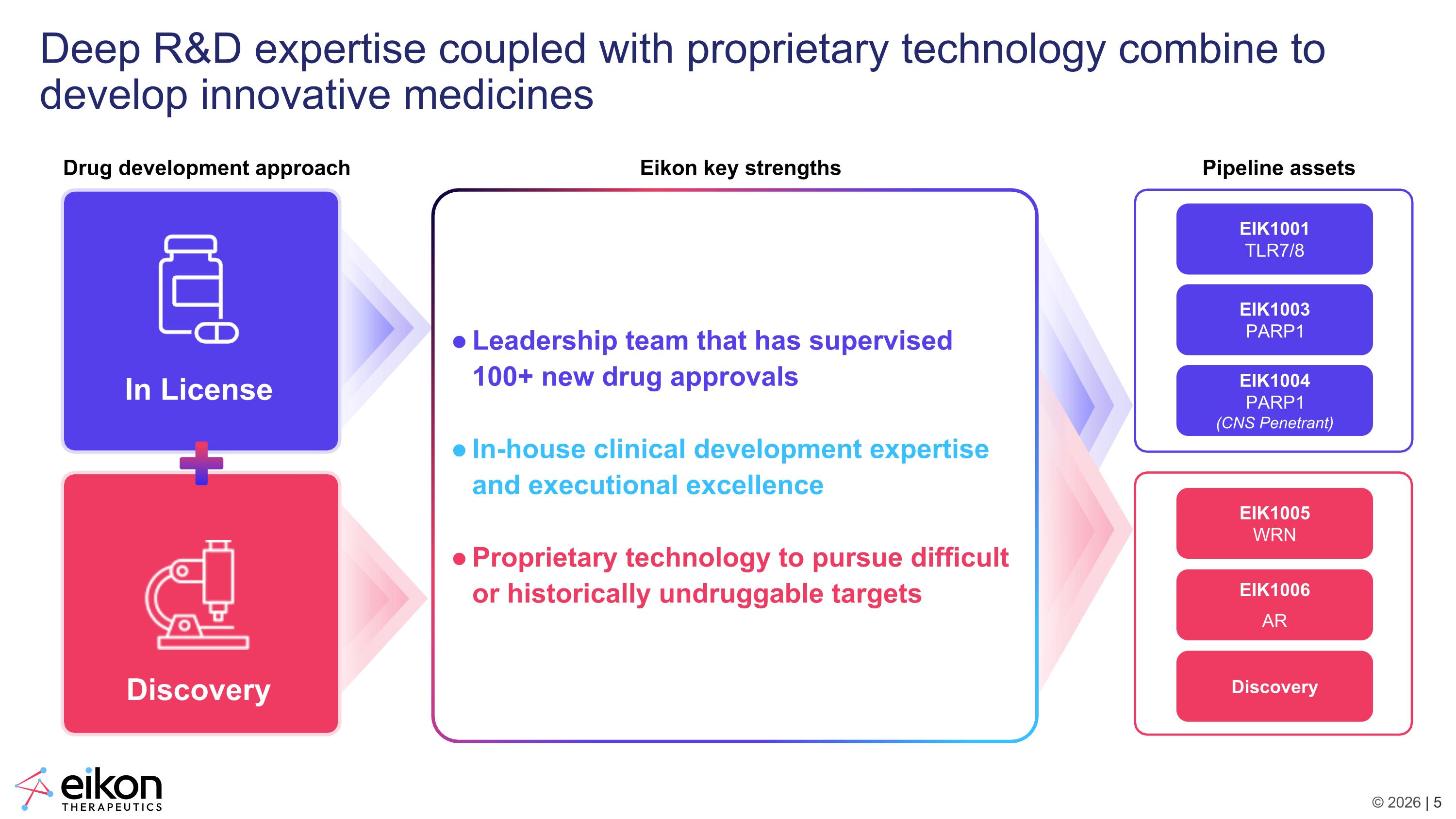
Deep R&D expertise coupled with proprietary technology combine to develop innovative medicines Eikon key strengths Leadership team that has supervised 100+ new drug approvals In-house clinical development expertise and executional excellence Proprietary technology to pursue difficult or historically undruggable targets Drug development approach EIK1005 WRN EIK1003 PARP1 EIK1001 TLR7/8 EIK1004 PARP1 (CNS Penetrant) EIK1006 AR Discovery Pipeline assets In License Discovery

1This Phase 2/3 trial is designed to proceed to completion, subject to interim analysis by a data monitoring committee, and to form the basis for registration; 2 Phase 2 safety and efficacy study nearing completion; United States Food and Drug Administration, or FDA, has allowed us to proceed with the Phase 2/3 registrational trial; 3 NSQ = Non-squamous cohort, SQ = Squamous cohort, Phase 2 readout for NSQ cohort expected to be presented at ASCO 2026; 4 Greater China: China, Hong Kong, Macau, Taiwan; 5Milestone achieved February, 2026; Note: Readouts are contingent on acceptance for presentation at one of several major medical conferences Eikon’s pipeline includes multiple indications addressing significant unmet medical needs We are also actively pursuing additional discovery research in oncology and neurodegeneration Melanoma1 NSCLC2 EIK1005 WRN EIK1003 PARP1 EIK1001 TLR7/8 Candidate/ Program Pre-Clinical Phase 1 Phase 2 Phase 3 Target Indication(s) Ownership Next Anticipated Milestone EIK1004 PARP1 (CNS Penetrant) Wholly owned Ovarian, Breast, Prostate, Pancreatic Solid Tumors with and without Brain Metastases MSI-high Tumors Global exclusive license excluding Greater China4 Global exclusive license excluding Greater China4 Target EIK1006 AR Wholly owned Prostate Cancer Phase 2/3 First Patient Dosed, 2H 2026 First Interim Analysis, 2H 2026 Phase 1 First Patient Dosed, Q1 20265 IND Submission, End of 2026 1A and 1C Data Readouts, 2H 2026 Completion of Phase 1/2 Dose Escalation, 2H 2026 Phase 2 Data (NSQ SQ)3 Readout, 2H 2026 Global exclusive license Data to be presented at ASCO 2026 ✽ ✽ ✽
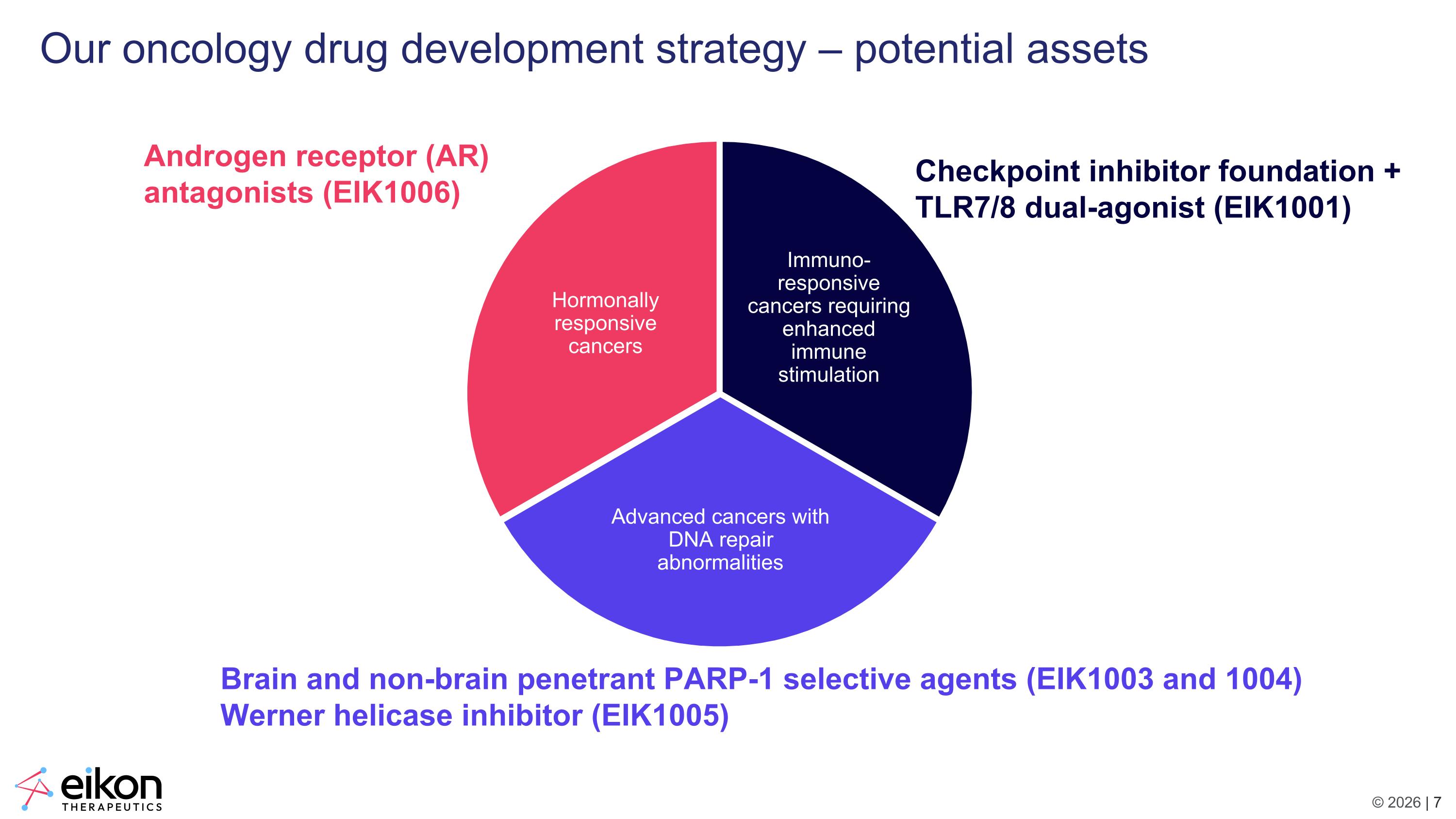
Our oncology drug development strategy – potential assets Immuno-responsive cancers requiring enhanced immune stimulation Advanced cancers with DNA repair abnormalities Hormonally responsive cancers Androgen receptor (AR) antagonists (EIK1006) Checkpoint inhibitor foundation + TLR7/8 dual-agonist (EIK1001) Brain and non-brain penetrant PARP-1 selective agents (EIK1003 and 1004) Werner helicase inhibitor (EIK1005)

EIK1001 – TLR7/8 Dual-Agonist
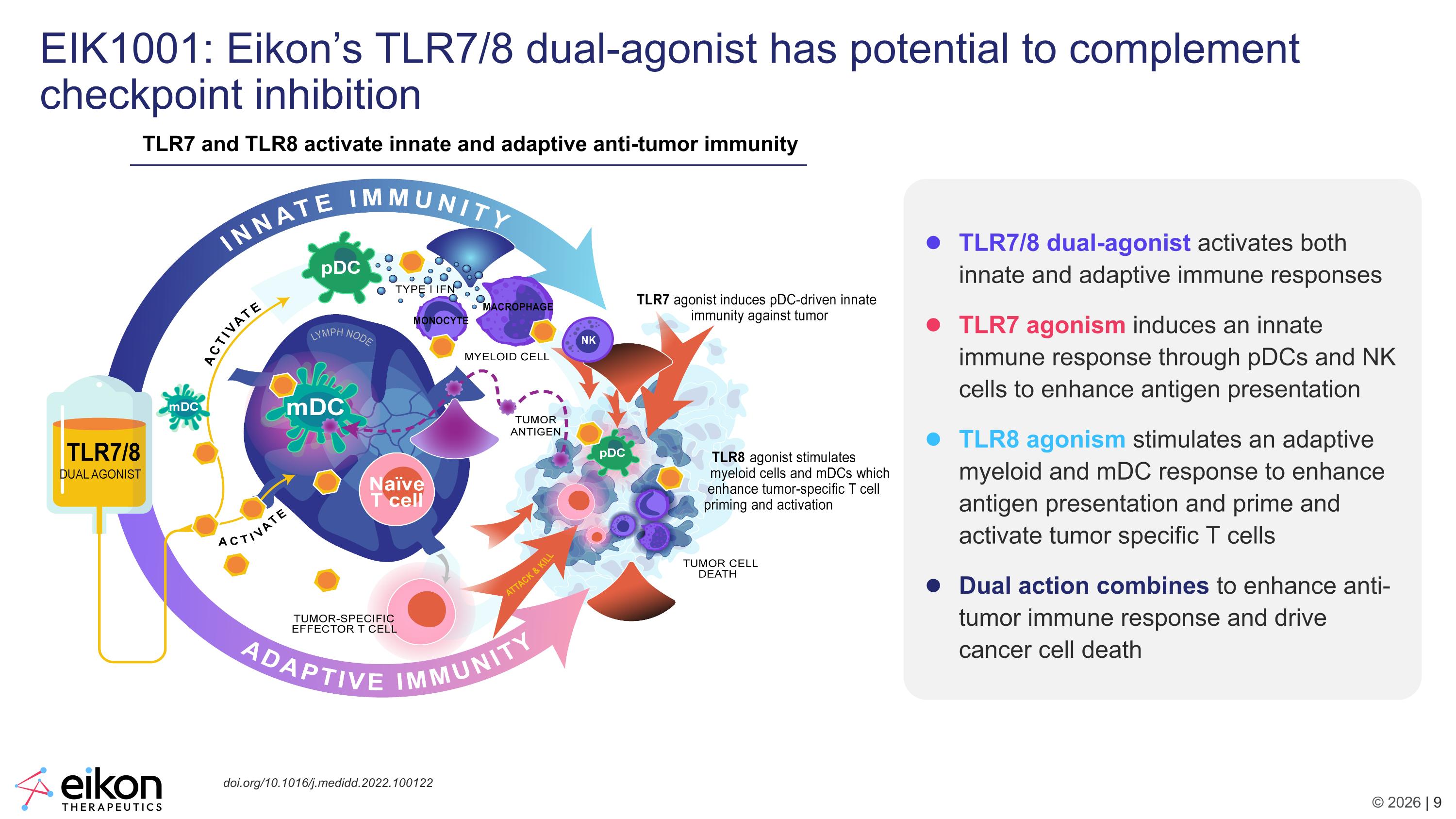
EIK1001: Eikon’s TLR7/8 dual-agonist has potential to complement checkpoint inhibition TLR7 and TLR8 activate innate and adaptive anti-tumor immunity TLR7/8 dual-agonist activates both innate and adaptive immune responses TLR7 agonism induces an innate immune response through pDCs and NK cells to enhance antigen presentation TLR8 agonism stimulates an adaptive myeloid and mDC response to enhance antigen presentation and prime and activate tumor specific T cells Dual action combines to enhance anti-tumor immune response and drive cancer cell death doi.org/10.1016/j.medidd.2022.100122

EIK1001 introduces a systemic proinflammatory signal which may complement traditional checkpoint inhibitor-based regimens Dual agonism stimulates innate and adaptive responses Orthogonal mechanism of action to checkpoint inhibitors Cutaneous administration of resiquimod (EIK1001) has been shown to provoke responses in cutaneous T-cell lymphoma and basal cell carcinoma1 Monotherapy activity observed in Phase 1 study Observed to be generally well-tolerated when dosed systemically in Eikon’s large (>400) patient-exposure database Differentiation of EIK1001 EIK1001 is designed to be an Active Anti-Neoplastic Agent EIK1001 TLR7/8 Field 1doi: 10.1182/blood-2015-02-630335; doi: 10.3390/pharmaceutics14102076

EIK1001 development strategy maximizes potential impact across significant indications Currently conducting three clinical trials: NSCLC Phase 2 exploratory/tolerability study of triplet (chemo, EIK1001, pembrolizumab), initiated in Q1 2024 Advanced Melanoma Phase 2/3 registrational trial in combination with pembrolizumab, initiated in Q2 2025 NSCLC Phase 2/3 registrational trial (chemo, EIK1001, pembrolizumab), initiated in Q1 2026 Potential to explore additional indications: Highly immunogenic (hot tumors where immune therapy has been tremendously successful) Moderately and low immunogenic (broader populations where current immune therapies leave room for improvement or lack activity altogether)
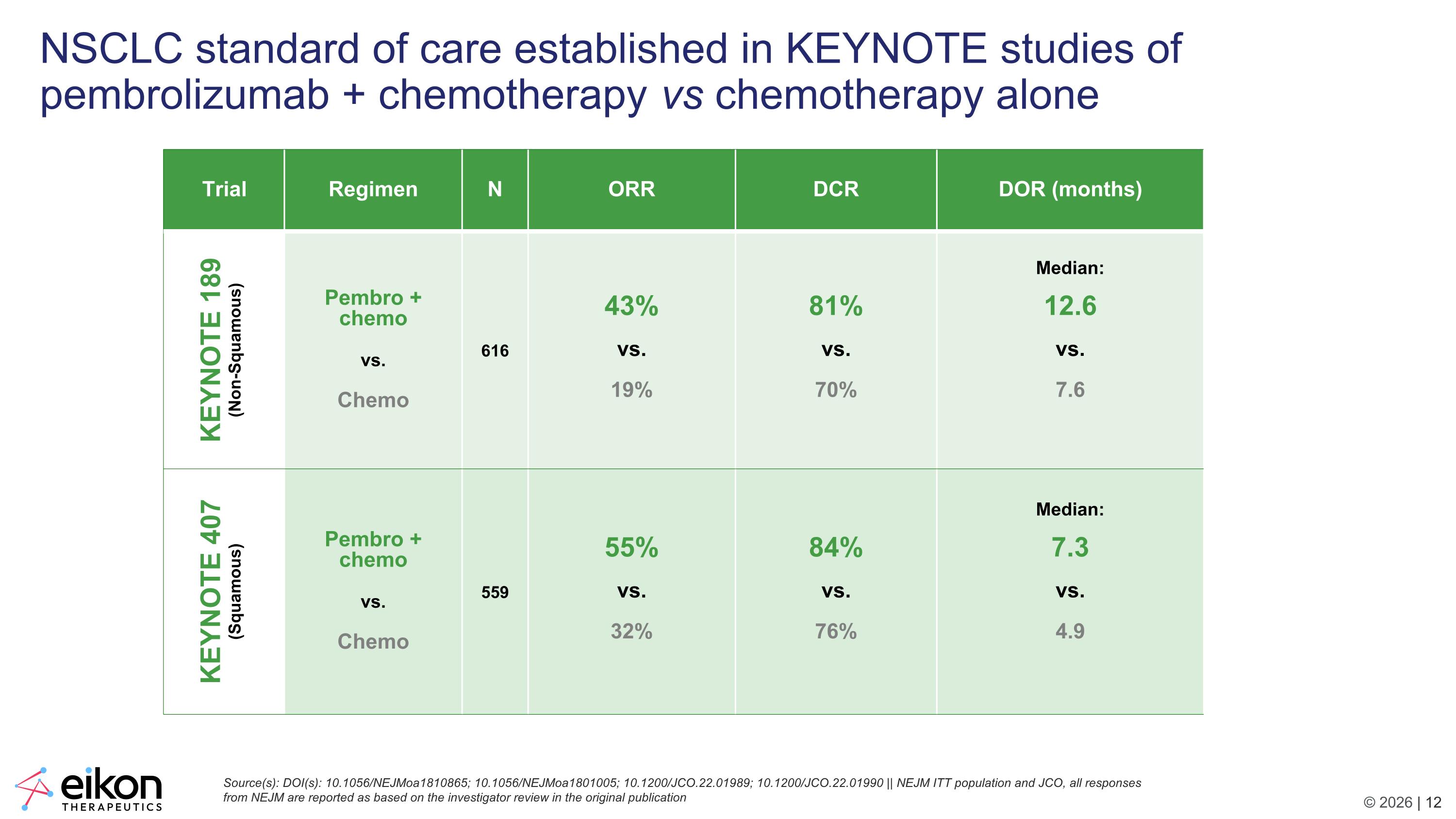
NSCLC standard of care established in KEYNOTE studies of pembrolizumab + chemotherapy vs chemotherapy alone Source(s): DOI(s): 10.1056/NEJMoa1810865; 10.1056/NEJMoa1801005; 10.1200/JCO.22.01989; 10.1200/JCO.22.01990 || NEJM ITT population and JCO, all responses from NEJM are reported as based on the investigator review in the original publication Trial Regimen N ORR DCR DOR (months) KEYNOTE 189 (Non-Squamous) Pembro + chemo vs. Chemo 616 43% vs. 19% 81% vs. 70% Median: 12.6 vs. 7.6 KEYNOTE 407 (Squamous) Pembro + chemo vs. Chemo 559 55% vs. 32% 84% vs. 76% Median: 7.3 vs. 4.9
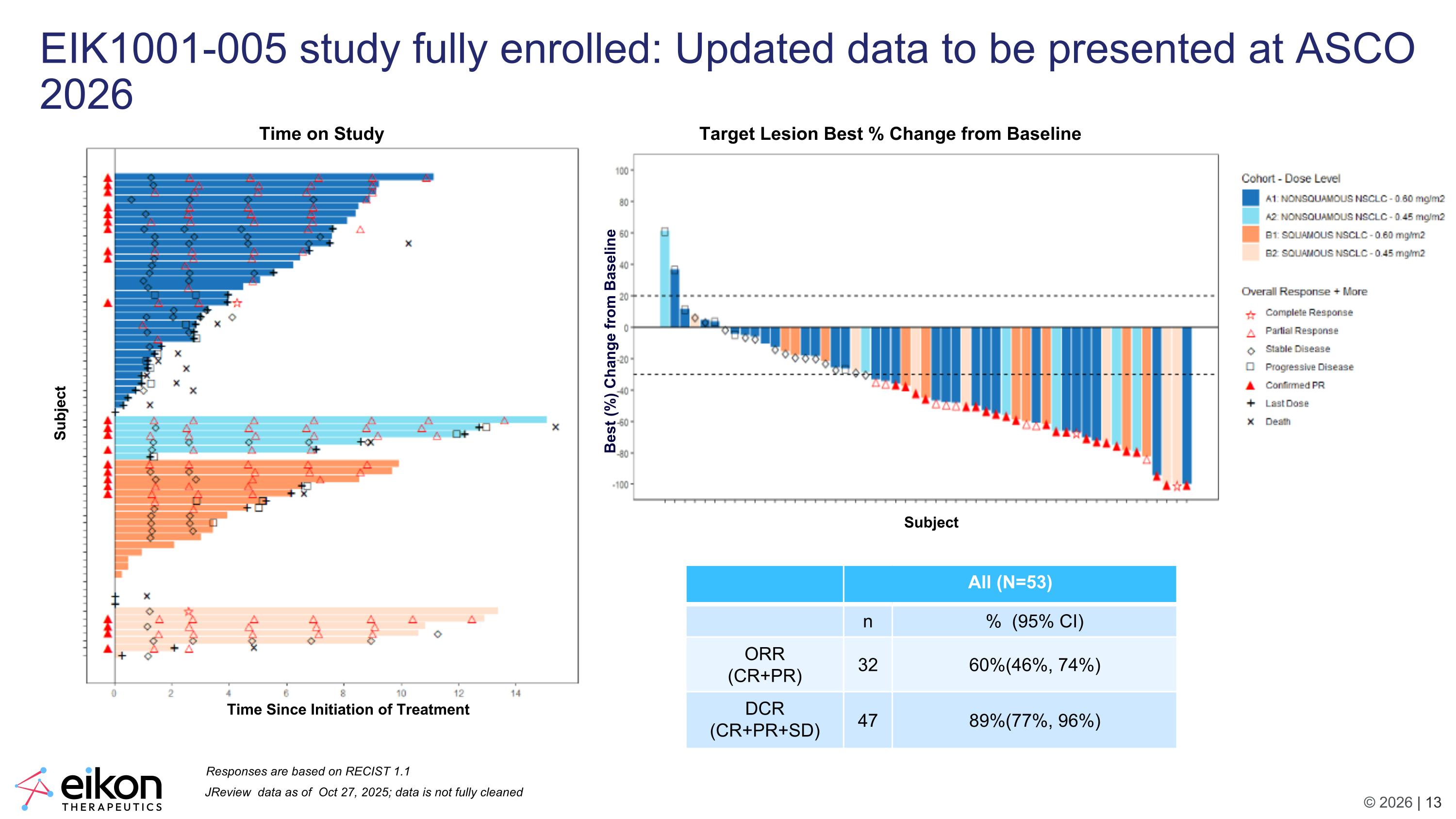
JReview data as of Oct 27, 2025; data is not fully cleaned All (N=53) n % (95% CI) ORR (CR+PR) 32 60%(46%, 74%) DCR (CR+PR+SD) 47 89%(77%, 96%) Responses are based on RECIST 1.1 Target Lesion Best % Change from Baseline Time on Study EIK1001-005 study fully enrolled: Updated data to be presented at ASCO 2026 Subject Subject Time Since Initiation of Treatment Best (%) Change from Baseline
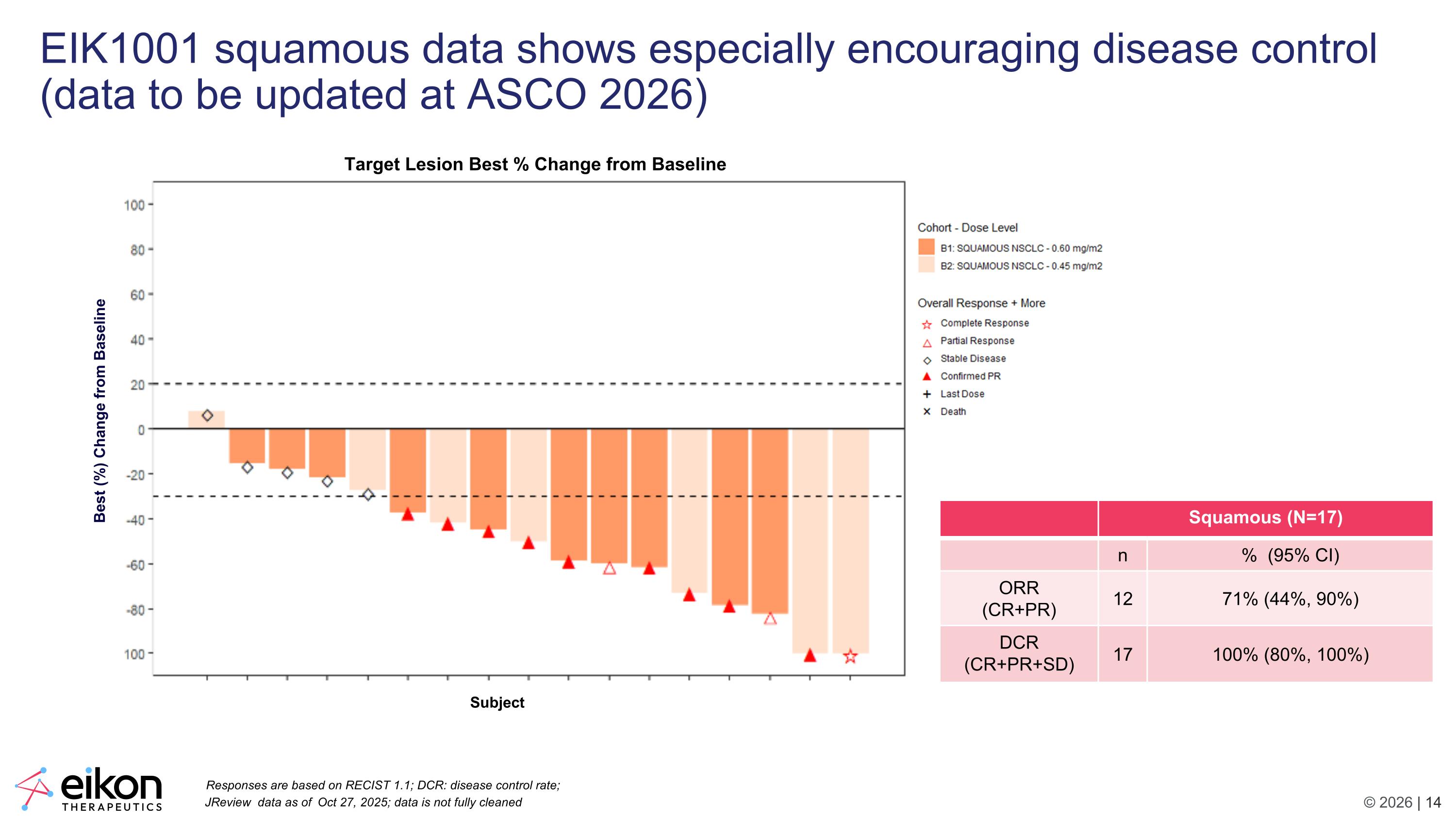
EIK1001 squamous data shows especially encouraging disease control (data to be updated at ASCO 2026) Squamous (N=17) n % (95% CI) ORR (CR+PR) 12 71% (44%, 90%) DCR (CR+PR+SD) 17 100% (80%, 100%) Subject Best (%) Change from Baseline Target Lesion Best % Change from Baseline JReview data as of Oct 27, 2025; data is not fully cleaned Responses are based on RECIST 1.1; DCR: disease control rate;

EIK1003/EIK1004: PARP1-Selective Inhibitors for Treatment of Malignancy
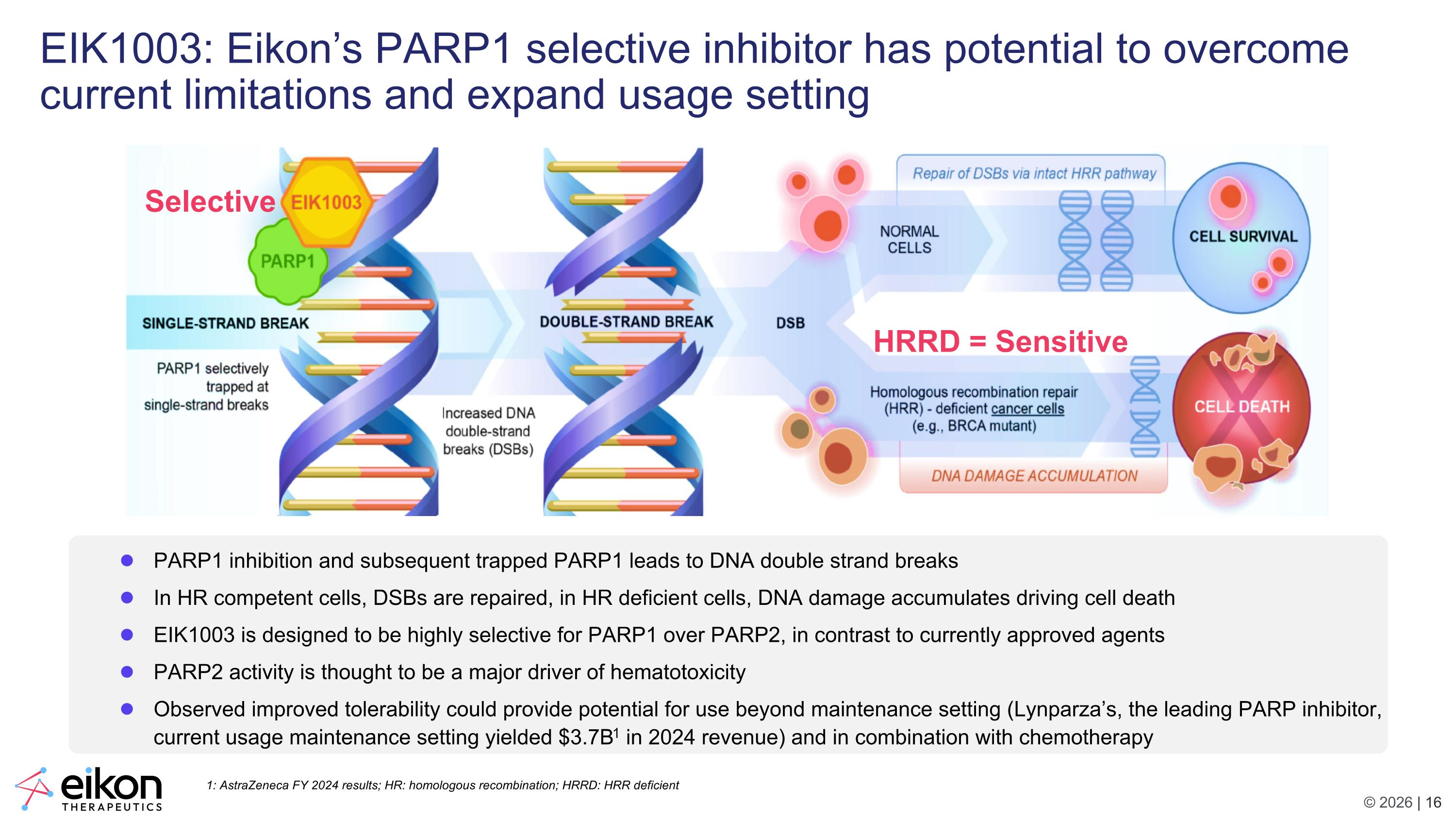
HRRD = Sensitive Selective EIK1003: Eikon’s PARP1 selective inhibitor has potential to overcome current limitations and expand usage setting PARP1 inhibition and subsequent trapped PARP1 leads to DNA double strand breaks In HR competent cells, DSBs are repaired, in HR deficient cells, DNA damage accumulates driving cell death EIK1003 is designed to be highly selective for PARP1 over PARP2, in contrast to currently approved agents PARP2 activity is thought to be a major driver of hematotoxicity Observed improved tolerability could provide potential for use beyond maintenance setting (Lynparza’s, the leading PARP inhibitor, current usage maintenance setting yielded $3.7B1 in 2024 revenue) and in combination with chemotherapy 1: AstraZeneca FY 2024 results; HR: homologous recombination; HRRD: HRR deficient
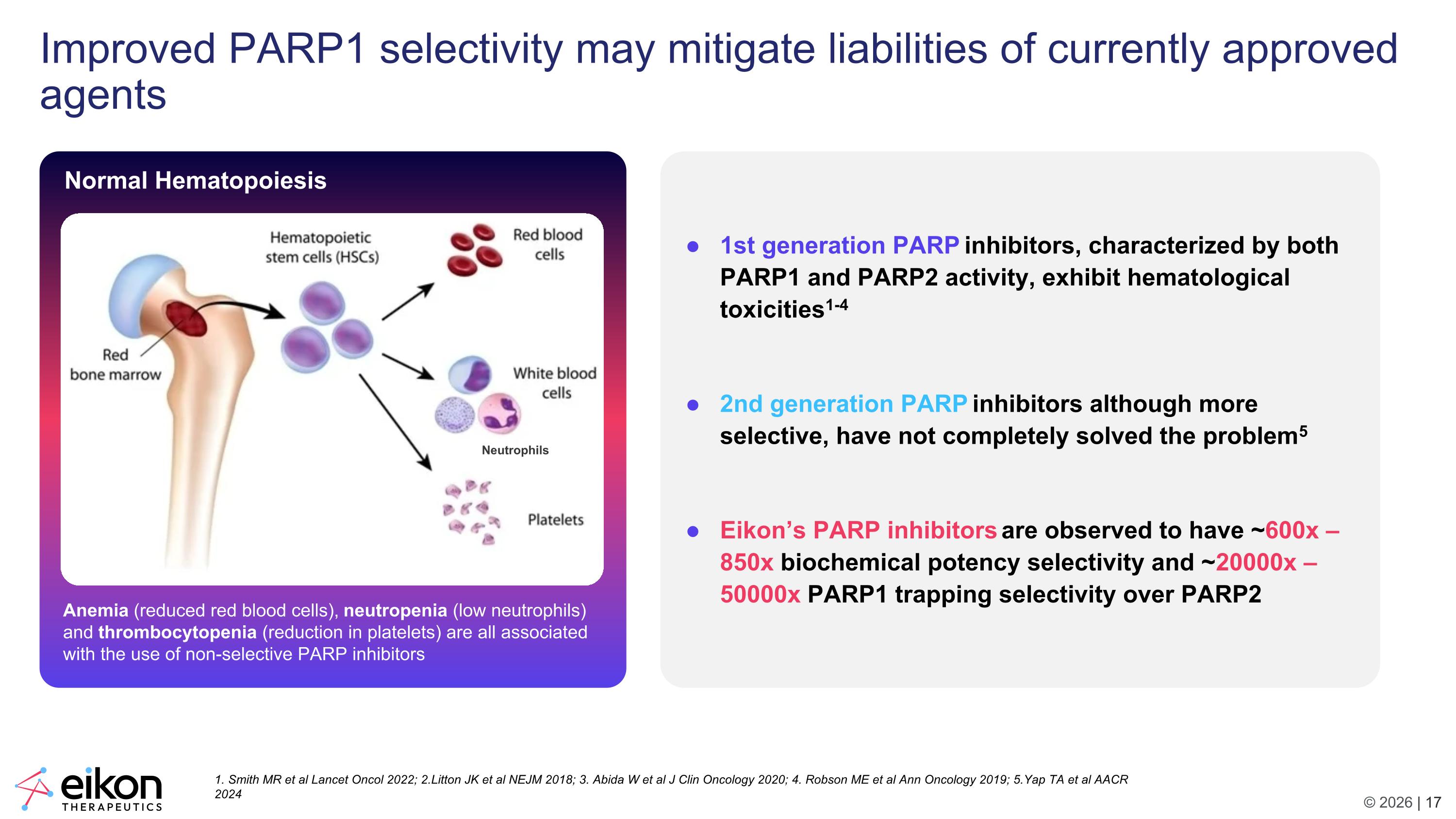
Improved PARP1 selectivity may mitigate liabilities of currently approved agents Normal Hematopoiesis Anemia (reduced red blood cells), neutropenia (low neutrophils) and thrombocytopenia (reduction in platelets) are all associated with the use of non-selective PARP inhibitors 1. Smith MR et al Lancet Oncol 2022; 2.Litton JK et al NEJM 2018; 3. Abida W et al J Clin Oncology 2020; 4. Robson ME et al Ann Oncology 2019; 5.Yap TA et al AACR 2024 Neutrophils 1st generation PARP inhibitors, characterized by both PARP1 and PARP2 activity, exhibit hematological toxicities1-4 2nd generation PARP inhibitors although more selective, have not completely solved the problem5 Eikon’s PARP inhibitors are observed to have ~600x – 850x biochemical potency selectivity and ~20000x – 50000x PARP1 trapping selectivity over PARP2

EIK1003: Recent data provides support for improved tolerability versus conventional PARP inhibitors Phase 1/2 Trial Initial Dose Escalation Fully Enrolled: Monotherapy and combination data (Cohorts 1A & 1C) to be presented at ASCO 2026 EIK1003 Phase 2 randomized dose optimization trial currently dosing patients Additional combination cohort (Cohort 1D) is designed to explore tolerability with platinum-based chemotherapeutic agents Learnings to Date: Once daily dosing Encouraging tolerability profile observed in combination studies with paclitaxel (breast, ovarian) or abiraterone (prostate) Tumor responses observed at multiple dose levels for monotherapy and in combination with standard-of-care treatments Potential for use beyond maintenance setting
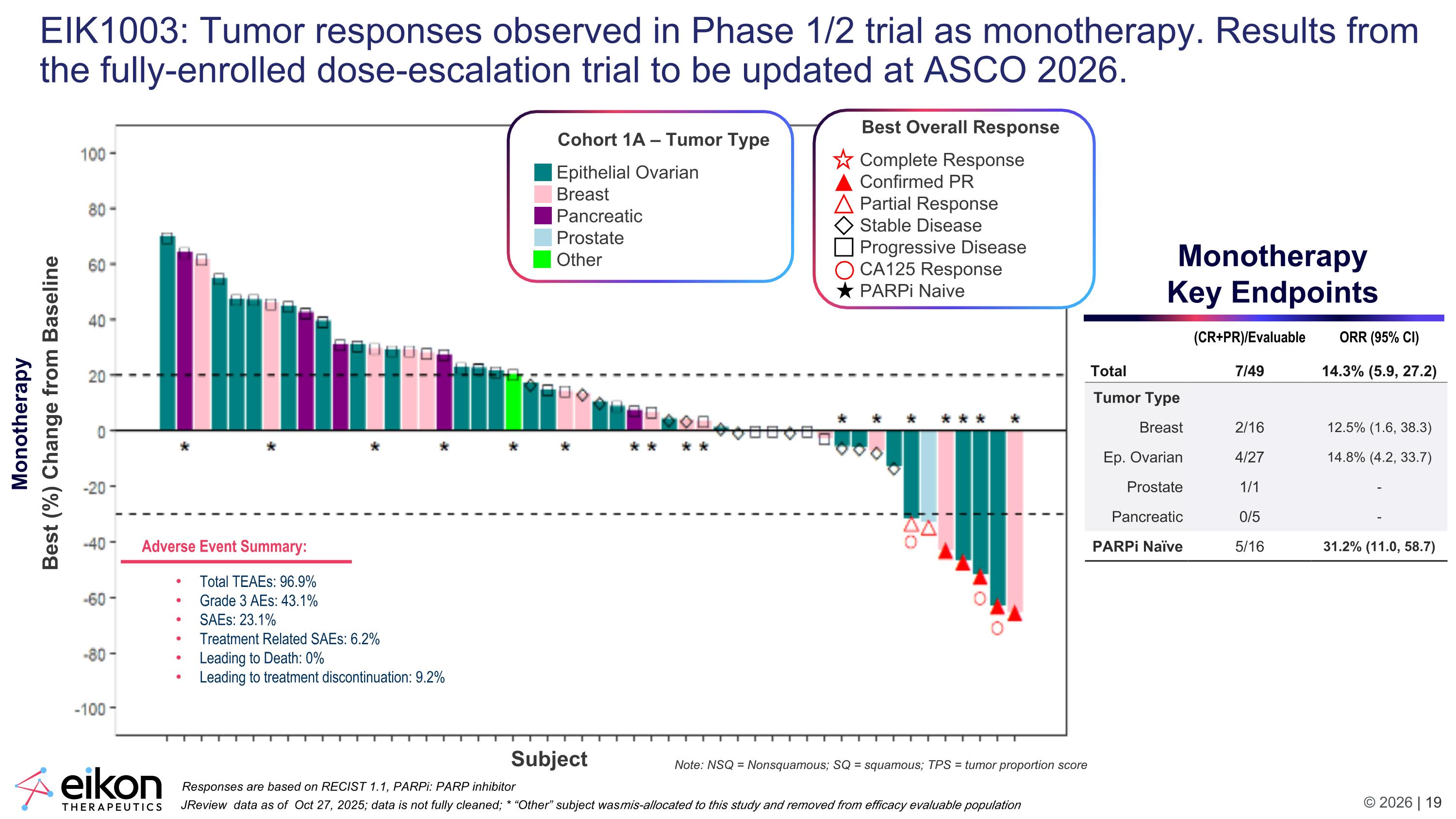
EIK1003: Tumor responses observed in Phase 1/2 trial as monotherapy. Results from the fully-enrolled dose-escalation trial to be updated at ASCO 2026. Monotherapy Key Endpoints Adverse Event Summary: Total TEAEs: 96.9% Grade 3 AEs: 43.1% SAEs: 23.1% Treatment Related SAEs: 6.2% Leading to Death: 0% Leading to treatment discontinuation: 9.2% (CR+PR)/Evaluable ORR (95% CI) Total 7/49 14.3% (5.9, 27.2) Tumor Type Breast 2/16 12.5% (1.6, 38.3) Ep. Ovarian 4/27 14.8% (4.2, 33.7) Prostate 1/1 - Pancreatic 0/5 - PARPi Naïve 5/16 31.2% (11.0, 58.7) Note: NSQ = Nonsquamous; SQ = squamous; TPS = tumor proportion score Best (%) Change from Baseline JReview data as of Oct 27, 2025; data is not fully cleaned; * “Other” subject was mis-allocated to this study and removed from efficacy evaluable population Responses are based on RECIST 1.1, PARPi: PARP inhibitor Subject Cohort 1A – Tumor Type Epithelial Ovarian Breast Pancreatic Prostate Other Best Overall Response Complete Response Confirmed PR Partial Response Stable Disease Progressive Disease CA125 Response PARPi Naive Monotherapy
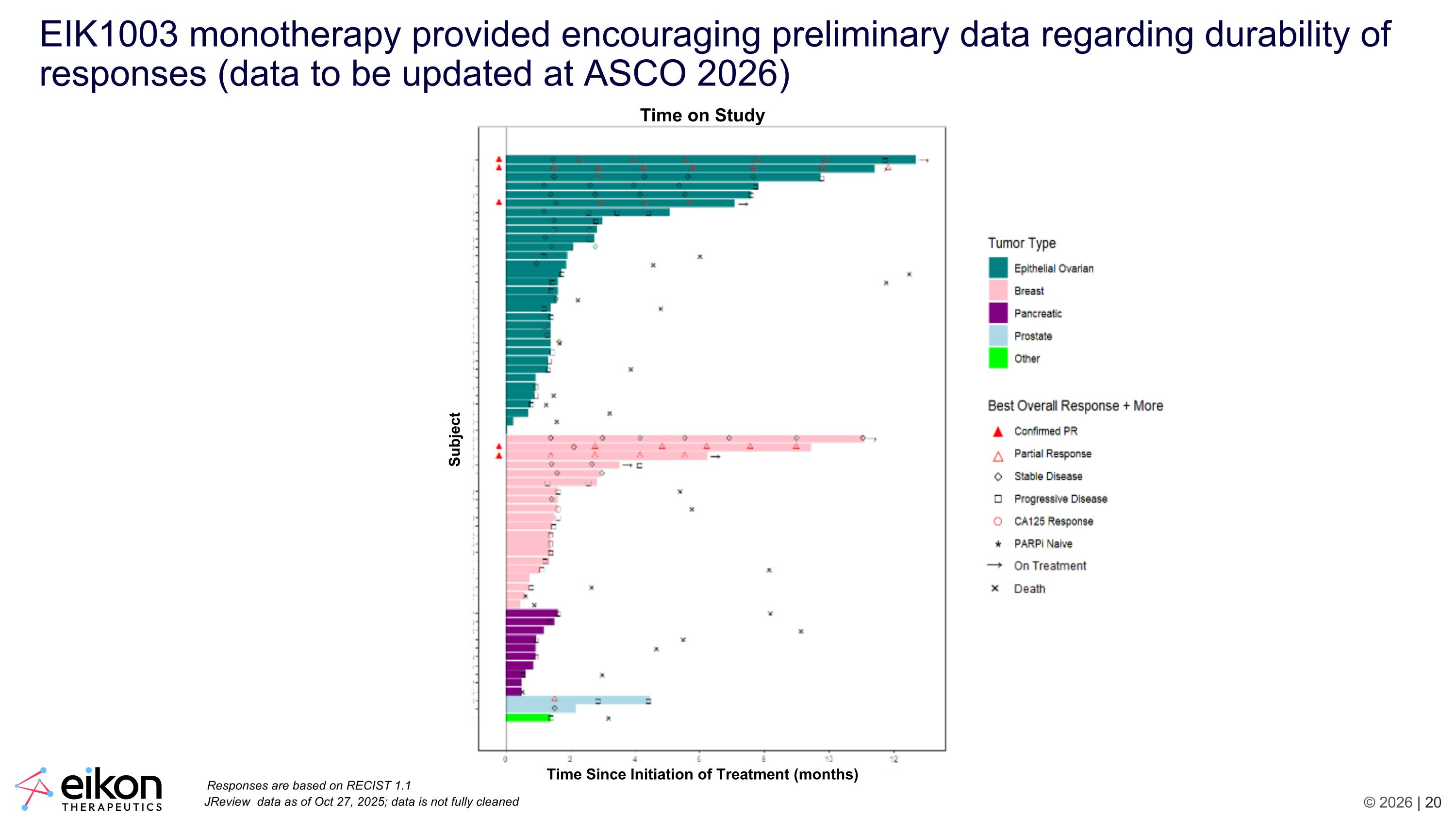
EIK1003 monotherapy provided encouraging preliminary data regarding durability of responses (data to be updated at ASCO 2026) JReview data as of Oct 27, 2025; data is not fully cleaned Responses are based on RECIST 1.1 Time on Study Subject Time Since Initiation of Treatment (months)
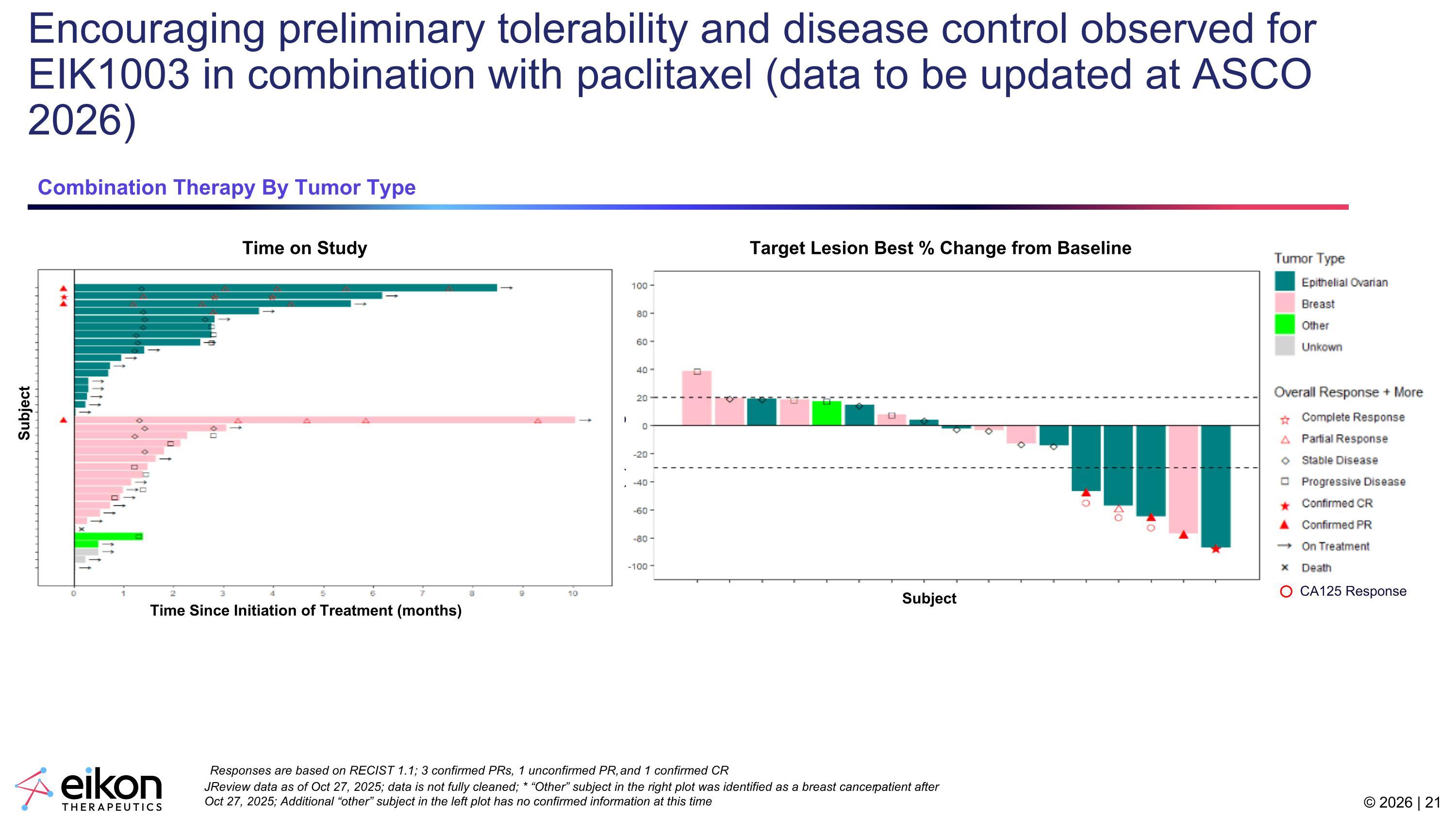
Encouraging preliminary tolerability and disease control observed for EIK1003 in combination with paclitaxel (data to be updated at ASCO 2026) JReview data as of Oct 27, 2025; data is not fully cleaned; * “Other” subject in the right plot was identified as a breast cancer patient after Oct 27, 2025; Additional “other” subject in the left plot has no confirmed information at this time Responses are based on RECIST 1.1; 3 confirmed PRs, 1 unconfirmed PR, and 1 confirmed CR Combination Therapy By Tumor Type Target Lesion Best % Change from Baseline Time on Study Subject Subject Best (%) Change from Baseline CA125 Response Time Since Initiation of Treatment (months)

Eikon Technology Provides Novel Scientific Insights and Permits Rapid Lead Optimization
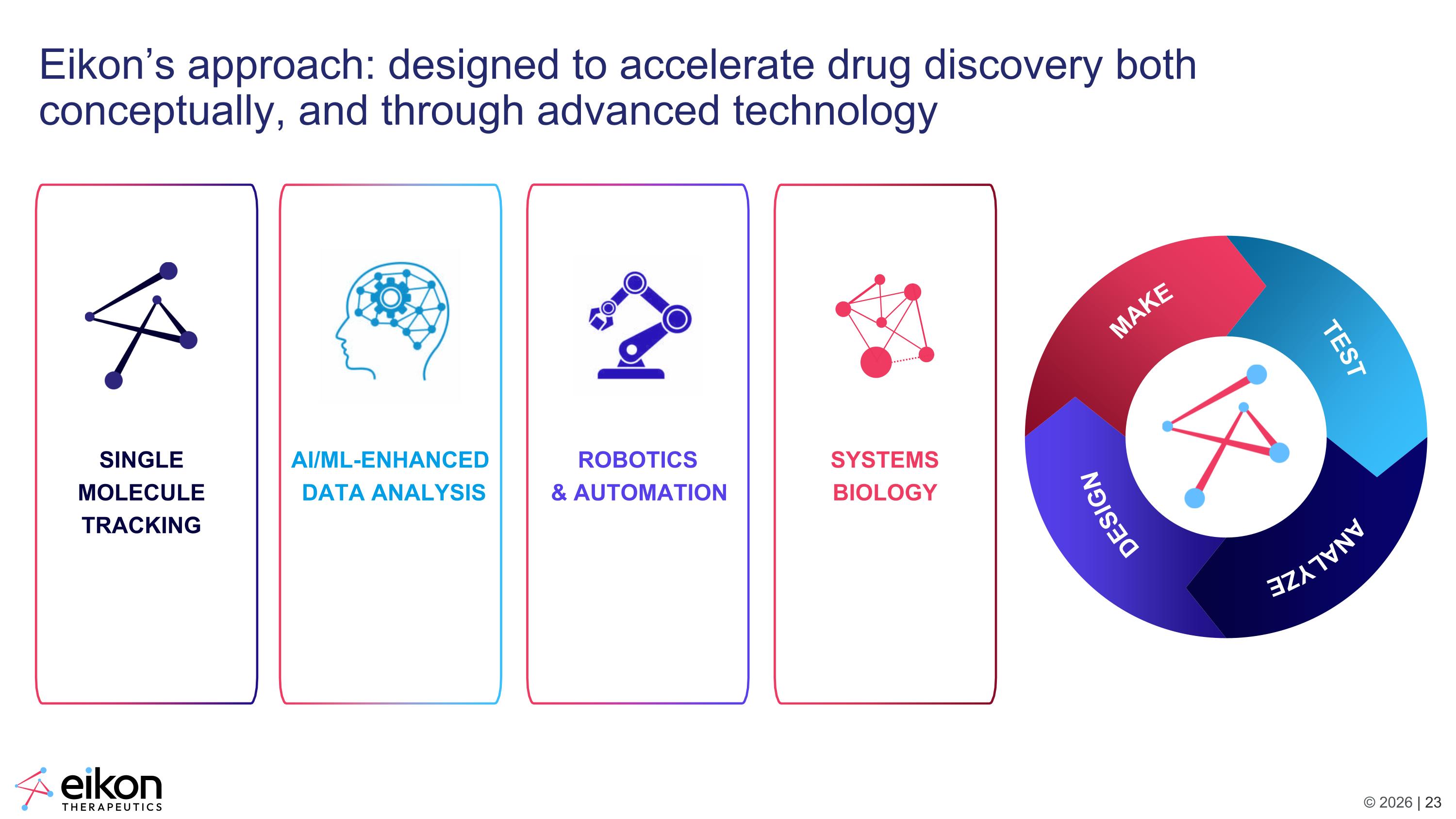
MAKE TEST ANALYZE DESIGN Eikon’s approach: designed to accelerate drug discovery both conceptually, and through advanced technology ROBOTICS & AUTOMATION SYSTEMS BIOLOGY SINGLE MOLECULE TRACKING AI/ML-ENHANCED DATA ANALYSIS
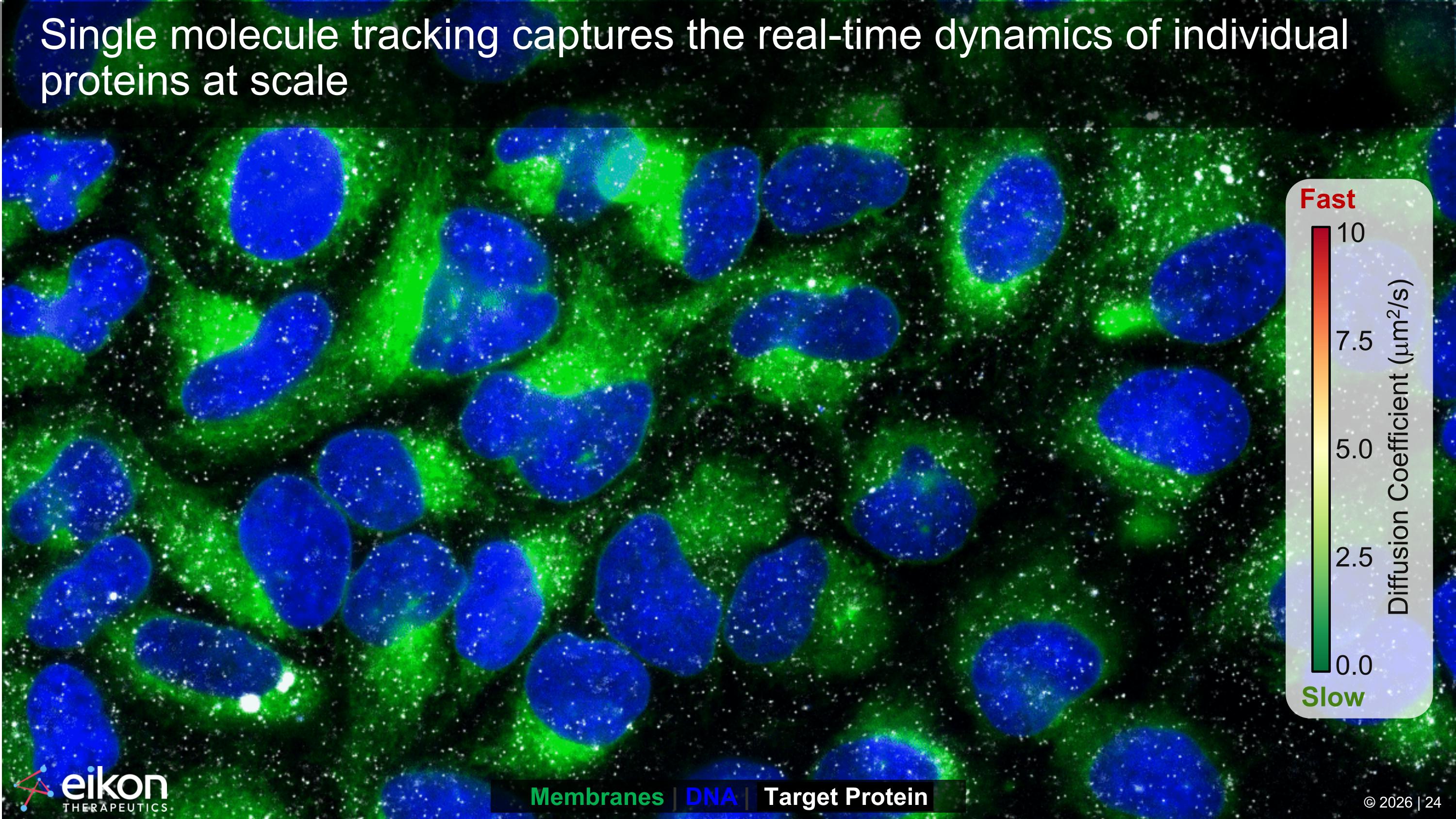
Single molecule tracking captures the real-time dynamics of individual proteins at scale 0.0 2.5 5.0 7.5 10 Fast Slow Diffusion Coefficient (mm2/s) Membranes | DNA | Target Protein © 2026 | 24
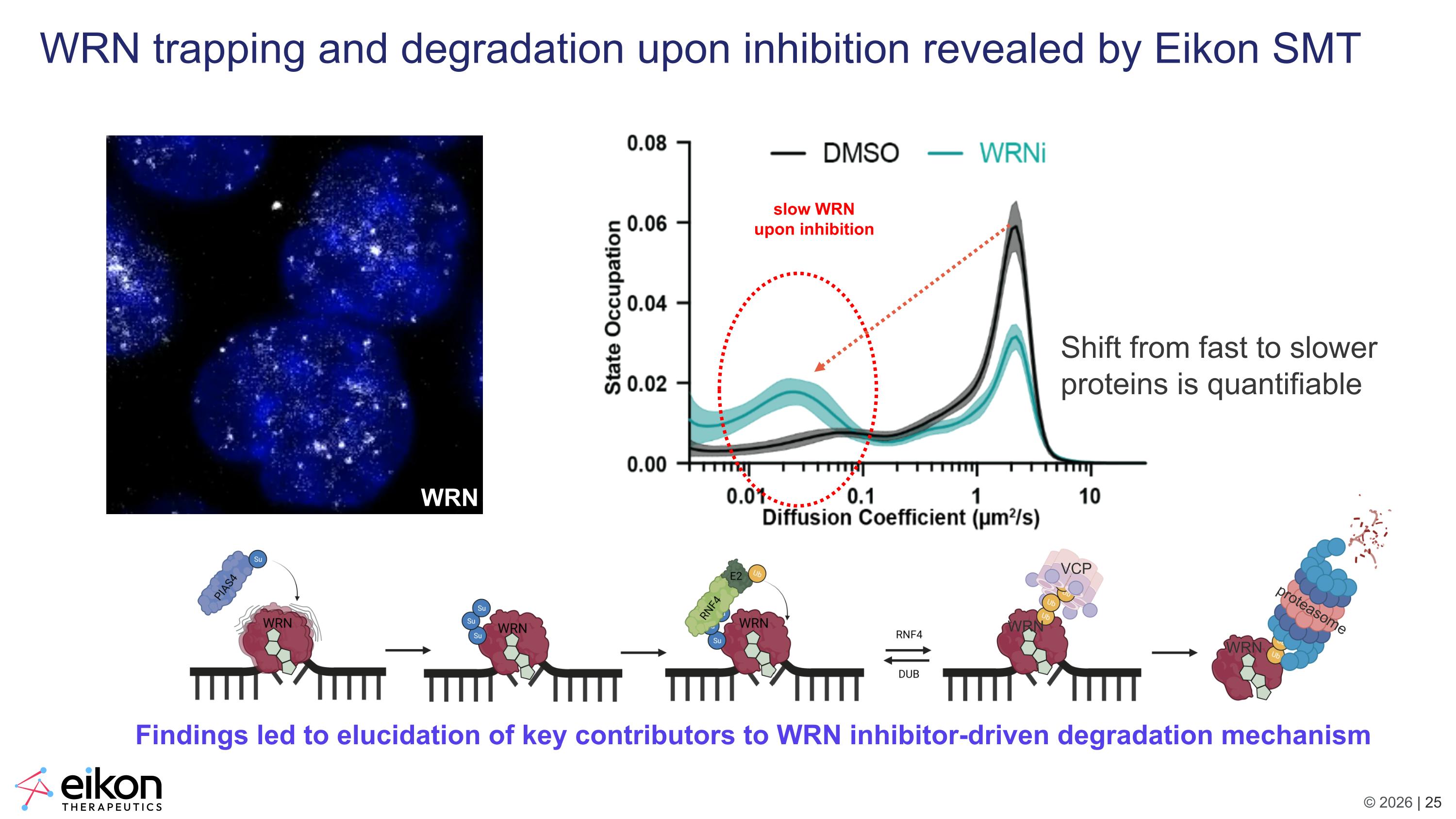
WRN trapping and degradation upon inhibition revealed by Eikon SMT VCP proteasome WRN WRN Findings led to elucidation of key contributors to WRN inhibitor-driven degradation mechanism slow WRN upon inhibition Shift from fast to slower proteins is quantifiable WRN
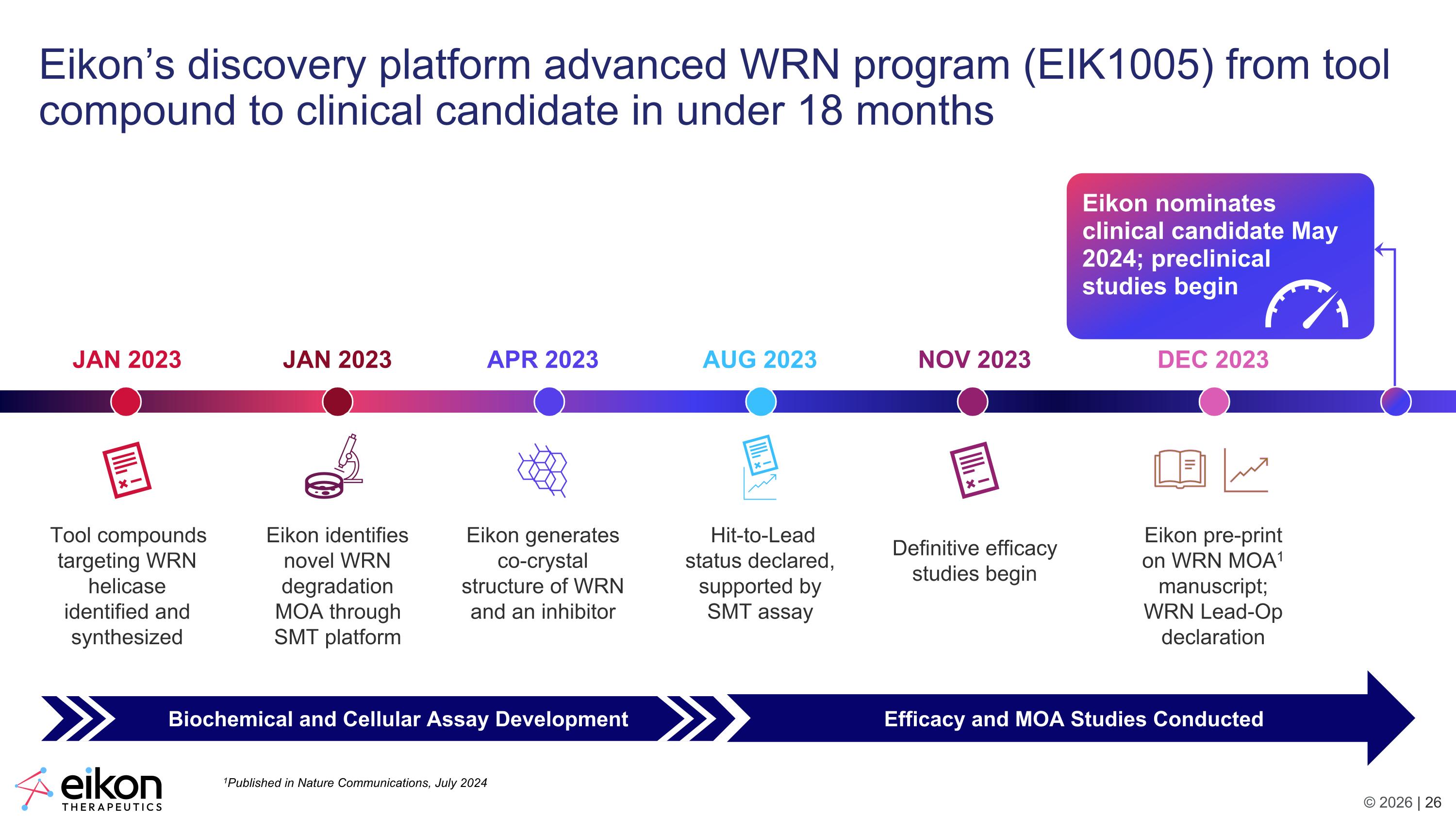
Eikon’s discovery platform advanced WRN program (EIK1005) from tool compound to clinical candidate in under 18 months JAN 2023 Tool compounds targeting WRN helicase identified and synthesized JAN 2023 Eikon identifies novel WRN degradation MOA through SMT platform APR 2023 Eikon generates co-crystal structure of WRN and an inhibitor AUG 2023 Hit-to-Lead status declared, supported by SMT assay NOV 2023 Definitive efficacy studies begin DEC 2023 Eikon pre-print on WRN MOA1 manuscript; WRN Lead-Op declaration Biochemical and Cellular Assay Development Efficacy and MOA Studies Conducted Eikon nominates clinical candidate May 2024; preclinical studies begin 1Published in Nature Communications, July 2024
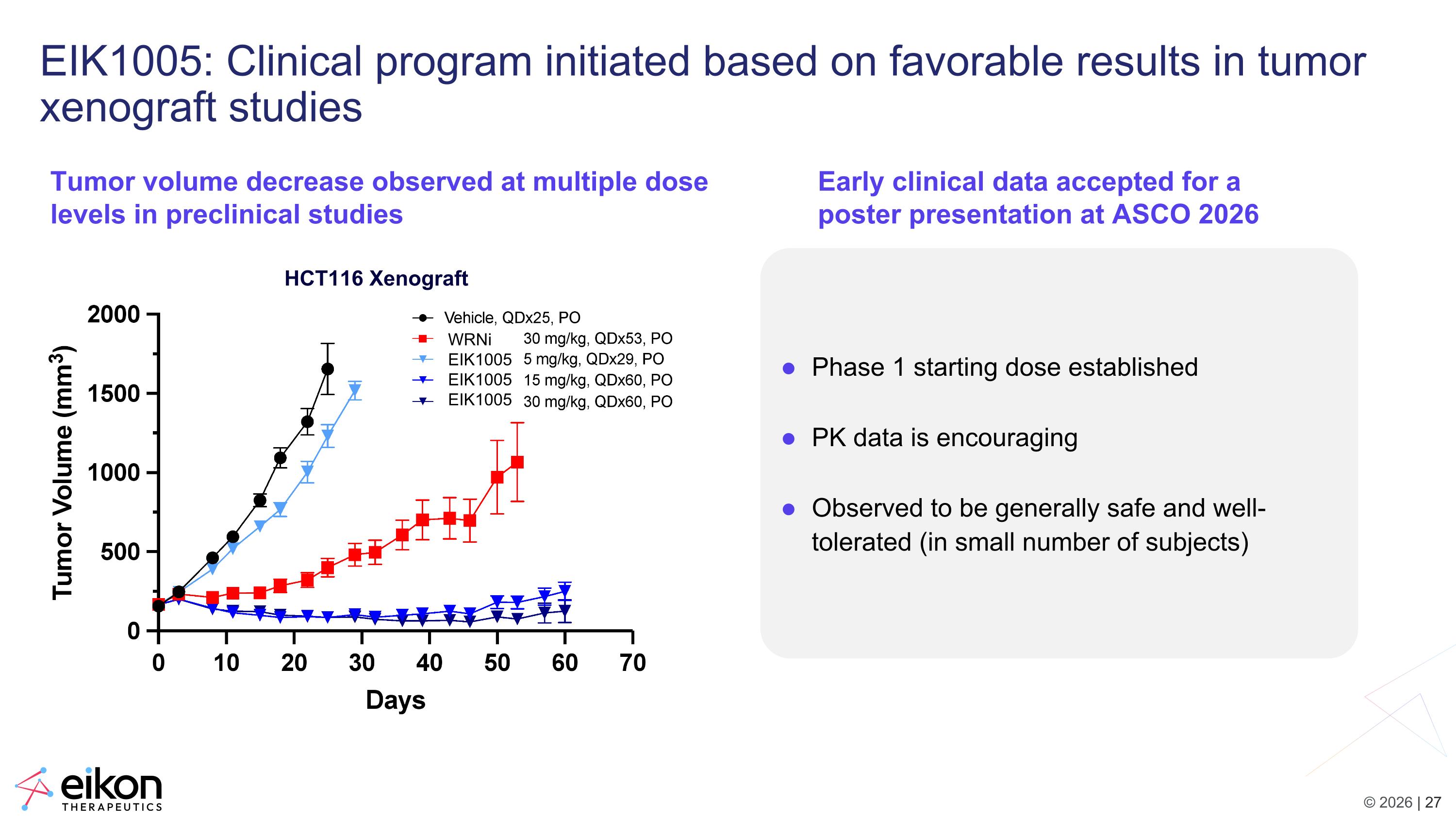
EIK1005: Clinical program initiated based on favorable results in tumor xenograft studies Phase 1 starting dose established PK data is encouraging Observed to be generally safe and well-tolerated (in small number of subjects) Tumor volume decrease observed at multiple dose levels in preclinical studies Early clinical data accepted for a poster presentation at ASCO 2026 HCT116 Xenograft WRNi EIK1005 EIK1005 EIK1005
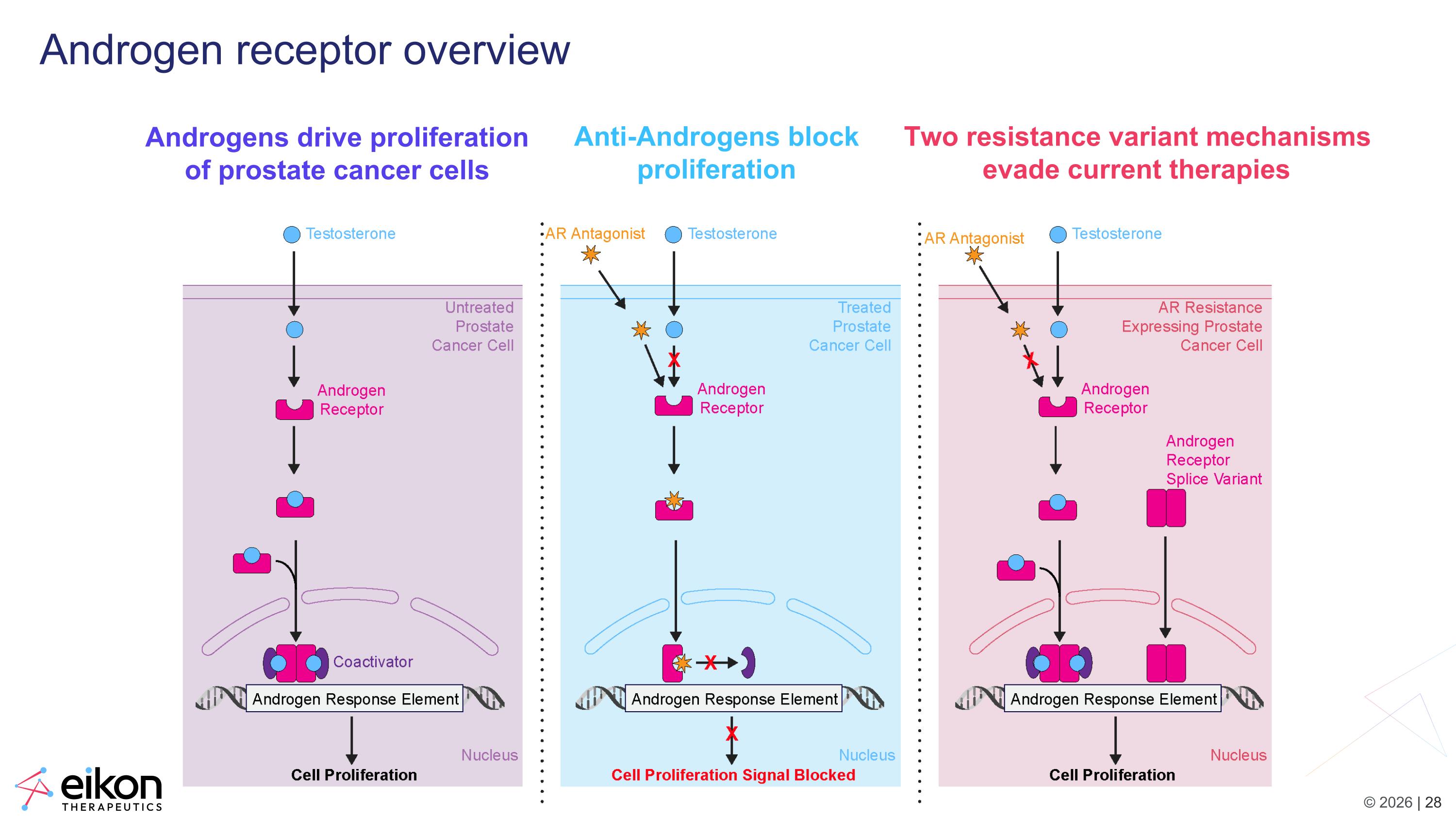
Androgen receptor overview Androgens drive proliferation of prostate cancer cells Anti-Androgens block proliferation Two resistance variant mechanisms evade current therapies
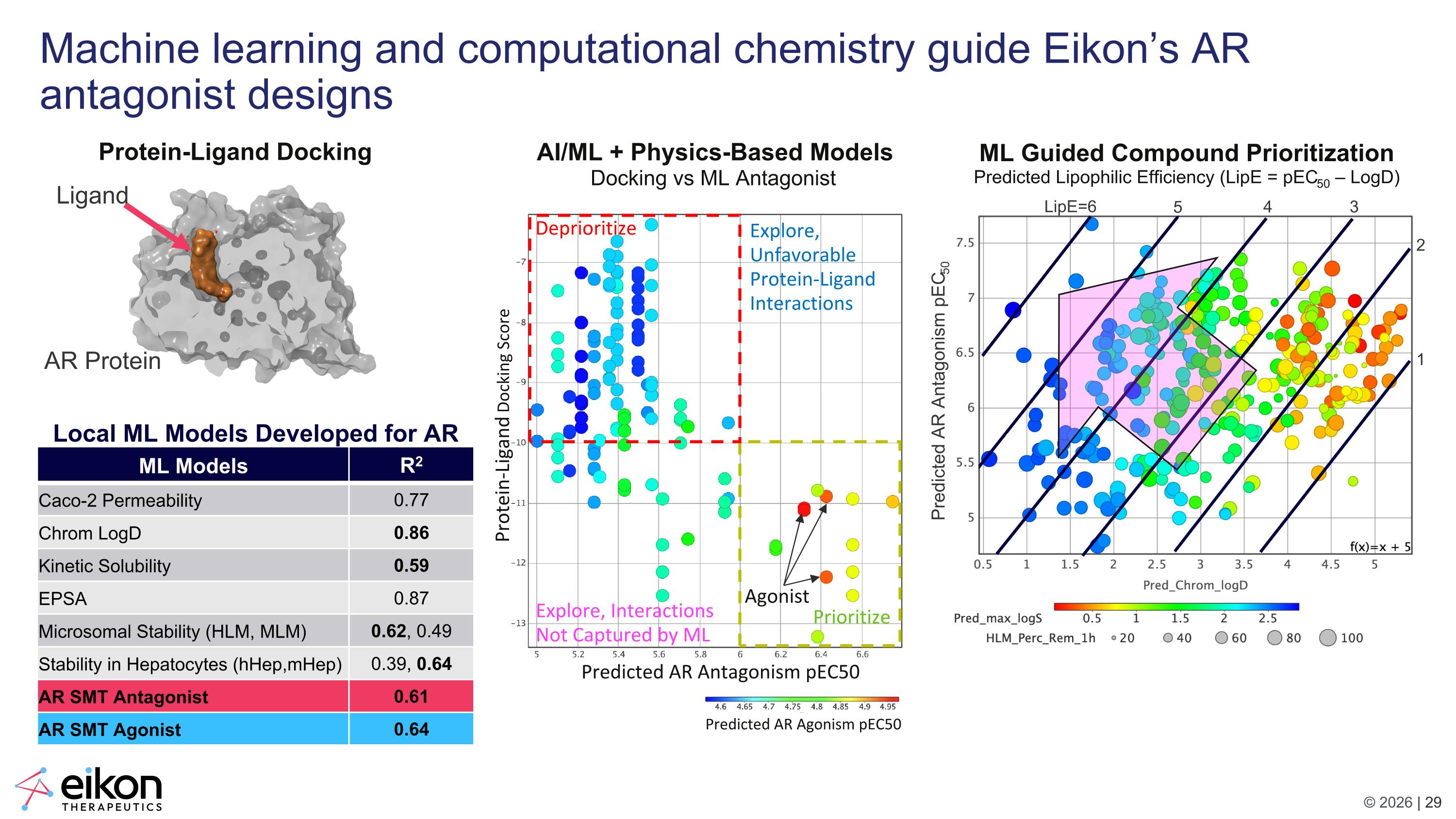
AI/ML + Physics-Based Models Docking vs ML Antagonist Deprioritize Prioritize Predicted AR Antagonism pIC50 Predicted AR Agonism pEC50 Predicted AR Antagonism pEC50 Protein-Ligand Docking Score Agonist Explore, Interactions Not Captured by ML Explore, Unfavorable Protein-Ligand Interactions ML Guided Compound Prioritization Predicted Lipophilic Efficiency (LipE = pEC50 – LogD) LipE=6 5 4 3 2 1 Machine learning and computational chemistry guide Eikon’s AR antagonist designs Protein-Ligand Docking Predicted AR Antagonism pEC50 Ligand AR Protein ML Models R2 Caco-2 Permeability 0.77 Chrom LogD 0.86 Kinetic Solubility 0.59 EPSA 0.87 Microsomal Stability (HLM, MLM) 0.62, 0.49 Stability in Hepatocytes (hHep,mHep) 0.39, 0.64 AR SMT Antagonist 0.61 AR SMT Agonist 0.64 Local ML Models Developed for AR

Eikon’s AR programs aim to address limitations of current therapies EIK1006 (AR clinical candidate) observed to inhibit tumor growth, reduce PSA levels and alter AR gene expression in preclinical models Observed activity across a range of emerging resistant variants EIK1006 declared clinical candidate in November 2025; preclinical studies now tracking ahead of schedule, expected to enable IND submission by the end of 2026

Conclusion
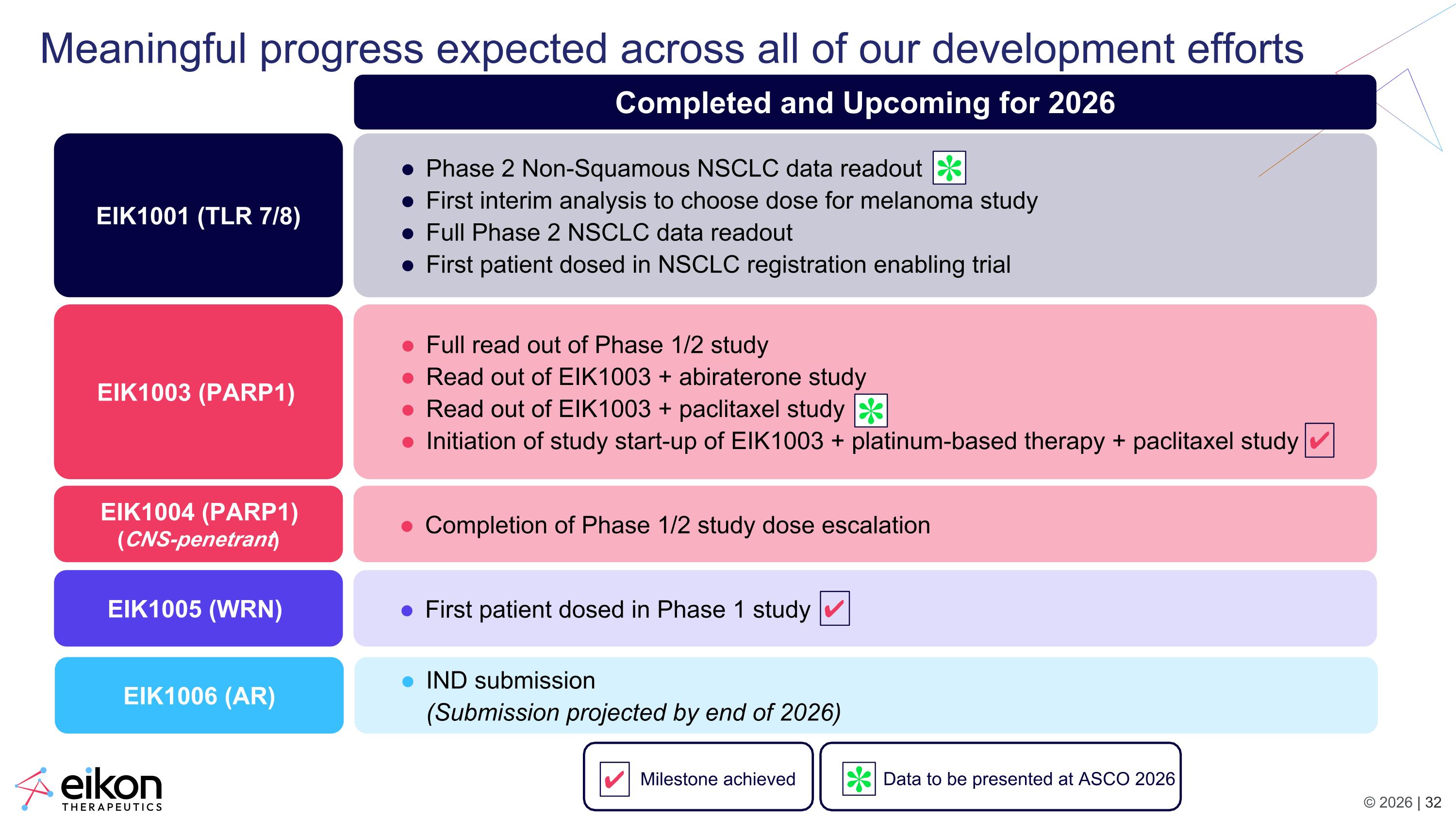
Meaningful progress expected across all of our development efforts EIK1001 (TLR 7/8) EIK1003 (PARP1) EIK1004 (PARP1) (CNS-penetrant) EIK1005 (WRN) Phase 2 Non-Squamous NSCLC data readout First interim analysis to choose dose for melanoma study Full Phase 2 NSCLC data readout First patient dosed in NSCLC registration enabling trial Full read out of Phase 1/2 study Read out of EIK1003 + abiraterone study Read out of EIK1003 + paclitaxel study Initiation of study start-up of EIK1003 + platinum-based therapy + paclitaxel study Completion of Phase 1/2 study dose escalation First patient dosed in Phase 1 study EIK1006 (AR) IND submission (Submission projected by end of 2026) Data to be presented at ASCO 2026 Completed and Upcoming for 2026 ✔ Milestone achieved ✔ ✔ ✽ ✽ ✽

Led by world-renowned drug developers Eikon Therapeutics highlights Technology platform, centered around proprietary single-molecule tracking (SMT) Clinical-stage programs have generated early signs of encouraging clinical activity Internally identified and strategically tailored product candidates advancing into clinical investigation 1 2 3 4
