

Pioneering Precision Peptides for Endocrine and Metabolic Diseases Obesity Day May 11, 2026 EXHIBIT 99.2

Introduction Kent Hawryluk President & CEO

MBX Obesity Day 2026 Agenda Welcome/Introductions Kent Hawryluk, MBX President & CEO 1 Market Landscape & Opportunity Katherine H. Saunders, M.D., Co-Founder, FlyteHealth; Faculty, Weill Cornell Medicine 2 MBX PEP™ Platform Overview Richard DiMarchi, Ph.D., Distinguished Professor of Chemistry at Indiana University, MBX Co-founder 3 MBX 4291 Initial Phase 1 Data Sam Azoulay, M.D., MBX Chief Medical Officer 4 Amycretin Candidate Nomination Richard DiMarchi 5 Imapextide Phase 2a Results Kent Hawryluk 6 Priorities/Conclusion Kent Hawryluk 7 Q&A/Lunch All 8

Disclaimer This presentation includes forward looking statements. All statements other than statements of historical facts contained in this presentation, including statements regarding our product candidates, preclinical study and/or clinical trial timelines, including projected data announcements, future results of operations and financial position, strategy and plans, industry environment, potential growth opportunities, and our expectations for future operations, are forward looking statements The words “ believe,” “ may,” “will,” “ estimate,” “ continue,” “ anticipate,” “ design,” “ “ expect,” “ could,” “ plan,” “ potential,” “ predict,” “ seek,” “ should,” “would,” or the negative version of these words and similar expressions are intended to identify forward looking statements. We have based these forward-looking statements on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, strategy, short- and long-term business operations and objectives, and financial needs. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including but not limited to, our ability to develop and advance our programs and product candidates, our regulatory approvals and filings, and other risks, uncertainties and assumptions identified in our filings with the Securities and Exchange Commission. Moreover, we operate in a very competitive and rapidly changing environment. New risks emerge from time to time, and it is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward looking statements we may make. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this presentation may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. You should not rely upon forward looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances reflected in the forward-looking statements will be achieved or occur. Moreover, except as required by law, neither we nor any other person assumes responsibility for the accuracy and completeness of the forward-looking statements. We undertake no obligation to update publicly any forward-looking statements for any reason after the date of this presentation to conform these statements to actual results or to changes in our expectations, unless required by law. This presentation contains estimates and other information concerning our industry, our business and the markets for our product candidates. Information that is based on estimates, market research or similar methodologies, including prevalence studies which are extrapolated to broader populations, is inherently subject to uncertainties, and actual events or circumstances may differ materially from events and circumstances that are assumed in this information. Unless otherwise expressly stated, we obtained this industry, business, market and other data from our own internal estimates and research as well as from reports, research surveys, studies and similar data prepared by market research firms and other third parties, industry, medical and general publications, government data and similar sources. Industry publications and surveys generally state that the information contained therein has been obtained from sources believed to be reliable. Although we believe the industry and market data to be reliable as of the date of this presentation, this information could prove to be inaccurate. Industry and market data could be wrong because of the method by which sources obtained their data and because information cannot always be verified with complete certainty due to the limits on the availability and reliability of raw data, the voluntary nature of the data gathering process and other limitations and uncertainties. While we are responsible for the accuracy of such information and believe our internal company research as to such matters is reliable and the market definitions are appropriate, neither such research nor these definitions have been verified by any independent source.
MBX: Pioneering Precision Peptides for Endocrine and Metabolic Diseases Multiple clinical-stage programs, each designed for differentiation in multibillion dollar markets Extended duration of action Consistent drug exposures Less frequent dosing Unique PK profile to simplify titration regimens Once-weekly canvuparatide data support potential best-in-class profile in chronic hypoparathyroidism Results from Phase 2 trial accepted for oral presentation at ENDO in June 2026 Phase 3 trial on track to initiate in Q3 Obesity pipeline designed for once-monthly administration, fewer titration steps and improved tolerability MBX 4291 PK shows potential for true once-monthly dosing 12-week Phase 1 MAD results on track for Q4 MBX 5765 (amycretin) in IND-enabling studies Nomination of triple agonist on track for Q3 Clinically validated PEP platform unlocks vast potential of peptide therapeutics Catalyst-rich 2026 with substantial value inflection opportunities Well capitalized with $440 million in cash, expected to provide runway into 20291 1 Unaudited cash, cash equivalents and marketable securities as of March 31, 2026

Obesity Market Landscape Katherine H. Saunders, M.D. Co-Founder, FlyteHealth Faculty, Weill Cornell Medicine

Worldwide Obesity Prevalence Obesity rates have doubled since 1990 25% of the World’s Population is projected to have obesity by 2035 390 million children and adolescents overweight 1 in 8 people worldwide are living with obesity 160 million children living with obesity 890 million adults living with obesity 2.5 billion adults worldwide overweight According to World Health Organization and World Obesity Federation https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight https://www.worldobesity.org/about/about-obesity/prevalence-of-obesity Ahmed SK, Mohammed RA. Obesity: Prevalence, causes, consequences, management, preventive strategies and future research directions. Metabol Open. 2025 Jun 14;27:100375. 35 million children less than 5yrs are overweight
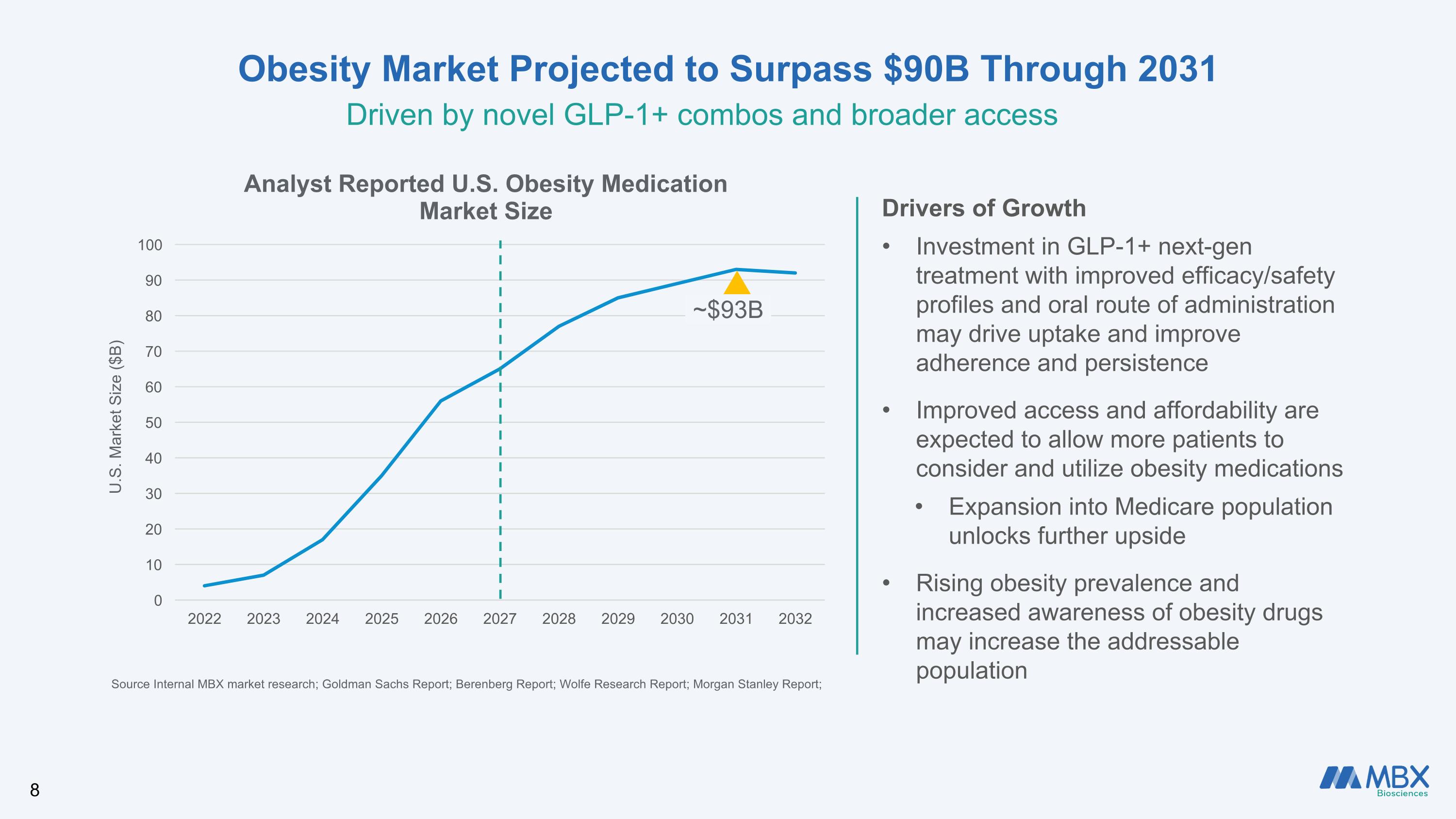
Obesity Market Projected to Surpass $90B Through 2031 Driven by novel GLP-1+ combos and broader access Drivers of Growth Investment in GLP-1+ next-gen treatment with improved efficacy/safety profiles and oral route of administration may drive uptake and improve adherence and persistence Improved access and affordability are expected to allow more patients to consider and utilize obesity medications Expansion into Medicare population unlocks further upside Rising obesity prevalence and increased awareness of obesity drugs may increase the addressable population Source Internal MBX market research; Goldman Sachs Report; Berenberg Report; Wolfe Research Report; Morgan Stanley Report; U.S. Market Size ($B) ~$93B
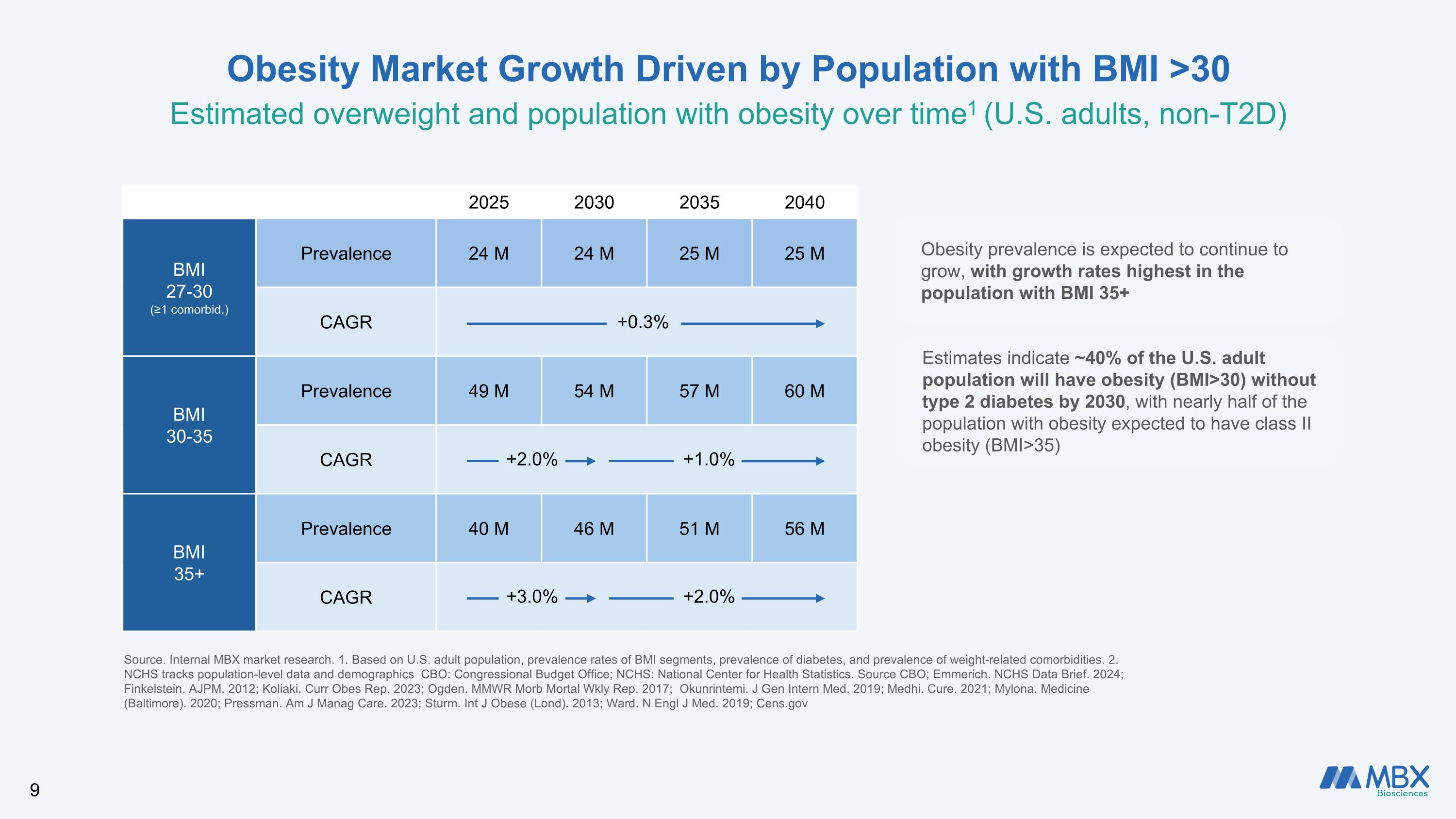
Obesity Market Growth Driven by Population with BMI >30 Estimated overweight and population with obesity over time1 (U.S. adults, non-T2D) Obesity prevalence is expected to continue to grow, with growth rates highest in the population with BMI 35+ Estimates indicate ~40% of the U.S. adult population will have obesity (BMI>30) without type 2 diabetes by 2030, with nearly half of the population with obesity expected to have class II obesity (BMI>35) Source. Internal MBX market research. 1. Based on U.S. adult population, prevalence rates of BMI segments, prevalence of diabetes, and prevalence of weight-related comorbidities. 2. NCHS tracks population-level data and demographics CBO: Congressional Budget Office; NCHS: National Center for Health Statistics. Source CBO; Emmerich. NCHS Data Brief. 2024; Finkelstein. AJPM. 2012; Koliaki. Curr Obes Rep. 2023; Ogden. MMWR Morb Mortal Wkly Rep. 2017; Okunrintemi. J Gen Intern Med. 2019; Medhi. Cure. 2021; Mylona. Medicine (Baltimore). 2020; Pressman. Am J Manag Care. 2023; Sturm. Int J Obese (Lond). 2013; Ward. N Engl J Med. 2019; Cens.gov 2025 2030 2035 2040 BMI 27-30 (≥1 comorbid.) Prevalence 24 M 24 M 25 M 25 M CAGR BMI 30-35 Prevalence 49 M 54 M 57 M 60 M CAGR BMI 35+ Prevalence 40 M 46 M 51 M 56 M CAGR +0.3% +2.0% +1.0% +3.0% +2.0%

Unmet Needs Suggest Potential for Future Differentiation Source: Internal MBX market research. AOM: Anti-obesity Medication; ROA: Route of Administration. Source: Abdel-Bary. Obes Med. 2025; Aronne. JAMA. 2023; Wilding. Diabetes Obes Metab. 2022; Clearview Analysis Clinical data show weight loss plateau at ~20% at ~1 year, which may be insufficient for patients with class III obesity (BMI>40) Response 1 Real-world studies indicate that patients discontinuing GLP-1 therapies are prone to rapid weight regain, highlighting a need for weight strategies Prevention of Weight Regain 2 Improved tolerability may enhance adherence, expand long-term use, and decrease discontinuations in real world settings Tolerability 3 Less frequent and flexible dosing, simplified regimens and oral formulations can support long-term management and improve patient persistence Convenience 4 Preserving muscle while reducing fat is still a key unmet need for improving health outcomes Weight Loss Quality 5 Developing treatments with proven benefits and safety in subpopulations (e.g., pediatric, comorbid, elderly) can improve health beyond weight loss and facilitate in enabling access to AOMs Outcomes 6

Gastrointestinal Adverse Events Remain a Key Limitation of Incretin-Based Therapies GI-related tolerability is a well-known on-target side effect of incretin therapies (e.g., semaglutide, tirzepatide) Nausea, vomiting, diarrhea are most frequent (25-44% in Phase 3 trials) Requires lengthy titration regimen to allow adaptation before getting to optimal efficacious dose Events often emerge during initiation and dose escalation Mechanism linked to gastric emptying/GI motility What we see clinically Why it matters Symptoms are often mild, but can be persistent Repeated GI events may impact adherence Key driver of treatment discontinuation Requires lengthy and gradual titration to improve tolerability May impact reaching optimal therapeutic dose Optimizing exposure may improve tolerability and decrease amount of required titration steps Source: Ismaiel, A., et al. Int J Obes 2025 Oct;49(10):1946-1957; Jastreboff AM, et al. N Engl J Med. 2022;387:205–216; Wilding JPH, et al. N Engl J Med. 2021;384:989–1002.
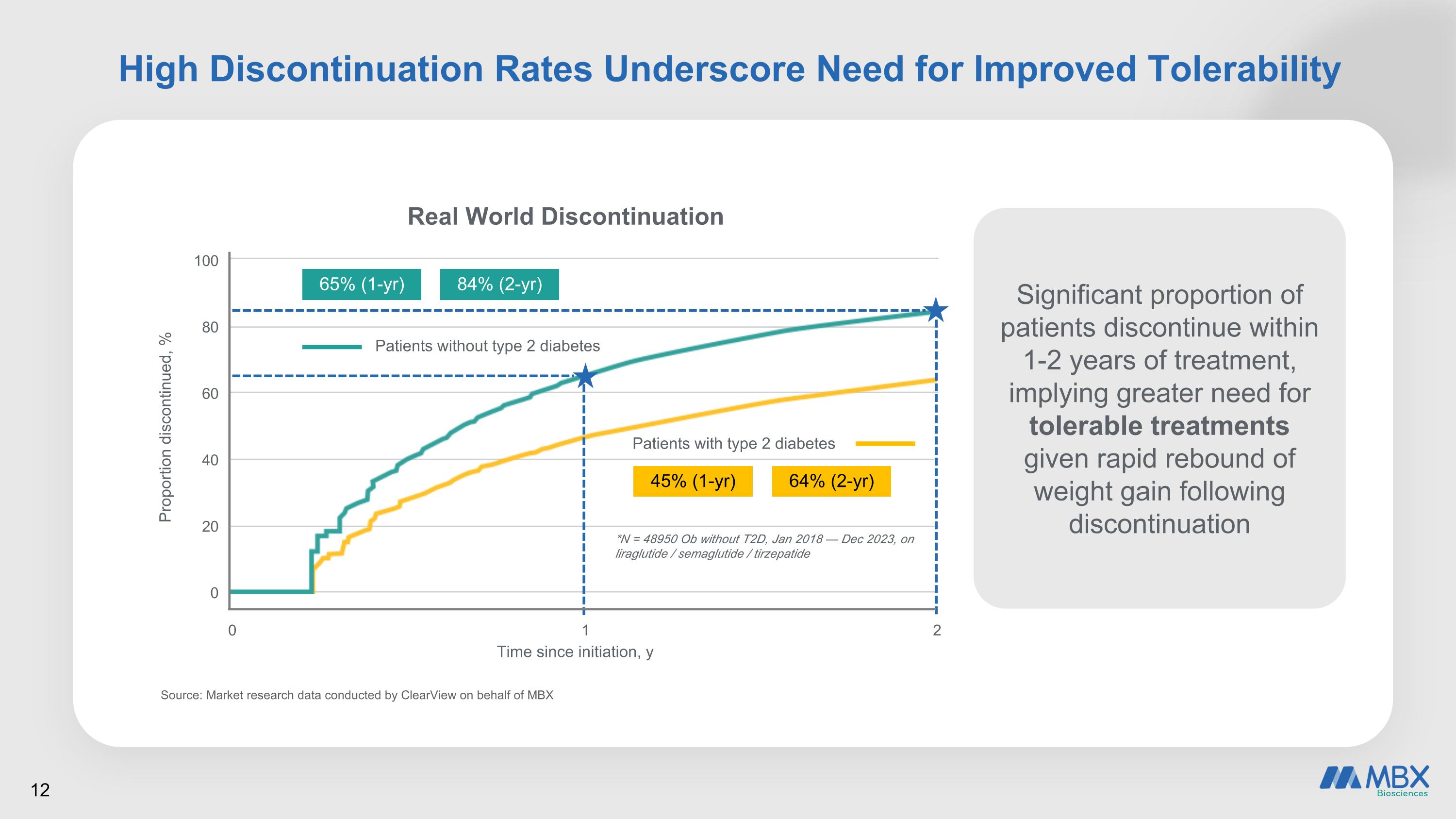
High Discontinuation Rates Underscore Need for Improved Tolerability Source: Market research data conducted by ClearView on behalf of MBX Proportion discontinued, % 100 80 60 40 20 0 0 1 2 Time since initiation, y Real World Discontinuation 65% (1-yr) 84% (2-yr) Patients without type 2 diabetes Patients with type 2 diabetes 45% (1-yr) 64% (2-yr) *N = 48950 Ob without T2D, Jan 2018 — Dec 2023, on liraglutide / semaglutide / tirzepatide Significant proportion of patients discontinue within 1-2 years of treatment, implying greater need for tolerable treatments given rapid rebound of weight gain following discontinuation

MBX Proprietary PEP™ Platform Richard DiMarchi, PhD MBX Scientific Co-Founder Distinguished Professor of Chemistry and Gill Chair in Biomolecular Sciences at Indiana University
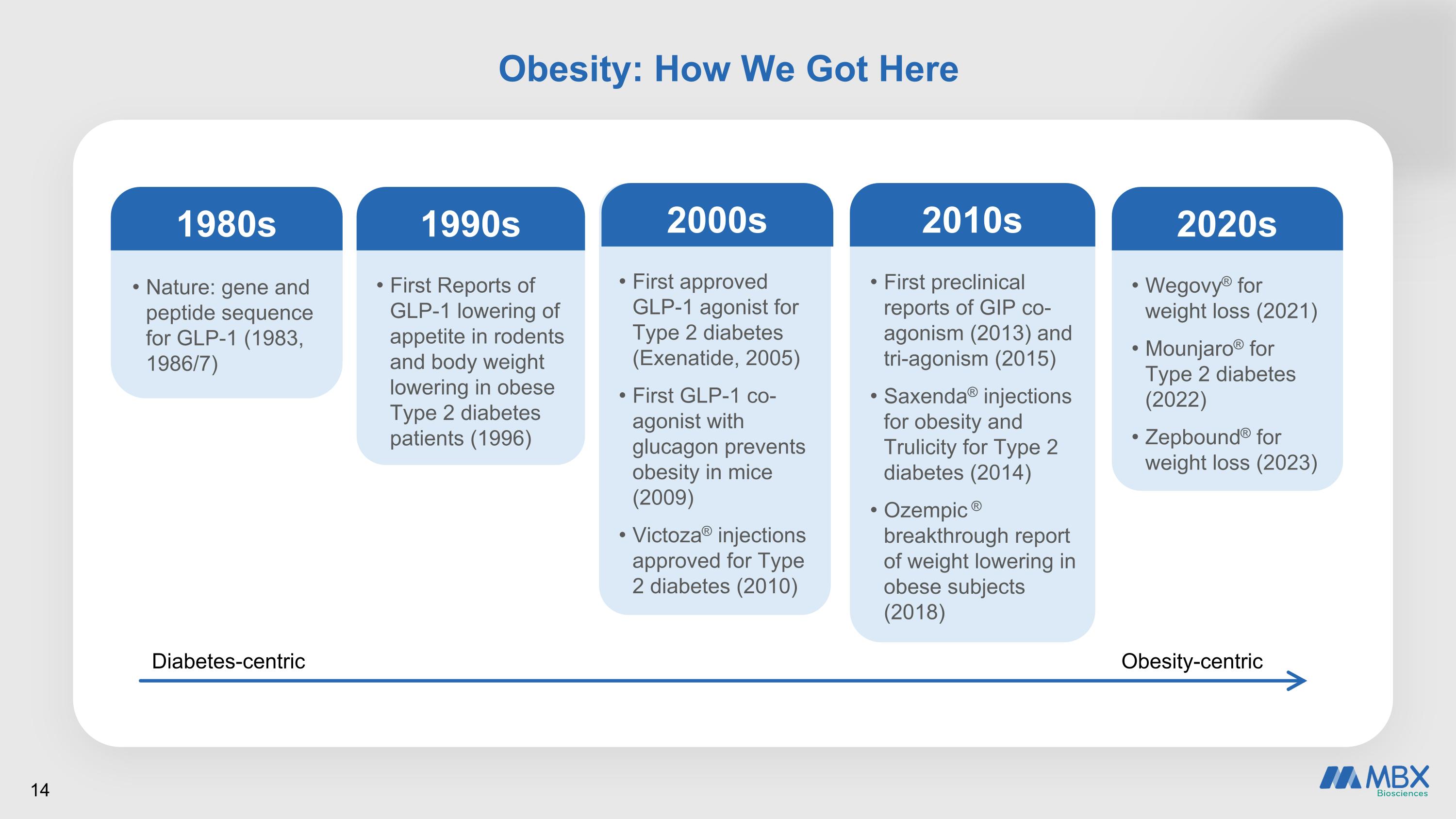
Obesity: How We Got Here Obesity-centric Nature: gene and peptide sequence for GLP-1 (1983, 1986/7) 1980s First Reports of GLP-1 lowering of appetite in rodents and body weight lowering in obese Type 2 diabetes patients (1996) 1990s First approved GLP-1 agonist for Type 2 diabetes (Exenatide, 2005) First GLP-1 co-agonist with glucagon prevents obesity in mice (2009) Victoza® injections approved for Type 2 diabetes (2010) 2000s First preclinical reports of GIP co-agonism (2013) and tri-agonism (2015) Saxenda® injections for obesity and Trulicity for Type 2 diabetes (2014) Ozempic ® breakthrough report of weight lowering in obese subjects (2018) 2010s Diabetes-centric Wegovy® for weight loss (2021) Mounjaro® for Type 2 diabetes (2022) Zepbound® for weight loss (2023) 2020s

Meaningful Weight Loss 1 Tolerability Improved Compliance 2 3 Patient-centric Drug-centric Obesity: Next Generation Performance
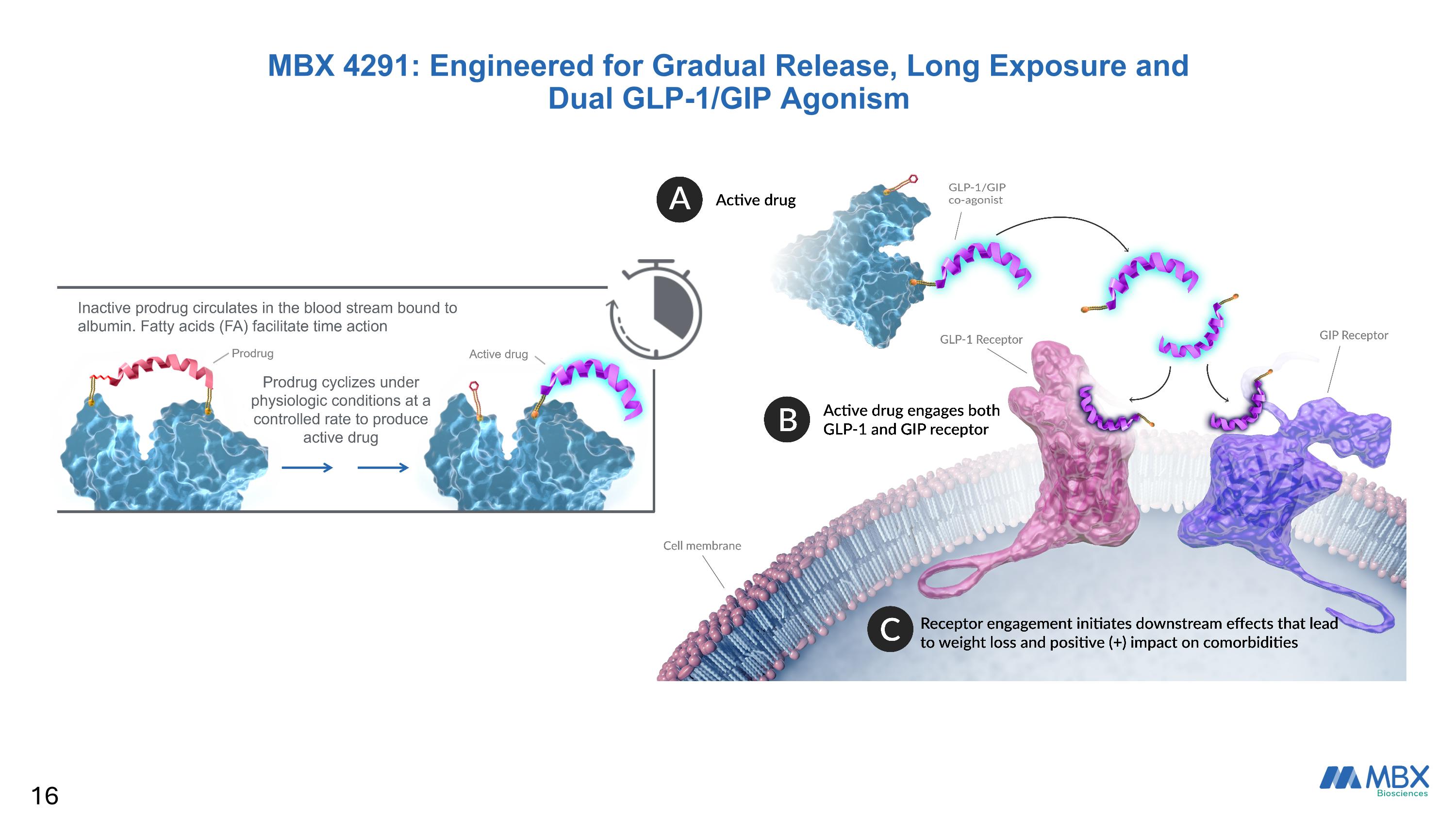
MBX 4291: Engineered for Gradual Release, Long Exposure and Dual GLP-1/GIP Agonism

Sam Azoulay, MD MBX Chief Medical Officer MBX 4291 GLP-1/GIP Agonist
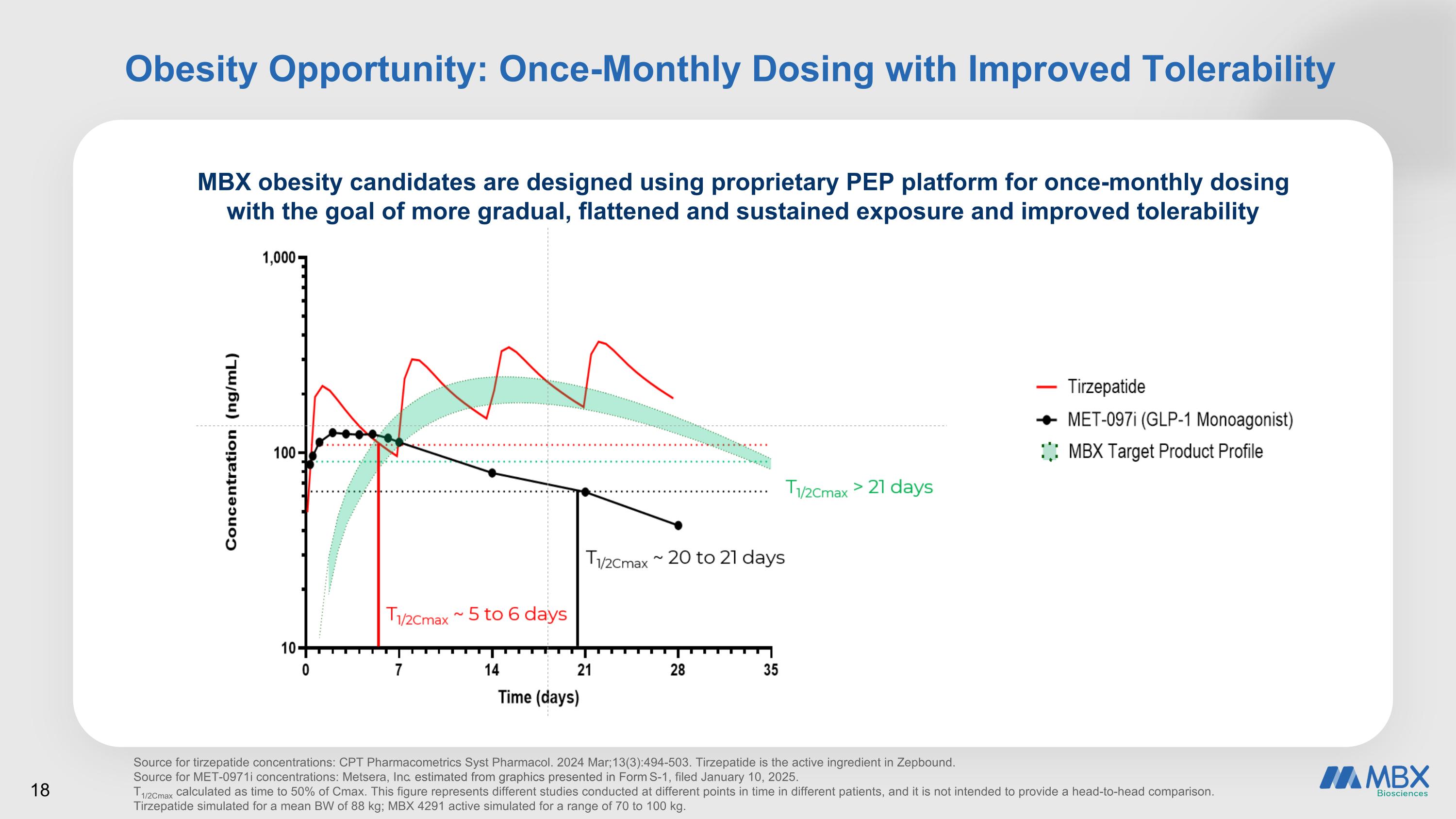
Obesity Opportunity: Once-Monthly Dosing with Improved Tolerability MBX obesity candidates are designed using proprietary PEP platform for once-monthly dosing with the goal of more gradual, flattened and sustained exposure and improved tolerability Source for tirzepatide concentrations: CPT Pharmacometrics Syst Pharmacol. 2024 Mar;13(3):494-503. Tirzepatide is the active ingredient in Zepbound. Source for MET-0971i concentrations: Metsera, Inc. estimated from graphics presented in Form S-1, filed January 10, 2025. T1/2Cmax calculated as time to 50% of Cmax. This figure represents different studies conducted at different points in time in different patients, and it is not intended to provide a head-to-head comparison. Tirzepatide simulated for a mean BW of 88 kg; MBX 4291 active simulated for a range of 70 to 100 kg.

SAD (Part A) MAD (Part B) MAD (Part C) Participants BMI > 30 Design N=40 N=24 N=60 5 Cohorts 3 Cohorts 2 Cohorts1 X X X Single dose (15 mg to 180 mg) 4 weekly doses followed by a single monthly dose 4 weekly doses followed by once monthly dosing for a total of 12 weeks MBX 4291 Ongoing Phase 1 Clinical Trial Primary: Assess safety and tolerability focusing on competitive gastrointestinal tolerability and streamlined dose titration Secondary: Determine pharmacokinetics suitable for monthly injection schedule Evaluate pharmacodynamics (i.e., through assessment of weight loss) Identify doses and titration regimen for Phase 2 Study Objectives MBX 4291 Placebo N=6 N=2 N=6 N=2 N=20 N=10 1 Planned second cohort added to evaluate additional doses/dosing regimens
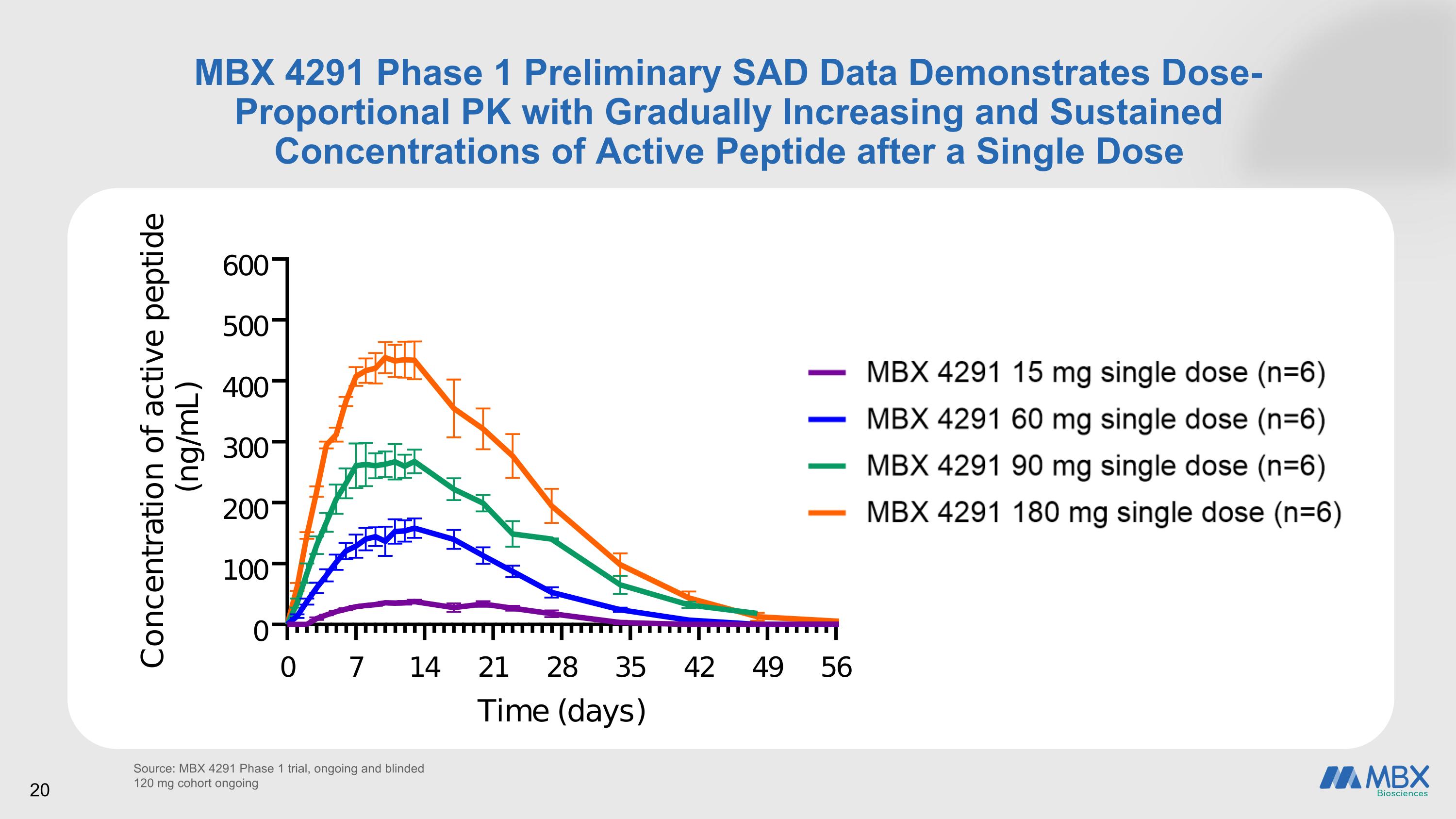
MBX 4291 Phase 1 Preliminary SAD Data Demonstrates Dose-Proportional PK with Gradually Increasing and Sustained Concentrations of Active Peptide after a Single Dose Source: MBX 4291 Phase 1 trial, ongoing and blinded 120 mg cohort ongoing
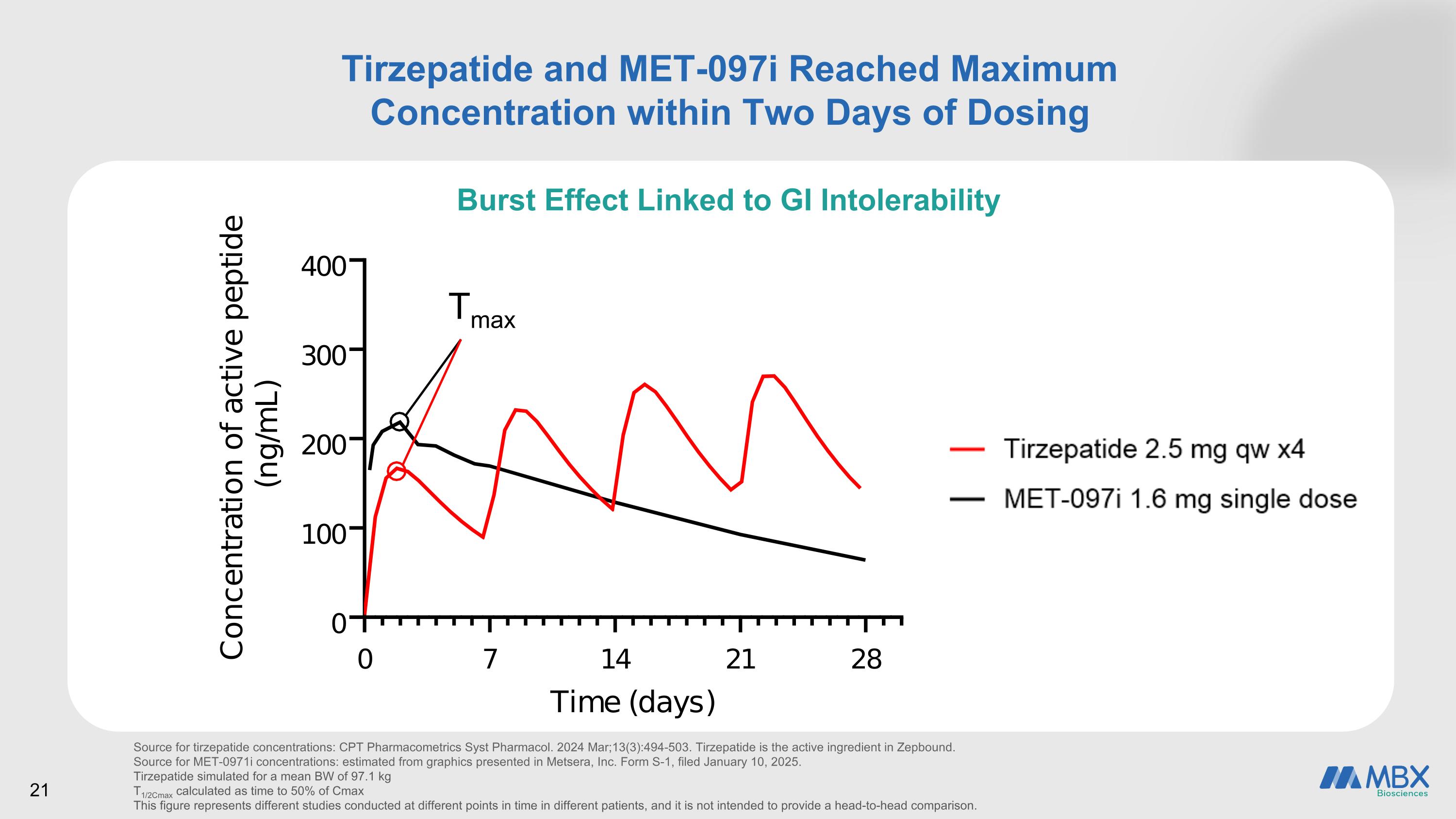
Tirzepatide and MET-097i Reached Maximum Concentration within Two Days of Dosing Tmax Source for tirzepatide concentrations: CPT Pharmacometrics Syst Pharmacol. 2024 Mar;13(3):494-503. Tirzepatide is the active ingredient in Zepbound. Source for MET-0971i concentrations: estimated from graphics presented in Metsera, Inc. Form S-1, filed January 10, 2025. Tirzepatide simulated for a mean BW of 97.1 kg T1/2Cmax calculated as time to 50% of Cmax This figure represents different studies conducted at different points in time in different patients, and it is not intended to provide a head-to-head comparison. 21 Burst Effect Linked to GI Intolerability
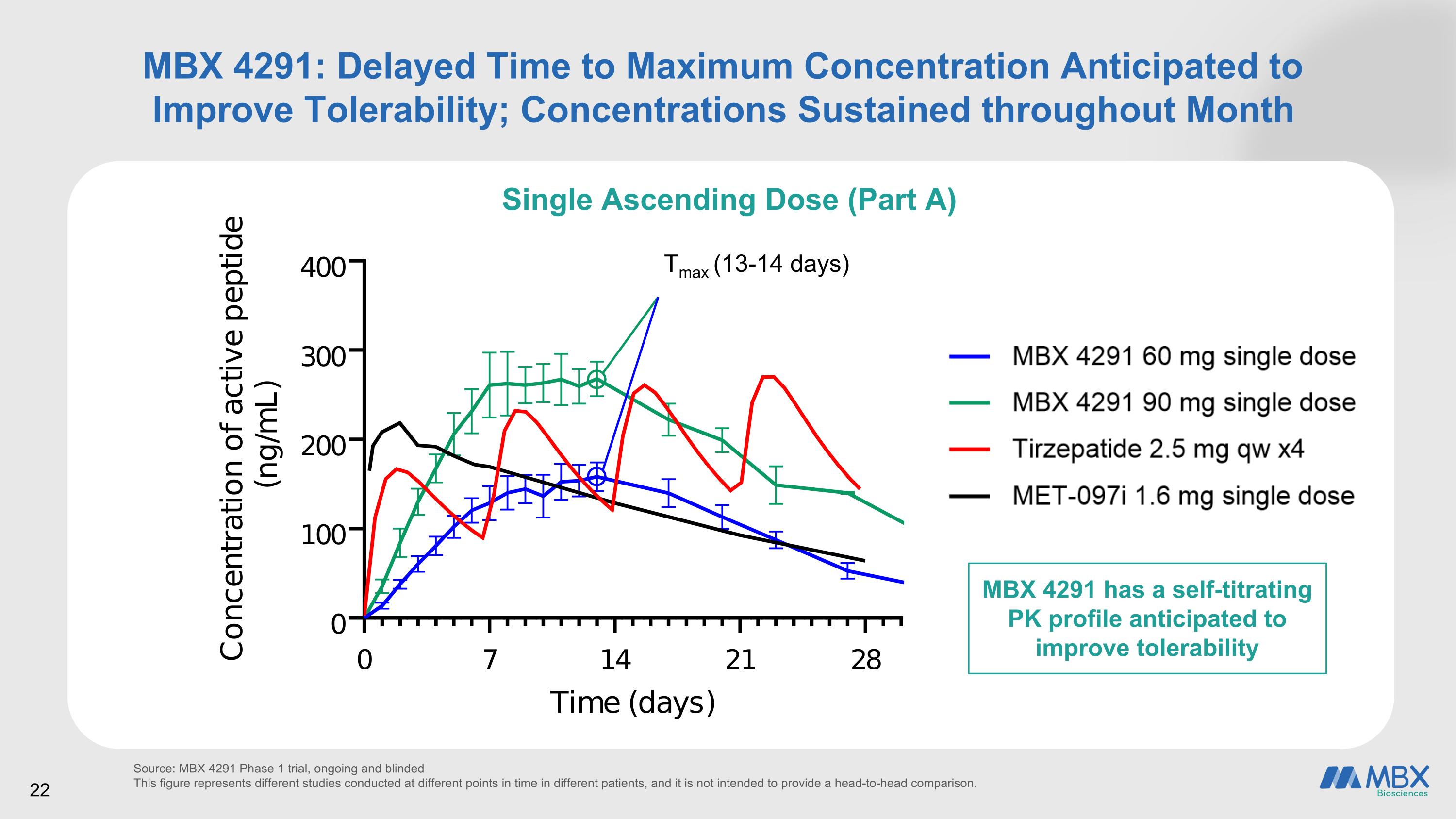
MBX 4291: Delayed Time to Maximum Concentration Anticipated to Improve Tolerability; Concentrations Sustained throughout Month Single Ascending Dose (Part A) Source: MBX 4291 Phase 1 trial, ongoing and blinded This figure represents different studies conducted at different points in time in different patients, and it is not intended to provide a head-to-head comparison. Tmax (13-14 days) 22 MBX 4291 has a self-titrating PK profile anticipated to improve tolerability

MBX 4291: Dose-Dependent GI-Related Adverse Events Were Predominantly Mild SAD All SAD 15 mg (N=8, incl pbo) 60 mg (N=8, incl pbo) 90 mg (N=8, incl pbo) 180 mg (N=8, incl pbo) N=32, incl pbo Number of Subjects with Treatment-Emergent Events of Nausea, Vomiting or Diarrhea 1 (12.5%) 0 3 (37.5%) 7 (87.5%) 11 (34%) Nausea (mild/moderate/severe) 0 0 1/1/0 (25%) 6/1/0 (87.5%) 7/2/0 (28%) Vomiting (mild/moderate/severe) 0 0 1/0/0 (12.5%) 4/1/0 (62.5%) 5/1/0 (19%) Diarrhea (mild/moderate/severe) 1/0/0 (12.5%) 0 2/0/0 (25%) 3/0/0 (37.5%) 6/0/0 (19%) Source: MBX 4291 Phase 1 trial, ongoing and blinded pbo = placebo 15 mg → 90 mg single doses were generally well tolerated, with low incidence of GI-related adverse events that were predominantly mild MTD reached with 180 mg single dose → associated with GI AEs in 7/8 participants, mostly mild 120 mg cohort ongoing Single Ascending Dose (Part A) 23
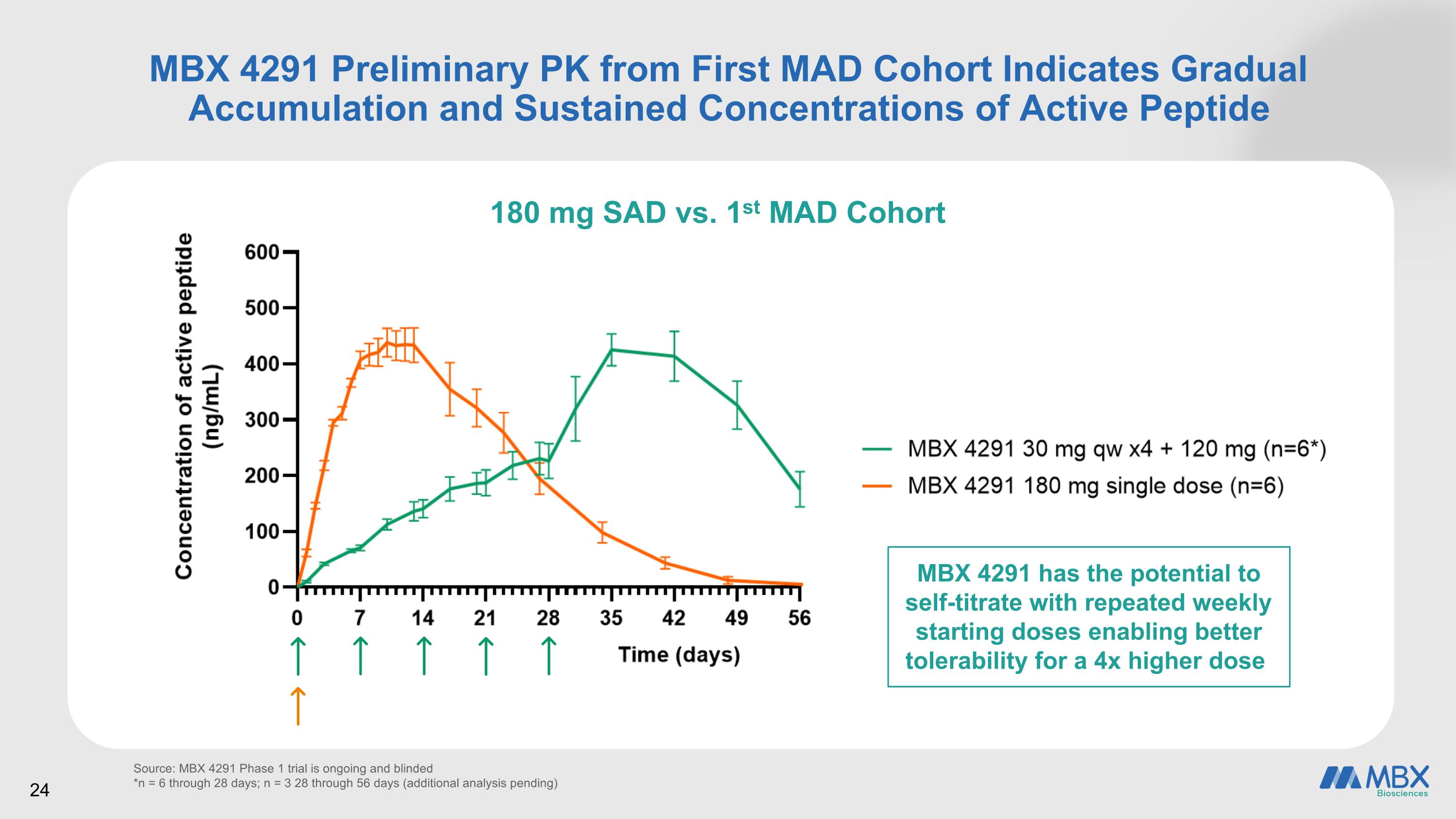
MBX 4291 Preliminary PK from First MAD Cohort Indicates Gradual Accumulation and Sustained Concentrations of Active Peptide Source: MBX 4291 Phase 1 trial is ongoing and blinded *n = 6 through 28 days; n = 3 28 through 56 days (additional analysis pending) 180 mg SAD vs. 1st MAD Cohort 24 MBX 4291 has the potential to self-titrate with repeated weekly starting doses enabling better tolerability for a 4x higher dose
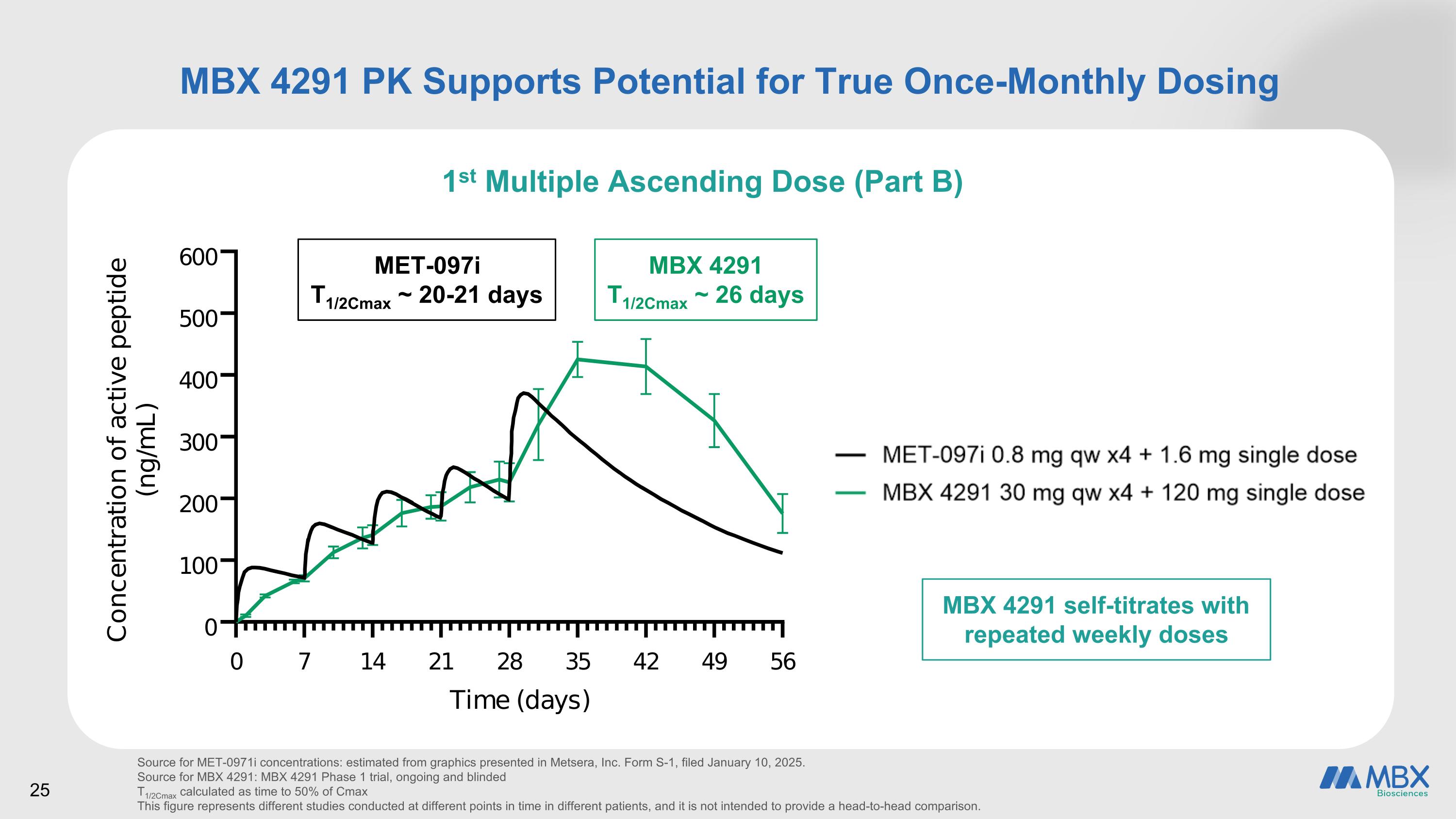
MBX 4291 PK Supports Potential for True Once-Monthly Dosing 1st Multiple Ascending Dose (Part B) Source for MET-0971i concentrations: estimated from graphics presented in Metsera, Inc. Form S-1, filed January 10, 2025. Source for MBX 4291: MBX 4291 Phase 1 trial, ongoing and blinded T1/2Cmax calculated as time to 50% of Cmax This figure represents different studies conducted at different points in time in different patients, and it is not intended to provide a head-to-head comparison. MET-097i T1/2Cmax ~ 20-21 days MBX 4291 T1/2Cmax ~ 26 days MBX 4291 self-titrates with repeated weekly doses 25

Mean weight loss of 7% (range 0-16%) at 8 weeks (n=8, including 2 placebo) MBX 4291 First MAD Cohort: Competitive Weight Loss and Tolerability with Potential for True Once-Monthly Dosing 30 mg qw x4 + 120 mg single dose Safety and Tolerability Only 1/8 subjects experienced an event of diarrhea, nausea or vomiting through 8 weeks One event of mild diarrhea following the first administration No nausea No vomiting No serious adverse events Source: MBX 4291 Phase 1 trial, ongoing and blinded

MBX 4291 Initial Phase 1 Data Summary MBX 4291 designed for once-monthly dosing with controlled, sustained concentrations and improved tolerability Data from the Phase 1 SAD show a PK profile supporting a self-titrating weekly induction regimen and potential for a true once-monthly regimen, including: Dose-proportional PK Sustained concentrations of active peptide for 28 days Dose-dependent GI-related adverse events across 4 dose cohorts, ranging from 15 to 180 mg Preliminary blinded data from the first Phase 1 MAD cohort following 4 weekly induction doses of 30 mg and a single 120 mg once-monthly dose indicates: Gradual accumulation of active peptide Mean weight loss of 7% (range 0-16%) at 8 weeks (n=8, 6 active, 2 placebo) Only one event of diarrhea, nausea or vomiting through 8 weeks 12-week Phase 1 MAD (Part C) results remain on track for Q4 2026

MBX 5765 Amycretin Richard DiMarchi, PhD MBX Scientific Co-Founder Distinguished Professor of Chemistry and Gill Chair in Biomolecular Sciences at Indiana University

GLP-1 GIP GCG DACRA (amylin + calcitonin) Improved efficacy compared to CagriSema or Semaglutide, alone or as mix Improved tolerability; it’s a prodrug Potential for a more simplified approval path compared to a mix of novel agonists MBX 5765: Multi-Agonist Amycretin Prodrug Designed for Once-Monthly Dosing, Superior Efficacy and Improved Tolerability Potency at the receptors equal to or greater than endogenous ligand except GCG DACRA = dual amylin and calcitonin receptor agonist Multi-Receptor Agonist Profile Enhanced Preclinical Potency vs. Benchmark 29
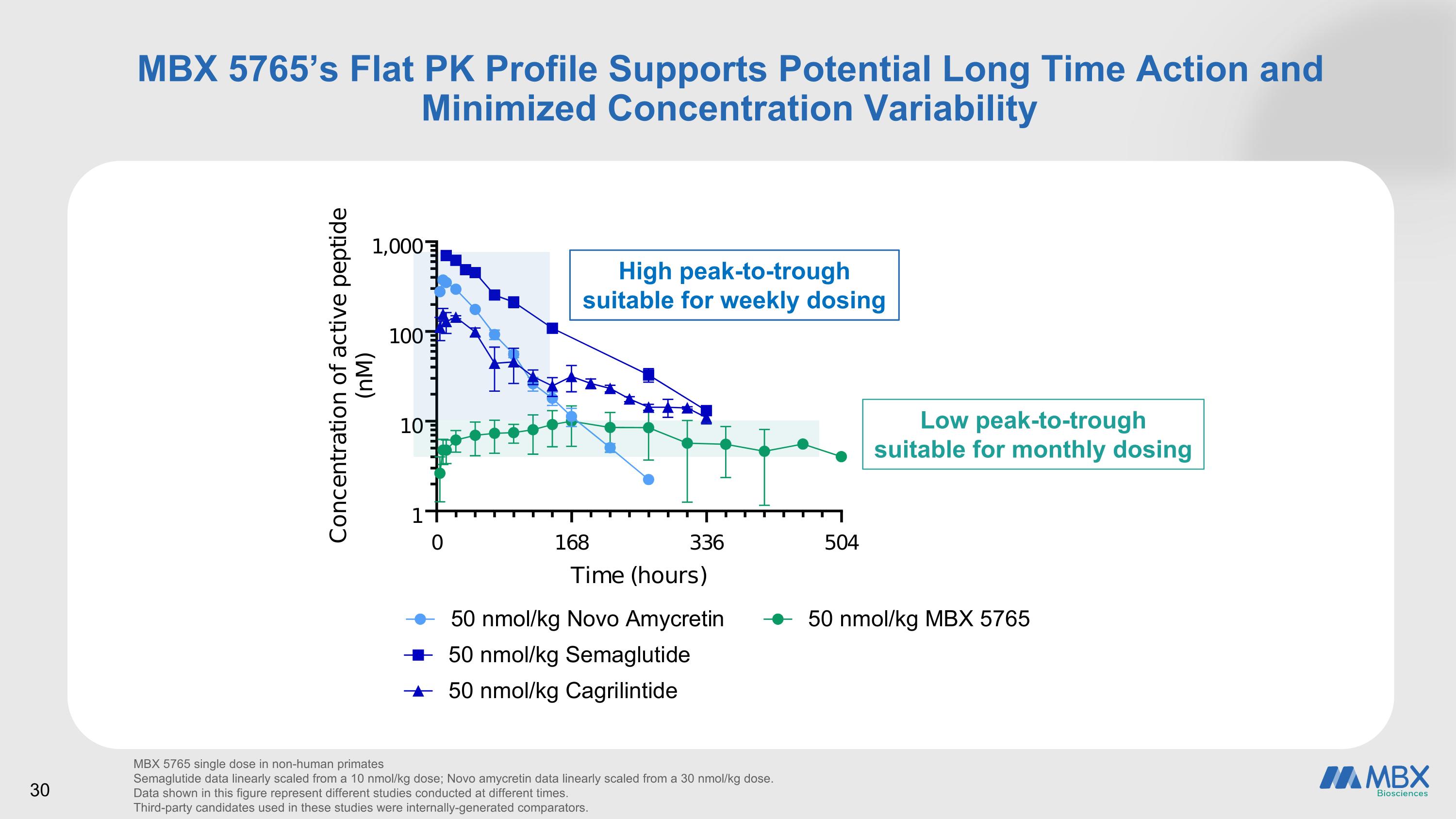
30 we MBX 5765’s Flat PK Profile Supports Potential Long Time Action and Minimized Concentration Variability MBX 5765 single dose in non-human primates Semaglutide data linearly scaled from a 10 nmol/kg dose; Novo amycretin data linearly scaled from a 30 nmol/kg dose. Data shown in this figure represent different studies conducted at different times. Third-party candidates used in these studies were internally-generated comparators. High peak-to-trough suitable for weekly dosing Low peak-to-trough suitable for monthly dosing
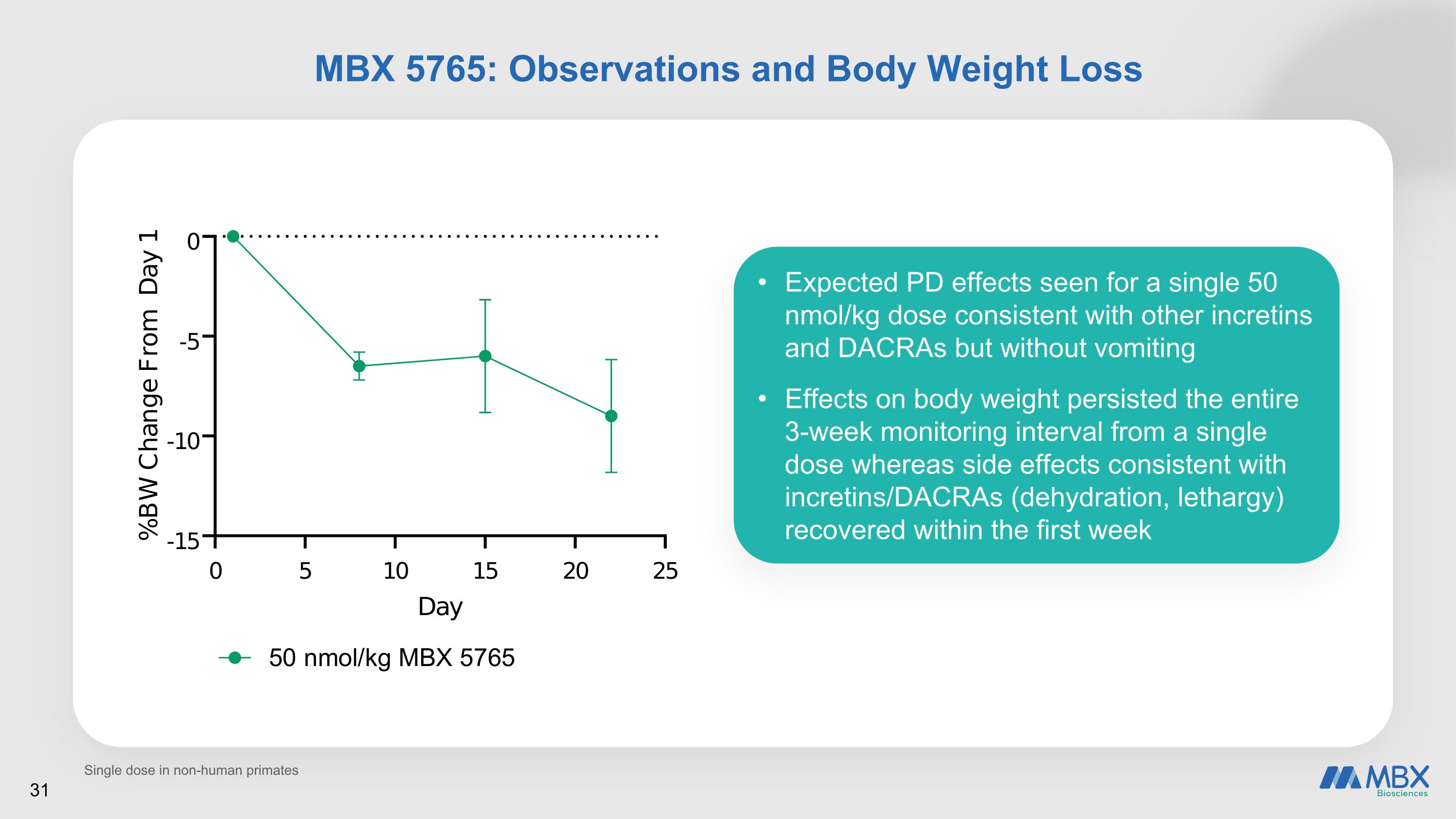
MBX 5765: Observations and Body Weight Loss Expected PD effects seen for a single 50 nmol/kg dose consistent with other incretins and DACRAs but without vomiting Effects on body weight persisted the entire 3-week monitoring interval from a single dose whereas side effects consistent with incretins/DACRAs (dehydration, lethargy) recovered within the first week 31 Single dose in non-human primates
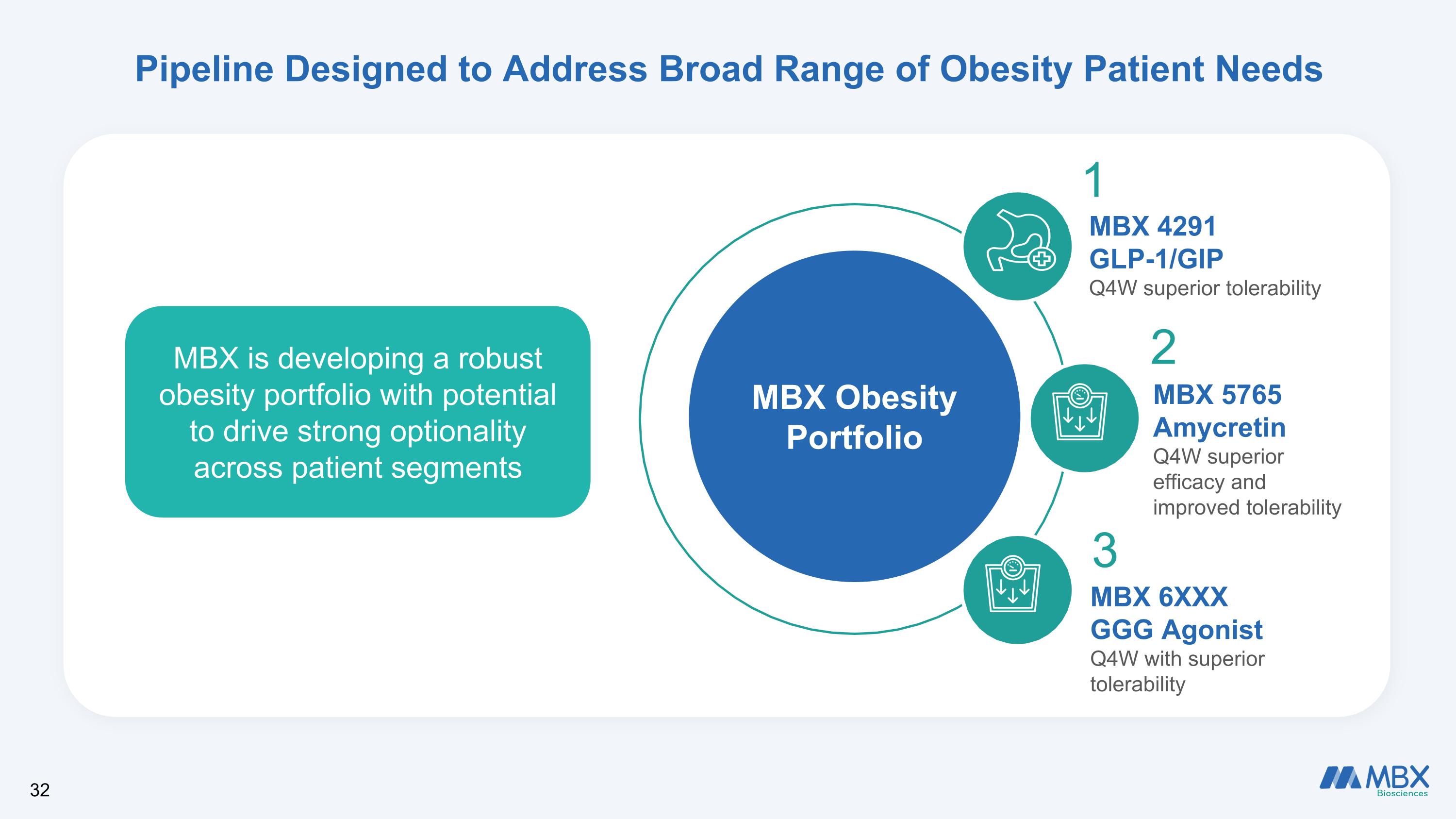
Pipeline Designed to Address Broad Range of Obesity Patient Needs MBX Obesity Portfolio MBX 4291 GLP-1/GIP Q4W superior tolerability MBX 5765 Amycretin Q4W superior efficacy and improved tolerability MBX 6XXX GGG Agonist Q4W with superior tolerability 1 2 3 MBX is developing a robust obesity portfolio with potential to drive strong optionality across patient segments 32

Conclusion Kent Hawryluk President & CEO
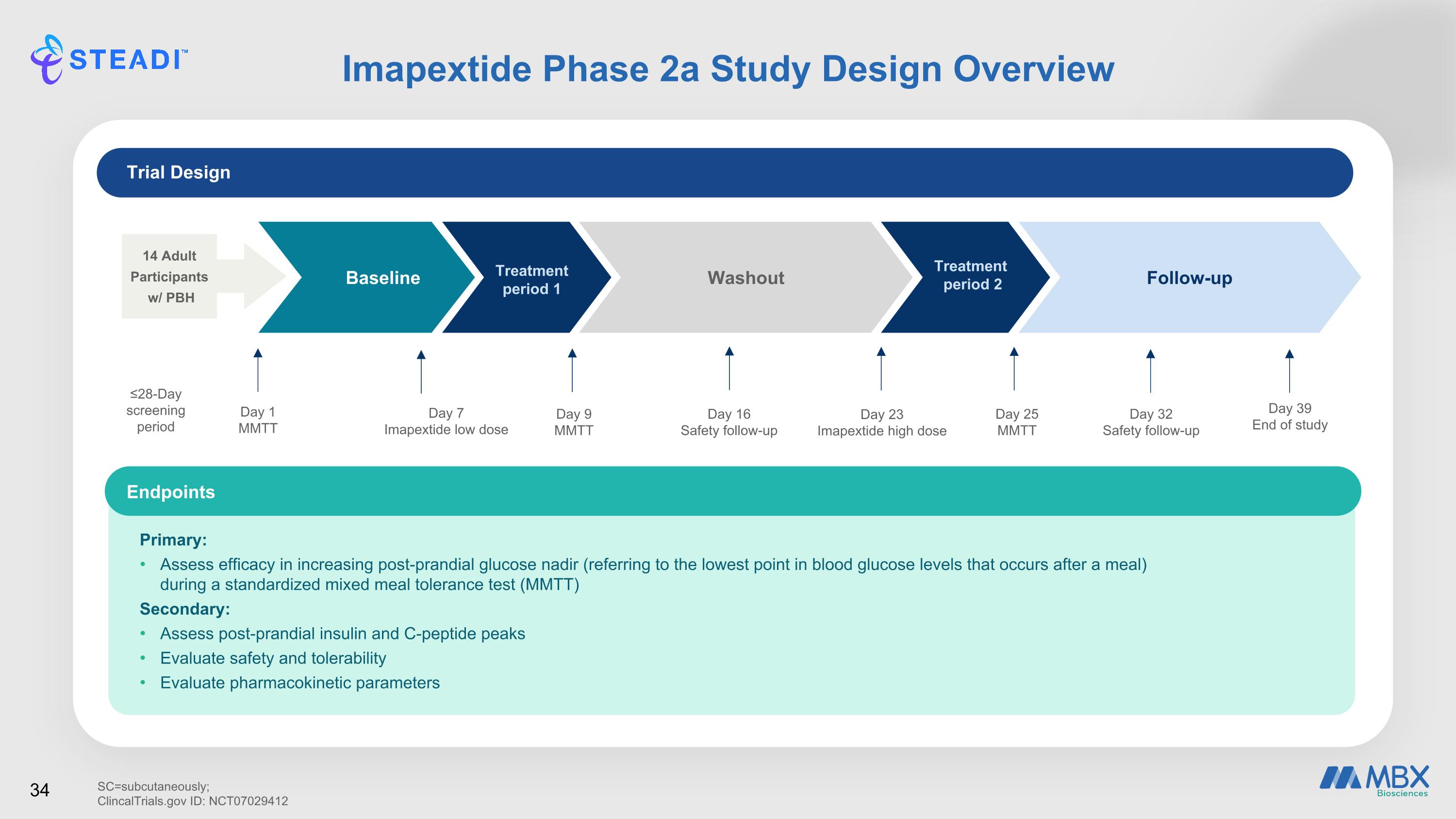
Imapextide Phase 2a Study Design Overview 14 Adult Participants w/ PBH Baseline Day 1 MMTT Treatment period 1 Day 7 Imapextide low dose Day 9 MMTT Washout Day 16 Safety follow-up Day 23 Imapextide high dose Day 25 MMTT Follow-up Day 32 Safety follow-up Day 39 End of study Treatment period 2 ≤28-Day screening period 34 Trial Design Primary: Assess efficacy in increasing post-prandial glucose nadir (referring to the lowest point in blood glucose levels that occurs after a meal) during a standardized mixed meal tolerance test (MMTT) Secondary: Assess post-prandial insulin and C-peptide peaks Evaluate safety and tolerability Evaluate pharmacokinetic parameters Endpoints SC=subcutaneously; ClincalTrials.gov ID: NCT07029412
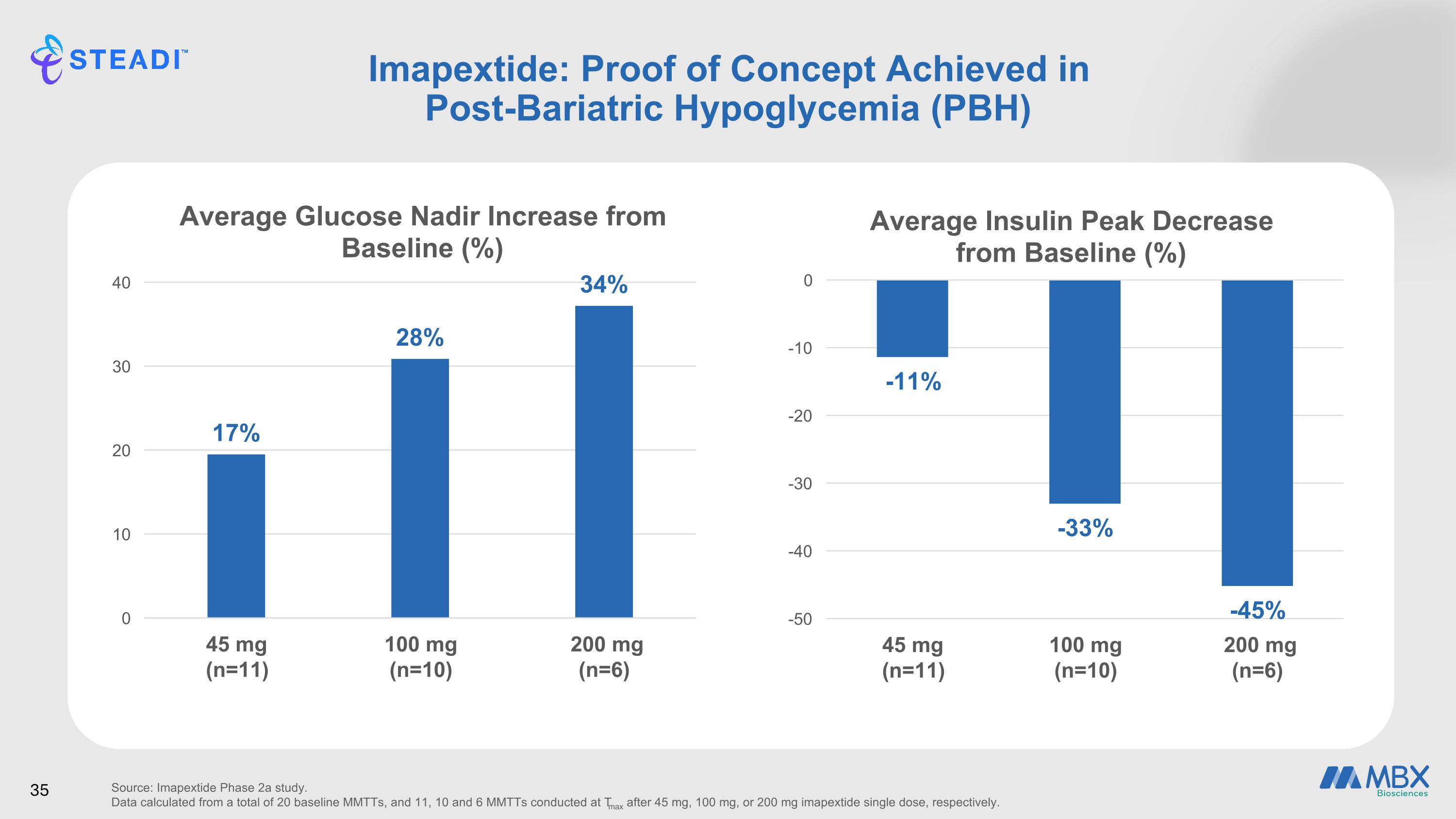
Imapextide: Proof of Concept Achieved in Post-Bariatric Hypoglycemia (PBH) Source: Imapextide Phase 2a study. Data calculated from a total of 20 baseline MMTTs, and 11, 10 and 6 MMTTs conducted at Tmax after 45 mg, 100 mg, or 200 mg imapextide single dose, respectively.

Expanding Pipeline of Novel Peptide-Based Drug Candidates Preliminary, blinded MBX 4291 data further demonstrate potential of PEP™ platform: PK supports potential for true once-monthly dosing Mean weight loss of 7% (range 0-16%) at 8 weeks in first MAD Part B cohort (n=8, including 2 placebo) MBX 4291 generally well tolerated; only one event of diarrhea, nausea or vomiting in first MAD Part B cohort MBX 4291 12-week MAD Part C results remain on track for Q4 2026 MBX 5765 nominated as amycretin prodrug development candidate designed for once-monthly dosing, superior efficacy and improved tolerability Prioritized resource and capital allocation to canvuparatide and obesity programs provides strongest opportunity to deliver long-term value

$440 million in cash expected to provide runway into 20291 Program Milestone Anticipated Timing Canvuparatide (MBX 2109) End-of-Phase 2 FDA Meeting Avail™ Phase 2 presentation and one-year OLE data ENDO 2026 Phase 3: Initiation Q3 2026 MBX 4291 (GLP-1/GIP) Phase 1: 12-week MAD results Q4 2026 MBX 5765 (amycretin) Nominate development candidate MBX 6XXX (GLP-1/GIP/GCGR) Nominate development candidate Q3 2026 Imapextide (MBX 1416) STEADI™ Phase 2a: Results 1 Unaudited cash, cash equivalents and marketable securities as of March 31, 2026 MBX: Catalyst-Rich Year

Q&A Thank You