
Exhibit 99.1

Revolutionary Alpha-Emitters Radiotherapy: Brain Tumors 1

2 This presentation (together with oral statements made in connection herewith, the "Presentation") is for informational purposes only to assist interested parties in making their own evaluation with respect to Alpha Tau Medical Ltd. ("Alpha Tau" or the "Company"). The information contained herein does not purport to be all-inclusive and neither the Company nor any of its respective subsidiaries, stockholders, shareholders, affiliates, representatives, control persons, partners, directors, officers, employees, advisers or agents make any representation or warranty, express or implied, as to the accuracy, completeness or reliability of the information contained in this Presentation. You should consult your own counsel and tax and financial advisors as to legal and related matters concerning the matters described herein, and, by accepting this Presentation, you confirm that you are not relying upon the information contained herein to make any decision. The reader shall not rely upon any statement, representation or warranty made by any other person, firm or corporation in making its investment or decision to invest in the Company. To the fullest extent permitted by law, in no circumstances will the Company or any of its subsidiaries, stockholders, shareholders, affiliates, representatives, control persons, partners, directors, officers, employees, advisers or agents be responsible or liable for any direct, indirect or consequential loss or loss of profit arising from the use of this Presentation, its contents, its omissions, reliance on the information contained within it, or on opinions communicated in relation thereto or otherwise arising in connection therewith. In addition, this Presentation does not purport to be all-inclusive or to contain all of the information that may be required to make a full analysis of the Company. The general explanations included in this Presentation cannot address, and are not intended to address, your specific investment objectives, financial situations or financial needs. Use of Data Certain information contained in this Presentation relates to or is based on studies, publications, surveys and the Company's own internal estimates and research. In addition, all of the market data included in this Presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while the Company believes its internal research is reliable, such research has not been verified by any independent source and none of the Company nor any of its affiliates nor any of its control persons, officers, directors, employees or representatives make any representation or warranty with respect to the accuracy of such information. The information contained in this Presentation is not intended to predict actual results, and no assurances are given with respect thereto. Past performance is no indication, guarantee or representation as to future events, results or performance. Forward-Looking Statements This presentation contains forward-looking statements, including without limitation, statements related to: the progress, timing and results of our clinical trials, including the results from trials of Alpha DaRT conducted by the Company, and the safety and efficacy of our development programs. Forward-looking statements can be identified by the words "believe," "anticipate," "continue," "estimate "project," "expect," "plan," "potential," "intends," "will," "would," "could," "should" or the negative or plural of these words or other similar expressions that are predictions or indicate future events, trends or prospects but the absence of these words does not necessarily mean that a statement is not forward-looking. In addition, any statements that refer to expectations, projections or other characterizations of future events or circumstances are forward-looking statements. Forward-looking statements are subject to risks and uncertainties that could cause actual results to differ materially and reported results should not be considered as an indication of future performance. Actual results and the timing of events could differ materially from those anticipated in the forward-looking statements as a result of these risks and uncertainties, which include, among others, those inherent in the preclinical and clinical development process and the regulatory approval process, the risks and uncertainties in commercialization and gaining market acceptance, the risks associated with protecting and defending our patents or other proprietary rights, the risk that our proprietary rights may be insufficient to protect our development programs, the risk that we will be unable to obtain necessary capital when needed on acceptable terms or at all, competition from other products or procedures, our reliance on third-parties to conduct our clinical and non-clinical trials, our reliance on any third-party suppliers to manufacture clinical, non-clinical and any future commercial supplies of our products, and increased regulatory requirements. These statements are subject to the risk that clinical trial data are subject to differing interpretations, and regulatory agencies, medical and scientific experts and others may not share Alpha Tau's views of the clinical study data. There can be no assurance that the clinical studies for our development programs will be successful in demonstrating safety and/or efficacy, that we will not encounter problems or delays in clinical development, or that any of our products will ever receive regulatory approval or be successfully commercialized. These forward-looking statements are based on information available to Alpha Tau as of the date of this presentation and speak only as of the date of this presentation. Alpha Tau disclaims any obligation to update these forward-looking statements, except as may be required by law. This presentation is for informational purposes only and does not constitute an offer to sell or a solicitation of an offer to purchase any securities of any nature whatsoever, and it may not be relied upon in connection with the purchase of securities. Trademarks This presentation may contain trademarks, service marks, trade names and copyrights of other companies, which are the property of their respective owners. Solely for convenience, some of the trademarks, service marks, trade names and copyrights referred to in this presentation may be listed without the TM, SM © or ® symbols, but Alpha Tau will assert, to the fullest extent under applicable law, the rights of the applicable owners, if any, to these trademarks, service marks, trade names and copyrights. Disclaimer
Today's Presenters 3 Raphi Levy CFO Alpha Tau Joshua Palmer, MD Professor, Radiation Oncology Ohio State University Comprehensive Cancer Center – The James Robert Den, MD CMO Alpha Tau Uzi Sofer CEO Alpha Tau

• Alpha DaRT Mechanism of Action • Preclinical Data • Clinical Approach • Clinical Trial 4 Agenda
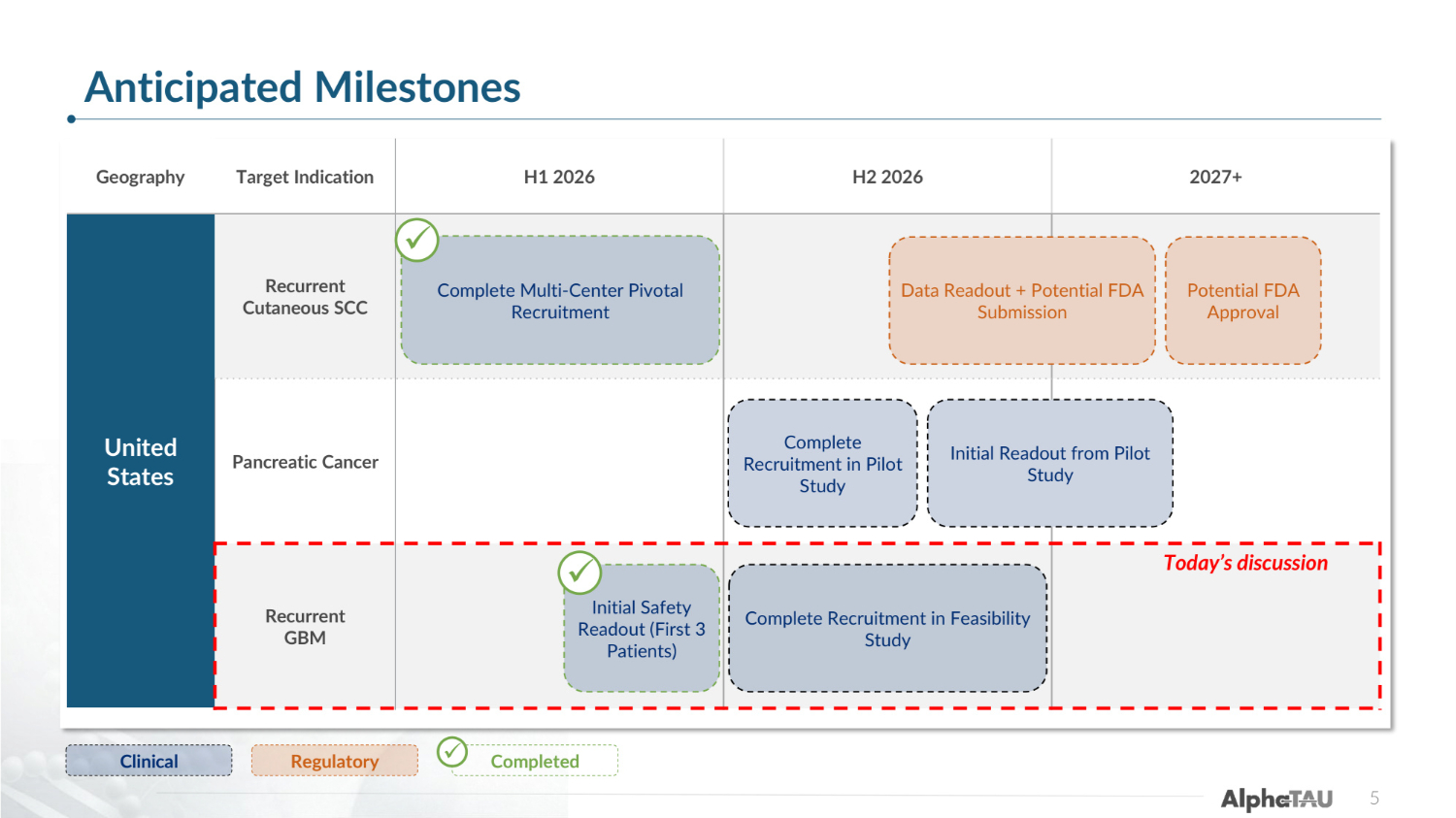
Geography Target Indication H1 2026 H2 2026 2027+ United States Recurrent Cutaneous SCC Pancreatic Cancer Recurrent GBM Clinical Regulatory Complete Multi-Center Pivotal Recruitment 5 Data Readout + Potential FDA Submission Complete Recruitment in Pilot Study Complete Recruitment in Feasibility Study Initial Safety Readout (First 3 Patients) ✓ Completed ✓ ✓ Initial Readout from Pilot Study Potential FDA Approval Today's discussion Anticipated Milestones

Alpha DaRT: Mechanism of Action 6

Types of Radioactive Decay Due to the mass of the alpha particle, in comparison to beta particle, alpha has a low penetration power. This means that the outside layer of the human skin, for example, can block these particles. 7
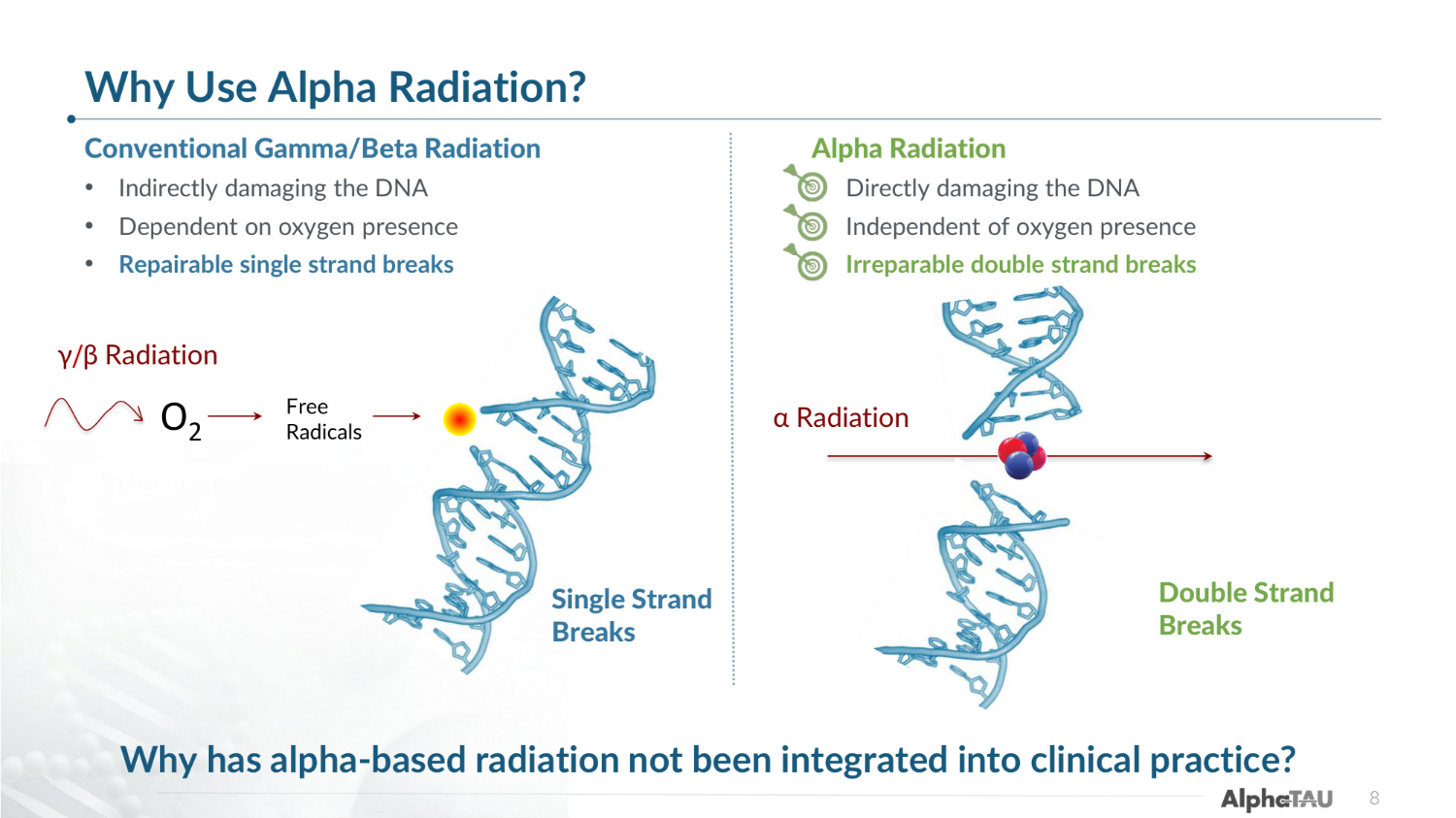
8 Conventional Gamma/Beta Radiation • Indirectly damaging the DNA • Dependent on oxygen presence • Repairable single strand breaks Alpha Radiation • Directly damaging the DNA • Independent of oxygen presence • Irreparable double strand breaks γ/β Radiation Single Strand Breaks Double Strand Breaks α Radiation O2 Free Radicals Why has alpha-based radiation not been integrated into clinical practice? Why Use Alpha Radiation?
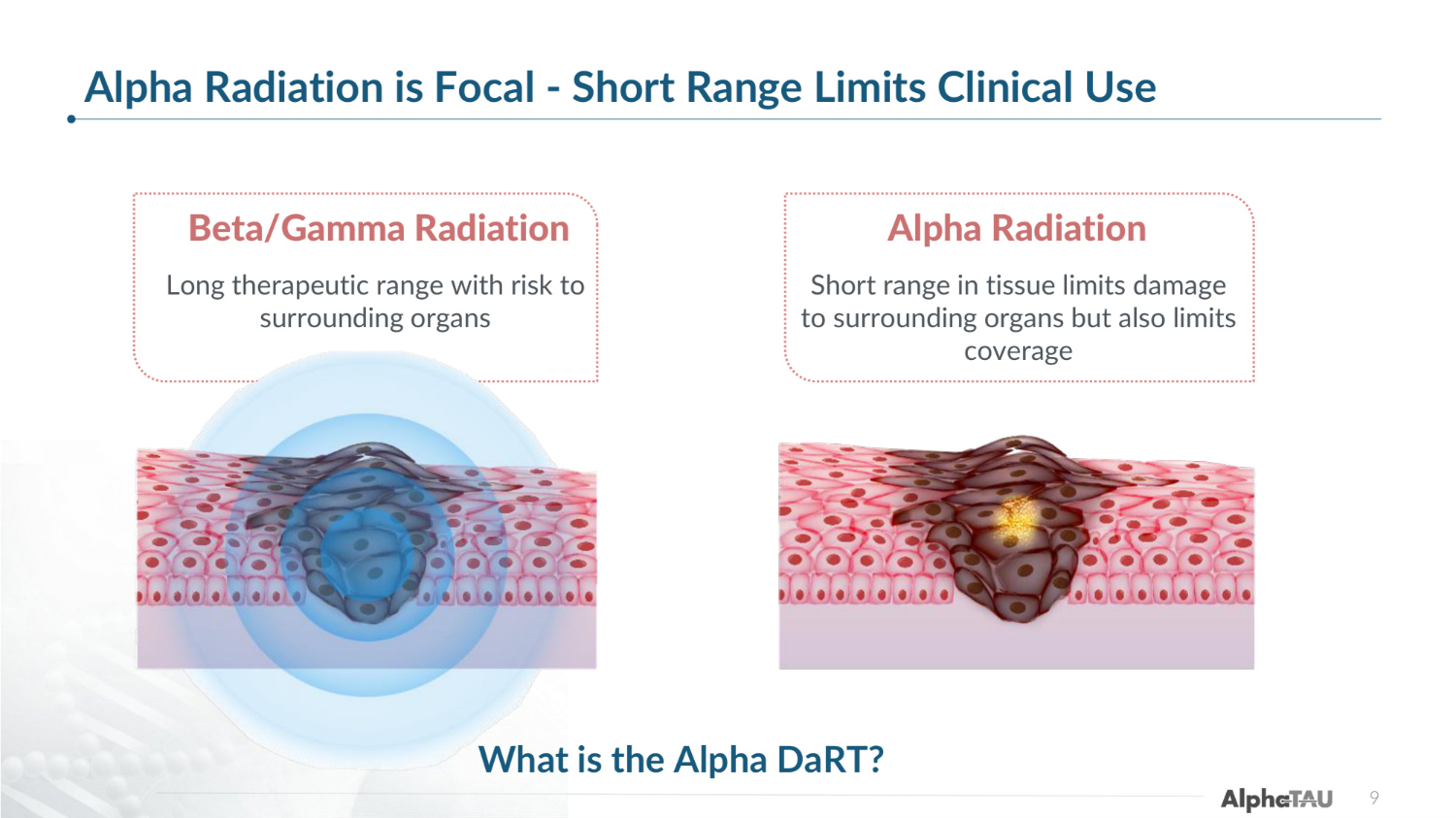
Healthy Tissue Healthy Tissue Alpha Radiation is Focal - Short Range Limits Clinical Use 9 Alpha Radiation Short range in tissue limits damage to surrounding organs but also limits coverage Beta/Gamma Radiation Long therapeutic range with risk to surrounding organs What is the Alpha DaRT?

Alpha DaRT - Diffusing Alpha-emitters Radiation Therapy https://www.youtube.com/watch?v=nwfzJHm0fTQ 10
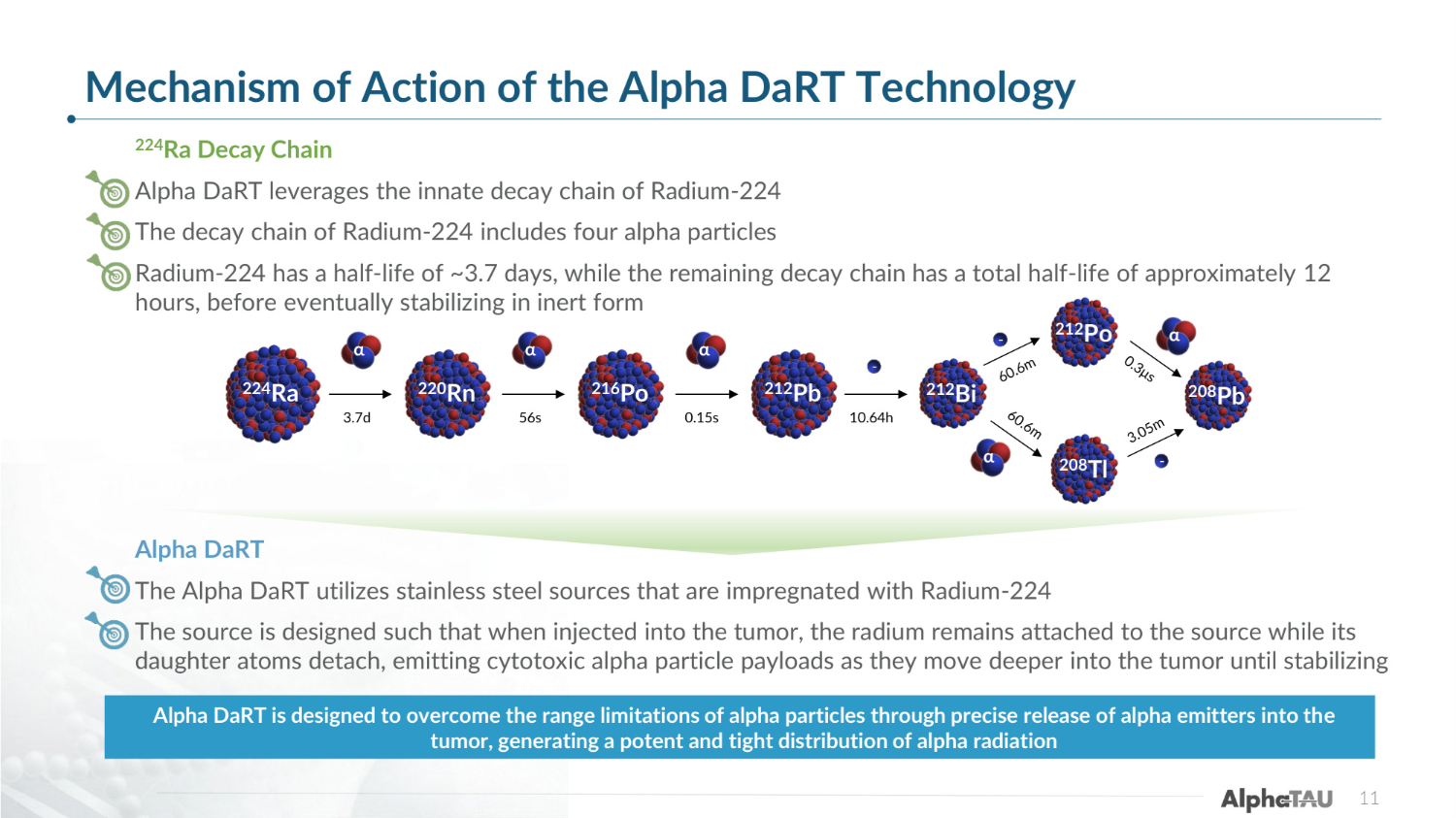
Mechanism of Action of the Alpha DaRT Technology 224Ra Decay Chain Alpha DaRT leverages the innate decay chain of Radium-224 The decay chain of Radium-224 includes four alpha particles Radium-224 has a half-life of ~3.7 days, while the remaining decay chain has a total half-life of approximately 12 hours, before eventually stabilizing in inert form Alpha DaRT The Alpha DaRT utilizes stainless steel sources that are impregnated with Radium-224 The source is designed such that when injected into the tumor, the radium remains attached to the source while its daughter atoms detach, emitting cytotoxic alpha particle payloads as they move deeper into the tumor until stabilizing Alpha DaRT is designed to overcome the range limitations of alpha particles through precise release of alpha emitters into the tumor, generating a potent and tight distribution of alpha radiation α 224Ra 3.7d 220Rn 216Po 212Pb 212Bi 56s 0.15s 10.64h α α 212Po 208Pb 208Tl α α - - - 11

Brain Preclinical Studies – Small Animal 12
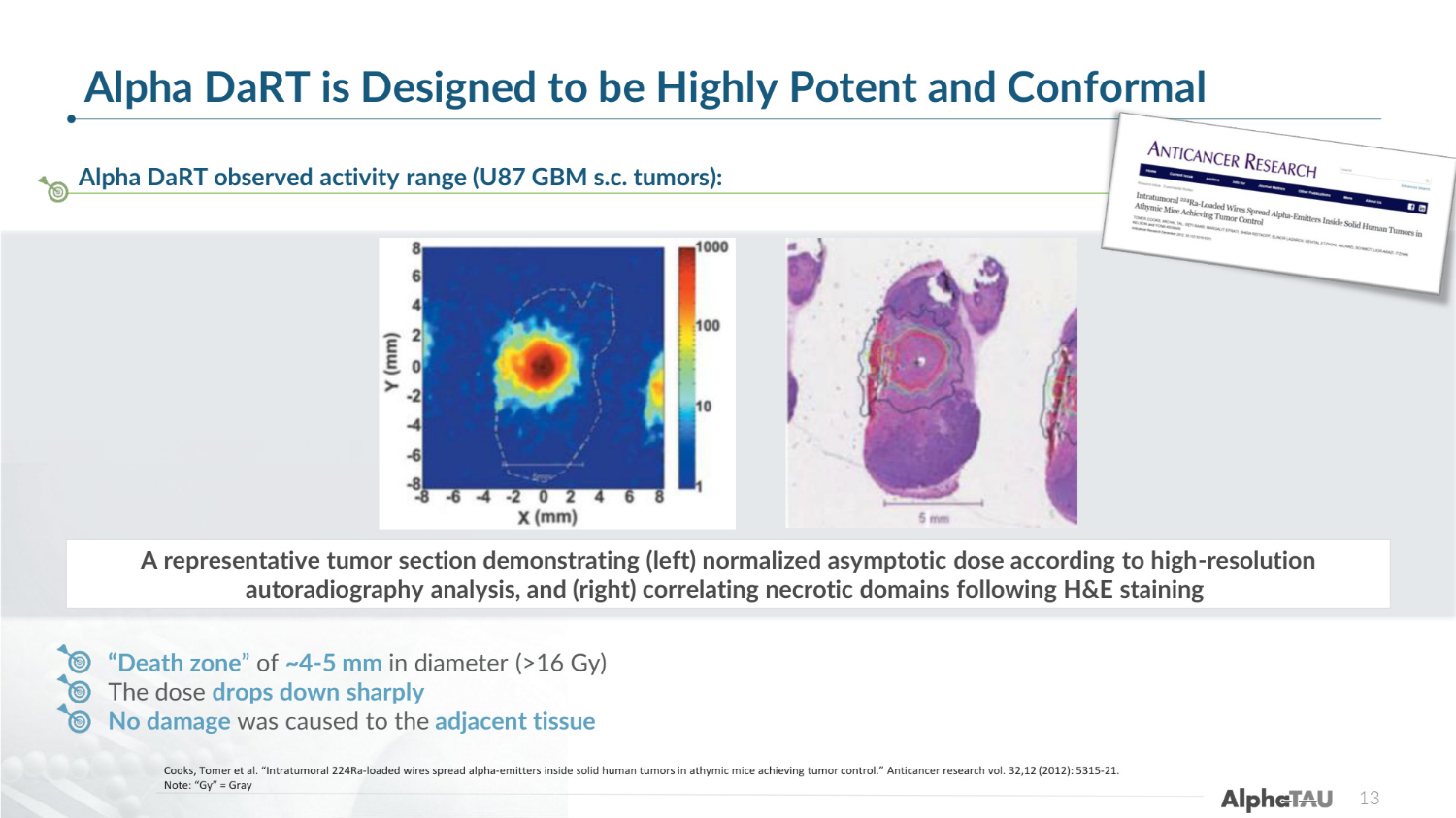
Alpha DaRT is Designed to be Highly Potent and Conformal 13 Alpha DaRT observed activity range (U87 GBM s.c. tumors): Cooks, Tomer et al. "Intratumoral 224Ra-loaded wires spread alpha-emitters inside solid human tumors in athymic mice achieving tumor control." Anticancer research vol. 32,12 (2012): 5315-21. Note: "Gy" = Gray "Death zone" of ~4-5 mm in diameter (>16 Gy) The dose drops down sharply No damage was caused to the adjacent tissue A representative tumor section demonstrating (left) normalized asymptotic dose according to high-resolution autoradiography analysis, and (right) correlating necrotic domains following H&E staining
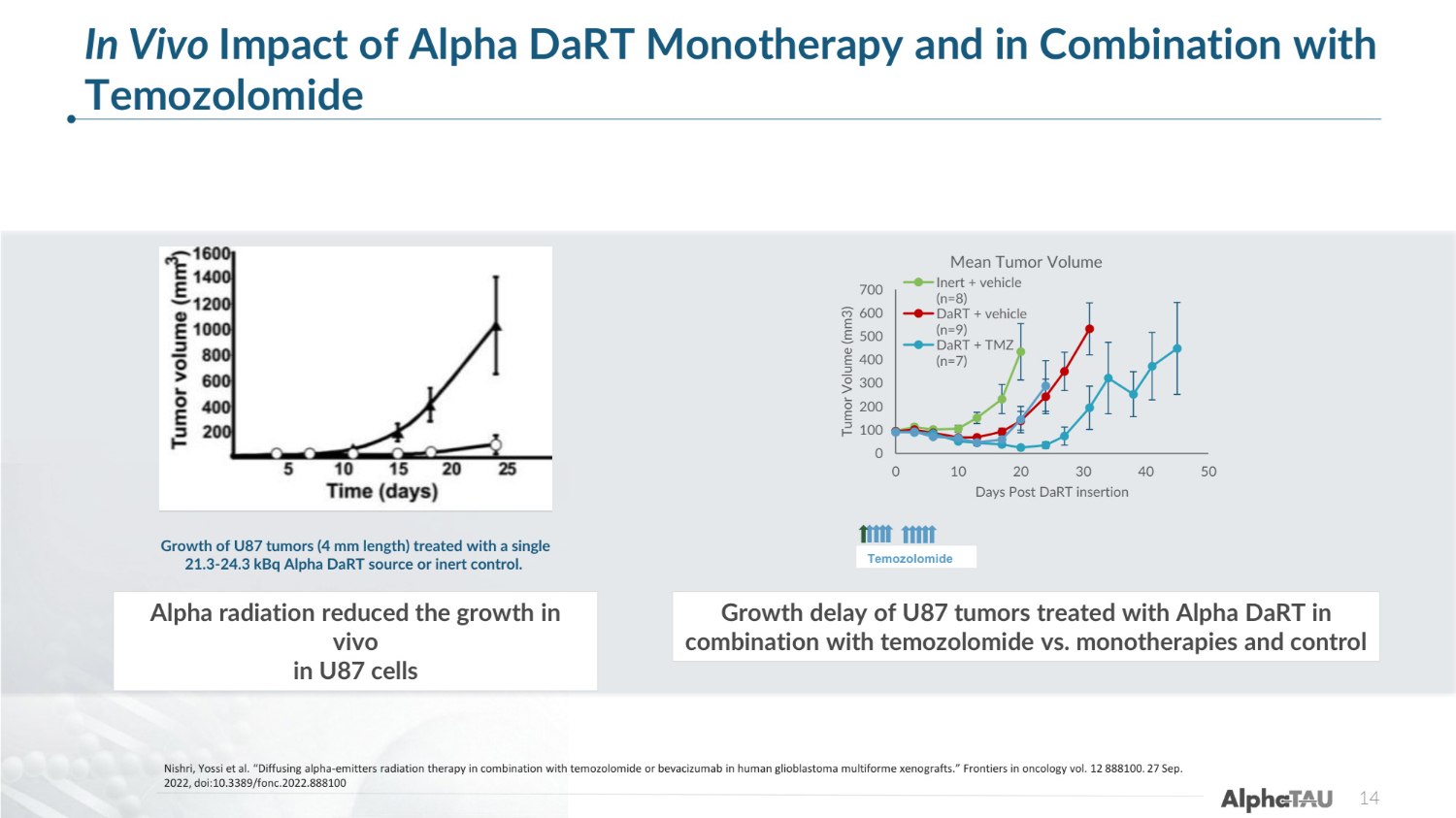
14 Nishri, Yossi et al. "Diffusing alpha-emitters radiation therapy in combination with temozolomide or bevacizumab in human glioblastoma multiforme xenografts." Frontiers in oncology vol. 12 888100. 27 Sep. 2022, doi:10.3389/fonc.2022.888100 In Vivo Impact of Alpha DaRT Monotherapy and in Combination with Temozolomide Growth of U87 tumors (4 mm length) treated with a single 21.3-24.3 kBq Alpha DaRT source or inert control. 0 100 200 300 400 500 600 700 0 10 20 30 40 50 Tumor Volume (mm3) Days Post DaRT insertion Mean Tumor Volume Inert + vehicle (n=8) DaRT + vehicle (n=9) DaRT + TMZ (n=7) Temozolomide Alpha radiation reduced the growth in vivo in U87 cells Growth delay of U87 tumors treated with Alpha DaRT in combination with temozolomide vs. monotherapies and control
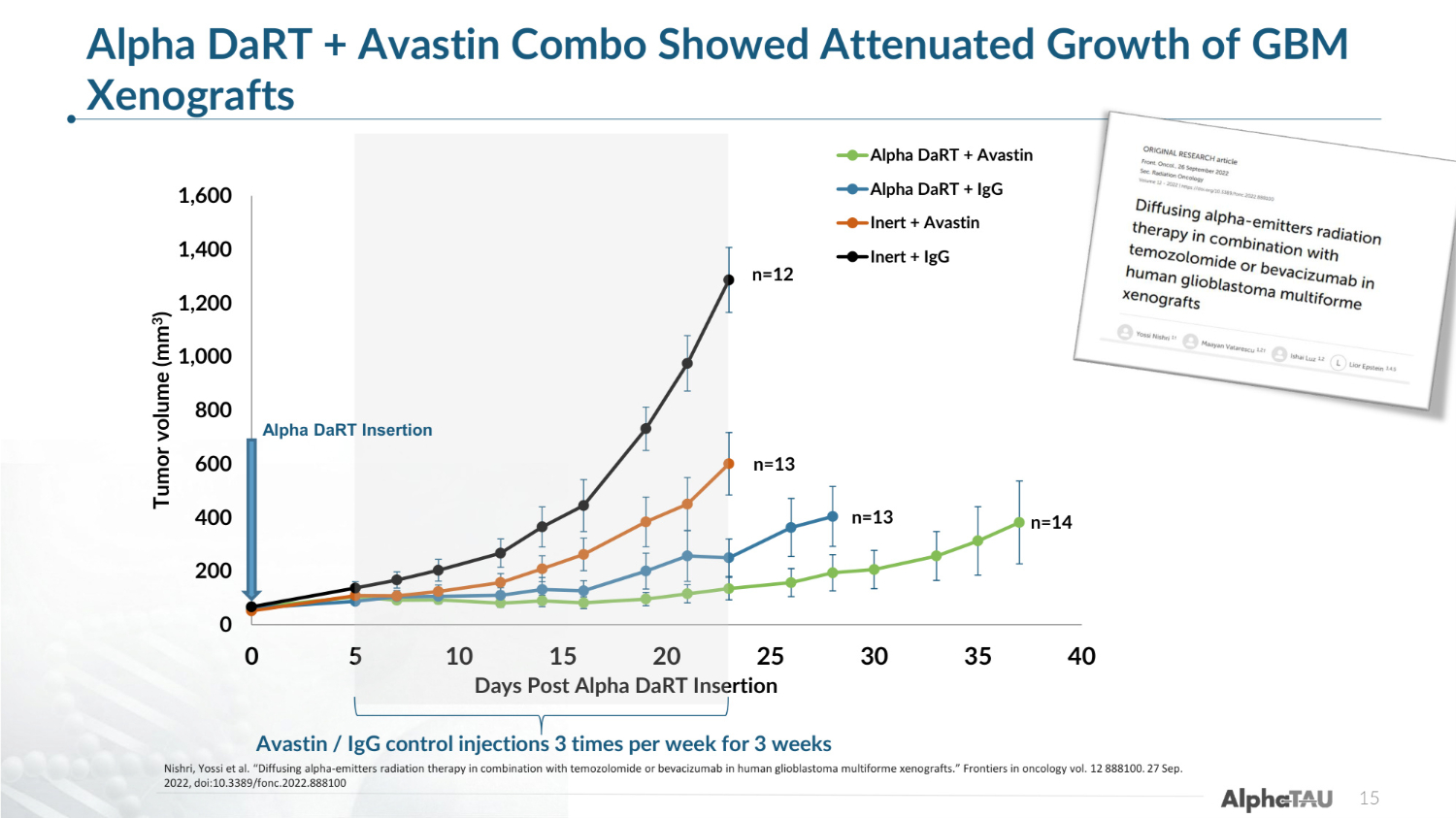
0 200 400 600 800 1,000 1,200 1,400 1,600 0 5 10 15 20 25 30 35 40 Tumor volume (mm3) Days Post Alpha DaRT Insertion Alpha DaRT + Avastin Alpha DaRT + IgG Inert + Avastin Inert + IgG n=14 n=13 n=13 n=12 Avastin / IgG control injections 3 times per week for 3 weeks Alpha DaRT + Avastin Combo Showed Attenuated Growth of GBM Xenografts 15 Alpha DaRT Insertion Nishri, Yossi et al. "Diffusing alpha-emitters radiation therapy in combination with temozolomide or bevacizumab in human glioblastoma multiforme xenografts." Frontiers in oncology vol. 12 888100. 27 Sep. 2022, doi:10.3389/fonc.2022.888100
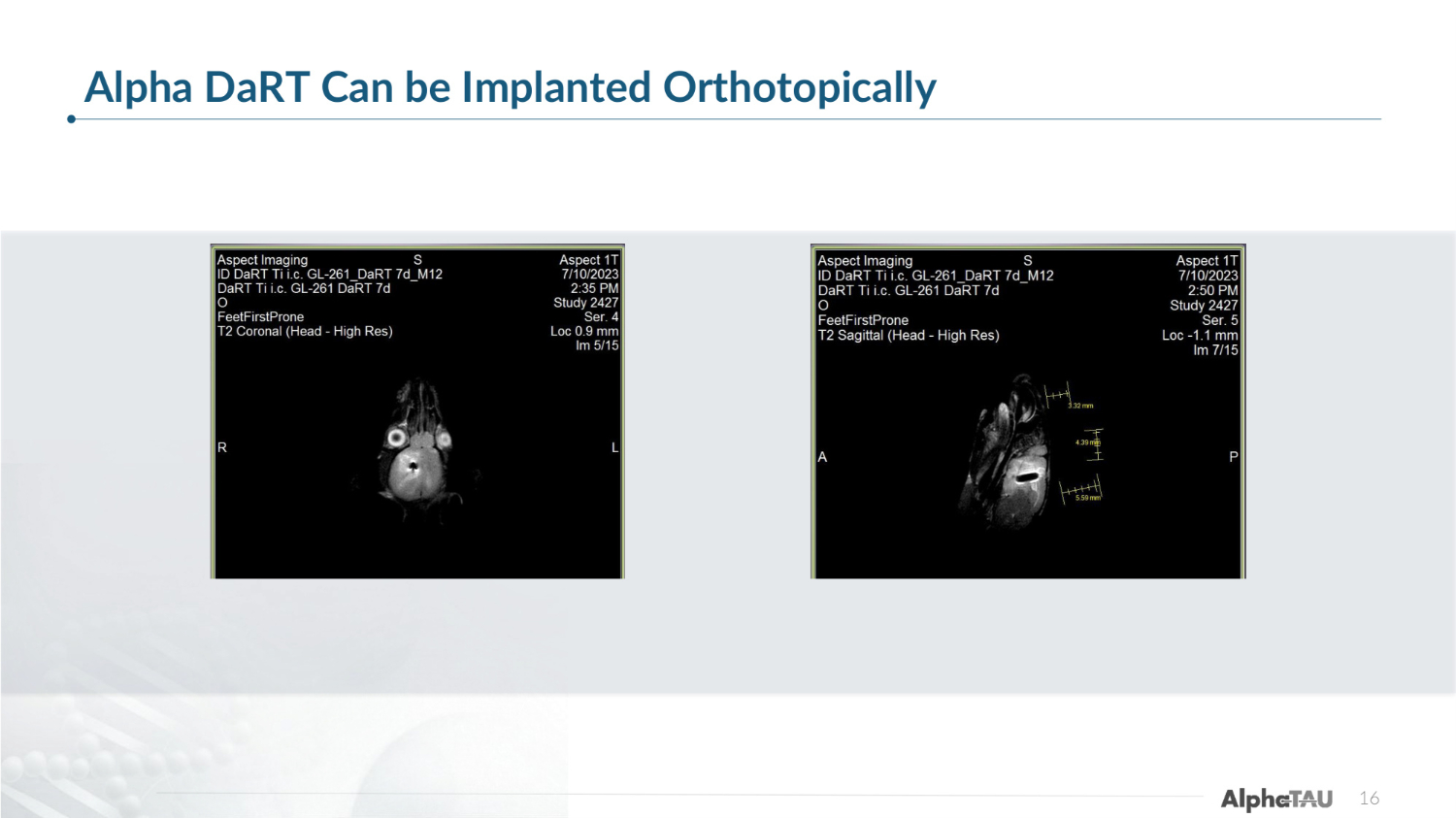
16 Alpha DaRT Can be Implanted Orthotopically

Brain Preclinical Studies – Large Animal 17
Alpha Radial Applicator 18 https://www.youtube.com/watch?v=IJY965J0xMk

19 MR Image of an Alpha DaRT Source in a Phantom Model A designated applicator allowed the delivery of 1-3 clusters of 7 sources in each hemisphere
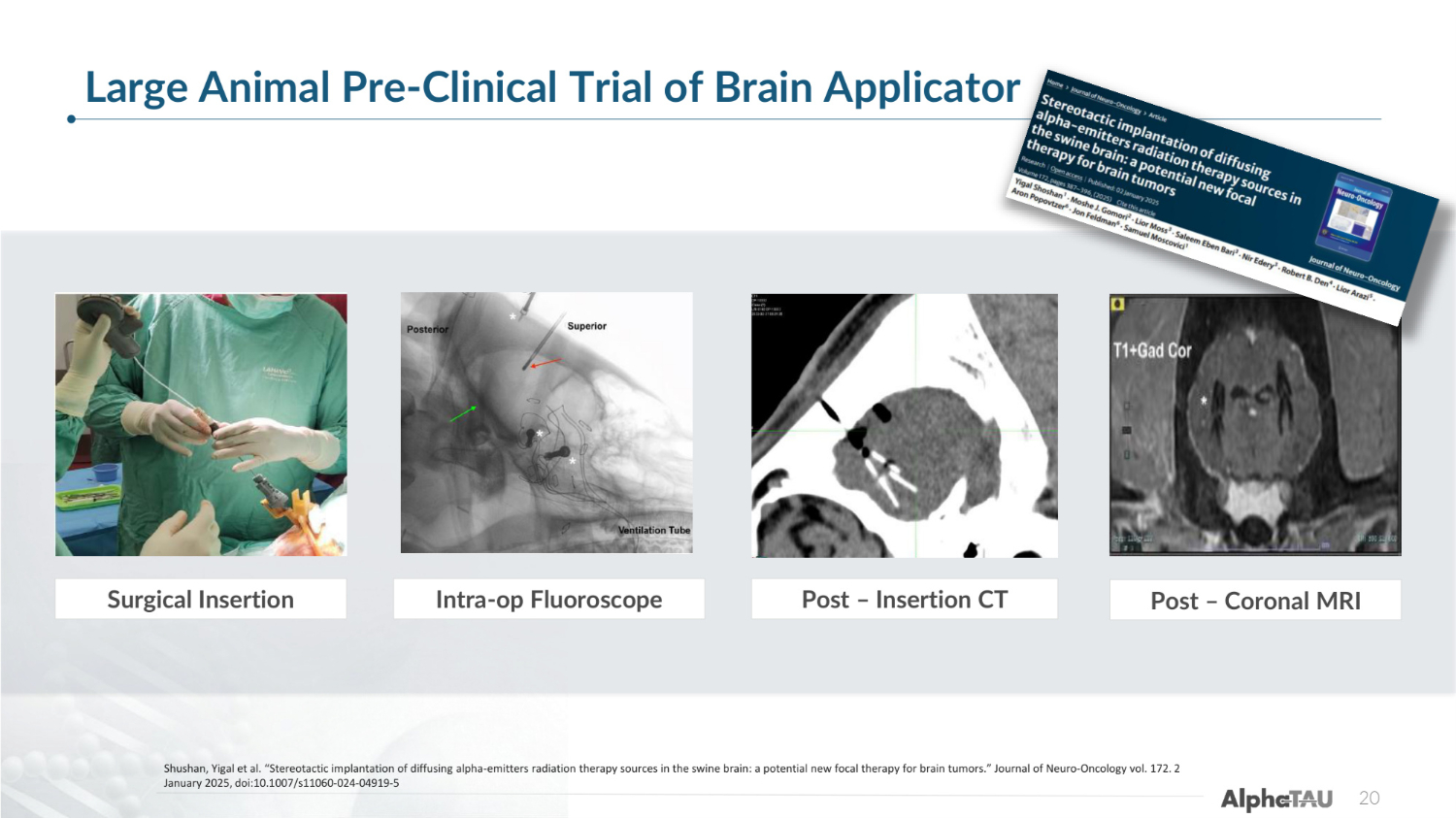
20 Surgical Insertion Post – Insertion CT Intra-op Fluoroscope Post – Coronal MRI Large Animal Pre-Clinical Trial of Brain Applicator Shushan, Yigal et al. "Stereotactic implantation of diffusing alpha-emitters radiation therapy sources in the swine brain: a potential new focal therapy for brain tumors." Journal of Neuro-Oncology vol. 172. 2 January 2025, doi:10.1007/s11060-024-04919-5

Image-guided stereotactic Alpha DaRT deployment and implantation in the swine brain was demonstrated to be feasible and safe. The configuration required by the treatment plan was efficiently implemented using a designated applicator. In contrast to other radiotherapies, no tissue changes in distant sites were observed. This is a major potential advantage of the suggested therapy, especially for patients who were previously treated with radiation and cannot receive additional external radiation treatments. Based on these safety findings, Alpha DaRT is being studied in clinical trials for patients with recurrent brain tumors. 21 Highlights from Pre-Clinical Brain Studies

Alpha DaRT Interim Data First 3 Patients in CTP-GBM-01 22
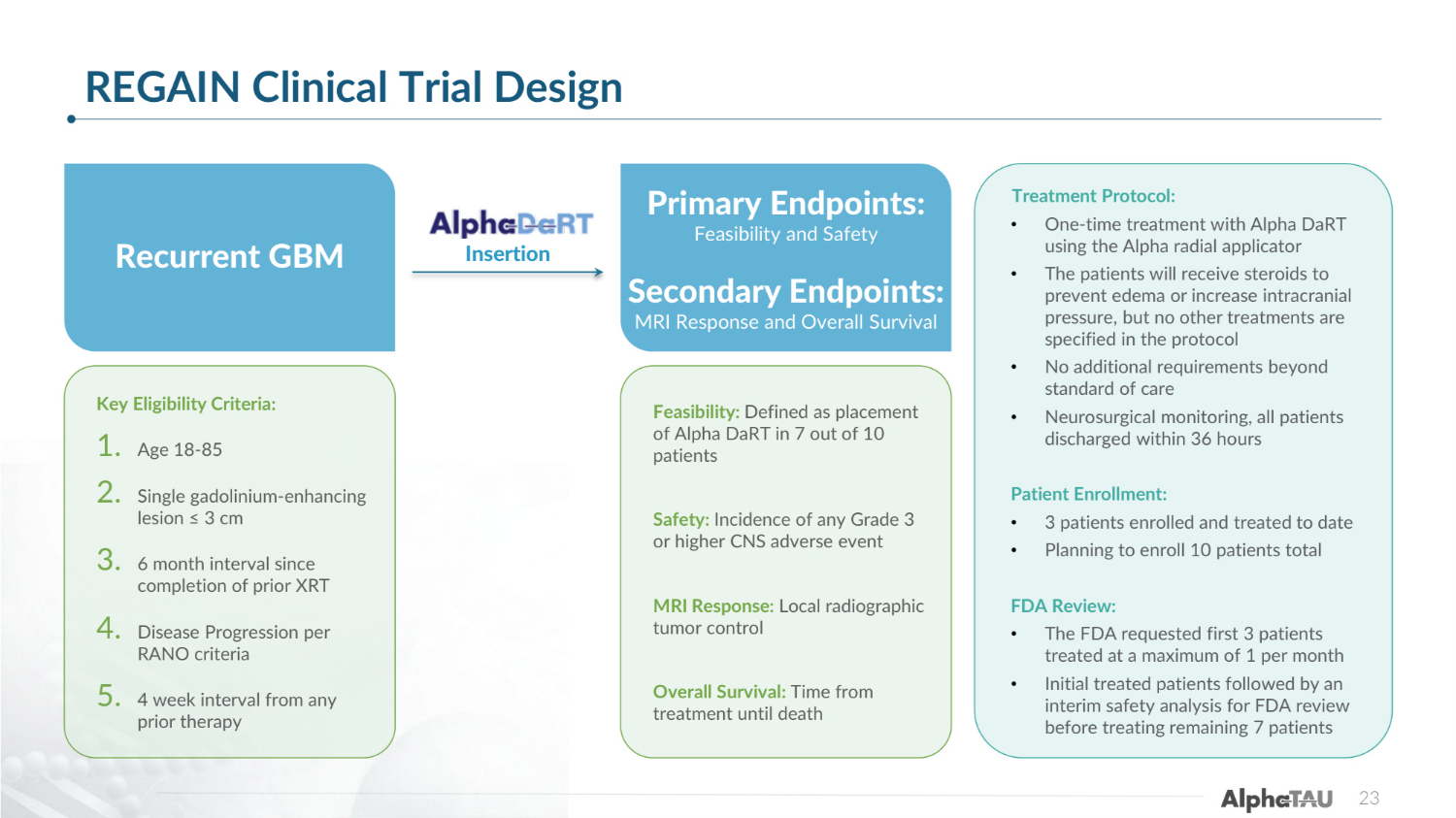
23 REGAIN Clinical Trial Design Recurrent GBM Key Eligibility Criteria: 1. Age 18-85 2. Single gadolinium-enhancing lesion ≤ 3 cm 3. 6 month interval since completion of prior XRT 4. Disease Progression per RANO criteria 5. 4 week interval from any prior therapy Feasibility: Defined as placement of Alpha DaRT in 7 out of 10 patients Safety: Incidence of any Grade 3 or higher CNS adverse event MRI Response: Local radiographic tumor control Overall Survival: Time from treatment until death Primary Endpoints: Feasibility and Safety Secondary Endpoints: MRI Response and Overall Survival Insertion Treatment Protocol: • One-time treatment with Alpha DaRT using the Alpha radial applicator • The patients will receive steroids to prevent edema or increase intracranial pressure, but no other treatments are specified in the protocol • No additional requirements beyond standard of care • Neurosurgical monitoring, all patients discharged within 36 hours Patient Enrollment: • 3 patients enrolled and treated to date • Planning to enroll 10 patients total FDA Review: • The FDA requested first 3 patients treated at a maximum of 1 per month • Initial treated patients followed by an interim safety analysis for FDA review before treating remaining 7 patients

24 Baseline Characteristics of First Three Patients Treated Recurrent GBM Patient 1 Patient 2 Patient 3 Patient 1 Patient 2 Patient 3 Age 56 71 56 Sex Male Male Male Initial Diagnosis WHO grade 4 glioma IDH wild type, MGMT unmethylated with FGFR:TACC fusion Right frontal WHO grade 4 glioma IDH wild type WHO grade 4 glioma IDH wild type, MGMT hypermethylated Date of Initial Diagnosis Feb 2024 May 2021 Feb 2025 Number of Recurrences 3 2 1

25 Patient 1 • 56-year-old male with a WHO grade 4 glioma IDH wild type, MGMT unmethylated with FGFR:TACC fusion • Initial Diagnosis was in 2/2024, Gross Total Resection in 2/11/24 • Post operative course: • 60 Gy in 30 fractions with temozolomide completed 3/19/24-4/30/24 • 1st recurrence 10/24 underwent LITT and Gallium Maltolate trial • 2nd recurrence underwent surgery revealing recurrent GBM • 3rd recurrence led to Alpha DaRT trial • He has baseline left homonymous hemianopsia Patient 1 Patient 2 Patient 3
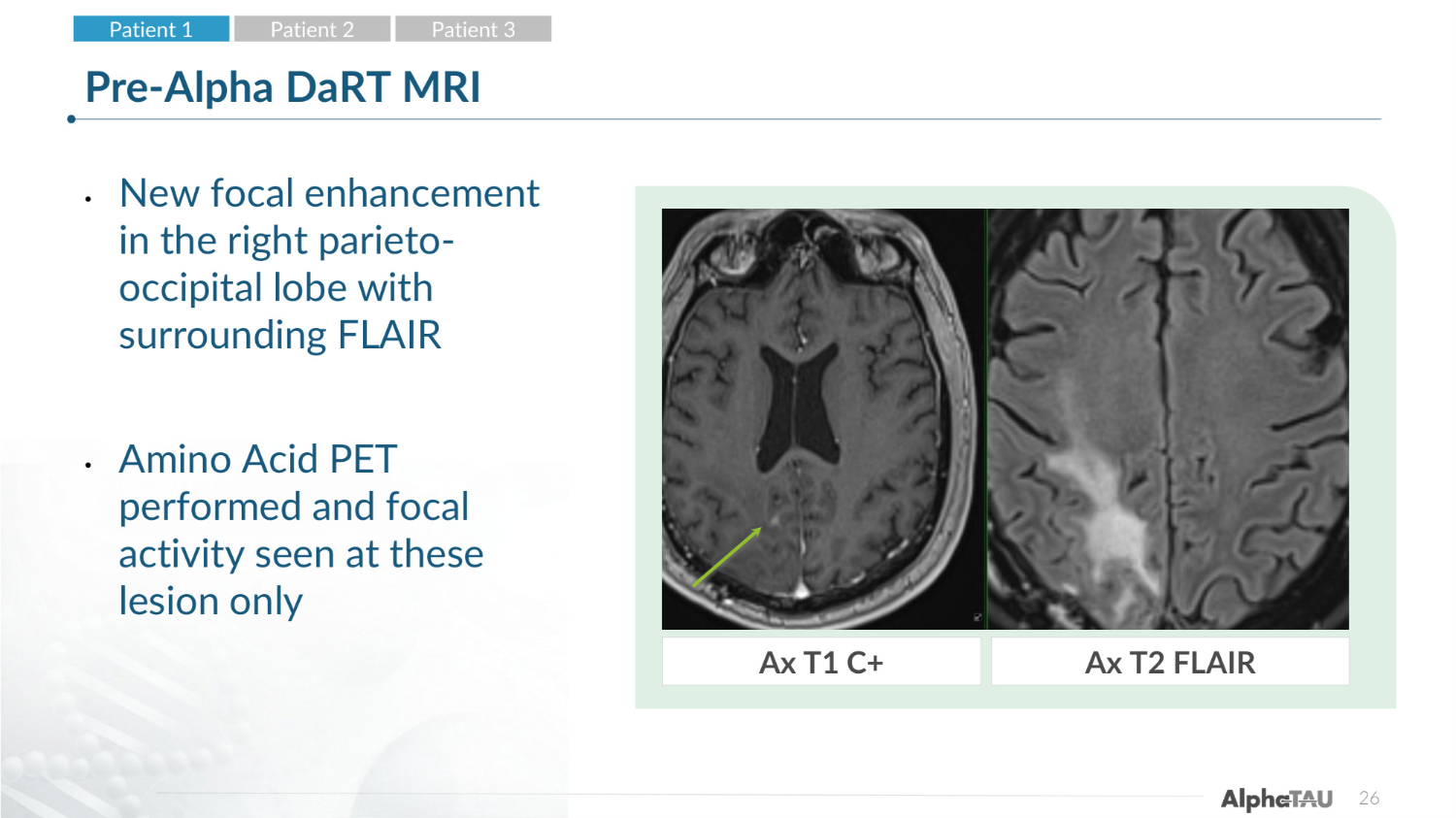
26 Pre-Alpha DaRT MRI • New focal enhancement in the right parieto- occipital lobe with surrounding FLAIR • Amino Acid PET performed and focal activity seen at these lesion only Ax T1 C+ Ax T2 FLAIR Patient 1 Patient 2 Patient 3
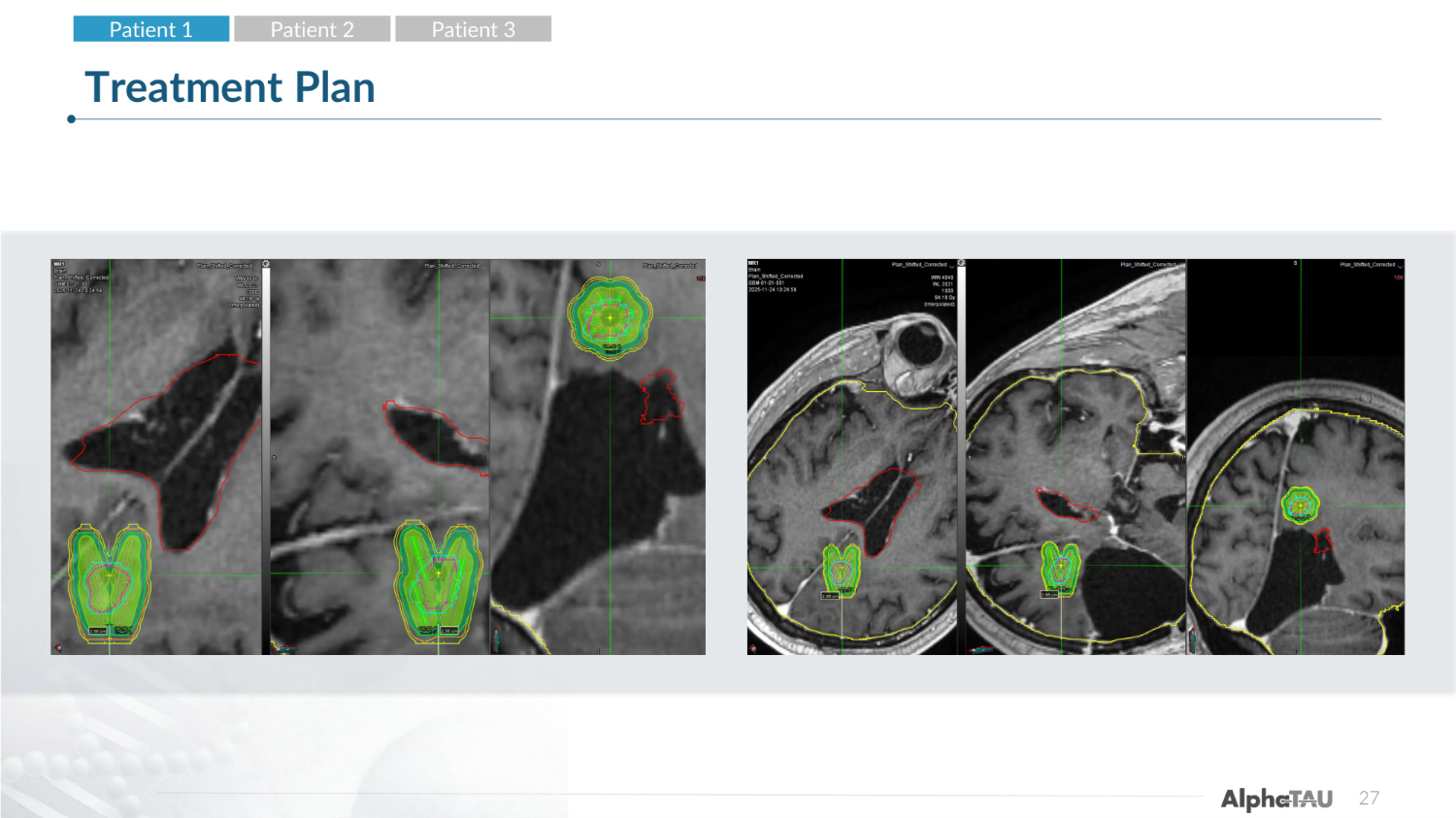
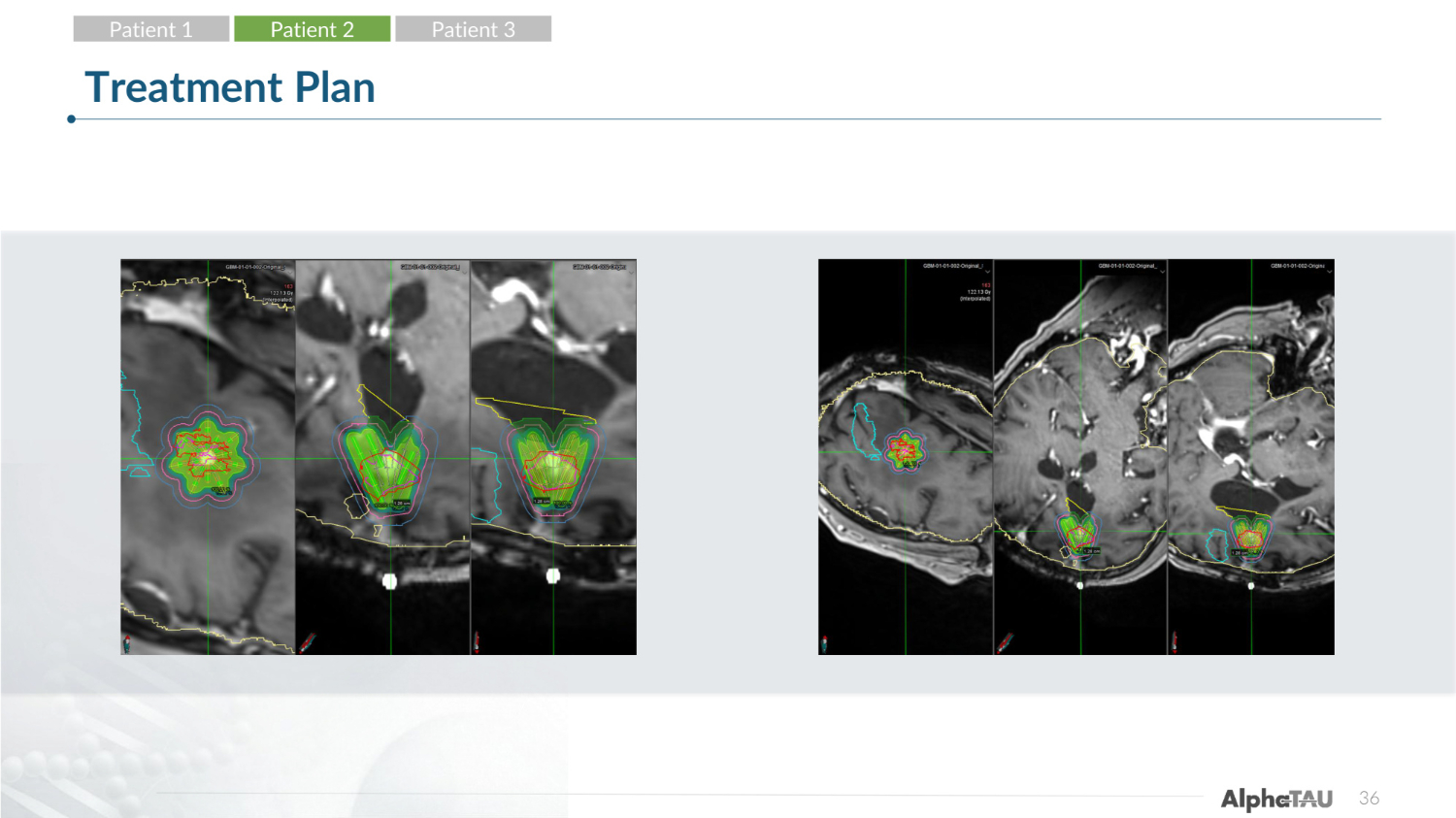
27 Treatment Plan Patient 1 Patient 2 Patient 3
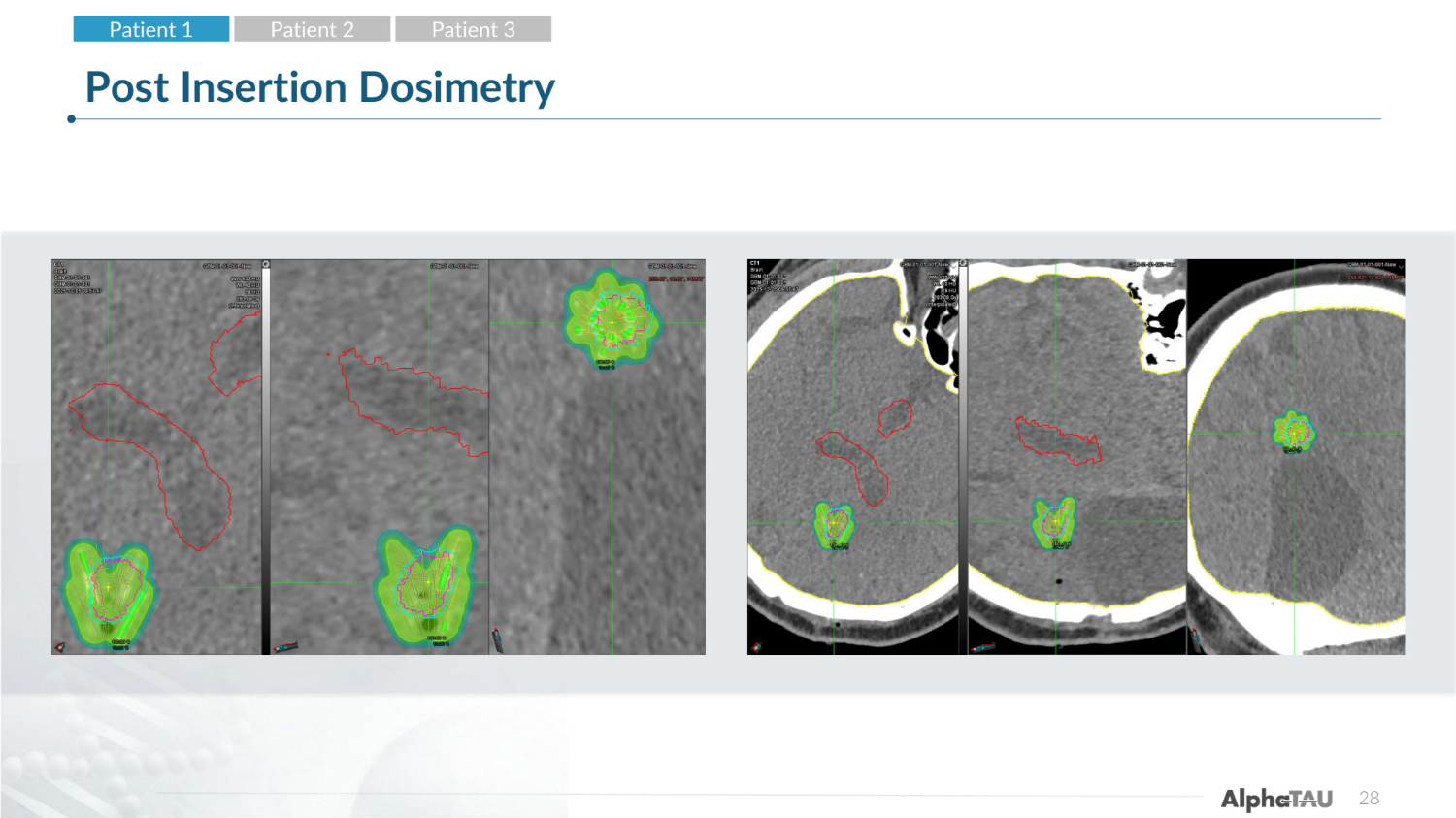
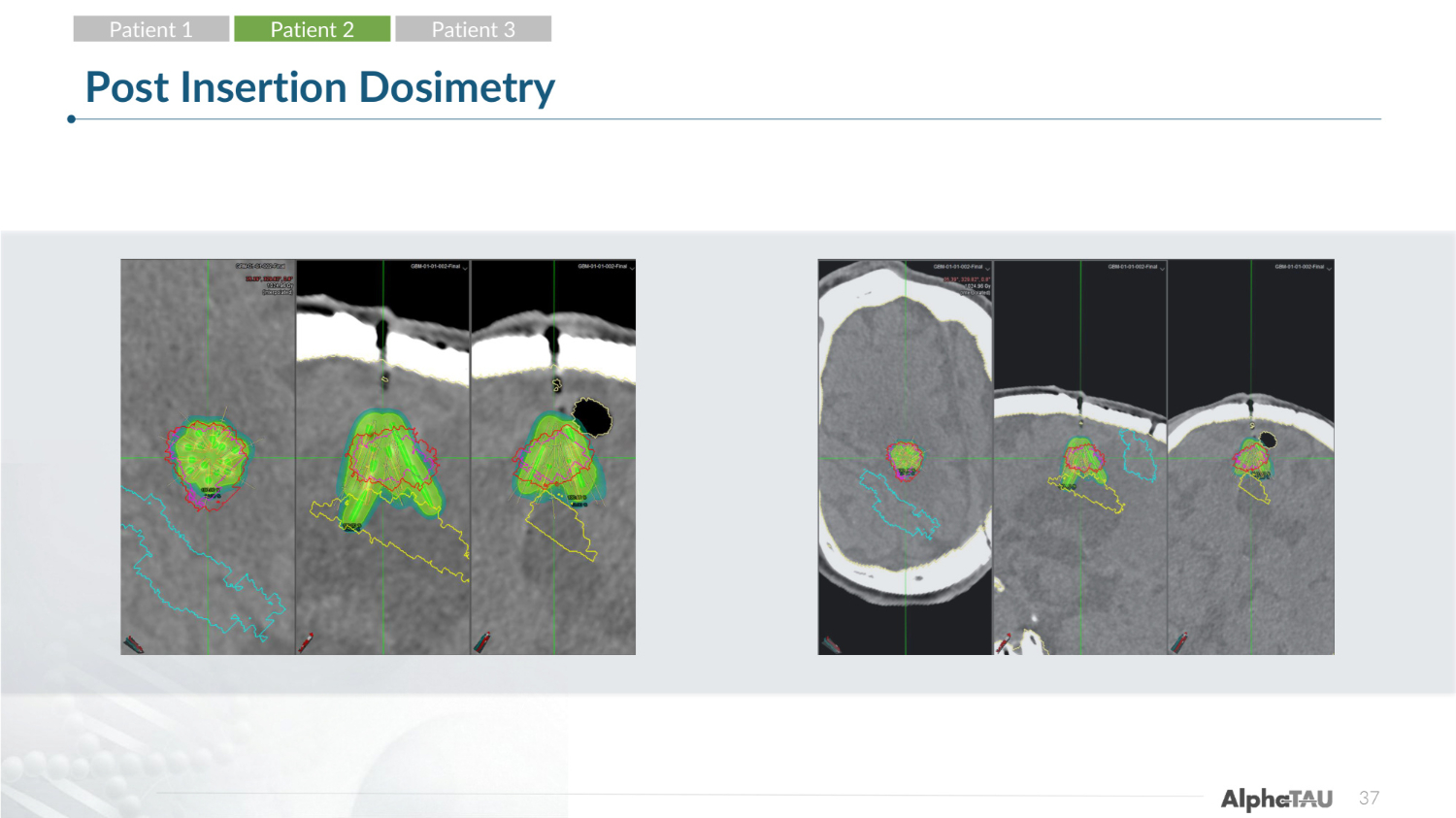
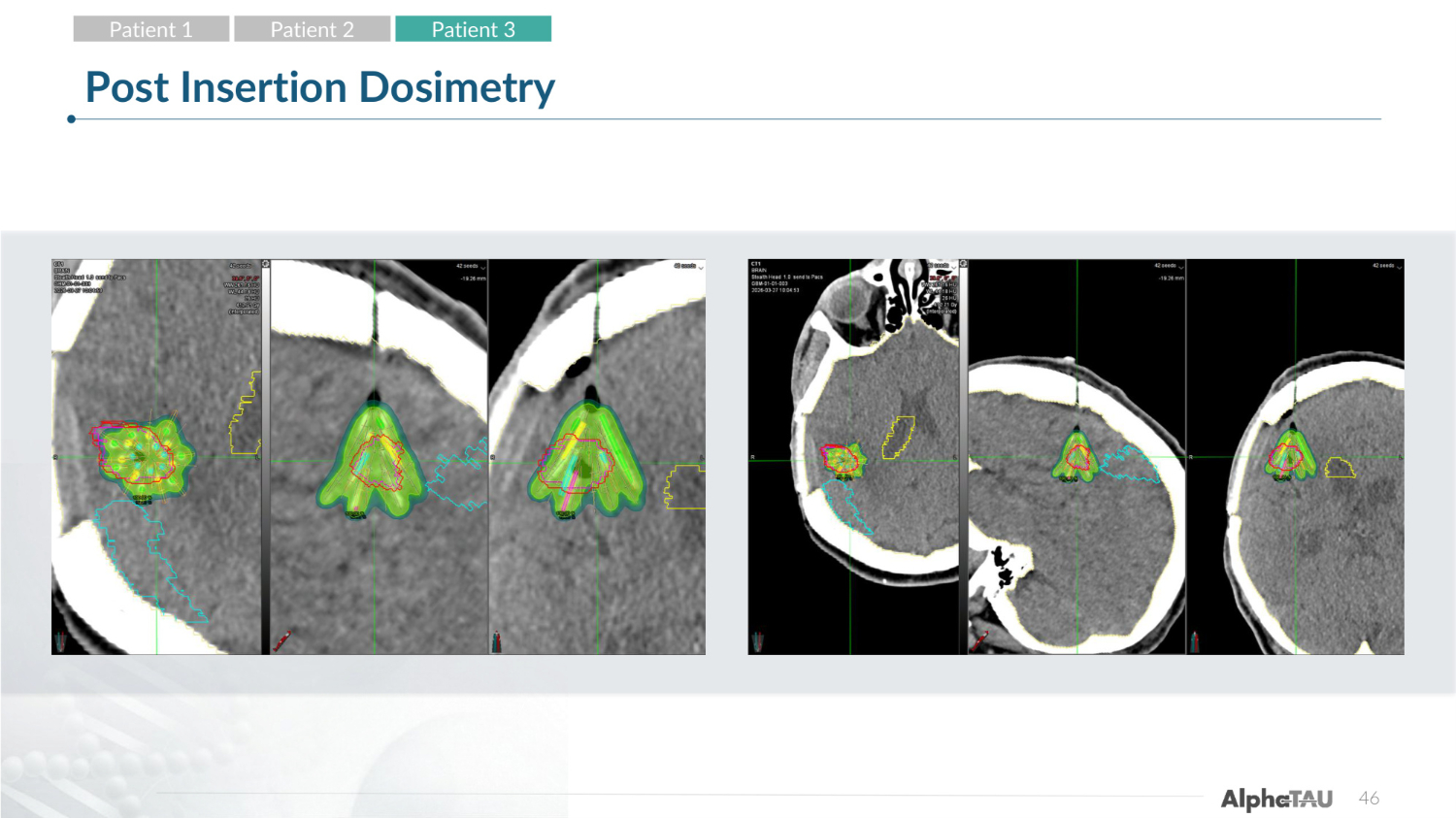
28 Post Insertion Dosimetry Patient 1 Patient 2 Patient 3
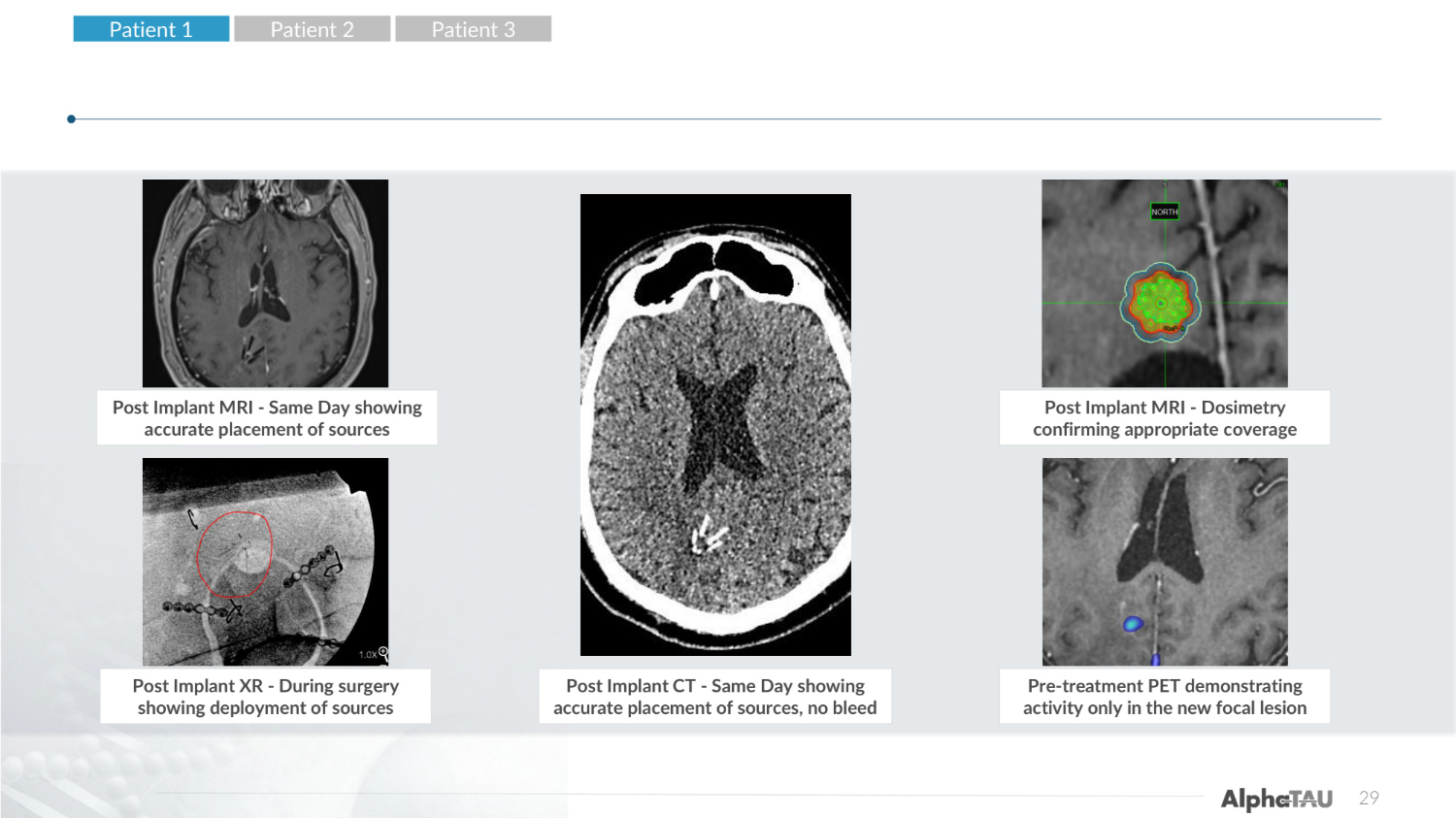
Post Implant CT-Same Day Showing accurate placement of seeds, no bleed Pre-treatment PET demonstrating activity only in the new focal lesion 29 Post Implant MRI - Same Day showing accurate placement of sources Post Implant XR - During surgery showing deployment of sources Post Implant CT - Same Day showing accurate placement of sources, no bleed Post Implant MRI - Dosimetry confirming appropriate coverage Pre-treatment PET demonstrating activity only in the new focal lesion Patient 1 Patient 2 Patient 3
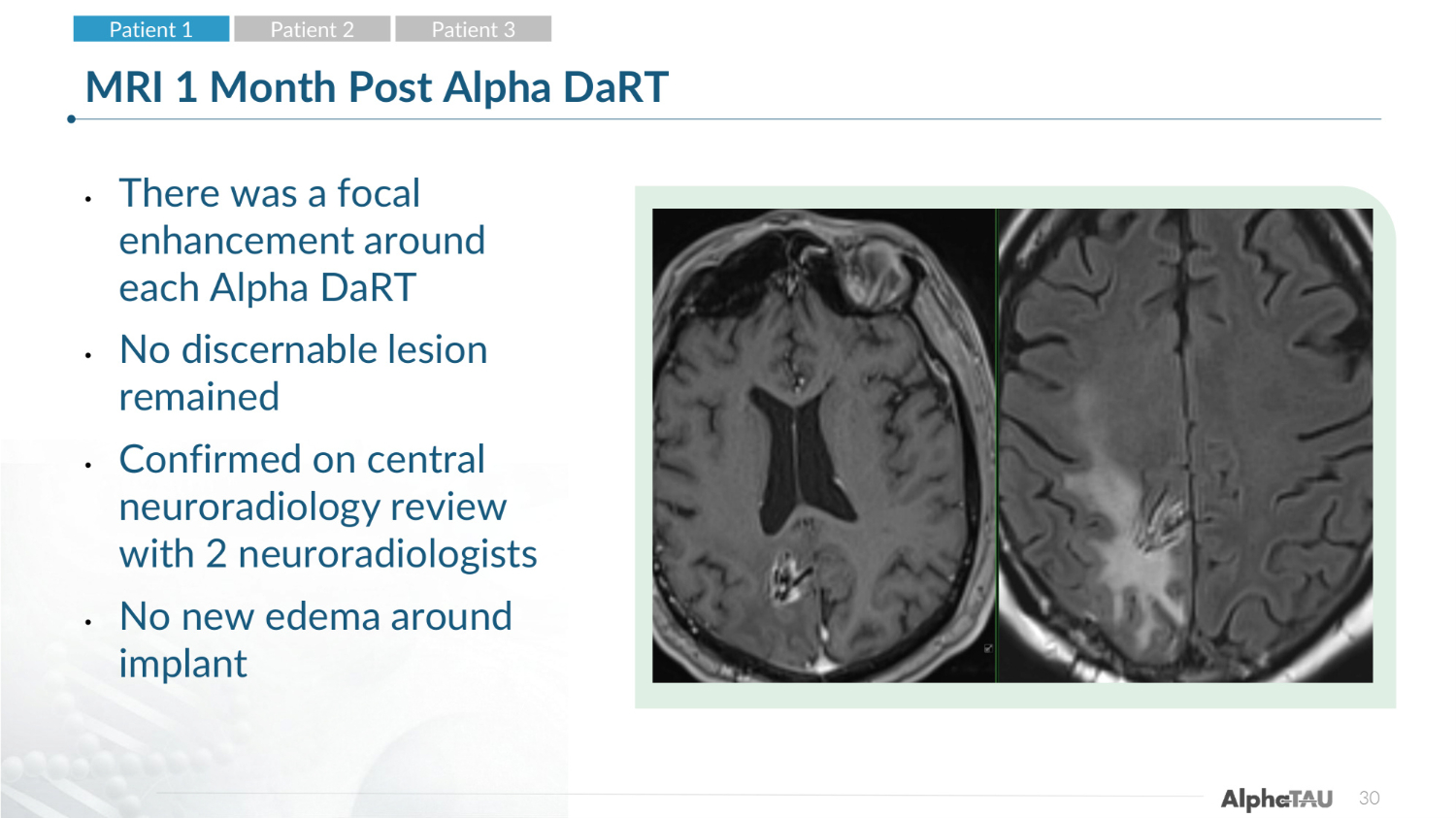
30 MRI 1 Month Post Alpha DaRT • There was a focal enhancement around each Alpha DaRT • No discernable lesion remained • Confirmed on central neuroradiology review with 2 neuroradiologists • No new edema around implant Patient 1 Patient 2 Patient 3
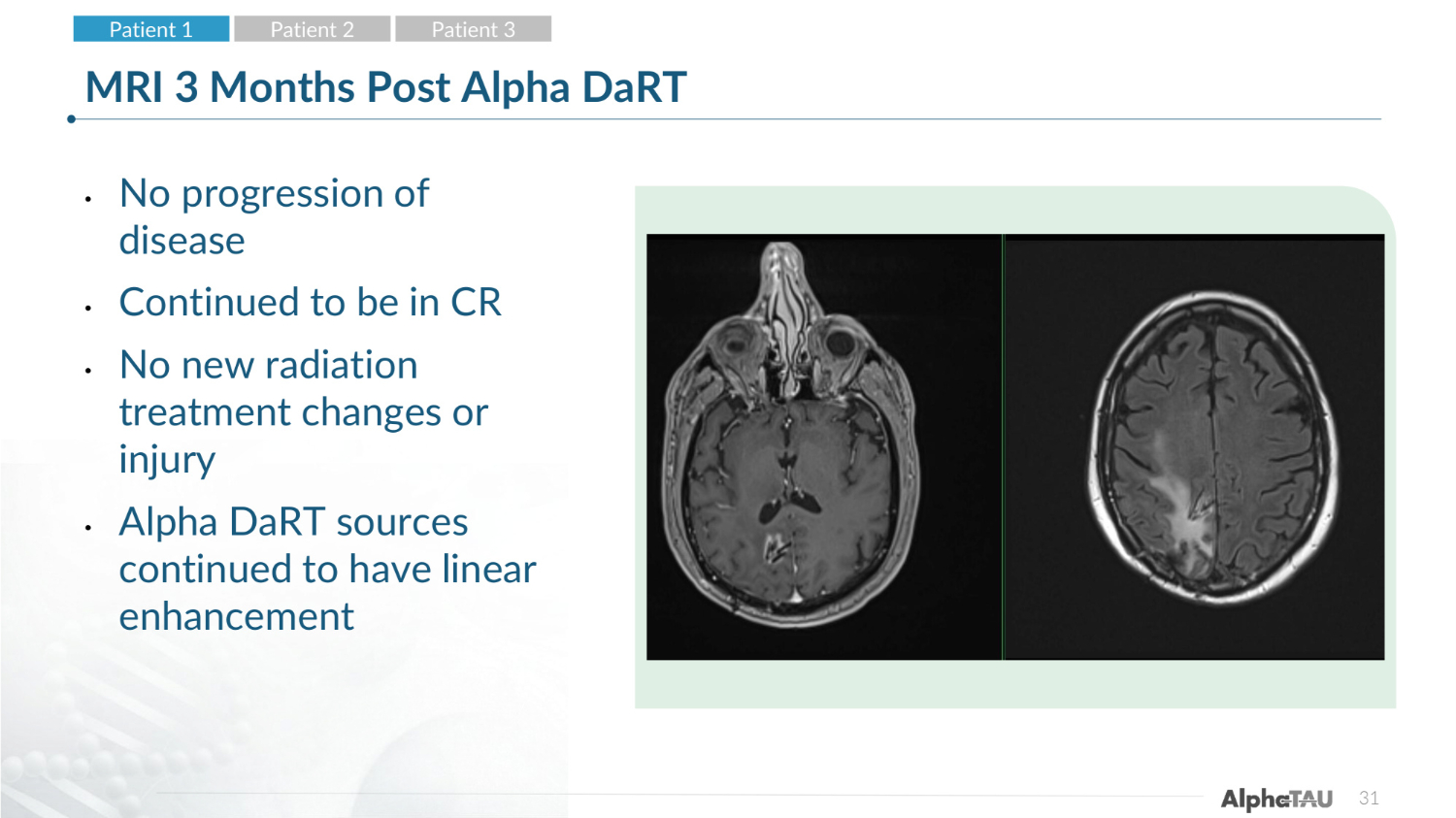
31 MRI 3 Months Post Alpha DaRT • No progression of disease • Continued to be in CR • No new radiation treatment changes or injury • Alpha DaRT sources continued to have linear enhancement Patient 1 Patient 2 Patient 3

32 Adverse Events and Current Status* • He developed no dose limiting toxicities, no symptoms from treatment • NED (no evidence of disease), with complete response (CR) in the lesion Patient 1 Patient 2 Patient 3 * Interim Data as of 3-May-2026

33 Patient 2 • 71-year-old male with an initial diagnosis in 5/2021 of a right frontal WHO grade 4 glioma IDH wild type, who underwent gross total resection and completed chemo-RT with 60 Gy in 30 fractions 6/21- 8/11/21 and temozolomide • The patient had seizures post surgery • This right frontal was stable. • He developed a left frontal lesion in 5/2025 and underwent gross total resection on 5/20/25 demonstrating a WHO grade 4 glioma IDH wild type • He underwent 35 GyE in 10 fractions intensity modulated proton therapy completed on 7/22/25 • On 2/2/26 he developed a local recurrence in the right frontal lobe Patient 1 Patient 2 Patient 3
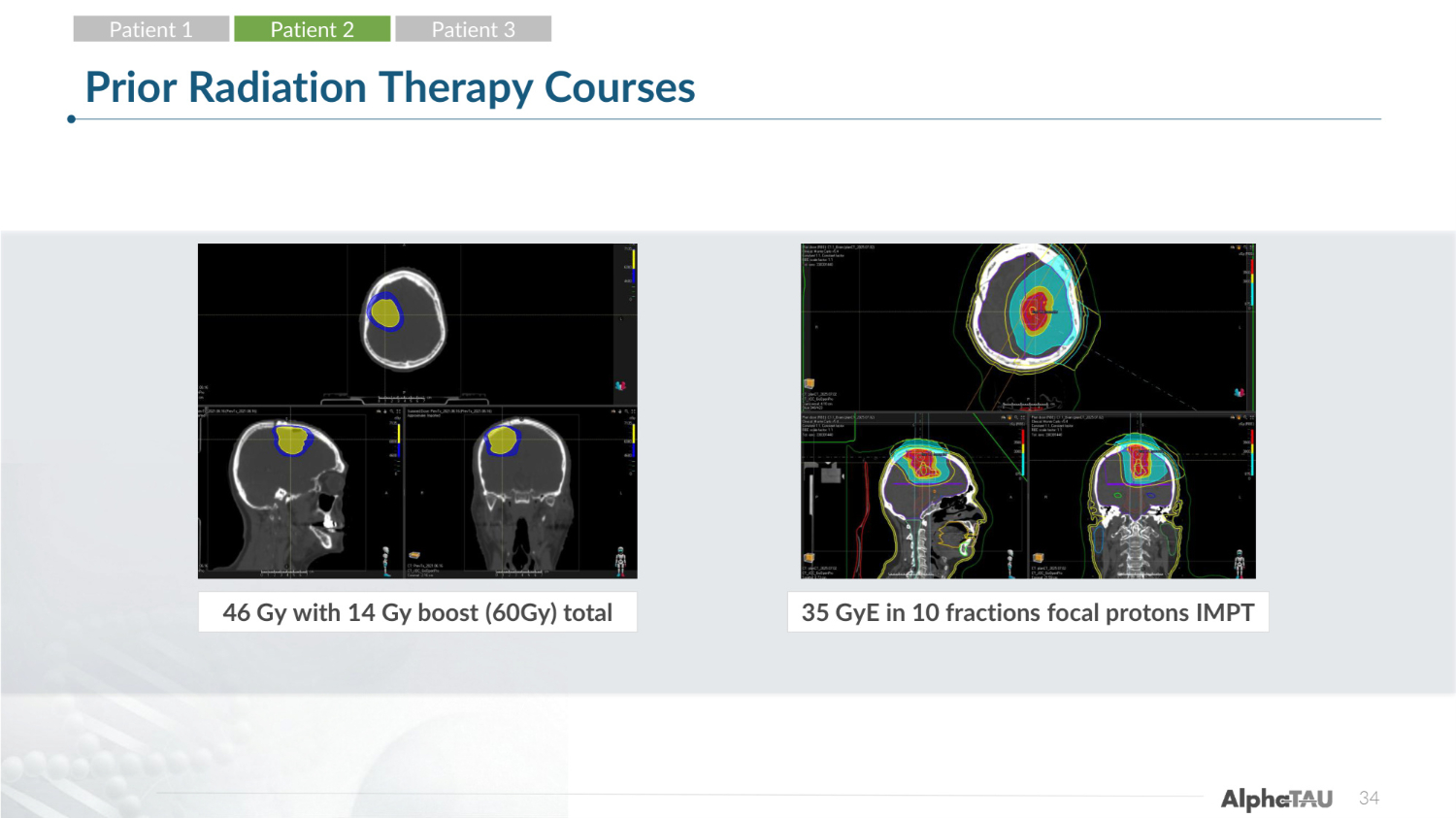
34 Prior Radiation Therapy Courses 46 Gy with 14 Gy boost (60Gy) total 35 GyE in 10 fractions focal protons IMPT Patient 1 Patient 2 Patient 3
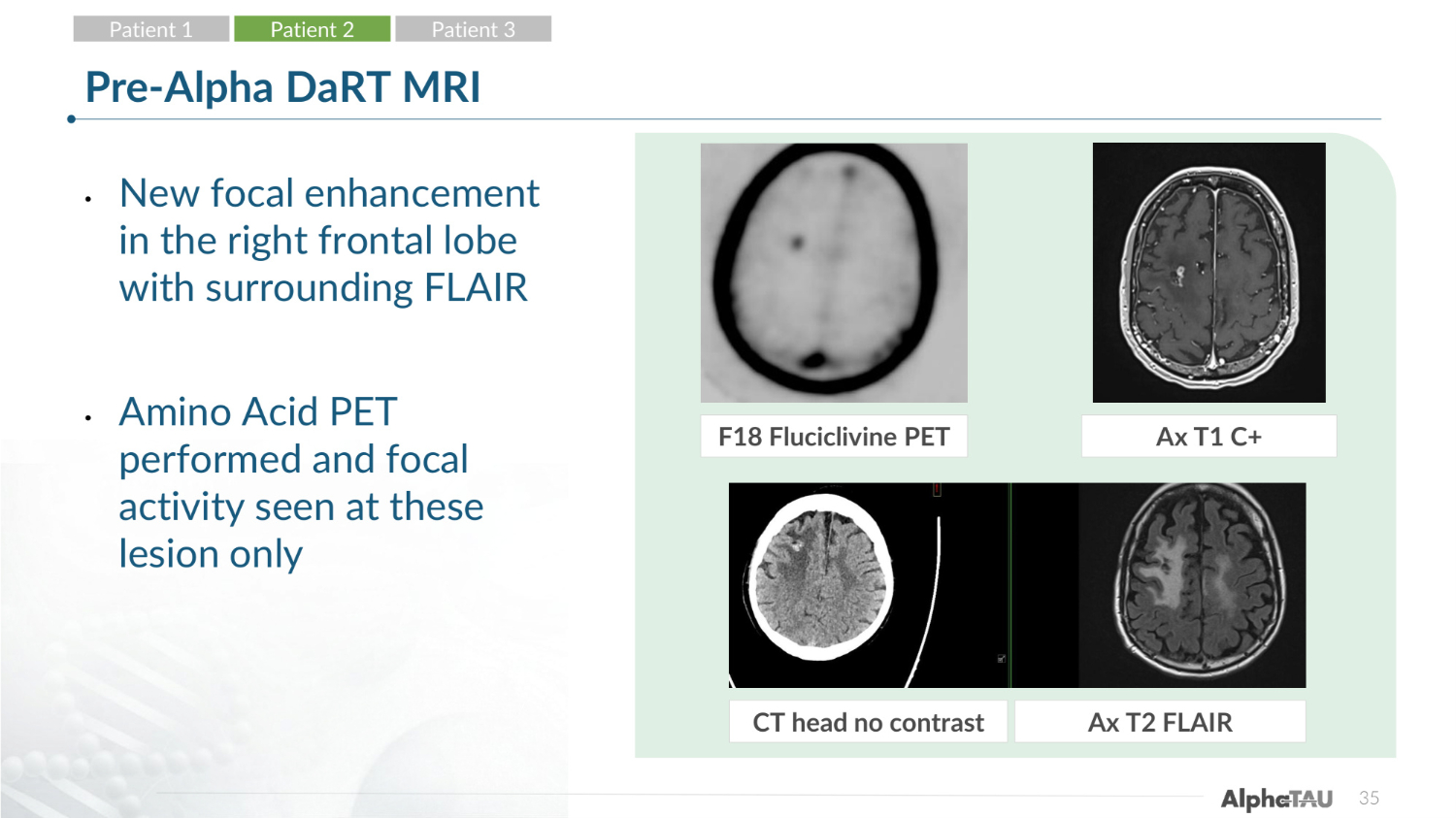
35 Pre-Alpha DaRT MRI • New focal enhancement in the right frontal lobe with surrounding FLAIR • Amino Acid PET performed and focal activity seen at these lesion only F18 Fluciclivine PET Ax T1 C+ CT head no contrast Ax T2 FLAIR Patient 1 Patient 2 Patient 3
36 Treatment Plan Patient 1 Patient 2 Patient 3
37 Post Insertion Dosimetry Patient 1 Patient 2 Patient 3
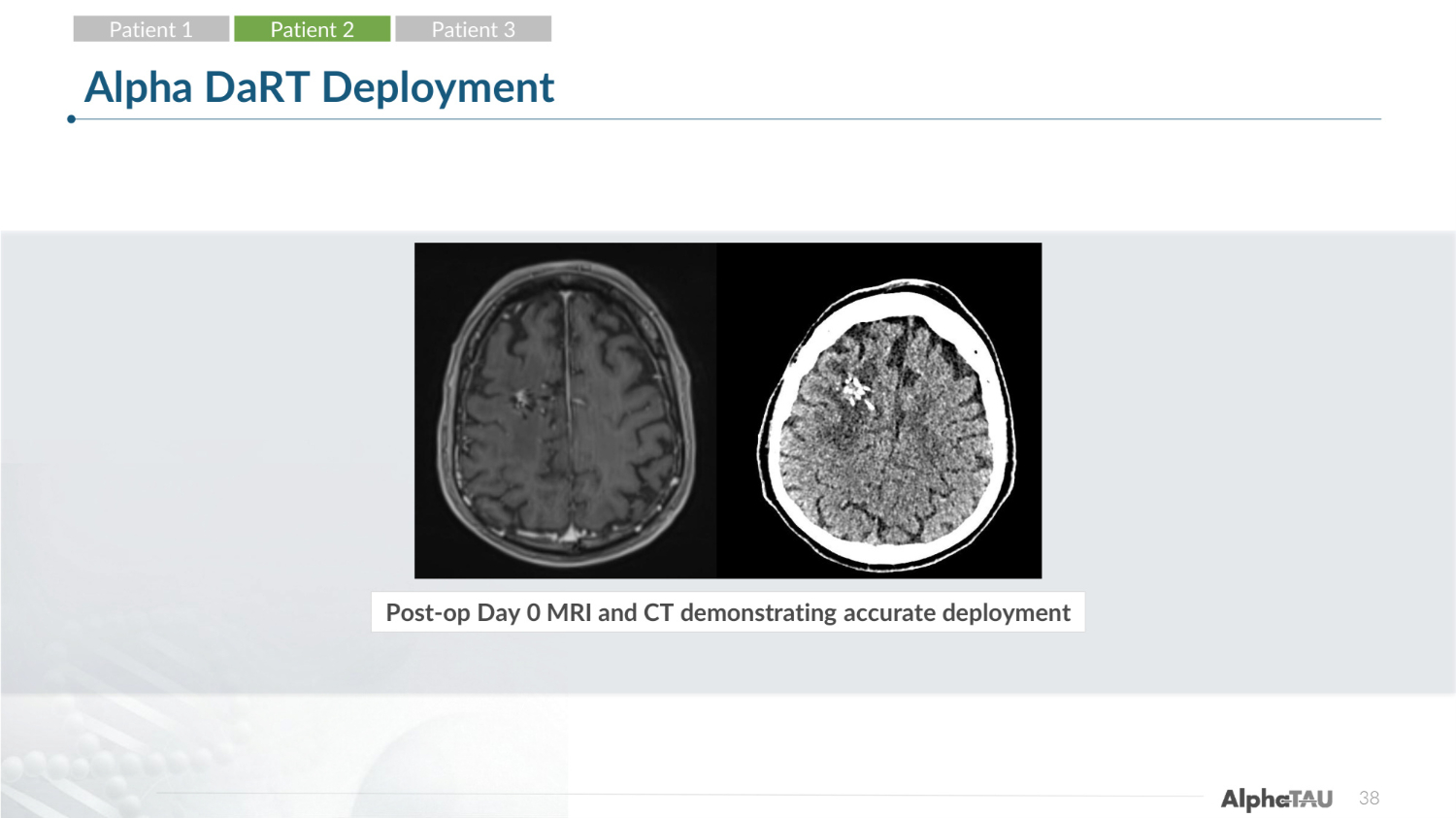
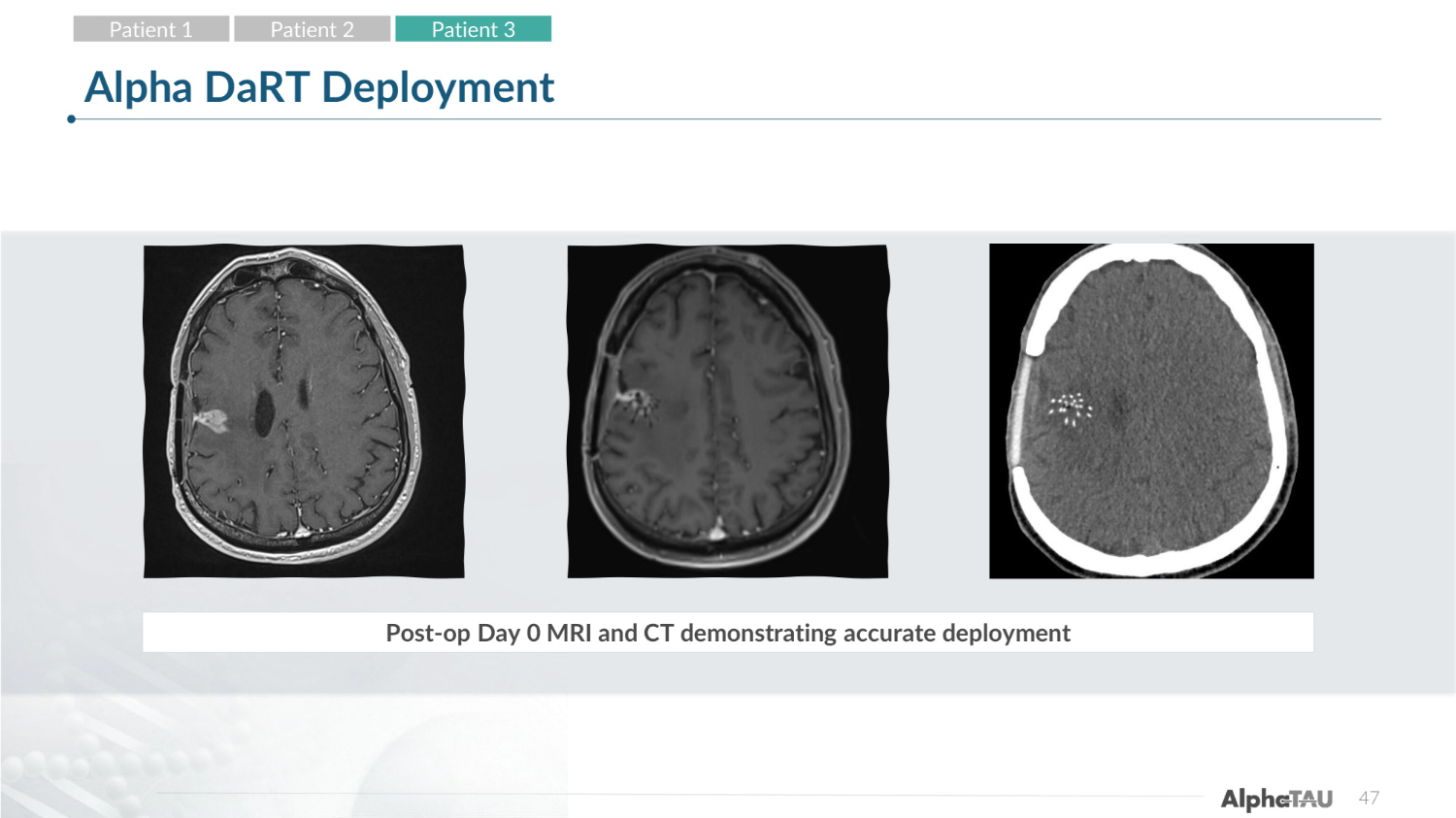
38 Alpha DaRT Deployment Post-op Day 0 MRI and CT demonstrating accurate deployment Patient 1 Patient 2 Patient 3
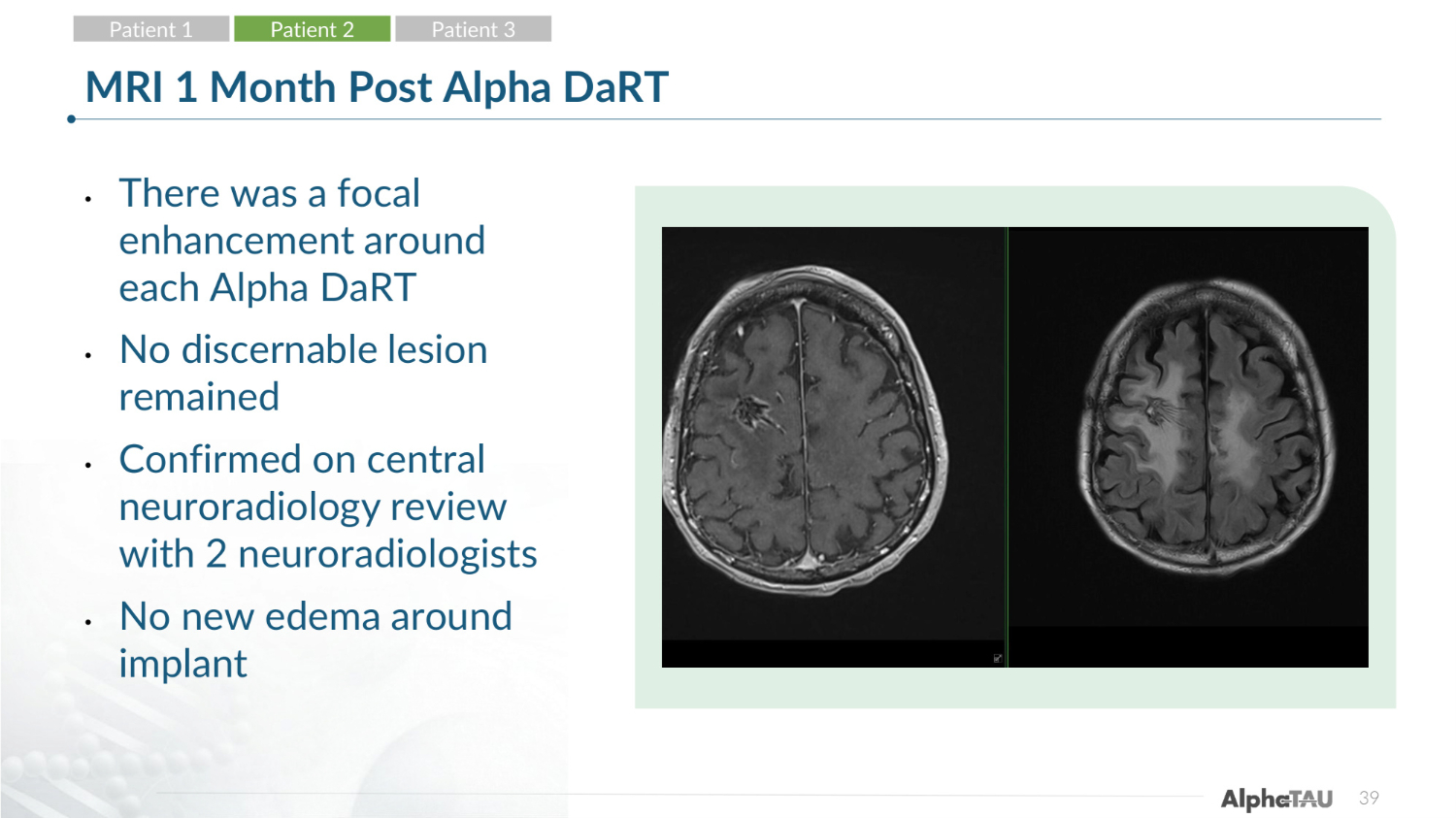
39 MRI 1 Month Post Alpha DaRT • There was a focal enhancement around each Alpha DaRT • No discernable lesion remained • Confirmed on central neuroradiology review with 2 neuroradiologists • No new edema around implant Patient 1 Patient 2 Patient 3
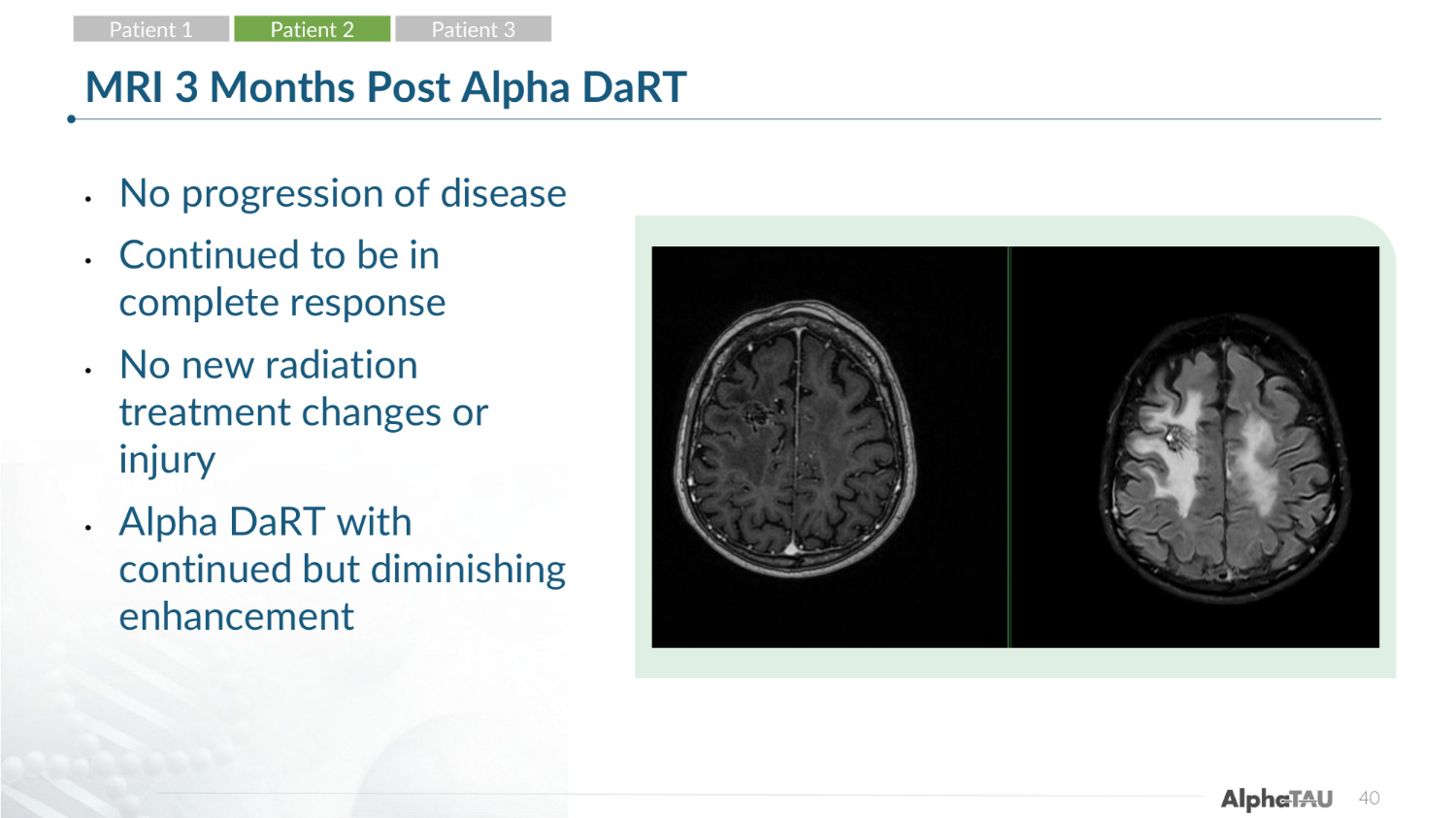
40 MRI 3 Months Post Alpha DaRT • No progression of disease • Continued to be in complete response • No new radiation treatment changes or injury • Alpha DaRT with continued but diminishing enhancement Patient 1 Patient 2 Patient 3

41 Adverse Events • He developed a grade 3 seizure with Todd's paralysis 10 days post Alpha DaRT, this occurred during his steroid taper • This required a 48-hour hospitalization • As previously noted, the patient had a history of seizures • Symptoms improved to baseline on steroids and additional imaging showed normalizing edema to pretreatment extent • Doing well and returned to his neurologic baseline* Patient 1 Patient 2 Patient 3 * Interim Data as of 3-May-2026

• 56 yo M with WHO grade 4 glioma IDH wt, MGMT hypermethylated, Ki67-40%, DL1 0% • He underwent a subtotal resection and underwent a reoperation (gross total resection) 1 month later due to early progression noted on imaging, and developed post op empyema with klebsiella • He has a history of seizures since his first surgery • He underwent post op Chemo RT with 60 Gy and temozolomide • During adjuvant temozolomide noted to have progressive enhancement in the prior surgical resection • Amino Acid PET positive and RANO progressive based on size and consensus review 42 Patient 3 Patient 1 Patient 2 Patient 3
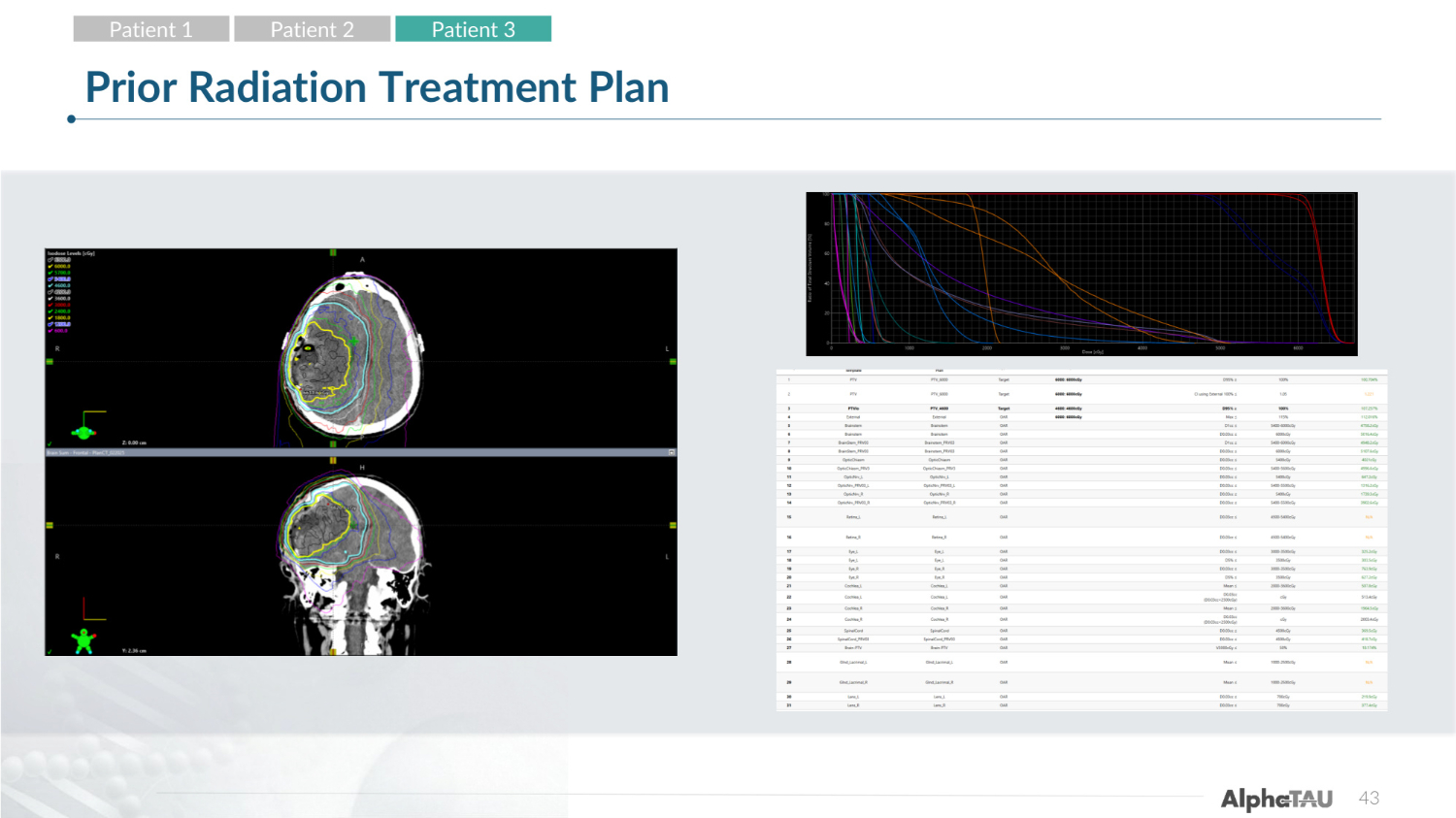
43 Prior Radiation Treatment Plan Patient 1 Patient 2 Patient 3
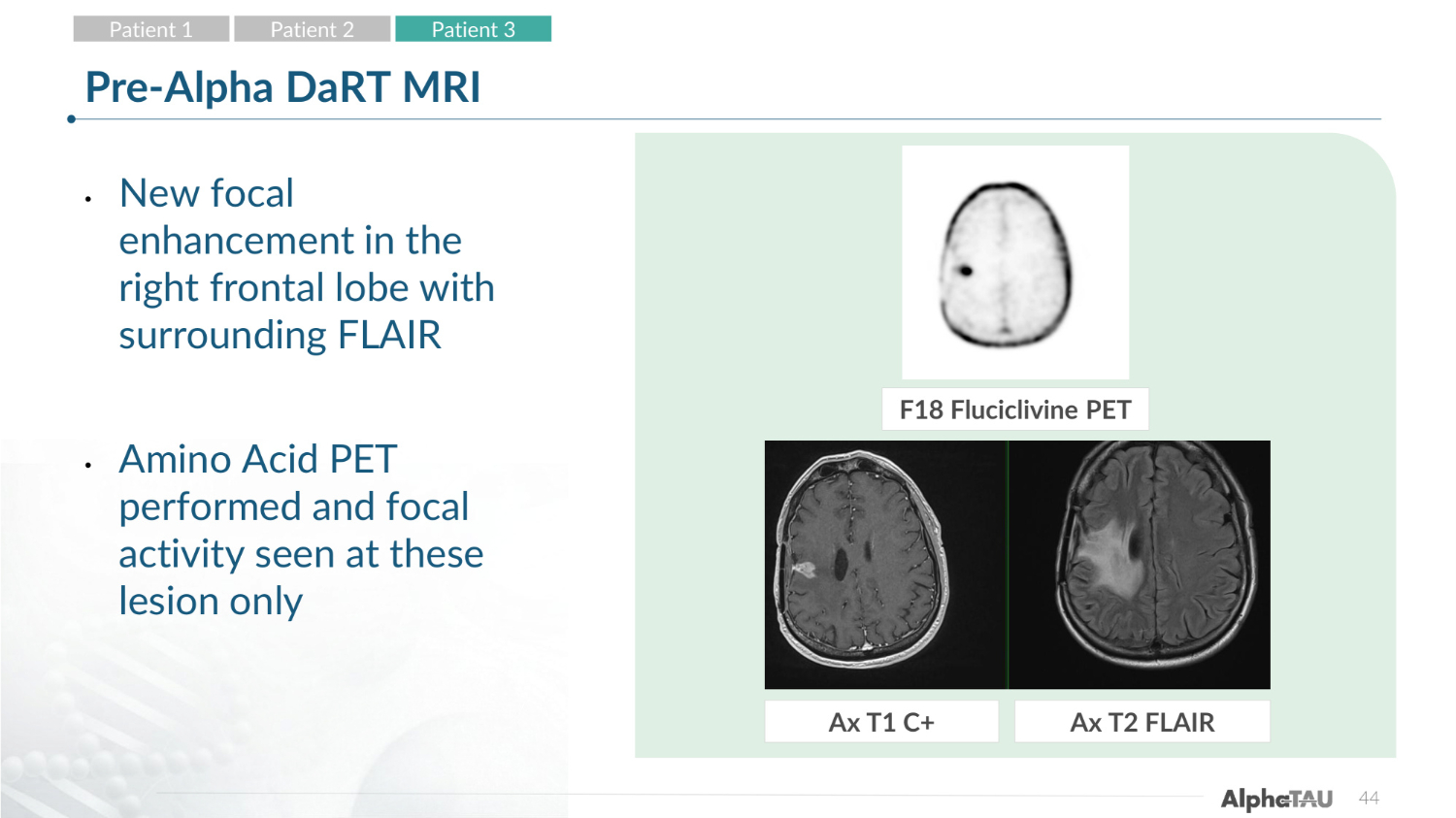
44 Pre-Alpha DaRT MRI • New focal enhancement in the right frontal lobe with surrounding FLAIR • Amino Acid PET performed and focal activity seen at these lesion only F18 Fluciclivine PET Ax T1 C+ Ax T2 FLAIR Patient 1 Patient 2 Patient 3
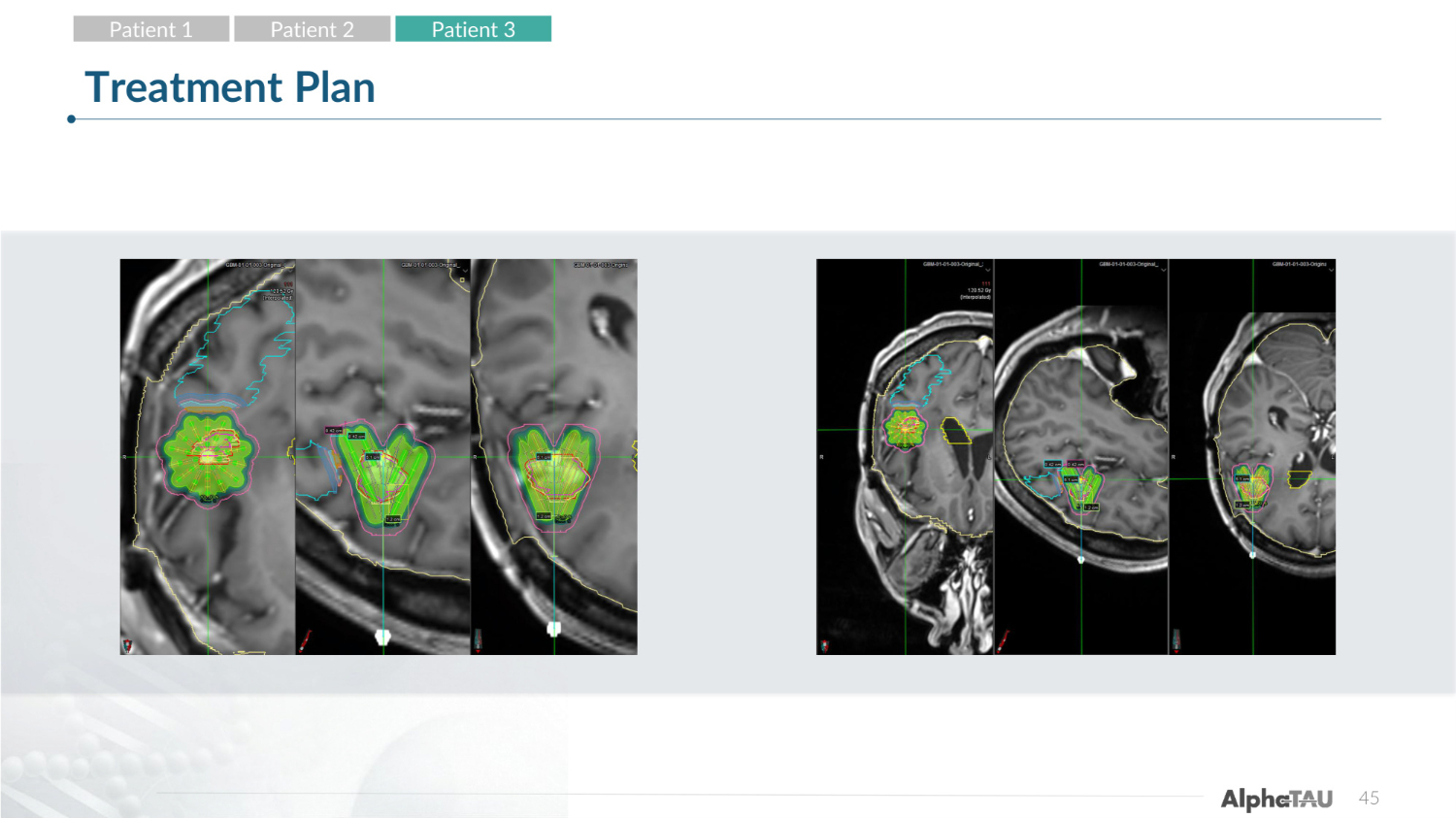
45 Treatment Plan Patient 1 Patient 2 Patient 3
46 Post Insertion Dosimetry Patient 1 Patient 2 Patient 3
47 Alpha DaRT Deployment Post-op Day 0 MRI and CT demonstrating accurate deployment Patient 1 Patient 2 Patient 3
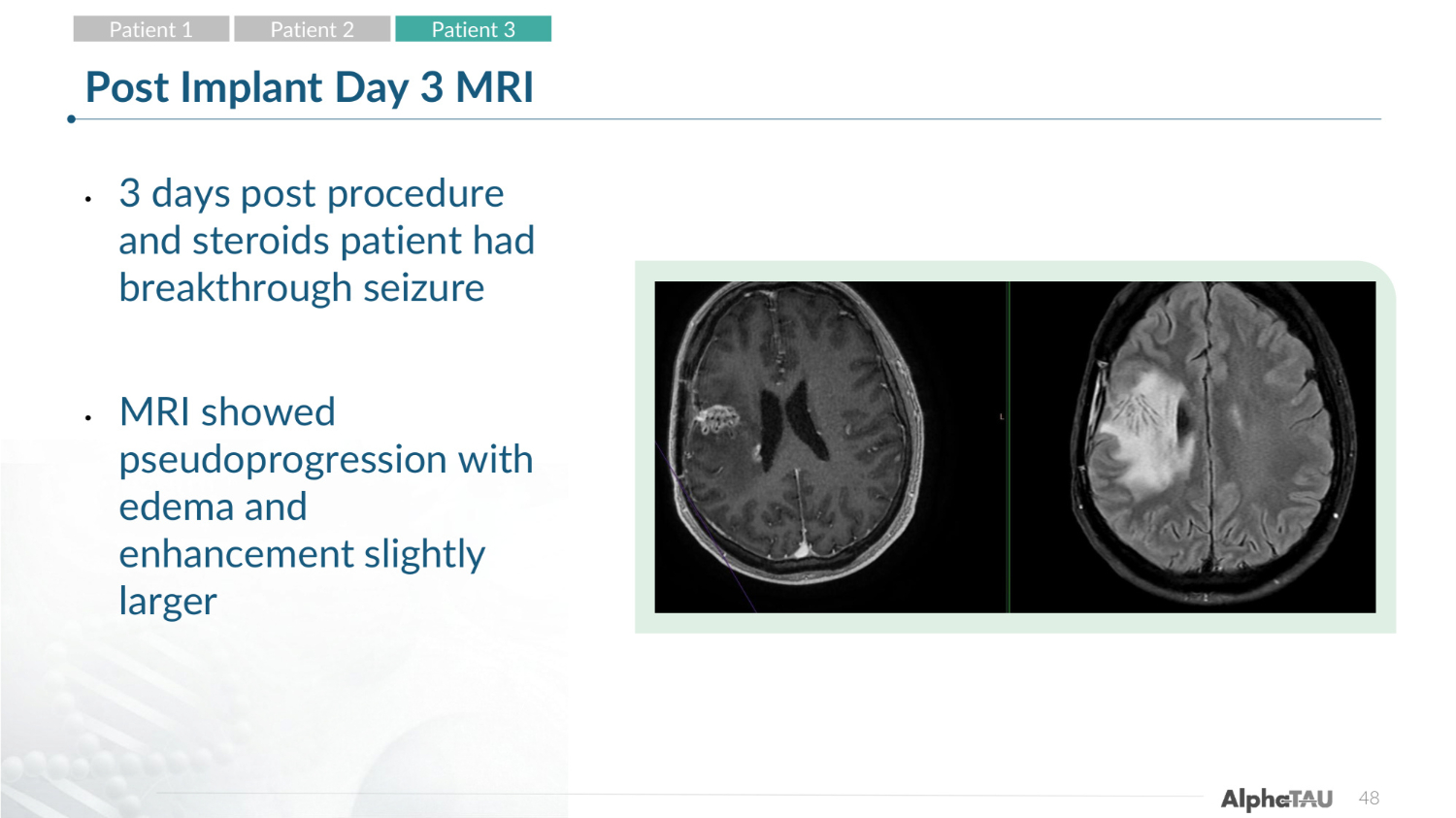
48 Post Implant Day 3 MRI • 3 days post procedure and steroids patient had breakthrough seizure • MRI showed pseudoprogression with edema and enhancement slightly larger Patient 1 Patient 2 Patient 3
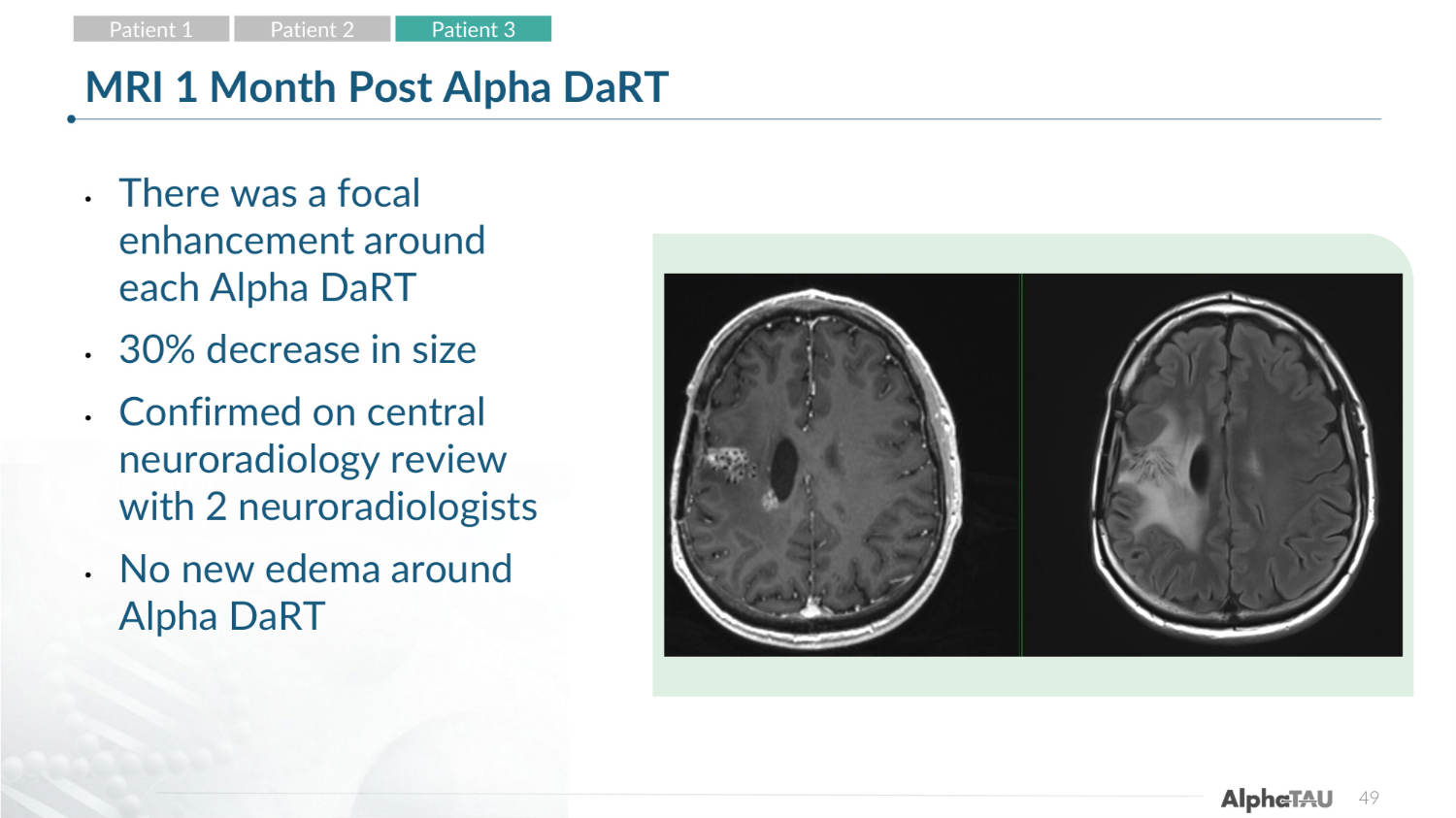
49 MRI 1 Month Post Alpha DaRT • There was a focal enhancement around each Alpha DaRT • 30% decrease in size • Confirmed on central neuroradiology review with 2 neuroradiologists • No new edema around Alpha DaRT Patient 1 Patient 2 Patient 3

50 Adverse Events* • He developed a grade 2 seizure • His symptoms improved to baseline on steroids and additional imaging showed normalizing edema to pretreatment extent • Doing well and back to his neurologic baseline • His most recent response was Stable Disease per RANO criteria with a 30% reduction in size on 1 month MRI Patient 1 Patient 2 Patient 3 * Interim Data as of 3-May-2026

51 51 Summary* • Three patients have been treated • There were no unanticipated associated side effects noted • All patients' tumors have responded with 2 complete responses and one stable disease (30% size reduction) • Plan for completion of enrollment of 10 patients on this pilot study pending FDA review * Interim Data as of 3-May-2026

Questions? 52