

aTyr Pharma Advancing tRNA synthetase biology Efzofitimod in Pulmonary Sarcoidosis and Beyond May 11, 2026 © 2026 aTyr Pharma Exhibit 99.1

Forward-Looking Statements The following slides and any accompanying oral presentation contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. The use of words such as “may,” “might,” “will,” “should,” “expect,” “plan,” “anticipate,” “believe,” “estimate,” “project,” “intend,” “future,” “potential,” “opportunity,” or “continue,” and other similar expressions are intended to identify forward-looking statements. For example, all statements regarding: the potential therapeutic benefits of proteins derived from tRNA synthetase genes and our product candidates and development programs; the ability to successfully advance our product candidates and undertake certain development activities (such as the initiation of clinical trials, clinical trial enrollment, the conduct of clinical trials and announcement of clinical results) and accomplish certain development goals, and the timing of such events; the potential market opportunity for our product candidates; our ability to receive regulatory approvals for, and commercialize, our product candidates; our ability to identify and discover additional product candidates; potential activities and payments under collaboration agreements; and the ability of our intellectual property portfolio to provide protection are forward-looking statements. All forward-looking statements are based on estimates and assumptions by our management that, although we believe to be reasonable, are inherently uncertain. All forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those that we expected. These risks, uncertainties and other factors are more fully described in our filings with the U.S. Securities and Exchange Commission, including our Annual Report on Form 10-K, our subsequently filed Quarterly Reports on Form 10-Q, and in our other filings. The forward-looking statements in this presentation speak only as of the date of this presentation and neither we nor any other person assume responsibility for the accuracy and completeness of any forward-looking statement. We undertake no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law. We own various U.S. federal trademark applications and unregistered trademarks, including our company name. All other trademarks or trade names referred to in this presentation are the property of their respective owners. Solely for convenience, the trademarks and trade names in this presentation are referred to without the symbols ® and ™, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. This presentation discusses product candidates that are under clinical study and which have not yet been approved for marketing by the U.S. Food and Drug Administration. No representation is made as to the safety or effectiveness of these product candidates for the uses for which they are being studied. This presentation also contains estimates and other statistical data made by independent parties and by us relating to market size and growth and other data about our industry. This data involved a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. | © 2026 aTyr Pharma

FDA Type C Meeting Informs Future Efzofitimod Development Background EFZO-FIT missed its primary endpoint but demonstrated a safety profile consistent with previous trials and durable benefit on multiple clinically meaningful, prespecified quality of life measures aTyr was granted a Type C meeting to discuss the EFZO-FIT data, clinical development plan and validation of KSQ-L as a fit-for-purpose endpoint for pulmonary sarcoidosis Discussion focused on: Endpoints (FVC; KSQ-L) Our rationale for FVC as endpoint in target patient population Dosing (Q3W) Safety monitoring Benefit / risk profile FDA Type C Meeting Meeting objective: align on the path forward for efzofitimod in pulmonary sarcoidosis Abbreviations: FDA, Food and Drug Administration; KSQ-L, King’s Sarcoidosis Questionnaire-Lung; FVC, forced vital capacity | © 2026 aTyr Pharma

FDA Type C Meeting Informs Future Efzofitimod Development FDA indicated support for FVC and KSQ-L as clinically meaningful endpoints in pulmonary sarcoidosis FDA indicated FVC to be a direct measure of how patients function FDA indicated KSQ-L to be a direct measure of how patients feel, and recommended the inclusion of wheezing and cough severity as separate measures of patient symptoms along with new anchors to interpret the clinically meaningful threshold, based on further content validation Our conclusion: FVC is a more appropriate primary endpoint for C-006, considering FDA has not fully endorsed KSQ-L as fit-for-purpose FDA reviewed data presenting the rationale for utilizing FVC as an endpoint in the target population for C-006 based on data from EFZO-FIT FDA Type C Meeting Meeting outcome: Company to continue development of efzofitimod in pulmonary sarcoidosis incorporating FDA feedback; Company to pursue with new Phase 3 study (C-006) Abbreviations: FDA, Food and Drug Administration; KSQ-L, King’s Sarcoidosis Questionnaire-Lung; FVC, forced vital capacity | © 2026 aTyr Pharma
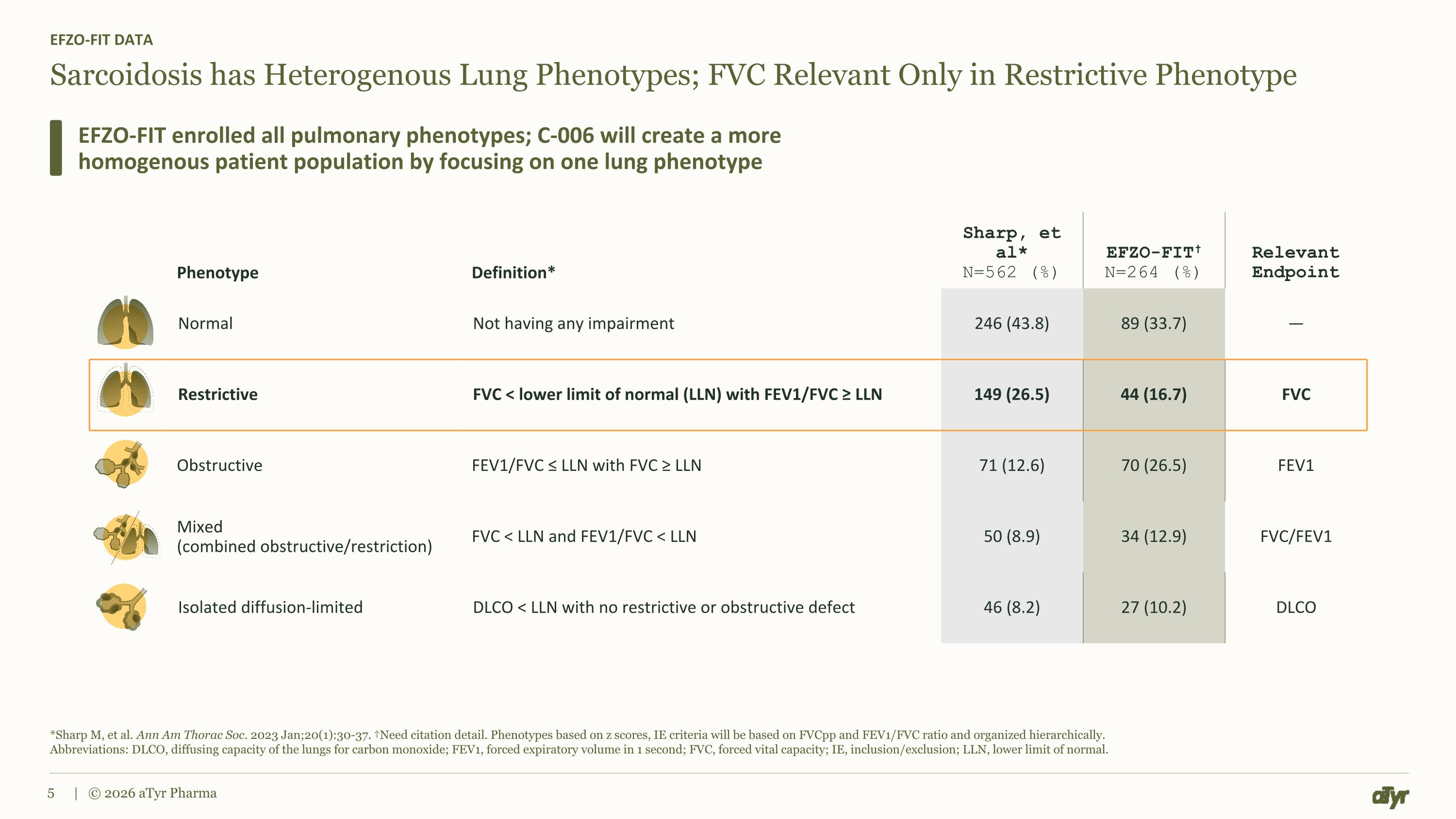
Sarcoidosis has Heterogenous Lung Phenotypes; FVC Relevant Only in Restrictive Phenotype EFZO-FIT DATA EFZO-FIT enrolled all pulmonary phenotypes; C-006 will create a more homogenous patient population by focusing on one lung phenotype *Sharp M, et al. Ann Am Thorac Soc. 2023 Jan;20(1):30-37. †Need citation detail. Phenotypes based on z scores, IE criteria will be based on FVCpp and FEV1/FVC ratio and organized hierarchically. Abbreviations: DLCO, diffusing capacity of the lungs for carbon monoxide; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; IE, inclusion/exclusion; LLN, lower limit of normal. | © 2026 aTyr Pharma Phenotype Definition* Sharp, et al* N=562 (%) EFZO-FIT† N=264 (%) Relevant Endpoint Normal Not having any impairment 246 (43.8) 89 (33.7) — Restrictive FVC < lower limit of normal (LLN) with FEV1/FVC ≥ LLN 149 (26.5) 44 (16.7) FVC Obstructive FEV1/FVC ≤ LLN with FVC ≥ LLN 71 (12.6) 70 (26.5) FEV1 Mixed (combined obstructive/restriction) FVC < LLN and FEV1/FVC < LLN 50 (8.9) 34 (12.9) FVC/FEV1 Isolated diffusion-limited DLCO < LLN with no restrictive or obstructive defect 46 (8.2) 27 (10.2) DLCO
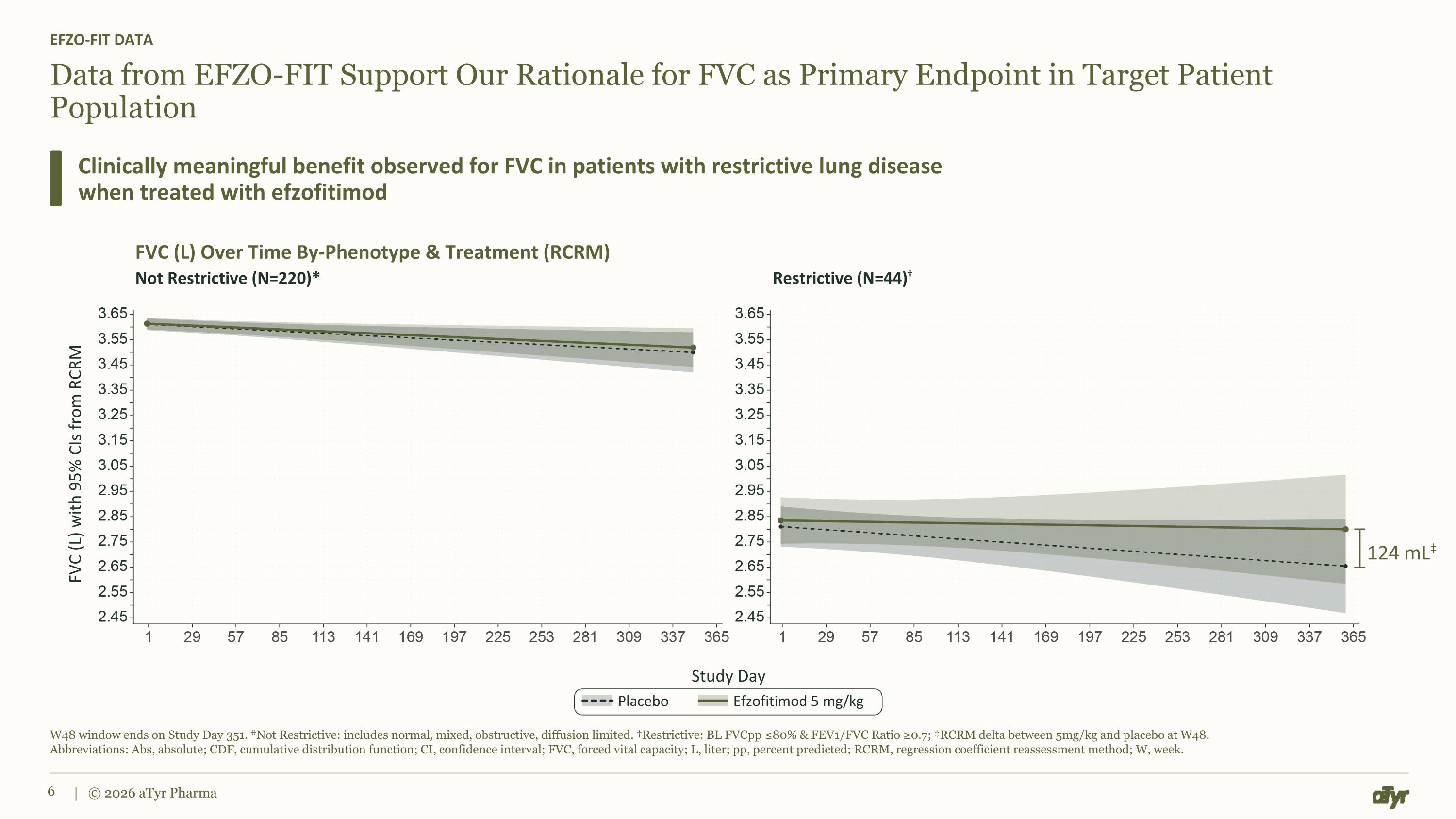
Data from EFZO-FIT Support Our Rationale for FVC as Primary Endpoint in Target Patient Population EFZO-FIT DATA Clinically meaningful benefit observed for FVC in patients with restrictive lung disease when treated with efzofitimod W48 window ends on Study Day 351. *Not Restrictive: includes normal, mixed, obstructive, diffusion limited. †Restrictive: BL FVCpp ≤80% & FEV1/FVC Ratio ≥0.7; ‡RCRM delta between 5mg/kg and placebo at W48. Abbreviations: Abs, absolute; CDF, cumulative distribution function; CI, confidence interval; FVC, forced vital capacity; L, liter; pp, percent predicted; RCRM, regression coefficient reassessment method; W, week. | © 2026 aTyr Pharma FVC (L) with 95% CIs from RCRM Study Day FVC (L) Over Time By-Phenotype & Treatment (RCRM) Not Restrictive (N=220)* Restrictive (N=44)† Placebo Efzofitimod 5 mg/kg 124 mL‡

FDA Type C Meeting Informs Future Efzofitimod Development FDA acknowledged reasonableness of the proposed Q3W dosing in the proposed patient population in C-006 Aligned with FDA on incorporating additional risk mitigation strategies in the protocol Safety surveillance for the potential for development of anti-synthetase syndrome will be closely and prospectively monitored Data Monitoring Committee will be in place Benefit / risk profile Efzofitimod’s benefit / risk profile supports continued development with Q3W dosing in proposed population Company plans to file IND for C-006 in June 2026 FDA Type C Meeting Meeting outcome: Company to continue development of efzofitimod in pulmonary sarcoidosis incorporating FDA feedback; Company to pursue with new Phase 3 study (C-006) Abbreviations: FDA, Food and Drug Administration; KSQ-L, King’s Sarcoidosis Questionnaire-Lung; FVC, forced vital capacity, IND, investigational new drug | © 2026 aTyr Pharma

Go-Forward Development Plan Leverages Insights from EFZO-FIT and Engagement with FDA ATYR1923-C-006 Study design to incorporate key changes intended to demonstrate compelling evidence of efficacy and safety on clinically meaningful endpoint Abbreviations: FVC, forced vital capacity; Q3W, every three weeks; Q4W, every four weeks. | © 2026 aTyr Pharma All lung phenotypes Forced steroid taper 5.0 mg/kg Q4W Steroid reduction at week 48 Study design Primary endpoint Dosing Restrictive lung phenotype Stable background treatment FVC at week 48 5.0 mg/kg Q3W EFZO-FIT New Trial
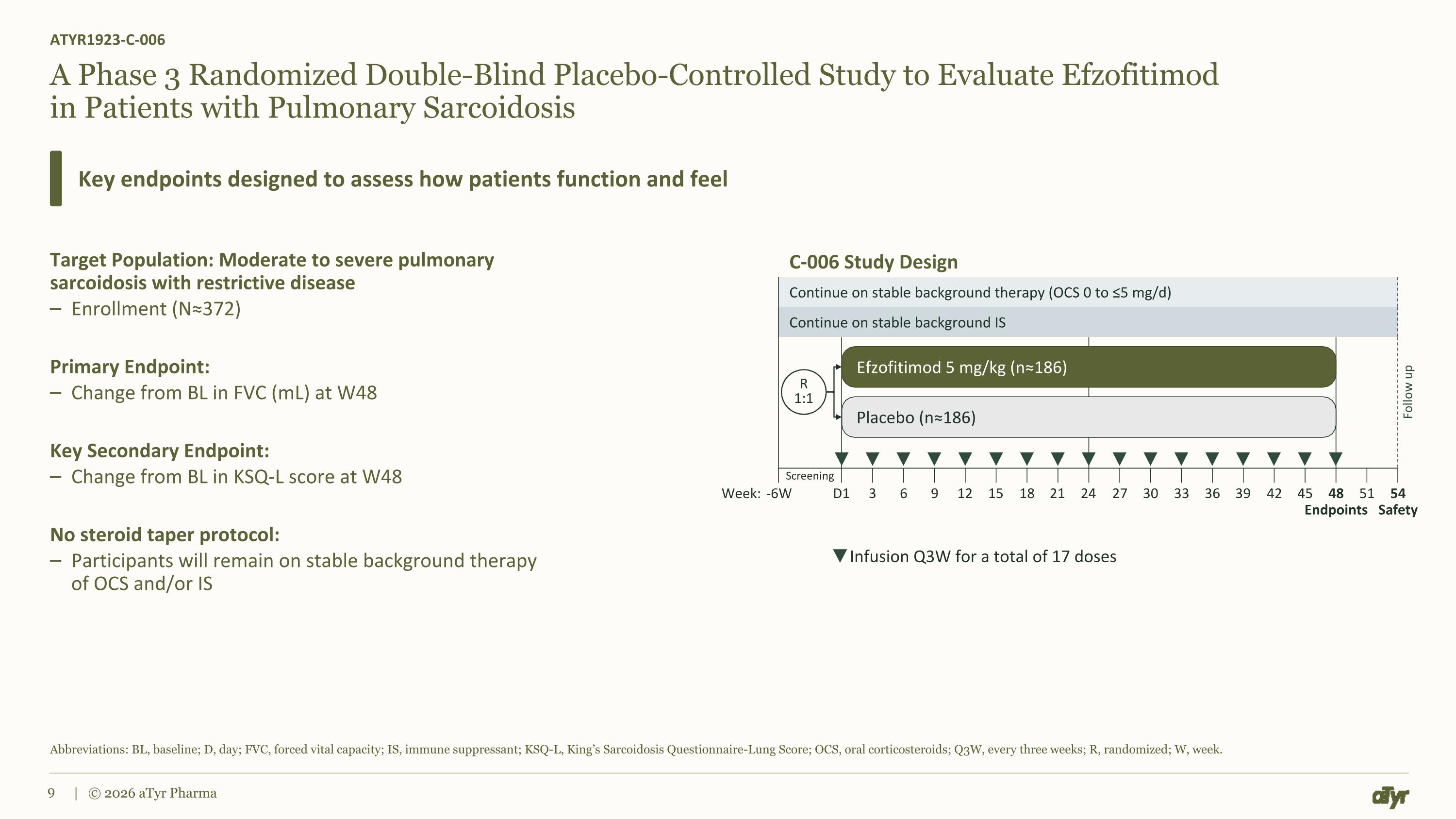
Continue on stable background therapy (OCS 0 to ≤5 mg/d) Continue on stable background IS Screening A Phase 3 Randomized Double-Blind Placebo-Controlled Study to Evaluate Efzofitimod in Patients with Pulmonary Sarcoidosis Target Population: Moderate to severe pulmonary sarcoidosis with restrictive disease Enrollment (N≈372) Primary Endpoint: Change from BL in FVC (mL) at W48 Key Secondary Endpoint: Change from BL in KSQ-L score at W48 No steroid taper protocol: Participants will remain on stable background therapy of OCS and/or IS ATYR1923-C-006 Key endpoints designed to assess how patients function and feel Abbreviations: BL, baseline; D, day; FVC, forced vital capacity; IS, immune suppressant; KSQ-L, King’s Sarcoidosis Questionnaire-Lung Score; OCS, oral corticosteroids; Q3W, every three weeks; R, randomized; W, week. | © 2026 aTyr Pharma Efzofitimod 5 mg/kg (n≈186) Placebo (n≈186) R 1:1 Follow up Infusion Q3W for a total of 17 doses -6W 6 9 12 15 18 21 24 27 30 33 48 Endpoints 54 Safety D1 Week: 3 36 39 42 45 51 C-006 Study Design
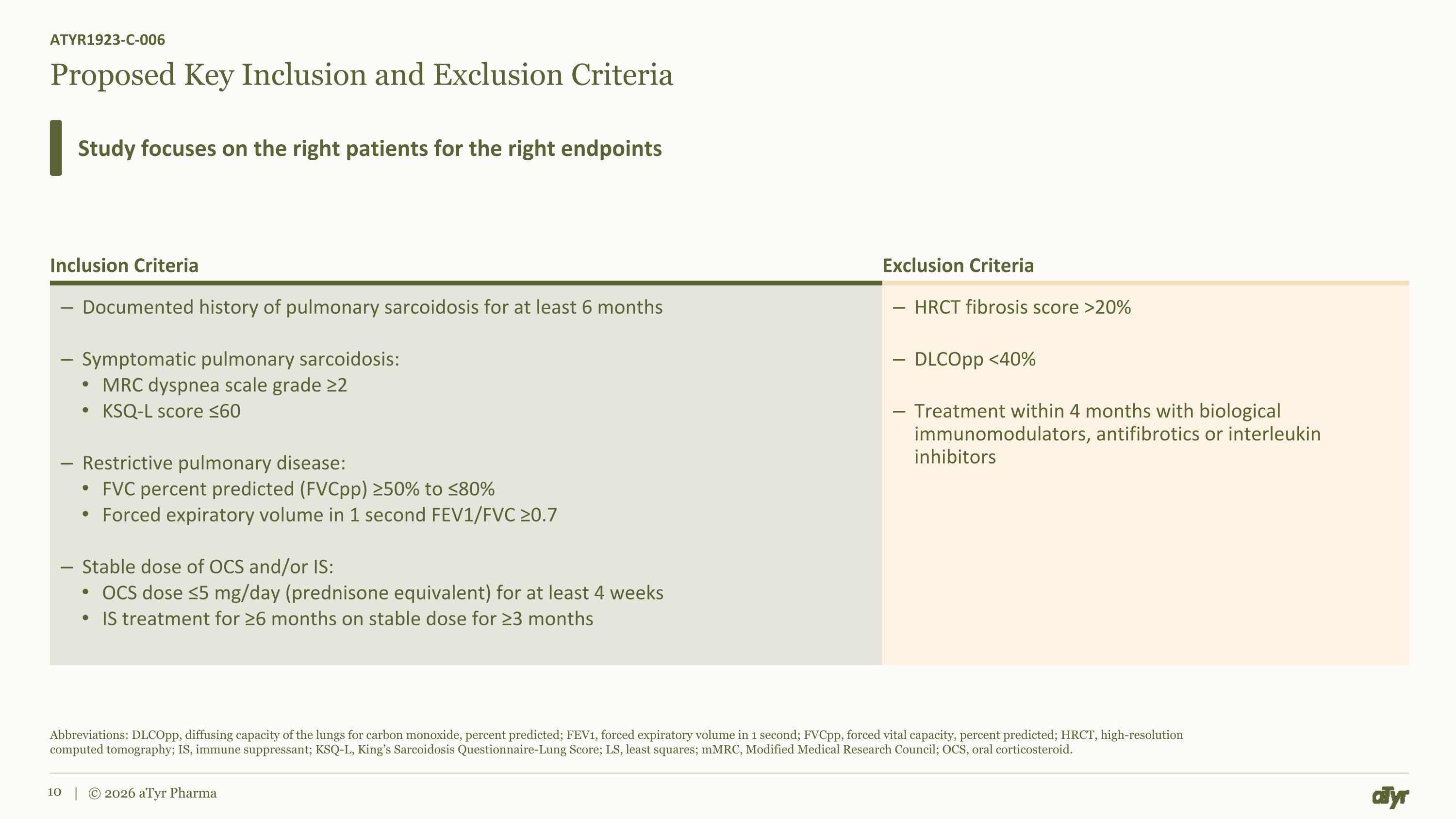
Proposed Key Inclusion and Exclusion Criteria ATYR1923-C-006 Study focuses on the right patients for the right endpoints Abbreviations: DLCOpp, diffusing capacity of the lungs for carbon monoxide, percent predicted; FEV1, forced expiratory volume in 1 second; FVCpp, forced vital capacity, percent predicted; HRCT, high-resolution computed tomography; IS, immune suppressant; KSQ-L, King’s Sarcoidosis Questionnaire-Lung Score; LS, least squares; mMRC, Modified Medical Research Council; OCS, oral corticosteroid. | © 2026 aTyr Pharma Inclusion Criteria Exclusion Criteria Documented history of pulmonary sarcoidosis for at least 6 months Symptomatic pulmonary sarcoidosis: MRC dyspnea scale grade ≥2 KSQ-L score ≤60 Restrictive pulmonary disease: FVC percent predicted (FVCpp) ≥50% to ≤80% Forced expiratory volume in 1 second FEV1/FVC ≥0.7 Stable dose of OCS and/or IS: OCS dose ≤5 mg/day (prednisone equivalent) for at least 4 weeks IS treatment for ≥6 months on stable dose for ≥3 months HRCT fibrosis score >20% DLCOpp <40% Treatment within 4 months with biological immunomodulators, antifibrotics or interleukin inhibitors
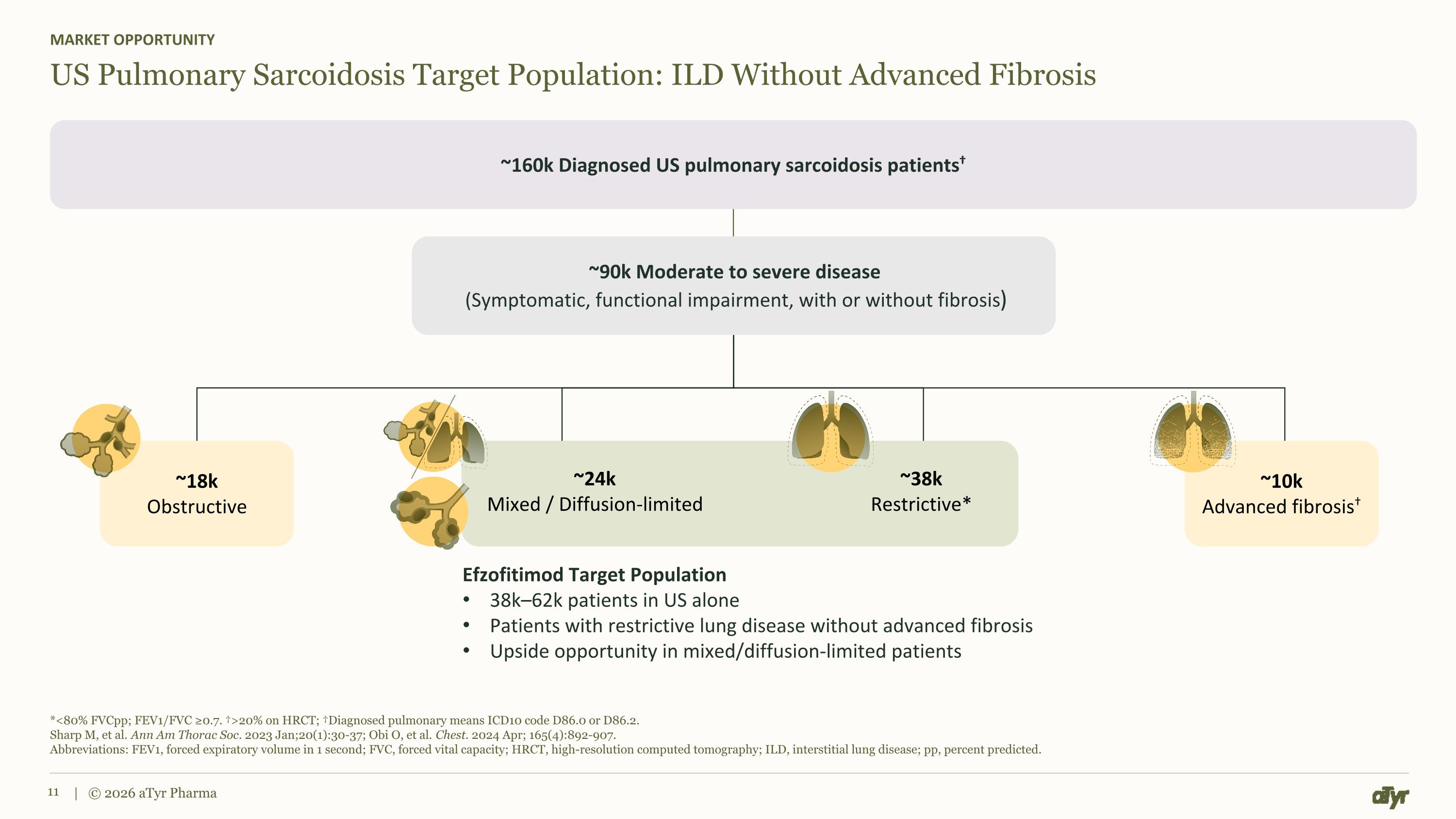
*<80% FVCpp; FEV1/FVC ≥0.7. †>20% on HRCT; †Diagnosed pulmonary means ICD10 code D86.0 or D86.2. Sharp M, et al. Ann Am Thorac Soc. 2023 Jan;20(1):30-37; Obi O, et al. Chest. 2024 Apr; 165(4):892-907. Abbreviations: FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; HRCT, high-resolution computed tomography; ILD, interstitial lung disease; pp, percent predicted. | © 2026 aTyr Pharma US Pulmonary Sarcoidosis Target Population: ILD Without Advanced Fibrosis MARKET OPPORTUNITY ~160k Diagnosed US pulmonary sarcoidosis patients† ~90k Moderate to severe disease (Symptomatic, functional impairment, with or without fibrosis) Efzofitimod Target Population 38k–62k patients in US alone Patients with restrictive lung disease without advanced fibrosis Upside opportunity in mixed/diffusion-limited patients ~18k Obstructive ~24k Mixed / Diffusion-limited ~38k Restrictive* ~10k Advanced fibrosis†
| © 2026 aTyr Pharma Thank You