
Exhibit 99.1

Copyright © 2023 Allarity Therapeutics. All rights reserved. Nasdaq: ALLR Dual inhibition of PARP and WNT: A novel drug - biomarker combination in clinical trials Thomas Jensen, CEO

2 Allarity Therapeutics Legal Statement This presentation is provided for informational purposes only and is subject to change. This presentation is furnished as Exh ibi t 99.1 to the Company’s Current Report on Form 8 - K pursuant to Item 7.01. The information in this presentation shall not be deemed “filed” for purposes of Section 18 of the Securities E xch ange Act of 1934, nor incorporated by reference into any filing under the Securities Act of 1933 or the Exchange Act, except as expressly set forth by specific reference in such filing. The in formation contained herein does not purport to be all - inclusive. The data contained herein is derived from internal and external sources, and may rely upon assumptions, stated or otherwise, and forward - looking statements discussed below. No representation is made as to the reasonableness of the assumptions made within or the accuracy or completeness of any forward - lo oking statements, modeling or information contained herein. Allarity Therapeutics, Inc. (collectively “Allarity Therapeutics,” “Allarity,” or the “Company”) assume no obligation to update the information in this presentation. This material is not for the benefit of, and does not convey any rights or remedies for the benefit of, any holder of securities or any other person. Thi s material is not intended to provide the basis for evaluation of any transaction and does not purport to contain all information that may be required and should not be considered a recommend ati on or opinion of any kind with respect to any transaction. No Offer or Solicitation. This material shall not constitute an offer to sell or a solicitation of an offer to buy any securities, nor shall there be a ny sale of such securities in any state or jurisdiction in which such offer, solicitation, or sale would be unlawful prior to registration or qualification under the se cur ities laws of such state or jurisdiction. No offer of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the Securities Act of 1933, as amended. Forward - Looking Statements. This presentation contains “forward - looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Forward - looking statements provide Allarity’s current expectations or forecasts of future events. The words “anticipates,” “believe,” “contin ue, ” “could,” “estimate,” “expect,” “intends,” “may,” “might,” “plan,” “possible,” “potential,” “predicts,” “project,” “should,” “would” and similar expressions may identify forward - looking statement s, but the absence of these words does not mean that a statement is not forward - looking. These forward - looking statements include, but are not limited to, statements relating to interim clinical data readouts, regula tory milestones, and the ongoing clinical development of stenoparib and its DRP® companion diagnostic. Any forward - looking statements in this presentatio n are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and ad versely from those set forth in or implied by such forward - looking statements. These risks and uncertainties include, but are not limited to, the risk that that the Company is unable to raise suf ficient capital to fund its ongoing and prospective clinical trials and operations, the risk that results of a clinical study do not necessarily predict final results and that one or more of th e c linical outcomes may materially change following more comprehensive reviews of the data, and as more patient data become available, the risk that results of a clinical study are s ubj ect to interpretation and additional analyses may be needed and/or may contradict such results, the receipt of regulatory approval for stenoparib, the drug - specific DRP ® companion diagnostic for stenoparib , if approved, the successful commercialization of such products, the risk of cessation or delay of any of the ongoing or planned clinical trials and/or ou r d evelopment of our product candidate, and the risk that the results of previously conducted studies will not be repeated or observed in ongoing or future studies involving our therapeutic candi dat es. For a discussion of other risks and uncertainties, and other important factors, any of which could cause our actual results to differ from those contained in the forward - looking statements, see the section entitled “Risk Factors” in our Form 10 - K annual report filed with the Securities and Exchange Commission (the ‘SEC’) on March 31, 2025 , available at the Securities and Exchange Commission’s website at www.sec.gov , and as well as discussions of potential risks, uncertainties and other important factors in the Company’s subsequent filings with the Securitie s and Exchange Commission. All information in this presentation is as of the date of the presentation , and the Company undertakes no duty to update this information unless required by law. Any financial projections in this presentation are forward - looking statements that are based on assumptions that are inherently subject to significant uncertainties and contingencies, many of which are beyond Allarity’s control. While all projections are necessarily speculative, Allarity believes that the preparatio n o f prospective financial information involves increasingly higher levels of uncertainty the further out the projection extends from the date of preparation. The assumptions and estimates unde rly ing the projected results are inherently uncertain and are subject to a wide variety of significant business, economic and competitive risks and uncertainties that could cause actual r esu lts to differ materially from those contained in the projections. The inclusion of projections in this communication should not be regarded as an indication that Al larity or their representatives, considered or consider the projections to be a reliable prediction of future events.

3 Allarity Therapeutics Corporate Overview Allarity Therapeutics: A Unique Approach to Advancing a Unique Cancer Therapy » Allarity was founded on its proprietary gene expression technology - the Drug Response Predictor (DRP) o DRPs have successfully been developed for dozens of individual cancer drugs » Allarity has exclusive world - wide rights to Stenoparib (2X - 121, E7449) a drug originally developed by Eisai Pharma » Stenoparib has a unique mechanism of therapeutic action, inhibiting PARP and the WNT pathway » Unlike 1 st generation PARP inhibitors, Stenoparib shows a favorable myelotoxicity profile » Stenoparib given twice daily has shown durable clinical benefit in Advanced, Recurrent Ovarian Cancer o Phase 2 study in platinum resistant or refractory Ovarian Cancer o Unique clinical activity - provides clinical benefit regardless of BRCA status, prior PARPi treatment o Shows extended median Overall Survival - 22 months by K - M analysis o Newest treatments advanced for Platinum Resistant Ovarian cancer show 16 months mOS » Currently Enrolling 2 new Phase 2 trials o Monotherapy in Platinum Resistant Ovarian Cancer, 2 dose cohorts, started summer 2025 o In combination with Temozolomide in relapsed SCLC - randomized phase 2 fully funded by the VA

Our DRP ® Companion Diagnostics Platform
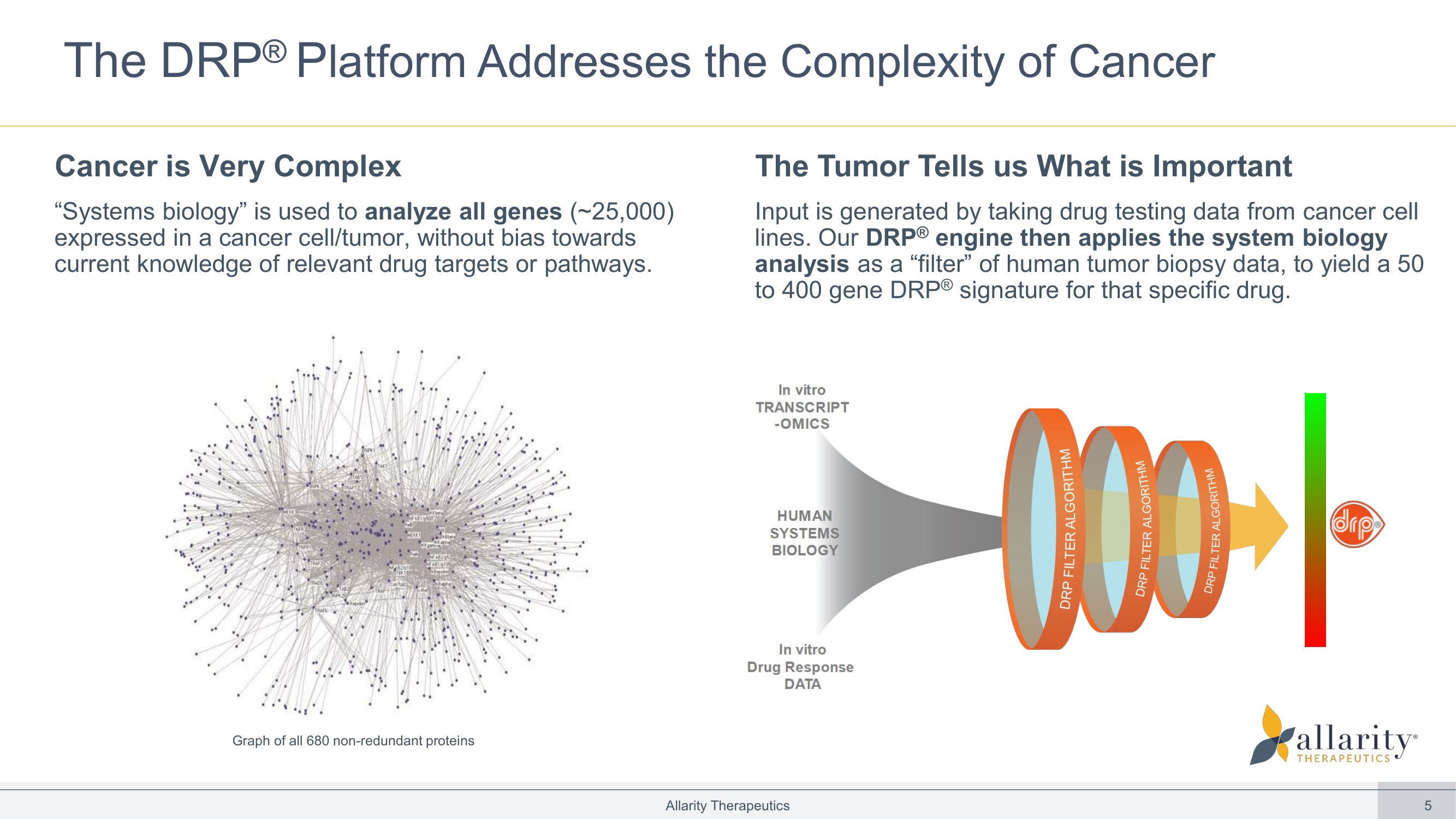
5 In vitro TRANSCRIPT -OMICS HUMAN SYSTEMS BIOLOGY In vitro Drug Response DATA Multi Gene DRP DRUG BLIND CLINICAL VALIDATION 14 The DRP ® P latform Addresses the Complexity of Cancer Cancer is Very Complex “Systems biology” is used to analyze all genes (~25,000) expressed in a cancer cell/tumor, without bias towards current knowledge of relevant drug targets or pathways. The Tumor Tells us What is Important Input is generated by taking drug testing data from cancer cell lines. Our DRP ® engine then applies the system biology analysis as a “filter” of human tumor biopsy data, to yield a 50 to 400 gene DRP ® signature for that specific drug. Allarity Therapeutics Graph of all 680 non - redundant proteins
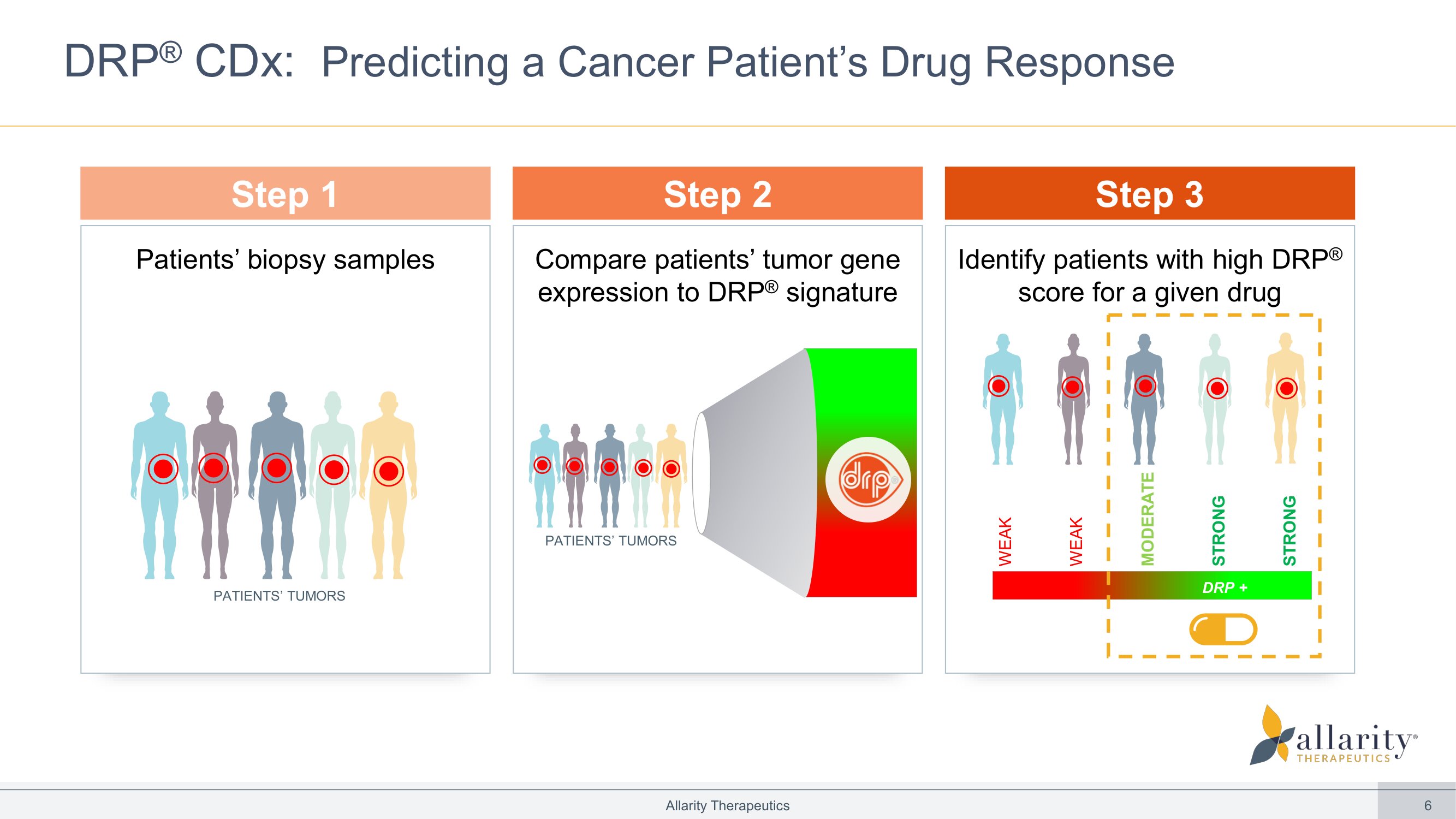
6 Compare patients’ tumor gene expression to DRP ® signature Allarity Therapeutics DRP ® CDx: Predicting a Cancer Patient’s Drug Response Identify patients with high DRP ® score for a given drug STRONG WEAK Patients’ biopsy samples Step 1 Step 2 Step 3 WEAK MODERATE STRONG PATIENTS’ TUMORS DRP + PATIENTS’ TUMORS

7 PR SD24w+ SD24w− PD 4 0 5 0 6 0 7 0 8 0 9 0 1 0 0 Results of LiPlaCis Phase II Trial Best overall response D R P p r e d i c t e d L i P l a C i s s e n s i t i v i t y PR SD24w+ SD24w− PD 4 0 5 0 6 0 7 0 8 0 9 0 1 0 0 CC=0.40, P=0.007 Allarity Therapeutics . * Nielsen et al: Predictive biomarker for cisplatin in prospective phase 2 of liposomal cisplatin in metastatic breast cancer . ASCO 2023 abstract 3130 LiPlaCis : Prospective enrollment based on DRP identifies patients with greatest clinical benefit Results of LiPlaCis Phase II Trial * Allarity’s LiPlaCis DRP ® was used to screen patients for inclusion into trial based on diagnostic biopsy. Metastatic Breast Cancer patients with a DRP score above 33 were treated with LiPlaCis (N=37). All responders had a DRP score >80 (ORR was 25% above this cutoff, 0% below) The mean DRP score for SD>24w was 80.
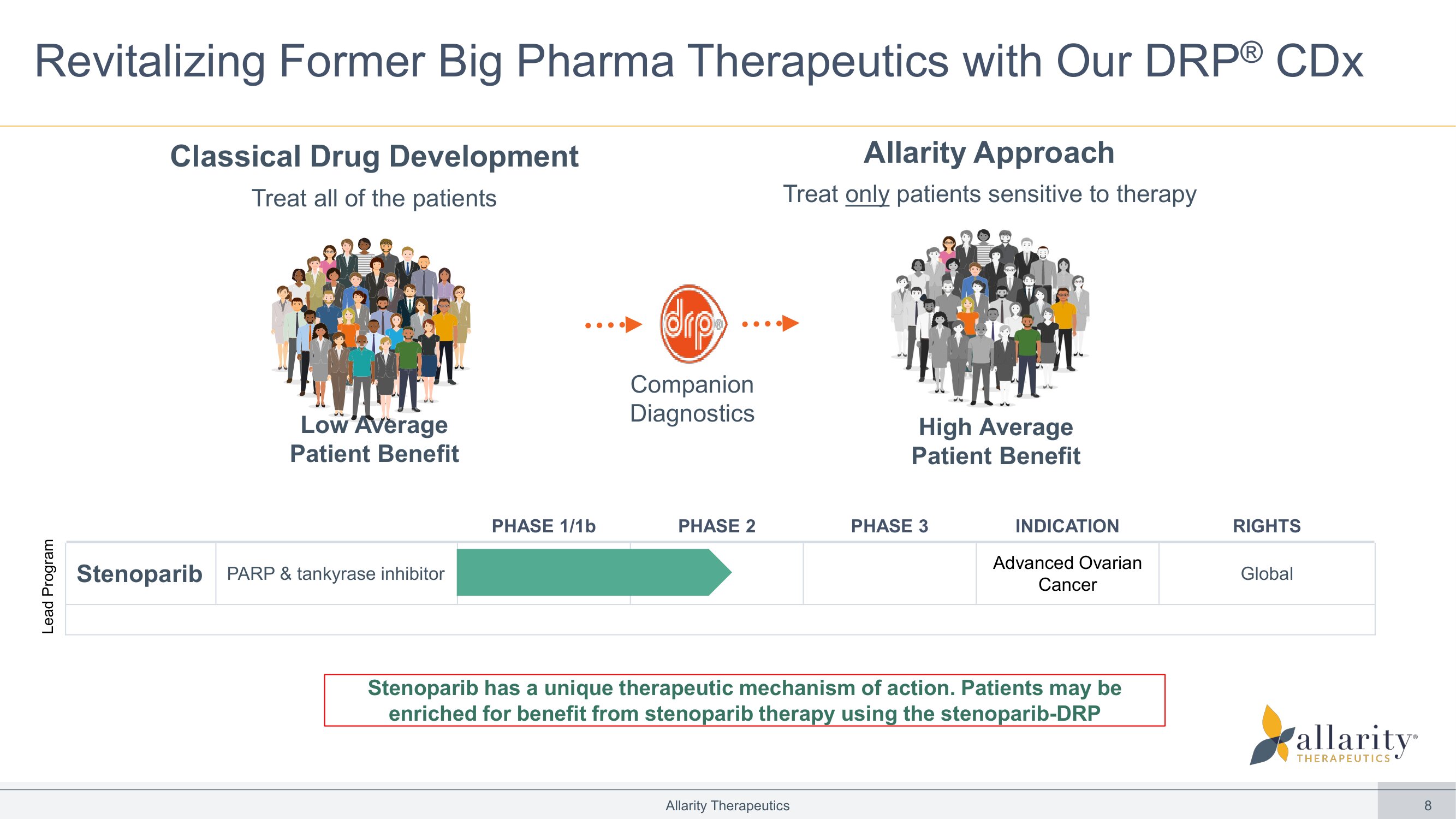
8 Allarity Therapeutics Revitalizing Former Big Pharma Therapeutics with Our DRP ® CDx Companion Diagnostics Low Average Patient Benefit High Average Patient Benefit Classical Drug Development Treat all of the patients Allarity Approach Treat only patients sensitive to therapy RIGHTS INDICATION PHASE 3 PHASE 2 PHASE 1/1b Global Advanced Ovarian Cancer PARP & tankyrase inhibitor Stenoparib Lead Program Stenoparib has a unique therapeutic mechanism of action. Patients may be enriched for benefit from stenoparib therapy using the stenoparib - DRP

Ongoing Phase 2 Clinical trials: Stenoparib monotherapy in recurrent Platinum Resistant Ovarian cancer as well as Relapsed Small Cell Lung Cancer in combination with Temozolomide
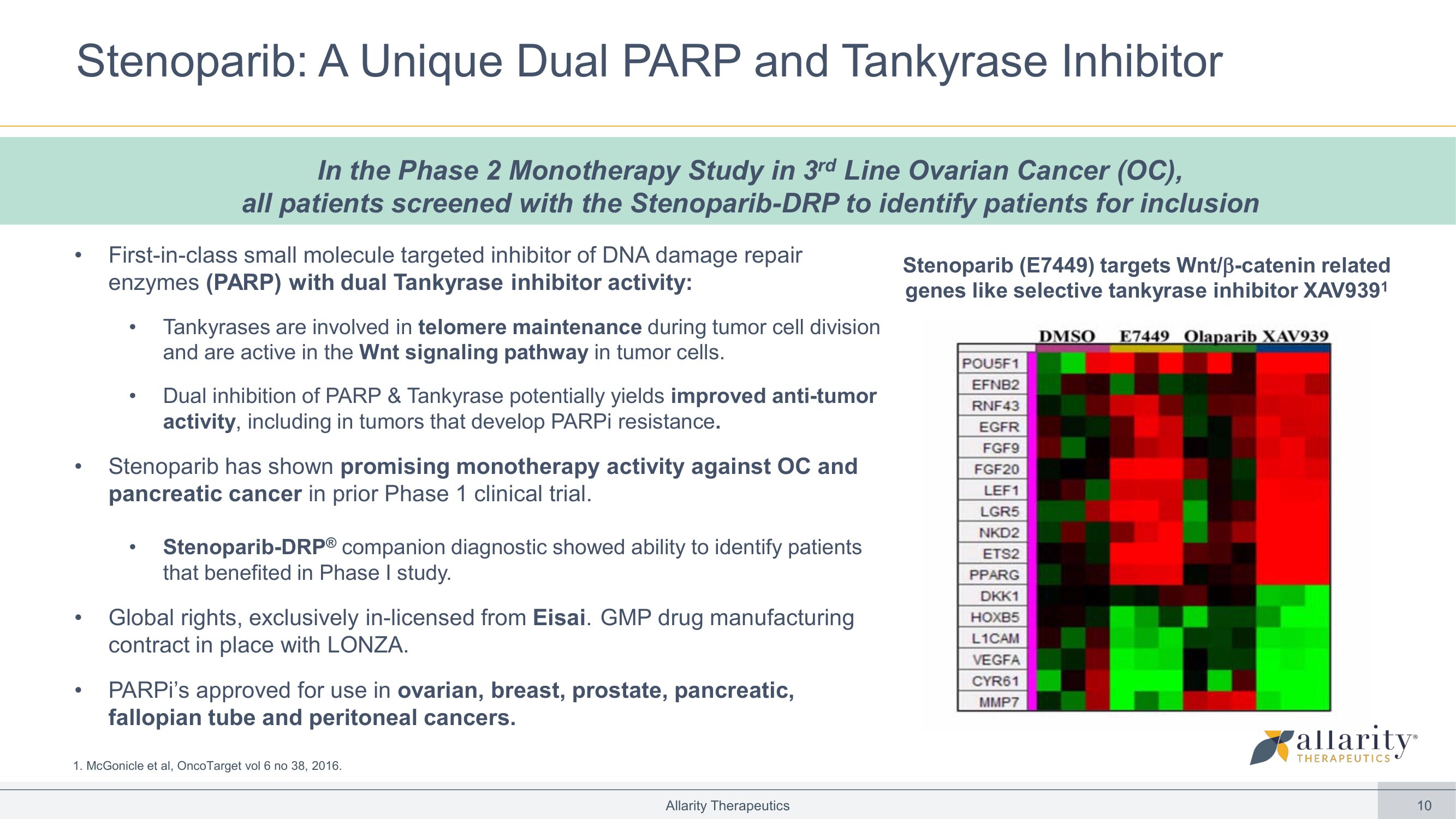
10 • First - in - class small molecule targeted inhibitor of DNA damage repair enzymes (PARP) with dual Tankyrase inhibitor activity: • Tankyrases are involved in telomere maintenance during tumor cell division and are active in the Wnt signaling pathway in tumor cells. • Dual inhibition of PARP & Tankyrase potentially yields improved anti - tumor activity , including in tumors that develop PARPi resistance . • Stenoparib has shown promising monotherapy activity against OC and pancreatic cancer in prior Phase 1 clinical trial. • Stenoparib - DRP ® companion diagnostic showed ability to identify patients that benefited in Phase I study. • Global rights, exclusively in - licensed from Eisai . GMP drug manufacturing contract in place with LONZA. • PARPi’s approved for use in ovarian, breast, prostate, pancreatic, fallopian tube and peritoneal cancers. Allarity Therapeutics 1. McGonicle et al, OncoTarget vol 6 no 38, 2016. Stenoparib: A Unique Dual PARP and Tankyrase Inhibitor In the Phase 2 Monotherapy Study in 3 rd Line Ovarian Cancer (OC), all patients screened with the Stenoparib - DRP to identify patients for inclusion Stenoparib (E7449) targets Wnt/ - catenin related genes like selective tankyrase inhibitor XAV939 1 0 2,000 4,000 6,000 8,000 10,000 12,000 2019A 2026E Ovarian cancer Pancreatic cancer

11 Allarity Therapeutics 1. Lou Stancato et al: AACR Poster 2026. Stenoparib: Wnt / - catenin pathway modulation Active (non - phosphorylated) Beta Catenin TCF - LEF Promoter Reporter Activity (Colo320 72 hrs )
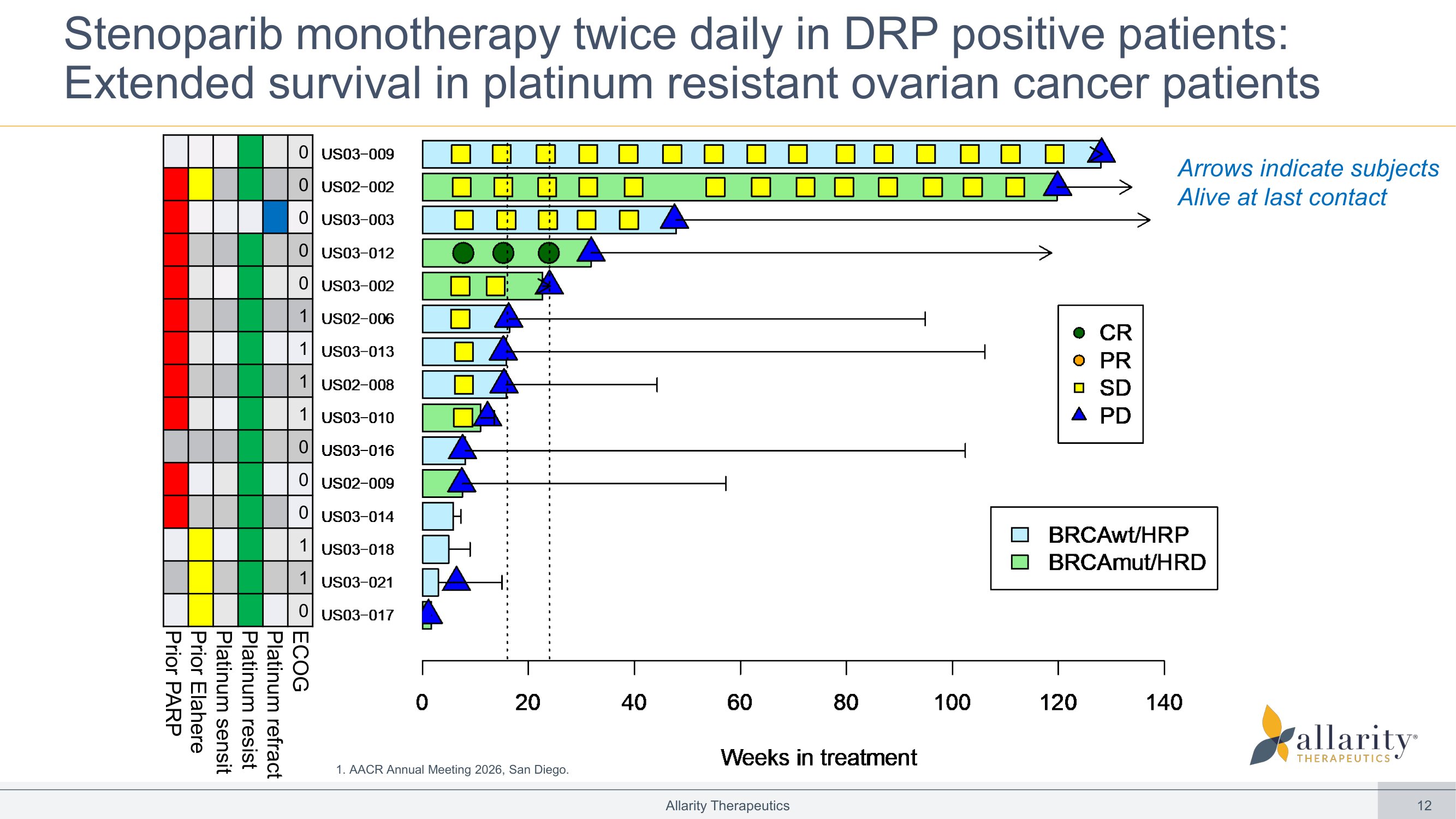
12 Stenoparib monotherapy twice daily in DRP positive patients: Extended survival in platinum resistant ovarian cancer patients Allarity Therapeutics 0 0 0 0 0 1 1 1 1 0 0 0 1 1 0 ECOG Platinum refract Platinum resist Platinum sensit Prior Elahere Prior PARP Arrows indicate subjects A live at last contact US03−017 US03−021 US03−018 US03−014 US02−009 US03−016 US03−010 US02−008 US03−013 US02−006 US03−002 US03−012 US03−003 US02−002 US03−009 BRCAwt/HRP BRCAmut/HRD Weeks in treatment 0 20 40 60 80 100 120 140 CR PR SD PD 1. AACR Annual Meeting 2026, San Diego.
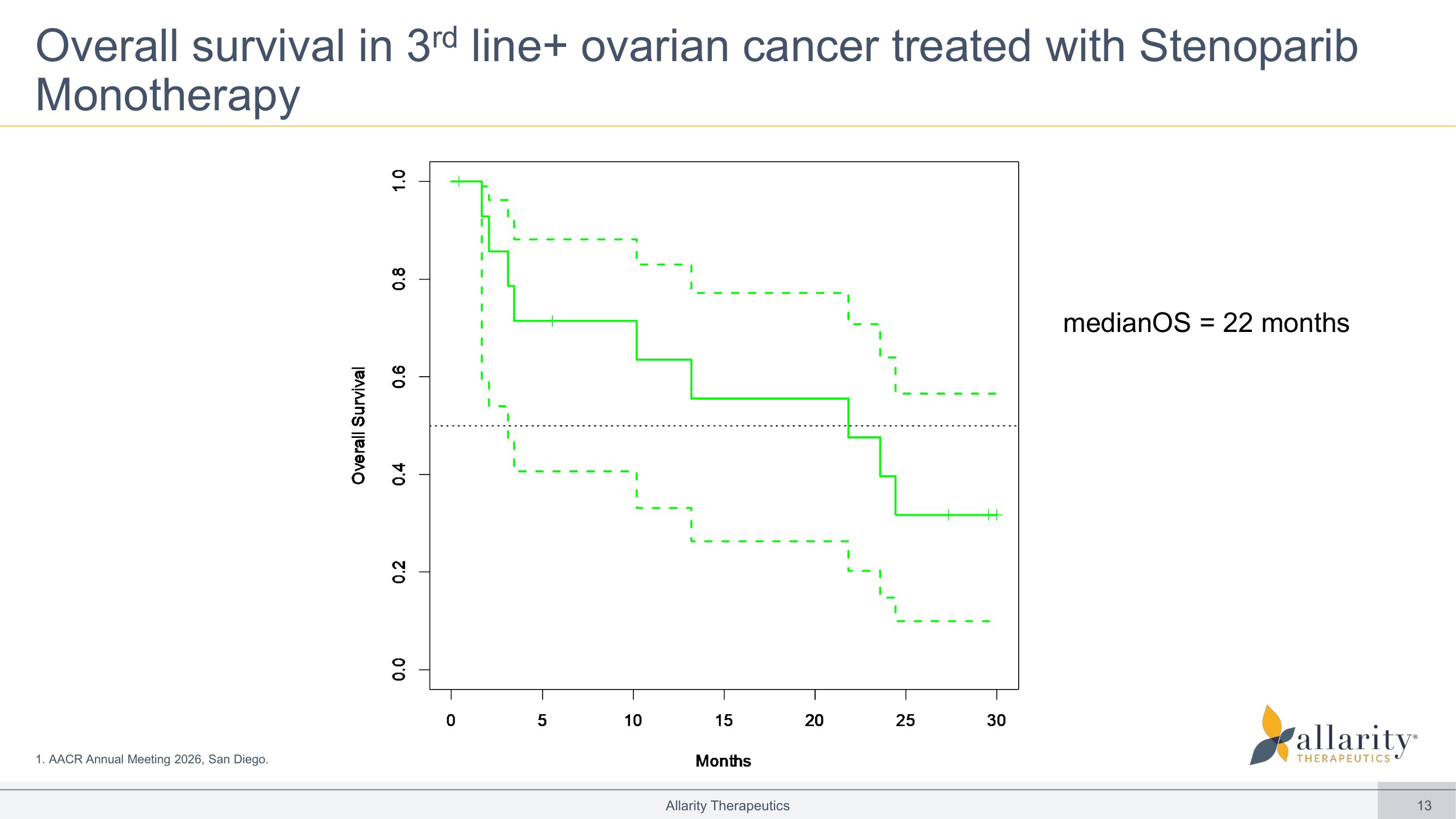
13 Overall survival in 3 rd line+ ovarian cancer treated with Stenoparib Monotherapy Allarity Therapeutics 0 5 10 15 20 25 30 0 . 0 0 . 2 0 . 4 0 . 6 0 . 8 1 . 0 Months O v e r a l l S u r v i v a l m edianOS = 22 months 1. AACR Annual Meeting 2026, San Diego.
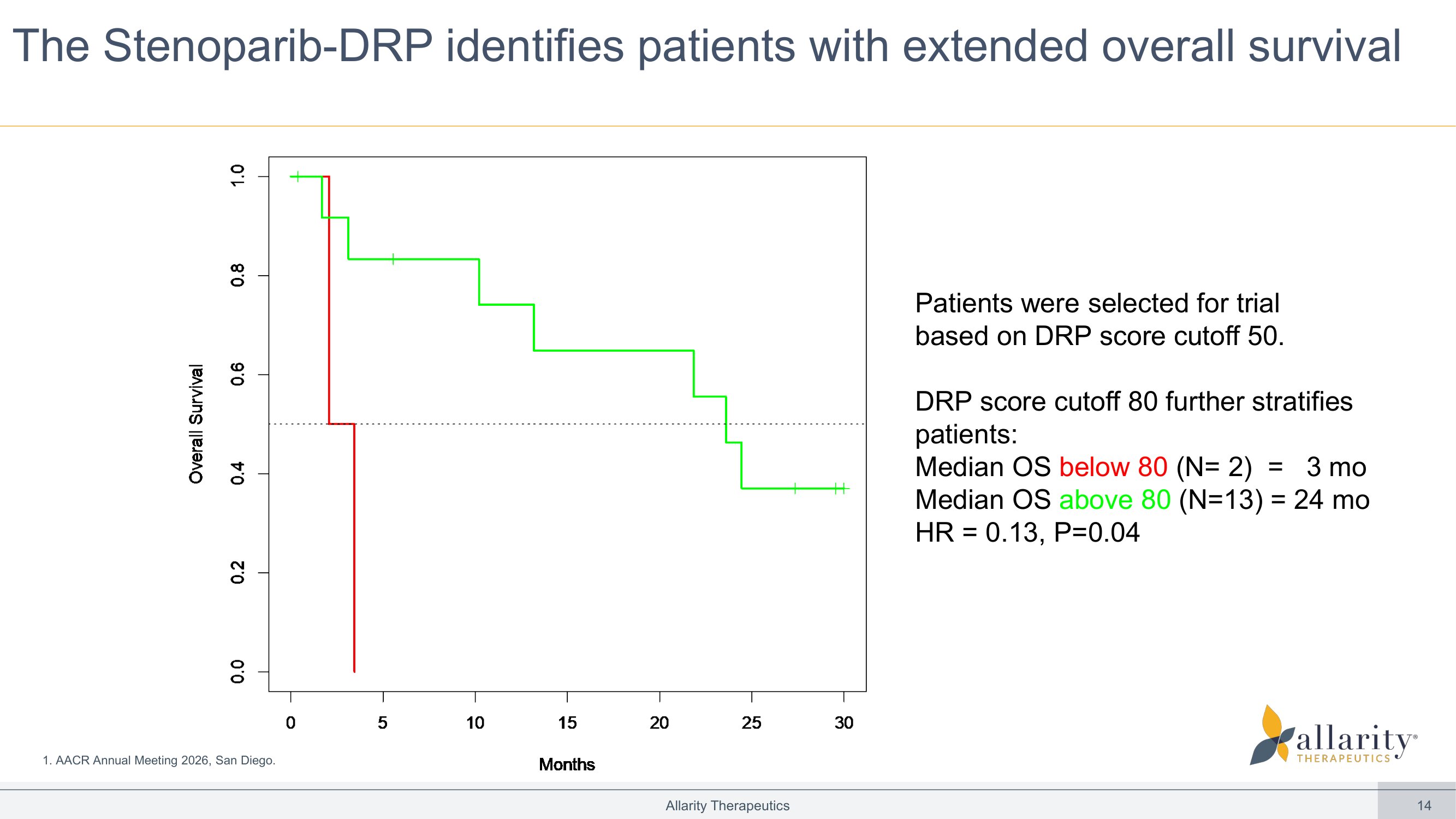
14 The Stenoparib - DRP identifies patients with extended overall survival Allarity Therapeutics Patients were selected for trial based on DRP score cutoff 50. DRP score cutoff 80 further stratifies patients: Median OS below 80 (N= 2) = 3 mo M edian OS above 80 (N=13) = 24 mo HR = 0.13, P=0.04 0 5 10 15 20 25 30 0 . 0 0 . 2 0 . 4 0 . 6 0 . 8 1 . 0 Months O v e r a l l S u r v i v a l 1. AACR Annual Meeting 2026, San Diego.

15 New Protocol for Phase 2 Trial in Ovarian Cancer: Stenoparib Monotherapy in Platinum Resistant or Ineligible Ovarian Cancer Allarity Therapeutics Corporate Overview Objective: Solidify Dose for Pivotal Trial, Establish Cut - point for DRP Pre - selection in Pivotal Trial Patients with Platinum resistant or Platinum ineligible disease » No more than 1L of chemo beyond declaration of Platinum Resistance except ADCs » No Platinum Refractory Pts or Pts with Ascites » BRCA mutant and BRCA wt patients accepted No DRP pre - selection - DRP will be run retrospectively in newer biopsy tissues to define cut - point as CDx Randomization to one of two BID dose cohorts to address FDA’s Project Optimus Edict » 200mg + 400mg » 400mg + 400mg Safety and Efficacy » Response Rate, Clinical benefit Rate, Progression Free Survival Up to 12 Trial Sites Enrolling Now

Backup slides
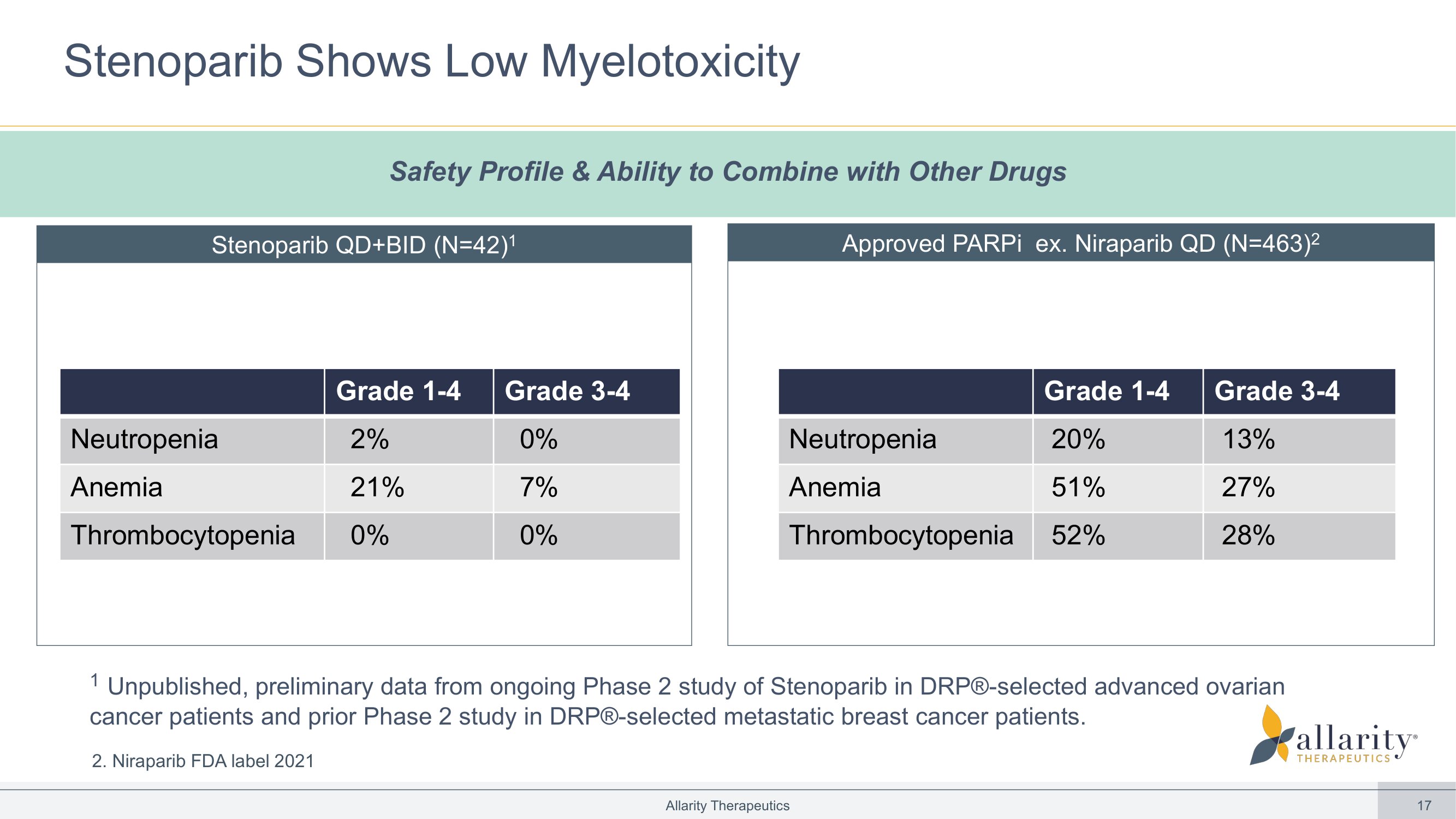
17 Stenoparib Shows Low Myelotoxicity Allarity Therapeutics Stenoparib QD+BID (N=42) 1 Approved PARPi ex. Niraparib QD (N=463) 2 Grade 3 - 4 Grade 1 - 4 0% 2% Neutropenia 7% 21% Anemia 0% 0% Thrombocytopenia Grade 3 - 4 Grade 1 - 4 13% 20% Neutropenia 27% 51% Anemia 28% 52% Thrombocytopenia Safety Profile & Ability to Combine with Other Drugs 1 Unpublished, preliminary data from ongoing Phase 2 study of Stenoparib in DRP® - selected advanced ovarian cancer patients and prior Phase 2 study in DRP® - selected metastatic breast cancer patients. 2. Niraparib FDA label 2021
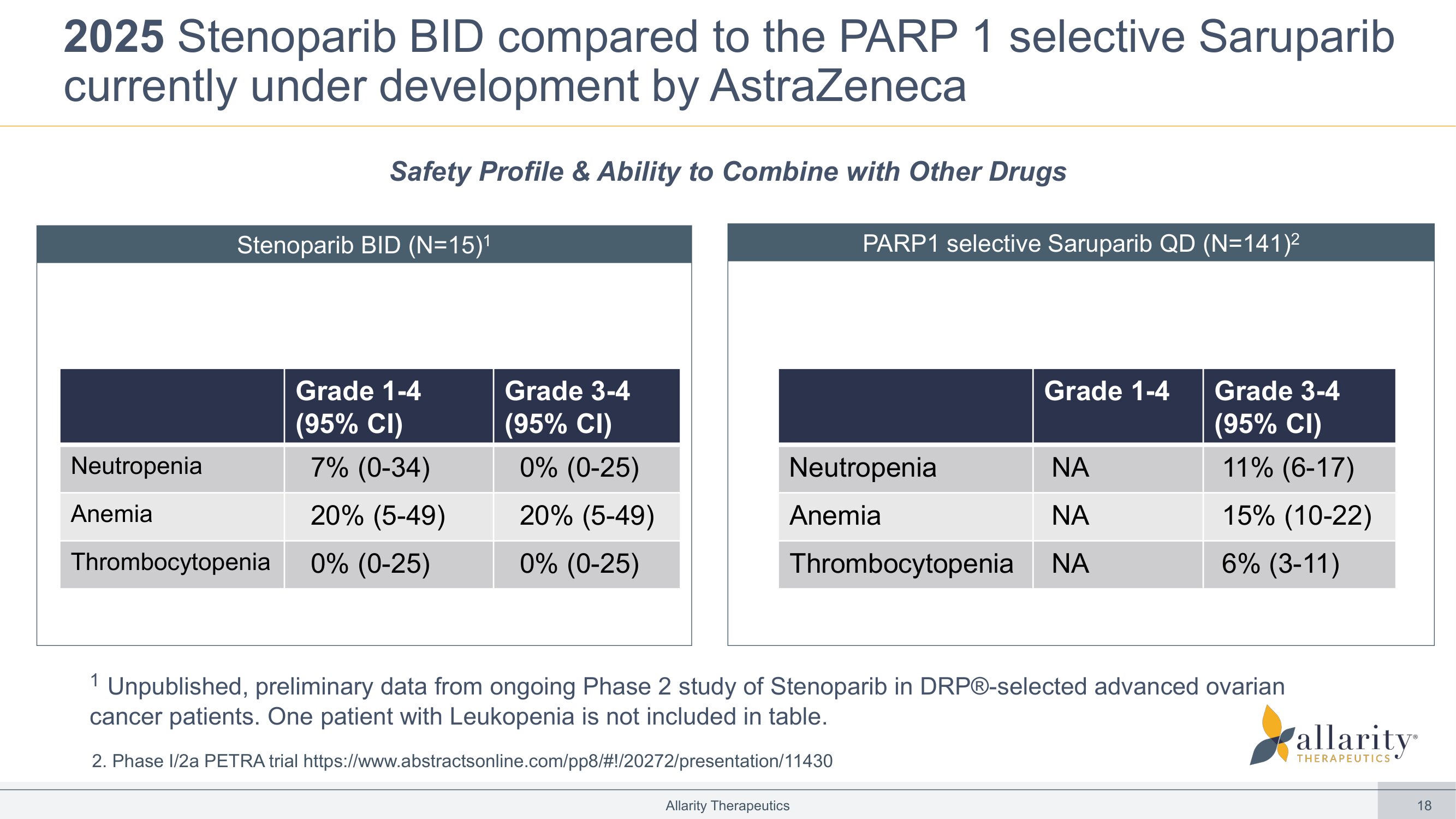
18 2025 Stenoparib BID compared to the PARP 1 selective Saruparib currently under development by AstraZeneca Allarity Therapeutics Stenoparib BID (N=15) 1 PARP1 selective Saruparib QD (N=141) 2 Grade 3 - 4 (95% CI) Grade 1 - 4 (95% CI) 0% (0 - 25) 7% (0 - 34) Neutropenia 20% (5 - 49) 20% (5 - 49) Anemia 0% (0 - 25) 0% (0 - 25) Thrombocytopenia Grade 3 - 4 (95% CI) Grade 1 - 4 11% (6 - 17) NA Neutropenia 15% (10 - 22) NA Anemia 6% (3 - 11) NA Thrombocytopenia Safety Profile & Ability to Combine with Other Drugs 1 Unpublished, preliminary data from ongoing Phase 2 study of Stenoparib in DRP® - selected advanced ovarian cancer patients. One patient with Leukopenia is not included in table. 2. Phase I/2a PETRA trial https :// www.abstractsonline.com /pp8/#!/20272/ presentation /11430
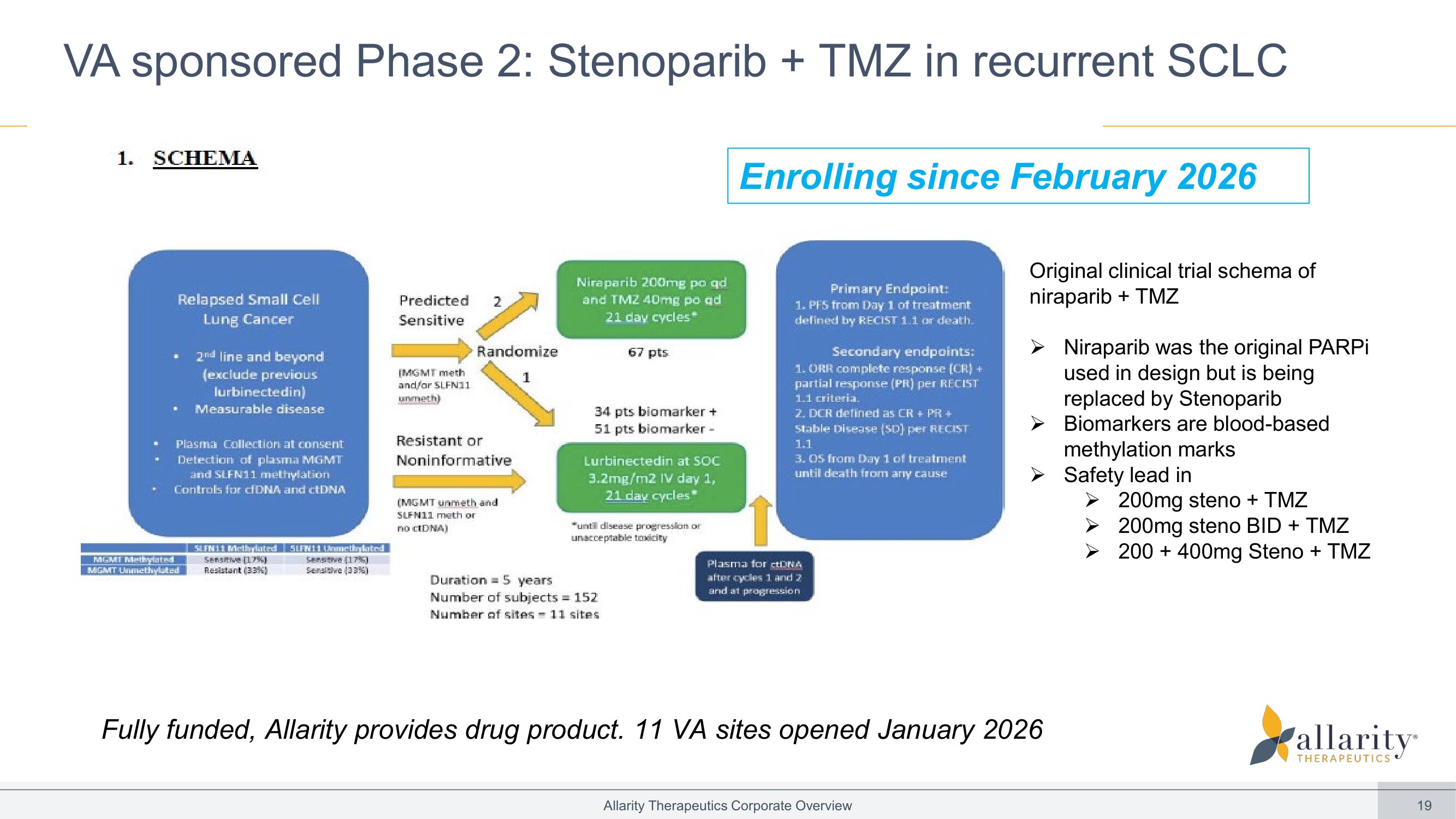
19 Allarity Therapeutics Corporate Overview VA sponsored Phase 2: Stenoparib + TMZ in recurrent SCLC Fully funded, Allarity provides drug product. 11 VA sites opened January 2026 Original clinical trial schema of niraparib + TMZ » Niraparib was the original PARPi used in design but is being replaced by Stenoparib » Biomarkers are blood - based methylation marks » Safety lead in » 200mg steno + TMZ » 200mg steno BID + TMZ » 200 + 400mg Steno + TMZ Enrolling since February 2026

20 Allarity Therapeutics • The DRP technology has been successful in identifying patients who may benefit from cancer drugs in a number of distinct indications • DRP has been prospectively used in a trial of LiPlaCis where it was able to enrich for responding patients • DRP has been retrospectively evaluated in a Phase III trial of dovitinib • DRP is being used to prospectively select patients in the phase II trial of stenoparib monotherapy in ovarian cancer • Stenoparib monotherapy (BID) shows durable clinical benefit in advanced ovarian cancer patients • Stenoparib is being advanced with the DRP toward potential registration in Ovarian Cancer Allarity’s DRP may identify patients most likely to benefit from anti - cancer therapies

Thomas Jensen CEO tjensen@allarity.com allarity.com NASDAQ: ALLR
