Exhibit 99.2

CORPORATE PRESENTATION AAV + RENAL STRATEGY May 2026

Important Notice and Disclaimer This presentation has been prepared by InflaRx N.V.
(“InflaRx” or “we”). This presentation is made for informational purposes only and does not constitute an offer to sell or a solicitation of an offer to buy securities in any jurisdiction in which such offer or solicitation would be unlawful
prior to the registration or qualification of such securities under the securities law of any such jurisdiction. Cautionary statement regarding forward looking statements This presentation contains forward-looking statements that involve
substantial risks and uncertainties. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “estimate,” “believe,”
“predict,” “potential” or “continue” or the negative of these terms or other similar expressions intended to identify statements about the future. These statements speak only as of the date of this presentation and involve known and unknown
risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking
statements. We based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These
forward-looking statements include, without limitation, statements about the following: the success of our future clinical trials for vilobelimab’s treatment of other debilitating or life-threatening inflammatory indications, including ARDS;
the potential strategic transactions or collaborations, including a potential partnership of izicopan, or vilobelimab for PG; the success of our future clinical trials for izicopan, and whether such clinical results will reflect results seen
in previously conducted preclinical studies and clinical trials; the timing, progress and results of preclinical studies and clinical trials of vilobelimab, izicopan and any other product candidates, including for the development of
vilobelimab in several indications and other virally induced ARDS, HS and CSU, and statements regarding the timing of initiation and completion of studies or trials and related preparatory work, the period during which the results of the
trials will become available, the costs of such trials and our research and development programs generally; our interactions with and the receptiveness and approval by regulators regarding the results of clinical trials and potential
regulatory approval or authorization pathways, including our BLA submission for GOHIBIC (vilobelimab); the timing and outcome of any discussions or submission of filings for regulatory approval of vilobelimab, izicopan or any other product
candidate, and the timing of and our ability to obtain and maintain full regulatory approval and/or market authorization of vilobelimab or izicopan for any indication; our ability to leverage our proprietary anti-C5a and anti-C5aR
technologies to discover and develop therapies to treat complement-mediated immunological and inflammatory diseases; our ability to protect, maintain and enforce our intellectual property protection for vilobelimab, izicopan and any other
product candidates, and the scope of such protection; whether the FDA or any comparable foreign regulatory authority will accept or agree with the number, design, size, conduct or implementation of our clinical trials, including any proposed
primary or secondary endpoints for such trials; the success of our future clinical trials for vilobelimab, izicopan and any other product candidates and whether such clinical results will reflect results seen in previously conducted
preclinical studies and clinical trials; our expectations regarding the size of the patient populations for, the market opportunity for, the medical need for and clinical utility of vilobelimab, izicopan or any other product candidates, if
approved or authorized for commercial use; our manufacturing capabilities and strategy, including the scalability and cost of our manufacturing methods and processes and the optimization of our manufacturing methods and processes, and our
ability to continue to rely on our existing third-party manufacturers and our ability to engage additional third-party manufacturers for our planned future clinical trials and for commercial supply of any development candidate in the United
States and Europe; our estimates of our expenses, ongoing losses, future revenue, capital requirements and our needs for or ability to obtain additional financing; our ability to defend against liability claims resulting from the testing of
our product candidates in the clinic or, if, approved or authorized, any commercial sales; if any of our product candidates obtain regulatory approval or authorization, our ability to comply with and satisfy ongoing drug regulatory
obligations and continued regulatory overview; our ability to comply with enacted and future legislation in seeking marketing approval or commercialization; our future growth and ability to compete, which depends on our retaining key
personnel and recruiting additional qualified personnel; our competitive position and the development of and projections relating to our competitors in the development of C5a and C5aR inhibitors and other therapeutic products being developed
in similar medical conditions in which vilobelimab, izicopan or any other of our product candidates is being developed or our industry; and other risk factors discussed under the “ITEM 3. Key information––Risk factors” section of our Annual
Report on Form 20-F. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward-looking
statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking
statements. You should refer to the “ITEM 3. Key information––Risk factors” section of our Annual Report and risks described in our subsequent SEC filings for a discussion of important factors that may cause our actual results to differ
materially from those expressed or implied by our forward-looking statements. Moreover, we operate in an evolving environment. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict
all risk factors and uncertainties. As a result of these factors, we cannot assure you that the forward-looking statements in this presentation or in our Annual Report will prove to be accurate. Except as required by applicable law, we do not
plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. You should, however, review the factors and risks and other
information we describe in the reports we will file from time to time with the SEC after the date of this presentation. CONTROLLING INFLAMMATION

Important Notice and Disclaimer Information and sources Certain information contained
in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources, as well as InflaRx’s own internal estimates and research. While we believe these third-party sources to be
reliable as of the date of this presentation, we have not independently verified, and make no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. All market data and
other information from third-party sources involve a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Further, while we believe our own internal research is reliable,
such research has not been verified by any independent source. About InflaRx InflaRx (Nasdaq: IFRX) is a biopharmaceutical company pioneering anti-inflammatory therapeutics by applying its proprietary anti-C5a and anti-C5aR technologies to
discover, develop and commercialize highly potent and specific inhibitors of the complement activation factor C5a and its receptor, C5aR. C5a is a powerful inflammatory mediator involved in the progression of a wide variety of inflammatory
diseases. InflaRx‘s lead program is izicopan (INF904), an orally administered small molecule inhibitor of C5a-induced signaling via the C5a receptor, which has shown promising PK/PD characteristics as well as therapeutic potential in Phase 1
and Phase 2a clinical studies. The company is developing izicopan for the treatment of several inflammatory diseases, including hidradenitis suppurativa (HS). The Company has also developed vilobelimab, a novel, intravenously delivered,
first-in-class, anti-C5a monoclonal antibody that selectively binds to free C5a and has demonstrated disease-modifying clinical activity and tolerability in multiple clinical studies. InflaRx was founded in 2007, and the group has offices
and subsidiaries in Jena and Munich, Germany, as well as Ann Arbor, MI, USA. For further information, please visit www.inflarx.de. InflaRx GmbH (Germany) and InflaRx Pharmaceuticals Inc. (USA) are wholly owned subsidiaries of InflaRx N.V.
(together, InflaRx). CONTROLLING INFLAMMATION

Izicopan: A next-generation C5aR inhibitor for AAV and broader renal Izicopan is an
oral inhibitor of C5a/C5aR, a critical driver of the inflammatory cascade C5aR has been validated from a clinical, regulatory and commercial perspective in AAV Further validation in AAV provided by promising clinical data with C5a
antibodies vilobelimab and BDB-001 AAV market growth and regulatory scrutiny related to the marketed comparator, avacopan, has provided InflaRx a clear and present opportunity with its next generation C5aR inhibitor, izicopan By improving
upon the significant limitations of the marketed comparator, izicopan has potential best-in-class properties Faster onset of action and higher target coverage leading to potentially differentiated efficacy Cleaner safety profile with low
risk of liver toxicity and drug-drug interactions More convenient dosing with the potential for lower pill burden and once-daily dosing Given its class-leading properties izicopan has the potential to improve upon the ~$1.3B peak sales
estimate* of avacopan Izicopan could also address significant unmet needs and large commercial markets across broader I&I, including renal indications such as aHUS, IgAN, C3 glomerulopathy, and others Strong IP position and a team with
proven track record of delivering clinical and regulatory successes I&I [immunology & inflammation]. AAV [ANCA-associated vasculitis]. aHUS [atypical hemolytic uremic syndrome]. IgAN [immunoglobulin-A nephropathy]. * FactSet
estimates, March 2026. 4

Indication PreClin Phase 1 Phase 2 Phase 3 MARKET STATUS &
Milestones ANCA-associated vasculitis other renal indications HS + CSU broader I&I Phase 2b planning ongoing Additional renal indications such as aHUS, IgAN, C3G, and others Topline Phase 2a reported Additional indications in
immuno-dermatology, hematology, neurology and others izicopan Oral C5aR inhibitor I&I InflaRx Pipeline: Targeting the C5a–C5aR1 axis to control inflammation vilobelimab C5a inhibitor critical COVID-19 SARS-CoV-2-induced
ARDS broader ARDS US EUA granted* Approved by European Commission* Phase 2 “Just Breathe” ASPR/BARDA clinical platform study Critical Care DERM vilobelimab C5a inhibitor pyoderma gangrenosum Phase 3 reported** OTHER IFX002 C5a
inhibitor vilobelimab life-cycle approach For optimized use in chronic inflammatory indications *Commercial partnering and distribution options in the US and EU being considered. ** Phase 3 stopped early for futility at interim
analysis, FDA interaction considered. aHUS [atypical hemolytic uremic syndrome]. IgAN [immunoglobulin-A nephropathy]. C3G [anti-C3 glomerulopathy]. 5
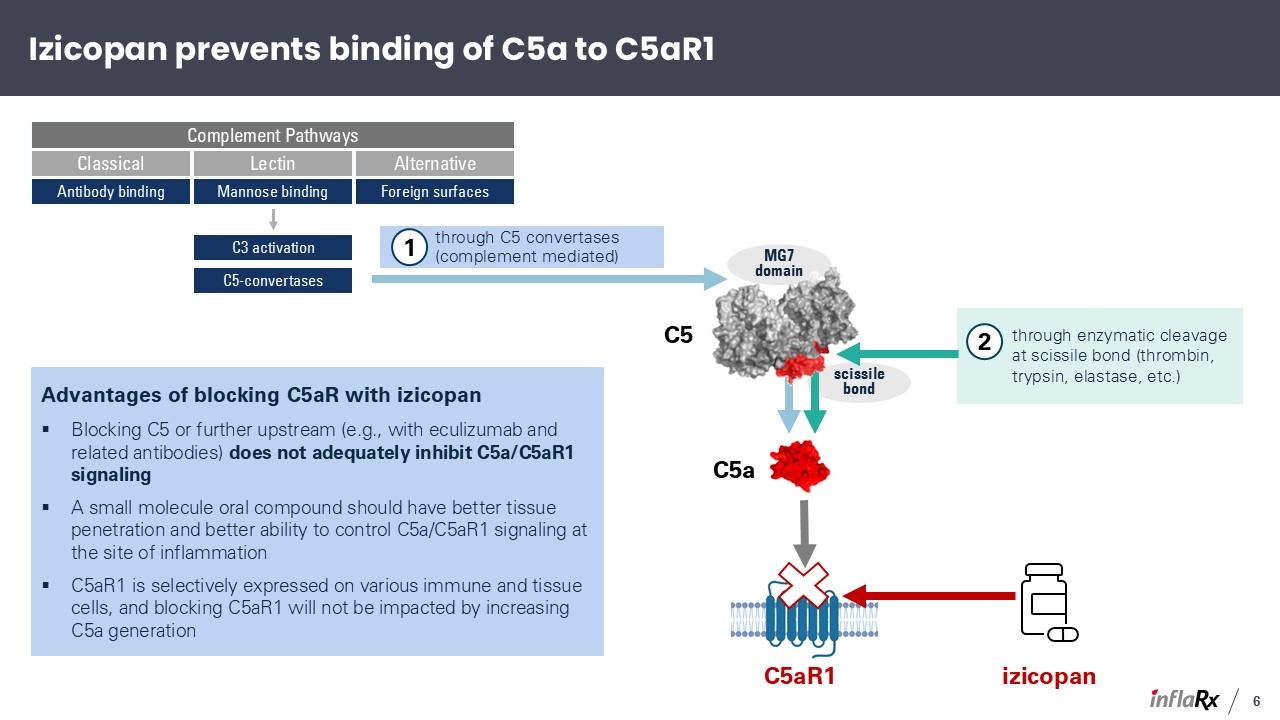
Izicopan prevents binding of C5a to C5aR1 Advantages of blocking C5aR with
izicopan Blocking C5 or further upstream (e.g., with eculizumab and related antibodies) does not adequately inhibit C5a/C5aR1 signaling A small molecule oral compound should have better tissue penetration and better ability to control
C5a/C5aR1 signaling at the site of inflammation C5aR1 is selectively expressed on various immune and tissue cells, and blocking C5aR1 will not be impacted by increasing C5a generation scissile bond MG7 domain C5a C5aR1 through C5
convertases (complement mediated) through enzymatic cleavage at scissile bond (thrombin, trypsin, elastase, etc.) Classical Lectin Alternative Complement Pathways Antibody binding Mannose binding Foreign surfaces C3
activation C5-convertases 1 2 C5 izicopan 6
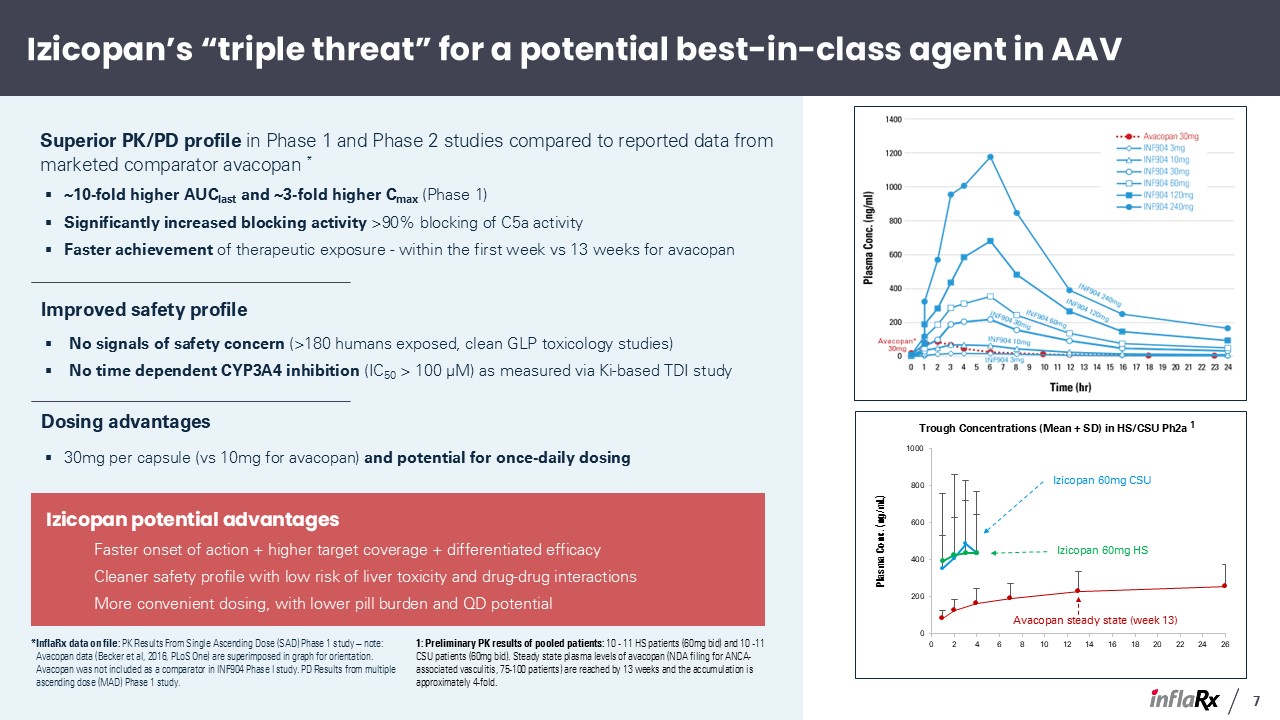
*InflaRx data on file: PK Results From Single Ascending Dose (SAD) Phase 1 study – note:
Avacopan data (Becker et al, 2016, PLoS One) are superimposed in graph for orientation. Avacopan was not included as a comparator in INF904 Phase I study. PD Results from multiple ascending dose (MAD) Phase 1 study. Izicopan’s “triple
threat” for a potential best-in-class agent in AAV Superior PK/PD profile in Phase 1 and Phase 2 studies compared to reported data from marketed comparator avacopan * ~10-fold higher AUClast and ~3-fold higher Cmax (Phase 1) Significantly
increased blocking activity >90% blocking of C5a activity Faster achievement of therapeutic exposure - within the first week vs 13 weeks for avacopan Dosing advantages 30mg per capsule (vs 10mg for avacopan) and potential for
once-daily dosing Improved safety profile No signals of safety concern (>180 humans exposed, clean GLP toxicology studies) No time dependent CYP3A4 inhibition (IC50 > 100 μM) as measured via Ki-based TDI study Izicopan potential
advantages Faster onset of action + higher target coverage + differentiated efficacy Cleaner safety profile with low risk of liver toxicity and drug-drug interactions More convenient dosing, with lower pill burden and QD
potential Avacopan steady state (week 13) Izicopan 60mg CSU Izicopan 60mg HS 1: Preliminary PK results of pooled patients: 10 - 11 HS patients (60mg bid) and 10 -11 CSU patients (60mg bid). Steady state plasma levels of avacopan (NDA
filing for ANCA-associated vasculitis, 75-100 patients) are reached by 13 weeks and the accumulation is approximately 4-fold. 7
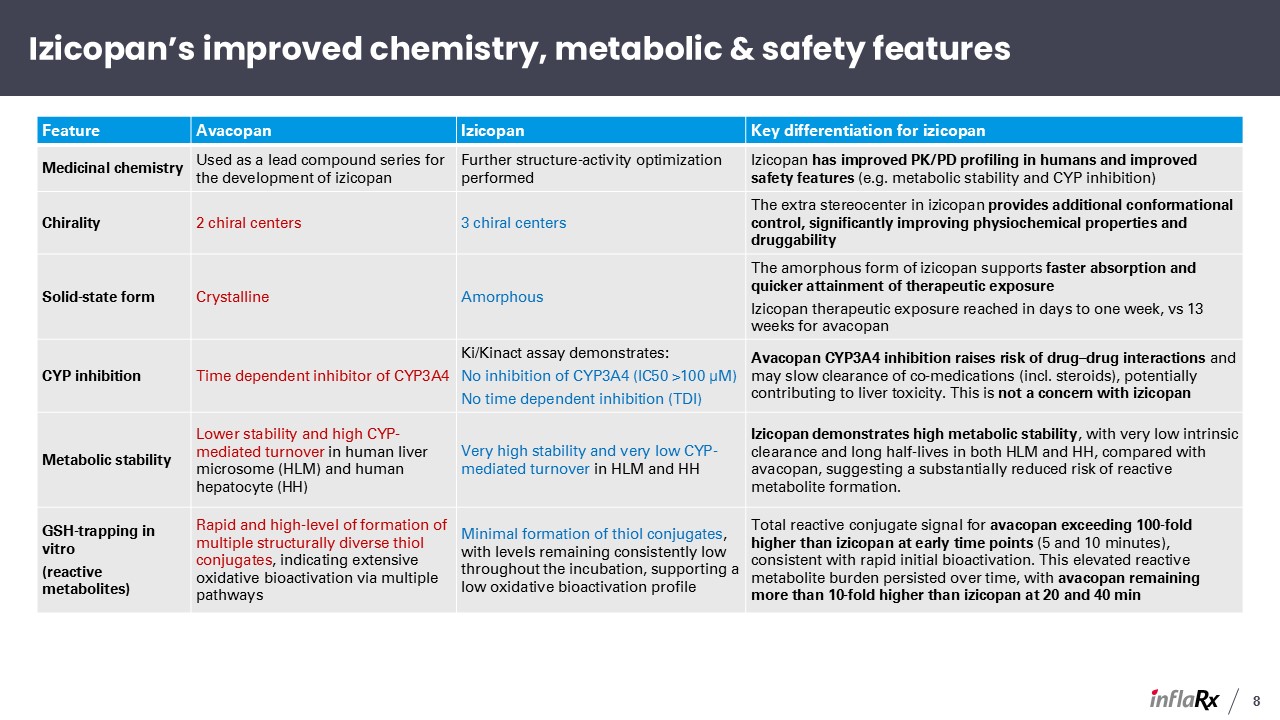
Feature Avacopan Izicopan Key differentiation for izicopan Medicinal chemistry Used
as a lead compound series for the development of izicopan Further structure-activity optimization performed Izicopan has improved PK/PD profiling in humans and improved safety features (e.g. metabolic stability and CYP
inhibition) Chirality 2 chiral centers 3 chiral centers The extra stereocenter in izicopan provides additional conformational control, significantly improving physiochemical properties and druggability Solid-state
form Crystalline Amorphous The amorphous form of izicopan supports faster absorption and quicker attainment of therapeutic exposure Izicopan therapeutic exposure reached in days to one week, vs 13 weeks for avacopan CYP inhibition Time
dependent inhibitor of CYP3A4 Ki/Kinact assay demonstrates: No inhibition of CYP3A4 (IC50 >100 μM) No time dependent inhibition (TDI) Avacopan CYP3A4 inhibition raises risk of drug–drug interactions and may slow clearance of
co-medications (incl. steroids), potentially contributing to liver toxicity. This is not a concern with izicopan Metabolic stability Lower stability and high CYP-mediated turnover in human liver microsome (HLM) and human hepatocyte
(HH) Very high stability and very low CYP-mediated turnover in HLM and HH Izicopan demonstrates high metabolic stability, with very low intrinsic clearance and long half-lives in both HLM and HH, compared with avacopan, suggesting a
substantially reduced risk of reactive metabolite formation. GSH-trapping in vitro (reactive metabolites) Rapid and high-level of formation of multiple structurally diverse thiol conjugates, indicating extensive oxidative bioactivation via
multiple pathways Minimal formation of thiol conjugates, with levels remaining consistently low throughout the incubation, supporting a low oxidative bioactivation profile Total reactive conjugate signal for avacopan exceeding 100-fold
higher than izicopan at early time points (5 and 10 minutes), consistent with rapid initial bioactivation. This elevated reactive metabolite burden persisted over time, with avacopan remaining more than 10-fold higher than izicopan at 20 and
40 min Izicopan’s improved chemistry, metabolic & safety features 8
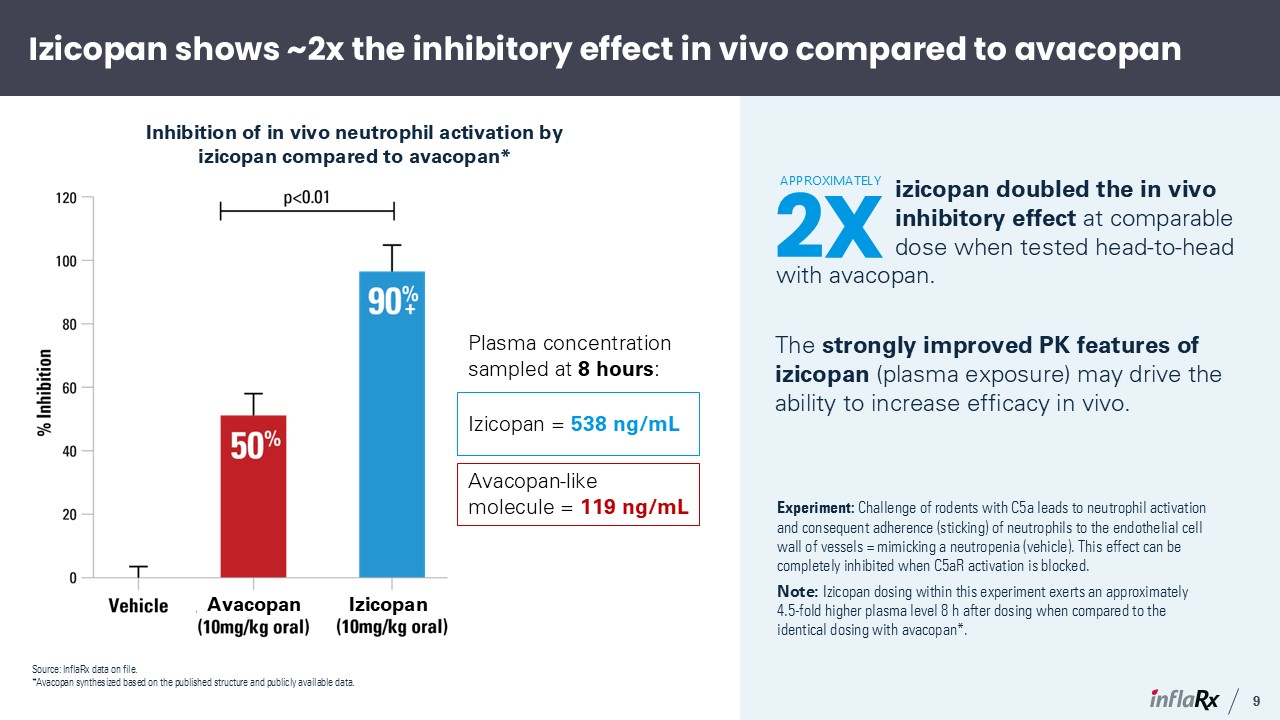
Izicopan shows ~2x the inhibitory effect in vivo compared to avacopan Inhibition of in
vivo neutrophil activation by izicopan compared to avacopan* Experiment: Challenge of rodents with C5a leads to neutrophil activation and consequent adherence (sticking) of neutrophils to the endothelial cell wall of vessels = mimicking a
neutropenia (vehicle). This effect can be completely inhibited when C5aR activation is blocked. Note: Izicopan dosing within this experiment exerts an approximately 4.5-fold higher plasma level 8 h after dosing when compared to the identical
dosing with avacopan*. Plasma concentration sampled at 8 hours: izicopan doubled the in vivo inhibitory effect at comparable dose when tested head-to-head with avacopan. The strongly improved PK features of izicopan (plasma exposure) may
drive the ability to increase efficacy in vivo. Izicopan = 538 ng/mL Avacopan-like molecule = 119 ng/mL 2X APPROXIMATELY + 9 Izicopan Source: InflaRx data on file. *Avacopan synthesized based on the published structure and publicly
available data. Avacopan

ANCA-associated vasculitis (AAV) CONTROLLING INFLAMMATION
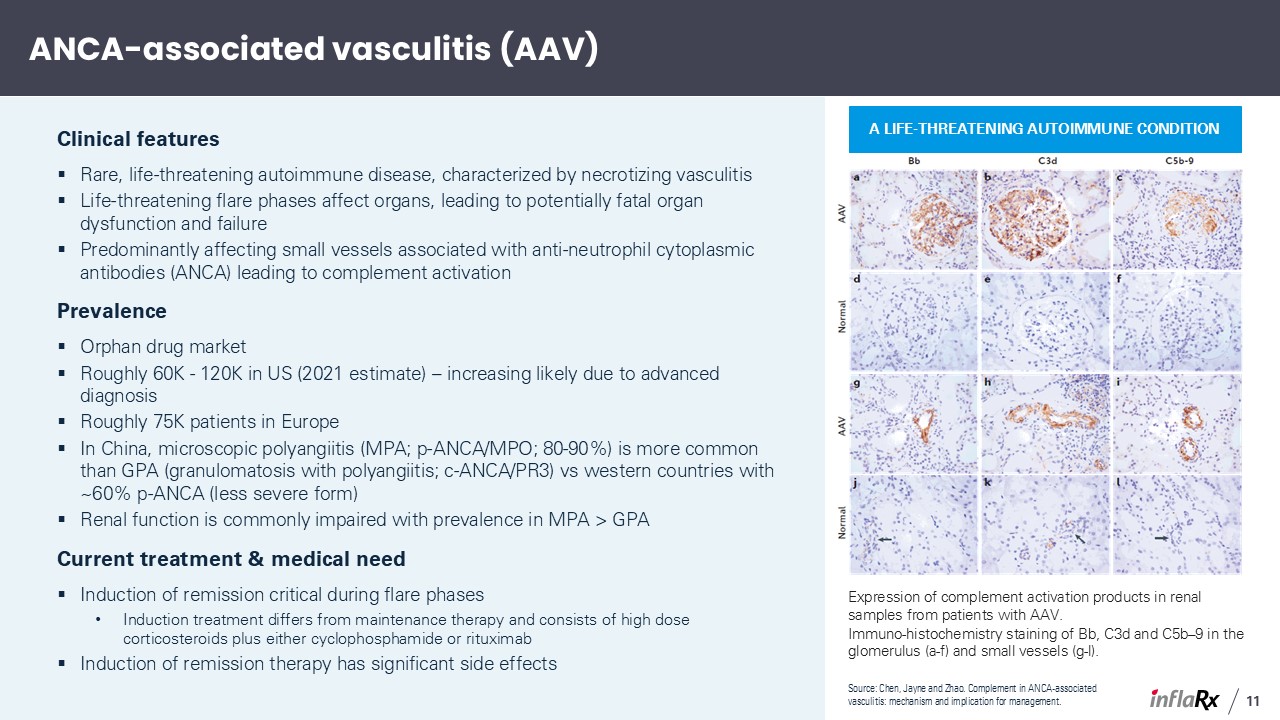
ANCA-associated vasculitis (AAV) Clinical features Rare, life-threatening autoimmune
disease, characterized by necrotizing vasculitis Life-threatening flare phases affect organs, leading to potentially fatal organ dysfunction and failure Predominantly affecting small vessels associated with anti-neutrophil cytoplasmic
antibodies (ANCA) leading to complement activation Prevalence Orphan drug market Roughly 60K - 120K in US (2021 estimate) – increasing likely due to advanced diagnosis Roughly 75K patients in Europe In China, microscopic polyangiitis
(MPA; p-ANCA/MPO; 80-90%) is more common than GPA (granulomatosis with polyangiitis; c-ANCA/PR3) vs western countries with ~60% p-ANCA (less severe form) Renal function is commonly impaired with prevalence in MPA > GPA Current treatment
& medical need Induction of remission critical during flare phases Induction treatment differs from maintenance therapy and consists of high dose corticosteroids plus either cyclophosphamide or rituximab Induction of remission therapy
has significant side effects Source: Chen, Jayne and Zhao. Complement in ANCA-associated vasculitis: mechanism and implication for management. A life-threatening autoimmune condition Expression of complement activation products in renal
samples from patients with AAV. Immuno-histochemistry staining of Bb, C3d and C5b–9 in the glomerulus (a-f) and small vessels (g-l). 11
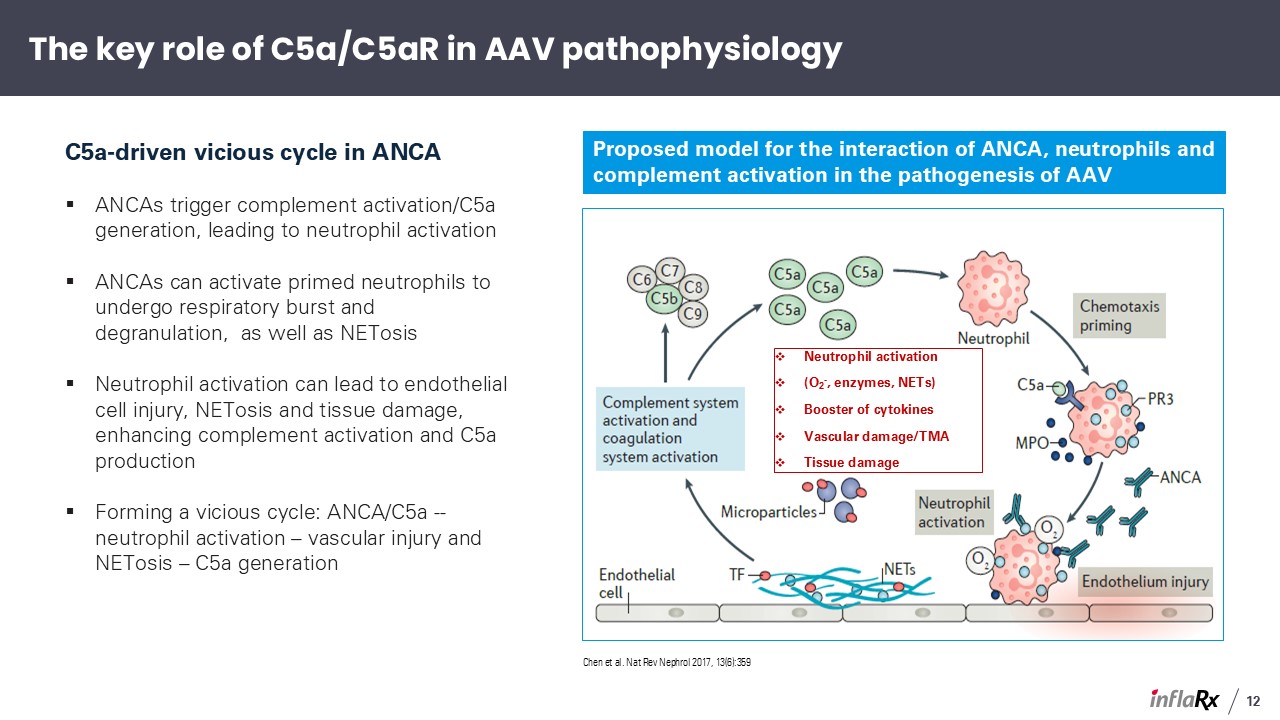
C5a-driven vicious cycle in ANCA ANCAs trigger complement activation/C5a generation,
leading to neutrophil activation ANCAs can activate primed neutrophils to undergo respiratory burst and degranulation, as well as NETosis Neutrophil activation can lead to endothelial cell injury, NETosis and tissue damage, enhancing
complement activation and C5a production Forming a vicious cycle: ANCA/C5a -- neutrophil activation – vascular injury and NETosis – C5a generation Proposed model for the interaction of ANCA, neutrophils and complement activation in the
pathogenesis of AAV Chen et al. Nat Rev Nephrol 2017, 13(6):359 Neutrophil activation (O2-, enzymes, NETs) Booster of cytokines Vascular damage/TMA Tissue damage The key role of C5a/C5aR in AAV pathophysiology 12
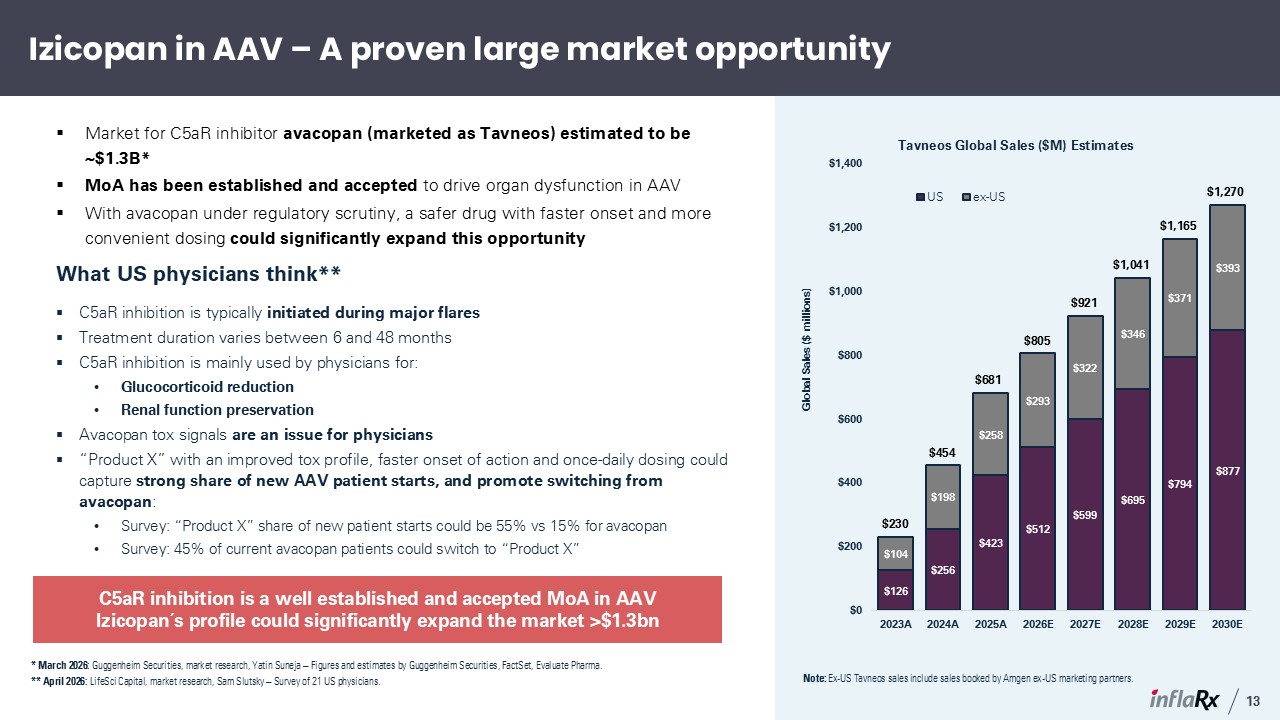
Izicopan in AAV – A proven large market opportunity * March 2026: Guggenheim
Securities, market research, Yatin Suneja – Figures and estimates by Guggenheim Securities, FactSet, Evaluate Pharma. ** April 2026: LifeSci Capital, market research, Sam Slutsky – Survey of 21 US physicians. What US physicians
think** C5aR inhibition is typically initiated during major flares Treatment duration varies between 6 and 48 months C5aR inhibition is mainly used by physicians for: Glucocorticoid reduction Renal function preservation Avacopan tox
signals are an issue for physicians “Product X” with an improved tox profile, faster onset of action and once-daily dosing could capture strong share of new AAV patient starts, and promote switching from avacopan: Survey: “Product X” share
of new patient starts could be 55% vs 15% for avacopan Survey: 45% of current avacopan patients could switch to “Product X” C5aR inhibition is a well established and accepted MoA in AAV Izicopan´s profile could significantly expand the
market >$1.3bn Market for C5aR inhibitor avacopan (marketed as Tavneos) estimated to be ~$1.3B* MoA has been established and accepted to drive organ dysfunction in AAV With avacopan under regulatory scrutiny, a safer drug with faster
onset and more convenient dosing could significantly expand this opportunity $230 $454 $681 $805 $921 $1,041 $1,165 $1,270 Note: Ex-US Tavneos sales include sales booked by Amgen ex-US marketing partners. 13

Izicopan in AAV – Capturing the opportunity Steroid-reducing / rapid taper
label Avacopan label does not state steroid-sparing Demonstrate early benefit (potentially label relevant) Early reduction of proteinuria (w4) Improve eGFR (w26 / w52) Improve time to remission (BVAS-0) Clean label (no liver tox) Lower
capsule intake or once-daily dosing “Steroids have much more toxicity than just infection. Steroids are incredible medicines - they help control these conditions, but used long-term and high dose cumulative use of these medications are
extraordinarily toxic to the body, so you want to lower their use to zero, if possible.” – interview # 2 Goal: ‘Rock the Boat’ Scenario Development improvements to focus on izicopan advantages Key Development Improvements Overall
Goal Increase signal delta to placebo especially during early major flare phase while demonstrating a rapid steroid tapering / reduction Study Design Aspects Enrolling fast = get patients on izicopan early in the flare No mandatory GC
tapering before initiation of dosing Enrich for / allow patients with severe renal impairment Fix SOC remission induction treatment to rituximab Aggressive rapid steroid taper in treatment arm(s) tested versus standard steroid tapering in
placebo arm GC [glucocorticoids]. SOC [standard of care]. 14 * IFRX proprietary market research.

GC indiv. GC indiv. Izicopan AAV Phase 2 trial – Base case / current planning stage
BVAS [Birmingham Vasculitis Assessment Score]. CYC [cyclophosphamide]. GC [glucocorticoids]. IZI [izicopan]. RTX [rituximab]. SOC [standard of care maintenance]. NGAL [neutrophil gelatinase-associated lipocalin]. MCP-1 [monocyte
chemoattractant protein-1]. TEAEs [treatment emergent adverse events]. SAEs [serious adverse events]. TRAEs [treatment-related adverse events]. Arm 3:* IZI dose 1 BID + Rapid GC Taper + RTX Arm 1: Placebo + Standard GC Taper + RTX w
26 IZI dose 1 BID / QD + RTX Placebo + RTX Remission Maintenance Remission Induction Screening GC indiv 2 4 6 8 10 12 14 16 18 20 0 2 4 6 8 10 12 14 16 18 20 0 Phase 2 over 12 mos with unblinded analysis for
primary endpoint at wk 26 (6 mos), GC tapering according to SOC (PEXIVAS), izicopan arm rapid GC tapering within 4-6 wks * Possibility to add an additional dosing arm with same GC tapering regimen switching to QD earlier – will depend on
regulatory discussions w 52 Arm 2: IZI dose 1 BID + Standard GC Taper + RTX Izi dose 1 BID + RTX 2 4 6 8 10 12 14 16 18 20 0 Endpoint analyses Primary: Safety TEAEs, SAEs, TRAEs, infections + descriptive BVAS-0
non-inferiority week 26 Secondary efficacy focus Early Proteinuria reduction: UACR delta ≥30% to placebo at Wk 4 (≥ 20% at w 2) eGFR week 26 / 52 Time to BVAS-0 (clinical remission) + # of patients with BVAS-0 per early time
points Secondary other Pharmacokinetics (PK) Time in clinical remission 12 mos GC infections / toxicity Urine NGAL / MCP-1, other biomarkers n = approximately 30-50 per arm 15
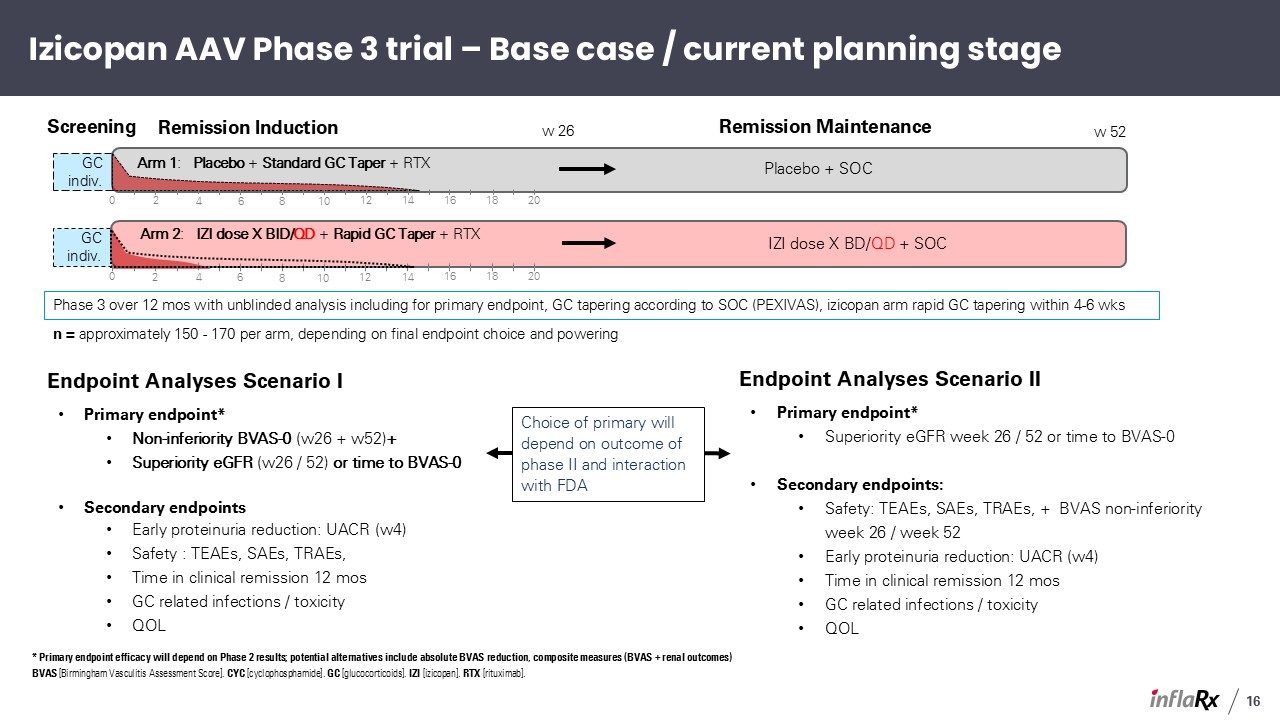
GC indiv. GC indiv. Izicopan AAV Phase 3 trial – Base case / current planning
stage Arm 2: IZI dose X BID/QD + Rapid GC Taper + RTX Arm 1: Placebo + Standard GC Taper + RTX w 26 IZI dose X BD/QD + SOC Placebo + SOC Remission Maintenance Remission
Induction Screening 2 4 6 8 10 12 14 16 18 20 0 2 4 6 8 10 12 14 16 18 20 0 Phase 3 over 12 mos with unblinded analysis including for primary endpoint, GC tapering according to SOC (PEXIVAS), izicopan arm rapid GC
tapering within 4-6 wks w 52 Endpoint Analyses Scenario I Primary endpoint* Non-inferiority BVAS-0 (w26 + w52)+ Superiority eGFR (w26 / 52) or time to BVAS-0 Secondary endpoints Early proteinuria reduction: UACR (w4) Safety : TEAEs,
SAEs, TRAEs, Time in clinical remission 12 mos GC related infections / toxicity QOL Endpoint Analyses Scenario II Primary endpoint* Superiority eGFR week 26 / 52 or time to BVAS-0 Secondary endpoints: Safety: TEAEs, SAEs, TRAEs, +
BVAS non-inferiority week 26 / week 52 Early proteinuria reduction: UACR (w4) Time in clinical remission 12 mos GC related infections / toxicity QOL Choice of primary will depend on outcome of phase II and interaction with FDA n =
approximately 150 - 170 per arm, depending on final endpoint choice and powering * Primary endpoint efficacy will depend on Phase 2 results; potential alternatives include absolute BVAS reduction, composite measures (BVAS + renal
outcomes) BVAS [Birmingham Vasculitis Assessment Score]. CYC [cyclophosphamide]. GC [glucocorticoids]. IZI [izicopan]. RTX [rituximab]. 16

Izicopan in AAV – Exploring potential for expedited development Concurrent with AAV
planning and in accordance with the fluid regulatory environment for avacopan, InflaRx is evaluating the feasibility of an expedited clinical and regulatory path for izicopan in AAV Options for expedited clinical and regulatory path could
include Use of a “seamless” trial strategy for a ”one-shot” Phase 2 - 3 registrational trial approach Seamless trial strategy could explore 2 doses in its first phase, including once-daily dosing, transitioning to a second phase focusing on
one dosing regimen in the Phase 3 part Potential to reduce time to the commercial market by 6 to 12 months 17

Additional renal indications Development opportunities CONTROLLING INFLAMMATION
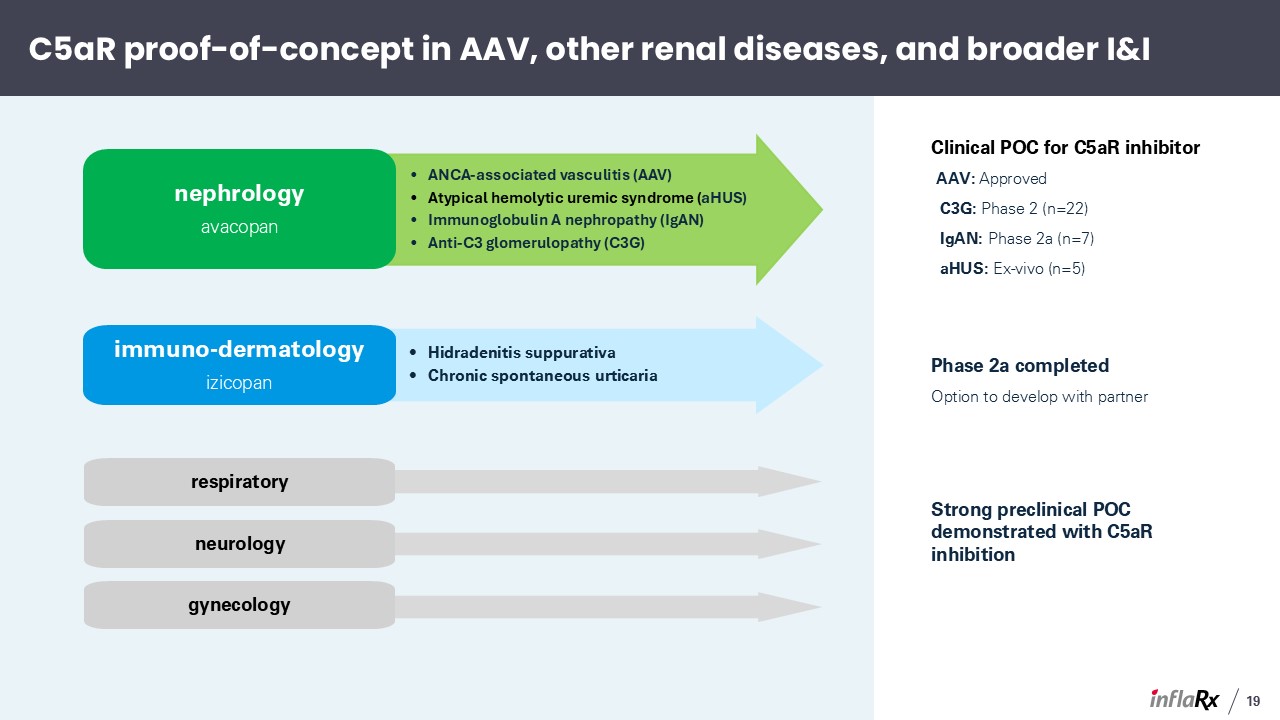
C5aR proof-of-concept in AAV, other renal diseases, and broader I&I ANCA-associated
vasculitis (AAV) Atypical hemolytic uremic syndrome (aHUS) Immunoglobulin A nephropathy (IgAN) Anti-C3 glomerulopathy (C3G) nephrology avacopan Clinical POC for C5aR inhibitor AAV: Approved C3G: Phase 2 (n=22) IgAN: Phase 2a
(n=7) aHUS: Ex-vivo (n=5) Hidradenitis suppurativa Chronic spontaneous urticaria immuno-dermatology izicopan Phase 2a completed Option to develop with partner Strong preclinical POC demonstrated with C5aR
inhibition respiratory gynecology neurology 19

Potential izicopan renal disease PoC basket studies aHUS Compelling biology with
strong evidence for C5a/neutrophil–driven endothelial injury Fast POC with clear, regulator-accepted TMA response (biomarker) endpoint with rapid efficacy readout Izicopan: differentiated safety with no vaccination, low infection risk, oral
therapy Monotherapy potential or as add-on / rescue to anti-C5 IgAN Accelerated approval via biomarker surrogate (proteinuria), with eGFR stabilization as confirmatory endpoint Autoimmune disease with immune complex–driven pathology,
where C5a/C5aR1 is a clearly defined key disease mechanism. Aligned with growing adoption of complement therapies in this kidney disease Izicopan: no vaccination required, and low infection risk compared with other complement inhibitor such
as iptacopan. C3G Accelerated approval pathway based on biomarker surrogate (proteinuria), with eGFR stabilization as supportive endpoint. Strong proof-of-concept data from avacopan support C5a/C5aR1 as a validated therapeutic target
pathway. Aligned with growing adoption of complement therapies in this kidney disease Izicopan could have oral convenience, no vaccination requirement and low infection risk compared with pegcetacoplan. A strategy to conduct studies in
regions where InflaRx can rapidly generate data, including China, for a strategic advantage in indications such as IgAN, aHUS, and C3G Large patient populations, centralized nephrology networks, and strong expertise in complement-mediated
kidney disease trials Strong potential for rapid recruitment and efficient proof-of-concept generation aHUS [atypical hemolytic uremic syndrome]. IgAN [immunoglobulin-A nephropathy]. C3G [anti-C3 glomerulopathy]. 20

Renal indications aHUS (atypical hemolytic uremic syndrome) CONTROLLING INFLAMMATION
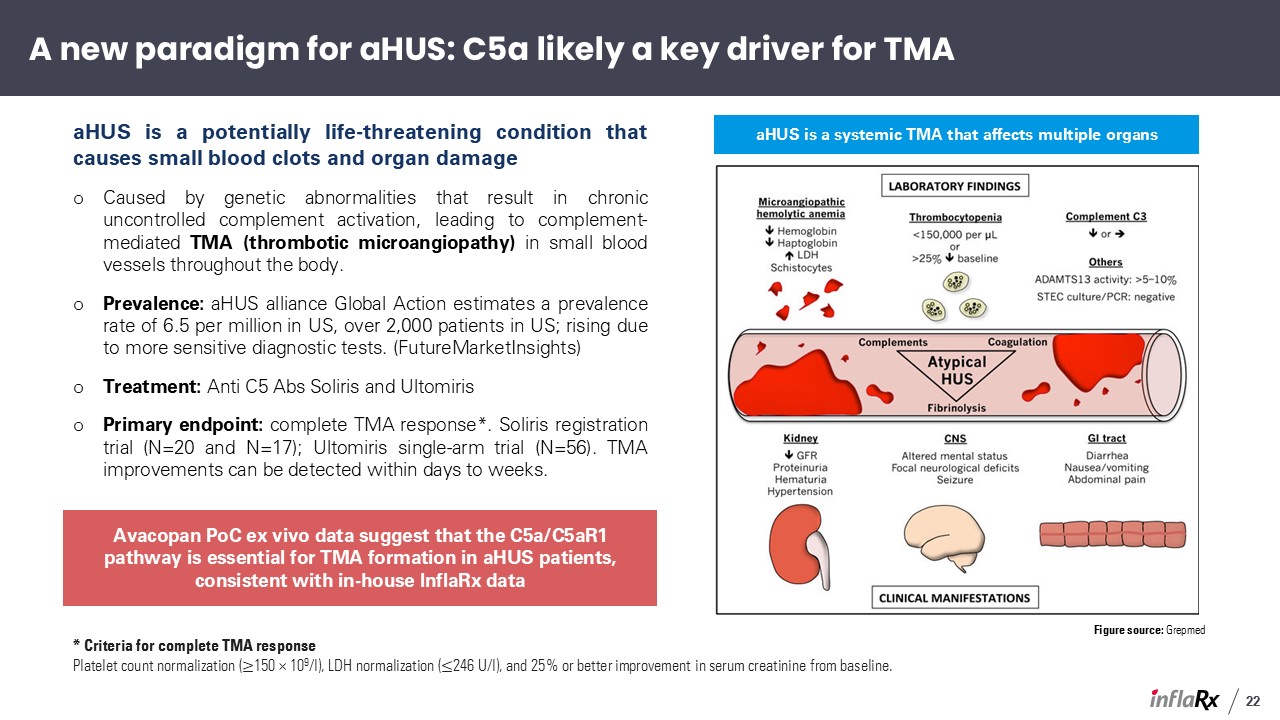
aHUS is a potentially life-threatening condition that causes small blood clots and organ
damage Caused by genetic abnormalities that result in chronic uncontrolled complement activation, leading to complement-mediated TMA (thrombotic microangiopathy) in small blood vessels throughout the body. Prevalence: aHUS alliance Global
Action estimates a prevalence rate of 6.5 per million in US, over 2,000 patients in US; rising due to more sensitive diagnostic tests. (FutureMarketInsights) Treatment: Anti C5 Abs Soliris and Ultomiris Primary endpoint: complete TMA
response*. Soliris registration trial (N=20 and N=17); Ultomiris single-arm trial (N=56). TMA improvements can be detected within days to weeks. Figure source: Grepmed aHUS is a systemic TMA that affects multiple organs * Criteria for
complete TMA response Platelet count normalization (≥150 × 109/l), LDH normalization (≤246 U/l), and 25% or better improvement in serum creatinine from baseline. A new paradigm for aHUS: C5a likely a key driver for TMA Avacopan PoC ex
vivo data suggest that the C5a/C5aR1 pathway is essential for TMA formation in aHUS patients, consistent with in-house InflaRx data 22
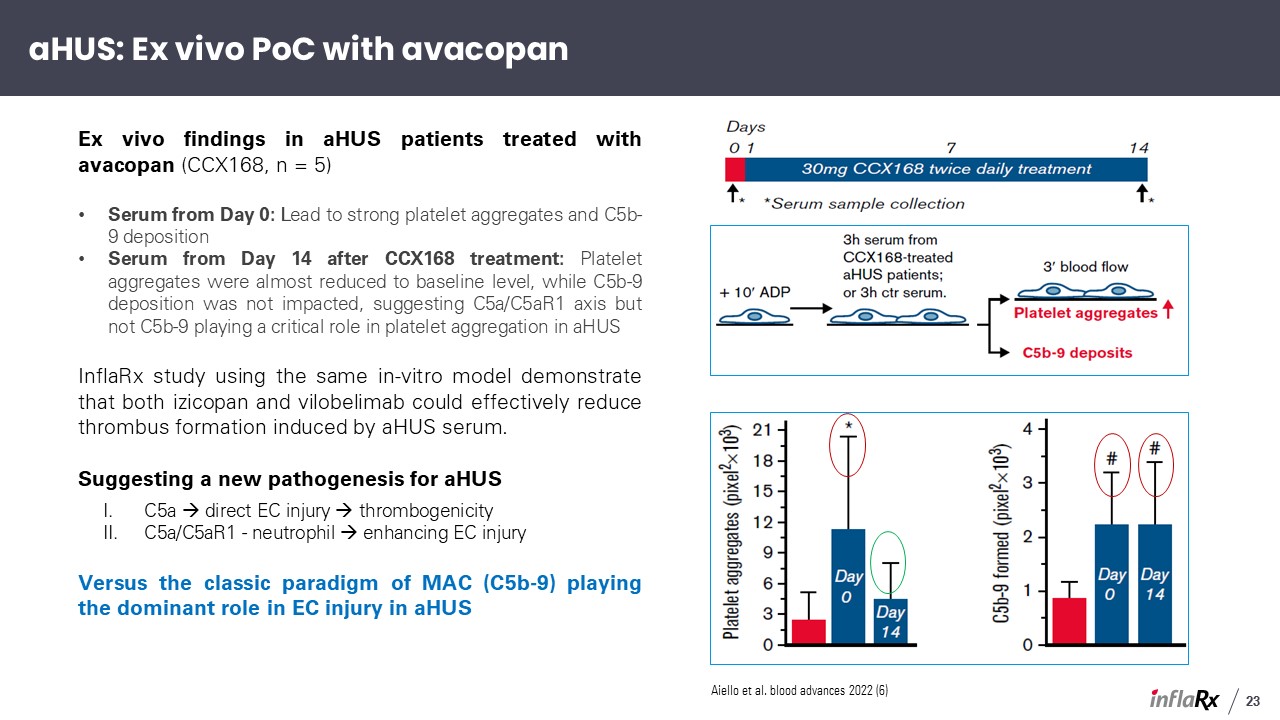
Ex vivo findings in aHUS patients treated with avacopan (CCX168, n = 5) Serum from Day
0: Lead to strong platelet aggregates and C5b-9 deposition Serum from Day 14 after CCX168 treatment: Platelet aggregates were almost reduced to baseline level, while C5b-9 deposition was not impacted, suggesting C5a/C5aR1 axis but not C5b-9
playing a critical role in platelet aggregation in aHUS InflaRx study using the same in-vitro model demonstrate that both izicopan and vilobelimab could effectively reduce thrombus formation induced by aHUS serum. Suggesting a new
pathogenesis for aHUS C5a direct EC injury thrombogenicity C5a/C5aR1 - neutrophil enhancing EC injury Versus the classic paradigm of MAC (C5b-9) playing the dominant role in EC injury in aHUS Aiello et al. blood advances 2022
(6) aHUS: Ex vivo PoC with avacopan 23

Renal indications IgA nephropathy (IgAN) CONTROLLING INFLAMMATION
IgAN is an autoimmune condition in which IgA deposits in the glomeruli of the kidneys,
impacting filtration Prevalence5: US: ~85k; EU5: ~180k; China: ~800K; Japan: ~85k IgAN is the most common cause of primary glomerulonephritis (GN)3 It may also occur in association with other conditions, including lupus nephritis and
thrombotic microangiopathy. Disease course and symptoms Typically, slow-progressing; may ultimately lead to acute kidney injury and chronic kidney disease, including end-stage renal disease (ESRD) Approximately 25% of patients develop ESRD
over the disease course1,2 Complement activation is a key driver of kidney inflammation and injury in IgAN, highlighting an unmet need for a safe and effective complement inhibitor to control complement-driven damage4 Iptacopan, an oral
factor B inhibitor, has achieved regulatory approval Ultomiris (ravulizumab), an anti-C5 antibody, has recently demonstrated statistically significant reductions in proteinuria, meeting the primary endpoint in Phase 3 clinical
studies Source: NIDDK Sources: 1: Mayo Clinic. 2: NIDDK. 3: UpToDate. 4: Duval et al. Nephrol dial transplant, 2023 (38). 5: CDA, Clarivate, Opti-Brand PMR, Adelphi, ZS conjoint 2024, Spherix, data for year 2025. IgAN: An autoimmune
condition with complement activation 25
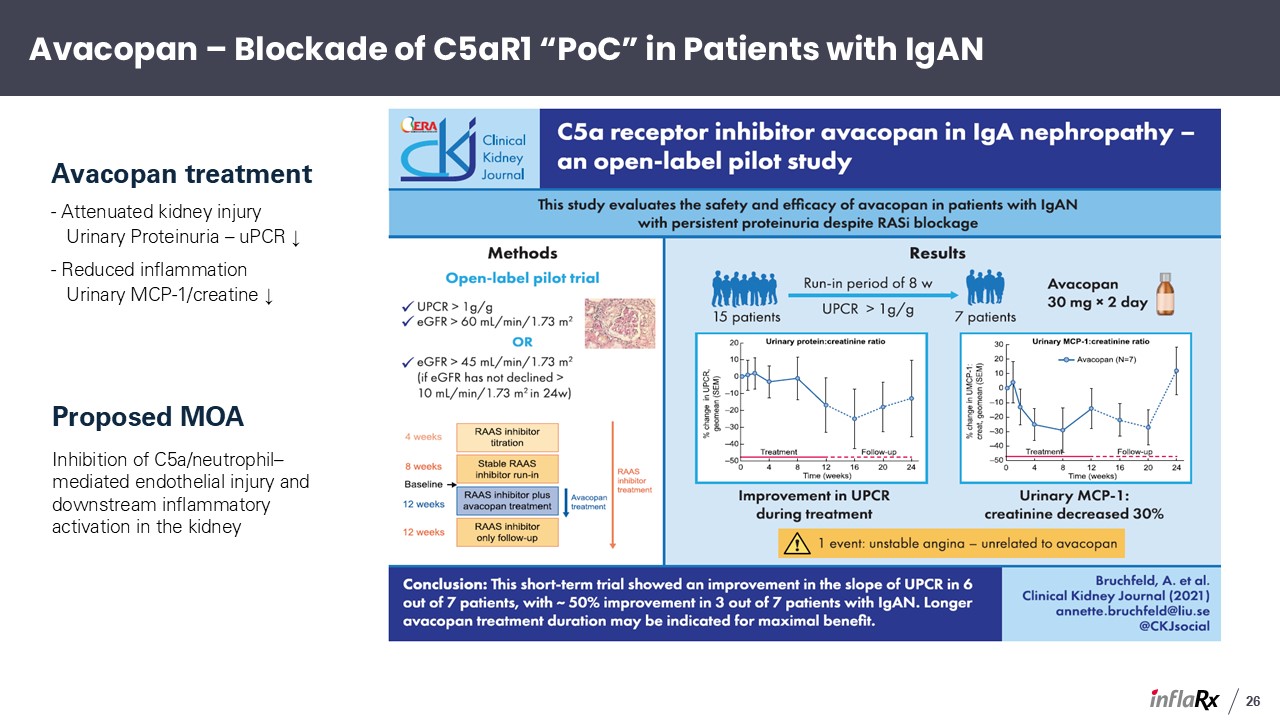
Avacopan treatment - Attenuated kidney injury Urinary Proteinuria – uPCR ↓ -
Reduced inflammation Urinary MCP-1/creatine ↓ Proposed MOA Inhibition of C5a/neutrophil–mediated endothelial injury and downstream inflammatory activation in the kidney Avacopan – Blockade of C5aR1 “PoC” in Patients with IgAN 26

Renal indications C3 glomerulopathy (C3G) CONTROLLING INFLAMMATION
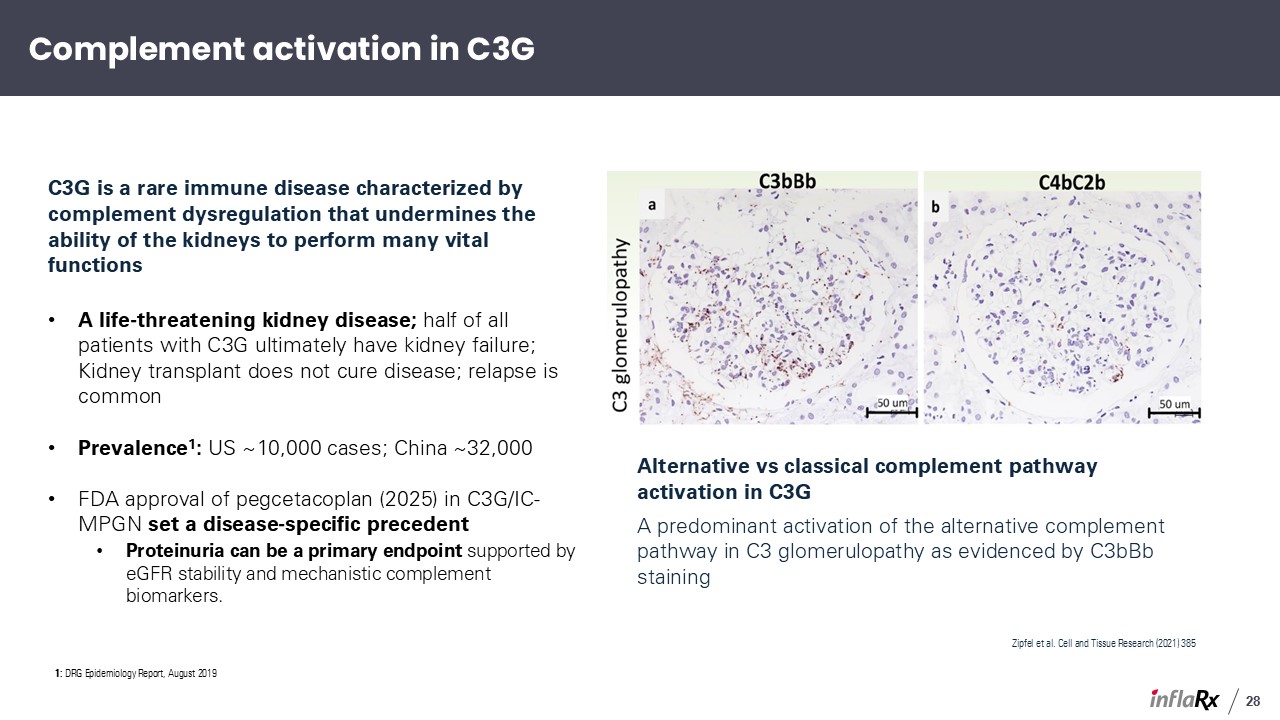
C3G is a rare immune disease characterized by complement dysregulation that undermines
the ability of the kidneys to perform many vital functions A life-threatening kidney disease; half of all patients with C3G ultimately have kidney failure; Kidney transplant does not cure disease; relapse is common Prevalence1: US ~10,000
cases; China ~32,000 FDA approval of pegcetacoplan (2025) in C3G/IC-MPGN set a disease-specific precedent Proteinuria can be a primary endpoint supported by eGFR stability and mechanistic complement biomarkers. Alternative vs classical
complement pathway activation in C3G A predominant activation of the alternative complement pathway in C3 glomerulopathy as evidenced by C3bBb staining Zipfel et al. Cell and Tissue Research (2021) 385 Complement activation in C3G 28 1:
DRG Epidemiology Report, August 2019
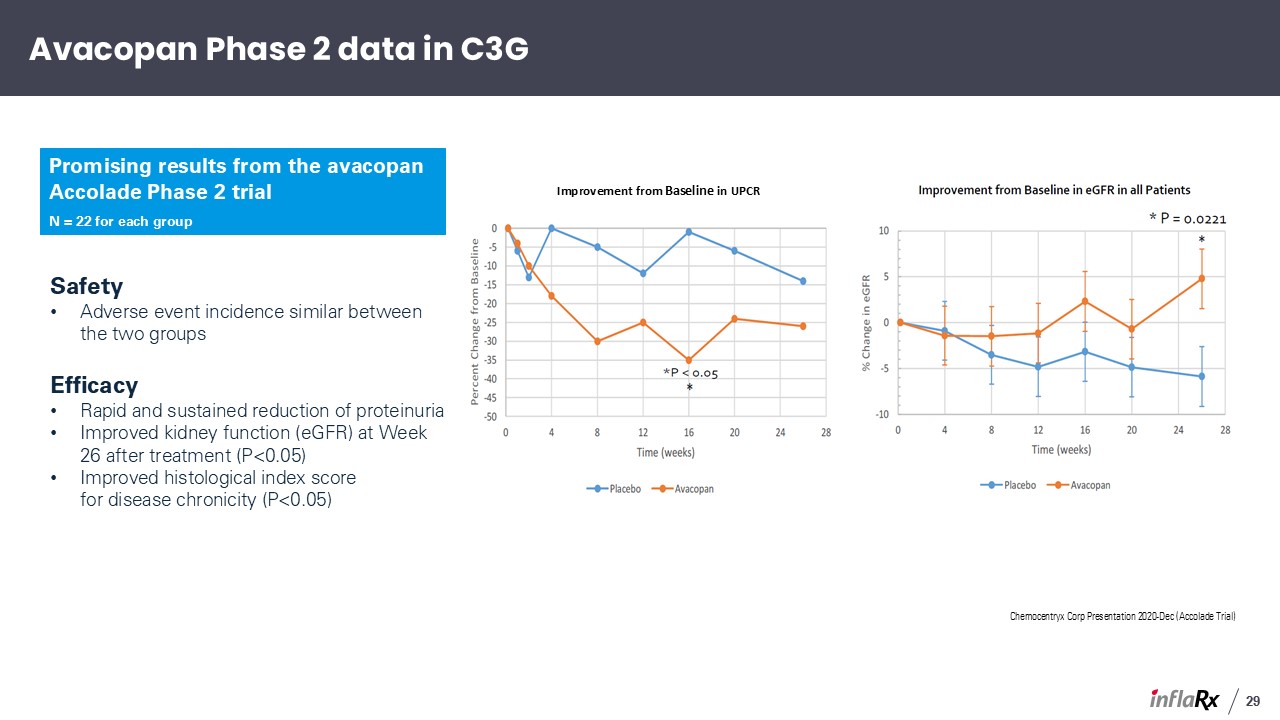
Chemocentryx Corp Presentation 2020-Dec (Accolade Trial) Safety Adverse event
incidence similar between the two groups Efficacy Rapid and sustained reduction of proteinuria Improved kidney function (eGFR) at Week 26 after treatment (P<0.05) Improved histological index score for disease chronicity
(P<0.05) Improvement from Baseline in UPCR Avacopan Phase 2 data in C3G Promising results from the avacopan Accolade Phase 2 trial N = 22 for each group 29

Fast PoC execution Streamlined regulatory pathways for rare diseases, unmet needs, and
strong KOL engagement support rapid enrollment Cost efficiency Lower clinical trial costs can improve capital efficiency and enable larger, faster PoC studies within the same budgets Established clinical expertise and network Strong
investigator networks including key opinion leaders and city centers such as Beijing University Hospital, and population centers, support efficient recruitment and high-quality trial execution Clear endpoint readouts Primary readouts based
on endpoint such as proteinuria reduction (UPCR), eGFR stabilization and optional biomarker profiling to confirm mechanism Potential izicopan renal disease PoC basket studies Izicopan has a differentiated safety and convenience profile
versus other complement inhibitors, including fast onset, oral administration, no vaccination requirement, and no increased infection risk, with potential to drive improved physician adoption, patient acceptance, and treatment
persistence Fast, focused, Phase 2 PoC studies: Smaller sizes, optimized for early go/no-go decision in these rare renal indications 30

Hidradenitis suppurativa (HS) CONTROLLING INFLAMMATION

FDA has acknowledged the importance of draining tunnels (dT) as a major contributor to
HS disease burden FDA supportive of development of a new endpoint in HS, the modified Hidradenitis clinical response score (mHiSCR) which, depending on validation work in a Phase 2b trial, could be used as primary endpoint in pivotal trials.
mHiSCR incorporates reduction of dT by requiring on overall 50% abscess, nodule and dT (ANdT) count reduction from baseline and separately, a 50% dT reduction from baseline as key criteria (next to no increase in abscesses and no new dT
development from baseline) InflaRx believes mHiSCR may be suitable to meaningfully control placebo response rates in HS while catering to izicopan´s MoA Phase 2b study could provide additional insight into the clinical utility of izicopan
in HS, help validate mHiSCR and dT reduction as a label relevant endpoints and establish a dose for Phase 3 HS remains a large market opportunity for izicopan: Currently further development is only envisioned in collaboration with a
partner FDA interactions indicate a viable path forward for izicopan in HS Phase 2a data indicate izicopan has biologic-like activity within just 4 weeks of treatment, with AN count reduction and HiSCR in line with successful Phase 3
comparators, fast and deep reduction in dT, and significant improvement in pain (NRS30) Izicopan shows promise to be a safe oral agent with biologic-like activity and a differentiated new mechanism of action and a market potential that
could exceed $1.5B per year1 1: IFRX proprietary market research, Clarivate. dT [draining tunnel]. Outcome of FDA interactions 32

Appendix CONTROLLING INFLAMMATION
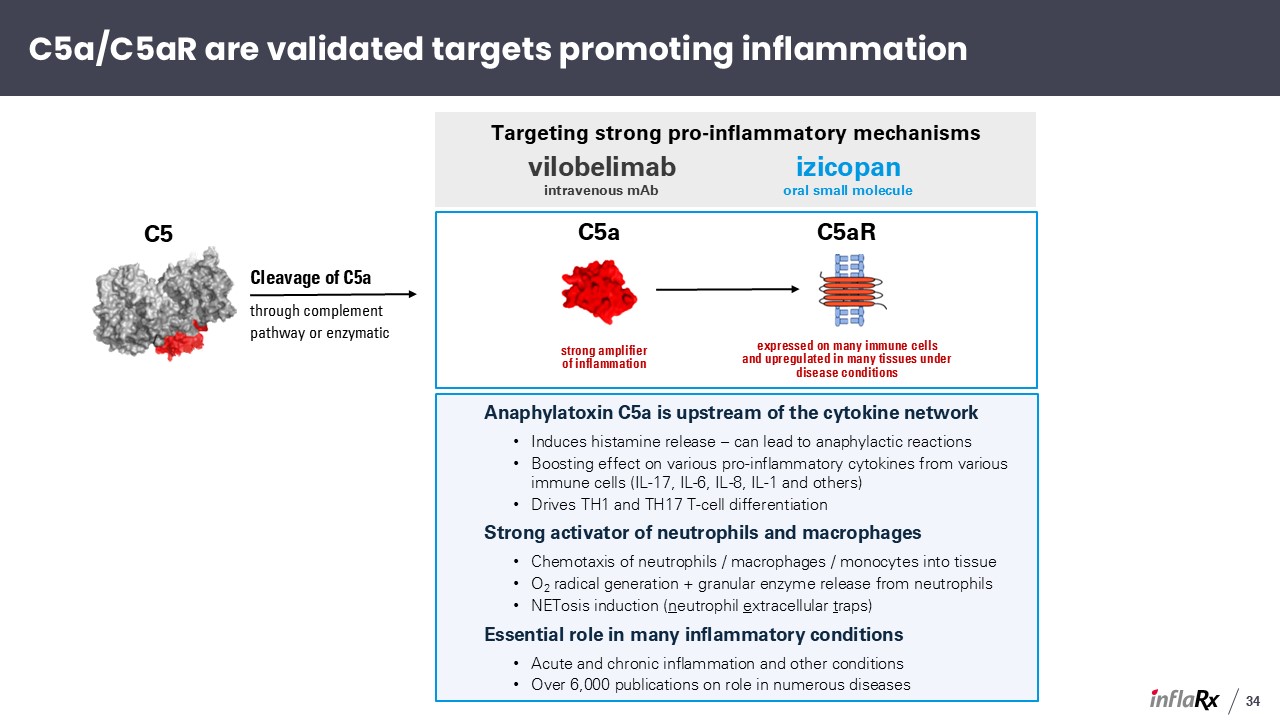
Anaphylatoxin C5a is upstream of the cytokine network Induces histamine release – can
lead to anaphylactic reactions Boosting effect on various pro-inflammatory cytokines from various immune cells (IL-17, IL-6, IL-8, IL-1 and others) Drives TH1 and TH17 T-cell differentiation Strong activator of neutrophils and
macrophages Chemotaxis of neutrophils / macrophages / monocytes into tissue O2 radical generation + granular enzyme release from neutrophils NETosis induction (neutrophil extracellular traps) Essential role in many inflammatory
conditions Acute and chronic inflammation and other conditions Over 6,000 publications on role in numerous diseases C5a strong amplifierof inflammation C5aR expressed on many immune cells and upregulated in many tissues under disease
conditions Targeting strong pro-inflammatory mechanisms vilobelimab intravenous mAb izicopan oral small molecule C5 Cleavage of C5a through complement pathway or enzymatic C5a/C5aR are validated targets promoting inflammation 34
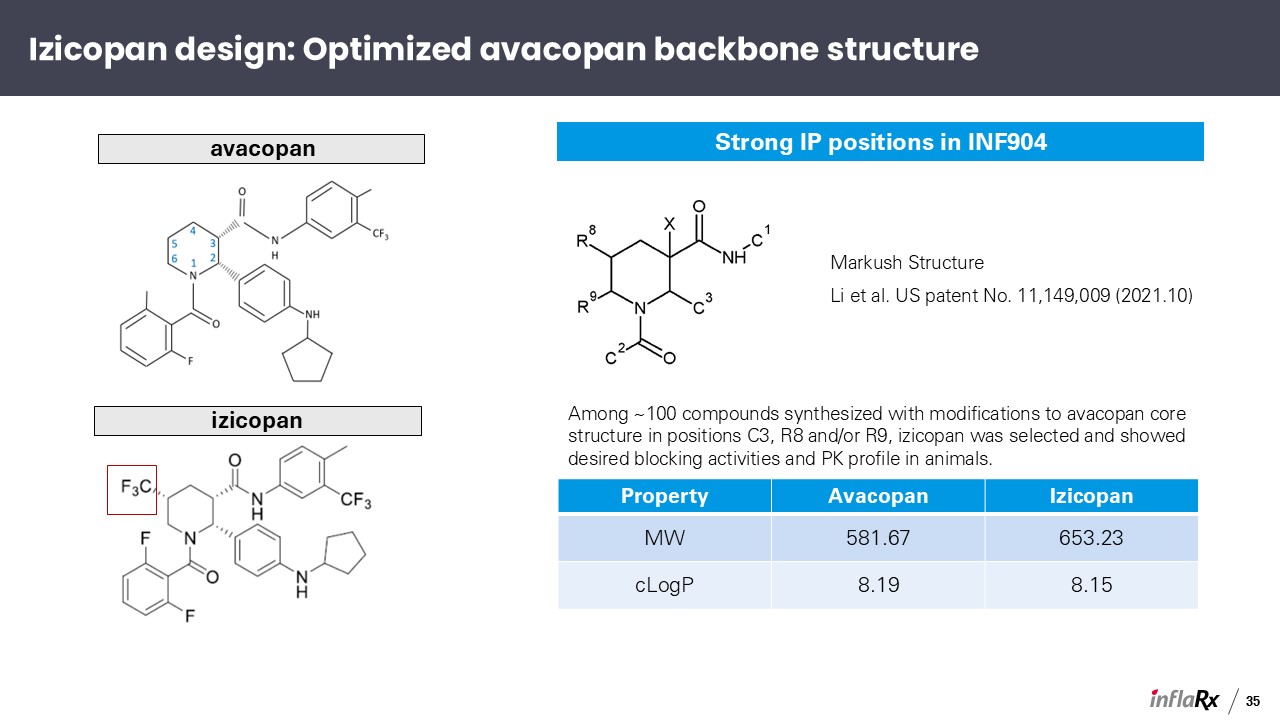
Strong IP positions in INF904 avacopan Among ~100 compounds synthesized with
modifications to avacopan core structure in positions C3, R8 and/or R9, izicopan was selected and showed desired blocking activities and PK profile in animals. Property Avacopan Izicopan MW 581.67 653.23 cLogP 8.19 8.15 Markush
Structure Li et al. US patent No. 11,149,009 (2021.10) izicopan Izicopan design: Optimized avacopan backbone structure 35
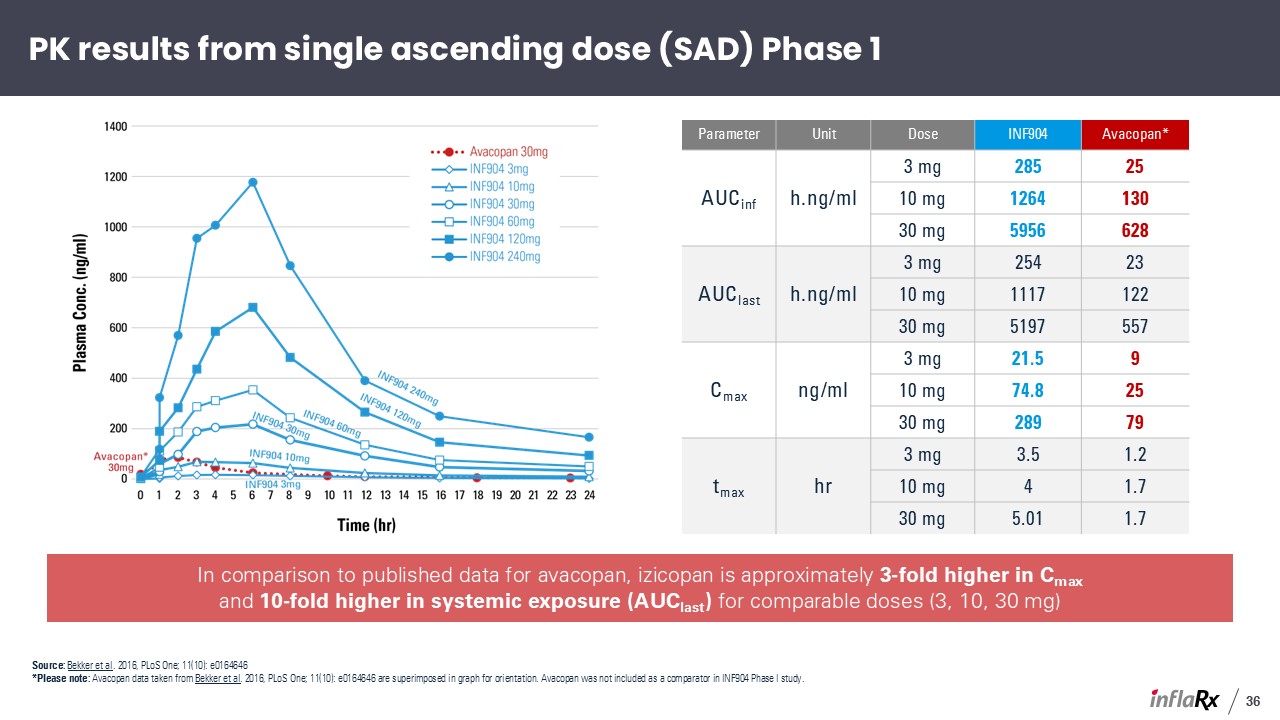
Parameter Unit Dose INF904 Avacopan* AUCinf h.ng/ml 3 mg 285 25 10
mg 1264 130 30 mg 5956 628 AUClast h.ng/ml 3 mg 254 23 10 mg 1117 122 30 mg 5197 557 Cmax ng/ml 3 mg 21.5 9 10 mg 74.8 25 30 mg 289 79 tmax hr 3 mg 3.5 1.2 10 mg 4 1.7 30 mg 5.01 1.7 In comparison to
published data for avacopan, izicopan is approximately 3-fold higher in Cmax and 10-fold higher in systemic exposure (AUClast) for comparable doses (3, 10, 30 mg) PK results from single ascending dose (SAD) Phase 1 36 Source: Bekker et al.
2016, PLoS One; 11(10): e0164646 *Please note: Avacopan data taken from Bekker et al. 2016, PLoS One; 11(10): e0164646 are superimposed in graph for orientation. Avacopan was not included as a comparator in INF904 Phase I study.
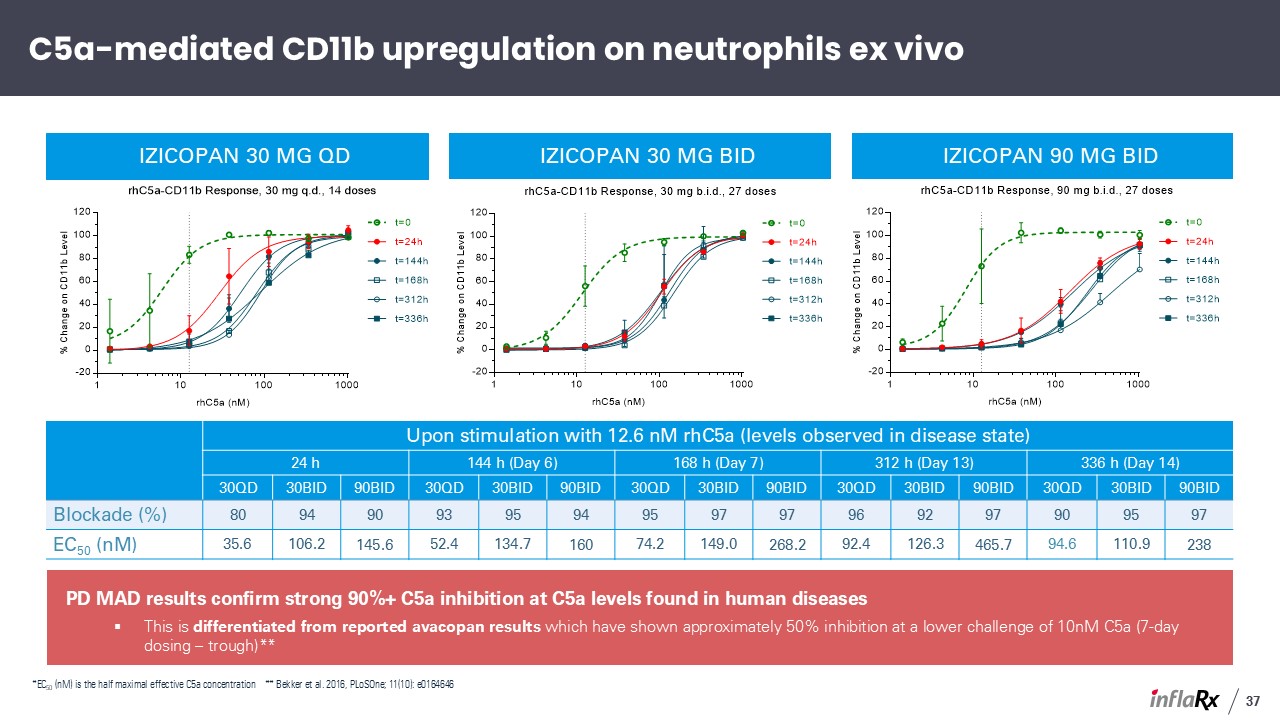
PD MAD results confirm strong 90%+ C5a inhibition at C5a levels found in human
diseases This is differentiated from reported avacopan results which have shown approximately 50% inhibition at a lower challenge of 10nM C5a (7-day dosing – trough)** Upon stimulation with 12.6 nM rhC5a (levels observed in disease
state) 24 h 144 h (Day 6) 168 h (Day 7) 312 h (Day 13) 336 h (Day 14) C5a (nM) 30QD 30BID 90BID 30QD 30BID 90BID 30QD 30BID 90BID 30QD 30BID 90BID 30QD 30BID 90BID Blockade (%)
80 94 90 93 95 94 95 97 97 96 92 97 90 95 97 EC50 (nM) 35.6 106.2 145.6 52.4 134.7 160 74.2 149.0 268.2 92.4 126.3 465.7 94.6 110.9 238 izicopan 30 mg QD izicopan 30 mg BID izicopan 90 mg BID
C5a-mediated CD11b upregulation on neutrophils ex vivo 37 *EC50 (nM) is the half maximal effective C5a concentration ** Bekker et al. 2016, PLoSOne; 11(10): e0164646

InflaRx N.V. CONTROLLING INFLAMMATION Email: IR@inflarx.com Tel:
+49-3641-508180 Fax: +49-3641-508181 www.inflarx.com Winzerlaer Str. 207745 Jena, Germany