Exhibit 99.2

® Developing Advanced Therapies for Cancer Patients
Exhibit 99.2

® Developing Advanced Therapies for Cancer Patients

Forward Looking Statements This presentation contains forward-looking statements (including within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended (the “Securities Act”) concerning Tempest Therapeutics, Inc. (“Tempest Therapeutics”). These statements may discuss goals, intentions and expectations as to future plans, trends, events, results of operations or financial condition, or otherwise, based on current beliefs of the management of Tempest Therapeutics, as well as assumptions made by, and information currently available to, management of Tempest Therapeutics. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as “may,” “will,” “should,” “would,” “could”, “expect,” “anticipate,” “plan,” “likely,” “believe,” “estimate,” “project,” “intend,” and other similar expressions. All statements that are not historical facts are forward-looking statements, including any statements regarding: the potential benefits of Tempest Therapeutics’ expanded oncology pipeline; the design, initiation, progress, timing, scope and results of clinical trials; the anticipated therapeutic benefit, opportunity to improve patient care, and regulatory development of Tempest Therapeutics’ product candidates; Tempest Therapeutics’ ability to deliver on potential value-creating milestones; the potential use of Tempest Therapeutics’ product candidates to treat additional indications; Tempest Therapeutics’ ability to achieve its operational plans; and the sufficiency of Tempest Therapeutics’ cash and cash equivalents. Forward-looking statements are based on current beliefs and assumptions that are subject to risks and uncertainties and are not guarantees of future performance. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including, without limitation: Tempest Therapeutics’ strategies, prospects, plans, expectations or objectives for future operations; the progress, scope or timing of the development of Tempest Therapeutics’ product candidates; the benefits that may be derived from any future products or the commercial or market opportunity with respect to any of Tempest Therapeutics’ future products; unexpected safety or efficacy data observed during preclinical or clinical trials; the possibility that results from prior clinical trials and preclinical studies may not necessarily be predictive of future results; past results may not be indicative of future results; clinical trial site activation or enrollment rates that are lower than expected; loss of key personnel; changes in expected or existing competition; changes in the regulatory environment; risks relating to volatility and uncertainty in the capital markets for biotechnology companies; unexpected litigation or other disputes; Tempest Therapeutics’ ability to protect its intellectual property rights; Tempest Therapeutics’ need for additional capital to fund its planned programs and operations and to continue to operate as a going concern; Tempest Therapeutics’ anticipated operations, financial position, ability to raise capital to fund operations, revenues, costs or expenses; statements regarding future economic conditions or performance; statements of belief and any statement of assumptions underlying any of the foregoing. These and other factors that may cause actual results to differ from those expressed or implied are discussed in greater detail in the “Risk Factors” section of the company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission (SEC) on March 30, 2026, as well as in other filings the company may make with the SEC in the future. Except as required by applicable law, Tempest Therapeutics undertakes no obligation to revise or update any forward-looking statement, or to make any other forward-looking statements, whether as a result of new information, future events or otherwise. These forward-looking statements should not be relied upon as representing Tempest Therapeutics’ views as of any date subsequent to the date of this press release and should not be relied upon as prediction of future events. In light of the foregoing, investors are urged not to rely on any forward-looking statement in reaching any conclusion or making any investment decision about any securities of Tempest Therapeutics. This presentation discusses product candidates that are under clinical study and which have not yet been approved for marketing by the U.S. Food and Drug Administration. No representation is made as to the safety or effectiveness of these product candidates for the use for which such product candidates are being studied. This presentation incorporates publicly-available third-party data that Tempest Therapeutics has not independently verified. There are risks inherent in conducting cross-trial comparisons and the results should be interpreted with caution. The presentation of such third-party data does not represent a head-to-head comparison of how TPST-2003 performed against any other third-party drug candidate or study. Rather, such third-party data has been pulled by us from publicly-available sources for supplemental informational purposes, only. Tempest Therapeutics cautions you that any comparisons against third-party data set forth herein should not be viewed as a side-by-side comparison, and you should not rely on the completeness or accuracy of Tempest Therapeutics’ presentation of the results of any third-party drug candidate in these slides, due to differences in study design, how other companies quantify or qualify eligibility criteria, and how results are recorded, among other distinguishing factors and uncertainties. Because Tempest Therapeutics may be unaware of or may not adequately present various distinguishing factors and uncertainties, the comparisons set forth herein may not properly present such third-party data, which may differ materially from the data as presented here. Investors are encouraged to independently review third party data and should not rely on Tempest Therapeutics’ presentation of such data (including any such data placed in comparison with the performance of TPST-2003) as a single measure to evaluate Tempest Therapeutics’ business. The trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of such products.
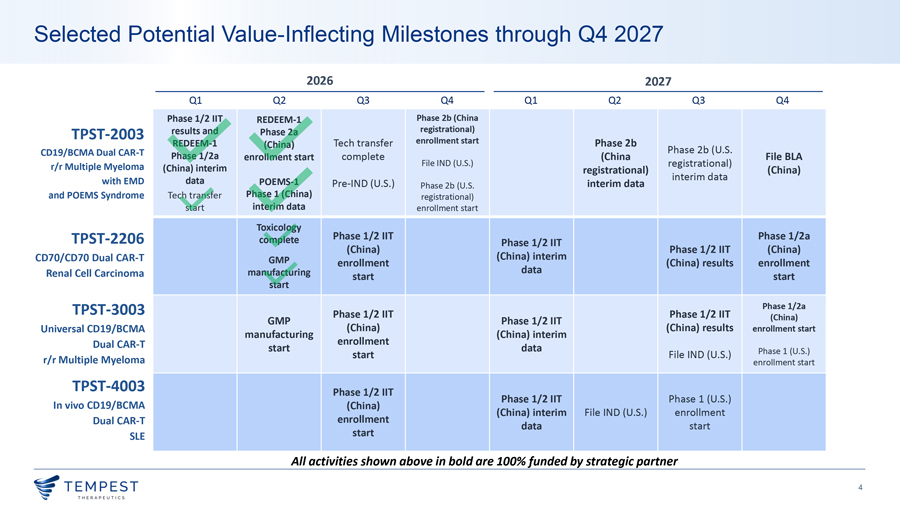
Selected Potential Value-Inflecting Milestones through Q4 2027 2026 2027 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Phase 1/2 IIT REDEEM-1 Phase 2b (China TPST-2003 results and Phase 2a registrational) REDEEM-1 Tech transfer enrollment start Phase 2b (China) CD19/BCMA Dual CAR-T Phase 2b (U.S. Phase 1/2a enrollment start complete File IND (U.S.) (China File BLA r/r Multiple Myeloma registrational) (China) interim registrational) interim data (China) with EMD data POEMS-1 Pre-IND (U.S.) interim data Phase 2b (U.S. and POEMS Syndrome Tech transfer Phase 1 (China) registrational) start interim data enrollment start Toxicology Phase 1/2 IIT Phase 1/2a TPST-2206 complete Phase 1/2 IIT (China) Phase 1/2 IIT (China) CD70/CD70 Dual CAR-T GMP (China) interim enrollment (China) results enrollment Renal Cell Carcinoma manufacturing data start start start TPST-3003 Phase 1/2 IIT Phase 1/2 IIT Phase 1/2a GMP Phase 1/2 IIT (China) Universal CD19/BCMA (China) (China) results enrollment start manufacturing (China) interim Dual CAR-T enrollment start data Phase 1 (U.S.) start File IND (U.S.) r/r Multiple Myeloma enrollment start TPST-4003 Phase 1/2 IIT Phase 1/2 IIT Phase 1 (U.S.) In vivo CD19/BCMA (China) (China) interim File IND (U.S.) enrollment Dual CAR-T enrollment data start SLE start All activities shown above in bold are 100% funded by strategic partner

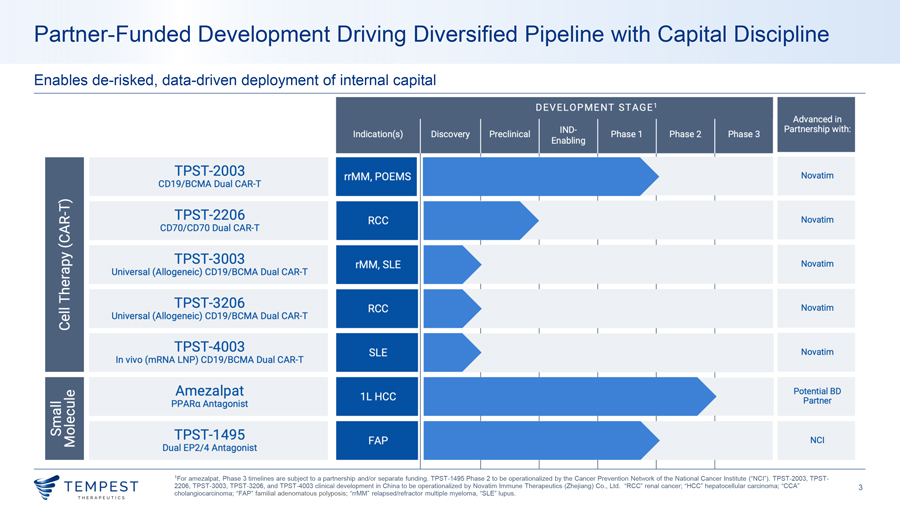
TPST-2003 Dual Targeting CD19/BCMA CAR-T

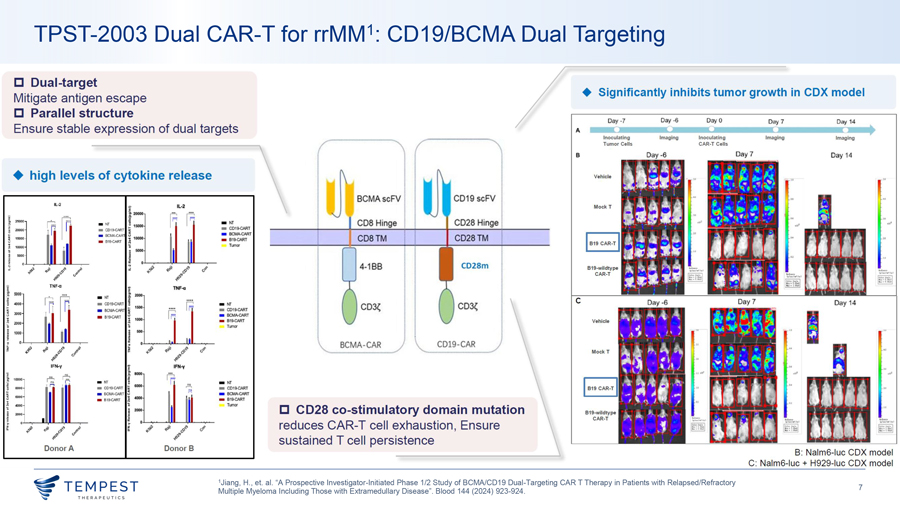
TPST-2003 CD19/BCMA CAR-T TPST-20031,2 is the world’s first parallel-structure dual-target CAR-T cell therapy for rrMM with EMD & POEMS syndrome • REDEEM-1 Phase 1/2a (rrMM) and POEMS-1 Phase 1 (POEMS syndrome) Dual-target CAR-T structure • 44 patient target – 32 in REDEEM-1 and 12 in POEMS-1 • 20 patients dosed as of May 6, 2026 – 13 in REDEEM-1, 7 in POEMS-1 • 100% CR rate among CAR-T-naïve patients (15/15) – REDEEM-1 (10/10 CR) and POEMS-1 (5/5 CRVEGF) efficacy-evaluable as of March 31, 2026 and January 31, 2026, respectively • No grade ≥3 CRS, no grade ≥3 ICANS in REDEEM-1, Phase 1 enrollment complete (12/12), Phase 2a currently enrolling, first patient dosed May 2, 2026 • Phase 1/2 Investigator-Initiated Trial (rrMM) – Enrollment complete (24 patients) • 100% ORR among all 19 patients with measurable disease at baseline, 89.5% CR rate (17/19), 100% CR rate at highest dose level (5/5) • Median PFS of 23.1 months across all patients (24/24), median PFS of 23.1 months in EMD patients (15/15) • All evaluable patients at month 12 were MRD-negative (5/5) 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory Multiple Myeloma Including Those with Extramedullary Disease”. Blood 144 (2024) 923- 924., 2Li, J., et. al. “REDEEM-1, A MULTICENTER OPEN-LABEL PHASE 1/2A STUDY OF A BCMA/CD19 DUAL-TARGETING CAR-T THERAPY IN PATIENTS WITH RELAPSED/REFRACTORY MULTIPLE MYELOMA INCLUDING THOSE 6 WITH EXTRAMEDULLARY DISEASE”. Cytotherapy 28 (2026) 1268., Interim data update, May 6, 2026. “CRS” Cytokine Release Syndrome, “EMD” Extramedullary Disease, “CR” Complete Response, “CRVEGF” Complete Response as measured by normalization of serum vascular endothelial growth factor levels, “ORR” Overall Response Rate, “PFS” Progression-Free Survival, “ICANS” Immune effector cell-associated neurotoxicity syndrome, “MRD” Minimal Residual Disease.
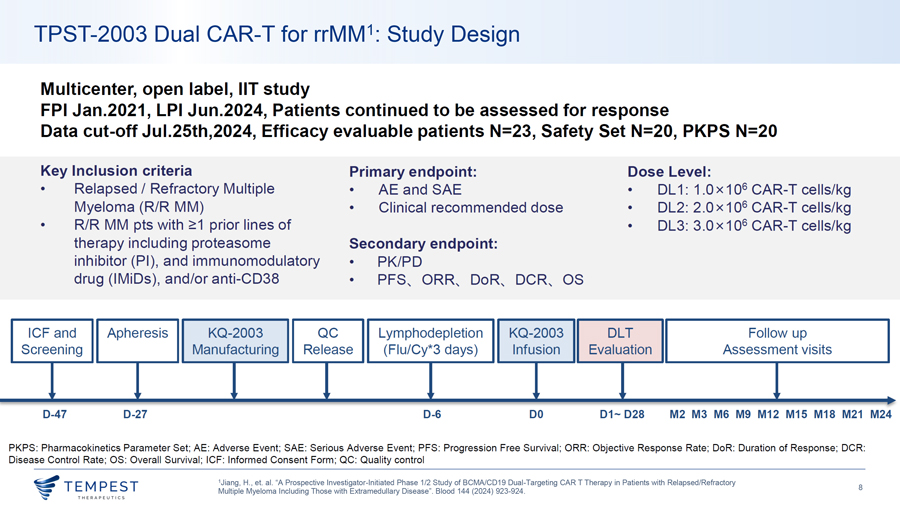
TPST-2003 Dual CAR-T for rrMM1: Study Design Multicenter, open label, IIT study FPI Jan.2021, LPI Jun.2024, Patients continued to be assessed for response Data cut-off Jul.25th,2024, Efficacy evaluable patients N=23, Safety Set N=20, PKPS N=20 Key Inclusion criteria Primary endpoint: Dose Level: • Relapsed/ Refractory Multiple • AE and SAE • DL 1: 1.0x106 CAR-T cells/kg Myeloma (R/R MM) • Clinical recommended dose • DL2: 2 .0 x 106 CAR-T cells/kg • R/R MM pts with ~1 prior lines of • DL3: 3.0x106 CAR-T cells/kg therapy including proteasome Secondary endpoint: inhibitor (Pl), and immunomodulatory • PK/PD drug (IMiDs), and/or anti-CD38 • PFS, ORR, DoR, OCR, OS ICF and Apheresis KQ-2003 QC Lymphodepletion KQ-2003 DLT Follow up Screening Manufacturing Release (Flu/Cy*3 days) Infusion Evaluation Assessment visits , , , ‘ ‘ ‘ ‘ ... ~ ‘ D-47 D-27 D-6 DO D1~ D28 M2 M3 MG M9 M12 M15 M18 M21 M24 PKPS : Pharmacokinetics Parameter Set; AE: Adverse Event; SAE: Serious Adverse Event; PFS: Progression Free Survival ; ORR: Objective Response Rate; DoR: Duration of Response; OCR: Disease Control Rate; OS: Overall Survival; ICF: Informed Consent Form ; QC: Quality control 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory ~TEMPEST 8 Multiple Myeloma Including Those with Extramedullary Disease”. Blood 144 (2024) 923-924. ~ THERAPEUT I CS
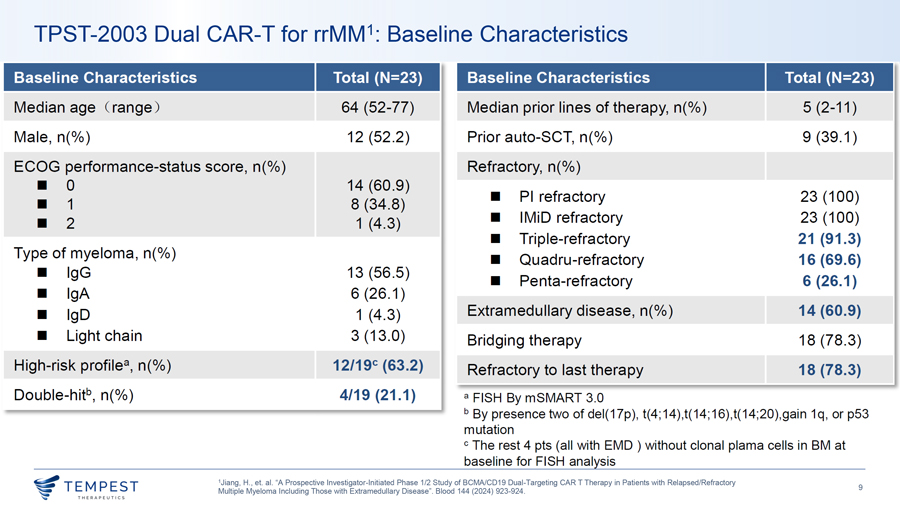
TPST-2003 Dual CAR-T for rrMM1: Baseline Characteristics Baseline Characteristics Total {N=23) Baseline Characteristics Total {N=23) Median age Crange ) 64 (52-77) Median prior lines of therapy, n(%) 5 (2-11) Male, n(%) 12 (52.2) Prior auto-SCT, n(%) 9 (39.1) ECOG performance-status score, n(%) Refractory, n(%) ¦ 0 14 (60.9) ¦ Pl refractory 23 (100) ¦ 1 8 (34.8) 2 ¦ IMiD refractory 23 (100) ¦ 1 (4.3) ¦ Triple-refractory 21 (91.3) Type of myeloma, n(%) ¦ Quadru-refractory 16 (69.6) ¦ lgG 13 (56.5) ¦ Penta-refractory 6 (26.1) ¦ lgA 6 (26.1) ¦ lgD 1 (4.3) Extramedullary disease, n(%) 14 (60.9) ¦ Light chain 3 (13.0) Bridging therapy 18 (78.3) High-risk profile8 , n(%) 12/19c {63.2) Refractory to last therapy 18 (78.3) Double-hitb, n(%) 4/19 (21.1) a FISH By mSMART 3.0 b By presence two of del(17p), t(4;14),t(14;16),t(14;20),gain 1q, or p53 mutation c The rest 4 pts (all with EMO ) without clonal plama cells in BM at baseline for FISH analysis 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory ~TEMPEST 9 Multiple Myeloma Including Those with Extramedullary Disease”. Blood 144 (2024) 923-924.
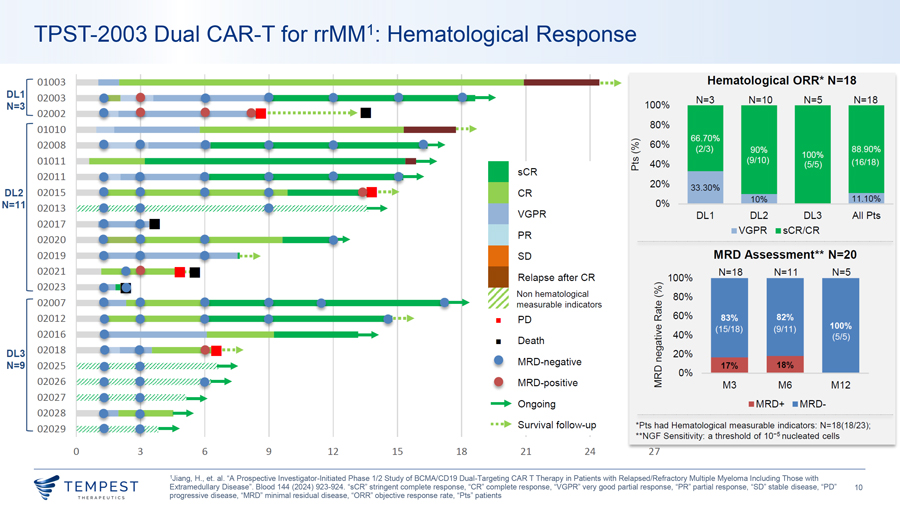
TPST-2003 Dual CAR-T for rrMM1: Hematological Response 01003 Hematological ORR* N=18 1 [ 02003 DL N=3 N=10 N=5 N=18 N=3 100% 02002 80% 01010 -02008 ~ 60% - 0 01011 CJ) 40% 0:: sCR 02011 20% 33.30% DL2 02015 CR N=11 0% 02013 1//////////////////////// ....... VGPR DL1 DL2 DL3 All Pts 02017 ¦ VGPR ¦ sCR/CR PR 02020 02019 SD MRD Assessment** N=20 02021 N= 18 N=11 N=5 Relapse after CR—100% 02023 Non hematological 0 ~ - 80% 02007 measurable indicators Q.) ro 02012 0:: 60% ¦ PD Q.) 02016 > 40% :;:; ¦ Death Ctl 02018 Ol DL3 Q.) 20% MRD-negative C N=9 02025 0 0% 0:: 02026 MRD-positive ~ M3 M6 M1 2 02027 ..... Ongoing ¦ M RD+ ¦ M RD- 02028 02029 •••► Survival fo llow-up *Pts had Hematological measurable indicators: N=18(18/23); **NGF Sensitivity: a threshold of 10-5 nucleated cells 0 3 6 9 12 15 18 21 24 27 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory Multiple Myeloma Including Those with ~TEMPEST Extramedullary Disease”. Blood 144 (2024) 923-924. “sCR” stringent complete response, “CR” complete response, “VGPR” very good partial response, “PR” partial response, “SD” stable disease, “PD” 10 ~ THERAPEUT I CS progressive disease, “MRD” minimal residual disease, “ORR” objective response rate, “Pts” patients
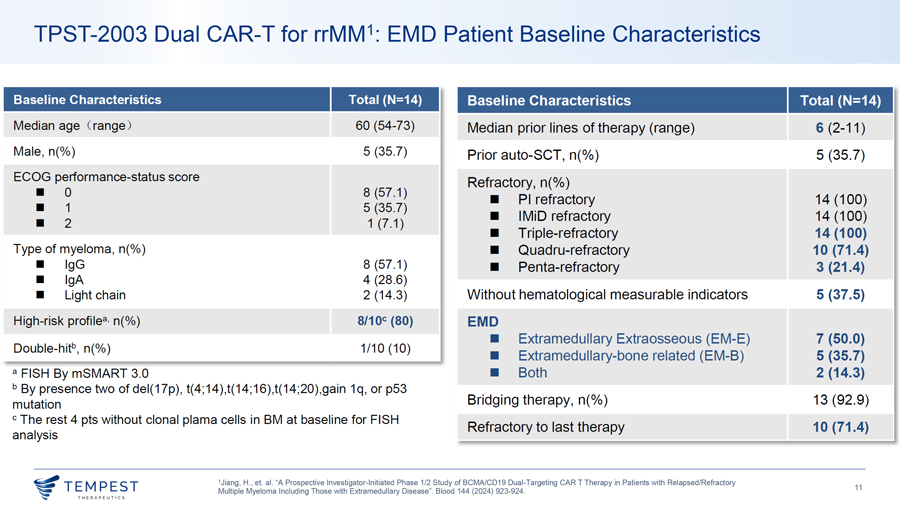
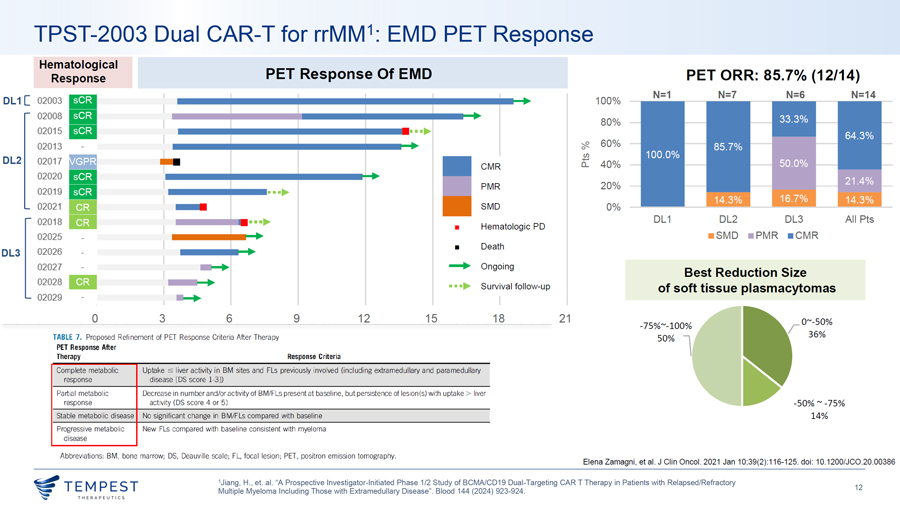
TPST-2003 Dual CAR-T for rrMM1: EMD PET Response Hematological PET Response Of EMD Response PET ORR: 85.7% (12/14) N=1 N=7 N=6 N=14 DL1[ 02003—100% 02008—80% 02015—•••► 02013 0 -;,12_ 60% DL2 Cl) 02017 0.... 40% CMR 02020 PMR 20% 02019 •••► 02021 SMD 0% 02018 Hematologic PD DL 1 DL2 DL3 All Pts •••► ¦ 02025 ¦ SMD ¦ PMR ¦ CMR ¦ Death DL3 02026 02027 —► Ongoing Best Reduction Size 02028 •••► Survival follow-up of soft tissue plasmacytomas 02029 - 0 3 6 9 12 15 18 21 ——·-···-······-···-· ·-· ——·—···· ·-······ -75%~-100% TABLE 7. Proposed Refinement of PET Response Criteria After Therapy PET Response After 50% l Therapy Response Criteria Complete metabolic Uptake c;; liver activity in BM sites and Fls previously involved (including extramedullary and paramedullary response disease [DS score 1-3]) Partial metabolic Decrease in number and/or activity of BM/Fls present at baseline, but persistence of lesion(s) with uptake > liver response activity (DS score 4 or 5) -50% ~ -75% Stable metabolic disease No significant change in BM/Fls compared with baseline 14% Progressive metabolic New Fls compared with baseline consistent with myeloma disease Abbreviations: BM, bone marrow; DS, Deauville scale; FL, focal lesion; PET, positron emission tomography. Elena Zamagni, et al. J Clin Oneal. 2021 Jan 10;39(2):1 16-125. doi: 10.1200/JC0.20.00386 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory ~TEMPEST 12 Multiple Myeloma Including Those with Extramedullary Disease”. Blood 144 (2024) 923-924.
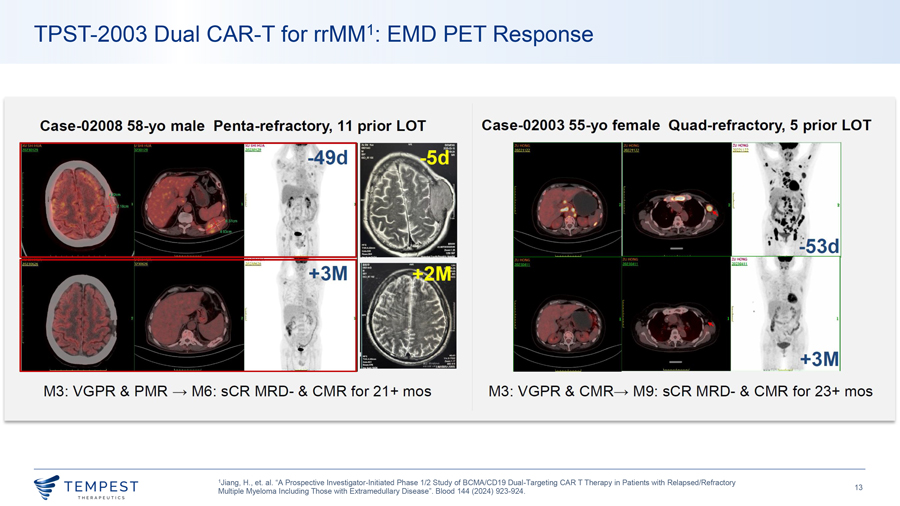
TPST-2003 Dual CAR-T for rrMM1: EMD PET Response Case-02008 58-yo male Penta-refractory, 11 prior LOT Case-02003 55-yo female Quad-refractory, 5 prior LOT M3: VGPR & PMR -c, M6: sCR MRD- & CMR for 21 + mos M3: VGPR & CMR-c, M9: sCR MRD- & CMR for 23+ mos 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory ~TEMPEST 13 Multiple Myeloma Including Those with Extramedullary Disease”. Blood 144 (2024) 923-924. ~ THERAPEUT I CS
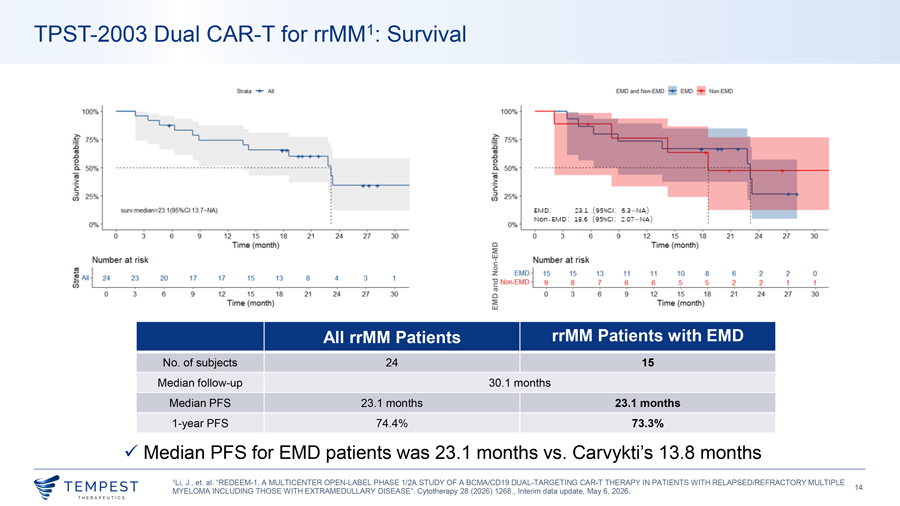
TPST-2003 Dual CAR-T for rrMM1: Survival All EMO NonEJ.,D 100% 100% ~ 15% li 111, ~· 2’ ~ 50%, iii, 2 > ;;;J, (I), ,5~ staVmelfian=23.1(95%ct 13.7--:W,,) E:M D: ::::;u ( 9 s • 1: 6.a- ) NOfl- EM D : S.6 ( 9511GI : :?.07—) 0% 0% 0 3 6 9 2 15 18 21 24 21 30 0 3 6 9 12 5 18 21 21 30 ·me, (month) EMD ime (mol’l1!h} Number at ris!k - Nu.mber a.t l’lsk ! : I Non 15 15 13 11 11 10 8 •6 .2 2 0 23 20 17 17 15 13 8 3 ED ~ !;Alll 2~ • and Non- II D 9 8 1 6 6 5 5 2 2 ‘ ‘ ‘ ‘ ‘ ‘ ‘ ‘ ‘ ‘ g, ‘ ‘ ‘ ‘ ‘ ‘ 0 J 6 9 12 15 18 2 1 24 27 30 0 J •6 12 15 18 21 24 27 J8 Time (monlh) EMD T“me ( om J All rrMM Patients rrMM Patients with EMD No. of subjects 24 15 Median follow-up 30.1 months Median PFS 23.1 months 23.1 months 1-year PFS 74.4% 73.3% ✓ Median PFS for EMD patients was 23.1 months vs. Carvykti’s 13.8 months 1Li, J., et. al. “REDEEM-1, A MULTICENTER OPEN-LABEL PHASE 1/2A STUDY OF A BCMA/CD19 DUAL-TARGETING CAR-T THERAPY IN PATIENTS WITH RELAPSED/REFRACTORY MULTIPLE ~TEMPEST 14 MYELOMA INCLUDING THOSE WITH EXTRAMEDULLARY DISEASE”. Cytotherapy 28 (2026) 1268., Interim data update, May 6, 2026. ~ THERAPEUT I CS
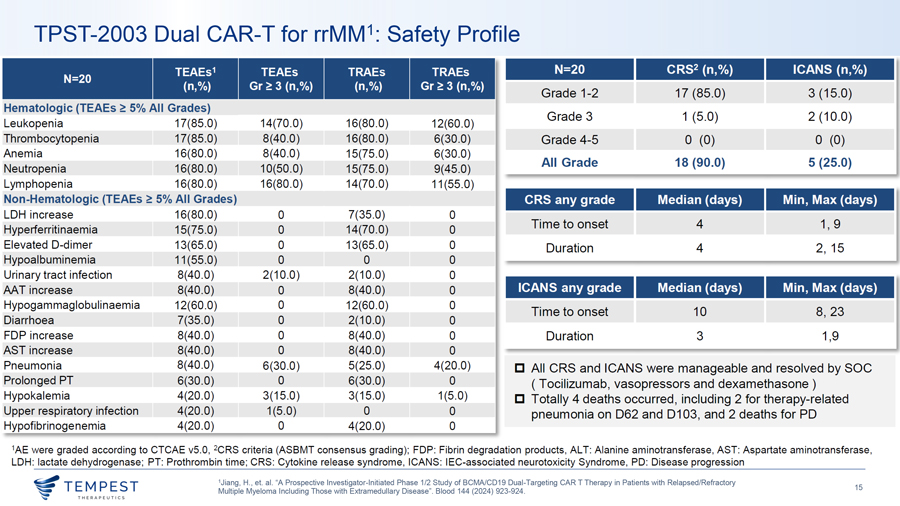
TPST-2003 Dual CAR-T for rrMM1: Safety Profile TEAEs1 TEAEs TRAEs TRAEs N=20 CRS2 (n,%) ICANS (n,%) N=20 (n,%) Gr~ 3 (n,%) (n,%) Gr~ 3 (n,%) Grade 1-2 17 (85.0) 3 (15.0) Hematologic (TEAEs ~ 5% All Grades) Grade 3 1 (5.0) 2 (10.0) Leukopenia 17(85.0) 14(70.0) 16(80.0) 12(60.0) Thrombocytopenia 17(85.0) 8(40.0) 16(80.0) 6(30.0) Grade 4-5 0 (0) 0 (0) Anemia 16(80.0) 8(40.0) 15(75.0) 6(30.0) All Grade 18 (90.0) 5 (25.0) Neutropenia 16(80.0) 10(50.0) 15(75.0) 9(45.0) Lymphopenia 16(80.0) 16(80.0) 14(70.0) 11 (55.0) Non-Hematologic (TEAEs ~ 5% All Grades) CRS any grade Median (days) Min, Max (days) LOH increase 16(80.0) 0 7(35.0) 0 Time to onset 4 1, 9 Hyperferritinaemia 15(75.0) 0 14(70.0) 0 Elevated D-dimer 13(65.0) 0 13(65.0) 0 Duration 4 2, 15 Hypoalbuminemia 11 (55.0) 0 0 0 Urinary tract infection 8(40.0) 2(10.0) 2(10.0) 0 MT increase 8(40.0) 0 8(40.0) 0 ICANS any grade Median (days) Min, Max (days) Hypogam maglobuli naemia 12(60.0) 0 12(60.0) 0 Diarrhoea 7(35.0) 0 2(10.0) Time to onset 10 8, 23 0 FOP increase 8(40.0) 0 8(40.0) 0 Duration 3 1,9 AST increase 8(40.0) 0 8(40.0) 0 Pneumonia 8(40.0) 6(30.0) 5(25.0) 4(20.0) □ All CRS and ICANS were manageable and resolved by SOC Prolonged PT 6(30.0) 0 6(30.0) 0 ( Tocilizumab, vasopressors and dexamethasone) Hypokalemia 4(20.0) 3(15.0) 3(15.0) 1(5.0) □ Totally 4 deaths occurred , including 2 for therapy-related Upper respiratory infection 4(20.0) 1(5.0) 0 0 pneumonia on 062 and 0103, and 2 deaths for PD Hypofibrinogenemia 4(20.0) 0 4(20.0) 0 1AE were graded according to CTCAE v5.0, 2CRS criteria (ASBMT consensus grading); FOP: Fibrin degradation products, ALT: Alanine aminotransferase, AST: Aspartate aminotransferase, LOH: lactate dehydrogenase; PT: Prothrombin time; CRS: Cytokine release syndrome, ICANS: !EC-associated neurotoxicity Syndrome, PD: Disease progression 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory ~TEMPEST 15 Multiple Myeloma Including Those with Extramedullary Disease”. Blood 144 (2024) 923-924. ~ THERAPEUT I CS
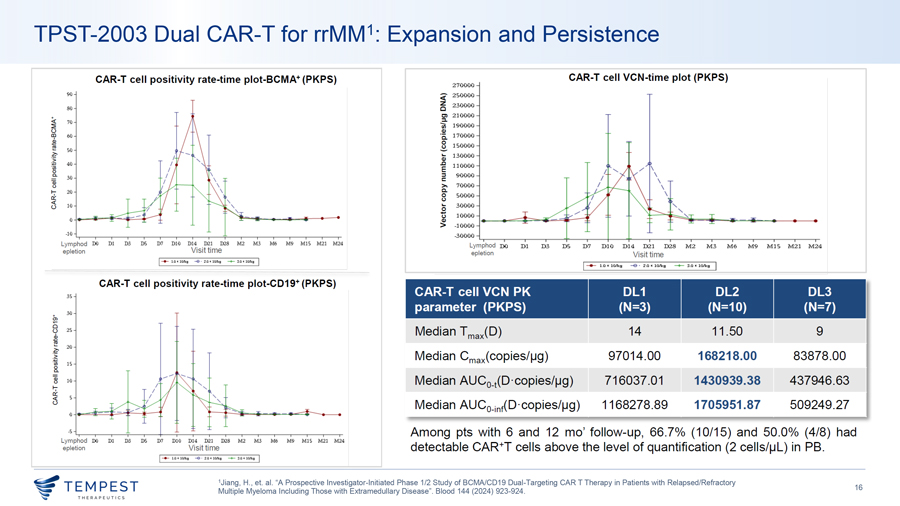
TPST-2003 Dual CAR-T for rrMM1: Expansion and Persistence CAR-T cell positivity rate-time plot-BCMA+ (PKPS) CAR-T cell VCN-time plot (PKPS) 270000 90 <( 250000 ~ 230000 80 Cl 210000 :::1. ~ ~ 70 ui 190000 u QI 00 60 ‘ii 170000 2 8 150000 ~ so ~ ai 130000 1 40 ‘iii i 110000 \ 0 a. 30 ~ 90000 \ ai >, 70000 \ \ ~ “ 20 C. 0 50000 \ u ,_ t3 10 0 30000 .... u 10000 QI > -10000 -10 ..,.__—————-~———-~ -30000 Lymphod DO DI D3 DS D7 DIO Dl4 D21 D28 M2 M3 M6 M9 MIS M21 M24 Lymphod DO D1 D3 DS D7 D10 D14 D21 D28 M2 M3 M6 M9 M15 M21 M24 epletion Visit time epletion Visit time I-+- l .01ClOlq: -0 · 2.0~lOJ’itJ -+- ‘.lll~ l lllq I 1 —- l.Q IC 1O/k,g -e. 2.0 IC 101kg -+- 3.0 IC 10,‘kg I CAR-T cell positivity rate-time plot-CD19+ (PKPS) CAR-T cell VCN PK DL1 DL2 DL3 35 parameter (PKPS) (N=3) (N=10) (N=7) a, 30 8 25 Median T max(D) 14 11 .50 9 d, ! 20 ·;; Median Cmax(copies/µg) 97014.00 168218.00 83878.00 ~ 15 8_ ] 10 Median AUC _ lD ·copies/µg) 716037.01 1430939.38 437946.63 1-;- 0 Cl:’. u <I: 5 Median AUCo-int(D ·copies/µg) 1168278.89 1705951.87 509249 .27 -5 Among pts with 6 and 12 mo’ follow-up, 66.7% (10/15) and 50 .0% (4/8) had ..,.__———-+———-~~~-~-~~ Lymphod DO DI D3 DS D7 DIO Dl4 D21 D28 M2 M3 M6 M9 MIS M21 M24 epletion Visit time detectable CAR+T cells above the level of quantification (2 cells/µL) in PB. I-+- l .0 ~10/ki -e—2.0• IO~ -+- :SD ~lO~ I 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory ~TEMPEST 16 Multiple Myeloma Including Those with Extramedullary Disease”. Blood 144 (2024) 923-924. ~ THERAPEUT I CS

TPST-2003 Dual CAR-T for rrMM: Conclusions TPST-20031,2 is the world’s first parallel-structure dual-target CAR-T cell therapy for rrMM with EMD & POEMS syndrome • REDEEM-1 Phase 1/2a (rrMM) and POEMS-1 Phase 1 (POEMS syndrome) • 44 patient target – 32 in REDEEM-1 and 12 in POEMS-1 • 20 patients dosed as of May 6, 2026 – 13 in REDEEM-1, 7 in POEMS-1 • 100% CR rate among CAR-T-naïve patients (15/15) – REDEEM-1 (10/10 CR) and POEMS-1 (5/5 CRVEGF) efficacy-evaluable as of March 31, 2026 and January 31, 2026, respectively • No grade ≥3 CRS, no grade ≥3 ICANS in REDEEM-1, Phase 1 enrollment complete (12/12), Phase 2a currently enrolling, first patient dosed May 2, 2026 • Phase 1/2 Investigator-Initiated Trial (rrMM) – Enrollment complete (24 patients) • 100% ORR among all 19 patients with measurable disease at baseline, 89.5% CR rate (17/19), 100% CR rate at highest dose level (5/5) • Median PFS of 23.1 months across all patients (24/24), median PFS of 23.1 months in EMD patients (15/15) • All evaluable patients at month 12 were MRD-negative (5/5) 1Jiang, H., et. al. “A Prospective Investigator-Initiated Phase 1/2 Study of BCMA/CD19 Dual-Targeting CAR T Therapy in Patients with Relapsed/Refractory Multiple Myeloma Including Those with ~TEMPEST Extramedullary Disease”. Blood 144 (2024) 923-924., 2Li, J., et. al. “REDEEM-1, A MULTICENTER OPEN-LABEL PHASE 1/2A STUDY OF A BCMA/CD19 DUAL-TARGETING CAR-T THERAPY IN 17 THERAPEUT I CS PATIENTS WITH RELAPSED/REFRACTORY MULTIPLE MYELOMA INCLUDING THOSE WITH EXTRAMEDULLARY DISEASE”. Cytotherapy 28 (2026) 1268., Interim data update, May 6, 2026.
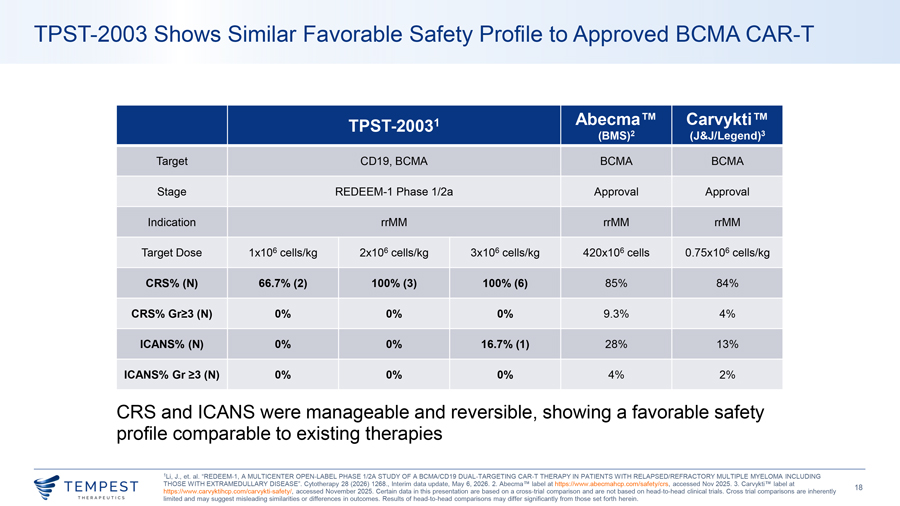
TPST-2003 Shows Similar Favorable Safety Profile to Approved BCMA CAR-T TPST-20031 Abecma Carvykti (BMS)2 (J&J/Legend)3 Target CD19, BCMA BCMA BCMA Stage REDEEM-1 Phase 1/2a Approval Approval Indication rrMM rrMM rrMM Target Dose 1x106 cells/kg 2x106 cells/kg 3x106 cells/kg 420x106 cells 0.75x106 cells/kg CRS% (N) 66.7% (2) 100% (3) 100% (6) 85% 84% CRS% Gr≥3 (N) 0% 0% 0% 9.3% 4% ICANS% (N) 0% 0% 16.7% (1) 28% 13% ICANS% Gr ≥3 (N) 0% 0% 0% 4% 2% CRS and ICANS were manageable and reversible, showing a favorable safety profile comparable to existing therapies 1Li, J., et. al. “REDEEM-1, A MULTICENTER OPEN-LABEL PHASE 1/2A STUDY OF A BCMA/CD19 DUAL-TARGETING CAR-T THERAPY IN PATIENTS WITH RELAPSED/REFRACTORY MULTIPLE MYELOMA INCLUDING THOSE WITH EXTRAMEDULLARY DISEASE”. Cytotherapy 28 (2026) 1268., Interim data update, May 6, 2026. 2. Abecma TM label at https://www.abecmahcp.com/safety/crs, accessed Nov 2025. 3. Carvykti TM label at ~TEMPEST 18
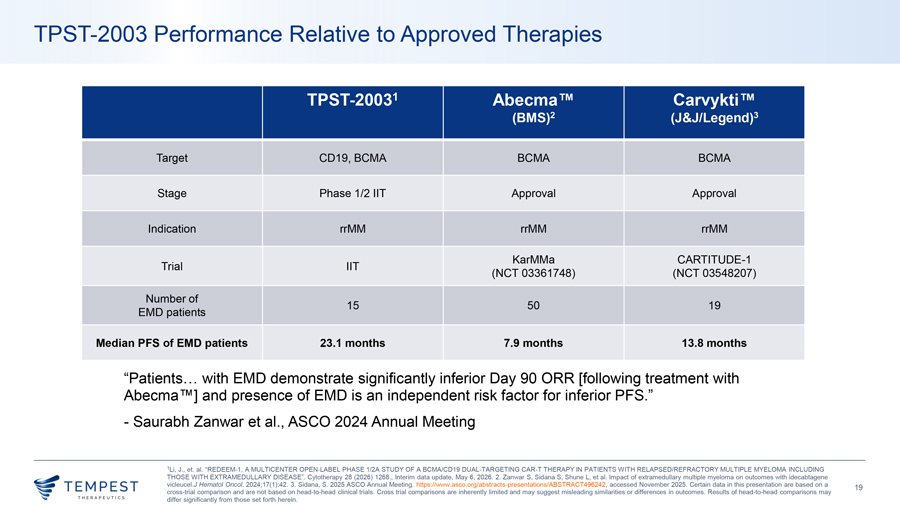
TPST-2003 Performance Relative to Approved Therapies TPST-20031 Abecma Carvykti (BMS)2 (J&J/Legend)3 Target CD19, BCMA BCMA BCMA Stage Phase 1/2 IIT Approval Approval Indication rrMM rrMM rrMM KarMMa CARTITUDE-1 Trial IIT (NCT 03361748) (NCT 03548207) Number of 15 50 19 EMD patients Median PFS of EMD patients 23.1 months 7.9 months 13.8 months “Patients… with EMD demonstrate significantly inferior Day 90 ORR [following treatment with Abecma ] and presence of EMD is an independent risk factor for inferior PFS.” - Saurabh Zanwar et al., ASCO 2024 Annual Meeting 1Li, J., et. al. “REDEEM-1, A MULTICENTER OPEN-LABEL PHASE 1/2A STUDY OF A BCMA/CD19 DUAL-TARGETING CAR-T THERAPY IN PATIENTS WITH RELAPSED/REFRACTORY MULTIPLE MYELOMA INCLUDING THOSE WITH EXTRAMEDULLARY DISEASE”. Cytotherapy 28 (2026) 1268., Interim data update, May 6, 2026. 2. Zanwar S, Sidana S, Shune L, et al. Impact of extramedullary multiple myeloma on outcomes with idecabtagene vicleucel.J Hematol Oncol. 2024;17(1):42. 3. Sidana, S. 2025 ASCO Annual Meeting. https://www.asco.org/abstracts-presentations/ABSTRACT496242, accessed November 2025. Certain data in this presentation are based on a 19 cross-trial comparison and are not based on head-to-head clinical trials. Cross trial comparisons are inherently limited and may suggest misleading similarities or differences in outcomes. Results of head-to-head comparisons may differ significantly from those set forth herein.

Small Molecule Programs Amezalpat (TPST-1120) First-in-Class PPAR�� Antagonist TPST-1495 First-in-Class Dual EP2/4 Antagonist



® Developing Advanced Therapies for Cancer Patients May 6, 2026