
Exhibit 99.2

Courageous Innovation Dedicated to Bringing Game - Changing Gene Therapies to Market and Working Even Harder to Provide Access to Patients Globally

2 This presentation contains forward - looking statements within the meaning of The Private Securities Litigation Reform Act of 1995 , including, but not limited to, strategy, business plans and objectives for Ocugen’s clinical programs, plans and timelines fo r t he preclinical and clinical development of Ocugen’s product candidates, including the therapeutic potential, clinical benefits and potential sa fety thereof, expectations regarding timing, success and data announcements of current ongoing preclinical and clinical trials, th e a bility to initiate new clinical programs, statements regarding qualitative assessments of available data, potential benefits, expectati ons for ongoing clinical trials, anticipated regulatory filings and anticipated development timelines, which are subject to risks and un certainties. We may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “estimates,” “anticipat es, ” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” or other words that convey uncertainty of future ev ent s or outcomes to identify these forward - looking statements. Such statements are subject to numerous important factors, risks, and uncertainties that may cause actual events or results to differ materially from our current expectations, including, but not lim ited to, the risks that preliminary, interim and top - line clinical trial results may not be indicative of, and may differ from, final clinica l data; that unfavorable new clinical trial data may emerge in ongoing clinical trials or through further analyses of existing clinical tr ial data; that earlier non - clinical and clinical data and testing of may not be predictive of the results or success of later clinical trials; and that that clinical trial data are subject to differing interpretations and assessments, including by regulatory authorities. These and other risks and uncertainties are more fully described in our annual and periodic filings with the Securities and E xch ange Commission (SEC), including the risk factors described in the section entitled “Risk Factors” in the quarterly and annual rep ort s that we file with the SEC. Any forward - looking statements that we make in this presentation speak only as of the date of this presentati on. Except as required by law, we assume no obligation to update forward - looking statements contained in this presentation whether a s a result of new information, future events, or otherwise, after the date of this presentation. Forward Looking Statement Ocugen - First Quarter 2026 Business Update

3 • Announced offering of $115 million aggregate principal amount ( $130 million if overallotment is exercised) of 6.75% Convertible Senior Notes due 2034 in a private offering to qualified institutional buyers • $32.7 million of net proceeds from the recently announced Convertible Senior Notes to retire the Avenue debt (12.5% interest rate) • Ocugen expects to use the remaining net proceeds from the offering for general corporate purposes, which is expected to extend cash runway into 2028 Financing Private Offering of $115 Million of Convertible Senior Notes Ocugen - First Quarter 2026 Business Update

4 Targeting Three Biologics License Applications (BLAs) in Three Years Pipeline Phase I Phase II Phase III Target BLA/ MAA * Submission Program 300, 000 (U.S. + EU) Gene - agnostic targeting >100 genes, broad indication ABCA4 - associated retinopathies caused by 1,200+ mutations Advanced dry age - related macular degeneration ( d AMD ) Phase 3 in Progress (Largest Orphan Gene Therapy Clinical Trial) Phase 2/3 Pivotal Confirmatory Clinical Trial in Progress Plan to Initiate Phase 3 in 2026 2026 2027 2028 100, 000 (U.S. + EU) 2 - 3 million (U.S. + EU) Retinitis Pigmentosa Stargardt Disease Geographic Atrophy OCU400 OCU410ST OCU410 Designations: RMAT 1 , ODD 2 , OMPD 3 & ATMP 4 Designation: ODD, RPDD 5 & OMPD Designation: ATMP 1 Regenerative Medicine Advanced Therapy (RMAT); 2 Orphan Drug Designation (ODD); 3 Orphan Medicinal Product Designation (OMPD); 4 Advance Therapy Medicinal Products (ATMP); 5 Rare Paediatric Disease Designation (RPDD); Rolling Submission * Market Authorization Application will follow BLA submission Phase 3 Enrollment Completed Phase 2/3 Enrollment Completed Ocugen - First Quarter 2026 Business Update

Breakthrough technology designed to address many rare diseases as well as complex diseases that affect millions Modifier Gene Therapy Platform

OCU400 Retinitis Pigmentosa (RP) Broad indication, gene - agnostic, targets 100+ genes
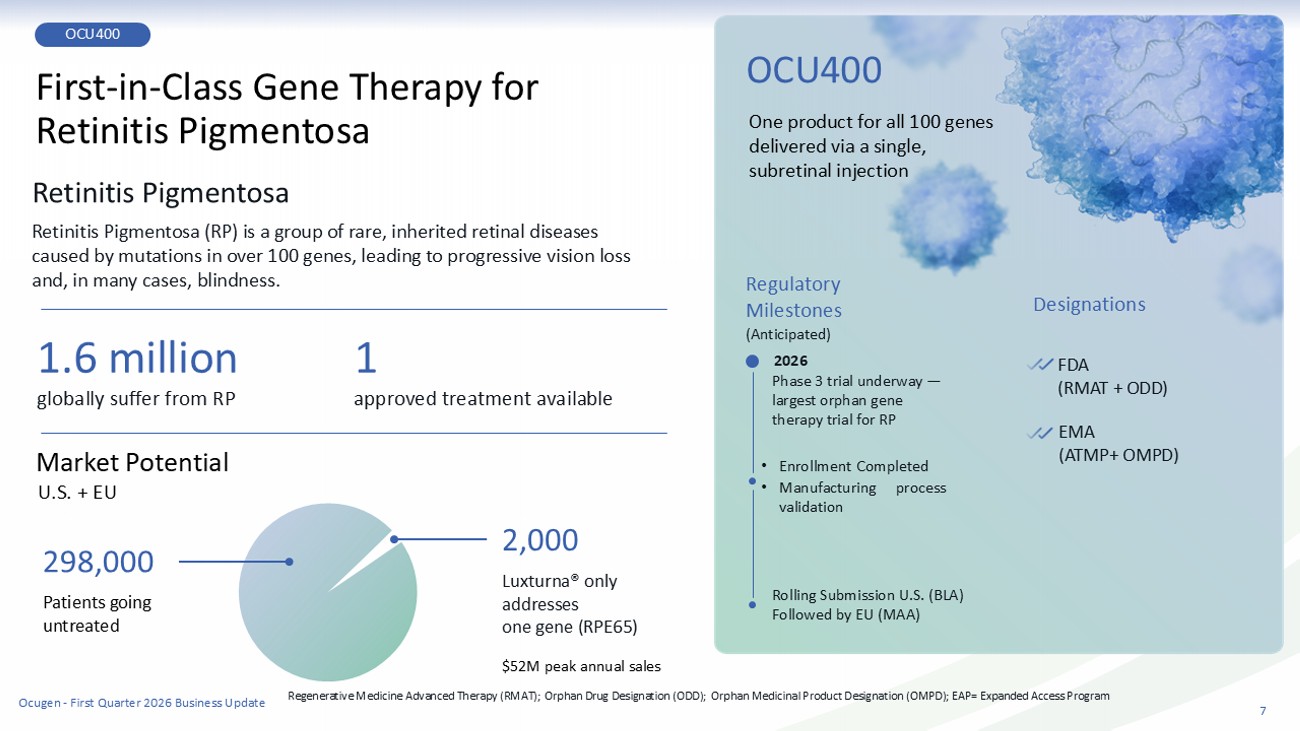
7 OCU400 First - in - Class Gene Therapy for Retinitis Pigmentosa Retinitis Pigmentosa Retinitis Pigmentosa (RP) is a group of rare, inherited retinal diseases caused by mutations in over 100 genes, leading to progressive vision loss and, in many cases , blindness. 1.6 million 2,000 1 Market Potential U.S. + EU approved treatment ava ila ble globally suffer from RP $52 M peak annual sales Luxturna® only addresses one gene (RPE65) Regulatory Milestone s (Anticipated) One product for all 100 genes delivered via a single, subretinal injection Designations OCU400 Phase 3 trial underway — largest orphan gene therapy trial for RP • Enrollment Completed • Manufacturing process validation Rolling Submission U.S. (BLA) Followed by EU (MAA) FDA (RMAT + ODD) EMA (ATMP+ OMPD) Patients going untreated 298,000 Regenerative Medicine Advanced Therapy (RMAT); Orphan Drug Designation (ODD); Orphan Medicinal Product Designation (OMPD); EAP= Expanded Access Program 2026 Ocugen - First Quarter 2026 Business Update
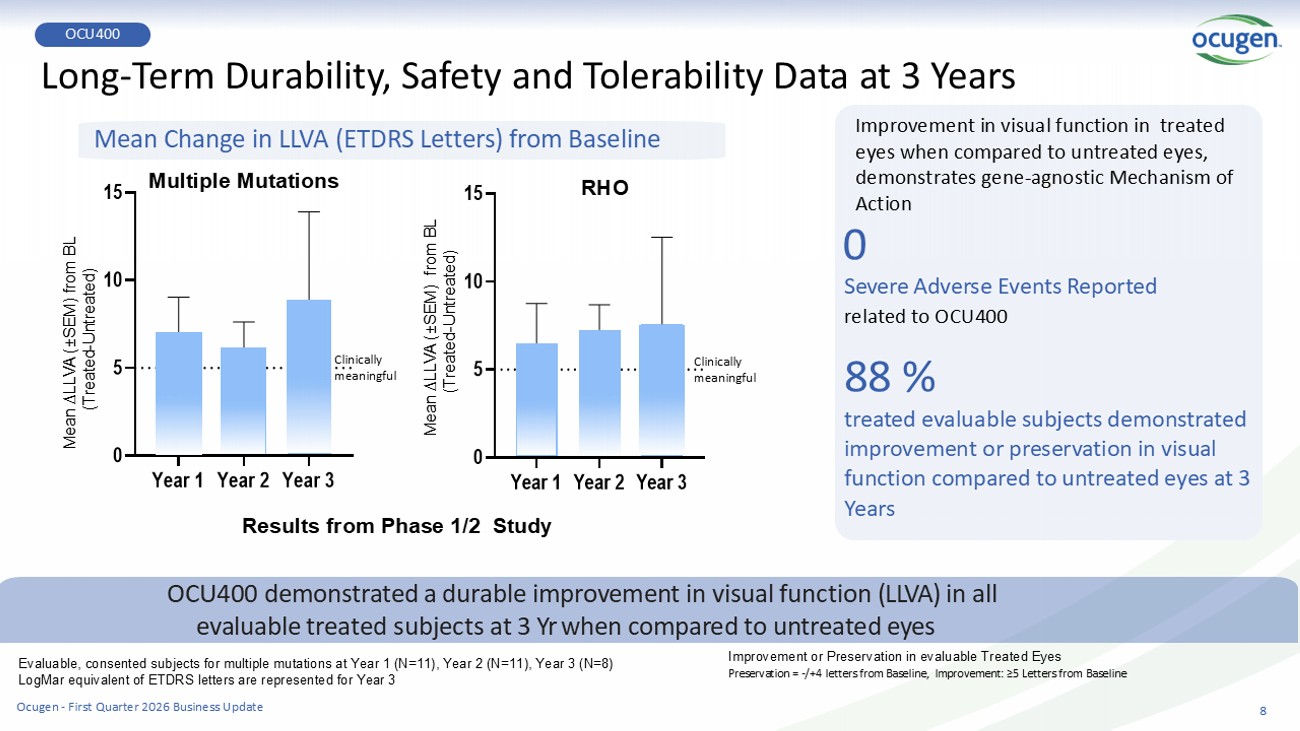
8 Long - Term Durability, Safety and Tolerability Data at 3 Years Year 1 Year 2 Year 3 0 5 10 15 Year 1 Year 2 Year 3 0 5 10 15 OCU400 demonstrated a durable improvement in visual function (LLVA) in all evaluable treated subjects at 3 Yr when compared to untreated eyes Mean Change in LLVA (ETDRS Letters) from Baseline Results from Phase 1/2 Study Improvement in visual function in treated eyes when compared to untreated eyes, demonstrates gene - agnostic Mechanism of Action 0 Severe Adverse Events Reported related to OCU400 88 % treated evalu a ble subjects demonstrated improvement or preservation in visual function compared to untreated eyes at 3 Years Mean ∆LLVA ( “ SEM) from BL (Treated - Untreated) Mean ∆LLVA ( “ SEM) from BL (Treated - Untreated) Multiple Mutations RHO Evaluable, consented subjects for multiple mutations at Year 1 (N=11), Year 2 (N=11), Year 3 (N=8) LogMar equivalent of ETDRS letters are represented for Year 3 Improvement or Preservation in evaluable Treated Eyes Preservation = - /+4 letters from Baseline, Improvement: ≥5 Letters from Baseline Clinically meaningful Clinically meaningful OCU400 Ocugen - First Quarter 2026 Business Update
OCU410ST Stargardt Disease ABCA4 - associated retinopathies >1,200 mutations

OCU410ST First - in - Class Gene Therapy for Stargardt Disease Stargardt Disease A rare IRD associated with 1,200+ mutations of the ABCA4 gene 1 million 0 Market Potential U.S. + EU approved treatments available globally suffer from ABCA4 - associated retinopathies Regulatory Milestone s (Completed/anticipated) for upregulation of networks of key genes improving the cell environment and survival with a single, subretinal injection Designations OCU410ST 2025 Initiated pivotal Phase 2 / 3 2027 Topline Data, BLA submission FDA ( RPD D + ODD ) EMA (ATMP+ OMPD) 100% of Patients going untreated 100,000 Potential Patients Advanced Therapeutic Medicinal Product (ATMP); Orphan Drug Designation (ODD); Orphan Medicinal Product Designation (OMPD) 2026 • Enrollment Completed Interim analysis 10 Ocugen - First Quarter 2026 Business Update

11 GARDian3 - Phase 2/3 Pivotal Confirmatory Trial OCU410ST Endpoints Trial Design Randomized 2:1 (N=51) DSMB 4 - week Data Reviews Functional improvement in vision vs. control eye Assessed by LLVA and BCVA EZ analysis (exploratory) Target Population Early - to late - stage disease population Including pediatrics ( 3 + years) 12 - month change in atrophic lesion size from baseline vs. control Measured in mm 2 by fundus autofluorescence (FAF) 34 Treatment Group 3 п 10 10 vg per eye in 200 µL 17 Control Group No Treatment First Second 17 17 All Subjects M TD Established in Phase 1 Primary Secondary Adaptive design with sample size re - estimation [3Q2026] Interim Analysis: 24 subjects complete 8 months post - OCU410ST (16 treated and 8 controls) DMC Interim - Data Analysis Ocugen - First Quarter 2026 Business Update

OCU410 Geographic Atrophy Advanced dry age - related macular degeneration ( dAMD )

13 OCU410 First - in - Class Gene Therapy for GA Patients Geographic Atrophy Geographic Atrophy (GA) is an advanced form of dry AMD. GA causes irreversible degeneration of retina cells in the macula, leading to loss of central vision. ~8 million 2 Market Size U.S. + EU approved treatments available that address only 1 of the 4 pathways involved in disease progression globally suffer from advanced dAMD Regulatory Milestone s (Anticipated) Designed to address all four pathways associated with GA without 6 - 12 injections per year and related side effects Designation s OCU410 2026 Initiate Phase 3 2028 Topline data, BLA submission EMA (ATMP) SYFO V RE ® and IZERVAY ® > $ 1 B combined annual sales 2 - 3M Patients Recent Milestone Positive 12 - month Phase 2 data Approved Products in US 2026 Phase 2 Topline data released 2027 Complete enrollment Ocugen - First Quarter 2026 Business Update

14 1 Akula et al. Gene Ther 2024; MOA= Mechanism of Action: anti - drusen activity (improves retinal function), anti - inflammatory (suppresses inflammation in HMC3 cells), anti - oxidative (improves ARPE19 cell survival), anti - complement (increases Cd59 protein) OCU410 Aims to Disrupt GA Treatment Driven by a Novel MOA Driving global change at the patient level (2 - 3M patients in U.S. and EU ) GA Patient Experience RORA 4 - way MOA 1 Addresses all disease pathways – marketed therapies only address the complement system OCU410 Ocugen - First Quarter 2026 Business Update
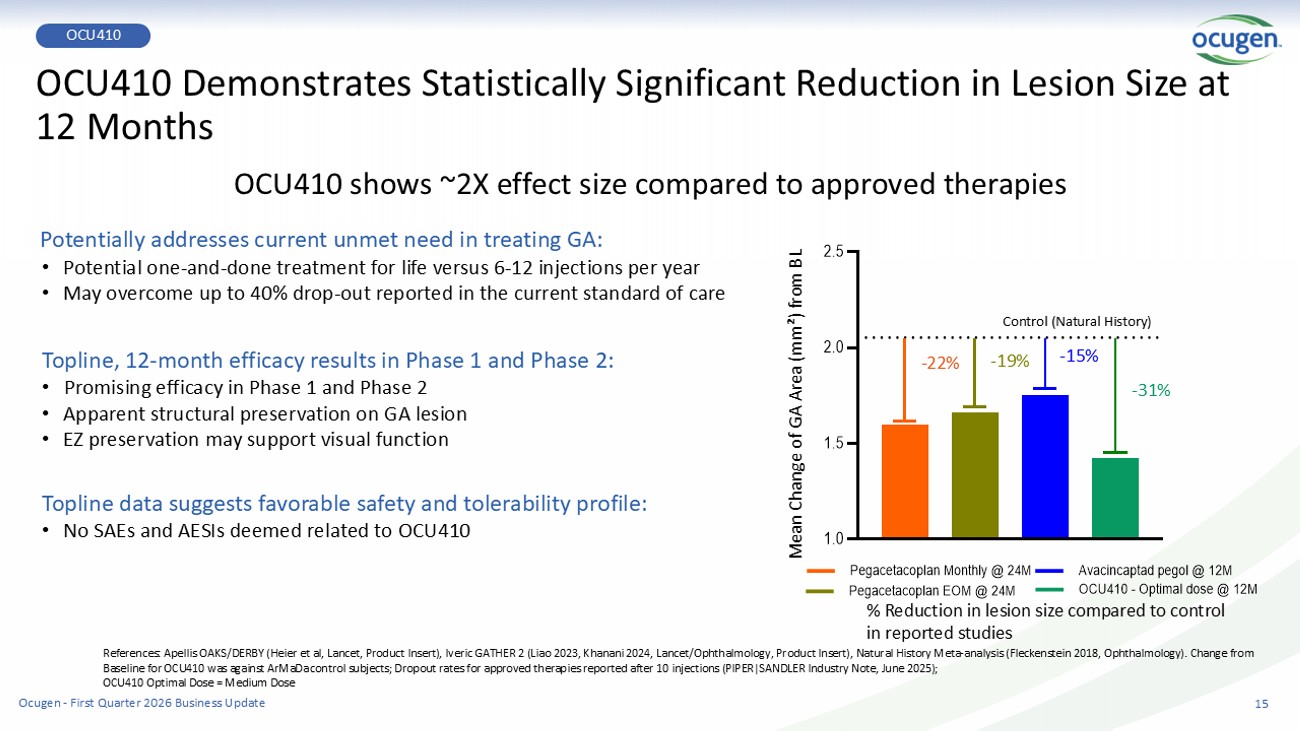
OCU410 Demonstrates Statistically Significant Reduction in Lesion Size at 12 Months OCU410 shows ~2X effect size compared to approved therapies References: Apellis OAKS/DERBY (Heier et al, Lancet, Product Insert), Iveric GATHER 2 (Liao 2023, Khanani 2024, Lancet/Ophthalmology, Product Insert), Natural History Meta - analysis (Fleckenstein 2018, Oph thalmology). Change from Baseline for OCU410 was against ArMaDa control subjects; Dropout rates for approved therapies reported after 10 injections (PIPER|SANDLER Industry Note, June 2025); OCU410 Optimal Dose = Medium Dose Potentially addresses current unmet need in treating GA: • Potential one - and - done treatment for life versus 6 - 12 injections per year • May overcome up to 40% drop - out reported in the current standard of care Topline, 12 - month efficacy results in Phase 1 and Phase 2: • Promising efficacy in Phase 1 and Phase 2 • Apparent structural preservation on GA lesion • EZ preservation may support visual function Topline data suggests favorable safety and tolerability profile: • No SAEs and AESIs deemed related to OCU410 % Reduction in lesion size compared to control in reported studies 1.0 1.5 2.0 2.5 Pegacetacoplan Monthly @ 24M Pegacetacoplan EOM @ 24M Avacincaptad pegol @ 12M OCU410 - Optimal dose @ 12M Control (Natural History) - 22% - 19% - 15% - 31% Mean Change of GA Area (mm²) from BL 15 OCU410 Ocugen - First Quarter 2026 Business Update
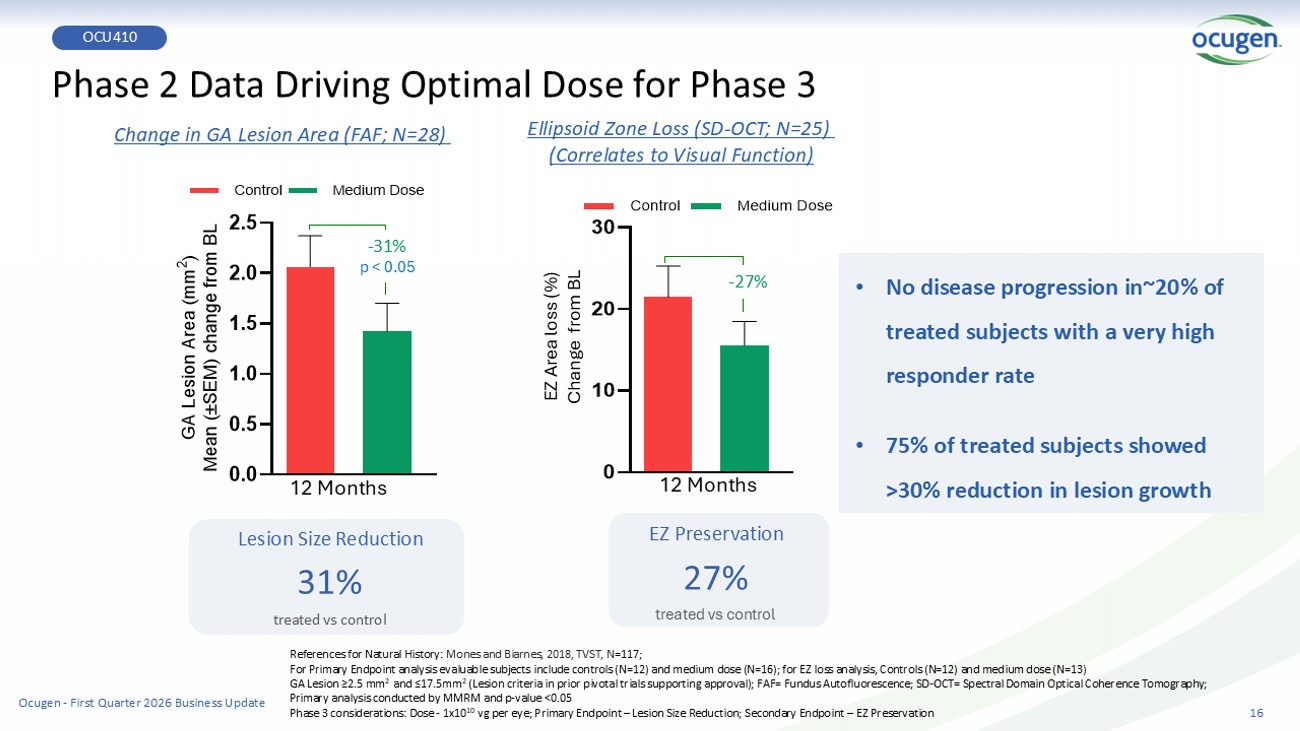
0 10 20 30 Control Medium Dose Lesion Size Reduction 31% treated vs control Phase 2 Data Driving Optimal Dose for Phase 3 References for Natural History: Mones and Biarnes , 2018, TVST, N =117; For Primary Endpoint analysis evaluable subjects include controls (N=12) and medium dose (N=16); for EZ loss analysis, Contro ls (N=12) and medium dose (N=13) GA Lesion ≥2.5 mm 2 and ≤17.5mm 2 (Lesion criteria in prior pivotal trials supporting approval); FAF= Fundus Autofluorescence; SD - OCT= Spectral Domain Optical Co herence Tomography; P rimary analysis conducted by MMRM and p - value <0.05 Phase 3 considerations: Dose - 1x10 10 vg per eye; Primary Endpoint – Lesion Size Reduction; Secondary Endpoint – EZ Preservation EZ Preservation 27% treated vs control 0.0 0.5 1.0 1.5 2.0 2.5 G A L e s i o n A r e a ( m m 2 ) M e a n ( ± S E M ) c h a n g e f r o m B L Control Medium Dose - 31% Ellipsoid Zone Loss (SD - OCT; N=25) (Correlates to Visual F unction) - 27% Change in GA Lesion Area (FAF; N=28) EZ Area loss (%) Change from BL p < 0.05 16 • No disease progression in~20% of treated subjects with a very high responder rate • 75% of treated subjects showed >30% reduction in lesion growth OCU410 12 Months 12 Months Ocugen - First Quarter 2026 Business Update
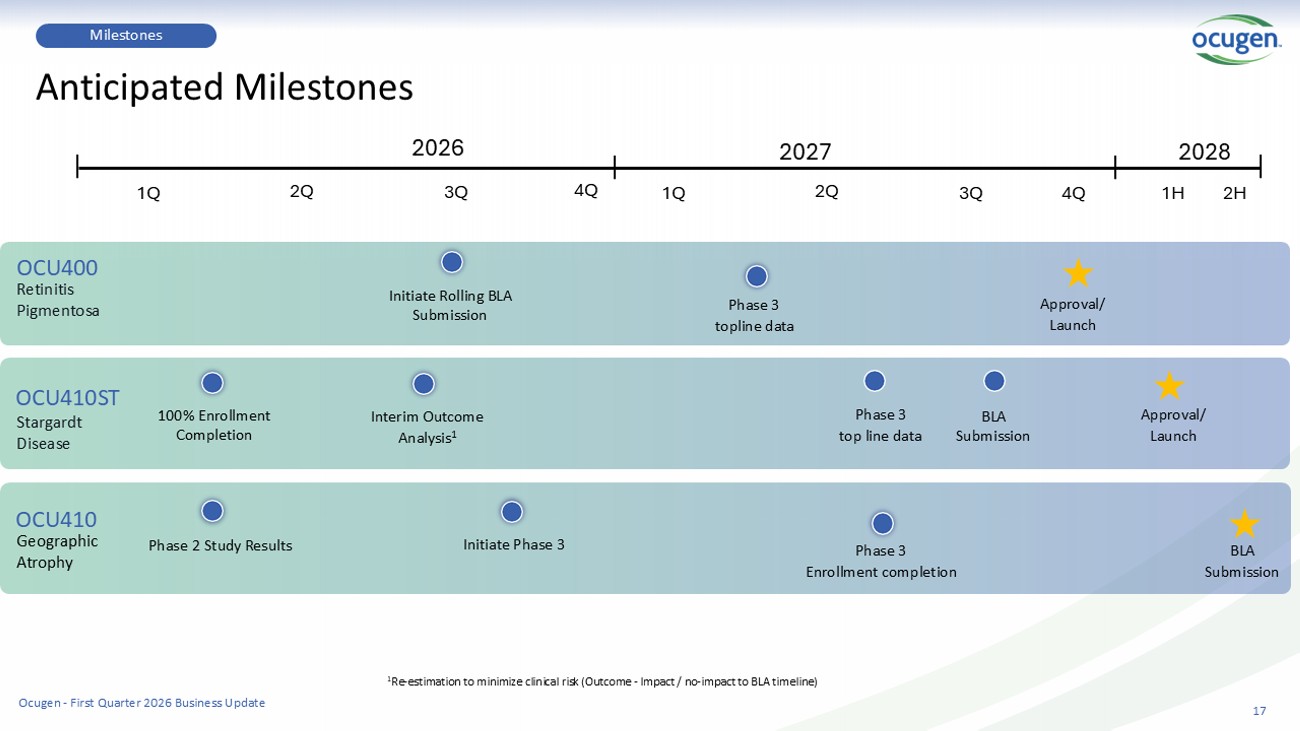
3Q 17 Milestones Anticipated Milestones Retinitis Pigmentosa Stargardt Disease Geographic Atrophy OCU400 OCU410ST OCU410 100% Enrollment Completion Interim Outcome Analysis 1 Initiate Rolling BLA Submission Phase 3 topline data Initiate Phase 3 Phase 2 Study Results 2026 2027 1Q 2Q 3Q 4Q 1Q 2Q Phase 3 top line data BLA Submission Approval/ Launch Approval/ Launch BLA Submission 4Q 1H 2H 2028 Phase 3 Enrollment completion 1 Re - estimation to minimize clinical risk (Outcome - Impact / no - impact to BLA timeline) Ocugen - First Quarter 2026 Business Update
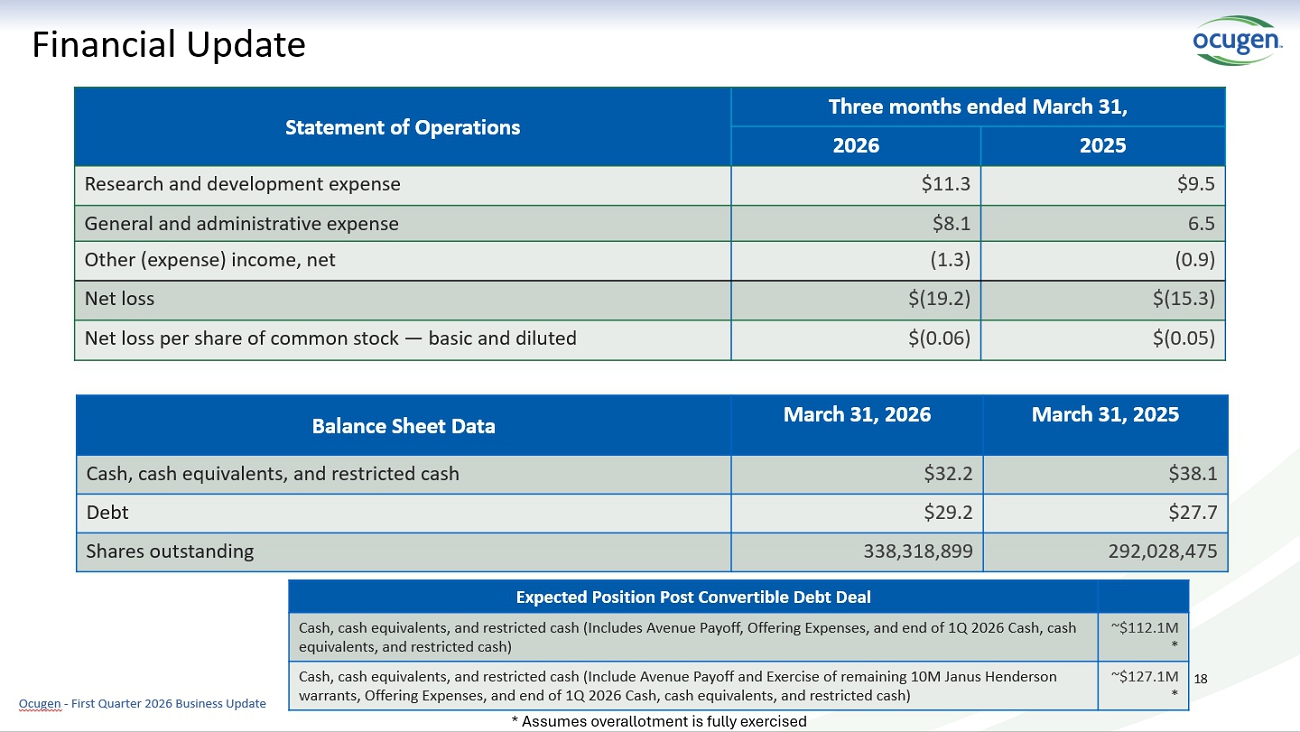
Financial Update Three months ended March 31, Statement of Operations 2025 2026 $9.5 $11.3 Research and development expense 6.5 $8.1 General and administrative expense (0.9) (1.3) Other (expense) income, net $(15.3) $(19.2) Net loss $(0.05) $(0.06) Net loss per share of common stock — basic and diluted March 31, 2025 March 31, 2026 Balance Sheet Data $38.1 $32.2 Cash, cash equivalents, and restricted cash $27.7 $29.2 Debt 292,028,475 338,318,899 Shares outstanding 18 Ocugen - First Quarter 2026 Business Update Expected Position Post Convertible Debt Deal ~$112.1M * Cash, cash equivalents, and restricted cash (Includes Avenue Payoff, Offering Expenses, and end of 1Q 20256 Cash, cash equivalents, and restricted cash) ~$127.1M * Cash, cash equivalents, and restricted cash (Include Avenue Payoff and Exercise of remaining 10M Janus Henderson warrants, Offering Expenses, and end of 1Q 2026 Cash, cash equivalents, and restricted cash) * Assumes overallotment is fully exercised

Advancing cures for blindness IR @ocugen.com

Q&A