Exhibit 10.1
CERTAIN CONFIDENTIAL PORTIONS OF THIS EXHIBIT HAVE BEEN OMITTED AND REPLACED WITH “[***]”. SUCH IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS (I) NOT MATERIAL AND (II) THE TYPE THAT THE REGISTRANT CUSTOMARILY AND ACTUALLY TREATS AS PRIVATE AND CONFIDENTIAL.
LICENSE AND COLLABORATION AGREEMENT
This License and Collaboration Agreement (this “Agreement”), dated January 11, 2026 (the “Effective Date”), is entered into by and between: Nuvation Bio Inc., a Delaware corporation having its office and place of business at 1500 Broadway, Suite 1401, New York, NY 10036, USA (“Nuvation”); and Eisai Co., Ltd., a Japanese corporation having its office and place of business at 4-6-10 Koishikawa, Bunkyo-ku, Tokyo 112-8088, Japan (“Eisai”). Nuvation and Eisai are each referred to herein by name, individually as a “Party”, or collectively as the “Parties”.
RECITALS:
Whereas, Nuvation owns or controls certain rights to the Licensed Product (hereinafter defined);
Whereas, Eisai desires to exploit the Licensed Product in the Field in the Territory (each as hereinafter defined); and
Whereas, Nuvation desires to grant to Eisai, and Eisai desires to obtain from Nuvation, an exclusive license under the Licensed IP (hereinafter defined) to exploit the Licensed Product in the Field in the Territory on the terms and conditions set forth in this Agreement.
Now, Therefore, in consideration of the foregoing recitals and the mutual agreements and covenants set forth herein, the Parties agree as follows:
As used in this Agreement, each capitalized term used herein shall have the meaning set forth below unless context clearly and unambiguously dictates otherwise.
1.1“AB-106-G319 Study” means the clinical study for the Licensed Compound titled: “A Phase 3 Multicenter Double-blind Randomized Study of Taletrectinib versus Placebo in Patients with ROS1-Fusion Positive Stage IB-IIIA Non-Small Cell Lung Cancer Who Have Undergone Complete Tumor Resection”.
1
Page of SECTIONPAGES \* MERGEFORMAT 55
1.2“AB-106-G319 Study IP” has the meaning set forth in Section 4.5.2.
1.3“Abandoned Licensed Patent” has the meaning set forth in Section 8.2.1(c).
1.4“Affiliate” means, with respect to a legal person or entity, any other legal person or entity that controls, is controlled by or is under common control with such legal person or entity, for so long as such control exists. For purposes of this definition only, “control” or “Control” shall mean: (a) beneficial ownership (direct or indirect) of more than fifty percent (50%) [***]; or (b) the de facto ability to control or direct the management of such person or entity.
1.5“Anti-Bribery Requirement” means any Applicable Law, the principal purpose of which is to address prohibitions on bribery or other unlawful payments in the Territory, including the Foreign Corrupt Practices Act in the US and all equivalent laws in any other applicable jurisdiction.
1.6“Applicable Law” means any law, statute, ordinance, code (including applicable pharma codes of practice such as the PhRMA Code on Interactions with Healthcare Professionals and the ABPI Code of Practice for the Pharmaceutical Industry), rule or regulation that has been enacted by a Governmental Authority (including, without limitation, any Regulatory Authority) and is in force as of the Effective Date or comes into force during the Term, in each case to the extent that the same is applicable to the performance by the Parties of their respective obligations under this Agreement.
1.7“Arbitration Rules” has the meaning set forth in Section 13.2.1.
1.8“Business Day” means any day other than a Saturday, a Sunday, or a day on which commercial banks located in New York, USA or Tokyo, Japan are authorized or required by law to remain closed.
1.9“Calendar Quarter” means each period of three (3) consecutive calendar months, ending March 31, June 30, September 30, and December 31, respectively.
1.10“Calendar Year” means the period of time beginning on January 1 and ending December 31, except for the first year which shall begin on the Effective Date and end on December 31 of the Calendar Year in which the Effective Date is encompassed and the last year of the Term shall begin on January 1 of the last Calendar Year of the Term and end on the last day of the Term.
1.11“CDMO” means a contract development and manufacture organization, which is a company that provides support to the pharmaceutical, biotechnology, and medical device industries in the form of development and manufacturing services outsourced on a contract basis.
1.12“Clinical Subcommittee” has the meaning set forth in Section 3.7.
1.13“Clinical Trial” means any clinical testing of a Licensed Product in human subjects (including the New Indication Study, and AB-106-G319 Study), in accordance with GCP, where applicable, and designed to generate data in support or maintenance of a Marketing Approval
2
Page of SECTIONPAGES \* MERGEFORMAT 55
application (including data relating to the safety, tolerability, pharmacological activity, pharmacokinetics, dose ranging or efficacy of such Licensed Product).
1.16“Commercially Reasonable Efforts” means, [***].
1.17“Commercialization” or “Commercialize” means any and all activities associated with using, marketing, promoting, distributing, offering for sale, selling, having sold, and importing Licensed Product(s) and shall include activities (a) related to the submission of filings with an applicable Reimbursement Authority to seek and obtain Pricing Approval; or (b) required to fulfill ongoing regulatory obligations after receipt of a Marketing Approval.
1.18“Commercial Supply Agreement” has the meaning set forth in Section 6.4.
1.19“Competing Product” means [***].
1.20“Conditional Marketing Approval” means Marketing Approval of a Licensed Product by the European Commission on the basis of an application made to the EMA pursuant to Article 14-a of Regulation (EC) No. 726/2004 or a successor regulation thereto.
1.21“Confidential Information” has the meaning set forth in Section 9.1.
1.22“Control” means, with respect to an item of information or Intellectual Property Rights, possession of the ability (whether by license or ownership, or through an Affiliate having possession by license or ownership) to disclose and grant a license or sublicense under such information and Intellectual Property Rights within the scope set forth in this Agreement, without violating the terms of any written agreement with any Third Party. “Controlled” has the corelative meaning.
1.23“Cover”, “Covering” or “Covered” means, with respect to a Patent, that, but for ownership by or a license granted to a Party under a Valid Claim included in such Patent, the practice by such Party of an invention claimed in such Patent would infringe such Valid Claim (or in the case of a Patent that is a patent application, would infringe a Valid Claim in such patent application if it were to issue as a patent).
1.24“CRO” means a contract research organization, which is a company that provides support to the pharmaceutical, biotechnology, and medical device industries in the form of research services outsourced on a contract basis.
1.25“Daiichi Sankyo” means Daiichi Sankyo Company, Limited.
1.26“Development” or “Develop” means any and all non-clinical, pre-clinical, clinical and regulatory activities with respect to the Licensed Compound, the Licensed Product(s) or companion diagnostics therefor, including (a) Clinical Trials (including investigator-initiated trials and Clinical Trials conducted after Marketing Approval), and (b) such activities relating to the
3
Page of SECTIONPAGES \* MERGEFORMAT 55
submission of filings with applicable Regulatory Authorities to seek, obtain and maintain a Marketing Approval (except Pricing Approvals). For clarity, Development excludes Commercialization.
1.27“Distributor” means any Third Party that purchases Licensed Product in final form (or in a form suitable solely to package or label such Licensed Product purchased in bulk form) from Eisai or its Affiliates or Sublicensees for resale, in circumstances where such Third Party is appointed by Eisai or its Affiliate or Sublicensee as a distributor to distribute, market or resell such Licensed Product in one or more countries in the Territory and such Third Party takes title to such Licensed Product and pays Eisai or its Affiliate or Sublicensee a purchase price for the Licensed Product for resale in such country(ies) (which may include a discount from any official list or reimbursed price to cover the Third Party’s services fee or margin) but such Third Party does not make any subsequent or additional royalty, profit-share, revenue-share, or milestone based payments (excluding any subsequent reconciliation process for the purchase price) to Eisai or any of its Affiliates or Sublicensees with respect to its resale of such Licensed Product, including any such Third Party that is granted ancillary rights to further Develop, package or obtain Marketing Approvals of such Licensed Product in such country(ies) solely to distribute, market or sell such Licensed Product in such country(ies) without any royalty, profit-share, revenue-share, or milestone based payments (excluding any subsequent reconciliation process for the purchase price) to Eisai, its Affiliate or Sublicensees (beyond the purchase price of Licensed Product for resale in such country(ies)) for the grant of such rights. For the avoidance of doubt, any wholesaler or reseller of a Licensed Product is a “Distributor” if such entity meets the requirements of this definition.
1.28“DMF” means a Drug Master File as more fully defined in 21 C.F.R. § 314.420 in the United States or similar documents filed with a Regulatory Authority in another jurisdiction.
1.29“DS License Agreement” means that certain Third Party License by and between Nuvation and Daiichi Sankyo, dated as of December 7, 2018 and any amendment thereof. For clarity, the DS License Agreement was originally entered into by and between AnHeart Therapeutics LLC (formerly known as AnHeart Therapeutics Inc.) and Daiichi Sankyo on December 7, 2018 and was assigned by AnHeart Therapeutics LLC to Nuvation pursuant to that certain Assignment and Assumption Agreement between AnHeart Therapeutics LLC and Nuvation, dated July 1, 2024.
1.30“Effective Date” has the meaning set forth in the preamble.
1.31“Eisai” has the meaning set forth in the preamble.
1.32“Eisai IP” means the Eisai Patents, the Eisai Know-How, and Eisai’s interest in the Joint IP.
1.33“Eisai Know-How” means Know-How that is related to the Licensed Compound or the Licensed Product(s), including any Invention, and conceived, discovered, developed, made or reduced to practice by or on behalf of employees, agents, or consultants of Eisai, its Affiliates or its or their Sublicensees during the Term.
4
Page of SECTIONPAGES \* MERGEFORMAT 55
1.34“Eisai Patents” means all Patents conceived, discovered, developed, made or reduced to practice by or on behalf of employees, agents, or consultants of Eisai, its Affiliates or Sublicensees in connection with the performance of its obligations and exercise of its rights under this Agreement during the Term that (a) include a Valid Claim Covering an Invention; or (b) otherwise Cover the manufacturing, using, selling, offering for sale, or importing of the Licensed Product(s).
1.35“Eisai Related Parties” has the meaning set forth in Section 12.6.5.
1.36“EMA” means the European Medicines Agency or any successor agency thereto.
1.37“Europe” means (a) the economic, scientific and political organization of member states of the European Union as it may be constituted from time to time, which as of the Effective Date consists of Austria, Belgium, Bulgaria, Croatia, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Luxembourg, Malta, The Netherlands, Poland, Portugal, Romania, Slovakia, Slovenia, Spain, Sweden and that certain portion of Cyprus included in such organization (the “European Union”), (b) the United Kingdom of Great Britain and Northern Ireland, (c) any member country of the European Economic Area that is not otherwise a member of the European Union, (d) Albania, Andorra, Armenia, Azerbaijan, Belarus, Bosnia and Herzegovina, Georgia, Kosovo, Moldova, Monaco, Montenegro, North Macedonia, San Marino, Serbia, Switzerland, Ukraine, Vatican City, and (e) any country not otherwise included in clauses (a), (b), (c), or (d) that participates in the unified filing system under the auspices of the EMA.
1.38“European Commission” means the European Commission or any successor entity that is responsible for granting Marketing Approvals authorizing the sale of pharmaceuticals in the European Union.
1.39“European Economic Area” means the European Economic Area as its membership may be constituted from time to time, and any successor thereto, and which, as of the Effective Date, is composed of the members of the European Union together with Iceland, Liechtenstein and Norway.
1.40“Executive Officer” means (a) with respect to Eisai, Eisai’s Chairman & Chief Executive Officer, EMEA or their designee, and (b) with respect to Nuvation, Nuvation’s Chief Financial Officer or their designee.
1.41“Extending” means taking any actions to extend the term of a Patent, including preparing and filing requests for patent term extensions, supplemental protection certificates, pediatric exclusivity or similar protections. “Extend” and “Extension” have correlative meaning.
1.42“Field” means all human therapeutic uses of the Licensed Compound that derive therapeutic effect from binding to or modulating (a) the ROS proto-oncogene 1 (“ROS1”), (b) the neurotrophic tyrosine receptor kinase (“NTRK”), or (c) anaplastic lymphoma kinase pathways, in each case (a) through (c) for the diagnosis, prevention, or treatment of any indication.
1.43“First Commercial Sale” means on a country-by-country basis the date on which the Licensed Product is first sold by Eisai, its Affiliate or Sublicensees to Third Parties for use or
5
Page of SECTIONPAGES \* MERGEFORMAT 55
consumption by end users in the applicable country of the Territory after all required Marketing Approvals have been granted, or such sale is otherwise permitted, by the relevant Regulatory Authority in the applicable country of the Territory, excluding (a) any sale to an Affiliate or Sublicensee, unless such Affiliate or Sublicensee is the last person or entity in the distribution chain of the Licensed Product; or (b) any use of such Licensed Product in Clinical Trials or non-clinical development activities with respect to such Licensed Product by or on behalf of a Party, or disposal or transfer of such Licensed Product for a bona fide charitable purpose, compassionate use, or samples if no monetary consideration is received for such use or transfer.
1.44“Fiscal Year” means each twelve-month period from 1 April and ending on 31 March in the following Calendar Year.
1.45“GCP” means the applicable standards for clinical trials for pharmaceuticals, as set forth in the ICH guidelines and applicable regulations promulgated thereunder, as amended from time to time, and such standards of good clinical practice as are required by the FDA, European Union and other relevant organizations and governmental agencies in countries in which a Licensed Product is intended to be sold to the extent such standards are not less stringent than the ICH guidelines.
1.47“GLP” means the applicable then-current standards for laboratory activities for pharmaceuticals or biologicals, as set forth in 21 C.F.R. part 58, as amended from time to time, or guidance documents promulgated thereunder, together with any similar standards of good laboratory practice as are required by any Regulatory Authority in any applicable jurisdiction.
1.48“GMP” means the applicable then-current quality assurance standards that ensure that pharmaceutical products are consistently produced and controlled in accordance with the quality standards appropriate to their intended use as defined in 21 C.F.R. § 210 and 211, European Directive 2017/1572 supplementing Directive 2001/83/EC, EU GMP, Annex 16, Japanese drug manufacturing regulations, ICH guidelines, and other applicable guidance or equivalent laws in other jurisdictions.
1.49“Governmental Authority” means any agency, department, authority, body, institution or other instrumentality of any national, state, country, city or other political subdivision.
1.50“GxP” means, collectively, all applicable good practice quality guidelines and regulations, encompassing such internationally recognized standards as GMP, Good Clinical Practice (GCP), GLP, Good Distribution Practice (GDP), Good Quality Practice (GQP), Good Vigilance Practice (GVP), Good Post-Marketing Study Practice (GPSP), Good Review Practice (GRP), and any other applicable practice, in each case (a) as such terms are defined from time to time by the FDA or other applicable Governmental Authorities pursuant to its regulations, guidelines or otherwise and (b) applicable from time to time to the Development, Manufacturing, Commercialization, importing or other exploitation of the Licensed Compound or Licensed Product(s) or any intermediate thereof pursuant to Applicable Law.
6
Page of SECTIONPAGES \* MERGEFORMAT 55
1.51“Indemnify” has the meaning set forth in Section 11.1.
1.52“Initial Indication” means locally advanced or metastatic ROS1 positive non-small cell lung cancer with a first-line or a line-agnostic label.
1.53“Intellectual Property Rights” means rights to Know-How, Patents, copyright and related rights, moral rights, trade marks, domain names, rights in get up and trade dress, goodwill and the right to sue for passing off or unfair competition, database rights, rights in designs and all other intellectual property rights in each case whether registered or unregistered and including all applications and rights to apply for and be granted, renewals or extensions of, and rights to claim priority from, such rights and all similar or equivalent rights or forms of protection which subsist or shall subsist now or in the future in any part of the world.
1.54“Invention” means (a) any new or useful process, machine, manufacture, or composition of matter that is (i) related to the Licensed Compound or the Licensed Product(s), or (ii) otherwise necessary for Commercialization of the Licensed Compound or the Licensed Product(s), and (b) any improvement, enhancement, modification, or derivative work to any Licensed IP that is conceived or first reduced to practice or first demonstrated to have utility during the Term in connection with either Parties’ activities.
1.55“Joint IP” has the meaning set forth in Section 8.1.
1.56“Joint Patents” has the meaning set forth in Section 8.1.
1.57“Know-How” means confidential or proprietary technical or business information, including (a) any proprietary scientific or technical information, results and data of any type whatsoever, in any tangible or intangible form whatsoever, including databases, safety information, practices, methods, techniques, specifications, instructions, processes, formulations, formulae, methods, protocols, expertise, knowledge, know-how, inventions, discoveries, improvements, trade secrets, skill, experience, test data including pharmacological, pharmaceutical, physical, medicinal chemistry, biological, chemical, biochemical, toxicological and clinical test data (including patient report forms, preliminary and final investigators’ reports, statistical analyses, expert opinions and reports, safety and other electronic databases, Regulatory Filings and communications, and the like), analytical and quality control data, stability data, studies and procedures, and manufacturing process and development information and descriptions, results and data and (b) any proprietary biological, chemical or physical materials, that exists as of the Effective Date or is discovered, developed or acquired during the Term, and whether patentable or unpatentable. Know-How does not include any Patents.
1.58“Licensed Compound” means the compound claimed in the Nuvation Patent(s) set forth in Schedule 1.58, which is identified by the internal Nuvation compound code AB-106 or INN name taletrectinib with the molecular structure set forth in Schedule 1.58, and any salts, hydrates, solvates, esters, and stereoisomers of such compound.
1.59“Licensed IP” means the Nuvation Patents, the Nuvation Know-How, the Other Nuvation IP, and Nuvation’s interest in the Joint IP.
1.60“Licensed Patents” means the Nuvation Patents and the Joint Patents.
7
Page of SECTIONPAGES \* MERGEFORMAT 55
1.61“Licensed Product” means any product, in any formulation, dosage form, presentation or mode of administration, that constitutes, incorporates, comprises or contains the Licensed Compound.
1.62“MAH” means the holder of a Marketing Approval.
1.63“Manufacturing” or “Manufacture” has the meaning set forth in Section 2.1.2.
1.65“Marketing Approval” means, with respect to a Licensed Product or companion diagnostic and a country in the Territory, all approvals, licenses, registrations, or authorizations of the applicable Regulatory Authority or Reimbursement Authority that are necessary for the manufacturing, use, storage, import, transport, sale, offer for sale and other exploitation of such Licensed Product or companion diagnostic in such country in the Territory, including, if applicable, Pricing Approvals, any accelerated or conditional approvals and authorizations necessary for the sale of the Licensed Product.
1.66“Material Commercial Issue” has the meaning set forth in Section 6.3.
1.67“Material Commercial Issue Notice” has the meaning set forth in Section 6.3.
1.68“Mediation Rules” has the meaning set forth in Section 13.3.
1.69“Medical Affairs Activities” means non-promotional activities conducted by a medical affairs function directed to health care practitioners, such as physicians, pharmacists, nurse practitioners, and any other individual or entity that is a health care practitioner (as determined in accordance with the Applicable Law) and health care organizations, to provide each such health care practitioner or healthcare organization with objective, scientific information to educate and inform, whether such information is provided directly, through congresses or venues for peer-to-peer scientific discourse, or otherwise.
1.70“Medical Subcommittee” has the meaning set forth in Section 3.9.
1.71“NDA” means a new drug application filed with the Regulatory Authority in the Territory to obtain permission to administer a Licensed Product to humans.
1.72“Net Sales” means the aggregate gross amounts invoiced for Licensed Product sold by Eisai, its Affiliates or Sublicensees (but not Distributors) (each a “Selling Party”) in finished product form, packaged and labeled for sale in arm’s length transactions to Third Parties (including Distributors) under this Agreement, less the following deductions from such gross amounts:
1.72.1normal and customary trade, cash and other discounts and allowances actually allowed by the Selling Party and taken by the customer;
1.72.2credits, price adjustments or allowances actually granted to the customer for damaged goods, returns or rejections of a Licensed Product;
8
Page of SECTIONPAGES \* MERGEFORMAT 55
1.72.3sales taxes or similar taxes, including duties, customary compulsory payments to governmental authorities or other governmental charges, levies or rebates (for example, Contribution M in France) imposed on the sale of a Licensed Product (including value added taxes or other governmental charges, but excluding any income taxes), to the extent the Selling Party is not otherwise entitled to a credit or a refund for such taxes, duties or payments made;
1.72.4commission, chargeback payments, rebates, fees, and other adjustments, including those granted on price adjustments, billing errors, reimbursements or similar payments granted or given to wholesalers, other distributors or Third Party logistics providers, buying groups, health insurance carriers or other institutions, including those paid in connection with such sales to any governmental entity; and
1.72.5any invoiced freight, shipping, insurance and other transportation charges.
1.72.6Sales between or among Eisai, its Affiliates and Sublicensees (but not Distributors) shall be excluded from the computation of Net Sales if such sales are not intended for end use, but Net Sales will include the subsequent final sales to Third Parties (including Distributors) by Eisai, its Affiliates or Sublicensees (but not Distributors).
1.72.7Net Sales does not include samples or Licensed Product for compassionate use and the like; for the avoidance of doubt, the Licensed Product for compassionate use shall include Licensed Products donated under any patient assistance program.
1.72.8Net Sales, as set forth in this definition, will be calculated from the books and records of the Selling Party by applying the Selling Party’s standard accounting practices, in accordance with generally accepted accounting principles, used by the Selling Party, as consistently applied in its respective audited financial statements.
1.72.9If Eisai, or its Affiliate sells or transfers units of a Licensed Product in conjunction with any other product, and in so doing sells or transfers such units for an amount less than the sum of the weighted average selling price for such units of such Licensed Product sold separately, for the purposes of determining Net Sales from such sales or transfers, Net Sales shall be, subject to the deductions from gross amounts provided in Section 1.72.1 through Section 1.72.5 above, based upon the price of such Licensed Product sold to a similar size customer ordering a similar volume of units of the Licensed Product under similar terms and conditions, but sold separately.
1.73“New Indication Study” has the meaning set forth in Section 4.5.4.
1.74“New Indication Study Know-How” has the meaning set forth in Section 4.5.4.
1.75“Nuvation” has the meaning set forth in the preamble.
1.76“Nuvation Cure Period” has the meaning set forth in Section 12.3.1.
1.77“Nuvation Know-How” means any Know-How Controlled by Nuvation or any of its Affiliates as of the Effective Date or at any time during the Term (a) that is necessary or
9
Page of SECTIONPAGES \* MERGEFORMAT 55
reasonably useful for the Development, Manufacture and Commercialization of any Licensed Product in the Field in the Territory, or (b) that is otherwise necessary or reasonably useful to exercise the licenses granted under Section 2.1.2(b) or Section 2.1.3.
1.78“Nuvation Patents” means all Patents Controlled by Nuvation or any of its Affiliates as of the Effective Date or at any time during the Term (a) that include a Valid Claim Covering the Licensed Compound or any Licensed Product, (b) that are otherwise necessary or reasonably useful for the Development, Manufacture and Commercialization of any Licensed Product in the Field in the Territory, or (c) that are otherwise necessary or reasonably useful to exercise the licenses granted under Section 2.1.2(b) or Section 2.1.3. The Nuvation Patents as of the Effective Date in the Territory are set forth in Schedule 1.78.
1.79“Other Nuvation IP” means (a) the trademarks set forth in Schedule 1.79 and, (b) to the extent not included in Nuvation Know-How, any Intellectual Property Rights (including in the packaging and package inserts of the Licensed Products) Controlled by Nuvation or any of its Affiliates as of the Effective Date or at any time during the Term, in each case (a) and (b) (i) that are necessary or reasonably useful for the Development, Manufacture and Commercialization of any Licensed Product in the Field in the Territory, or (ii) that are otherwise necessary or reasonably useful to exercise the licenses granted under Section 2.1.2(b) or Section 2.1.3.
1.80“Out-of-Pocket Costs” means amounts paid to Third Parties in respect of goods, services or other materials. For clarity, Out-of-Pocket Costs do not include payments for internal costs; salaries or benefits; facilities; utilities; general office or facility supplies; insurance; information technology; capital expenditures or the like.
1.81“Outside of Territory Patents” means all Patents Controlled by Nuvation or any of its Affiliates as of the Effective Date or at any time during the Term outside of the Territory that include a Valid Claim Covering the Licensed Compound or any Licensed Product.
1.82“Patents” means any of the following: (a) any issued and unexpired patent, including without limitation, any inventor’s certificate, substitution, extension, re-registration, confirmation, reissue, re-examination, re-validation, renewal or any similar governmental grant for protection of inventions (including, but not limited to, patent term extensions, pediatric exclusivity or supplementary protection certificate) and all other post-grant forms of any of the foregoing; (b) any patent application including, without limitation, any continuation, divisional, substitution, continuation-in-part, provisional applications, converted provisional applications and all other pre-grant forms of any of the foregoing; and (c) all foreign counterparts of any of the foregoing.
1.83“Pricing Approval” means all approvals, licenses, registrations or authorizations of the applicable Reimbursement Authority.
1.84“Prosecuting” means preparing, filing, prosecuting, obtaining the issuance of, maintaining or renewing a Patent, including any interferences, reissue proceedings, reexaminations, oppositions, invalidation proceedings, opposition of validity or enforceability challenges, national validation filings (for example, with respect to European Patents and Unitary
10
Page of SECTIONPAGES \* MERGEFORMAT 55
Patents) and patent listing procedures (e.g. in Canada). “Prosecute” and “Prosecution” have correlative meaning.
1.85“Quality Agreement” has the meaning set forth in Section 6.5.
1.86“Regulatory Authority” means any federal, national, multinational (including for example the EMA or the European Commission), state, provincial or local regulatory agency, department, bureau, or other governmental entity with authority over the research, development, manufacture, commercialization, or other use (including the granting of Marketing Approvals) of the Licensed Product(s) or companion diagnostic in the Territory.
1.87“Regulatory Exclusivity” means exclusivity (other than Patent protection or Patent-related exclusivity) granted or afforded by Applicable Law or by a Regulatory Authority in the applicable country or jurisdiction that confers exclusive marketing rights or data exclusivity rights with respect to a Licensed Product in such country or jurisdiction and prevents a Third Party from submitting a regulatory filing and marketing or selling such Licensed Product without the prior written consent of the MAH, including under national implementations of Article 10 of Directive 2001/83/EC, or rights similar thereto in any other jurisdiction.
1.88“Regulatory Filings” means any filing, application, or submission (including clinical study results or transparency submissions) with any Regulatory Authority or Reimbursement Authority, including authorizations, variations, approvals or clearances arising from the foregoing, including the DMF, any orphan drug designation application, pediatric application, any NDA, any application for a Marketing Approval, Marketing Approvals, any safety-related filings, and all other filings, reports or correspondence or communication with or from the relevant Regulatory Authority or Reimbursement Authority including minutes or written feedback of any material meetings, telephone conferences or discussions with the relevant Regulatory Authority or Reimbursement Authority, in each case, with respect to a Licensed Product in the Territory.
1.89“Regulatory Milestone Payment” has the meaning set forth in Section 7.2.
1.90“Regulatory Subcommittee” has the meaning set forth in Section 3.8.
1.91“Reimbursement Authority” means any federal, national, provincial or local regulatory agency, department, bureau, or other entity with authority over establishing whether a biopharmaceutical or pharmaceutical product may be reimbursed or the prices that can be charged, paid or reimbursed for a biopharmaceutical or pharmaceutical product.
1.92[***] has the meaning set forth in Section 4.5.3.
1.93[***] has the meaning set forth in Section 4.5.3.
1.94[***] has the meaning set forth in Section 4.5.3.
1.95“Royalty Payment” has the meaning set forth in Section 7.4.
1.96“Royalty Report” has the meaning set forth in Section 7.7.
11
Page of SECTIONPAGES \* MERGEFORMAT 55
1.97“Royalty Term” has the meaning set forth in Section 7.5.
1.98“Sales Milestone Payment” has the meaning set forth in Section 7.3.
1.99“Subcommittee” has the meaning set forth in Section 3.6.
1.100“Subcommittee Member” has the meaning set forth in Section 3.6.
1.101“Sublicense” means an agreement into which Eisai enters with an Affiliate or Third Party (other than a Distributor) for the purpose of (a) granting certain rights under the Licensed IP, including without limitation the Nuvation Patents or Nuvation Know-How, or (b) forbearing the exercise of any rights granted to Eisai under this Agreement.
1.102“Sublicensee” means a Third Party (other than a Distributor) with whom Eisai enters into a Sublicense.
1.103“Term” has the meaning set forth in Section 12.1.
1.104“Territory” means, [***], the countries within the regions listed in the table in Schedule 1.1041.104 to this Agreement.
1.105“Third Party” means any legal person or entity other than a Party or an Affiliate of a Party.
1.106“Upfront Payment” has the meaning set forth in Section 7.1.
1.107“U.S.” or “United States” means the United States of America and all of its territories and possessions.
1.108“Valid Claim” means [***].
1.109“Wind-Down Period” has the meaning set forth in Section 12.6.5.
2.1.1Exclusive License to Eisai. In consideration of and subject to the terms and conditions of this Agreement (including Section 8.6), Nuvation grants to Eisai a sublicensable (in accordance with and subject to Section 2.2), royalty-bearing, exclusive (even as to Nuvation and its Affiliates) right and license or sublicense, as applicable, under the Licensed IP to Develop and Commercialize the Licensed Product(s) in the Field and in the Territory.
2.1.2Non-Exclusive Licenses to Eisai. In consideration of and subject to the terms and conditions of this Agreement, Nuvation grants to Eisai a sublicensable (in accordance with and subject to Section 2.2), royalty-bearing, non-exclusive right and license or sublicense, as applicable, under the Licensed IP to: (a) make (including packaging or labelling), have made (including packaging or labelling), import, export, and keep (collectively “Manufacturing” or
12
Page of SECTIONPAGES \* MERGEFORMAT 55
“Manufacture” (as applicable)) any Licensed Product in the Territory; and (b) Manufacture any Licensed Product outside of the Territory solely for the purpose of exploiting the Licensed Product in the Territory pursuant to the exclusive license granted to Eisai under Section 2.1.1 above.
2.1.3Global Congresses and JCC-Approved Pre-Clinical Studies. Notwithstanding that Eisai’s license to Develop any Licensed Product in the Field is limited to the Territory pursuant to Section 2.1.1, the Parties acknowledge and agree that Eisai shall have (a) the right to participate in any global congresses that are organized outside of the Territory for any Licensed Product, subject to review and coordination with Nuvation via the JCC in accordance with Article 3; and (b) subject to approval by the JCC in accordance with Article 3 below, a non-exclusive right to conduct non-clinical and pre-clinical activities in respect of Licensed Product or the Licensed Compound outside of the Territory (and where the results of which are Controlled by Eisai these would be deemed to be Eisai IP) solely for either Development or Commercializing the Licensed Product in the Territory [***] and such right shall not, under any circumstances, grant Eisai any right to Commercialize the Licensed Product outside of the Territory.
2.2Sublicenses; Subcontracting.
2.2.1Eisai may grant Sublicenses of those rights granted by Nuvation under Section 2.1 to Eisai’s Affiliates and Third Parties through multiple tiers; provided, that Eisai shall obtain the prior written consent of Nuvation, which consent shall not be unreasonably withheld or delayed, prior to (a) Eisai granting any Sublicense to a Sublicensee and (b) a Sublicensee or Affiliate of Eisai granting any additional tier of Sublicense, provided that if Nuvation does not notify Eisai of its denial or approval of a proposed sublicensee within [***] calendar days of receiving the request from Eisai, such sublicensee shall be deemed approved. For clarity, Nuvation’s prior written consent is not required for Eisai to grant a Sublicense to any of its Affiliates. Eisai shall ensure that each Sublicensee and applicable Affiliate is bound by a written Sublicense that is consistent with, and expressly made subject to, the applicable terms and conditions of this Agreement. Eisai shall remain responsible and be liable to Nuvation for any breach of the terms of this Agreement by Eisai’s Affiliates and Sublicensees without regard to whether Nuvation consents to such Sublicense.
2.2.2Eisai and its Affiliates and Sublicensees may engage Third Party subcontractors (including Distributors) to exercises their respective rights and perform their respective obligations under this Agreement, provided that (a) Eisai shall remain responsible and liable to Nuvation for the performance by any such Third Party subcontractor engaged by Eisai or its Affiliates or Sublicensees and (b) Eisai shall ensure that, prior to any such engagement, each such Third Party subcontractor is subject to a binding written agreement containing terms and conditions that enable Eisai to comply with its obligations under this Agreement, including obligations of confidentiality, obligations relating to Intellectual Property Rights, and the right for Nuvation to perform (or have performed) audits to the extent provided under this Agreement and as otherwise may be necessary to confirm compliance with the applicable terms of this Agreement. In connection with the engagement of any such Third Party subcontractor (including any Distributor), Eisai or its Affiliate or Sublicensee may grant a sublicense of its rights under Section 2.1, and, provided that such sublicense is in the ordinary course of business and is incidental or ancillary to the performance of the Third Party subcontractor, such grant of rights shall not be deemed a “Sublicense” under this Agreement and such Third Party subcontractor shall not be
13
Page of SECTIONPAGES \* MERGEFORMAT 55
deemed a “Sublicensee” under this Agreement. For clarity, (a) a grant of rights to use the trademarks set forth in Schedule 1.79 in accordance with the terms of this Agreement by a Distributor in connection with the distribution, marketing or resale of Licensed Products shall be deemed incidental to the performance of such Distributor, and (b) a non-exclusive, freedom-to-operate sublicense granted to CROs and CDMOs engaged by Eisai solely to enable such CROs and CDMOs to perform services on behalf of Eisai which does not include the right for such CROs and CDMOs to Commercialize Licensed Products shall be deemed shall be deemed incidental to the performance of such services by such CROs and CDMOs.
2.3.1Nuvation will remain responsible for and shall satisfy all obligations arising under licenses from Third Parties (each, a “Third Party License”) executed prior to the Effective Date. Each Party shall promptly notify the other Party if it becomes aware of any Third Party Know-How or Patents that are necessary to Develop, Manufacture or Commercialize a Licensed Product in the Territory (the “Third Party IP”). As between the Parties, [***].
2.4.1If the DS License Agreement is terminated in whole or in part with respect to the Licensed Compound or Licensed Product in the Territory, Nuvation shall promptly notify Eisai upon its delivery of a notice of termination of the DS License Agreement or upon its receipt of such a notice and Eisai, its Affiliates or Sublicensees may at its sole option negotiate a direct license of the right to continue to use the Licensed IP licensed to Nuvation under the DS License Agreement to the full extent contemplated by this Agreement (a “Direct License”). If Eisai wishes to obtain a Direct License from Daiichi Sankyo, Eisai shall promptly notify Nuvation of the same (and prior to the effective date of the termination of the DS License Agreement in the event it is made aware of such termination prior to such date). Upon request from Eisai, Nuvation shall [***].
2.4.2In the event that the DS License Agreement is terminated (other than due to any material breach stemming from any act or omission of Eisai or its Affiliates) and Eisai obtains a Direct License, then Eisai shall [***].
2.5Nuvation’s Reserved Rights. Notwithstanding anything in this Agreement to the contrary, the Parties acknowledge and agree that Nuvation retains a limited right to practice and use the Licensed IP solely to the extent necessary or useful to enable Nuvation to (a) assist Eisai with regulatory and clinical activities agreed in accordance with this Agreement for the Licensed Product in the Territory for the purpose of obtaining a Marketing Approval for a Licensed Product, (b) conduct Clinical Trials in the Territory in the Field, provided the study plan and budget for such studies are approved by the JCC in accordance with Article 3 and further provided that under no circumstances shall such right grant Nuvation any rights to Commercialize the Licensed Product in the Territory, (c) participate in any global congresses that are organized in the Territory in respect of any Licensed Product, subject to review and coordination with Eisai via the JCC in accordance with this Agreement, and (d) otherwise exercise its rights and perform its obligations as set forth in and in accordance with this Agreement, which for clarity shall not include the right to Commercialize the Licensed Product in the Territory.
14
Page of SECTIONPAGES \* MERGEFORMAT 55
2.6Eisai License to Nuvation. Eisai hereby grants Nuvation a non-exclusive, [***] perpetual, irrevocable, right and license, with the right to grant sublicenses through multiple tiers, under (a) the Eisai IP, (b) the AB-106-G319 Study IP Controlled by Eisai, (c) [***], (d) the New Indication Study IP Controlled by Eisai, and (e) [***], in each case, to Develop and Commercialize the Licensed Product(s) outside the Territory. For clarity, the foregoing license shall survive termination or expiration of this Agreement.
2.7No Other Rights. Except as otherwise expressly provided in this Agreement, under no circumstances will a Party, as a result of this Agreement, obtain any ownership interest, license right or other right in any Know-How, Patent, or other Intellectual Property Rights of the other Party or any of its Affiliates, including items owned, controlled, developed, or acquired by the other Party or any of its Affiliates, or provided by the other Party to the first Party at any time pursuant to this Agreement.
2.8Exclusivity. On a country-by-country basis in the Territory, during the Royalty Term for such country, neither Party shall, directly or through its Affiliates, Third Party contractors or sublicensees (including Sublicensees), Commercialize any Competing Product in such country.
3.1Joint Collaboration Committee. Within [***] days of the Effective Date, the Parties shall each appoint (a) one person to coordinate its activities under this Agreement (each an “Alliance Manager”) and (b) an equal number of, and in any case no more than [***] senior executives to establish a joint collaboration committee (the “JCC”), which will act as a forum for information exchange, monitoring, and discussion of issues, best practices, and ideas with respect to any Licensed Product in the Territory. Each Party may change its representatives to the JCC, in its sole discretion, effective upon written notice to the other Party designating such change. The representatives from each Party shall have appropriate technical credentials, experience and knowledge pertaining to, and ongoing familiarity with, the Licensed Products and their Development, Manufacturing and Commercialization. The Parties’ respective Alliance Managers will take turns in being responsible for calling and chairing meetings of the JCC (at times that are convenient for both Parties’ JCC representatives), circulating agendas (which shall include all agenda items proposed by both Parties and standing agenda items with respect to (i) intellectual property (including in respect of matters referred to in Article 8) and (ii) Development Updates) and performing administrative tasks required to assure efficient operation of the JCC but the Party responsible in a given turn shall not have any extra or additional vote. Nuvation will take the first turn with respect to the first meeting of the JCC.
3.2Function and Powers of the JCC.
3.2.1Discussion and Coordination. The JCC, or its designated Subcommittee, will:
(a)review and coordinate the Parties’ participation with respect to any Licensed Product in any global congresses (including, by way of example, ASCO, ESMO and
15
Page of SECTIONPAGES \* MERGEFORMAT 55
World Lung) organized within or outside the Territory where the Licensed Product is the subject of presentations, booths and stands, seminars, advisory boards panel discussions, satellite symposiums or posters at such global congress;
(b)review and align on and ensure the compliance of any material data disclosures, claims and messages which relate to the Licensed Product prepared by or on behalf of either Party for use in any global congress organized within or outside the Territory, and such materials are to be provided for review no later than [***] weeks before the congress is due to be held;
(c)review any expanded access programs with respect to Licensed Products in the Territory;
(d)review and discuss Development Updates;
(e)review and discuss Eisai’s Medical Affairs Activities with respect to the Licensed Products in the Territory;
(f)discuss and coordinate the transfer of Nuvation Know-How pursuant to Section 5.1;
(g)review and discuss Commercialization Updates;
(h)review the manufacturing strategy for the Licensed Compound or Licensed Products, and review the operation of the Commercial Supply Agreement regarding the supply of the Licensed Compound or Licensed Products;
(i)coordinate and review Nuvation’s assistance provided pursuant to Section 4.1.3 and in connection with Eisai’s activities under Section 4.1 with respect to the Licensed Product in the Field in the Territory;
(j)review and consider [***];
(k)review and consider the need for obtaining a license to any Third Party Know-How or Patents that could be reasonably useful for the Development, Manufacture or Commercialization of the Licensed Product(s) for the Initial Indication in the Territory and the costs related thereto; and
(l)consider additional suitable matters for review and discussion, which shall be considered on a case-by-case basis and recorded in the JCC meeting minutes.
3.2.2Approval. The JCC directly, or through its designated Subcommittee, will review and discuss ahead of the JCC’s decision to approve, or not:
(a)any changes to the manufacturing process for a Licensed Product required by any Regulatory Authority in the Territory and establish a plan for implementing such changes;
16
Page of SECTIONPAGES \* MERGEFORMAT 55
(b)any investigator-initiated trials with respect to Licensed Products in the Territory (whilst taking into account that such investigator-initiated trials are, pursuant to Applicable Law, to be approved by medical, and not commercial, functions);
(c)study plans for the AB-106-G319 Study in the Territory (following Eisai’s relevant approval under Section 4.5.2), [***], and any New Indication Study (including which Party shall be responsible for the performance of such New Indication Study), and the coordination and performance thereof;
(d)corresponding budgets for the AB-106-G319 Study in the Territory (following Eisai’s relevant approval under Section 4.5.2), [***] or New Indication Study;
(e)any proposed study plan and budget for other Clinical Trials in the Field proposed to be conducted by Nuvation in the Territory;
(f)any proposed Eisai study plan to conduct non-clinical or pre-clinical activities in respect of a Licensed Product or the Licensed Compound outside of the Territory solely for either Development or Commercializing of the Licensed Product in the Territory, [***];
(g)the external assistance described in Section 4.1.3 proposed by Nuvation and associated Out-Of-Pocket Costs;
(h)any scientific presentations and publications that disclose material data generated inside and outside of the Territory with respect to the Licensed Compound or Licensed Product(s), such approval right to be limited to the first such presentation or publication of such data only;
(i)the resolution of any Material Commercial Issues; and
(j)additional suitable and material matters for review, discussion and approval shall be considered on a case-by-case basis and recorded in the JCC meeting minutes.
3.3Meetings. The JCC will meet at least once per Calendar Quarter for so long as the JCC remains in effect. The JCC may conduct such meetings by telephone, videoconference, or in person. Each Party may call special meetings of the JCC with at least [***] Business Days’ prior written notice, or a shorter time period in exigent circumstances, to resolve particular matters requested by such Party that are within the purview of the JCC. Meetings of the JCC are effective only if at least [***] representative of each Party participates in such meeting. Any decision from such meeting shall be documented and shall not be effective until shared and approved (or deemed to be approved) by both Parties’ respective Alliance Managers as provided below in this Section 3.3. Each Party may invite a reasonable number of other participants, in addition to its representatives, to attend JCC meetings in a non‑voting capacity; provided, that if either Party intends to have any Third Party (including any consultant) attend such a meeting, such Party shall: (a) provide prior written notice to the other Party and shall ensure in advance that such Third Party is bound by confidentiality and non-use obligations and intellectual property assignment obligations in each case consistent with the terms of this Agreement; and (b) bear the cost of such Third Party attending such a meeting. Each Party is responsible for its own expenses incurred in connection with participating in and attending all such meetings. The Alliance Manager of the
17
Page of SECTIONPAGES \* MERGEFORMAT 55
Party calling the meeting or his/her designee shall keep minutes of each JCC meeting that records in writing all decisions made, action items assigned or completed and other appropriate matters. Such Alliance Manager or his/her designee shall send meeting minutes to the other Party’s Alliance Manager promptly after a meeting for review. Such Alliance Manager shall have [***] Business Days from receipt in which to comment on and to approve or reject the minutes. If such Alliance Manager, within such time period, does not notify the other Party’s Alliance Manager or his/her designee that they do not approve of the minutes, the minutes shall be deemed to have been approved by such Alliance Manager. Each Party shall bear the cost of its respective JCC members attending such JCC meetings.
3.4Decisions. Except as set forth herein expressly, the JCC will make decisions by consensus, with the representatives of each Party having, collectively, one (1) vote on behalf of that Party. The representatives of each Party on the JCC shall take into reasonable consideration the recommendations and concerns of each Party and shall endeavor to make any decisions before the JCC in good faith and using reasonable business judgment. If, despite the foregoing good faith efforts, the JCC cannot reach approvals by consensus, then either Party may refer such dispute to the Executive Officers for resolution. If consensus cannot be reached with respect to a decision within [***] days after attempted resolution by the Executive Officers, then:
3.4.1subject to Section 3.4.3, Nuvation shall have final decision-making authority with respect to [***];
3.4.2except as set forth in Section 3.4.1, Eisai shall have final decision-making authority with respect to [***];
3.4.3with respect to [***], such matter will be referred to confidential mediation in accordance with Section 13.3 and if the matter is not resolved in accordance with Section 13.3, [***]; and
3.4.4with respect to [***], (i) neither Party shall have final decision-making authority, (ii) such matter will be referred to [***].
3.5Authority. The JCC shall only have the powers assigned expressly to them in this Article 3 and elsewhere in this Agreement (or in the case of Subcommittees, as expressly assigned to them by the JCC). Each Party retains the rights, powers, and discretion granted to it under this Agreement and neither Party may delegate or vest such rights, powers, or discretion in the JCC or any other person or committee unless expressly provided for in this Agreement or the Parties expressly so agree in writing. The JCC (and either Party in exercising its final decision-making authority at the JCC) shall not have the power to amend, waive or modify any term of this Agreement, and no decision of the JCC may be in contravention of any terms and conditions of this Agreement. It is understood and agreed that issues to be formally decided by the JCC are limited to those specific issues that are expressly provided in this Agreement to be decided by the JCC.
3.6Subcommittees. The JCC may, from time to time, establish subcommittees to coordinate and oversee particular projects or activities (for example, by identifying and developing solutions to issues arising within their remit) under this Agreement (each a “Subcommittee”),
18
Page of SECTIONPAGES \* MERGEFORMAT 55
including the Clinical Subcommittee described in Section 3.7, the Regulatory Subcommittee described in Section 3.8 and the Medical Subcommittee described in Section 3.9 and any manufacturing subcommittee described in the Commercial Supply Agreement. Each Subcommittee shall be comprised of such number of representatives from each Party as the JCC decides from time to time, each with appropriate seniority and functional expertise and two of which shall serve as the co-chairs of such Subcommittee to enable such representative to make decisions on behalf of the relevant Party in respect of issues falling within the jurisdiction of such Subcommittee (“Subcommittee Member”). The quorum for a meeting of any Subcommittee shall be no less than half of such Subcommittee Members from each Party including the relevant co-chairs. Each Subcommittee will meet at a frequency to be agreed between the Subcommittee co-chairs for so long as the Subcommittee remains in effect. The Parties acknowledge and agree that they anticipate such Subcommittee Members will communicate on a daily or weekly basis as appropriate outside of such meetings. Each Subcommittee will report and where applicable provide its recommendations through the co-chairs to the JCC as appropriate, including to notify the JCC of any issues that cannot be resolved by the Subcommittee for resolution by the JCC. The JCC may dissolve any Subcommittee which is no longer necessary or useful. Each Party shall bear the cost of its respective Subcommittee Members attending such Subcommittee meetings.
3.7Clinical Subcommittee. Within [***] days of the Effective Date, the JCC may establish a joint clinical Subcommittee (the “Clinical Subcommittee”) in accordance with Section 3.6 which shall report to the JCC. The Clinical Subcommittee shall have responsibility for making recommendations to the JCC regarding scientific presentations and publications that disclose data generated inside and outside of the Territory with respect to the Licensed Compound or Clinical Trials (such approval right shall only apply in respect of the first such presentation or publication of such data), overseeing and coordinating the activities described in Section 4.5, sharing Development Updates or any other activities as may otherwise be determined by the JCC.
3.8Regulatory Subcommittee. Within [***] days of the Effective Date, the JCC may establish a joint regulatory Subcommittee (the “Regulatory Subcommittee”) in accordance with Section 3.6 which shall report to the JCC. The Regulatory Subcommittee shall have responsibility for overseeing and coordinating the activities described in Section 4.1.3 or any other activities as may otherwise be determined by the JCC.
3.9Medical Subcommittee. Within [***] days of the Effective Date, the JCC may establish a joint medical Subcommittee (the “Medical Subcommittee”) in accordance with Section 3.6 which shall report to the JCC. The Medical Subcommittee shall have responsibility for overseeing and coordinating and making recommendations to the JCC regarding Medical Affairs Activities. This may include publications and the results of evidence generation from phase 4 Clinical Trials and investigator initiated trials either inside and outside of the Territory with respect to the Licensed Compound or Licensed Product(s) (such approval right shall only apply in respect of the first such presentation or publication of such data), Medical Affairs materials which relate to the Licensed Product prepared by, or on behalf of, either Party for use in any global congress organized within or outside the Territory and any expanded access programs with respect to Licensed Products in the Territory.
3.10Discontinuation of the JCC. The JCC and any Subcommittees will automatically disband upon termination or expiry of this Agreement. Once disbanded, all approval rights of the
19
Page of SECTIONPAGES \* MERGEFORMAT 55
JCC, or final decision-making authority granted to a Party pursuant to this Agreement, shall become approval rights of the corresponding Party (i.e., mutual agreement by the Parties or final decision-making authority by a Party).
4.Regulatory Responsibilities; Development
4.1.1Subject to the terms of this Agreement, from the Effective Date, as between the Parties, Eisai, either directly or through its Affiliates or other agents, shall have the right (but shall not be obliged) to (a) seek, obtain, and maintain Marketing Approvals for all Licensed Products in the Territory, subject to the terms of the Commercial Supply Agreement, at Eisai’s sole cost and expense in accordance with Section 4.1.3 and (b) hold and own all Regulatory Filings including the Marketing Approvals in the Territory as MAH in the Territory. As between the Parties, Eisai shall have sole discretion to determine any Regulatory Filings, market access initiatives, commercial launch activities, distribution, safety measures, and any other activities relating to any Licensed Product in the Territory.
4.1.3From the Effective Date, at the reasonable request of Eisai, Nuvation will promptly provide reasonable assistance to Eisai in connection with Eisai’s activities under Section 4.1 with respect to the Licensed Product in the Field in the Territory, including by (a) promptly (and in any event within [***] Business Days of such request or earlier if so required by the Regulatory Authority) providing reasonably necessary and accurate information, analysis and documentation, including information that relates to regulatory, quality, non-clinical and clinical data to the extent in Nuvation’s or its Affiliates’ possession or Control and using Commercially Reasonable Efforts to obtain such provision from its subcontractors (including CROs and other Third Party vendors for example imaging vendors, systems vendor (EDC, IRT etc.), PK/PD vendors or biostats vendors), (b) preparing for, and if required attending, meetings in connection with Regulatory Filings, (c) assisting in preparing and filing Regulatory Filings for Marketing Approvals with Regulatory Authorities, (d) preparing for the Health Technology Assessment dossier for centralized Joint Clinical Assessment, (e) assisting with responding to questions raised by a Regulatory Authority following application for Marketing Approval and grant of Marketing Approval, (f) assisting with responding to regulatory issues raised by a Regulatory Authority whether during an inspection, audit or otherwise; and (g) in accordance with the Quality Agreement (where applicable), promptly providing necessary information, analysis and documentation in Nuvation’s possession or Control, and using Commercially Reasonable Efforts to obtain provision of necessary information, analysis and documentation from its subcontractors (including CROs or CDMOs) in such subcontractors’ possession or Control, to the extent required with respect to any regulatory issues raised by a Regulatory Authority in a country of the Territory concerning the Manufacture of the Licensed Compound or Licensed Product; provided that, in each case ((a) to (g)) and, subject to Section 4.1.5, Eisai will promptly reimburse [***] of Nuvation’s reasonable Out-Of-Pocket Costs (provided that such costs are approved by the JCC under Article 3 and are actually incurred by Nuvation for the foregoing assistance and provided further that Nuvation shall not be required to incur any Out-of-Pocket Costs in relation to the foregoing assistance that are not reimbursed by Eisai) but excluding any Out-of-Pocket Costs
20
Page of SECTIONPAGES \* MERGEFORMAT 55
incurred by Nuvation to resolve verified deficiencies identified by Regulatory Authorities solely related to the information, documentation and analysis provided by or on behalf of Nuvation, including its CROs and CDMOs (which costs shall be borne by Nuvation). Nuvation acknowledges and agrees that any related internal costs incurred by Nuvation or its Affiliates for the foregoing assistance shall be borne solely by Nuvation. During the Term, Nuvation and/or its Affiliates shall maintain the rights to all data used in Regulatory Filings filed by Eisai with respect to the Licensed Products so that Nuvation and/or its Affiliates retain Control of such data insofar as such data are relevant to Development and Commercialization in the Territory.
4.1.4If Nuvation reasonably feels that the level of support requested by Eisai pursuant to Section 4.1.3 has extended beyond what is reasonable, this will be discussed and be renegotiated in good faith at the JCC [***].
4.1.5Notwithstanding Section 4.1.1 or Section 4.1.3, if, in connection with Eisai seeking, obtaining, or maintaining Marketing Approval for a Licensed Product, [***] then the matter shall be referred to the JCC for timely resolution in accordance with Article 3. If as part of the resolution [***], the JCC agrees to modify [***], then the JCC shall establish a plan for implementing such modification and for the reimbursement of costs in association therewith, provided that all reasonable Out-of-Pocket Costs actually incurred by Nuvation for such implementation shall [***]. Any related internal costs incurred by Nuvation or its Affiliates shall be borne solely by Nuvation.
4.2Development Updates. At each regularly scheduled JCC meeting or Clinical Subcommittee meeting, each Party shall reasonably inform the other as to the progress and results of its and its Affiliates’ and Sublicensees’ Development of the Licensed Products in the Territory and its plans for upcoming Development activities, including all planned Clinical Trials in the Territory and the draft protocols for such Clinical Trials. At least [***] Business Days before each such meeting, each Party shall provide the JCC with a written report summarizing such Development activities, the results thereof, and the upcoming plans therefor (each, a “Development Update”). Each Development Update shall cover the applicable subject matter at a reasonable level of detail sufficient to enable each Party to determine the other Party’s compliance with its obligations hereunder. In addition, each Party shall make available to the other Party such additional information about its Development activities as Nuvation may reasonably request from time to time.
4.3Regulatory Exclusivity. If Eisai decides to seek Regulatory Exclusivity in the Territory, Eisai will be responsible, at its sole expense, for preparing and filing such requests with the applicable Regulatory Authority. Nuvation will, upon Eisai’s reasonable request, provide reasonable support to Eisai in preparing and filing such requests at [***].
4.4Medical Affairs. Eisai will, at its sole cost, be responsible for the execution of Medical Affairs Activities in accordance with a strategy based upon Nuvation’s global medical affairs strategy as reasonably adapted for the Territory and as set out in an annual medical affairs plan, and as discussed at the JCC and will provide quarterly updates to the JCC regarding such Medical Affairs Activities and progress against such annual plan. Notwithstanding the foregoing, including investigator-initiated trials and expanded access programs with respect to Licensed Product(s) in the Territory shall be subject to approval by the JCC.
21
Page of SECTIONPAGES \* MERGEFORMAT 55
4.5Development Activities.
4.5.1Development. Subject to Sections 4.5.2, 4.5.3 and 4.5.4 Eisai shall, at its own cost and expense, have the discretion over the Development of the Licensed Products it undertakes in the Field in the Territory and shall comply with all Applicable Law and, where applicable, GxP for any such Development activities. Nuvation shall comply with all Applicable Law and, where applicable, GxP for its Development activities contemplated under this Agreement. If either Party reasonably believes that the other Party’s Development activities are being conducted in a manner that would have, or would be reasonably expected to have, a material adverse effect on the Development or Commercialization of such Licensed Product in such first Party’s respective territory(ies), then such first Party may raise the matter to the JCC for good faith discussion.
4.5.2The AB-106-G319 Study. The Parties acknowledge that Nuvation sponsors and manages the AB-106-G319 Study in countries outside of the Territory. Nuvation agrees not to conduct the AB-106-G319 Study in countries within the Territory, unless approved by Eisai. If Eisai approves the conduct of the AB-106-G319 Study in countries within the Territory Nuvation shall continue to sponsor and manage the AB-106-G319 Study as extended to such countries, provided that Eisai and Nuvation shall thereafter share the costs of the AB-106-G319 Study as such costs pertain to the activities in the countries in the Territory portion on a [***] basis, subject to approval by the JCC with respect to a study plan and the Parties’ mutual agreement in writing with respect to a corresponding budget for such AB-106-G319 Study in the Territory. Any Know-How, Patents, or other Intellectual Property Rights generated in the course of conducting the AB-106-G319 Study, including the results thereof (the “AB-106-G319 Study IP”), that is solely invented by or on behalf of Nuvation shall be deemed to be Nuvation Know-How, Nuvation Patents, or Other Nuvation IP (as applicable) and subject to the license in Section 2.1, any AB-106-G319 Study IP that is solely invented by or on behalf of Eisai shall be subject to the license in Section 2.6, and any AB-106-G319 Study IP that is jointly invented by or behalf of Nuvation, on one hand, and by or on behalf of Eisai, on the other hand, shall be Joint IP and subject to the licenses in Section 2.1 and Section 2.6 as applicable.
4.5.4New Indication Studies in the Territory. For any Clinical Trial in any country or countries in the Territory for a Licensed Product other than the AB-106-G319 Study or [***] (a “New Indication Study”), subject to approval by the JCC with respect to a study plan and the Parties’ mutual agreement in writing with respect to a corresponding budget for such New Indication Study, Eisai and Nuvation shall share the costs of such New Indication Study on a [***] basis. Prior to the initiation of each New Indication Study, the Parties shall discuss in good faith via the JCC which Party shall sponsor such New Indication Study. Any Know-How, Patents, or other Intellectual Property Rights generated in the course of conducting such New Indication Study, including the results thereof (the “New Indication Study IP”), that is solely invented by or on behalf of Nuvation shall be deemed to be Nuvation Know-How, Nuvation Patents, or Other Nuvation IP (as applicable) and subject to the license in Section 2.1, any New Indication Study IP that is invented solely by or on behalf of Eisai shall be subject to the license in Section 2.6, and any New Indication Study IP that is jointly invented by or behalf of Nuvation, on one hand, and
22
Page of SECTIONPAGES \* MERGEFORMAT 55
by or on behalf of Eisai, on the other hand, shall be Joint IP and subject to the licenses in Section 2.1 and Section 2.6 as applicable.
4.6Right of Reference. Each Party shall have the right to cross-reference the other Party’s or its Affiliate’s DMF, if any, and any other Regulatory Filings submitted by or on behalf of Nuvation anywhere in the world related to the Licensed Products, and to access such Regulatory Filings and any data and Know-How therein and use such data and Know-How solely in connection with the performance of its obligations and exercise of its rights under this Agreement, including inclusion of such data and Know-How in its own Regulatory Filings. Nuvation hereby grants to Eisai a “Right of Reference,” as that term is defined in 21 C.F.R. § 314.3(b) in the United States, or an equivalent right of access/reference in the Territory, to any data, including Nuvation’s, its Affiliates’ or its sublicensees’ clinical dossiers, in each case, to the extent Controlled by Nuvation or its Affiliates that relates to any Licensed Product(s) for use by Eisai, its Affiliates and Sublicensees to Commercialize, Manufacture and Develop Licensed Product(s) in the Field in the Territory pursuant to this Agreement. Eisai hereby grants to Nuvation a Right of Reference or an equivalent right of access/reference outside the Territory, to any data, including Eisai’s clinical dossiers, Controlled by Eisai, its Affiliate or its Sublicensee that relates to any Licensed Product(s) for use by Nuvation and its Affiliates to Commercialize, Manufacture and Develop Licensed Product(s) in the Field outside the Territory. The Party granting such Right of Reference shall provide a signed statement to this effect, if requested by the other Party, in accordance with 21 C.F.R. § 314.50(g)(3) or the equivalent as required or otherwise provide appropriate notification of such right of such other Party to the applicable Regulatory Authority.
4.7Safety Data Exchange Agreement. Within [***] days following the Effective Date, the Parties shall agree upon a safety data exchange agreement prepared by Eisai specifying a procedure for the mutual exchange of adverse event reports and safety information associated with Licensed Compound and Licensed Products. Prior to the safety data exchange agreement being executed, (a) if Eisai becomes aware of any adverse event or event of special interest in respect of the Licensed Product, Eisai will report such event to Nuvation at AER@nuvationbio.com within [***] Business Days or [***] calendar days, whichever is earlier, and (b) Nuvation will, from the filing of the first Regulatory Filing for a Marketing Approval (excluding Pricing Approvals) in the Territory, send reportable individual case safety reports (ICSRs) in respect of countries in the Territory in which there is a Marketing Approval for the Licensed Product to Eisai in writing within [***] calendar days of receipt as well as any related safety information including relevant quality, non-clinical, clinical, post marketing information or details to Eisai for Eisai to make any required submissions to applicable Regulatory Authorities in the Territory.
5.1Promptly after the Effective Date and in any event within [***] Business Days of the Effective Date, Nuvation, at its sole cost, will transfer to Eisai or to a CRO or CDMO selected by Eisai and reasonably approved by Nuvation, (such approval not to be unreasonably withheld, conditioned or delayed), all documents set forth in Schedule 5.1 hereto, all Nuvation Know-How and documents that (a) exist as of the Effective Date, (b) are necessary or reasonably useful to Commercialize, Develop, Manufacture and for Medical Affairs Activities in respect of any Licensed Product(s) in the Field in the Territory, and (c) in Nuvation’s or its Affiliates’ Control,
23
Page of SECTIONPAGES \* MERGEFORMAT 55
and provide Eisai with a copy of all embodiments of the Nuvation Know-How in a format reasonably accessible to Eisai. Such transfer will be effected through a virtual data room or other reasonable means including File Transfer Protocol (FTP) as determined by the Parties.
6.1.1Subject to the terms of this Agreement, Eisai shall be solely responsible for Commercialization of each Licensed Product in the Field in the Territory during the Term, at its own cost and expense. Eisai shall financially book all sales of the Licensed Product in the Territory. Upon Marketing Approval of a Licensed Product(s) in the Territory for the Initial Indication, Eisai shall, either directly or through its Affiliates, Sublicensees or Distributors, use Commercially Reasonable Efforts to Commercialize at least one Licensed Product in the Field in the Territory in accordance with the approved labeling therefor. Subject to Section 6.3, the Parties agree Eisai retains sole and complete decision-making authority as to whether to launch a Licensed Product in a country of the Territory.
6.1.2Upon Nuvation’s reasonable request in writing made through the JCC, Eisai shall provide Nuvation with an opportunity to review a copy of any promotional materials produced by Eisai’s EMEA regional team (including without limitation materials used on the Internet and social media), artwork and patient leaflets, and packaging or product labelling for the Licensed Product(s) that Eisai intends to use or uses in the course of the Commercialization of the Licensed Product(s) in the Territory and Eisai shall consider in good faith any reasonable comments provided by Nuvation.
6.2Commercialization Updates. Eisai shall provide Nuvation with an annual Commercialization plan and keep Nuvation reasonably informed through the JCC or as otherwise agreed as to the progress and results of its and its Affiliates’ and Sublicensees’ Commercialization of the Licensed Products in the Territory against such plan and any material updates to or deviations from such plan, [***]. At least [***] Business Days before each JCC meeting, Eisai shall provide the JCC with a written report summarizing such Commercialization activities, the status, progress and results thereof (each, a “Commercialization Update”). Each Commercialization Update shall cover the applicable subject matter at a reasonable level of detail sufficient to enable Nuvation to reasonably determine Eisai’s Commercialization efforts and its compliance with its obligations hereunder. In addition, Eisai shall make available to Nuvation such reasonable additional information about its Commercialization activities as may be reasonably requested from time to time.
6.3Material Commercial Issues. With respect to the Commercialization of the Licensed Products in the Territory by Eisai, its Affiliates or its or their Sublicensees, if Nuvation has [***] (a “Material Commercial Issue”), then Nuvation shall provide Eisai with a written notice that presents such demonstrable, objective and verifiable grounds in respect of such Material Commercial Issue (a “Material Commercial Issue Notice”). Upon delivery and receipt of a Material Commercial Issue Notice, the Parties shall convene a meeting of the JCC within [***] Business Days to discuss such Material Commercial Issue and resolve the same in accordance with Section 3.4. Where the relevant Commercialization activity to which the Material Commercial
24
Page of SECTIONPAGES \* MERGEFORMAT 55
Issue relates is [***] and provided further that if, despite good faith discussions, the Parties are unable to agree [***]. The Parties acknowledge and agree that, at regular intervals to be determined by the JCC (and in any event no less than every [***] years), the Parties shall in good faith review and consider (via the JCC) [***], taking into account all relevant factors, including all Applicable Law, [***]. In the event the Parties agree to [***], the Parties shall execute an amendment to this Agreement in accordance with Section 14.7.
6.4Commercial Supply Agreement. Nuvation will manufacture and supply all Licensed Compound and Licensed Product(s) in compliance with the terms and conditions set forth in a commercial supply agreement executed by the Parties [***] after the Effective Date (the “Commercial Supply Agreement”). In addition to customary commercial and contractual provisions, the Commercial Supply Agreement will include: [***].
6.5Quality Agreement. [***], Eisai, Nuvation or its local agent shall enter into a mutually-agreed upon quality agreement (provided that the first draft shall be prepared by Eisai) setting forth the responsibilities and procedures associated with the Licensed Compound or the Licensed Product(s) regarding recalls, changes, deviations, complaint handling, quality-specific audit rights with respect to compliance with all applicable GxP and other quality-related matters (the “Quality Agreement”).
7.1Upfront Payment. Eisai will pay Nuvation a non-refundable, non-creditable payment of €50,000,000 (the “Upfront Payment”). Nuvation shall issue an invoice for the Upfront Payment to Eisai promptly on or promptly after the Effective Date and Eisai will pay such invoice within [***] Business Days of receipt of such invoice.
7.2Regulatory Milestone Payment. Eisai will pay Nuvation a one-time, non-refundable, non-creditable payment of €25,000,000 following the receipt by Eisai, its Affiliate or Sublicensee of a Conditional Marketing Approval or other Marketing Approval, whichever occurs first, for a Licensed Product in the European Economic Area for the Initial Indication (the “Regulatory Milestone Payment”). Within [***] Business Days of achievement of such milestone event, Eisai will notify Nuvation of such achievement and Nuvation shall issue an invoice for the Regulatory Milestone Payment to Eisai promptly on or after such achievement. Eisai will pay such invoice pursuant to Section 7.8. In no case shall the Regulatory Milestone Payment be paid more than once.
7.3Sales Milestone Payments. Eisai will pay to Nuvation the following one-time non-refundable, non-creditable payments upon the first achievement of the following levels of annual Net Sales of all Licensed Product(s) in the Territory (each a “Sales Milestone Payment”). Eisai will notify Nuvation of such achievement in the Royalty Report for the Calendar Quarter ending March 31 of such Fiscal Year and Nuvation shall issue an invoice for the Sales Milestone Payment to Eisai promptly on or after receipt of the applicable Royalty Report. Eisai will pay such invoice pursuant to Section 7.8. In no case shall any Sales Milestone Payment be paid more than once.
|
|
Annual Net Sales in the Territory |
Payment Amount |
25
Page of SECTIONPAGES \* MERGEFORMAT 55
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
7.4Royalty Payments. During the Royalty Term, Eisai will pay Nuvation the following royalties on the aggregate Net Sales of all Licensed Product(s) in the Territory (the “Royalty Payment”). Royalties owed to Nuvation will be calculated for all Licensed Product(s) sold in the Territory in aggregate. Nuvation shall issue an invoice for the Royalty Payment to Eisai promptly after receipt of the Royalty Report and Eisai will pay such invoice pursuant to Section 7.8.
|
|
Net Sales |
Royalty Rate |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
7.5Term of Royalty Payments. On a Licensed Product-by-Licensed Product and country-by-country (of the Territory) basis, Eisai’s obligation to pay royalties pursuant to Section 7.4 shall begin on the date of the First Commercial Sale of such Licensed Product in such country and continuing until the later of [***] (the “Royalty Term”). [***].
7.6.1[***]. On a Licensed Product-by-Licensed Product and country-by-country basis, [***], the royalty rate applicable to Net Sales of such Licensed Product in such country shall be reduced by [***] for the remainder of the Royalty Term in such country in respect of such Licensed Product.
7.6.2[***]. On a Licensed Product-by-Licensed Product and country-by-country basis, in the event a Licensed Product is generating Net Sales in a country in the Territory during the Royalty Term at a time when such Licensed Product [***] in such country, then the royalty rate applicable to Net Sales of such Licensed Product in such country shall be reduced by [***].
7.6.3Reduction for Third Party Licenses. Notwithstanding Nuvation’s obligations under Section 2.3 and without prejudice to any other remedies available to Eisai at law or in equity, on a Licensed Product-by-Licensed Product and country-by-country basis, if Eisai or any of its Affiliates or Sublicensees obtains a Third Party License for Third Party IP that is (a) [***].
7.6.4Royalty Floor. Notwithstanding the application of Section 6.3, Section 7.6.1, Section 7.6.2, Section 7.6.3, or Section 12.7, in no case shall the effective rate of the Royalty Payment payable to Nuvation be less than [***] of Net Sales while royalties are payable by
26
Page of SECTIONPAGES \* MERGEFORMAT 55
Nuvation under the DS License Agreement (“Royalty Floor Period”). For clarity, if Nuvation and Daiichi Sankyo enter into any amendments pursuant to the DS License Agreement and as a result of such amendment the royalty term under the DS License Agreement is extended, this Section 7.6.4 shall not apply during any such extended royalty term.
7.6.5Set Off. Each Party has the right at all times to retain and set off against all amounts due and owing to the other Party as determined in accordance with Article 13 or as otherwise agreed between the Parties. If the damages to be set off are expressed in different currencies, the applicable Party may convert such damages in accordance with Section 7.10. Any exercise by a Party of its rights under this Section 7.6.5 shall not limit or affect any other rights or remedies available to it under this Agreement or otherwise.
7.7Royalty Reports. Within [***] calendar days after the close of the Calendar Quarter during which the corresponding Net Sales are recognized, Eisai will deliver a report (a “Royalty Report”) specifying in the aggregate: (a) Net Sales; (b) royalties payable; and (c) where applicable, Sales Milestone Payments solely in respect of the Royalty Report for the Calendar Quarter ending March 31. Eisai will deliver a forecast of Net Sales and royalties to be paid to Nuvation for each of the next [***] Calendar Quarters within [***] calendar days from the beginning of the Fiscal Year. Eisai shall send the first such report within [***] calendar days of receiving the first Marketing Approval in the Territory which will include forecast Net Sales for the next [***] Calendar Quarters, provided always that such forecast reports will be non-binding in nature.
7.8Payment Schedule. Unless otherwise provided herein, all payments under this Agreement, including the Regulatory Milestone Payment, all Sales Milestone Payments and all Royalty Payments, will be due after [***] calendar days of receipt of the invoice by Eisai from Nuvation except that the Upfront Payment shall be due after [***] Business Days of receipt of the invoice by Eisai from Nuvation. For clarity, all invoices may be delivered electronically by email.
7.9Payment Method. All payments due to Nuvation under this Agreement will be made by bank wire transfer in immediately available funds to an account shown in invoices to be issued to Eisai. All payments hereunder shall be made in euros. All references to “€” or “Euros” herein refer to the currency administered by the European Central Bank and the Eurosystem. Eisai shall be responsible for paying all transfer and other fees related to completing all bank wire transfers required under this Agreement, except for the transfer fee imposed by the bank designated by Nuvation.
7.10Currency Conversion. Unless otherwise agreed by the Parties in writing, if any currency conversion is required in connection with the calculation of amounts payable hereunder, such conversion will be made using the average of the buying and selling exchange rate for conversion of the foreign currency and Euros, quoted for current transactions reported in [***] for the last Business Day of each month of the Calendar Quarter to which such payment pertains.
7.11Late Payments. Either Party shall pay interest to the other Party on the aggregate amount of any payments that are not paid on or before the date such payments are due under this Agreement at a rate per annum (but with such annual interest accruing on a monthly basis) equal to the [***] for the applicable period, to the extent permitted by Applicable Law, calculated on the
27
Page of SECTIONPAGES \* MERGEFORMAT 55
number of days such payment is delinquent. This Section 7.11 will in no way limit any other remedies available to the other Party.
7.12.1Withholding Taxes. If Eisai is required to withhold any tax to the tax or revenue authorities in the Territory regarding any payment to Nuvation, such amount may be deducted from the payment to be made by Eisai, provided that Eisai takes all reasonable and lawful actions to avoid or minimize such withholding and promptly notifies Nuvation so that Nuvation may also take lawful actions to avoid or minimize such withholding. Eisai will promptly furnish Nuvation with copies of any tax certificate or other documentation evidencing such withholding, as necessary to enable Nuvation to support a claim, if permissible, for income tax credit for any amount so withheld. Each Party agrees to cooperate with the other Party in claiming exemptions from such deductions or withholdings under any agreement or treaty in effect from time to time.
7.12.2Indirect Taxes. All amounts to be paid by Eisai to Nuvation pursuant to this Agreement shall be exclusive of any indirect taxes (such as value added tax, sales tax, consumption tax and other similar taxes) occurred due to the payment of such amount, which will be paid by Eisai upon receipt of a valid indirect tax invoice. For the avoidance of doubt, the invoice amount shall be inclusive of the payment amount under this Agreement and the indirect taxes, and irrespective of whether the sums may be netted for settlement purposes.
7.13Records. Eisai will keep and will cause its Affiliates and its or their Sublicensees, if any, to keep, complete, true and accurate books of accounts and records, in compliance with Applicable Law and the terms and conditions of this Agreement, sufficient to determine and establish the calculation of Net Sales and royalties payable under this Agreement for a period of [***] years after the year in which the sale of the Licensed Product(s) generating the same occurred.
7.14Inspection of Records. At the request of Nuvation upon reasonable notice to Eisai, its Affiliates and its or their Sublicensees, will permit an independent certified public accountant appointed by Nuvation and reasonably acceptable to Eisai, to inspect the books and records described in Section 7.13, provided that such inspection shall not be more than once each Calendar Year, and the books and records for any given time period shall not be inspected more than once within a Calendar Year. Nuvation shall submit a reasonable audit plan, including audit scope, to Eisai for review and comment prior to audit implementation. Any inspection conducted under this Section 7.14 will be at Nuvation’s expense, unless such inspection reveals any underpayment of [***] or more of any amount due to Nuvation during the audited period, in which case all reasonable external costs of such inspection will be paid by Eisai. If the independent certified public accountant determines that additional royalties or milestones are owed under this Agreement, during such period, Eisai will pay Nuvation the additional royalties or milestones, within [***] calendar days from the date of receipt of Nuvation’s invoice for the additional royalties or milestones based on the independent certified public accountants’ written report received by Eisai; provided, that, if the independent certified public accountant determines that the royalties or milestones were overpaid, during such period, Nuvation will pay Eisai the amount of such overpayment within [***] calendar days of the date the independent certified public accountants’ written report is received by Nuvation.
28
Page of SECTIONPAGES \* MERGEFORMAT 55
7.15Compliance Audit. Nuvation shall have the right, not more than once per Calendar Year, upon [***] Business Days’ (or such other longer period mutually agreed between the Parties in writing) prior written notice and during normal business hours, to audit all applicable functions operated by: [***]; and such audit shall be of the protocols and procedures used (or proposed to be used) by such entities to Develop or Commercialize (as applicable) the Licensed Product(s) pursuant to its rights and obligations under this Agreement, including, but not limited to, any product quality maintenance, product security, storage, transportation, distribution, and the materials and equipment used therein (in addition to the associated protocols and procedures). Nuvation may be accompanied by any independent technical experts or consultants that Nuvation deems appropriate, provided that such persons are subject to confidentiality obligations no less restrictive than those set forth in this Agreement. Eisai shall provide Nuvation with reasonable assistance in performing such audit, including access to key associated personnel to discuss any associated queries of Nuvation. Eisai shall not charge any costs or expenses to Nuvation for any such audit. Nuvation is responsible for its direct costs of performing such audits. If any items are identified as requesting follow-up action, then Eisai shall respond to Nuvation in writing within [***] days (or such other longer period mutually agreed between the Parties in writing) following receipt of such request by Nuvation, and the response shall state actions Eisai plans to take to correct any such matters and the proposed timing(s) in relation thereto. Notwithstanding the foregoing limitation to performing audits once during each calendar year, Nuvation may perform more than one audit in a Calendar Year, (a) in order for Eisai to demonstrate that any items requiring follow-up action have been resolved or (b) in the event Nuvation has any reasonable quality or compliance-related concerns with respect to Eisai’s performance in the Development or Commercialization of the Licensed Product(s) and is able to provide written evidence demonstrating the same.
8.1Ownership of Intellectual Property Rights. Subject to the licenses and other rights granted herein, as between the Parties, each Party will retain all right, title and interest in and to, and ownership or rights of, all Patents and other Intellectual Property Rights conceived, discovered, developed, reduced to practice, or otherwise made by or on behalf of such Party (or its Affiliates, or its or their licensees or Sublicensees, as applicable) and not by or on behalf of the other Party (or its Affiliates, or its or their licensees or Sublicensees, as applicable). Each Party shall promptly disclose to the other Party any Invention or other material Intellectual Property Rights related to the Licensed Compound or Licensed Products which such first Party acquires, generates, develops, devises, conceives, or first reduces to practice or writing in connection with the performance of its obligations and exercise of its rights under this Agreement during the Term. Inventorship of Inventions and other Intellectual Property Rights created, developed, conceived or reduced to practice after the Effective Date under this Agreement will be determined under the intellectual property laws of the jurisdiction where such creation, development, conception, discovery or making occurs, and ownership of such Inventions and other Intellectual Property Rights shall follow inventorship. For clarity, (a) any such Inventions or other Intellectual Property Rights created, developed, conceived or reduced to practice after the Effective Date under this Agreement both by (or on behalf of) Nuvation (or its Affiliates) and Eisai (or its Affiliates) jointly and (b) any Controlled Patents claiming, disclosing, or otherwise Covering such Inventions or Know-How (“Joint Patents” and together with the Inventions and other Intellectual Property Rights described in clause (a), the “Joint IP”), in each case ((a) and (b)), shall be jointly owned
29
Page of SECTIONPAGES \* MERGEFORMAT 55
by the Parties. Each Party shall promptly disclose to the other Party in writing, and shall cause its Affiliates to so disclose, the development, making, conception or reduction to practice of any Joint IP. Subject to the licenses granted in this Agreement, each Party is entitled to practice the Joint IP for all purposes on a worldwide royalty-free basis, and to license such Joint IP through multiple tiers, without consent of the other Party (where consent is required by Applicable Law, such consent is deemed hereby granted) and without a duty of accounting to the other Party.
8.2Filing, Prosecution and Maintenance.
8.2.1Licensed Patents in the Territory.
(a)Prosecution and Extension. Using counsel mutually agreed by both Parties, [***] shall be responsible, at its sole expense, for Prosecuting and Extending the Licensed Patents in the Territory. As of the Effective Date, the Parties hereby acknowledge and agree that [***], and their foreign counterparts and, to the extent that they have been instructed in respect of the relevant Licensed Patent as at the Effective Date, correspondent firms are approved by the Parties to serve as counsel for Prosecuting and Extending the Licensed Patents in the Territory.
(b)Notification and Consultation. [***] shall consult with [***] and keep [***] reasonably and promptly informed of the status of such Prosecuting and Extending activities, shall provide all material correspondence from any Governmental Authority in connection therewith within [***] weeks of receipt of such correspondence and shall consider in good faith [***] views on overall patent portfolio strategy for the Licensed Products. [***] shall provide [***] with drafts of all proposed material filings and correspondence to any Governmental Authority with respect to the Licensed Patents in the Territory for [***] review and comment at least [***] weeks prior to the submission of any proposed filings or correspondence. [***] shall confer with [***] and consider in good faith [***] comments prior to submitting such filings or correspondence.
(c)Abandonment. If [***] decides that it will no longer Prosecute or Extend a Licensed Patent, [***] will promptly give [***] written notice of its decision (and, in any event, such notice must be given at least [***] calendar days in advance of any filing or payment deadline, or any expiration, termination or other relevant deadline that requires action (“Abandonment Deadline”)) and, upon the request of [***], will discuss in good faith such determination. [***] shall take into account any reasonable input from [***] regarding the decision to cease Prosecution or Extension of any Licensed Patent, provided that [***] shall have final authority to determine whether to continue to Prosecute such Licensed Patent. Upon [***] determination not to continue to Prosecute or Extend such Licensed Patent such Licensed Patent, (an “Abandoned Licensed Patent”), [***] shall use Commercially Reasonable Efforts to allow [***], at its option, to assume control over and continue Prosecuting or Extension of such Abandoned Licensed Patent [***] at [***] sole cost and expense. If [***] assumes control over the Prosecution or Extension, (a) [***] shall have the right, but no obligation, to use counsel of its choice Prosecute or Extend any Abandoned Licensed Patent(s) (for example, in defending such Abandoned Licensed Patent(s) from any challenges to their validity or enforceability, including responding to oppositions, reissue or reexamination proceedings, or interferences, brought by any Third Party, whether before a patent authority or judicial body) in the Territory and (b) [***] will take such action as may be reasonably requested by [***], including providing reasonable
30
Page of SECTIONPAGES \* MERGEFORMAT 55
assistance to [***] and executing any documents as may reasonably be required and delivering to [***] copies of all files related to such Abandoned Licensed Patent(s), for [***] to Prosecute or Extend the Abandoned Licensed Patents. Abandoned Licensed Patents shall not be used to calculate the Royalty Term under Section 7.5.
(d)New Licensed Patents. [***] shall share an up-to-date version of Schedule 1.78 with [***] promptly after it obtains such new Licensed Patents.
(e)Registration of Licenses and Assignments. [***] shall, at its sole cost, register:
(i)any license granted to Nuvation of a Licensed Patent from another Third Party patent holder; and
(ii)any assignment to Nuvation of a Licensed Patent from another Third Party,
at the relevant patent offices in each jurisdiction within the Territory (a) that is set forth in Schedule 8.2.1(e) promptly following the Effective Date, (b) promptly where the Parties acting reasonably and in good faith mutually agree such registration is required by Applicable Law, or (c) promptly upon [***] reasonable request where required for [***] to institute an infringement suit, initiate administrative proceedings, or take other appropriate action against a Third Party for any alleged infringement of any Licensed Patents or recover damages or costs in connection with such action.
(f)Registration of Licenses Granted to Eisai under this Agreement. [***] shall take such action as may be reasonably requested by [***], including providing reasonable assistance to [***] and executing any documents as may reasonably be required, for [***] to register any license granted to Eisai under this Agreement. All such assistance shall be taken at [***] cost save that [***] shall pay [***] Out-of-Pocket Costs incurred by [***] in providing such assistance.
8.2.2Licensed Patents Outside of the Territory. As between the Parties, Nuvation shall (a) have the sole right, at its sole expense, to Prosecute and Extend the Outside of Territory Patents and (b) have the first right, at its sole expense, to Prosecute and Extend the Joint Patents outside of the Territory. Nuvation shall keep Eisai reasonably and promptly informed of the status of Prosecution and Extension, if any, of the Outside of Territory Patents. Nuvation shall keep Eisai reasonably and promptly informed of the status of Prosecution and Extension, if any, of the Joint Patents outside of the Territory and provide Eisai a reasonable opportunity to review and comment on all proposed Prosecuting or Extending activities with respect to the Joint Patents outside of the Territory where such activities may have a material effect on Eisai’s rights under this Agreement or Manufacture, Development and Commercialization of the Licensed Product(s) in the Field and in the Territory. Nuvation shall consult with Eisai and consider all of Eisai’s comments on such proposed activities in good faith.
31
Page of SECTIONPAGES \* MERGEFORMAT 55
8.2.3For the avoidance of doubt, nothing in this Section 8.2 will be interpreted to limit or otherwise impair Nuvation’s right to compromise or settle any indemnified claim controlled by Nuvation in accordance with Section 11.1.
(a)Prosecution and Extension. Eisai shall be responsible, at its sole expense, for Prosecuting or Extending the Eisai Patents in and outside of the Territory.
(b)Notification and Consultation. Eisai shall consult with Nuvation and keep Nuvation reasonably and promptly informed of the status of such Prosecuting and Extending activities, shall provide all material correspondence from any Governmental Authority in connection therewith within [***] weeks of receipt of such correspondence and shall consider in good faith Nuvation’s views on overall patent portfolio strategy for the Licensed Products. Eisai shall provide Nuvation with drafts of all proposed material filings and correspondence to any Governmental Authority with respect to the Eisai Patents outside the Territory for Nuvation’s review and comment at least [***] weeks prior to the submission of any proposed filings or correspondence. Eisai shall confer with Nuvation and consider in good faith Nuvation’s comments prior to submitting such filings or correspondence.
(c)Abandonment. If Eisai decides that it will no longer Prosecute or Extend an Eisai Patent, Eisai will give Nuvation notice of its decision reasonably in advance of any expiration, termination or other relevant deadline sufficient for Nuvation to take any necessary actions to preserve the rights in such Eisai Patent (and, in any event, such notice must be given at least [***] calendar days in advance of the Abandonment Deadline), and, upon the request of Nuvation, Eisai will discuss in good faith such determination. Eisai shall take into account any reasonable input from Nuvation regarding the decision to cease Prosecution or Extension of any Eisai Patent, provided that Eisai shall have final authority to determine whether to continue to Prosecute or Extend such Eisai Patent in its applicable territory. Upon Eisai’s determination not to continue to Prosecute or Extend such Eisai Patent (such Eisai Patent, an “Abandoned Eisai Patent”), Nuvation shall have the right, at its option, to assume control over and continue Prosecuting or Extending such Abandoned Eisai Patent, at Nuvation’s sole expense. If Nuvation assumes control over the Prosecution or Extension such election, (a) Nuvation shall have the right, but no obligation, to defend any Abandoned Eisai Patent(s) from any challenges to their validity or enforceability, including responding to oppositions, reissue or reexamination proceedings, or interferences, brought by any Third Party, whether before a patent authority or judicial body in the Territory; (b) Eisai will reasonably cooperate with such other Party, including providing reasonable assistance to such other Party (including executing any documents as may reasonably be required), in the defense of Abandoned Eisai Patents that Cover the Licensed Compound or Licensed Product(s) and (c) such Abandoned Eisai Patent shall be treated as a Licensed Patent and the Parties’ respective rights and obligations under this Agreement in respect of Licensed Patents shall apply to such Abandoned Eisai Patent, other than those respective rights and obligations set forth under Article 7. For clarity, Abandoned Eisai Patents shall not be used to calculate the Royalty Term under Section 7.5. For the avoidance of doubt, nothing in this Section 8.2.4(b) will be interpreted to limit or otherwise impair Eisai’s right to compromise or settle any indemnified claim controlled by the Eisai in accordance with Article 11.
32
Page of SECTIONPAGES \* MERGEFORMAT 55
8.3Defense of Infringement Claims by Third Parties.
8.3.1Except to the extent a Third Party files an indemnifiable claim pursuant to which Nuvation is obligated to Indemnify [***], if a Third Party files or threatens to file an infringement claim against either Party or both Parties related to the Licensed Compound or a Licensed Product(s) in the Territory, (a) the Party that became aware of such claim or threatened claim shall promptly notify the other Party and the Parties shall promptly meet to consider such claim or threatened claim and the appropriate course of action and (b) [***] shall have the right but no obligation to lead the defense of any such claim, at its sole expense, and with legal counsel mutually agreed by both Parties. Should [***] reasonably and in good faith decide not to actively defend or fail to defend any such claim, suit, or proceeding by a Third Party relating to any Licensed Compound or Licensed Product in the Territory within [***] calendar days of notice of such claim, suit, or proceeding, and in any case at least [***] calendar days in advance of any filing or payment deadline, or other relevant deadline that requires action in order to adequately defend such claim, then [***] will be entitled to take over, at its option, the defense of such proceedings. If [***] is the leading Party in any such claim, suit or proceeding, [***] shall reimburse [***] of [***] costs and expenses incurred in respect of such claim, suit or proceeding, provided that [***] shall pay [***] of any amount it recovers from a Third Party claimant in respect of such claim, suit or proceeding until [***] contributions have been fully repaid and any remaining recovery shall be retained by [***] one hundred percent (100%).
8.3.2If the leading Party finds that the other Party’s assistance is necessary or desirable to any such suit, such other Party may cooperate in such defense at [***] sole cost and expense. The leading Party shall consider any comments from the other Party in good faith in relation to the defense of such claim. Each Party shall reasonably cooperate with the Party conducting the defense of the claim. Each Party shall keep the other Party reasonably informed of all material developments in connection with any such claim, suit, or proceeding, and the Parties shall reasonably cooperate in conducting the defense of any such claim and may, if appropriate, agree on and enter into a common interest agreement. Neither Party shall enter into any settlement in relation to such claim in a manner that imposes any Out-of-Pocket Costs, obligations or liability on, or involves any admission by or materially adversely affects, the other Party without such other Party’s prior written consent, not to be unreasonably withheld, conditioned or delayed. Neither Party shall make any admission of liability, agreement or compromise in relation to such claim without such other Party’s prior written consent, not to be unreasonably withheld, conditioned or delayed.
8.4Enforcement Actions Against Third Parties.
8.4.1Notification. If either Party learns of any infringement, unauthorized use, misappropriation or ownership claim, or threatened infringement of any Licensed Patents, Outside of Territory Patents or Eisai Patents by a Third Party with respect to the Licensed Compound or Licensed Product(s) anywhere within or outside of the Territory (respectively a “Licensed Patent Infringement”, an “Outside of Territory Patents Infringement”, and an “Eisai Patent Infringement”), such Party will promptly notify the other Party in writing and will promptly provide the other Party with available evidence of such infringement or other such claim.
33
Page of SECTIONPAGES \* MERGEFORMAT 55
8.4.2Control; Reimbursement of Expenses.
(a)Licensed Patents. As between the Parties and using counsel mutually agreed by both Parties, [***] will have the first right, but not the obligation, to institute an infringement suit, initiate administrative proceedings, or take other appropriate action against a Third Party for any alleged infringement of any Licensed Patents anywhere within the Territory and [***]. [***], upon [***] request, shall assist and cooperate in such enforcement, including by providing any information regarding technical details or other knowledge related to the Commercialization of the Licensed Compound or any Licensed Product in the Territory, at [***] sole cost and expense. [***] shall provide [***] with drafts of all proposed submissions to any court with respect to the Licensed Patent Infringement for [***] review and comment at least [***] weeks prior to the filing of such proposed submission. [***] shall confer with [***] and consider in good faith [***] comments prior to submitting such submission. If [***] (i) elects not to bring such legal action against a Licensed Patent Infringement (the decision of which [***] shall inform [***] promptly and in no event more than [***] days following the notification of the Licensed Patent Infringement under Section 8.4.1) or (ii) [***] otherwise fails to bring such legal action against a Licensed Patent Infringement within [***] days following the notification of the Licensed Patent Infringement under Section 8.4.1, then as between the Parties, [***] shall have the right to bring and control any legal action in connection with such Licensed Patent Infringement.
(b)Eisai Patents. Using counsel mutually agreed by both Parties, [***] will have the first right, but not the obligation, to institute an infringement suit, initiate administrative proceedings, or take other appropriate action against a Third Party for any alleged infringement of any Eisai Patents anywhere within or outside of the Territory. Upon [***] request, [***] shall assist and cooperate in such enforcement, including by providing any information regarding technical details or other knowledge related to the Licensed Compound or any Licensed Product in the Territory, at [***] sole cost and expense. [***] shall provide [***] with drafts of all proposed material submissions to any court with respect to the Eisai Patent Infringement for [***] review and comment at least [***] weeks prior to the filing of such proposed submission. [***] shall confer with [***] and consider in good faith [***] comments prior to submitting such submission. If [***] (i) elects not to bring such legal action against an Eisai Patent Infringement (the decision of which [***] shall inform [***] of promptly and in no event more than [***] days following the notification of the Eisai Patent Infringement under Section 8.4.1) or (ii) [***] otherwise fails to bring such legal action against an Eisai Patent Infringement within [***] days following the notification of the Eisai Patent Infringement under Section 8.4.1 [***] shall have the right to bring and control any legal action in connection with such Eisai Patent Infringement at its sole cost and expense as it reasonably determines appropriate after consultation with [***].
(c)Damages. Any damages paid by Third Parties as a result of such an enforcement action (whether by way of settlement or otherwise) in relation to a Licensed Patent Infringement or Eisai Patent Infringement will be [***]:
(i)Licensed Patent Infringement. [***].
(ii)Eisai Patent Infringement in the Territory. With respect to Eisai Patent Infringement in the Territory, (A) if [***] is the enforcing Party, [***] and (B) if [***]
34
Page of SECTIONPAGES \* MERGEFORMAT 55
is the enforcing Party, such remaining amount will be allocated [***]to Nuvation and [***] to Eisai.
(iii)Eisai Patent Infringement outside the Territory. With respect to Eisai Patent Infringement outside the Territory, such remaining amount will be allocated [***] to the enforcing Party and [***] to the non-enforcing Party.
(d)Information. In relation to the enforcement of any Licensed Patent Infringement or any Eisai Patent Infringement, the enforcing Party shall keep the non-enforcing Party informed of the status of such proceedings and, upon request, shall (to the extent permitted under Applicable Law) provide the non-enforcing Party, at no cost to the non-enforcing Party, with copies of all documents filed in, and all material written communications between the parties to such proceedings. In relation to the enforcement of any Outside of Territory Patent Infringement, Nuvation shall keep Eisai reasonably and promptly informed of the status of such proceedings.
8.5.1Eisai will have the right to decide, at its sole discretion, the brand name and brand logo under which the Licensed Product(s) will be marketed in the Territory, and the right to, at its own expense, apply for, register, maintain or abandon the trademark(s) corresponding the said brand name and brand logo in the Territory. Upon Eisai’s reasonable request and Nuvation’s consent (not to be unreasonably withheld, conditioned, or delayed), Nuvation will reasonably assist Eisai in connection with Eisai’s activities under this Section 8.5, provided that Eisai will promptly reimburse [***] of Nuvation’s Out-of-Pocket Costs incurred in providing the foregoing assistance.
8.5.2Except for any trademarks comprising the Other Nuvation IP, Eisai will own all trademarks corresponding to the said brand name and brand logo used to market the Licensed Product(s) in the Territory (excluding ones identical or similar to Other Nuvation IP).
8.5.3If Eisai intends to use a trademark set forth in Schedule 1.79, then Eisai shall provide written notice to Nuvation and Eisai shall not use any such trademark until the JCC has established and approved a style guide for such trademark that comports with Nuvation’s existing trademark usage policy. Once the JCC has established and approved such a style guide, then Eisai may use such trademark under the license granted in Section 2.1.1 in accordance with such style guide, and with respect to any such trademark, Nuvation shall (a) take reasonable steps to diligently maintain any existing trademark registration set forth in Schedule 1.79 and (b) diligently prosecute to registration any pending application as set forth in Schedule 1.79, provided that, Eisai will promptly reimburse [***] of Nuvation’s reasonably incurred Out-of-Pocket Costs in relation to the activities described in (b). Upon Eisai’s reasonable written request, Nuvation shall (i) diligently prosecute to registration, using counsel mutually agreed between the Parties, other trademark registrations requested by Eisai for trademark(s) that comprise the Other Nuvation IP in the Territory and thereafter (ii) take all reasonable steps to diligently maintain any and all such trademark registrations, provided that, in each case ((i) and (ii)), Eisai will promptly reimburse [***] of Nuvation’s reasonably incurred Out-of-Pocket Costs. Nuvation shall provide Eisai with copies of receipts for renewal fees in respect of any such trademark registrations on Eisai’s request.
35
Page of SECTIONPAGES \* MERGEFORMAT 55
8.6Recordal. Nuvation shall, to the extent required under Applicable Law and when requested by Eisai, at its own cost, record licenses to trademarks comprised in Other Nuvation IP granted under Section 2.1 and Section 2.2 in the relevant registries in the Territory and Eisai shall provide reasonable assistance to enable Nuvation to do so.
8.7Domain Names. If, at any time after the Effective Date, Eisai identifies a domain name comprising the Other Nuvation IP which Eisai intends to use, Nuvation shall, upon Eisai’s written request, negotiate in good faith to agree and enter into an agreement effecting the transfer of such domain name to Eisai. Nuvation shall take all actions reasonably necessary to effect such transfer, and Eisai shall reimburse Nuvation for [***] Out-of-Pocket Costs reasonably incurred by Nuvation in connection with such transfer.
9.1Confidential Information. Except to the extent expressly authorized by this Article 9 or otherwise agreed in a writing signed by both Parties, each Party (the “Receiving Party”) shall, during and [***] years after the Term of this Agreement, keep confidential and not publish or otherwise disclose or use for any purpose other than as explicitly provided for in this Agreement any confidential and proprietary information or materials, patentable or otherwise, in any form (written, oral, photographic, electronic, magnetic, or otherwise) that are disclosed or provided to a Receiving Party, its Affiliate or Sublicensee, as applicable, by the other Party or an Affiliate or Sublicensee of the other Party (each a “Disclosing Party”) in the course of performing its obligations under this Agreement or otherwise in connection with this Agreement, including, but not limited to, any trade secrets, Know-How, product specifications, formulae, processes, techniques and information relating to the Disclosing Party’s past, present and future marketing, financial, and research and development activities for any product of the Disclosing Party and the pricing and customer lists, customer data and other customer information thereof (collectively, “Confidential Information”). Confidential Information of each Party includes the terms and conditions of this Agreement.
9.2Exceptions. Notwithstanding the foregoing, Confidential Information does not include information or materials to the extent that it can be established by the Receiving Party that such information or material:
9.2.1is already lawfully known to the Receiving Party, other than under an obligation of confidentiality at the time of disclosure by the Disclosing Party as evidenced by written records kept in the ordinary course of business, or other documentary proof of actual use by the Receiving Party;
9.2.2is generally available to the public at the time of its disclosure to the Receiving Party;
9.2.3becomes generally available to the public after its disclosure to the Receiving Party and other than through any act or omission of the Receiving Party, its Affiliates, or, with respect to Eisai, its Sublicensees in violation of this Agreement;
36
Page of SECTIONPAGES \* MERGEFORMAT 55
9.2.4is independently developed by the Receiving Party as demonstrated by documented evidence prepared contemporaneously with such independent development; or
9.2.5is lawfully disclosed to the Receiving Party, other than under an obligation of confidentiality, by a Third Party who had no obligation not to disclose such information to others.
9.3Authorized Disclosure.
9.3.1Notwithstanding Section 9.1, the Receiving Party may disclose Confidential Information of the Disclosing Party:
(a)to its respective employees, officers, directors, consultants, and advisors, and to the employees, officers, directors, consultants, and advisors of such Receiving Party’s Affiliates, who have a need to know such Confidential Information to the extent such disclosure is reasonably necessary in connection with performing its obligations or exercising its rights under this Agreement and have an obligation to treat such Confidential Information as confidential under terms no less restrictive than those set forth herein;
(b)in the case of Nuvation, to its (sub)licensees (including Sublicensees) who have a need to know such Confidential Information to the extent such disclosure is reasonably necessary in connection with such (sub)licensee performing any activities on behalf of Nuvation in connection with its obligations under this Agreement and have an obligation to treat such Confidential Information as confidential under terms no less restrictive than those set forth herein. For clarity, Nuvation shall not be permitted to disclose Eisai’s Confidential Information to any (sub)licensees that are not engaged in performing activities on behalf of Nuvation in connection with its obligations under this Agreement;
(c)in the case of Eisai, to its (sub)licensees (including Sublicensees) or potential (sub)licensees who have a need to know such Confidential Information to the extent such disclosure is reasonably necessary in connection with performing its obligations or exercising its rights under this Agreement and have an obligation to treat such Confidential Information as confidential under terms no less restrictive than those set forth herein;
(d)as required by any court or other Governmental Authority or as otherwise required by Applicable Law (including, solely with respect to the terms of this Agreement, any such disclosures as are required by the rules or regulations of the United States Securities and Exchange Commission or similar regulatory authority in a country other than the United States or of any stock exchange or listing entity); provided, that, the Receiving Party provides the Disclosing Party with prompt notice of such requirement so that, to the extent legally permissible, the Disclosing Party may seek a protective order or other appropriate remedy, then the Receiving Party may furnish only that portion of the Confidential Information which the Receiving Party is legally compelled to disclose;
(e)to Governmental Authorities to facilitate the issuance of Marketing Approvals for Licensed Product; provided that reasonable measures are taken to assure confidential treatment of such information;
37
Page of SECTIONPAGES \* MERGEFORMAT 55
(f)to Regulatory Authorities where such Confidential Information is required by Regulatory Authorities in connection with any Regulatory Filing, so long as such Regulatory Filing is permitted by and made in accordance with the terms of this Agreement; provided that reasonable measures shall be taken to assure confidential treatment of such information to the extent practicable and consistent with Applicable Law; and
(g)to a patent authority as may be reasonably necessary for purposes of obtaining or enforcing a Patent, so long as obtaining or enforcing such Patent is permitted by and done in accordance with the terms of this Agreement; provided, however, that reasonable measures shall be taken to assure confidential treatment of such information to the extent practicable.
9.3.2Notwithstanding Section 9.1 and without limiting Section 9.3.1, each Party may disclose the terms and conditions of this Agreement:
(a)to bona fide potential acquirors, existing and bona fide potential investors, existing and bona fide potential lenders, provided that such information is disclosed only on a need-to-know basis and under obligations of confidentiality and non-use that are at least as stringent as those obligations of confidentiality and non-use of the set forth in this Article 9 (which confidentiality and non-use obligations shall subsist for a period that is commercially reasonable for the acquisition, investment, or loan being contemplated (in any event no less than [***] years following the disclosure) and requires the destruction of all tangible materials comprising, bearing, or containing any Confidential Information of the Disclosing Party that are such potential lenders’ possession or control and provide written certification of such destruction); and
(b)to existing or bona fide potential strategic partners, collaborators, or (sub)licensees (including Sublicensees), provided that (i) such information is disclosed only on a need-to-know basis and under confidentiality provisions at least as stringent as those in this Agreement; (ii) any and all commercially sensitive information is redacted from the copy of the Agreement to be disclosed (such redactions to be approved by the other Party) unless there is a requirement under Applicable Law or prior written consent is given by the other Party, to disclose an unredacted copy of the Agreement; (iii) access to such redacted version of the Agreement shall be made via a secure and encrypted data room; and (iv) access to such redacted version of the Agreement shall cease in the event the proposed transaction to which the requirement to disclose the terms and conditions of the Agreement terminates or does not proceed.
9.4Receiving Party’s Obligations for Authorized Disclosure. Any authorized disclosures under Section 9.3 shall not affect the Receiving Party’s confidentiality obligations under Section 9.1, and the Receiving Party shall remain bound by such obligations except to the extent permitted under Section 9.3. The Receiving Party shall be liable to the Disclosing Party for any unauthorized disclosure by any Third Party to whom the Receiving Party discloses the Confidential Information pursuant to Section 9.3.1 and Section 9.3.2.
9.5.1Subject to the terms of this Agreement and review and approval by the JCC in accordance with Article 3, each Party shall have the right to participate in and make publications
38
Page of SECTIONPAGES \* MERGEFORMAT 55
at any global congress organized within or outside the Territory which relate to the Licensed Product.
9.5.2Subject to the review and approval by the JCC (including Clinical or Medical Subcommittee if applicable) in accordance with Article 3, each Party shall have the right to make scientific presentations and publications (including, but not limited to, any publication prepared in connection with the performance of Medical Affairs Activities) that disclose data that is either (a) generated entirely within the Territory or (b) generated both within and outside the Territory, in either case ((a) or (b)), with respect to the Licensed Compound and Licensed Product(s) and as is outlined in an annual publications plan that each Party shall provide to the other Party. If a Party intends to make such a scientific presentation or publication, then such Party will provide a copy of such draft presentation or publication to the other Party reasonably in advance (and being no less than [***] Business Days) of such presentation or publication, and the JCC (or the applicable) Subcommittee shall convene to review, discuss, and approve such presentation or publication.
9.5.3As part of the JCC review described in Section 9.5.2, either Party may inform the other Party that (a) the proposed presentation or publication contains the Confidential Information of the non-publishing Party, in which case the publishing Party shall delete such Confidential Information or (b) its proposed presentation or publication could be reasonably expected to have a material adverse effect on any Prosecution of any Licensed Patent or Eisai Patent, in which case the publishing Party shall delay such proposed presentation publication for a reasonable period being no longer than [***] days to permit the timely preparation and first filing of patent application(s) covering the information involved.
9.6Press Releases. The Parties may choose to issue a joint press release on or promptly following the Effective Date, such press release to be in the form set forth in Schedule 9.6. Neither Party may issue any press release relating this Agreement without the other Party’s prior written approval, which approval will not be unreasonably withheld or delayed. If such approval is not granted or denied by a Party within [***] Business Days after a request from the other Party for such approval that accompanies a final draft of the press release, such approval will be deemed granted.
9.7Restrictions on Use. During and [***] years after the Term, the Receiving Party shall not use, and shall ensure that its Affiliates, and its and their licensees or Sublicensees, as applicable, do not use any Confidential Information disclosed to it by a Disclosing Party or otherwise received or accessed in the course of performing its obligations under this Agreement for any purpose other than as expressly provided herein.
9.8Return/Destruction of Confidential Information. Upon request from and at the election of the Disclosing Party, the Receiving Party will return (at Disclosing Party’s expense) or destroy all tangible materials comprising, bearing, or containing any Confidential Information of the Disclosing Party relating to the terminated Licensed Product(s) that are in the Receiving Party’s or its Affiliates’ possession or control and provide written certification of such destruction (except to the extent any information is the Confidential Information of both Parties or to the extent that the Receiving Party has the continuing right to use the Confidential Information under this Agreement, including under the licenses granted under Section 2.1); provided, that, (a) the
39
Page of SECTIONPAGES \* MERGEFORMAT 55
Receiving Party may retain one copy of such Confidential Information for its legal archives mandatorily required by Applicable Law; and (b) Eisai may retain any Clinical Trial data in respect of any Clinical Trial for which it is the named sponsor, for a period of [***] following completion of such Clinical Trial or such other period required by Applicable Law.
10.Representations, Warranties and Covenants.
10.1Representations and Warranties of Both Parties. Each Party represents and warrants to the other, as of the Effective Date, that:
10.1.1it is duly organized and validly existing under the laws of its jurisdiction of incorporation, and has full corporate power and authority to enter into this Agreement and to carry out the provisions hereof;
10.1.2it is duly authorized to execute and deliver this Agreement and to perform its obligations hereunder and that it has the right to grant to the other Party the licenses and sublicenses granted pursuant to this Agreement, and the person or persons executing this Agreement on its behalf has been duly authorized to do so by all requisite corporate action. For the avoidance of doubt, Nuvation warrants that it has obtained written approval from Daiichi Sankyo for the entry into and execution of this Agreement;
10.1.3this Agreement is legally binding upon it and, upon execution by the other Party, shall be enforceable in accordance with its terms except to the extent that enforceability may be limited by applicable bankruptcy, insolvency or other laws affecting the enforcement of creditors’ rights generally and subject to the general principles of equity (regardless of whether enforcement is sought in a court of law or equity);
10.1.4the execution, delivery and performance of this Agreement by such Party (i) does not knowingly conflict with or is not prevented by any agreement, instrument or understanding, oral or written, to which it is a party or by which it may be bound (including the DS License Agreement), nor (ii) violate any material law or regulation of any governmental agency or Regulatory Authority having jurisdiction over it;
10.1.5it has not granted any right to any Third Party that would knowingly conflict with the rights granted to the other Party hereunder;
10.1.6it has not been debarred under the Generic Drug Enforcement Act of 1992 (21 U.S.C. § 301 et seq.), is not under investigation for debarment action, has not been disqualified as an investigator pursuant to 21 C.F.R. § 312.70, does not have a disqualification hearing pending and is not currently employing any person or entity that has been so debarred or disqualified to perform any of its obligations under this Agreement. It shall promptly notify the other Party if it is so debarred or disqualified and shall terminate any so debarred or disqualified individual’s or entity’s participation in the performance of any of its obligations under this Agreement promptly upon its awareness of such debarment or disqualification; and
10.1.7it is not aware of any action, suit or inquiry or investigation instituted by any person or Governmental Authority that questions or threatens the validity of this Agreement.
40
Page of SECTIONPAGES \* MERGEFORMAT 55
10.2Representations, Warranties and Undertakings of Nuvation. Nuvation represents, warrants and undertakes to Eisai, as of the Effective Date:
10.2.1all necessary consents, approvals and authorizations (and all notices to, and filings with) of all Governmental Authorities and Regulatory Authorities and other Third Party (including Daiichi Sankyo) required to be obtained or provided by Nuvation, in connection with the execution, delivery and performance of this Agreement have been obtained and provided;
10.2.2other than any Third Party License that has been obtained by Nuvation as of the Effective Date, Nuvation is not aware of any requirement of a Third Party License for the Commercialization, Manufacture or Development of the Licensed Compound or any Licensed Product for the Initial Indication in the Territory as contemplated as of the Effective Date;
10.2.3except as set forth in Schedule 10.2, there are no legal claims, judgments or settlements against or owed by Nuvation or, written legal claims or pending or threatened litigation against Nuvation, in each case relating to (i) the Licensed IP, or (ii) the Licensed Compound or Licensed Product(s) and their use;
10.2.4Nuvation has not received any notice from a Third Party alleging infringement or misappropriation of the Intellectual Property Rights or other property rights of any Third Party due to the manufacture, use, sale, offer to sell, or import of the Licensed Compound or Licensed Product in the Territory, and to its knowledge, the Licensed Compound, Licensed Product and the practice of Licensed IP will not infringe or misappropriate the intellectual or other property rights of any Third Party;
10.2.5there are no legal claims, judgments or settlements against or owed by Nuvation or pending legal claims or litigation, in each case relating to the Nuvation Patents and to its knowledge, the Nuvation Patents are subsisting, valid and enforceable and Nuvation has not received any notice challenging the inventorship, ownership, validity or enforceability of the Nuvation Patents;
10.2.6Nuvation Controls the Nuvation Patents and has the right to grant to Eisai the rights, license and sublicense that it purports to grant hereunder and has not granted any Third Party rights that would interfere or be inconsistent with Eisai’s rights hereunder;
10.2.7to its knowledge, Nuvation is not aware of any infringement or misappropriation by a Third Party of the Nuvation Patents in the Territory;
10.2.8Schedule 1.78 is a complete and correct list of all Nuvation Patents in the Territory as of the Effective Date;
10.2.9the trademark registrations as set forth in Schedule 1.79 are valid, subsisting and, to Nuvation’s knowledge, enforceable;
10.2.10to Nuvation’s knowledge, the material Nuvation Patents set forth in Schedule 1.78 (a) have been diligently prosecuted in the respective patent offices in the Territory in accordance with Applicable Law, and (b) have been filed and maintained and all applicable fees have been paid;
41
Page of SECTIONPAGES \* MERGEFORMAT 55
10.2.11Nuvation has provided to Eisai a true, correct and complete copy of the DS License Agreement, including any and all amendments, restatements, side letters, and other modifications thereto, as is in effect as of the Effective Date;
10.2.12the DS License Agreement (a) has not been the subject of any breach or default by Nuvation or, to Nuvation’s knowledge any other party to it or of any event which, with the giving of notice or lapse of time, would constitute a default, and (b) is not the subject of any claim, dispute or proceeding, pending or threatened and to Nuvation’s knowledge, there are no circumstances which are likely to give rise to a breach, claim, dispute or proceeding.
10.2.13Nuvation has not prior to the Effective Date filed any Marketing Approval applications for the Licensed Product in the Territory.
10.2.14Nuvation has disclosed to Eisai all Regulatory Authority inspection material findings in respect of the Licensed Compound and/or Licensed Product that, to Nuvation’s reasonable opinion, may materially impact a Regulatory Filing.
10.3Covenants of Nuvation. Nuvation agrees as follows:
10.3.1Nuvation shall, and shall cause its Affiliates to, comply with all Applicable Law with respect to the performance of its or their obligations and the activities envisaged under this Agreement. Without limitation to the foregoing, Nuvation shall, and shall cause each of its Affiliates to, in all material respects, conform its and their practices and procedures relating to the performance of its or their obligations and the activities envisaged under this Agreement to any Applicable Law.
10.3.2Nuvation shall comply with all applicable Anti-Bribery Requirements and shall not make any payment, either directly or indirectly, of money or other benefits or assets to Governmental Authorities or officials or physicians, or make any payment to, or receive any payment from employees or consultants of Eisai, or other related business entities or persons, where it would constitute a violation of Anti-Bribery Requirements. Regardless of its legality, Nuvation shall make no payment either directly or indirectly to the abovementioned Governmental Authorities, entities and persons if such payment is for the purpose of influencing decisions or actions related to the subject matter of this Agreement.
10.3.3Nuvation shall provide reasonable assistance as reasonably requested by Eisai (at Eisai’s expense) to assist Eisai in performing any activity for the purpose of compliance with the applicable Anti-Bribery Requirements or other regulatory requirements to which this Agreement relates.
10.3.4Subject to Section 8.2.1(c), Nuvation shall continue to diligently Prosecute or Extend the Licensed IP during the Term and shall not dispose of any Licensed IP during the Term in a manner that would have a material and adverse impact on the rights granted to Eisai hereunder.
10.3.5Nuvation shall not knowingly or materially breach the terms of the DS License Agreement.
42
Page of SECTIONPAGES \* MERGEFORMAT 55
10.3.6Nuvation shall not terminate the DS License Agreement in the Territory without the prior written consent of Eisai.
10.4Representations and Warranties of Eisai. Eisai represents and warrants to Nuvation, as of the Effective Date:
10.4.1that it is not actively researching, developing, or commercializing a Competing Product in the Territory;
10.4.2that it is in compliance with applicable Anti-Bribery Requirements in the Territory;
10.4.3that no agent, broker, investment banker, financial advisor, firm, individual, corporation, partnership, limited liability company, joint venture, association, trust, unincorporated organization or other entity is or shall be entitled, as a result of this Agreement, to any broker’s, finder’s, financial advisor’s or other similar fee or commission (or reimbursement of expenses) in connection with this Agreement; and
10.4.4all necessary consents, approvals and authorizations (and all notices to, and filings with) of all Governmental Authorities and Regulatory Authorities and other Third Party required to be obtained or provided by Eisai, in connection with the execution, delivery and performance of this Agreement have been obtained and provided.
10.5Covenants of Eisai. Eisai agrees as follows:
10.5.1Eisai shall, and shall cause its Affiliates and the Sublicensees to, comply with all Applicable Law with respect to the performance of its or their obligations and the activities envisaged under this Agreement, including the Commercialization of the Licensed Product(s). Without limitation to the foregoing, Eisai shall, and shall cause each of its Affiliates and the Sublicensees to, in all material respects, conform its and their practices and procedures relating to the performance of its or their obligations and the activities envisaged under this Agreement, including the Commercialization of the Licensed Product(s) to any Applicable Law.
10.5.2Eisai shall comply with all applicable Anti-Bribery Requirements and shall not make any payment, either directly or indirectly, of money or other benefits or assets to Governmental Authorities or officials or physicians, or make any payment to, or receive any payment from employees or consultants of Nuvation, or other related business entities or persons, where it would constitute a violation of Anti-Bribery Requirements. Regardless of its legality, Eisai shall make no payment either directly or indirectly to the abovementioned entities and persons if such payment is for the purpose of influencing decisions or actions related to the subject matter of this Agreement.
10.5.3Eisai shall make Commercially Reasonable Efforts to obtain Marketing Approval of Licensed Products for the Initial Indication.
10.5.4Eisai shall provide reasonable assistance as reasonably requested by Nuvation (at Nuvation’s expense) to assist Nuvation in performing any activity for the purpose of
43
Page of SECTIONPAGES \* MERGEFORMAT 55
compliance with the applicable Anti-Bribery Requirements or other regulatory requirements to which this Agreement relates.
10.5.5Without Nuvation’s prior written consent, Eisai shall not Develop or Commercialize in the Territory a pharmaceutical product which is comprised of the Licensed Compound and one or more additional active pharmaceutical or biological ingredients either (a) co-formulated as a single formulation or (b) co-packaged in a single package or container intended for coordinated use and sold for a single price.
10.6Disclaimer of Warranties. EXCEPT AS EXPRESSLY SET FORTH IN THIS ARTICLE 10, NEITHER PARTY MAKES ANY REPRESENTATIONS OR EXTENDS ANY WARRANTIES OF ANY KIND UNDER THIS AGREEMENT (INCLUDING WITH RESPECT TO ANY MATERIALS PROVIDED UNDER THIS AGREEMENT), EITHER EXPRESS OR IMPLIED, INCLUDING, BUT NOT LIMITED TO, WARRANTIES OF MERCHANTABILITY, QUALITY, FITNESS FOR A PARTICULAR PURPOSE, NON-INFRINGEMENT, OR VALIDITY OF PATENT CLAIMS, WHETHER ISSUED OR PENDING (AS APPLICABLE).
10.7Limitation of Liability. IN NO EVENT SHALL EITHER PARTY BE LIABLE TO THE OTHER PARTY OR ANY OTHER PERSON FOR INCIDENTAL, CONSEQUENTIAL, EXEMPLARY, PUNITIVE, MULTIPLE OR OTHER INDIRECT DAMAGES INCLUDING FOR LOSS OF PROFITS, LOSS OF DATA OR LOSS OF USE, ARISING OUT OF THIS AGREEMENT, WHETHER BASED UPON WARRANTY, CONTRACT, TORT, STATUTE, STRICT LIABILITY OR OTHERWISE, EVEN IF SUCH PARTY HAS BEEN ADVISED OF THE POSSIBILITY OF SUCH DAMAGES OR LOSSES. THE FOREGOING LIMITATION OF LIABILITY, HOWEVER, SHALL NOT APPLY TO: EITHER PARTY’S [***].
11.1Indemnification by Nuvation. Nuvation will defend, hold harmless and indemnify (collectively “Indemnify”) Eisai and its Affiliates, and their respective Sublicensees, Distributors, agents, directors, contractors, representatives, officers and employees (collectively “Eisai Indemnitees”) from and against any liability or expense, including without limitation reasonable legal expenses and attorneys’ fees, (collectively “Losses”) resulting from suits, claims, actions and demands, in each case brought by a Third Party (each, a “Third Party Claim”) to the extent relating to or arising from: (a) the breach by Nuvation of any representation, warranty, covenant or agreement made by Nuvation in this Agreement; (b) Nuvation’s material breach of the DS License Agreement or the termination of the DS License Agreement (in each case, other than due to any act or omission of Eisai or its Affiliates); (c) a claim that (i) the Commercialization of the Licensed Product or (ii) the practice of Licensed IP as authorized under this Agreement, in each case ((i) and (ii)) for the Initial Indication and in the Territory infringes any Intellectual Property Rights, including Patents, of any Third Party; or (d) any gross negligence or willful misconduct by any Nuvation Indemnitee. Nuvation’s obligation to Indemnify the Eisai Indemnitees pursuant to this Section 11.1 will not apply to the extent that any such Losses arise from the negligence, willful misconduct or wrongful acts or omissions of any Eisai Indemnitee or
44
Page of SECTIONPAGES \* MERGEFORMAT 55
are Losses for which Eisai is obligated to Indemnify an Nuvation Indemnitee pursuant to Section 11.2.
11.2Indemnification by Eisai. Eisai will Indemnify Nuvation and its Affiliates, and their respective agents, directors, contractors, representatives, officers and employees (collectively “Nuvation Indemnitees”) from and against any and all Losses resulting from Third Party Claims to the extent relating to or arising from: (a) the breach by Eisai of any representation, warranty, covenant or agreement made by Eisai under this Agreement; (b) any gross negligence or willful misconduct by any Eisai Indemnitee; (c) the exploitation by any Eisai Indemnitee of: (i) the Licensed Compound or (ii) any Licensed Product, in each case ((i) and (ii)) [***] or (d) Losses arising from or relating [***] for Commercialization of the Licensed Compound or any Licensed Product(s) in the Territory. Eisai’s obligation to Indemnify the Nuvation Indemnitees pursuant to this Section 11.2 will not apply to the extent that any such Losses arise from the negligence, willful misconduct or wrongful acts or omissions of any Nuvation Indemnitee, or are Losses for which Nuvation is obligated to Indemnify the Eisai Indemnitees pursuant to Section 11.1.
11.3Procedure. To be eligible to be indemnified hereunder, any Eisai Indemnitee under Section 11.1, or Nuvation Indemnitee under Section 11.2, as the case may be (an “Indemnitee”) seeking indemnification, must provide the indemnifying Party with prompt notice of the Third Party Claim giving rise to the claimed indemnification obligation and must assign the exclusive ability to defend or settle any such claim to the indemnifying Party; provided that any delay or failure to provide such notice shall not constitute a waiver or release of, or otherwise limit, the Indemnitee’s rights to indemnification under Section 11.1 or Section 11.2 as applicable, except to the extent that such delay or failure materially prejudices the indemnifying Party’s ability to defend against the relevant claims. Notwithstanding the foregoing, the indemnifying Party may not enter into any settlement that admits fault, wrongdoing or damages on the part of the Indemnitee without such Indemnitee’s written consent, provided that such consent shall not be unreasonably withheld or delayed. The Indemnitee will cooperate with reasonable requests from the indemnifying Party, at the indemnifying Party’s expense, and will have the right to participate, at its own expense and with counsel of its choice, in the defense of any claim or suit that has been assumed by the indemnifying Party. Without affecting or limiting the indemnifying Party’s right to control the defense of the Third Party Claim, if the Indemnitee elects to engage separate counsel, the Parties shall cooperate in defending or settling such claims.
11.4Allocation. If a claim is based in part on an indemnified claim, as described in Section 11.1 or Section 11.2, and in part on a non-indemnified claim, or is based in part on a claim described in Section 11.1 and in part on a claim described in Section 11.2, any payments and reasonable attorney fees incurred in connection with such claims will be apportioned between the Parties in accordance with the degree of fault attributable to each Party.
11.5Insurance. During the Term and for [***] years thereafter, Nuvation will maintain a policy of insurance at levels sufficient to support its indemnification obligations, but in any case, such insurance must provide adequate coverage for clinical trials liability, products liability, and comprehensive general liability. During the Term and for [***] years thereafter, Eisai will maintain a policy of insurance or the equivalent amount in self-insurance at levels sufficient to support its indemnification obligations and obligations in relation to the Commercialization of the
45
Page of SECTIONPAGES \* MERGEFORMAT 55
Licensed Product(s) in the Territory. Upon the request of either Party, the other Party shall provide evidence of such insurance.
12.1Term. This Agreement is effective as of the Effective Date and, unless earlier terminated pursuant to the other provisions of this Article 12, will continue in full force and effect on a Licensed Product-by-Licensed Product basis until the expiration of the Royalty Term with respect to such Licensed Product (the “Term”). Upon expiration of the Royalty Term with respect to a Licensed Product, but not earlier termination of this Agreement, the licenses granted to by Nuvation to Eisai pursuant to Article 2 with respect to such Licensed Product shall continue and convert into non-exclusive, royalty-free, fully-paid up, irrevocable and perpetual (subject to the provisions of the rest of this Agreement, including Article 12) licenses in the Field in the Territory.
12.2Termination by Nuvation.
12.2.1Nuvation may terminate this Agreement, without prejudice to any other remedies available to it at law or in equity, if Eisai or its Affiliates commit a material breach of this Agreement that, in the case of a material breach capable of remedy, have not been remedied within [***] calendar days (or such other longer period mutually agreed between the Parties in writing) of receiving a notice from Nuvation identifying the breach and requiring its remedy, or if such material breach cannot be cured within such period, Eisai does not commence and diligently continue actions to cure such breach during such period, provided that in no event shall such material breach be permitted to last more than [***] days (or such other longer period mutually agreed between the Parties in writing).
12.2.2To the extent permitted by Applicable Law, Nuvation may terminate this Agreement immediately if: (a) Eisai becomes insolvent, or makes or seeks to make or arrange an assignment for the benefit of creditors; (b) proceedings in voluntary bankruptcy are initiated by or on behalf of Eisai or proceedings in involuntary bankruptcy are initiated against Eisai (and, in the case of any such involuntary proceeding, not dismissed within [***] calendar days (or such other longer period mutually agreed between the Parties in writing)); or (c) a receiver or trustee of Eisai’s property is appointed and not discharged within [***] calendar days (or such other longer period mutually agreed between the Parties in writing).
12.2.3Nuvation may terminate this Agreement immediately upon written notice if Eisai or its Affiliate initiates or joins any challenge, whether in a court of law or in an administrative proceeding, to the validity or enforceability of a Licensed Patent.
12.3Termination by Eisai.
12.3.1Eisai may terminate this Agreement, without prejudice to any other remedies available to it at law or in equity, if Nuvation or its Affiliates commit a material breach of this Agreement that, in the case of a material breach capable of remedy, has not been remedied within [***] calendar days of receiving a notice from Eisai identifying the breach and requiring its remedy (the “Nuvation Cure Period”), or if such material breach cannot be cured within the Nuvation Cure Period, Nuvation does not commence, during the Nuvation Cure Period, and
46
Page of SECTIONPAGES \* MERGEFORMAT 55
diligently continue actions to cure such breach, provided that in no event shall such material breach be permitted to last more than [***] days.
12.3.2Eisai may terminate this Agreement, without prejudice to any other remedies available to it at law or in equity, if (a) Nuvation or its Affiliates commit a material breach of the DS License Agreement (other than any material breach stemming from any act or omission of Eisai or its Affiliates) and Nuvation or its Affiliates fail to cure such material breach within the applicable cure period or (b) if the DS License Agreement is terminated.
12.3.3To the extent permitted by Applicable Law, Eisai may terminate this Agreement immediately if: (a) Nuvation becomes insolvent, or makes or seeks to make or arrange an assignment for the benefit of creditors; (b) proceedings in voluntary bankruptcy are initiated by or on behalf of Nuvation or proceedings in involuntary bankruptcy are initiated against Nuvation (and, in the case of any such involuntary proceeding, not dismissed within [***] calendar days (or such other longer period mutually agreed between the Parties in writing)); or (c) a receiver or trustee of Nuvation’s property is appointed and not discharged within [***] calendar days (or such other longer period mutually agreed between the Parties in writing).
12.3.4Eisai may terminate this Agreement in its entirety, at Eisai’s sole discretion, upon [***] months prior written notice if: (a) there is a demonstrated material lack of safety for human use due to significant toxicity of the Licensed Compound or Licensed Products (other than adverse events known to Eisai as of the Effective Date); or (b) where a Conditional Marketing Approval or other Marketing Approval for the Initial Indication is not obtained from the EMA by [***] except where such failure to obtain Marketing Approval is as a result of Eisai’s acts or omissions or failure to use Commercially Reasonable Efforts to seek such Marketing Approval.
12.3.5Eisai may terminate this Agreement on a country-by-country basis, at Eisai’s sole discretion, [***].
12.4Accrued Obligations/Survival. Expiration or termination of this Agreement for any reason does not release either Party from any obligation or liability which, at the time of such expiration or termination, has already accrued to the other Party or which is attributable to a period prior to such expiration or termination, provided that [***], provided further that, if (a) [***] cures the applicable breach during the [***] Cure Period or (b) the arbitrators appointed in accordance with Section 13.2 or an appropriate Governmental Authority determines that Nuvation has not materially breached the Agreement, then, in each case ((a) and (b)), [***]. For the avoidance of doubt, Article 1 (for the purpose of interpreting this Agreement), Section 2.6, Article 7 (solely to the extent payable as of the effective date of expiration or termination), Section 8.1, Section 8.3 (solely with respect to the defense of claims concerning activities performed under this Agreement prior to the effective date of expiration or termination, regardless of whether such claims are brought before or after such expiration or termination), Section 8.4 (solely with respect to infringement actions concerning activities performed under this Agreement prior to the effective date of expiration or termination, regardless of whether such actions are brought before or after such expiration or termination), Article 9, Section 10.7, Article 11, Section 12.1, this Section 12.4, Section 12.5, Section 12.6, Article 13, and Article 14 shall survive any expiration or termination of this Agreement for any reason.
47
Page of SECTIONPAGES \* MERGEFORMAT 55
12.5Return or Destruction of Confidential Information. Upon expiration or termination of this Agreement, the Receiving Party shall return to the Disclosing Party, or at the Disclosing Party’s request, destroy, all of the Disclosing Party’s or any of its Affiliates’ Confidential Information, except to the extent that the Receiving Party is otherwise required to retain a copy for compliance with Applicable Law or in accordance with its ordinary computer archive policies. Notwithstanding the foregoing, (a) Eisai shall not be required to return or destroy Confidential Information of Nuvation to the extent such Confidential Information continues to be licensed to Eisai pursuant to Section 12.1 and (b) Nuvation shall not be required to return or destroy Confidential Information of Eisai to the extent such Confidential Information is licensed to Nuvation pursuant to Section 12.6.
12.6Other Consequences of Termination. In the event of termination, but not expiration, of this Agreement for any reason:
12.6.1with respect to the Licensed Compound and Licensed Product(s) all licenses granted to Eisai under Article 2 will automatically terminate and shall revert to Nuvation, except (a) as necessary for Eisai to exercise its rights and fulfil its obligations during the Wind-Down Period or (b) with respect to the Licensed IP other than the Licensed IP licensed to Nuvation under the DS License Agreement to allow Eisai to, acting in both good-faith and reasonably, negotiate a Direct License under Section 2.4.2, until the earlier of (i) [***] months from the date of termination; or (ii) conclusion of any such negotiations with Daiichi Sankyo;
12.6.2effective upon termination Eisai hereby grants to Nuvation a non-exclusive, perpetual, irrevocable and sublicensable (through multiple tiers) license to use and practice any Eisai IP that is necessary for the Commercialization of the Licensed Compound or Licensed Product(s) in the Field in the Territory, provided that [***] the Parties shall negotiate in good faith for period of [***] days with respect to [***], provided further that if the Parties do not agree [***] within [***] days of the effective date of the termination of this Agreement (or such longer period as may be mutually agreed by the Parties in writing), then [***];
12.6.3effective upon termination Eisai hereby grants to Nuvation an exclusive, perpetual, irrevocable, [***]and sublicensable (through multiple tiers) license to use solely in connection with such the exploitation of the Licensed Compound or Licensed Product(s) any trademarks, brand names and brand logos Controlled by Eisai or its Affiliates and used by Eisai, its Affiliates or Sublicensees in connection with the Commercialization of the Licensed Compound or Licensed Product(s) prior to the effective date of termination (other than Eisai’s company-specific names and company-specific logos) provided always Nuvation will reimburse [***] of Eisai’s costs incurred in respect of the registration and maintenance of any such trademarks, brand names and brand logos where Nuvation has requested in writing that Eisai register and maintain such trademarks, brand names and brand logos following the effective date of termination;
12.6.4Eisai will, at Nuvation’s written request: (a) promptly transfer to Nuvation or its designee copies of all data, reports, records, and materials that relate to the Licensed Compound and Licensed Product(s) including materials related to Medical Affairs Activities; (b) return to Nuvation all relevant records and materials in Eisai’s possession or control containing Confidential Information of Nuvation relating to the Licensed Product(s); (c) assign, and will cause its Affiliates to assign, each trademark which has been used to Commercialize the Licensed
48
Page of SECTIONPAGES \* MERGEFORMAT 55
Product(s) in the Territory; provided that, Nuvation will receive no rights to any Eisai name or any trademark, trade name, or logo of Eisai itself; (d) assign any Regulatory Filings, including the Marketing Approval, to Nuvation or its designee; and (e) reasonably cooperate with Nuvation or its designee to facilitate a smooth, orderly, and prompt transition of the Commercialization of the Licensed Product(s) to Nuvation or its designee upon termination; and
12.6.5if this Agreement is terminated after the First Commercial Sale by Eisai, its Affiliates or its or their Sublicensees, of a Licensed Product in the Territory, Eisai, its Affiliates, Sublicensees, subcontractors or other related parties (the “Eisai Related Parties”) may, if required by Applicable Law or requested by a Regulatory Authority, continue to distribute and sell the Licensed Product(s) in the Territory, in accordance with the terms and conditions of this Agreement, for the longer of (a) the period of time required by Applicable Law or requested by a Regulatory Authority and (b) the shorter of: (i) the time it takes for Eisai to exhaust its inventory of Licensed Products and components thereof at the time of termination; and (ii) one year after termination or such longer period as required by Applicable Law or requested by a Regulatory Authority (the “Wind-Down Period”). During the Wind-Down Period, Nuvation shall continue to supply Eisai with the Licensed Product(s) in accordance with the terms and price set out in the Commercial Supply Agreement in place as at the date of termination and in the quantity necessary for Eisai to comply with the requirements of Applicable Law or the request from a Regulatory Authority, and Eisai shall continue to make any and all applicable payments to Nuvation for the terminated Licensed Product sold or disposed by Eisai or the Eisai Related Parties in accordance with the terms set out in the Commercial Supply Agreement. After the Wind-Down Period, Eisai or Eisai Related Parties shall not sell the terminated Licensed Product in the Territory. Within [***] calendar days of expiration of the Wind-Down Period, Eisai shall notify Nuvation of any quantity of the terminated Licensed Product remaining in Eisai’s and Eisai Related Parties’ inventory, to the extent any exist, and Nuvation shall, at Eisai’s request, purchase any such quantities of the terminated Licensed Product and components thereof, as applicable, from Eisai and Eisai Related Parties at [***]. [***] shall be responsible for the shipping of any such inventory and components thereof. Upon termination of the Wind-Down Period, the Commercial Supply Agreement shall terminate.
12.7Alternative Remedy in Lieu of Termination. [***].
13.1Between the Parties. Except for matters falling with the scope of the JCC’s authority and decision-making rights in accordance with Section 3.4 or the Parties’ inability to agree on the terms of the reversion royalties pursuant to Section 12.6.2, in the event of a dispute between the Parties arising out of or related to the terms of this Agreement, either Party may request that the Parties engage in good faith discussions to resolve such dispute. Within [***] calendar days of such request, each Party will appoint an appropriate representative to engage discussions to resolve the dispute in a mutually acceptable manner. Such representatives will have a reasonable level of expertise in the subject matter of the dispute and possess the requisite authority to resolve the dispute. If such representatives are unable to resolve the dispute within [***] calendar days, either Party may provide a written request to submit the dispute for discussions between Executive Officers appointed by the respective chief executive officers of
49
Page of SECTIONPAGES \* MERGEFORMAT 55
each Party. If the Executive Officers are unable to resolve the dispute within [***] calendar days after referral, either Party may provide a written request to refer the dispute for arbitration.
13.2Arbitration. [***], the dispute shall be referred to and finally resolved by arbitration in the following manner:
13.2.1The dispute shall be conducted according to the Rules of Arbitration of the [***] (the “Arbitration Rules”) in effect as of the Effective Date, except as they may be modified herein or by mutual agreement by the Parties in writing. The seat of arbitration shall be [***]. All arbitration proceedings shall be conducted by [***] arbitrators unless otherwise mutually agreed by the Parties. [***].
13.2.2The arbitral proceedings shall be conducted in English. To the extent that the Arbitration Rules are in conflict with the provisions of this Section 13.2, including the provisions concerning the appointment of the arbitrator, the provisions of this Section 13.2 shall prevail.
13.2.3Each Party shall cooperate with the other Party in the arbitration by making full disclosure of and providing complete access to all information and documents requested by such other Party in connection with such arbitral proceedings, subject only to any confidentiality obligations binding on such Party.
13.2.4The award of the arbitral tribunal shall be final and binding upon the Parties, and the prevailing Party may apply to a court of competent jurisdiction for enforcement of such award.
13.2.5The existence, nature, and results of, as well as any documents relating to, any arbitration shall be treated as Confidential Information by the Parties.
13.2.6Before the initiation of an arbitration as set forth in Section 13.2 or while the arbitration is pending, each Party shall have the right to seek interim injunctive or other equitable relief from a court of competent jurisdiction that may be necessary to avoid irreparable harm, maintain the status quo or preserve the subject matter of the arbitration.
13.3Mediation. [***], in each case, such matters may be referred by either Party to confidential mediation under the [***] (the “Mediation Rules”) in effect as of the Effective Date. The seat of the mediation shall be [***] and the Parties will nominate the mediator in accordance with the Mediation Rules. Each Party shall bear their own costs in the conduct of such mediation. The existence, nature, and results of, as well as any documents relating to, any mediation shall be treated as Confidential Information by the Parties. The Parties shall use good faith efforts to resolve any matters [***] are to be settled in accordance with mediation, provided that, (a) if any matters [***] are not resolved within [***]days after the appointment of the mediator, then [***], and (b) if any matters [***] are not resolved within [***] days after the appointment of the mediator, then [***].
13.4Baseball Arbitration. [***].
50
Page of SECTIONPAGES \* MERGEFORMAT 55
13.4.1Procedure. Any inability of the Parties to agree on the terms of the reversion royalties as set forth in Section 12.6.2 will be submitted to an independent Third Party expert (a “Third Party Expert”) acceptable to the Parties having at least [***] years of experience in the licensing of biopharmaceutical compounds or products and who has not had any material business relationship with either Party in the [***] months prior to appointment. The Parties shall use reasonable efforts to agree on the Third Party Expert within [***] days after either Party designates the dispute for baseball arbitration. [***]. Each Party shall bear its own costs incurred in the arbitration and shall equally share the costs of the Third Party Expert [***].
13.4.2Proposal of Terms. Within [***] days of selection of the Third Party Expert, each Party will deliver to both the Third Party Expert and the other Party a detailed written proposal setting forth (i) all terms the Parties have agreed upon with respect to the reversion royalties, and (ii) all of such Party’s proposed additional terms with respect to the reversion royalties (the “Proposed Terms”) and a memorandum in support thereof, not exceeding five (5) pages in length (the “Support Memorandum”). The Parties will also provide the Third Party Expert with a copy of this Agreement, as amended through such date. Within [***] days after receipt of the other Party’s Proposed Terms and Support Memorandum, each Party may submit to the Third Party Expert (with a copy to the other Party) a response to the other Party’s Proposed Terms and Support Memorandum, such response not exceeding five (5) pages in length. Neither Party may have any other communications (either written or oral) with the Third Party Expert; provided that the Third Party Expert may, in its discretion, convene a hearing to ask questions of the Parties and hear oral argument arid discussion regarding each Party’s Proposed Terms and Support Memorandum, at which time each Party will have an agreed upon time to argue and present witnesses in support of its Proposed Terms.
13.4.3Selection of Proposed Terms. Within [***] days after the Third Party Expert is appointed, the Third Party Expert will select one of the two Proposed Terms (without modification) provided by the Parties which most closely reflects and takes into account all relevant factors, including without limitation [***]. The Third Party Expert will make its decision known to both Parties as promptly as possible by delivering written notice to both Parties. The decision of the Third Party Expert will be final and binding on the Parties, and specific performance may be ordered by any court or arbitral body of competent jurisdiction.
14.Miscellaneous Provisions
14.1Relationship of the Parties. Nuvation and Eisai agree that the relationship between them established by this Agreement is that of independent contractors. The Parties further agree that this Agreement does not, is not intended to, and should not be construed to establish an employment, agency, partnership, joint venture, or any other relationship between them. Except as may be specifically provided herein, neither Party has any right, power, or authority, nor may they represent themselves as having any right, power, or authority, to assume, create or incur any expense, liability, or obligation, express or implied, on behalf of the other Party, or otherwise act as an agent for the other Party for any purpose.
14.2No Third Party Beneficiaries. No person or entity other than Nuvation, Eisai, and their respective Affiliates and permitted assignees may be deemed an intended beneficiary or have any right to enforce any obligation of this Agreement.
51
Page of SECTIONPAGES \* MERGEFORMAT 55
14.3Assignments. Neither Party may assign this Agreement or any of its rights or obligations hereunder to any Third Party without the written consent of the other Party, provided that each Party may assign this Agreement (a) to an Affiliate and (b) in connection with a merger or the acquisition of all or substantially all of its assets. Subject to the foregoing, the terms and conditions of this Agreement will be binding on and inure to the benefit of the successors and permitted assigns of the Parties. Any attempted assignment not in accordance with the terms of this Agreement will be void and of no legal force or effect.
14.4Performance by Affiliates. Either Party may perform its obligations under this Agreement through one or more Affiliates without the need to obtain prior approval from the other Party. Each Party remain solely responsible for the performance of its obligations under this Agreement by its Affiliate(s) and for any breach of the terms of this Agreement by its Affiliate(s).
14.5No Implied Waivers; Rights Cumulative. The failure of either Party to assert a right hereunder or to insist upon compliance with any term or condition of this Agreement does not constitute a waiver of that right or excuse a similar subsequent failure to perform such term or condition. Any term or condition of this Agreement may be waived at any time by the Party that is entitled to the benefit thereof, but no such waiver is effective unless set forth in a written instrument duly executed by or on behalf of the Party waiving such term or condition. No waiver by any Party of any term or condition of this Agreement, in any one or more instances, may be deemed to be or construed as a waiver of the same or any other term or condition of this Agreement on any future occasion. Except as expressly set forth in this Agreement, all rights and remedies available to a Party, whether under this Agreement or afforded by law or otherwise, are cumulative and not in the alternative to any other rights or remedies that may be available to such Party.
14.6Severability. If any provision of this Agreement is held to be invalid, illegal, or unenforceable under law in any jurisdiction, the Parties will negotiate in good faith a valid, legal, and enforceable substitute provision that most nearly reflects the original intent of the Parties and all other provisions of this Agreement will remain in full force and effect in such jurisdiction and will be liberally construed in order to carry out the intentions of the Parties as nearly as may be possible. A holding of invalidity, illegality, or unenforceability of a provision in one jurisdiction will not affect the validity, legality, or enforceability of such provision in any other jurisdiction.
14.7Entire Agreement; Amendments. This Agreement, together with all appendices, constitutes the entire agreement between the Parties with respect to the subject matter hereof, and supersedes all previous arrangements, whether written or oral, with respect to the subject matter contained herein. Any amendment or modification to this Agreement must be made in a writing signed by both Parties.
14.8Force Majeure. Neither Party will be liable to the other Party for failure or delay in performing of any of its obligations under this Agreement for the time and to the extent such failure or delay is caused by; epidemic, pandemics, plague, chemical or radioactive contamination, riot, civil commotion, rebellion, blockade, insurrection, invasion, war, acts of war (whether war be declared or not), terrorist acts, earthquake, tsunami, hurricane, tornado, storm, typhoon, lightning, volcanic eruption, landslide, flood, fire, acts of God, labor strike, or governmental acts, embargoes, or restriction, or other cause that is beyond the reasonable control of the affected Party. The Party affected by such force majeure must provide the other Party with full particulars thereof (including
52
Page of SECTIONPAGES \* MERGEFORMAT 55
its best estimate of the likely extent and duration of the interference with its activities) as soon as reasonably practicable. The affected Party will use Commercially Reasonable Efforts to overcome the difficulties created by the force majeure and to resume performance of its obligations as soon as practicable. In such event, the Parties will meet and discuss promptly to determine an equitable solution to minimize or accommodate the effects of any such event, including the possibility of terminating this Agreement.
14.9Governing Law. This Agreement shall be governed by, and any disputes, claims or controversies arising out of or in connection with this Agreement, including any question regarding its formation, existence, validity, enforceability, performance, interpretation, or termination, shall be resolved in accordance with, the laws of [***].
14.10.1Any notice, request, delivery, approval or consent required or permitted to be given under this Agreement shall be in writing and shall be deemed to have been sufficiently given if: (a) delivered in person; (b) transmitted by email (receipt verified); or (c) [***] calendar days after it was sent by registered letter or express courier service, return receipt requested (or its equivalent), provided that no postal strike or other disruption is then in effect or comes into effect within [***] calendar days after such mailing.
14.10.2Any notice, request, delivery, approval or consent to the Party required or permitted to be given under this Agreement shall be directed at its address or email shown below or such other address or email as such Party shall have last given by notice to the other Party. If such notice, request, delivery, approval, or consent is transmitted by email, the sending Party shall also transmit an identical copy of such documentation to the receiving Party by express courier service at the address shown below or to such other addresses and emails as a Party may designate by written notice.
If to Eisai, addressed to:
Eisai Co., Ltd.14-6-10
Koishikawa, Bunkyo-ku
Tokyo 112-8088, JAPAN
Attn: [***]
Email: [***]
With a copy (which shall not constitute notice) to:
Eisai Europe Limited
EMEA Knowledge Centre
Mosquito Way
Hatfield, Hertfordshire
AL10 9SN, ENGLAND
Attn: [***]
Email: [***]
53
Page of SECTIONPAGES \* MERGEFORMAT 55
If to Nuvation, addressed to:
Nuvation Bio Inc.
1500 Broadway, Suite 1401
New York, NY 10036, USA
Attn: [***]
Email: [***]
With a copy (which shall not constitute notice) to:
Cooley LLP
55 Hudson Yards
New York, NY 10001, USA
Attn: [***]
Email: [***]
14.11Bankruptcy. All rights, licenses and options granted under this Agreement are, and shall otherwise be deemed to be, for purposes of Section 365(n) of Title 11, U.S. Code (the “Bankruptcy Code”) and any foreign equivalent thereof, licenses of rights to “intellectual property” as defined under Section 101 of the Bankruptcy Code or such foreign equivalent. In the event of commencement of a bankruptcy proceeding by or against Nuvation under the Bankruptcy Code or such foreign equivalent, Eisai will be (a) permitted to fully exercise all of Nuvation’s rights and elections under the U.S. Bankruptcy Code and such foreign equivalent in any country having jurisdiction over a Party or its assets, (b) entitled to a complete copy of or access to in order to make a complete copy of all intellectual property that is licensed to Eisai hereunder, including a duplicate of all records (in whatever form existing, whether in hardcopy, electronic, photographic, or otherwise), a reasonable quantity of biological or chemical materials for reproducing, and any samples, prototypes, or other embodiments of such intellectual property not already in Eisai’s possession and to which Eisai would otherwise have been or be entitled pursuant to this Agreement, and (c) Nuvation will as promptly as reasonably possible but in any event within [***] days of Eisai’s request, deliver all of the foregoing to Eisai or permit Eisai to access of the foregoing to make a copy or collect and take possession of the foregoing.
14.12No Strict Construction. This Agreement has been prepared jointly by the Parties and should not be strictly construed against either Party.
14.13Interpretation. The captions and headings in this Agreement are for convenience only and are to be of no force or effect in construing or interpreting any of the provisions of this Agreement. Unless specified to the contrary, references to Articles, Sections or Appendices mean those particular Articles, Sections and Appendices to this Agreement and references to this Agreement include all attachments hereto. Unless context otherwise clearly requires, whenever used in this Agreement: (a) the words “include” or “including” shall be construed as incorporating, also, “but not limited to” or “without limitation;” (b) the word “day” or “year” means a calendar day or year unless otherwise specified; (c) the word “notice” means notice in writing (whether or not specifically stated); (d) the words “hereof,” “herein,” “hereby” and derivative or similar words refer to this Agreement (including any exhibits); (e) the word “or” shall be construed as the inclusive meaning identified with the phrase “and/or;”; (f) words using the singular or plural
54
Page of SECTIONPAGES \* MERGEFORMAT 55
number also include the plural or singular number, respectively; and (g) references to any specific law, rule or regulation, or article, section or other division thereof, shall be deemed to include the then-current amendments thereto or any replacement law, rule or regulation thereof.
14.14Counterparts. This Agreement may be executed in any number of counterparts, each of which will be deemed an original, and all of which together, will constitute one and the same.
14.15Use of Name. Except as otherwise provided herein, neither Party has any right, express or implied, to use the name or other designation of the other Party or any other trade name, trademark, or logo of the other Party in any manner or for any purpose in connection with the performance of this Agreement, except for use in connection with notices or filings required by law, rule, or regulation.
14.16Costs. Except as otherwise expressly provided in this Agreement, each Party shall pay costs incurred by it in connection with the entering into and operation of this Agreement.
[REMAINDER OF PAGE INTENTIONALLY LEFT BLANK]
55
Page of SECTIONPAGES \* MERGEFORMAT 55
IN WITNESS WHEREOF, Nuvation and Eisai have caused this Agreement to be executed by their duly authorized representatives as of the Effective Date.
|
|
By: Name: Toshitaka Asano Title: Vice President, Business and Global Alliance, Eisai Co., Ltd. |
By: Name: David Hung, M.D. Title: CEO |
[***] [***] |
[***] [***] |
|
|
[***] [***] [***] |
|
[***] [***] |
|
Signature Page to License and Collaboration Agreement
Schedule 1.58
Licensed Compound
The molecular structure of taletrectinib:
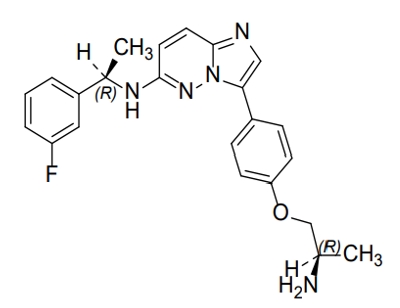
Schedule 1.78
Nuvation Patents
[***]
|
|
|
|
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
|
|
|
|
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***]
[***]
|
|
|
|
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***]
|
|
|
|
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***]
|
|
|
|
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
Schedule 1.79
Other Nuvation IP
|
|
|
|
|
|
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
|
|
|
|
|
|
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
Schedule 1.104
Territory
|
Europe |
Russia |
Türkiye |
Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, the United Arab Emirates, Israel, Jordan, Iran, Iraq, Libya, Lebanon, Egypt and Sudan |
Morocco, Algeria, and Tunisia |
Australia |
New Zealand |
Canada |
Singapore, Philippines, Indonesia, Thailand, Malaysia, Vietnam and India |
Schedule 4.1.2
[***]
|
|
|
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
Schedule 5.1
Technology Transfer
Eisai shall be responsible for any translations at its own cost.
|
|
|
[***] |
[***] |
[***] |
|
|
[***] |
|
|
|
|
[***] |
|
|
|
|
|
|
|
|
[***] |
|
Schedule 8.2.1(e)
Registration of DS License Agreement
[***]
Schedule 9.6
Joint Press Release


Nuvation Bio and Eisai Enter into Exclusive Licensing Agreement for Taletrectinib in Europe and Additional Countries Outside U.S., China and Japan
Strategic collaboration expands taletrectinib global reach with a leading oncology partner
Eisai will receive exclusive development, registration and commercialization rights for taletrectinib for the treatment of ROS1-positive non-small cell lung cancer in Europe, Middle East, Canada, Australia, New Zealand, Singapore, the Philippines, Indonesia, Thailand, Malaysia, Vietnam and India
Nuvation Bio will receive double-digit tiered royalties up to the high-teens as a percentage of taletrectinib sales in the licensed territories, in addition to up to €195 million (approx. USD $230 million) in upfront and milestone payments
Taletrectinib is already approved in the U.S., China and Japan for advanced ROS1-positive non-small cell lung cancer
NEW YORK AND TOKYO—January 12, 2026— Nuvation Bio Inc. (NYSE: NUVB, Corporate Headquarters: New York, NY, CEO: David Hung, M.D., “Nuvation Bio”), a global oncology company focused on tackling some of the toughest challenges in cancer treatment, and Eisai Co., Ltd (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”), a human-centered global leading research-based pharmaceutical company working in the neurology and oncology therapeutic areas, today announced an exclusive license and collaboration agreement that significantly expands the long-term global footprint of taletrectinib (marketed as IBTROZI® in the U.S. and Japan). Taletrectinib is a highly selective, next-generation oral treatment currently approved for patients living with advanced ROS1-positive (ROS1+) non-small cell lung cancer (NSCLC) in the U.S., China and Japan. Eisai will now have exclusive development, registration and commercialization rights for taletrectinib for the treatment of ROS1+ NSCLC in Europe, the Middle East, North Africa, Russia, Turkey, Canada, Australia, New Zealand, Singapore, the Philippines, Indonesia, Thailand, Malaysia, Vietnam and India. Nuvation Bio will continue to lead global development and retain full U.S. commercial rights, maintaining its strong focus on U.S. launch activities and ongoing pivotal studies of taletrectinib across early- and late-stage ROS1+ NSCLC.
“Our partnership with Eisai represents a major global expansion milestone for taletrectinib, strengthening the long-term potential of this clinically meaningful treatment option for even more patients living with ROS1+ NSCLC,” said David Hung, M.D., Founder, President, and Chief Executive Officer of Nuvation Bio. “With Eisai’s world-class infrastructure and track record of bringing innovative medicines to market in major regions, we believe the expanse of this collaboration illustrates the commercial potential of taletrectinib while accelerating the opportunity for providers and patients around the world to access this important treatment option.”
Under the terms of the exclusive license and collaboration agreement, Nuvation Bio will receive €50 million (approx. USD $60 million) upfront and up to €145 million (approx. USD $170 million) in
regulatory and commercial milestone payments, as well as double-digit tiered royalties up to the high-teens as a percentage of future net sales in the licensed territories. Following the upfront payment, Nuvation Bio will receive the first milestone payment of €25 million (approx. USD $30 million) from this transaction upon achievement of EU regulatory approval (conditional or full) of taletrectinib. The USD amounts are approximated based on an exchange rate of EUR1= USD1.2.
“With its efficacy and safety profile, we believe taletrectinib has the potential to become a standard of care for patients with ROS1+ NSCLC in the EU and beyond, as it is already becoming the standard of care in the U.S. just six months from approval,” said Terushige Iike, Chief Business Officer of Eisai Co., Ltd. “We are thrilled to partner with Nuvation Bio and prioritize taletrectinib as our flagship oncology product in NSCLC, bringing this innovative medicine to patients as quickly as possible.”
A Marketing Authorization Application (MAA) for the treatment of advanced ROS1+ NSCLC is expected to be filed in Europe in the first half of 2026. Additional filings are then planned for Canada and other regions.
In June 2025, the U.S. Food and Drug Administration (FDA) granted full approval to taletrectinib for the treatment of locally advanced or metastatic ROS1+ NSCLC across lines of therapy, following a Priority Review and double Breakthrough Therapy designations. Taletrectinib is also approved for patients with advanced ROS1+ NSCLC in Japan, where it is marketed by Nippon Kayaku, and in China, where it is marketed by Innovent Biologics under the brand name DOVBLERON®.
About ROS1+ NSCLC
Each year, more than one million people globally are diagnosed with non-small cell lung cancer (NSCLC), the most common form of lung cancer. It is estimated that approximately 2% of patients with NSCLC have ROS1+ disease. About 35% of patients newly diagnosed with metastatic ROS1+ NSCLC have tumors that have spread to their brain. The brain is also the most common site of disease progression, with about 50% of previously treated patients developing central nervous system (CNS) metastases.
About Taletrectinib
Taletrectinib is an oral, potent, CNS-active, selective, next-generation ROS1 inhibitor therapy. On June 11, 2025, following Priority Review and Breakthrough Therapy designations for both TKI-naïve and TKI-pretreated disease, the U.S. Food and Drug Administration (FDA) approved taletrectinib for the treatment of adult patients with locally advanced or metastatic ROS1+ NSCLC. Learn more about taletrectinib in the U.S. at IBTROZI.com.
About the TRUST Clinical Program
The TRUST clinical program comprises three registrational studies evaluating the safety and efficacy of taletrectinib. TRUST-I (NCT04395677) and TRUST-II (NCT04919811) are Phase 2 single-arm studies evaluating taletrectinib for the treatment of adults with advanced ROS1+ NSCLC in China (N=173) and globally (N=189), respectively. The primary endpoint of both studies is confirmed objective response rate (cORR) as assessed by an independent review committee. TRUST‑IV (NCT07154706) is a Phase 3 placebo-controlled study evaluating taletrectinib for the adjuvant treatment of adults with resected early-stage ROS1+ NSCLC. The study will enroll approximately 180 patients in the U.S., Canada, Europe, Japan and China. The primary endpoint is disease-free survival as determined by investigator, and the primary completion date is estimated to be in 2030. Nuvation Bio is also sponsoring TRUST-III (NCT06564324), a confirmatory randomized Phase 3 study evaluating taletrectinib versus crizotinib in 138 patients in China with advanced ROS1+ NSCLC who have not previously received ROS1 TKIs.
U.S. Indication
IBTROZI is indicated for the treatment of adult patients with locally advanced or metastatic ROS1+ non-small cell lung cancer (NSCLC).
IMPORTANT SAFETY INFORMATION FOR IBTROZI® (taletrectinib)
WARNINGS AND PRECAUTIONS
Hepatotoxicity: Hepatotoxicity, including drug-induced liver injury and fatal adverse reactions, can occur. 88% of patients experienced increased AST, including 10% Grade 3/4. 85% of patients experienced increased ALT, including 13% Grade 3/4. Fatal liver events occurred in 0.6% of patients. Median time to first onset of AST or ALT elevation was 15 days (range: 3 days to 20.8 months).
Increased AST or ALT each led to dose interruption in 7% of patients and dose reduction in 5% and 9% of patients, respectively. Permanent discontinuation was caused by increased AST, ALT, or bilirubin each in 0.3% and by hepatotoxicity in 0.6% of patients.
Concurrent elevations in AST or ALT ≥3 times the ULN and total bilirubin ≥2 times the ULN, with normal alkaline phosphatase, occurred in 0.6% of patients.
Interstitial Lung Disease (ILD)/Pneumonitis: Severe, life-threatening, or fatal ILD or pneumonitis can occur. ILD/pneumonitis occurred in 2.3% of patients, including 1.1% Grade 3/4. One fatal ILD case occurred at the 400 mg daily dose. Median time to first onset of ILD/pneumonitis was 3.8 months (range: 12 days to 11.8 months).
ILD/pneumonitis led to dose interruption in 1.1% of patients, dose reduction in 0.6% of patients, and permanent discontinuation in 0.6% of patients.
QTc Interval Prolongation: QTc interval prolongation can occur, which can increase the risk for ventricular tachyarrhythmias (e.g., torsades de pointes) or sudden death. IBTROZI prolongs the QTc interval in a concentration-dependent manner.
In patients who received IBTROZI and underwent at least one post baseline ECG, QTcF increase of >60 msec compared to baseline and QTcF >500 msec occurred in 13% and 2.6% of patients, respectively. 3.4% of patients experienced Grade ≥3. Median time from first dose of IBTROZI to onset of ECG QT prolongation was 22 days (range: 1 day to 38.7 months). Dose interruption and dose reduction each occurred in 2.8% of patients.
Significant QTc interval prolongation may occur when IBTROZI is taken with food, strong and moderate CYP3A inhibitors, and/or drugs with a known potential to prolong QTc. Administer IBTROZI on an empty stomach. Avoid concomitant use with strong and moderate CYP3A inhibitors and/or drugs with a known potential to prolong QTc.
Hyperuricemia: Hyperuricemia can occur and was reported in 14% of patients, with 16% of these requiring urate-lowering medication without pre-existing gout or hyperuricemia. 0.3% of patients experienced Grade ≥3. Median time to first onset was 2.1 months (range: 7 days to 35.8 months). Dose interruption occurred in 0.3% of patients.
Myalgia with Creatine Phosphokinase (CPK) Elevation: Myalgia with or without CPK elevation can occur. Myalgia occurred in 10% of patients. Median time to first onset was 11 days (range: 2 days to 10 months).
Concurrent myalgia with increased CPK within a 7-day time period occurred in 0.9% of patients. Dose interruption occurred in 0.3% of patients with myalgia and concurrent CPK elevation.
Skeletal Fractures: IBTROZI can increase the risk of fractures. ROS1 inhibitors as a class have been associated with skeletal fractures. 3.4% of patients experienced fractures, including 1.4% Grade 3. Some fractures occurred in the setting of a fall or other predisposing factors. Median time to first onset of fracture was 10.7 months (range: 26 days to 29.1 months). Dose interruption occurred in 0.3% of patients.
Embryo-Fetal Toxicity: Based on literature, animal studies, and its mechanism of action, IBTROZI can cause fetal harm when administered to a pregnant woman.
ADVERSE REACTIONS
Among patients who received IBTROZI, the most frequently reported adverse reactions (≥20%) were diarrhea (64%), nausea (47%), vomiting (43%), dizziness (22%), rash (22%), constipation (21%), and fatigue (20%).
The most frequently reported Grade 3/4 laboratory abnormalities (≥5%) were increased ALT (13%), increased AST (10%), decreased neutrophils (5%), and increased creatine phosphokinase (5%).
DRUG INTERACTIONS
•Strong and Moderate CYP3A Inhibitors/CYP3A Inducers and Drugs that Prolong the QTc Interval: Avoid concomitant use.
•Gastric Acid Reducing Agents: Avoid concomitant use with PPIs and H2 receptor antagonists. If an acid-reducing agent cannot be avoided, administer locally acting antacids at least 2 hours before or 2 hours after taking IBTROZI.
OTHER CONSIDERATIONS
•Pregnancy: Please see important information in Warnings and Precautions under Embryo-Fetal Toxicity.
•Lactation: Advise women not to breastfeed during treatment and for 3 weeks after the last dose.
•Effect on Fertility: Based on findings in animals, IBTROZI may impair fertility in males and females. The effects on animal fertility were reversible.
•Pediatric Use: The safety and effectiveness of IBTROZI in pediatric patients has not been established.
•Photosensitivity: IBTROZI can cause photosensitivity. Advise patients to minimize sun exposure and to use sun protection, including broad-spectrum sunscreen, during treatment and for at least 5 days after discontinuation.
Please see accompanying full U.S. Prescribing Information.
About Nuvation Bio
Nuvation Bio is a global oncology company focused on tackling some of the toughest challenges in cancer treatment with the goal of developing therapies that create a profound, positive impact on patients’ lives. Our diverse pipeline includes taletrectinib (IBTROZI®), a next-generation ROS1 inhibitor; safusidenib, a brain-penetrant IDH1 inhibitor; NUV-868, a BD2-selective BET inhibitor; and an innovative drug-drug conjugate (DDC) program.
Nuvation Bio was founded in 2018 by biopharma industry veteran David Hung, M.D., who previously founded Medivation, Inc., which brought to patients one of the world’s leading prostate cancer medicines. Nuvation Bio has offices in New York, San Francisco, Boston, and Shanghai. For more information, visit www.nuvationbio.com or follow the company on LinkedIn and X (@nuvationbioinc).
About Eisai Co., Ltd.
Eisai's Corporate Concept is "to give first thought to patients and people in the daily living domain, and to increase the benefits that health care provides." Under this Concept (also known as human health care (hhc) Concept), we aim to effectively achieve social good in the form of relieving anxiety over health and reducing health disparities. With a global network of R&D facilities, manufacturing sites and marketing subsidiaries, we strive to create and deliver innovative products to target diseases with high unmet medical needs, with a particular focus in our strategic areas of Neurology and Oncology.
In addition, we demonstrate our commitment to the elimination of neglected tropical diseases (NTDs), which is a target (3.3) of the United Nations Sustainable Development Goals (SDGs), by working on various activities together with global partners.
For more information about Eisai, please visit www.eisai.com (for global headquarters: Eisai Co., Ltd.), and connect via X, LinkedIn and Facebook. The website and social media channels are intended for audiences outside of the UK and Europe.
Forward-Looking Statements of Nuvation Bio
Certain statements included in this press release that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements are sometimes accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements regarding IBTROZI’S therapeutic and commercial potential, our expectations for a MAA filing for IBTROZI in Europe and the timing thereof, and the receipt and timing of a regulatory and commercial milestone payment under our license and collaboration agreement with Eisai. These statements are based on various assumptions, whether or not identified in this press release, and on the current expectations of the management team of Nuvation Bio and are not predictions of actual performance. These forward-looking statements are subject to a number of risks and uncertainties that may cause actual results to differ from those anticipated by the forward-looking statements, including but not limited to the challenges associated with conducting drug discovery and commercialization, and initiating or conducting clinical studies due to, among other things, difficulties or delays in the regulatory process, enrolling subjects or manufacturing or acquiring necessary products; the emergence or worsening of adverse events or other undesirable side effects; risks associated with preliminary and interim data, which may not be representative of more mature data; physician and patient behavior; and competitive developments. Risks and uncertainties facing Nuvation Bio are described more fully in its Form 10-Q filed with the SEC on November 3, 2025 under the heading “Risk Factors,” and other documents that Nuvation Bio has filed or will file with the SEC. You are cautioned not to place undue reliance on the forward-looking statements, which speak only as of the date of this press release. Nuvation Bio disclaims any obligation or undertaking to update, supplement or revise any forward-looking statements contained in this press release.
Nuvation Bio Contacts
Investor Contact
JR DeVita
ir@nuvationbio.com
Media Contact
Kaitlyn Nealy
media@nuvationbio.com
Eisai Contacts
Media Contact
Eisai Co., Ltd.
Public Relations Department
TEL: +81 (0)3-3817-5120
Investor Contact
Eisai Co., Ltd.
Investor Relations Department
TEL: +81 (0) 3-3817-5122


