
Exhibit 99.2

©2026 Oruka Therapeutics ©2026 ORUKA THERAPEUTICS EVERLAST - A Interim Data ORKA - 001 Phase 2a trial in psoriasis April 27, 2026

©2026 Oruka Therapeutics 2 Disclaimers The information contained in this presentation has been prepared by Oruka Therapeutics, Inc. (the “Company”) and contains inf orm ation pertaining to the business and operations of the Company. The information contained in this presentation: (a) is provided as at the date hereof, is subject to change without notice, and is based on p ubl icly available information, internally developed data as well as third party information from other sources; (b) does not purport to contain all the information that may be necessary or desirable to fully and accurately ev aluate an investment in the Company; (c) is not to be considered as a recommendation by the Company that any person make an investment in the Company; (d) is for information purposes only and shall not constitu te an offer to buy, sell, issue or subscribe for, or the solicitation of an offer to buy, sell or issue, or subscribe for any securities of the Company in any jurisdiction in which such offer, solicitation or sale would be unlawful. Where any opinion or belief is expressed in this presentation, it is based on certain assumptions and limitations and is an expression of present opinion or belief only. This presentation should not be c ons trued as legal, financial or tax advice to any individual, as each individual’s circumstances are different. This document is for informational purposes only and should not be considered a solicitation or rec ommendation to purchase, sell or hold a security. Forward - Looking Information Certain information set forth in this presentation contains “forward - looking statements” within the meaning of applicable United States securities legislation. Except for statements of historical fact, certain information contained herein constitutes forward - looking statements, which include but are not limited to statements regarding: expectations regarding the efficacy, durability of effect, dosing interval and safety of our product candidates; expectations regarding our plans for clinical trials and research and development programs, including the timing of clinical trials and data readouts; the time periods over which the Company’s capital resources will be sufficient to fund its anticipated operations; the Company’s business strategy objectives and goals; and ma nag ement’s assessment of future plans and operations, which are based on current internal expectations, estimates, projections, assumptions and beliefs, which may prove to be incorrect. Forward - looking stateme nts are neither historical facts nor assurances of future performance. Forward - looking statements are based on a number of factors and assumptions made by management and considered reasonable at the time suc h information is provided, and forward - looking statements involve known and unknown risks, uncertainties and other factors that may cause the actual results, performance or achievements to be materiall y d ifferent from those expressed or implied by the forward - looking statements including those uncertainties and factors described under the heading “Risk Factors” and “Cautionary Note Regarding Forward - Looking Statem ents” in the Company’s most recent filings with the SEC, including its Annual Report on Form 10 - K, its Quarterly Reports on Form 10 - Q and its other filings with the SEC , as well as discussions of potential risks, uncertainties by the Company from time to time, as well as risk factors associated with companies that operate in the biopharma industry, including those associated with the uncertainties of drug d eve lopment. All of the forward - looking statements made in this presentation are qualified by these cautionary statements and other cautionary statements or other factors contained herein. Although management believe s t hat the expectations conveyed by forward - looking statements herein are reasonable based on information available on the date such forward - looking statements are made, there can be no assurance that f orward looking statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. The Company undertakes no obligation to update forw ard - looking statements if circumstances or management’s estimates or opinions should change except as required by applicable securities laws. The forward - looking statements contained herein are presented fo r the purposes of assisting readers in understanding the Company’s plans, objectives and goals and may not be appropriate for other purposes. The reader is cautioned not to place undue reliance on fo rwa rd - looking statements. Industry Information This presentation also contains or references certain industry data that is based upon information from independent industry pub lications, market research, and surveys and other publicly available sources. Although the Company believes these sources to be generally reliable, such information is subject to interpretation and canno t b e verified with complete certainty due to limits on the availability and reliability of data, the voluntary nature of the data gathering process and other inherent limitations and uncertainties. The Company has not inde pen dently verified any of the data from third party sources referred to in this presentation and accordingly, the Company makes no representation or warranty as to the origin, validity, accuracy, completen ess , currency or reliability of the information in this presentation.

©2026 Oruka Therapeutics 3 Agenda Lawrence Klein, PhD Chief Executive Officer Introduction Joana Goncalves, MBChB Chief Medical Officer ORKA - 001 EVERLAST - A Phase 2a interim results Lawrence Klein, PhD Chief Executive Officer Understanding ORKA - 001's potential Lawrence Klein, CEO Joana Goncalves, CMO Analyst Q&A


©2026 Oruka Therapeutics 5 Two programs that could set a new standard in psoriatic disease Abbreviations: HS, hidradenitis suppurativa; PsA, psoriatic arthritis; PsO, psoriasis ORKA - 001 ORKA - 002 Ultra - long - acting IL - 17A/F mAb with potential for Q6M dosing in PsO/PsA and Q3M dosing in HS Ultra - long - acting IL - 23p19 mAb with potential for annual dosing, superior efficacy, and off - treatment remission Phase 2a in PsO TODAY: 16 - week data Dose - ranging Phase 2b to support Phase 3 initiation; data expected in 2027 Phase 2 in PsO; data expected in 2027 Phase 2 start in HS anticipated in 2H 2026

©2026 Oruka Therapeutics 6 PsO is a $30B+ market where better biologics consistently win Notes & Sources: (1) Evaluate Pharma global net sales estimates, excluding psoriatic arthritis 0 $31B market today for biologics and other advanced therapies, expected to grow to $39B by 2030 1 0 Bimzelx launch (~$1.4B in PsO alone in 2 nd year) shows that better biologics continue to win, even when launched by a non - incumbent 0 A once - or twice - yearly IL - 23p19 antibody with improved efficacy has the potential to become the preferred medicine in psoriasis 0 New orals have not reached the efficacy of modern biologics, but will likely expand the market, as Otezla did with the first generation of biologics
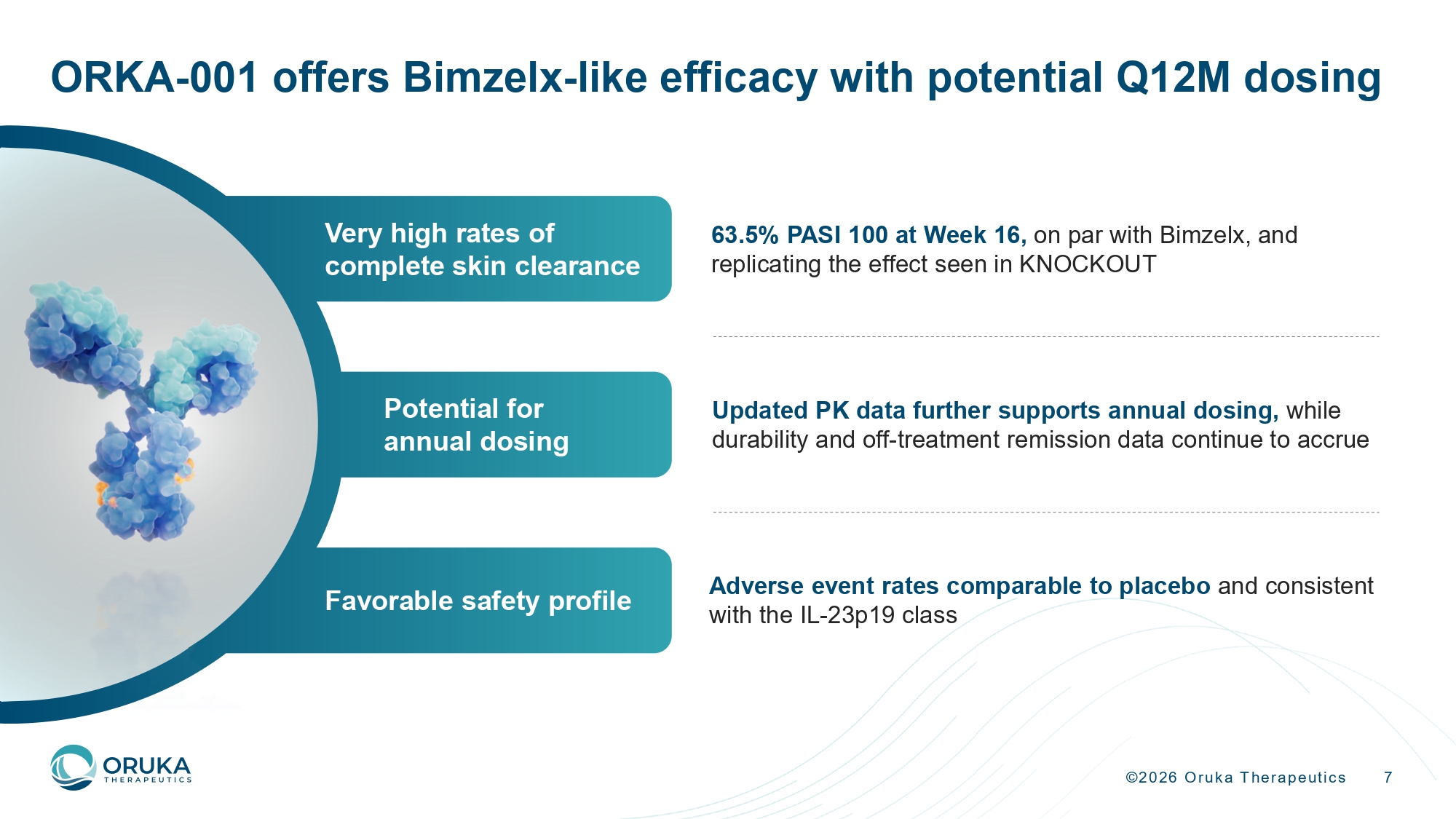
©2026 Oruka Therapeutics 7 ORKA - 001 offers Bimzelx - like efficacy with potential Q12M dosing 63.5% PASI 100 at Week 16, on par with Bimzelx, and replicating the effect seen in KNOCKOUT Adverse event rates comparable to placebo and consistent with the IL - 23p19 class Updated PK data further supports annual dosing, while durability and off - treatment remission data continue to accrue Very high rates of complete skin clearance Potential for annual dosing Favorable safety profile

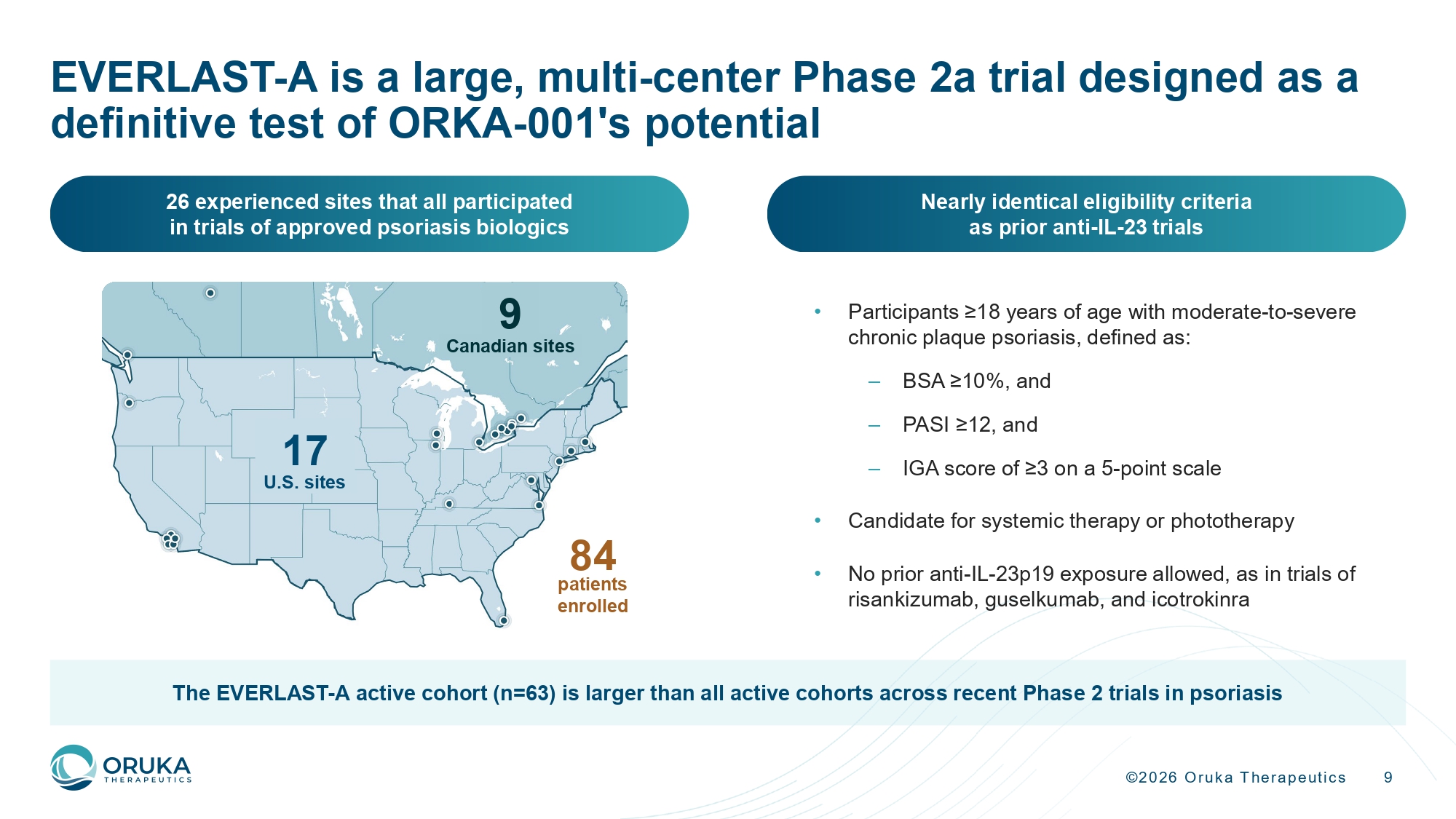
©2026 Oruka Therapeutics 9 26 experienced sites that all participated in trials of approved psoriasis biologics Nearly identical eligibility criteria as prior anti - IL - 23 trials • Participants ≥18 years of age with moderate - to - severe chronic plaque psoriasis, defined as: – BSA ≥10%, and – PASI ≥12, and – IGA score of ≥3 on a 5 - point scale • Candidate for systemic therapy or phototherapy • No prior anti - IL - 23p19 exposure allowed, as in trials of risankizumab, guselkumab, and icotrokinra 17 U.S. sites 9 Canadian sites 84 patients enrolled The EVERLAST - A active cohort (n=63) is larger than all active cohorts across recent Phase 2 trials in psoriasis EVERLAST - A is a large, multi - center Phase 2a trial designed as a definitive test of ORKA - 001's potential
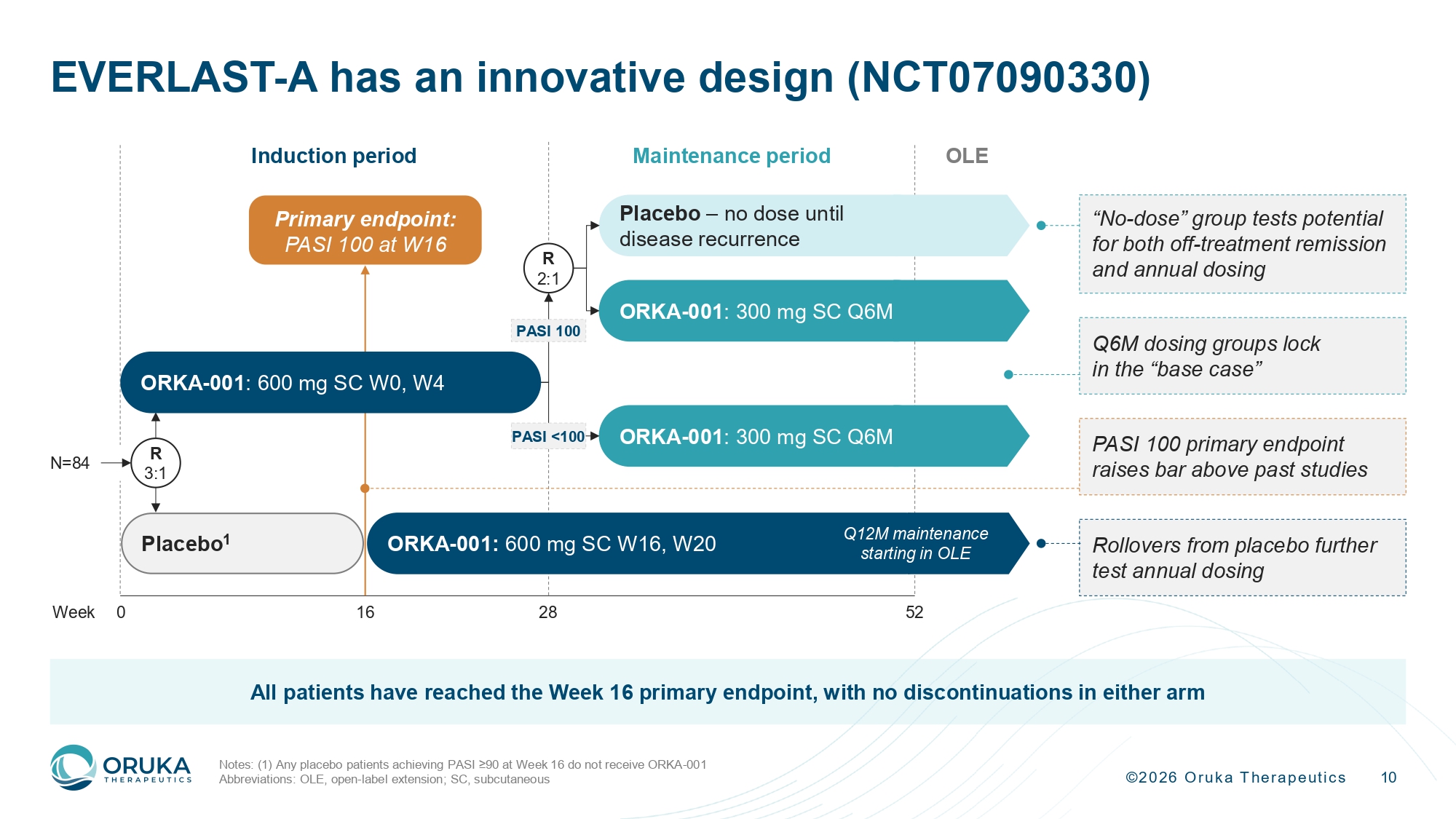
©2026 Oruka Therapeutics 10 EVERLAST - A has an innovative design (NCT07090330) Notes: (1) Any placebo patients achieving PASI ≥90 at Week 16 do not receive ORKA - 001 Abbreviations: OLE, open - label extension; SC, subcutaneous OLE 16 Induction period Placebo 1 Maintenance period Placebo – no dose until disease recurrence R 3:1 28 52 Week 0 Primary endpoint: PASI 100 at W16 ORKA - 001 : 300 mg SC Q6M R 2:1 ORKA - 001 : 300 mg SC Q6M ORKA - 001: 600 mg SC W16, W20 N=84 PASI 100 PASI <100 Q12M maintenance starting in OLE ORKA - 001 : 600 mg SC W0, W4 “No - dose” group tests potential for both off - treatment remission and annual dosing Q6M dosing groups lock in the “base case” PASI 100 primary endpoint raises bar above past studies Rollovers from placebo further test annual dosing All patients have reached the Week 16 primary endpoint, with no discontinuations in either arm
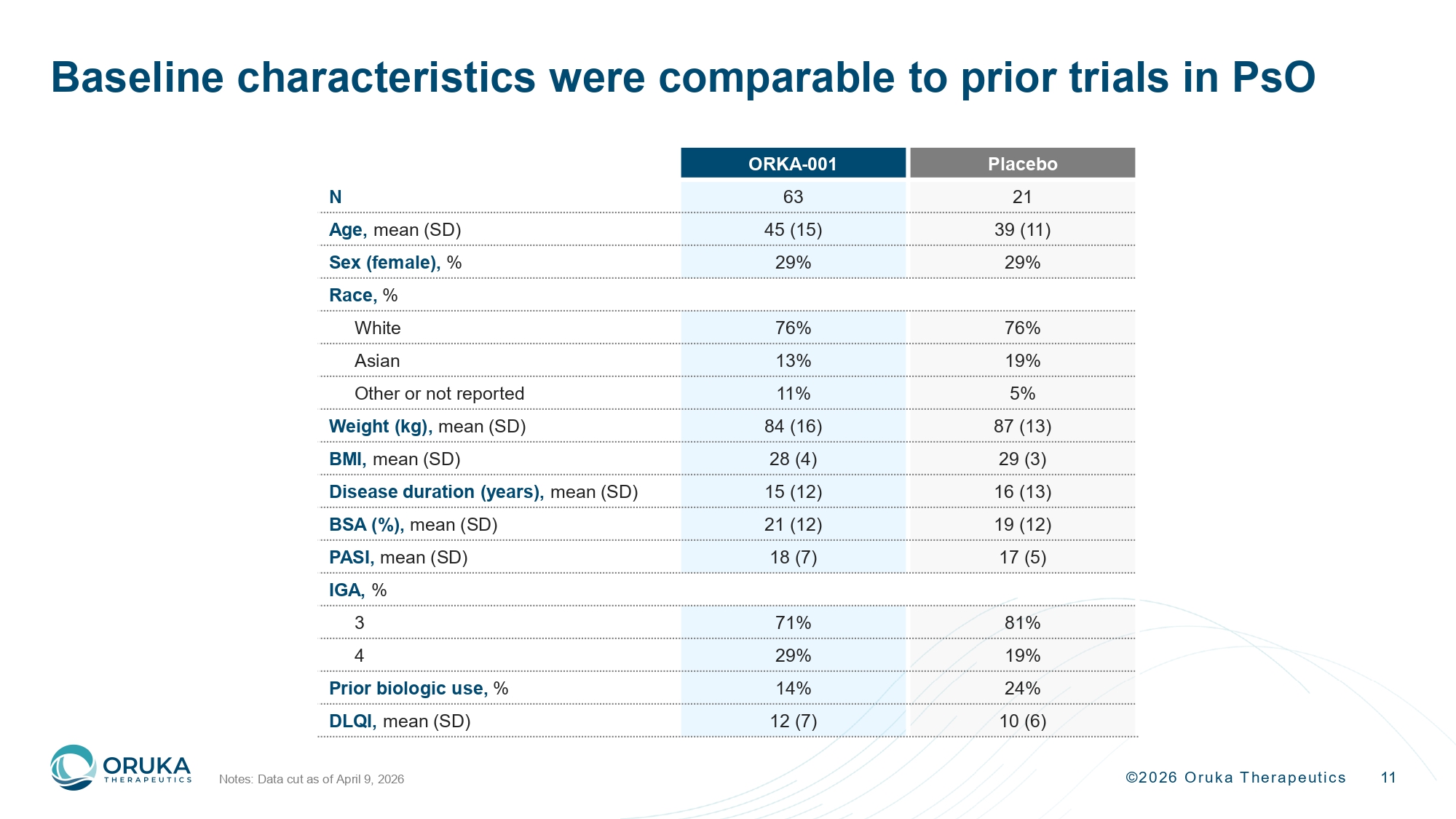
©2026 Oruka Therapeutics 11 Baseline characteristics were comparable to prior trials in PsO Notes: Data cut as of April 9, 2026 Placebo ORKA - 001 21 63 N 39 (11) 45 (15) Age, mean (SD) 29% 29% Sex (female), % Race, % 76% 76% White 19% 13% Asian 5% 11% Other or not reported 87 (13) 84 (16) Weight (kg), mean (SD) 29 (3) 28 (4) BMI, mean (SD) 16 (13) 15 (12) Disease duration (years), mean (SD) 19 (12) 21 (12) BSA (%), mean (SD) 17 (5) 18 (7) PASI, mean (SD) IGA, % 81% 71% 3 19% 29% 4 24% 14% Prior biologic use, % 10 (6) 12 (7) DLQI, mean (SD)
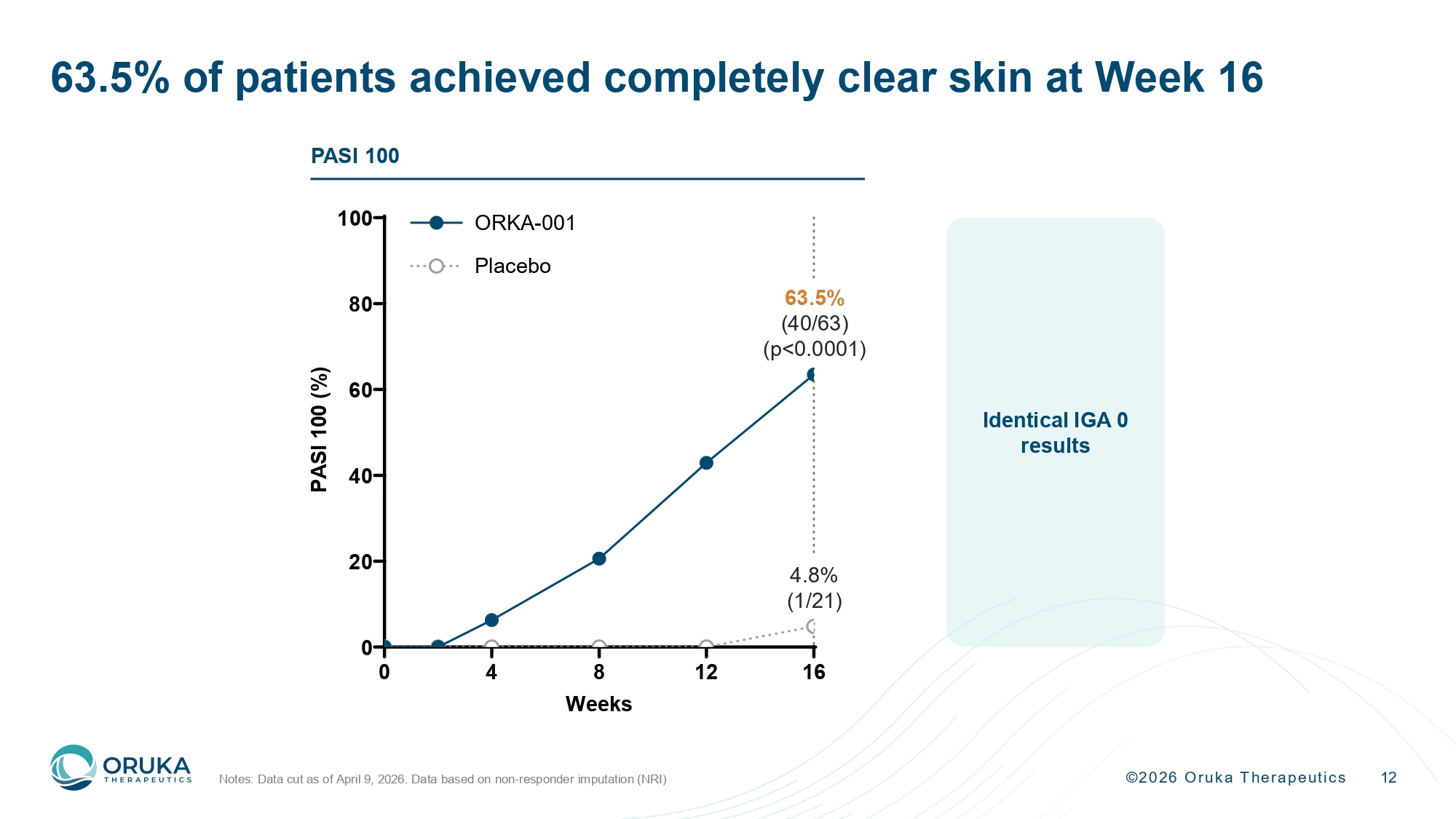
©2026 Oruka Therapeutics 12 63.5% of patients achieved completely clear skin at Week 16 Notes: Data cut as of April 9, 2026. Data based on non - responder imputation (NRI) Identical IGA 0 results 0 4 8 12 16 0 20 40 60 80 100 Weeks P A S I 1 0 0 ( % ) ORKA-001 Placebo 63.5% (40/63) (p<0.0001) 4.8% (1/21) PASI 100
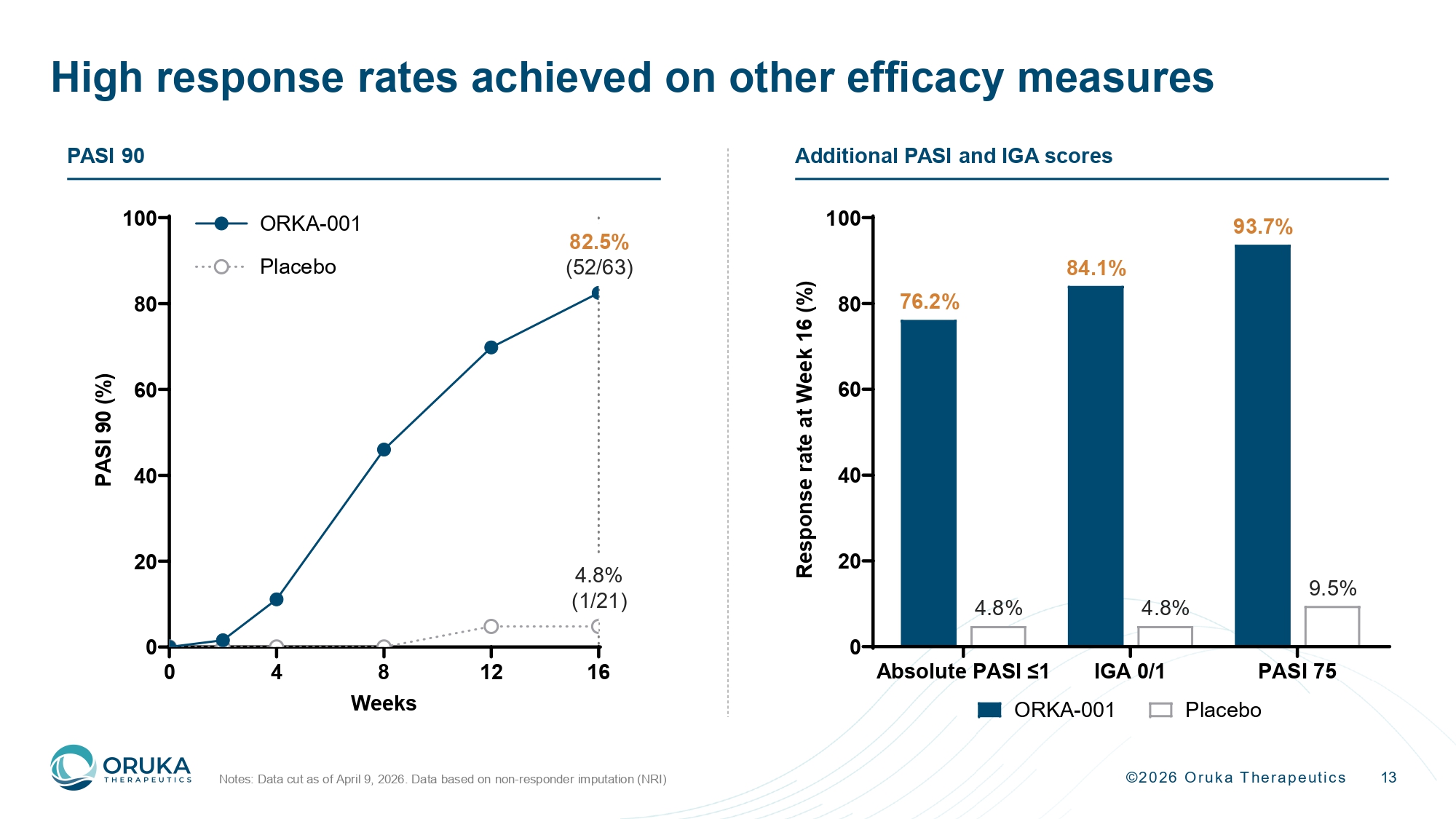
©2026 Oruka Therapeutics Absolute PASI ≤1 IGA 0/1 PASI 75 0 20 40 60 80 100 R e s p o n s e r a t e a t W e e k 1 6 ( % ) ORKA-001 Placebo 13 High response rates achieved on other efficacy measures Notes: Data cut as of April 9, 2026. Data based on non - responder imputation (NRI) 76.2% 4.8% 93.7% 9.5% 0 4 8 12 16 0 20 40 60 80 100 Weeks P A S I 9 0 ( % ) ORKA-001 Placebo 82.5% (52/63) 4.8% (1/21) 84.1% 4.8% PASI 90 Additional PASI and IGA scores
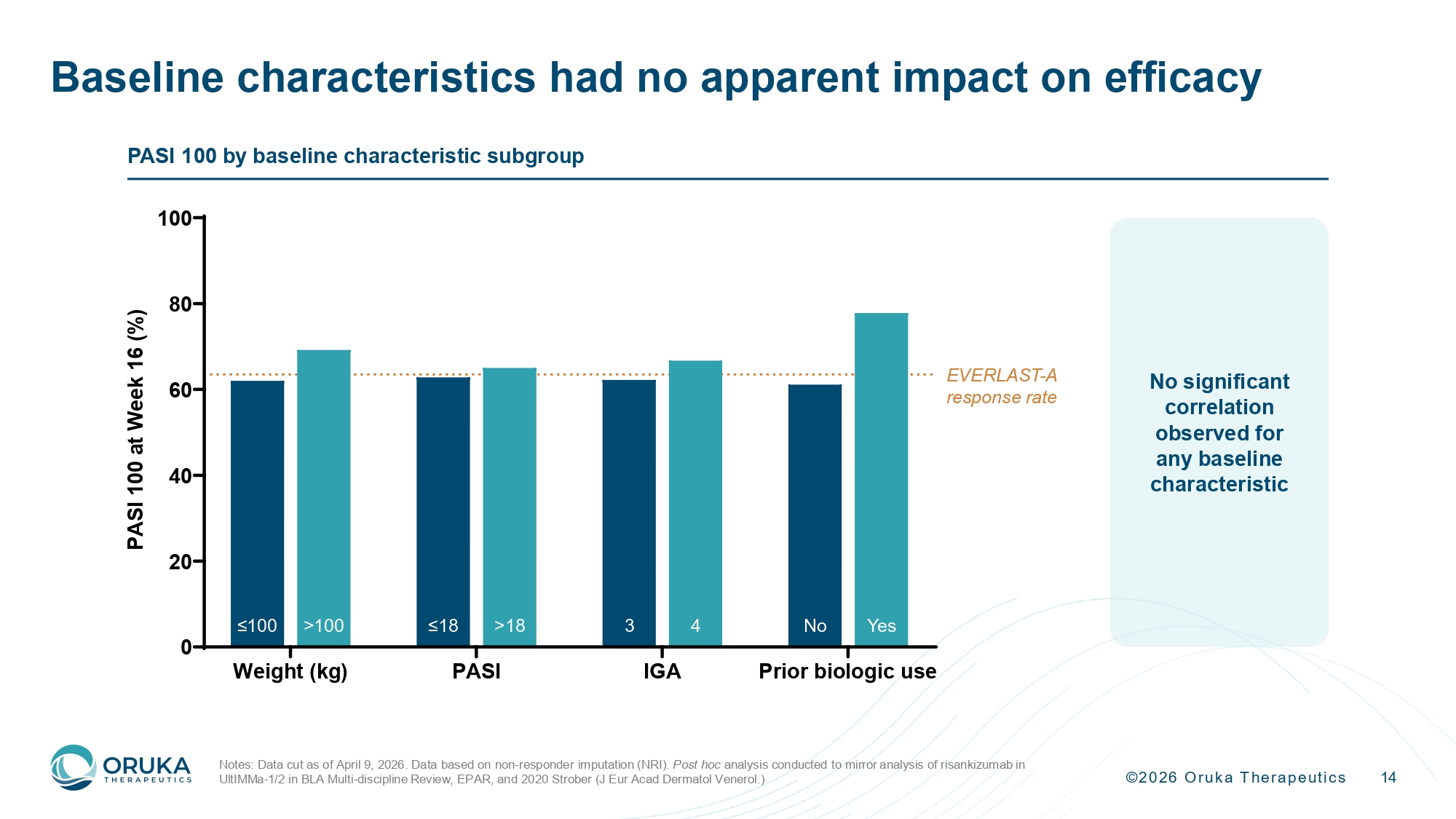
©2026 Oruka Therapeutics 14 Baseline characteristics had no apparent impact on efficacy Notes: Data cut as of April 9, 2026. Data based on non - responder imputation (NRI). Post hoc analysis conducted to mirror analysis of risankizumab in UltIMMa - 1/2 in BLA Multi - discipline Review, EPAR, and 2020 Strober (J Eur Acad Dermatol Venerol.) PASI 100 by baseline characteristic subgroup Weight (kg) PASI IGA Prior biologic use 0 20 40 60 80 100 P A S I 1 0 0 a t W e e k 1 6 ( % ) ≤18 >18 3 4 No Yes ≤100 >100 EVERLAST - A response rate No significant correlation observed for any baseline characteristic
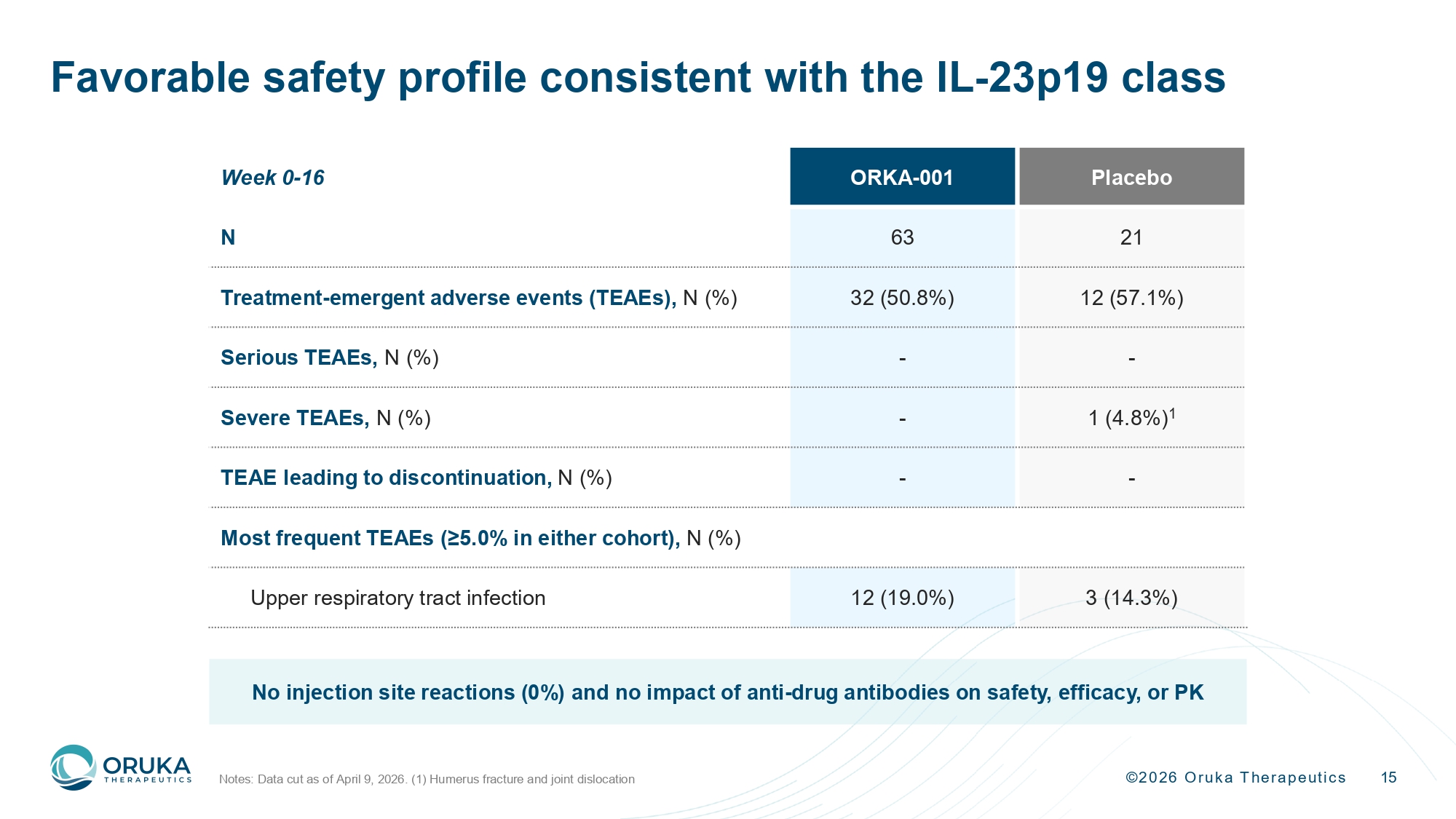
©2026 Oruka Therapeutics 15 Favorable safety profile consistent with the IL - 23p19 class Notes: Data cut as of April 9, 2026. (1) Humerus fracture and joint dislocation Placebo ORKA - 001 Week 0 - 16 21 63 N 12 (57.1%) 32 (50.8%) Treatment - emergent adverse events (TEAEs), N (%) - - Serious TEAEs, N (%) 1 (4.8%) 1 - Severe TEAEs, N (%) - - TEAE leading to discontinuation, N (%) Most frequent TEAEs (≥5.0% in either cohort), N (%) 3 (14.3%) 12 (19.0%) Upper respiratory tract infection No injection site reactions (0%) and no impact of anti - drug antibodies on safety, efficacy, or PK

©2026 Oruka Therapeutics 16 Upcoming EVERLAST data to further elucidate ORKA - 001’s profile EVERLAST - B 16 - week data is intended to support Phase 3 initiation Primary endpoint readout 2027 Longer - term durability data 2H 2026 • Efficacy and durability ‒ Week 28 efficacy (PASI 100, PASI 90, etc.) ‒ 52 - week follow - up for a subset of patients to support annual dosing and off - treatment remission • Updated safety data • Efficacy across dose levels ‒ Week 16 efficacy (primary endpoint) (PASI 100, PASI 90, etc.) • Safety data up to Week 16

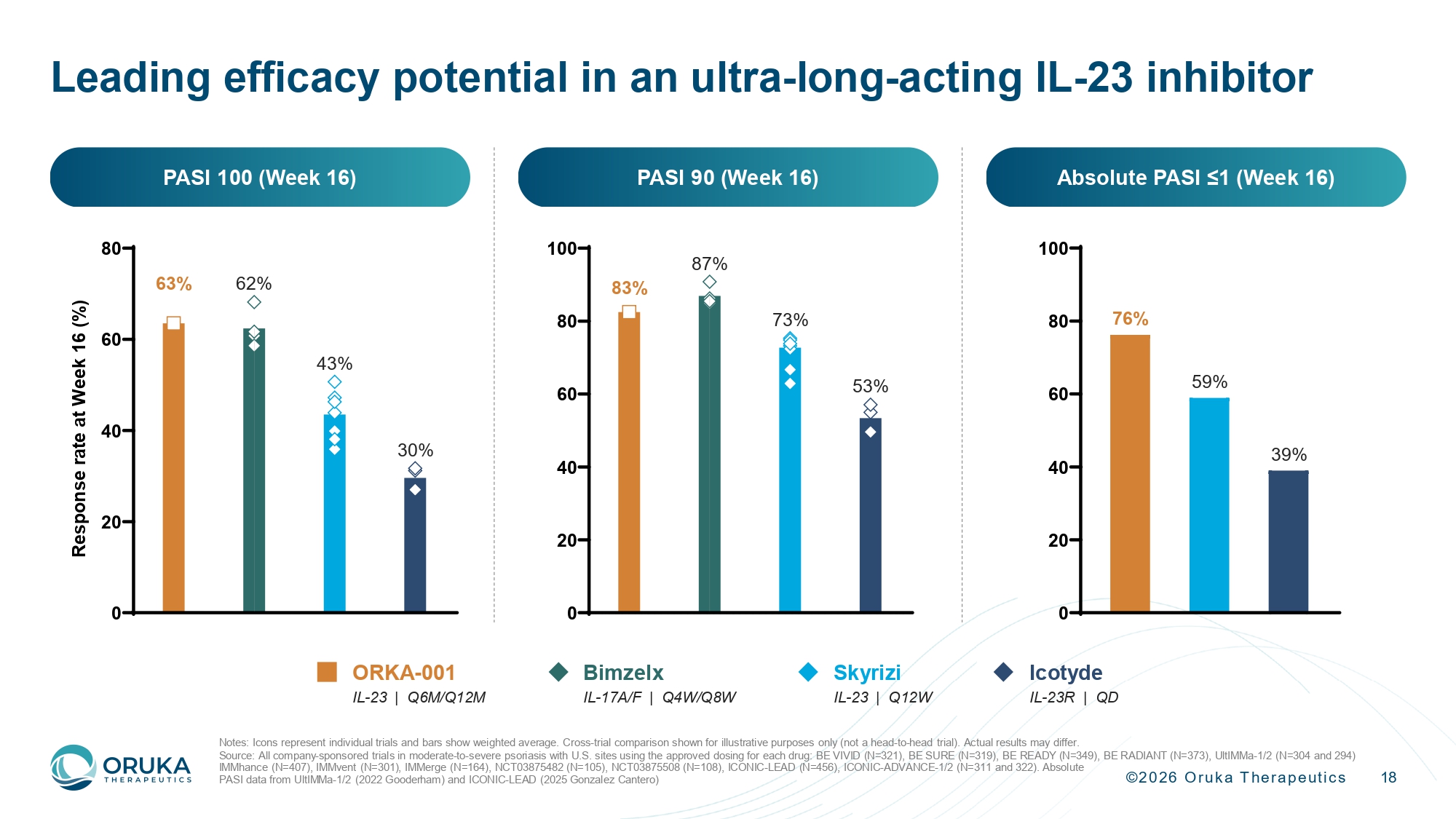
©2026 Oruka Therapeutics 18 Leading efficacy potential in an ultra - long - acting IL - 23 inhibitor IMMhance (N=407), IMMvent (N=301), IMMerge (N=164), NCT03875482 (N=105), NCT03875508 (N=108), ICONIC - LEAD (N=456), ICONIC - ADVANC E - 1/2 (N=311 and 322). Absolute PASI data from UltIMMa - 1/2 (2022 Gooderham) and ICONIC - LEAD (2025 Gonzalez Cantero) 0 20 40 60 80 100 83% 87% 73% 53% 0 20 40 60 80 R e s p o n s e r a t e a t W e e k 1 6 ( % ) 63% 62 % 43 % 30 % Absolute PASI ≤1 (Week 16) PASI 100 (Week 16) PASI 90 (Week 16) 59% 39 % 76% ORKA - 001 IL - 23 | Q6M/Q12M Skyrizi IL - 23 | Q12W Bimzelx IL - 17A/F | Q4W/Q8W Icotyde IL - 23R | QD Notes: Icons represent individual trials and bars show weighted average. Cross - trial comparison shown for illustrative purposes only (not a head - to - head trial). Actual results may differ. Source: All company - sponsored trials in moderate - to - severe psoriasis with U.S. sites using the approved dosing for each drug: BE VIVID (N=321), BE SURE (N=319), BE READY (N=349), BE RADIANT (N=373), UltIMMa - 1/2 (N=304 and 294)
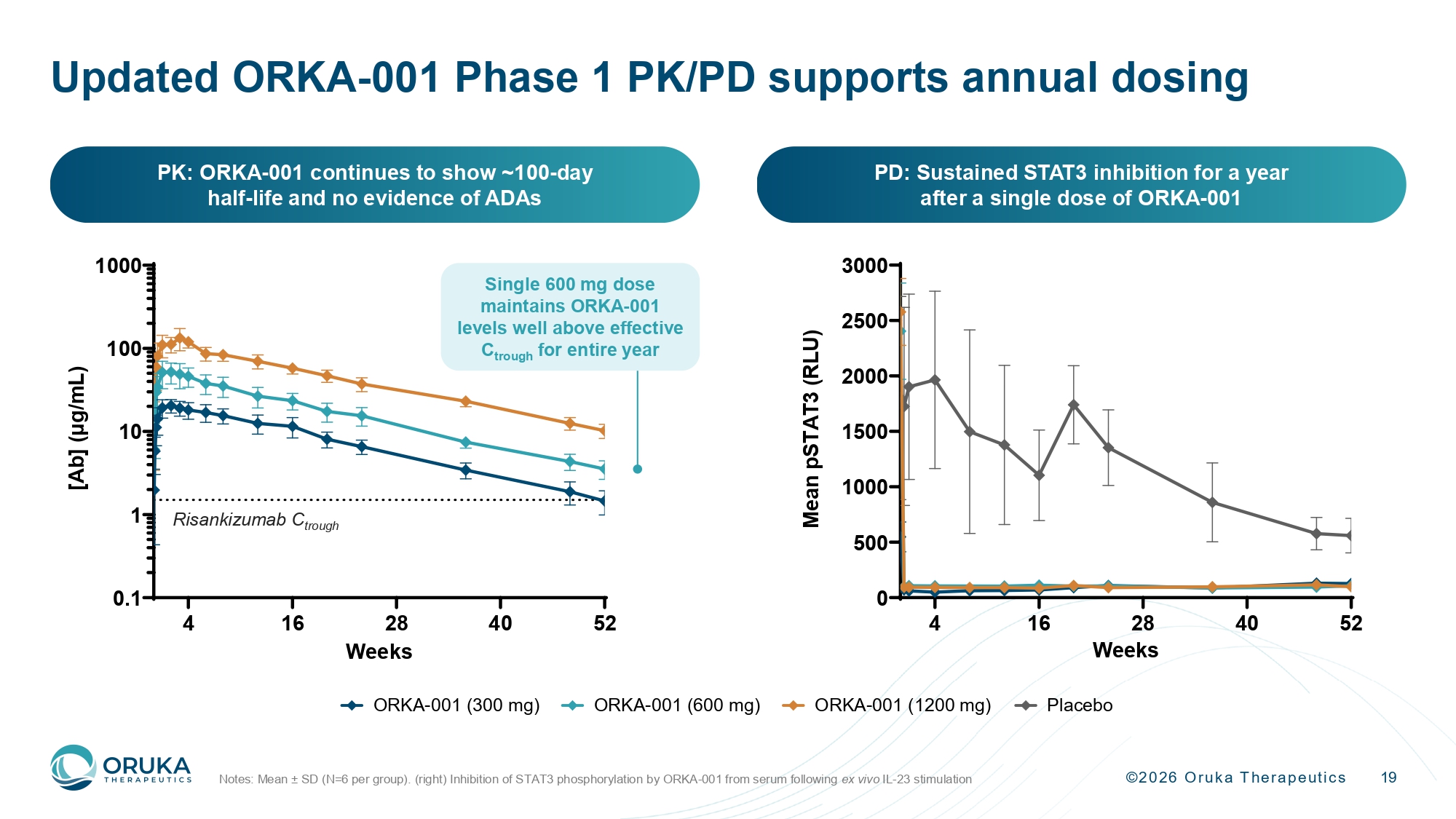
©2026 Oruka Therapeutics 19 Updated ORKA - 001 Phase 1 PK/PD supports annual dosing Notes: Mean ± SD (N=6 per group). (right) Inhibition of STAT3 phosphorylation by ORKA - 001 from serum following ex vivo IL - 23 stimulation 4 16 28 40 52 0 500 1000 1500 2000 2500 3000 Weeks M e a n p S T A T 3 ( R L U ) PD: Sustained STAT3 inhibition for a year after a single dose of ORKA - 001 PK: ORKA - 001 continues to show ~100 - day half - life and no evidence of ADAs 0 8 16 24 32 40 48 0 500 1000 1500 2000 2500 3000 Weeks M e a n p S T A T 3 ( R L U ) Placebo ORKA-001 (300 mg) ORKA-001 (600 mg) ORKA-001 (1200 mg) Risankizumab C trough 4 16 28 40 52 0.1 1 10 100 1000 Weeks [ A b ] ( μ g / m L ) Single 600 mg dose maintains ORKA - 001 levels well above effective C trough for entire year
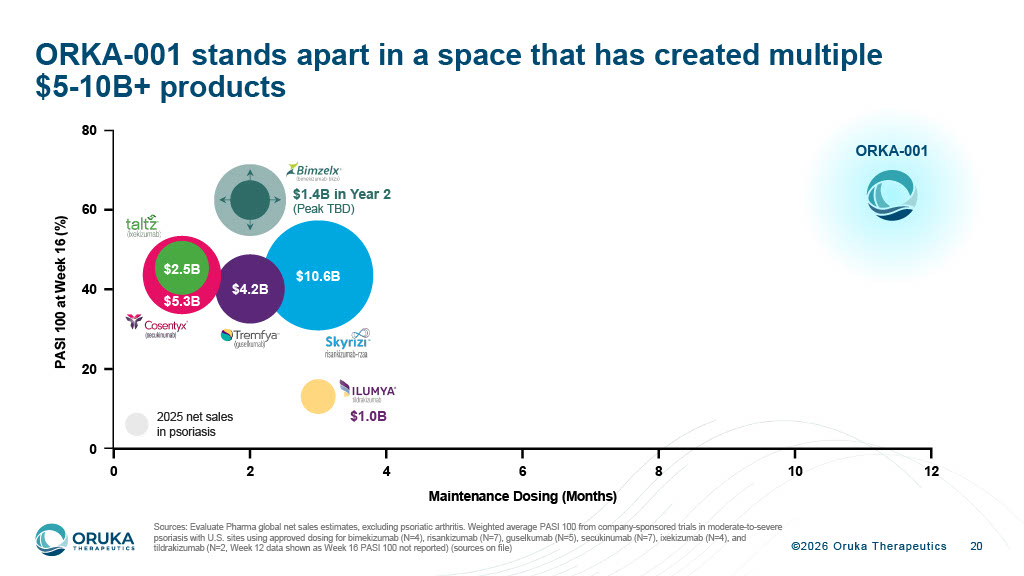
©2026 Oruka Therapeutics 0 2 4 6 8 10 12 0 20 40 60 80 Maintenance Dosing (Months) P A S I 1 0 0 a t W e e k 1 6 ( % ) $10.6B $4.2B $5.3B $2.5B $1.0B $1.4B in Year 2 (Peak TBD) 2025 net sales in psoriasis 20 ORKA - 001 stands apart in a space that has created multiple $5 - 10B+ products Sources: Evaluate Pharma global net sales estimates, excluding psoriatic arthritis. Weighted average PASI 100 from company - spons ored trials in moderate - to - severe psoriasis with U.S. sites using approved dosing for bimekizumab (N=4), risankizumab (N=7), guselkumab (N=5), secukinumab (N=7 ), ixekizumab (N=4), and tildrakizumab (N=2, Week 12 data shown as Week 16 PASI 100 not reported) (sources on file) ORKA - 001
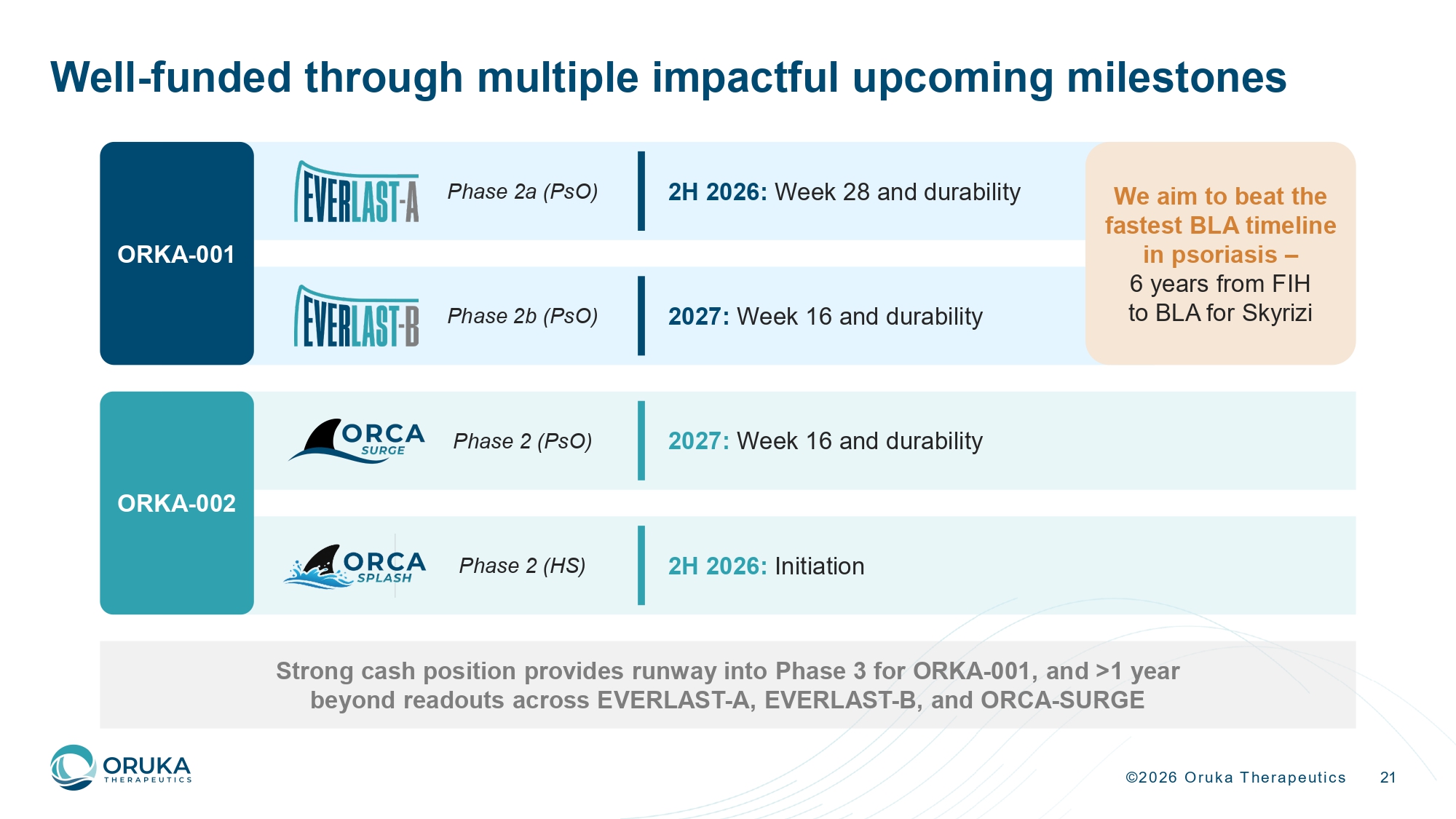
©2026 Oruka Therapeutics 21 Well - funded through multiple impactful upcoming milestones Strong cash position provides runway into Phase 3 for ORKA - 001, and >1 year beyond readouts across EVERLAST - A, EVERLAST - B, and ORCA - SURGE Phase 2 (PsO) 2027: Week 16 and durability Phase 2a (PsO) 2H 2026: Week 28 and durability Phase 2b (PsO) 2027: Week 16 and durability ORKA - 001 We aim to beat the fastest BLA timeline in psoriasis – 6 years from FIH to BLA for Skyrizi 2H 2026: Initiation Phase 2 (HS) ORKA - 002
