
Study 302 Phase 2/3 Topline Data in Males with Mild-to-Moderate Pattern Hair Loss April 2026

agenda 2 1 Opening Remarks 2 Study ‘302’ results 3 KOL Discussion 4 VDPHL01 Commercial Opportunity 5 Closing Remarks 6 Q&A Reid Waldman, M.D. Chief Executive Officer, Veradermics Mark Neumann Chief Commercial & Strategy Officer, Veradermics Dominic Carrano Chief Financial Officer, Veradermics Maryanne Makredes Senna, M.D. Veradermics Scientific Advisory Board Member Beth Israel Lahey Health, Harvard Medical School MANE Speakers Joined by APRIL 2026

This presentation contains forward-looking statements that involve substantial risks and uncertainties. All statements other than historical factual information are forward-looking statements, including without limitation statements regarding our product development activities for VDPHL01 and ongoing clinical trials; the ability of clinical trials to demonstrate safety and efficacy of VDPHL01; the beneficial characteristics, and the potential safety, efficacy and therapeutic effects of VDPHL01; our ability to develop and advance our potential future product candidates and programs; our ability to pursue and execute our strategy for our indications, business, programs and technology; our ability to leverage existing programs and to progress additional programs, the timing of investigational new drug application submissions; the timing of and our ability to obtain and maintain regulatory approval of our product candidates; our ability to compete with companies currently selling, marketing or engaged in the development of treatments for diseases that our product candidates are designed to target, including pattern hair loss (PHL); our estimates regarding the size and growth potential of the commercial opportunity for VDPHL01 and our current product candidates or other product candidates we may identify and pursue, and our ability to serve those markets; our and our collaborators’ ability to protect our intellectual property for our products; our ability to enter into future license agreements and collaborations; regulatory developments; objectives for future operations and other estimates contained herein. In some cases, you can identify forward-looking statements because they contain words such as “may,” “will,” “shall,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these words or other similar expressions that concern our expectations, strategy, plans or intentions, although not all forward-looking statements are accompanied by such words. Forward-looking statements are based on assumptions and assessments made by our management in light of their experience and perceptions of historical trends, current conditions, expected future developments and other factors they believe to be appropriate, and speak only as of the date of this presentation. Disclaimer 3 Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or other events to be materially different from any future results, performance or other events expressed or implied by the forward-looking statements. Given these uncertainties, you should not place undue reliance on forward-looking statements. Our actual future results, performance or other events may be materially different from what we expect. Except as required by law, we assume no obligation to update these forward-looking statements, or to update the reasons actual results could differ materially from those anticipated in these forward- looking statements, even if new information becomes available in the future. Market data and industry information used throughout this presentation are based on management’s knowledge of the industry and the good faith estimates of management. We also relied, to the extent available, upon management’s review of independent industry surveys and publications and other publicly available information prepared by a number of third-party sources. All of the market data and industry information used in this presentation involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. Although we believe that these sources are reliable as of their respective dates, we cannot guarantee the accuracy or completeness of this information, and we have not independently verified this information. Projections, assumptions and estimates of our future performance and the future performance of the industry in which we operate are necessarily subject to a high degree of uncertainty and risk due to a variety of factors. These and other factors could cause results to differ materially from those expressed in our estimates and beliefs and in the estimates prepared by independent parties. The trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of such products. This presentation discusses potential future product candidates that are investigational only and have not yet been approved for marketing by the U.S. Food and Drug Administration. No representation is made as to the safety or effectiveness of these potential future product candidates for the use for which such potential future product candidates are being studied. APRIL 2026
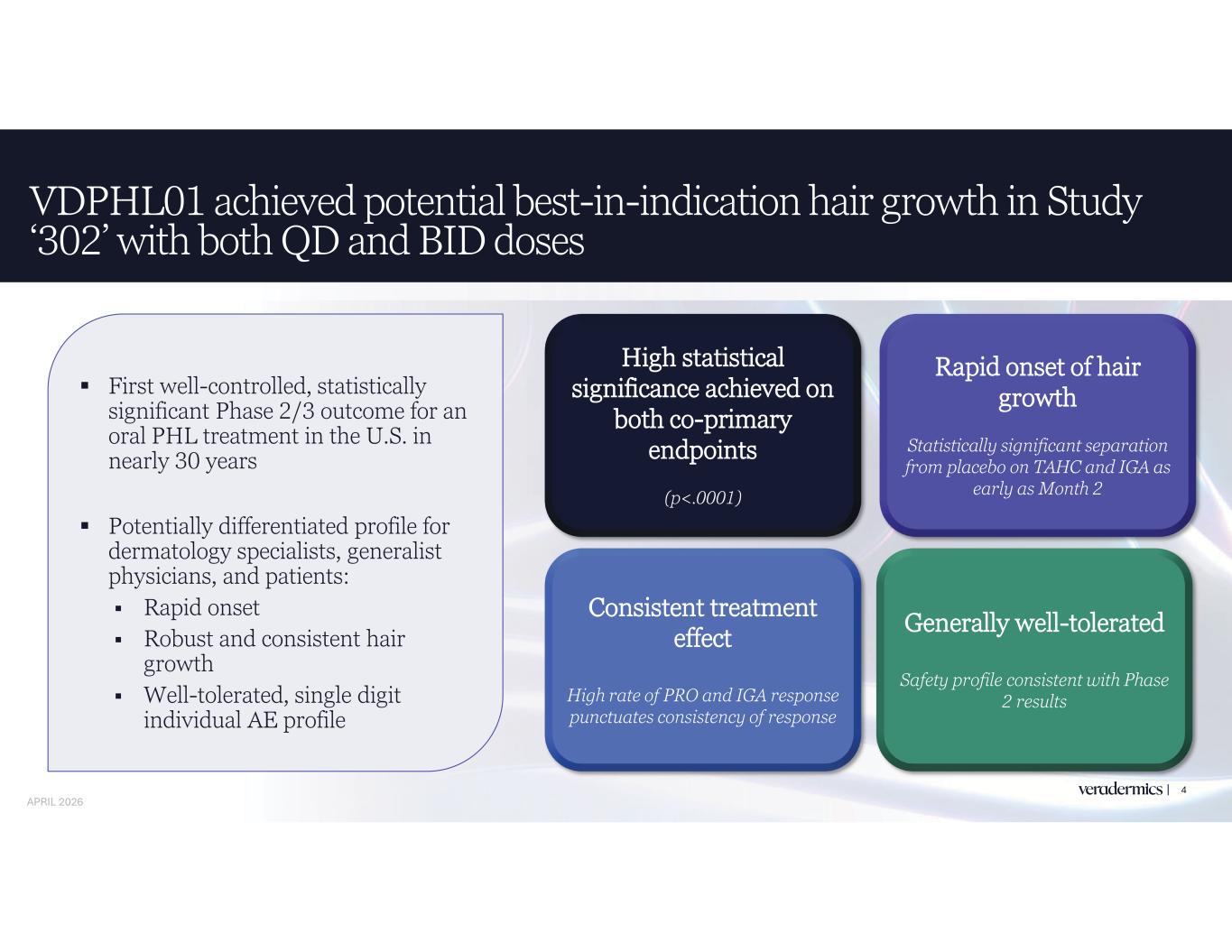
4 First well-controlled, statistically significant Phase 2/3 outcome for an oral PHL treatment in the U.S. in nearly 30 years Potentially differentiated profile for dermatology specialists, generalist physicians, and patients: Rapid onset Robust and consistent hair growth Well-tolerated, single digit individual AE profile VDPHL01 achieved potential best-in-indication hair growth in Study ‘302’ with both QD and BID doses High statistical significance achieved on both co-primary endpoints (p<.0001) Consistent treatment effect High rate of PRO and IGA response punctuates consistency of response Rapid onset of hair growth Statistically significant separation from placebo on TAHC and IGA as early as Month 2 Generally well-tolerated Safety profile consistent with Phase 2 results APRIL 2026
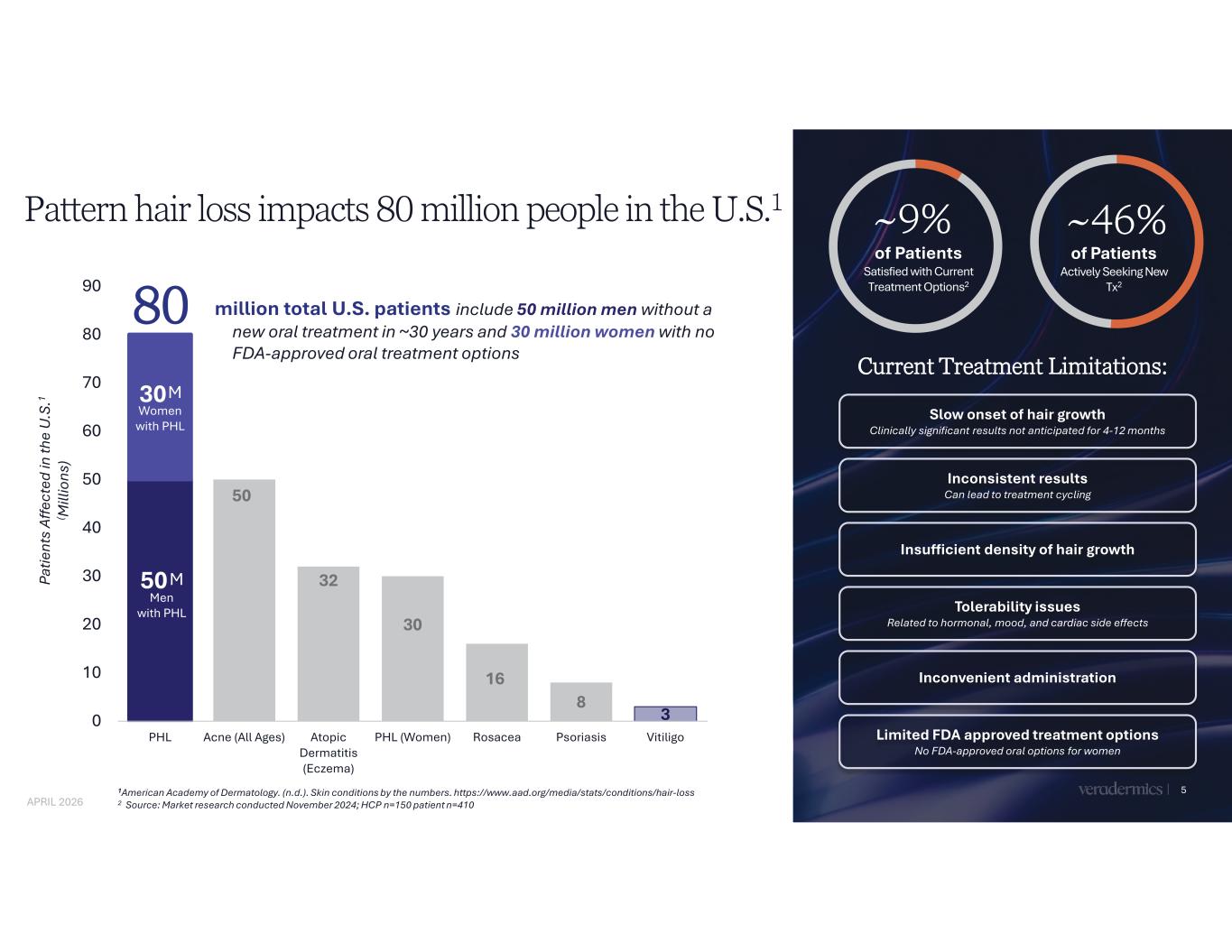
50 50 32 30 16 8 3 80 0 10 20 30 40 50 60 70 80 90 PHL Acne (All Ages) Atopic Dermatitis (Eczema) PHL (Women) Rosacea Psoriasis Vitiligo Pattern hair loss impacts 80 million people in the U.S.1 5 million total U.S. patients include 50 million men without a new oral treatment in ~30 years and 30 million women with no FDA-approved oral treatment options M Men with PHL Pa tie nt s Af fe ct ed in th e U .S .1 ( M ill io ns ) 1American Academy of Dermatology. (n.d.). Skin conditions by the numbers. https://www.aad.org/media/stats/conditions/hair-loss 2 Source: Market research conducted November 2024; HCP n=150 patient n=410 Current Treatment Limitations: M Women with PHL 30 of Patients Actively Seeking New Tx2 ~46% of Patients Satisfied with Current Treatment Options2 ~9% Slow onset of hair growth Clinically significant results not anticipated for 4-12 months Inconsistent results Can lead to treatment cycling Insufficient density of hair growth Tolerability issues Related to hormonal, mood, and cardiac side effects Inconvenient administration Limited FDA approved treatment options No FDA-approved oral options for women APRIL 2026
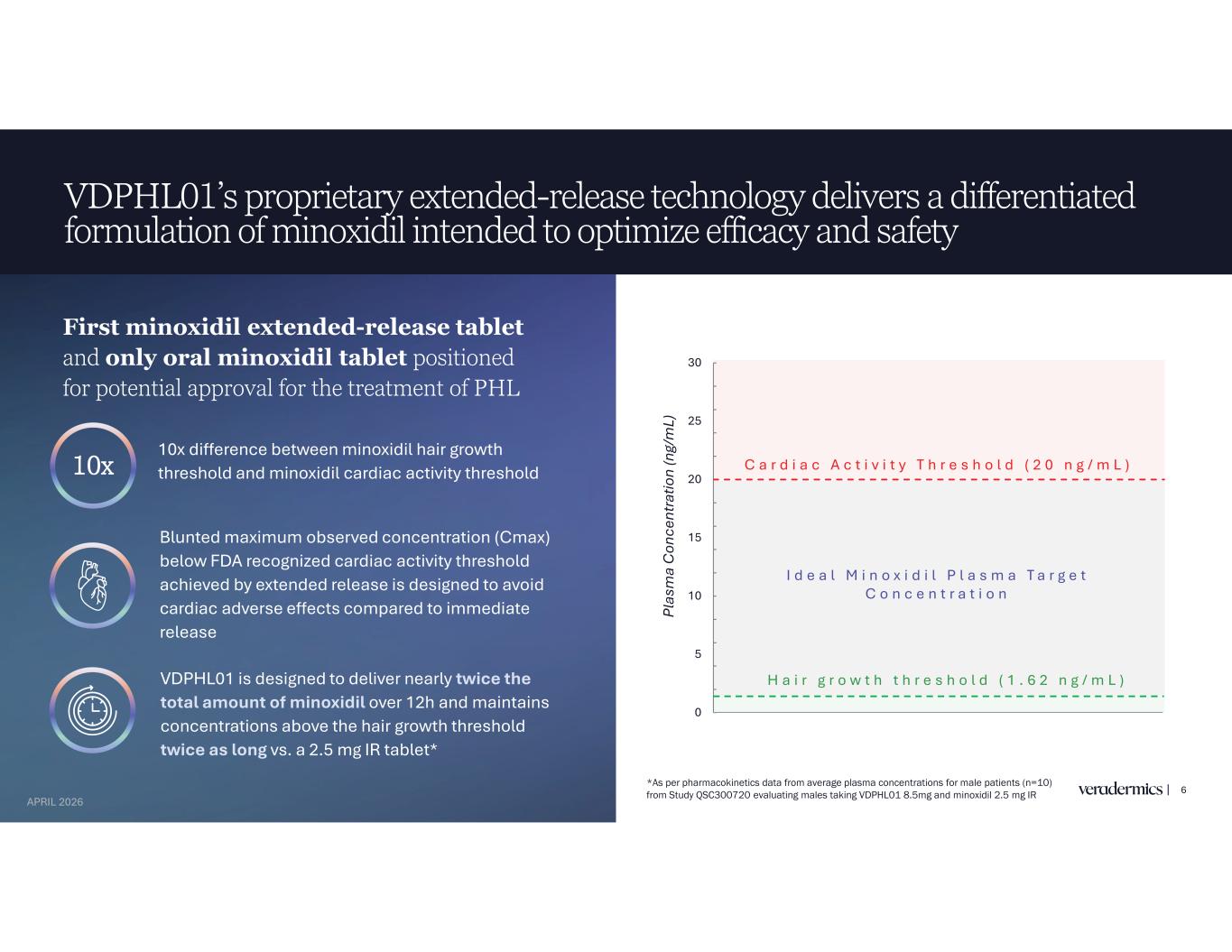
VDPHL01’s proprietary extended-release technology delivers a differentiated formulation of minoxidil intended to optimize efficacy and safety 6 10x difference between minoxidil hair growth threshold and minoxidil cardiac activity threshold VDPHL01 is designed to deliver nearly twice the total amount of minoxidil over 12h and maintains concentrations above the hair growth threshold twice as long vs. a 2.5 mg IR tablet* First minoxidil extended-release tablet and only oral minoxidil tablet positioned for potential approval for the treatment of PHL 10x Blunted maximum observed concentration (Cmax) below FDA recognized cardiac activity threshold achieved by extended release is designed to avoid cardiac adverse effects compared to immediate release *As per pharmacokinetics data from average plasma concentrations for male patients (n=10) from Study QSC300720 evaluating males taking VDPHL01 8.5mg and minoxidil 2.5 mg IR 0 5 10 15 20 25 30 Pl as m a C on ce nt ra tio n (n g/ m L) C a r d i a c A c t i v i t y T h r e s h o l d ( 2 0 n g / m L ) H a i r g r o w t h t h r e s h o l d ( 1 . 6 2 n g / m L ) I d e a l M i n o x i d i l P l a s m a T a r g e t C o n c e n t r a t i o n APRIL 2026
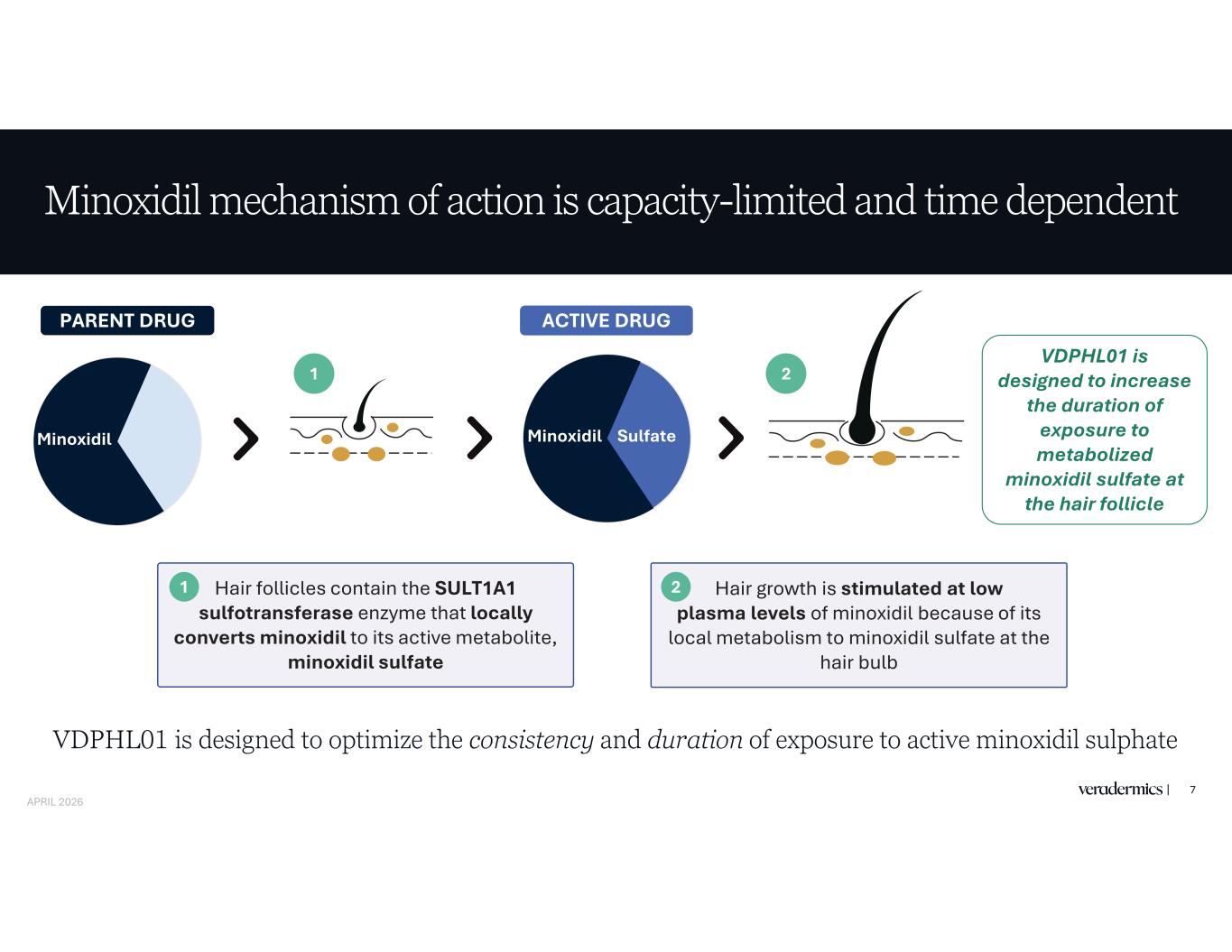
Minoxidil mechanism of action is capacity-limited and time dependent 7 Minoxidil Hair follicles contain the SULT1A1 sulfotransferase enzyme that locally converts minoxidil to its active metabolite, minoxidil sulfate Hair growth is stimulated at low plasma levels of minoxidil because of its local metabolism to minoxidil sulfate at the hair bulb 2 VDPHL01 is designed to increase the duration of exposure to metabolized minoxidil sulfate at the hair follicle PARENT DRUG ACTIVE DRUG 1 1 2 Minoxidil Sulfate VDPHL01 is designed to optimize the consistency and duration of exposure to active minoxidil sulphate APRIL 2026
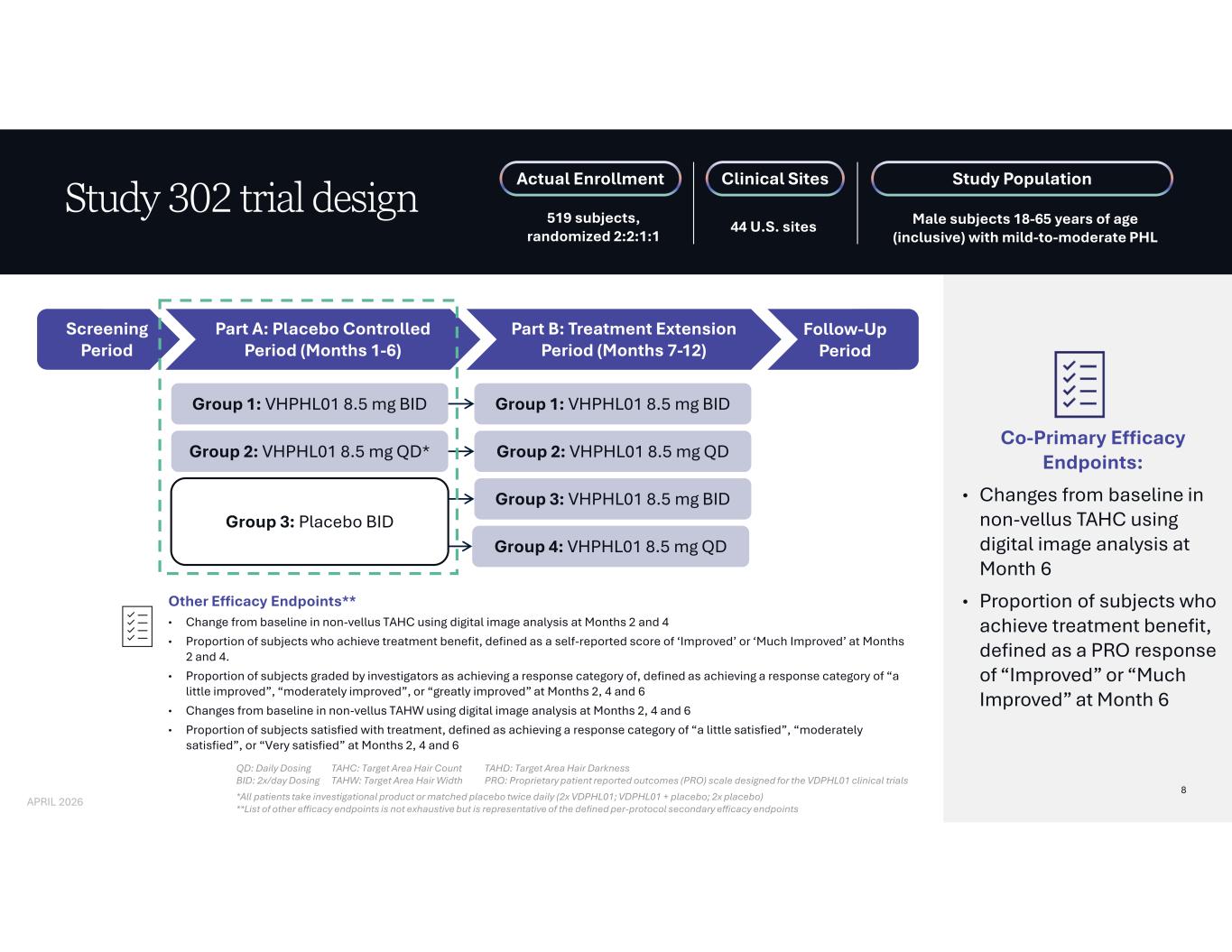
Co-Primary Efficacy Endpoints: • Changes from baseline in non-vellus TAHC using digital image analysis at Month 6 • Proportion of subjects who achieve treatment benefit, defined as a PRO response of “Improved” or “Much Improved” at Month 6 Study 302 trial design QD: Daily Dosing BID: 2x/day Dosing *All patients take investigational product or matched placebo twice daily (2x VDPHL01; VDPHL01 + placebo; 2x placebo) **List of other efficacy endpoints is not exhaustive but is representative of the defined per-protocol secondary efficacy endpoints TAHC: Target Area Hair Count TAHW: Target Area Hair Width TAHD: Target Area Hair Darkness PRO: Proprietary patient reported outcomes (PRO) scale designed for the VDPHL01 clinical trials Other Efficacy Endpoints** • Change from baseline in non-vellus TAHC using digital image analysis at Months 2 and 4 • Proportion of subjects who achieve treatment benefit, defined as a self-reported score of ‘Improved’ or ‘Much Improved’ at Months 2 and 4. • Proportion of subjects graded by investigators as achieving a response category of, defined as achieving a response category of “a little improved”, “moderately improved”, or “greatly improved” at Months 2, 4 and 6 • Changes from baseline in non-vellus TAHW using digital image analysis at Months 2, 4 and 6 • Proportion of subjects satisfied with treatment, defined as achieving a response category of “a little satisfied”, “moderately satisfied”, or “Very satisfied” at Months 2, 4 and 6 Actual Enrollment 519 subjects, randomized 2:2:1:1 Clinical Sites 44 U.S. sites Study Population Male subjects 18-65 years of age (inclusive) with mild-to-moderate PHL Screening Period Part A: Placebo Controlled Period (Months 1-6) Part B: Treatment Extension Period (Months 7-12) Group 1: VHPHL01 8.5 mg BID Group 2: VHPHL01 8.5 mg QD* Group 3: Placebo BID Group 1: VHPHL01 8.5 mg BID Group 2: VHPHL01 8.5 mg QD Group 3: VHPHL01 8.5 mg BID Group 4: VHPHL01 8.5 mg QD Follow-Up Period 8 APRIL 2026
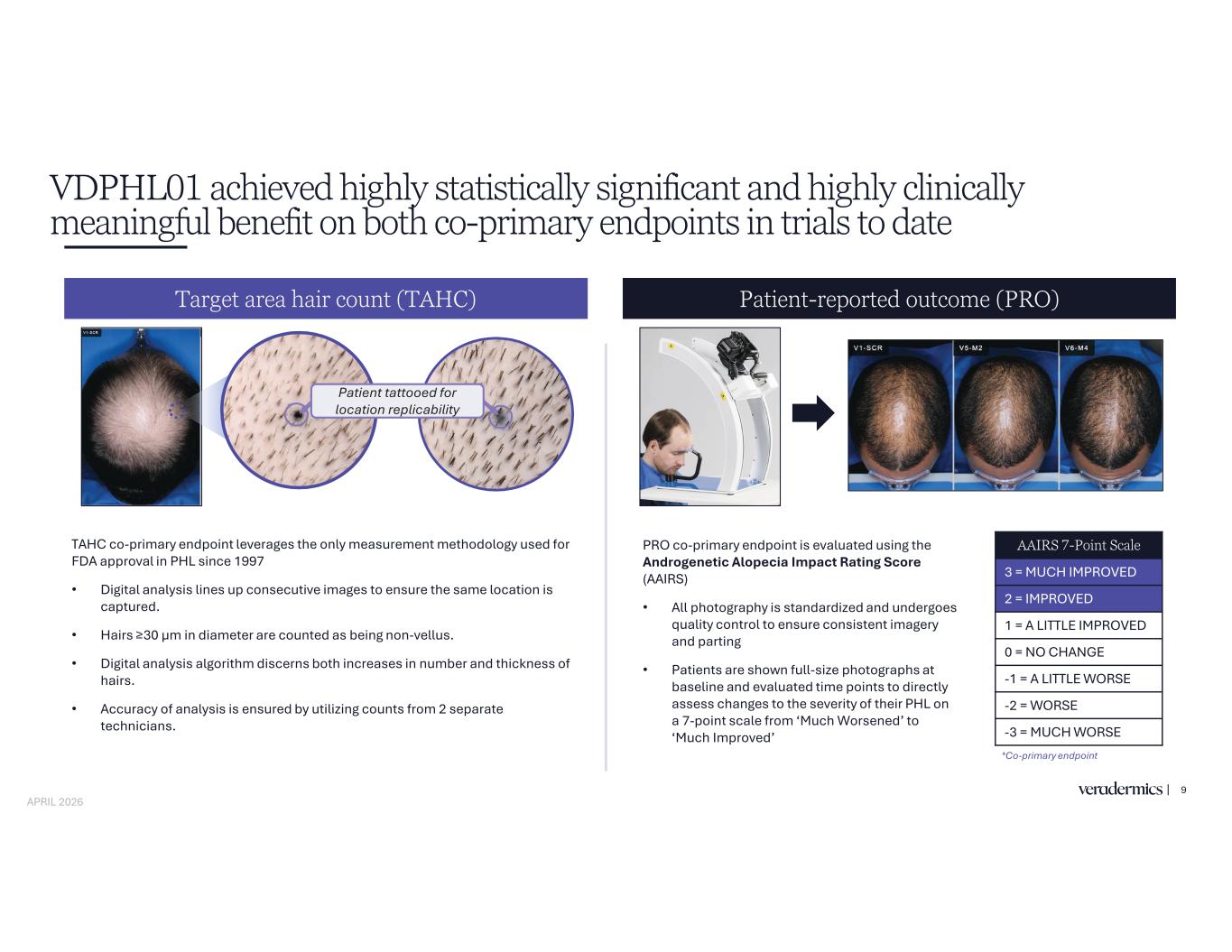
TAHC co-primary endpoint leverages the only measurement methodology used for FDA approval in PHL since 1997 • Digital analysis lines up consecutive images to ensure the same location is captured. • in diameter are counted as being non-vellus. • Digital analysis algorithm discerns both increases in number and thickness of hairs. • Accuracy of analysis is ensured by utilizing counts from 2 separate technicians. VDPHL01 achieved highly statistically significant and highly clinically meaningful benefit on both co-primary endpoints in trials to date Target area hair count (TAHC) Patient-reported outcome (PRO) 9 Patient tattooed for location replicability PRO co-primary endpoint is evaluated using the Androgenetic Alopecia Impact Rating Score (AAIRS) • All photography is standardized and undergoes quality control to ensure consistent imagery and parting • Patients are shown full-size photographs at baseline and evaluated time points to directly assess changes to the severity of their PHL on a 7-point scale from ‘Much Worsened’ to ‘Much Improved’ AAIRS 7-Point Scale 3 = MUCH IMPROVED 2 = IMPROVED 1 = A LITTLE IMPROVED 0 = NO CHANGE -1 = A LITTLE WORSE -2 = WORSE -3 = MUCH WORSE *Co-primary endpoint APRIL 2026
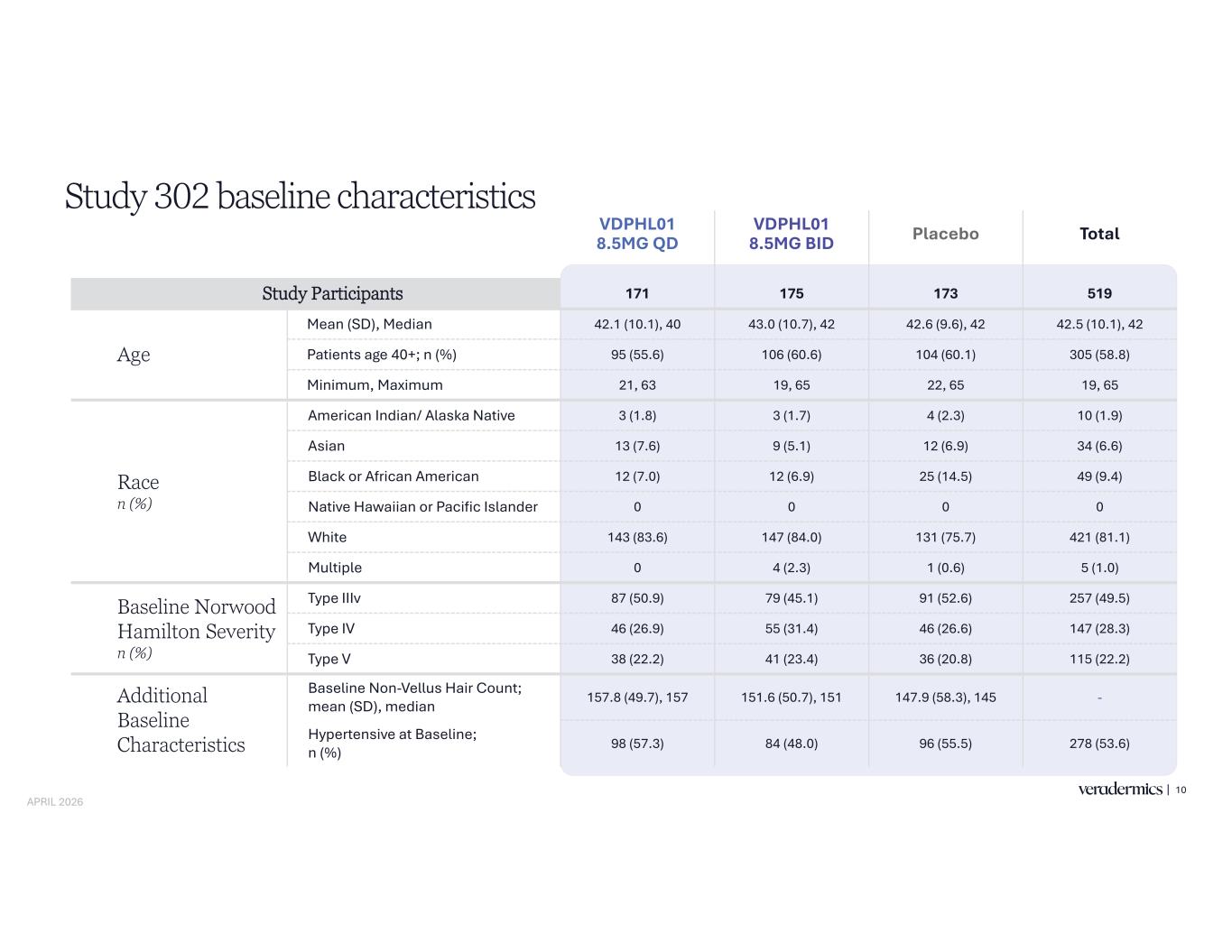
VDPHL01 8.5MG QD VDPHL01 8.5MG BID Placebo Total Study Participants 171 175 173 519 Age Mean (SD), Median 42.1 (10.1), 40 43.0 (10.7), 42 42.6 (9.6), 42 42.5 (10.1), 42 Patients age 40+; n (%) 95 (55.6) 106 (60.6) 104 (60.1) 305 (58.8) Minimum, Maximum 21, 63 19, 65 22, 65 19, 65 Race n (%) American Indian/ Alaska Native 3 (1.8) 3 (1.7) 4 (2.3) 10 (1.9) Asian 13 (7.6) 9 (5.1) 12 (6.9) 34 (6.6) Black or African American 12 (7.0) 12 (6.9) 25 (14.5) 49 (9.4) Native Hawaiian or Pacific Islander 0 0 0 0 White 143 (83.6) 147 (84.0) 131 (75.7) 421 (81.1) Multiple 0 4 (2.3) 1 (0.6) 5 (1.0) Baseline Norwood Hamilton Severity n (%) Type IIIv 87 (50.9) 79 (45.1) 91 (52.6) 257 (49.5) Type IV 46 (26.9) 55 (31.4) 46 (26.6) 147 (28.3) Type V 38 (22.2) 41 (23.4) 36 (20.8) 115 (22.2) Additional Baseline Characteristics Baseline Non-Vellus Hair Count; mean (SD), median 157.8 (49.7), 157 151.6 (50.7), 151 147.9 (58.3), 145 - Hypertensive at Baseline; n (%) 98 (57.3) 84 (48.0) 96 (55.5) 278 (53.6) Study 302 baseline characteristics 10 APRIL 2026
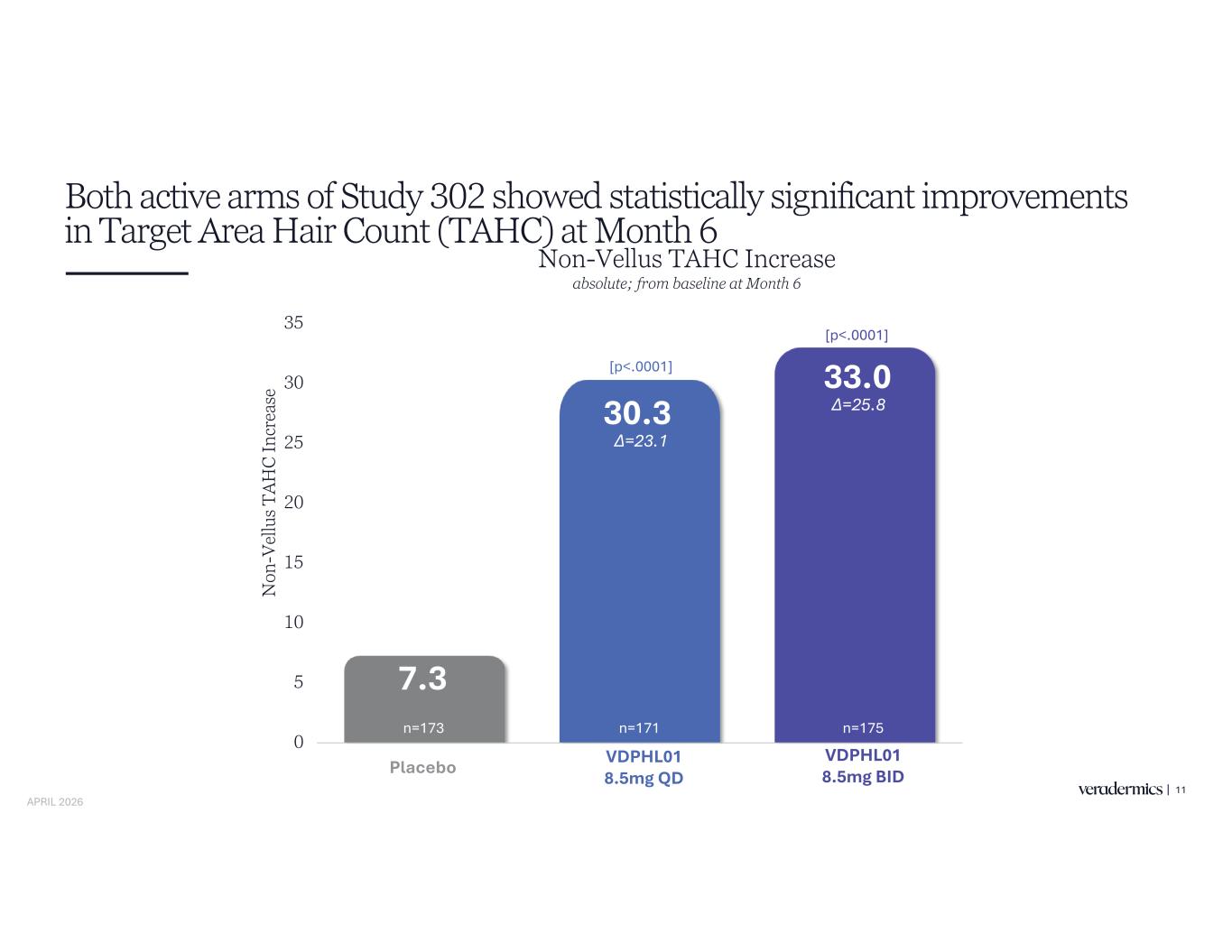
Both active arms of Study 302 showed statistically significant improvements in Target Area Hair Count (TAHC) at Month 6 11 7.3 30.3 33.0 0 5 10 15 20 25 30 35 Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID N on -V el lu s T A H C In cr ea se Non-Vellus TAHC Increase absolute; from baseline at Month 6 [p<.0001] [p<.0001] Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID n=171 n=175n=173 APRIL 2026
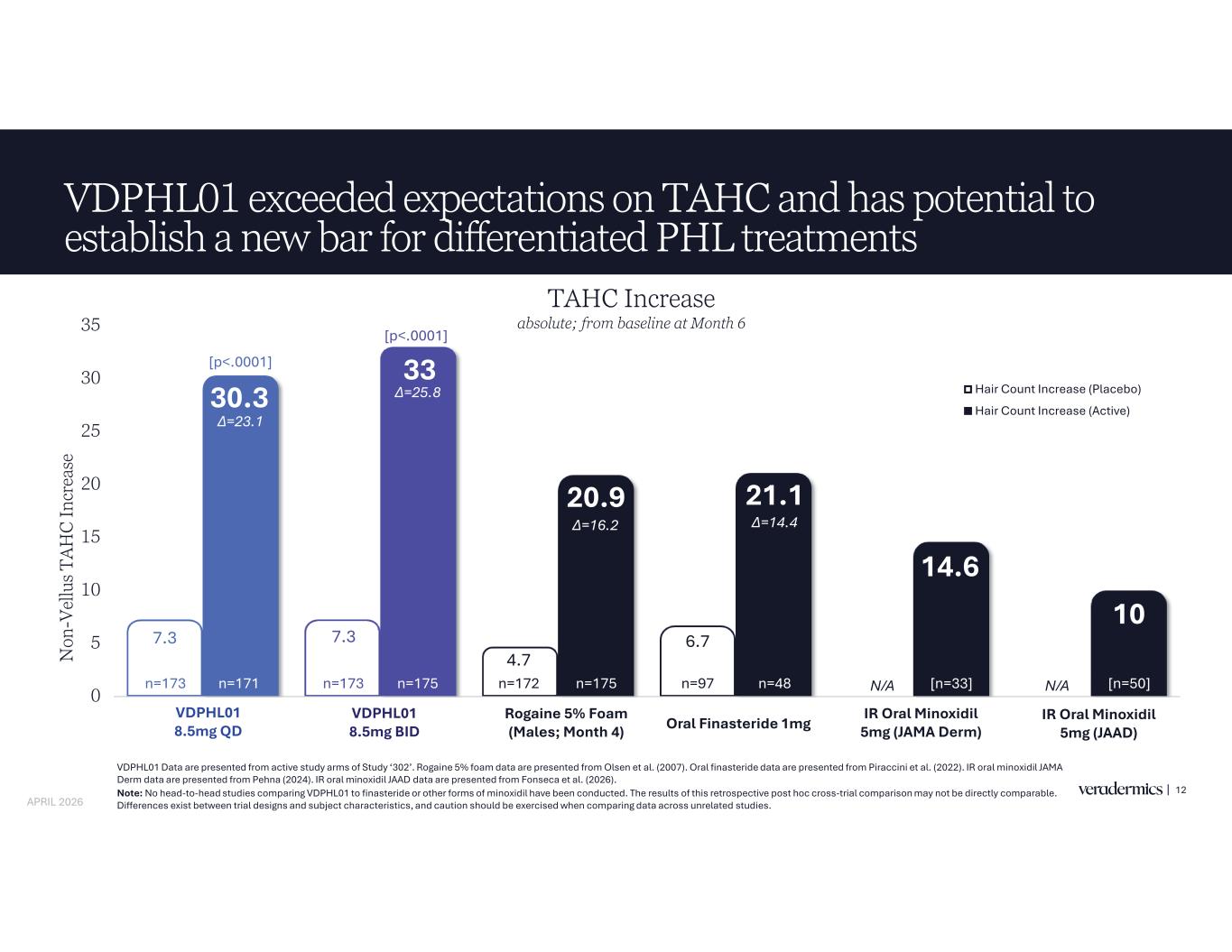
7.3 7.3 4.7 6.7 30.3 33 20.9 21.1 14.6 10 0 5 10 15 20 25 30 35 VDPHL01 8.5mg QD VDPHL01 8.5mg BID Rogaine 5% Foam (Males; Month 4) Oral Finasteride IR Oral Minoxidil 5mg (JAMA Derm) IR Oral Minoxidil 5mg (JAAD) N on -V el lu s T A H C In cr ea se Hair Count Increase (Placebo) Hair Count Increase (Active) VDPHL01 exceeded expectations on TAHC and has potential to establish a new bar for differentiated PHL treatments 12 [n=33] VDPHL01 Data are presented from active study arms of Study ‘302’. Rogaine 5% foam data are presented from Olsen et al. (2007). Oral finasteride data are presented from Piraccini et al. (2022). IR oral minoxidil JAMA Derm data are presented from Pehna (2024). IR oral minoxidil JAAD data are presented from Fonseca et al. (2026). Note: No head-to-head studies comparing VDPHL01 to finasteride or other forms of minoxidil have been conducted. The results of this retrospective post hoc cross-trial comparison may not be directly comparable. Differences exist between trial designs and subject characteristics, and caution should be exercised when comparing data across unrelated studies. [n=180*] [p<.0001] [p<.0001] N/A N/An=173 n=173 n=97n=172 [n=50][n=33]n=171 n=175 n=175 n=48 VDPHL01 8.5mg QD VDPHL01 8.5mg BID ine 5% Foam (Males; Month 4) Oral Finasteride 1mg IR Oral Minoxidil 5mg (JAMA Derm) IR Oral Minoxidil 5mg (JAAD) TAHC Increase absolute; from baseline at Month 6 APRIL 2026
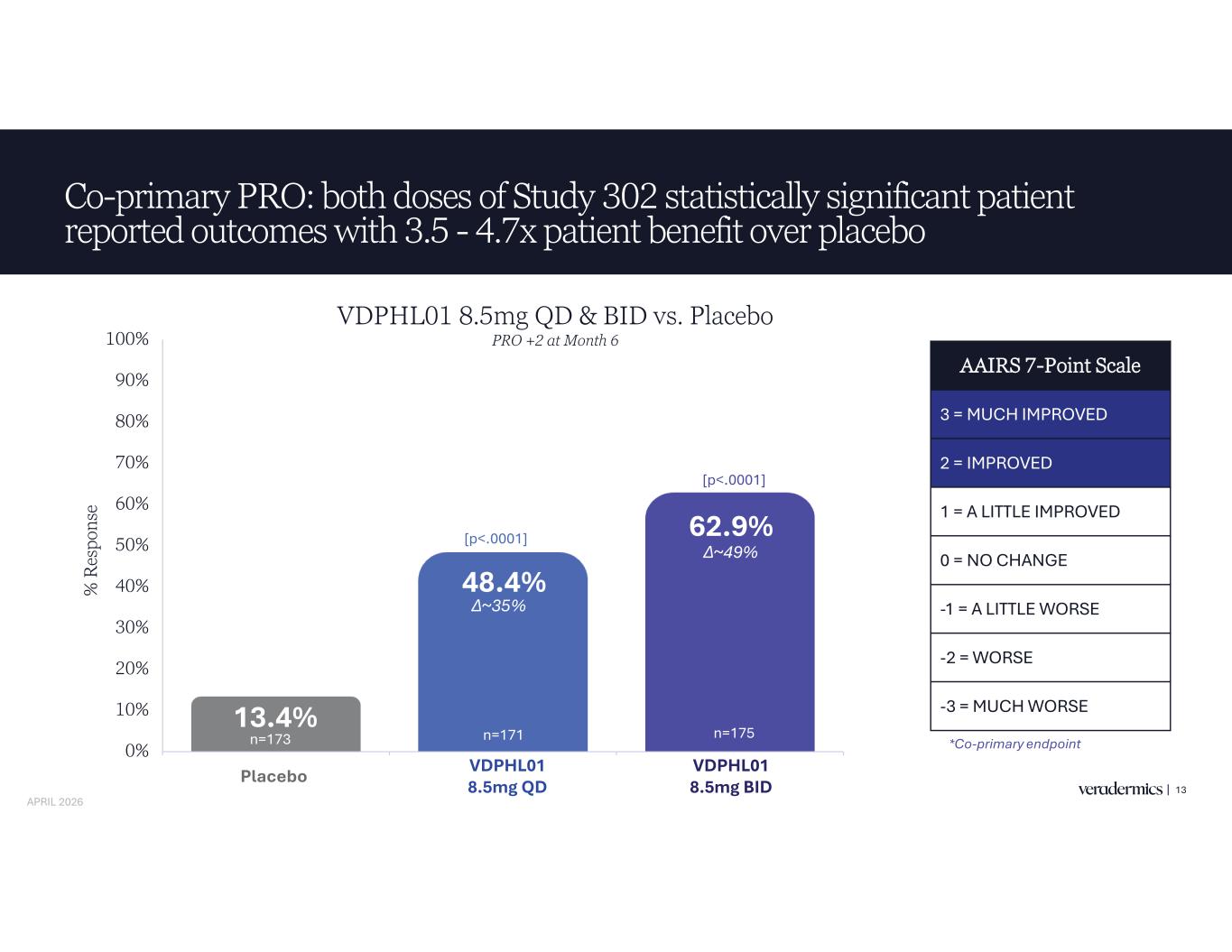
Co-primary PRO: both doses of Study 302 statistically significant patient reported outcomes with 3.5 - 4.7x patient benefit over placebo 13 13.4% 48.4% 62.9% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID % R es po ns e AAIRS 7-Point Scale 3 = MUCH IMPROVED 2 = IMPROVED 1 = A LITTLE IMPROVED 0 = NO CHANGE -1 = A LITTLE WORSE -2 = WORSE -3 = MUCH WORSE [p<.0001] [p<.0001] *Co-primary endpointn=173 Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID VDPHL01 8.5mg QD & BID vs. Placebo PRO +2 at Month 6 n=171 n=175 APRIL 2026
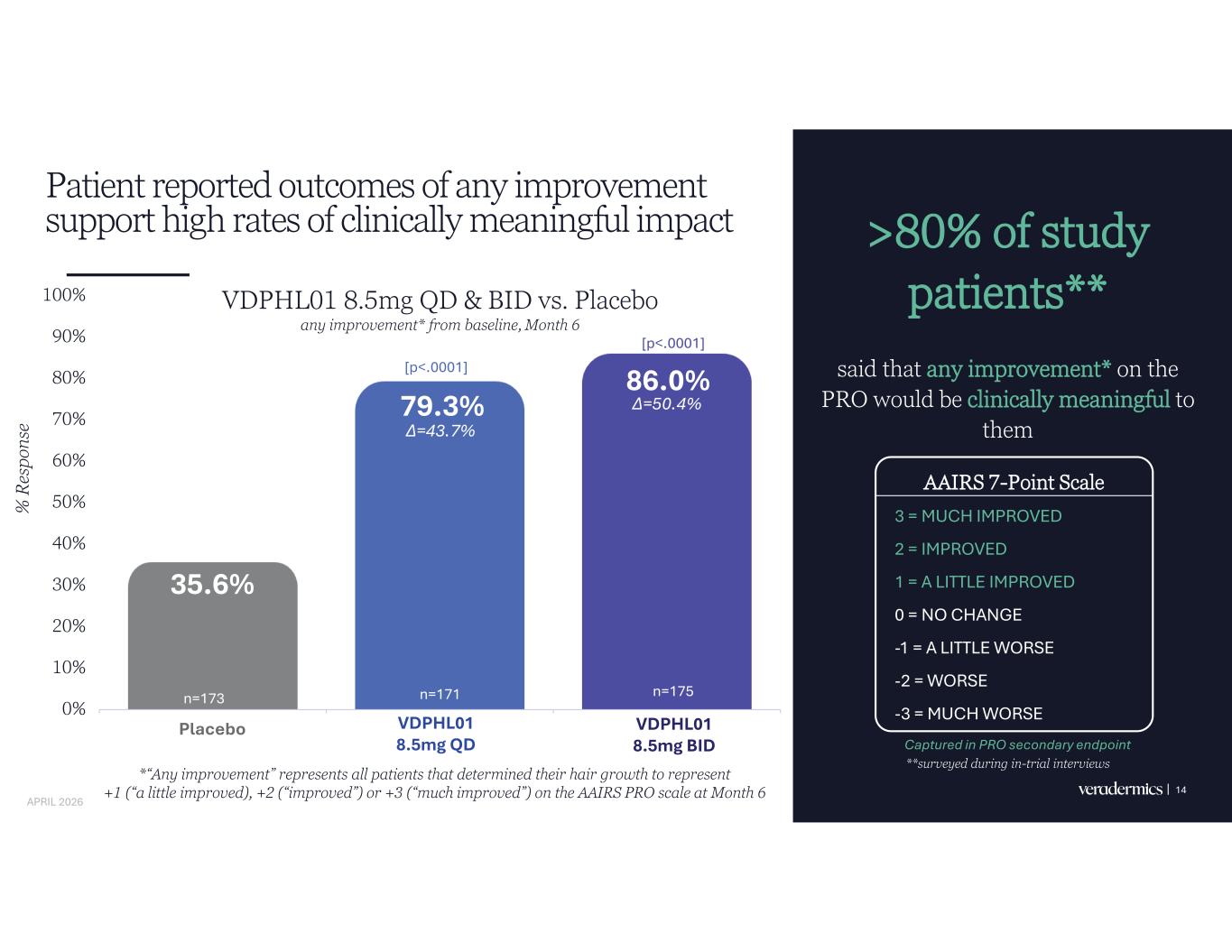
Patient reported outcomes of any improvement support high rates of clinically meaningful impact >80% of study patients** said that any improvement* on the PRO would be clinically meaningful to them 14 35.6% 79.3% 86.0% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID % R es po ns e *“Any improvement” represents all patients that determined their hair growth to represent +1 (“a little improved), +2 (“improved”) or +3 (“much improved”) on the AAIRS PRO scale at Month 6 AAIRS 7-Point Scale 3 = MUCH IMPROVED 2 = IMPROVED 1 = A LITTLE IMPROVED 0 = NO CHANGE -1 = A LITTLE WORSE -2 = WORSE -3 = MUCH WORSE Captured in PRO secondary endpoint [p<.0001] **surveyed during in-trial interviews [p<.0001] n=173 n=171 n=175 VDPHL01 8.5mg QD & BID vs. Placebo any improvement* from baseline, Month 6 Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID APRIL 2026
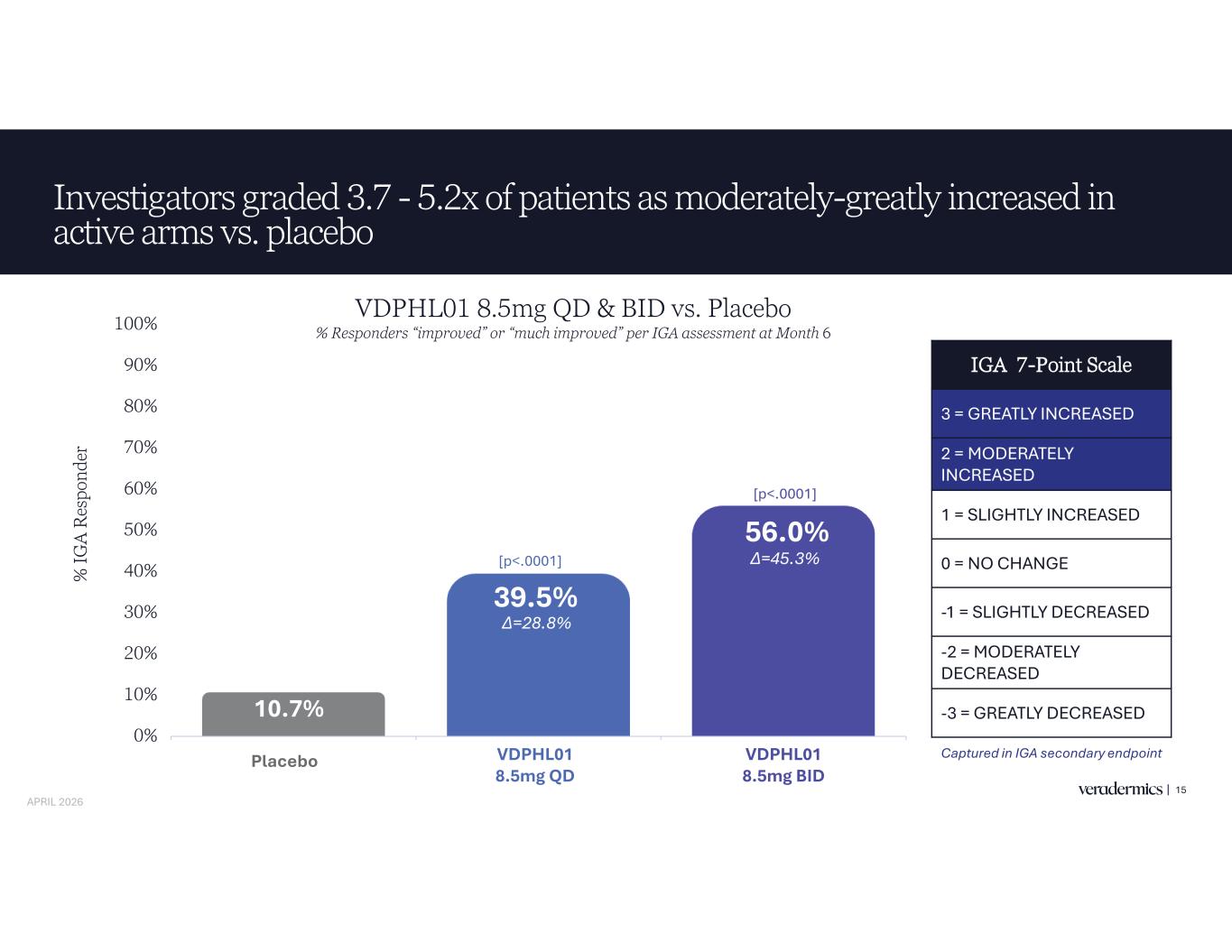
Investigators graded 3.7 - 5.2x of patients as moderately-greatly increased in active arms vs. placebo 15 10.7% 39.5% 56.0% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID % IG A R es po nd er [p<.0001] IGA 7-Point Scale 3 = GREATLY INCREASED 2 = MODERATELY INCREASED 1 = SLIGHTLY INCREASED 0 = NO CHANGE -1 = SLIGHTLY DECREASED -2 = MODERATELY DECREASED -3 = GREATLY DECREASED Captured in IGA secondary endpoint [p<.0001] VDPHL01 8.5mg QD & BID vs. Placebo % Responders “improved” or “much improved” per IGA assessment at Month 6 Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID APRIL 2026
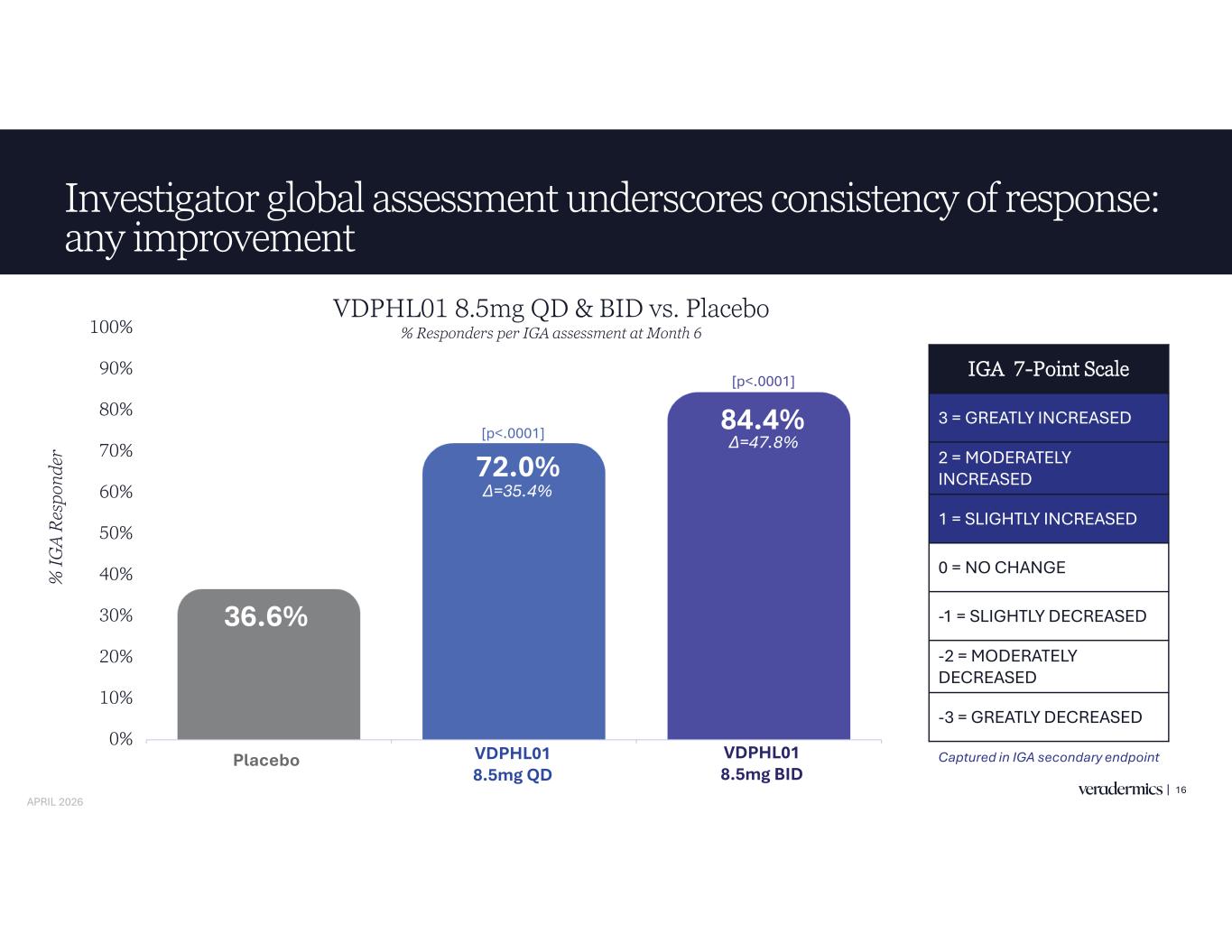
Investigator global assessment underscores consistency of response: any improvement 16 36.6% 72.0% 84.4% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID % IG A R es po nd er [p<.0001] [p<.0001] IGA 7-Point Scale 3 = GREATLY INCREASED 2 = MODERATELY INCREASED 1 = SLIGHTLY INCREASED 0 = NO CHANGE -1 = SLIGHTLY DECREASED -2 = MODERATELY DECREASED -3 = GREATLY DECREASED Captured in IGA secondary endpoint VDPHL01 8.5mg QD & BID vs. Placebo % Responders per IGA assessment at Month 6 Placebo VDPHL01 8.5mg QD VDPHL01 8.5mg BID APRIL 2026
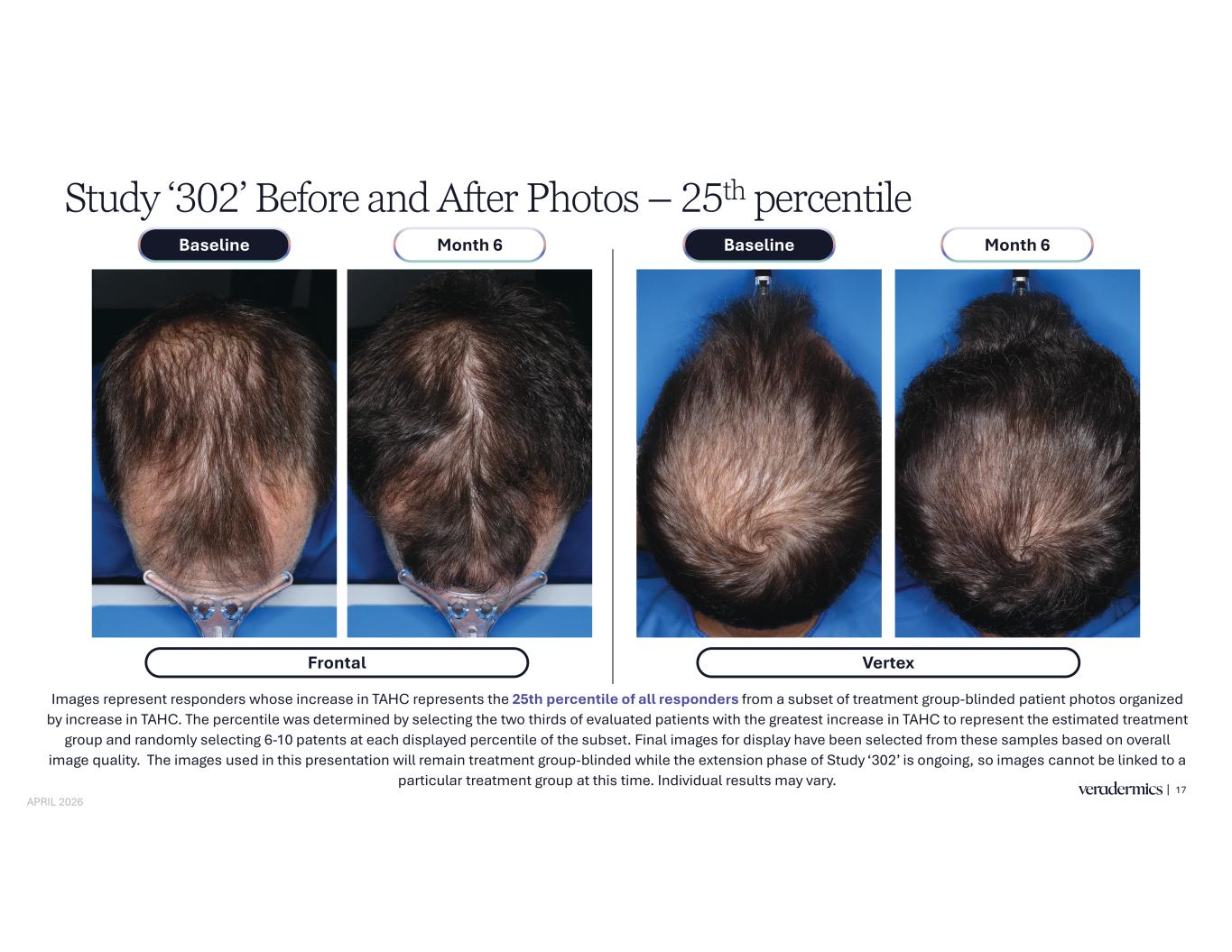
Study ‘302’ Before and After Photos – 25th percentile 17 Images represent responders whose increase in TAHC represents the 25th percentile of all responders from a subset of treatment group-blinded patient photos organized by increase in TAHC. The percentile was determined by selecting the two thirds of evaluated patients with the greatest increase in TAHC to represent the estimated treatment group and randomly selecting 6-10 patents at each displayed percentile of the subset. Final images for display have been selected from these samples based on overall image quality. The images used in this presentation will remain treatment group-blinded while the extension phase of Study ‘302’ is ongoing, so images cannot be linked to a particular treatment group at this time. Individual results may vary. Frontal Month 6Baseline Month 6Baseline Vertex APRIL 2026
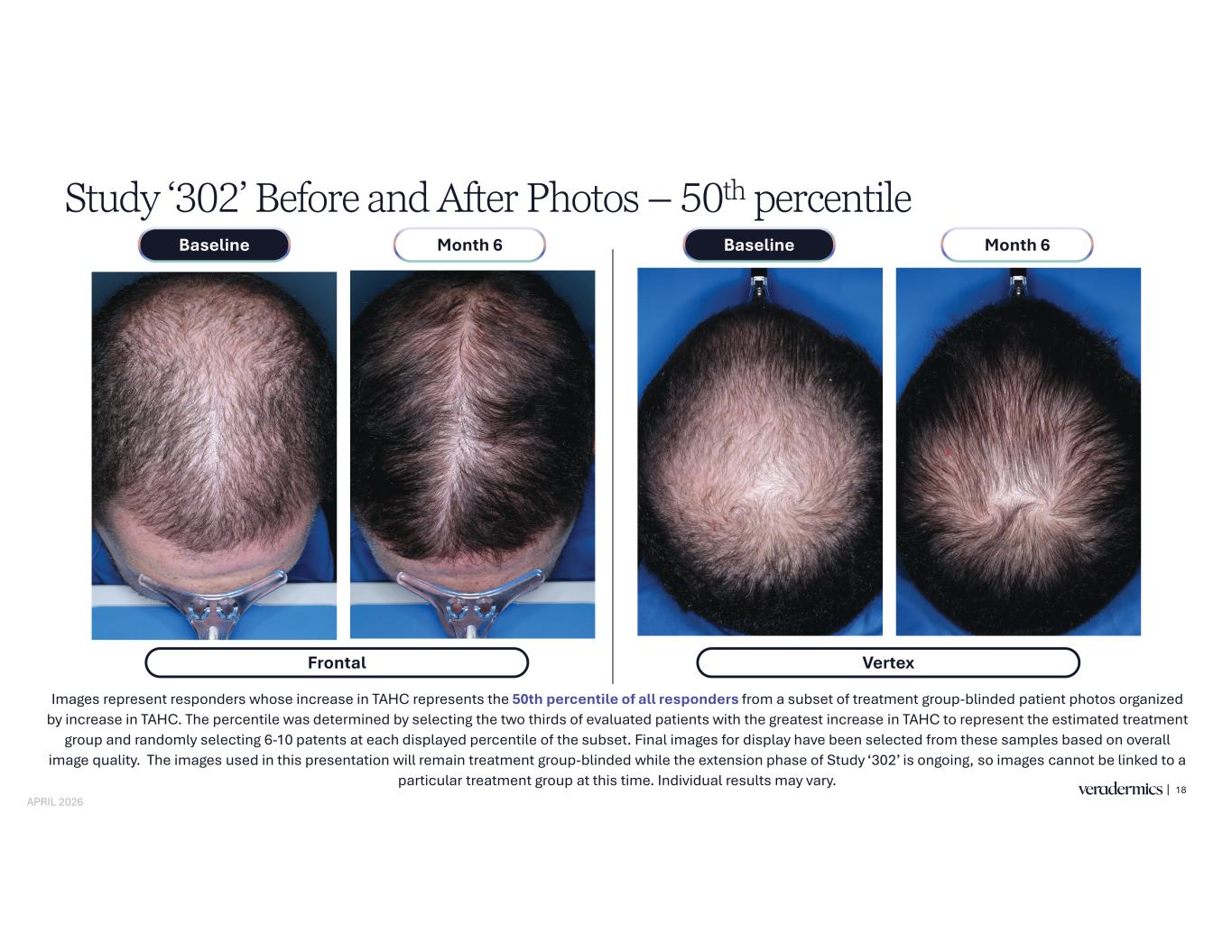
Study ‘302’ Before and After Photos – 50th percentile 18 Images represent responders whose increase in TAHC represents the 50th percentile of all responders from a subset of treatment group-blinded patient photos organized by increase in TAHC. The percentile was determined by selecting the two thirds of evaluated patients with the greatest increase in TAHC to represent the estimated treatment group and randomly selecting 6-10 patents at each displayed percentile of the subset. Final images for display have been selected from these samples based on overall image quality. The images used in this presentation will remain treatment group-blinded while the extension phase of Study ‘302’ is ongoing, so images cannot be linked to a particular treatment group at this time. Individual results may vary. Frontal Month 6Baseline Month 6Baseline Vertex APRIL 2026
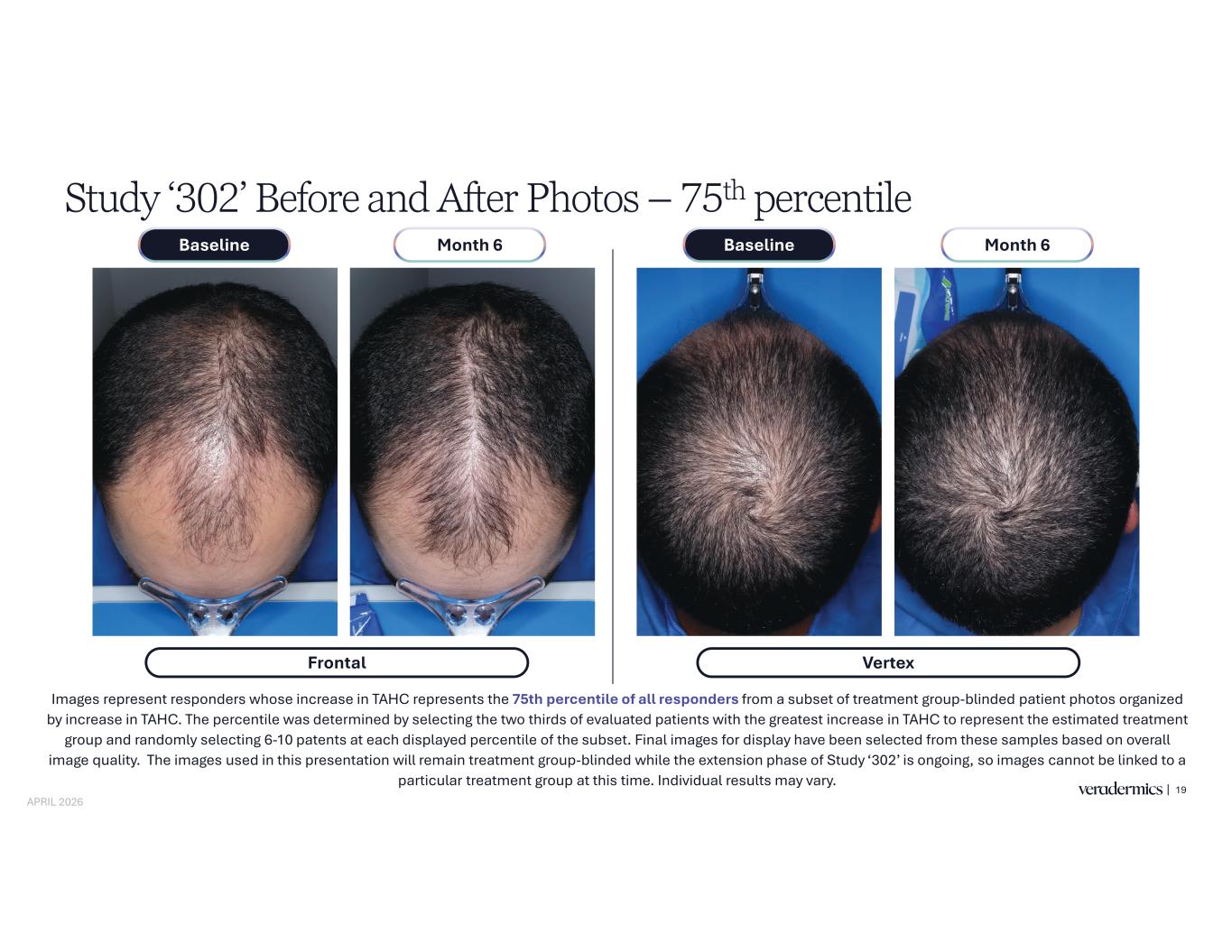
Study ‘302’ Before and After Photos – 75th percentile 19 Images represent responders whose increase in TAHC represents the 75th percentile of all responders from a subset of treatment group-blinded patient photos organized by increase in TAHC. The percentile was determined by selecting the two thirds of evaluated patients with the greatest increase in TAHC to represent the estimated treatment group and randomly selecting 6-10 patents at each displayed percentile of the subset. Final images for display have been selected from these samples based on overall image quality. The images used in this presentation will remain treatment group-blinded while the extension phase of Study ‘302’ is ongoing, so images cannot be linked to a particular treatment group at this time. Individual results may vary. Frontal Month 6Baseline Month 6Baseline Vertex APRIL 2026

Study '302' demonstrated a well-tolerated and safe profile • No treatment-related SAEs • No adverse events of special interest (AESI) of cardiac origin • Overall TEAE rates in active treatment arms were similar to placebo, generally tolerable, and occurred at low to mid single digit rates at most • No clinically significant differences in heart rate, blood pressure, or ECG changes compared to placebo • Lack of observed shedding 20 APRIL 2026
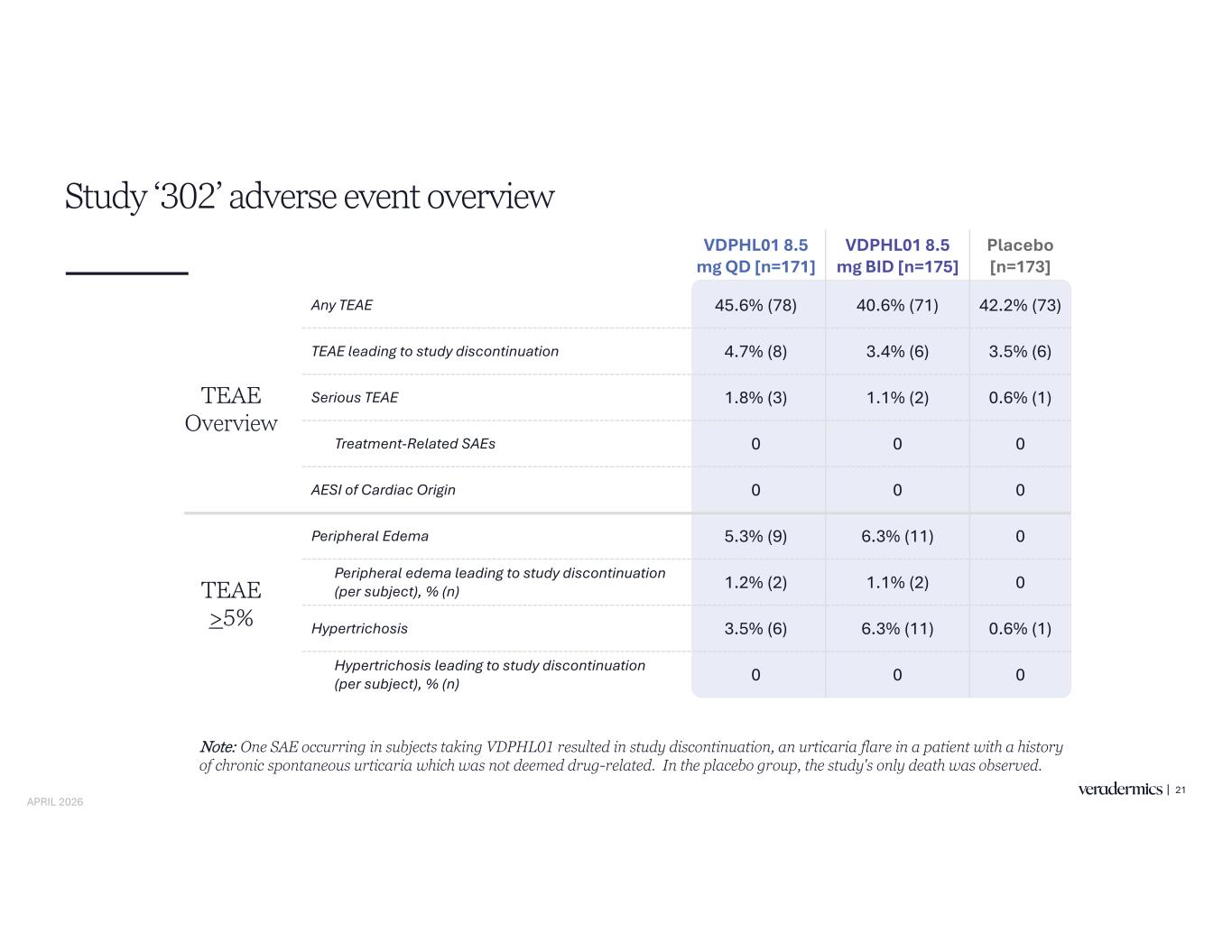
VDPHL01 8.5 mg QD [n=171] VDPHL01 8.5 mg BID [n=175] Placebo [n=173] Any TEAE 45.6% (78) 40.6% (71) 42.2% (73) TEAE leading to study discontinuation 4.7% (8) 3.4% (6) 3.5% (6) Serious TEAE 1.8% (3) 1.1% (2) 0.6% (1) Treatment-Related SAEs 0 0 0 AESI of Cardiac Origin 0 0 0 Peripheral Edema 5.3% (9) 6.3% (11) 0 Peripheral edema leading to study discontinuation 1.2% (2) 1.1% (2) 0 Hypertrichosis 3.5% (6) 6.3% (11) 0.6% (1) Hypertrichosis leading to study discontinuation 0 0 0 TEAE Overview TEAE >5% Study ‘302’ adverse event overview 21 Note: One SAE occurring in subjects taking VDPHL01 resulted in study discontinuation, an urticaria flare in a patient with a history of chronic spontaneous urticaria which was not deemed drug-related. In the placebo group, the study's only death was observed. APRIL 2026
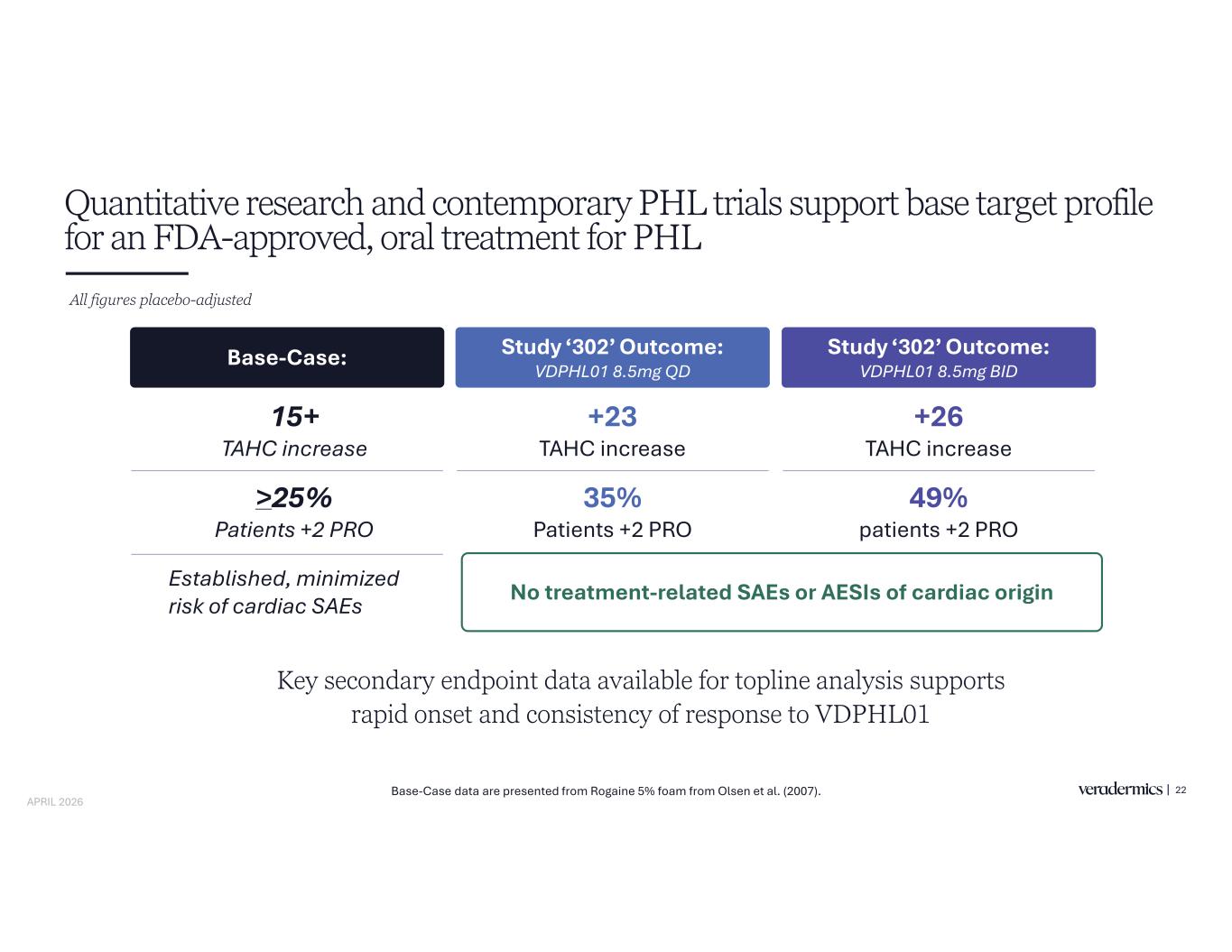
Quantitative research and contemporary PHL trials support base target profile for an FDA-approved, oral treatment for PHL All figures placebo-adjusted Base-Case: Study ‘302’ Outcome: Study ‘302’ Outcome: 15+ TAHC increase >25% Patients +2 PRO No treatment-related SAEs or AESIs of cardiac origin +23 TAHC increase 35% Patients +2 PRO +26 TAHC increase 49% patients +2 PRO Established, minimized risk of cardiac SAEs Key secondary endpoint data available for topline analysis supports rapid onset and consistency of response to VDPHL01 22Base-Case data are presented from Rogaine 5% foam from Olsen et al. (2007). APRIL 2026

23 VDPHL01 profile establishes a potential new bar for differentiation across multiple key product characteristics Fast Consistent Intense Generally Well-Tolerated Convenient Oral Administration 79.3% - 86.0% 48.4% - 62.9% Average non-vellus hair count change of 30.3 - 33.0 hairs/cm2 (QD/BID) No treatment-related SAEs; no AESIs of cardiac origin; AE-related discontinuation rates favorable vs. existing oral PHL therapies Favorable vs. topical alternatives1 Superiority vs. placebo on TAHC and IGA from Month 2 onwards 1Supported by third-party research Potential for the first oral PHL approval in males in the U.S. ~30 years APRIL 2026

KOL Discussion Maryanne Makredes Senna, M.D. Beth Israel Lahey Health, Harvard Medical School 24 APRIL 2026

VDPHL01 Phase 2/3 Clinical Trial Topline: Results from Market Opinion Study April 2026 Presented by: Mark Neumann, Chief Commercial and Strategy Officer
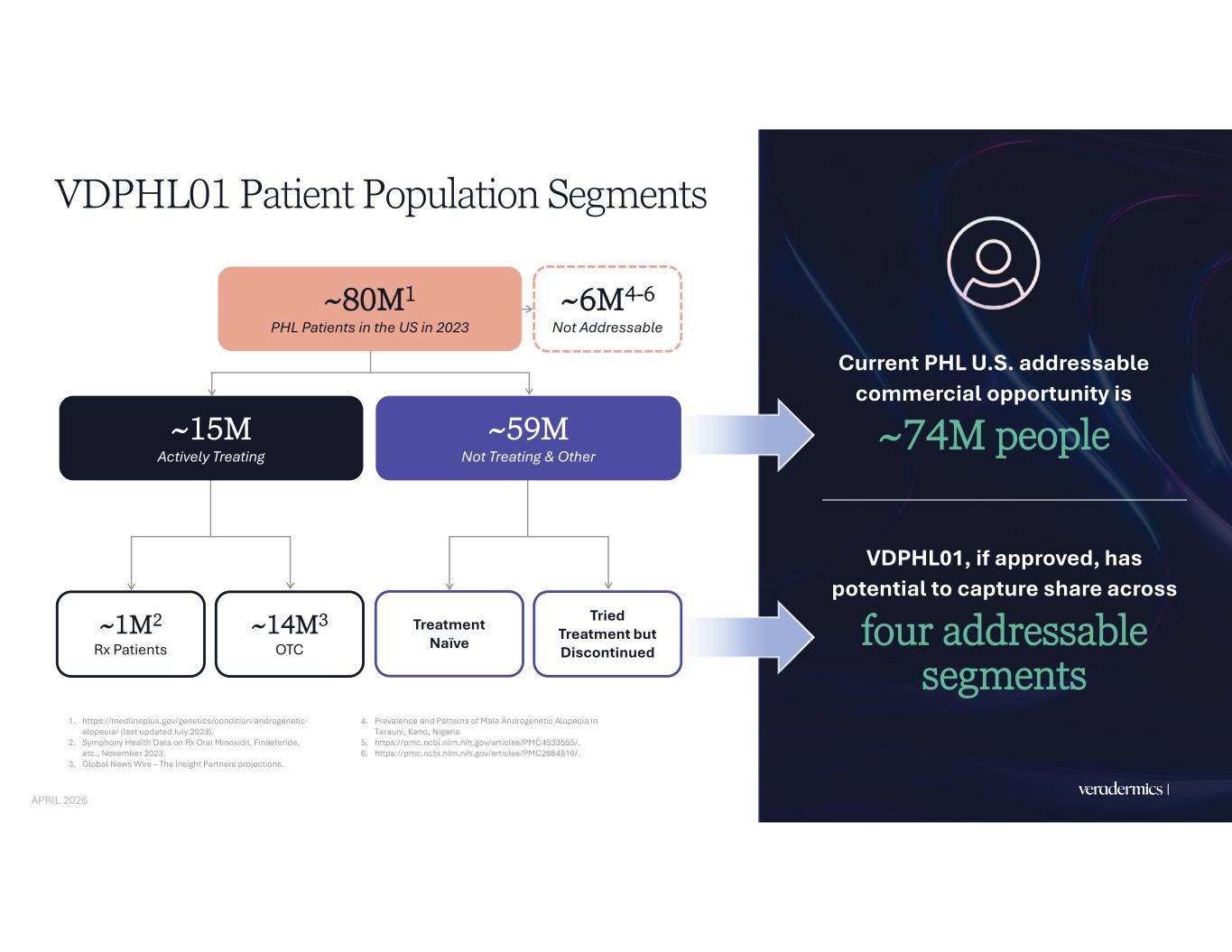
Veradermics | Internal Use Only – Proprietary and Confidential VDPHL01 Patient Population Segments Current PHL U.S. addressable commercial opportunity is ~74M people VDPHL01, if approved, has potential to capture share across four addressable segments 26 1. https://medlineplus.gov/genetics/condition/androgenetic- alopecia/ (last updated July 2023). 2. Symphony Health Data on Rx Oral Minoxidil, Finasteride, etc., November 2023. 3. Global News Wire – The Insight Partners projections. 4. Prevalence and Patterns of Male Androgenetic Alopecia in Tarauni, Kano, Nigeria 5. https://pmc.ncbi.nlm.nih.gov/articles/PMC4533555/. 6. https://pmc.ncbi.nlm.nih.gov/articles/PMC2684510/. ~80M1 PHL Patients in the US in 2023 ~15M Actively Treating ~59M Not Treating & Other ~1M2 Rx Patients ~14M3 OTC Treatment Naïve Tried Treatment but Discontinued ~6M4-6 Not Addressable APRIL 2026
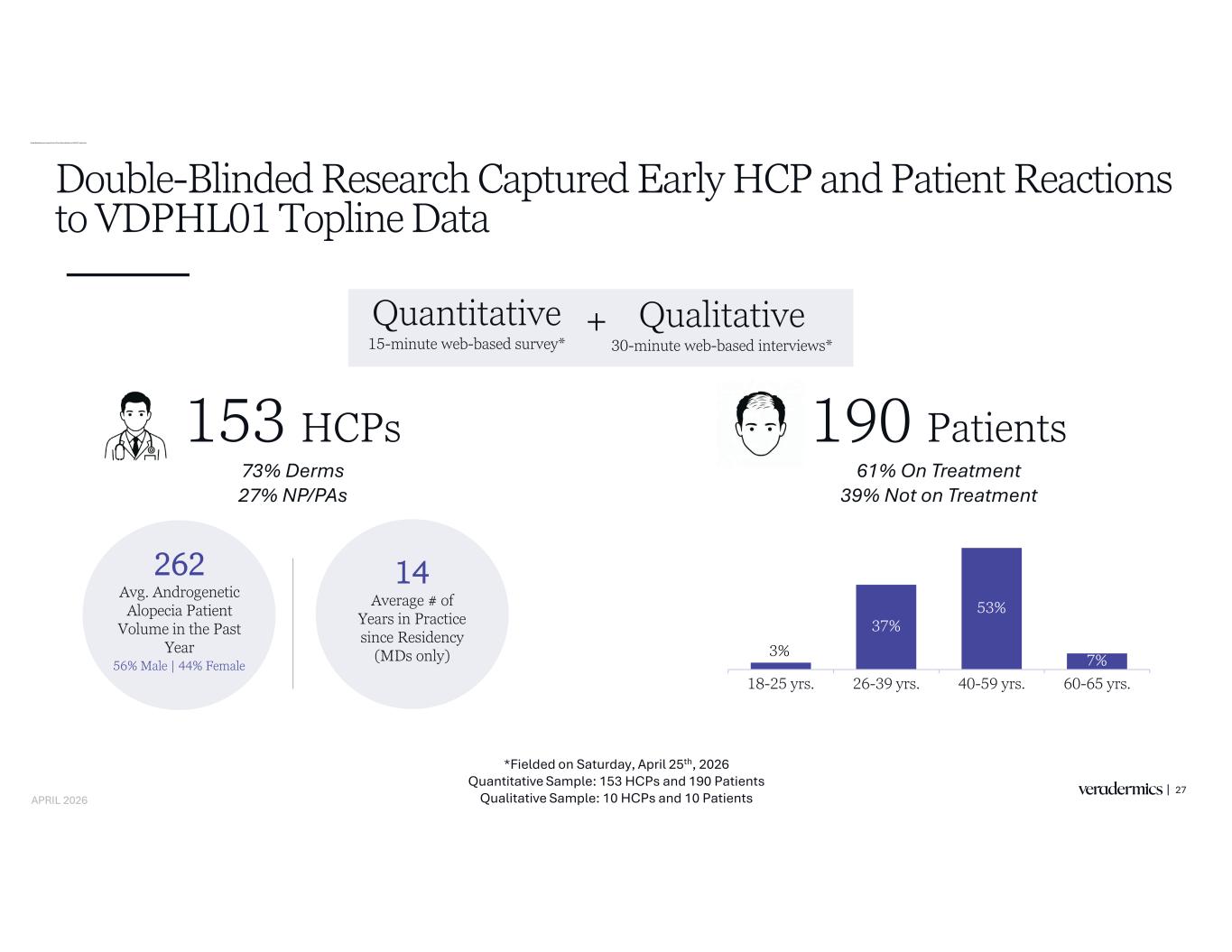
Double-Blinded Research Captured Early HCP and Patient Reactions to VDPHL01 Topline Data Double-Blinded Research Captured Early HCP and Patient Reactions to VDPHL01 Topline Data 27 Quantitative 15-minute web-based survey* 262 Avg. Androgenetic Alopecia Patient Volume in the Past Year 3% 37% 53% 7% 18-25 yrs. 26-39 yrs. 40-59 yrs. 60-65 yrs. 153 HCPs 190 Patients 14 Average # of Years in Practice since Residency (MDs only) *Fielded on Saturday, April 25th, 2026 Quantitative Sample: 153 HCPs and 190 Patients Qualitative Sample: 10 HCPs and 10 Patients Qualitative 30-minute web-based interviews* + 56% Male | 44% Female APRIL 2026
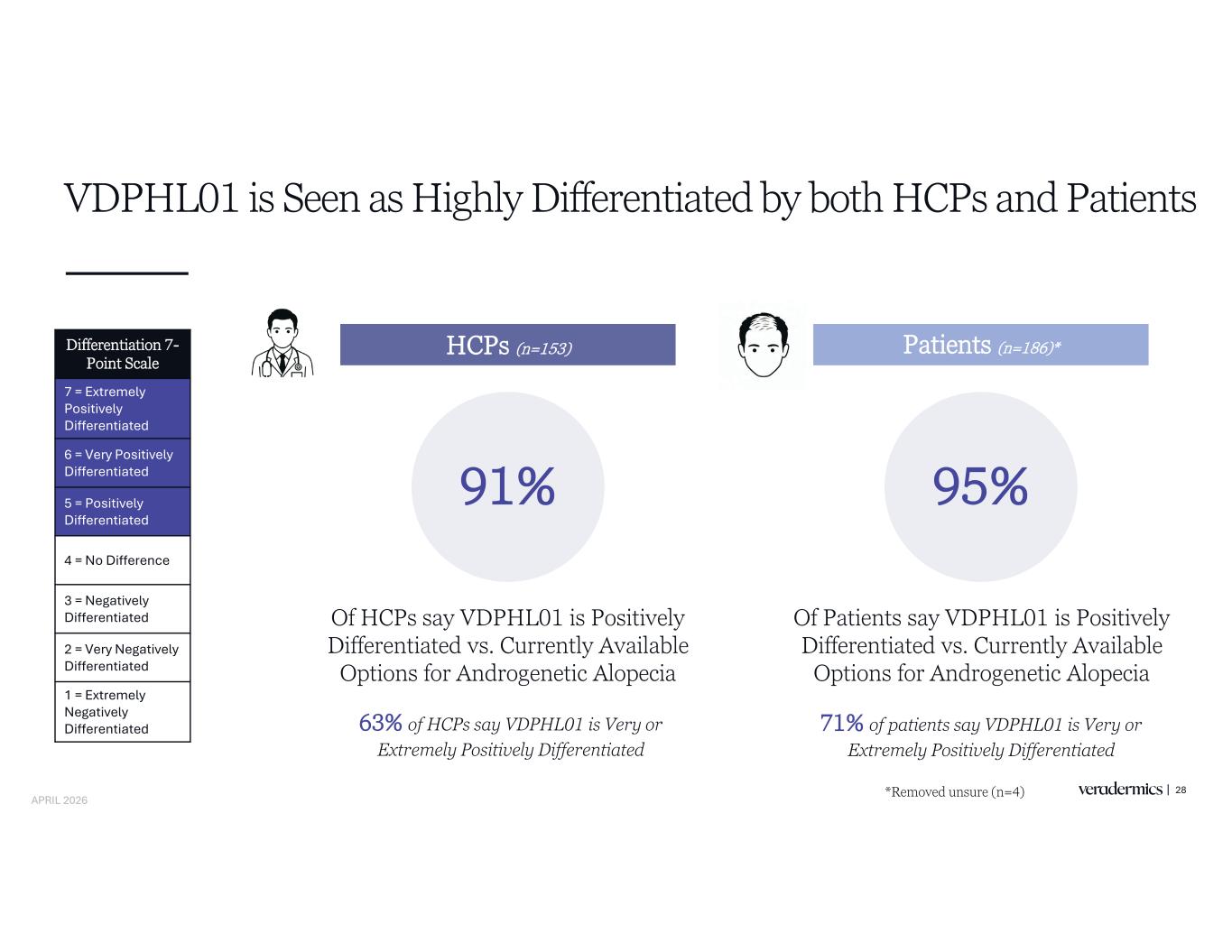
VDPHL01 is Seen as Highly Differentiated by both HCPs and Patients 28 HCPs (n=153) Patients (n=186)* 91% Differentiation 7- Point Scale 7 = Extremely Positively Differentiated 6 = Very Positively Differentiated 5 = Positively Differentiated 4 = No Difference 3 = Negatively Differentiated 2 = Very Negatively Differentiated 1 = Extremely Negatively Differentiated Of HCPs say VDPHL01 is Positively Differentiated vs. Currently Available Options for Androgenetic Alopecia 95% Of Patients say VDPHL01 is Positively Differentiated vs. Currently Available Options for Androgenetic Alopecia 63% of HCPs say VDPHL01 is Very or Extremely Positively Differentiated 71% of patients say VDPHL01 is Very or Extremely Positively Differentiated *Removed unsure (n=4) APRIL 2026
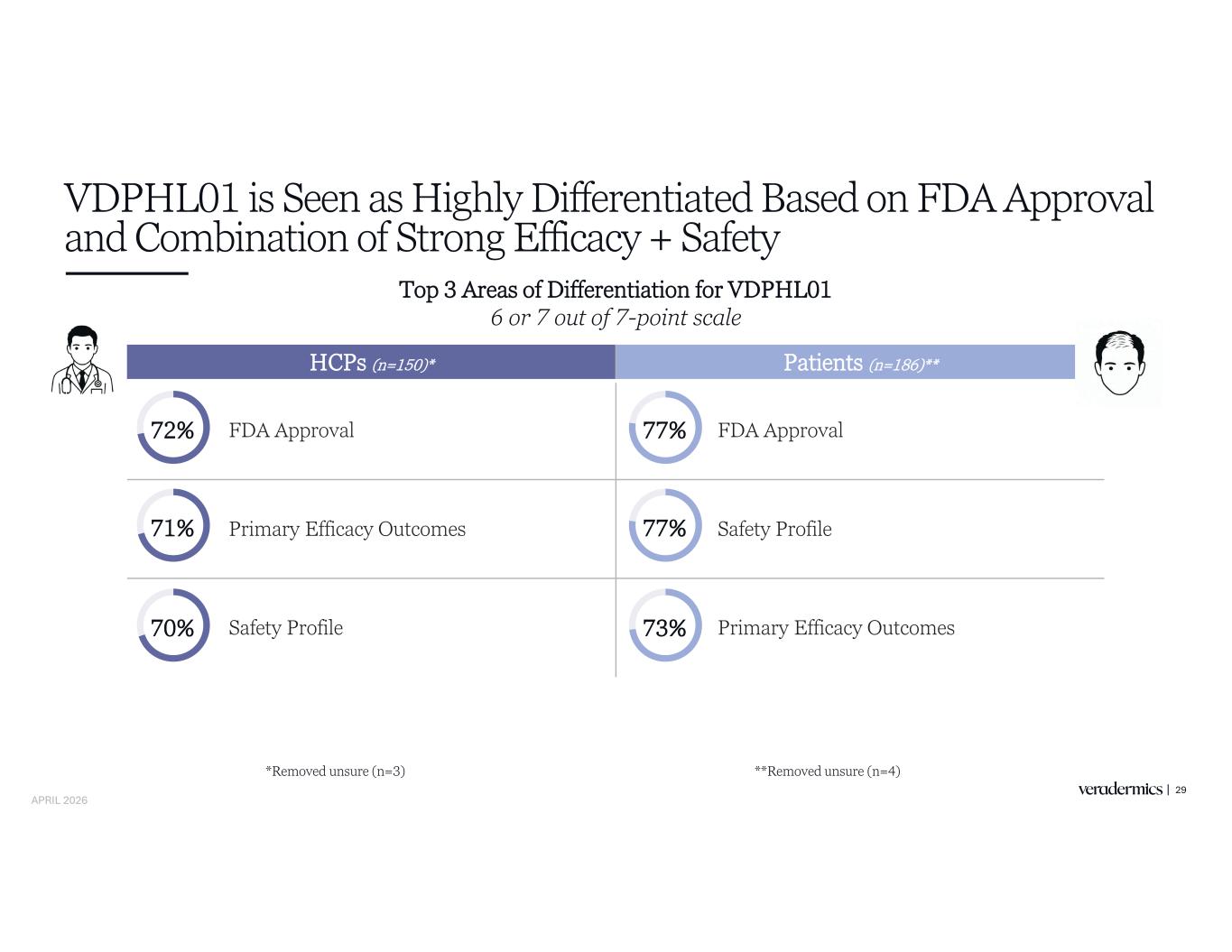
VDPHL01 is Seen as Highly Differentiated Based on FDA Approval and Combination of Strong Efficacy + Safety 29 Top 3 Areas of Differentiation for VDPHL01 6 or 7 out of 7-point scale HCPs (n=150)* Patients (n=186)** FDA Approval FDA Approval Primary Efficacy Outcomes Safety Profile Safety Profile Primary Efficacy Outcomes 72% 71% 70% 77% 77% 73% **Removed unsure (n=4)*Removed unsure (n=3) APRIL 2026
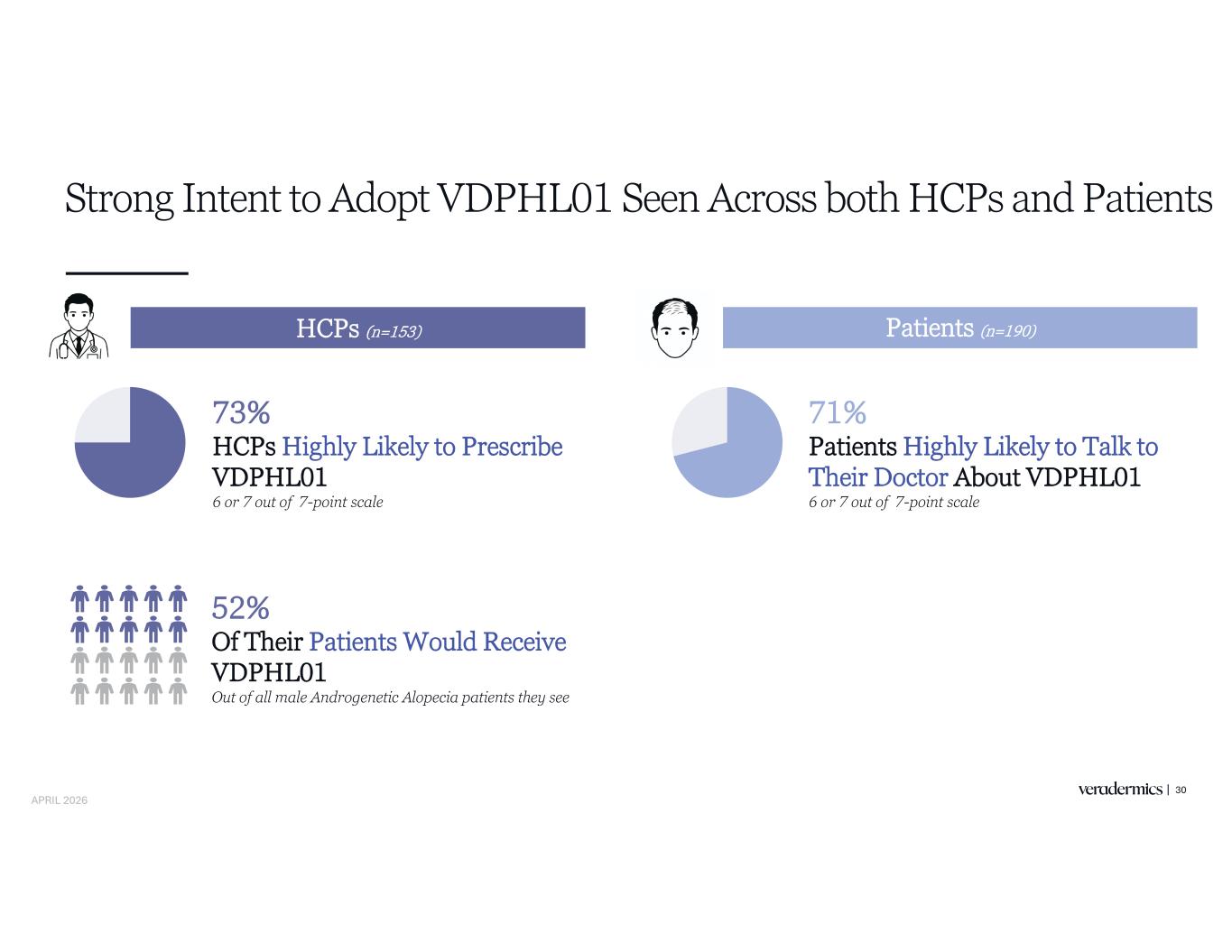
Strong Intent to Adopt VDPHL01 Seen Across both HCPs and Patients 30 HCPs (n=153) Patients (n=190) 71% Patients Highly Likely to Talk to Their Doctor About VDPHL01 6 or 7 out of 7-point scale 73% HCPs Highly Likely to Prescribe VDPHL01 6 or 7 out of 7-point scale 52% Of Their Patients Would Receive VDPHL01 Out of all male Androgenetic Alopecia patients they see APRIL 2026
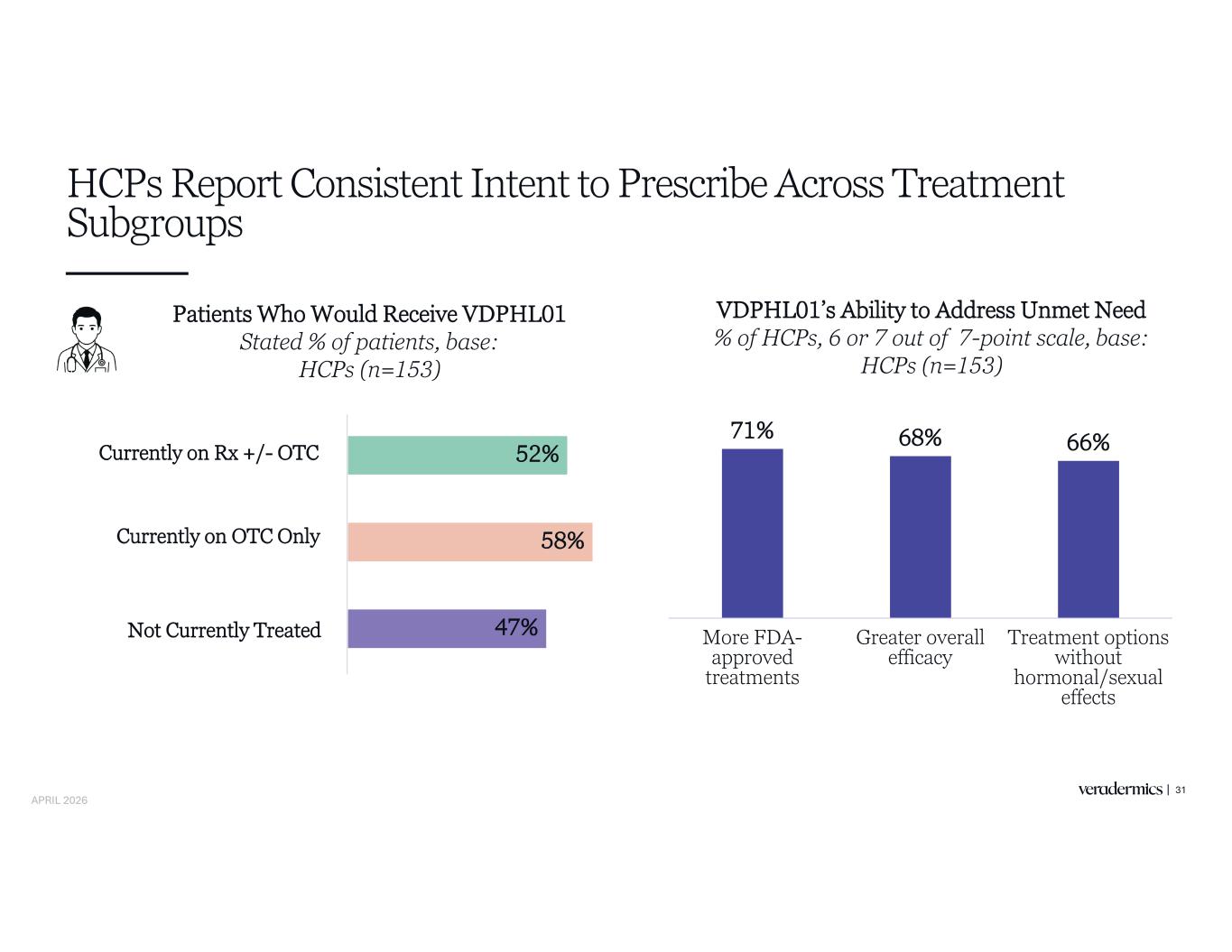
HCPs Report Consistent Intent to Prescribe Across Treatment Subgroups 31 Patients Who Would Receive VDPHL01 Stated % of patients, base: HCPs (n=153) 52% 58% 47% VDPHL01’s Ability to Address Unmet Need % of HCPs, 6 or 7 out of 7-point scale, base: HCPs (n=153) 71% 68% 66% More FDA- approved treatments Greater overall efficacy Treatment options without hormonal/sexual effects Currently on Rx +/- OTC Currently on OTC Only Not Currently Treated APRIL 2026
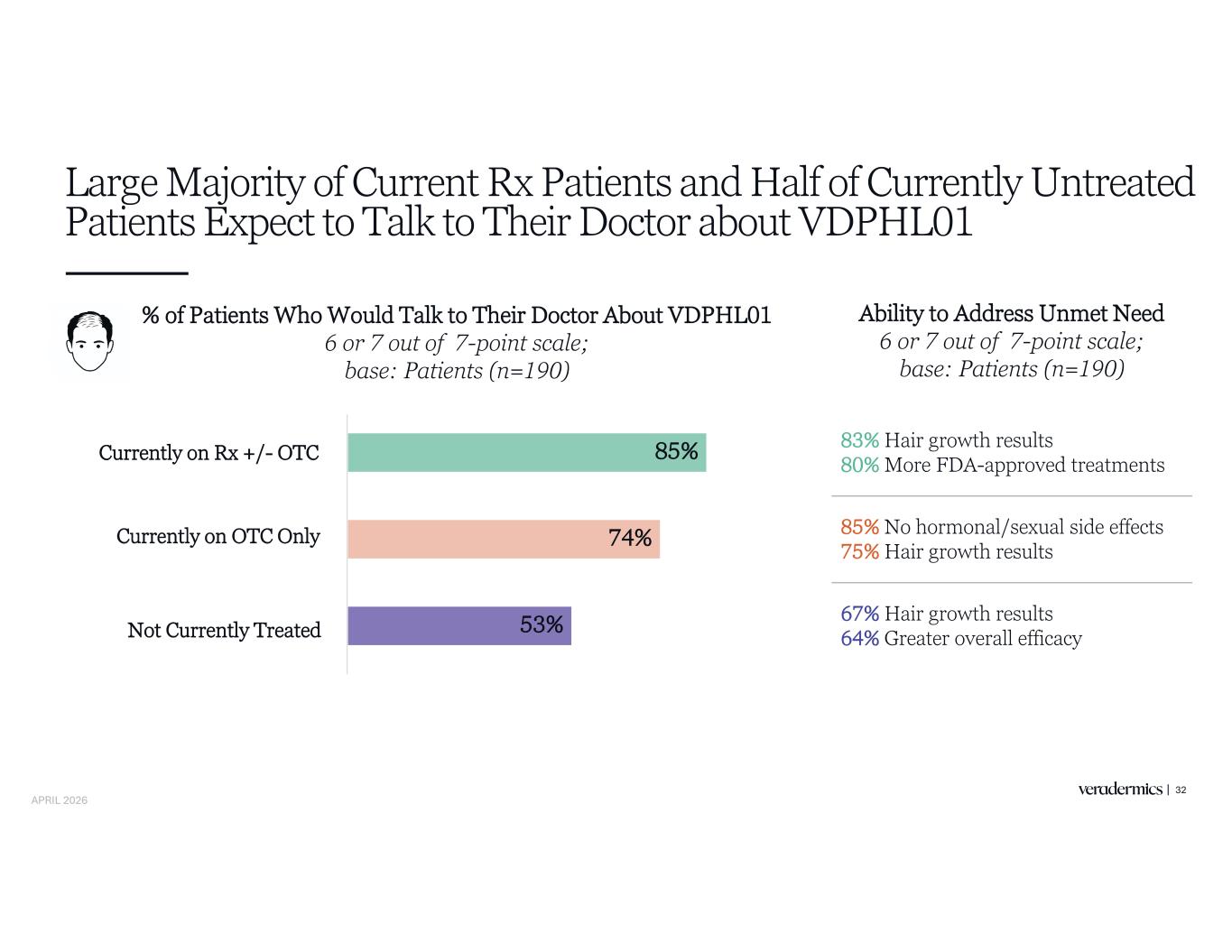
Large Majority of Current Rx Patients and Half of Currently Untreated Patients Expect to Talk to Their Doctor about VDPHL01 32 % of Patients Who Would Talk to Their Doctor About VDPHL01 6 or 7 out of 7-point scale; base: Patients (n=190) Ability to Address Unmet Need 6 or 7 out of 7-point scale; base: Patients (n=190) 83% Hair growth results 80% More FDA-approved treatments 85% No hormonal/sexual side effects 75% Hair growth results 67% Hair growth results 64% Greater overall efficacy Currently on Rx +/- OTC Currently on OTC Only Not Currently Treated 53% 74% 85% APRIL 2026
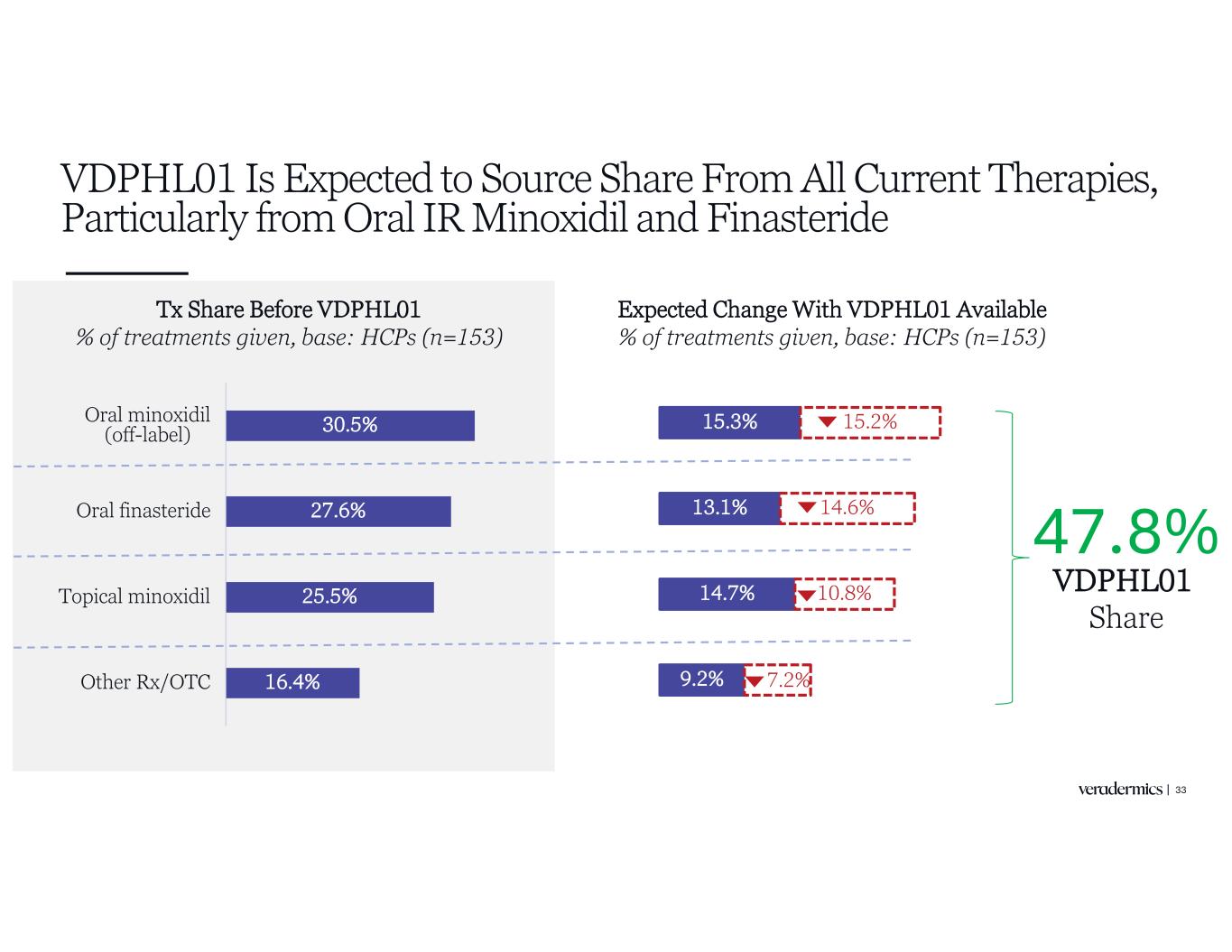
15.3% 13.1% 14.7% 9.2% 15.2% 14.6% 10.8% 7.2% VDPHL01 Is Expected to Source Share From All Current Therapies, Particularly from Oral IR Minoxidil and Finasteride 33 Tx Share Before VDPHL01 % of treatments given, base: HCPs (n=153) 30.5% 27.6% 25.5% 16.4% Oral minoxidil (off-label) Oral finasteride Topical minoxidil Other Rx/OTC Expected Change With VDPHL01 Available % of treatments given, base: HCPs (n=153) VDPHL01 Share 47.8%

Patient/Physician Voice from Interviews Highlight the Unique Opportunity for VDPHL01 34 Impressive Efficacy Patient “This is like superpowered minoxidil, maybe it's even to the 3rd degree because there's already topical oral minoxidil and now this one. I would say it's like a super powered Minoxidil.” - Oral Minoxidil / Oral Dutasteride Patient Patient Dermatologist Dermatologist Differentiated Safety Differentiated Efficacy Extended Release / Mechanism “The big reason I don't use Rx treatments is avoiding scary side effects, which is why I've mostly gone the topical route… [Product X side effects] seem mild compared to finasteride, which seemed pretty scary and are the reason I haven't tried it.” –Current OTC user “This is a better version of the current oral minoxidil that is more effective as monotherapy while also maintaining or even reducing some of the important side effects... I would describe it as a game changer.” – Community Dermatologist “I really like the extended release because usually that means it's better tolerated. The efficacy is better as with a lot of other conditions...” – Community Dermatologist

Closing Remarks 35 APRIL 2026

36 VDPHL01 profile establishes a potential new bar for differentiation across multiple key product characteristics Fast Consistent Intense Generally Well-Tolerated Convenient Oral Administration 79.3% - 86.0% 48.4% - 62.9% Average non-vellus hair count change of 30.3 - 33.0 hairs/cm2 (QD/BID) No treatment-related SAEs; no AESIs of cardiac origin; AE-related discontinuation rates favorable vs. existing oral PHL therapies Favorable vs. topical alternatives1 Superiority vs. placebo on TAHC and IGA from Month 2 onwards 1Supported by third-party research Potential for the first oral PHL approval in males in the U.S. ~30 years APRIL 2026

“So definitely on the top of my head, my hair has gotten a lot thicker. It is covering a lot more. And like, I have noticed it, but others have noticed it as well. I'm – I'm getting compliments about my hair.” It's thicker. You can't see my hair thinness as easily as you used to.… Because I used to be able to – if I'm standing for a mirror in the sunlight, you can see right through it, where now it's – it looks a lot thicker.” “The bald spot or bald area has decreased in size…. And the rest of my hair, especially the front…seems to be fuller and thicker.” “I would say just not thinking about it quite as much. So like I definitely have like gotten out of the shower, dried my hair, and then just like left the house, like went to the store, went to wherever and just decided not to care about it because I think I was like, Okay, like it's better than it was six months ago” …I definitely…wear [a hat] more for…casual comfort nature…versus… the need to wear it… because I enjoy wearing hats now versus the need to wear a hat” “…it’s very comforting that I don’t have to worry about [hair coverage], and [do] less prep [to my hair] before I leave the house” 37 Study ‘302’ patient quotes: Quoted patients are treatment group-blinded while the extension phase of Study ‘302’ is ongoing, so quotes cannot be linked to a particular treatment group at this time. Individual results may vary. APRIL 2026

Meaningful Study ‘302’ results position VDPHL01 as a potential foundational treatment for PHL Upcoming Milestones Veradermics anticipates: • Male confirmatory Phase 3 data (Study ‘304’) in the second half of 2026; • Study 302 Part B data in the second half of 2026 • Additional Study ‘207’ data in 2026 38 Phase 2/3 topline data support VDPHL01 as a potential best-in-class therapy for PHL Results delivered robust hair growth to patients who have grown accustomed to limitations when seeking to treat pattern hair loss Speed and consistency of effect further differentiate profile from current treatment options characterized by slow onset and varied outcomes APRIL 2026
