
Exhibit 99.2

0 April 2026 Corporate Presentation NASDAQ: ANL

1 Disclaimer This presentation has been prepared by Adlai Nortye Ltd. (the "Company") for informational purposes only and does not constitute an offer to sell or the solicitation of an offer to buy any securities. This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts, including statements regarding our business strategy, product candidates, preclinical and clinical development plans for each of our product candidates including AN9025, AN4035 and AN8025, anticipated milestones, potential therapeutic benefits, market opportunities, and other future events or prospects, are forward- looking statements. These statements may be identified by words such as "may," "will," "expect," "anticipate," "believe," "plan," "potential," "estimate," or similar expressions. Forward-looking statements are based on current expectations and assumptions and are subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statements. These risks include, but are not limited to, risks related to the discovery and development of our product candidates, the timing, progress, and likelihood of success of our preclinical studies and clinical trials, regulatory approval processes, reliance on third parties for manufacturing, research and development activities, intellectual property protection, competitive developments, our ability to obtain additional financing. Our product candidates are in preclinical or at various stages of clinical development. Our assumptions regarding the development potential of these product candidates are based in large part on preclinical data or early clinical readouts, and such data may not be predictive of future clinical outcomes. We may observe materially different results as we conduct further preclinical studies and any planned or ongoing clinical trials. Preclinical findings, including pharmacology, target engagement, biomarkers, efficacy, tolerability, safety, and translational data, may not be replicated in humans. Our development activities are subject to significant risks and uncertainties, including potential delays in the initiation, enrollment, data readout, and completion of preclinical studies and clinical trials, and the possibility of unexpected adverse events or insufficient efficacy, which may delay or prevent regulatory approval or commercialization. In addition, comparisons of product candidates between the Company and third parties based on preclinical and clinical data, are inherently limited due to differences in methodologies and models, experimental conditions, study design and patient populations, such comparisons should be interpreted with caution. This presentation concerns product candidates that are under preclinical or clinical investigation and have not been approved for marketing by the U.S. Food and Drug Administration (FDA) or any other regulatory authority. These product candidates are currently limited by federal law to investigational use, and no representation is made as to their safety or effectiveness for the purposes for which they are being investigated. Additional risks and uncertainties are described in the Company's prior filings with the U.S. Securities and Exchange Commission(SEC), including under the heading "Risk Factors" in the Company's annual report on Form 20-F for the year ended December 31, 2025, and any subsequent filings with the SEC. This presentation speaks only as of its date, the Company undertakes no obligation to update any forward-looking statements or other information contained herein, except as required by law. All copyrights and trademarks used herein are the property of their respective owners.

2 Product MOA Indication Discovery IND Enabling Phase 1a Phase 1b Phase 2 Phase 3 Upcoming Milestone Worldwide Rights AN9025 pan- RAS(ON)i RAS-addicted tumors Ph1 dose escalation completion in H1 2027 Ex-China1 AN4035 CEACAM5 pan- RAS(ON)i ADC CEACAM5- enriched RAS- addicted solid tumors IND submission in mid-2026 Global AN8025 αPD-L1 /Lag3v /CD86v Advanced tumors Ph1 dose escalation completion in 2026 YE Global Other Assets AN4005 Small molecule PD-L1i Advanced tumors Clinical update presentation at conference expected in H2 2026 Ex-China2 Palupiprant (AN0025) EP4 Neoadj RC (+CRT)3 Topline results of Ph2 study in H1 2027 Global except Japan, Korea, Taiwan and Southeast Asian Countries LA EC (+CRT)3 Clinical update in H2 2026 Pipeline Chart: Abbreviations: MOA = mechanism of action; Lag3v: Lag 3 variant; CD86v: CD86 variant; IND = Investigational New Drug; RC = rectal cancer; CRT = chemoradiation therapy; LA = locally advanced; EC = esophageal cancer Note 1. ASK Pharm holds exclusive rights to develop, manufacture, and commercialize AN9025 in mainland China, Hong Kong and Macao; 2. Xiamen Biotime Biotechnology Co., Ltd., holds exclusive rights to develop, manufacture, and commercialize AN4005 in mainland China, Hong Kong, Macao and Taiwan; 3. Investigator Initiated Trials

3 AN9025: An Oral Small-Molecule Pan-RAS(ON) Inhibitor
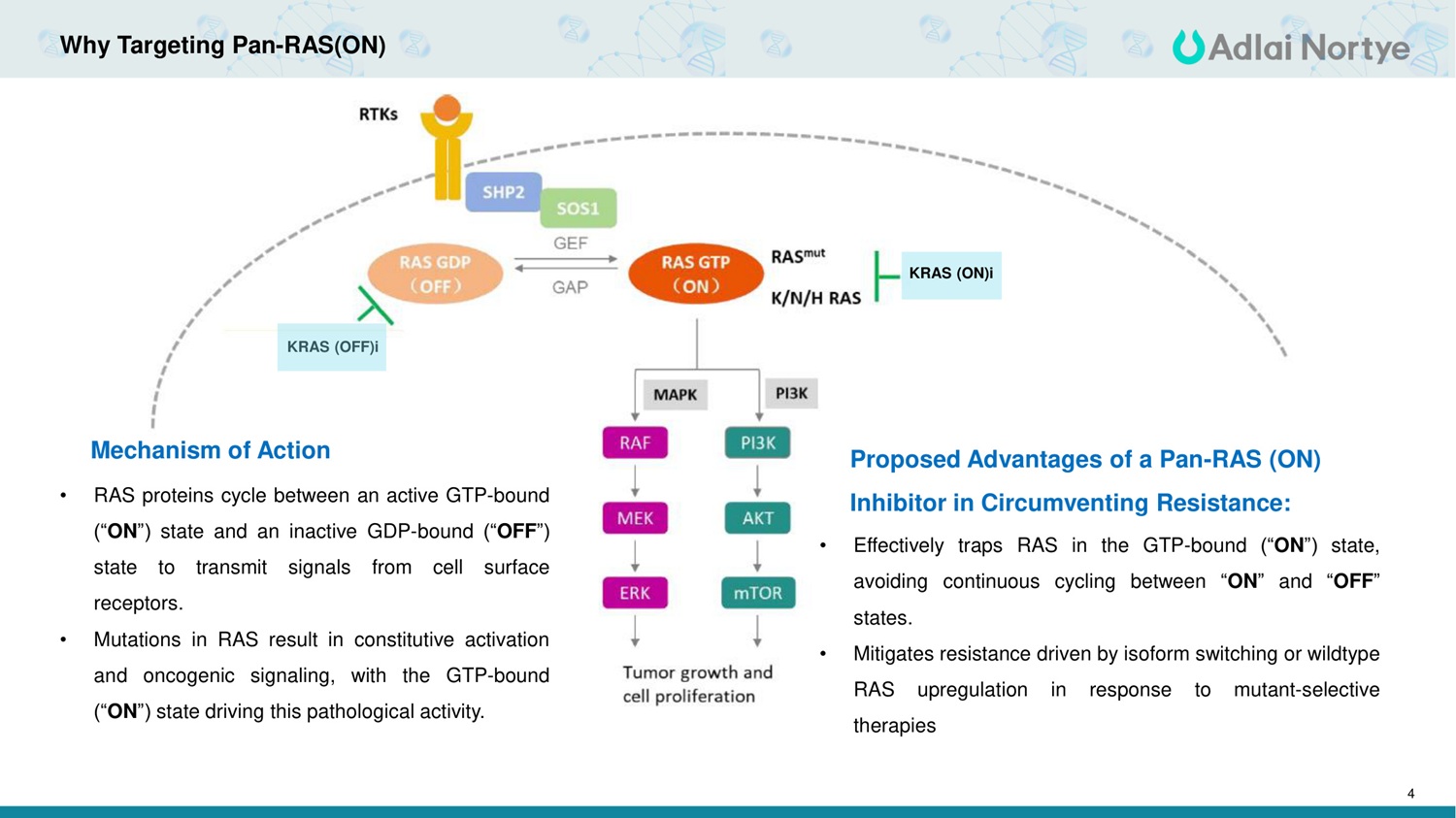
4 Why Targeting Pan-RAS(ON) KRAS (OFF)i KRAS (ON)i • RAS proteins cycle between an active GTP-bound ("ON") state and an inactive GDP-bound ("OFF") state to transmit signals from cell surface receptors. • Mutations in RAS result in constitutive activation and oncogenic signaling, with the GTP-bound ("ON") state driving this pathological activity. • Effectively traps RAS in the GTP-bound ("ON") state, avoiding continuous cycling between "ON" and "OFF" states. • Mitigates resistance driven by isoform switching or wildtype RAS upregulation in response to mutant-selective therapies Mechanism of Action Proposed Advantages of a Pan-RAS (ON) Inhibitor in Circumventing Resistance:
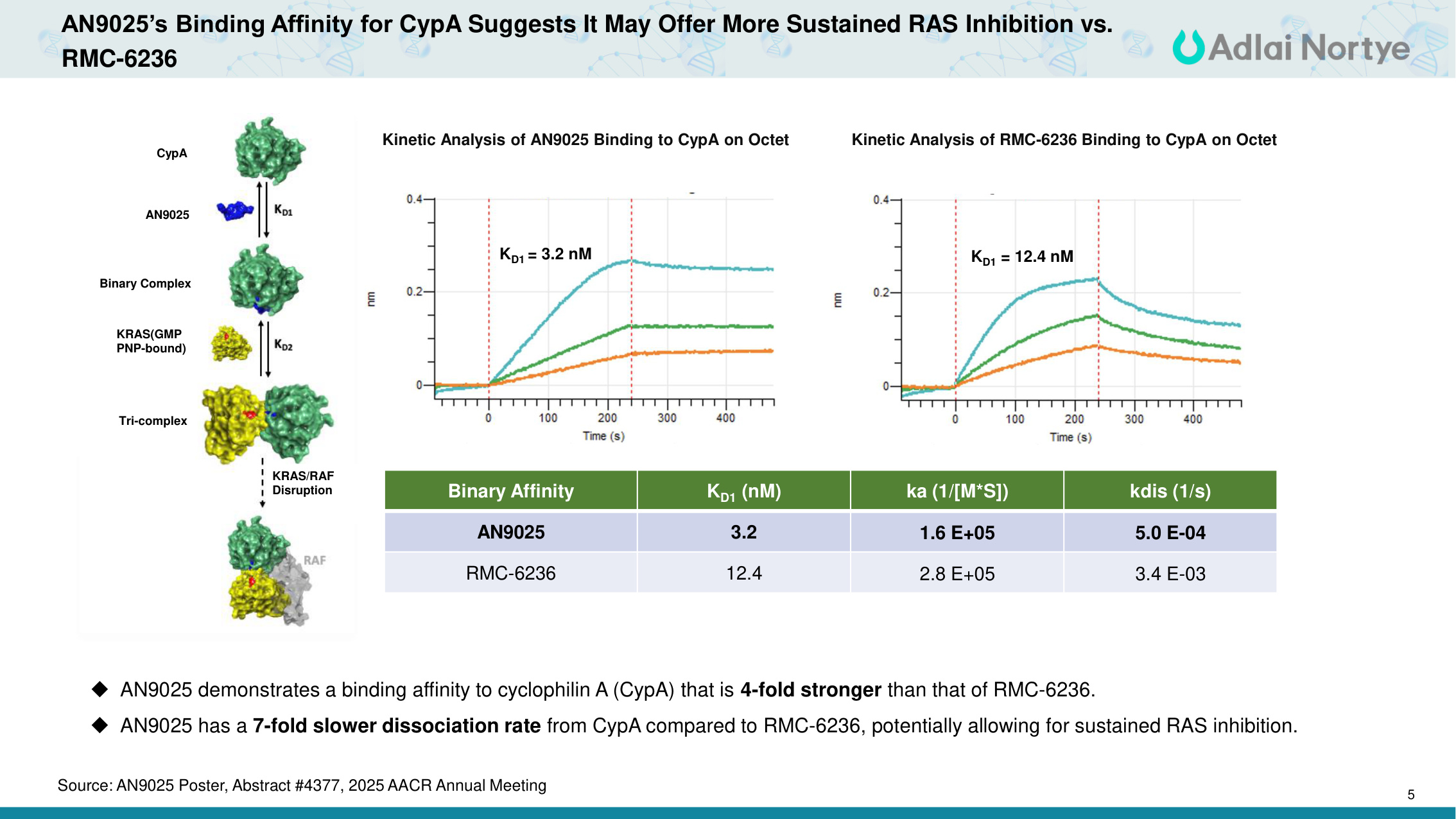
5 AN9025's Binding Affinity for CypA Suggests It May Offer More Sustained RAS Inhibition vs. RMC-6236 KD1 = 3.2 nM Kinetic Analysis of AN9025 Binding to CypA on Octet Binary Affinity KD1 (nM) ka (1/[M*S]) kdis (1/s) AN9025 3.2 1.6 E+05 5.0 E-04 RMC-6236 12.4 2.8 E+05 3.4 E-03 AN9025 demonstrates a binding affinity to cyclophilin A (CypA) that is 4-fold stronger than that of RMC-6236. AN9025 has a 7-fold slower dissociation rate from CypA compared to RMC-6236, potentially allowing for sustained RAS inhibition. KD1 = 12.4 nM Kinetic Analysis of RMC-6236 Binding to CypA on Octet CypA AN9025 Binary Complex KRAS(GMP PNP-bound) Tri-complex KRAS/RAF Disruption Source: AN9025 Poster, Abstract #4377, 2025 AACR Annual Meeting
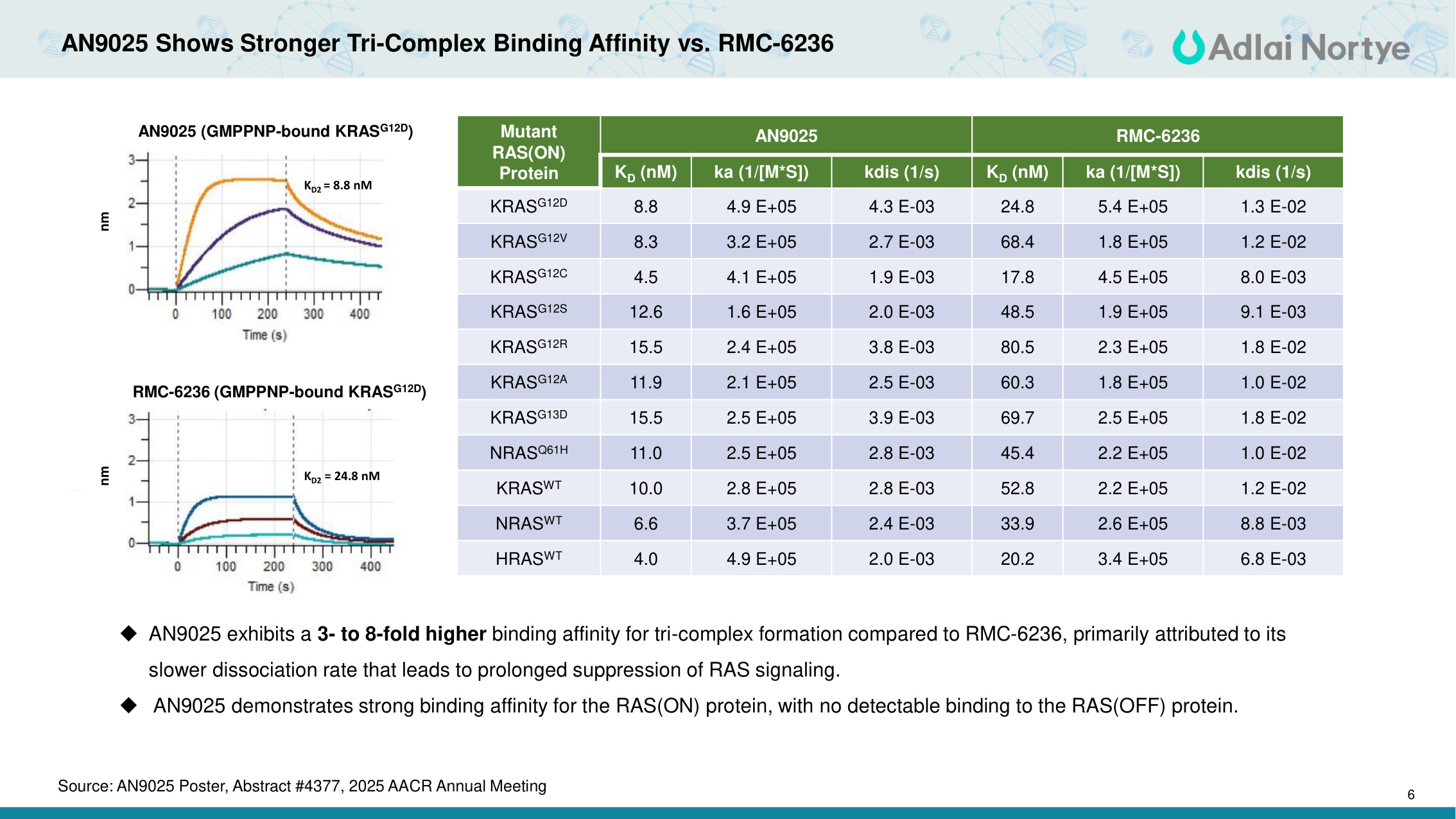
6 AN9025 Shows Stronger Tri-Complex Binding Affinity vs. RMC-6236 KD2 = 24.8 nM Mutant RAS(ON) Protein AN9025 RMC-6236 KD (nM) ka (1/[M*S]) kdis (1/s) KD (nM) ka (1/[M*S]) kdis (1/s) KRASG12D 8.8 4.9 E+05 4.3 E-03 24.8 5.4 E+05 1.3 E-02 KRASG12V 8.3 3.2 E+05 2.7 E-03 68.4 1.8 E+05 1.2 E-02 KRASG12C 4.5 4.1 E+05 1.9 E-03 17.8 4.5 E+05 8.0 E-03 KRASG12S 12.6 1.6 E+05 2.0 E-03 48.5 1.9 E+05 9.1 E-03 KRASG12R 15.5 2.4 E+05 3.8 E-03 80.5 2.3 E+05 1.8 E-02 KRASG12A 11.9 2.1 E+05 2.5 E-03 60.3 1.8 E+05 1.0 E-02 KRASG13D 15.5 2.5 E+05 3.9 E-03 69.7 2.5 E+05 1.8 E-02 NRASQ61H 11.0 2.5 E+05 2.8 E-03 45.4 2.2 E+05 1.0 E-02 KRASWT 10.0 2.8 E+05 2.8 E-03 52.8 2.2 E+05 1.2 E-02 NRASWT 6.6 3.7 E+05 2.4 E-03 33.9 2.6 E+05 8.8 E-03 HRASWT 4.0 4.9 E+05 2.0 E-03 20.2 3.4 E+05 6.8 E-03 AN9025 exhibits a 3- to 8-fold higher binding affinity for tri-complex formation compared to RMC-6236, primarily attributed to its slower dissociation rate that leads to prolonged suppression of RAS signaling. AN9025 demonstrates strong binding affinity for the RAS(ON) protein, with no detectable binding to the RAS(OFF) protein. AN9025 (GMPPNP-bound KRASG12D) KD2 = 8.8 nM RMC-6236 (GMPPNP-bound KRASG12D) nm nm Source: AN9025 Poster, Abstract #4377, 2025 AACR Annual Meeting
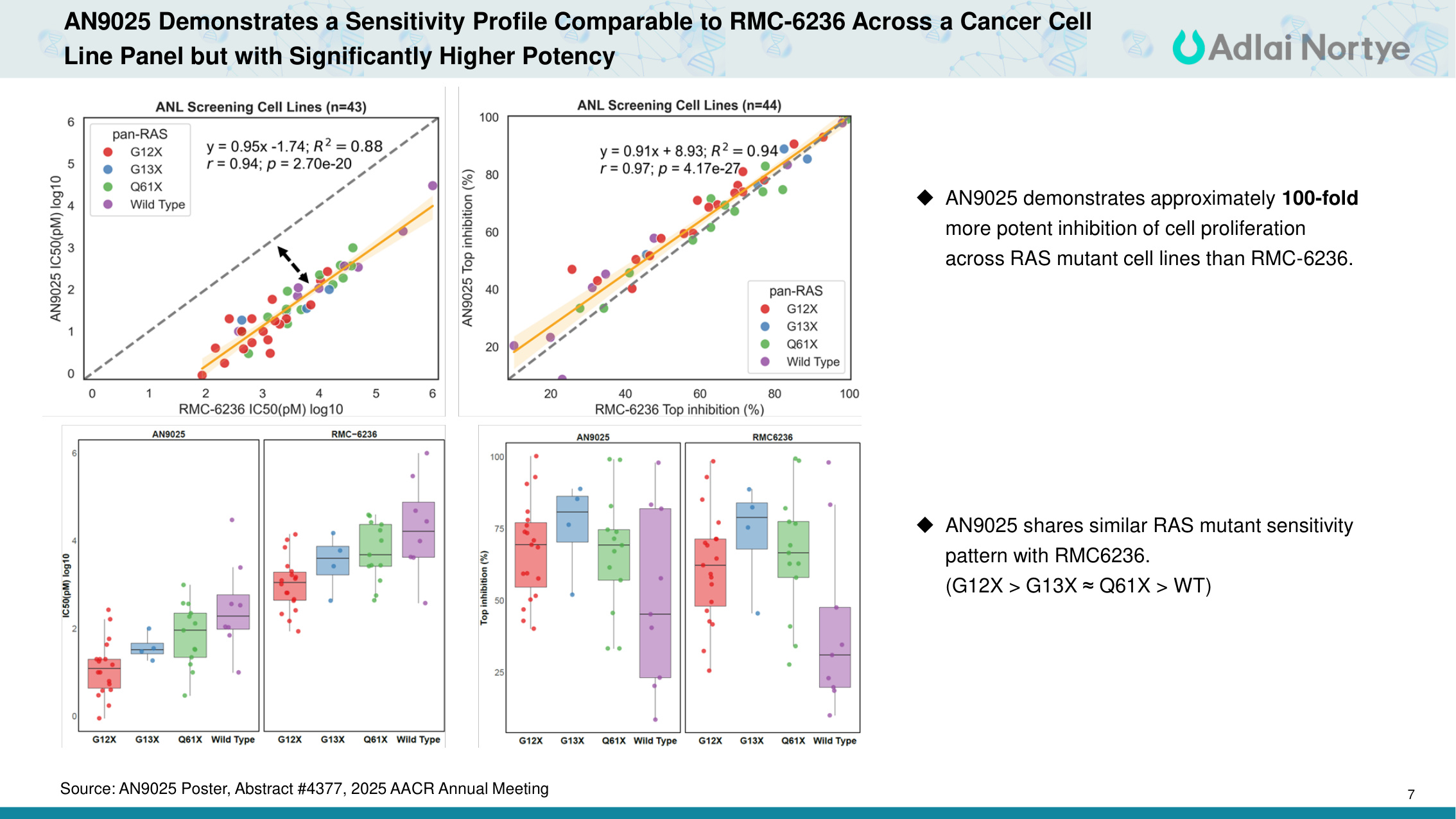
7 AN9025 Demonstrates a Sensitivity Profile Comparable to RMC-6236 Across a Cancer Cell Line Panel but with Significantly Higher Potency AN9025 demonstrates approximately 100-fold more potent inhibition of cell proliferation across RAS mutant cell lines than RMC-6236. AN9025 shares similar RAS mutant sensitivity pattern with RMC6236. (G12X > G13X ≈Q61X > WT) Source: AN9025 Poster, Abstract #4377, 2025 AACR Annual Meeting
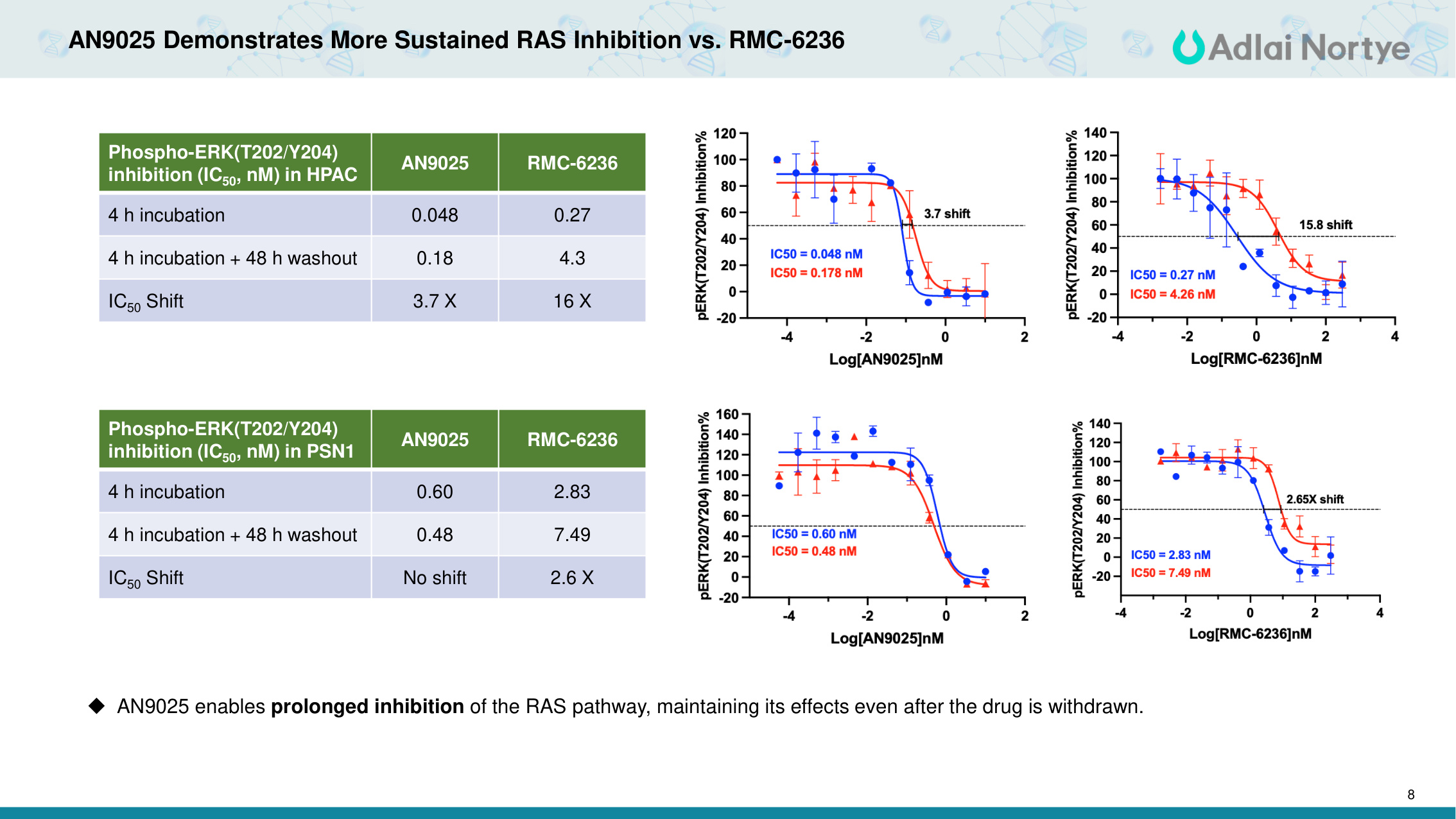
8 AN9025 Demonstrates More Sustained RAS Inhibition vs. RMC-6236 Phospho-ERK(T202/Y204) inhibition (IC50, nM) in HPAC AN9025 RMC-6236 4 h incubation 0.048 0.27 4 h incubation + 48 h washout 0.18 4.3 IC50 Shift 3.7 X 16 X AN9025 enables prolonged inhibition of the RAS pathway, maintaining its effects even after the drug is withdrawn. Phospho-ERK(T202/Y204) inhibition (IC50, nM) in PSN1 AN9025 RMC-6236 4 h incubation 0.60 2.83 4 h incubation + 48 h washout 0.48 7.49 IC50 Shift No shift 2.6 X
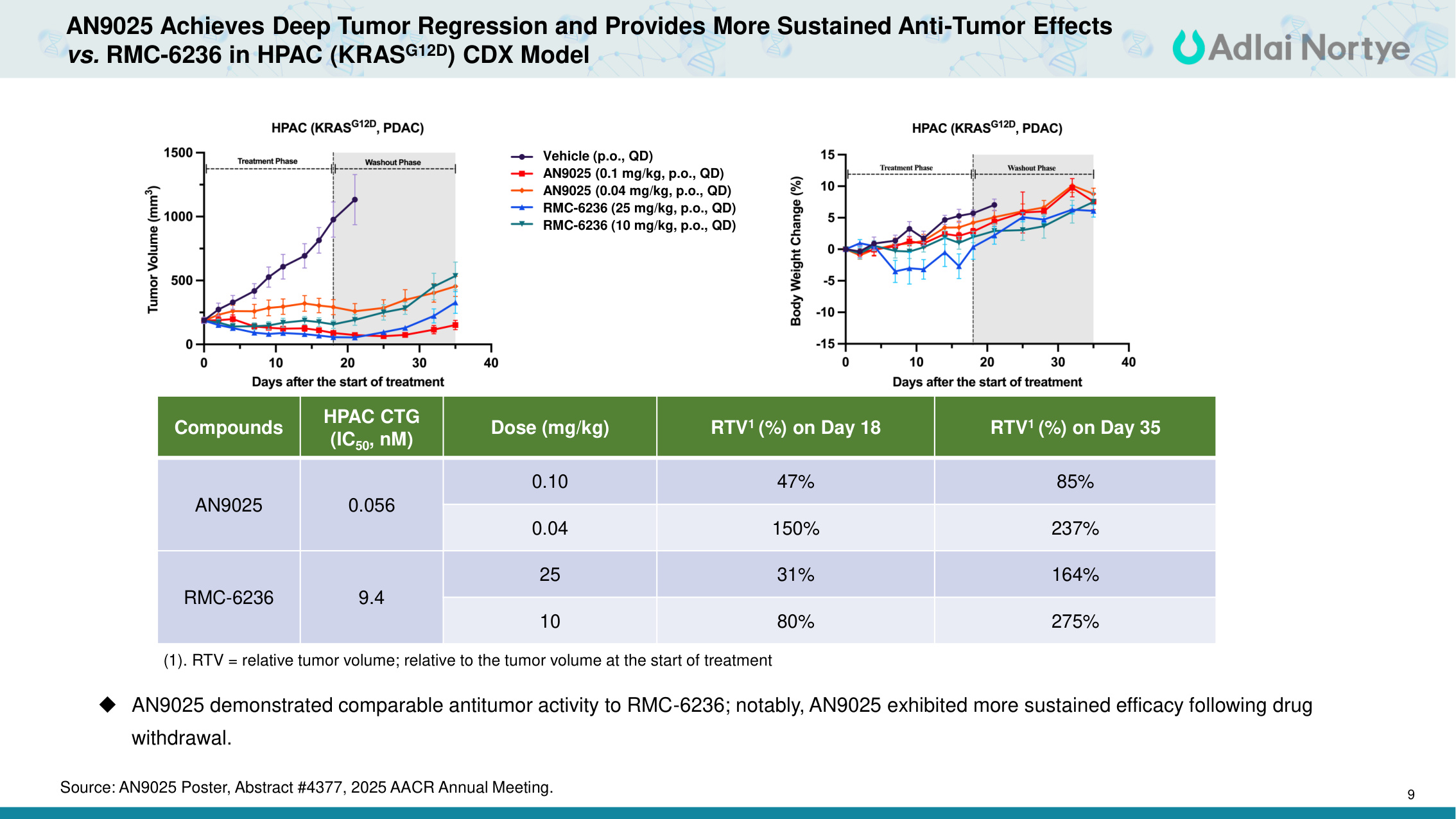
9 AN9025 Achieves Deep Tumor Regression and Provides More Sustained Anti-Tumor Effects vs. RMC-6236 in HPAC (KRASG12D) CDX Model AN9025 demonstrated comparable antitumor activity to RMC-6236; notably, AN9025 exhibited more sustained efficacy following drug withdrawal. Compounds HPAC CTG (IC50, nM) Dose (mg/kg) RTV1 (%) on Day 18 RTV1 (%) on Day 35 AN9025 0.056 0.10 47% 85% 0.04 150% 237% RMC-6236 9.4 25 31% 164% 10 80% 275% (1). RTV = relative tumor volume; relative to the tumor volume at the start of treatment Vehicle (p.o., QD) AN9025 (0.1 mg/kg, p.o., QD) AN9025 (0.04 mg/kg, p.o., QD) RMC-6236 (25 mg/kg, p.o., QD) RMC-6236 (10 mg/kg, p.o., QD) Source: AN9025 Poster, Abstract #4377, 2025 AACR Annual Meeting.
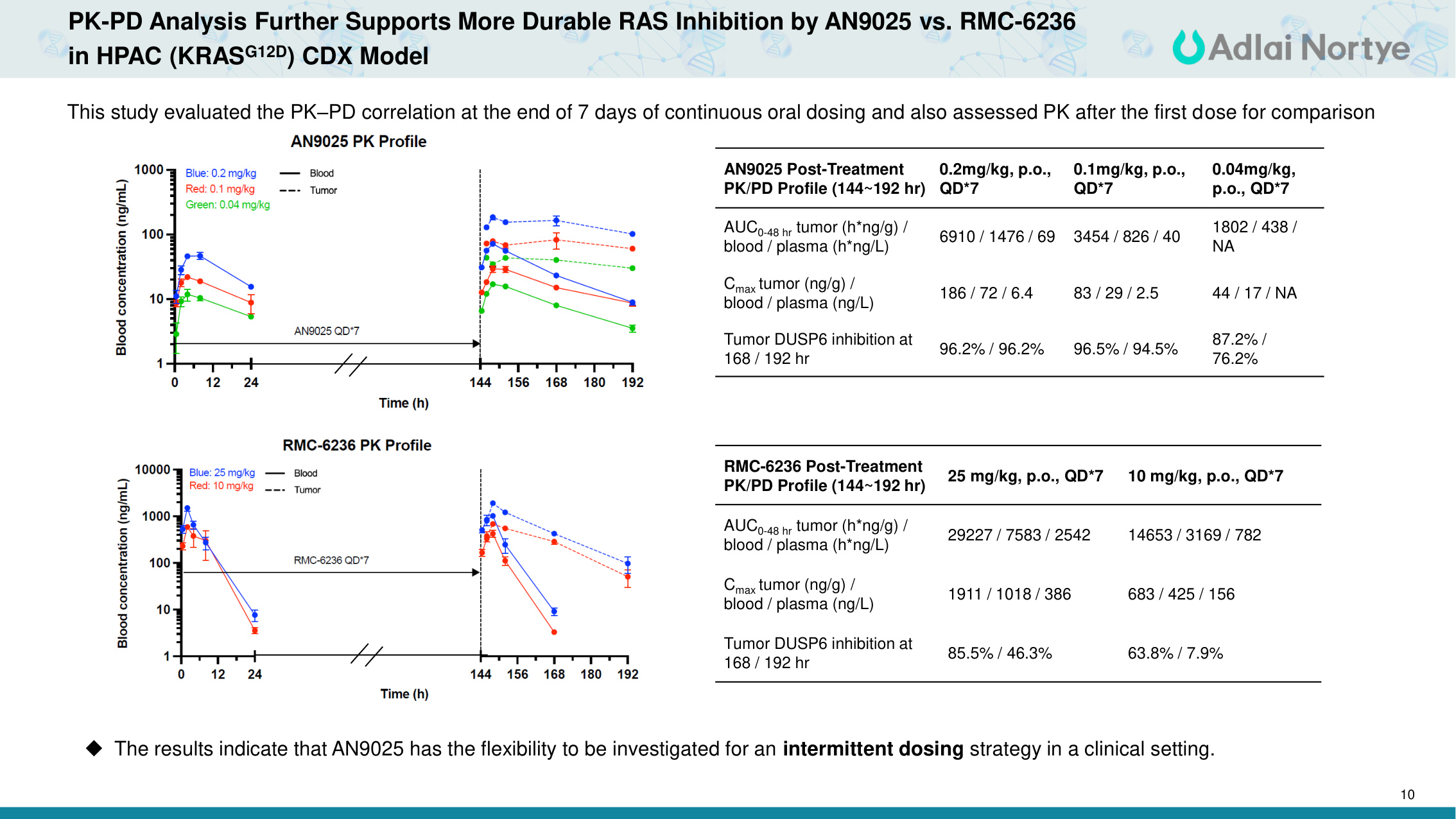
10 PK-PD Analysis Further Supports More Durable RAS Inhibition by AN9025 vs. RMC-6236 in HPAC (KRASG12D) CDX Model This study evaluated the PK–PD correlation at the end of 7 days of continuous oral dosing and also assessed PK after the first dose for comparison The results indicate that AN9025 has the flexibility to be investigated for an intermittent dosing strategy in a clinical setting. AN9025 Post-Treatment PK/PD Profile (144~192 hr) 0.2mg/kg, p.o., QD*7 0.1mg/kg, p.o., QD*7 0.04mg/kg, p.o., QD*7 AUC0-48 hr tumor (h*ng/g) / blood / plasma (h*ng/L) 6910 / 1476 / 69 3454 / 826 / 40 1802 / 438 / NA Cmax tumor (ng/g) / blood / plasma (ng/L) 186 / 72 / 6.4 83 / 29 / 2.5 44 / 17 / NA Tumor DUSP6 inhibition at 168 / 192 hr 96.2% / 96.2% 96.5% / 94.5% 87.2% / 76.2% RMC-6236 Post-Treatment PK/PD Profile (144~192 hr) 25 mg/kg, p.o., QD*7 10 mg/kg, p.o., QD*7 AUC0-48 hr tumor (h*ng/g) / blood / plasma (h*ng/L) 29227 / 7583 / 2542 14653 / 3169 / 782 Cmax tumor (ng/g) / blood / plasma (ng/L) 1911 / 1018 / 386 683 / 425 / 156 Tumor DUSP6 inhibition at 168 / 192 hr 85.5% / 46.3% 63.8% / 7.9%
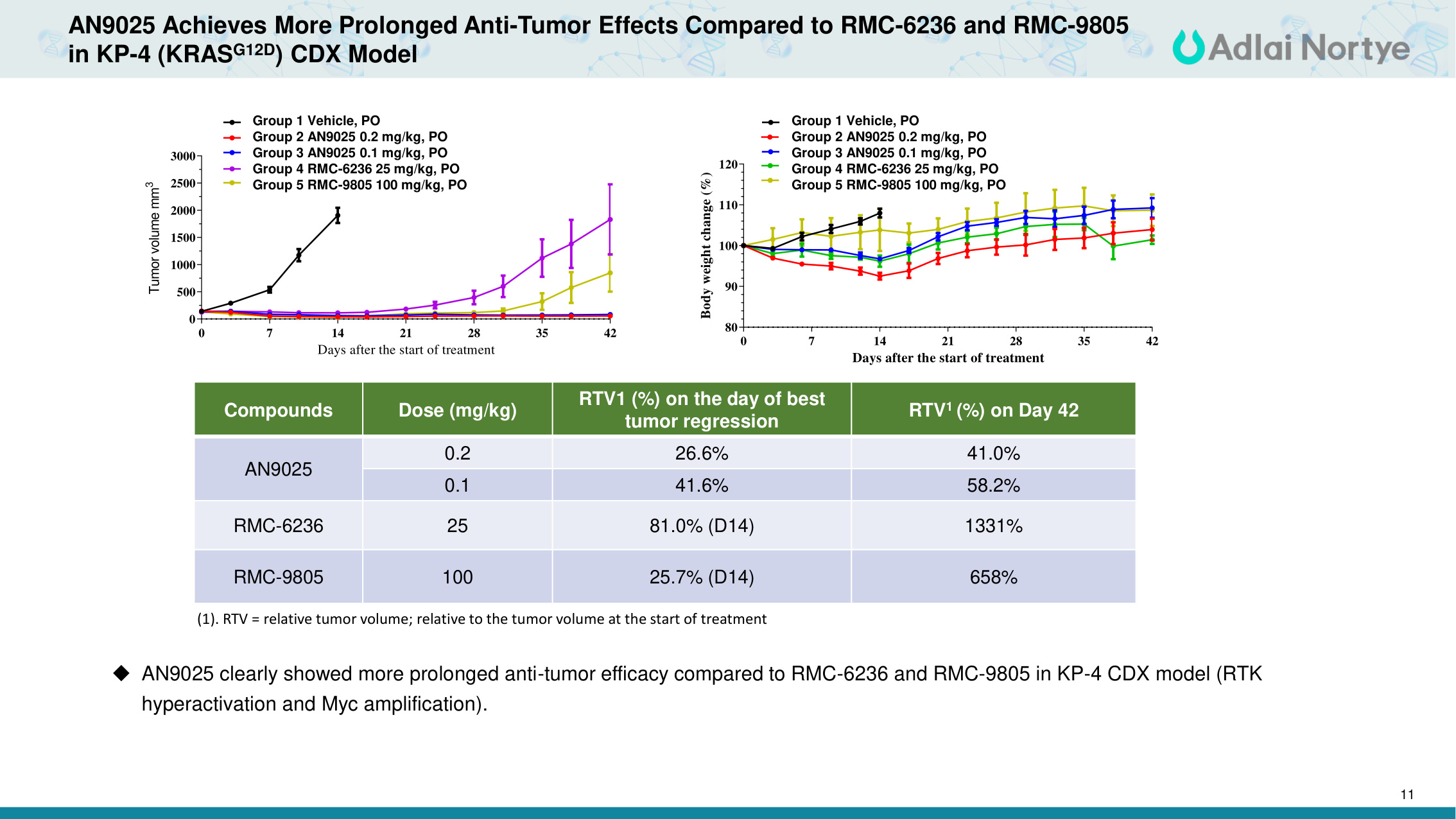
11 AN9025 Achieves More Prolonged Anti-Tumor Effects Compared to RMC-6236 and RMC-9805 in KP-4 (KRASG12D) CDX Model Compounds Dose (mg/kg) RTV1 (%) on the day of best tumor regression RTV1 (%) on Day 42 AN9025 0.2 26.6% 41.0% 0.1 41.6% 58.2% RMC-6236 25 81.0% (D14) 1331% RMC-9805 100 25.7% (D14) 658% Days after the start of treatment Tumor volume mm3 0 7 14 21 28 35 42 0 500 1000 1500 2000 2500 3000 Group 1 Vehicle, PO Group 2 AN9025 0.2 mg/kg, PO Group 3 AN9025 0.1 mg/kg, PO Group 4 RMC-6236 25 mg/kg, PO Group 5 RMC-9805 100 mg/kg, PO Days after the start of treatment Body weight change (%) 0 7 14 21 28 35 42 80 90 100 110 120 Group 1 Vehicle, PO Group 2 AN9025 0.2 mg/kg, PO Group 3 AN9025 0.1 mg/kg, PO Group 4 RMC-6236 25 mg/kg, PO Group 5 RMC-9805 100 mg/kg, PO (1). RTV = relative tumor volume; relative to the tumor volume at the start of treatment AN9025 clearly showed more prolonged anti-tumor efficacy compared to RMC-6236 and RMC-9805 in KP-4 CDX model (RTK hyperactivation and Myc amplification). Group 1 Vehicle, PO Group 2 AN9025 0.2 mg/kg, PO Group 3 AN9025 0.1 mg/kg, PO Group 4 RMC-6236 25 mg/kg, PO Group 5 RMC-9805 100 mg/kg, PO Group 1 Vehicle, PO Group 2 AN9025 0.2 mg/kg, PO Group 3 AN9025 0.1 mg/kg, PO Group 4 RMC-6236 25 mg/kg, PO Group 5 RMC-9805 100 mg/kg, PO
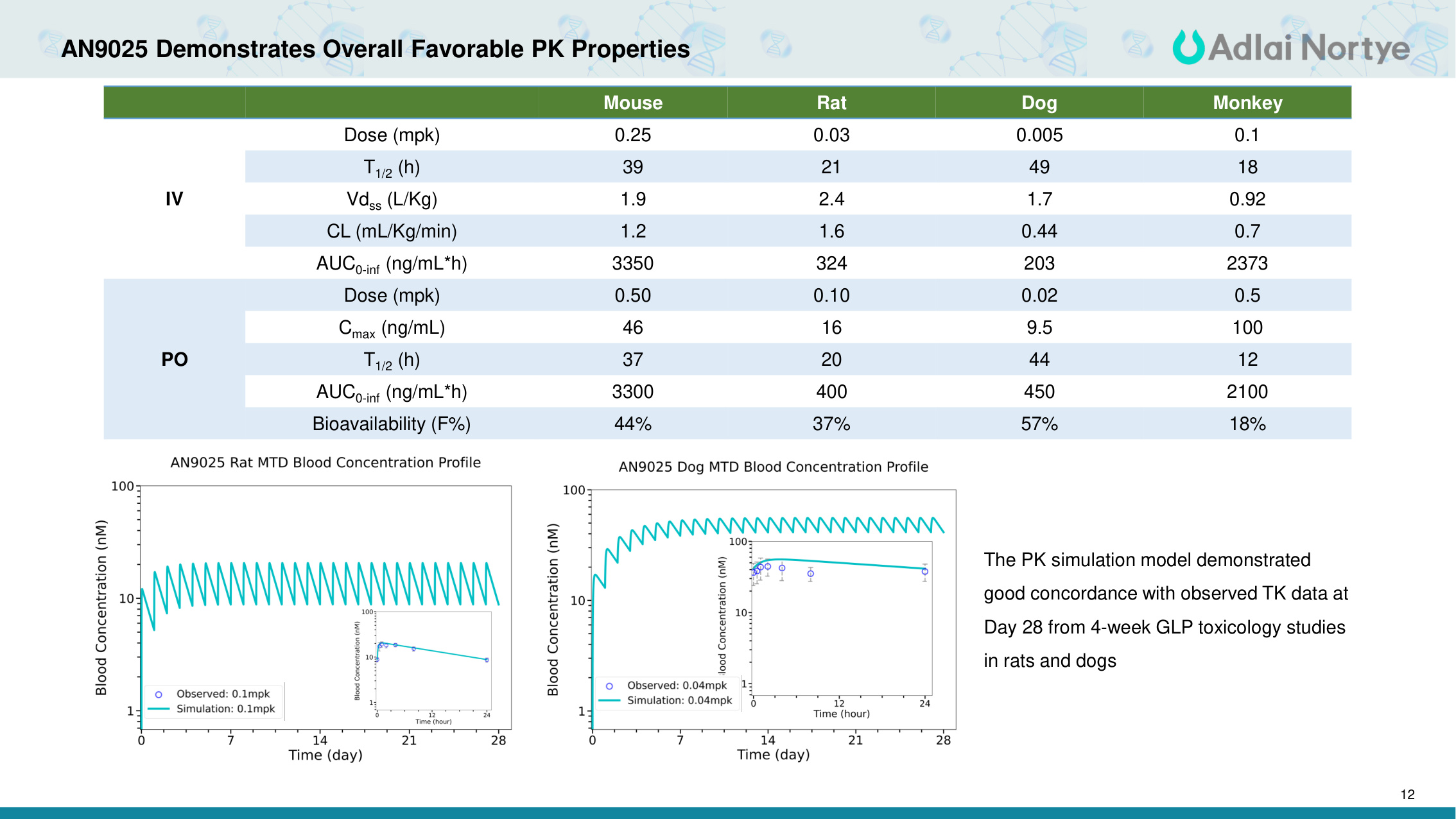
12 AN9025 Demonstrates Overall Favorable PK Properties Mouse Rat Dog Monkey IV Dose (mpk) 0.25 0.03 0.005 0.1 T1/2 (h) 39 21 49 18 Vdss (L/Kg) 1.9 2.4 1.7 0.92 CL (mL/Kg/min) 1.2 1.6 0.44 0.7 AUC0-inf (ng/mL*h) 3350 324 203 2373 PO Dose (mpk) 0.50 0.10 0.02 0.5 Cmax (ng/mL) 46 16 9.5 100 T1/2 (h) 37 20 44 12 AUC0-inf (ng/mL*h) 3300 400 450 2100 Bioavailability (F%) 44% 37% 57% 18% The PK simulation model demonstrated good concordance with observed TK data at Day 28 from 4-week GLP toxicology studies in rats and dogs
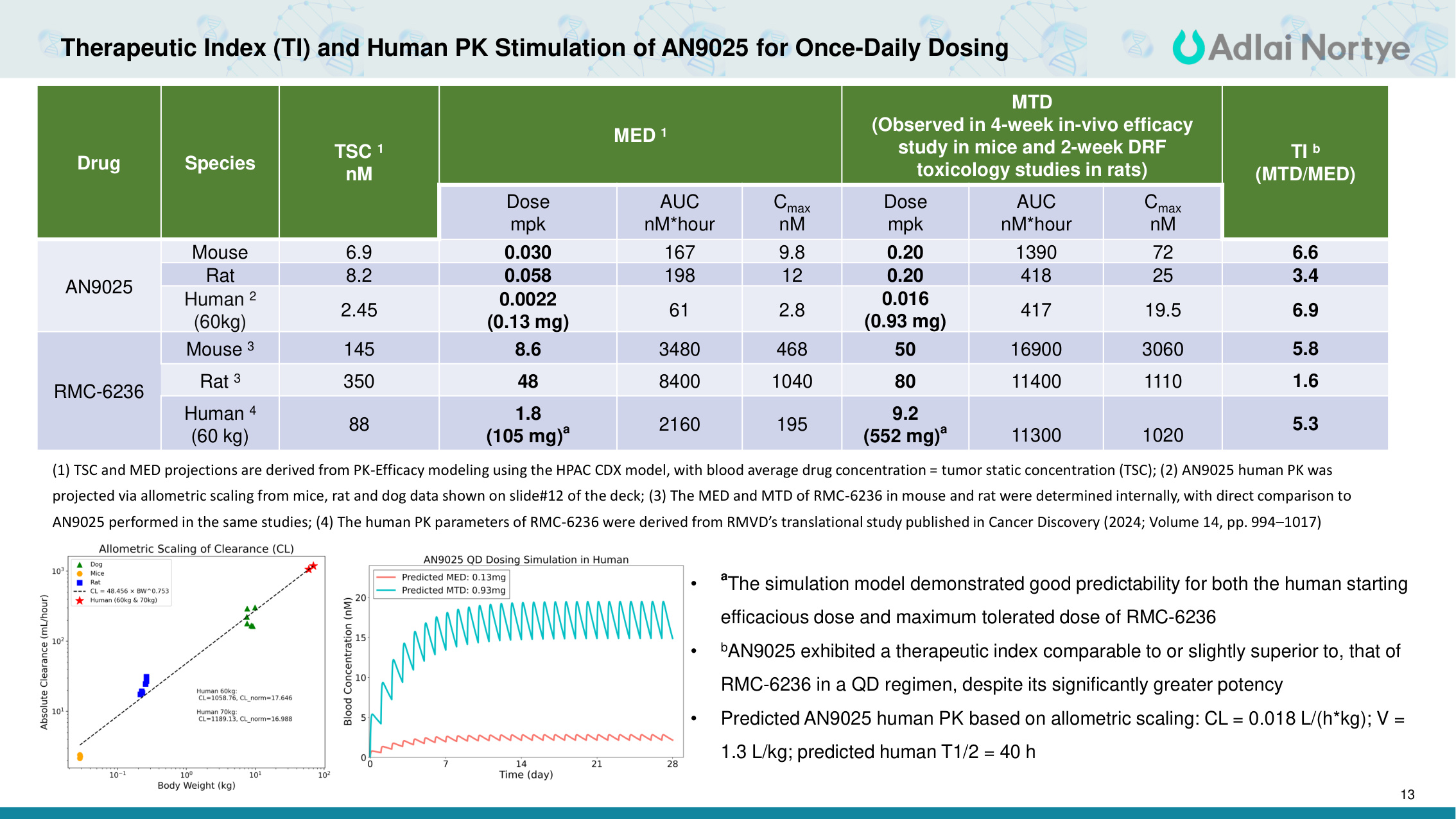
13 Therapeutic Index (TI) and Human PK Stimulation of AN9025 for Once-Daily Dosing Drug Species TSC 1 nM MED 1 MTD (Observed in 4-week in-vivo efficacy study in mice and 2-week DRF toxicology studies in rats) TI b (MTD/MED) Dose mpk AUC nM*hour Cmax nM Dose mpk AUC nM*hour Cmax nM AN9025 Mouse 6.9 0.030 167 9.8 0.20 1390 72 6.6 Rat 8.2 0.058 198 12 0.20 418 25 3.4 Human 2 (60kg) 2.45 0.0022 (0.13 mg) 61 2.8 0.016 (0.93 mg) 417 19.5 6.9 RMC-6236 Mouse 3 145 8.6 3480 468 50 16900 3060 5.8 Rat 3 350 48 8400 1040 80 11400 1110 1.6 Human 4 (60 kg) 88 1.8 (105 mg)a 2160 195 9.2 (552 mg)a 11300 1020 5.3 (1) TSC and MED projections are derived from PK-Efficacy modeling using the HPAC CDX model, with blood average drug concentration = tumor static concentration (TSC); (2) AN9025 human PK was projected via allometric scaling from mice, rat and dog data shown on slide#12 of the deck; (3) The MED and MTD of RMC-6236 in mouse and rat were determined internally, with direct comparison to AN9025 performed in the same studies; (4) The human PK parameters of RMC-6236 were derived from RMVD's translational study published in Cancer Discovery (2024; Volume 14, pp. 994–1017) • aThe simulation model demonstrated good predictability for both the human starting efficacious dose and maximum tolerated dose of RMC-6236 • bAN9025 exhibited a therapeutic index comparable to or slightly superior to, that of RMC-6236 in a QD regimen, despite its significantly greater potency • Predicted AN9025 human PK based on allometric scaling: CL = 0.018 L/(h*kg); V = 1.3 L/kg; predicted human T1/2 = 40 h
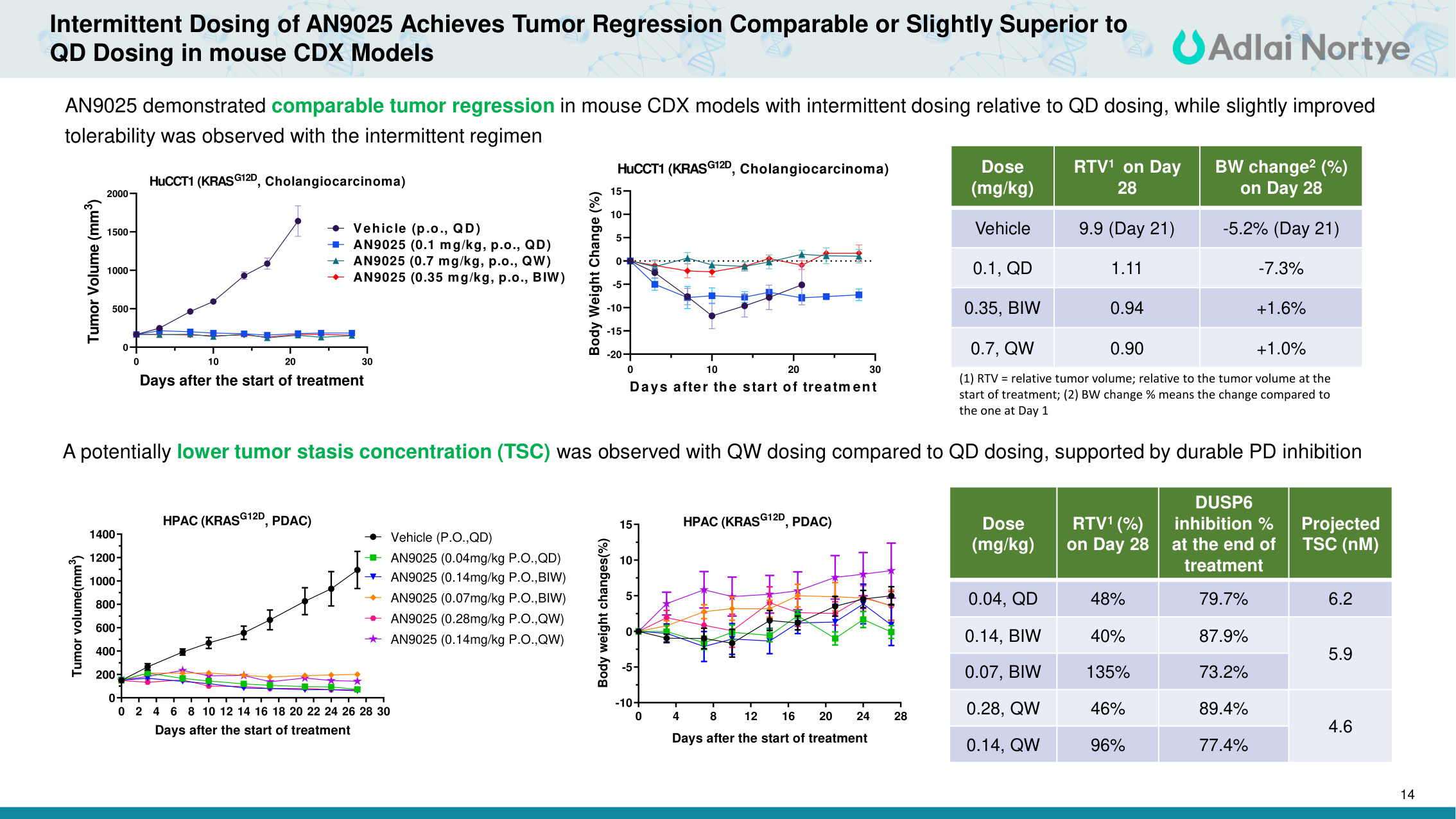
14 A potentially lower tumor stasis concentration (TSC) was observed with QW dosing compared to QD dosing, supported by durable PD inhibition 0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30 0 200 400 600 800 1000 1200 1400 Days after the start of treatment Tumor volume(mm3) Vehicle (P.O.,QD) AN9025 (0.04mg/kg P.O.,QD) AN9025 (0.14mg/kg P.O.,BIW) AN9025 (0.07mg/kg P.O.,BIW) AN9025 (0.28mg/kg P.O.,QW) AN9025 (0.14mg/kg P.O.,QW) HPAC (KRASG12D, PDAC) 0 4 8 12 16 20 24 28 -10 -5 0 5 10 15 Days after the start of treatment Body weight changes(%) HPAC (KRASG12D, PDAC) Dose (mg/kg) RTV1 (%) on Day 28 DUSP6 inhibition % at the end of treatment Projected TSC (nM) 0.04, QD 48% 79.7% 6.2 0.14, BIW 40% 87.9% 5.9 0.07, BIW 135% 73.2% 0.28, QW 46% 89.4% 4.6 0.14, QW 96% 77.4% 0 10 20 30 -20 -15 -10 -5 0 5 10 15 Days after the start of treatm ent Body Weight Change (%) HuCCT1 (KRASG12D, Cholangiocarcinoma) Dose (mg/kg) RTV1 on Day 28 BW change2 (%) on Day 28 Vehicle 9.9 (Day 21) -5.2% (Day 21) 0.1, QD 1.11 -7.3% 0.35, BIW 0.94 +1.6% 0.7, QW 0.90 +1.0% (1) RTV = relative tumor volume; relative to the tumor volume at the start of treatment; (2) BW change % means the change compared to the one at Day 1 0 10 20 30 0 500 1000 1500 2000 Days after the start of treatment Tumor Volume (mm3) Vehicle (p.o., Q D) AN9025 (0.1 mg/kg, p.o., QD) AN9025 (0.7 mg/kg, p.o., QW) AN9025 (0.35 mg/kg, p.o., BIW) HuCCT1 (KRASG12D, Cholangiocarcinoma) AN9025 demonstrated comparable tumor regression in mouse CDX models with intermittent dosing relative to QD dosing, while slightly improved tolerability was observed with the intermittent regimen Intermittent Dosing of AN9025 Achieves Tumor Regression Comparable or Slightly Superior to QD Dosing in mouse CDX Models
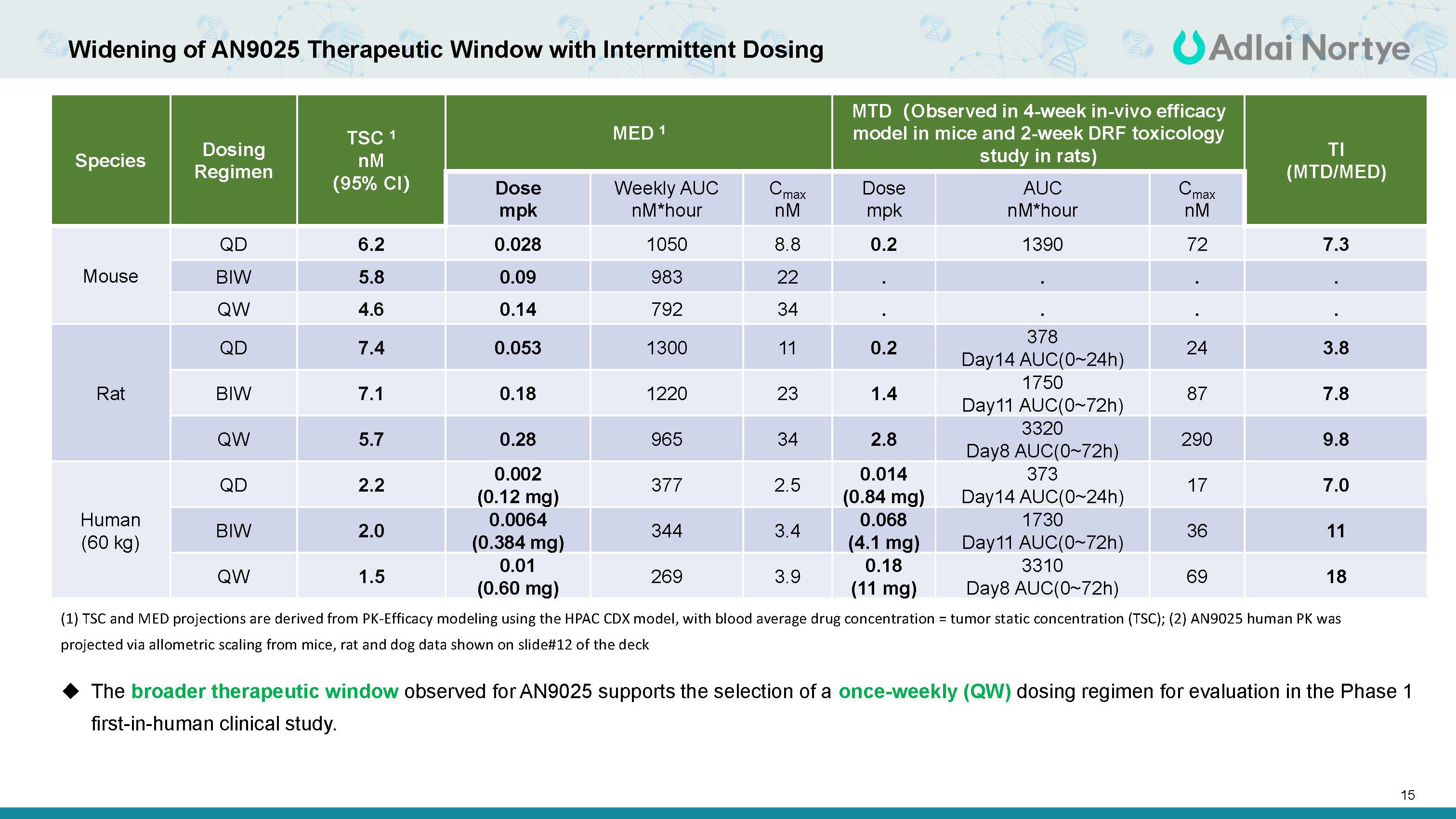
15 Widening of AN025 Therapeutic Window with Intermittent Dosing Species Dosing Regimen TSC 1 nM (95% CI) MED 1 MTD(Observed in 4-week in-vivo efficacy model in mice and 2-week DRF toxicology study in rats) TI (MTD/MED) Dose mpk Weekly AUC nM*hour Cmax nM Dose mpk AUC nM*hour Cmax nM Mouse QD 6.2 0.028 1050 8.8 0.2 1390 72 7.3 BIW 5.8 0.09 983 22 . . . . QW 4.6 0.14 792 34 . . . . Rat QD 7.4 0.053 1300 11 0.2 378 Day14 AUC(0~24h) 24 3.8 BIW 7.1 0.18 1220 23 1.4 1750 Day11 AUC(0~72h) 87 7.8 QW 5.7 0.28 965 34 2.8 3320 Day8 AUC(0~72h) 290 9.8 Human (60 kg) QD 2.2 0.002 (0.12 mg) 377 2.5 0.014 (0.84 mg) 373 Day14 AUC(0~24h) 17 7.0 BIW 2.0 0.0064 (0.384 mg) 344 3.4 0.068 (4.1 mg) 1730 Day11 AUC(0~72h) 36 11 QW 1.5 0.01 (0.60 mg) 269 3.9 0.18 (11 mg) 3310 Day8 AUC(0~72h) 69 18 (1) TSC and MED projections are derived from PK-Efficacy modeling using the HPAC CDX model, with blood average drug concentration = tumor static concentration (TSC); (2) AN9025 human PK was projected via allometric scaling from mice, rat and dog data shown on slide#12 of the deck The broader therapeutic window observed for AN9025 supports the selection of a once-weekly (QW) dosing regimen for evaluation in the Phase 1 first-in-human clinical study.

16 Summary of AN9025 AN9025 holds best-in-class potential within the Pan-RAS(ON) inhibitor category: • Exhibited a binding affinity for cyclophilin A (CypA) that is 4-folder stronger than that of RMC-6236, driven by a slower dissociation rate from CypA, which may enable more sustained RAS inhibition • Showed 3- to 8-fold higher binding affinity for tri-complex formation, also attributable to slower dissociation, potentially leading to prolonged suppression of RAS signaling • Demonstrated 100-fold more potent inhibition of cellular proliferation and provided more sustained RAS inhibition compared to RMC-6236 • Induced deep tumor regression, superior and more sustained anti-tumor effects compared to RMC-6236 in mouse CDX models covering different RAS mutant types • Demonstrated favorable overall DMPK properties, with 20–55% bioavailability across multiple species and a 2- to 5-fold longer half-life compared with RMC-6236 • Demonstrated a therapeutic index comparable to or slightly superior to RMC-6236 in QD regimen despite significantly improved potency The first patient has been dosed in early February 2026 in the ongoing global Phase 1 clinical trial in the US and China. Initiation of the intermittent dosing cohort is anticipated in mid-2026, with completion of full dose escalation expected by H1 2027.

17 AN4035: A Novel CEACAM5 Pan-RAS(ON)i ADC
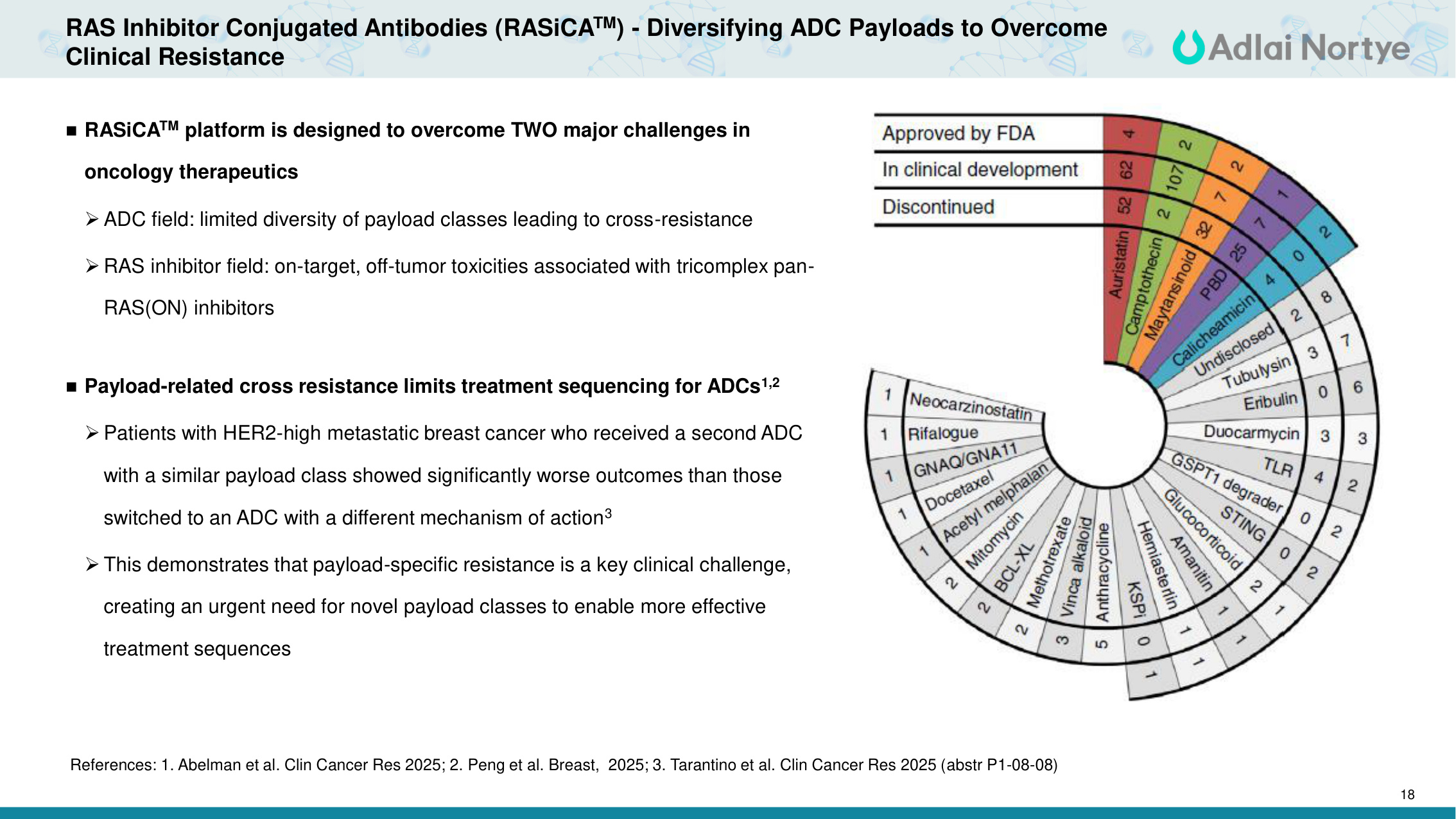
18 RAS Inhibitor Conjugated Antibodies (RASiCATM) - Diversifying ADC Payloads to Overcome Clinical Resistance References: 1. Abelman et al. Clin Cancer Res 2025; 2. Peng et al. Breast, 2025; 3. Tarantino et al. Clin Cancer Res 2025 (abstr P1-08-08) RASiCATM platform is designed to overcome TWO major challenges in oncology therapeutics ADC field: limited diversity of payload classes leading to cross-resistance RAS inhibitor field: on-target, off-tumor toxicities associated with tricomplex pan- RAS(ON) inhibitors Payload-related cross resistance limits treatment sequencing for ADCs1,2 Patients with HER2-high metastatic breast cancer who received a second ADC with a similar payload class showed significantly worse outcomes than those switched to an ADC with a different mechanism of action3 This demonstrates that payload-specific resistance is a key clinical challenge, creating an urgent need for novel payload classes to enable more effective treatment sequences
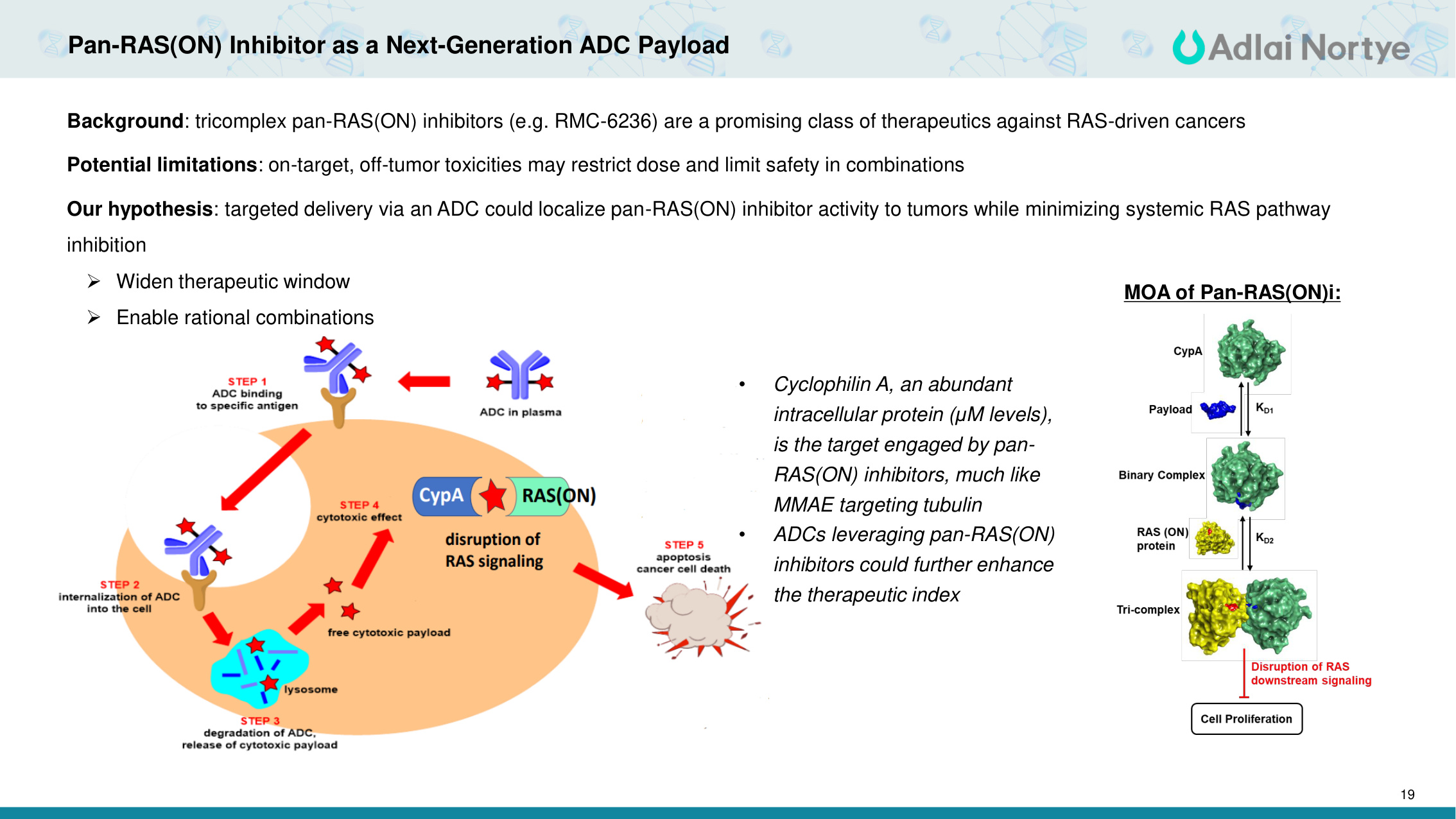
19 Pan-RAS(ON) Inhibitor as a Next-Generation ADC Payload • Cyclophilin A, an abundant intracellular protein (μM levels), is the target engaged by pan- RAS(ON) inhibitors, much like MMAE targeting tubulin • ADCs leveraging pan-RAS(ON) inhibitors could further enhance the therapeutic index MOA of Pan-RAS(ON)i: Background: tricomplex pan-RAS(ON) inhibitors (e.g. RMC-6236) are a promising class of therapeutics against RAS-driven cancers Potential limitations: on-target, off-tumor toxicities may restrict dose and limit safety in combinations Our hypothesis: targeted delivery via an ADC could localize pan-RAS(ON) inhibitor activity to tumors while minimizing systemic RAS pathway inhibition Widen therapeutic window Enable rational combinations
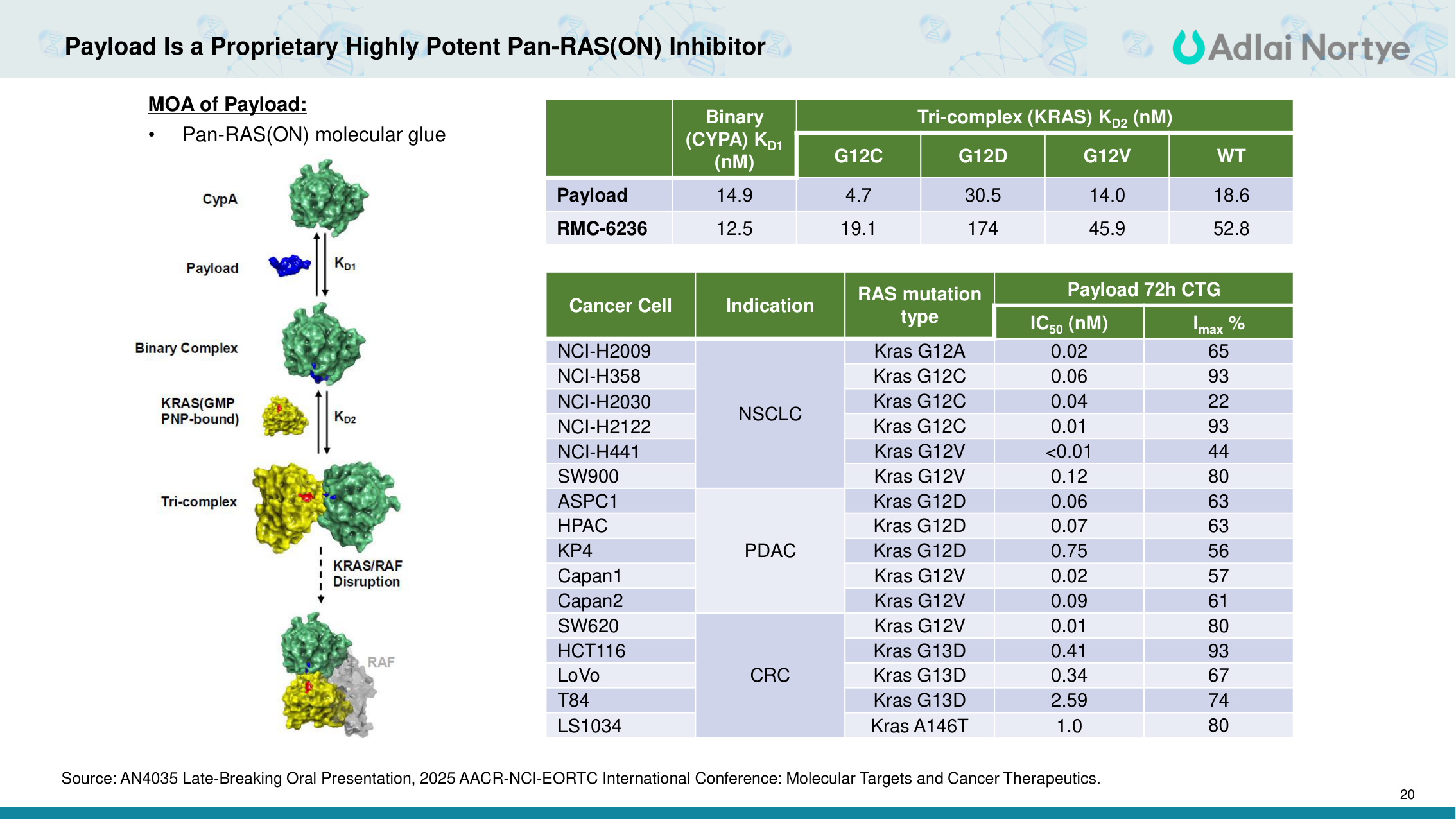
20 Payload Is a Proprietary Highly Potent Pan-RAS(ON) Inhibitor Binary (CYPA) KD1 (nM) Tri-complex (KRAS) KD2 (nM) G12C G12D G12V WT Payload 14.9 4.7 30.5 14.0 18.6 RMC-6236 12.5 19.1 174 45.9 52.8 Cancer Cell Indication RAS mutation type Payload 72h CTG IC50 (nM) Imax % NCI-H2009 NSCLC Kras G12A 0.02 65 NCI-H358 Kras G12C 0.06 93 NCI-H2030 Kras G12C 0.04 22 NCI-H2122 Kras G12C 0.01 93 NCI-H441 Kras G12V <0.01 44 SW900 Kras G12V 0.12 80 ASPC1 PDAC Kras G12D 0.06 63 HPAC Kras G12D 0.07 63 KP4 Kras G12D 0.75 56 Capan1 Kras G12V 0.02 57 Capan2 Kras G12V 0.09 61 SW620 CRC Kras G12V 0.01 80 HCT116 Kras G13D 0.41 93 LoVo Kras G13D 0.34 67 T84 Kras G13D 2.59 74 LS1034 Kras A146T 1.0 80 MOA of Payload: • Pan-RAS(ON) molecular glue Source: AN4035 Late-Breaking Oral Presentation, 2025 AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics.
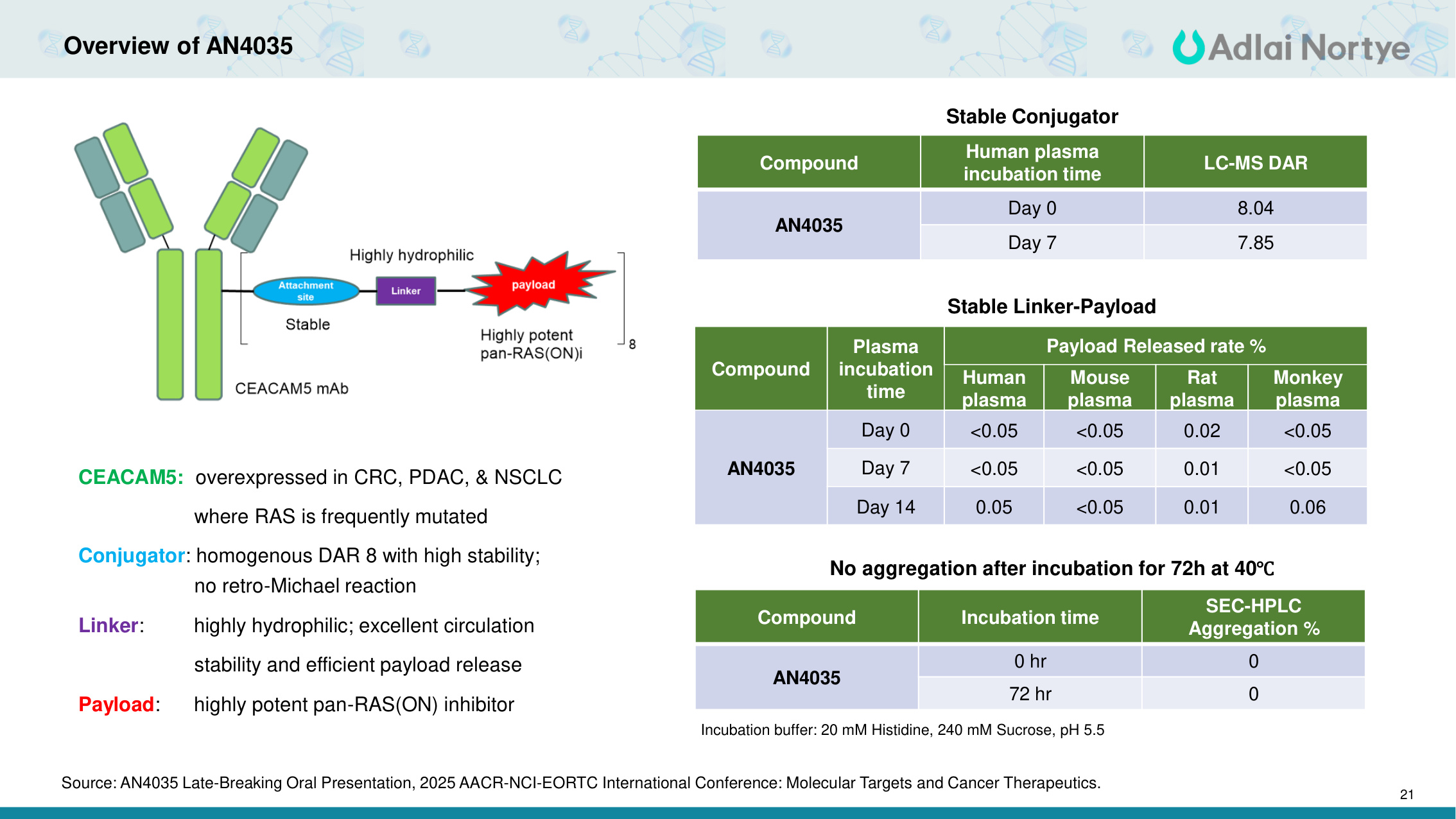
21 Overview of AN4035 CEACAM5: overexpressed in CRC, PDAC, & NSCLC where RAS is frequently mutated Conjugator: homogenous DAR 8 with high stability; no retro-Michael reaction Linker: highly hydrophilic; excellent circulation stability and efficient payload release Payload: highly potent pan-RAS(ON) inhibitor Compound Human plasma incubation time LC-MS DAR AN4035 Day 0 8.04 Day 7 7.85 Stable Conjugator Compound Plasma incubation time Payload Released rate % Human plasma Mouse plasma Rat plasma Monkey plasma AN4035 Day 0 <0.05 <0.05 0.02 <0.05 Day 7 <0.05 <0.05 0.01 <0.05 Day 14 0.05 <0.05 0.01 0.06 Stable Linker-Payload No aggregation after incubation for 72h at 40℃ Compound Incubation time SEC-HPLC Aggregation % AN4035 0 hr 0 72 hr 0 Incubation buffer: 20 mM Histidine, 240 mM Sucrose, pH 5.5 Source: AN4035 Late-Breaking Oral Presentation, 2025 AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics.
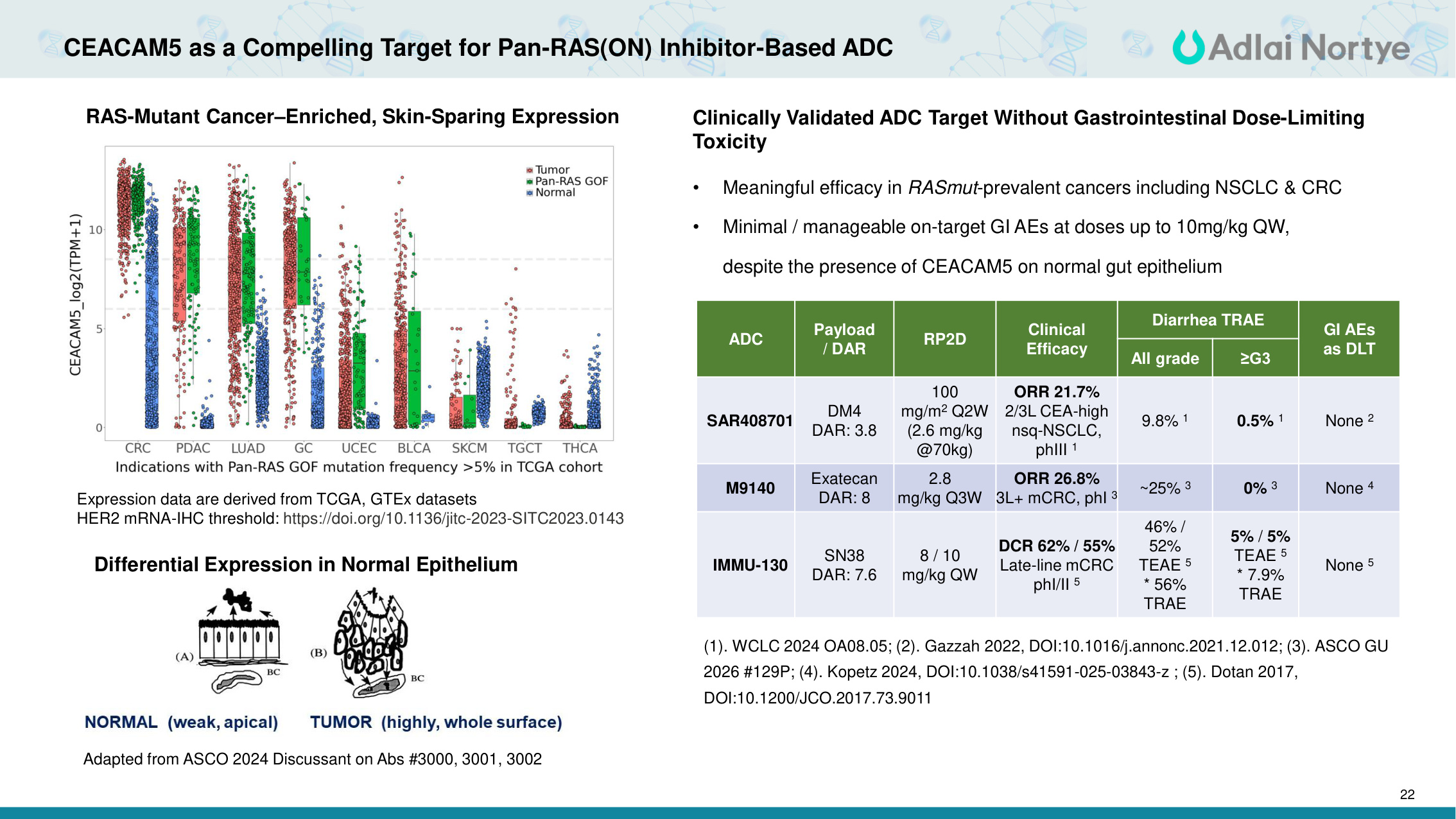
22 CEACAM5 as a Compelling Target for Pan-RAS(ON) Inhibitor-Based ADC RAS-Mutant Cancer–Enriched, Skin-Sparing Expression Differential Expression in Normal Epithelium Adapted from ASCO 2024 Discussant on Abs #3000, 3001, 3002 Expression data are derived from TCGA, GTEx datasets HER2 mRNA-IHC threshold: https://doi.org/10.1136/jitc-2023-SITC2023.0143 Clinically Validated ADC Target Without Gastrointestinal Dose-Limiting Toxicity • Meaningful efficacy in RASmut-prevalent cancers including NSCLC & CRC • Minimal / manageable on-target GI AEs at doses up to 10mg/kg QW, despite the presence of CEACAM5 on normal gut epithelium ADC Payload / DAR RP2D Clinical Efficacy Diarrhea TRAE GI AEs as DLT All grade ≥G3 SAR408701 DM4 DAR: 3.8 100 mg/m2 Q2W (2.6 mg/kg @70kg) ORR 21.7% 2/3L CEA-high nsq-NSCLC, phIII 1 9.8% 1 0.5% 1 None 2 M9140 Exatecan DAR: 8 2.8 mg/kg Q3W ORR 26.8% 3L+ mCRC, phI 3 ~25% 3 0% 3 None 4 IMMU-130 SN38 DAR: 7.6 8 / 10 mg/kg QW DCR 62% / 55% Late-line mCRC phI/II 5 46% / 52% TEAE 5 * 56% TRAE 5% / 5% TEAE 5 * 7.9% TRAE None 5 (1). WCLC 2024 OA08.05; (2). Gazzah 2022, DOI:10.1016/j.annonc.2021.12.012; (3). ASCO GU 2026 #129P; (4). Kopetz 2024, DOI:10.1038/s41591-025-03843-z ; (5). Dotan 2017, DOI:10.1200/JCO.2017.73.9011
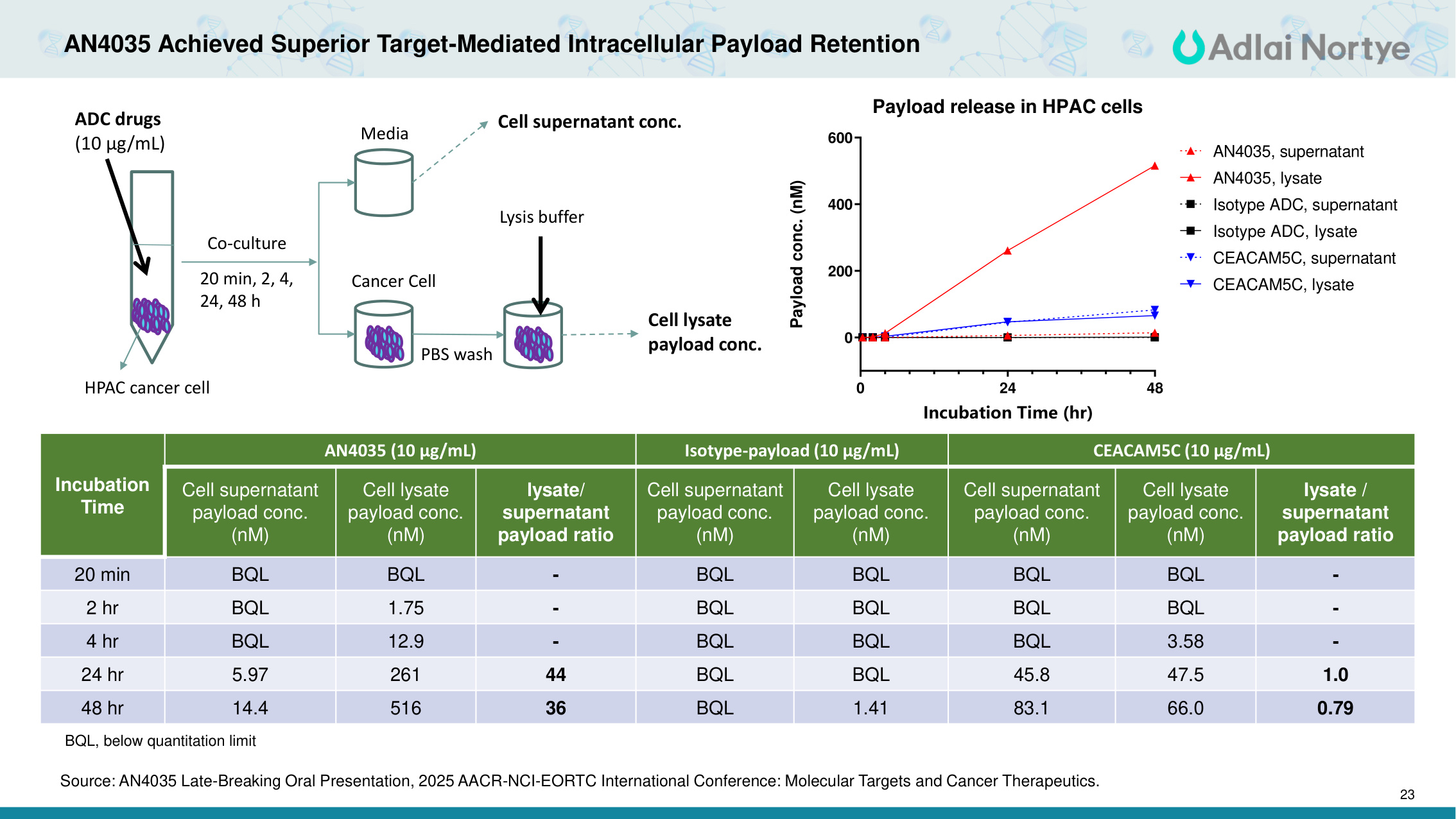
23 AN4035 Achieved Superior Target-Mediated Intracellular Payload Retention Incubation Time AN4035 (10 μg/mL) Isotype-payload (10 μg/mL) CEACAM5C (10 μg/mL) Cell supernatant payload conc. (nM) Cell lysate payload conc. (nM) lysate/ supernatant payload ratio Cell supernatant payload conc. (nM) Cell lysate payload conc. (nM) Cell supernatant payload conc. (nM) Cell lysate payload conc. (nM) Iysate / supernatant payload ratio 20 min BQL BQL - BQL BQL BQL BQL - 2 hr BQL 1.75 - BQL BQL BQL BQL - 4 hr BQL 12.9 - BQL BQL BQL 3.58 - 24 hr 5.97 261 44 BQL BQL 45.8 47.5 1.0 48 hr 14.4 516 36 BQL 1.41 83.1 66.0 0.79 BQL, below quantitation limit HPAC cancer cell Co-culture 20 min, 2, 4, 24, 48 h Media PBS wash Cancer Cell ADC drugs (10 μg/mL) Lysis buffer Cell supernatant conc. Cell lysate payload conc. 0 24 48 0 200 400 600 Payload release in HPAC cells Incubation Time (hr) Payload conc. (nM) AN4035, supernatant AN4035, lysate Isotype ADC, supernatant Isotype ADC, Iysate CEACAM5C, supernatant CEACAM5C, lysate Source: AN4035 Late-Breaking Oral Presentation, 2025 AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics.
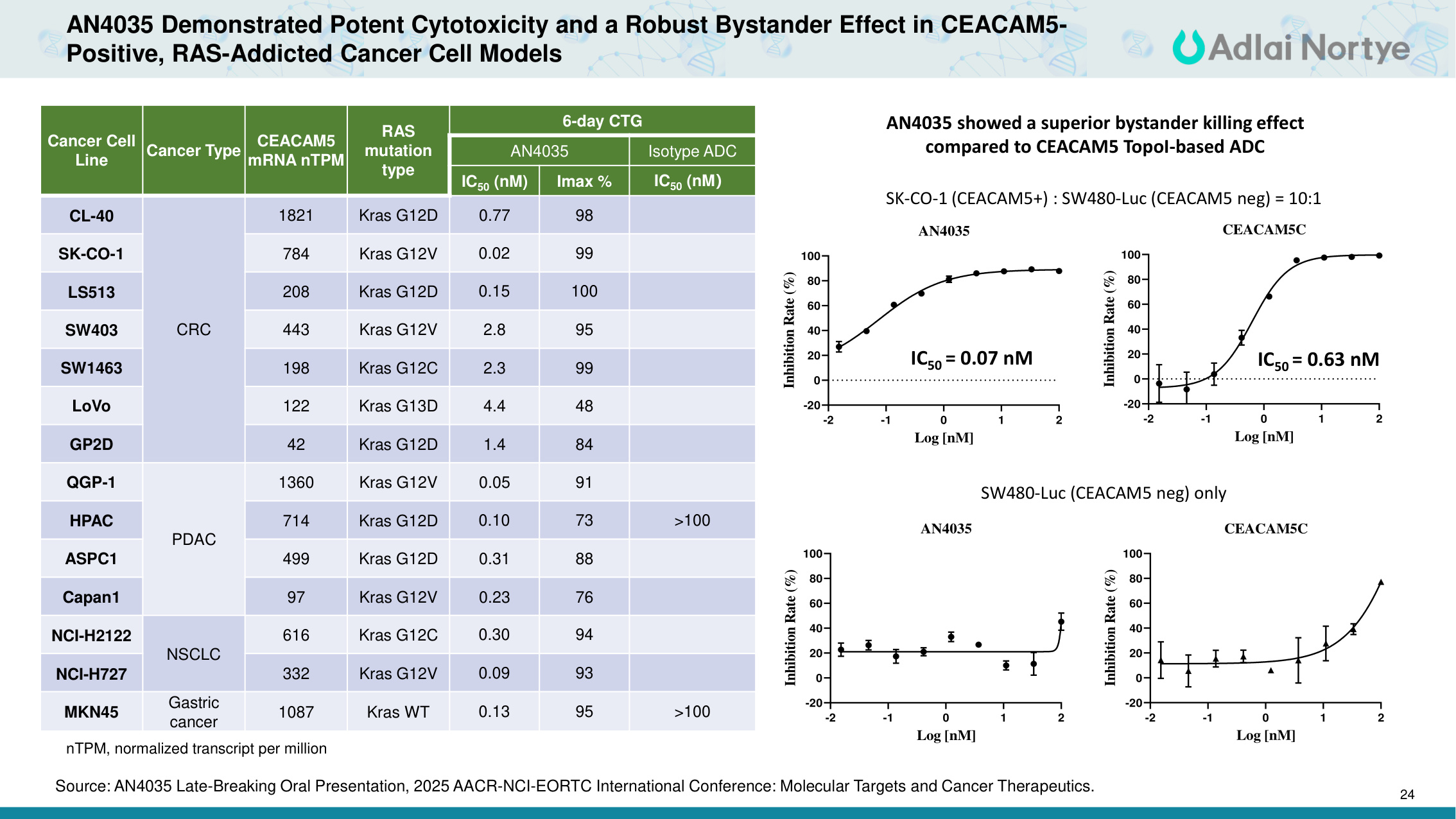
24 AN4035 Demonstrated Potent Cytotoxicity and a Robust Bystander Effect in CEACAM5- Positive, RAS-Addicted Cancer Cell Models Cancer Cell Line Cancer Type CEACAM5 mRNA nTPM RAS mutation type 6-day CTG AN4035 Isotype ADC IC50 (nM) Imax % IC50 (nM) CL-40 CRC 1821 Kras G12D 0.77 98 SK-CO-1 784 Kras G12V 0.02 99 LS513 208 Kras G12D 0.15 100 SW403 443 Kras G12V 2.8 95 SW1463 198 Kras G12C 2.3 99 LoVo 122 Kras G13D 4.4 48 GP2D 42 Kras G12D 1.4 84 QGP-1 PDAC 1360 Kras G12V 0.05 91 HPAC 714 Kras G12D 0.10 73 >100 ASPC1 499 Kras G12D 0.31 88 Capan1 97 Kras G12V 0.23 76 NCI-H2122 NSCLC 616 Kras G12C 0.30 94 NCI-H727 332 Kras G12V 0.09 93 MKN45 Gastric cancer 1087 Kras WT 0.13 95 >100 AN4035 showed a superior bystander killing effect compared to CEACAM5 TopoI-based ADC SK-CO-1 (CEACAM5+) : SW480-Luc (CEACAM5 neg) = 10:1 SW480-Luc (CEACAM5 neg) only -2 -1 0 1 2 -20 0 20 40 60 80 100 AN4035 Log [nM] Inhibition Rate (%) -2 -1 0 1 2 -20 0 20 40 60 80 100 CEACAM5C Log [nM] Inhibition Rate (%) -2 -1 0 1 2 -20 0 20 40 60 80 100 AN4035 Log [nM] Inhibition Rate (%) -2 -1 0 1 2 -20 0 20 40 60 80 100 CEACAM5C Log [nM] Inhibition Rate (%) IC50 = 0.07 nM IC50 = 0.63 nM nTPM, normalized transcript per million Source: AN4035 Late-Breaking Oral Presentation, 2025 AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics.
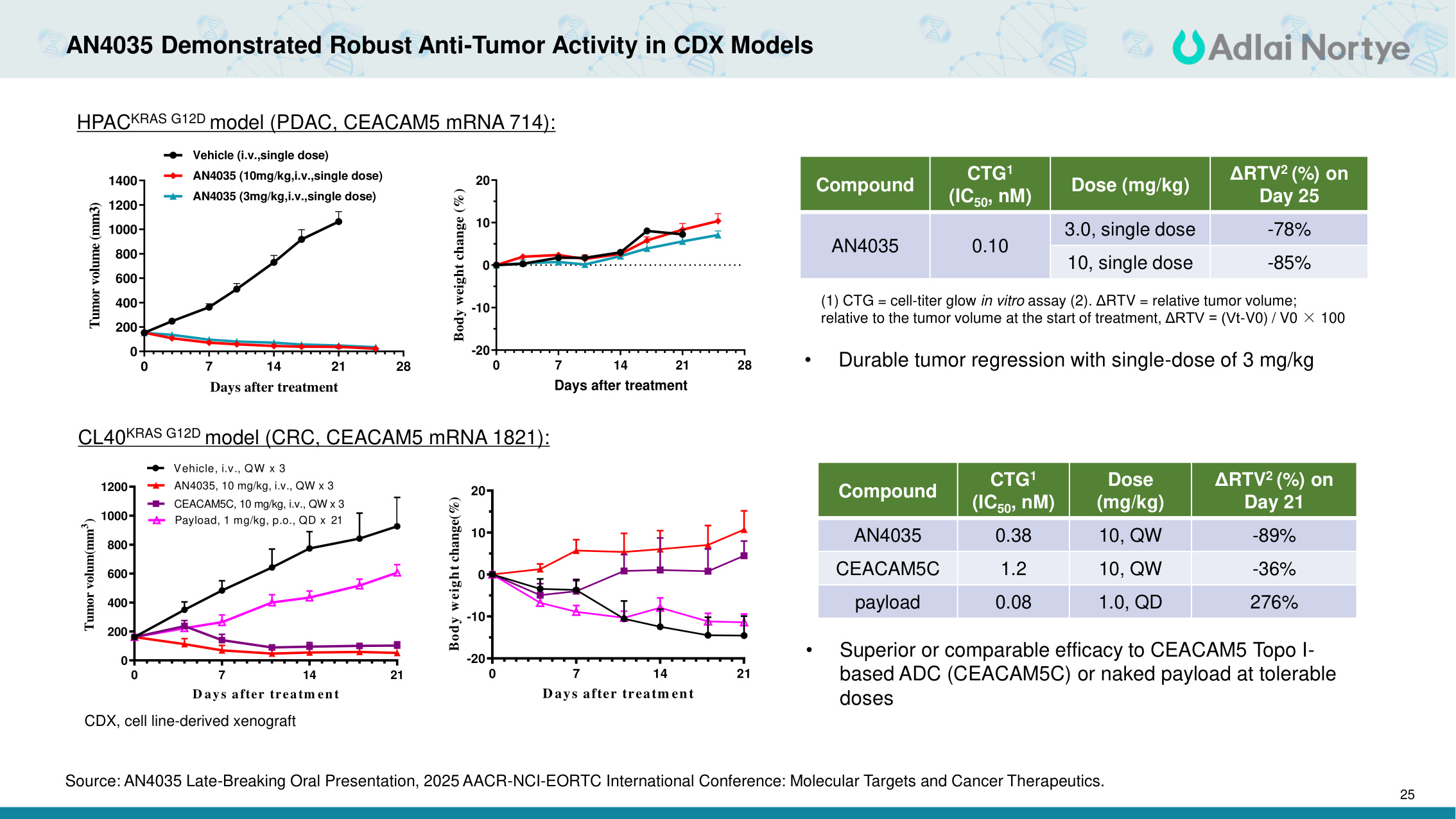
25 AN4035 Demonstrated Robust Anti-Tumor Activity in CDX Models Compound CTG1 (IC50, nM) Dose (mg/kg) ΔRTV2 (%) on Day 25 AN4035 0.10 3.0, single dose -78% 10, single dose -85% (1) CTG = cell-titer glow in vitro assay (2). ΔRTV = relative tumor volume; relative to the tumor volume at the start of treatment, ΔRTV = (Vt-V0) / V0 × 100 HPACKRAS G12D model (PDAC, CEACAM5 mRNA 714): CL40KRAS G12D model (CRC, CEACAM5 mRNA 1821): 0 7 14 21 0 200 400 600 800 1000 1200 D ays after treatm en t Tumor volum(mm3) Vehicle, i.v., QW x 3 CEACAM5C, 10 mg/kg, i.v., QW x 3 AN4035, 10 mg/kg, i.v., QW x 3 Payload, 1 mg/kg, p.o., QD x 21 0 7 14 21 -20 -10 0 10 20 D ays after treatm ent Body weight change(%) Compound CTG1 (IC50, nM) Dose (mg/kg) ΔRTV2 (%) on Day 21 AN4035 0.38 10, QW -89% CEACAM5C 1.2 10, QW -36% payload 0.08 1.0, QD 276% • Durable tumor regression with single-dose of 3 mg/kg • Superior or comparable efficacy to CEACAM5 Topo I- based ADC (CEACAM5C) or naked payload at tolerable doses 0 7 14 21 28 -20 -10 0 10 20 Days after treatment Body weight change (%) 0 7 14 21 28 0 200 400 600 800 1000 1200 1400 Days after treatment Tumor volume (mm3) Vehicle (i.v.,single dose) AN4035 (10mg/kg,i.v.,single dose) AN4035 (3mg/kg,i.v.,single dose) CDX, cell line-derived xenograft Source: AN4035 Late-Breaking Oral Presentation, 2025 AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics.
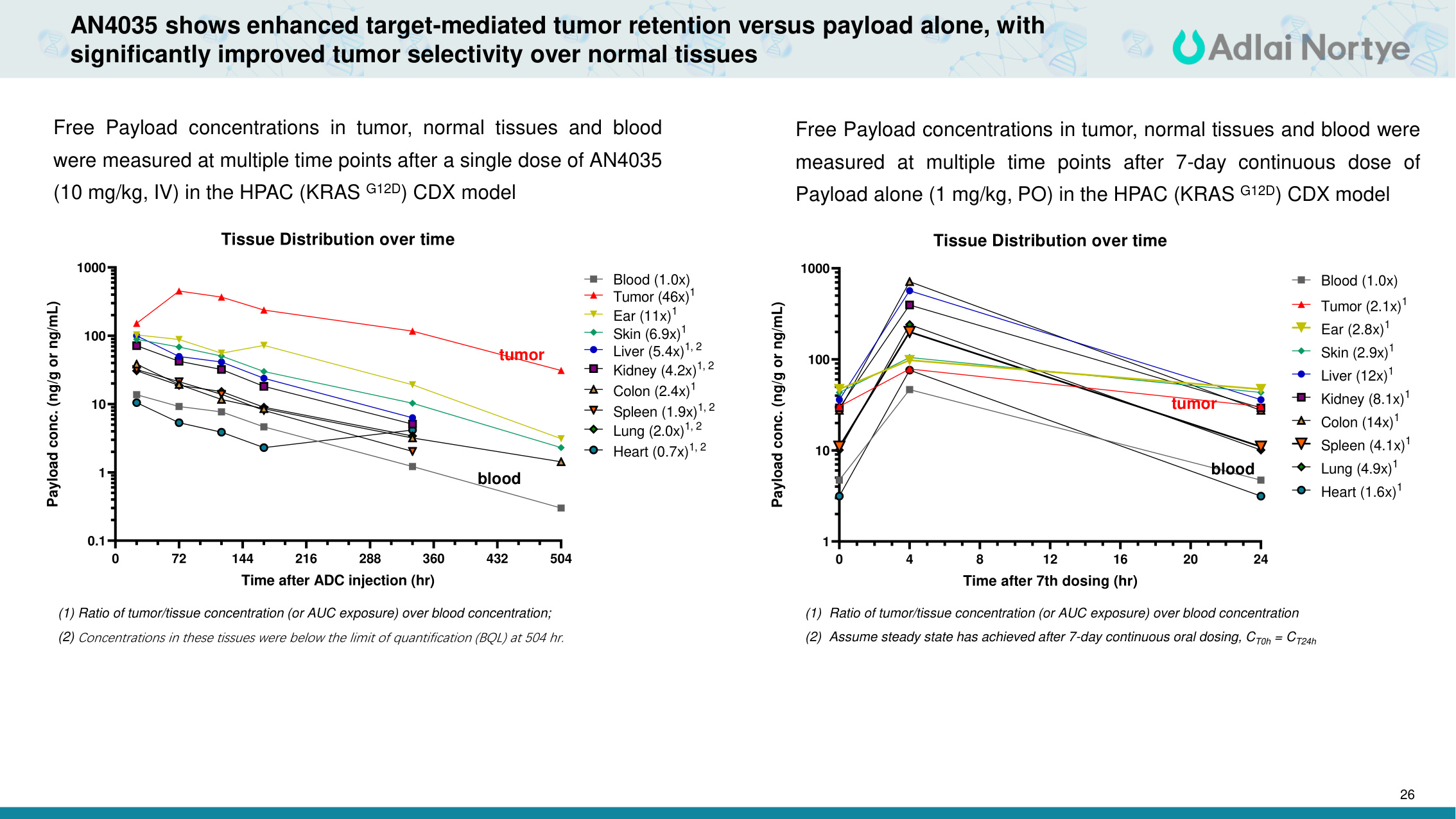
26 AN4035 shows enhanced target-mediated tumor retention versus payload alone, with significantly improved tumor selectivity over normal tissues (1) Ratio of tumor/tissue concentration (or AUC exposure) over blood concentration; (2) Concentrations in these tissues were below the limit of quantification (BQL) at 504 hr. 0 72 144 216 288 360 432 504 0.1 1 10 100 1000 Tissue Distribution over time Time after ADC injection (hr) Payload conc. (ng/g or ng/mL) Blood (1.0x) Tumor (46x)1 Ear (11x)1 Skin (6.9x)1 Liver (5.4x)1, 2 Kidney (4.2x)1, 2 Colon (2.4x)1 Spleen (1.9x)1, 2 Lung (2.0x)1, 2 Heart (0.7x)1, 2 Free Payload concentrations in tumor, normal tissues and blood were measured at multiple time points after a single dose of AN4035 (10 mg/kg, IV) in the HPAC (KRAS G12D) CDX model (1) Ratio of tumor/tissue concentration (or AUC exposure) over blood concentration (2) Assume steady state has achieved after 7-day continuous oral dosing, CT0h = CT24h Free Payload concentrations in tumor, normal tissues and blood were measured at multiple time points after 7-day continuous dose of Payload alone (1 mg/kg, PO) in the HPAC (KRAS G12D) CDX model 0 4 8 12 16 20 24 1 10 100 1000 Tissue Distribution over time Time after 7th dosing (hr) Payload conc. (ng/g or ng/mL) Blood (1.0x) Tumor (2.1x)1 Ear (2.8x)1 Skin (2.9x)1 Liver (12x)1 Kidney (8.1x)1 Colon (14x)1 Spleen (4.1x)1 Lung (4.9x)1 Heart (1.6x)1 blood tumor blood tumor
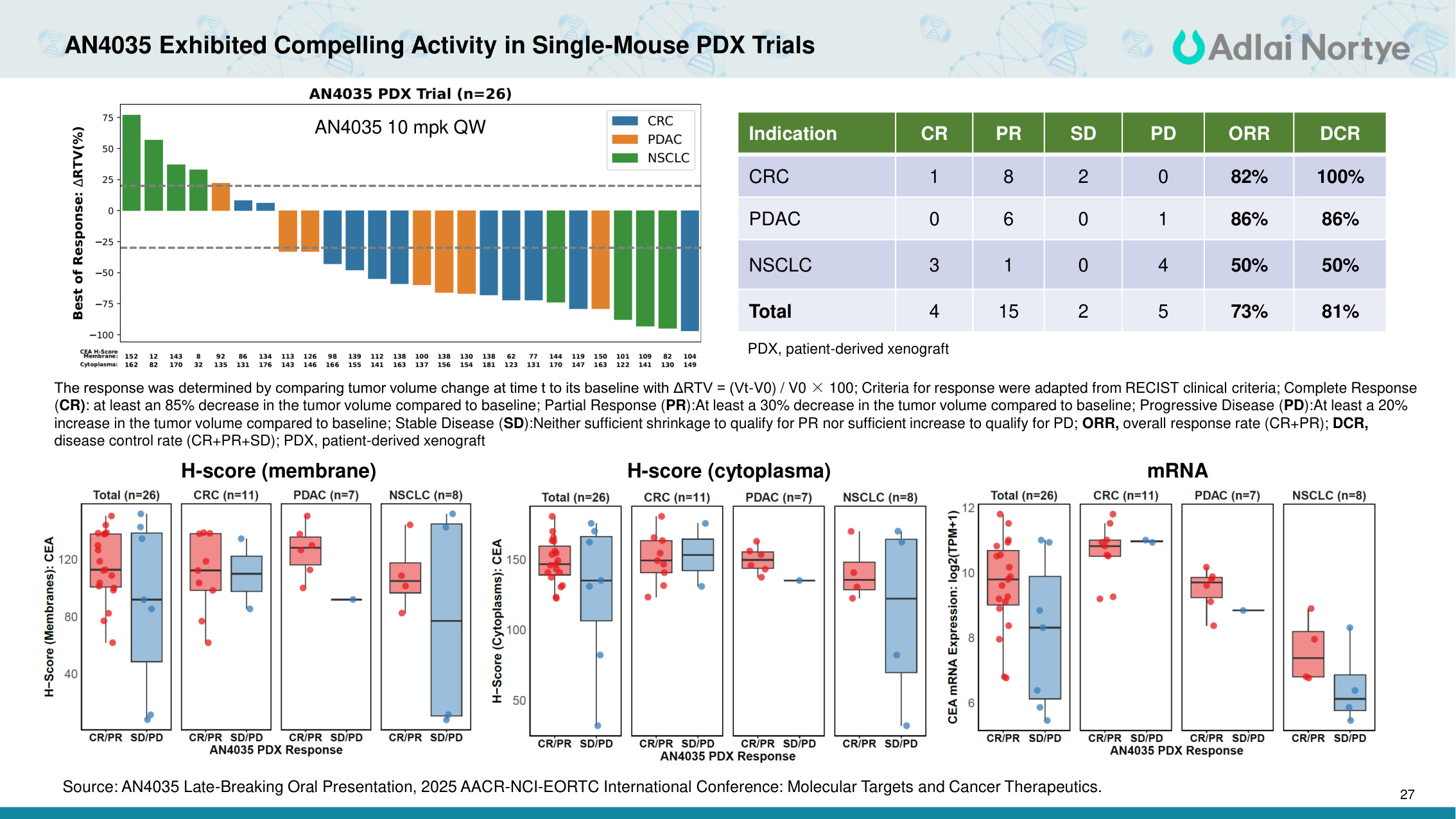
27 AN4035 Exhibited Compelling Activity in Single-Mouse PDX Trials 27 The response was determined by comparing tumor volume change at time t to its baseline with ΔRTV = (Vt-V0) / V0 × 100; Criteria for response were adapted from RECIST clinical criteria; Complete Response (CR): at least an 85% decrease in the tumor volume compared to baseline; Partial Response (PR):At least a 30% decrease in the tumor volume compared to baseline; Progressive Disease (PD):At least a 20% increase in the tumor volume compared to baseline; Stable Disease (SD):Neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD; ORR, overall response rate (CR+PR); DCR, disease control rate (CR+PR+SD); PDX, patient-derived xenograft Indication CR PR SD PD ORR DCR CRC 1 8 2 0 82% 100% PDAC 0 6 0 1 86% 86% NSCLC 3 1 0 4 50% 50% Total 4 15 2 5 73% 81% H-score (membrane) H-score (cytoplasma) mRNA PDX, patient-derived xenograft AN4035 10 mpk QW Source: AN4035 Late-Breaking Oral Presentation, 2025 AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics.
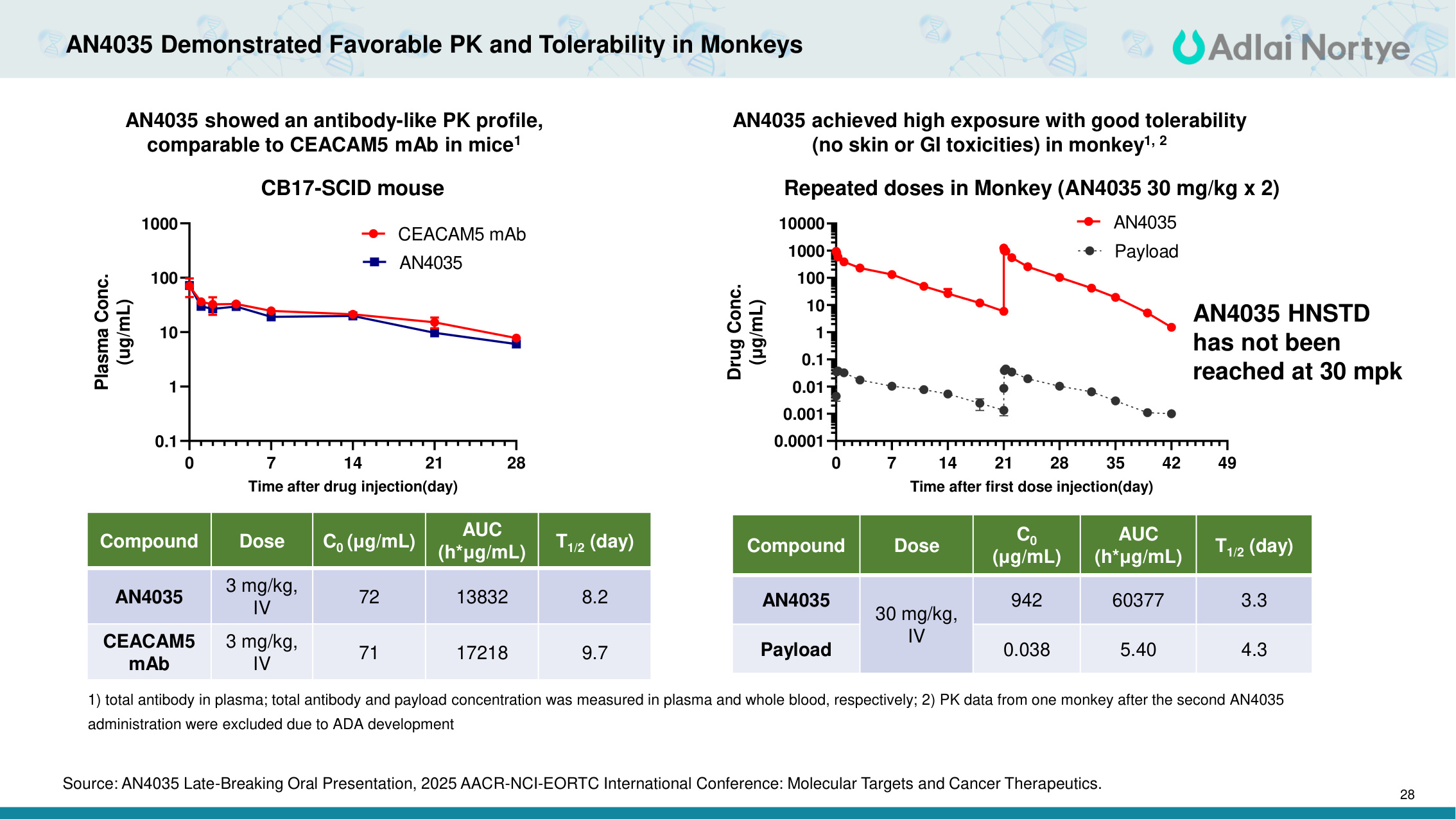
28 0 7 14 21 28 0.1 1 10 100 1000 CB17-SCID mouse Time after drug injection(day) Plasma Conc. (ug/mL) CEACAM5 mAb AN4035 AN4035 Demonstrated Favorable PK and Tolerability in Monkeys AN4035 showed an antibody-like PK profile, comparable to CEACAM5 mAb in mice1 Compound Dose C0 (μg/mL) AUC (h*μg/mL) T1/2 (day) AN4035 3 mg/kg, IV 72 13832 8.2 CEACAM5 mAb 3 mg/kg, IV 71 17218 9.7 AN4035 achieved high exposure with good tolerability (no skin or GI toxicities) in monkey1, 2 1) total antibody in plasma; total antibody and payload concentration was measured in plasma and whole blood, respectively; 2) PK data from one monkey after the second AN4035 administration were excluded due to ADA development 0 7 14 21 28 35 42 49 0.0001 0.001 0.01 0.1 1 10 100 1000 10000 Repeated doses in Monkey (AN4035 30 mg/kg x 2) Time after first dose injection(day) Drug Conc. (μg/mL) AN4035 Payload Compound Dose C0 (μg/mL) AUC (h*μg/mL) T1/2 (day) AN4035 30 mg/kg, IV 942 60377 3.3 Payload 0.038 5.40 4.3 AN4035 HNSTD has not been reached at 30 mpk Source: AN4035 Late-Breaking Oral Presentation, 2025 AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics.

29 Summary of AN4035 AN4035 is a potential first-in-class CEACAM5-targeting ADC armed with a highly potent pan-RAS(ON) inhibitor payload: • Favorable thermal and plasma stability with desirable pharmacokinetic properties • Strong intracellular payload retention, driving nanomolar to picomolar cytotoxicity in CEACAM5-positive/RAS-addicted cancer cell lines, along with a potent bystander-killing effect • Robust anti-tumor activity with deep regression in CEACAM5-positive/RAS-addicted CDX and PDX models, as monotherapy or in combination • Enhanced target-mediated tumor retention versus payload alone, with significantly improved tumor selectivity over normal tissues • Favorable therapeutic index in cynomolgus monkeys IND submission is on track to be completed by mid-2026