EXHIBIT 2.1
ASSET PURCHASE AGREEMENT
This ASSET PURCHASE AGREEMENT (“Agreement”) is entered into and made effective as of this 10th day of April 2026 (“Effective Date”) by and between VisionWave Holdings, Inc., a Delaware corporation with a place of business at 300 Delaware Ave., Suite 210 #301, Wilmington, DE 19801 (“Purchaser”), and Dream America Marketing Services, Ltda., a Costa Rican company with Persona Jurídica No. 3-102-912534 and a place of business at Davivienda Bldg, 1st fl. Meridiano Business Center. Escazu, SJ 10203. Costa Rica (“Seller”) (each of Seller and Purchaser is defined herein as a “Party”, and collectively referred to as the “Parties”).
WITNESSETH:
WHEREAS, Seller owns certain intellectual property assets related to the technology known as xClibre, as more particularly described in the detailed schedule attached hereto as Exhibit A (the “IP Schedule”), which has been valued by an independent third party at $60 million; and
WHEREAS, Purchaser desires to purchase from Seller, and Seller desires to sell and assign to Purchaser, such intellectual property assets set forth in Exhibit A hereto, on the terms and conditions set forth herein.
NOW, THEREFORE, in consideration of the premises and of the mutual covenants set forth herein, the sufficiency and receipt of which the Parties hereby acknowledge, the Parties, intending to be legally bound, hereby agree as follows:
ARTICLE I
DEFINITIONS
1.1 Capitalized Terms. In addition to those terms defined in the body of this Agreement, the following capitalized terms shall have the meanings set forth below:
(a) “Affiliate” means a legal entity that Controls, is Controlled by, or is under common Control with a Party. Such legal entity shall constitute an Affiliate of the Party only when and for so long as the Control exists.
(b) “Assigned IP” means each of the following, whether or not pending, issued, expired, abandoned or closed: (a) the intellectual property listed on Exhibit A (“Listed IP”), attached hereto and incorporated herein, (b) any and all intellectual property that is part of the same families as the Listed IP, (c) any and all of the inventions, invention disclosures, and discoveries existing as of the Effective Date to the extent disclosed or claimed in subitems (a) and (b) above, and (d) any rights of priority created by such intellectual property under any treaty relating thereto. For clarity, Assigned IP includes all patents, patent applications, trademarks, copyrights, trade secrets, know-how, software, and other proprietary rights related to the xClibre technology.
(c) “Assignment Agreements” means any executed agreements assigning, changing, confirming or correcting ownership (including without limitation original assignment agreements) of any part, portion or all rights in the Assigned IP from the Inventor(s) and/or any prior owner to any prior owner or Seller.
(d) “Bona Fide Owned and Controlled” means for the purpose of a design that ownership and control was not transferred or provided for purposes of providing a license to cover an offering of a third party under the licenses granted herein.
(e) “Change of Control” means (a) any transaction or series of transactions whereby any person or entity directly or indirectly acquires Control of another person or entity; or (b) the consummation (whether directly or indirectly through one or more intermediaries) of a sale or other disposition of all or substantially all of another person’s or entity’s assets in any single transaction or series of related transactions.
(f) “Closing” means the consummation of the transactions contemplated by this Agreement.
(g) “Closing Date” means the date that is no later than five (5) Business Days after the satisfaction or waiver of all conditions set forth in Article VIII, or such other date as the Parties may mutually agree in writing.
(h) “Control” (including the correlative meanings of the terms “Controls”, “Controlled by” and “under common Control with”) means the direct or indirect ownership of more than fifty percent (50%) of an entity, or the possession, directly or indirectly, of the power to direct or cause the direction of the management or policies of a person or an entity, whether through the ability to exercise voting power, by contract or otherwise.
(i) “Disclaimer Issue” means a terminal disclaimer (including under 35 U.S.C. Sec. 253 or 37 CFR 1.321 or the equivalent laws or regulation of any other patent authority) that exists or is or should reasonably be required to be made in a patent or patent application to address a double patenting issue, including such an issue raised in a judicial or administrative proceeding (including any proceeding with the U.S. Patent and Trademark Office or any corresponding foreign patent authority).
(j) “Encumbrance” means with respect to any of the Assigned IP, any mortgage, lien, pledge, charge, Commitment, security interest, express or implied license, Grant, judgment, stipulation, court order or decree, or other restriction regarding transfer or licensing, or any other commitment to a third party which would result in any such Encumbrance whether currently existing or arising in the future.
(k) “Governmental Entity” means any court, administrative agency or commission or other federal, state, county, local or foreign governmental authority, instrumentality, agency commission or subdivision thereof, including but not limited to the U.S. Patent and Trademark Office (“PTO”) and the European Patent Office (“EPO”).
(l) “Grant” means a license, waiver of any rights of enforcement (including but not limited to any covenant not to sue, covenant not to assert, or standstill agreement), release of any claim, or other grant of any right.
(m) “Importation Information” means an electronic file provided by Seller containing all information requested regarding the Assigned IP.
(n) “including” means including without limitation.
(o) “IP Schedule” means the detailed schedule of all Assigned IP to be delivered by Seller to Purchaser prior to Closing.
(p) “or” means “and/or”.
(q) “Inventor” means each of the named inventors of each of the Assigned IP as well as any inventor who should be or should have been named on each of the Assigned IP.
(r) “Nasdaq Shareholder Approval” means the approval of the issuance of the Purchase Shares by the holders of a majority of the votes cast by VWAV’s stockholders in accordance with Nasdaq Listing Rule 5635.
(s) “Patent Documents” means (i) all prosecution files (physical and electronic) and docket reports (capturing a time period no shorter than ninety (90) days following the Effective Date) for all of the Assigned IP in the possession or control of Seller, its counsel or its agents; (ii) all Assignment Agreements; (iii) all documents, records and files in the possession or control of Seller, its counsel or its agents (and including any and all of each Inventor) with respect to (A) the conception and reduction to practice (and diligence in reduction to practice) of the inventions of any of the Assigned IP, (B) the disclosure of, acquisition, prosecution, registration, reissuance, correction, enforcement, defense, and maintenance of the Assigned IP (including without limitation ribbon copies of any letters patent), (C) Seller’s marking activities and program(s) with respect to the Assigned IP, and (D) Seller’s licensing or sales activities with respect to the Assigned IP; and (iv) all other material documentation or information in the possession or control of Seller, its counsel or its agents related to the Assigned IP. Notwithstanding the foregoing, Patent Documents shall exclude any documents or information in clauses (i) - (iv) of this Section which are subject to the doctrines of attorney-client privilege, attorney work-product, joint defense, common interest and/or any other applicable privilege or immunity (collectively, “Common Interest Privilege”) if providing such documents or information to Purchaser cannot be accomplished in a manner that protects such Common Interest Privilege.
2
(t) “Patent Family” means a set comprised of all Patents (a) that are linked or entitled to be linked through one or more claims of benefit or priority pursuant to 35 U.S.C. §§ 120 or 119 (or the equivalent laws or regulation of any other patent authority) or by a terminal disclaimer pursuant to 35 U.S.C. § 253 or 37 CFR § 1.321 (or the equivalent laws or regulation of any other patent authority) or (b) that are, or are entitled to be, foreign counterparts, reissues, divisionals, extensions, continuations or continuations-in-part with respect to any other Patent in such set.
(u) “Patents” means any United States, foreign or international patents and patent applications, patents and patent applications resulting or issuing therefrom, certificates of invention, utility models or any other grants by any Governmental Entity for the protection of inventions, including all non-provisionals, provisional, reissues, divisionals, continuations, continuations-in-part, re-examinations and extensions of any of the foregoing; provided, however, that when the term “Patent” is used in the context of, or to refer to, a particular patent or patent application, or a patent or patent application on a schedule, the term shall mean only that particular patent or patent application, as the case may be.
(v) “Seller Product” means any product associated with the Assigned IP, which as of the Effective Date, meets all of the following: (i) the design of the product is Bona Fide Owned and Controlled by Seller, is sold by the Seller, or in development with the intention by Seller to commercialize, and (ii) in the absence of a license or other authorization, the product would infringe one or more of the Assigned IP.
(w) “Subsidiary” means a legal entity that is Controlled by a Party. Such legal entity shall constitute a Subsidiary of the Party only when and for so long as the Control exists.
(x) “Transfer Documents” means the fully executed transfer documents, in a form approved by Purchaser suitable for filing with the relevant Governmental Authority, in each jurisdiction where the Assigned IP issued from or have been filed, as the case may be, in each case to record the change of ownership of the Assigned IP from Seller to Purchaser. Unless otherwise directed by Purchaser, Transfer Documents for U.S. Assigned IP shall be as provided in the form of Exhibit B (“IP Assignment”).
ARTICLE II
SALE AND TRANSFER OF ASSIGNED IP AND COOPERATION
2.1 Transfer. Effective as of the Effective Date, Seller hereby irrevocably sells, transfers, conveys and assigns, and shall cause its Affiliates to irrevocably sell, transfer, convey and assign, to Purchaser (or its designee, as to any or all of the Assigned IP), and Purchaser hereby acquires from Seller or its Affiliates, all right, title and interest in and to (i) all Assigned IP, including without limitation the right to sue, license and collect and receive all income, royalties, damages, payments due, injunctive relief and any other settlements or remedies (including, without limitation, causes of action and rights to damages and payments for past, present and future infringements, violations or misappropriations) for infringement, violation or misappropriation of any of the Assigned IP; (ii) any and all corresponding rights that, now or after the date hereof, may be secured throughout the world, including rights to renew, reissue, continue or divide any of the Assigned IP; (iii) all Patent Documents; (iv) all Assignment Agreements; and (v) all causes of action (whether known or unknown or whether currently pending, filed, or otherwise) and other enforcement rights under, or on account of, any of the Assigned IP, including, without limitation, all causes of action and rights to damages and payments for past, present and future infringements, violations or misappropriations, and the right to sue therefor under the laws of any jurisdiction worldwide.
2.2 Consideration.
(a) Total Consideration. In consideration for the sale, transfer, conveyance, and assignment of the Assigned IP pursuant to Section 2.1, Purchaser shall pay to Seller aggregate consideration consisting of (i) Seven Million (7,000,000) shares of Purchaser’s common stock, par value $0.01 per share (the “Purchase Shares”), and (ii) a Promissory Note, in the form attached hereto as Exhibit C, in the principal amount of Six Million United States Dollars (US$6,000,000) (the “Note”).
3
(b) Delivery at Closing. At Closing, Purchaser shall:
(i) issue and deliver to Seller Three Million Five Hundred Thousand (3,500,000) of the Purchase Shares (the “Closing Shares”); and
(ii) execute and deliver the Note to Seller.
The Closing Shares shall be issued in book-entry form or, if requested by Seller, in certificated form, and shall bear customary legends reflecting applicable securities laws restrictions.
(c) Nasdaq Shareholder Approval Covenant. Purchaser shall use its commercially reasonable efforts to obtain the Nasdaq Shareholder Approval as promptly as practicable after the Effective Date following the Closing Date. The Contingent Shares shall not be issued until the Nasdaq Shareholder Approval has been obtained and all other applicable Nasdaq and SEC requirements have been satisfied.
(d) Post-Closing Assignment to Subsidiary. Within thirty (30) days following the Closing Date (or such later date as mutually agreed), Purchaser shall cause the Assigned IP to be assigned, transferred, and conveyed to a newly formed wholly-owned subsidiary corporation incorporated under the laws of the State of Nevada named “xClibre Inc.” or such other name as is reasonably acceptable to Purchaser and available under Nevada law (the “Subsidiary” or “xClibre”). Purchaser shall provide Seller with evidence reasonably satisfactory to Seller of such assignment (including recorded assignments with applicable intellectual property offices) and of xClibre’s status as a wholly-owned subsidiary of Purchaser.
(e) Security Interest in Pledged Equity. Concurrently with the assignment described in Section 2.2(d), Purchaser shall cause xClibre to issue to Purchaser, and Purchaser shall pledge and grant to Seller a first-priority perfected security interest in, sixty percent (60%) of the issued and outstanding equity interests of xClibre (the “Pledged Equity”). Such pledge shall be effected pursuant to a Pledge and Security Agreement in a form agreeable to the Parties (the “Pledge Agreement”), which shall include customary provisions for a transaction of this type, including, without limitation, delivery of stock certificates (if certificated), irrevocable stock powers executed in blank, UCC-1 financing statements, and control agreements if applicable. Seller’s security interest in the Pledged Equity shall secure Purchaser’s and xClibre’s obligations under this Agreement, the Note, and the Contingent Shares issuance.
(f) Contingent Shares and results of Proof Of Concept (“POC”) being conducted at C.M. Facility. The remaining Three Million Five Hundred Thousand (3,500,000) Purchase Shares (the “Contingent Shares”) shall be issued and delivered to Seller promptly upon receipt of Purchaser’s final POC results and the Nasdaq Shareholder Approval. Purchaser shall use its commercially reasonable efforts to obtain the POC Approval from C.M. management as soon as practicable following the Closing), but in no event later than nine (9) months after the Closing Date.
(g) Remedy for Failure to Obtain POC Approval. If the POC approval is not obtained within nine (9) months after the Closing Date, then:
(i) Purchaser shall promptly cause the Pledged Equity to be transferred to Seller (or its designee) free and clear of all Encumbrances (other than securities laws restrictions), including by delivering stock certificates, executed stock powers, and termination of any financing statements;
(ii) upon such transfer, Seller’s security interest shall be automatically released and the Pledge Agreement terminated; and
(iii) Seller shall retain full ownership of the Closing Shares previously issued and the Note (including all rights to payment thereunder), without any obligation to return, cancel, or forfeit the same.
For the avoidance of doubt, no alternative consideration shall be negotiated or provided in lieu of the Contingent Shares in such event.
4
(h) Further Assurances. Each Party shall execute and deliver such additional documents and take such further actions as may be reasonably requested by the other Party to effectuate the provisions of this Section 2.2, including filings necessary to perfect or release the security interest in the Pledged Equity.
2.3 Cooperation. Seller agrees to cooperate fully with Purchaser and its designees, at Purchaser’s expense, in connection with the prosecution, maintenance, enforcement, and defense of the Assigned IP, including but not limited to executing any additional documents necessary to perfect Purchaser’s title thereto, providing testimony, and assist in any legal proceedings related to the Assigned IP.
2.4 Transfer Documents and Patent Documents. Within ten (10) business days after the Effective Date, Seller shall deliver to Purchaser the Transfer Documents and all Patent Documents.
2.5 Importation Information. Within ten (10) business days after the Effective Date, Seller shall deliver to Purchaser the Importation Information.
2.6 Tax Matters. Purchaser shall be entitled to deduct and withhold from any consideration payable hereunder any amounts required to be deducted or withheld under applicable tax law (including Costa Rican and U.S. tax law). Any withheld amounts shall be treated as delivered to Seller. Seller shall provide Purchaser with all forms and certifications necessary to minimize or eliminate withholding.
ARTICLE III
REPRESENTATIONS AND WARRANTIES OF SELLER
Seller represents and warrants to Purchaser as follows:
3.1 Organization and Authority. Seller is a sociedad de responsabilidad limitada duly organized, validly existing, and in good standing under the laws of the Republic of Costa Rica, and has full corporate power and authority to own the Assigned IP, to enter into this Agreement, and to consummate the transactions contemplated hereby, including the assignment and transfer of the Assigned IP to Purchaser. The execution, delivery, and performance of this Agreement by Seller have been duly authorized by all necessary corporate action.
3.2 Title to Assigned IP. Seller is the sole and exclusive owner of the entire right, title, and interest in and to the Assigned IP (including all registered patents, patent applications, trademarks, trademark applications, copyrights, and copyright applications listed in the Schedules), free and clear of all Encumbrances (except as disclosed in the Schedules). The Assigned IP constitutes all intellectual property rights used in or necessary for the conduct of the business as currently conducted. Upon consummation of the transactions contemplated hereby, Purchaser will acquire good, valid, and marketable title to the Assigned IP, free and clear of all Encumbrances.
3.3 No Conflicts; Consents. Neither the execution, delivery, nor performance of this Agreement by Seller, nor the consummation of the transactions contemplated hereby (including the assignment of the Assigned IP), will (a) conflict with, violate, or result in a breach of Seller’s organizational documents, (b) conflict with, violate, or result in a breach or default under any agreement, instrument, license, permit, or understanding (oral or written) to which Seller is a party or by which Seller or the Assigned IP is bound, (c) violate any applicable law or order, or (d) result in the creation of any Encumbrance on the Assigned IP. No consent, approval, or authorization of any third party or governmental authority is required for the assignment of the Assigned IP to Purchaser.
3.4 Validity and Enforceability. Each item of registered Assigned IP is subsisting, in full force and effect, valid, and enforceable. To Seller’s Knowledge, the unregistered Assigned IP (including know-how and trade secrets) is valid and enforceable. There are no pending or, to Seller’s Knowledge, threatened actions, suits, proceedings, oppositions, interferences, reexaminations, reissues, invalidity challenges, or other challenges to the validity, enforceability, ownership, or registrability of any Assigned IP. No item of Assigned IP has been abandoned, canceled, or adjudged invalid or unenforceable in whole or in part.
5
3.5 No Infringement by Third Parties; No Litigation. To Seller’s Knowledge, no third party is infringing, misappropriating, or otherwise violating any Assigned IP. There are no pending or, to Seller’s Knowledge, threatened claims, actions, or proceedings by Seller alleging infringement of the Assigned IP, and Seller has not sent any notice of infringement to any third party. There are no pending or, to Seller’s Knowledge, threatened claims, actions, suits, or proceedings against Seller alleging that the Assigned IP or the use thereof infringes, misappropriates, or violates the intellectual property rights of any third party.
3.6 No Infringement by Assigned IP. The Assigned IP does not infringe, misappropriate, or otherwise violate, and the exploitation or use of the Assigned IP has not infringed, misappropriated, or violated, the intellectual property rights of any third party.
3.7 Maintenance. All necessary registration, maintenance, renewal fees, annuities, and other payments due with respect to the Assigned IP have been timely paid in full, all required documents and certificates have been filed with the relevant authorities, and the Assigned IP is subsisting and in good standing. No renewal or maintenance deadlines fall due within 120 days following the Closing Date (except as disclosed in the Schedules).
3.8 No Terminal Disclaimer or Disclaimer Issues. No item of Assigned IP is subject to any terminal disclaimer, statutory disclaimer, or other disclaimer that limits its term, scope, enforceability, or value. There are no other “Disclaimer Issues” (including any filings or obligations that could shorten the term or impair enforceability) with respect to the Assigned IP.
3.9 Assignment Agreements and Chain of Title. Seller has obtained valid, enforceable, and duly executed written assignments from all inventors, authors, employees, consultants, and other persons who contributed to the creation or development of the Assigned IP, sufficient to vest full and exclusive ownership in Seller (or its predecessors). All such Assignment Agreements are listed in the Schedules and copies have been provided to Purchaser. There are no gaps or defects in the chain of title to the Assigned IP.
3.10 No Prior Grants or Obligations. Seller has not granted, and there are no outstanding, any licenses, options, covenants not to sue, or other rights or interests (exclusive or non-exclusive) in or to the Assigned IP to any third party. The Assigned IP is not subject to any co-ownership, joint development, or government funding obligations that would restrict Purchaser’s exclusive use or ownership post-Closing.
3.11 Accredited Investor Status. Seller represents and warrants that it is an “accredited investor” as defined in Rule 501(a) of Regulation D promulgated under the Securities Act of 1933, as amended (the “Securities Act”). Specifically, Seller meets one or more of the following criteria (as indicated in the disclosure schedule delivered concurrently herewith):
(a) Seller is an entity with total assets in excess of US$7,000,000 that was not formed for the specific purpose of acquiring the securities or other consideration issued by Purchaser in connection with this Agreement;
(b) Seller is an entity in which all of the equity owners are accredited investors under Rule 501(a); or
(c) Seller otherwise qualifies as an accredited investor under one of the categories set forth in Rule 501(a)(1) through (8) of Regulation D.
Seller has such knowledge, sophistication, and experience in business and financial matters that it is capable of evaluating the merits and risks of receiving the Promissory Note (and any other securities issued as consideration hereunder) as partial consideration for the Assigned IP. Seller is able to bear the economic risk of such investment and has been furnished with or has had access to all information it considers necessary to make an informed investment decision with respect thereto.
3.12 Full Disclosure. Seller has disclosed to Purchaser all material information regarding the Assigned IP, including all prior art known to Seller that could affect patentability, all correspondence with patent/trademark offices material to validity or scope, and all third-party claims or notices received relating to the Assigned IP. There are no facts or circumstances known to Seller that could materially adversely affect the value, validity, enforceability, or Purchaser’s use of the Assigned IP.
6
ARTICLE IV
REPRESENTATIONS AND WARRANTIES OF PURCHASER
Purchaser represents and warrants to Seller as follows:
4.1 Organization and Authority. Purchaser is a corporation duly organized, validly existing, and in good standing under the laws of the State of Delaware. Purchaser has full corporate power and authority to execute and deliver this Agreement, the Purchase Shares and the Note, to consummate the transactions contemplated hereby (including the acquisition of the Assigned IP and the issuance of, the Purchase Shares and the Note as partial consideration), and to perform its obligations hereunder and thereunder. The execution, delivery, and performance of this Agreement, the Purchase Shares and the Note by Purchaser have been duly authorized by all necessary corporate action on the part of Purchaser, and no other corporate proceedings are necessary for such authorization.
4.2 No Conflicts; Consents. Neither the execution, delivery, nor performance of this Agreement by Purchaser, nor the consummation of the transactions contemplated hereby or thereby (including the issuance of , the Purchase Shares and the Note), will (a) conflict with, violate, or result in a breach of Purchaser’s certificate of incorporation or bylaws (or equivalent organizational documents), (b) conflict with, violate, result in a breach or default under, or give rise to any right of termination, cancellation, or acceleration of any obligation under any agreement, instrument, license, permit, or understanding (oral or written) to which Purchaser is a party or by which Purchaser or its assets are bound, (c) violate any applicable law, rule, regulation, judgment, order, or decree of any governmental authority, or (d) result in the creation of any Encumbrance upon any assets of Purchaser. No consent, approval, waiver, or authorization of, or filing with, any third party or governmental authority is required in connection with the execution, delivery, or performance by Purchaser of this Agreement or the consummation of the transactions contemplated hereby or thereby.
4.3 Issuance of Note and Consideration Securities. The Note, when issued and delivered as provided herein, will constitute a valid and binding obligation of Purchaser, enforceable against Purchaser in accordance with its terms (subject to applicable bankruptcy, insolvency, reorganization, moratorium, or similar laws affecting creditors’ rights generally and general principles of equity). The Purchase Shares, when issued and delivered in accordance with this Agreement, will be duly authorized, validly issued, fully paid, and non-assessable, and will be issued in compliance with all applicable federal and state securities laws, free and clear of any Encumbrances (other than those arising under applicable securities laws or as set forth in this Agreement).
4.4 Securities Compliance. The Purchase Shares, when issued, will be duly authorized, validly issued, fully paid and non-assessable and issued in compliance with all applicable federal and state securities laws. The issuance does not and will not require any registration under the Securities Act or any state blue-sky laws except pursuant to any registration rights granted hereunder. Purchaser is not in violation of any Nasdaq listing requirements that would prevent the issuance or listing of the Purchase Shares.
4.5 No Material Adverse Effect. Since December 31, 2025, there has been no Material Adverse Effect on Purchaser’s business, financial condition or results of operations that would reasonably be expected to materially impair Purchaser’s ability to consummate the transactions contemplated hereby.
ARTICLE V
COVENANTS
5.1 Further Assurances; Cooperation.
(a) From and after the Closing, each Party shall, at the reasonable request of the other Party and without additional consideration, execute, acknowledge, and deliver (or cause to be executed, acknowledged, and delivered) such additional instruments, documents, and assignments, and take such further actions as may be reasonably required to carry out the intent and purposes of this Agreement, including to vest in Purchaser full right, title, and interest in and to the Assigned IP worldwide, to record the assignment of the Assigned IP with relevant governmental authorities (including patent, trademark, and copyright offices), and to perfect, protect, or enforce the Assigned IP.
7
(b) Seller shall promptly provide Purchaser with all original files, records, prosecution histories, correspondence, and other materials relating to the Assigned IP in Seller’s possession or control.
(c) For a period of 12 months following Closing (or longer if reasonably required), Seller shall provide reasonable transition assistance to Purchaser regarding the Assigned IP, including responding to inquiries about prosecution history, prior art, or inventorship, at no additional cost (except reimbursement of reasonable out-of-pocket expenses).
5.2 Confidentiality.
Each Party shall treat as confidential and not disclose to any third party (except as required by law or with the prior written consent of the other Party) the terms of this Agreement, the existence of the transactions contemplated hereby, and any non-public information received from the other Party. This obligation shall survive Closing for a period of three (3) years (or indefinitely with respect to trade secrets). The receiving Party may disclose such information to its affiliates, advisors, financiers, or representatives who need to know and are bound by equivalent confidentiality obligations.
5.3 Public Announcements.
No Party shall issue or cause the publication of any press release or other public announcement or communication with respect to this Agreement or the transactions contemplated hereby without the prior written consent of the other Party (which consent shall not be unreasonably withheld, conditioned, or delayed), except as may be required by applicable law, regulation, or Nasdaq listing rules (in which case the disclosing Party shall provide the other Party with a reasonable opportunity to review and comment on such disclosure in advance). The Parties shall cooperate in good faith to prepare and agree upon any joint press release regarding the transaction if mutually desired.
5.4 Tax Cooperation.
Each Party shall cooperate fully with the other in connection with the filing of tax returns and any audit or proceeding relating to the Assigned IP or the transactions contemplated hereby, including providing necessary information and documents.
ARTICLE VI
INDEMNIFICATION
Seller shall defend, indemnify, and hold harmless Purchaser, its affiliates, and their respective officers, directors, employees, agents, successors, and permitted assigns (collectively, the “Purchaser Indemnitees”) from and against any and all losses, damages, liabilities, obligations, claims, demands, actions, suits, proceedings, judgments, settlements, interest, awards, penalties, fines, costs, and expenses (including reasonable attorneys’ fees, expert fees, and court costs) (collectively, “Losses”) incurred or suffered by any Purchaser Indemnitee arising out of, relating to, or resulting from:
(a) any breach or inaccuracy of any representation or warranty made by Seller in this Agreement (including the Schedules hereto);
(b) any breach or non-performance of any covenant or agreement of Seller contained in this Agreement;
(c) any third-party claim alleging infringement, misappropriation, or violation of intellectual property rights based on the Assigned IP (regardless of knowledge qualifiers in representations);
(d) any taxes, duties, or governmental charges imposed on Seller or relating to the Assigned IP or the business prior to Closing; or
(e) any fraud, intentional misrepresentation, or willful breach by Seller.
6.2 Indemnification by Purchaser.
8
Purchaser shall defend, indemnify, and hold harmless Seller, its affiliates, and their respective officers, directors, employees, agents, successors, and permitted assigns (collectively, the “Seller Indemnitees”) from and against any and all Losses incurred or suffered by any Seller Indemnitee arising out of, relating to, or resulting from:
(a) any breach or inaccuracy of any representation or warranty made by Purchaser in this Agreement;
(b) any breach or non-performance of any covenant or agreement of Purchaser contained in this Agreement (including payment obligations under the Promissory Note); or
(c) any fraud, intentional misrepresentation, or willful breach by Purchaser.
6.3 Procedures for Indemnification.
(a) Notice. Promptly after receipt by an Indemnitee of notice of any third-party claim or the commencement of any action or proceeding for which indemnification may be sought, such Indemnitee shall notify the Indemnifying Party in writing; provided, however, that failure to give timely notice shall not relieve the Indemnifying Party of its obligations except to the extent it is actually prejudiced thereby.
(b) Defense. The Indemnifying Party shall have the right to assume and control the defense of any third-party claim with counsel reasonably acceptable to the Indemnitee, at the Indemnifying Party’s expense. The Indemnitee may participate in (but not control) the defense at its own expense. The Indemnifying Party shall not settle any claim without the Indemnitee’s prior written consent (not to be unreasonably withheld) if such settlement imposes liability on, or requires admission from, the Indemnitee.
(c) Direct Claims. For non-third-party claims, the Indemnitee shall provide written notice specifying the basis and amount (if known), and the Indemnifying Party shall respond within [30] days.
6.4 Survival.
The representations and warranties in this Agreement shall survive Closing for a period of 18 months, except:
(a) Fundamental Representations (Sections 3.1 Organization and Authority, 3.2 Title to Assigned IP, 4.1 Organization and Authority) shall survive for 5 years (or the applicable statute of limitations);
(b) IP-specific representations (Sections 3.4–3.10) shall survive for 3 years; and
(c) Covenants shall survive indefinitely or until fully performed.
Claims for fraud or intentional misrepresentation shall survive indefinitely.
6.5 Limitations on Indemnification.
(a) Basket. No indemnification shall be payable by Seller for breaches of representations and warranties (other than Fundamental Representations or fraud) until aggregate Losses exceed US$100,000 (the “Basket”), after which the Indemnifying Party shall be liable for all Losses (including those comprising the Basket).
(b) Cap. The aggregate liability of Seller for indemnification claims (other than for Fundamental Representations, fraud, or covenants) shall not exceed 100% of the total Purchase Price (the “Cap”). Liability for Fundamental Representations and covenants shall be capped at the total Purchase Price. There shall be no cap for fraud or intentional misrepresentation.
9
(c) Total Consideration. For purposes of calculating the Cap, the “Purchase Price” shall mean the principal amount of the Note ($6,000,000) plus the fair market value of any Purchase Shares or other consideration delivered at Closing (valued as of the Closing Date using the closing price of Purchaser’s common stock on Nasdaq, if applicable).
(d) Other Limitations. Losses shall be calculated net of insurance recoveries or tax benefits actually received. No punitive damages except if awarded to a third party.
6.6 Exclusive Remedy.
Except for claims based on fraud or equitable remedies (e.g., specific performance), the indemnification rights in this Article VI shall be the sole and exclusive remedy for any Losses arising from this Agreement.
ARTICLE VII
MISCELLANEOUS
7.1 Confidentiality. The Parties shall maintain as strictly confidential this Agreement and any proprietary information disclosed under, or as a result of or during the negotiation of, this Agreement, which obligation shall survive the consummation of the transactions contemplated herein, and shall only use such information for the purpose of performing under and/or enforcing this Agreement or the Assigned IP, except that each Party, or its Affiliates, may disclose or use this Agreement or any such proprietary information as follows:
(a) As reasonably necessary to prosecute or enforce the Assigned IP;
(b) as reasonably necessary for Purchaser to record or otherwise perfect Purchaser’s interest in the Assigned IP;
(c) to the extent required by law;
(d) to the extent such information is public information, except as a result of the breach of this Section 7.1;
(e) as is required by a court or an arbitral order which has been precipitated by a third party request; provided, that the entity making such disclosure or use shall seek appropriate confidentiality protections (e.g., having such disclosures covered by a protective order or other comparable protections) prior to making such disclosure or use;
(f) to satisfy SEC, Nasdaq or other statutory, regulatory, taxation, or administrative requirements;
(g) in a legal proceeding between the Parties or their Affiliates;
(h) to a potential acquirer, in connection with a potential acquisition of all or any material part of any business of such Party; or
(i) in confidence, to its accountants, bankers, attorneys, or their Affiliates.
Notwithstanding the foregoing, the Parties acknowledge that Purchaser or its Affiliates shall have the right, at its sole discretion, to publish and distribute a press release or a Current Report on Form 8-K or Periodic Report filed pursuant to the Securities Exchange Act of 1934, as amended, announcing the execution of this Agreement, describing the material terms hereof and filing such Agreement as an exhibit thereto.
7.2 Expenses. Except as otherwise provided in this Agreement, each Party will pay all fees and expenses incurred by it in connection with this Agreement and the transactions contemplated hereby.
10
7.3 Governing Law/Venue. This Agreement is governed by the laws of the State of California, excluding its conflict-of-laws principles. The state and federal courts in the State of California shall have exclusive jurisdiction over any claim, suit or proceeding (each, a “Proceeding”) related to this Agreement (including without limitation the breach or threatened breach thereof), and each Party irrevocably (a) consents to the jurisdiction of such courts for any Proceeding, (b) consents to service of process in any Proceeding in such courts by globally recognized overnight courier service at the address set forth above, as well as other means of service permitted by law; and (c) waives any objections on the grounds of venue, residence, domicile or inconvenient forum to any Proceeding brought in such courts.
7.4 Waivers. The failure of any Party to insist upon the performance of any of the terms or conditions of this Agreement or to exercise any right hereunder, shall not be construed as a waiver or relinquishment of any such right, term or condition. No waiver by any Party of any provision of this Agreement or any default, misrepresentation, or breach of warranty or covenant hereunder shall be valid unless the same shall be in writing and signed by the Party making such waiver.
7.5 Severability. The provisions of this Agreement shall be severable, and if any of them are held invalid or unenforceable, then that provision shall be construed to the maximum extent permitted by law. The invalidity or unenforceability of one provision shall not necessarily affect any other.
7.6 Notices. All notices or other communications required or permitted under this Agreement shall be in writing and shall be delivered by personal delivery, registered mail, return receipt requested, or a qualified overnight delivery service addressed as set forth above. All such notices shall be deemed delivered at the time of delivery, except a facsimile shall be deemed delivered at the time of electronic confirmation of delivery.
7.7 Asset Purchase. The transaction contemplated under this Agreement is strictly an asset purchase, and Purchaser is not taking any assignment of any debt, obligation, or other Encumbrance on any of the Assigned IP.
7.8 Entire Agreement/Amendment. This Agreement contains the complete and final agreement between the Parties, and supersedes all previous understandings, relating to the subject matter hereof whether oral or written. This Agreement may only be modified by a written agreement signed by duly authorized representatives of the Parties.
7.9 No Assignment. Except for Purchaser’s right to assign or delegate this Agreement and its rights and duties as set forth herein to a present or future Affiliate, neither this Agreement nor any rights or duties under this Agreement may be assigned or delegated, in whole or in part, by either Party without the prior written consent of the non-assigning Party, which consent may be withheld by the non-assigning Party for any or no reason; provided, however, Seller may assign the Purchaser Shares and the Note to its shareholders. Any attempted assignment and/or delegation in breach of this Section 7.9 will be null and void. The Parties understand and agree that a Change of Control event is considered an assignment for the purposes of this Section 7.9.
7.10 Survival. Notwithstanding anything in this Agreement to the contrary except as set forth in Section 6.1, all representations, warranties, obligations, responsibilities, terms or conditions which by a fair reading of their nature are intended to survive shall be deemed to survive.
7.11 Limitation on Consequential Damages. EXCEPT IN THE CASE OF FRAUD BY SELLER, NEITHER PARTY SHALL BE LIABLE TO THE OTHER FOR LOSS OF PROFITS, OR ANY OTHER INDIRECT OR SPECIAL, CONSEQUENTIAL, PUNITIVE OR INCIDENTAL DAMAGES, HOWEVER CAUSED, EVEN IF ADVISED OF THE POSSIBILITY OF SUCH DAMAGE. THE PARTIES ACKNOWLEDGE THAT THESE LIMITATIONS ON POTENTIAL LIABILITIES WERE AN ESSENTIAL ELEMENT IN SETTING CONSIDERATION UNDER THIS AGREEMENT.
7.12 Arm’s Length Negotiations. Each Party herein expressly represents and warrants to all other Parties hereto that (a) before executing this Agreement, said Party has fully informed itself of the terms, contents, conditions and effects of this Agreement; (b) said Party has relied solely and completely upon its own judgment in executing this Agreement; (c) said Party has had the opportunity to seek and has obtained the advice of its own legal, tax and business advisors before executing this Agreement; (d) said Party has acted voluntarily and of its own free will in executing this Agreement; and (e) this Agreement is the result of arm’s length negotiations conducted by and among the Parties and their respective counsel.
11
7.13 Amendments. Every right and remedy provided herein shall be cumulative with every other right and remedy, whether conferred herein, at law, or in equity, and may be enforced concurrently herewith, and no waiver by any Party of the performance of any obligation by the other shall be construed as a waiver of the same or any other default then, theretofore, or thereafter occurring or existing. This Agreement may be amended by a writing signed by all Parties hereto.
7.14 Remedies. The remedies provided in this Agreement shall be cumulative and in addition to all other remedies available under this Agreement, at law or in equity (including a decree of specific performance and/or other injunctive relief).
7.15 Counterparts, Effect of Facsimile, Emailed and Photocopied Signatures. This Agreement and any signed agreement or instrument entered into in connection with this Agreement, and any amendments hereto or thereto, may be executed in one or more counterparts, all of which shall constitute one and the same instrument. Any such counterpart, to the extent delivered by means of a facsimile machine or by .pdf, .tif, .gif, .jpeg or similar attachment to electronic mail (any such delivery, an “Electronic Delivery”) shall be treated in all manner and respects as an original executed counterpart and shall be considered to have the same binding legal effect as if it were the original signed version thereof delivered in person. At the request of any Party, each other Party shall re execute the original form of this Agreement and deliver such form to all other Parties. No Party shall raise the use of Electronic Delivery to deliver a signature or the fact that any signature or agreement or instrument was transmitted or communicated through the use of Electronic Delivery as a defense to the formation of a contract, and each such Party forever waives any such defense, except to the extent such defense relates to lack of authenticity.
7.16 Assignment to Wholly-Owned Subsidiary of Purchaser. The Parties agree and confirm, and the Seller acknowledges and agrees, that the Purchaser may assign this Agreement and its rights hereunder to a wholly-owned subsidiary, and Seller agrees to execute whatever documents and materials as the Purchaser may reasonably request, to effect such transfer and assignment. Such subsidiary shall be bound by the terms of this Agreement relating to the Assigned IP as if a party hereto upon such assignment, and Purchaser shall take whatever action as may be necessary for such subsidiary to be bound thereby, and comply with such applicable terms.
7.17 Public Filings. Purchaser shall be entitled, without prior consent of Seller, to file this Agreement (or a form of it) and a press release describing the material terms hereof as exhibits to a Current Report on Form 8-K and in any other filings required by the Securities Exchange Act of 1934, as amended, or Nasdaq rules.
ARTICLE VIII
CONDITIONS TO CLOSING
8.1 Conditions to Obligations of Purchaser. The obligation of Purchaser to consummate the transactions contemplated hereby is subject to the satisfaction (or waiver by Purchaser in its sole discretion) of the following conditions on or prior to the Closing Date:
(a) All representations and warranties of Seller contained in this Agreement (including the Schedules and Exhibits) shall be true and correct in all material respects (or, in the case of any representation or warranty qualified by materiality, “Material Adverse Effect,” or similar qualification, in all respects) as of the Effective Date and as of the Closing Date (except for representations and warranties expressly made as of a specific date, which shall be true and correct as of such date);
(b) Seller shall have delivered to Purchaser the complete and accurate IP Schedule, all Patent Documents, all Transfer Documents (in recordable form satisfactory to Purchaser), the Importation Information, and all other deliverables required under Section 2.4 and Section 2.5, each in form and substance satisfactory to Purchaser in its reasonable discretion;
(c) No Material Adverse Effect with respect to the Assigned IP or the business related thereto shall have occurred since the Effective Date;
(d) Purchaser shall have received a favorable legal opinion from counsel to Seller qualified to practice in the Republic of Costa Rica, in form and substance satisfactory to Purchaser and its U.S. counsel, covering due organization, valid existence, good standing, corporate power and authority, due authorization and execution of this Agreement and the Transfer Documents, and valid transfer of good, valid, and marketable title to the Assigned IP free and clear of all Encumbrances;
12
(e) The Nasdaq Shareholder Approval (if required under Nasdaq Listing Rule 5635 or any other applicable rule) shall have been obtained for the issuance of the Purchase Shares (including the Contingent Shares) in accordance with applicable law and Nasdaq requirements;
(f) All consents, approvals, filings, and notices required by applicable law or any Governmental Entity (including any required filings with the U.S. Patent and Trademark Office, Costa Rican intellectual property registries, or any other foreign patent office) shall have been obtained or made and shall be in full force and effect;
(g) There shall be no action, suit, proceeding, or investigation pending or threatened by any Governmental Entity or third party that seeks to restrain, enjoin, or prohibit the consummation of the transactions contemplated hereby or that would reasonably be expected to result in a Material Adverse Effect on the Assigned IP; and
(h) Seller shall have delivered such other customary closing deliverables as Purchaser may reasonably request, including a certificate of an officer of Seller certifying as to the satisfaction of the foregoing conditions, incumbency, and resolutions authorizing the transactions.
8.2 Conditions to Obligations of Seller. The obligation of Seller to consummate the transactions contemplated hereby is subject to the satisfaction (or waiver by Seller) of the following conditions on or prior to the Closing Date, subject in all respects to Purchaser’s prior reasonable approval:
(a) All representations and warranties of Purchaser contained in this Agreement shall be true and correct in all material respects (or, in the case of any representation or warranty qualified by materiality, in all respects) as of the Effective Date and as of the Closing Date (except for representations and warranties expressly made as of a specific date, which shall be true and correct as of such date);
(b) Purchaser shall have performed and complied in all material respects with all covenants, agreements, and obligations required to be performed or complied with by it under this Agreement at or prior to the Closing;
(c) Purchaser shall have delivered (or caused to be delivered) to Seller (i) the Closing Shares, duly authorized, validly issued, fully paid, and non-assessable, and (ii) the executed Promissory Note in the form of Exhibit C;
(d) There shall be no order, injunction, decree, or other legal restraint issued by any Governmental Entity of competent jurisdiction that restrains or prohibits the consummation of the transactions contemplated hereby; and
(e) Purchaser shall have delivered a certificate of an officer of Purchaser certifying as to the satisfaction of the foregoing conditions, incumbency, and resolutions authorizing the transactions.
8.3 Mutual Conditions to Closing. The respective obligations of each Party to consummate the transactions contemplated hereby are subject to the satisfaction (or waiver by both Parties) of the following conditions:
(a) No statute, rule, regulation, order, decree, or injunction shall have been enacted, entered, promulgated, or enforced by any Governmental Entity that prohibits or makes illegal the consummation of the transactions contemplated hereby; and
(b) All applicable waiting periods (and any extensions thereof) under any applicable antitrust, competition, or similar laws shall have expired or been terminated (if any such laws are applicable).
8.4 Frustration of Closing Conditions. Neither Party may rely on the failure of any condition set forth in this Article VIII to be satisfied if such failure was caused by such Party’s failure to act in good faith or to use its commercially reasonable efforts to consummate the transactions contemplated hereby as required by this Agreement.
[Remainder of page left intentionally blank. Signature page follows.]
13
IN WITNESS WHEREOF, the Parties have executed this Agreement by their duly authorized representatives as of the dates below to be effective as of the Effective Date.
| PURCHASER: | ||
| VISIONWAVE HOLDINGS, INC. | ||
| By: | ||
| Name: | Douglas Davis | |
| Title: | CEO & Executive Chairman | |
| SELLER: | ||
| DREAM AMERICA MARKETING SERVICES, LTDA. | ||
| By: | ||
| Name: | Cynthia Elena Mora | |
| Title: | Manager & Director | |
14
EXHIBIT A
All intellectual property rights owned by Seller relating to the xClibre technology, including but not limited to:
- All patents and patent applications related to xClibre;
- All trademarks, service marks, and trade names related to xClibre;
- All copyrights in software, documentation, and other materials related to xClibre;
- All trade secrets, discoveries, know-how, inventions, processes, improvements, enhancements, derivative works and proprietary information related to xClibre;
- all software, source code, object code, algorithms, heuristics, and mathematical methodologies;
- all SDKs, APIs, documentation, benchmarks, test results, and development roadmaps;
- all worldwide rights to use, modify, commercialize, sublicense, distribute, and exploit the foregoing in any field of use;
- Any other intellectual property rights, whether registered or unregistered, pertaining to the xClibre technology.
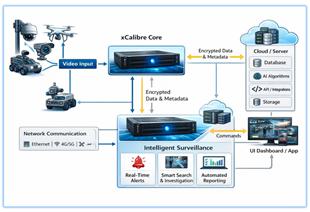
xCalibre Security is an AI video security and video-as-a-sensor platform intended to convert existing IP cameras into continuously-learning “AI agents,” delivering real-time alerts, rapid forensic search, and operational dashboards via an edge-first architecture (Core appliances) plus cloud management services. For VisionWave, the strategic value is not to become another VMS (Video Management System) vendor. The value is to add a mature, security-hardened visual perception layer that can operate as (a) a redundancy channel to RF-derived sensing (Vision RF) and (b) an event-to-action pipeline that accelerates detection, prioritization, response, and auditability across multi-domain autonomous systems (interceptor drones, Argus, UGVs, and fixed-site deployments). Preliminary recommendation: proceed with a verify-first, integrate-by-interface approach. Run a tightly-scoped technical and operational evaluation focused on accuracy and false-alert performance in harsh conditions, end-to-end latency, offline operation, cybersecurity posture, and API/SDK integration into VisionWave C2 and autonomy pipelines. Conditional on measured outcomes, consider a product partnership/OEM embedding path or a deeper strategic relationship
xClibre System – Image/Video Security:

15
EXHIBIT B
IP ASSIGNMENT
FOR VALUE RECEIVED, DREAM AMERICA MARKETING SERVICES, LTDA. (the “Assignor”), hereby sells, assigns, transfers, and sets over unto VisionWave Holdings, Inc. (the “Assignee”), its successors and assigns, the entire right, title, and interest in and to the Assigned IP as defined in the Asset Purchase Agreement dated April 10, 2026, between Assignor and Assignee, including the right to sue for past infringement and to collect damages therefor.
16
IN WITNESS WHEREOF, the Assignor has executed this Assignment as of the 10 day of April, 2026.
DREAM AMERICA MARKETING SERVICES, LTDA.
| By: | ||
| Name: | Cynthia Elena Mora | |
| Title: | Manager & Director |
State of ___________ )
County of _________ ) ss.
On this ____ day of ___________, 2026, before me personally appeared Cynthia Elena Mora, to me known to be the person described in and who executed the foregoing instrument, and acknowledged that he executed the same as his free act and deed.
________________________________
Notary Public
My Commission Expires: __________
17
Exhibit C
PROMISSORY NOTE
Principal Amount: US$6,000,000.00
Date: April 10, 2026
FOR VALUE RECEIVED, including as partial consideration for the purchase of certain assets pursuant to that certain Asset Purchase Agreement dated as of even date herewith (the “Asset Purchase Agreement”), the undersigned, VisionWave Holdings, Inc. (the “Maker” or “Payor”), hereby unconditionally promises to pay to the order of DREAM AMERICA MARKETING SERVICES, LTDA. (the “Holder” or “Payee”), or its assigns, the principal sum of Six Million United States Dollars (US$6,000,000), together with interest thereon as provided herein.
| 1. | Interest. Interest shall accrue on the outstanding principal balance from the date hereof at the rate of twelve percent (12%) per annum, calculated on the basis of a 365-day year and the actual number of days elapsed (simple interest); provided, however, upon the payment of every $1,000,000 in principal the interest rate shall be decreased by 1%. |
| 2. | Payment. The entire outstanding principal amount, together with all accrued and unpaid interest thereon, shall be due and payable in full on April 10, 2027 (the “Maturity Date”). Payment shall be made in lawful money of the United States of America by wire transfer of immediately available funds to an account designated in writing by the Holder. |
| 3. | Prepayment. The Maker may prepay this Note in whole or in part at any time without premium or penalty. Any partial prepayment shall be applied first to accrued and unpaid interest and then to principal. |
| 4. | Place of Payment. All payments hereunder shall be made to the Holder at such address as the Holder may designate in writing from time to time. |
| 5. | Default. If the Maker fails to pay any amount due hereunder when due, or if any material event of default occurs under this Note or the Asset Purchase Agreement, the entire unpaid principal balance and all accrued interest shall, at the option of the Holder, become immediately due and payable without further notice or demand. |
| 6. | Costs of Collection. The Maker agrees to pay all costs and expenses of collection, including reasonable attorneys’ fees, incurred by the Holder in enforcing this Note. |
| 7. | Waivers. The Maker waives presentment, demand, protest, notice of dishonor, and all other notices or demands in connection with this Note (except as expressly required under the Asset Purchase Agreement). |
| 8. | Governing Law. This Note shall be governed by and construed in accordance with the laws of the California, United States of America, without regard to conflict of laws principles. |
| 9. | Severability. If any provision of this Note is held invalid or unenforceable, the remaining provisions shall continue in full force and effect. |
| 10. | Maximum Lawful Rate. Notwithstanding any provision to the contrary, in no event shall the interest or any other amount payable hereunder exceed the maximum rate of interest permitted by applicable law (the “Maximum Lawful Rate”). Any interest paid in excess of the Maximum Lawful Rate shall be applied to the reduction of principal or, if no principal remains outstanding, refunded to the Maker. |
| 11. | Conflict with Asset Purchase Agreement. In the event of any conflict or inconsistency between the terms of this Note and the Asset Purchase Agreement, the terms of the Asset Purchase Agreement shall control. |
| 12. | Securities Law Matters. Any transfer or assignment of this Note by the Holder shall be made in compliance with all applicable federal and state securities laws. The Maker shall have no obligation to issue replacement Notes or register any transfer unless the Holder delivers an opinion of counsel reasonably satisfactory to the Maker that such transfer is exempt from registration under the Securities Act of 1933, as amended. |
| 13. | Electronic Execution. This Note and any amendments may be executed by electronic signature (including DocuSign or .pdf) and shall have the same legal effect as original wet-ink signatures. |
18
IN WITNESS WHEREOF, the Maker has caused this Promissory Note to be executed by its duly authorized representative as of the date first above written.
| VISIONWAVE HOLDINGS, INC. | ||
| By: | ||
| Name: | Douglas Davis | |
| Title: | CEO & Executive Chairman | |
19