
Protein degraded. Disease targeted. Lives transformed. April 2026

Forward-looking Statements and Intellectual Property 2 FORWARD-LOOKING STATEMENTS The following presentation contains forward-looking statements. All statements other than statements of historical fact are forward-looking statements, which are often indicated by terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “goal,” “intend,” “look forward to,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “will,” “would” and similar expressions. These forward-looking statements include, but are not limited to, statements regarding the therapeutic potential of C4 Therapeutics, Inc.’s technology and products. These forward-looking statements are not promises or guarantees and involve substantial risks and uncertainties. Among the factors that could cause actual results to differ materially from those described or projected herein include uncertainties associated generally with research and development, clinical trials and related regulatory reviews and approvals, as well as the fact that the product candidates that we are developing or may develop may not demonstrate success in clinical trials. Prospective investors are cautioned not to place undue reliance on these forward- looking statements. The forward-looking statements included in this presentation speak only as of the date hereof and are subject to a variety of risks and uncertainties, including those set forth in our most recent and future filings with the Securities and Exchange Commission. Our actual results could vary significantly from those anticipated in this presentation, and our financial condition and results of operations could be materially adversely affected. C4 Therapeutics, Inc., undertakes no obligation to update or revise the information contained in this presentation, whether as a result of new information, future events or circumstances or otherwise. This presentation also contains estimates, projections and other information concerning the markets for C4 Therapeutics, Inc.’s product candidates, including data regarding the estimated size of those markets, and the incidence and prevalence of certain medical conditions and patient use of medicines. Information that is based on estimates, forecasts, projections, market research, or similar methodologies is inherently subject to uncertainties and actual events, and circumstances may differ materially from events and circumstances reflected in this information. Unless otherwise expressly stated, the Company obtained this industry, business, market and other data from reports, research surveys, clinical trials studies and similar data prepared by market research firms and other third parties, from industry, medical and general publications, from other publicly available information, and from government data and similar sources. INTELLECTUAL PROPERTY C4 Therapeutics, Inc., owns various registered and unregistered trademarks and service marks in the U.S. and internationally, including, without limitation, C4 THERAPEUTICS, our housemark logo, the name of our TORPEDO platform, and the names of our BIDAC and MONODAC degrader products. All trademarks, service marks, or trade names referred to in this presentation that we do not own are the property of their respective owners. Solely for convenience, the trademarks, service marks, and trade names in this presentation are referred to without the symbols ®, SM and , but those references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights to these trademarks, service marks, or trade names. © 2026 C4 Therapeutics, Inc.

Advancing Differentiated TPD Medicines and Building a Sustainable Pipeline of High-value Degraders To Achieve Our Vision 3© 2026 C4 Therapeutics, Inc. BEST-IN-CLASS AND FIRST-IN-CLASS DEGRADERS. VALIDATED PATHWAYS. LARGE MARKET OPPORTUNITIES Multiple myeloma (MM); Non-small cell lung cancer (NSCLC) High Value Clinical Oncology Portfolio Discovery Strategy Now Focused on INN (Inflammation, Neuroinflammation, and Neurodegeneration) Financial Strength to Execute Advancing two clinical degraders • A potential best-in- class IKZF1/3 degrader for MM • An EGFR L858R degrader for NSCLC Progressing potential first-in-class degraders focused on INN diseases to build a sustainable pipeline Cash runway expected to end of 2028 beyond key value inflection points across portfolio Vision: To become a fully integrated biopharmaceutical company
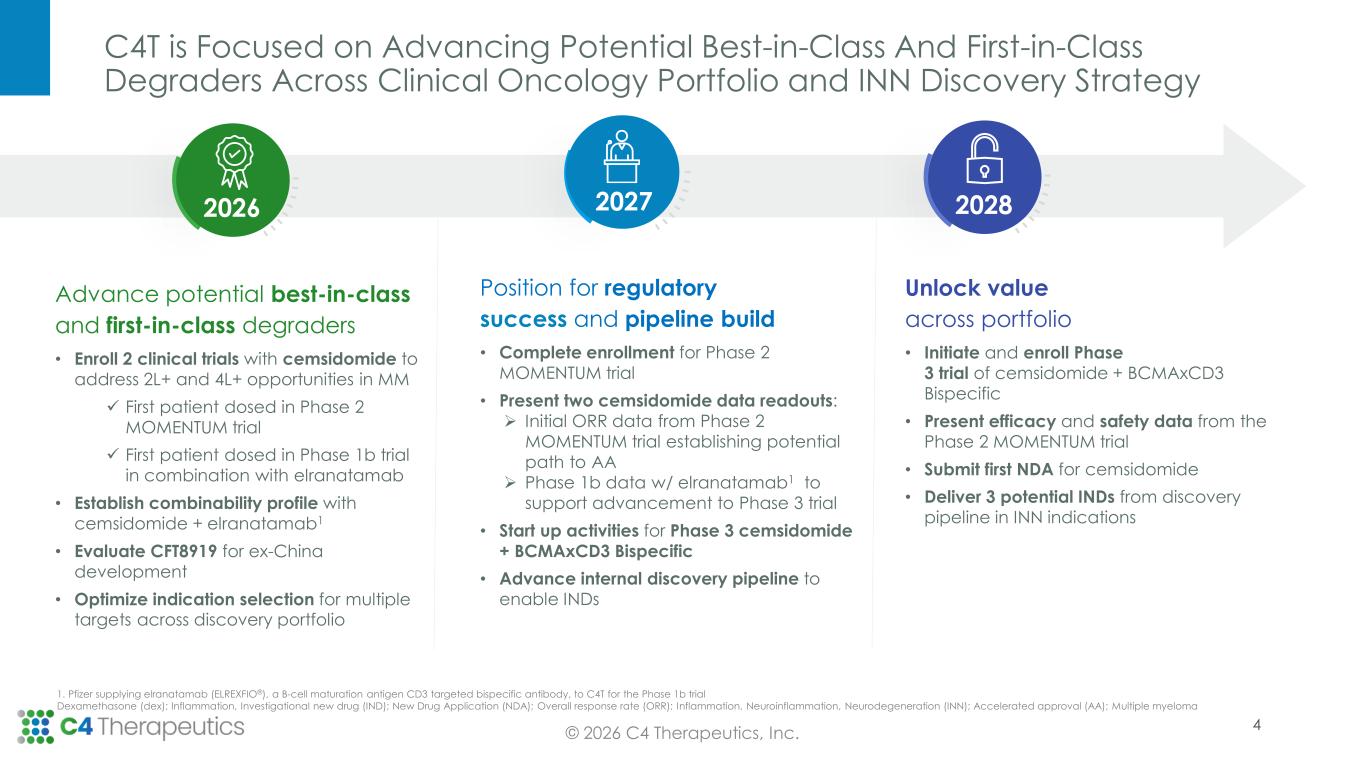
C4T is Focused on Advancing Potential Best-in-Class And First-in-Class Degraders Across Clinical Oncology Portfolio and INN Discovery Strategy © 2026 C4 Therapeutics, Inc. Advance potential best-in-class and first-in-class degraders • Enroll 2 clinical trials with cemsidomide to address 2L+ and 4L+ opportunities in MM ✓ First patient dosed in Phase 2 MOMENTUM trial ✓ First patient dosed in Phase 1b trial in combination with elranatamab • Establish combinability profile with cemsidomide + elranatamab1 • Evaluate CFT8919 for ex-China development • Optimize indication selection for multiple targets across discovery portfolio Unlock value across portfolio • Initiate and enroll Phase 3 trial of cemsidomide + BCMAxCD3 Bispecific • Present efficacy and safety data from the Phase 2 MOMENTUM trial • Submit first NDA for cemsidomide • Deliver 3 potential INDs from discovery pipeline in INN indications Position for regulatory success and pipeline build • Complete enrollment for Phase 2 MOMENTUM trial • Present two cemsidomide data readouts: ➢ Initial ORR data from Phase 2 MOMENTUM trial establishing potential path to AA ➢ Phase 1b data w/ elranatamab1 to support advancement to Phase 3 trial • Start up activities for Phase 3 cemsidomide + BCMAxCD3 Bispecific • Advance internal discovery pipeline to enable INDs 202820272026 1. Pfizer supplying elranatamab (ELREXFIO®), a B-cell maturation antigen CD3 targeted bispecific antibody, to C4T for the Phase 1b trial Dexamethasone (dex); Inflammation, Investigational new drug (IND); New Drug Application (NDA); Overall response rate (ORR); Inflammation, Neuroinflammation, Neurodegeneration (INN); Accelerated approval (AA); Multiple myeloma 4
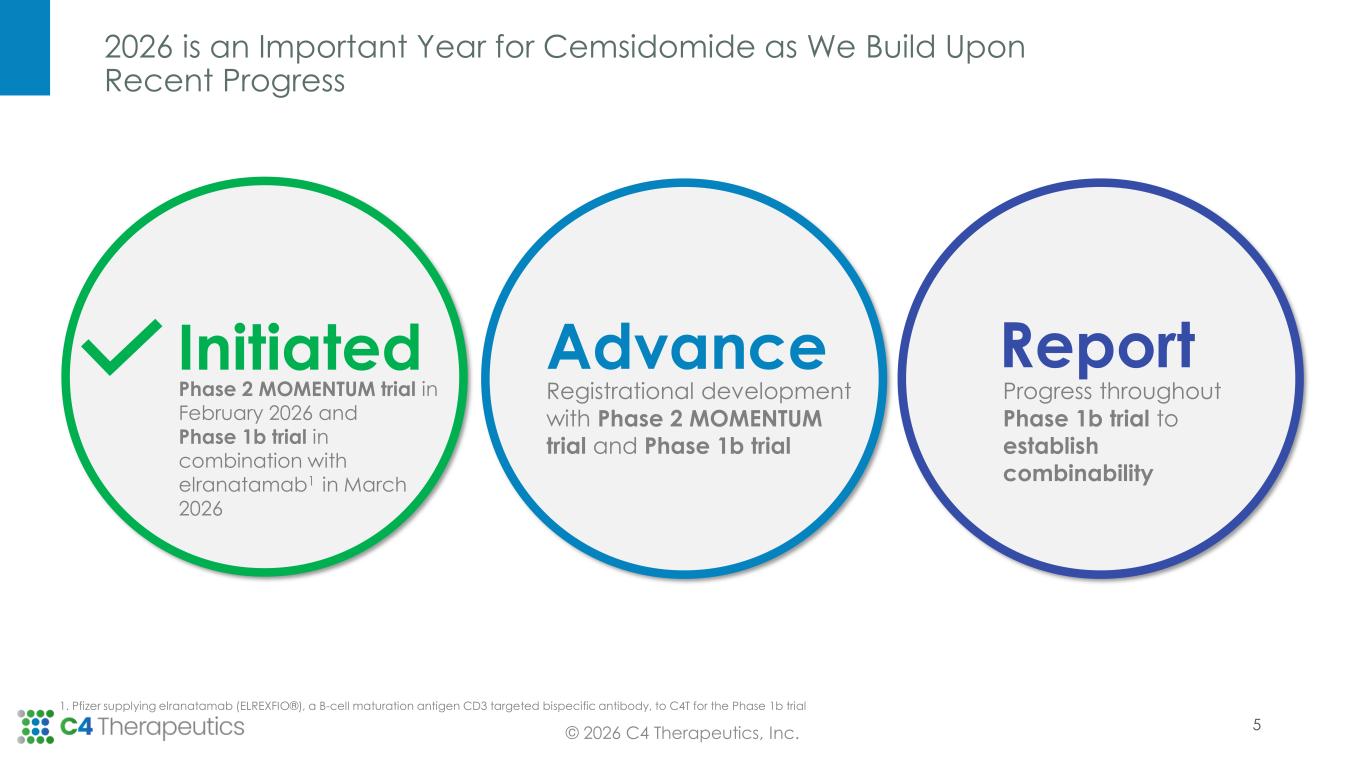
2026 is an Important Year for Cemsidomide as We Build Upon Recent Progress © 2026 C4 Therapeutics, Inc. Initiated Phase 2 MOMENTUM trial in February 2026 and Phase 1b trial in combination with elranatamab1 in March 2026 Advance Registrational development with Phase 2 MOMENTUM trial and Phase 1b trial Report Progress throughout Phase 1b trial to establish combinability 5 1. Pfizer supplying elranatamab (ELREXFIO®), a B-cell maturation antigen CD3 targeted bispecific antibody, to C4T for the Phase 1b trial
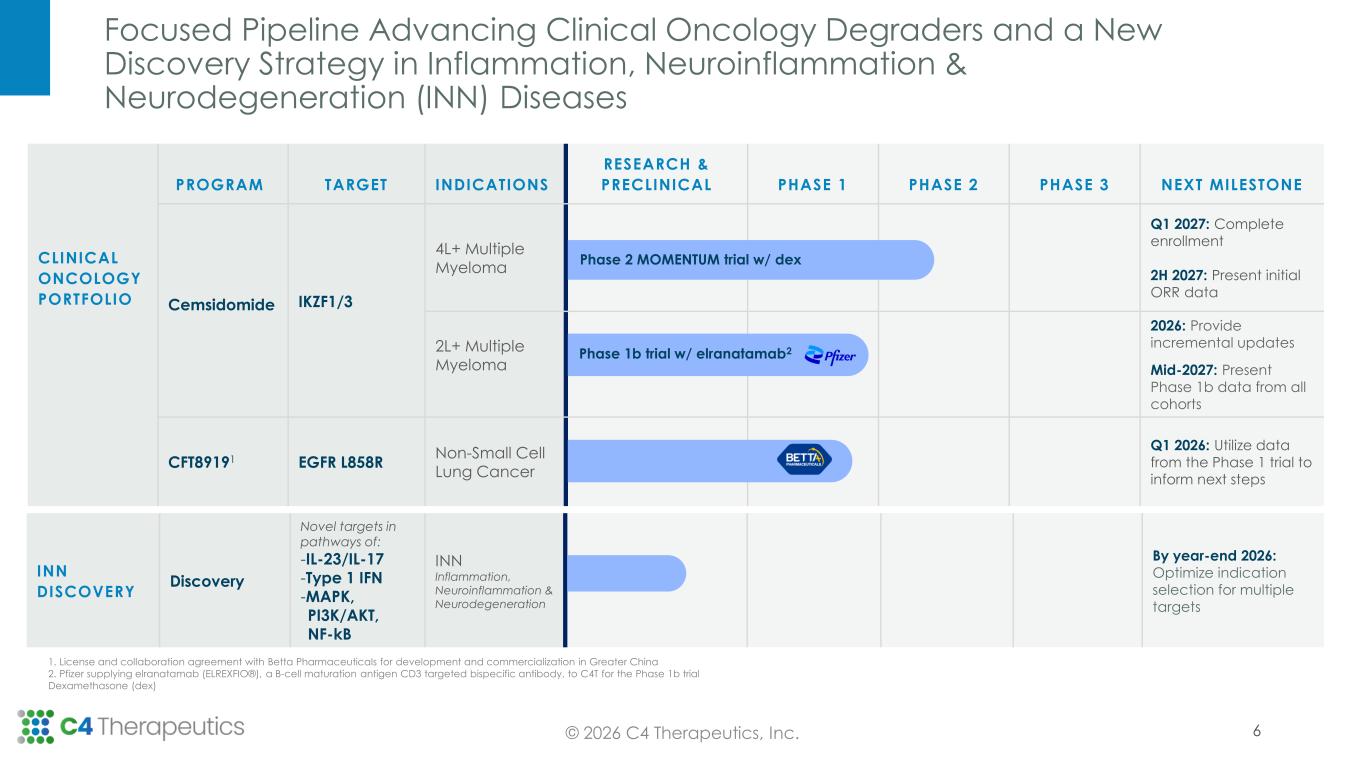
Focused Pipeline Advancing Clinical Oncology Degraders and a New Discovery Strategy in Inflammation, Neuroinflammation & Neurodegeneration (INN) Diseases 6© 2026 C4 Therapeutics, Inc. INN DISCOVERY Discovery Novel targets in pathways of: -IL-23/IL-17 -Type 1 IFN -MAPK, PI3K/AKT, NF-kB INN Inflammation, Neuroinflammation & Neurodegeneration By year-end 2026: Optimize indication selection for multiple targets 1. License and collaboration agreement with Betta Pharmaceuticals for development and commercialization in Greater China 2. Pfizer supplying elranatamab (ELREXFIO®), a B-cell maturation antigen CD3 targeted bispecific antibody, to C4T for the Phase 1b trial Dexamethasone (dex) CLINICAL ONCOLOGY PORTFOLIO PROGRAM TARGET INDICATIONS RESEARCH & PRECLINICAL PHASE 1 PHASE 2 PHASE 3 NEXT MILESTONE Cemsidomide IKZF1/3 4L+ Multiple Myeloma Q1 2027: Complete enrollment 2H 2027: Present initial ORR data 2L+ Multiple Myeloma 2026: Provide incremental updates Mid-2027: Present Phase 1b data from all cohorts CFT89191 EGFR L858R Non-Small Cell Lung Cancer Q1 2026: Utilize data from the Phase 1 trial to inform next steps Phase 2 MOMENTUM trial w/ dex Phase 1b trial w/ elranatamab2
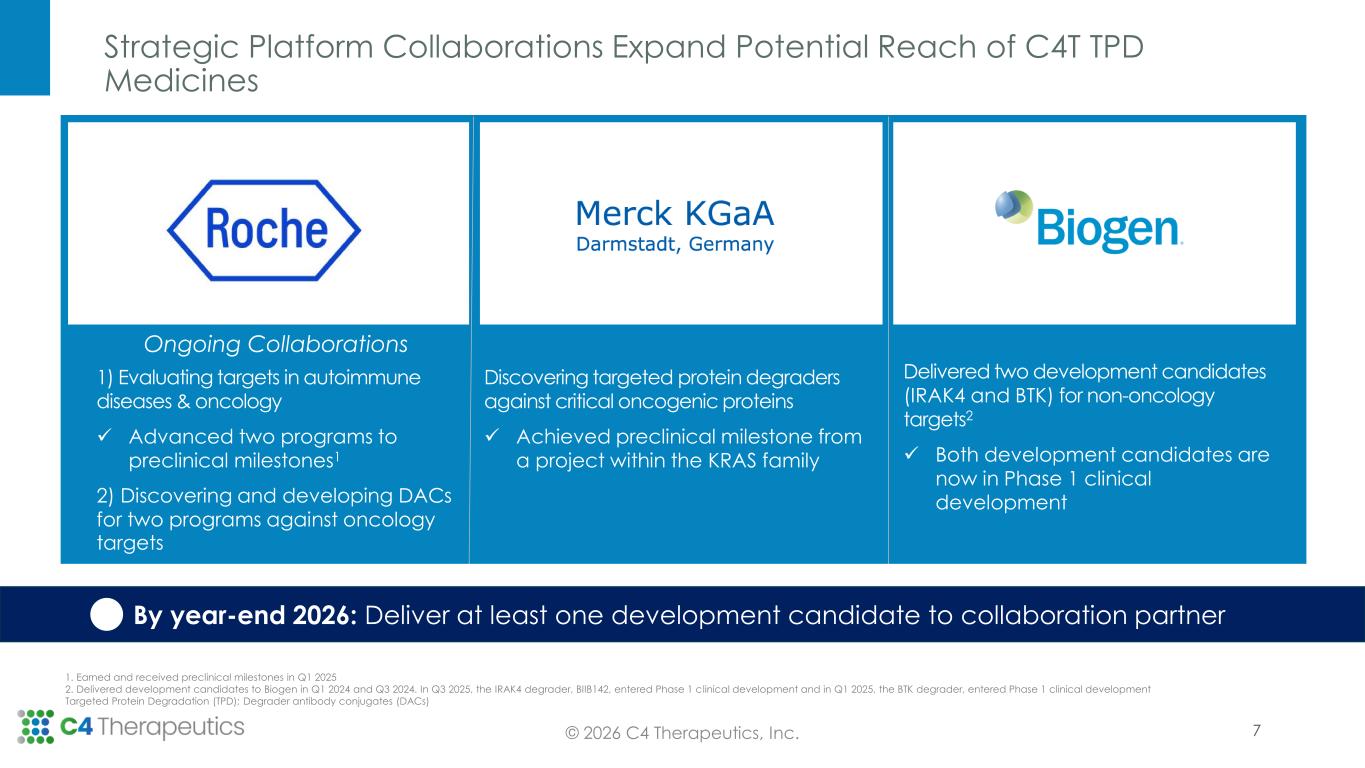
Strategic Platform Collaborations Expand Potential Reach of C4T TPD Medicines 7© 2026 C4 Therapeutics, Inc. 1. Earned and received preclinical milestones in Q1 2025 2. Delivered development candidates to Biogen in Q1 2024 and Q3 2024. In Q3 2025, the IRAK4 degrader, BIIB142, entered Phase 1 clinical development and in Q1 2025, the BTK degrader, entered Phase 1 clinical development Targeted Protein Degradation (TPD); Degrader antibody conjugates (DACs) By year-end 2026: Deliver at least one development candidate to collaboration partner 1) Evaluating targets in autoimmune diseases & oncology ✓ Advanced two programs to preclinical milestones1 2) Discovering and developing DACs for two programs against oncology targets Discovering targeted protein degraders against critical oncogenic proteins ✓ Achieved preclinical milestone from a project within the KRAS family Delivered two development candidates (IRAK4 and BTK) for non-oncology targets2 ✓ Both development candidates are now in Phase 1 clinical development Ongoing Collaborations

Cemsidomide IKZF1/3 Degrader Multiple Myeloma

Cemsidomide is Positioned for Success in Multiple Myeloma 9© 2026 C4 Therapeutics, Inc. Efficient cemsidomide registrational development path with the potential for two accelerated approvals is differentiated from other IKZF1/3 degraders and focused on where the landscape is evolving Cemsidomide has a potential best-in-class profile among other IKZF1/3 degraders, including CELMoDs® , in a large and growing multiple myeloma market with a clinically and commercially de-risked MOA Despite recent approval for immune-based therapies in the MM landscape, IKZF1/3 are central drivers of MM development and progression, thus IKZF1/3 degraders will remain relevant across multiple lines and in combinations Multiple myeloma (MM) CELMoDs® is a registered trademarks of BMS
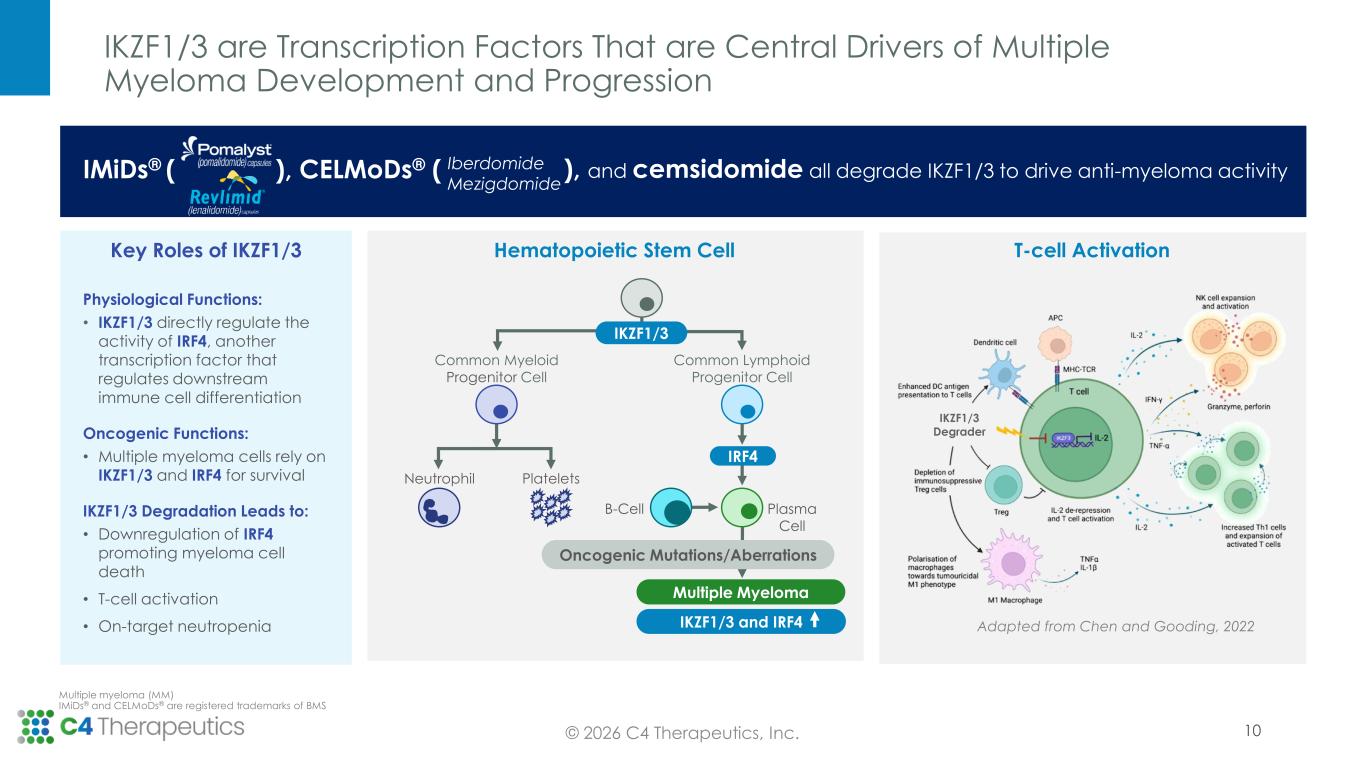
IKZF1/3 are Transcription Factors That are Central Drivers of Multiple Myeloma Development and Progression © 2026 C4 Therapeutics, Inc. Multiple myeloma (MM) IMiDs® and CELMoDs® are registered trademarks of BMS 10 Physiological Functions: • IKZF1/3 directly regulate the activity of IRF4, another transcription factor that regulates downstream immune cell differentiation Oncogenic Functions: • Multiple myeloma cells rely on IKZF1/3 and IRF4 for survival IKZF1/3 Degradation Leads to: • Downregulation of IRF4 promoting myeloma cell death • T-cell activation • On-target neutropenia Key Roles of IKZF1/3 Common Myeloid Progenitor Cell Common Lymphoid Progenitor Cell Neutrophil Platelets B-Cell Plasma Cell Oncogenic Mutations/Aberrations Multiple Myeloma IRF4 IKZF1/3 and IRF4 IKZF1/3 Hematopoietic Stem Cell T-cell Activation Adapted from Chen and Gooding, 2022 IKZF1/3 Degrader IMiDs® ( ), CELMoDs® ( Iberdomide Mezigdomide ), and cemsidomide all degrade IKZF1/3 to drive anti-myeloma activity
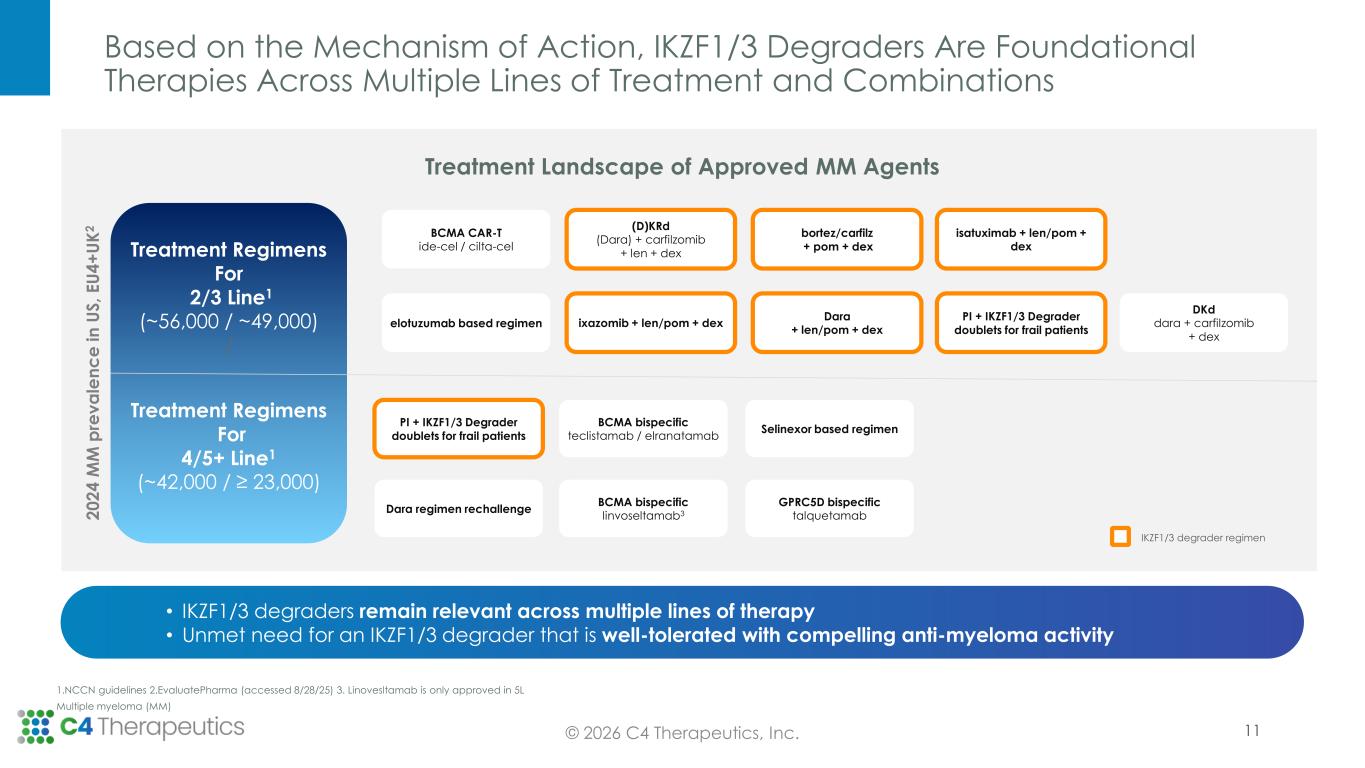
Based on the Mechanism of Action, IKZF1/3 Degraders Are Foundational Therapies Across Multiple Lines of Treatment and Combinations 11© 2026 C4 Therapeutics, Inc. bortez/carfilz + pom + dex BCMA CAR-T ide-cel / cilta-cel Dara + len/pom + dex PI + IKZF1/3 Degrader doublets for frail patients (D)KRd (Dara) + carfilzomib + len + dex DKd dara + carfilzomib + dex elotuzumab based regimen isatuximab + len/pom + dex ixazomib + len/pom + dex BCMA bispecific teclistamab / elranatamab PI + IKZF1/3 Degrader doublets for frail patients Selinexor based regimen GPRC5D bispecific talquetamab Dara regimen rechallenge BCMA bispecific linvoseltamab32 0 2 4 M M p re v a le n c e i n U S , E U 4 + U K 2 Treatment Landscape of Approved MM Agents IKZF1/3 degrader regimen 1.NCCN guidelines 2.EvaluatePharma (accessed 8/28/25) 3. Linovesltamab is only approved in 5L Multiple myeloma (MM) • IKZF1/3 degraders remain relevant across multiple lines of therapy • Unmet need for an IKZF1/3 degrader that is well-tolerated with compelling anti-myeloma activity Treatment Regimens For 2/3 Line1 (~56,000 / ~49,000) / Treatment Regimens For 4/5+ Line1 (~42,000 / ≥ 23,000)
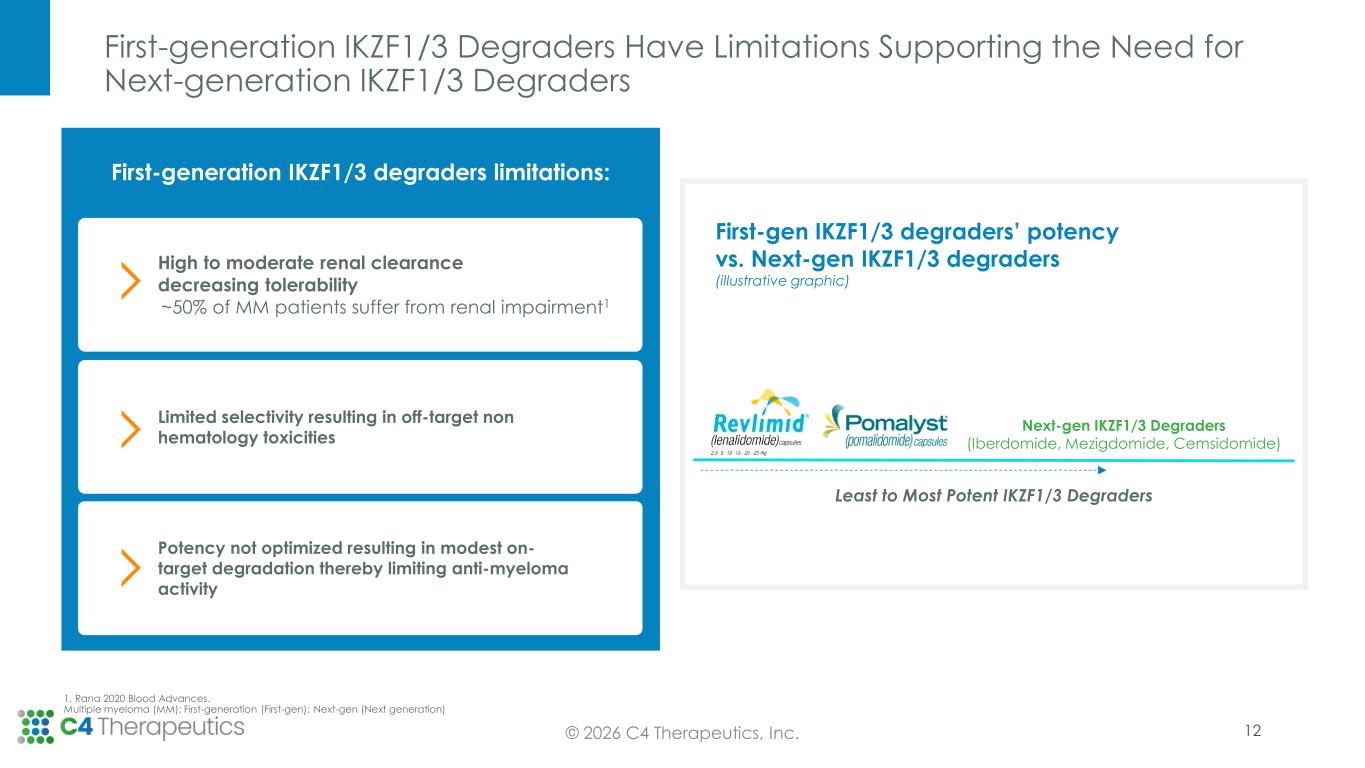
First-generation IKZF1/3 Degraders Have Limitations Supporting the Need for Next-generation IKZF1/3 Degraders 12© 2026 C4 Therapeutics, Inc. High to moderate renal clearance decreasing tolerability ~50% of MM patients suffer from renal impairment1 First-gen IKZF1/3 degraders’ potency vs. Next-gen IKZF1/3 degraders (illustrative graphic) First-generation IKZF1/3 degraders limitations: 1. Rana 2020 Blood Advances. Multiple myeloma (MM); First-generation (First-gen); Next-gen (Next generation) Potency not optimized resulting in modest on- target degradation thereby limiting anti-myeloma activity Limited selectivity resulting in off-target non hematology toxicities Least to Most Potent IKZF1/3 Degraders Next-gen IKZF1/3 Degraders (Iberdomide, Mezigdomide, Cemsidomide)
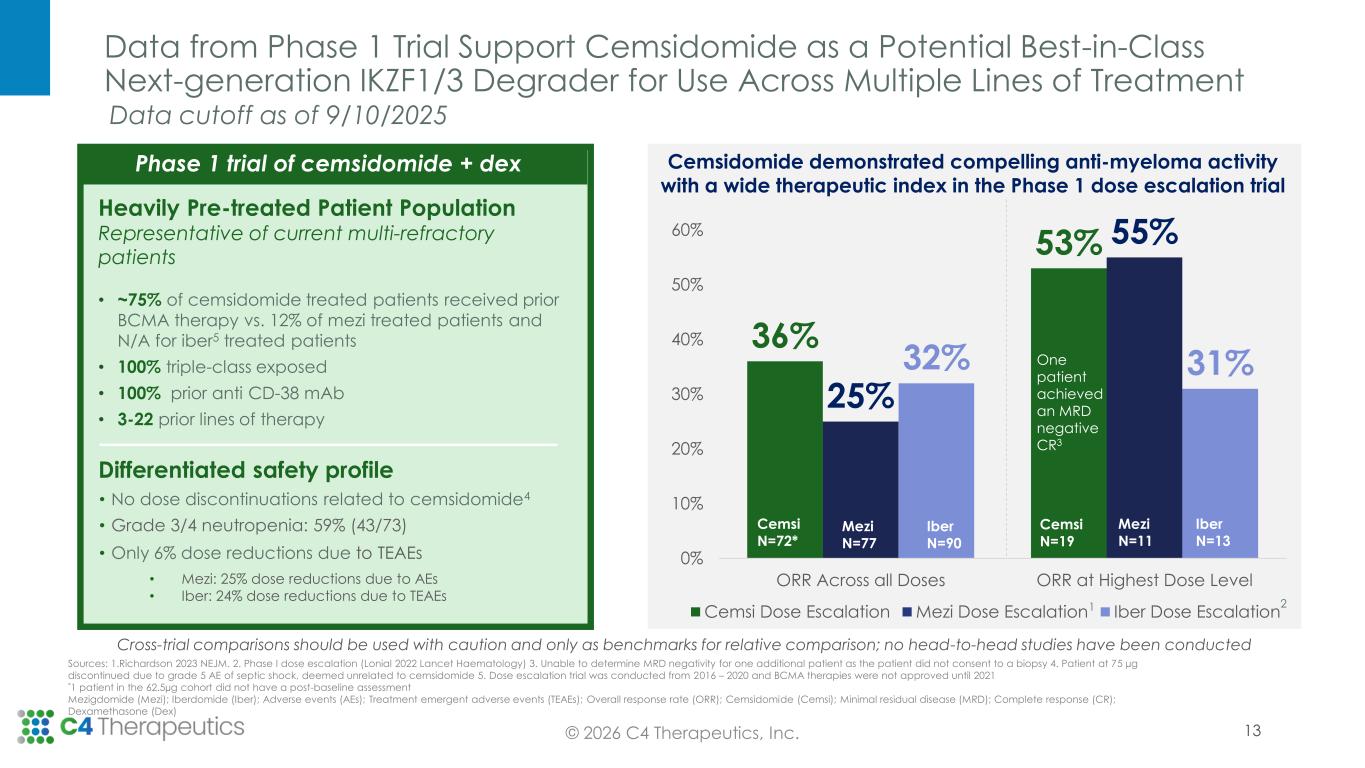
Data from Phase 1 Trial Support Cemsidomide as a Potential Best-in-Class Next-generation IKZF1/3 Degrader for Use Across Multiple Lines of Treatment 13© 2026 C4 Therapeutics, Inc. Heavily Pre-treated Patient Population Representative of current multi-refractory patients • ~75% of cemsidomide treated patients received prior BCMA therapy vs. 12% of mezi treated patients and N/A for iber5 treated patients • 100% triple-class exposed • 100% prior anti CD-38 mAb • 3-22 prior lines of therapy Differentiated safety profile • No dose discontinuations related to cemsidomide4 • Grade 3/4 neutropenia: 59% (43/73) • Only 6% dose reductions due to TEAEs • Mezi: 25% dose reductions due to AEs • Iber: 24% dose reductions due to TEAEs Sources: 1.Richardson 2023 NEJM. 2. Phase I dose escalation (Lonial 2022 Lancet Haematology) 3. Unable to determine MRD negativity for one additional patient as the patient did not consent to a biopsy 4. Patient at 75 µg discontinued due to grade 5 AE of septic shock, deemed unrelated to cemsidomide 5. Dose escalation trial was conducted from 2016 – 2020 and BCMA therapies were not approved until 2021 *1 patient in the 62.5µg cohort did not have a post-baseline assessment Mezigdomide (Mezi); Iberdomide (Iber); Adverse events (AEs); Treatment emergent adverse events (TEAEs); Overall response rate (ORR); Cemsidomide (Cemsi); Minimal residual disease (MRD); Complete response (CR); Dexamethasone (Dex) 36% 53% 25% 55% 32% 31% 0% 10% 20% 30% 40% 50% 60% ORR Across all Doses ORR at Highest Dose Level Cemsi Dose Escalation Mezi Dose Escalation Iber Dose Escalation Cemsi N=72* Mezi N=77 Cemsi N=19 Mezi N=11 Iber N=90 Iber N=13 1 2 Cemsidomide demonstrated compelling anti-myeloma activity with a wide therapeutic index in the Phase 1 dose escalation trial One patient achieved an MRD negative CR3 Phase 1 trial of cemsidomide + dex Cross-trial comparisons should be used with caution and only as benchmarks for relative comparison; no head-to-head studies have been conducted Data cutoff as of 9/10/2025

14© 2026 C4 Therapeutics, Inc. Cemsidomide Has the Potential to Capture a Valuable Portion of the Large Global Multiple Myeloma Market Sources: 1. Evaluate Pharma (8/14/2025) 2. Health Advances (2022), ClearView (2023), and C4T analysis Dexamethasone (dex) 2 Total global projected MM market is $46B by 20301 Cemsidomide’s market opportunity has potential to increase with additional combinations Cemsidomide has potential for multibillion dollar opportunities across multiple lines of therapy 2 Cemsidomide + BCMAxCD3 Bispecific (2L+) and Cemsidomide + dex (4L+) Estimated peak revenue for initial cemsidomide opportunity $2.5-$4B

Cemsidomide + Dexamethasone Has the Potential to Address a Large and Growing 4L+ Patient Population with a High Unmet Need © 2026 C4 Therapeutics, Inc. Large Market in a Growing Patient Population with High Unmet Needs Majority of MM Patients Continue to Progress Despite Novel Treatment Options: • Despite high initial response rates, 2/3 of CARVYKTI-treated patients relapse before 5 years1 • Later lines are expected to grow as patients live longer on newer treatments but ultimately progress - Median PFS range for patients treated with BiTEs: 7.5- 17.2 months2 Sources: 1. Legend Biotech Press Release June 3, 2025 (https://investors.legendbiotech.com/news-releases/news-release-details/legend-biotech-unveils-groundbreaking-5-year-survival-data) 2. https://www.jnjmedicalconnect.com/media/attestation/congresses/oncology/2024/ims/longterm-followup-from-the-phase-12-majestec1-trial-of-teclistamab-in-patients-with-relapsedrefracto.pdf; https://www.pfizer.com/news/press-release/press-release- detail/elrexfiotm-shows-median-overall-survival-more-two-years ; https://www.jnjmedicalconnect.com/products/talvey/medical-content/talvey-monumental1-mmy1001-study Overall Survival (OS); Mechanism of Action (MOA); Dexamethasone (dex) Phase 1 cemsidomide + dex trial in heavily pre- treated patients, de-risks Phase 2 MOMENTUM trial in the same population CEMSIDOMIDE DEVELOPMENT RATIONALE IN 4L+ 1 IKZF1/3 Remains A Key Validated MOA 2 3 Efficient Regulatory Path 15 Current treatment options have limited uptake due to their modest efficacy and poor tolerability
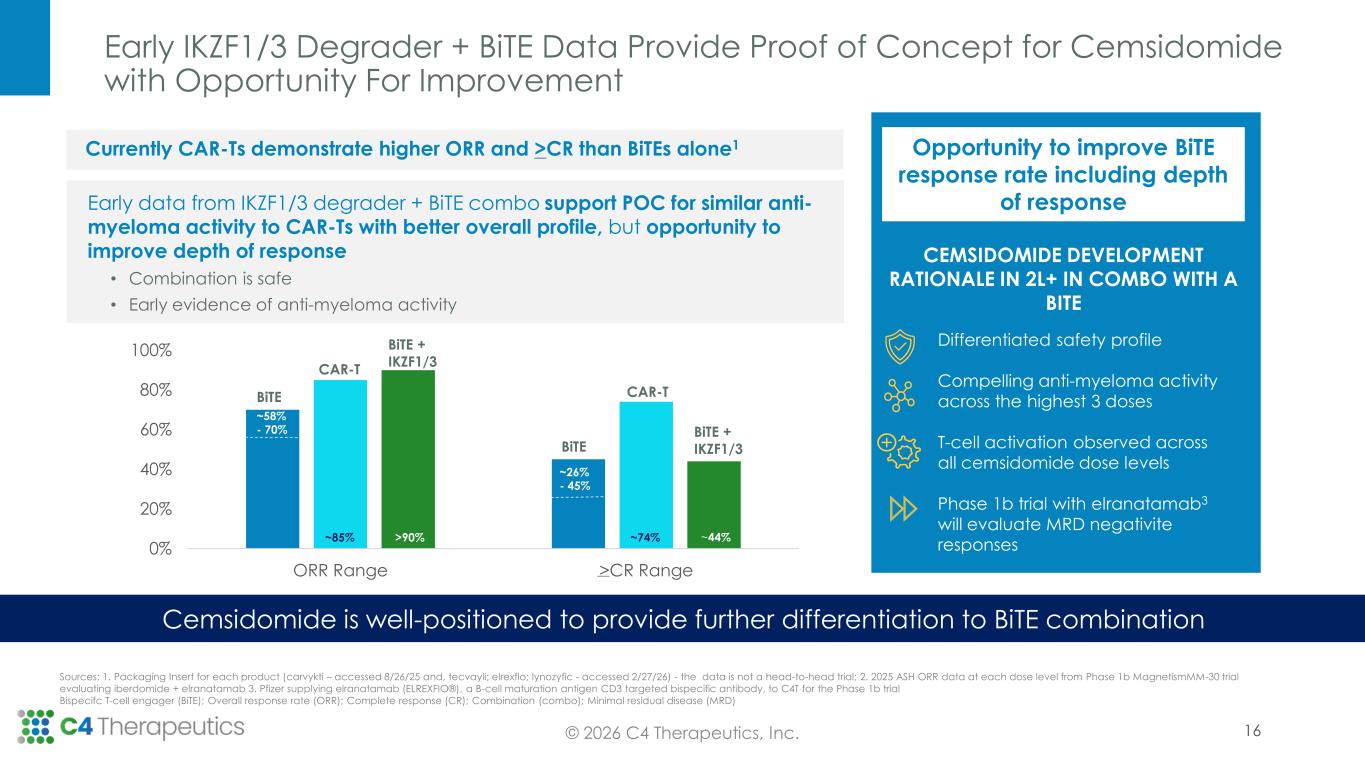
0% 20% 40% 60% 80% 100% ORR Range >CR Range Early IKZF1/3 Degrader + BiTE Data Provide Proof of Concept for Cemsidomide with Opportunity For Improvement © 2026 C4 Therapeutics, Inc. Currently CAR-Ts demonstrate higher ORR and >CR than BiTEs alone1 Early data from IKZF1/3 degrader + BiTE combo support POC for similar anti- myeloma activity to CAR-Ts with better overall profile, but opportunity to improve depth of response Cemsidomide is well-positioned to provide further differentiation to BiTE combination Sources: 1. Packaging Insert for each product (carvykti – accessed 8/26/25 and, tecvayli; elrexflo; lynozyfic - accessed 2/27/26) - the data is not a head-to-head trial; 2. 2025 ASH ORR data at each dose level from Phase 1b MagnetismMM-30 trial evaluating iberdomide + elranatamab 3. Pfizer supplying elranatamab (ELREXFIO®), a B-cell maturation antigen CD3 targeted bispecific antibody, to C4T for the Phase 1b trial Bispecifc T-cell engager (BiTE); Overall response rate (ORR); Complete response (CR); Combination (combo); Minimal residual disease (MRD) CEMSIDOMIDE DEVELOPMENT RATIONALE IN 2L+ IN COMBO WITH A BITE Opportunity to improve BiTE response rate including depth of response 16 • Combination is safe • Early evidence of anti-myeloma activity Differentiated safety profile Compelling anti-myeloma activity across the highest 3 doses T-cell activation observed across all cemsidomide dose levels Phase 1b trial with elranatamab3 will evaluate MRD negativite responses 74% ~58% - 70% CAR-T BiTE BiTE + IKZF1/3 BiTE + IKZF1/3 CAR-T BiTE ~74%>90% ~26% - 45% ~44%~85%
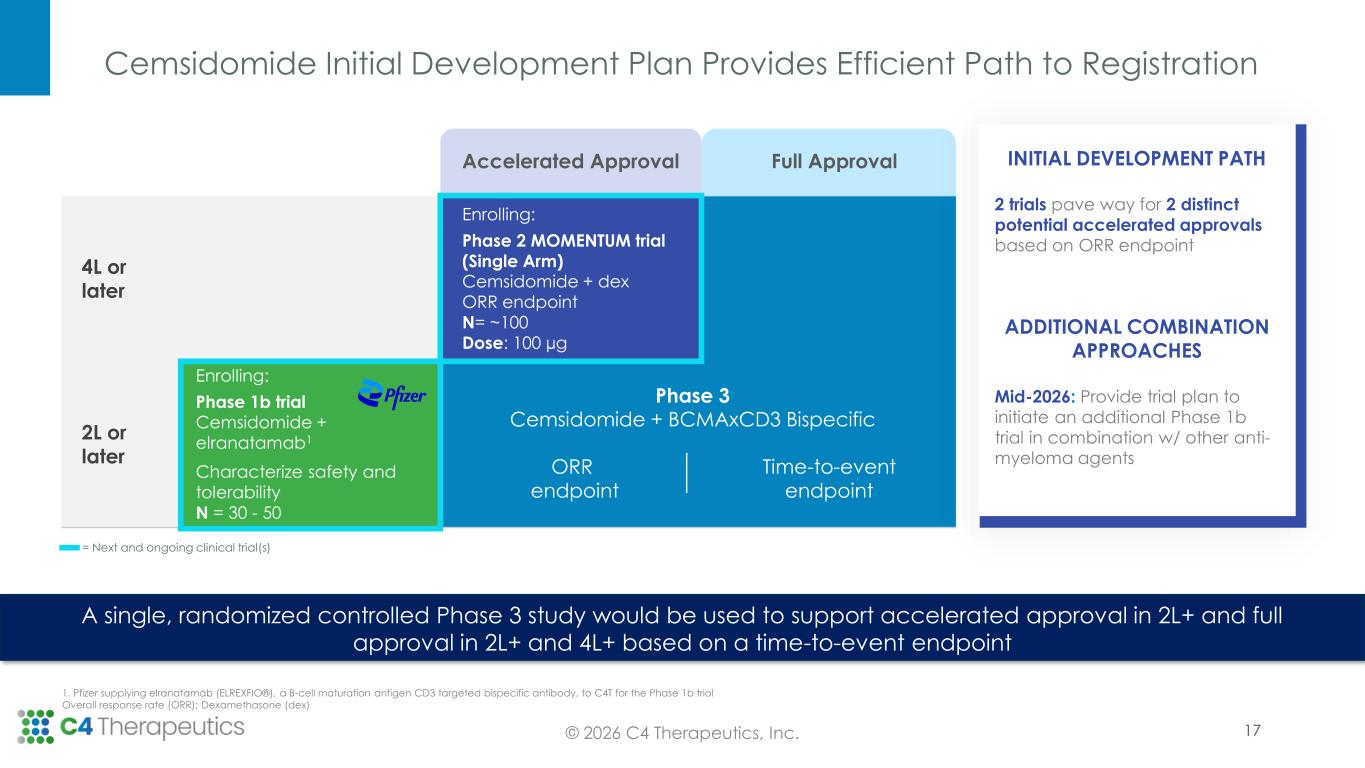
Cemsidomide Initial Development Plan Provides Efficient Path to Registration © 2026 C4 Therapeutics, Inc. A single, randomized controlled Phase 3 study would be used to support accelerated approval in 2L+ and full approval in 2L+ and 4L+ based on a time-to-event endpoint 1. Pfizer supplying elranatamab (ELREXFIO®), a B-cell maturation antigen CD3 targeted bispecific antibody, to C4T for the Phase 1b trial Overall response rate (ORR); Dexamethasone (dex) 17 Accelerated Approval Full Approval 4L or later Enrolling: Phase 2 MOMENTUM trial (Single Arm) Cemsidomide + dex ORR endpoint N= ~100 Dose: 100 µg Time-to-event endpoint 2L or later Enrolling: Phase 1b trial Cemsidomide + elranatamab1 Characterize safety and tolerability N = 30 - 50 ORR endpoint INITIAL DEVELOPMENT PATH 2 trials pave way for 2 distinct potential accelerated approvals based on ORR endpoint ADDITIONAL COMBINATION APPROACHES Mid-2026: Provide trial plan to initiate an additional Phase 1b trial in combination w/ other anti- myeloma agents Phase 3 Cemsidomide + BCMAxCD3 Bispecific = Next and ongoing clinical trial(s)

Phase 2 MOMENTUM Trial of Cemsidomide + Dex in 4L+ MM Now Enrolling Patients © 2026 C4 Therapeutics, Inc. Dexamethasone (dex); Overall response rate (ORR); Fourth line (4L); Recommended Phase 2 dose (RP2D); Overall response (ORR); International Myeloma Working Group (IMWG); Once daily (QD) 18 Endpoints: ORR per IMWG response criteria assessed by independent review committee • 20% increase over a background rate of 20% RP2D: 100 µg Schedule: QD 14/14 Potential for accelerated approval PHASE 2 MOMENTUM TRIAL DESIGN: 2H 2027: Phase 2 initial ORR data Phase 2 MOMENTUM Cemsidomide + dex (single arm) 4L+ N = ~100 Dose: 100 µg QD Enrollment Expected to Complete in Q1 2027

Phase 1b Trial is Evaluating Safety and Tolerability of Cemsidomide in Combination With Elranatamab, With Data From All Cohorts Expected in Mid-2027 © 2026 C4 Therapeutics, Inc. 1. Pfizer will supply elranatamab (ELREXFIO®), a B-cell maturation antigen CD3 targeted bispecific antibody, to C4T for its upcoming Phase 1b trial Dexamethasone (dex); Once daily (QD); Once weekly (QW) PHASE 1b TRIAL DESIGN: Primary Objectives: Characterize the safety and tolerability of cemsidomide in combination with elranatamab Dosing Regimen: • Cemsidomide: QD 14/14 • Dexamethasone: QW through cycle 4 • Elranatamab 1 Key Differentiators: • Evaluated with dex, which may help manage neutropenic complications • Focused on evaluating MRD negativity rates to demonstrate depth of response 19 Cemsidomide Dose Level: 75 µg + Elranatamab If 75 µg is declared safe, potential to simultaneously evaluate 50 µg and 100 µg Cemsidomide Dose Level: 50 µg + Elranatamab Cemsidomide Dose Level: 100 µg + Elranatamab Potential to expand at each dose level once combination is declared safe Trial Initiated in Q1; Enrollment Ongoing 2026: Provide incremental updates throughout Phase 1b dose escalation Elranatamab step-up dosing without cemsidomide

Cemsidomide Has a Potential Best-in-Class Profile To Be Used Across Multiple Lines of Treatment © 2026 C4 Therapeutics, Inc. • Orally bioavailable degrader with differentiated safety & tolerability profile with class-leading anti-myeloma activity ✓ 53% ORR at the highest dose level (100 µg) and 40% ORR at the second highest dose level (75 µg) ✓ 36% ORR across all doses evaluated, demonstrating a wide therapeutic window ✓ No discontinuations related to cemsidomide and minimal disruptive adverse events Potential best-in-class profile (Phase 1 cemsidomide + dex data) • Potential $2.5 - $4B1 peak revenue in combination with a BCMA BiTE in the 2L+ and with dexamethasone in 4L+ as an initial opportunity • Peak revenue has potential to increase with additional combinations Large addressable market opportunity • Initial opportunity focused on two distinct opportunities for accelerated approval in 2L+ and 4L+ • Differentiated development path focused on where the market is evolving Efficient regulatory path 1. Health Advances (2022), ClearView (2023), and C4T analysis 20 Data cutoff as of 9/10/2025

Discovery Inflammation, Neuroinflammation, & Neurodegeneration (INN)

New Discovery Strategy Focused on Inflammation, Neuroinflammation & Neurodegeneration (INN) with First-in-Class Potential in Clinically Validated Pathways Uniquely Suited for TPD 22© 2026 C4 Therapeutics, Inc. Targeted Protein Degradation (TPD); Central nervous system (CNS) Leveraging C4T’s success Maximizing value through target selection Deliver degraders with first-in-class potential that are CNS penetrant C4T HAS CONSISTENTLY DEVELOPED ORALY BIOAVAILABLE HIGHLY CATALYTIC HETEROBIVALENT DEGRADERS THAT… • Penetrate the blood brain barrier to achieve high central nervous system exposures and compelling efficacy in central nervous system models • Control target protein levels through finely-tuned degrader kinetics TARGET-TO-DISEASE LINK: • Selecting targets that modulate clinically validated pathways in inflammation, neuroinflammation, and neurodegeneration (INN) to enhance efficacy focusing on early clinical validation and growing valuing through indication expansion STRONG DEGRADER RATIONALE: • Strong competitive positioning • Clear and compelling advantage for a degrader over an inhibitor EXPANDED CAPABILITIES: • Extended capabilities to identify molecular glue degraders for targets with and without G- and RT-loops by utilizing DNA-encoded library (DEL) technology

Focused on Inflammation, Neuroinflammation & Neurodegeneration (INN) to Address High Unmet Needs in a Large Patient Population with a Clear TPD Advantage © 2026 C4 Therapeutics, Inc. Central nervous system (CNS), Pharmacodynamic (PD); Targeted Protein Degradation (TPD); Mechanism of action (MOA) 1. Based on preclinical evidence and working hypothesis 23 Deploying TPD where the MOA is uniquely positioned to have an advantage over inhibitors to help benefit patients in a large market Degraders have the potential to outperform inhibitors in efficacy and safety in CNS diseases1 Fast path to clinical proof-of-concept, including early validation based on PD markers in healthy volunteers Normalize elevated protein levels without the need for complete elimination of the target Large market opportunities with high unmet medical needs
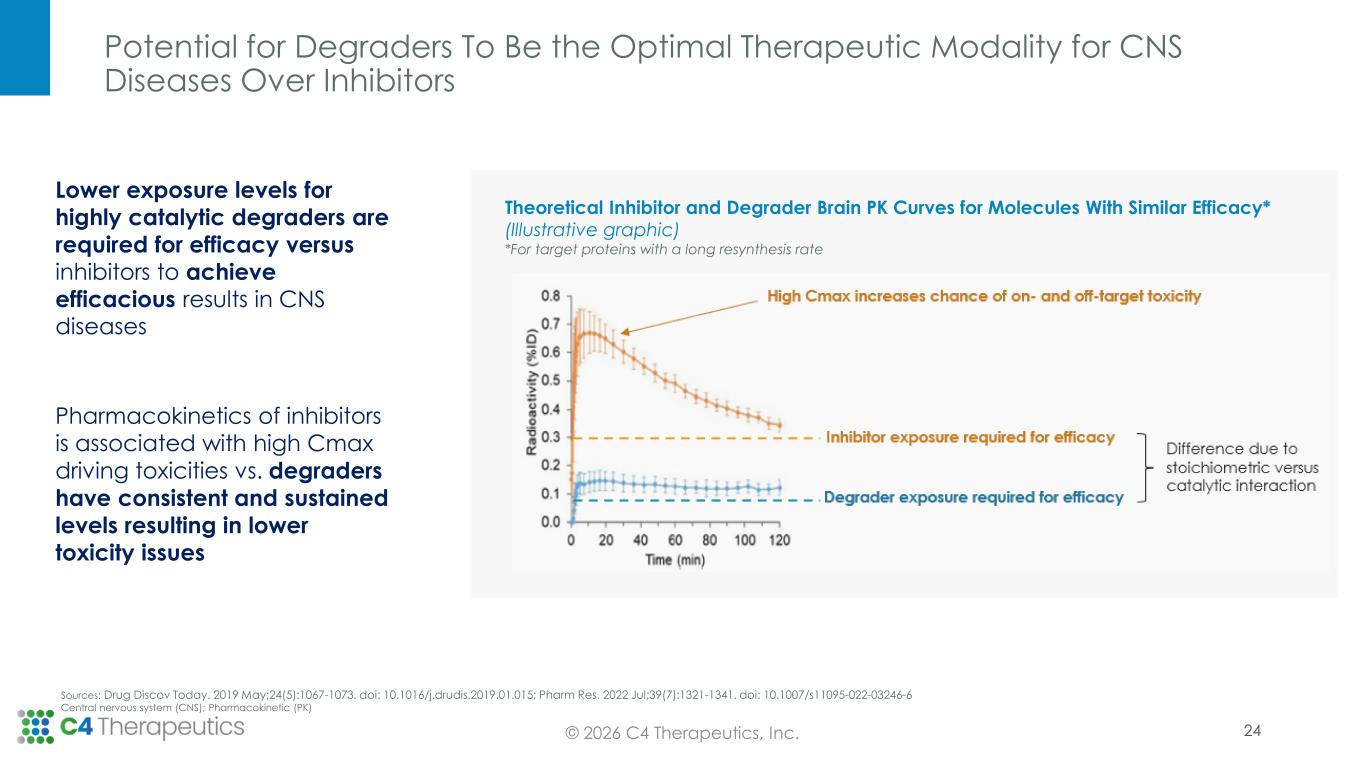
Potential for Degraders To Be the Optimal Therapeutic Modality for CNS Diseases Over Inhibitors 24 Theoretical Inhibitor and Degrader Brain PK Curves for Molecules With Similar Efficacy* (Illustrative graphic) *For target proteins with a long resynthesis rate Lower exposure levels for highly catalytic degraders are required for efficacy versus inhibitors to achieve efficacious results in CNS diseases Pharmacokinetics of inhibitors is associated with high Cmax driving toxicities vs. degraders have consistent and sustained levels resulting in lower toxicity issues Sources: Drug Discov Today. 2019 May;24(5):1067-1073. doi: 10.1016/j.drudis.2019.01.015; Pharm Res. 2022 Jul;39(7):1321-1341. doi: 10.1007/s11095-022-03246-6 Central nervous system (CNS); Pharmacokinetic (PK) © 2026 C4 Therapeutics, Inc.
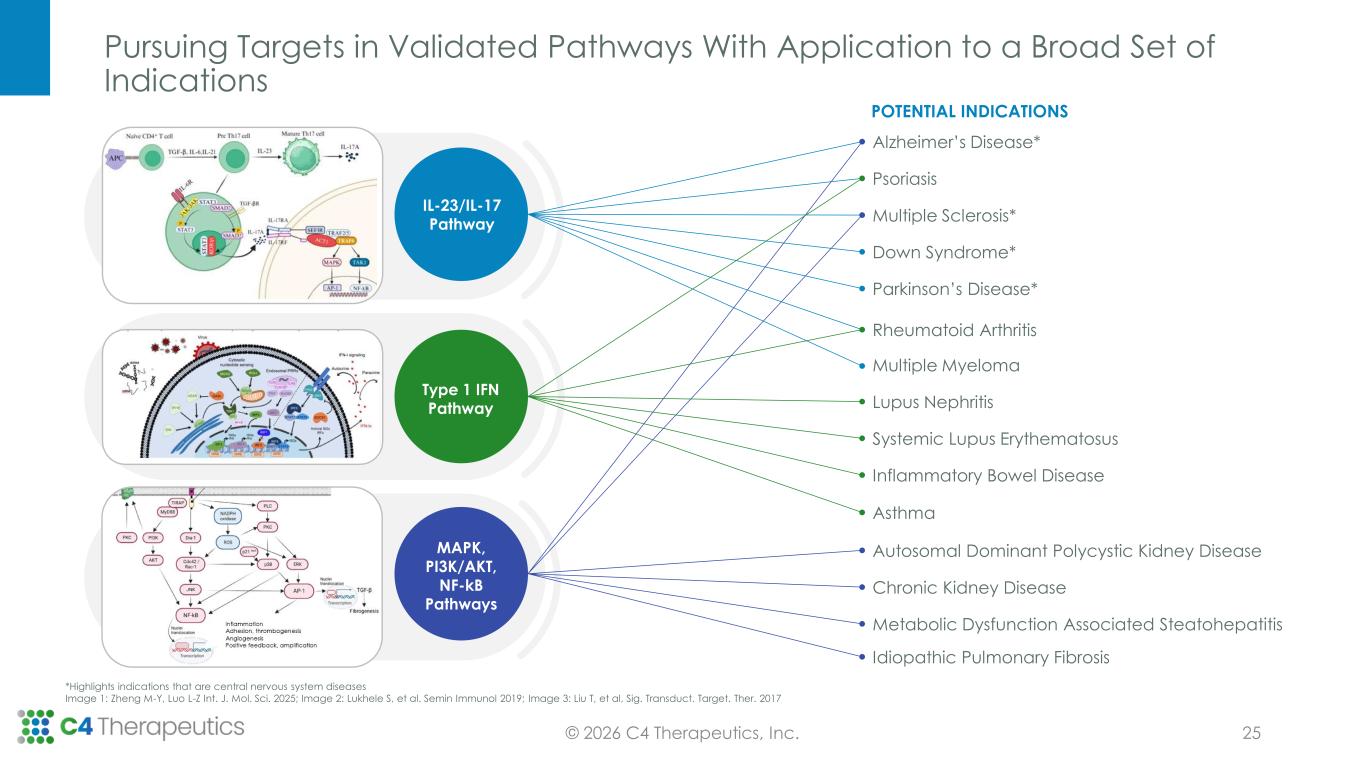
Pursuing Targets in Validated Pathways With Application to a Broad Set of Indications © 2026 C4 Therapeutics, Inc. *Highlights indications that are central nervous system diseases Image 1: Zheng M-Y, Luo L-Z Int. J. Mol. Sci. 2025; Image 2: Lukhele S, et al. Semin Immunol 2019; Image 3: Liu T, et al, Sig. Transduct. Target. Ther. 2017 25 Alzheimer’s Disease* Psoriasis Multiple Sclerosis* Down Syndrome* Parkinson’s Disease* Rheumatoid Arthritis Multiple Myeloma Lupus Nephritis Systemic Lupus Erythematosus Inflammatory Bowel Disease Asthma Autosomal Dominant Polycystic Kidney Disease Chronic Kidney Disease Metabolic Dysfunction Associated Steatohepatitis Idiopathic Pulmonary Fibrosis POTENTIAL INDICATIONS IL-23/IL-17 Pathway Type 1 IFN Pathway MAPK, PI3K/AKT, NF-kB Pathways

Multiple Strategic Milestones Expected to Advance Cemsidomide as a Potential Best-in-Class IKZF1/3 Degrader and Discovery Strategy Focused on Novel Targets in Clinically Validated Pathways 26© 2026 C4 Therapeutics, Inc. 2026 2027 - 2028 Cemsi + dex (4L+) ✓ Q1: Initiate the Phase 2 MOMENTUM trial of cemsidomide • Mid-2026: Present further analysis of the data from the completed Phase 1 trial • Q1 2027: Complete enrollment for Phase 2 MOMENTUM trial • 2H 2027: Present initial ORR data for the Phase 2 MOMENTUM trial • Mid-2028: Present ORR data and indices of durability and safety for the Phase 2 MOMENTUM trial • By year-end 2028: Submit new drug application Cemsi combination (2L+) ✓ Q1: Initiate the Phase 1b trial in combination w/ elranatamab1 • 2026: Provide incremental updates throughout Phase 1b dose escalation in combination w/ elranatamab1 • Mid-2026: Provide trial plan to initiate an additional Phase 1b trial in combination w/ other anti-myeloma agents • Mid-2027: Present Phase 1b data from all cohorts in combination w/ elranatamab1 • By early 2028: Initiate the Phase 3 trial in combination with a BCMAxCD3 Bispecific CFT8919 • Q1: Utilize data from the Phase 1 dose escalation trial to inform ex-China clinical development Discovery (INN & Collaborations) • By year-end: Deliver at least one development candidate to a collaboration partner • By year-end: Advance existing collaborations toward key milestones • 2027: Advance internal discovery pipeline to enable INDs • By year-end 2028: Deliver up to three investigational new drug applications Inflammation, Neuroinflammation & Neurodegeneration (INN) 1. Pfizer supplying elranatamab (ELREXFIO®), a B-cell maturation antigen CD3 targeted bispecific antibody, to C4T for the Phase 1b trial