
1 ASX: MSB; Nasdaq: MESO. I April 8, 2026 Welcome to R&D Day 1 Exhibit 99.2

2 This presentation includes forward-looking statements and forecasts that relate to future events or our future financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to differ materially from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. We make such forward- looking statements pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. All statements other than statements of historical facts contained in this presentation are forward-looking statements. Words such as, but not limited to, “believe,” “expect,” “anticipate,” “estimate,” “intend,” “plan,” “targets,” “likely,” “will,” “would,” “could,” and similar expressions or phrases identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and future events , recent changes in regulatory laws, and financial trends that we believe may affect our financial condition, results of operation, business strategy and financial needs. These statements may relate to, but are not limited to: expectations with respect to sales and revenue, expectations regarding the safety or efficacy of, or potential applications for, Mesoblast's adult stem cell technologies; expectations regarding the strength of Mesoblast's intellectual property, the timeline for Mesoblast's regulatory approval process, and the scalability and efficiency of manufacturing processes; expectations about Mesoblast's ability to grow its business and statements regarding its relationships with current and potential future business partners and future benefits of those relationships; statements concerning Mesoblast's share price or potential market capitalization; and statements concerning Mesoblast's capital requirements and ability to raise future capital, among others. Forward-looking statements should not be read as a guarantee of future performance or results, and actual results may differ from the results anticipated in these forward-looking statements, and the differences may be material and adverse. You should read this presentation together with our financial statements and the notes related thereto, as well as the risk factors, in our most recently filed reports with the SEC or on our website. Uncertainties and risks that may cause Mesoblast's actual results, performance or achievements to be materially different from those which may be expressed or implied by such statements, include, without limitation: risks inherent in the development and commercialization of potential products; uncertainty of clinical trial results or regulatory approvals or clearances; government regulation; the need for future capital; dependence upon collaborators; and protection of our intellectual property rights, among others. Accordingly, you should not place undue reliance on these forward-looking statements. We do not undertake any obligations to publicly update or revise any forward-looking statements, whether as a result of new information, future developments or otherwise. Cautionary Note Regarding Forward Looking Statements

3 Presenting KOL Perspective Welcome Paul Hughes Overview Silviu Itescu, MD Ryoncil® Flagship Growth Marcelo Santoro, DBA Joanne Kurtzberg, MD Adult SR-aGvHD Michael Schuster, MBA, MS Susan Prockop, MD Chronic Lower Back Pain Roger Brown Doug Beall, MD Inflammatory Heart Failure Ken Borow, MD Emerson Perin, MD Eric Rose, MD Pediatric Label Extension Michael Schuster, MBA, MS Next Generation Platforms Dan Devine, JD, MBA Saad Kenderian, MB Financial and Manufacturing James O’Brien / Justin Horst Wrap-up and Closing Remarks Silviu Itescu, MD Audience Q&A Agenda

4 Meet Today’s Invited Experts – Key Opinion Leaders Eric Rose, MD, Chief Medical Officer World-renowned heart surgeon and scientist made history when he performed the first successful pediatric heart transplant Joanne Kurtzberg, MD Jerome Harris Distinguished Professor of Pediatrics Professor of Pathology Duke University School of Medicine Susan Prockop, MD Director, Clinical and Translational Research (Stem Cell Transplant Program, DFCI/BCH Cancer and Blood Disorders Center) Emerson C. Perin, MD, PhD Medical Director of The Texas Heart Institute Professor of Medicine, Baylor College of Medicine Douglas P. Beall, MD Chief of Services, Comprehensive Specialty Care; Director of Research Clinical Investigations LLC Saad Kenderian, MD, ChB Assistant Professor of Oncology, Medicine and Immunology at Mayo Clinic

55 Strategic Overview Silviu Itescu Chief Executive

6 Mesoblast: Global Leader in Allogeneic (Off-the-Shelf) Cellular Medicines First FDA approval more than 1,100 patents & applications Robust Pipeline • World leader in developing allogeneic (off-the-shelf) cellular medicines for the treatment of severe and life- threatening inflammatory conditions • Locations in United States, Australia, and Singapore • Listed on NASDAQ (MESO) and ASX (MSB) • Developing product candidates for distinct indications based on its remestemcel-L and rexlemestrocel-L stromal cell technology platforms • First FDA-approved product launched successfully • Extensive global intellectual property portfolio with protection extending through to at least 2044 in all major markets • FDA-inspected commercial scale manufacturing process and facilities THREE Additional Major Phase 3 Assets
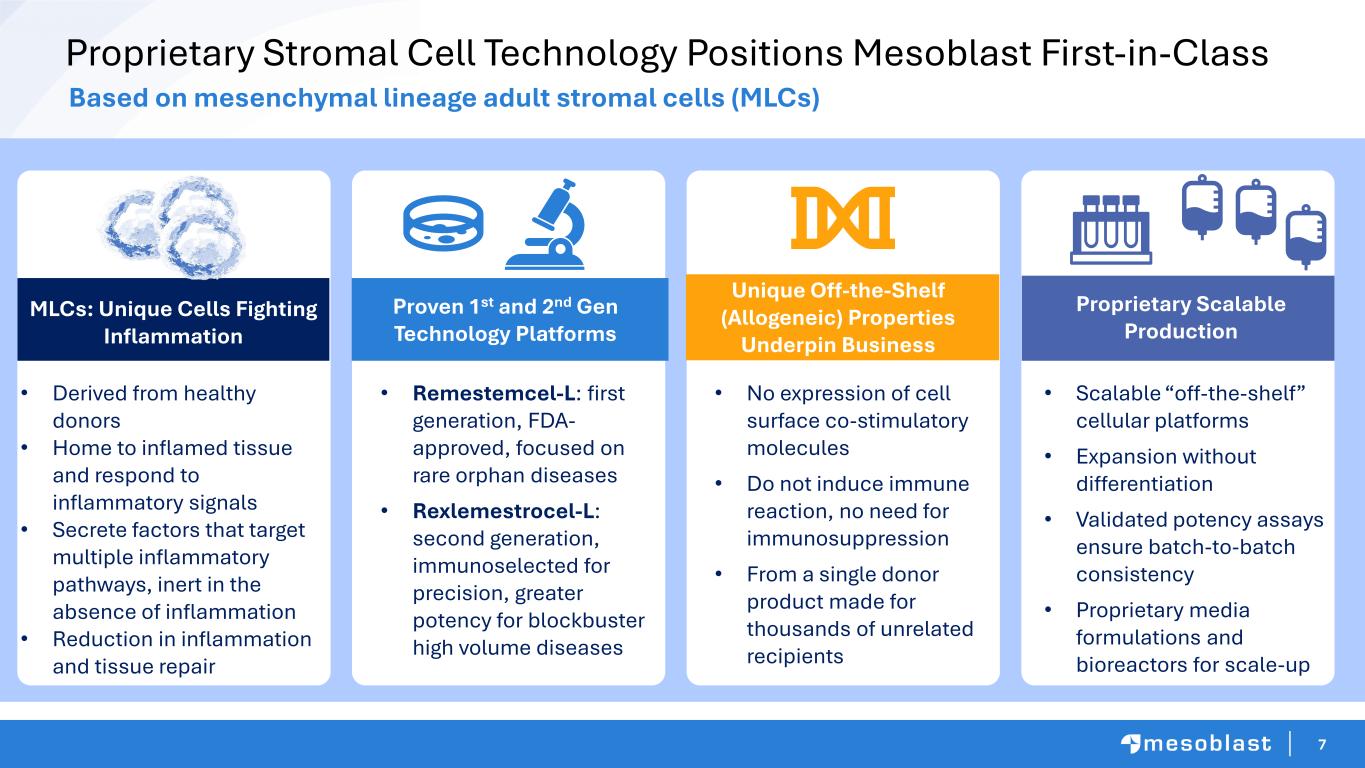
7 Proprietary Stromal Cell Technology Positions Mesoblast First-in-Class MLCs Derived from healthy bone marrow Home to inflamed tissue and respond to inflammatory signals Secrete factors thattarget multiple inflammatory pathways, inert in the absence of inflammation Reduction in inflammation and tissue repair Based on mesenchymal lineage adult stromal cells (MLCs) • Derived from healthy donors • Home to inflamed tissue and respond to inflammatory signals • Secrete factors that target multiple inflammatory pathways, inert in the absence of inflammation • Reduction in infla mation and tissue repair • Remestemcel-L: first generation, FDA- approved, focused on rare orphan diseases • Rexlemestrocel-L: second generation, immunoselected for precision, greater potency for blockbuster high volume diseases • No expression of cell surface co-stimulatory molecules • Do not induce immune reaction, no need for immunosuppression • From a single donor product made for thousands of unrelated recipients • Scalable “off-the-shelf” cellular platforms • Expansion without differentiation • Validated potency assays ensure batch-to-batch consistency • Proprietary media formulations and bioreactors for scale-up MLCs: Unique Cells Fighting Inflammation Proven 1st and 2nd Gen Technology Platforms Unique Off-the-Shelf (Allogeneic) Properties Underpin Business Proprietary Scalable Production
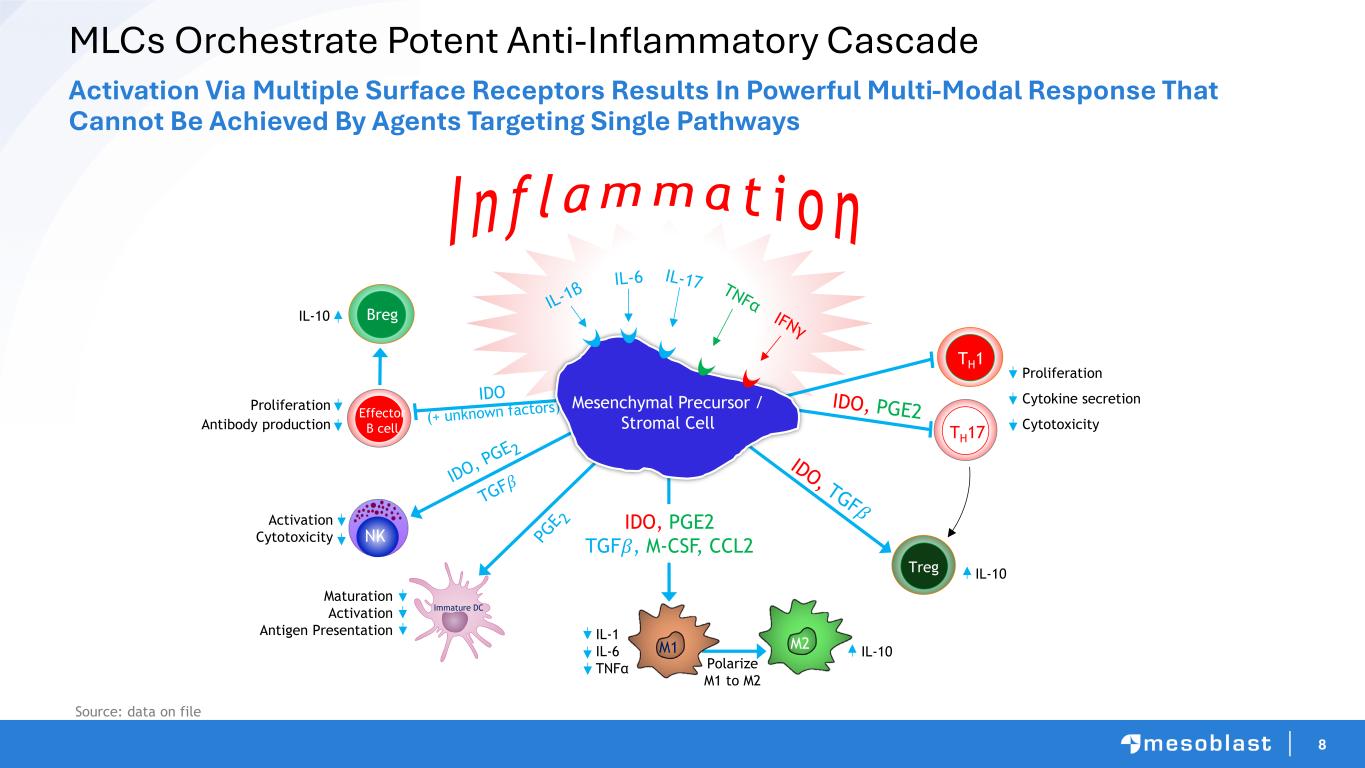
8 MLCs Orchestrate Potent Anti-Inflammatory Cascade Activation Via Multiple Surface Receptors Results In Powerful Multi-Modal Response That Cannot Be Achieved By Agents Targeting Single Pathways Source: data on file Effector B cell M2 Breg Treg I n f l amma t i o n NK Activation Cytotoxicity Maturation Activation Antigen Presentation Proliferation Antibody production IDO, PGE2 TGF𝛽, M-CSF, CCL2 IL-10 TH17 Proliferation Cytokine secretion Cytotoxicity IL-10 TH1 M1 Immature DC IL-1 IL-6 TNFα IL-10 Mesenchymal Precursor / Stromal Cell Polarize M1 to M2
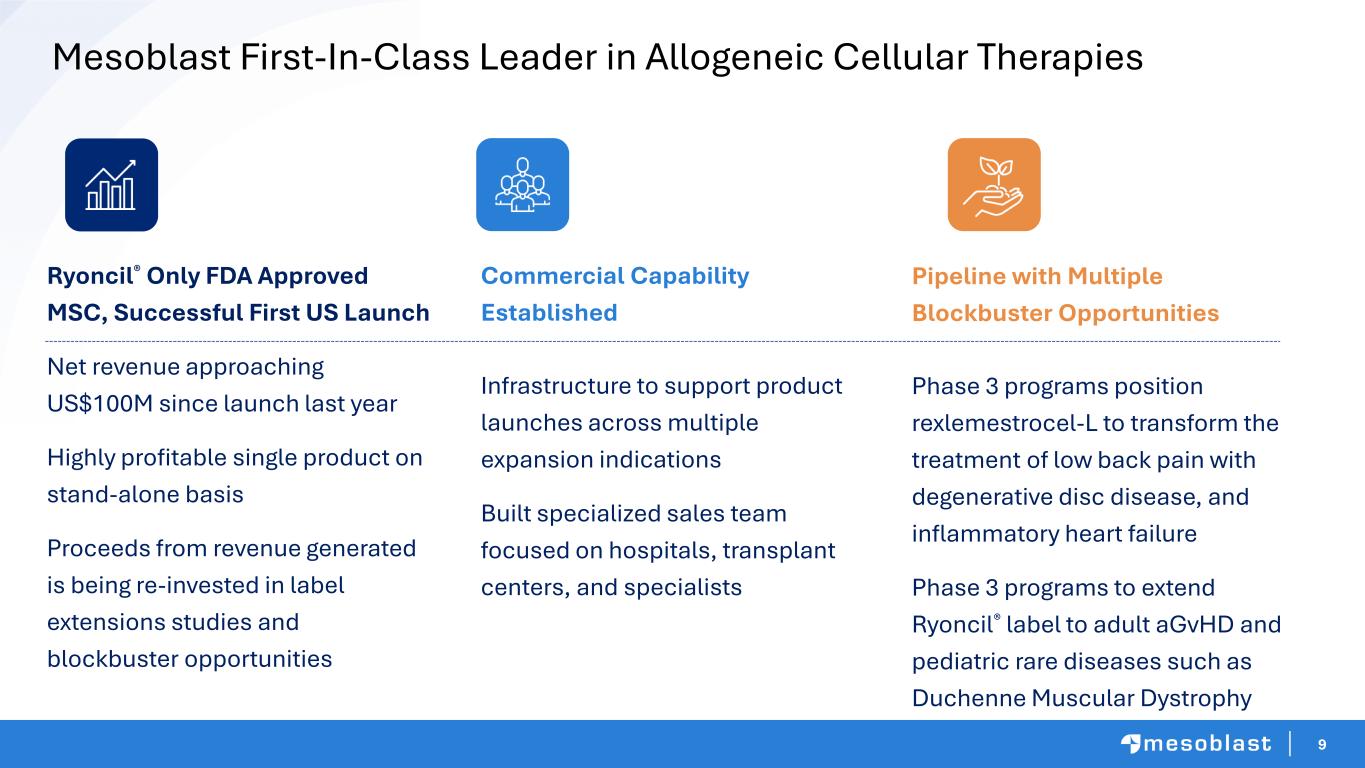
9 Mesoblast First-In-Class Leader in Allogeneic Cellular Therapies Ryoncil® Only FDA Approved MSC, Successful First US Launch Net revenue approaching US$100M since launch last year Highly profitable single product on stand-alone basis Proceeds from revenue generated is being re-invested in label extensions studies and blockbuster opportunities Pipeline with Multiple Blockbuster Opportunities Phase 3 programs position rexlemestrocel-L to transform the treatment of low back pain with degenerative disc disease, and inflammatory heart failure Phase 3 programs to extend Ryoncil® label to adult aGvHD and pediatric rare diseases such as Duchenne Muscular Dystrophy Commercial Capability Established Infrastructure to support product launches across multiple expansion indications Built specialized sales team focused on hospitals, transplant centers, and specialists

10 RYONCIL Profitability to Fund Growth Pipeline Successful U.S. commercial launch of RYONCIL • Q3 FY26 gross revenue US$35.3M, net revenue US$30.3M • RYONCIL net revenue approaching US$100 million since launch last year • RYONCIL gross profit 1H FY26, excluding amortization expense, was US$44.2M. Direct selling costs were US$7.7M Strong operating performance in the period allowed us to invest in: • R&D, including to support the Phase 3 trial on the blockbuster chronic low back pain indication • Clinical programs for lifecycle extension • Commercial manufacturing of Ryoncil® inventory as well as for launch of second- generation product

11 What We Have Built - Commercial Capability ✓ Established commercial infrastructure to support product launches ✓ Built specialized sales and marketing teams focused on hospitals and transplant centers ✓ Implemented market access and payer engagement strategies to support reimbursement and adoption ✓ Established strong manufacturing and supply chain capabilities ensuring scalable, consistent cell therapy production and delivery ✓ Leveraged clinical data and real-world evidence to support product positioning and physician uptake ✓ Established medical affairs and education programs to drive clinician awareness and training ✓ Ensured leadership position is maintained by continued access to cutting-edge technology
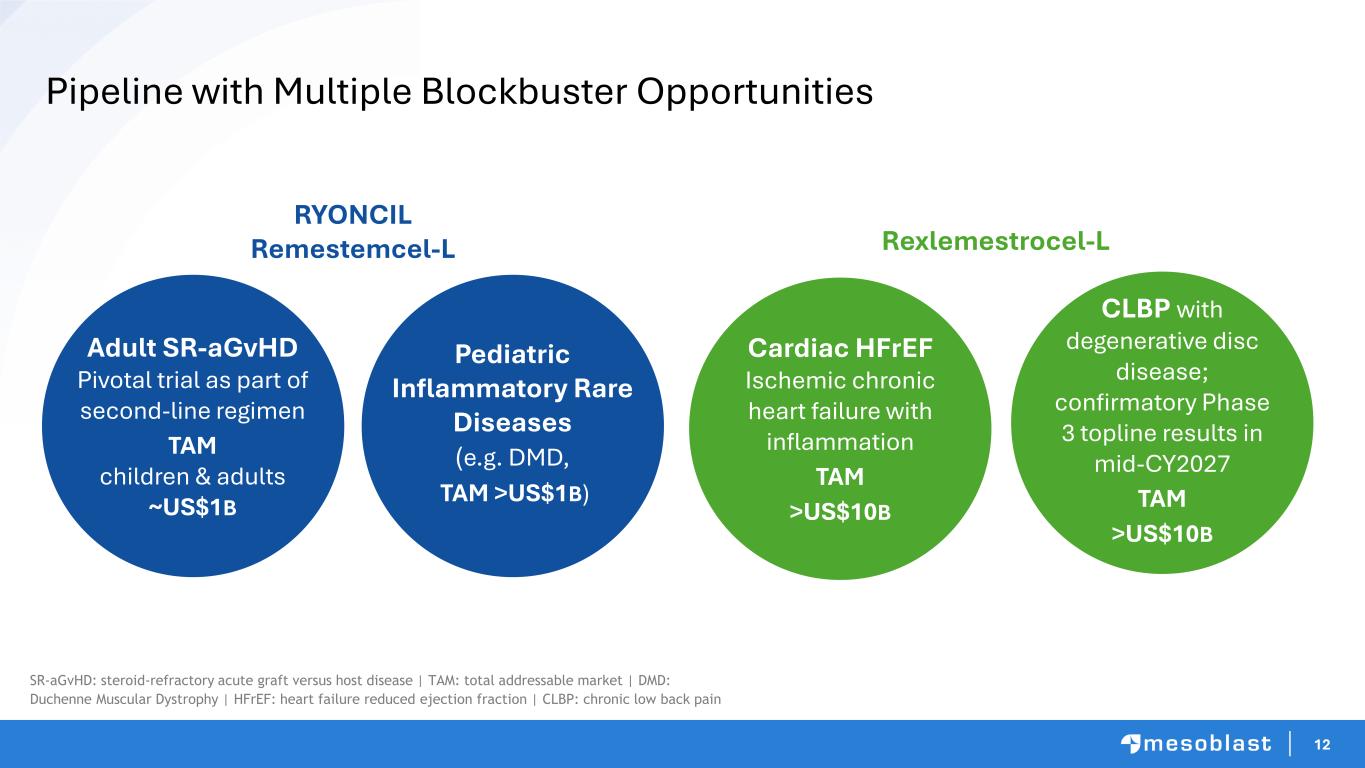
12 Pipeline with Multiple Blockbuster Opportunities Adult SR-aGvHD Pivotal trial as part of second-line regimen TAM children & adults ~US$1B Cardiac HFrEF Ischemic chronic heart failure with inflammation TAM >US$10B CLBP with degenerative disc disease; confirmatory Phase 3 topline results in mid-CY2027 TAM >US$10B RYONCIL Remestemcel-L Rexlemestrocel-L Pediatric Inflammatory Rare Diseases (e.g. DMD, TAM >US$1B) SR-aGvHD: steroid-refractory acute graft versus host disease | TAM: total addressable market | DMD: Duchenne Muscular Dystrophy | HFrEF: heart failure reduced ejection fraction | CLBP: chronic low back pain
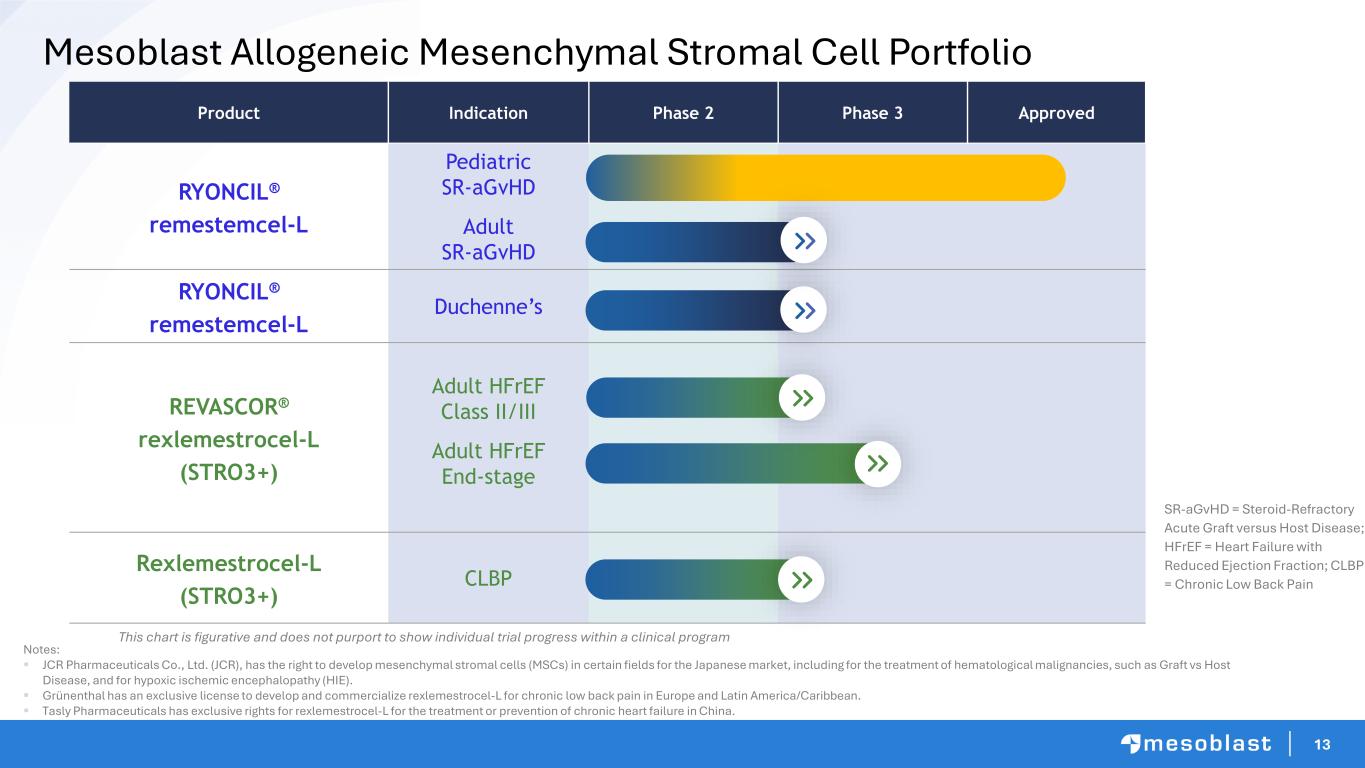
13 Mesoblast Allogeneic Mesenchymal Stromal Cell Portfolio Product Indication Phase 2 Phase 3 Approved RYONCIL® remestemcel-L Pediatric SR-aGvHD Adult SR-aGvHD RYONCIL® remestemcel-L Duchenne’s REVASCOR® rexlemestrocel-L (STRO3+) Adult HFrEF Class II/III Adult HFrEF End-stage Rexlemestrocel-L (STRO3+) CLBP SR-aGvHD = Steroid-Refractory Acute Graft versus Host Disease; HFrEF = Heart Failure with Reduced Ejection Fraction; CLBP = Chronic Low Back Pain This chart is figurative and does not purport to show individual trial progress within a clinical program Notes: ▪ JCR Pharmaceuticals Co., Ltd. (JCR), has the right to develop mesenchymal stromal cells (MSCs) in certain fields for the Japanese market, including for the treatment of hematological malignancies, such as Graft vs Host Disease, and for hypoxic ischemic encephalopathy (HIE). ▪ Grünenthal has an exclusive license to develop and commercialize rexlemestrocel-L for chronic low back pain in Europe and Latin America/Caribbean. ▪ Tasly Pharmaceuticals has exclusive rights for rexlemestrocel-L for the treatment or prevention of chronic heart failure in China.

14 Anticipated Major Upcoming Milestones Blockbuster CLBP • Complete enrollment of pivotal trial end of April • Top-line primary endpoint mid-CY2027 • BLA filing for FDA approval Q3 CY2027 • Potential FDA approval and US launch Q2 CY2028 CHF: regulatory strategy to gain approval for blockbuster indication • File BLA with FDA for end-stage heart failure patients on LVADs this quarter • Leverage approval to initiate confirmatory trial in NYHA II/III HFrEF Label extension for RYONCIL • Adult SR-aGvHD partnered with BMT-CTN network, trial initiated, to complete in 12-18 months • Pediatric Duchenne’s partnered with PPMD advocacy group, Phase 3 IND cleared by FDA SR-aGvHD: steroid-refractory acute graft versus host disease | CHF: chronic heart failure | BLA: Biologics License Application | LVAD: left ventricular assist device | HFrEF: heart failure reduced ejection fraction | NYHA: New York Heart Association | CLBP: chronic low back pain | PPMD: Parent Project Muscular Dystrophy
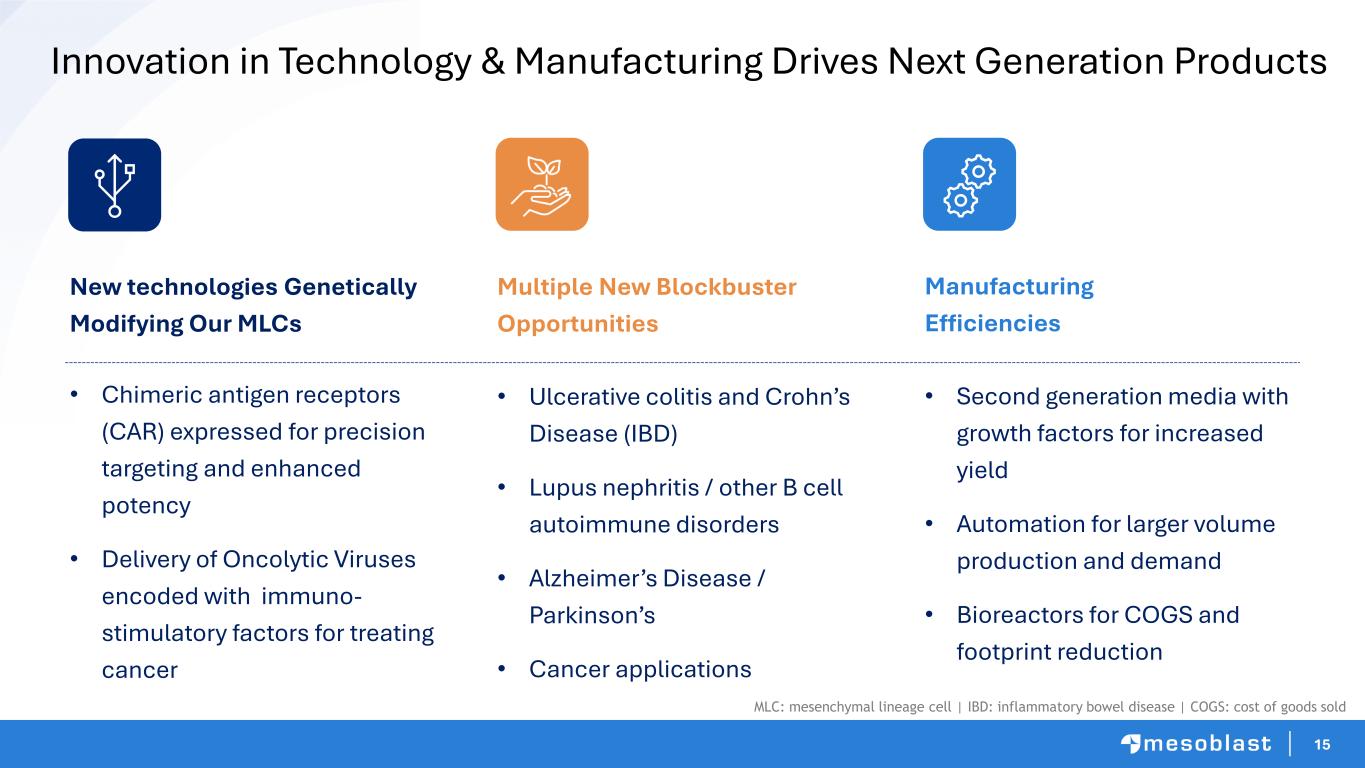
15 Innovation in Technology & Manufacturing Drives Next Generation Products New technologies Genetically Modifying Our MLCs • Chimeric antigen receptors (CAR) expressed for precision targeting and enhanced potency • Delivery of Oncolytic Viruses encoded with immuno- stimulatory factors for treating cancer Multiple New Blockbuster Opportunities • Ulcerative colitis and Crohn’s Disease (IBD) • Lupus nephritis / other B cell autoimmune disorders • Alzheimer’s Disease / Parkinson’s • Cancer applications Manufacturing Efficiencies • Second generation media with growth factors for increased yield • Automation for larger volume production and demand • Bioreactors for COGS and footprint reduction MLC: mesenchymal lineage cell | IBD: inflammatory bowel disease | COGS: cost of goods sold

16 • MSCs safe and effective treatments to transform outcomes • First-in-class platform technology approved by FDA, unlocks many opportunities • Massive barriers to entry include dominant IP, clinical trial outcomes, robust manufacturing, proprietary potency assays and FDA approval • Well cashed-up, profits from RYONCIL being re-invested into follow-on opportunities • Blockbuster potential with major near-term milestones with back pain and heart failure products • Innovative next generation technologies to genetically modify our cells provide potential for even greater efficacy Key Takeaways

Pediatric Steroid Refractory Acute Graft versus Host Disease Marcelo Santoro Chief Commercial Officer

18 Redefining What’s Possible for SR-aGvHD Children and Their Loved Ones

19 Successful First Year of RYONCIL • Ryoncil is transforming outcomes in pediatric SR-aGvHD, with strong positive feedback from treating physicians • Commercial uptake has exceeded expectations, outperforming benchmarks from comparable successful launches • We have established the capability to scale, with a clear path to doubling our US revenue from RYONCIL • Adult indication represents a major opportunity, representing a 3-fold increase in TAM and revenue potential
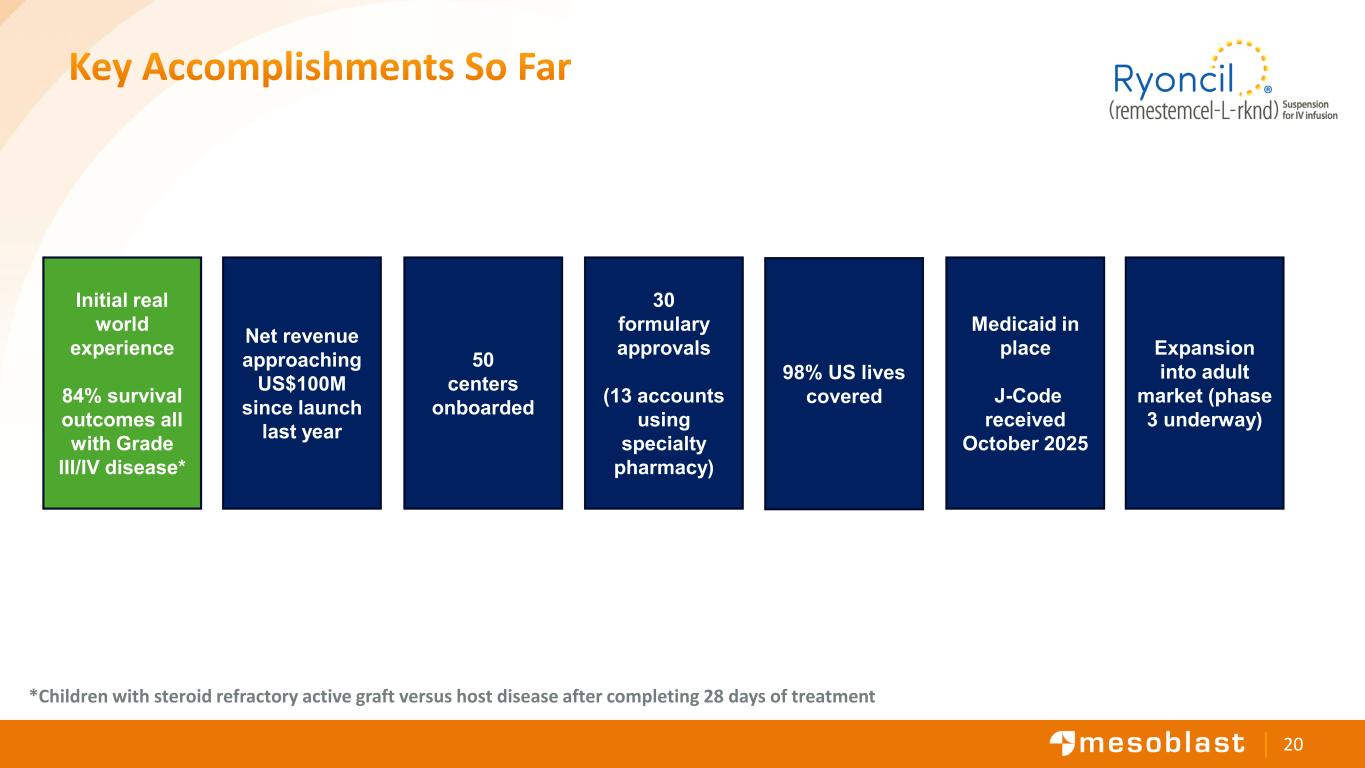
20 Key Accomplishments So Far 98% US lives covered Medicaid in place J-Code received October 2025 50 centers onboarded Net revenue approaching US$100M since launch last year Initial real world experience 84% survival outcomes all with Grade III/IV disease* 30 formulary approvals (13 accounts using specialty pharmacy) Expansion into adult market (phase 3 underway) *Children with steroid refractory active graft versus host disease after completing 28 days of treatment
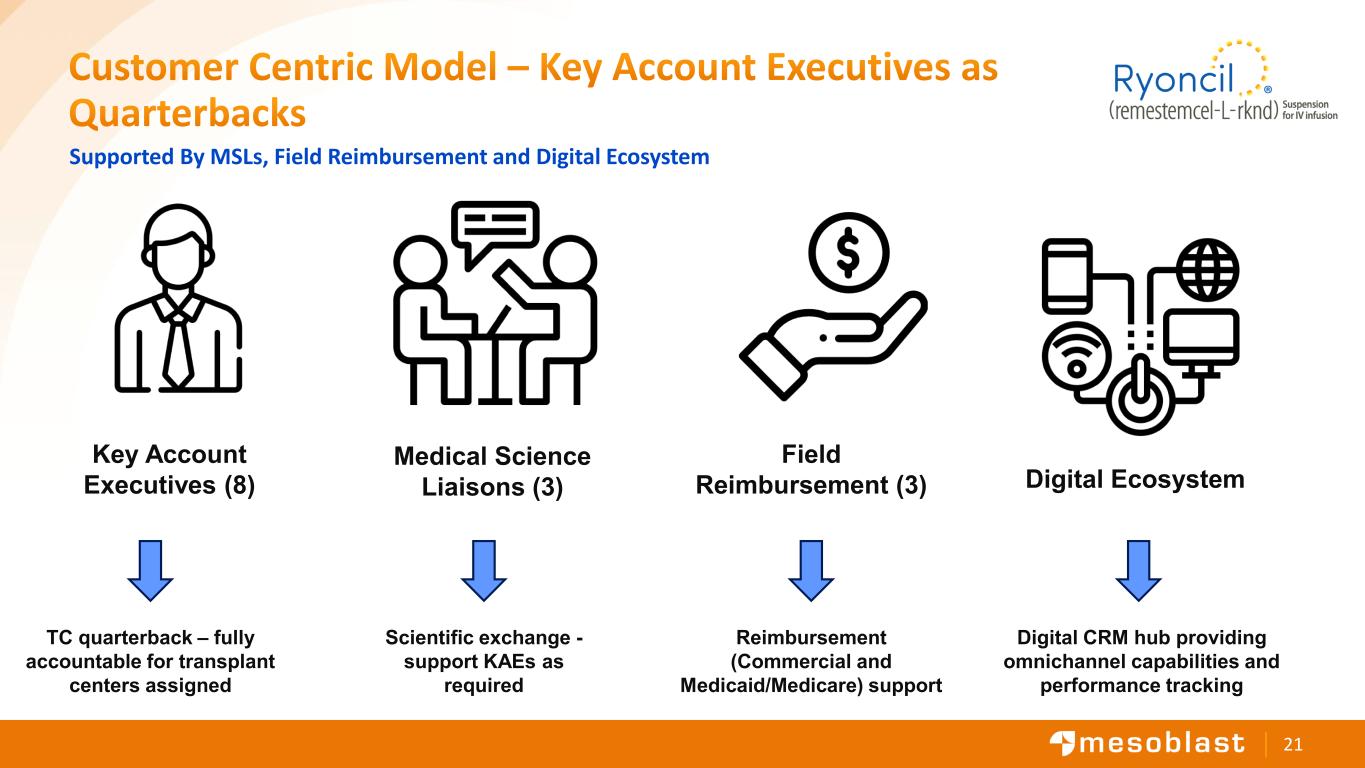
21 Customer Centric Model – Key Account Executives as Quarterbacks Supported By MSLs, Field Reimbursement and Digital Ecosystem Digital Ecosystem Key Account Executives (8) TC quarterback – fully accountable for transplant centers assigned Medical Science Liaisons (3) Scientific exchange - support KAEs as required Digital CRM hub providing omnichannel capabilities and performance tracking Field Reimbursement (3) Reimbursement (Commercial and Medicaid/Medicare) support
22 Revenue Growth Plan Next target: Doubling net revenue Approaching US$100M net revenue since launch Expand Ryoncil utilization across all 64 targeted centers, driving breadth and depth Ryoncil available at the site of care for immediate dispensing Broader Ryoncil utilization in first line after steroids

23 Three Strategic Priorities for Continued Growth in FY 2027 Empower caregivers to demand Ryoncil® for their children Reinforce superior patient outcomes in first-line Proactively identify and prioritize appropriate patients

24 Key Takeaways • Approaching US$100M in net revenues since launch last year, strategy in place to double revenue • TAM for children and adults in SR-aGvHD alone is >US$1.0 billion • Leveraging the commercial capability to support RYONCIL label extensions • Opportunity to expand ex-US at US Most Favored Nation pricing

25 Ryoncil in Adult GvHD Michael Schuster Head of Business Development
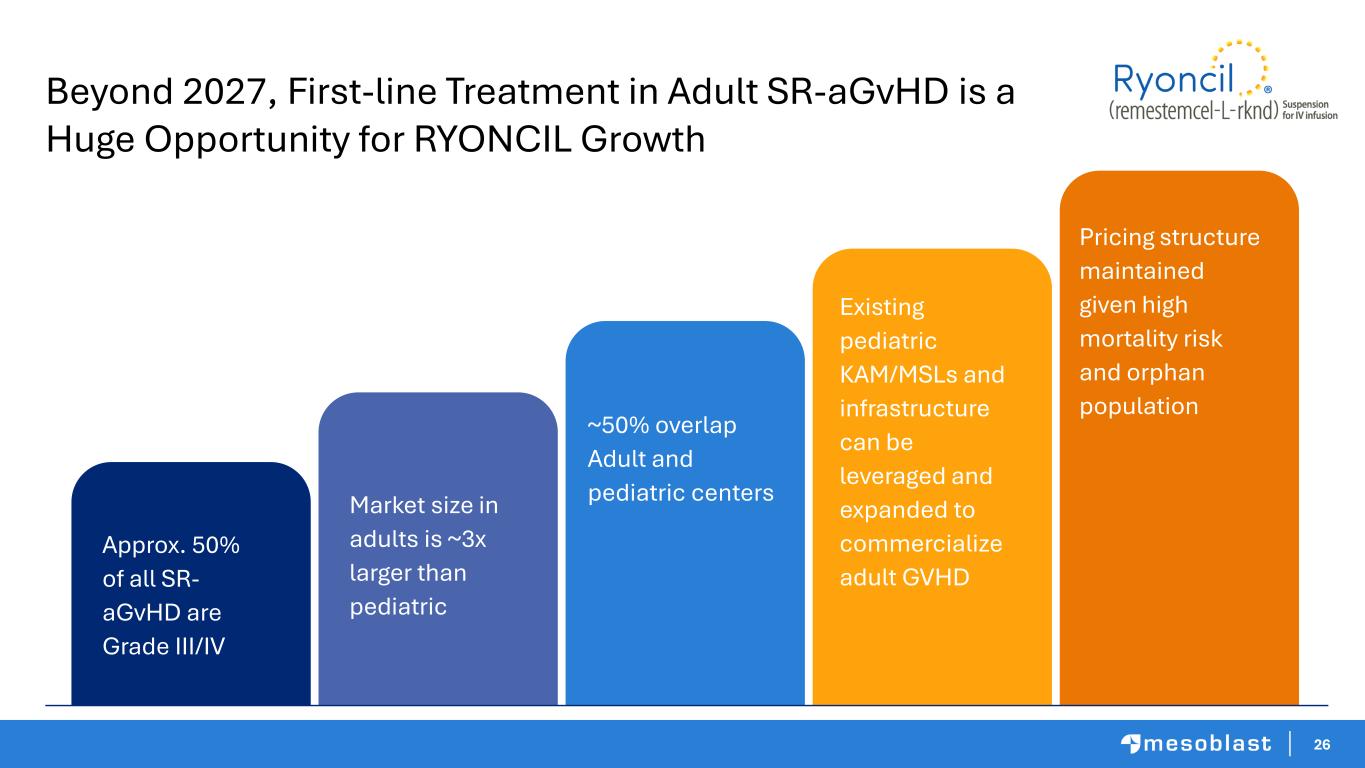
26 Beyond 2027, First-line Treatment in Adult SR-aGvHD is a Huge Opportunity for RYONCIL Growth Approx. 50% of all SR- aGvHD are Grade III/IV Market size in adults is ~3x larger than pediatric ~50% overlap Adult and pediatric centers Existing pediatric KAM/MSLs and infrastructure can be leveraged and expanded to commercialize adult GVHD Pricing structure maintained given high mortality risk and orphan population
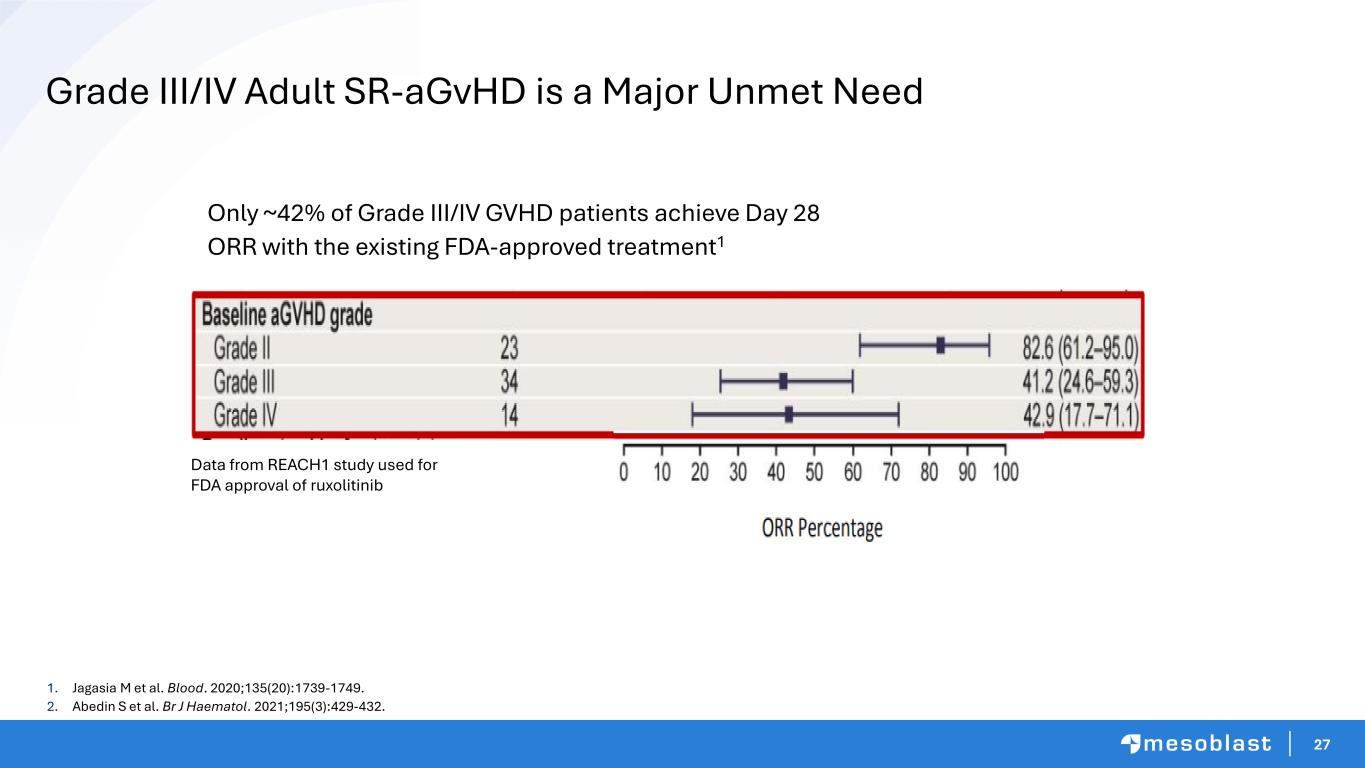
27 1. Jagasia M et al. Blood. 2020;135(20):1739-1749. 2. Abedin S et al. Br J Haematol. 2021;195(3):429-432. Only ~42% of Grade III/IV GVHD patients achieve Day 28 ORR with the existing FDA-approved treatment1 Data from REACH1 study used for FDA approval of ruxolitinib Grade III/IV Adult SR-aGvHD is a Major Unmet Need
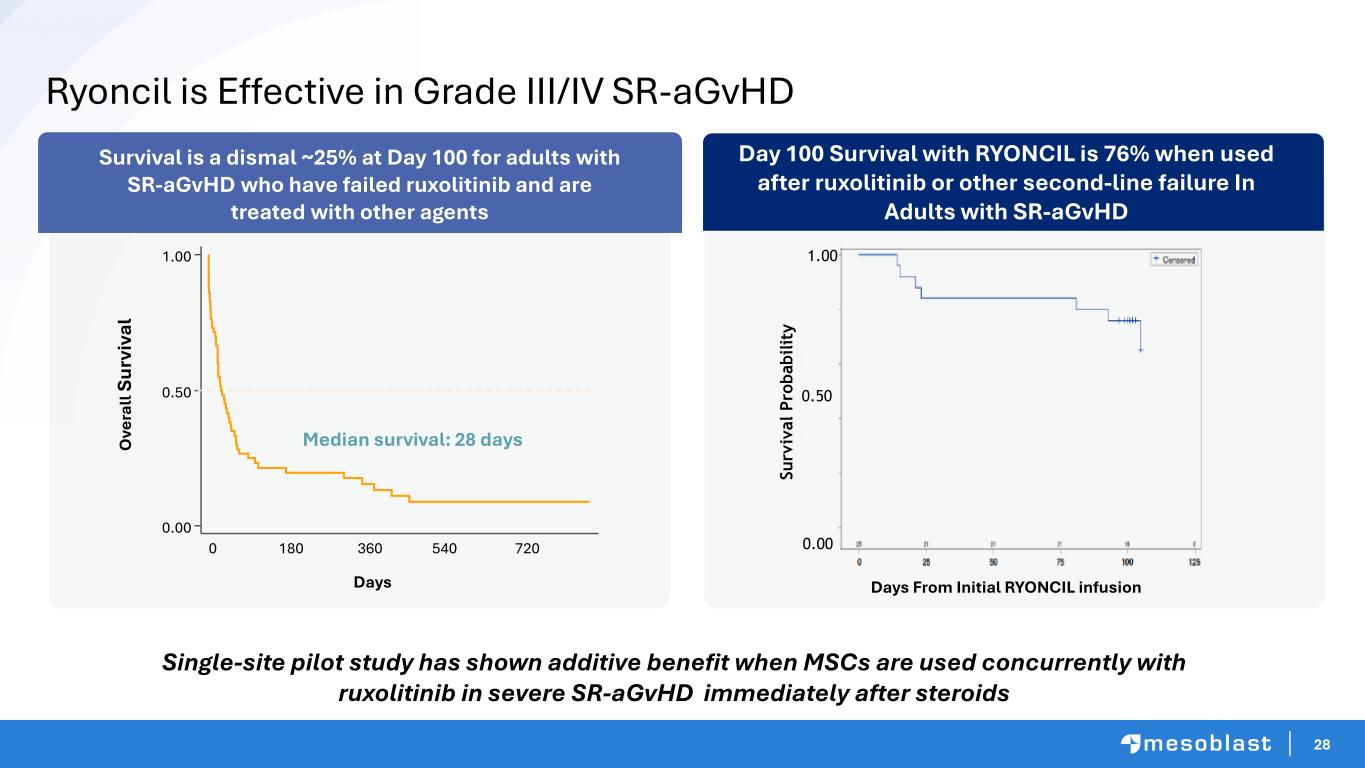
28 Day 100 Survival with RYONCIL is 76% when used after ruxolitinib or other second-line failure In Adults with SR-aGvHD 0 180 540 720 Days 360 0.00 0.50 1.00 O ve ra ll Su rv iv al Median survival: 28 days Days From Initial RYONCIL infusion S u rv iv a l P ro b a b il it y 0.00 0.50 1.00 Ryoncil is Effective in Grade III/IV SR-aGvHD Survival is a dismal ~25% at Day 100 for adults with SR-aGvHD who have failed ruxolitinib and are treated with other agents Single-site pilot study has shown additive benefit when MSCs are used concurrently with ruxolitinib in severe SR-aGvHD immediately after steroids
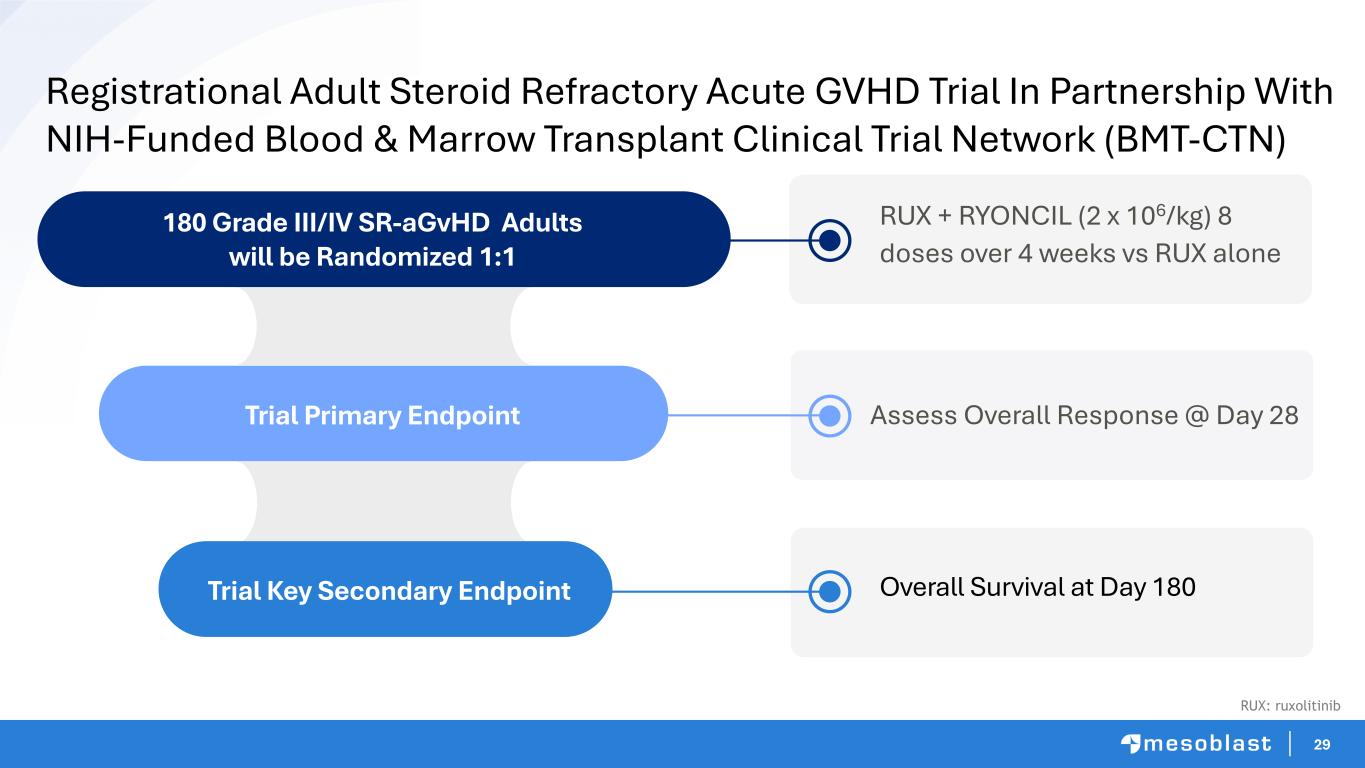
29 Registrational Adult Steroid Refractory Acute GVHD Trial In Partnership With NIH-Funded Blood & Marrow Transplant Clinical Trial Network (BMT-CTN) 180 Grade III/IV SR-aGvHD Adults will be Randomized 1:1 Trial Key Secondary Endpoint Trial Primary Endpoint Assess Overall Response @ Day 28 RUX + RYONCIL (2 x 106/kg) 8 doses over 4 weeks vs RUX alone Overall Survival at Day 180 RUX: ruxolitinib

30 • Mesoblast will oversee trial execution using NIH-sponsored BMT-CTN network of major hospitals • DSMB approval and NMDP Central IRB clearance • Patients to be recruited across 45 U.S. centers (representing ≈5,000 annual allo transplants) • First sites to be activated this quarter • Expected duration 18 months, with interim analysis at 12 months Adult SR-aGvHD Trial Timelines Interim analysis planned for early success when first 102 patients (57% enrolled) have reached Day 28 (primary endpoint) 57% BMT-CTN: Blood & Marrow Transplant Clinical Trial Network | NIH: National Institutes of Health | DSMB: Data and Safety Monitoring Board | IRB: Institutional Review Board | allo: allogeneic

31 • Partnership with the BMT-CTN consortium will subsidize cost and create end- user awareness for market adoption • Potential to shorten time to launch by six months through successful interim analysis • Label extension in adult SR-aGvHD maintains existing Ryoncil pricing structure • Leverages existing commercial infrastructure for pediatric • Adult SR-aGvHD represents a market opportunity 3x that of pediatric SR-aGVHD Label Expansion into Adult GvHD Provides Significant Revenue Upside

32 32 KOL Perspective Susan Prockop, MD Dana Farber Cancer Institute/Boston Children’s Hospital Director, Clinical and Translational Research Stem Cell Transplant Program, DFCI/BCH Cancer and Blood Disorders Center Adult aGvHD KOL Perspective Joanne Kurtzberg, MD Duke University School of Medicine Jerome Harris Distinguished Professor of Pediatrics Professor of Pathology Duke University School of Medicine

33 Jess, this is the commercial section – per our call: • Orange bar on the bottom – pls keep for all slides in this section • Header text: please keep the same color for each slide • Logos: please keep as placed (even if a duplicate on the slides) • Slide 16 icons: ok to drop in size • Slide 17-18: please keep same box colors • Slide 19: glad for an alternative image • These notes apply to slides 13 – 23 • Ok to swap the font Rexlemestrocel-L for Chronic Lower Back Pain Roger Brown Head of Musculoskeletal

34 Rexlemestrocel-L for CLBP: Huge Commercial Opportunity ~35M patients in US suffer from CLBP of which ~60% is due to degenerative disc disease (DDD) ~7M US patients have moderate / severe DDD within 5 years of diagnosis that is refractory to medical therapies including opioids P3 trial results to confirm first trial pain reduction at 12 months expected mid- CY2027 Potential approval following Priority Review Q2 CY2028 10% of prevalent market penetration represents US$14B per year in product sales Filing BLA with FDA in Q3 CY2027 Substantial Unmet Need Timelines to CLBP Commercial Launch
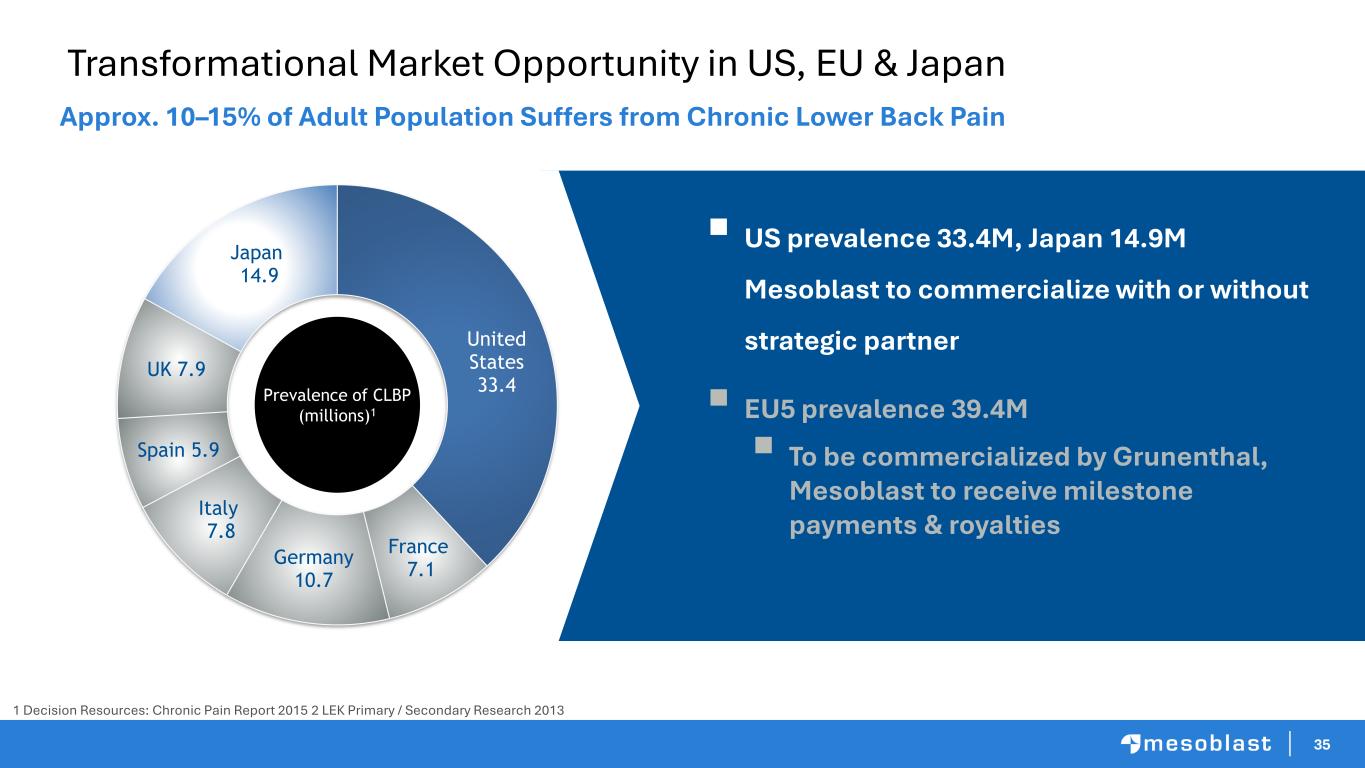
35 Transformational Market Opportunity in US, EU & Japan Approx. 10–15% of Adult Population Suffers from Chronic Lower Back Pain United States 33.4 France 7.1 Germany 10.7 Italy 7.8 Spain 5.9 UK 7.9 Japan 14.9 Prevalence of CLBP (millions)1 ▪ US prevalence 33.4M, Japan 14.9M Mesoblast to commercialize with or without strategic partner ▪ EU5 prevalence 39.4M ▪ To be commercialized by Grunenthal, Mesoblast to receive milestone payments & royalties 1 Decision Resources: Chronic Pain Report 2015 2 LEK Primary / Secondary Research 2013
36 McCann MR and Seguin CA. Notochord Cells in Intervertebral Disc Development and Degeneration. J. Dev. Biol. 2016, 4(1), 3 Inflammation is at the Core of Pain in Degenerative Disc Disease
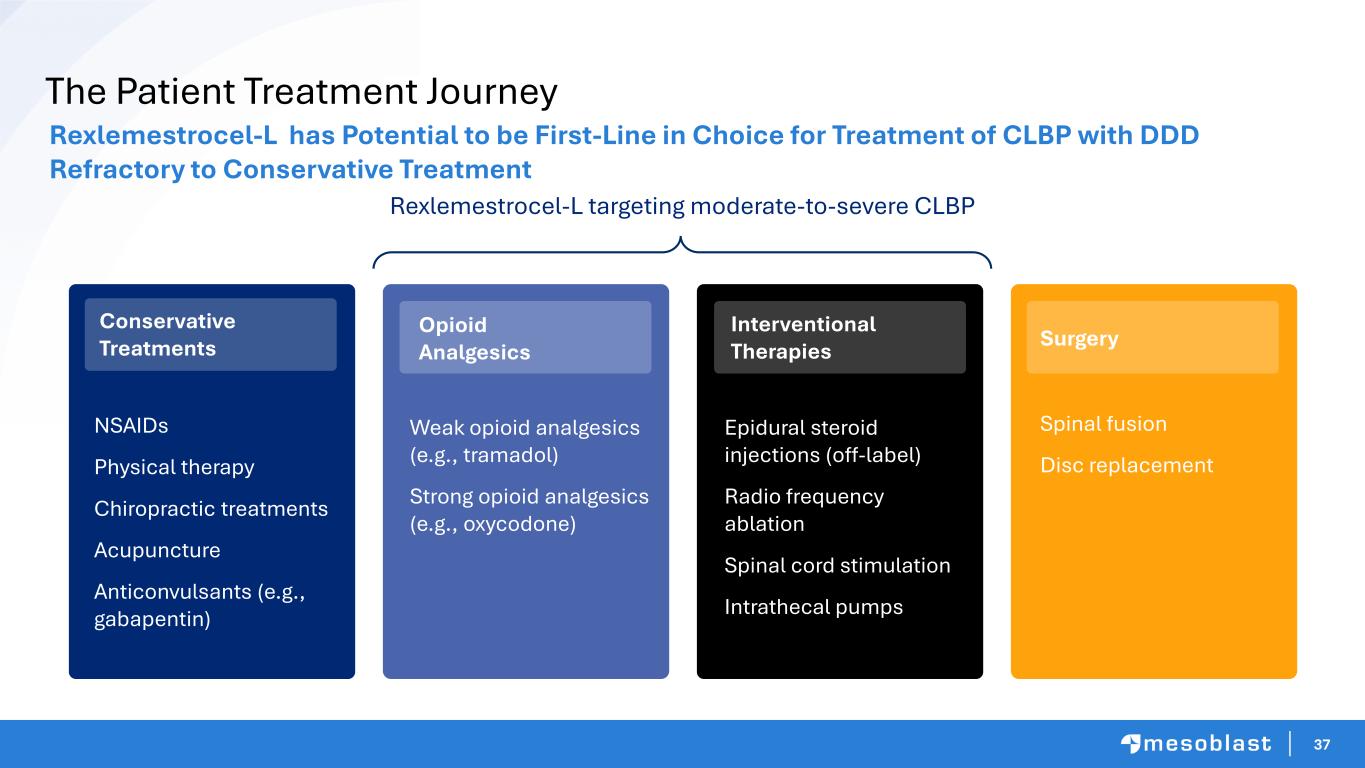
37 The Patient Treatment Journey Rexlemestrocel-L has Potential to be First-Line in Choice for Treatment of CLBP with DDD Refractory to Conservative Treatment Rexlemestrocel-L targeting moderate-to-severe CLBP NSAIDs Physical therapy Chiropractic treatments Acupuncture Anticonvulsants (e.g., gabapentin) Epidural steroid injections (off-label) Radio frequency ablation Spinal cord stimulation Intrathecal pumps Conservative Treatments Opioid Analgesics Weak opioid analgesics (e.g., tramadol) Strong opioid analgesics (e.g., oxycodone) Interventional Therapies Surgery Spinal fusion Disc replacement
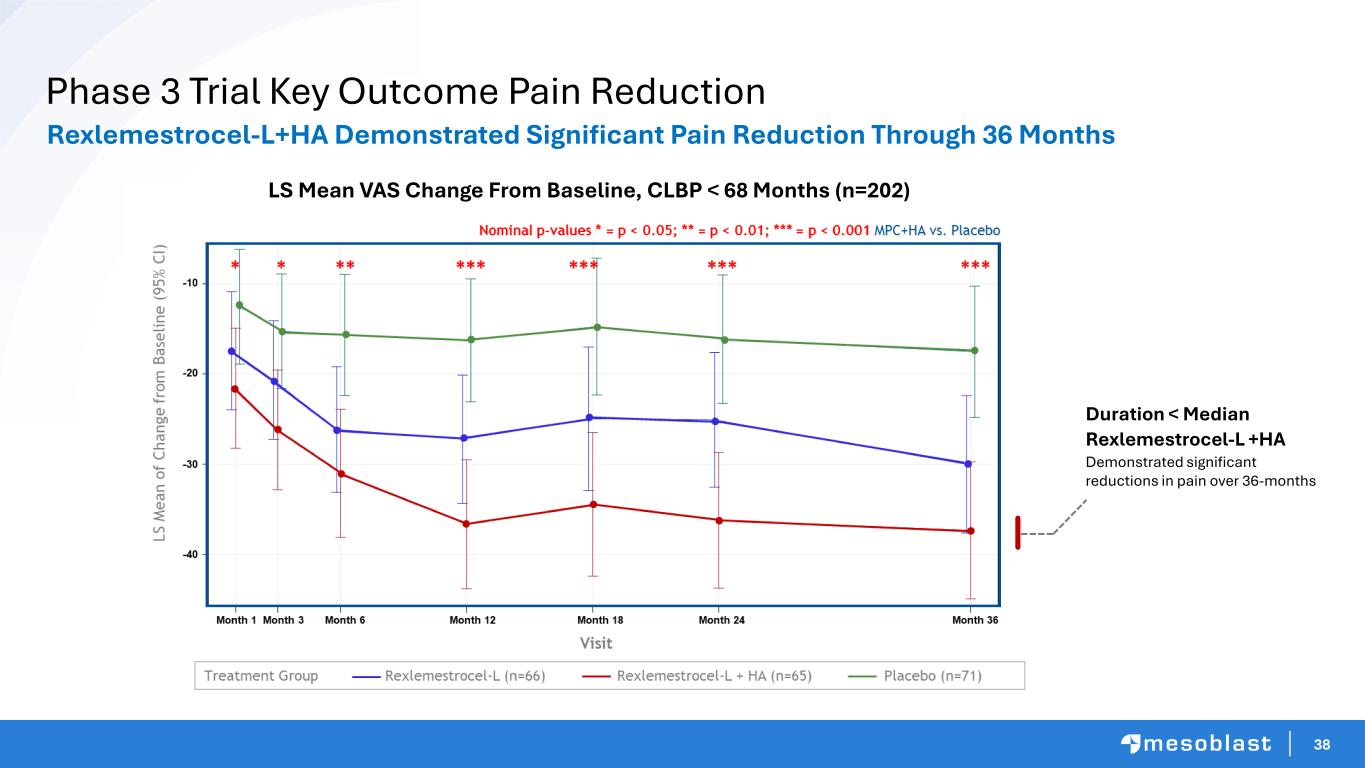
38 Phase 3 Trial Key Outcome Pain Reduction Duration < Median Rexlemestrocel-L +HA Demonstrated significant reductions in pain over 36-months LS Mean VAS Change From Baseline, CLBP < 68 Months (n=202) Rexlemestrocel-L+HA Demonstrated Significant Pain Reduction Through 36 Months
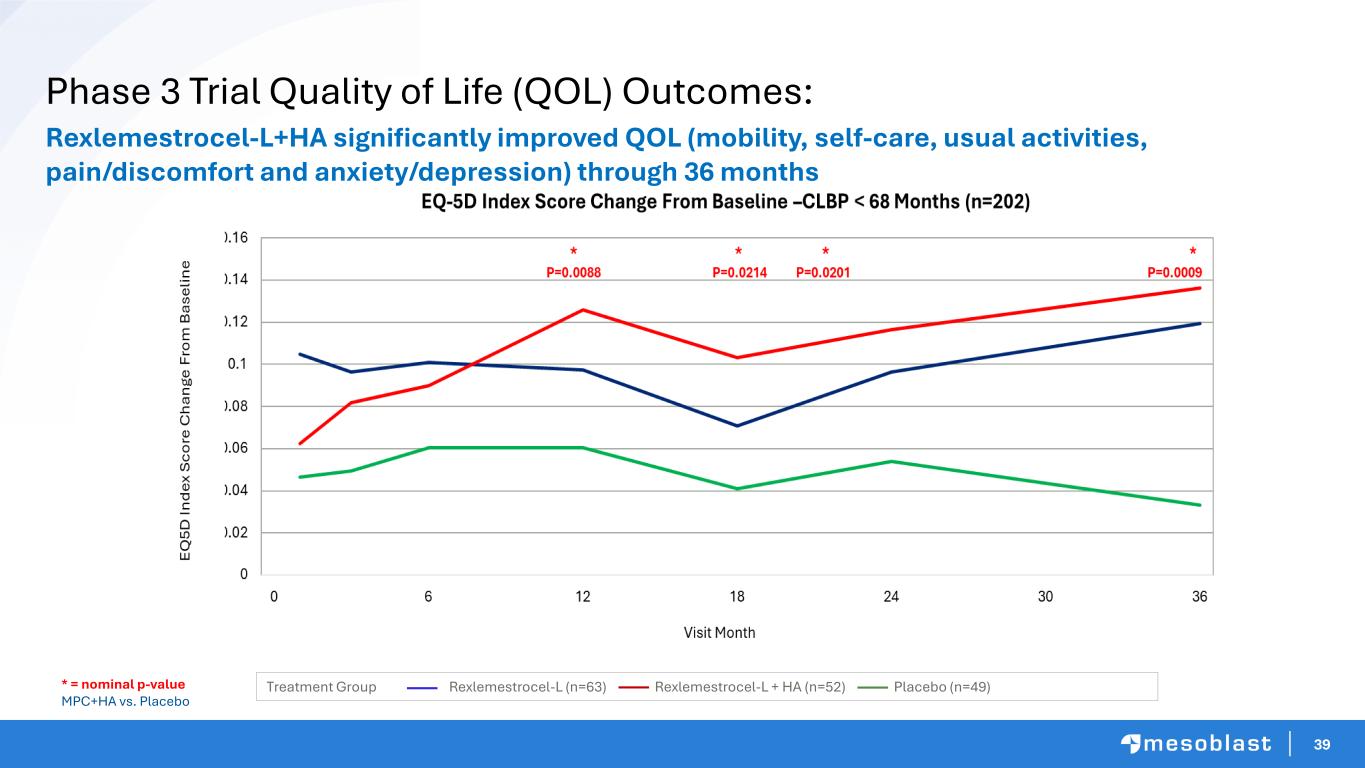
39 Phase 3 Trial Quality of Life (QOL) Outcomes: Rexlemestrocel-L+HA significantly improved QOL (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) through 36 months * = nominal p-value MPC+HA vs. Placebo Treatment Group Rexlemestrocel-L (n=63) Rexlemestrocel-L + HA (n=52) Placebo (n=49)
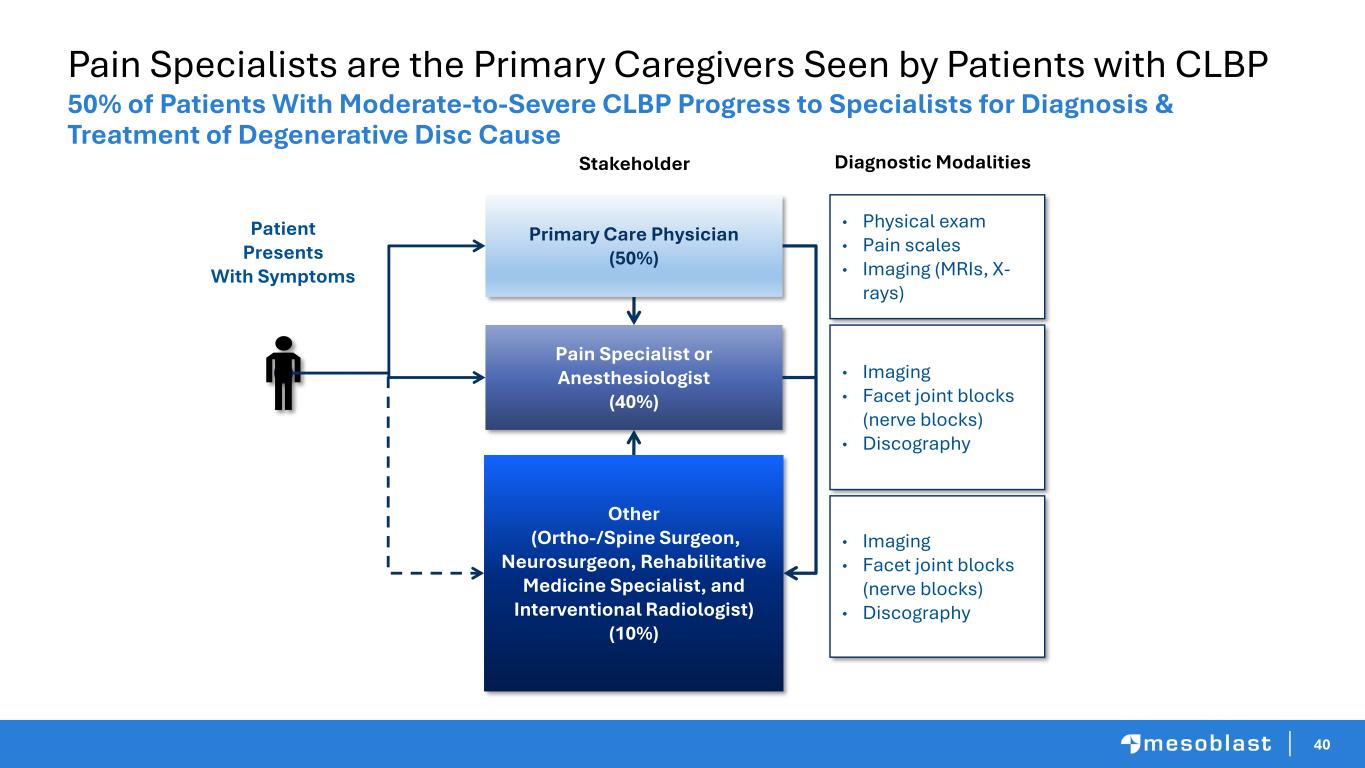
40 Pain Specialists are the Primary Caregivers Seen by Patients with CLBP 50% of Patients With Moderate-to-Severe CLBP Progress to Specialists for Diagnosis & Treatment of Degenerative Disc Cause Primary Care Physician (50%) Other (Ortho-/Spine Surgeon, Neurosurgeon, Rehabilitative Medicine Specialist, and Interventional Radiologist) (10%) Patient Presents With Symptoms Pain Specialist or Anesthesiologist (40%) • Physical exam • Pain scales • Imaging (MRIs, X- rays) • Imaging • Facet joint blocks (nerve blocks) • Discography • Imaging • Facet joint blocks (nerve blocks) • Discography Stakeholder Diagnostic Modalities
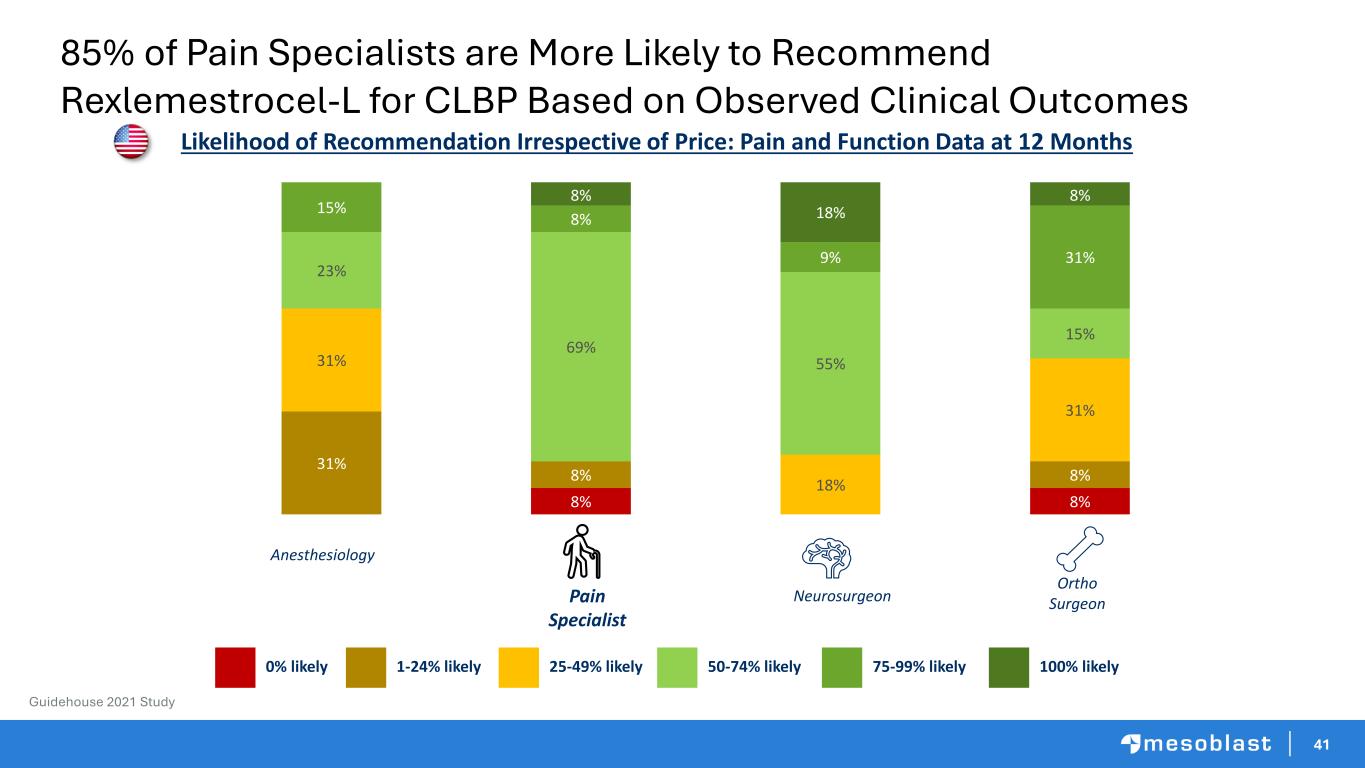
41 Pain Specialist Neurosurgeon Ortho Surgeon 8% 8% 31% 8% 8% 31% 18% 31% 23% 69% 55% 15% 15% 8% 9% 31% 8% 18% 8% Likelihood of Recommendation Irrespective of Price: Pain and Function Data at 12 Months 50-74% likely25-49% likely1-24% likely0% likely 75-99% likely 100% likely Anesthesiology Guidehouse 2021 Study 85% of Pain Specialists are More Likely to Recommend Rexlemestrocel-L for CLBP Based on Observed Clinical Outcomes
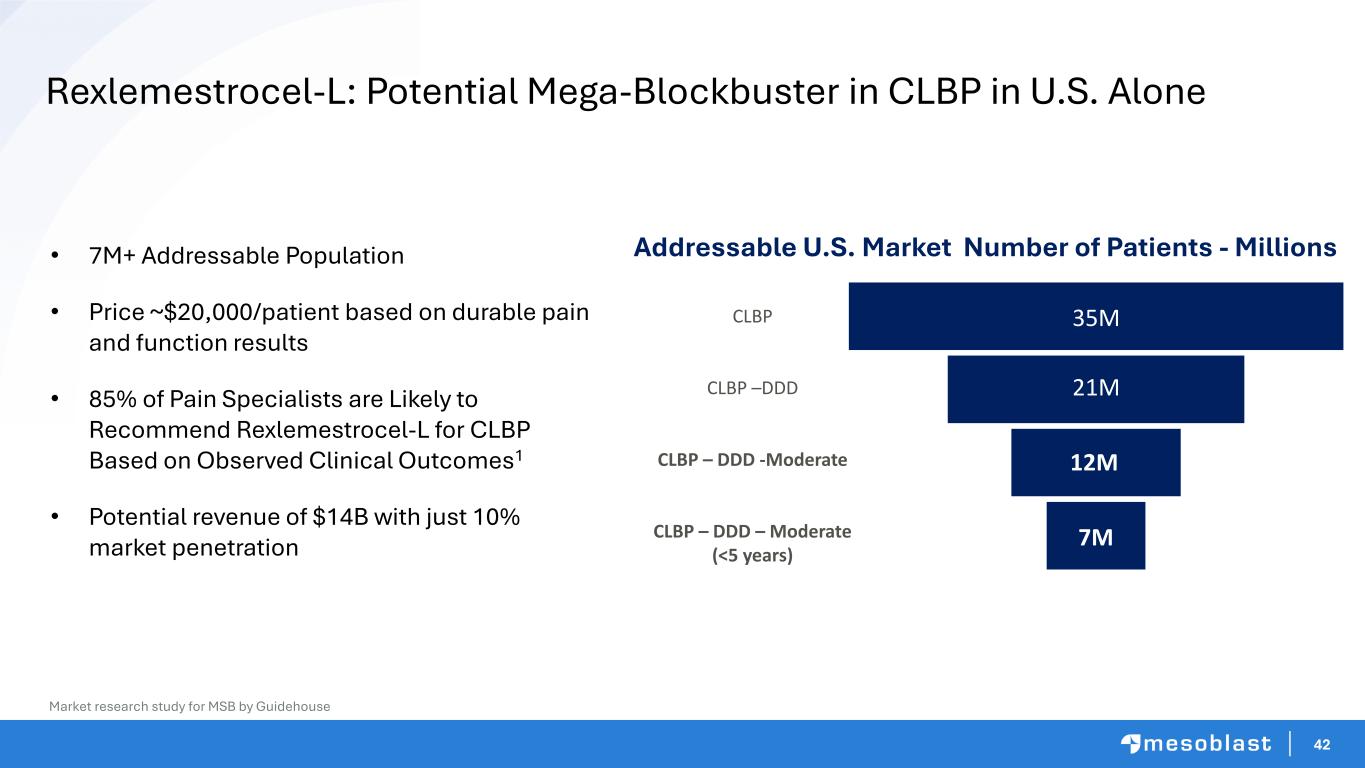
42 • 7M+ Addressable Population • Price ~$20,000/patient based on durable pain and function results • 85% of Pain Specialists are Likely to Recommend Rexlemestrocel-L for CLBP Based on Observed Clinical Outcomes1 • Potential revenue of $14B with just 10% market penetration Market research study for MSB by Guidehouse Rexlemestrocel-L: Potential Mega-Blockbuster in CLBP in U.S. Alone Addressable U.S. Market Number of Patients - Millions 35M 21M 12M 7M CLBP CLBP –DDD CLBP – DDD -Moderate CLBP – DDD – Moderate (<5 years)

43 Launch timeline for rexlemestrocel-L in CLBP 300 Patient 1:1 Placebo-Controlled Phase 3 Pivotal Trial in DDD Completes Enrollment 12 Month Primary Endpoint of Pain Reduction Top-Line Results BLA Submission to FDA Potential Approval Following Priority Review APR 2026 Mid CY2027 Q3 CY2027 Q2 CY2028

44 Go To Market - Phased Launch Based on Milestones Achievement Higher investments Lower investments At Launch - Focus on Main Accounts and Limited Consumer Spend • Prioritize and focus on main accounts (pain managers) representing 70%+ of the volume • Limited primary care engagement – digital only • Limited consumer engagement – digital only • Limited contracting with payers and GPOs Phase 2 – Expand to all Accounts and Increase Consumer Spend • Upon achievement of milestones, increase depth and breadth to all accounts (pain managers) – 100% of volume • Target selected primary care offices and expand reach to all primary care physicians online • Expand consumer engagement – omnichannel • Increase contracting with payers and GPOs Phase 3 – Direct to Consumer (DTC) • Upon achievement of milestones, deploy DTC campaign to accelerate uptake • Maintain depth and breadth to all accounts - 100% of volume (pain managers and selected primary care offices) • Optimize contracting with payers and GPOs

4545 Doug P. Beall, MD Clinical Investigations LLC Chief of Services, Comprehensive Specialty Care; Director of Research Clinical Investigations LLC Chronic Lower Back Pain KOL Perspective

4646 Chronic Heart Failure/LVAD Ken Borow Head of Cardiovascular

47 Chronic Heart Failure: Rising Incidence with High Morbidity and Mortality • Cardiovascular (CV) disease associated with inflammation is the leading cause of death in the U.S.1 • Heart failure (HF) in the United States is projected to affect >7 million adults by the year 2030. Approximately 50% of these people will have heart failure with reduced ejection fraction (HFrEF)2 • Chronic heart failure is a progressive disease with mortality that approaches 50% at 5 years and at least 75% after an initial hospitalization2-4 1. Muntner BEJ, et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation. Feb 19, 2019. 2. United States Food & Drug Administration. Treatment for Heart Failure: Endpoints for Drug Development. Draft Guidance. June 2019. 3. Taylor CJ, et al. Trends in survival after a diagnosis of heart failure in the United Kingdom 2000-2017: population-based cohort study. BMJ. 2019;364:I223. 4. Shah KS, et al. Heart Failure with Preserve, Borderline, and Reduced Ejection Fraction; 5-Year Outcomes. JACC. 2017;Nov12. 47
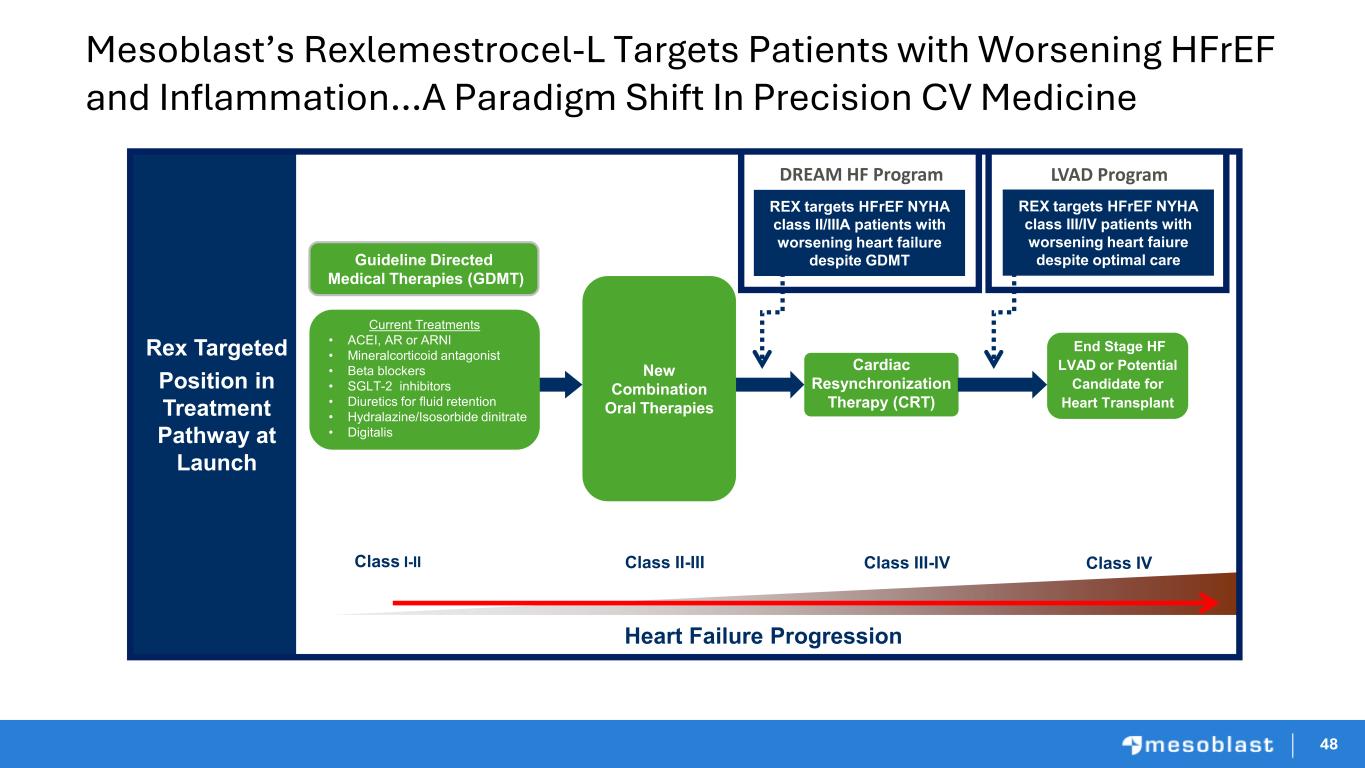
48 Mesoblast’s Rexlemestrocel-L Targets Patients with Worsening HFrEF and Inflammation…A Paradigm Shift In Precision CV Medicine Rex Targeted Position in Treatment Pathway at Launch Heart Failure Progression Class I-II Class IV Current Treatments • ACEI, AR or ARNI • Mineralcorticoid antagonist • Beta blockers • SGLT-2 inhibitors • Diuretics for fluid retention • Hydralazine/Isosorbide dinitrate • Digitalis Cardiac Resynchronization Therapy (CRT) End Stage HF LVAD or Potential Candidate for Heart Transplant Guideline Directed Medical Therapies (GDMT) New Combination Oral Therapies REX targets HFrEF NYHA class II/IIIA patients with worsening heart failure despite GDMT REX targets HFrEF NYHA class III/IV patients with worsening heart faiure despite optimal care Class II-III Class III-IV DREAM HF Program LVAD Program 48
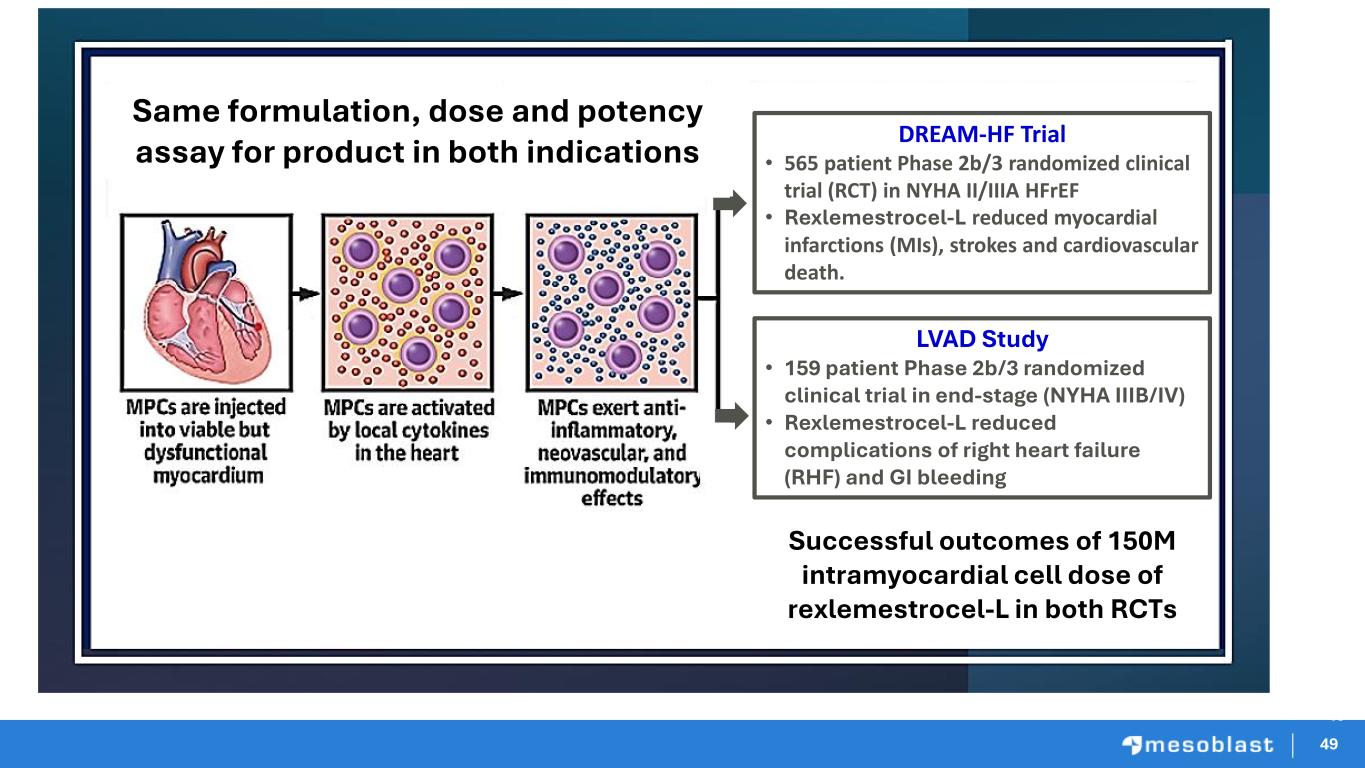
49 49 Successful outcomes of 150M intramyocardial cell dose of rexlemestrocel-L in both RCTs DREAM-HF Trial • 565 patient Phase 2b/3 randomized clinical trial (RCT) in NYHA II/IIIA HFrEF • Rexlemestrocel-L reduced myocardial infarctions (MIs), strokes and cardiovascular death. LVAD Study • 159 patient Phase 2b/3 randomized clinical trial in end-stage (NYHA IIIB/IV) • Rexlemestrocel-L reduced complications of right heart failure (RHF) and GI bleeding 49 Same formulation, dose and potency assay for product in both indications
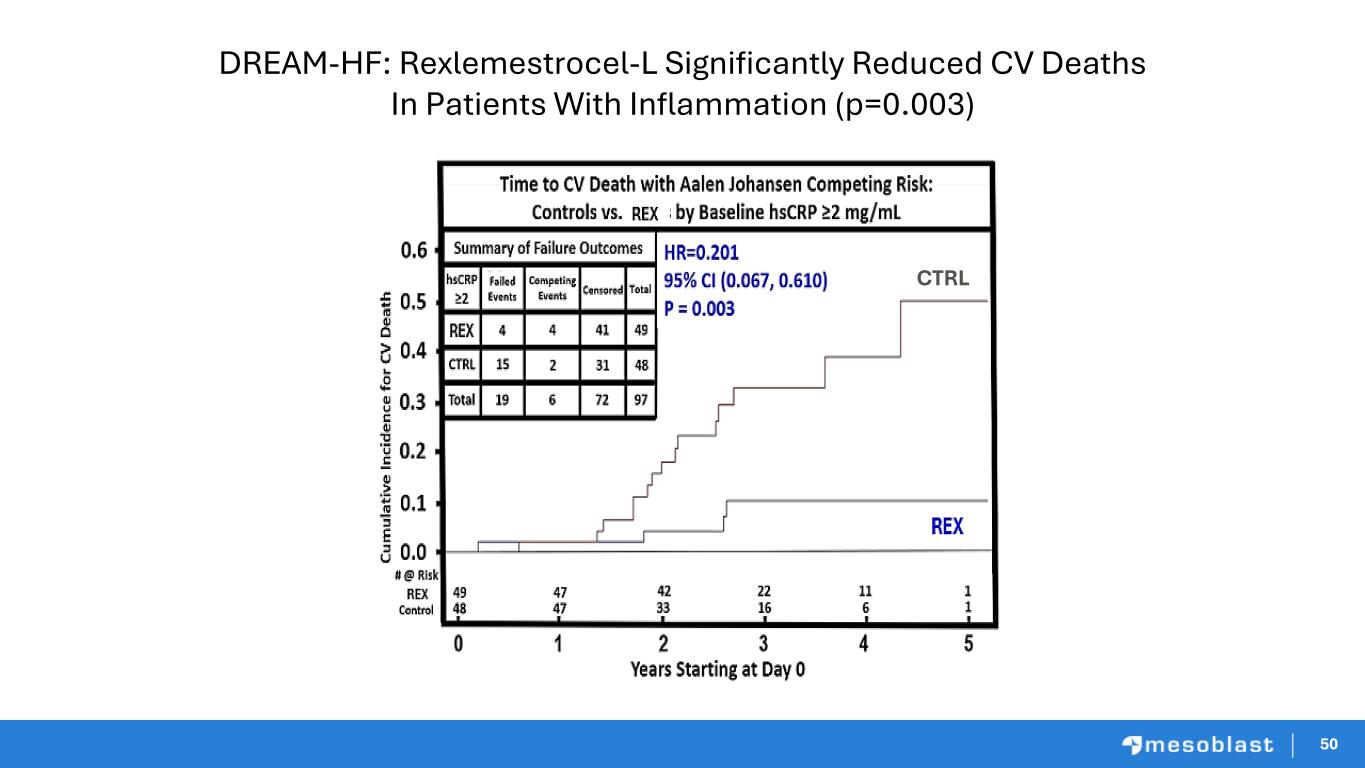
50 DREAM-HF: Rexlemestrocel-L Significantly Reduced CV Deaths In Patients With Inflammation (p=0.003) 50 CTRL
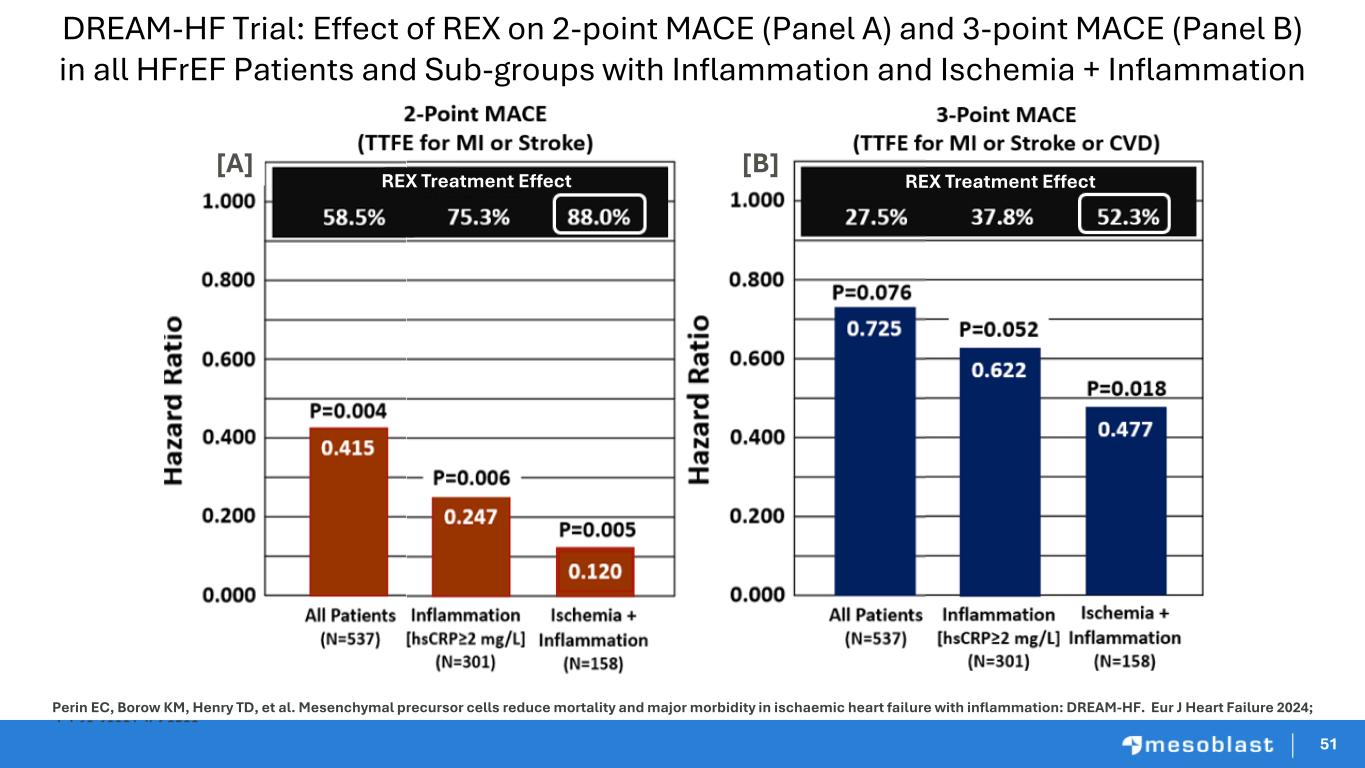
51 Perin EC, Borow KM, Henry TD, et al. Mesenchymal precursor cells reduce mortality and major morbidity in ischaemic heart failure with inflammation: DREAM-HF. Eur J Heart Failure 2024; doi:10.1002/ejhf.3522 [A] [B] REX Treatment EffectREX Treatment Effect DREAM-HF Trial: Effect of REX on 2-point MACE (Panel A) and 3-point MACE (Panel B) in all HFrEF Patients and Sub-groups with Inflammation and Ischemia + Inflammation 51
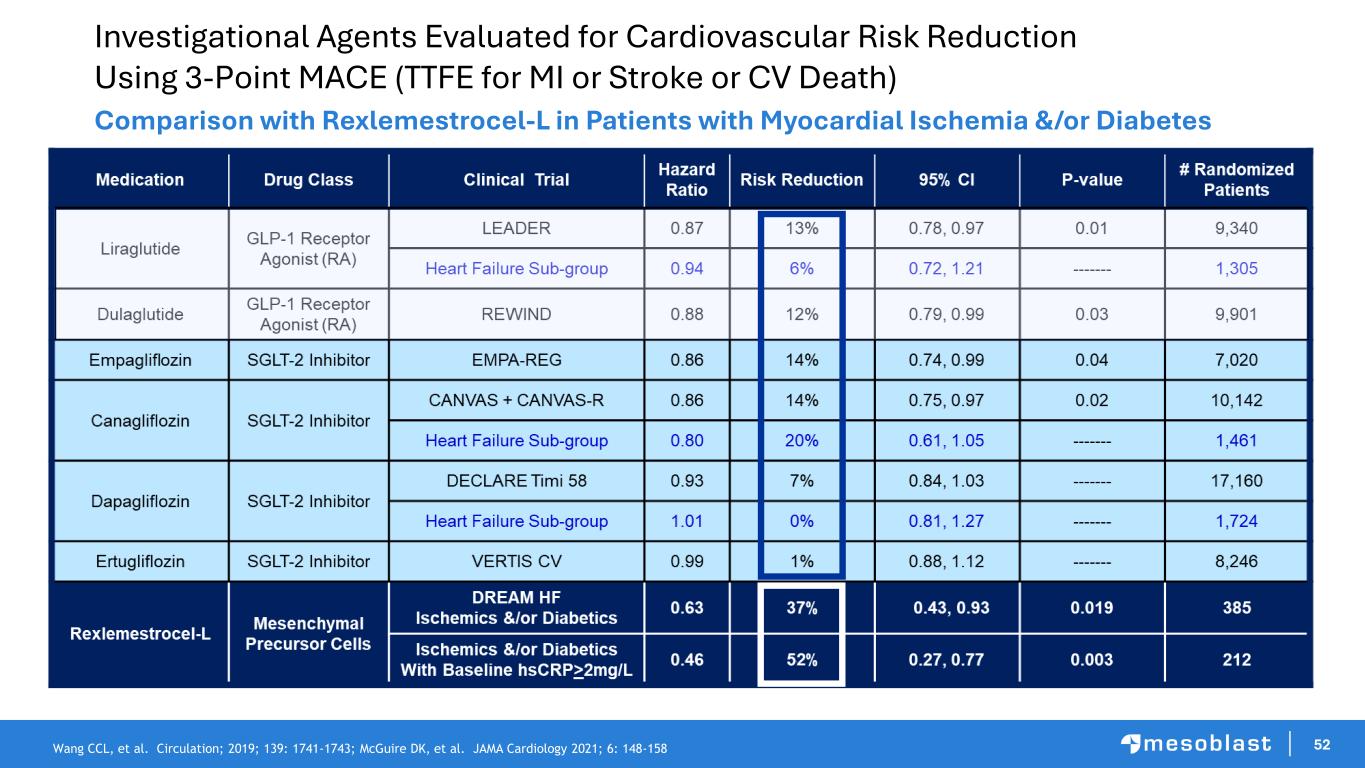
52 Investigational Agents Evaluated for Cardiovascular Risk Reduction Using 3-Point MACE (TTFE for MI or Stroke or CV Death) Comparison with Rexlemestrocel-L in Patients with Myocardial Ischemia &/or Diabetes Wang CCL, et al. Circulation; 2019; 139: 1741-1743; McGuire DK, et al. JAMA Cardiology 2021; 6: 148-158
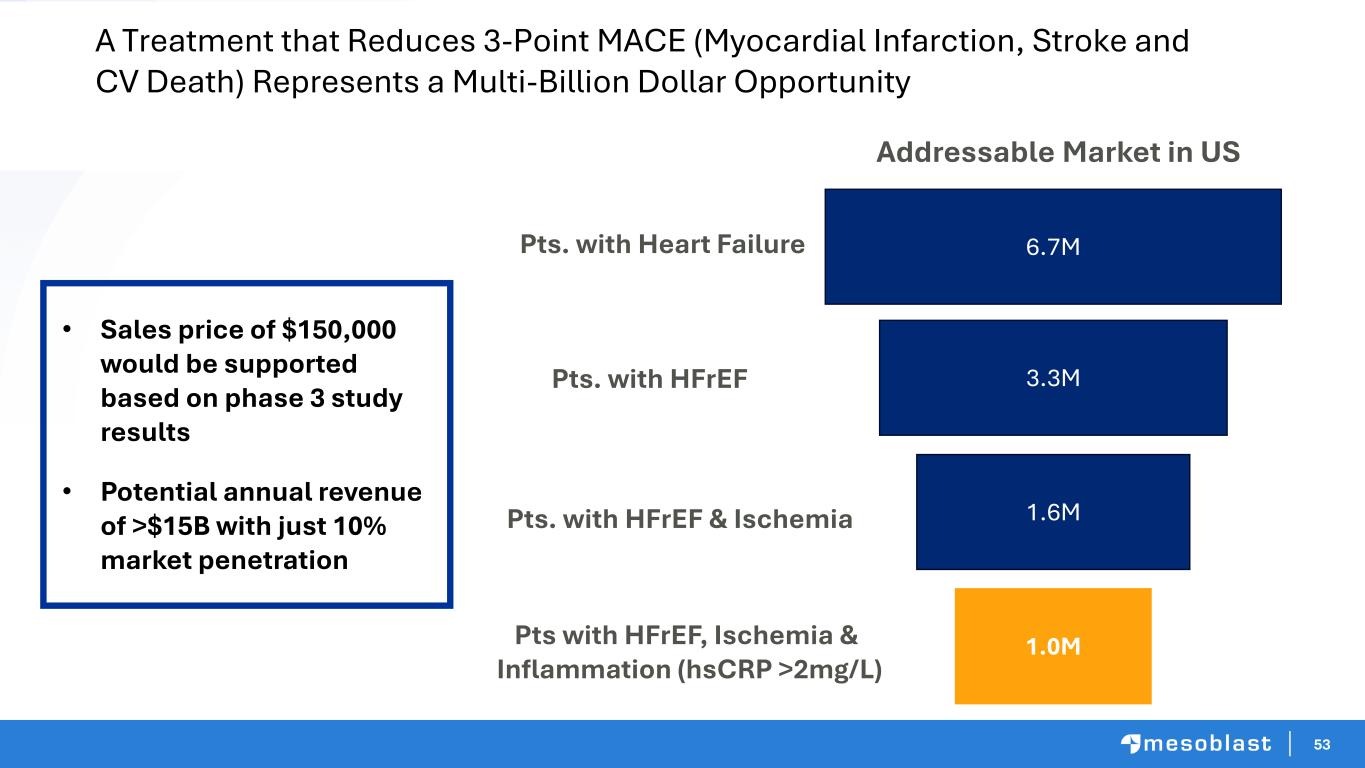
53 A Treatment that Reduces 3-Point MACE (Myocardial Infarction, Stroke and CV Death) Represents a Multi-Billion Dollar Opportunity 6.7M 3.3M 1.6M 1.0M Pts. with Heart Failure Pts. with HFrEF Pts. with HFrEF & Ischemia Pts with HFrEF, Ischemia & Inflammation (hsCRP >2mg/L) Addressable Market in US • Sales price of $150,000 would be supported based on phase 3 study results • Potential annual revenue of >$15B with just 10% market penetration

5454 End-stage Chronic HFrEF with LVAD • Despite an LVAD in the left ventricle, progressive right heart failure (RHF) continues due to ongoing inflammation of the right ventricle • Progressive RHF occurs in 15-30% of patients and is the primary cause of multi-organ failure and death • A further complication of RHF is potentially life-threatening major mucosal bleeding events (MMBE), seen in ~30% of patients and the main cause of recurrent hospitalizations
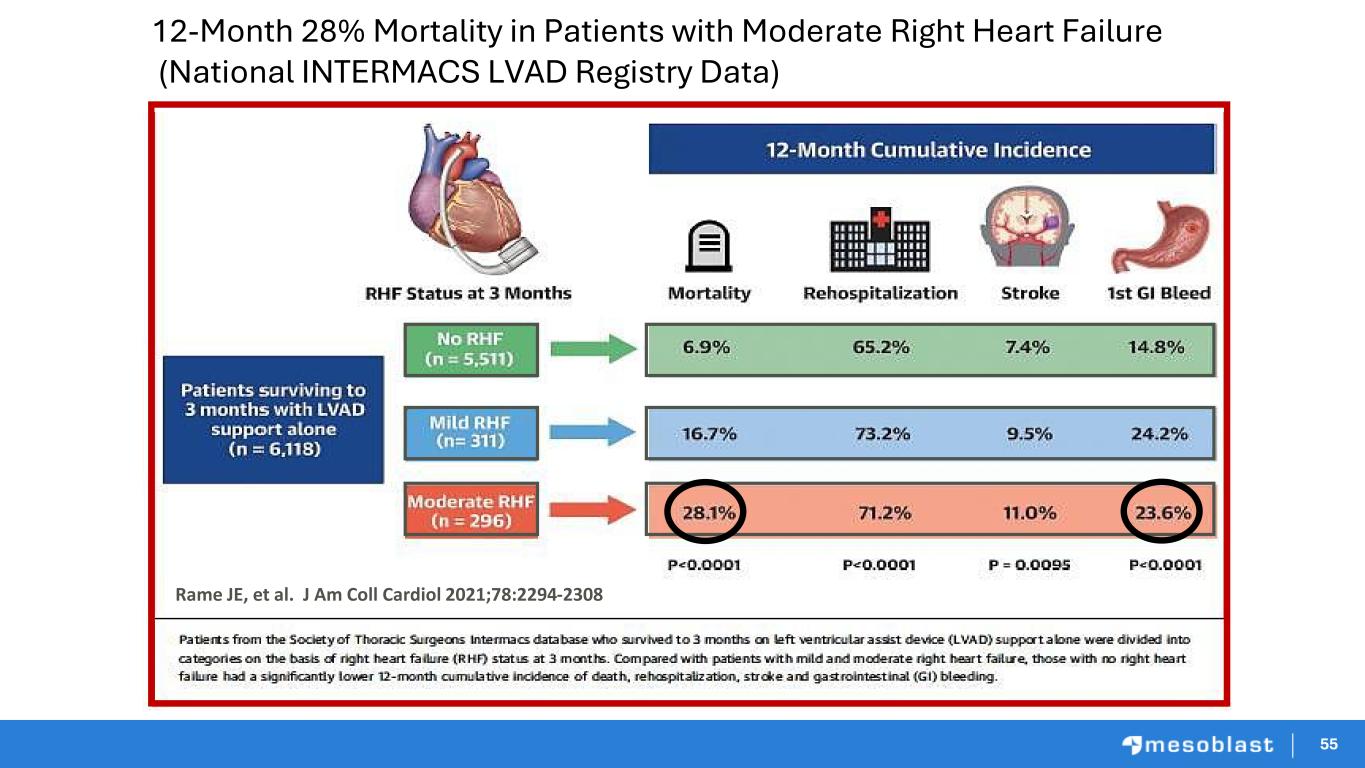
55 12-Month 28% Mortality in Patients with Moderate Right Heart Failure (National INTERMACS LVAD Registry Data) Rame JE, et al. J Am Coll Cardiol 2021;78:2294-2308 5
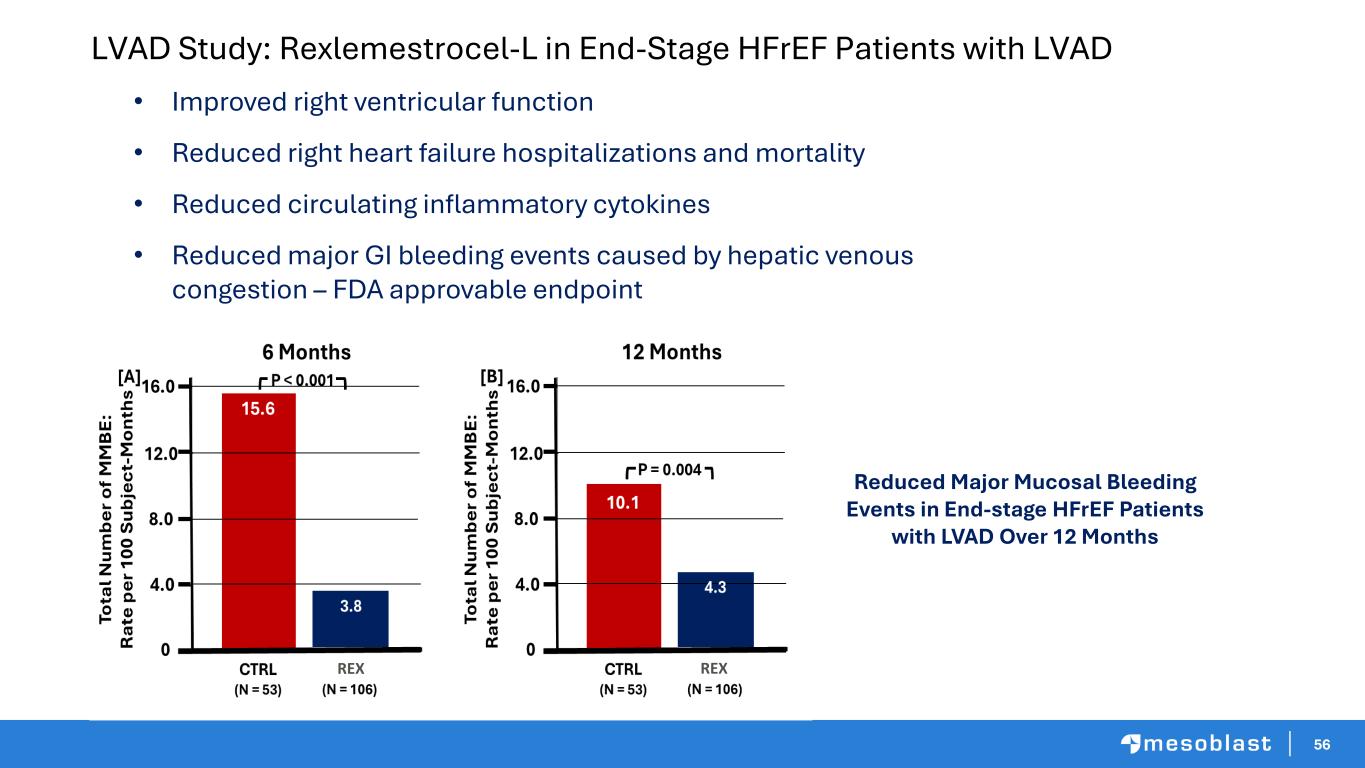
56 LVAD Study: Rexlemestrocel-L in End-Stage HFrEF Patients with LVAD • Improved right ventricular function • Reduced right heart failure hospitalizations and mortality • Reduced circulating inflammatory cytokines • Reduced major GI bleeding events caused by hepatic venous congestion – FDA approvable endpoint Reduced Major Mucosal Bleeding Events in End-stage HFrEF Patients with LVAD Over 12 Months REXREX

5757 CHF Franchise: Tactics & Strategy • File with FDA for full approval for rex-L in patients with LVADs • Filing is based on GI Bleeding being an FDA-acknowledged indication and having received Orphan Drug designation • FDA’s stated preference for randomized controlled trials has resulted in Mesoblast seeking a full FDA approval pathway, with benefit that an additional confirmatory trial is not required • FDA approval of Rex-L in patients with LVADs will facilitate subsequent approval of Mesoblast’s pre-LVAD (NYHA Class II/IIIA) chronic HFrEF patients via label extension GI = gastrointestinal | CMC = chemistry, manufacturing & controls | HFrEF = heart failure reduced ejection fraction
58 LVAD Approval Represents A Near Term Orphan Opportunity That Supports Label Extension Into The Larger Heart Failure Market 6,700,000 500,000 2,500 2,000 Pts. with Heart Failure NYHA Class IV Pts. Receiving an LVAD LVAD Patients with Ischemia Addressable LVAD Market in US • Sales price of $200,000 would be supported based on phase 3 results and reduction in cardiac ICU hospitalizations • Total addressable market potential is $400M • Approximately 190 LVAD certified centers in the US

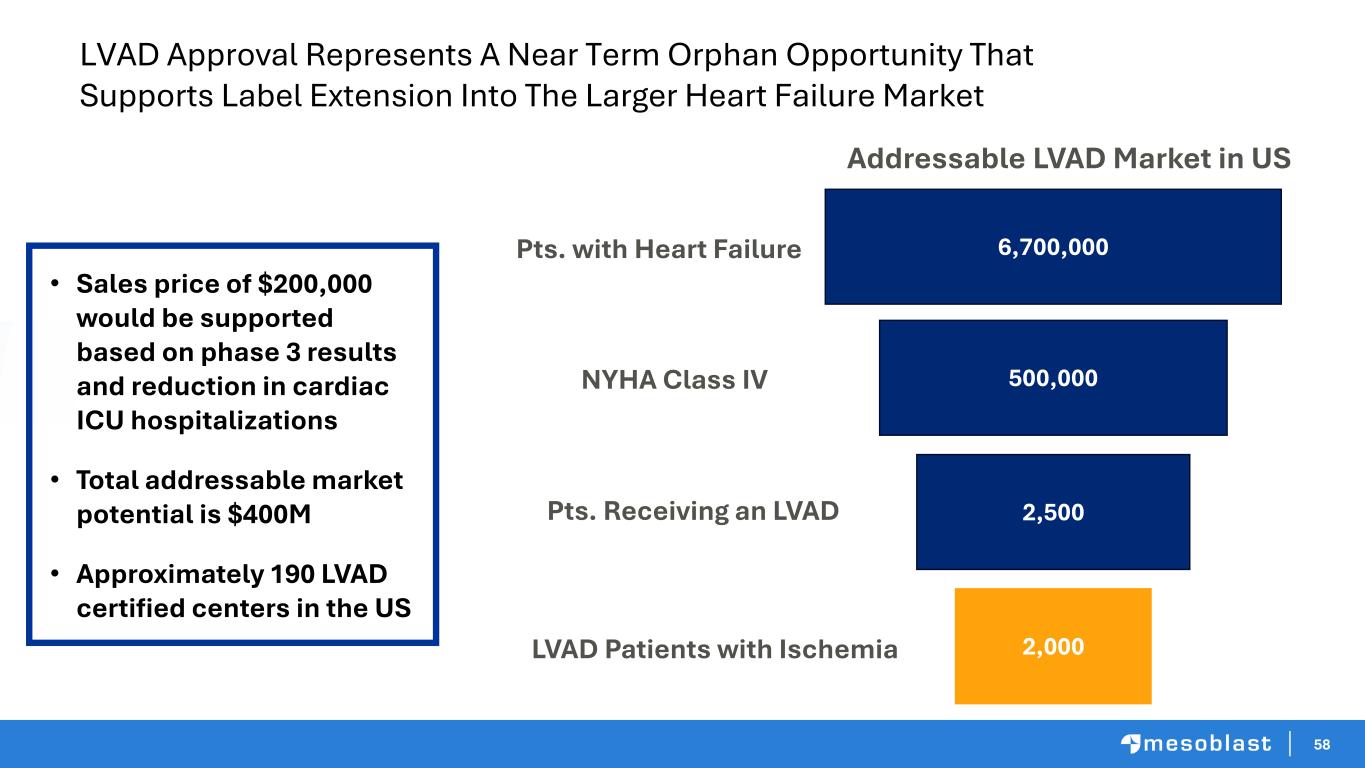
59 59 Inflammatory Heart Failure KOL Perspective Emerson Perin, MD, PhD, FACC Baylor College of Medicine Medical Director of The Texas Heart Institute Professor of Medicine, Baylor College of Medicine Eric Rose, MD Emerson C. Perin, MD, PhD Eric Rose, MD Chief Medical Officer, Mesoblast Chief Medical Officer World-renowned heart surgeon and scientist made history when he performed the first successful pediatric heart transplant

6060 Leveraging FDA Approval for Ryoncil for New Pediatric Label Extension Indications Michael Schuster Head of Business Development

61 • DMD is a genetic X-linked muscle disease with lack of dystrophin (a stabilizing protein essential for muscle strength) which ~15,000 patients in US • Inflammation of skeletal muscle and fibrosis results in loss of ambulation by 12 years old • Steroids are used in every patient for life and are the only agents proven to delay loss of ambulation and improve survival • Dystrophin gene therapy replacement has only shown modest benefit Duchenne Muscular Dystrophy (DMD): An Inflammatory Disease with Unmet Need

62 • RYONCIL has proven efficacy and safety in pediatric SR-aGvHD following steroid resistance, a disease of severe inflammation • RYONCIL has demonstrated benefit alone and with gene therapy in animal models of DMD • IND cleared by FDA for a trial that aims to extend RYONCIL label to ambulatory children with DMD • Label extension in DMD maintains existing RYONCIL pricing structure DMD: Revenue Growth Opportunity Leveraging RYONCIL’s Anti-Inflammatory Mechanism of Action
63 1. McGreevy JW, et al. Dis Model Mech. 2015 Mar;8(3):195-213. 2. Morrison J et al, Lab Invest 2000, 80:881–891. Administration of RYONCIL in mdx Mice Results in: • Down regulation of T cell, macrophage and neutrophil immune activation pathways • Increase in muscle regeneration pathways • Improvement in exercise-induced muscle fatigue Proprietary Mesoblast MSC Technology Improves Muscle Function in Duchenne Mice Serum CK M dx co ntr ol M dx M SC -tr ea te d 0 5 10 15 20 25 Serum creatine kinase (CK) levels Day 36 (14 days post last dose) S e ru m C K ( n g /m l) ✱ P=0.042 Ryoncil Reduces Serum Creatine Kinase (CK) Activity

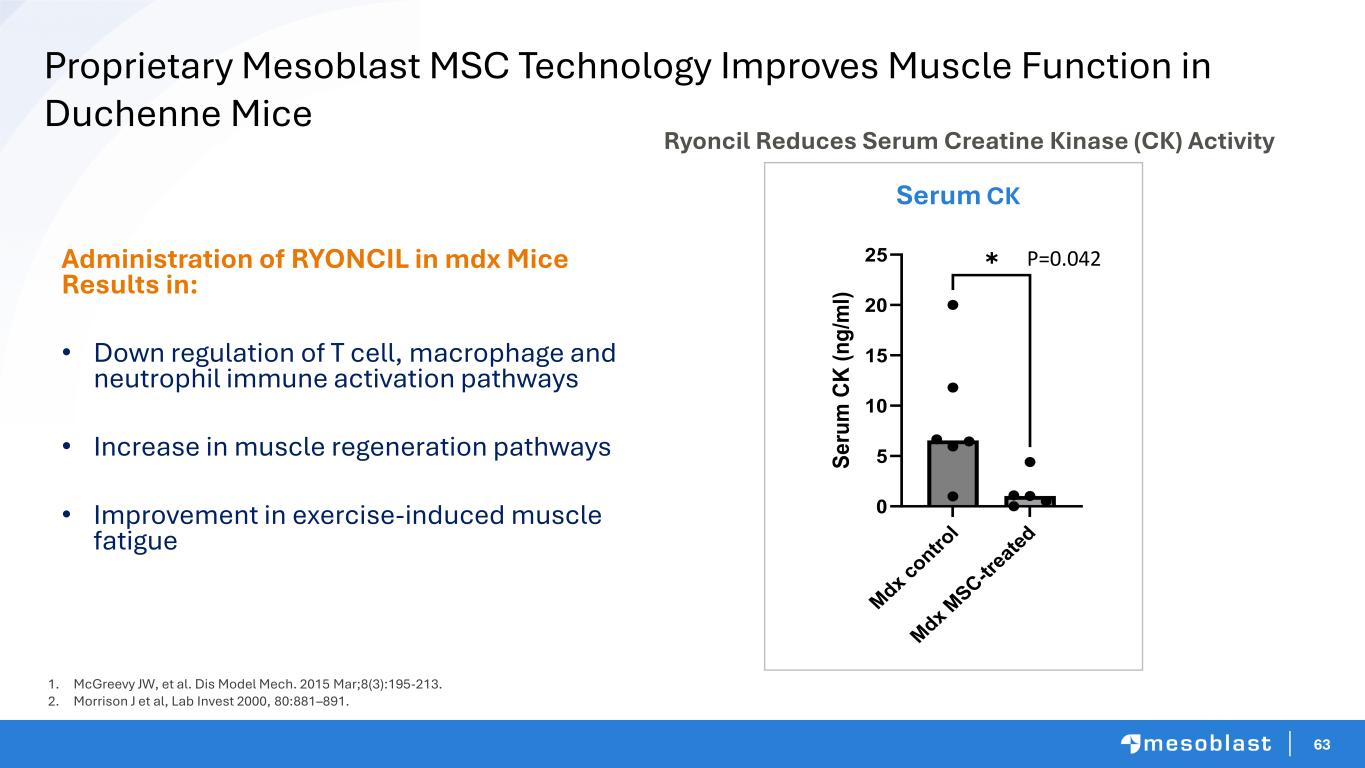
64 Mesoblast Proprietary MSC Combined with Micro Dose of Dystrophin Gene Therapy Shows Additive Benefit In Duchenne Dogs DMD Beagle - Control DMD Beagle 8 Months Post Treated Composite score of gait, mobility, limb or temporal muscle atrophy, drooling, macroglossia, and dysphagia Grading score HC DMD Non-treated dog 1 Severe Mild 6M 12M 18M 24M DMD dog AAV+MSC 0 5 10 15 20 25 2M AAV+MSC DMD Non-treated dog 2 Healthy Control dog
65 • Collaboration with Parent Project Muscular Dystrophy (PPMD) for patient identification and trial awareness; PPMD is the leading advocacy group for afflicted patients and their families • Expected to enroll over12 months • Prevalence of 5-9 year olds ~2,500 patients with ~500 new patients/year Clinical Development Plan IND Clearance Received from FDA for Phase 3 Registration Trial in Children Aged 5-9 Years with Duchenne Muscular Dystrophy Inclusion & Exclusion Criteria • Patients 5-9yo • All patients ongoing treatment with steroids for >6months • May have received gene therapy Ryoncil 7 iv infusions (2M/kg) N=38 RCT 1:1 Placebo N=38 Primary endpoint 9 month change in Time to Stand Velocity

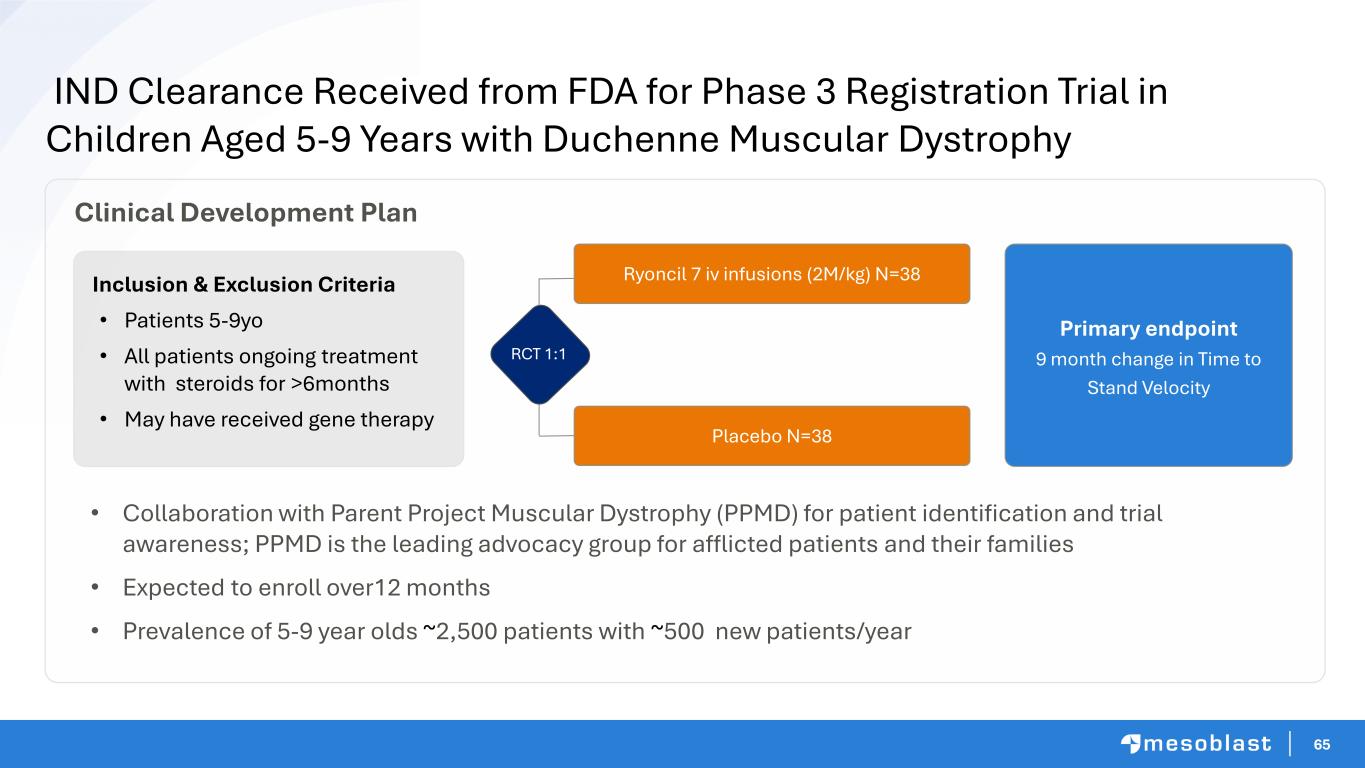
6666 Next Generation MSC Therapies Dan Devine Head of Special Projects

67 ➢ Mesoblast is leveraging the only FDA-approved MSC platform to create genetically- modified next gen products ➢ Enhanced (a) potency via payload ( ) or triggered pathway ( ), and (b) targeting via antigen binding moiety ( ) ➢ Two proprietary next gen platforms (patents through 2043) • Chimeric Antigen Receptor “CAR-MSCs” developed at Mayo Clinic (Dr. Dr. (Saad Kenderian)* • Oncolytic Virus “OV-MSCs” developed at Baylor College of Medicine (Dr. Malcolm Brenner)** ➢ IND-enabling development ongoing at each location; first IND in 1QCY2028 Next Generation Gene Modified MSCs – Enhanced Products for New Indications *Nat Biomed Eng. 2024 April ; 8(4): 443–460. doi:10.1038/s41551-024-01195-6;**https://pubmed.ncbi.nlm.nih.gov/36989357

68 CAR-MSC Platform • Leverage tech behind CAR-T therapies without issues re: (i) safety and (ii) manufacturing • Broad range of potential target antigens/intracellular pathway choices - plug and play system • Enhanced targeting/increased potency can reduce dosing/COGS for all future indications • Lead products target Lupus and UC/Crohn’s, significant/unmet medical needs • IND-enabling studies at Mayo Clinic; supervised by Dr. Kenderian Antigen/Product Target Tissue Potential Indication(s) Triggered Intracellular Pathway CD19-CAR-MSC CD19 B Cells Lupus CD28z, TLR3, TLR4, CD28, IFNy ECAD-CAR-MSC Inflamed tissue UC/Crohn’s MOG-CAR-MSC Myelin Sheath MS COL2A1-CAR-MSC Collagen RA AMPA-CAR-MSC Neurons Alzheimer's CAR-MSC-CCN1 Skin Wounds (A) Chimeric antigen receptor (CAR) targeting specific disease-associated antigen (B) Binding to antigen triggers selected intracellular pathway to enhance potency (C) Retain native properties of MSCs (A) (B) (C)
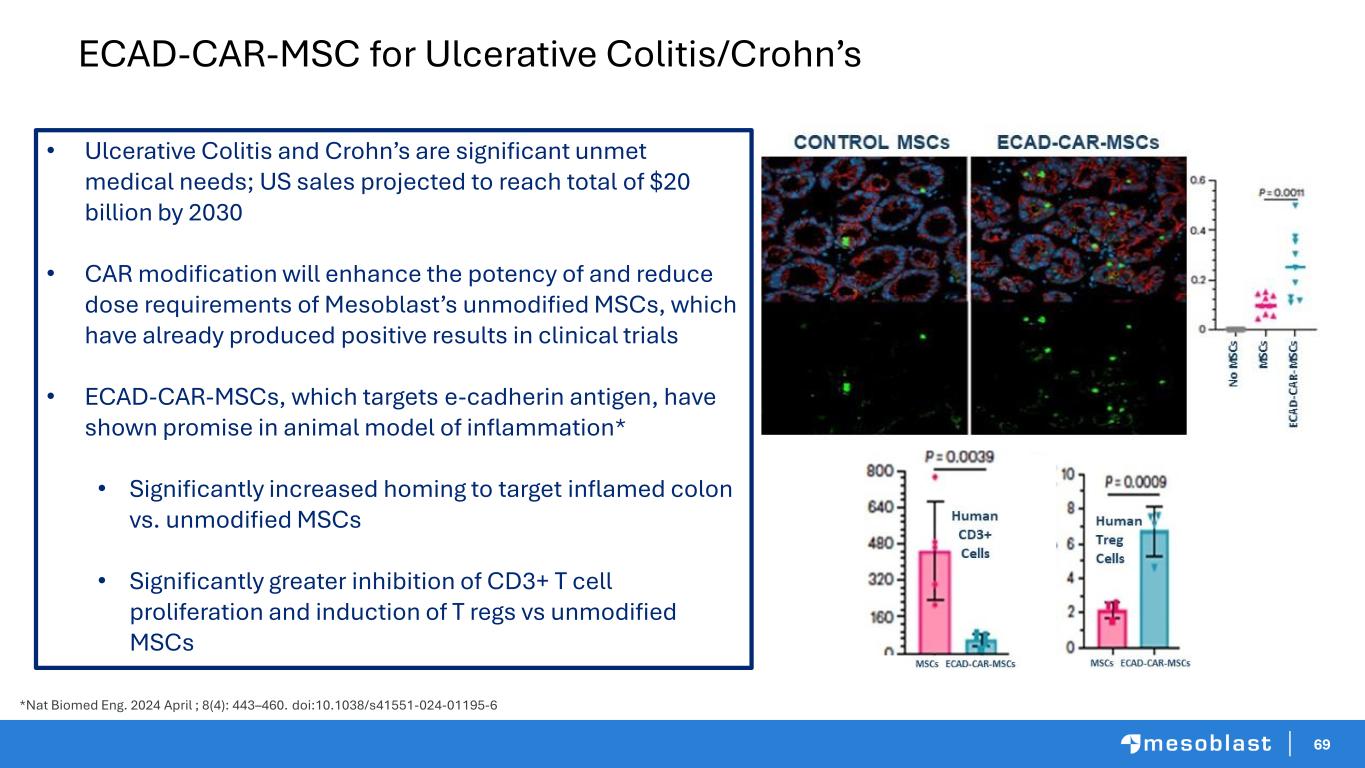
69 ECAD-CAR-MSC for Ulcerative Colitis/Crohn’s • Ulcerative Colitis and Crohn’s are significant unmet medical needs; US sales projected to reach total of $20 billion by 2030 • CAR modification will enhance the potency of and reduce dose requirements of Mesoblast’s unmodified MSCs, which have already produced positive results in clinical trials • ECAD-CAR-MSCs, which targets e-cadherin antigen, have shown promise in animal model of inflammation* • Significantly increased homing to target inflamed colon vs. unmodified MSCs • Significantly greater inhibition of CD3+ T cell proliferation and induction of T regs vs unmodified MSCs *Nat Biomed Eng. 2024 April ; 8(4): 443–460. doi:10.1038/s41551-024-01195-6
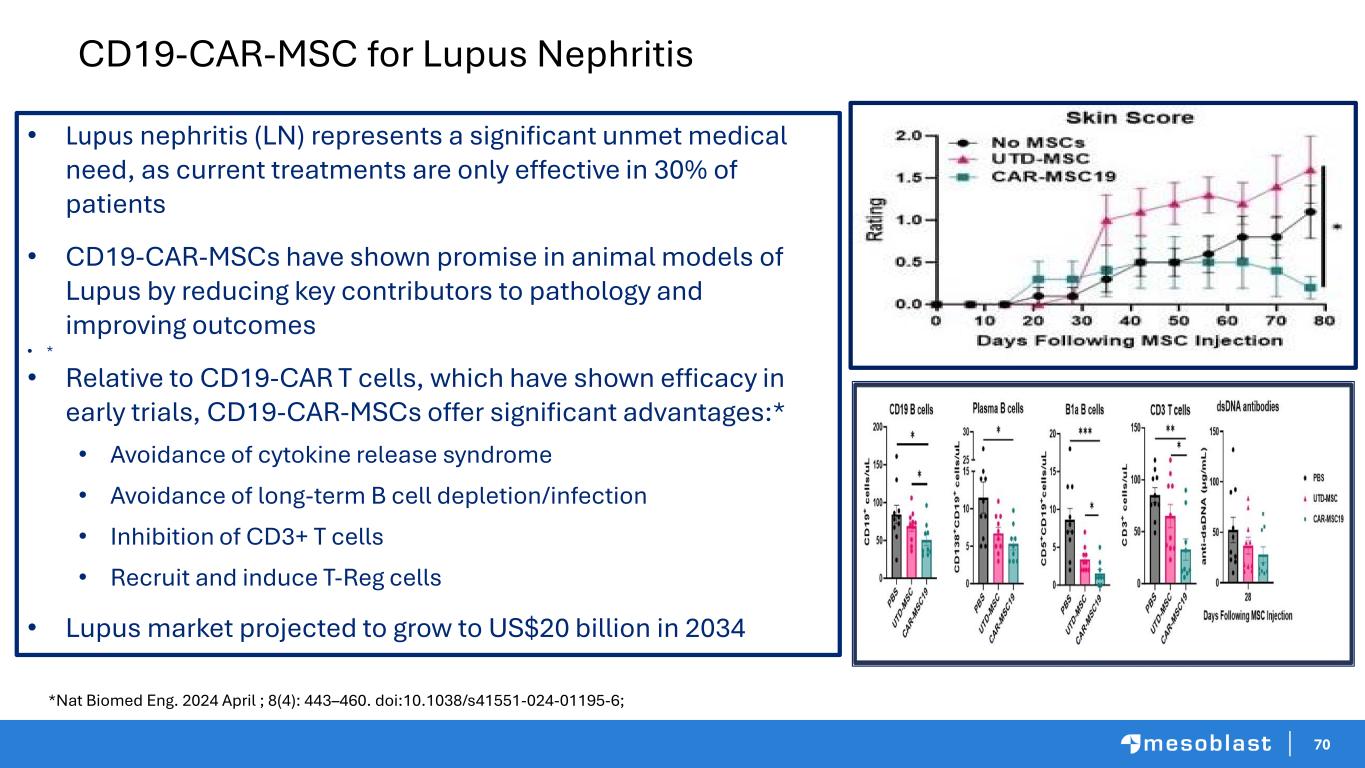
70 CD19-CAR-MSC for Lupus Nephritis • Lupus nephritis (LN) represents a significant unmet medical need, as current treatments are only effective in 30% of patients • CD19-CAR-MSCs have shown promise in animal models of Lupus by reducing key contributors to pathology and improving outcomes • * • Relative to CD19-CAR T cells, which have shown efficacy in early trials, CD19-CAR-MSCs offer significant advantages:* • Avoidance of cytokine release syndrome • Avoidance of long-term B cell depletion/infection • Inhibition of CD3+ T cells • Recruit and induce T-Reg cells • Lupus market projected to grow to US$20 billion in 2034 *Nat Biomed Eng. 2024 April ; 8(4): 443–460. doi:10.1038/s41551-024-01195-6;

71 OV-MSC Platform • Leverage technology behind CAR-T therapies without issues of safety and manufacturing • IND-enabling studies ongoing at Baylor College of Medicine, under supervision of Dr. Malcolm Brenner, Director of the Center for Cell and Gene Therapy at Baylor OV Payloads/Engagers Evaluated by BCM Chemokine Cytokine Checkpoint Inhibitor Engager MPI-1a RANTES IL2 IL7 IL12 IL15 IL21 PD-L1 CTLA-4 CD19 CD44v6 MUC-1 FAP HER2 • MSCs loaded with an oncolytic virus (OV) genetically engineered to produce (i) binary T-cell engager (BiTE) that engage tumor cells and CD3+ T cells; and (ii) anti-cancer payloads • MSCs protect payload as they navigate to targeted tissues • OV and payloads released at tumor site as MSCs go through oncolysis
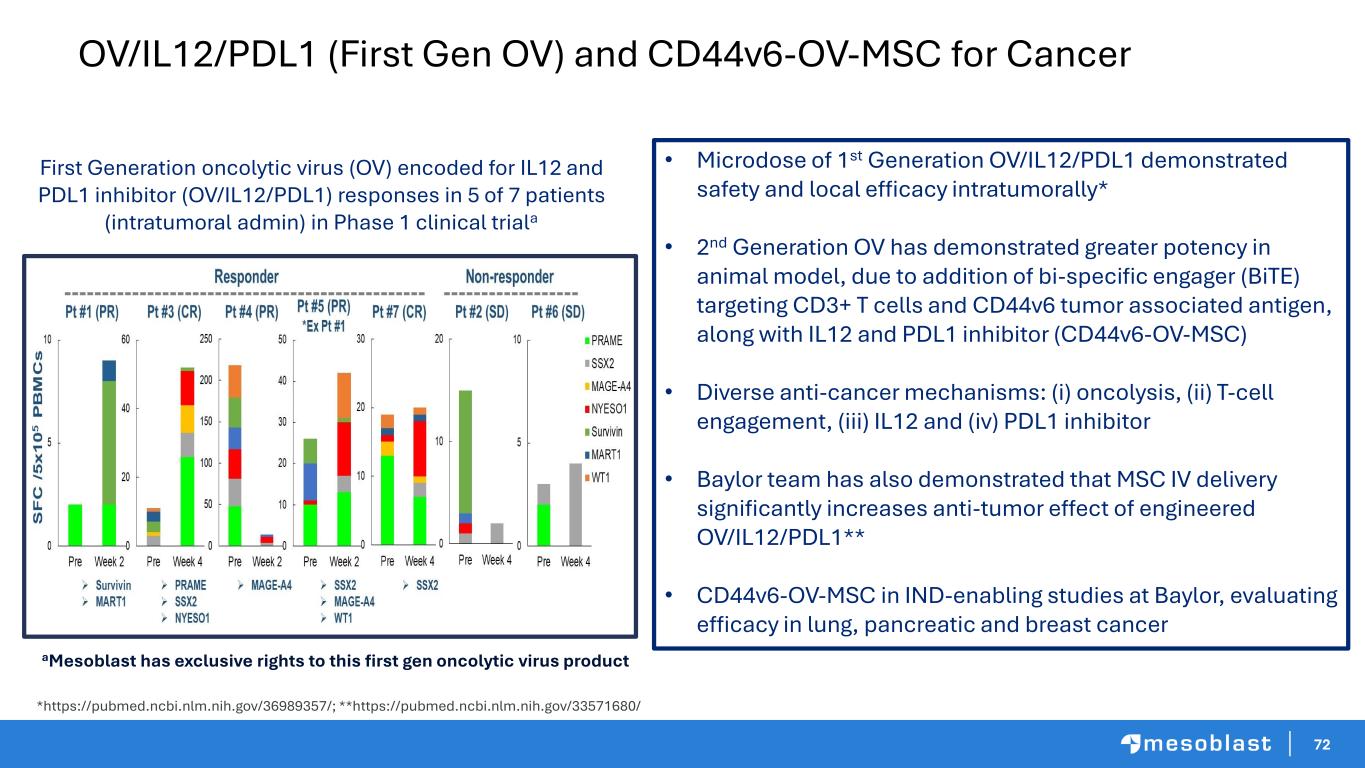
72 OV/IL12/PDL1 (First Gen OV) and CD44v6-OV-MSC for Cancer *https://pubmed.ncbi.nlm.nih.gov/36989357/; **https://pubmed.ncbi.nlm.nih.gov/33571680/ First Generation oncolytic virus (OV) encoded for IL12 and PDL1 inhibitor (OV/IL12/PDL1) responses in 5 of 7 patients (intratumoral admin) in Phase 1 clinical triala aMesoblast has exclusive rights to this first gen oncolytic virus product • Microdose of 1st Generation OV/IL12/PDL1 demonstrated safety and local efficacy intratumorally* • 2nd Generation OV has demonstrated greater potency in animal model, due to addition of bi-specific engager (BiTE) targeting CD3+ T cells and CD44v6 tumor associated antigen, along with IL12 and PDL1 inhibitor (CD44v6-OV-MSC) • Diverse anti-cancer mechanisms: (i) oncolysis, (ii) T-cell engagement, (iii) IL12 and (iv) PDL1 inhibitor • Baylor team has also demonstrated that MSC IV delivery significantly increases anti-tumor effect of engineered OV/IL12/PDL1** • CD44v6-OV-MSC in IND-enabling studies at Baylor, evaluating efficacy in lung, pancreatic and breast cancer

73 Next-Gen MSCs protect leadership position and enhance pipeline • Maintains leading competitive position well into the future (IP, technology, product performance) • Opens promising/valuable new commercial opportunities • Candidates for deployment of advanced manufacturing capabilities (e.g., bioreactors) Development of Next-Gen MSCs strategically aligned with current corporate priorities • Leading institutions can advance products through Phase 1 trials onsite • Building value w/o expansion of infrastructure • Maintains internal focus on core commercial and late-stage clinical trial focus Next-Gen MSC Technology Securing the Future While Aligned with Current Priorities

7474 Saad Kenderian, MD, ChB Mayo Clinic Assistant Professor of Oncology, Medicine and Immunology at Mayo Clinic Next Generation MSC Platforms KOL Perspective

7575 Justin Horst Head of Manufacturing Manufacturing
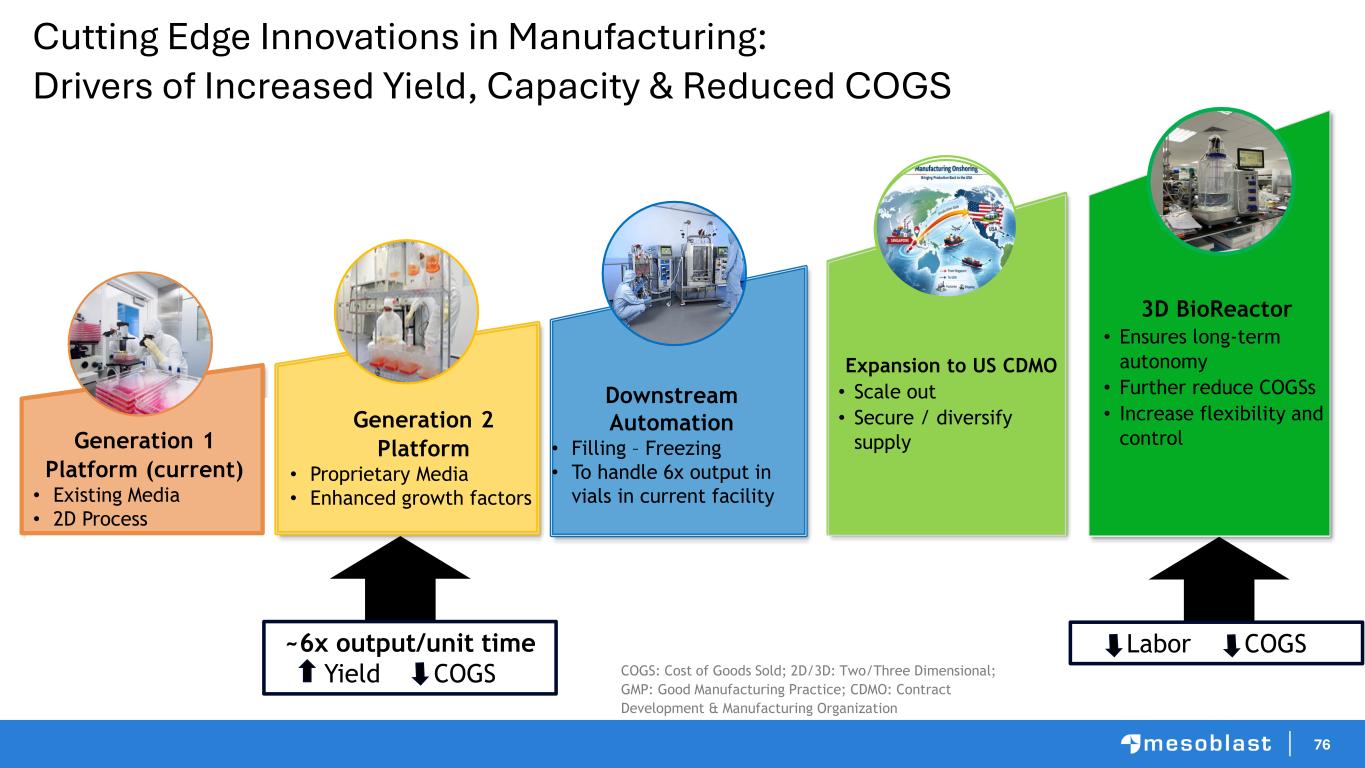
76 Cutting Edge Innovations in Manufacturing: Drivers of Increased Yield, Capacity & Reduced COGS Generation 2 Platform • Proprietary Media • Enhanced growth factors Expansion to US CDMO • Scale out • Secure / diversify supply 3D BioReactor • Ensures long-term autonomy • Further reduce COGSs • Increase flexibility and controlGeneration 1 Platform (current) • Existing Media • 2D Process Downstream Automation • Filling – Freezing • To handle 6x output in vials in current facility ~6x output/unit time Yield COGS COGS: Cost of Goods Sold; 2D/3D: Two/Three Dimensional; GMP: Good Manufacturing Practice; CDMO: Contract Development & Manufacturing Organization Labor COGS

77 Manufacturing activities adequately funded internally to support projected demand and further drive down COGS Manufacturing Activity & Strategic Initiatives Timeframe • Ensure RYONCIL commercial inventory supply meets projected growth in demand for pediatric and adult indications CDMO contracts in place for projected 3- year demand in commercial product • Introduce second generation manufacturing platform with proprietary media post first approvals for initial remestemcel-L and rexlemestrocel-l products to meet requirements of large volume indications (Adult GVHD, CLBP, CHF) 12-18 months • Introduce automation to match increased production scale 12-18 months • Validate U.S. manufacturing site post approvals Planned for FY2028

7878 James O’Brien Chief Financial Officer Financial

79 Strong Financial Position • Gross revenue Q3 FY26 was US$35.3M, with net revenue US$30.3M • RYONCIL gross profit for H1 FY26 was US$44.2M, with US$7.7M direct selling costs • Net operating cash usage for H1 FY26 was US$30.3M • Mesoblast expects to see reduction in net cash spend over the remainder of the fiscal period based on projected receipts from quarterly revenues • Cost discipline approach to managing spend in-line with revenue • Operating plan includes spend on Phase 3 programs, manufacturing for BLA filing and commercial inventory • New term loan totaling US$125M replaces existing higher- cost debt Cash balance US$130M at Dec 31, 2025 BLA: Biologics License Application (FDA)
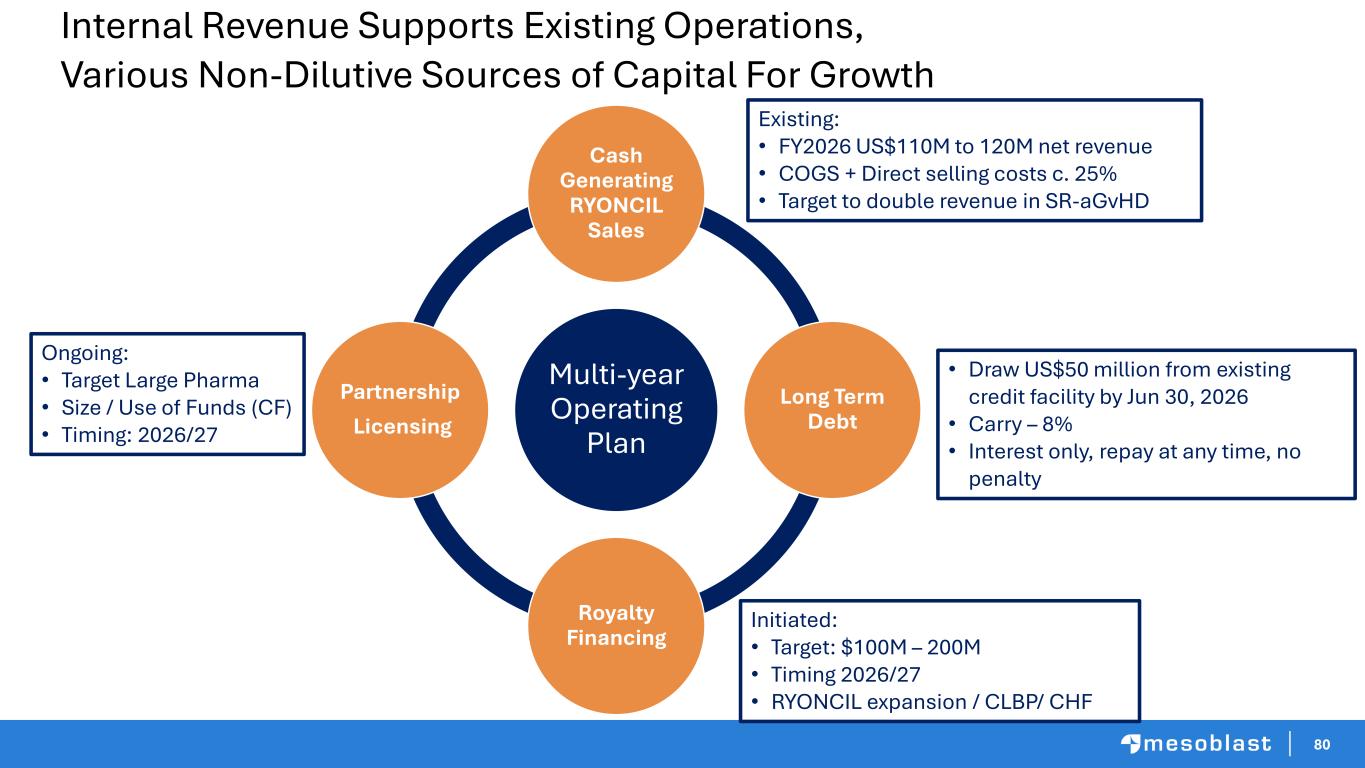
80 Internal Revenue Supports Existing Operations, Various Non-Dilutive Sources of Capital For Growth Multi-year Operating Plan Cash Generating RYONCIL Sales Long Term Debt Royalty Financing Partnership Licensing • Draw US$50 million from existing credit facility by Jun 30, 2026 • Carry – 8% • Interest only, repay at any time, no penalty Initiated: • Target: $100M – 200M • Timing 2026/27 • RYONCIL expansion / CLBP/ CHF Ongoing: • Target Large Pharma • Size / Use of Funds (CF) • Timing: 2026/27 Existing: • FY2026 US$110M to 120M net revenue • COGS + Direct selling costs c. 25% • Target to double revenue in SR-aGvHD

8181 Silviu Itescu Chief Executive Wrap-Up

82 • Multi-modal anti-inflammatory mechanism of action – means can act concurrently on multiple pathways of the immune system, not just one • Inert and self-regulating in absence of inflammation equals excellent safety profile • These features result in superior efficacy without concerns of off-target adverse events typical of small molecules or antibodies • Platform technology can be leveraged across many indications with high unmet needs • No requirement for matching or immunosuppression • Highly expandable and scalable manufacturing means product made from one donor can be used in thousands of unrelated recipients • These features underpin industrial scale supply chain with a sustainable high-margin business model MSC Magic – What Makes These Cells Special

83 • Globally IP portfolio >1,100 patents provide protection through >2044 • Dominant IP protects a cell type whose unique properties underpin a scalable commercial business model • First mover advantage – the first and only MSC approved by FDA • Completed large, US-based, randomized clinical trials which provide evidence of efficacy • Benchmark in complex manufacturing with IP protection, significant know-how advantage, demonstrated FDA alignment, scale-up capacity, and ability to leverage across many products • Next gen technology leadership to enhance tissue-homing characteristics and achieve even greater efficacy for existing products in areas such as inflammatory bowel disease, lupus nephritis and Alzheimer’s disease, and completely open new areas such as cancer Our ‘MOAT’ – What Sets Mesoblast Apart and Protects Our Market Leadership Position

84 Anticipated Major Upcoming Milestones RYONCIL – Commercial • Net revenue approaching US$100M since launch • Strategy in place to strongly grow revenue base • Well-funded to execute on operational plans, RYONCIL sales to fund growth pipeline • Focused on increasing penetration of pediatric SR-aGvHD market CHF: regulatory strategy to gain approval for blockbuster indication • File BLA with FDA for end-stage heart failure patients on LVADs this quarter • Leverage approval to initiate confirmatory trial in NYHA II/III HFrEF RYONCIL - Label Extension • Label extension to adult SR-aGvHD; 3x larger market vs pediatric • Adult SR-aGvHD trial in collaboration with BMT-CTN network, trial initiated, to complete in 12-18 months • Pediatric Duchenne’s in collaboration with PPMD patient advocates – FDA IND clearance received Rexlemestrocel-L - CLBP Blockbuster • Complete enrollment of pivotal trial end of April • Top-line primary endpoint mid-CY2027 • BLA filing for FDA approval Q3 CY2027 • Potential FDA approval and US launch Q2 CY2028

85 Thank You