

First-in-disease therapies for patients with rare diseases Corporate Presentation April 2026

Forward Looking Statements This presentation contains forward-looking statements of Palvella Therapeutics, Inc. (“the Company”) within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include all statements that are not historical facts, and in some cases, can be identified by terms such as “may,” “might,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “objective,” “anticipate,” “believe,” “estimate,” “predict,” “potential,” “continue,” “ongoing,” or the negative of these terms, or other comparable terminology intended to identify statements about the future. Forward-looking statements contained in this presentation include, but are not limited to, statements regarding the Company’s future financial or business performance, conditions, plans, prospects, trends or strategies and other financial and business matters, the Company’s current and prospective product candidates and any additional indications or platform candidates, the Company's planned research and development activities, the Company's planned clinical trials, including timing of receipt of data from the same, the planned regulatory framework for the Company's product candidates, the Company's ability and the ability of third-party manufacturers the Company engages to optimize and scale manufacturing, the strength of the Company's intellectual property portfolio, and projections of the Company’s future financial results and other metrics. Such forward-looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward looking statements. These forward-looking statements are based upon current estimates and assumptions of the Company and its management and are subject to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking statements contained in this presentation. Factors that may cause actual results to differ materially from current expectations include, but are not limited to: competition, the ability of the Company to grow and manage growth, maintain relationships with suppliers and retain its management and key employees; the success, cost and timing of the Company’s product development activities, studies and clinical trials; changes in applicable laws or regulations; the possibility that the Company may be adversely affected by other economic, business or competitive factors; the Company’s estimates of expenses and profitability; the evolution of the markets in which the Company competes; the ability of the Company to implement its strategic initiatives and continue to innovate its existing products; and the ability of the Company to defend its intellectual property. Nothing in this Presentation should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. You should not place undue reliance on forward-looking statements, which speak only as of the date they are made. The Company undertakes no duty to update these forward-looking statements. Industry and Market Data: The Company may from time to time provide estimates, projections and other information concerning its industry, the general business environment, and the markets for certain conditions, including estimates regarding the potential size of those markets and the estimated incidence and prevalence of certain medical conditions. Information that is based on estimates, forecasts, projections, market research or similar methodologies is inherently subject to uncertainties, and actual events, circumstances or numbers, including actual disease prevalence rates and market size, may differ materially from the information reflected in this presentation. Unless otherwise expressly stated, we obtained this industry, business information, market data, prevalence information and other data from reports, research surveys, studies and similar data prepared by market research firms and other third parties, industry, medical and general publications, government data, and similar sources, in some cases applying our own assumptions and analysis that may, in the future, prove not to have been accurate. Trademarks: This Presentation may contain trademarks, service marks, trade names and copyrights of other companies, which are the property of their respective owners. Solely for convenience, some of the trademarks, service marks, trade names and copyrights referred to in this Presentation may be listed without the TM, SM © or ® symbols, but the Company will assert, to the fullest extent under applicable law, the rights of the applicable owners, if any, to these trademarks, service marks, trade names and copyrights.

PALVELLA (pɑlʋelːɑ, Finnish): TO SERVE Building the leading rare disease biopharma company focused on developing and commercializing first-in-disease therapies for serious, rare skin diseases and vascular malformations
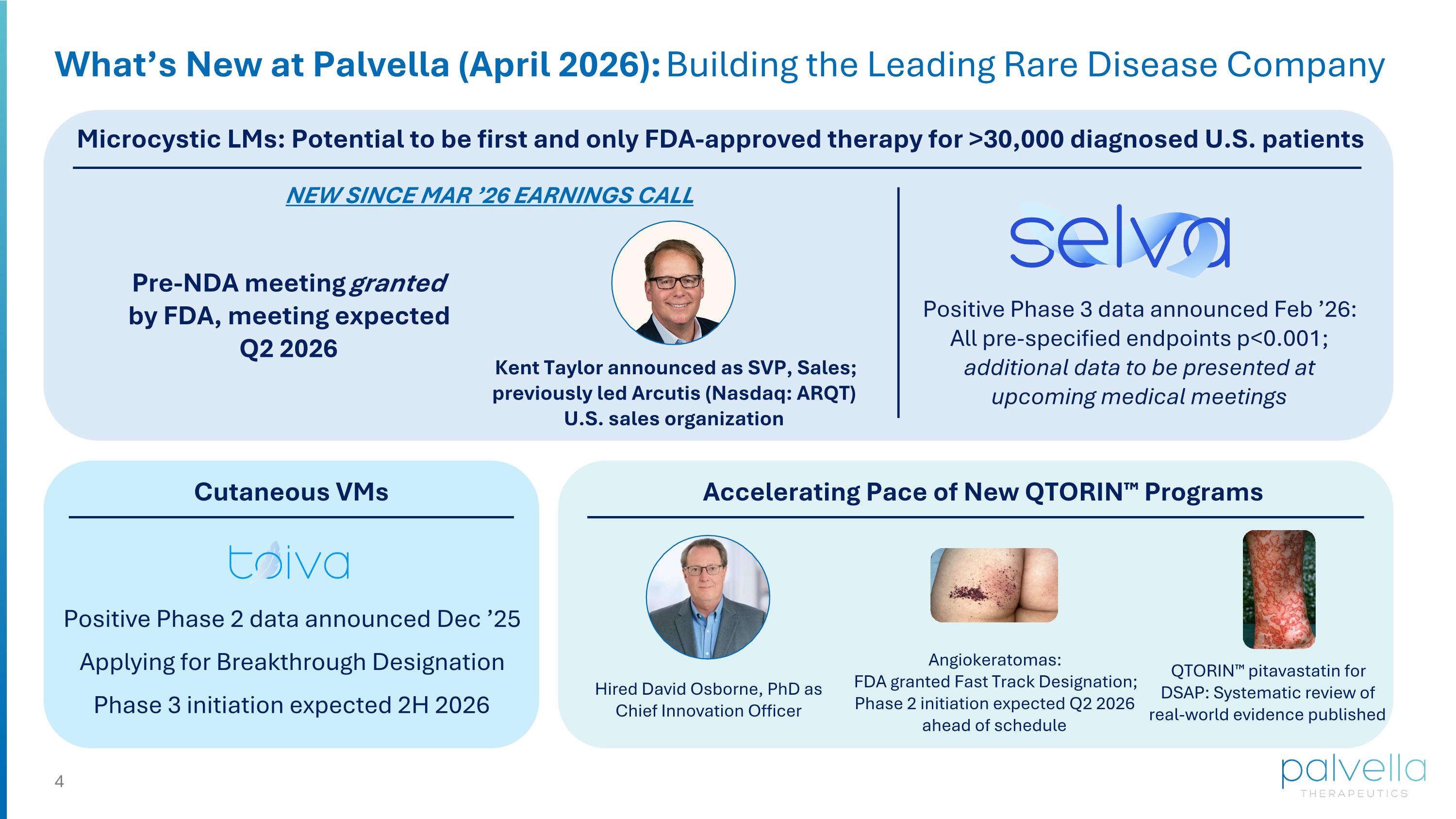
What’s New at Palvella (April 2026): Building the Leading Rare Disease Company Microcystic LMs: Potential to be first and only FDA-approved therapy for >30,000 diagnosed U.S. patients Positive Phase 3 data announced Feb ’26: All pre-specified endpoints p<0.001; additional data to be presented at upcoming medical meetings Pre-NDA meeting granted by FDA, meeting expected Q2 2026 Kent Taylor announced as SVP, Sales; previously led Arcutis (Nasdaq: ARQT) U.S. sales organization Cutaneous VMs Positive Phase 2 data announced Dec ’25 Applying for Breakthrough Designation Phase 3 initiation expected 2H 2026 Accelerating Pace of New QTORIN™ Programs Angiokeratomas: FDA granted Fast Track Designation; Phase 2 initiation expected Q2 2026 ahead of schedule Hired David Osborne, PhD as Chief Innovation Officer QTORIN™ pitavastatin for DSAP: Systematic review of real-world evidence published NEW SINCE MAR ’26 EARNINGS CALL
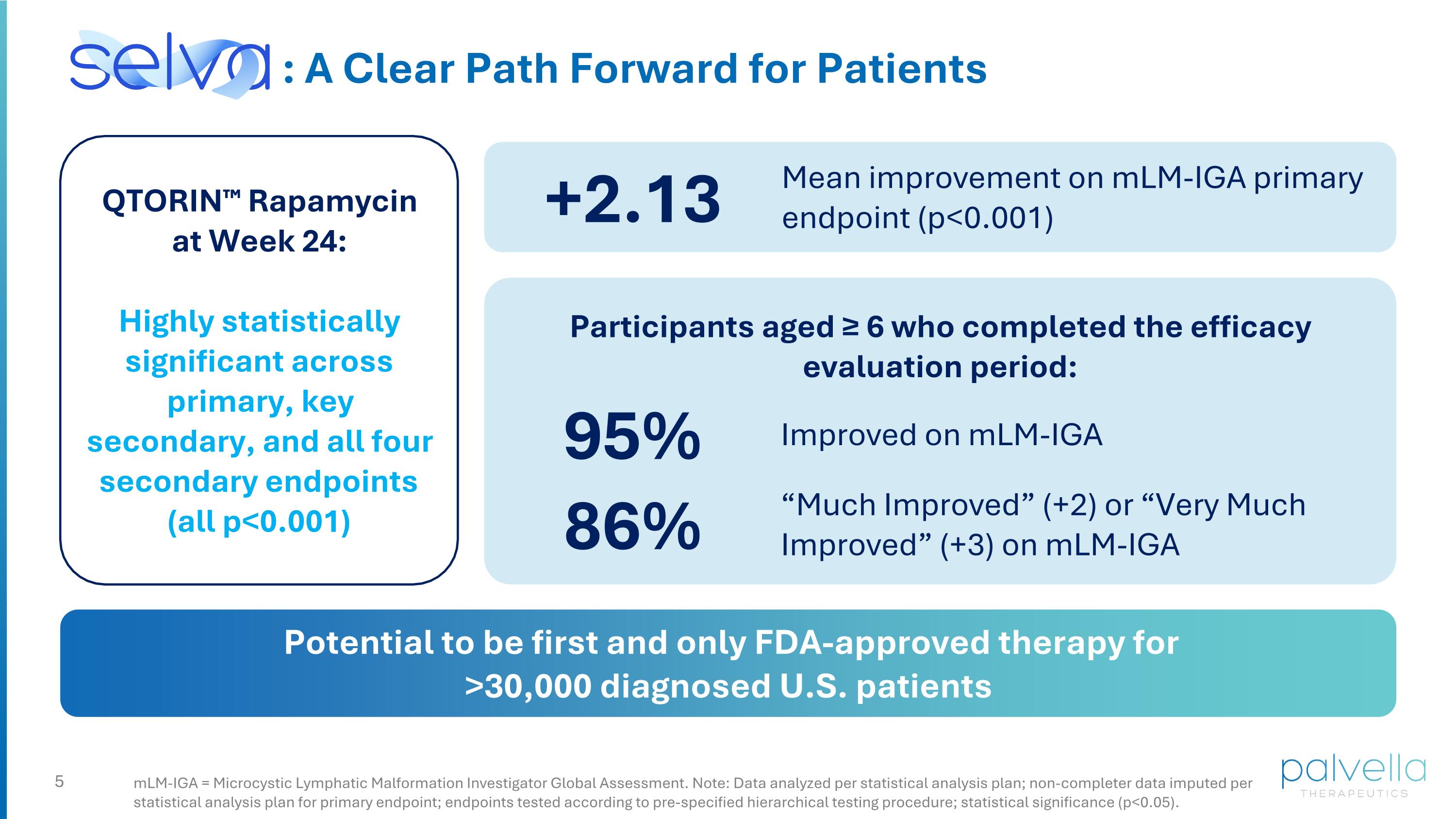
mLM-IGA = Microcystic Lymphatic Malformation Investigator Global Assessment. Note: Data analyzed per statistical analysis plan; non-completer data imputed per statistical analysis plan for primary endpoint; endpoints tested according to pre-specified hierarchical testing procedure; statistical significance (p<0.05). : A Clear Path Forward for Patients QTORIN™ Rapamycin at Week 24: Highly statistically significant across primary, key secondary, and all four secondary endpoints (all p<0.001) +2.13 Mean improvement on mLM-IGA primary endpoint (p<0.001) 95% Participants aged ≥ 6 who completed the efficacy evaluation period: Potential to be first and only FDA-approved therapy for >30,000 diagnosed U.S. patients 86% “Much Improved” (+2) or “Very Much Improved” (+3) on mLM-IGA Improved on mLM-IGA
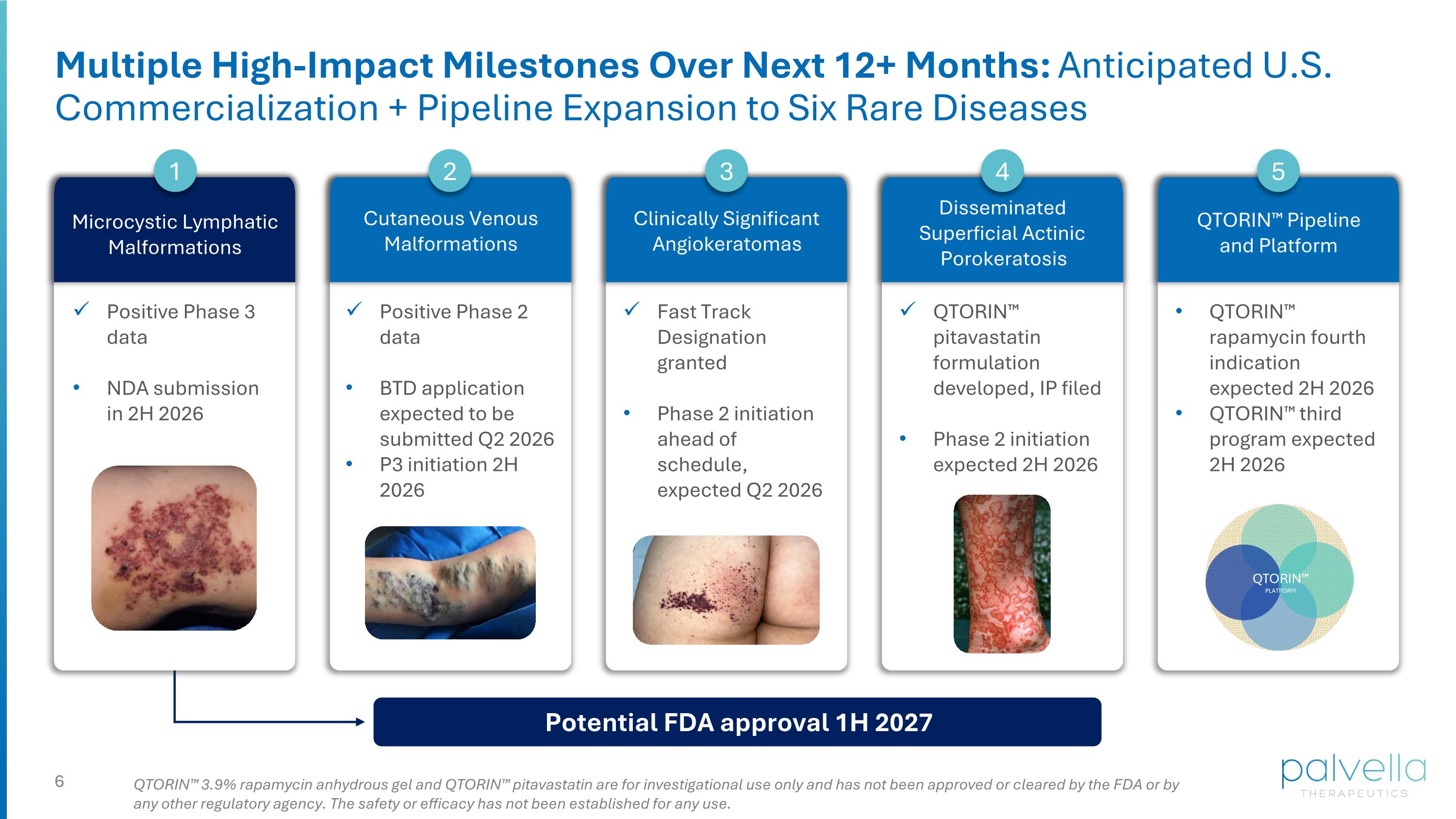
Multiple High-Impact Milestones Over Next 12+ Months: Anticipated U.S. Commercialization + Pipeline Expansion to Six Rare Diseases QTORIN™ 3.9% rapamycin anhydrous gel and QTORIN™ pitavastatin are for investigational use only and has not been approved or cleared by the FDA or by any other regulatory agency. The safety or efficacy has not been established for any use. Microcystic Lymphatic Malformations 1 Positive Phase 3 data NDA submission in 2H 2026 Cutaneous Venous Malformations 2 Positive Phase 2 data BTD application expected to be submitted Q2 2026 P3 initiation 2H 2026 Clinically Significant Angiokeratomas 3 Fast Track Designation granted Phase 2 initiation ahead of schedule, expected Q2 2026 Disseminated Superficial Actinic Porokeratosis 4 QTORIN™ pitavastatin formulation developed, IP filed Phase 2 initiation expected 2H 2026 QTORIN™ rapamycin fourth indication expected 2H 2026 QTORIN™ third program expected 2H 2026 QTORIN™ Pipeline and Platform 5 Potential FDA approval 1H 2027
What Makes Palvella Stand Apart Repeatably unlocking multi-billion dollar market opportunities in previously untreated orphan diseases …through: First-in-Disease Focus Rare Diseases with Clear Disease Biology Leveraging Existing Human Proof-of-Concept Data Innovative QTORIN™ Platform: On Target, In Tissue …resulting in first-in-disease therapies with potential in: Microcystic Lymphatic Malformations Cutaneous Venous Malformations Clinically Significant Angiokeratomas Disseminated Superficial Actinic Porokeratosis …with two more to come in 2026 Focused rare disease development model designed to reduce time and capital to FDA approval

OUR LEAD PRODUCT CANDIDATE QTORIN™ 3.9% RAPAMYCIN ANHYDROUS GEL
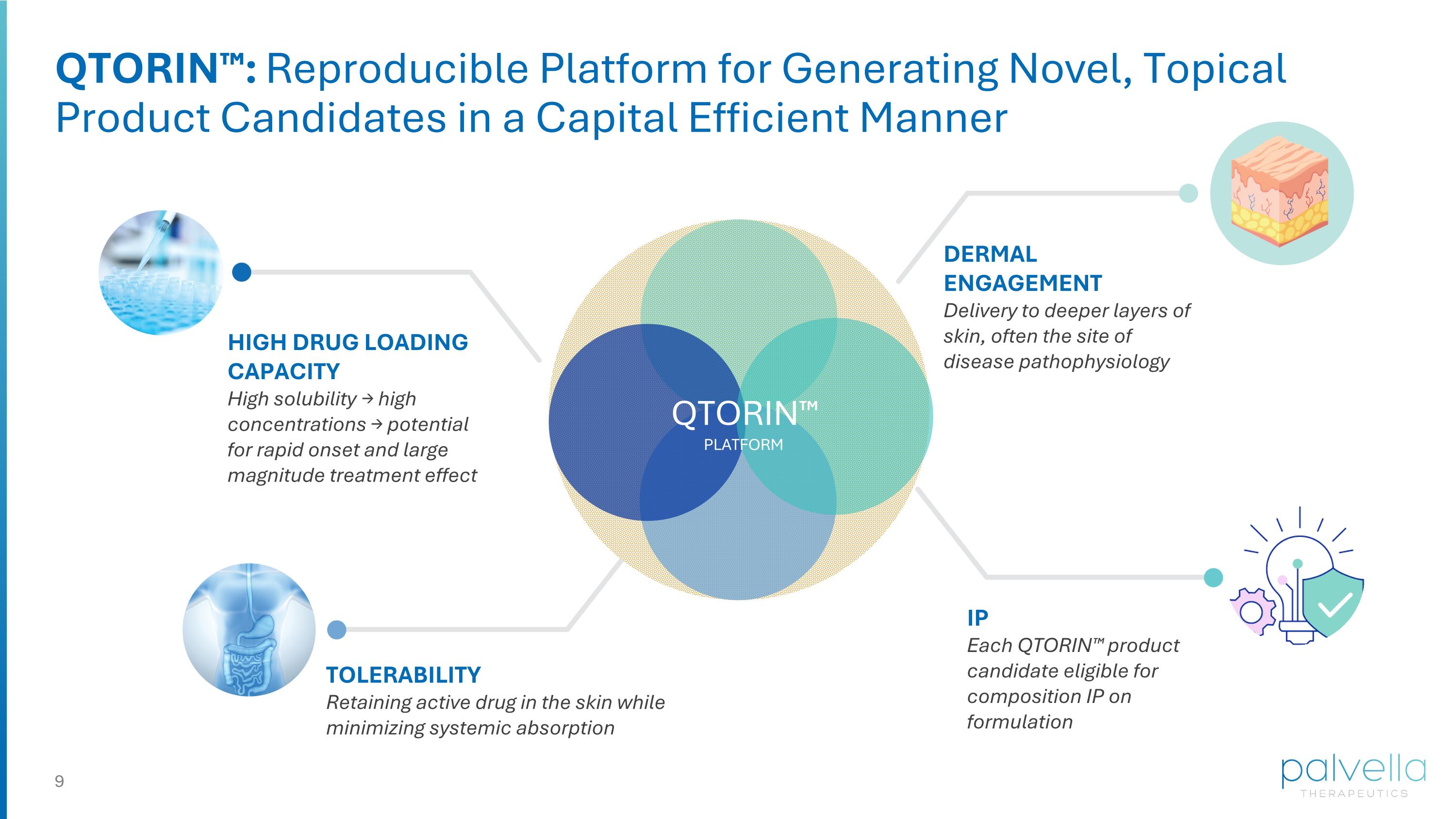
HIGH DRUG LOADING CAPACITY High solubility → high concentrations → potential for rapid onset and large magnitude treatment effect TOLERABILITY Retaining active drug in the skin while minimizing systemic absorption DERMAL ENGAGEMENT Delivery to deeper layers of skin, often the site of disease pathophysiology IP Each QTORIN™ product candidate eligible for composition IP on formulation QTORIN™: Reproducible Platform for Generating Novel, Topical Product Candidates in a Capital Efficient Manner QTORIN™ PLATFORM
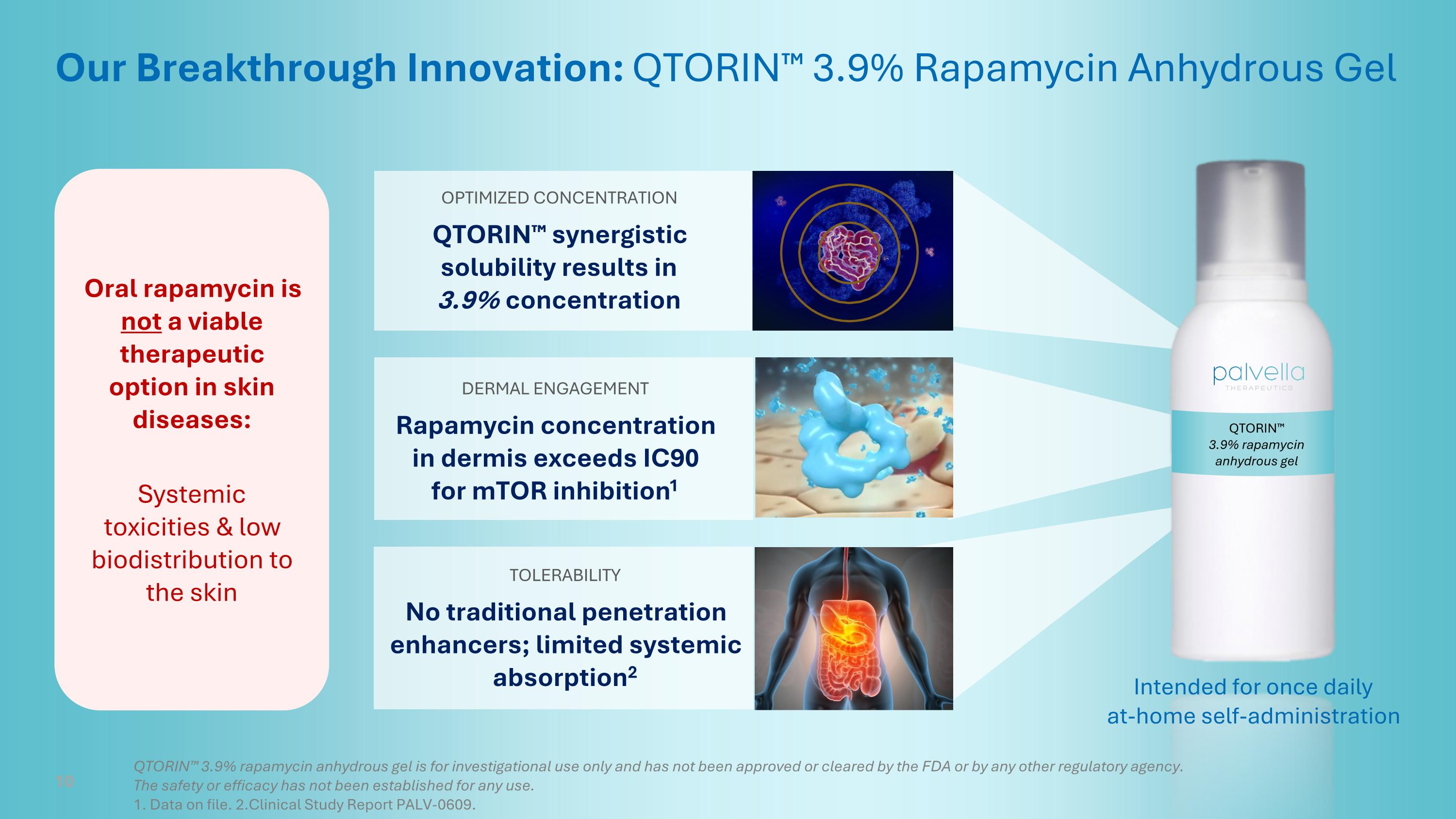
QTORIN™ 3.9% rapamycin anhydrous gel Our Breakthrough Innovation: QTORIN™ 3.9% Rapamycin Anhydrous Gel DERMAL ENGAGEMENT Rapamycin concentration in dermis exceeds IC90 for mTOR inhibition1 OPTIMIZED CONCENTRATION QTORIN™ synergistic solubility results in 3.9% concentration TOLERABILITY No traditional penetration enhancers; limited systemic absorption2 Intended for once daily at-home self-administration 10 QTORIN™ 3.9% rapamycin anhydrous gel is for investigational use only and has not been approved or cleared by the FDA or by any other regulatory agency. The safety or efficacy has not been established for any use. 1. Data on file. 2.Clinical Study Report PALV-0609. Oral rapamycin is not a viable therapeutic option in skin diseases: Systemic toxicities & low biodistribution to the skin
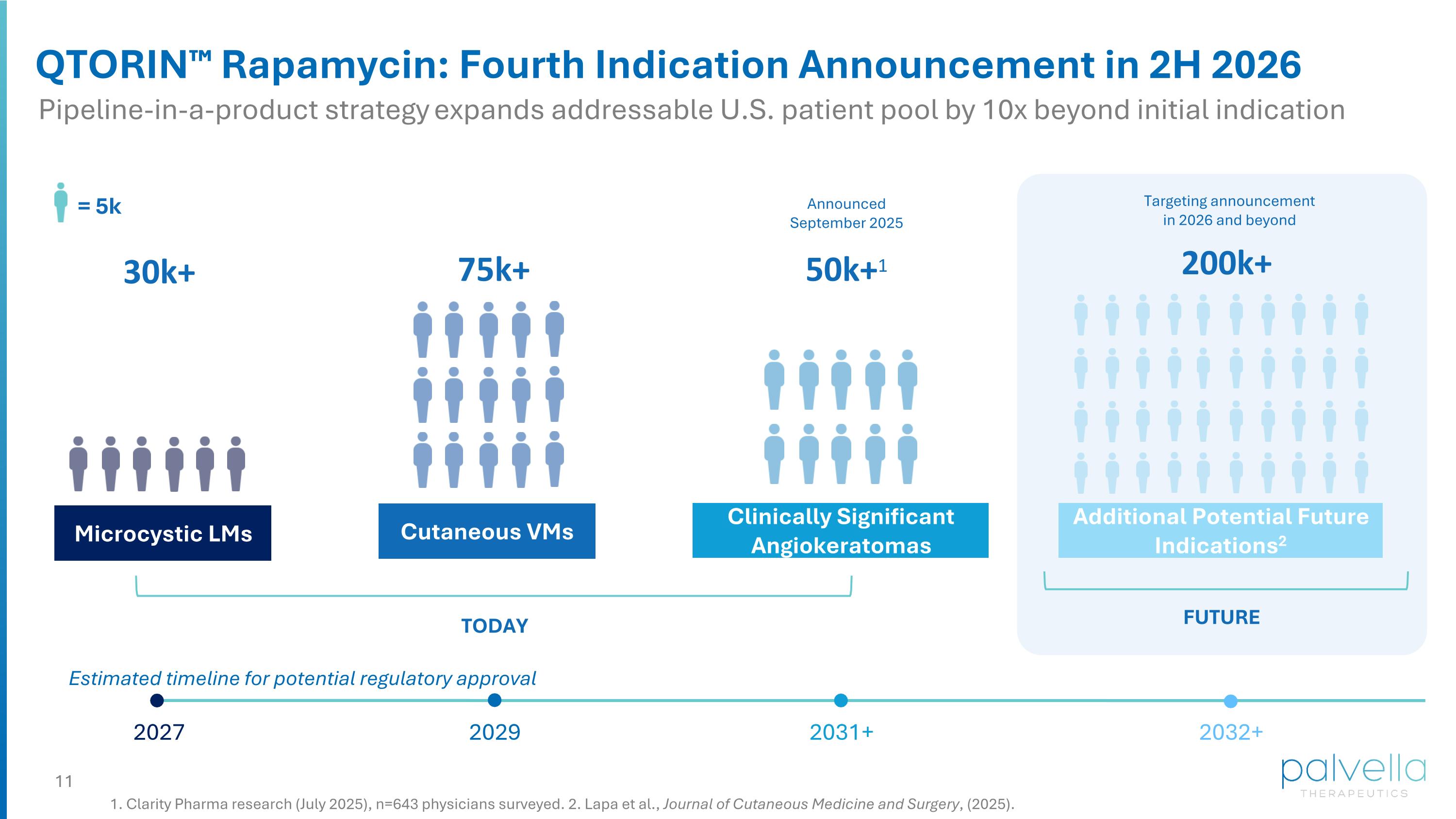
Microcystic LMs Cutaneous VMs Clinically Significant Angiokeratomas Additional Potential Future Indications2 TODAY FUTURE Announced September 2025 Targeting announcement in 2026 and beyond = 5k 30k+ 75k+ 200k+ 50k+1 QTORIN™ Rapamycin: Fourth Indication Announcement in 2H 2026 1. Clarity Pharma research (July 2025), n=643 physicians surveyed. 2. Lapa et al., Journal of Cutaneous Medicine and Surgery, (2025). Pipeline-in-a-product strategy expands addressable U.S. patient pool by 10x beyond initial indication Estimated timeline for potential regulatory approval 2031+ 2032+ 2027 2029
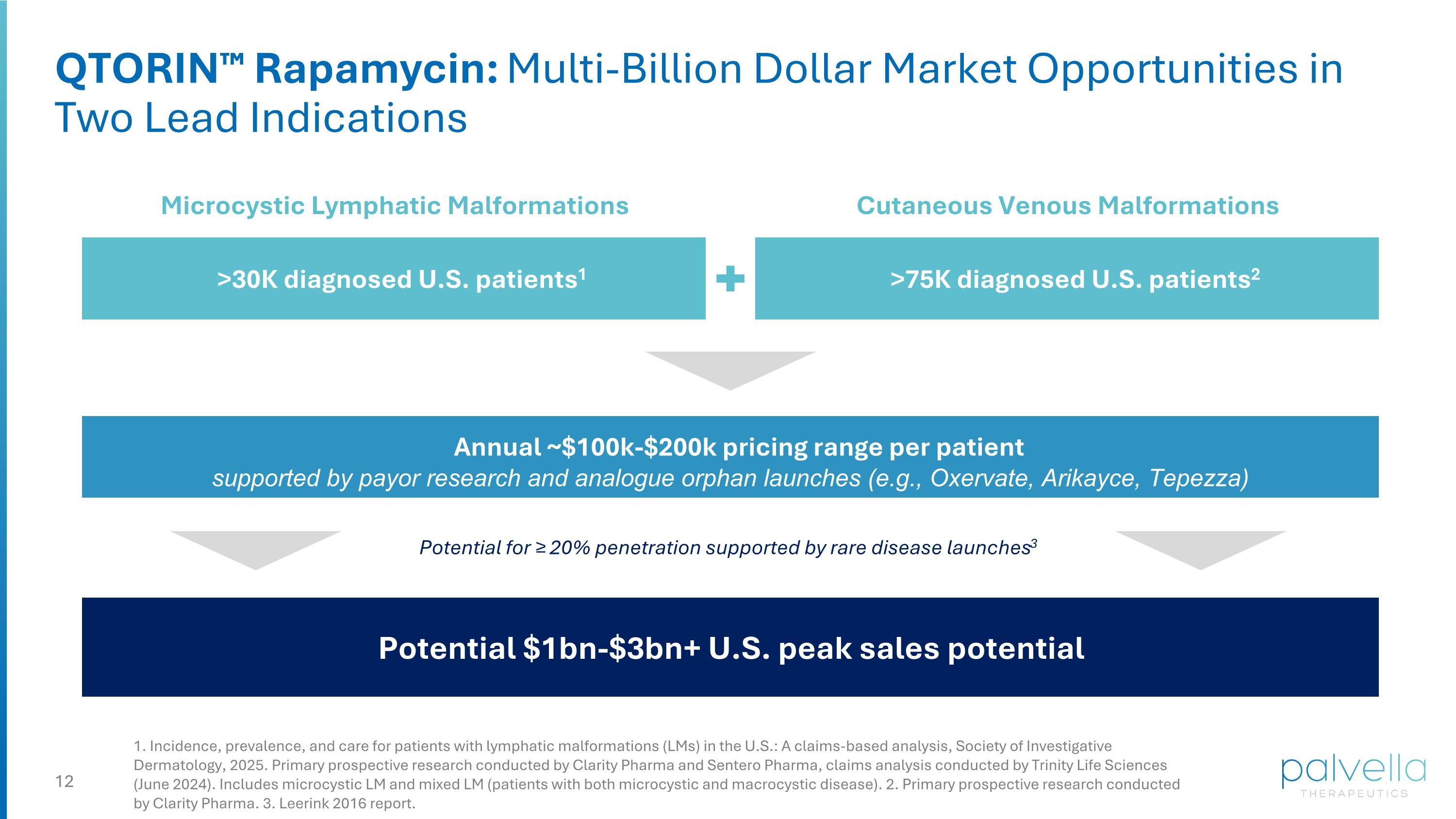
QTORIN™ Rapamycin: Multi-Billion Dollar Market Opportunities in Two Lead Indications >75K diagnosed U.S. patients2 Cutaneous Venous Malformations Potential $1bn-$3bn+ U.S. peak sales potential >30K diagnosed U.S. patients1 Microcystic Lymphatic Malformations Annual ~$100k-$200k pricing range per patient supported by payor research and analogue orphan launches (e.g., Oxervate, Arikayce, Tepezza) 1. Incidence, prevalence, and care for patients with lymphatic malformations (LMs) in the U.S.: A claims-based analysis, Society of Investigative Dermatology, 2025. Primary prospective research conducted by Clarity Pharma and Sentero Pharma, claims analysis conducted by Trinity Life Sciences (June 2024). Includes microcystic LM and mixed LM (patients with both microcystic and macrocystic disease). 2. Primary prospective research conducted by Clarity Pharma. 3. Leerink 2016 report. Potential for ≥ 20% penetration supported by rare disease launches3

QTORIN™ 3.9% RAPAMYCIN Microcystic Lymphatic Malformations FOR
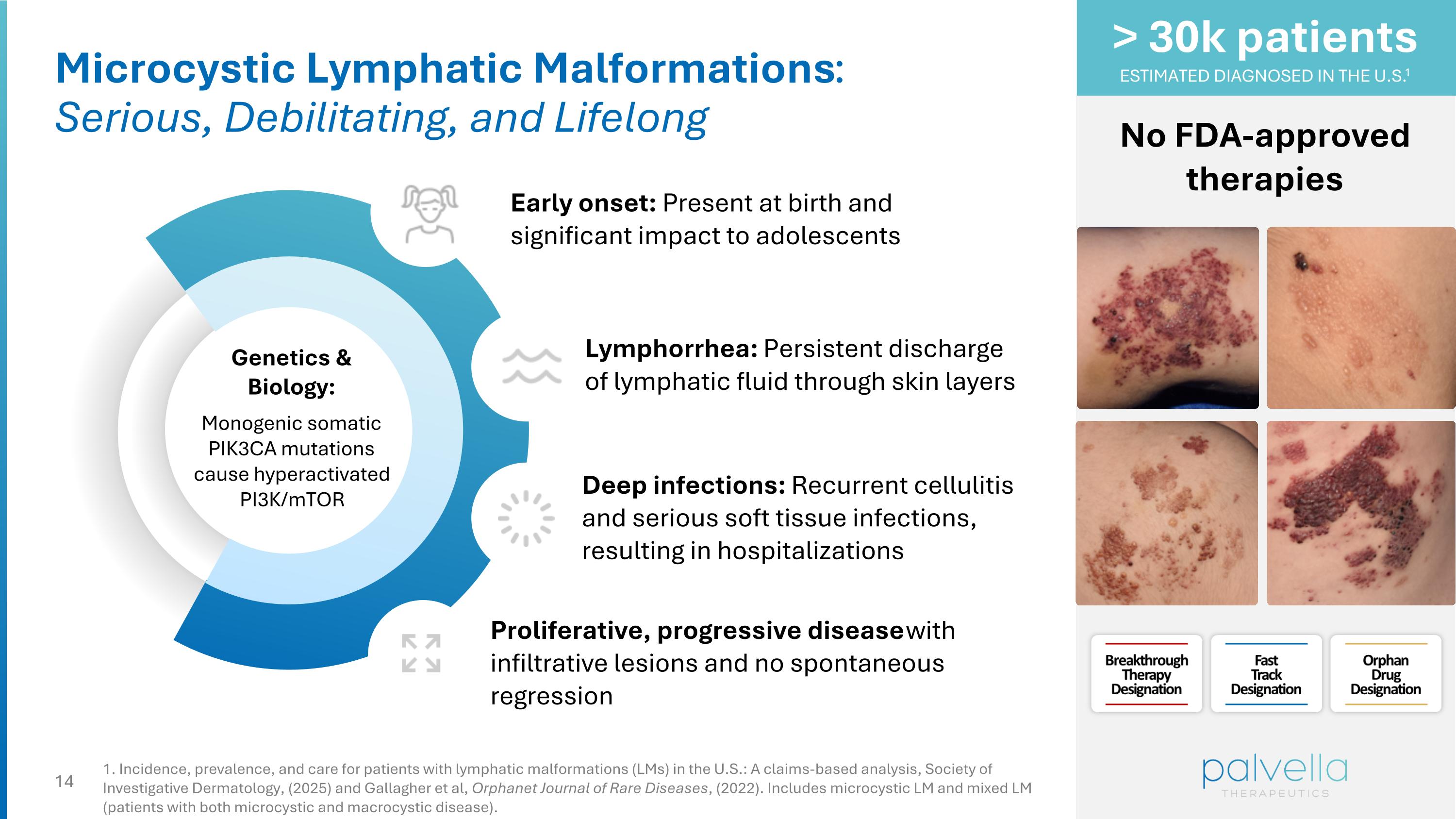
Microcystic Lymphatic Malformations: Serious, Debilitating, and Lifelong Genetics & Biology: Monogenic somatic PIK3CA mutations cause hyperactivated PI3K/mTOR Lymphorrhea: Persistent discharge of lymphatic fluid through skin layers Deep infections: Recurrent cellulitis and serious soft tissue infections, resulting in hospitalizations Proliferative, progressive disease with infiltrative lesions and no spontaneous regression No FDA-approved therapies > 30k patients ESTIMATED DIAGNOSED IN THE U.S.1 Early onset: Present at birth and significant impact to adolescents 1. Incidence, prevalence, and care for patients with lymphatic malformations (LMs) in the U.S.: A claims-based analysis, Society of Investigative Dermatology, (2025) and Gallagher et al, Orphanet Journal of Rare Diseases, (2022). Includes microcystic LM and mixed LM (patients with both microcystic and macrocystic disease).
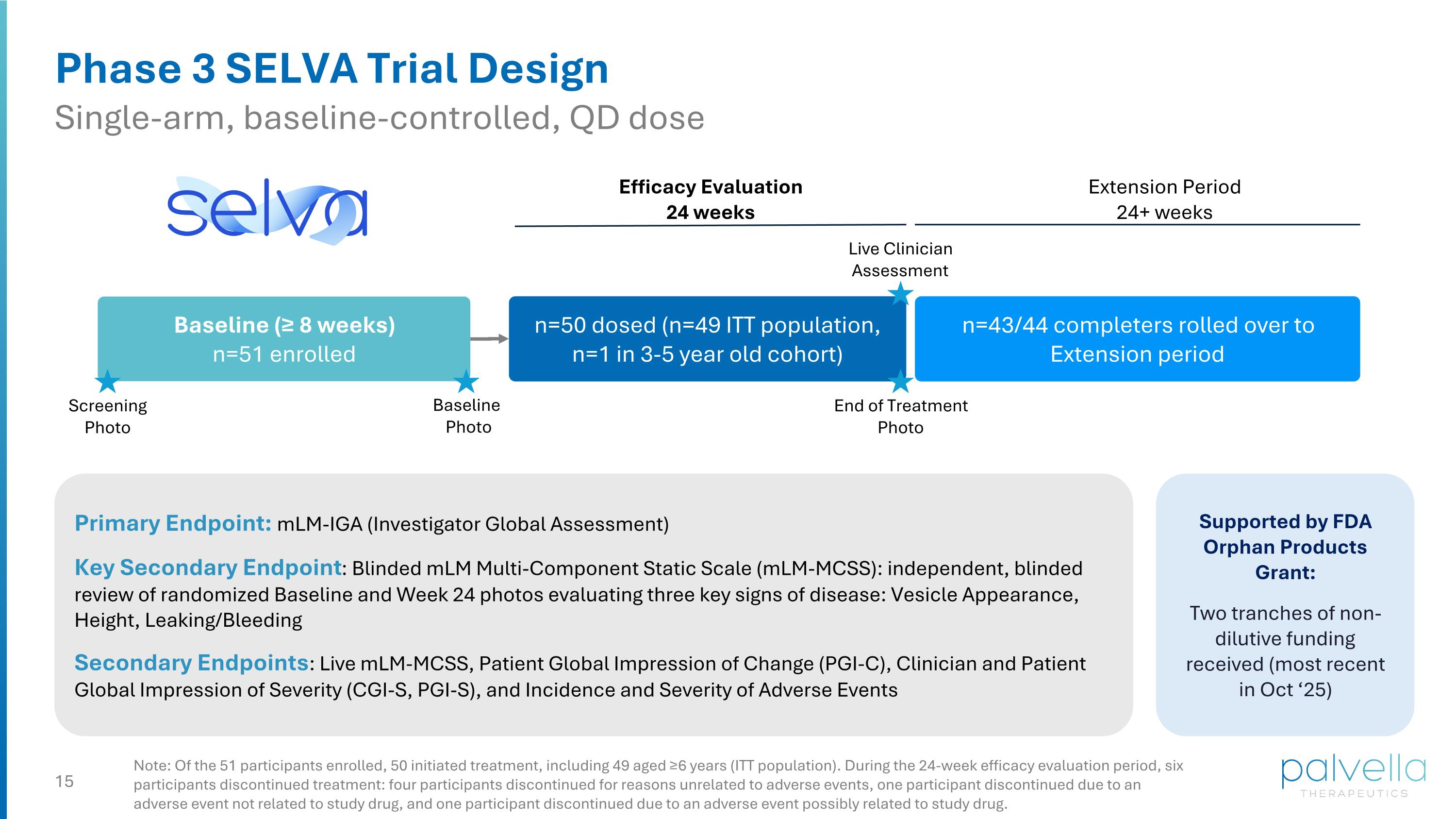
Phase 3 SELVA Trial Design Single-arm, baseline-controlled, QD dose Primary Endpoint: mLM-IGA (Investigator Global Assessment) Key Secondary Endpoint: Blinded mLM Multi-Component Static Scale (mLM-MCSS): independent, blinded review of randomized Baseline and Week 24 photos evaluating three key signs of disease: Vesicle Appearance, Height, Leaking/Bleeding Secondary Endpoints: Live mLM-MCSS, Patient Global Impression of Change (PGI-C), Clinician and Patient Global Impression of Severity (CGI-S, PGI-S), and Incidence and Severity of Adverse Events Note: Of the 51 participants enrolled, 50 initiated treatment, including 49 aged ≥6 years (ITT population). During the 24-week efficacy evaluation period, six participants discontinued treatment: four participants discontinued for reasons unrelated to adverse events, one participant discontinued due to an adverse event not related to study drug, and one participant discontinued due to an adverse event possibly related to study drug. n=50 dosed (n=49 ITT population, n=1 in 3-5 year old cohort) Extension Period 24+ weeks Efficacy Evaluation 24 weeks Baseline Photo Baseline (≥ 8 weeks) n=51 enrolled Live Clinician Assessment Supported by FDA Orphan Products Grant: Two tranches of non-dilutive funding received (most recent in Oct ‘25) n=43/44 completers rolled over to Extension period End of Treatment Photo Screening Photo

Upside Target Clinical Profile SELVA Outcome Statistically significant primary endpoint with mean mLM-IGA of ≥+1.5 at Week 24 Statistical significance on independent, blinded key secondary endpoint Safety profile: well-tolerated and similar to previous clinical trials Highly statistically significant with mean mLM-IGA of +2.13 at Week 24 (p<0.001) Highly statistically significant (p<0.001), including on all three clinical signs: vesicle appearance, height, leaking/bleeding Well-tolerated across both adult and pediatric patients, supporting chronic administration : QTORIN™ Rapamycin Exceeded Upside Case Profile Rollover into extension period in line with best-in-class drugs for rare diseases 98% of Week 24 completers (43/44) rolled over to Extension period Note: Data analyzed per statistical analysis plan; non-completer data handled via multiple imputation per statistical analysis plan; endpoints tested sequentially according to pre-specified hierarchical testing; statistical significance (p<0.05). Week 24 completers included 43 participants ≥6 years old and 1 participant 3-5 years old.

SELVA: Primary and Key Secondary Endpoints Achieved Mean Change at Week 24 (95% CI) p-value Primary Endpoint: Microcystic Lymphatic Malformation Investigator Global Assessment (mLM-IGA)* +2.13 (1.88, 2.38) p<0.001 Key Secondary Endpoint: Blinded Microcystic Lymphatic Malformation Multi-Component Static Scale** -3.36 (-4.34, -2.38) p<0.001 Note: Data analyzed per statistical analysis plan; non-completer data imputed per statistical analysis plan for primary endpoint; endpoints tested according to pre-specified hierarchical testing procedure; statistical significance (p<0.05). *Dynamic change scales (7-point scales ranging from "Very Much Worse" (-3) to "Very Much Improved" (+3); positive values indicate improvements from baseline) **mLM-MCSS (Sum of three static severity scales: Height, Leaking/Bleeding, Vesicle Appearance: Each scale rated "Clear or Almost Clear" (1) to "Very Severe" (5); total score 3-15. Test baseline to Week 24 change; negative values indicate improvements from baseline) Highly statistically significant across primary, key secondary, and all four secondary endpoints (all p<0.001)
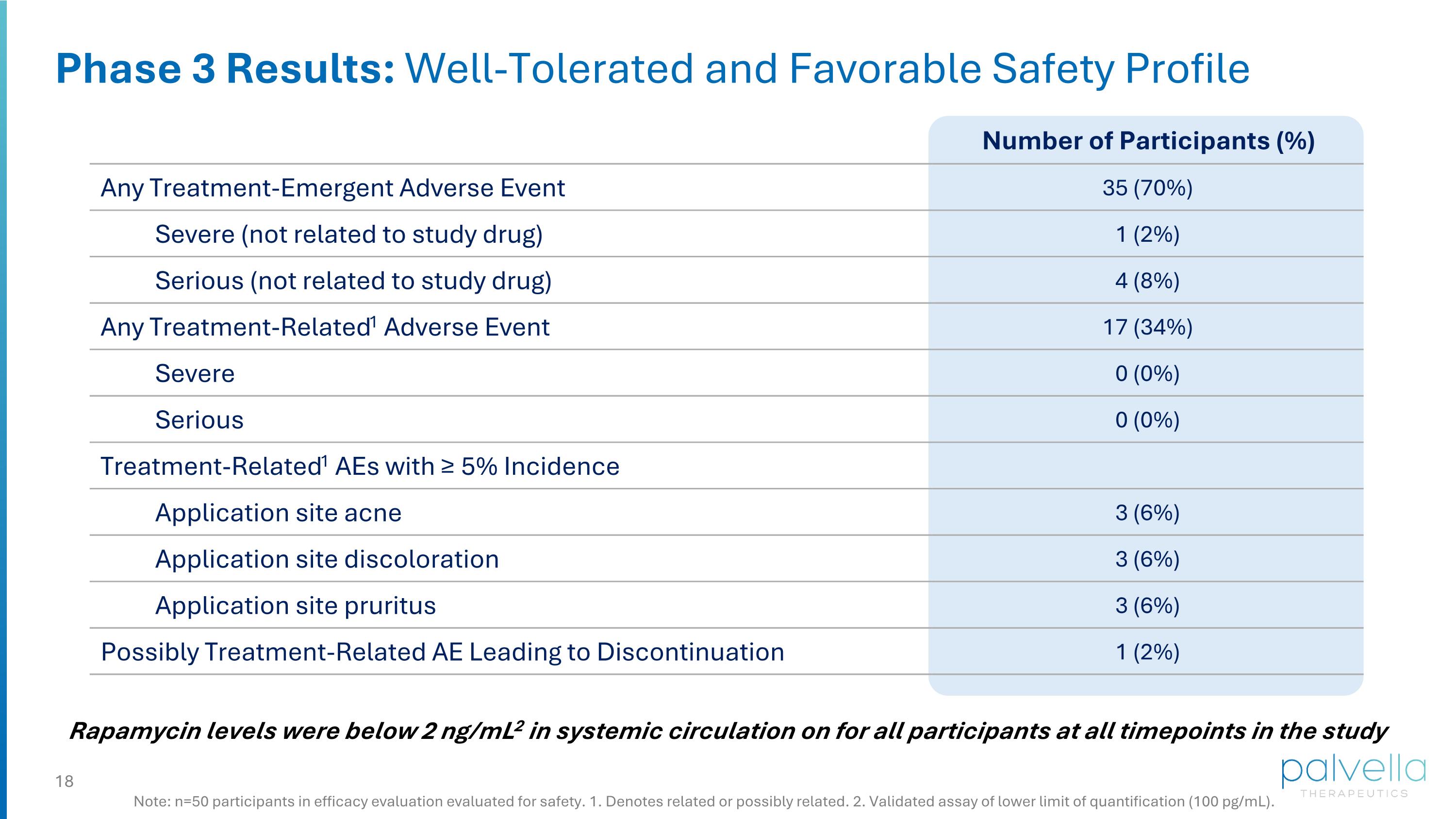
Phase 3 Results: Well-Tolerated and Favorable Safety Profile Rapamycin levels were below 2 ng/mL2 in systemic circulation on for all participants at all timepoints in the study Note: n=50 participants in efficacy evaluation evaluated for safety. 1. Denotes related or possibly related. 2. Validated assay of lower limit of quantification (100 pg/mL). Number of Participants (%) Any Treatment-Emergent Adverse Event 35 (70%) Severe (not related to study drug) 1 (2%) Serious (not related to study drug) 4 (8%) Any Treatment-Related1 Adverse Event 17 (34%) Severe 0 (0%) Serious 0 (0%) Treatment-Related1 AEs with ≥ 5% Incidence Application site acne 3 (6%) Application site discoloration 3 (6%) Application site pruritus 3 (6%) Possibly Treatment-Related AE Leading to Discontinuation 1 (2%)
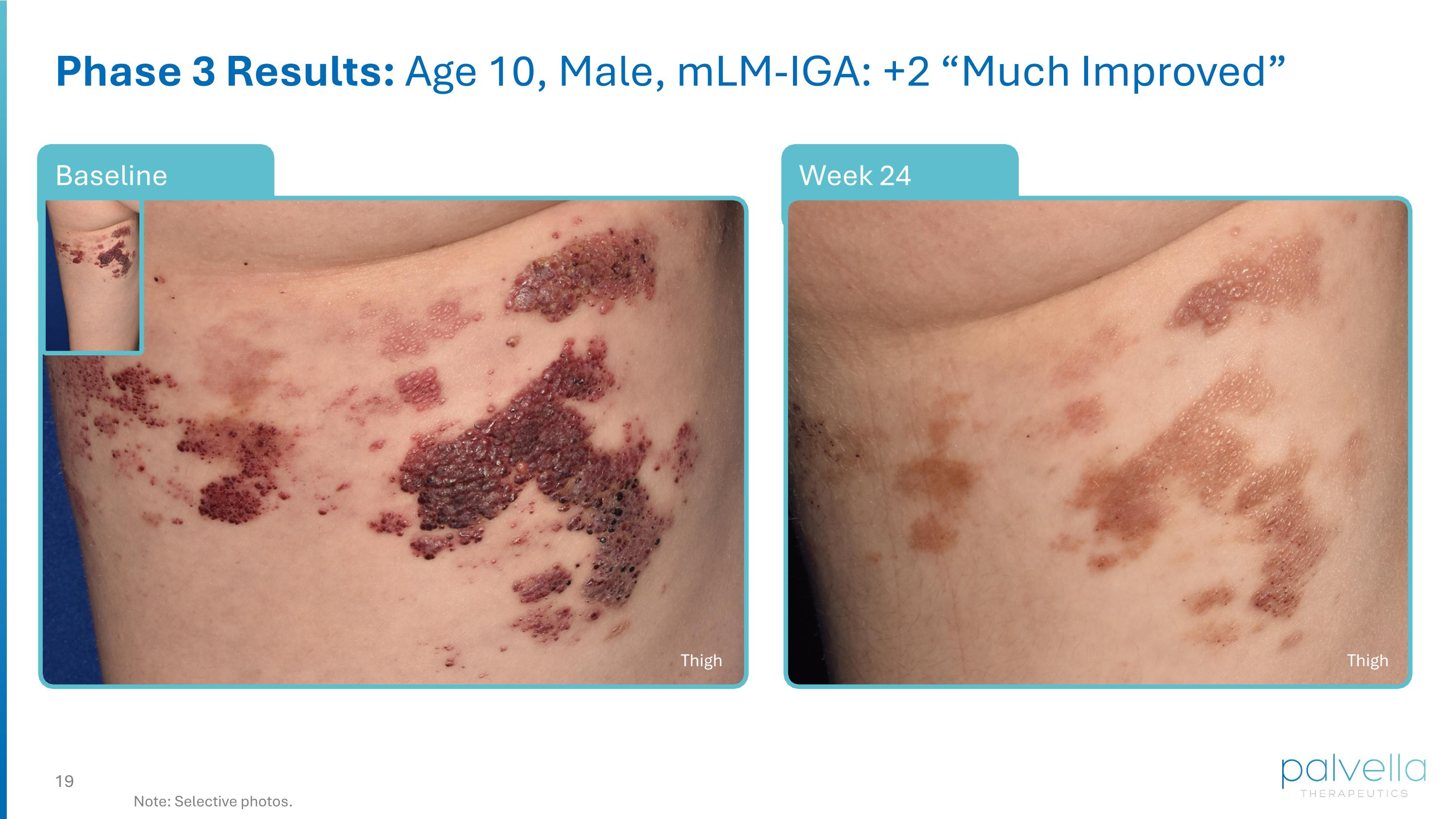
Week 24 Baseline Phase 3 Results: Age 10, Male, mLM-IGA: +2 “Much Improved” Note: Selective photos. Thigh Thigh
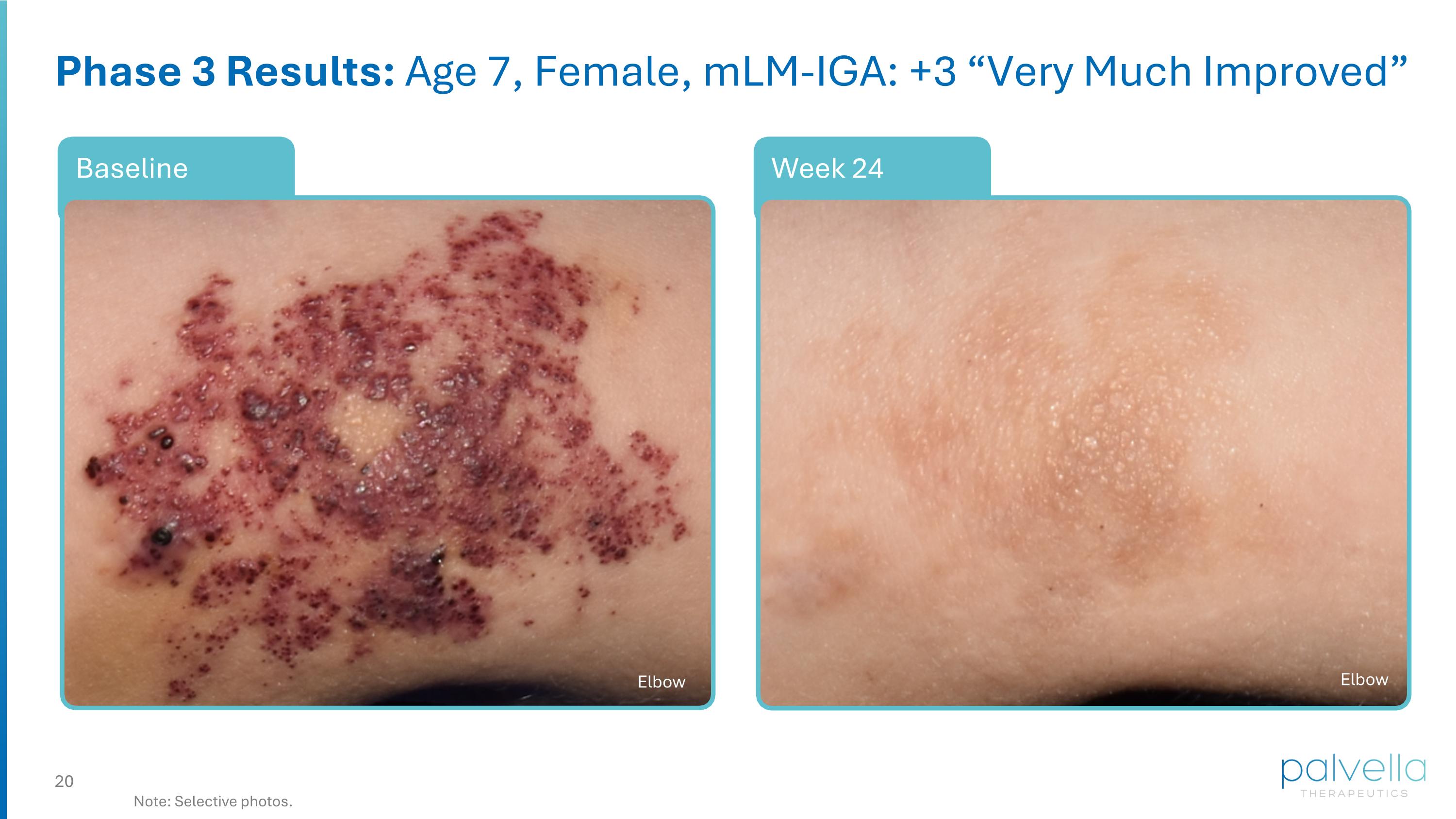
Week 24 Baseline Phase 3 Results: Age 7, Female, mLM-IGA: +3 “Very Much Improved” Note: Selective photos. Elbow Elbow
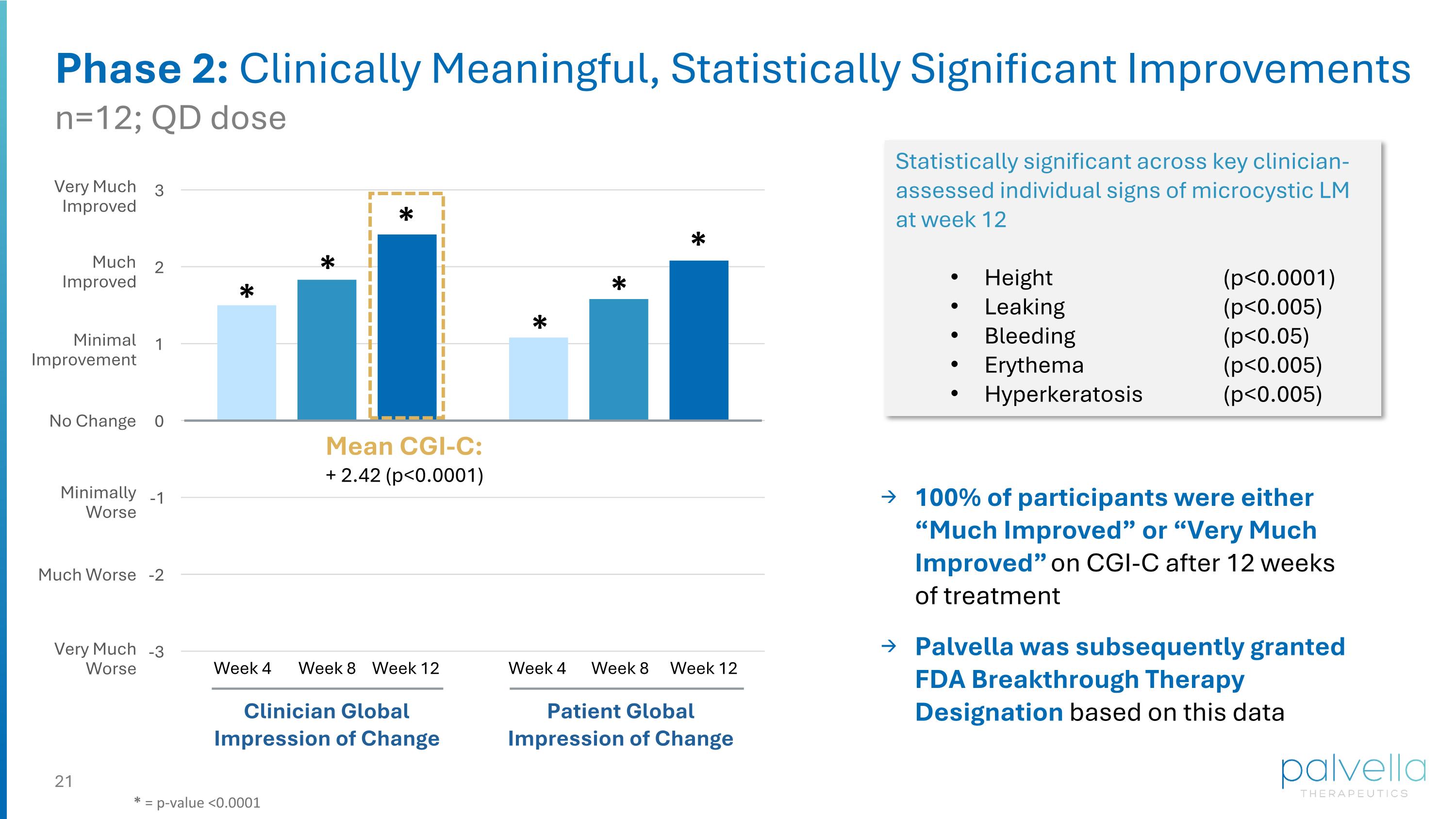
* * * * * * Statistically significant across key clinician-assessed individual signs of microcystic LM at week 12 Height (p<0.0001) Leaking (p<0.005) Bleeding (p<0.05) Erythema (p<0.005) Hyperkeratosis (p<0.005) Phase 2: Clinically Meaningful, Statistically Significant Improvements Mean CGI-C: + 2.42 (p<0.0001) Clinician Global Impression of Change Patient Global Impression of Change Week 4 Week 8 Week 12 Week 4 Week 8 Week 12 Very Much Improved Much Improved Minimal Improvement No Change Minimally Worse Much Worse Very Much Worse n=12; QD dose * = p-value <0.0001 100% of participants were either “Much Improved” or “Very Much Improved” on CGI-C after 12 weeks of treatment Palvella was subsequently granted FDA Breakthrough Therapy Designation based on this data

Phase 3 SELVA data highly statistically significant and clinically meaningful, confirming positive Phase 2 results 1 Seeking traditional approval (not accelerated) based on clinical endpoints; intent to seek broad label age 3 and above 2 Previously granted Breakthrough, Fast Track, and Orphan Designations; 505(b)(2) submission 3 Collaborating with FDA Office of Orphan Products Development, which is providing non-dilutive funding, to participate in Pre-NDA meeting 4 Regulatory: NDA Submission On Track for 2H 2026 Breakthrough, Fast Track, and Orphan Designations granted Pre-NDA meeting granted by FDA, with meeting expected Q2 2026 Since April 2025, there have been at least 10 drug approvals based on one or more single-arm studies, of which 8 were orphan-designated1 1. Hyman Phelps & McNamara analysis.
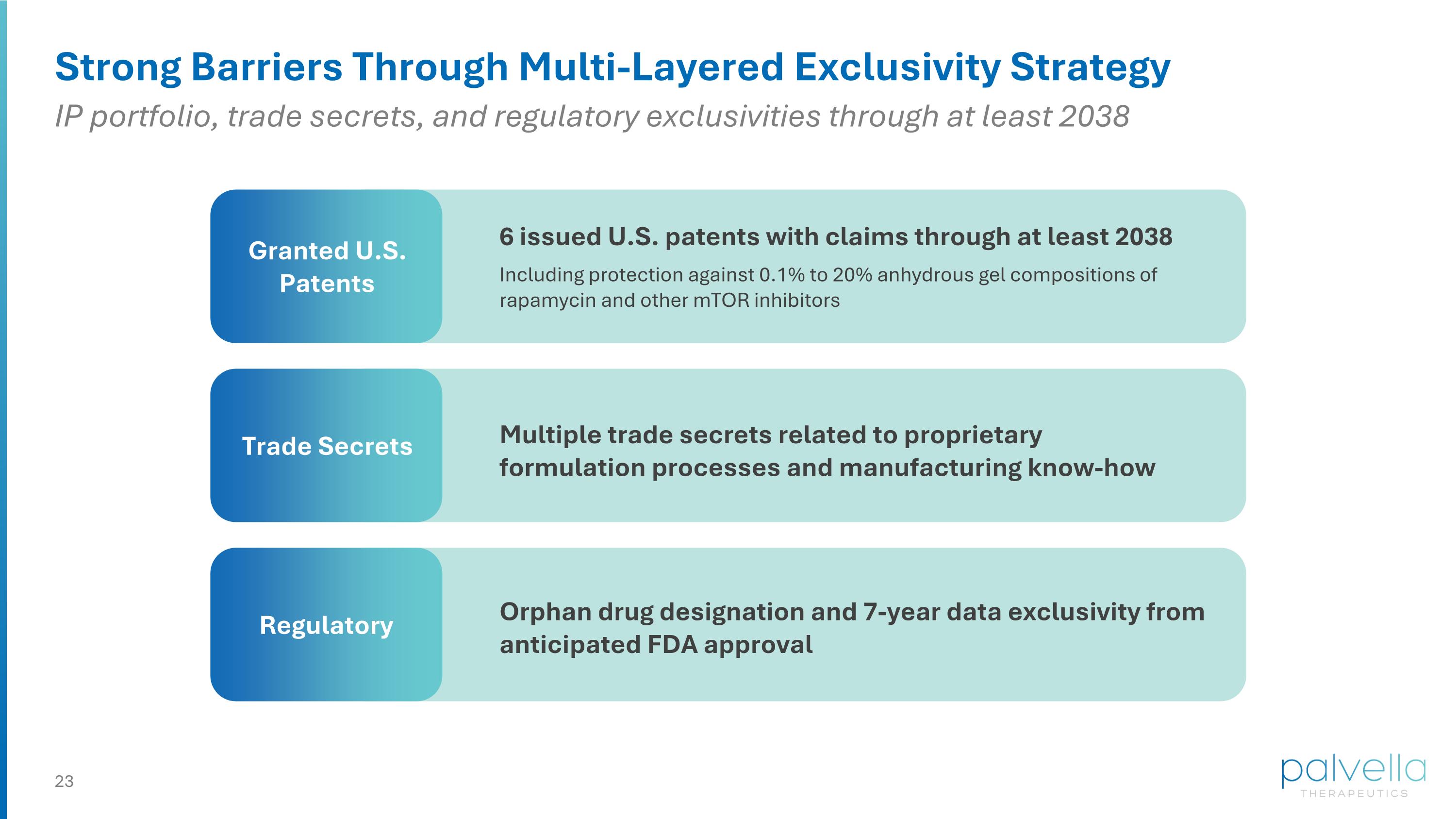
Strong Barriers Through Multi-Layered Exclusivity Strategy Granted U.S. Patents Trade Secrets Regulatory Orphan drug designation and 7-year data exclusivity from anticipated FDA approval Multiple trade secrets related to proprietary formulation processes and manufacturing know-how 6 issued U.S. patents with claims through at least 2038 Including protection against 0.1% to 20% anhydrous gel compositions of rapamycin and other mTOR inhibitors IP portfolio, trade secrets, and regulatory exclusivities through at least 2038

QTORIN™ 3.9% RAPAMYCIN U.S. Commercial Launch Planning

Microcystic LMs: Multi-billion Dollar, Uncontested U.S. Market Opportunity with Commercial Build-Out Underway Large orphan market: Claims analysis presented at 2025 Society for Investigative Dermatology (SID) Meeting verified > 30k estimated diagnosed U.S. patients, with >1,500 incident patients annually 1 Positioned to be first and only FDA-approved therapy; market research indicates strong intent to prescribe, including in pediatric population 2 Payor testing and orphan analogues validate expected orphan pricing corridor above $100k per patient per year 3 Concentrated prescriber base in vascular anomaly centers (VACs) & other clinics 4
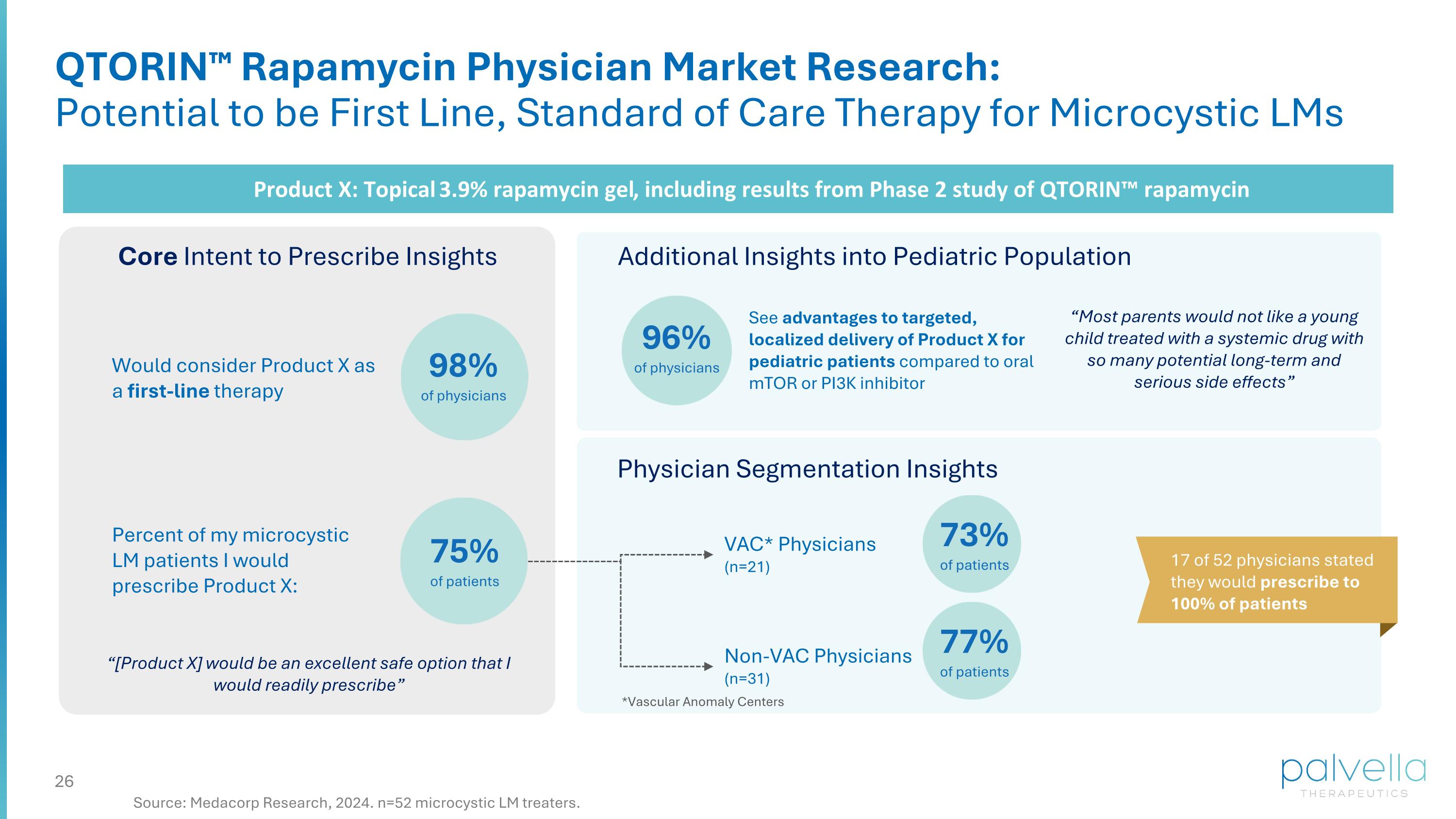
QTORIN™ Rapamycin Physician Market Research: Potential to be First Line, Standard of Care Therapy for Microcystic LMs Product X: Topical 3.9% rapamycin gel, including results from Phase 2 study of QTORIN™ rapamycin Core Intent to Prescribe Insights 98% of physicians Percent of my microcystic LM patients I would prescribe Product X: 75% of patients Additional Insights into Pediatric Population Physician Segmentation Insights VAC* Physicians (n=21) Non-VAC Physicians (n=31) 73% of patients 77% of patients *Vascular Anomaly Centers 17 of 52 physicians stated they would prescribe to 100% of patients “[Product X] would be an excellent safe option that I would readily prescribe” Would consider Product X as a first-line therapy 96% of physicians See advantages to targeted, localized delivery of Product X for pediatric patients compared to oral mTOR or PI3K inhibitor “Most parents would not like a young child treated with a systemic drug with so many potential long-term and serious side effects” Source: Medacorp Research, 2024. n=52 microcystic LM treaters.
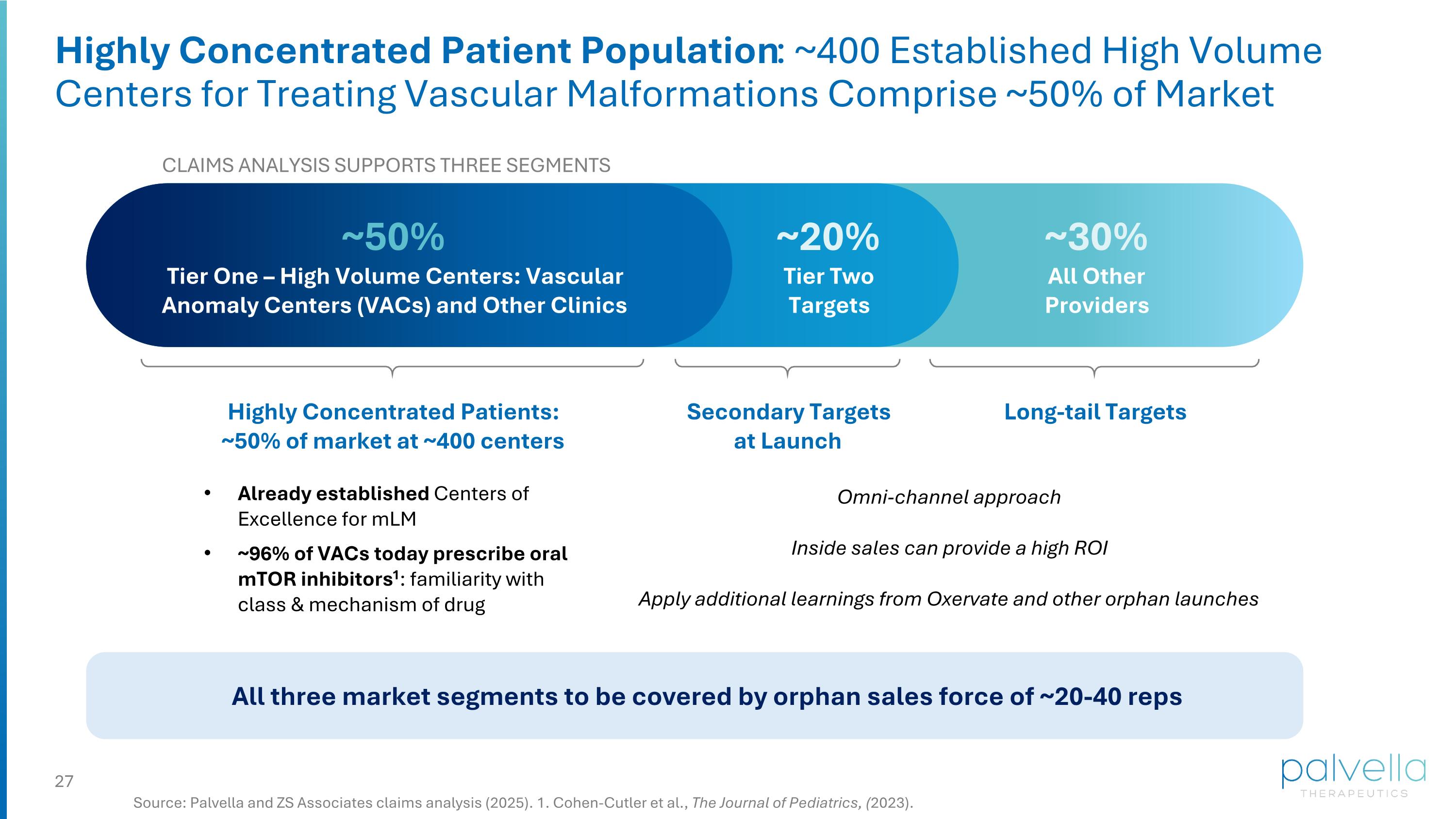
Highly Concentrated Patient Population: ~400 Established High Volume Centers for Treating Vascular Malformations Comprise ~50% of Market Source: Palvella and ZS Associates claims analysis (2025). 1. Cohen-Cutler et al., The Journal of Pediatrics, (2023). CLAIMS ANALYSIS SUPPORTS THREE SEGMENTS ~50% Tier One – High Volume Centers: Vascular Anomaly Centers (VACs) and Other Clinics Already established Centers of Excellence for mLM ~96% of VACs today prescribe oral mTOR inhibitors1: familiarity with class & mechanism of drug ~20% Tier Two Targets ~30% All Other Providers Highly Concentrated Patients: ~50% of market at ~400 centers Secondary Targets at Launch Long-tail Targets Omni-channel approach Inside sales can provide a high ROI Apply additional learnings from Oxervate and other orphan launches All three market segments to be covered by orphan sales force of ~20-40 reps
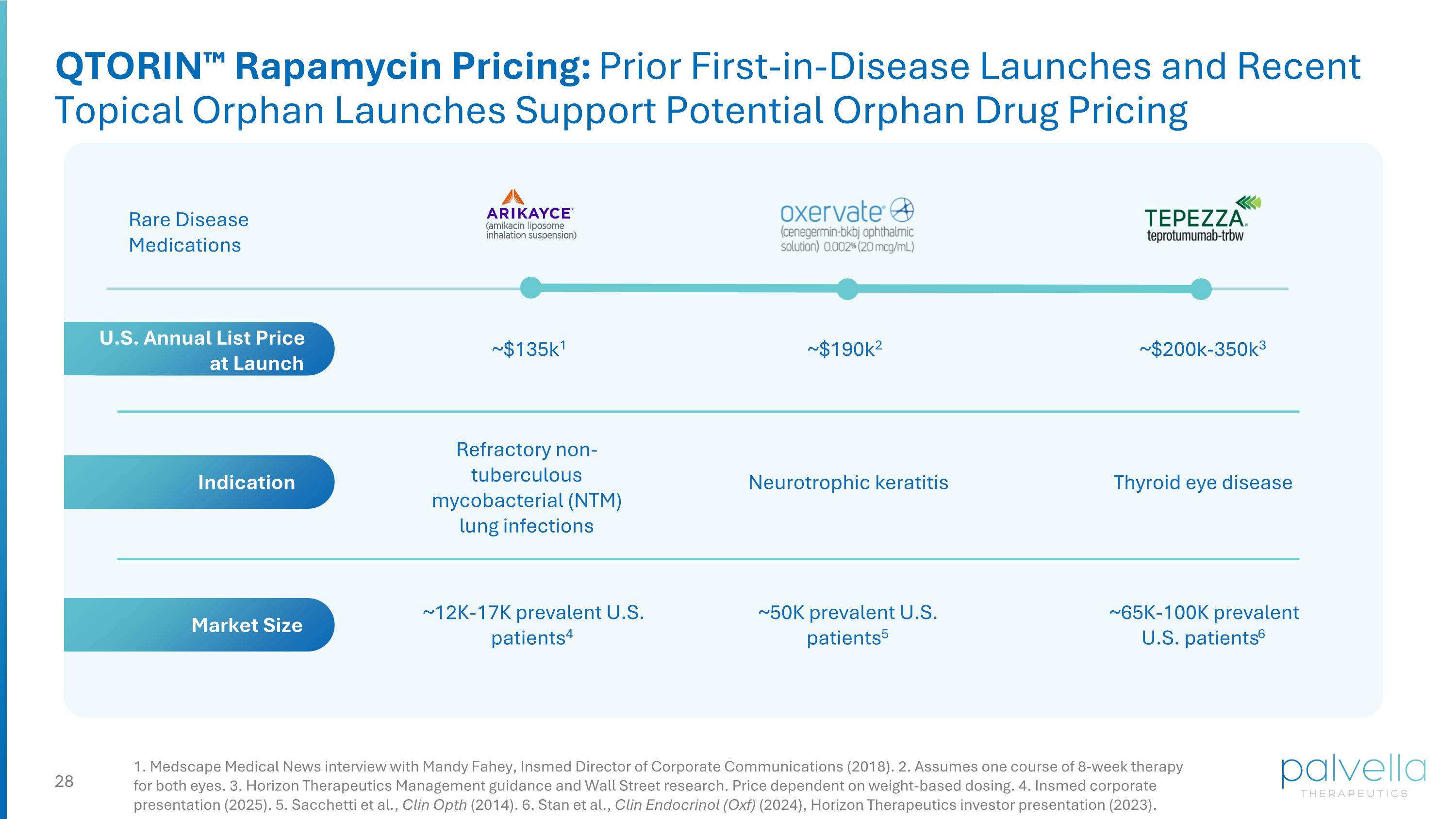
1. Medscape Medical News interview with Mandy Fahey, Insmed Director of Corporate Communications (2018). 2. Assumes one course of 8-week therapy for both eyes. 3. Horizon Therapeutics Management guidance and Wall Street research. Price dependent on weight-based dosing. 4. Insmed corporate presentation (2025). 5. Sacchetti et al., Clin Opth (2014). 6. Stan et al., Clin Endocrinol (Oxf) (2024), Horizon Therapeutics investor presentation (2023). ~$135k1 Rare Disease Medications Refractory non-tuberculous mycobacterial (NTM) lung infections Thyroid eye disease ~12K-17K prevalent U.S. patients4 ~65K-100K prevalent U.S. patients6 ~$200k-350k3 Indication Market Size U.S. Annual List Price at Launch QTORIN™ Rapamycin Pricing: Prior First-in-Disease Launches and Recent Topical Orphan Launches Support Potential Orphan Drug Pricing Neurotrophic keratitis ~50K prevalent U.S. patients5 ~$190k2

QTORIN™ 3.9% RAPAMYCIN Cutaneous Venous Malformations FOR
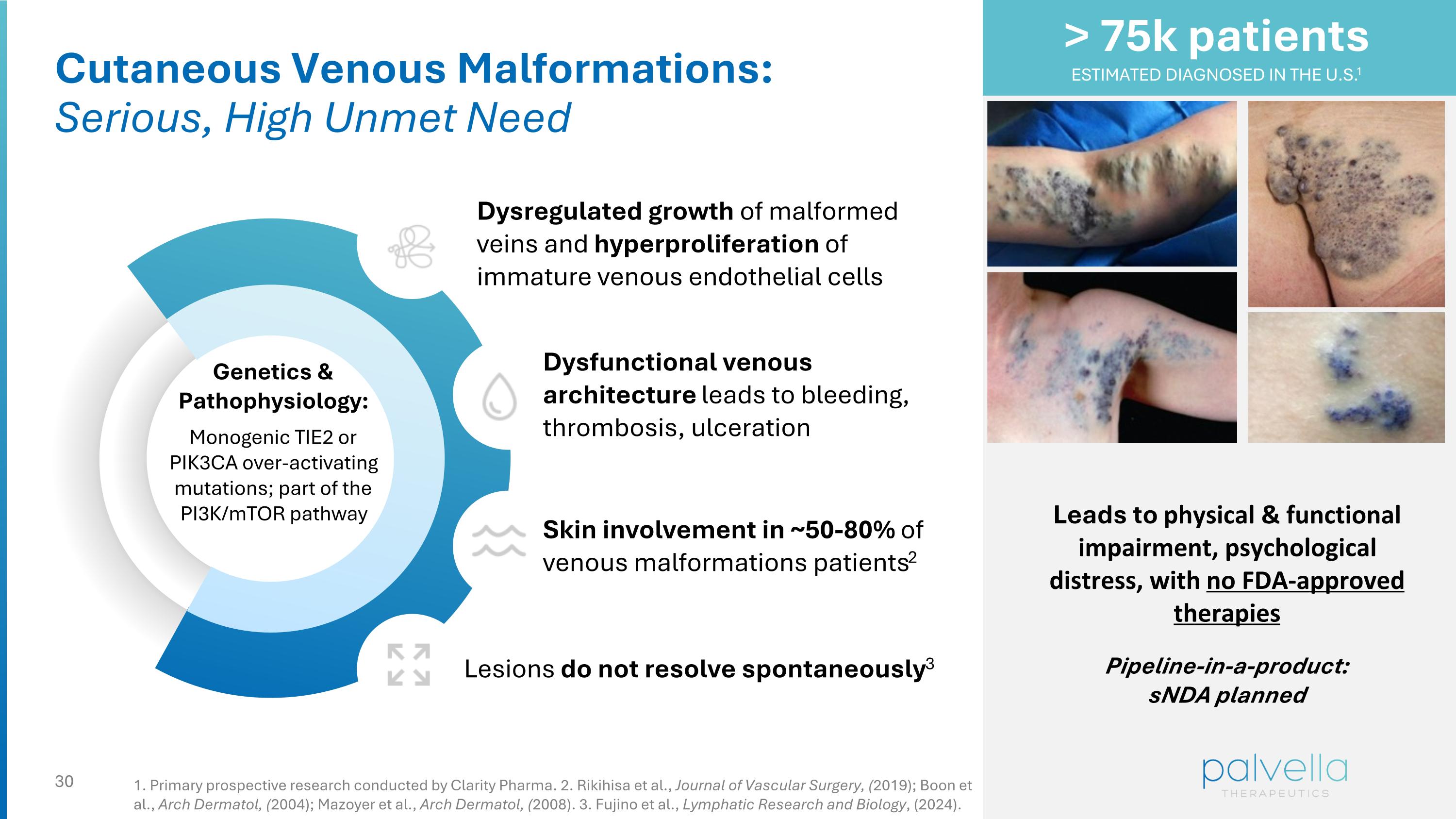
Cutaneous Venous Malformations: Serious, High Unmet Need Genetics & Pathophysiology: Monogenic TIE2 or PIK3CA over-activating mutations; part of the PI3K/mTOR pathway Dysregulated growth of malformed veins and hyperproliferation of immature venous endothelial cells Dysfunctional venous architecture leads to bleeding, thrombosis, ulceration Lesions do not resolve spontaneously3 Skin involvement in ~50-80% of venous malformations patients2 Leads to physical & functional impairment, psychological distress, with no FDA-approved therapies Pipeline-in-a-product: sNDA planned > 75k patients ESTIMATED DIAGNOSED IN THE U.S.1 1. Primary prospective research conducted by Clarity Pharma. 2. Rikihisa et al., Journal of Vascular Surgery, (2019); Boon et al., Arch Dermatol, (2004); Mazoyer et al., Arch Dermatol, (2008). 3. Fujino et al., Lymphatic Research and Biology, (2024).

Venous Malformations: Progress Towards the Potential First Targeted Therapy for Unaddressed Cutaneous Disease Known Genetics / Clear Biology Real-world Evidence Real-world evidence supports rapamycin’s off-label use in primarily internal manifestations of VMs… …however, poor patient outcomes persist in cutaneous disease Limaye et al (2009, 2015) Teng et al (2025) QTORIN™ RAPAMYCIN On Target, In Tissue Phase 2 TOIVA Data (2025) Plausible mechanism 3.9% concentration Dermal penetration Extensive CMC package QTORIN™ 3.9% rapamycin anhydrous gel is for investigational use only and has not been approved or cleared by the FDA or by any other regulatory agency. The safety or efficacy has not been established for any use.
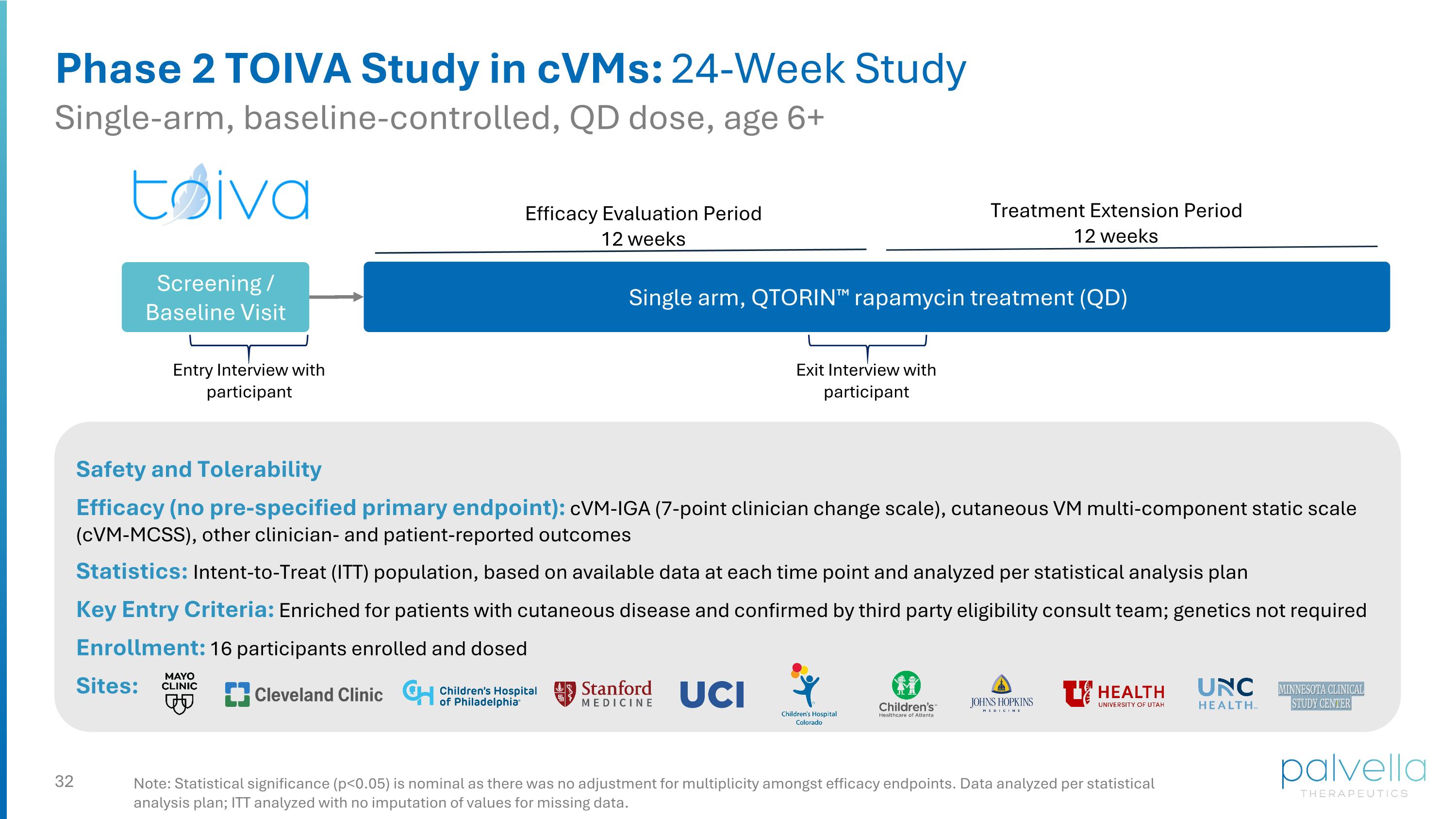
Phase 2 TOIVA Study in cVMs: 24-Week Study Single-arm, baseline-controlled, QD dose, age 6+ Safety and Tolerability Efficacy (no pre-specified primary endpoint): cVM-IGA (7-point clinician change scale), cutaneous VM multi-component static scale (cVM-MCSS), other clinician- and patient-reported outcomes Statistics: Intent-to-Treat (ITT) population, based on available data at each time point and analyzed per statistical analysis plan Key Entry Criteria: Enriched for patients with cutaneous disease and confirmed by third party eligibility consult team; genetics not required Enrollment: 16 participants enrolled and dosed Sites: Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; ITT analyzed with no imputation of values for missing data. Single arm, QTORIN™ rapamycin treatment (QD) Efficacy Evaluation Period 12 weeks Screening / Baseline Visit Exit Interview with participant Entry Interview with participant Treatment Extension Period 12 weeks
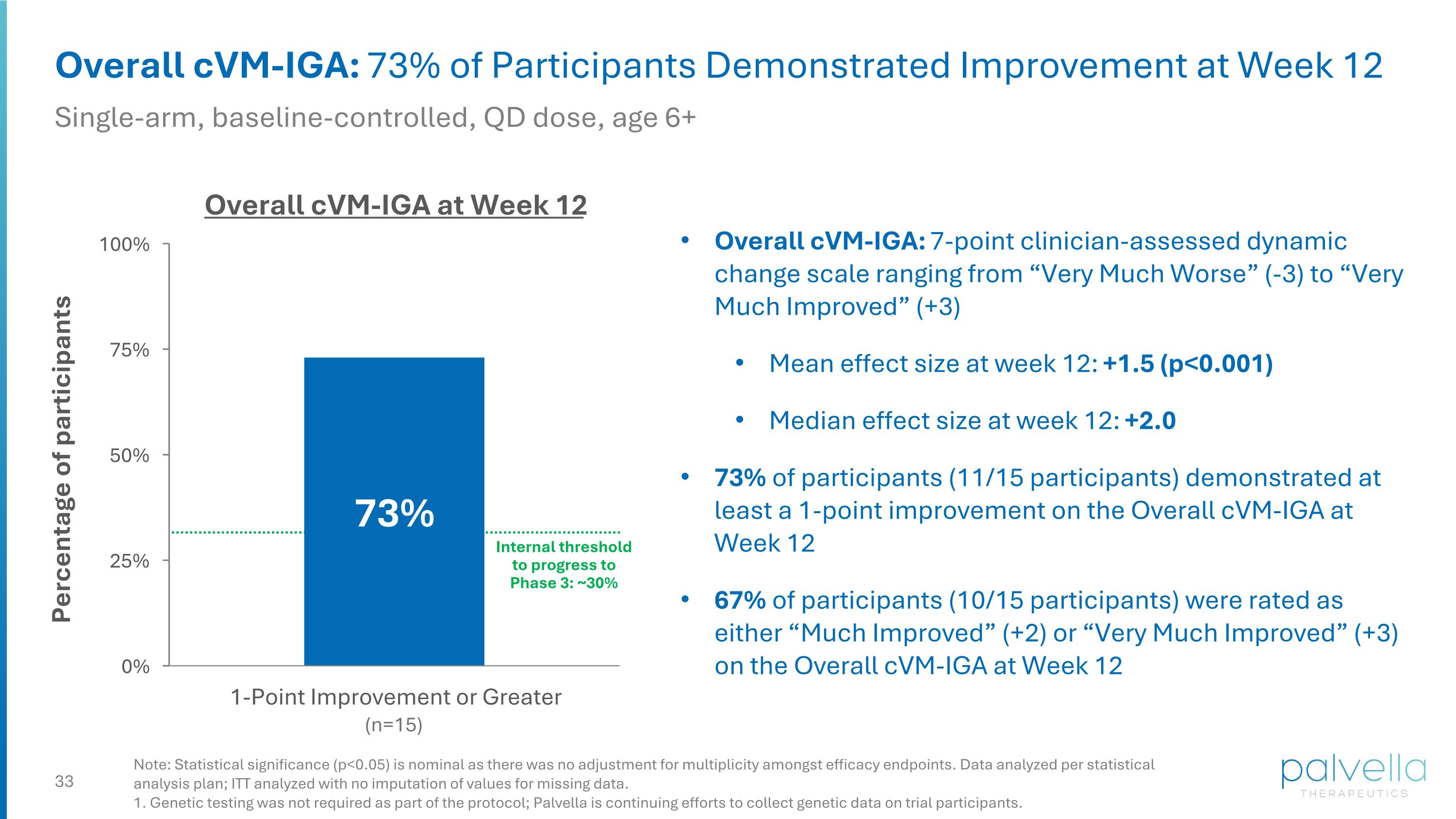
Overall cVM-IGA: 73% of Participants Demonstrated Improvement at Week 12 Single-arm, baseline-controlled, QD dose, age 6+ Overall cVM-IGA: 7-point clinician-assessed dynamic change scale ranging from “Very Much Worse” (-3) to “Very Much Improved” (+3) Mean effect size at week 12: +1.5 (p<0.001) Median effect size at week 12: +2.0 73% of participants (11/15 participants) demonstrated at least a 1-point improvement on the Overall cVM-IGA at Week 12 67% of participants (10/15 participants) were rated as either “Much Improved” (+2) or “Very Much Improved” (+3) on the Overall cVM-IGA at Week 12 Overall cVM-IGA at Week 12 (n=15) Internal threshold to progress to Phase 3: ~30% Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; ITT analyzed with no imputation of values for missing data. 1. Genetic testing was not required as part of the protocol; Palvella is continuing efforts to collect genetic data on trial participants.
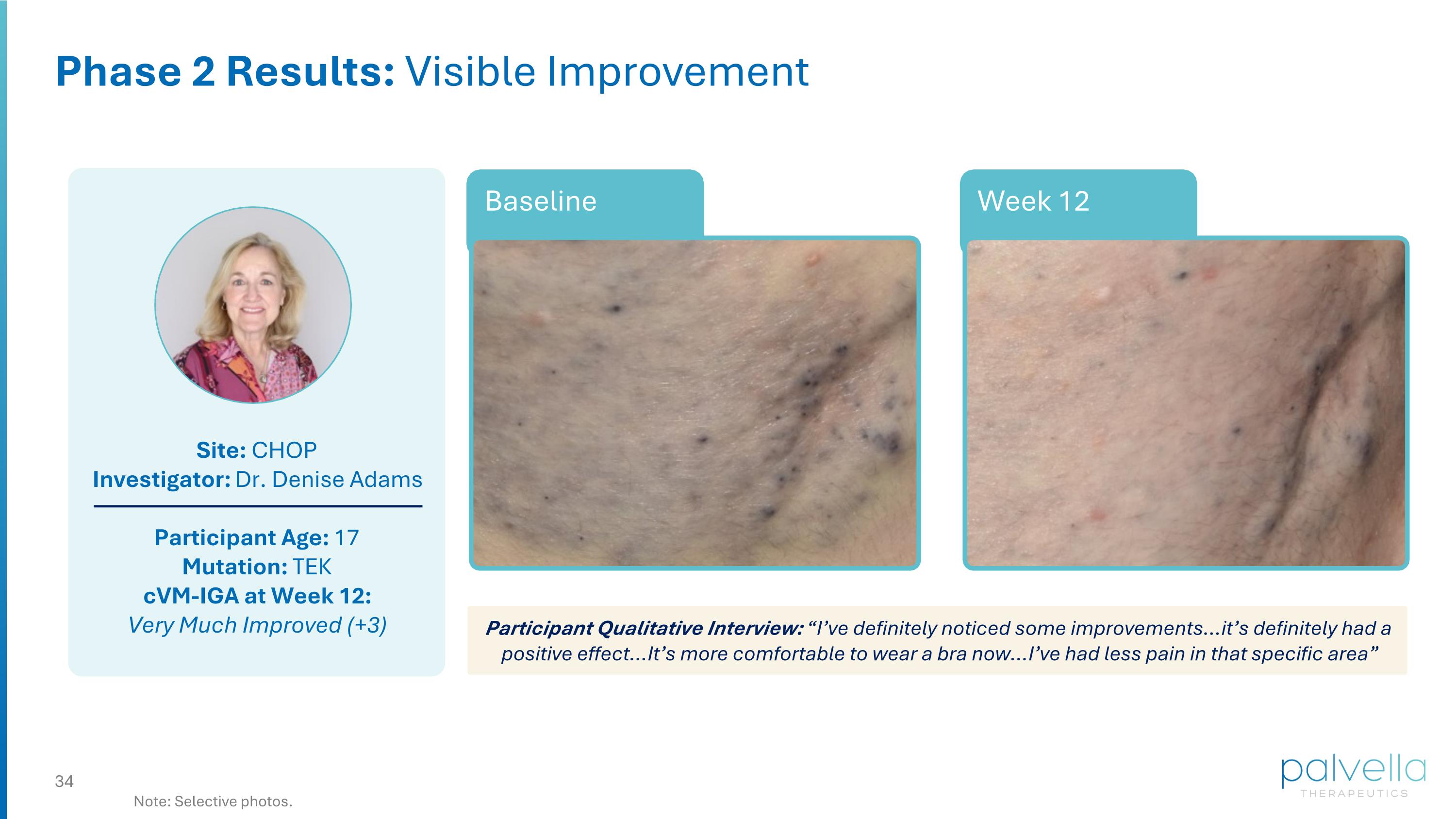
Site: CHOP Investigator: Dr. Denise Adams Participant Age: 17 Mutation: TEK cVM-IGA at Week 12: Very Much Improved (+3) Week 12 Baseline Phase 2 Results: Visible Improvement Participant Qualitative Interview: “I’ve definitely noticed some improvements…it’s definitely had a positive effect…It’s more comfortable to wear a bra now…I’ve had less pain in that specific area” Note: Selective photos.

Phase 2 Results: Safety and Tolerability QTORIN™ rapamycin was generally well-tolerated, similar to previous clinical trials Most common Treatment-Emergent Adverse Events were application site reactions (erythema, 25%) All Treatment-Related Adverse Events were moderate or mild (no severe events) Majority of AEs were mild No SAEs related to study drug No unexpected AEs Rapamycin levels were below the lower limit of quantification (2 ng/mL) in systemic circulation on a standard lab assay for all participants at all timepoints in the study Significantly below 5 ng/mL which is the lower boundary where rapamycin begins to exert immunosuppressive effects

QTORIN™ 3.9% RAPAMYCIN Clinically Significant Angiokeratomas FOR

> 50k patients ESTIMATED DIAGNOSED IN THE U.S.1 Clinically Significant Angiokeratomas: Superficial Lymphatic Malformations Persistent and extensive: Lesions can be large and increase in size, number, and extent over time Chronically debilitating lymphatic-derived skin lesions associated with bleeding, pain, and functional impairment Recurrent bleeding: Friction can cause fragile lesions to frequently bleed Disease Biology: Increased VEGF and mTOR signaling, leading to vessel dilation and hyperkeratosis Natural history: No tendency for spontaneous regression Wang et al., Journal of Cutaneous Pathology, (2014); Trindade et al., Am J Dermopathol, (2014); Prindaville et al., Pediatric Dermatology, (2017); Singh et al, Indian Journal of Dermatology, (2023); Caraffa et al, International Journal of Infection, (2025); Molla, Clinical, Cosmetic and Investigative Dermatology, (2024). Ivy H, Julian CA. Angiokeratoma Circumscriptum. Treasure Island (FL): StatPearls Publishing; 2025 Jan; Lapa et al., Journal of Cutaneous Medicine and Surgery, (2025). 1. Clarity Pharma research (July 2025), n=643 physicians surveyed. Palvella’s focus to include Fordyce, Solitary, Mibelli, and Circumscriptum subtypes No FDA-approved therapies Pipeline-in-a-product: sNDA planned Phase 2 trial initiation expected Q2 2026 ahead of schedule

QTORIN™ PITAVASTATIN Disseminated Superficial Actinic Porokeratosis FOR

> 50k patients ESTIMATED DIAGNOSED IN THE U.S.1 Disseminated Superficial Actinic Porokeratosis (DSAP): Chronic, Pre-Cancerous, and Progressive Persistent and extensive: Clonal proliferation of abnormal keratinocytes leads to increased number and size of lesions Risk of malignant transformation: Premalignant disease with transformation to non-melanoma skin cancers2 Genetics & Disease Biology: Autosomal dominant (primary) or de novo germline mutation leads to accumulation of toxic intermediates Natural history: Spontaneous regression is extremely rare2 No FDA-approved therapies Phase 2 trial initiation expected 2H 2026 1. Clarity Pharma research (April 2025), n=277 physicians surveyed. 2. Williams, Grant M., et al. “Porokeratosis.” StatPearls Publishing, 2024. Significant impact to quality of life: clinical signs include skin disfigurement, burning, and persistent itch

Unmet Need for First FDA-approved Topical Mevalonate Pathway Inhibitor for DSAP Proof-of-concept study, demonstrating a plausible mechanistic approach Significant need for an FDA-approved topical mevalonate pathway inhibitor Recently published systematic review in Clinical and Experimental Dermatology (Martini et al, 2026) included 24 case studies of off-label use of topical statin therapy in porokeratosis… …however, today poor patient outcomes persist due to lack of access and known variability in unapproved formulations which can limit safety, efficacy, and quality Oral statins are not a viable therapeutic option in DSAP: High first pass metabolism and/or sub-therapeutic biodistribution to the skin
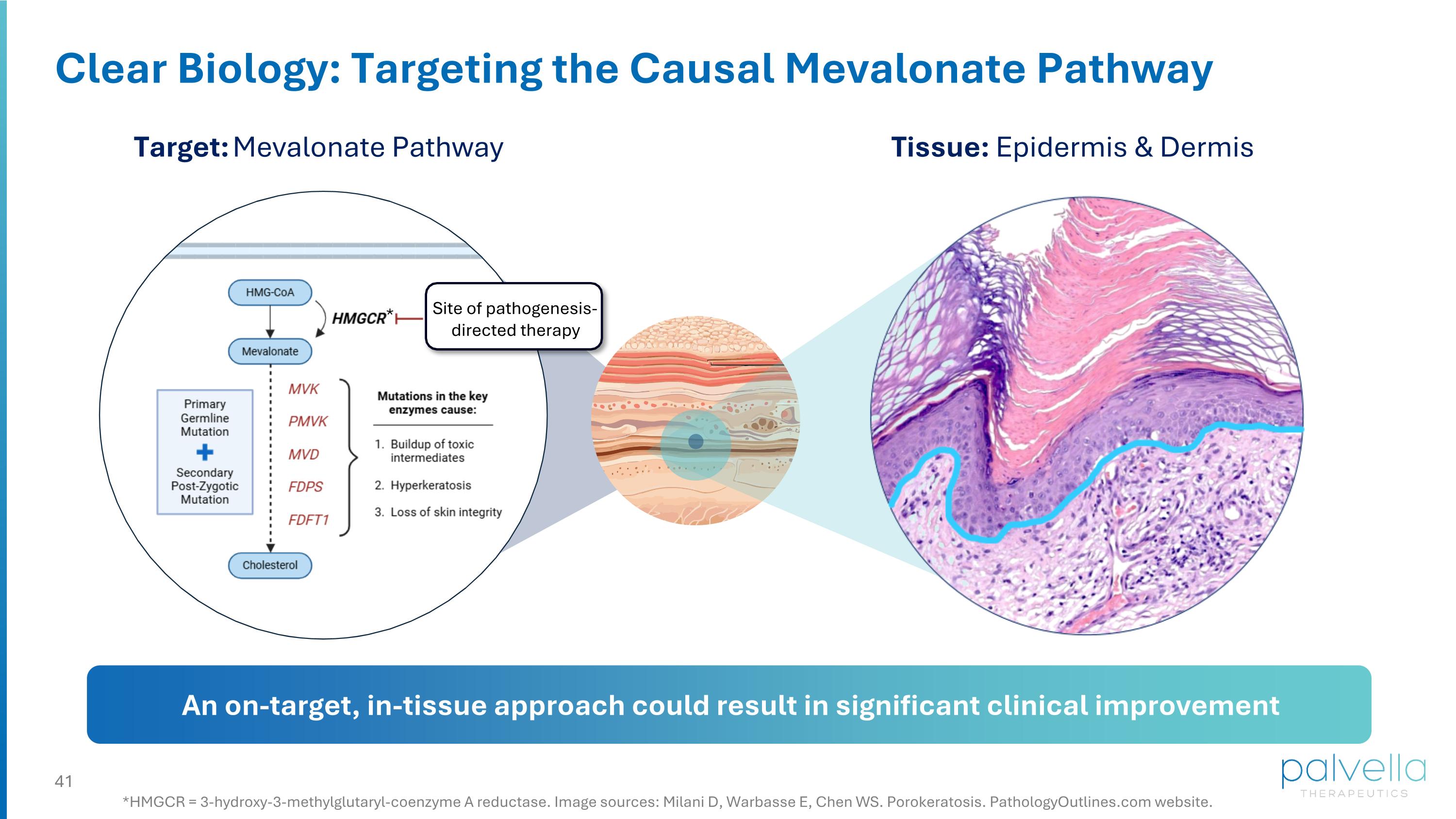
Clear Biology: Targeting the Causal Mevalonate Pathway Target: Mevalonate Pathway Tissue: Epidermis & Dermis Site of pathogenesis-directed therapy An on-target, in-tissue approach could result in significant clinical improvement *HMGCR = 3-hydroxy-3-methylglutaryl-coenzyme A reductase. Image sources: Milani D, Warbasse E, Chen WS. Porokeratosis. PathologyOutlines.com website. *
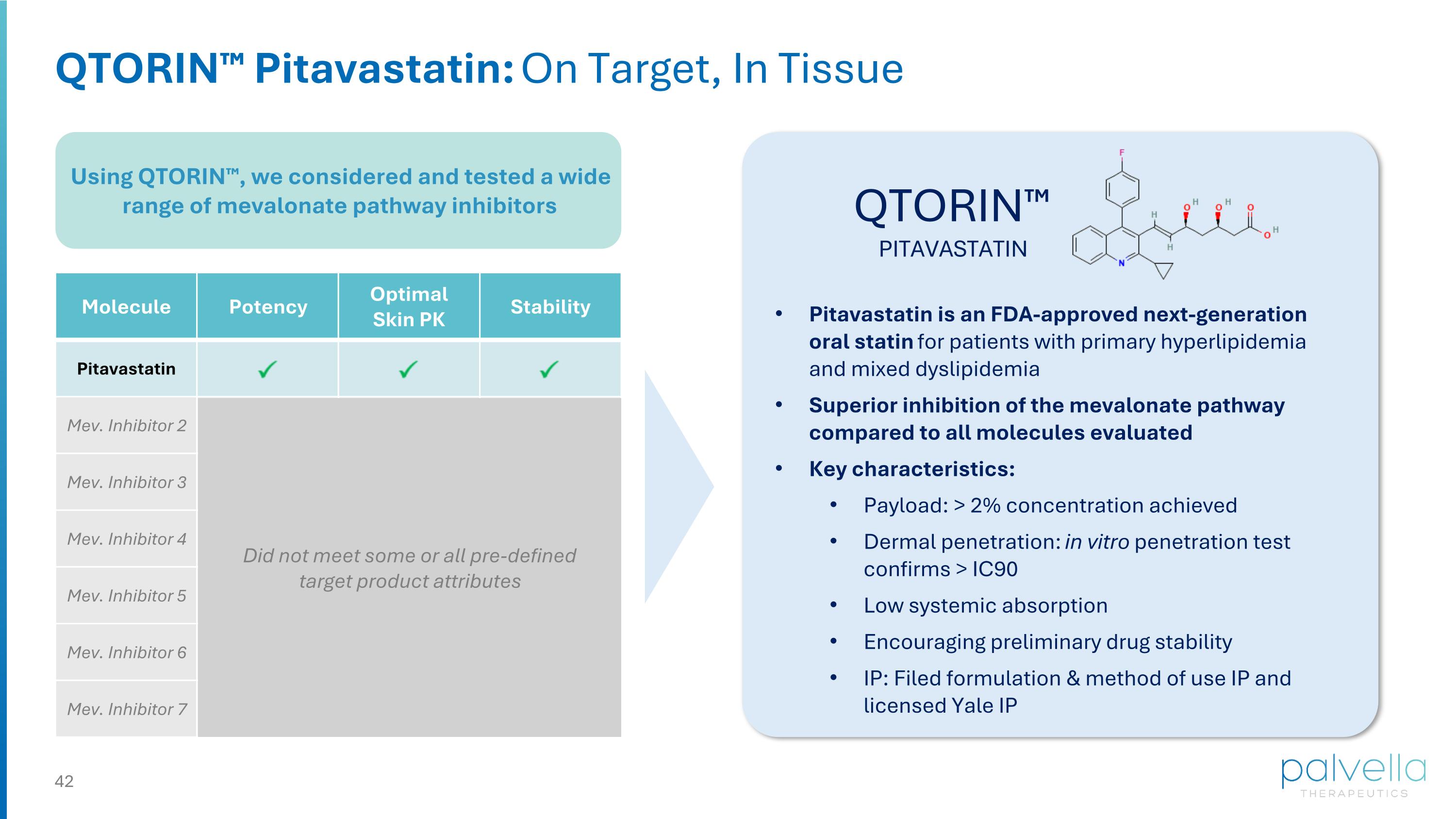
Using QTORIN™, we considered and tested a wide range of mevalonate pathway inhibitors QTORIN™ Pitavastatin: On Target, In Tissue QTORIN™ PITAVASTATIN Molecule Potency Optimal Skin PK Stability Pitavastatin Mev. Inhibitor 2 Mev. Inhibitor 3 Mev. Inhibitor 4 Mev. Inhibitor 5 Mev. Inhibitor 6 Mev. Inhibitor 7 Did not meet some or all pre-defined target product attributes Pitavastatin is an FDA-approved next-generation oral statin for patients with primary hyperlipidemia and mixed dyslipidemia Superior inhibition of the mevalonate pathway compared to all molecules evaluated Key characteristics: Payload: > 2% concentration achieved Dermal penetration: in vitro penetration test confirms > IC90 Low systemic absorption Encouraging preliminary drug stability IP: Filed formulation & method of use IP and licensed Yale IP

Platform QTORIN™

QTORIN™: New Product Development Engine VALIDATION OF QTORIN™ PLATFORM Two positive clinical study readouts (Phase 3 SELVA trial and Phase 2 TOIVA trial results) PLANNING TO PURSUE PLATFORM DESIGNATION We expect to apply for FDA’s Platform Technology Designation Program following QTORIN™ rapamycin’s targeted approval in 2027 SCALING OF PIPELINE WITH NEW QTORIN™ PROGRAMS Rapidly advancing and testing multiple molecules through QTORIN™ platform in capital- and time-efficient manner We plan to announce one new QTORIN™ program and one new QTORIN™ rapamycin indication later this year DAVID OSBORNE, PhD Chief Innovation Officer

Finance

Well-Capitalized with High-Quality Investor Participation in $230mm Oversubscribed Feb. 2026 Financing Following Positive SELVA Data Potential to Fund Through: QTORIN™ + 505(b)(2) + rare disease focus offers potential for attractive ROI NDA filing, FDA approval, and, if approved, U.S. launch for QTORIN™ rapamycin in microcystic LMs NDA filing for QTORIN™ rapamycin in cutaneous VMs Multiple Phase 2 data readouts from pipeline programs ~$274 million pro forma cash1 1. Pro forma cash reflects $58.0 million of cash and cash equivalents as of December 31, 2025, together with the $215.8 million of net proceeds from February 2026 public offering.

What Sets Palvella Apart: Building The Leader in Rare Skin Diseases and Vascular Malformations Positive Phase 3 data in microcystic lymphatic malformations, focused on NDA submission and potential U.S. commercial launch QTORIN™ rapamycin: potential to be first approved therapy and SOC in U.S. for microcystic LMs, cutaneous VMs, and angiokeratomas U.S. commercial opportunity: multi-billion dollar TAM in mLMs, with potential to expand addressable pool of patients by 10x Striving to be first for rare disease patients Deep pipeline of rare disease therapies: six diseases anticipated by year end 2026

Striving to be first for rare disease patients Thank You