

Neurocrine Biosciences to Acquire Soleno Therapeutics April 6, 2026 Exhibit 99.2

Forward-Looking Statements This communication contains forward-looking statements that involve risks and uncertainties relating to future events and the future performance of each of Soleno and Neurocrine, including statements relating to the ability to complete and the timing of completion of the transactions contemplated by the Agreement and Plan of Merger by and among Soleno, Neurocrine, and the other parties thereto (the “Merger Agreement”), including the anticipated occurrence, manner and timing of the proposed tender offer; the parties’ ability to satisfy the conditions to the consummation of the tender offer and the other conditions to the consummation of the subsequent merger set forth in the Merger Agreement; the possibility of any termination of the Merger Agreement; the prospective benefits of the proposed transaction; Neurocrine’s strategy, plans, objectives, expectations (financial or otherwise) and intentions with respect to its future financial results and growth potential, anticipated product portfolio, development programs and patent terms; the estimated occurrence of PWS; the estimated U.S. population of PWS patients; and other statements that are not historical facts. The forward-looking statements contained in this communication are based on current expectations and assumptions that are subject to risks and uncertainties which may cause actual results to differ materially from the forward-looking statements. These statements may contain words such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “future,” “intend,” “may,” “opportunity,” “plan,” “potential,” “project,” “seek,” “should,” “strategy,” “will,” “would” or other similar words and expressions indicating future results. Risks that may cause these forward-looking statements to be inaccurate include, without limitation: uncertainties as to the timing of the tender offer; uncertainties as to how many of Soleno’s stockholders will tender their stock in the offer; the possibility that competing offers or acquisition proposals will be made; the possibility that various closing conditions in the Merger Agreement may not be satisfied or waived; the difficulty of predicting the timing or outcome of regulatory approvals or actions, if any; the occurrence of any event, change or other circumstance that could give rise to the termination of the Merger Agreement; the possibility that the transaction does not close; risks related to the parties’ ability to realize the anticipated benefits of the proposed transaction, including the possibility that the expected benefits from the proposed acquisition will not be realized or will not be realized within the expected time period and that Neurocrine will not be able to integrate Soleno successfully or that such integration may be more difficult, time-consuming or costly than expected; disruption from the proposed transaction, making it more difficult for either company to conduct business as usual or maintain relationships with employees, customers, suppliers, other business partners or governmental entities; negative effects of this announcement or the consummation of the proposed transaction on the market price of Neurocrine’s common stock and/or Neurocrine’s operating results, including the possibility that if the parties do not achieve the perceived benefits of the proposed transaction as rapidly or to the extent anticipated by financial analysts or investors, the market price of Neurocrine’s common stock could decline; significant transaction costs; unknown or inestimable liabilities; the risk of litigation and/or regulatory actions related to the proposed transaction; Neurocrine’s ability to fund the proposed transaction; the time-consuming and uncertain regulatory approval process; the degree and pace of market uptake of Soleno’s commercial product, VYKATTM XR (diazoxide choline); the costly and time-consuming pharmaceutical product development process and the uncertainty of clinical success, including risks related to failure or delays in successfully initiating or completing clinical trials; global economic, financial, and healthcare system disruptions and the current and potential future negative impacts to the parties’ business operations and financial results; the sufficiency of Neurocrine’s cash flows and capital resources; Neurocrine’s ability to achieve targeted or expected future financial performance and results and the uncertainty of future tax, accounting and other provisions and estimates; and other risks and uncertainties affecting Neurocrine and Soleno, including those described from time to time under the caption “Risk Factors” and elsewhere in Neurocrine’s and Soleno’s respective filings and reports with the U.S. Securities and Exchange Commission (“SEC”), including their respective Annual Reports on Form 10-K for the fiscal year ended December 31, 2025 and subsequent Quarterly Reports on Form 10-Q and other filings filed with the SEC, as well as the Tender Offer Statement on Schedule TO and related tender offer documents to be filed by Neurocrine and its acquisition subsidiary, and the Solicitation/Recommendation Statement on Schedule 14D-9 to be filed by Soleno. Any forward-looking statements are made based on the current beliefs and judgments of Neurocrine’s and Soleno’s respective management teams, and the reader is cautioned not to rely on any forward-looking statements made by Neurocrine or Soleno. Except as required by law, Neurocrine and Soleno do not undertake any obligation to update (publicly or otherwise) any forward-looking statement, including without limitation any financial projection or guidance, whether as a result of new information, future events, or otherwise. This presentation shall not constitute an offer to sell or the solicitation of an offer to purchase any securities, nor shall there be any sale of securities in any states or jurisdictions in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction or an exemption therefrom. This presentation also contains estimates and other statistical data made by independent parties and by us relating to market size and growth and other data about our industry. These data involve a number of assumptions and limitations, and we make no representation as to the accuracy of such estimates. Projections, assumptions and estimates of the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. The trademarks included herein are the property of the owners thereof and are used for reference purposes only.

Additional Information About the Acquisition and Where to Find It The tender offer for all of the outstanding shares of Soleno described in this communication has not yet commenced. This communication is for informational purposes only, is not a recommendation and is neither an offer to purchase nor a solicitation of an offer to sell any securities, nor is it a substitute for the tender offer materials that Neurocrine and its acquisition subsidiary will file with the SEC upon commencement of the tender offer. A solicitation and offer to purchase outstanding shares of Soleno will only be made pursuant to an offer to purchase and related tender offer materials that Neurocrine and its acquisition subsidiary intend to file with the SEC. At the time that the tender offer is commenced, Neurocrine and its acquisition subsidiary will file a tender offer statement on Schedule TO, and Soleno will file a Solicitation/Recommendation Statement on Schedule 14D-9 with the SEC with respect to the tender offer. THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND CERTAIN OTHER TENDER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED ACQUISITION AND THE PARTIES THERETO. INVESTORS AND STOCKHOLDERS OF SOLENO ARE URGED TO READ THESE DOCUMENTS CAREFULLY WHEN THEY BECOME AVAILABLE (AND EACH AS IT MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME) BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION THAT INVESTORS AND STOCKHOLDERS OF SOLENO SHOULD CONSIDER BEFORE MAKING ANY DECISION REGARDING TENDERING THEIR SHARES OF COMMON STOCK IN THE TENDER OFFER. The tender offer materials (including the Offer to Purchase and the related Letter of Transmittal) will be made available at no expense on Neurocrine’s website at neurocrine.com/investors and (once they become available) will be mailed to the stockholders of Soleno free of charge. The Solicitation/Recommendation Statement and other documents filed with the SEC by Soleno will be available at no expense at Soleno’s website at investors.soleno.life. The information contained in, or that can be accessed through, Neurocrine’s and Soleno’s respective websites are not a part of, or incorporated by reference herein. The tender offer materials (including the Offer to Purchase and the related Letter of Transmittal), as well as the Solicitation/Recommendation Statement, will also be made available for free on the SEC’s website at www.sec.gov. Copies of those offer documents and all other documents filed by Neurocrine and Soleno will be made available at no charge by directing a request to the information agent for the tender offer, which will be named in the Schedule TO. In addition to the Offer to Purchase, the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, Neurocrine and Soleno each file annual, quarterly, and current reports, proxy statements and other information with the SEC. You may read any reports, statements or other information filed by Neurocrine or Soleno with the SEC for free on the SEC’s website at www.sec.gov.

Shared Focus on Rare Disease and Endocrinology Accelerates Growth and Commercial Diversification Positions Neurocrine For Sustained Value Creation Reinforces Neurocrine’s commitment to delivering medicines that are transformative for patients and families Provides opportunity for Neurocrine’s medical and commercial capabilities to help improve care for individuals with Prader-Willi syndrome (PWS) Adds VYKATTM XR, the first and only approved medicine for hyperphagia in PWS patients Immediately accelerates Neurocrine’s revenue growth and portfolio diversification strategy Expands high-growth portfolio to three first-in-class medicines, including INGREZZA® and CRENESSITY® Increases Neurocrine’s scale and growth profile to support sustained innovation and development Strong VYKAT XR IP expected to extend into the mid-2040s, providing a durable platform for long-term value creation Aligns well with Neurocrine’s emerging endocrinology portfolio Acquisition of Soleno: Strategic and Financial Fit Positions Neurocrine to Deliver Sustained Growth and Value

Purchase Price Neurocrine has entered into a definitive agreement to acquire Soleno Therapeutics in an all-cash transaction Purchase price of $53.00 per share represents a 34% premium to Soleno’s closing price on April 2nd, 2026 and 51% premium to the 30-day volume-weighted average price Funding Represents a $2.9B equity purchase price Transaction will be funded with cash on hand and Neurocrine plans to optimize its capital structure by taking on a modest amount of pre-payable debt. The transaction is not subject to any financing condition. Approvals & Timing Transaction was unanimously approved by the Neurocrine Board of Directors and unanimously approved by the Soleno Board of Directors among those directors present at the meeting Neurocrine will commence a cash tender offer to acquire all outstanding shares of Soleno Expected to close within 90 days of announcement, subject to satisfaction of customary closing conditions, including receipt of regulatory approvals Financial Overview: Attractive Transaction Terms

Commercial-stage rare disease biotechnology company Lead commercial asset, VYKAT XR granted Breakthrough Therapy Designation by the FDA in April 2024 VYKAT XR approved by the FDA in March 2025 as first and only drug to treat hyperphagia in patients with PWS 4 years and older VYKAT XR generated $190M in 2025 revenue for Soleno including $92M in Q4 alone Developing and Commercializing Novel Therapeutics for the Treatment of Rare Diseases Company Overview Soleno Therapeutics: Snapshot
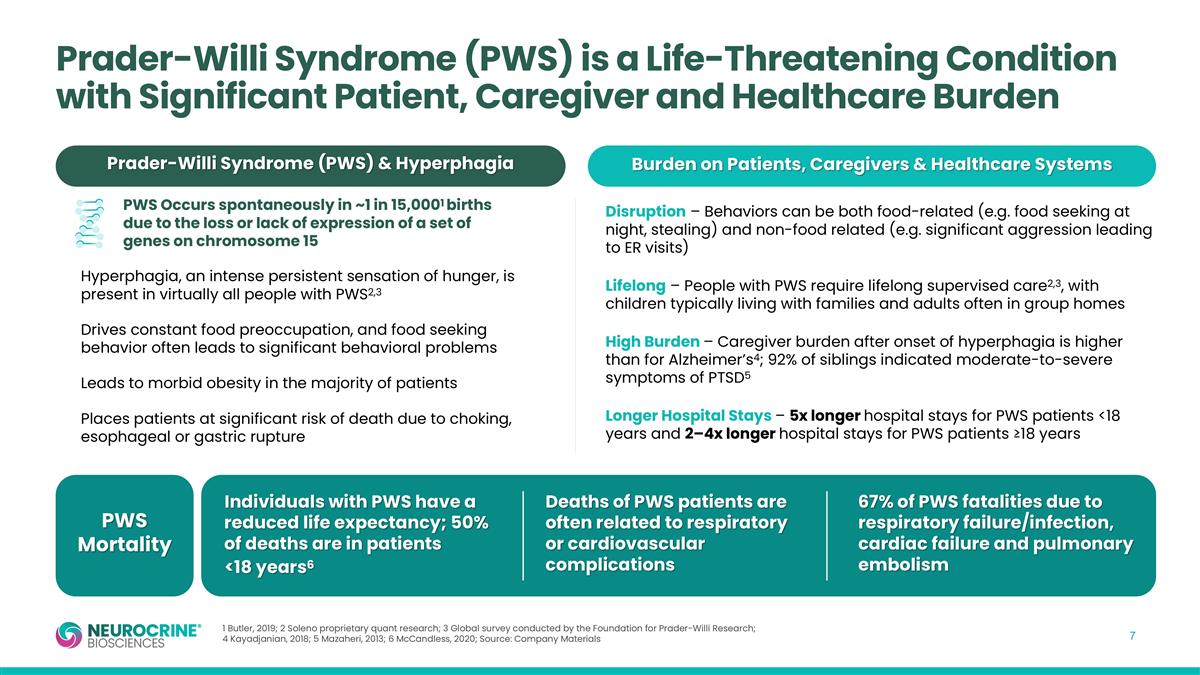
PWS Mortality Burden on Patients, Caregivers & Healthcare Systems Prader-Willi Syndrome (PWS) & Hyperphagia Disruption – Behaviors can be both food-related (e.g. food seeking at night, stealing) and non-food related (e.g. significant aggression leading to ER visits) Lifelong – People with PWS require lifelong supervised care2,3, with children typically living with families and adults often in group homes High Burden – Caregiver burden after onset of hyperphagia is higher than for Alzheimer’s4; 92% of siblings indicated moderate-to-severe symptoms of PTSD5 Longer Hospital Stays – 5x longer hospital stays for PWS patients <18 years and 2–4x longer hospital stays for PWS patients ≥18 years Hyperphagia, an intense persistent sensation of hunger, is present in virtually all people with PWS2,3 Drives constant food preoccupation, and food seeking behavior often leads to significant behavioral problems Leads to morbid obesity in the majority of patients Places patients at significant risk of death due to choking, esophageal or gastric rupture Individuals with PWS have a reduced life expectancy; 50% of deaths are in patients <18 years6 Deaths of PWS patients are often related to respiratory or cardiovascular complications 67% of PWS fatalities due to respiratory failure/infection, cardiac failure and pulmonary embolism PWS Occurs spontaneously in ~1 in 15,0001 births due to the loss or lack of expression of a set of genes on chromosome 15 1 Butler, 2019; 2 Soleno proprietary quant research; 3 Global survey conducted by the Foundation for Prader-Willi Research; 4 Kayadjanian, 2018; 5 Mazaheri, 2013; 6 McCandless, 2020; Source: Company Materials Prader-Willi Syndrome (PWS) is a Life-Threatening Condition with Significant Patient, Caregiver and Healthcare Burden

Indicated for the treatment of hyperphagia in adults and pediatric patients 4 years of age and older with Prader-Willi syndrome (PWS) APPROVED BY THE FDA IN MARCH 2025 FIRST-IN-CLASS VYKAT XR is a first-in-class treatment for hyperphagia in patients with PWS 4 years and older ROBUST DATA Clinical program demonstrated ability to significantly reduce hyperphagia and impact other PWS-related symptoms and co-morbidities STANDARD OF CARE VYKAT XR can become the standard of care for patients with PWS SAFETY Clinical trial safety profile supported by multiple years of experience VYKAT XR – Changing What It Means to Live with PWS
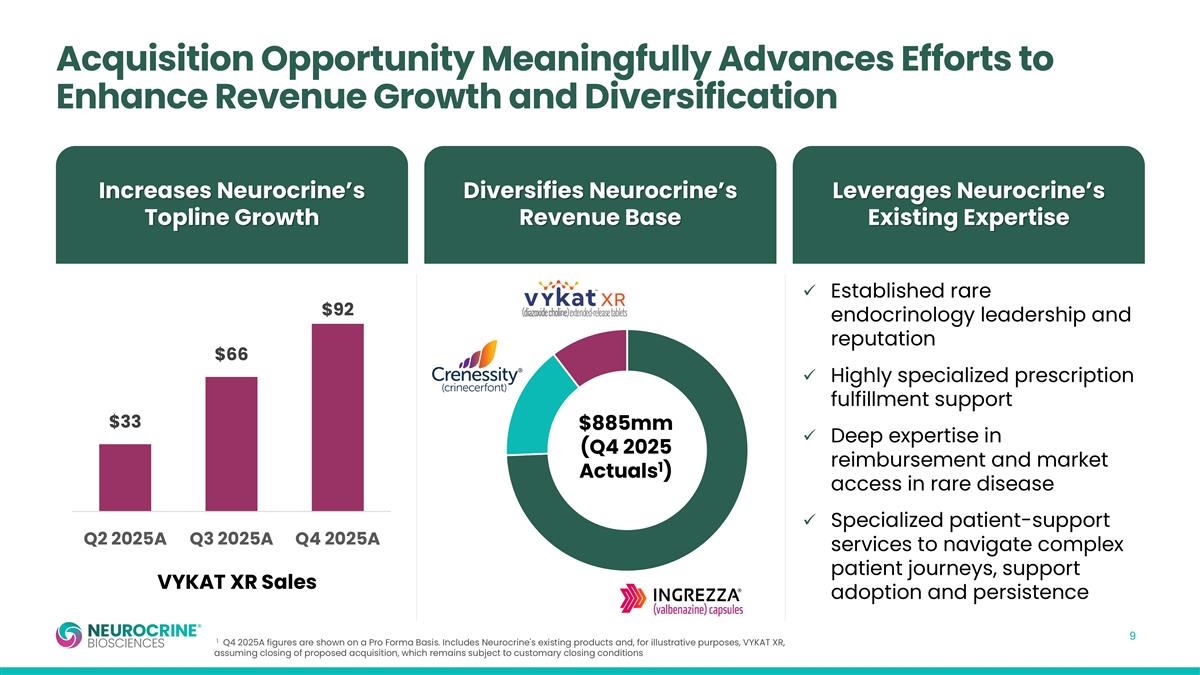
Acquisition Opportunity Meaningfully Advances Efforts to Enhance Revenue Growth and Diversification Established rare endocrinology leadership and reputation Highly specialized prescription fulfillment support Deep expertise in reimbursement and market access in rare disease Specialized patient-support services to navigate complex patient journeys, support adoption and persistence Increases Neurocrine’s Topline Growth Diversifies Neurocrine’s Revenue Base Leverages Neurocrine’s Existing Expertise $885mm (Q4 2025 Actuals1) VYKAT XR Sales 1 Q4 2025A figures are shown on a Pro Forma Basis. Includes Neurocrine's existing products and, for illustrative purposes, VYKAT XR, assuming closing of proposed acquisition, which remains subject to customary closing conditions
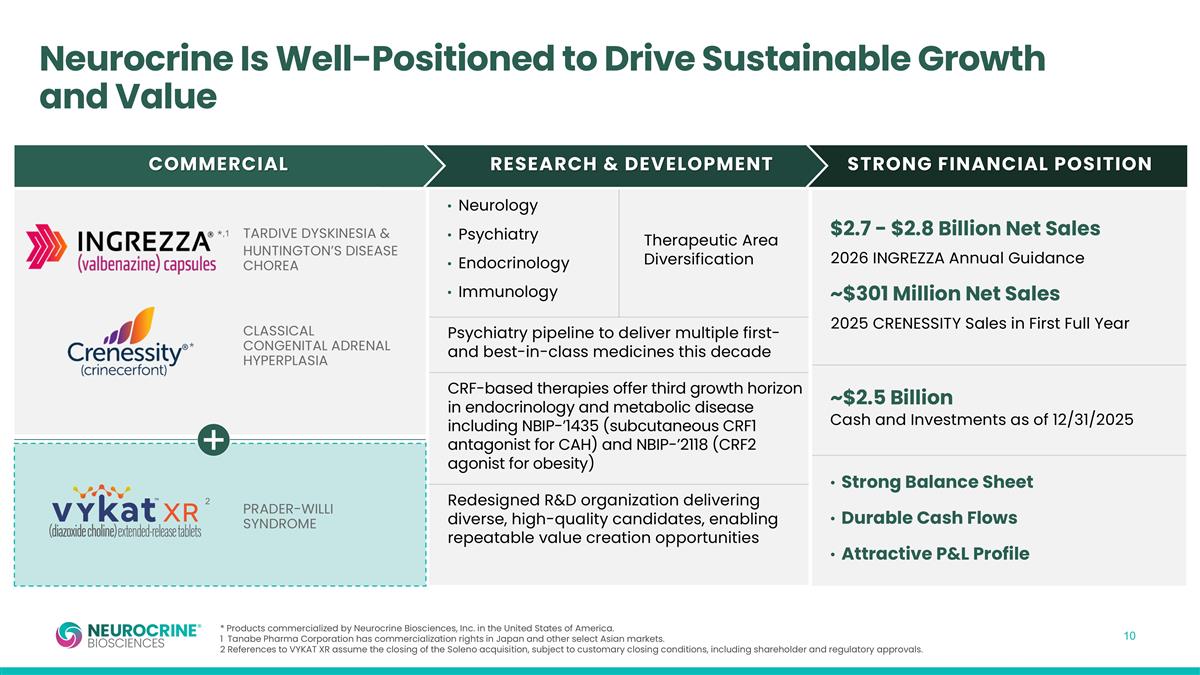
Neurocrine Is Well-Positioned to Drive Sustainable Growth and Value * Products commercialized by Neurocrine Biosciences, Inc. in the United States of America. 1 Tanabe Pharma Corporation has commercialization rights in Japan and other select Asian markets. 2 References to VYKAT XR assume the closing of the Soleno acquisition, subject to customary closing conditions, including shareholder and regulatory approvals. COMMERCIAL RESEARCH & DEVELOPMENT STRONG FINANCIAL POSITION Neurology Psychiatry Endocrinology Immunology Psychiatry pipeline to deliver multiple first- and best-in-class medicines this decade CRF-based therapies offer third growth horizon in endocrinology and metabolic disease including NBIP-’1435 (subcutaneous CRF1 antagonist for CAH) and NBIP-’2118 (CRF2 agonist for obesity) Redesigned R&D organization delivering diverse, high-quality candidates, enabling repeatable value creation opportunities $2.7 - $2.8 Billion Net Sales 2026 INGREZZA Annual Guidance ~$301 Million Net Sales 2025 CRENESSITY Sales in First Full Year ~$2.5 Billion Cash and Investments as of 12/31/2025 Strong Balance Sheet Durable Cash Flows Attractive P&L Profile Therapeutic Area Diversification TARDIVE DYSKINESIA & HUNTINGTON’S DISEASE CHOREA *,1 * PRADER-WILLI SYNDROME 2 CLASSICAL CONGENITAL ADRENAL HYPERPLASIA

1 Assumes the closing of the proposed acquisition of Soleno, subject to customary closing conditions Adds VYKAT XR, the First and Only Approved Medicine for the Treatment of Hyperphagia in PWS Expands and Strengthens Neurocrine’s Leadership in Rare Disease and Endocrinology Broadens Neurocrine’s Portfolio With Three First-in-Class Medicines, Including INGREZZA and CRENESSITY Strong IP Estate Expected to Extend into the Mid-2040s, Providing a Durable Platform for Long-Term Value Creation Increases Neurocrine’s Scale and Growth Profile to Support Sustained Innovation and Development Immediately Accelerates Neurocrine’s Revenue Growth Profile and Portfolio Diversification Strategy Acquisition of Soleno Will Strengthen Leadership in Endocrinology and Rare Disease1

Kyle Gano, Ph.D. Chief Executive Officer Matt Abernethy Chief Financial Officer Eric Benevich Chief Commercial Officer Sanjay Keswani, M.D. Chief Medical Officer Samir Siddhanti Vice President, Business Development & Strategy Q&A