

Korsana Biosciences Overview March 2026 Exhibit 99.2

Disclaimers The information contained in this presentation has been prepared by Korsana Biosciences, Inc. and its affiliates (“Korsana” or the “Company”) and contains information pertaining to the business and operations of the Company. The information contained in this presentation: (a) is provided as at the date hereof, is subject to change without notice, and is based on publicly available information, information obtained from third party sources, and internally developed data; (b) does not purport to contain all the information that may be necessary or desirable to fully and accurately evaluate an investment in the Company; (c) is not to be considered as a recommendation by the Company that any person make an investment in the Company; and (d) is for information purposes only. Where any opinion or belief is expressed in this presentation, it is based on certain assumptions and limitations and is an expression of present opinion or belief only. This presentation should not be construed as legal, financial, or tax advice to any individual, as each individual’s circumstances are different. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of the Company’s securities in any state or other jurisdiction in which such offer, solicitation, or sale would be unlawful prior to the registration or qualification under the securities laws of any such state or other jurisdiction. Although the Company believes the third-party sources and publicly available information in this presentation to be reliable, it has not independently verified and makes no representation or warranty, express or implied, as to the accuracy, completeness or timeliness of any third-party data. Accordingly, no representation is made as to, and no reliance should be placed on, any third-party data contained in this presentation. Forward-Looking Statements and Other Information Certain information set forth in this presentation contains “forward-looking statements” within the meaning of applicable United States securities legislation. Except for statements of historical fact, certain information contained herein constitutes forward-looking statements which include but are not limited to statements regarding: our business strategy, including our ability to develop potentially best-in-class therapies initially focused on neurodegenerative disorders like Alzheimer’s disease, including a potentially best-in class shuttled anti-Aβ therapy offering meaningful improvements over trontinemab; the efficacy, safety profile, dosing regime, convenience, half-life, and tolerability of KRSA-028; Korsana’s ongoing and future clinical development activities, including the expected timing of CTN and IND filings, healthy volunteer PK and CSF data, and interim clinical proof of concept data for KRSA-028; the expected timing of unveiling additional THETA™ enabled programs; estimated market sizes, potential growth opportunities, potential value creation and sample transactions; the length of time that the Company believes its existing cash resources will fund its operations, including expectations of cash runway extending into 2028; and management’s assessment of future plans and operations which are based on current internal expectations, estimates, projections, assumptions and beliefs, which may prove to be incorrect. Forward-looking statements can often be identified by the use of words such as “may”, “will”, “could”, “would”, “anticipate”, “believe”, “expect”, “intend”, “potential”, “estimate”, “scheduled”, “plans”, “planned”, “forecasts”, “goals” and similar expressions or the negatives thereof. Forward-looking statements are neither historical facts nor assurances of future performance. Forward-looking statements are based on a number of factors and assumptions made by management and considered reasonable at the time such information is provided, and forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause the actual results, performance or achievements to be materially different from those expressed or implied by the forward-looking statements, including: uncertainties and risks arising from regulatory feedback, including potential disagreement by regulatory authorities with our clinical trial design, interpretation of data and our ongoing or planned clinical trials for our product candidates; risks related to our patent portfolio and strategy, including our ability to obtain and protect patent rights or such rights being subject to challenges from third parties; the expected or potential impact of macroeconomic conditions; the implementation of changes in law, tariffs, sanctions, export or import controls, and other government measures that could impact our business operations; the impacts of adverse events or disappointing results in clinical trials of third parties, including our competitors developing product candidates that target similar mechanisms of action and/or indications as our product candidates; and discussions of potential risks, uncertainties, and other filings by the Company from time to time, as well as risk factors associated with companies that operate in the biopharma industry, including those associated with the uncertainties of drug development. All of the forward-looking statements made in this presentation are qualified by these cautionary statements and other cautionary statements or other factors contained herein. Although management believes that the expectations conveyed by forward-looking statements herein are reasonable based on information available on the date such forward-looking statements are made, there can be no assurance that forward-looking statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. The Company undertakes no obligation to update forward-looking statements if circumstances or management’s estimates or opinions should change except as required by applicable securities laws. The forward-looking statements contained herein are presented for the purpose of assisting readers in understanding the Company’s plan, objectives, and goals and may not be appropriate for other purposes. The reader is cautioned not to place undue reliance on forward-looking statements.

Transaction Summary Transaction: Transaction between Cyclerion Therapeutics, Inc. (CYCN), including its wholly owned subsidiaries, and Korsana Biosciences, Inc. (Korsana) Transaction Structure: CYCN to acquire 100% of Korsana equity interests in reverse-triangular merger with Korsana surviving the merger as a wholly-owned subsidiary of CYCN Rebrand: Post-closing, CYCN will be renamed Korsana Biosciences, Inc. Interim Operating Covenants: Customary interim covenants that limit both Korsana and CYCN to ordinary-course operations between signing and closing, subject to certain exceptions Survival: No survival of reps and warranties post-Closing Director / Officer Indemnification: Korsana (post-Closing) will remain obligated to indemnify CYCN D&Os post-Closing pursuant to pre-Closing rights. CYCN (pre-closing) required to procure six-year D&O insurance tail policy Outside Date: To be determined Timing: Closing expected to occur during mid-2026 (announcement expected week of March 30, 2026) CVR: At Closing, CYCN stockholders will receive CVRs (Contingent Value Rights) for certain cash proceeds received by Korsana from the sale of CYCN’s legacy assets Post-Closing Ownership: Korsana holders expected to own ~96.64% of combined enterprise (f.d.) and CYCN holders expected to own ~3.36%, assuming CYCN Net Cash at closing of $0.0m and prior to adjusting for Financing of ~$300m+ and other customary items Certain Closing Conditions: S-4/Proxy Statement: S-4/Proxy Statement review by SEC completed and mailing to Holders completed (see SEC Filings below) Reps Bringdown: Materiality scrape on MAE- and materiality-qualified reps, brought down to MAE standard; capitalization rep brought down flat, subject to de minimis exceptions; fundamental representations brought down in all material respects Interim covenants: Perform or comply in all material respects; no MAE Korsana Stockholder Approval: Requisite stockholder approval, including holders of a majority of the capital stock outstanding on an as-converted basis CYCN Stockholder Approval: Holders representing a majority of the votes cast where a quorum is present Financing: Financing proceeds of at least ~$150 million shall have been received by Korsana Nasdaq Application: Nasdaq application covering merger and Financing shares shall be approved HSR: Expiration of the applicable waiting period (if applicable) SEC Filings: Parties expected to mail S-4/Proxy Statement in June/July 2026 in respect of required CYCN vote Merger and Financing shares not registered on the S-4 to be registered on a resale S-1 registration statement following Closing Directors & Executive Officers to file Forms 3 & 4 following the Closing Support Agreements: Directors / officers and certain affiliated investors to sign support agreements, agreeing to vote in favor of and otherwise support the transaction Lock-Up Agreements: Continuing directors / officers and certain affiliated investors to sign 180-day lock-up agreements prohibiting (subject to certain exceptions) post-closing transactions in Korsana’s securities during the lock-up period Concurrent Financing: ~$150 million of proceeds from private financing (Financing) Registration Rights Agreement: Company agrees to register any merger shares and Financing shares not registered on the S-4 on a resale S-1 registration statement following the Closing Certain Closing Conditions (Subscription Agreement): Reverse Merger: Closing conditions under the merger agreement must have been met Reps: Subject to certain exceptions, representations and warranties generally brought down to MAE standard Interim covenants: Use commercially reasonable efforts to comply Closing: Expected to occur immediately prior to the closing of the reverse merger Merger Overview Post-Closing Ownership; Closing Financing Overview Other Workstreams
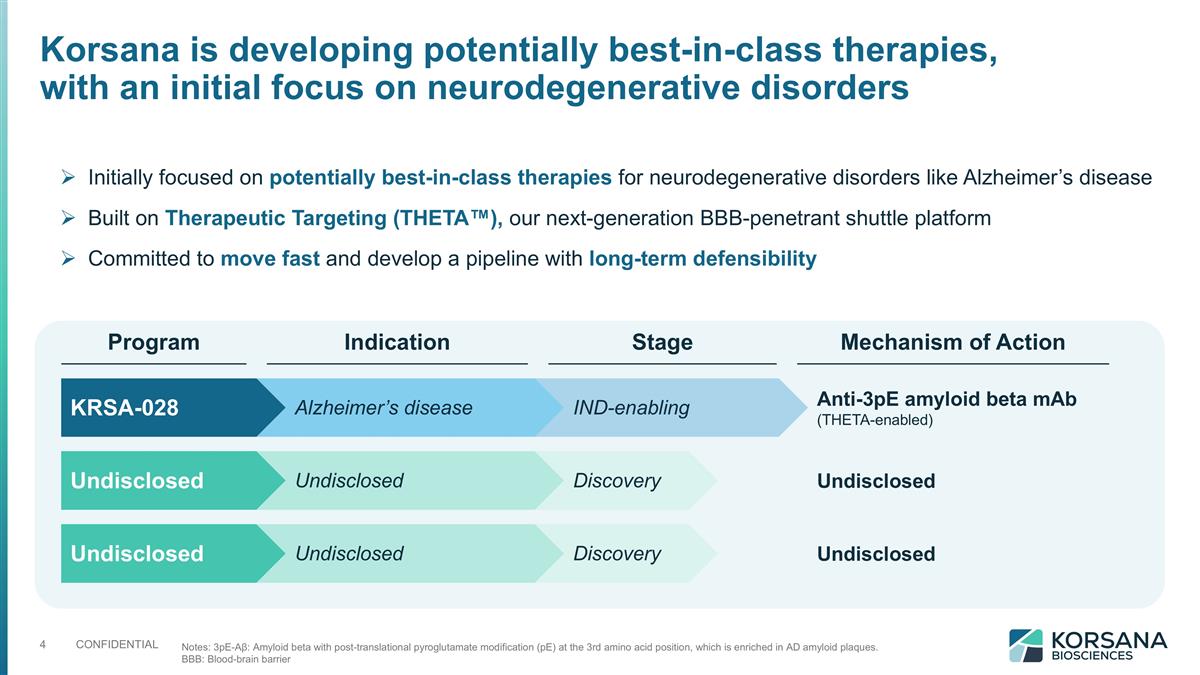
Korsana is developing potentially best-in-class therapies, with an initial focus on neurodegenerative disorders Initially focused on potentially best-in-class therapies for neurodegenerative disorders like Alzheimer’s disease Built on Therapeutic Targeting (THETA™), our next-generation BBB-penetrant shuttle platform Committed to move fast and develop a pipeline with long-term defensibility Notes: 3pE-Aβ: Amyloid beta with post-translational pyroglutamate modification (pE) at the 3rd amino acid position, which is enriched in AD amyloid plaques. BBB: Blood-brain barrier Program Indication Stage Mechanism of Action KRSA-028 Anti-3pE amyloid beta mAb (THETA-enabled) Alzheimer’s disease IND-enabling Undisclosed Undisclosed Undisclosed Discovery Undisclosed Undisclosed Undisclosed Discovery
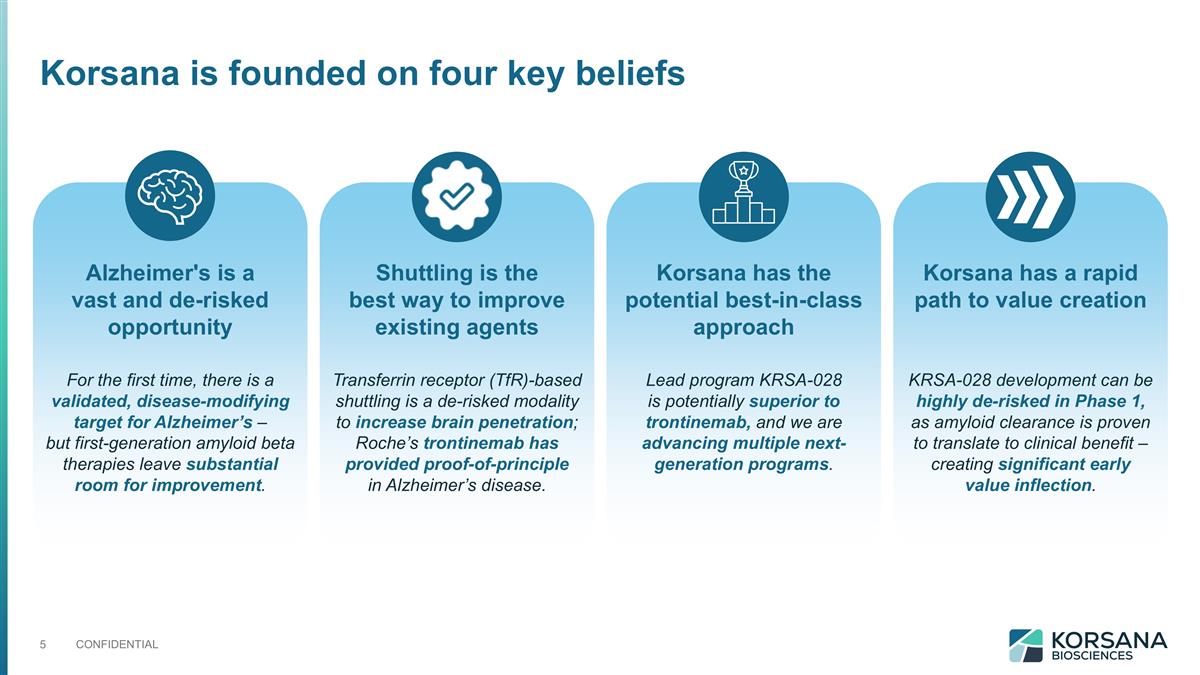
Korsana has a rapid path to value creation Korsana is founded on four key beliefs Alzheimer's is a vast and de-risked opportunity Shuttling is the best way to improve existing agents Korsana has the potential best-in-class approach Transferrin receptor (TfR)-based shuttling is a de-risked modality to increase brain penetration; Roche’s trontinemab has provided proof-of-principle in Alzheimer’s disease. For the first time, there is a validated, disease-modifying target for Alzheimer’s – but first-generation amyloid beta therapies leave substantial room for improvement. Lead program KRSA-028 is potentially superior to trontinemab, and we are advancing multiple next-generation programs. KRSA-028 development can be highly de-risked in Phase 1, as amyloid clearance is proven to translate to clinical benefit – creating significant early value inflection.

Alzheimer’s is a vast and de-risked opportunity
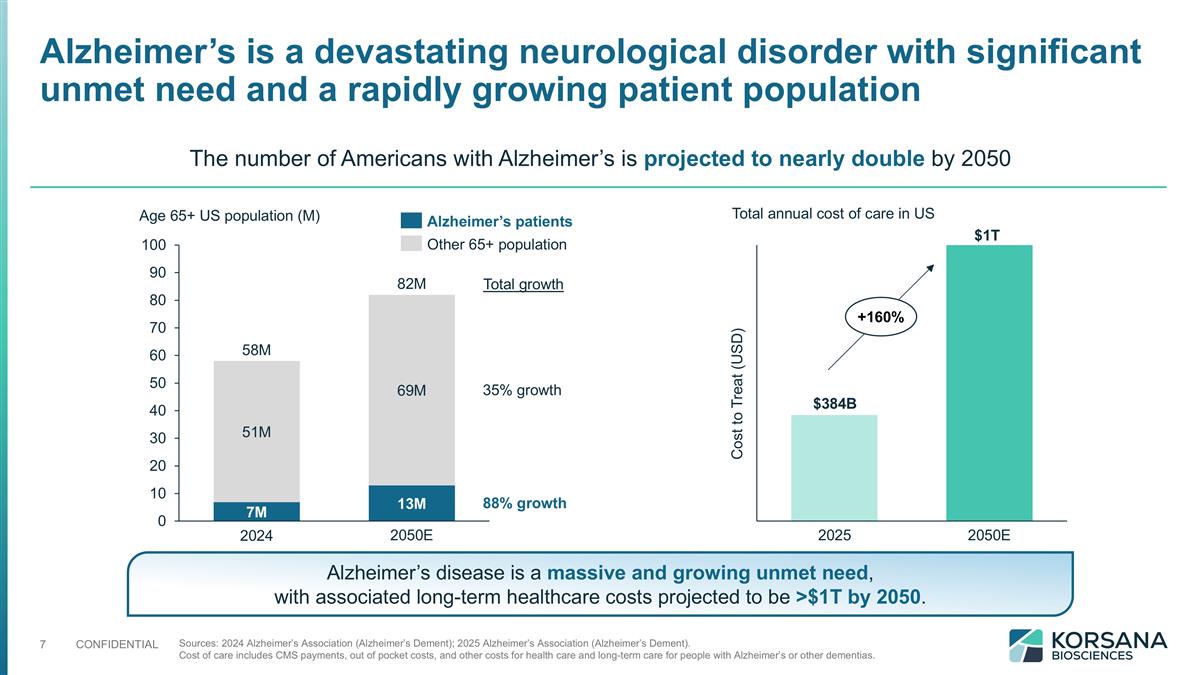
Alzheimer’s is a devastating neurological disorder with significant unmet need and a rapidly growing patient population Age 65+ US population (M) M M M M M M Total growth 35% growth 88% growth The number of Americans with Alzheimer’s is projected to nearly double by 2050 Sources: 2024 Alzheimer’s Association (Alzheimer’s Dement); 2025 Alzheimer’s Association (Alzheimer’s Dement). Cost of care includes CMS payments, out of pocket costs, and other costs for health care and long-term care for people with Alzheimer’s or other dementias. $384B $1T 2025 Total annual cost of care in US +160% Alzheimer’s disease is a massive and growing unmet need, with associated long-term healthcare costs projected to be >$1T by 2050.
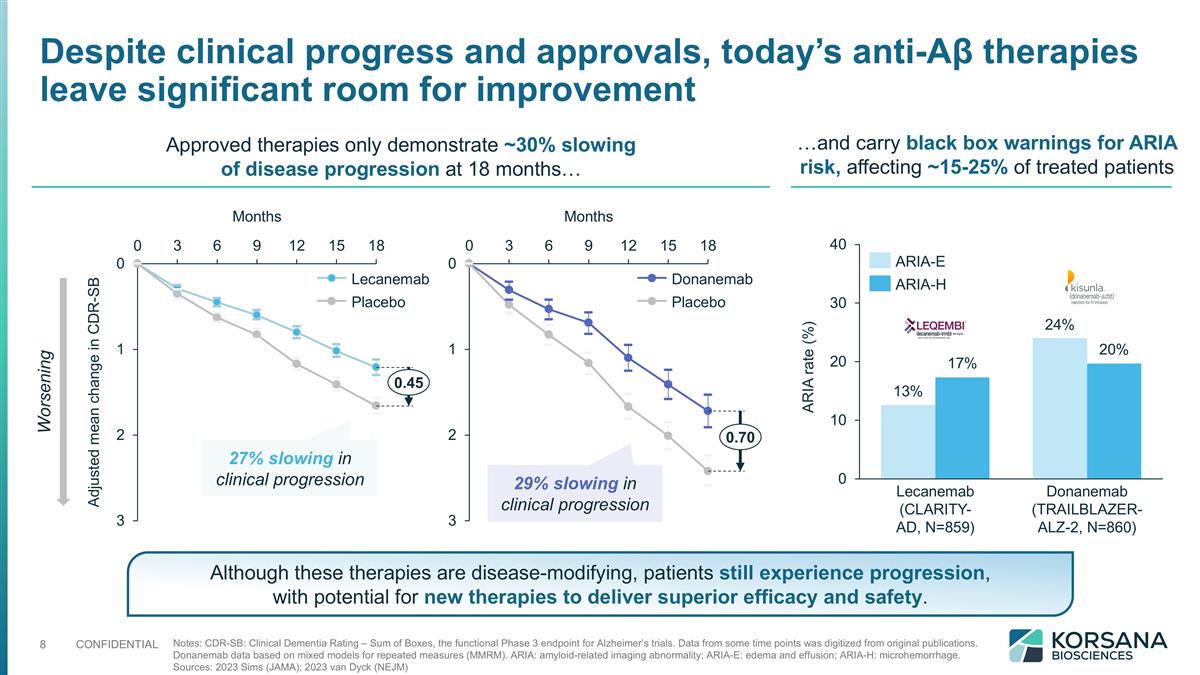
Notes: CDR-SB: Clinical Dementia Rating – Sum of Boxes, the functional Phase 3 endpoint for Alzheimer’s trials. Data from some time points was digitized from original publications. Donanemab data based on mixed models for repeated measures (MMRM). ARIA: amyloid-related imaging abnormality; ARIA-E: edema and effusion; ARIA-H: microhemorrhage. Sources: 2023 Sims (JAMA); 2023 van Dyck (NEJM) Approved therapies only demonstrate ~30% slowing of disease progression at 18 months… Adjusted mean change in CDR-SB Months 27% slowing in clinical progression Months 29% slowing in clinical progression …and carry black box warnings for ARIA risk, affecting ~15-25% of treated patients Worsening ARIA rate (%) Despite clinical progress and approvals, today’s anti-Aβ therapies leave significant room for improvement Although these therapies are disease-modifying, patients still experience progression, with potential for new therapies to deliver superior efficacy and safety.
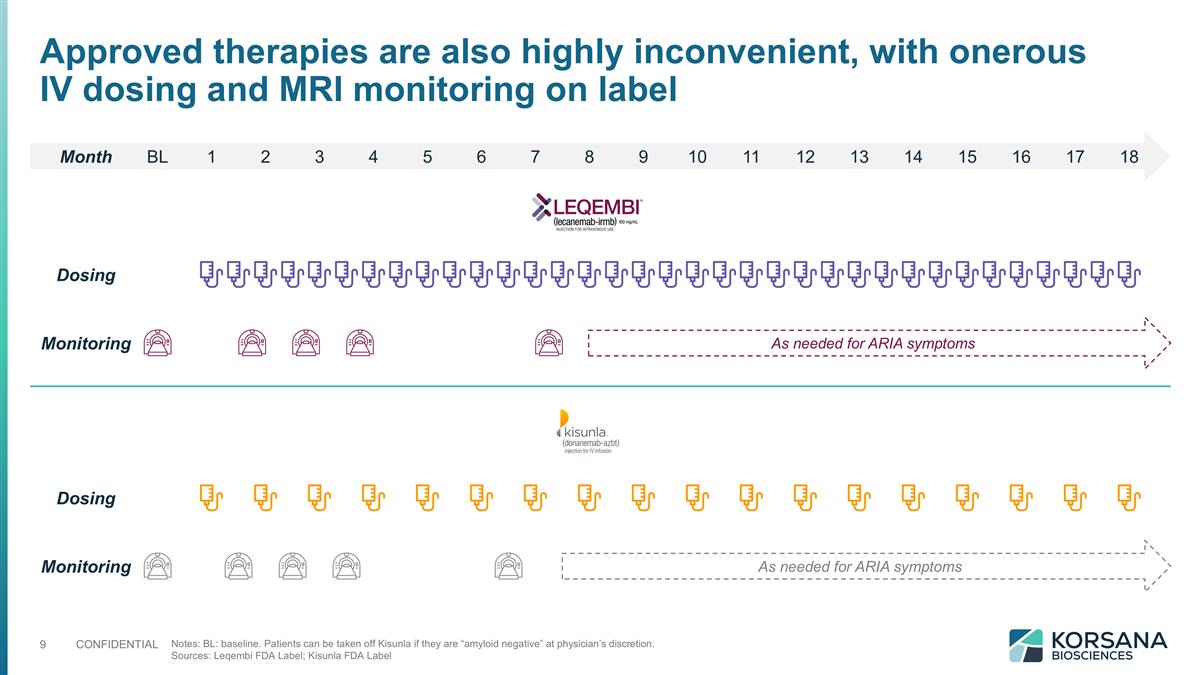
Notes: BL: baseline. Patients can be taken off Kisunla if they are “amyloid negative” at physician’s discretion. Sources: Leqembi FDA Label; Kisunla FDA Label Month BL 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Dosing Dosing Monitoring As needed for ARIA symptoms Monitoring As needed for ARIA symptoms Approved therapies are also highly inconvenient, with onerous IV dosing and MRI monitoring on label
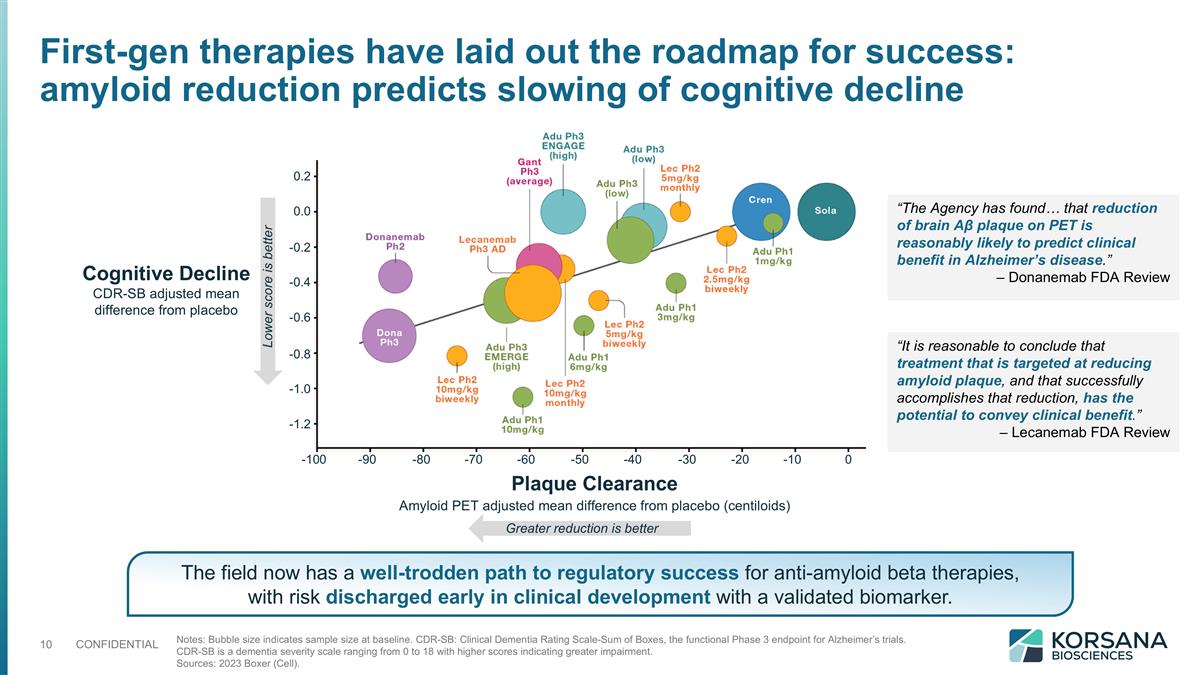
Notes: Bubble size indicates sample size at baseline. CDR-SB: Clinical Dementia Rating Scale-Sum of Boxes, the functional Phase 3 endpoint for Alzheimer’s trials. CDR-SB is a dementia severity scale ranging from 0 to 18 with higher scores indicating greater impairment. Sources: 2023 Boxer (Cell). Cognitive Decline CDR-SB adjusted mean difference from placebo “The Agency has found… that reduction of brain Aβ plaque on PET is reasonably likely to predict clinical benefit in Alzheimer’s disease.” – Donanemab FDA Review “It is reasonable to conclude that treatment that is targeted at reducing amyloid plaque, and that successfully accomplishes that reduction, has the potential to convey clinical benefit.” – Lecanemab FDA Review -100 -90 -80 -70 -60 -50 -40 -30 -20 -10 0 0.2 0.0 -0.2 -0.4 -0.6 -0.8 -1.0 -1.2 Lower score is better Greater reduction is better Plaque Clearance Amyloid PET adjusted mean difference from placebo (centiloids) First-gen therapies have laid out the roadmap for success: amyloid reduction predicts slowing of cognitive decline The field now has a well-trodden path to regulatory success for anti-amyloid beta therapies, with risk discharged early in clinical development with a validated biomarker.

Shuttling is the best way to improve existing agents
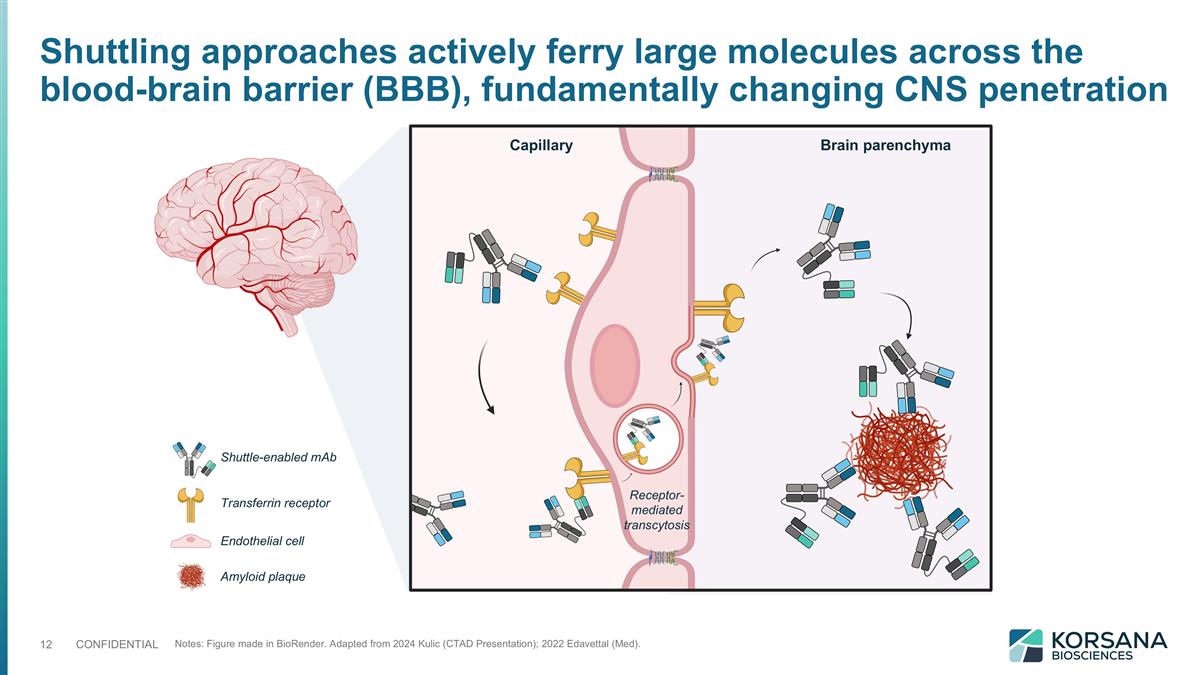
Notes: Figure made in BioRender. Adapted from 2024 Kulic (CTAD Presentation); 2022 Edavettal (Med). Shuttle-enabled mAb Transferrin receptor Endothelial cell Amyloid plaque Capillary Brain parenchyma Receptor-mediated transcytosis Shuttling approaches actively ferry large molecules across the blood-brain barrier (BBB), fundamentally changing CNS penetration
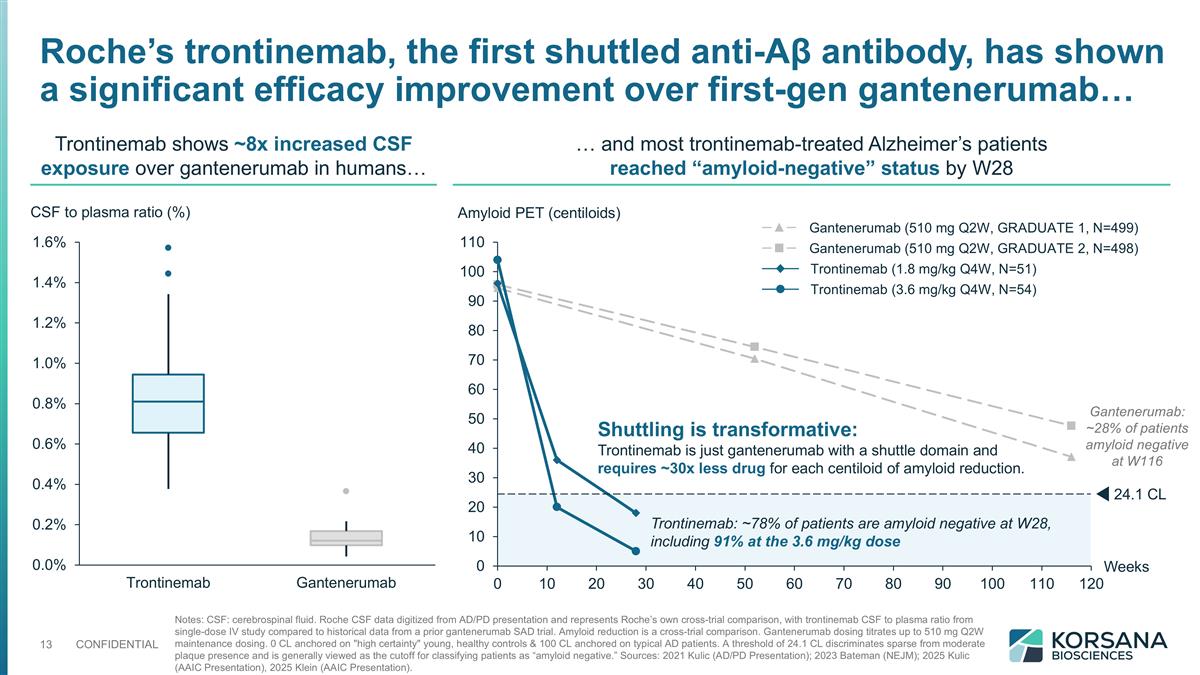
Notes: CSF: cerebrospinal fluid. Roche CSF data digitized from AD/PD presentation and represents Roche’s own cross-trial comparison, with trontinemab CSF to plasma ratio from single-dose IV study compared to historical data from a prior gantenerumab SAD trial. Amyloid reduction is a cross-trial comparison. Gantenerumab dosing titrates up to 510 mg Q2W maintenance dosing. 0 CL anchored on "high certainty" young, healthy controls & 100 CL anchored on typical AD patients. A threshold of 24.1 CL discriminates sparse from moderate plaque presence and is generally viewed as the cutoff for classifying patients as “amyloid negative.” Sources: 2021 Kulic (AD/PD Presentation); 2023 Bateman (NEJM); 2025 Kulic (AAIC Presentation), 2025 Klein (AAIC Presentation). CSF to plasma ratio (%) Trontinemab shows ~8x increased CSF exposure over gantenerumab in humans… … and most trontinemab-treated Alzheimer’s patients reached “amyloid-negative” status by W28 Roche’s trontinemab, the first shuttled anti-Aβ antibody, has shown a significant efficacy improvement over first-gen gantenerumab… Weeks Amyloid PET (centiloids) .1 CL Shuttling is transformative: Trontinemab is just gantenerumab with a shuttle domain and requires ~30x less drug for each centiloid of amyloid reduction. Gantenerumab: ~28% of patients amyloid negative at W116 Trontinemab: ~78% of patients are amyloid negative at W28, including 91% at the 3.6 mg/kg dose Trontinemab (1.8 mg/kg Q4W, N=51) Trontinemab (3.6 mg/kg Q4W, N=54) Gantenerumab (510 mg Q2W, GRADUATE 1, N=499) Gantenerumab (510 mg Q2W, GRADUATE 2, N=498)
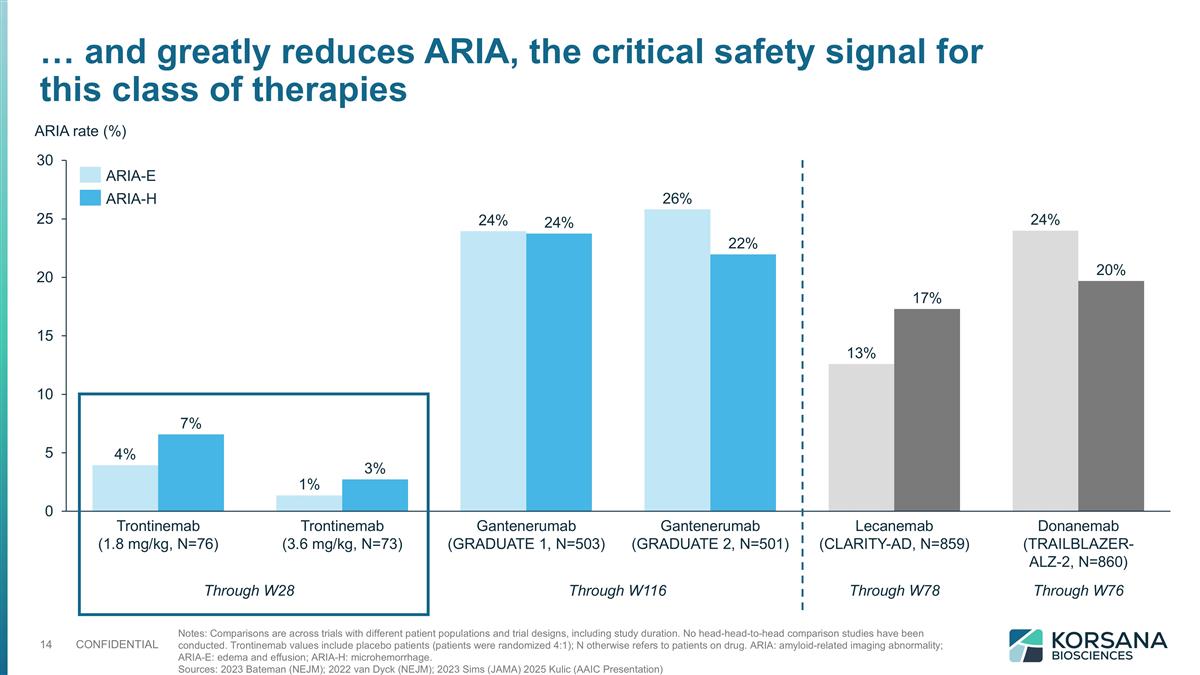
Notes: Comparisons are across trials with different patient populations and trial designs, including study duration. No head-head-to-head comparison studies have been conducted. Trontinemab values include placebo patients (patients were randomized 4:1); N otherwise refers to patients on drug. ARIA: amyloid-related imaging abnormality; ARIA-E: edema and effusion; ARIA-H: microhemorrhage. Sources: 2023 Bateman (NEJM); 2022 van Dyck (NEJM); 2023 Sims (JAMA) 2025 Kulic (AAIC Presentation) … and greatly reduces ARIA, the critical safety signal for this class of therapies ARIA rate (%) Through W28 Through W116 Through W78 Through W76
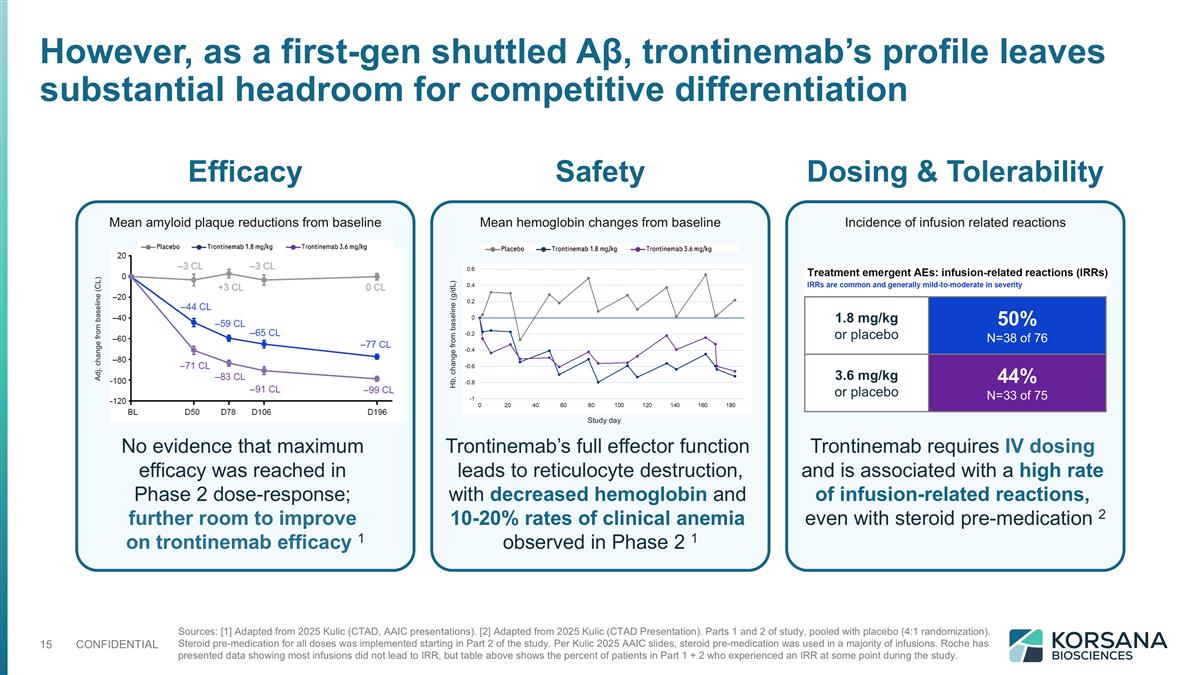
However, as a first-gen shuttled Aβ, trontinemab’s profile leaves substantial headroom for competitive differentiation Sources: [1] Adapted from 2025 Kulic (CTAD, AAIC presentations). [2] Adapted from 2025 Kulic (CTAD Presentation). Parts 1 and 2 of study, pooled with placebo (4:1 randomization). Steroid pre-medication for all doses was implemented starting in Part 2 of the study. Per Kulic 2025 AAIC slides, steroid pre-medication was used in a majority of infusions. Roche has presented data showing most infusions did not lead to IRR, but table above shows the percent of patients in Part 1 + 2 who experienced an IRR at some point during the study. Efficacy Safety Dosing & Tolerability No evidence that maximum efficacy was reached in Phase 2 dose-response; further room to improve on trontinemab efficacy 1 Trontinemab requires IV dosing and is associated with a high rate of infusion-related reactions, even with steroid pre-medication 2 Trontinemab’s full effector function leads to reticulocyte destruction, with decreased hemoglobin and 10-20% rates of clinical anemia observed in Phase 2 1 Mean hemoglobin changes from baseline Mean amyloid plaque reductions from baseline Incidence of infusion related reactions Hb, change from baseline (g/dL) Adj. change from baseline (CL) Study day 1.8 mg/kg or placebo 50% N=38 of 76 3.6 mg/kg or placebo 44% N=33 of 75

Korsana has the potential best-in-class approach

Key Value Drivers for KRSA-028 Korsana’s goal is to achieve a best-in-class shuttled Aβ therapy, offering meaningful improvements over trontinemab FAST, ROBUST AMYLOID REDUCTIONS Efficacy on par or greater than trontinemab Minimal ARIA risk, avoid hematologic AEs DIFFERENTIATED SAFETY PROFILE Low-volume subcutaneous autoinjector for infrequent dosing (Q4W or less) CONVENIENT, PATIENT-FRIENDLY DOSING
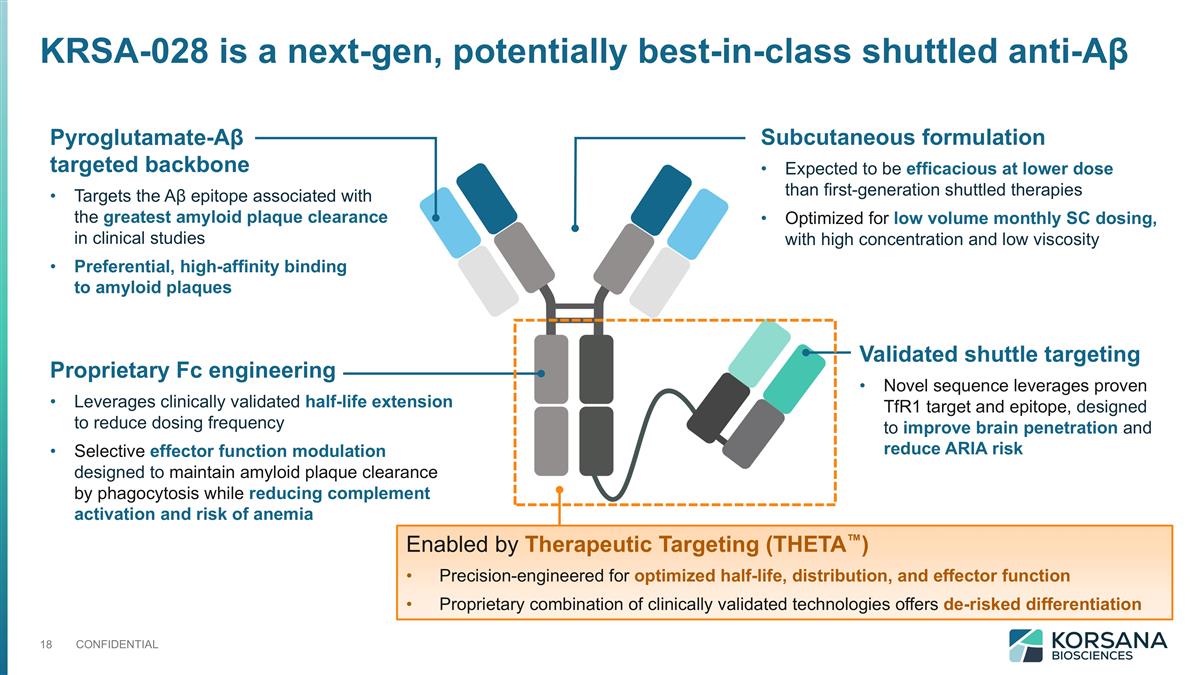
Proprietary Fc engineering Leverages clinically validated half-life extension to reduce dosing frequency Selective effector function modulation designed to maintain amyloid plaque clearance by phagocytosis while reducing complement activation and risk of anemia Subcutaneous formulation Expected to be efficacious at lower dose than first-generation shuttled therapies Optimized for low volume monthly SC dosing, with high concentration and low viscosity Validated shuttle targeting Novel sequence leverages proven TfR1 target and epitope, designed to improve brain penetration and reduce ARIA risk Enabled by Therapeutic Targeting (THETA™) Precision-engineered for optimized half-life, distribution, and effector function Proprietary combination of clinically validated technologies offers de-risked differentiation Pyroglutamate-Aβ targeted backbone Targets the Aβ epitope associated with the greatest amyloid plaque clearance in clinical studies Preferential, high-affinity binding to amyloid plaques KRSA-028 is a next-gen, potentially best-in-class shuttled anti-Aβ
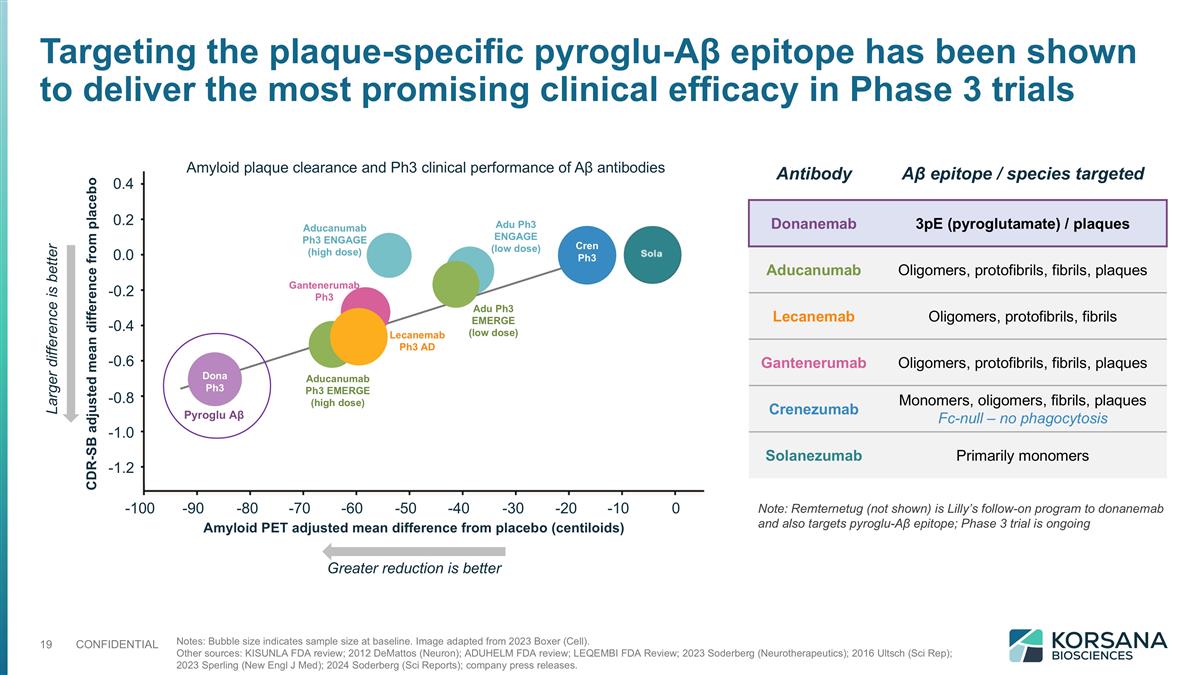
Notes: Bubble size indicates sample size at baseline. Image adapted from 2023 Boxer (Cell). Other sources: KISUNLA FDA review; 2012 DeMattos (Neuron); ADUHELM FDA review; LEQEMBI FDA Review; 2023 Soderberg (Neurotherapeutics); 2016 Ultsch (Sci Rep); 2023 Sperling (New Engl J Med); 2024 Soderberg (Sci Reports); company press releases. 0.4 0.2 0.0 -0.2 -0.4 -0.6 -0.8 -1.0 -1.2 Greater reduction is better Larger difference is better Amyloid PET adjusted mean difference from placebo (centiloids) CDR-SB adjusted mean difference from placebo -100 -90 -80 -70 -60 -50 -40 -30 -20 -10 0 Dona Ph3 Cren Ph3 Pyroglu Aβ Aducanumab Ph3 EMERGE (high dose) Aducanumab Ph3 ENGAGE (high dose) Adu Ph3 EMERGE (low dose) Adu Ph3 ENGAGE (low dose) Gantenerumab Ph3 Lecanemab Ph3 AD Amyloid plaque clearance and Ph3 clinical performance of Aβ antibodies Antibody Aβ epitope / species targeted Donanemab 3pE (pyroglutamate) / plaques Aducanumab Oligomers, protofibrils, fibrils, plaques Lecanemab Oligomers, protofibrils, fibrils Gantenerumab Oligomers, protofibrils, fibrils, plaques Crenezumab Monomers, oligomers, fibrils, plaques Fc-null – no phagocytosis Solanezumab Primarily monomers Targeting the plaque-specific pyroglu-Aβ epitope has been shown to deliver the most promising clinical efficacy in Phase 3 trials Note: Remternetug (not shown) is Lilly’s follow-on program to donanemab and also targets pyroglu-Aβ epitope; Phase 3 trial is ongoing
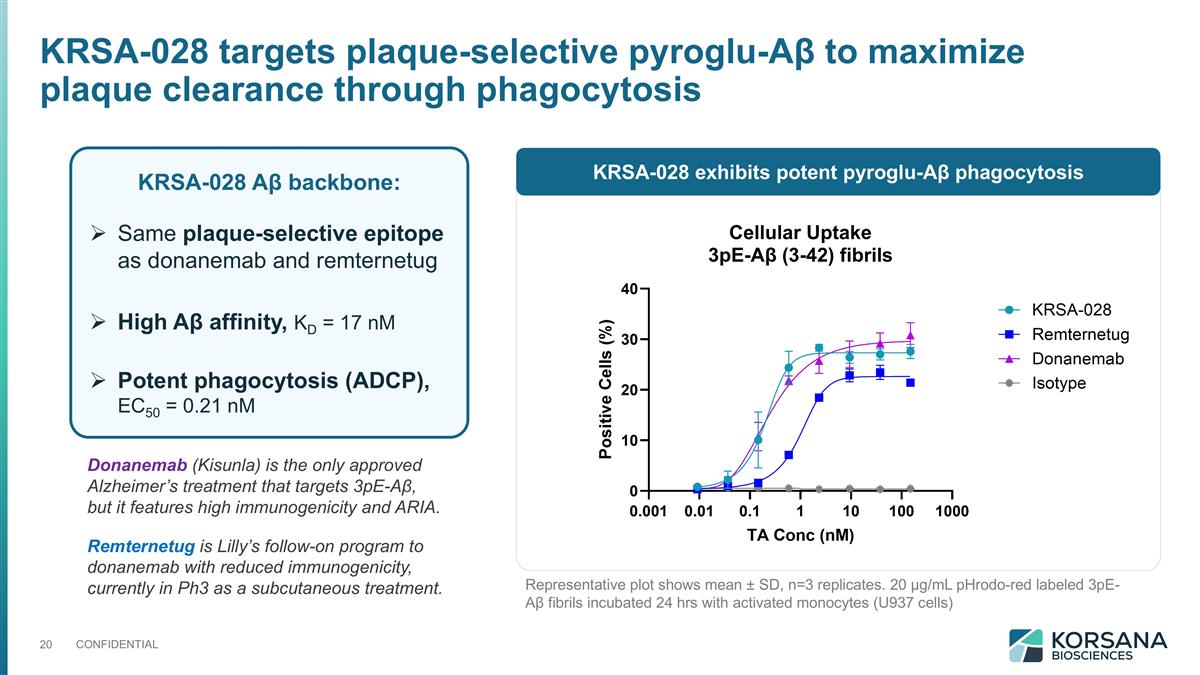
KRSA-028 Aβ backbone: Same plaque-selective epitope as donanemab and remternetug High Aβ affinity, KD = 17 nM Potent phagocytosis (ADCP), EC50 = 0.21 nM Representative plot shows mean ± SD, n=3 replicates. 20 µg/mL pHrodo-red labeled 3pE-Aβ fibrils incubated 24 hrs with activated monocytes (U937 cells) Donanemab (Kisunla) is the only approved Alzheimer’s treatment that targets 3pE-Aβ, but it features high immunogenicity and ARIA. Remternetug is Lilly’s follow-on program to donanemab with reduced immunogenicity, currently in Ph3 as a subcutaneous treatment. KRSA-028 targets plaque-selective pyroglu-Aβ to maximize plaque clearance through phagocytosis KRSA-028 exhibits potent pyroglu-Aβ phagocytosis
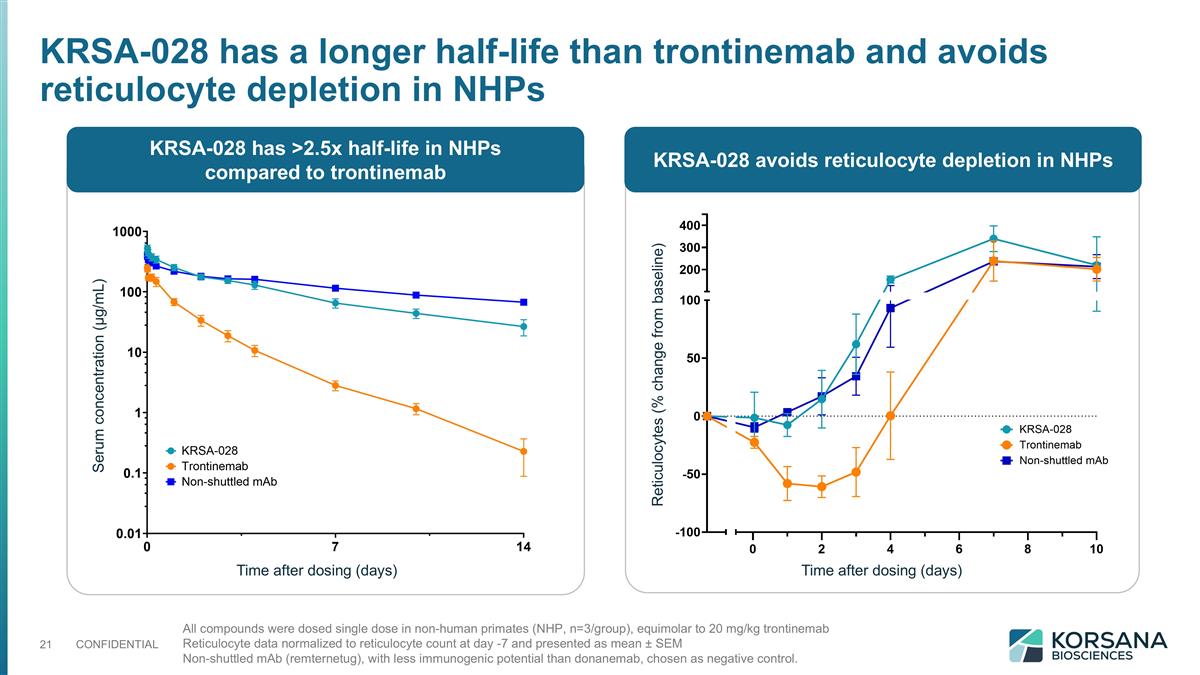
All compounds were dosed single dose in non-human primates (NHP, n=3/group), equimolar to 20 mg/kg trontinemab Reticulocyte data normalized to reticulocyte count at day -7 and presented as mean ± SEM Non-shuttled mAb (remternetug), with less immunogenic potential than donanemab, chosen as negative control. KRSA-028 has a longer half-life than trontinemab and avoids reticulocyte depletion in NHPs KRSA-028 has >2.5x half-life in NHPs compared to trontinemab KRSA-028 avoids reticulocyte depletion in NHPs Time after dosing (days) Time after dosing (days) Serum concentration (µg/mL) Reticulocytes (% change from baseline)
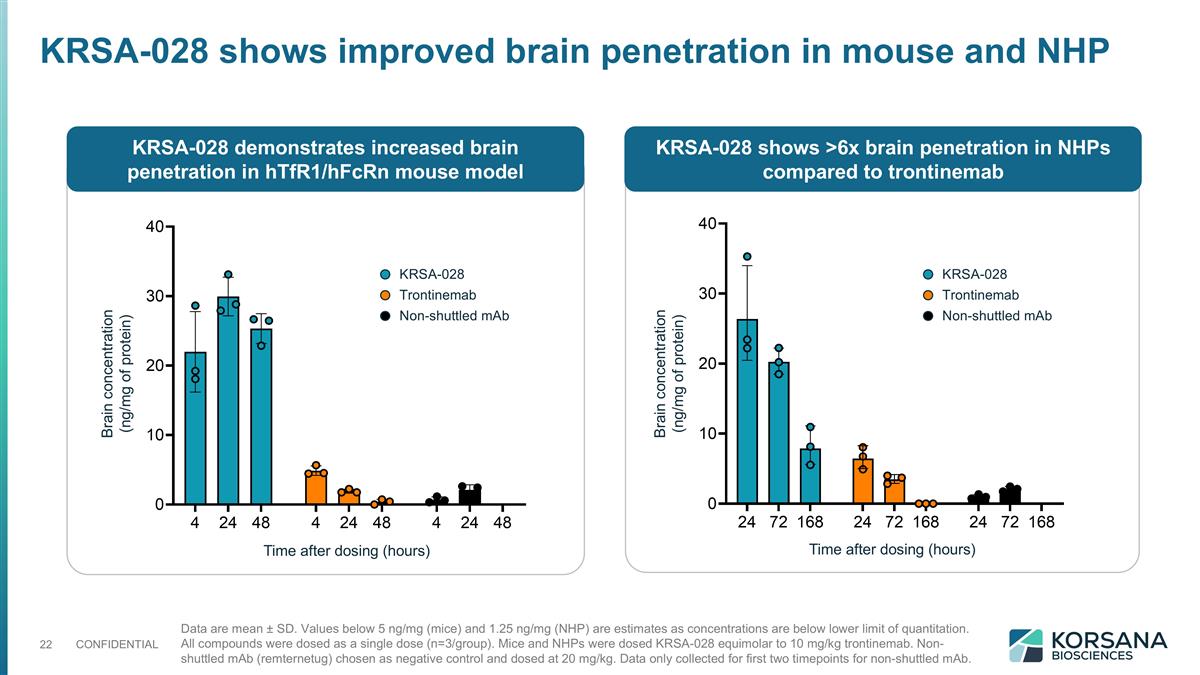
Data are mean ± SD. Values below 5 ng/mg (mice) and 1.25 ng/mg (NHP) are estimates as concentrations are below lower limit of quantitation. All compounds were dosed as a single dose (n=3/group). Mice and NHPs were dosed KRSA-028 equimolar to 10 mg/kg trontinemab. Non-shuttled mAb (remternetug) chosen as negative control and dosed at 20 mg/kg. Data only collected for first two timepoints for non-shuttled mAb. KRSA-028 shows improved brain penetration in mouse and NHP KRSA-028 demonstrates increased brain penetration in hTfR1/hFcRn mouse model KRSA-028 shows >6x brain penetration in NHPs compared to trontinemab Time after dosing (hours) Time after dosing (hours) Brain concentration (ng/mg of protein) Brain concentration (ng/mg of protein) KRSA-028 Trontinemab Non-shuttled mAb KRSA-028 Trontinemab Non-shuttled mAb
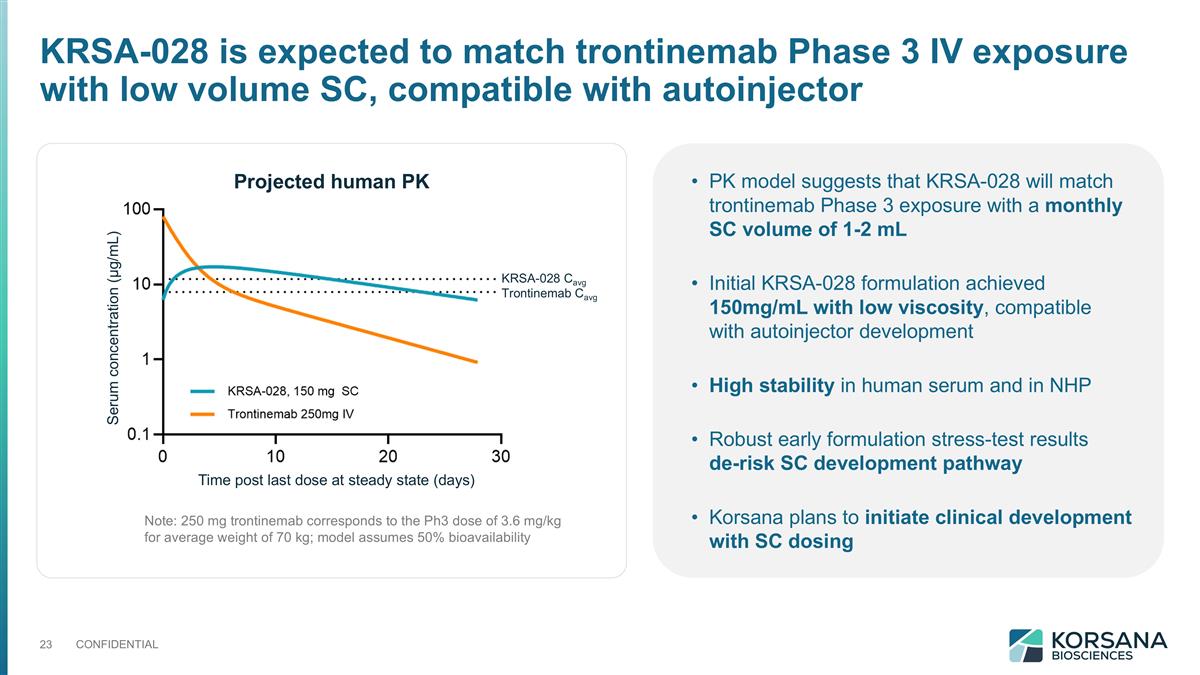
PK model suggests that KRSA-028 will match trontinemab Phase 3 exposure with a monthly SC volume of 1-2 mL Initial KRSA-028 formulation achieved 150mg/mL with low viscosity, compatible with autoinjector development High stability in human serum and in NHP Robust early formulation stress-test results de-risk SC development pathway Korsana plans to initiate clinical development with SC dosing Note: 250 mg trontinemab corresponds to the Ph3 dose of 3.6 mg/kg for average weight of 70 kg; model assumes 50% bioavailability KRSA-028 is expected to match trontinemab Phase 3 IV exposure with low volume SC, compatible with autoinjector KRSA-028 Cavg Trontinemab Cavg Projected human PK Time post last dose at steady state (days) Serum concentration (µg/mL)

We believe KRSA-028 preclinical data accelerate & de-risk development Precision engineered for a differentiated therapeutic profile Novel Aβ and TfR1 binding sequences retain key features of clinically validated molecules High affinity pyroglu-Aβ binding and clearance via ADCP leverage best-proven mechanism of efficacy TfR1 binding matches trontinemab epitope with similar affinity, leveraging the best-proven mechanism of brain distribution Clinically validated Fc modifications add half-life extension and effector function modulation to enable a lower dose and reduce anemia risk Early formulation work supports high concentration, low viscosity to enable low-volume SC Composition of matter patent applications filed

Korsana has a rapid path to value creation
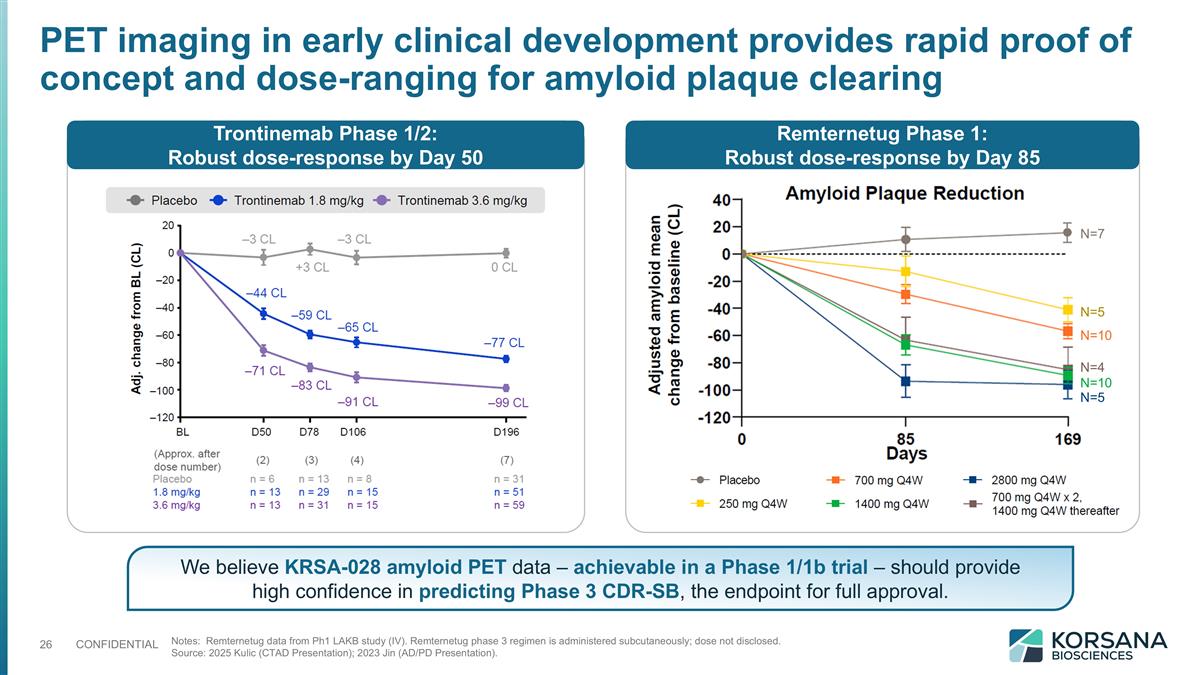
PET imaging in early clinical development provides rapid proof of concept and dose-ranging for amyloid plaque clearing Trontinemab Phase 1/2: Robust dose-response by Day 50 Remternetug Phase 1: Robust dose-response by Day 85 Notes: Remternetug data from Ph1 LAKB study (IV). Remternetug phase 3 regimen is administered subcutaneously; dose not disclosed. Source: 2025 Kulic (CTAD Presentation); 2023 Jin (AD/PD Presentation). N=7 N=5 N=10 N=4 N=10 N=5 We believe KRSA-028 amyloid PET data – achievable in a Phase 1/1b trial – should provide high confidence in predicting Phase 3 CDR-SB, the endpoint for full approval.

A confluence of scientific innovations and market dynamics are driving Alzheimer’s treatment to a major inflection point The future of Alzheimer’s treatment will center on a prevention-based paradigm, where patients are diagnosed before symptom onset and given a safe, convenient Aβ plaque-clearing therapy. Despite a slower than expected launch, sales of Aβ therapies are accelerating Upcoming catalysts in presymptomatic AD may lead to rapid market expansion Improved dose regimens (SC, dose titration) Blood-based biomarkers are gaining traction Increasing footprint of global approvals Future entrants (e.g., trontinemab) will further grow market Donanemab TRAILBLAZER-ALZ 3 (~2027) Lecanemab AHEAD 3-45 (~2028) Remternetug TRAILRUNNER-ALZ 3 (~2029) Trontinemab PrevenTRON (study start 2026) Sales expected to reach $1B in 2026, $5B+ by 2030 Data could greatly expand eligible patient pool Sources: Biogen SEC filings; Lilly SEC filings; VisibleAlpha
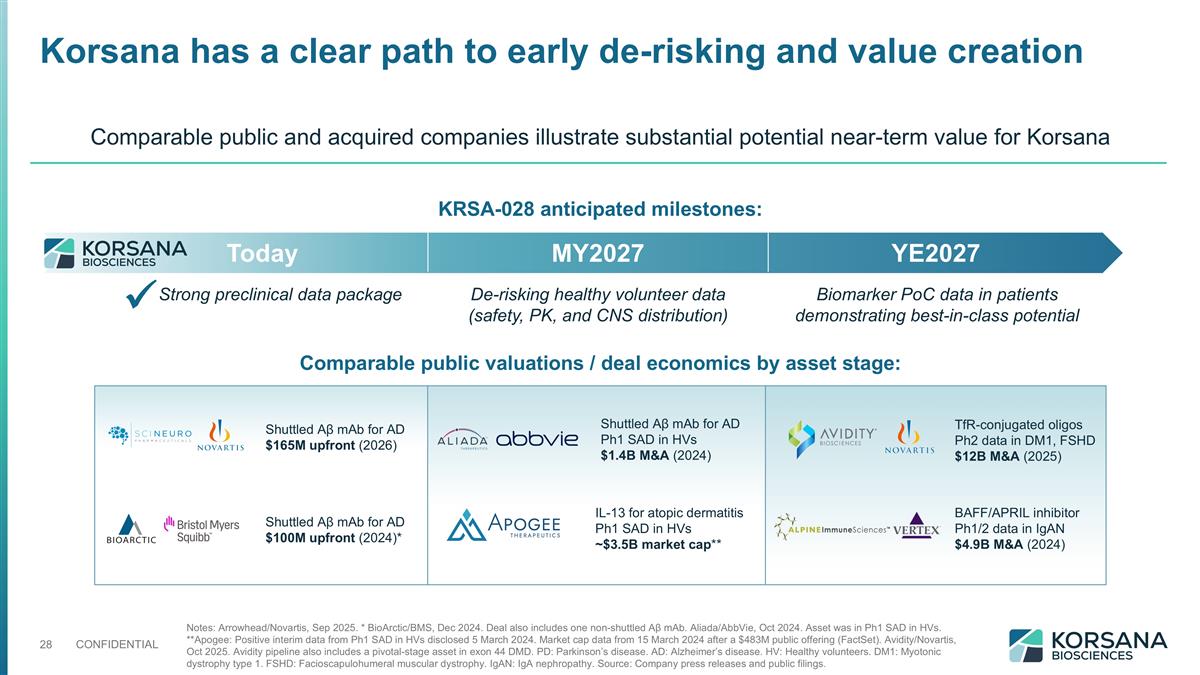
Strong preclinical data package De-risking healthy volunteer data (safety, PK, and CNS distribution) Biomarker PoC data in patients demonstrating best-in-class potential Today MY2027 YE2027 KRSA-028 anticipated milestones: Notes: Arrowhead/Novartis, Sep 2025. * BioArctic/BMS, Dec 2024. Deal also includes one non-shuttled Aβ mAb. Aliada/AbbVie, Oct 2024. Asset was in Ph1 SAD in HVs. **Apogee: Positive interim data from Ph1 SAD in HVs disclosed 5 March 2024. Market cap data from 15 March 2024 after a $483M public offering (FactSet). Avidity/Novartis, Oct 2025. Avidity pipeline also includes a pivotal-stage asset in exon 44 DMD. PD: Parkinson’s disease. AD: Alzheimer’s disease. HV: Healthy volunteers. DM1: Myotonic dystrophy type 1. FSHD: Facioscapulohumeral muscular dystrophy. IgAN: IgA nephropathy. Source: Company press releases and public filings. Korsana has a clear path to early de-risking and value creation ü Comparable public and acquired companies illustrate substantial potential near-term value for Korsana Comparable public valuations / deal economics by asset stage: Shuttled Aβ mAb for AD $100M upfront (2024)* IL-13 for atopic dermatitis Ph1 SAD in HVs ~$3.5B market cap** BAFF/APRIL inhibitor Ph1/2 data in IgAN $4.9B M&A (2024) Shuttled Aβ mAb for AD Ph1 SAD in HVs $1.4B M&A (2024) Shuttled Aβ mAb for AD $165M upfront (2026) TfR-conjugated oligos Ph2 data in DM1, FSHD $12B M&A (2025)
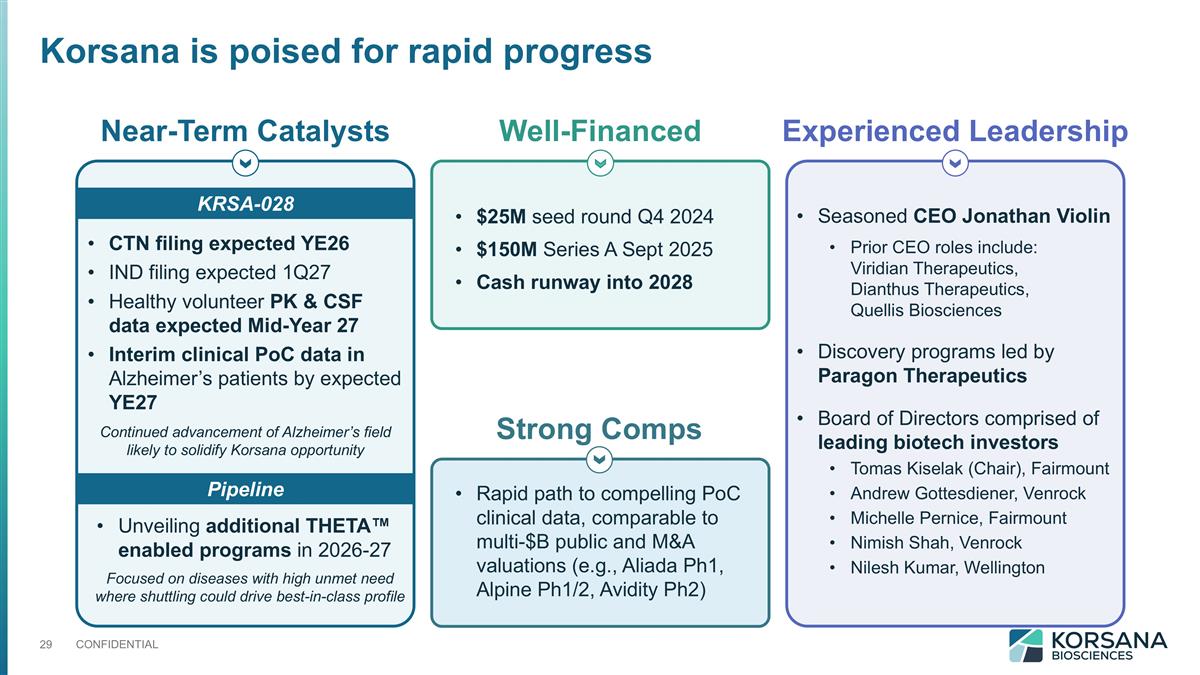
Near-Term Catalysts Well-Financed Experienced Leadership CTN filing expected YE26 IND filing expected 1Q27 Healthy volunteer PK & CSF data expected Mid-Year 27 Interim clinical PoC data in Alzheimer’s patients by expected YE27 $25M seed round Q4 2024 $150M Series A Sept 2025 Cash runway into 2028 Seasoned CEO Jonathan Violin Prior CEO roles include: Viridian Therapeutics, Dianthus Therapeutics, Quellis Biosciences Discovery programs led by Paragon Therapeutics Board of Directors comprised of leading biotech investors Tomas Kiselak (Chair), Fairmount Andrew Gottesdiener, Venrock Michelle Pernice, Fairmount Nimish Shah, Venrock Nilesh Kumar, Wellington KRSA-028 Unveiling additional THETA™ enabled programs in 2026-27 Pipeline Continued advancement of Alzheimer’s field likely to solidify Korsana opportunity Focused on diseases with high unmet need where shuttling could drive best-in-class profile Strong Comps Rapid path to compelling PoC clinical data, comparable to multi-$B public and M&A valuations (e.g., Aliada Ph1, Alpine Ph1/2, Avidity Ph2) Korsana is poised for rapid progress

Thank you

Appendix
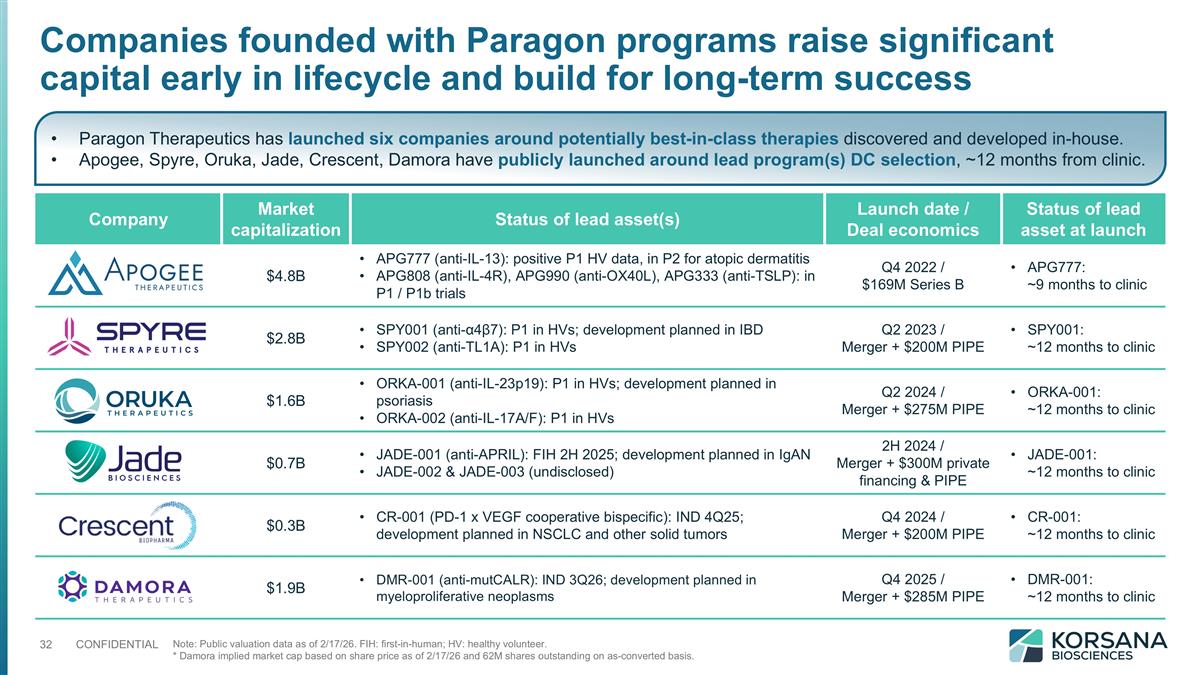
Company Market capitalization Status of lead asset(s) Launch date / Deal economics Status of lead asset at launch $4.8B APG777 (anti-IL-13): positive P1 HV data, in P2 for atopic dermatitis APG808 (anti-IL-4R), APG990 (anti-OX40L), APG333 (anti-TSLP): in P1 / P1b trials Q4 2022 / $169M Series B APG777: ~9 months to clinic $2.8B SPY001 (anti-α4β7): P1 in HVs; development planned in IBD SPY002 (anti-TL1A): P1 in HVs Q2 2023 / Merger + $200M PIPE SPY001: ~12 months to clinic $1.6B ORKA-001 (anti-IL-23p19): P1 in HVs; development planned in psoriasis ORKA-002 (anti-IL-17A/F): P1 in HVs Q2 2024 / Merger + $275M PIPE ORKA-001: ~12 months to clinic $0.7B JADE-001 (anti-APRIL): FIH 2H 2025; development planned in IgAN JADE-002 & JADE-003 (undisclosed) 2H 2024 / Merger + $300M private financing & PIPE JADE-001: ~12 months to clinic $0.3B CR-001 (PD-1 x VEGF cooperative bispecific): IND 4Q25; development planned in NSCLC and other solid tumors Q4 2024 / Merger + $200M PIPE CR-001: ~12 months to clinic $1.9B DMR-001 (anti-mutCALR): IND 3Q26; development planned in myeloproliferative neoplasms Q4 2025 / Merger + $285M PIPE DMR-001: ~12 months to clinic Companies founded with Paragon programs raise significant capital early in lifecycle and build for long-term success Note: Public valuation data as of 2/17/26. FIH: first-in-human; HV: healthy volunteer. * Damora implied market cap based on share price as of 2/17/26 and 62M shares outstanding on as-converted basis. Paragon Therapeutics has launched six companies around potentially best-in-class therapies discovered and developed in-house. Apogee, Spyre, Oruka, Jade, Crescent, Damora have publicly launched around lead program(s) DC selection, ~12 months from clinic.
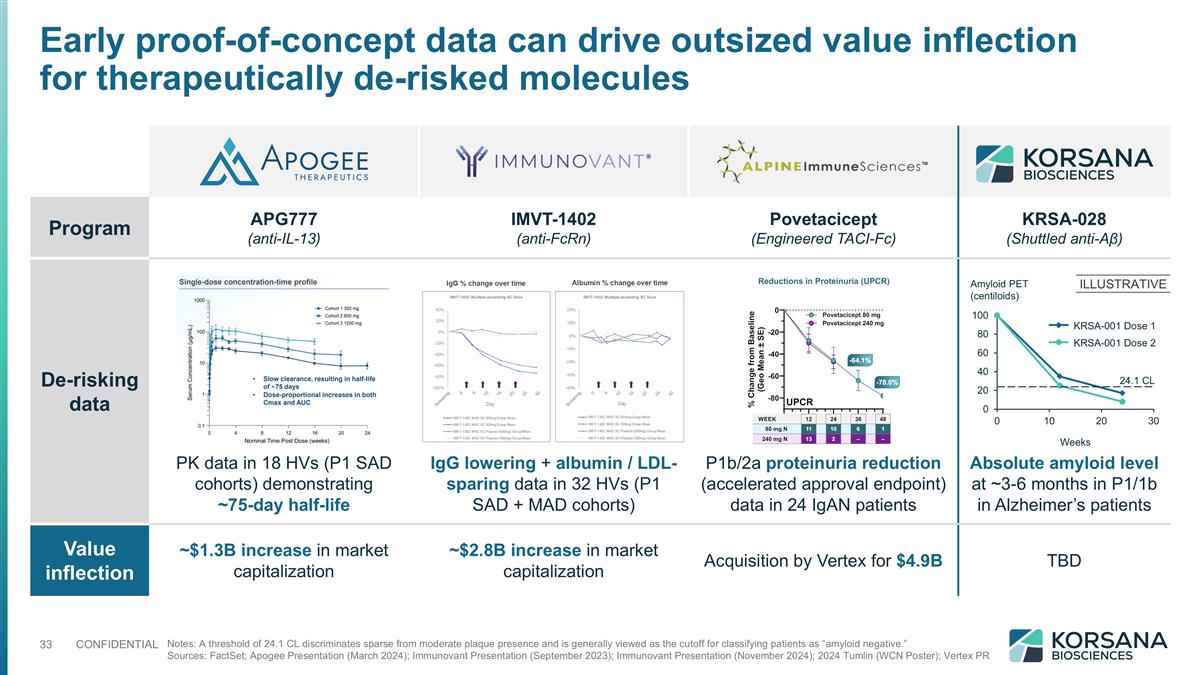
Program APG777 (anti-IL-13) IMVT-1402 (anti-FcRn) Povetacicept (Engineered TACI-Fc) KRSA-028 (Shuttled anti-Aβ) De-risking data PK data in 18 HVs (P1 SAD cohorts) demonstrating ~75-day half-life IgG lowering + albumin / LDL-sparing data in 32 HVs (P1 SAD + MAD cohorts) P1b/2a proteinuria reduction (accelerated approval endpoint) data in 24 IgAN patients Absolute amyloid level at ~3-6 months in P1/1b in Alzheimer’s patients Value inflection ~$1.3B increase in market capitalization ~$2.8B increase in market capitalization Acquisition by Vertex for $4.9B TBD Early proof-of-concept data can drive outsized value inflection for therapeutically de-risked molecules Notes: A threshold of 24.1 CL discriminates sparse from moderate plaque presence and is generally viewed as the cutoff for classifying patients as “amyloid negative.” Sources: FactSet; Apogee Presentation (March 2024); Immunovant Presentation (September 2023); Immunovant Presentation (November 2024); 2024 Tumlin (WCN Poster); Vertex PR ILLUSTRATIVE Weeks Amyloid PET (centiloids) 24.1 CL

Trontinemab ameliorates ARIA risk but introduces new problems; KRSA-028 was designed to deliver a best-in-class profile Kisunla, Leqembi Remternetug Trontinemab KRSA-028 Target Strong efficacy +/- ü ü ü Reduced ARIA risk û û ü ü Avoids anemia ü ü û ü Ease of admin û ü û ü Non-Shuttled Aβ mAbs Shuttled Aβ mAbs CONCEPTUAL
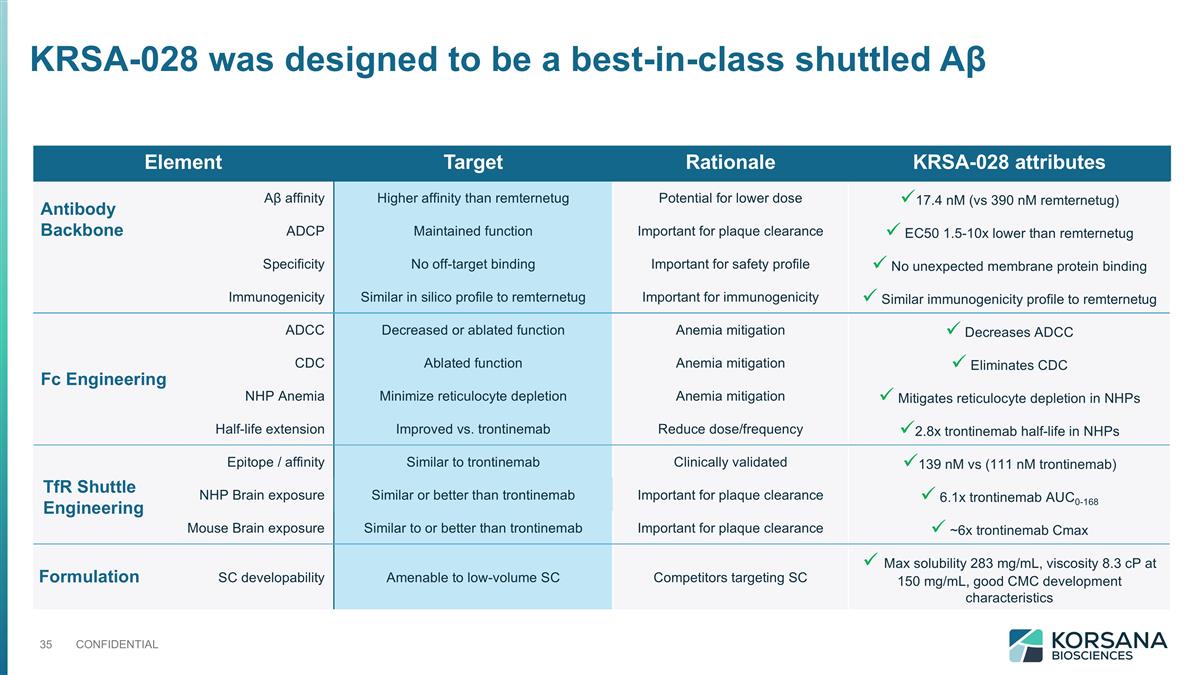
KRSA-028 was designed to be a best-in-class shuttled Aβ Element Target Rationale KRSA-028 attributes Aβ affinity Higher affinity than remternetug Potential for lower dose ü17.4 nM (vs 390 nM remternetug) ADCP Maintained function Important for plaque clearance ü EC50 1.5-10x lower than remternetug Specificity No off-target binding Important for safety profile ü No unexpected membrane protein binding Immunogenicity Similar in silico profile to remternetug Important for immunogenicity ü Similar immunogenicity profile to remternetug ADCC Decreased or ablated function Anemia mitigation ü Decreases ADCC CDC Ablated function Anemia mitigation ü Eliminates CDC NHP Anemia Minimize reticulocyte depletion Anemia mitigation ü Mitigates reticulocyte depletion in NHPs Half-life extension Improved vs. trontinemab Reduce dose/frequency ü2.8x trontinemab half-life in NHPs Epitope / affinity Similar to trontinemab Clinically validated ü139 nM vs (111 nM trontinemab) NHP Brain exposure Similar or better than trontinemab Important for plaque clearance ü 6.1x trontinemab AUC0-168 Mouse Brain exposure Similar to or better than trontinemab Important for plaque clearance ü ~6x trontinemab Cmax SC developability Amenable to low-volume SC Competitors targeting SC ü Max solubility 283 mg/mL, viscosity 8.3 cP at 150 mg/mL, good CMC development characteristics Antibody Backbone Fc Engineering TfR Shuttle Engineering Formulation
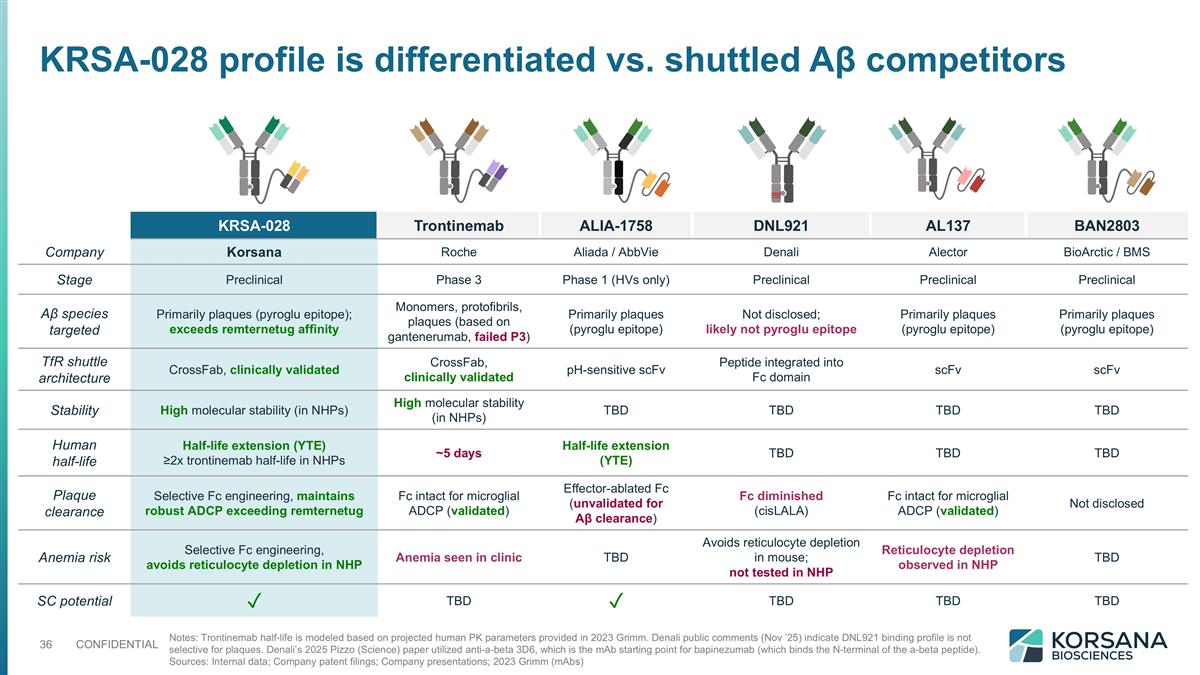
Notes: Trontinemab half-life is modeled based on projected human PK parameters provided in 2023 Grimm. Denali public comments (Nov ’25) indicate DNL921 binding profile is not selective for plaques. Denali’s 2025 Pizzo (Science) paper utilized anti-a-beta 3D6, which is the mAb starting point for bapinezumab (which binds the N-terminal of the a-beta peptide). Sources: Internal data; Company patent filings; Company presentations; 2023 Grimm (mAbs) KRSA-028 Trontinemab ALIA-1758 DNL921 AL137 BAN2803 Company Korsana Roche Aliada / AbbVie Denali Alector BioArctic / BMS Stage Preclinical Phase 3 Phase 1 (HVs only) Preclinical Preclinical Preclinical Aβ species targeted Primarily plaques (pyroglu epitope); exceeds remternetug affinity Monomers, protofibrils, plaques (based on gantenerumab, failed P3) Primarily plaques (pyroglu epitope) Not disclosed; likely not pyroglu epitope Primarily plaques (pyroglu epitope) Primarily plaques (pyroglu epitope) TfR shuttle architecture CrossFab, clinically validated CrossFab, clinically validated pH-sensitive scFv Peptide integrated into Fc domain scFv scFv Stability High molecular stability (in NHPs) High molecular stability (in NHPs) TBD TBD TBD TBD Human half-life Half-life extension (YTE) ≥2x trontinemab half-life in NHPs ~5 days Half-life extension (YTE) TBD TBD TBD Plaque clearance Selective Fc engineering, maintains robust ADCP exceeding remternetug Fc intact for microglial ADCP (validated) Effector-ablated Fc (unvalidated for Aβ clearance) Fc diminished (cisLALA) Fc intact for microglial ADCP (validated) Not disclosed Anemia risk Selective Fc engineering, avoids reticulocyte depletion in NHP Anemia seen in clinic TBD Avoids reticulocyte depletion in mouse; not tested in NHP Reticulocyte depletion observed in NHP TBD SC potential ✓ TBD ✓ TBD TBD TBD KRSA-028 profile is differentiated vs. shuttled Aβ competitors

Anticipated clinical profile based on available preclinical data Plaque clearance Higher Lower Anemia risk Higher AL137 Pronounced reticulocyte depletion in NHP Max dose response not reached; significant clinical anemia CONCEPTUAL Trontinemab We believe KRSA-028 is uniquely positioned in the shuttled Aβ competitive landscape Lower KRSA-028 Robust NHP data DNL921 Mouse data only ALIA-1758 No phagocytosis ALIA-1758: Clinical-stage shuttled Aβ being developed by AbbVie (via acquisition of Aliada Therapeutics). DNL921: Preclinical shuttled Aβ being developed by Denali Therapeutics. AL137: Preclinical shuttled Aβ being developed by Alector Therapeutics.
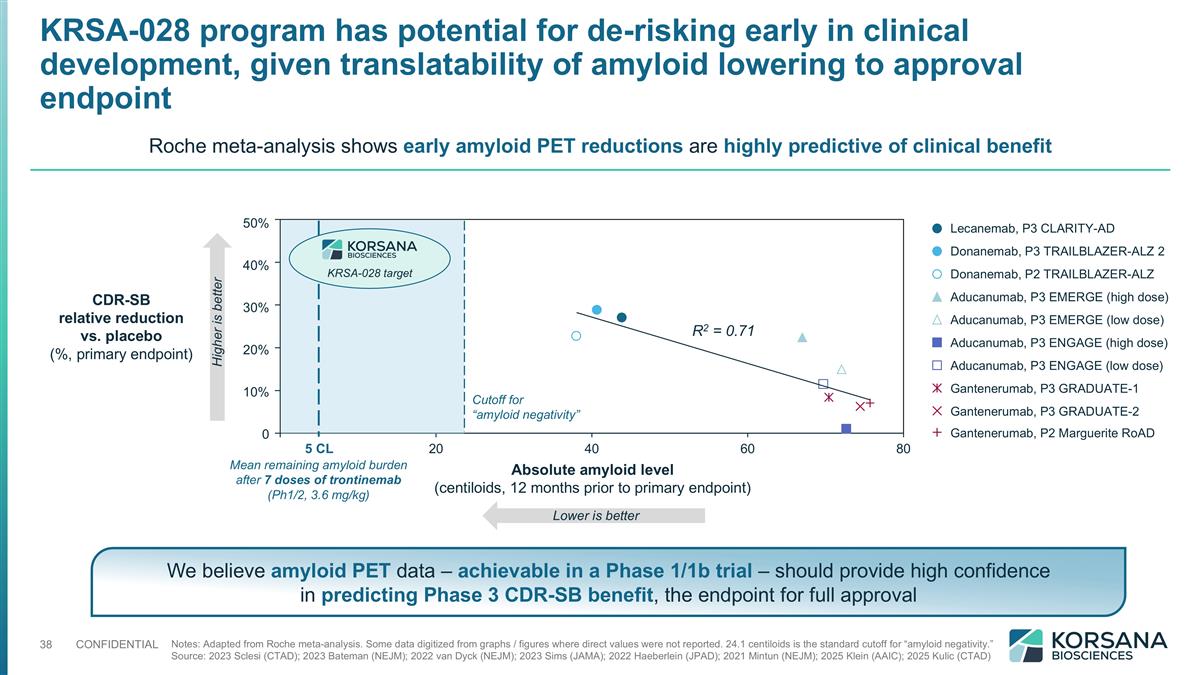
KRSA-028 program has potential for de-risking early in clinical development, given translatability of amyloid lowering to approval endpoint Notes: Adapted from Roche meta-analysis. Some data digitized from graphs / figures where direct values were not reported. 24.1 centiloids is the standard cutoff for “amyloid negativity.” Source: 2023 Sclesi (CTAD); 2023 Bateman (NEJM); 2022 van Dyck (NEJM); 2023 Sims (JAMA); 2022 Haeberlein (JPAD); 2021 Mintun (NEJM); 2025 Klein (AAIC); 2025 Kulic (CTAD) Roche meta-analysis shows early amyloid PET reductions are highly predictive of clinical benefit % % % % % R2 = 0.71 CDR-SB relative reduction vs. placebo (%, primary endpoint) Absolute amyloid level (centiloids, 12 months prior to primary endpoint) Lower is better Higher is better Mean remaining amyloid burden after 7 doses of trontinemab (Ph1/2, 3.6 mg/kg) 5 CL Cutoff for “amyloid negativity” KRSA-028 target We believe amyloid PET data – achievable in a Phase 1/1b trial – should provide high confidence in predicting Phase 3 CDR-SB benefit, the endpoint for full approval
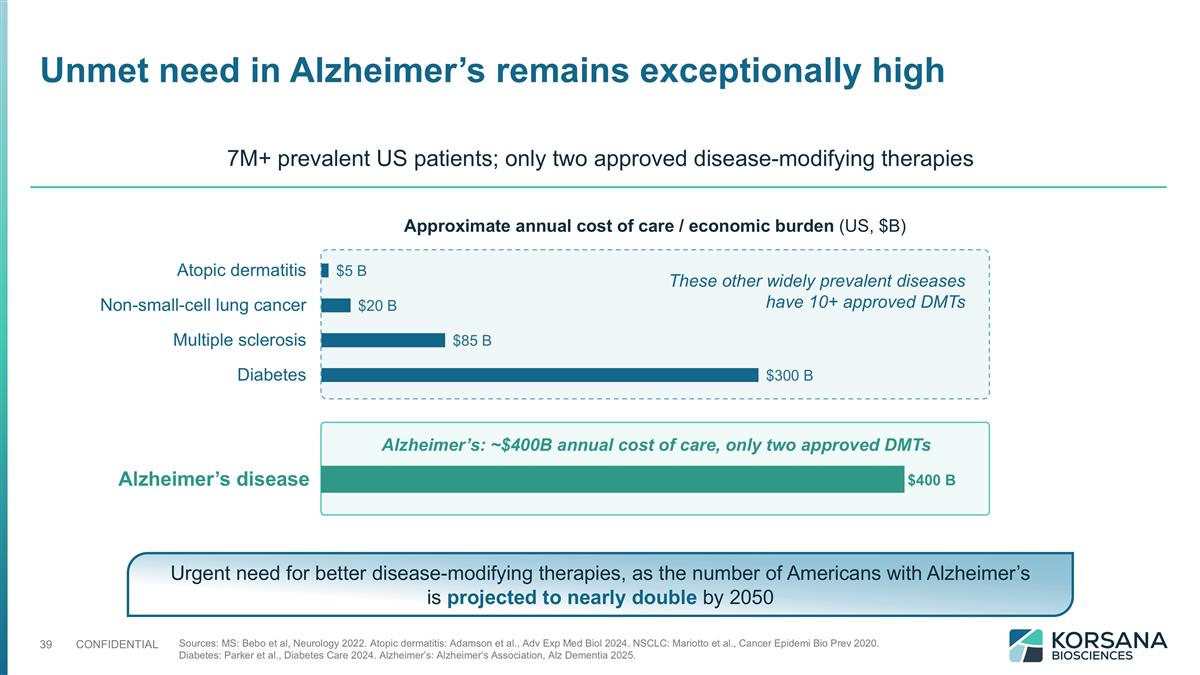
Unmet need in Alzheimer’s remains exceptionally high Approximate annual cost of care / economic burden (US, $B) Alzheimer’s disease Alzheimer’s: ~$400B annual cost of care, only two approved DMTs These other widely prevalent diseases have 10+ approved DMTs 7M+ prevalent US patients; only two approved disease-modifying therapies Sources: MS: Bebo et al, Neurology 2022. Atopic dermatitis: Adamson et al., Adv Exp Med Biol 2024. NSCLC: Mariotto et al., Cancer Epidemi Bio Prev 2020. Diabetes: Parker et al., Diabetes Care 2024. Alzheimer’s: Alzheimer’s Association, Alz Dementia 2025. Urgent need for better disease-modifying therapies, as the number of Americans with Alzheimer’s is projected to nearly double by 2050
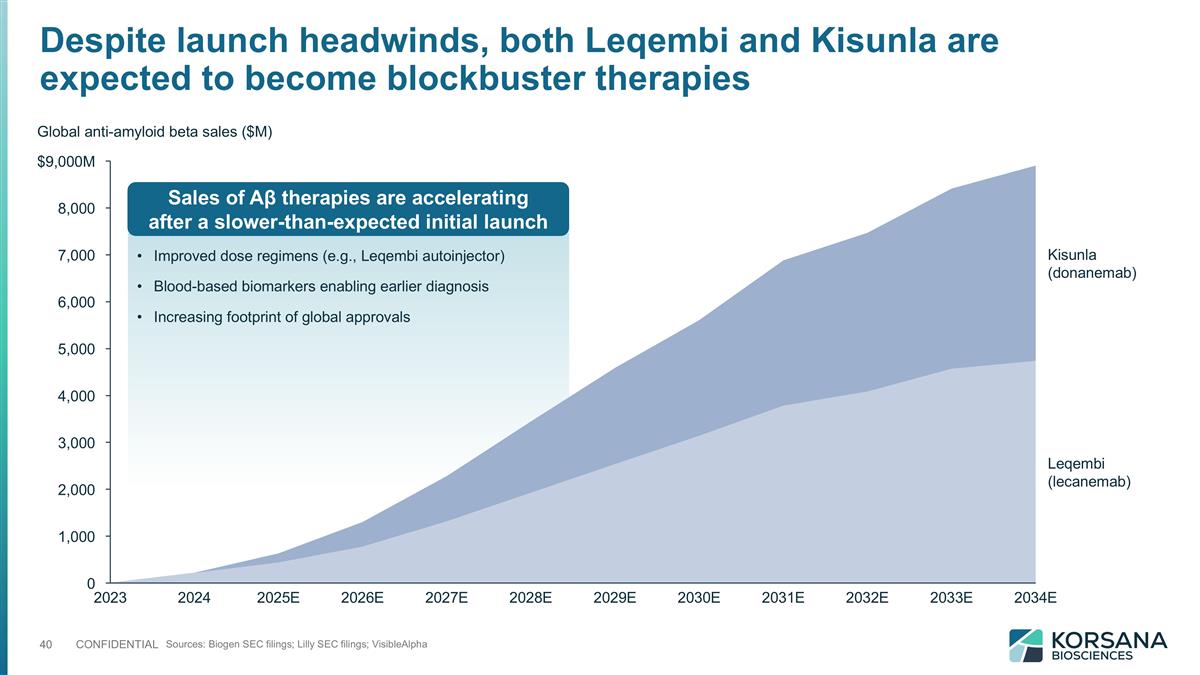
Improved dose regimens (e.g., Leqembi autoinjector) Blood-based biomarkers enabling earlier diagnosis Increasing footprint of global approvals Sources: Biogen SEC filings; Lilly SEC filings; VisibleAlpha $M Global anti-amyloid beta sales ($M) Sales of Aβ therapies are accelerating after a slower-than-expected initial launch Despite launch headwinds, both Leqembi and Kisunla are expected to become blockbuster therapies
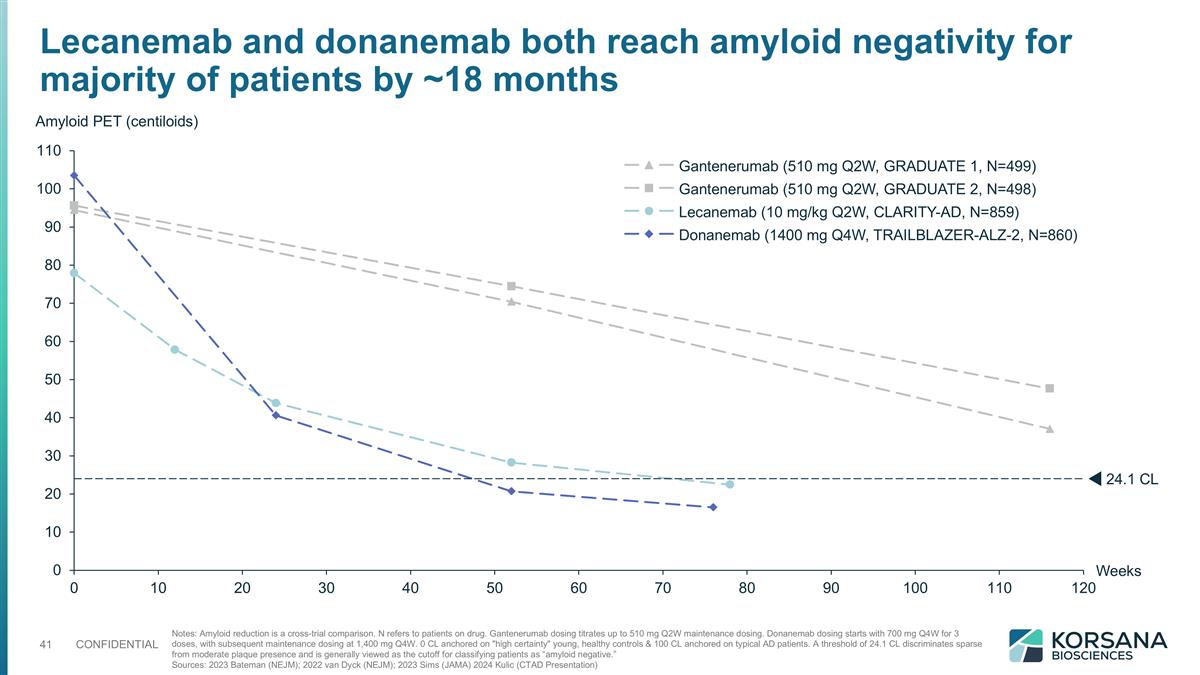
Lecanemab and donanemab both reach amyloid negativity for majority of patients by ~18 months Notes: Amyloid reduction is a cross-trial comparison. N refers to patients on drug. Gantenerumab dosing titrates up to 510 mg Q2W maintenance dosing. Donanemab dosing starts with 700 mg Q4W for 3 doses, with subsequent maintenance dosing at 1,400 mg Q4W. 0 CL anchored on "high certainty" young, healthy controls & 100 CL anchored on typical AD patients. A threshold of 24.1 CL discriminates sparse from moderate plaque presence and is generally viewed as the cutoff for classifying patients as “amyloid negative.” Sources: 2023 Bateman (NEJM); 2022 van Dyck (NEJM); 2023 Sims (JAMA) 2024 Kulic (CTAD Presentation) Weeks Amyloid PET (centiloids) .1 CL
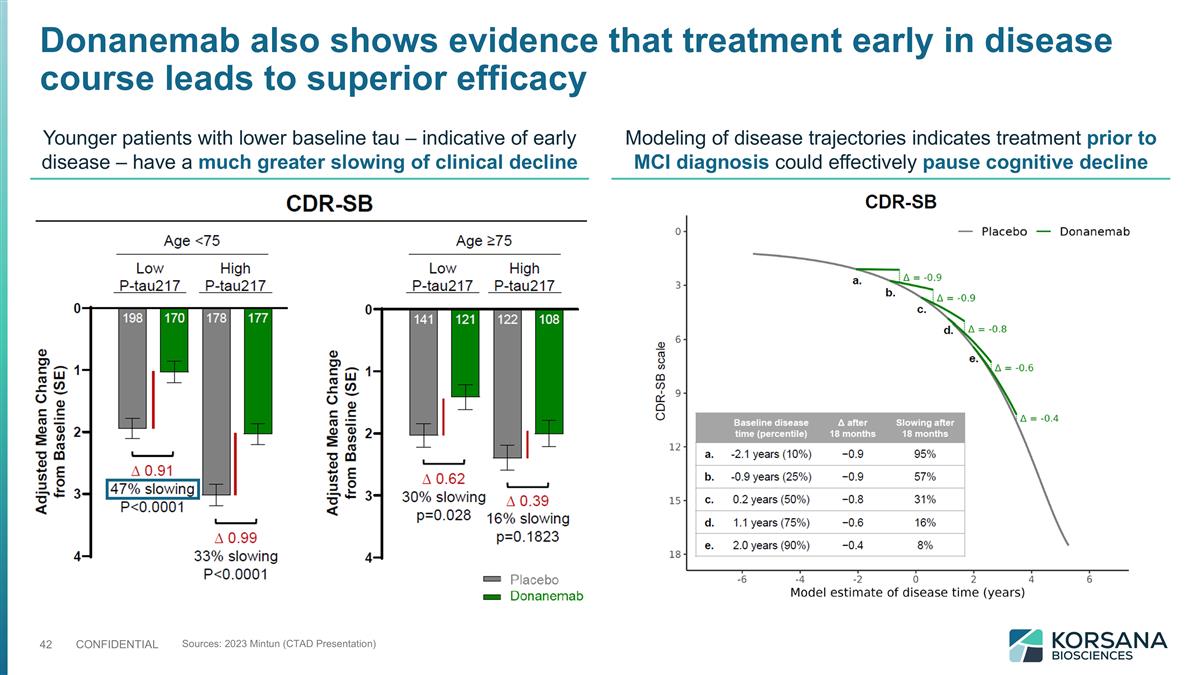
Donanemab also shows evidence that treatment early in disease course leads to superior efficacy Sources: 2023 Mintun (CTAD Presentation) Younger patients with lower baseline tau – indicative of early disease – have a much greater slowing of clinical decline Modeling of disease trajectories indicates treatment prior to MCI diagnosis could effectively pause cognitive decline
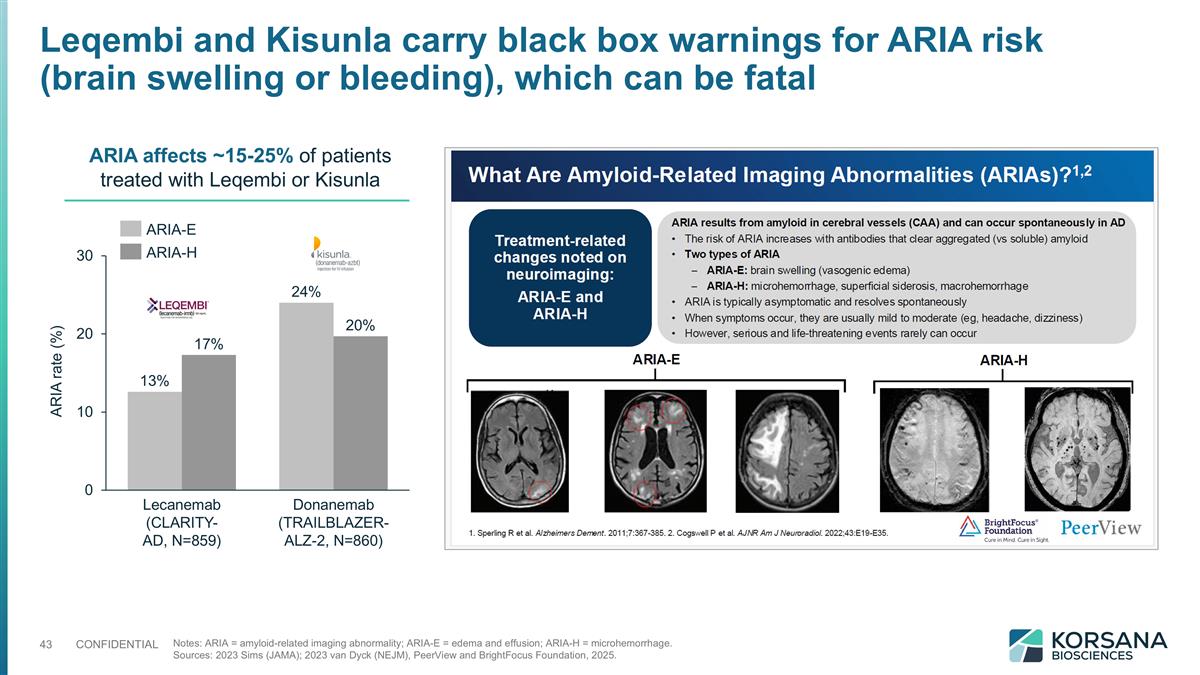
ARIA rate (%) ARIA affects ~15-25% of patients treated with Leqembi or Kisunla Notes: ARIA = amyloid-related imaging abnormality; ARIA-E = edema and effusion; ARIA-H = microhemorrhage. Sources: 2023 Sims (JAMA); 2023 van Dyck (NEJM), PeerView and BrightFocus Foundation, 2025. Leqembi and Kisunla carry black box warnings for ARIA risk (brain swelling or bleeding), which can be fatal
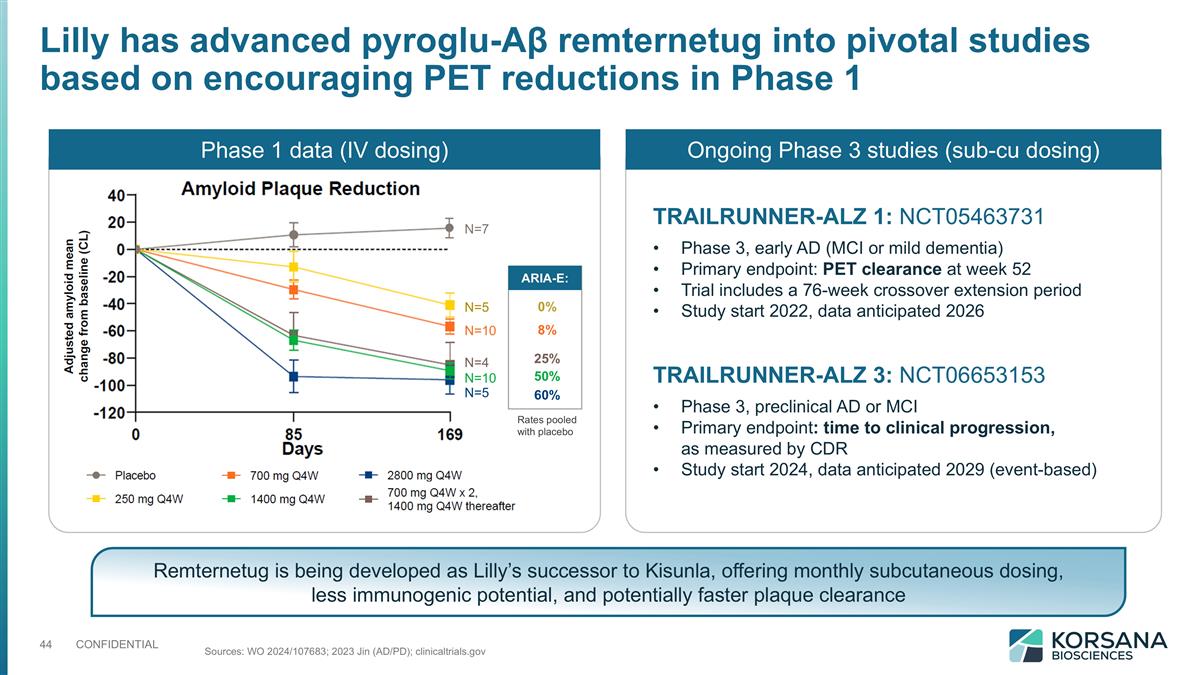
Lilly has advanced pyroglu-Aβ remternetug into pivotal studies based on encouraging PET reductions in Phase 1 N=7 N=5 N=10 N=4 N=10 N=5 ARIA-E: 0% 8% 25% 50% 60% Ongoing Phase 3 studies (sub-cu dosing) TRAILRUNNER-ALZ 1: NCT05463731 Phase 3, early AD (MCI or mild dementia) Primary endpoint: PET clearance at week 52 Trial includes a 76-week crossover extension period Study start 2022, data anticipated 2026 Sources: WO 2024/107683; 2023 Jin (AD/PD); clinicaltrials.gov TRAILRUNNER-ALZ 3: NCT06653153 Phase 3, preclinical AD or MCI Primary endpoint: time to clinical progression, as measured by CDR Study start 2024, data anticipated 2029 (event-based) Remternetug is being developed as Lilly’s successor to Kisunla, offering monthly subcutaneous dosing, less immunogenic potential, and potentially faster plaque clearance Phase 1 data (IV dosing) Rates pooled with placebo
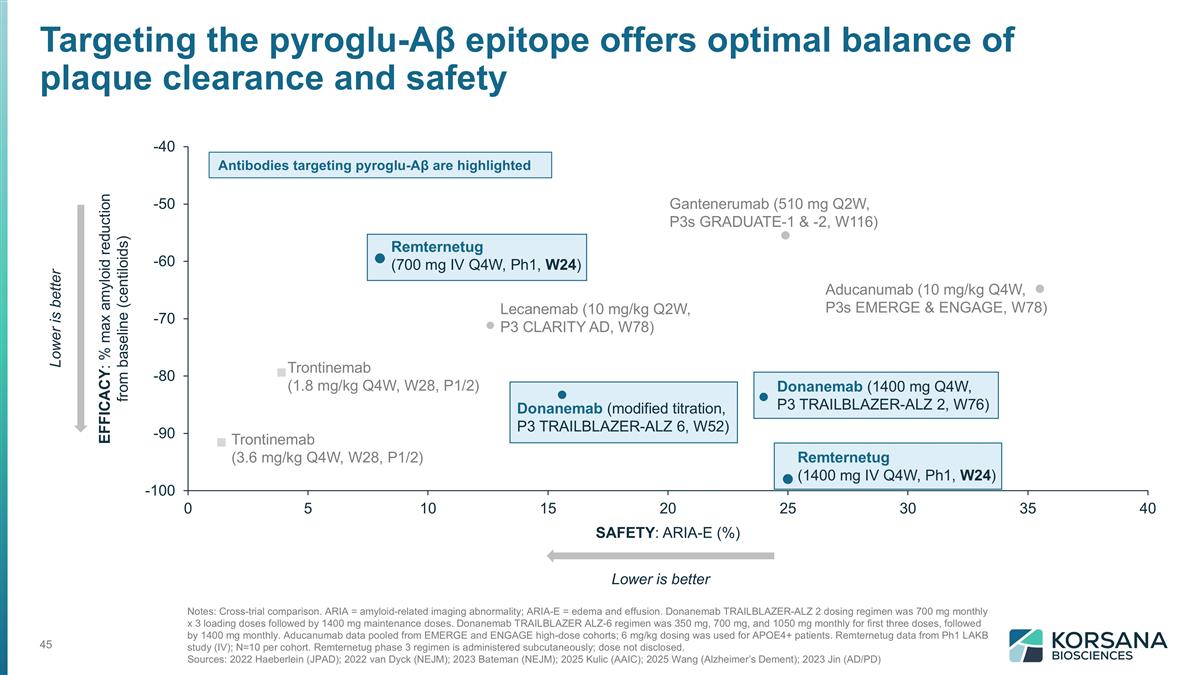
Notes: Cross-trial comparison. ARIA = amyloid-related imaging abnormality; ARIA-E = edema and effusion. Donanemab TRAILBLAZER-ALZ 2 dosing regimen was 700 mg monthly x 3 loading doses followed by 1400 mg maintenance doses. Donanemab TRAILBLAZER ALZ-6 regimen was 350 mg, 700 mg, and 1050 mg monthly for first three doses, followed by 1400 mg monthly. Aducanumab data pooled from EMERGE and ENGAGE high-dose cohorts; 6 mg/kg dosing was used for APOE4+ patients. Remternetug data from Ph1 LAKB study (IV); N=10 per cohort. Remternetug phase 3 regimen is administered subcutaneously; dose not disclosed. Sources: 2022 Haeberlein (JPAD); 2022 van Dyck (NEJM); 2023 Bateman (NEJM); 2025 Kulic (AAIC); 2025 Wang (Alzheimer’s Dement); 2023 Jin (AD/PD) Lower is better EFFICACY: Lower is better SAFETY: Remternetug (700 mg IV Q4W, Ph1, W24) Remternetug (1400 mg IV Q4W, Ph1, W24) Donanemab (modified titration, P3 TRAILBLAZER-ALZ 6, W52) Donanemab (1400 mg Q4W, P3 TRAILBLAZER-ALZ 2, W76) Lecanemab (10 mg/kg Q2W, P3 CLARITY AD, W78) Aducanumab (10 mg/kg Q4W, P3s EMERGE & ENGAGE, W78) Trontinemab (1.8 mg/kg Q4W, W28, P1/2) Trontinemab (3.6 mg/kg Q4W, W28, P1/2) Gantenerumab (510 mg Q2W, P3s GRADUATE-1 & -2, W116) Targeting the pyroglu-Aβ epitope offers optimal balance of plaque clearance and safety Antibodies targeting pyroglu-Aβ are highlighted
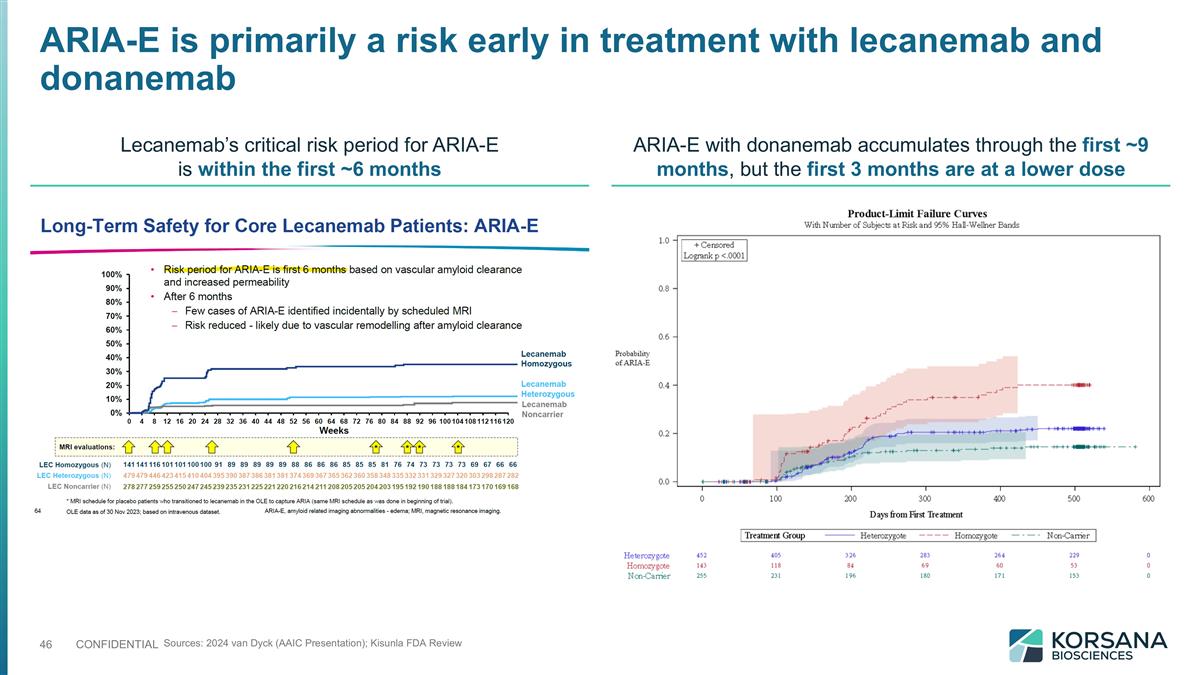
ARIA-E is primarily a risk early in treatment with lecanemab and donanemab Sources: 2024 van Dyck (AAIC Presentation); Kisunla FDA Review Lecanemab’s critical risk period for ARIA-E is within the first ~6 months ARIA-E with donanemab accumulates through the first ~9 months, but the first 3 months are at a lower dose
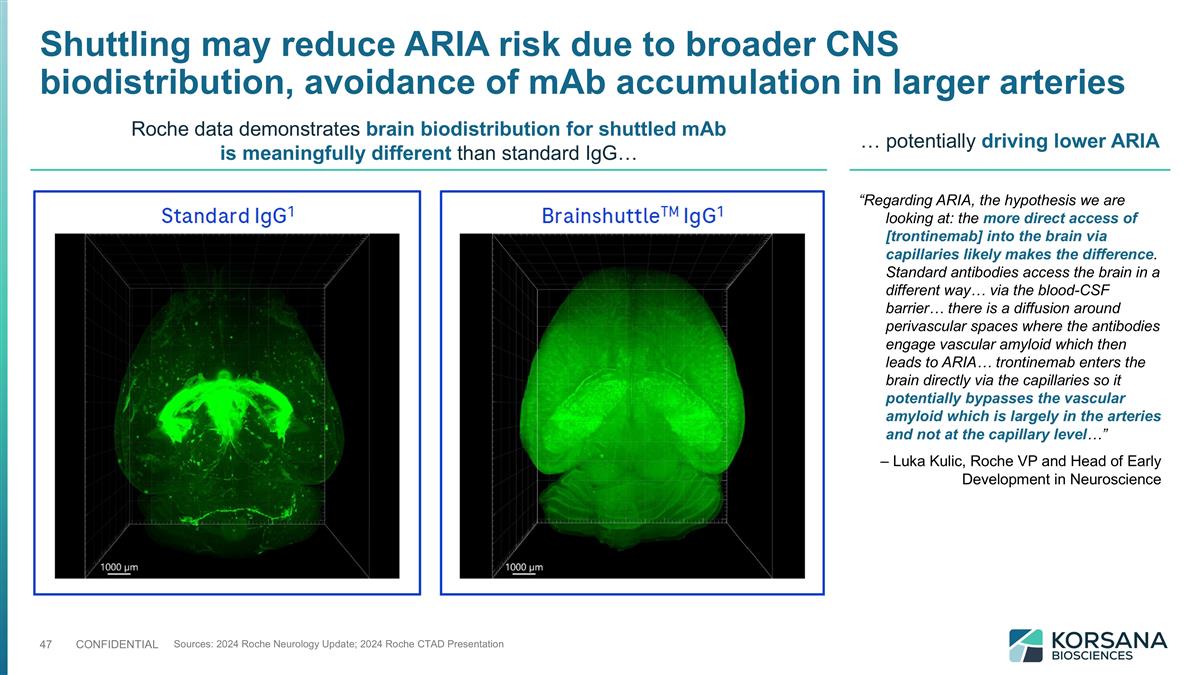
Shuttling may reduce ARIA risk due to broader CNS biodistribution, avoidance of mAb accumulation in larger arteries Sources: 2024 Roche Neurology Update; 2024 Roche CTAD Presentation Roche data demonstrates brain biodistribution for shuttled mAb is meaningfully different than standard IgG… … potentially driving lower ARIA “Regarding ARIA, the hypothesis we are looking at: the more direct access of [trontinemab] into the brain via capillaries likely makes the difference. Standard antibodies access the brain in a different way… via the blood-CSF barrier… there is a diffusion around perivascular spaces where the antibodies engage vascular amyloid which then leads to ARIA… trontinemab enters the brain directly via the capillaries so it potentially bypasses the vascular amyloid which is largely in the arteries and not at the capillary level…” – Luka Kulic, Roche VP and Head of Early Development in Neuroscience
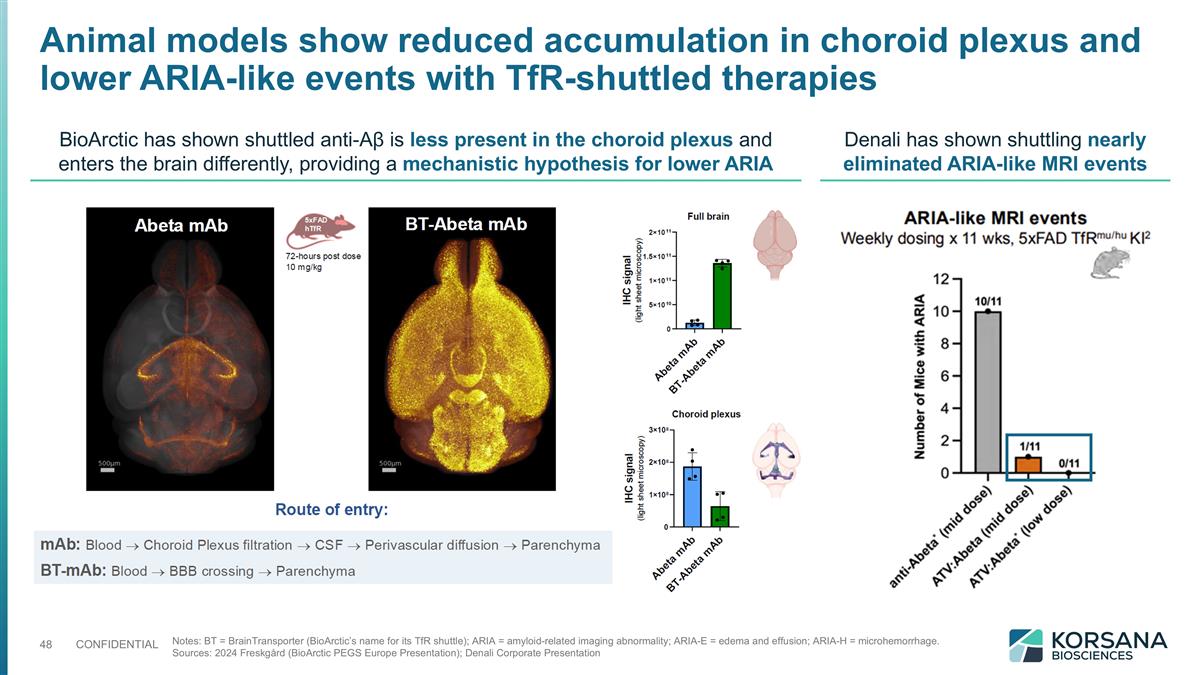
Animal models show reduced accumulation in choroid plexus and lower ARIA-like events with TfR-shuttled therapies Notes: BT = BrainTransporter (BioArctic’s name for its TfR shuttle); ARIA = amyloid-related imaging abnormality; ARIA-E = edema and effusion; ARIA-H = microhemorrhage. Sources: 2024 Freskgård (BioArctic PEGS Europe Presentation); Denali Corporate Presentation BioArctic has shown shuttled anti-Aβ is less present in the choroid plexus and enters the brain differently, providing a mechanistic hypothesis for lower ARIA Denali has shown shuttling nearly eliminated ARIA-like MRI events
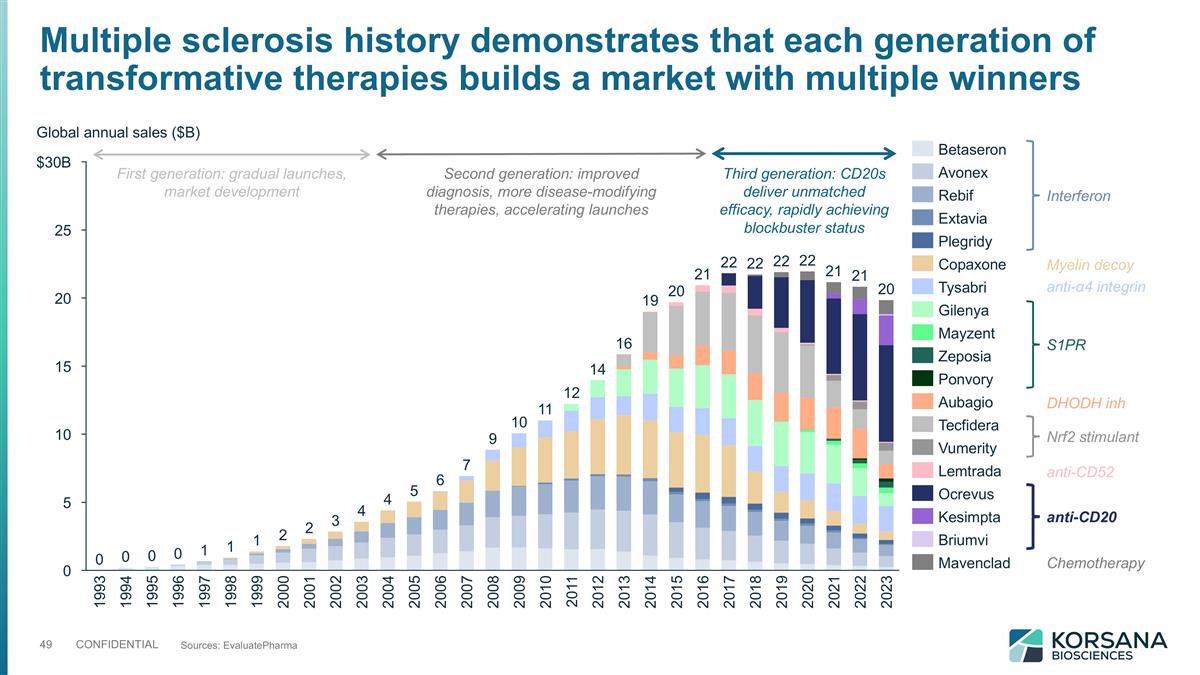
Multiple sclerosis history demonstrates that each generation of transformative therapies builds a market with multiple winners Sources: EvaluatePharma $B Global annual sales ($B) Interferon Myelin decoy anti-α4 integrin S1PR DHODH inh Nrf2 stimulant anti-CD52 anti-CD20 Chemotherapy First generation: gradual launches, market development Second generation: improved diagnosis, more disease-modifying therapies, accelerating launches Third generation: CD20s deliver unmatched efficacy, rapidly achieving blockbuster status
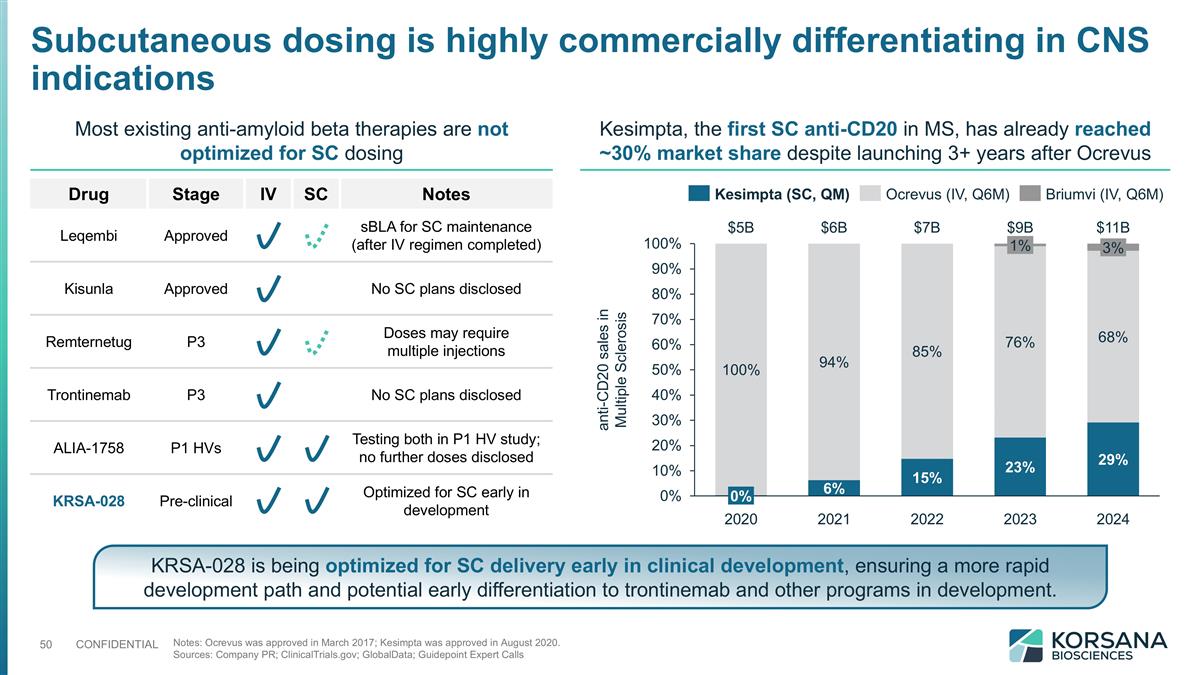
Subcutaneous dosing is highly commercially differentiating in CNS indications Notes: Ocrevus was approved in March 2017; Kesimpta was approved in August 2020. Sources: Company PR; ClinicalTrials.gov; GlobalData; Guidepoint Expert Calls KRSA-028 is being optimized for SC delivery early in clinical development, ensuring a more rapid development path and potential early differentiation to trontinemab and other programs in development. Kesimpta, the first SC anti-CD20 in MS, has already reached ~30% market share despite launching 3+ years after Ocrevus anti-CD20 sales in Multiple Sclerosis $B $B $B $B $B Most existing anti-amyloid beta therapies are not optimized for SC dosing Drug Stage IV SC Notes Leqembi Approved sBLA for SC maintenance (after IV regimen completed) Kisunla Approved No SC plans disclosed Remternetug P3 Doses may require multiple injections Trontinemab P3 No SC plans disclosed ALIA-1758 P1 HVs Testing both in P1 HV study; no further doses disclosed KRSA-028 Pre-clinical Optimized for SC early in development
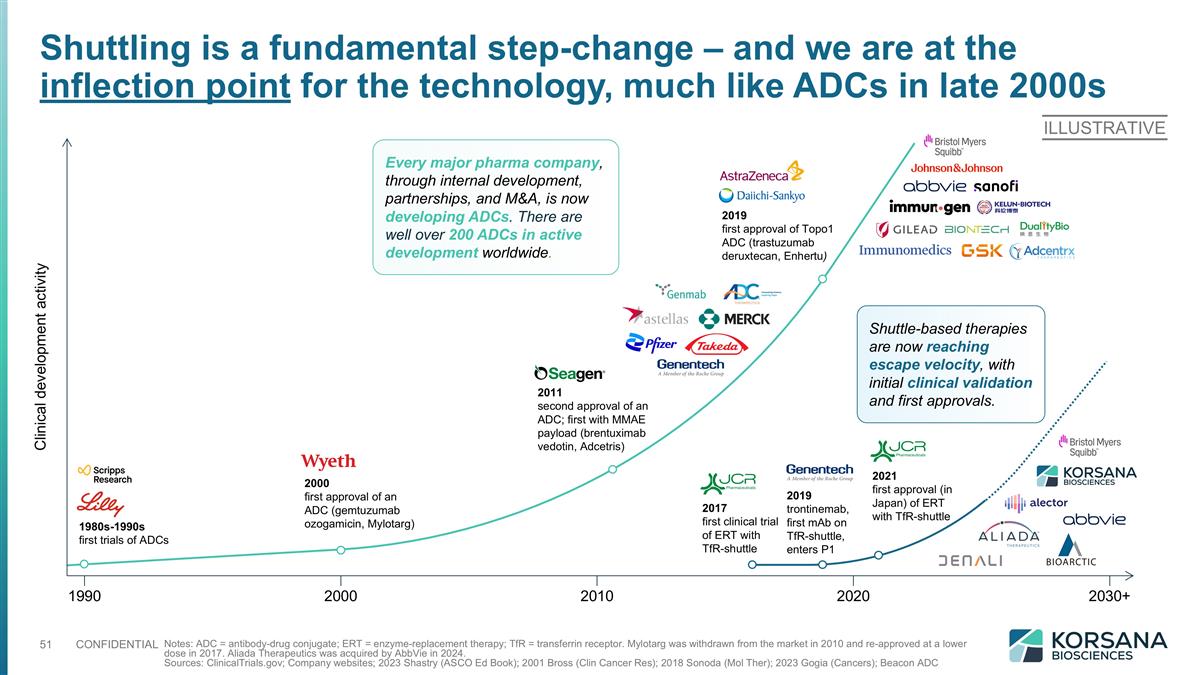
Shuttling is a fundamental step-change – and we are at the inflection point for the technology, much like ADCs in late 2000s Notes: ADC = antibody-drug conjugate; ERT = enzyme-replacement therapy; TfR = transferrin receptor. Mylotarg was withdrawn from the market in 2010 and re-approved at a lower dose in 2017. Aliada Therapeutics was acquired by AbbVie in 2024. Sources: ClinicalTrials.gov; Company websites; 2023 Shastry (ASCO Ed Book); 2001 Bross (Clin Cancer Res); 2018 Sonoda (Mol Ther); 2023 Gogia (Cancers); Beacon ADC 1980s-1990s first trials of ADCs 2000 first approval of an ADC (gemtuzumab ozogamicin, Mylotarg) 2011 second approval of an ADC; first with MMAE payload (brentuximab vedotin, Adcetris) 2019 first approval of Topo1 ADC (trastuzumab deruxtecan, Enhertu) Clinical development activity 1990 2000 2010 2020 2030+ 2017 first clinical trial of ERT with TfR-shuttle 2021 first approval (in Japan) of ERT with TfR-shuttle 2019 trontinemab, first mAb on TfR-shuttle, enters P1 ILLUSTRATIVE Shuttle-based therapies are now reaching escape velocity, with initial clinical validation and first approvals. Every major pharma company, through internal development, partnerships, and M&A, is now developing ADCs. There are well over 200 ADCs in active development worldwide.

Having a shuttle-enabled anti-Aβ, with additional pipeline potential, is rapidly becoming a necessity for neuro drug development Sources: Company websites; Company PR; Patent disclosures Company Active CNS programs Key neurodegeneration programs Shuttling platform Kisunla (anti-Aβ), remternetug (anti-Aβ) ABL partnership; Qinotto partnership; internal efforts Posdinemab (anti-tau mAb) failed Ph2 in AD Multiple TfR & CD98 patents; spun out Aliada Tavapadon (D1/D5 agonist), ABBV-1758 (shuttled anti-Aβ) Acquired Aliada for $1.4B in 2024 Semaglutide (GLP-1 agonist) failed Ph3 in AD Trontinemab (shuttled anti-Aβ), prasinezumab (anti-α-syn) Internally developed; Manifold Bio partnership MEDI1341 (anti-α-syn) MK-1167 (α7nAChR PAM); MK-2214 (anti-tau mAb) NIO752 (tau ASO), VHB937 (TREM2 stabilizer) Avidity $12B; partnered with BioArctic, Arrowhead, etc. ADEL-Y01 (anti-tau mAb) BMS-986446 (anti-tau mAb), BMS-986495 (undiscl. MoA) Partnered with BioArctic (preclinical anti-Aβ program) AL-101 (anti-SORT) Partnered with ABL Bio (platform)