Exhibit 2.1
CERTAIN CONFIDENTIAL INFORMATION CONTAINED IN THIS DOCUMENT, MARKED BY [***], HAS BEEN OMITTED BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) IS THE TYPE THAT LEONABIO, INC. TREATS AS PRIVATE AND CONFIDENTIAL.
LICENSE AGREEMENT
This License Agreement (the “Agreement”) is entered into as of December 18, 2025 (the “Effective Date”) between Sermonix Pharmaceuticals, Inc., a corporation organized under the laws of Delaware, with a place of business at 250 East Broad Street, Suite 250, Columbus, Ohio, 43215 (“Sermonix”) and Athira Pharma, Inc., a corporation organized under the laws of Delaware, with a place of business at 18706 N Creek Pkwy S, Suite 104, Bothell, WA 98011 (“Athira”). In addition, Sermonix and Athira each may be referred to herein individually as a “Party” or collectively as the “Parties.”
RECITALS
Whereas, Sermonix has developed certain oncology products, including that certain selective estrogen-receptor modulator known as Lasofoxifene, and Sermonix owns or controls certain patent, know-how and other intellectual property rights relating to Lasofoxifene;
Whereas, Athira wishes to obtain from Sermonix, and Sermonix is willing to grant to Athira, an exclusive license to develop, manufacture, commercialize and otherwise exploit Sermonix’s owned or controlled rights to Lasofoxifene and products containing Lasofoxifene outside of the Retained Territory, all on the terms and conditions set forth herein; and
Whereas, the Parties have negotiated this Agreement and its terms in good faith, at arms’ length, and for reasonably equivalent value.
Now Therefore, in consideration of the foregoing premises and the mutual covenants contained herein, the receipt and sufficiency of which are hereby acknowledged, the Parties hereby agree as follows:
Article 1
DEFINITIONS
Unless the context otherwise requires, the terms in this Agreement with initial letters capitalized shall have the meanings set forth below:
1.1“Accounting Standards” means U.S. Generally Accepted Accounting Principles (“GAAP”) or the International Financial Reporting Standards (“IFRS”), in each case as then-current at the relevant time and as consistently applied by the applicable Person.
1.2“Additional Clinical Trial” means a material Registrational Trial Initiated by or on behalf of Athira for a Licensed Product following [***].
1.3“Affiliate” means, with respect to a Person, any corporation or other business entity that directly or indirectly controls, is controlled by or is under common control with such Person, in each case, for so long as such control exists. As used in this definition, “control” (and, with
correlative meanings, the terms “controlled by” and “under common control with”) means (a) the direct or indirect beneficial ownership of more than 50% of the voting stock or other ownership interest of, or more than 50% interest in the income of, the applicable entity, or (b) the possession, directly or indirectly, of the power to direct the management or policies of the applicable entity, whether through the ownership of voting securities or other equity rights, by contract relating to voting rights or corporate governance, or otherwise. For the avoidance of doubt, for purposes of this Agreement, [***] and their Affiliates shall not be deemed an Affiliate of either Party, regardless whether such Person meets the criteria set forth in the preceding sentence or not.
1.4“Annual Net Sales” means, [***].
1.5“Applicable Laws” means applicable (with respect to the particular activity, task, or obligation under this Agreement to which such term applies) laws, statutes, rules, regulations, and other pronouncements having the effect of law of any Governmental Authority that may be in effect from time to time, including applicable rules, regulations, guidelines, or other requirements of any Regulatory Authority that may be in effect from time to time, including, where applicable, GCP, GLP and GMP.
1.6“Business Day” means a day other than Saturday, Sunday or any day on which banks located in Seattle, Washington, or Columbus, Ohio, are authorized or obligated to close. Whenever this Agreement refers to a number of days, such number shall refer to calendar days unless Business Days are specified.
1.7“Calendar Quarters” means the respective periods of three consecutive calendar months ending on March 31, June 30, September 30 and December 31, except that the first Calendar Quarter shall commence on the Effective Date and end on the first to occur of March 31, June 30, September 30 or December 31 after the Effective Date, and the last Calendar Quarter shall end on the last day of the Term.
1.8“Calendar Year” means each twelve (12) month period commencing on January 1 and ending on December 31, except that the first Calendar Year shall commence on the Effective Date and end on the first December 31 to occur after the Effective Date, and the last Calendar Year shall end on the last day of the Term.
1.10“Change of Control” means, with respect to a Party, (a) a merger, reorganization, combination or consolidation of such Party (or, if applicable, a parent Affiliate of such Party) with a Third Party that results in the holders of beneficial ownership of the voting securities or other voting interests of such Party (or, if applicable, a parent Affiliate of such Party) immediately prior to such merger, reorganization, combination or consolidation ceasing to hold beneficial ownership of at least fifty percent (50%) of the combined voting power of the surviving entity or the applicable parent of the surviving entity immediately after such merger, reorganization, combination or consolidation; (b) a transaction or series of related transactions in which a Third Party (together with its Affiliates), becomes the beneficial owner of fifty percent (50%) or more of the combined voting power of the outstanding securities or other voting interest of such Party or a parent Affiliate of such Party; or (c) the sale or other transfer (in one transaction or a series of
related transactions) to a Third Party of all or substantially all of such Party’s or its parent Affiliate’s assets to which this Agreement relates.
1.11“Clinical Trial” means any clinical testing of a Licensed Product in human subjects, including any Phase 3 Clinical Trial or Registrational Trial.
1.12“CMO” means any Third Party contract manufacturing organization, contract development and manufacturing organization or contract research organization that conducts any Manufacturing activities, including analysis of pharmaceutical compounds or products.
1.13“Commercialization” means all activities directed to marketing, promotion, detailing, sale or booking of sales, pricing, selling or distributing of any Licensed Product (including importing and exporting activities in connection therewith) or interacting with Regulatory Authorities regarding the foregoing, including all activities directed to obtaining Pricing and Reimbursement Approvals for a Licensed Product. The terms “Commercialize” and “Commercializing” have correlative meanings.
1.14“Commercially Reasonable Efforts” means: [***].
1.15“Committee” means the JSC or any subcommittee established after the Effective Date by the JSC, as applicable.
1.16“Competing Product” means any product that contains, comprises or incorporates any selective estrogen-receptor modulator, in all current and future formulations and in any dosage strengths, presentations or package configuration, and for any mode of administration.
1.17“Confidentiality Agreement” means the Mutual Nondisclosure Agreement between the Parties dated [***].
1.18“Control” or “Controlled” means the possession by a Party or its Affiliate (whether by ownership, license or otherwise, other than pursuant to this Agreement) of, (a) with respect to any materials or other tangible Know-How, the legal authority or right to physical possession of such materials or tangible Know-How, with the right to provide such materials or tangible Know-How to the other Party on the terms set forth herein, and (b) with respect to Patents, Regulatory Material, intangible Know-How, or other intellectual property rights, the legal authority or right to grant a license, sublicense, access, or right to use (as applicable) to the other Party under such Patents, Regulatory Material, intangible Know-How, or other intellectual property rights on the terms set forth herein, in each case ((a) and (b)), without breaching the terms of any agreement with a Third Party.
1.19“Cover” means, (a) with respect to any Know-How and the Licensed Compound or any Licensed Product, that in the absence of a Person having ownership of or a license to such Know-How, the Exploitation of such Licensed Compound or Licensed Product would misappropriate such Know-How or (b) with respect to a Patent and the Licensed Compound or any Licensed Product, that, in the absence of a Person having ownership of or a license to such Patent, the Exploitation of such Licensed Compound or Licensed Product would infringe a claim in such Patent or if such Patent is a patent application, would infringe a claim in such patent application if such patent application were to issue as a patent with such claim.
1.21“Data Safety Monitoring Board” means an independent data monitoring committee of individuals with relevant expertise that may be established to assess the progress of a Clinical Trial, the safety data, and the critical efficacy endpoints, and to recommend to the sponsor of a Clinical Trial whether to continue, modify, or stop such Clinical Trial.
1.22“Debarred/Excluded” means, with respect to a Person, that such Person (a) is debarred or suspended by the FDA pursuant to Section 306 of the FFDCA or is the subject of a conviction described in such section, (b) has been or is excluded from a federal or governmental health care program, debarred from federal contracting, or convicted of or pled nolo contendere to any felony, or to any federal or state legal violation (including misdemeanors) relating to prescription drug products or fraud, in each case in the U.S., (c) is the subject of an FDA debarment investigation or proceeding, or (d) is subject to any similar sanction or similar proceeding of any other Regulatory Authority outside the U.S.
1.23“Development” means all research and development activities for the Licensed Compound or any Licensed Product including (a) research, discovery, process development, pre-clinical studies, toxicology, pharmacokinetic and pharmacological studies, statistical analyses, assay development, protocol design and development, non-clinical activities, IND-enabling studies and Clinical Trials, (b) preparation, submission, review, and development of data or information and regulatory filings for the purpose of submission to a Regulatory Authority to obtain authorization to conduct Clinical Trials or to obtain, support, or maintain MAA Approval of a Licensed Product, and (c) all regulatory affairs related to any of the foregoing. The terms “Develop” and “Developing” have correlative meanings.
1.24“Dollars” or “$” means U.S. dollars, the lawful currency of the U.S.
1.25“Duke License Agreement” means that certain License Agreement dated November 8, 2016, by and between Duke University (“Duke”) and Sermonix (formerly known as Sermonix Pharmaceuticals, LLC), as amended.
1.27“ELAINE 3 Study” means the ongoing Phase 3 Clinical Trial sponsored by Sermonix with ClinicalTrials.gov Identifier: NCT05696626, with sites in the United States, Europe, Canada, Israel and Asia.
1.28“EMA” means the European Medicines Agency or any successor agency thereto.
1.29“European Union” means all countries or territories that are officially members of the European Union, as it may be constituted from time to time.
1.30“Executive Officer” means (a) with respect to Athira, [***] or his or her designee and (b) with respect to Sermonix, [***] or his designee.
1.31“Existing Upstream License Agreements” means the Duke License Agreement, and any other written agreement (other than the Ligand-Sermonix Agreement or the Henlius
Agreement) pursuant to which Sermonix or any of its Affiliates has obtained Control of any Licensed Technology from a Third Party as of the Effective Date. Exhibit 1.31 sets forth a list of all Existing Upstream License Agreements.
1.32“Exploit” means Develop, Manufacture, have Manufactured, use, practice, sell, offer for sale, import, export, Commercialize or otherwise exploit. “Exploitation” has the correlative meaning.
1.33“Ex-Territory Licensee” means any Third Party that has a license or sublicense from Sermonix or its Affiliate to Develop, Manufacture or Commercialize one or more Licensed Products in any country in the Retained Territory or any further (sub)licensee of such Third Party, provided that distributors, wholesalers, contract research organizations, contract sales organizations, CMOs and similar entities engaged on a fee-for-service basis to provide services to Sermonix or its Affiliate shall not be Ex-Territory Licensees for the purpose of this Agreement. As of the Effective Date, Henlius is an Ex-Territory Licensee and there are no other Ex-Territory Licensees.
1.34“FDA” means the U.S. Food and Drug Administration or any successor agency thereto.
1.35“FFDCA” means the United States Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 301 et seq., as amended from time to time, together with any rules, regulations and requirements promulgated thereunder (including all additions, supplements, extensions, and modifications thereto).
1.36“Field” means the treatment, palliation, diagnosis or prevention of any diseases and conditions in humans.
1.37“First Commercial Sale” means, with respect to a Licensed Product in a country in the Territory, the first sale of such Licensed Product to a Third Party in such country by Athira or its Affiliate for distribution, use or consumption in such country following receipt of Regulatory Approval of such Licensed Product in such country, provided that [***].
1.38“GCP” means the then-current good clinical practice standards, practices, and procedures promulgated or endorsed by the applicable Regulatory Authority as set forth in the guidelines imposed by such Regulatory Authority, as may be updated from time-to-time.
1.39“Generic Product” means, with respect to a Licensed Product, and on a country-by-country basis, any product (including a “generic product” or “generic drug”) approved by way of an abbreviated regulatory mechanism by the relevant Regulatory Authority in a country in reference to such Licensed Product, that in each case: (a) is sold in the same country (or is commercially available in the same country via import from another country) as such Licensed Product by [***], and (b) [***], in each case, as is [***], including, with respect to the U.S., a Licensed Product with an approved Abbreviated New Drug Application under Section 505(j) of 21 U.S.C. 355(j), or, outside the United States, in accordance with European Directive 2001/83/EC on the Community Code for medicinal products (Article 10(4) and Section 4, Part II of Annex I) and European Regulation EEC/2309/93 establishing the Community procedures for the authorization and evaluation of medicinal products, each as amended, and together with all
associated guidance, and any counterparts thereof or equivalent process inside or outside of the U.S. or European Union to the foregoing.
1.40“GLP” means the then-current good laboratory practice standards, practices, and procedures promulgated or endorsed by the applicable Regulatory Authority as set forth in the guidelines imposed by such Regulatory Authority, as may be updated from time-to-time.
1.41“GMP” means the then-current good manufacturing practice standards, practices, and procedures promulgated or endorsed by the applicable Regulatory Authority as set forth in the guidelines imposed by such Regulatory Authority, as may be updated from time-to-time.
1.42“Government Official” means any official, officer, employee, or representative of: (a) any federal, state, provincial, administrative division, county, or municipal government or any department or agency thereof; (b) any public international organization or any department or agency thereof; or (c) any company or other entity owned or controlled by any government or Governmental Authority.
1.43“Governmental Authority” means any court, commission, authority, department, ministry, official or other instrumentality of, or being vested with public authority under any law of, any country, region, state or local authority or any political subdivision thereof, or any association of countries.
1.44“Henlius” means Shanghai Henlius Biotech, Inc.
1.45“Henlius Agreement” means that certain Exclusive License Agreement dated January 10, 2024, by and between Henlius and Sermonix, as amended by Amendment No. 1 to Exclusive License Agreement dated June 3, 2024, Amendment No. 2 to Exclusive License Agreement dated June 11, 2025 and Amendment No. 3 to Exclusive License Agreement dated December 18, 2025 (the “Third Henlius Amendment”) and as may be amended from time to time.
1.46“Incremental Henlius Royalties” means all royalties payable to Sermonix pursuant to Section 5.3 of the Henlius Agreement following the Effective Date on account of sales of Licensed Products in any country in the Retained Territory, minus any royalties owed under the Ligand-Sermonix Agreement on account of such Licensed Product sales.
1.47“Initiate” means, with respect to a Clinical Trial, the [***] of the first human subject or patient in such Clinical Trial. “Initiation” means the act of Initiating a Clinical Trial.
1.48“Invention” means any process, method, composition of matter, article of manufacture, improvement, discovery, finding, or other Know-How, patentable or otherwise, that is conceived, developed, invented, discovered, reduced to practice, or otherwise generated (a) solely by or on behalf of either Party, its Affiliates or its or their Affiliates’ employees, agents or independent contractors or (b) jointly by or on behalf of both Parties, their Affiliates or their or their Affiliates’ employees, agents or independent contractors, in each case, during the Term in the performance of any activity under this Agreement or otherwise in the exercise of its (their) rights or carrying out its (their) obligations under this Agreement, including all rights, title and interest in and to the intellectual property rights therein.
1.49“Know-How” means any proprietary scientific or technical information, materials, results and data of any type whatsoever, in any tangible or intangible form whatsoever, including databases, safety information, practices, methods, techniques, specifications, formulations, formulae, knowledge, know-how, skill, experience, test data including pharmacological, medicinal chemistry, biological, chemical, biochemical, toxicological and clinical test data, analytical and quality control data, stability data, studies and procedures, and manufacturing process and development information, results and data.
1.50“Knowledge” means, with respect to a particular fact or matter and Sermonix, [***].
1.51“Licensed Compound” means the selective estrogen receptor modulator known as Lasofoxifene, which has the structure set forth on Exhibit 1.51.
1.52“Licensed Know-How” means all Know-How Controlled by Sermonix or its Affiliates as of the Effective Date or during the Term that (a) Cover the Licensed Compound or any Licensed Product (including the composition of matter, method of using, method of making or formulation thereof) or (b) are necessary or useful to Exploit the Licensed Compound or any Licensed Product in the Field in the Territory. Licensed Know-How includes (i) Sermonix’s interest in any Sole Invention or Joint Invention and (ii) all Know-How licensed to Sermonix or its Affiliates pursuant to the Duke License Agreement or Henlius Agreement.
1.53“Licensed Patents” means all Patents Controlled by Sermonix or its Affiliates as of the Effective Date or during the Term that (a) Cover the Licensed Compound or any Licensed Product (including the composition of matter, method of using, method of making or formulation thereof) or (b) are necessary or useful to Exploit the Licensed Compound or any Licensed Product in the Field in the Territory. Licensed Patents include (i) Sermonix’s interest in any Sermonix Sole Patents or Joint Patents in the Territory; (ii) Sermonix’s interest in any Patents licensed to Sermonix or its Affiliates pursuant to the Duke License Agreement or Henlius Agreement in the Territory; and (iii) Sermonix’s interest in (A) any Patents jointly owned by Sermonix and Lilly arising under the [***] and (B) any Patents jointly owned by Sermonix and the Regents of the University of California arising under the QLHC Agreement. Licensed Patents existing as of the Effective Date (“Existing Licensed Patents”) are set forth in Exhibit 1.53.
1.54“Licensed Product” means any product that contains, comprises or incorporates the Licensed Compound, either alone or in combination with other active or inactive components or ingredients, in all current and future formulations and in any dosages, strengths, presentations or package configuration, and for any mode of administration.
1.55“Licensed Technology” means Licensed Know-How and Licensed Patents.
1.56“Ligand-Athira Agreement” means that certain License Agreement dated December 18, 2025, by and between Ligand Pharmaceuticals Incorporated (“Ligand”) and Athira.
1.57“Ligand-Sermonix Agreement” means that certain Amended and Restated License Agreement dated December 18, 2025, by and between Ligand and Sermonix.
1.59“MAA” or “Marketing Authorization Application” means an application to the appropriate Regulatory Authority for approval (including conditional or accelerated approval) to market a pharmaceutical product in a particular country or jurisdiction and all amendments, supplements and variations thereto or thereof, including any New Drug Application (NDA) in the U.S. and equivalent applications in any country or jurisdiction outside the U.S., but excluding any application for Pricing and Reimbursement Approval.
1.60“MAA Approval” means approval by FDA, EMA or European Commission, or other Regulatory Authority of an MAA for a Licensed Product submitted to such Regulatory Authority by or on behalf of Athira, its Affiliate or Sublicensee, excluding Pricing and Reimbursement Approvals.
1.61“Manufacture” and “Manufacturing” mean activities directed to the synthesis, manufacturing, processing, filling, finishing, packaging, labeling, assembly, quality assurance, quality control, testing and release, post-marketing validation testing, inventory control and management, storing and shipping a Licensed Product, or any component or intermediate thereof, or placebo or comparator agent, as the case may be, including qualification, validation, scale-up, pre-clinical, clinical, and commercial manufacture and analytic development, product characterization, CMC development, and stability testing, but excluding activities directed to Development or Commercialization.
1.62“Net Proceeds” means an amount equal to [***] minus [***]. For clarity, Net Proceeds shall not include [***].
1.63“Net Sales” means [***] less the following deductions, [***]
In the event that a Licensed Product is sold in a package or formulated in combination with one or more other active ingredients that are not Licensed Products (a “Combination Product”), then [***].
1.64“Other Covered Party” means any political party or party official, or any candidate for political office.
1.65“Pass-Through Payments” means any payment owed [***] in each case that is triggered [***]. For clarity, the Pass-Through Payments shall exclude any payments owed [***], in each case that is triggered [***].
1.66“Patents” means (a) all national, regional and international patents and patent applications, including any provisional patent application, (b) any patent application claiming priority from such patent application or provisional patent applications, including divisions, continuations, continuations-in-part, additions, (c) any patent that has issued or in the future issues from any of the foregoing patent applications, including any utility or design patent or certificate of invention, and (d) re-issues, renewals, extensions, substitutions, re-examinations or restorations, registrations and revalidations, and supplementary protection certificates and equivalents to any of the foregoing.
1.67“Person” means any individual, sole proprietorship, corporation, joint venture, limited liability company, partnership, limited partnership, limited liability partnership, trust or any other private, public or governmental entity.
1.68“Phase 3 Clinical Trial” means a Clinical Trial of a Licensed Product that satisfies the requirements of 21 C.F.R. § 312.21(c) (or any amended or successor regulations) or that satisfies the requirements of similar laws or regulations in any applicable country or jurisdiction in the Territory other than the U.S. For clarity, Phase 3 Clinical Trial includes the ELAINE 3 Study.
1.69“Pricing and Reimbursement Approvals” means, with respect to a country in the Territory where the applicable Governmental Authority(ies) approves or determines pricing and/or pricing reimbursement for pharmaceutical or biologic products, the first receipt (or the first publication, if required to make effective) of such pricing and/or pricing reimbursement approval or determination by such Governmental Authority(ies) in such country.
1.70“Prosecution” means, with respect to a Patent, the filing, preparation, prosecution (including conducting all correspondence and interactions with any patent office and seeking, conducting and defending all derivation proceedings, inter partes reviews, post-grant reviews, reissue proceedings, reexaminations, and oppositions and similar proceedings), and maintenance thereof, including obtaining patent term extensions, supplemental protection certificates, or their equivalents with respect thereto. When used as a verb, “Prosecute” and “Prosecuting” mean to engage in Prosecution. “Prosecution,” “Prosecute,” and “Prosecuting” exclude any enforcement action with respect to a Patent.
1.71“QLHC Agreement” means that certain Clinical Trial Participation Agreement dated December 20, 2022 by and between Sermonix and Quantum Leap Health Care Collaborative.
1.72“Registrational Trial” means a Clinical Trial of a Licensed Product in any country: (a) that is a Phase 3 Clinical Trial; or (b) [***].
1.73“Regulatory Approval” means, with respect to a Licensed Product in a country or jurisdiction, all approvals, licenses, registrations or authorizations from the applicable Regulatory Authorities necessary for the Manufacture, distribution, marketing, import, export, or other Commercialization of such Licensed Product in such country or jurisdiction, including any MAA Approval and any Pricing and Reimbursement Approval.
1.74“Regulatory Authority” means any applicable Governmental Authority responsible for granting any Regulatory Approvals for a Licensed Product.
1.75“Regulatory Exclusivity” means, with respect to a Licensed Product in a country in the Territory, the period of time during which: (a) Athira or its Affiliate has been granted the exclusive legal right by a Regulatory Authority (or is otherwise entitled to the exclusive legal right by operation of Applicable Law) in such country to market and sell such Licensed Product, and such right precludes a Third Party from making such Licensed Product available for purchase for any indication; or (b) the data and information submitted by Athira or its Affiliate or Sublicensee to the relevant Regulatory Authority in such country for purposes of obtaining MAA Approval of
such Licensed Product may not be referenced, or relied upon in any way by a Third Party or such Regulatory Authority to support the Regulatory Authority approval or marketing of any Generic Product for such Licensed Product by a Third Party in such country, or if such data and information is referenced, or relied upon to support a Regulatory Authority approval granted to an unrelated Third Party in such country, the Generic Product for such Licensed Product may not be placed on the market for any indication.
1.76“Regulatory Materials” means all applications, submissions, dossiers, notifications, communications, correspondences, registrations, approvals, filings and other documents that are (a) made to, received from or otherwise conducted with a Regulatory Authority in order to Develop, Manufacture, market, sell or otherwise Commercialize or Exploit the Licensed Compound or any Licensed Product in any country or jurisdiction in the Territory and (b) Controlled by Sermonix or its Affiliates as of the Effective Date or during the Term. For clarity, Regulatory Materials include MAAs and Regulatory Approvals.
1.77“Retained Territory” means all countries and regions then included in the definition of “Territory” in Section 1.84 of the Henlius Agreement. As of the Effective Date, the Retained Territory means all countries and regions in geographical Asia that are listed in Exhibit 1.77. If the Henlius Agreement is amended during the Term [***], then [***]. If the Henlius Agreement is terminated during the Term, then [***].
1.78“Retained Territory Know-How” means any and all Know-How Controlled by Sermonix or its Affiliates as of the Effective Date or during the Term that (a) Cover the Licensed Compound or any Licensed Product, or (b) are necessary or useful to Develop, Manufacture and have Manufactured the Licensed Compound or any Licensed Product in the Field in the Retained Territory. Retained Territory Know-How includes (i) Sermonix’s interest in any Sole Invention or Joint Invention and (ii) all Know-How licensed to Sermonix or its Affiliates pursuant to the Duke License Agreement, Ligand-Sermonix Agreement or Henlius Agreement.
1.79“Retained Territory Patents” means any and all Patents in the Retained Territory Controlled by Sermonix or its Affiliates as of the Effective Date or during the Term that (a) Cover the Licensed Compound or any Licensed Product, or (b) are necessary or useful to Develop, Manufacture and have Manufactured the Licensed Compound or any Licensed Product in the Field in the Retained Territory. Retained Territory Patents includes (i) Sermonix’s interest in any Sermonix Sole Patents or Joint Patents in the Retained Territory and (ii) Sermonix’s interest in all Patents licensed to Sermonix or its Affiliates pursuant to the Duke License Agreement, Ligand-Sermonix Agreement or Henlius Agreement in the Retained Territory.
1.80“Retained Territory Technology” means all (a) Retained Territory Patents and (b) Retained Territory Know-How.
1.81“Serious Adverse Event” means an adverse drug experience or circumstance that results in any of the following outcomes: (a) death; (b) life-threatening event; (c) inpatient hospitalization or prolongation of existing hospitalization; (d) persistent or significant disability or incapacity or substantial disruption of the ability to conduct normal life functions; (e) a congenital anomaly/birth defect; (f) significant intervention required to prevent permanent impairment or damage; or (g) a medical event that may not result in death, be life-threatening or require
hospitalization but, based on appropriate medical judgment, that may jeopardize the patient or subject and may require medical or surgical intervention to prevent one of the outcomes described in clauses (a) through (e).
1.82“SPA” means that certain Securities Purchase Agreement, by and between Sermonix and Athira, dated on or around the date hereof.
1.83“Sublicensee” means a Third Party that has been granted a sublicense by Athira under the license granted to Athira pursuant to Section 2.1; provided that distributors, wholesalers, contract research organizations, contract sales organizations, CMOs and similar entities will not be Sublicensees for purposes of this Agreement.
1.84“Terminated Product” means a Licensed Product (and the Licensed Compound incorporated therein) with respect to which [***].
1.85“Terminated Territory” means (a) if this Agreement is terminated in its entirety pursuant to Section 12.2, the Territory as a whole; or (b) if this Agreement is terminated with respect to one (1) or more country(ies) in the Territory (but not in its entirety), the country(ies) with respect to which this Agreement has been terminated pursuant to Section 12.2.
1.86“Territory” means worldwide, excluding the Retained Territory.
1.87“Third Party” means any Person other than Sermonix, Athira or any of their respective Affiliates.
1.88“Trademark” means any word, name, symbol, color, shape, designation or any combination thereof, including any trademark, service mark, trade name, brand name, sub-brand name, domain name, trade dress, product configuration, program name, delivery form name, certification mark, collective mark, logo, tagline, slogan, design or business symbol, that functions as an identifier of source or origin, whether or not registered, and all statutory and common law rights therein and all registrations and applications therefor, together with all goodwill associated with, or symbolized by, any of the foregoing.
1.89“Trademark Assignment Agreement” means that certain Trademark Assignment Agreement, by and between Sermonix and Athira, dated on or around the date hereof.
1.90“U.S.” means United States of America, including all possessions and territories thereof.
1.91“Valid Claim” means, with respect to a particular country, a claim of [***] Licensed Patent (as may be adjusted through a patent term adjustment or extended through supplementary protection certificate or patent term extension or the like) in such country that has not been [***], and that has not been [***].
1.92Additional Definitions. The following table identifies the location of definitions set forth in various Sections of the Agreement:
|
|
Defined Terms |
Section |
AAA |
13.4(a) |
AAA Rules |
13.4(a) |
[***] |
2.7(b) |
Additional Service Provider Agreements |
3.4 |
Advances |
7.6 |
Assumed Liabilities |
7.2 |
Athira Controlled Patents |
8.4(a) |
Athira Indemnitees |
11.2 |
Athira Sole Patents |
8.1(b) |
Audited Party |
7.10 |
Auditing Party |
7.10 |
Auditor |
7.10 |
Claim |
11.1 |
Confidential Information |
9.1 |
Disclosing Party |
9.1 |
Dispute |
13.1 |
Dispute Referral |
13.3 |
[***] |
2.3(e) |
Excluded Matters |
13.1 |
Existing Inventory |
5.1 |
[***] |
10.2(i) |
Existing Service Provider Agreements |
3.4 |
GSDB |
4.6(b) |
Indemnified Party |
11.3 |
Indemnifying Party |
11.3 |
Infringement |
8.6(a) |
Initial Advances |
7.6 |
Initial Know-How Transfer |
3.3 |
Injunctive Relief |
13.5 |
Insolvency Event |
12.2(c) |
Joint Inventions |
8.1(b) |
Joint Patents |
8.1(b) |
Losses |
11.1 |
Manufacturing Technology Transfer |
5.2 |
Milestone Event |
7.3(a) |
Milestone Payment |
7.3(a) |
|
|
Defined Terms |
Section |
Monthly Advances |
7.6 |
Partnering Transaction |
7.7(a) |
Partnering Transaction Agreement |
7.7(a) |
Partnering Transaction Consideration |
7.7(a) |
Product Marks |
8.9 |
PV Agreement |
4.6(a) |
Receiving Party |
9.1 |
Remedial Action |
4.7 |
Representatives |
9.1 |
Royalty Payment |
7.4(a) |
Royalty Report |
7.5 |
Royalty Term |
7.4(b) |
Securities Regulator |
9.3(e) |
Sermonix Indemnitees |
11.1 |
Sermonix Sole Patents |
8.1(b) |
Service Provider Agreements |
3.4 |
Sole Inventions |
8.1(b) |
Subcontractor |
2.8 |
Term |
12.1 |
Third Party Infringement Claim |
8.5 |
Transferred Existing Regulatory Materials |
4.2 |
Article 2
LICENSES
2.1License to Athira. Subject to the terms and conditions of this Agreement, Sermonix hereby grants to Athira an exclusive (even as to Sermonix and its Affiliates), royalty-bearing license, with the right to grant sublicenses through multiple tiers in accordance with Section 2.2, under the Licensed Technology, to Develop, Manufacture and have Manufactured, Commercialize and otherwise Exploit the Licensed Compound and Licensed Products in the Field in the Territory. For clarity, the license granted to Athira under this Section 2.1 shall not include rights to Exploit any active pharmaceutical ingredient that is proprietary to Sermonix and is not the Licensed Compound.
2.2Right to Sublicense. Athira shall have the right to grant sublicenses through multiple tiers under the licenses granted to it under Section 2.1 to its Affiliates and Third Parties, provided that each sublicense to a Sublicensee shall be subject to a written agreement that is consistent with the terms and conditions of this Agreement. Athira shall notify Sermonix within [***] following the grant of a sublicense to a Sublicensee. Notwithstanding any sublicense, Athira shall remain primarily liable for the performance of Athira’s obligations under this Agreement by
any sublicensed Affiliate or Sublicensee, including but not limited to payment obligations set forth herein.
2.3Existing Upstream License Agreements.
(a)Athira acknowledges and agrees that certain rights granted to Athira under this Agreement are Controlled by Sermonix pursuant to the Existing Upstream License Agreements. Sermonix shall not (i) amend, modify, reject, or terminate any Existing Upstream License Agreement in a manner that would reasonably be expected to adversely affect Athira or its rights hereunder or to increase any obligations of Athira in connection therewith, in each case without the prior written consent of Athira, (ii) breach any Existing Upstream License Agreement or (iii) commit or omit to commit any act in a manner that would reasonably be expected to permit the counterparty of such Existing Upstream License Agreement to terminate such Existing Upstream License Agreement or otherwise diminish the scope or exclusivity of the licenses or other rights of Sermonix that are granted to Athira hereunder. Sermonix shall (A) immediately notify Athira in writing of any notice it receives that Sermonix has breached an Existing Upstream License Agreement, (B) keep Athira fully apprised of Sermonix’s plan for curing such breach and the implementation of such plan and (C) permit Athira to cure such breach on Sermonix’s behalf and at Sermonix’s expense.
(b)As between the Parties, Athira shall be solely responsible for [***]. As between the Parties, Sermonix shall be solely responsible for [***]. For clarity, Sermonix shall be solely responsible for [***].
(c)With respect to the Duke License Agreement, Athira [***].
(d)In order to enable Sermonix to comply with its obligation to [***], the Parties shall [***].
(e)The Parties acknowledge and agree that [***].
(a)In consideration for Athira’s payment and other obligations pursuant to this Agreement, Sermonix agrees that (i) all milestones payable to Sermonix pursuant to Section 5.2 of the Henlius Agreement for milestones achieved following the Effective Date are Athira’s property and will be paid by Henlius directly to Athira, and (ii) all Incremental Henlius Royalties are Athira’s property and will be paid by Henlius directly to Athira. For the avoidance of doubt, Sermonix shall retain [***], including any share of Sublicensing Income to which Sermonix may be entitled in the event that Henlius exercises its right to require Sermonix to effect the Buy Back (as such terms are defined in the Henlius Agreement).
(b)Sermonix (i) shall not amend, modify, reject, or terminate the Henlius Agreement in a manner that would reasonably be expected to adversely affect Athira or its rights hereunder or to increase any obligations of Athira in connection therewith, in each case without the prior written consent of Athira, (ii) shall not breach the Henlius Agreement, (iii) commit or omit to commit any act in a manner that would reasonably be expected to permit Henlius to terminate the Henlius Agreement or otherwise diminish the scope or exclusivity of the licenses or
other rights of Sermonix that are granted to Athira hereunder and (iv) shall [***] to cause or require Henlius to comply with its obligations under the Henlius Agreement. Sermonix shall (A) immediately notify Athira in writing of any notice it receives that Sermonix has breached the Henlius Agreement, (B) keep Athira fully apprised of Sermonix’s plan for curing such breach and the implementation of such plan and (C) permit Athira to cure such breach on Sermonix’s behalf and at Sermonix’s expense.
2.5No Other Rights; Retained Rights. Except as expressly set forth herein, no license or other right or interest under any Know-How, Patent or other intellectual property of a Party is granted (by implication or otherwise) to the other Party under this Agreement.
2.6Negative Covenant. Sermonix shall not, and shall cause its Affiliates to not, grant or convey any right to any Third Party (pursuant to a license grant, collaboration or services agreement, option grant, or otherwise) that would be in conflict with, limit the scope of, or otherwise adversely affect the licenses granted by Sermonix to Athira under Section 2.1 of this Agreement.
(a)During the Term, Sermonix will not, and will cause its Affiliates not to, engage in (independently or for or with any Third Party, including through any license or grant of rights to any Third Party) any Development, Manufacture, Commercialization or other Exploitation of (a) any Competing Product in the Territory and (b) any Competing Product (other than a Licensed Product) in the Retained Territory, in each case, other than (i) activities by or on behalf of Athira with the Licensed Compounds and Licensed Products as contemplated under this Agreement and (ii) any activities by or on behalf of Sermonix that are necessary for Sermonix to satisfy its obligations under the Henlius Agreement.
2.8Subcontracting. Each Party may engage one or more Third Party subcontractors to perform services in furtherance of the performance of its obligations or exercise of its rights under this Agreement, including any Third Party contract research organization, contract sales organization, CMO, wholesaler or distributor (including a distributor that is engaged to conduct promotional activities with respect to the Licensed Products on such Party’s behalf and under such Party’s control) (each, a “Subcontractor”); provided that neither Party will engage any Subcontractor that has been Debarred/Excluded and no engagement of any Subcontractor will relieve the engaging Party of its obligations under this Agreement or any liability hereunder. Each agreement pursuant to which a Party engages a Subcontractor will contain obligations of confidentiality and non-use regarding Confidential Information that are consistent with those set forth in Article 9, provided that the duration of confidentiality obligation may be reasonable and customary in the industry for similar agreements and will include terms that are consistent with the intellectual property provisions set forth in this Agreement, subject to exceptions for Subcontractor background intellectual property to the extent reasonable and customary in the industry for similar agreements.
Article 3
DEVELOPMENT
3.1General. As between the Parties, Athira shall (itself or through its Affiliates or Sublicensees) have the sole right, at its sole cost and expense, to conduct all Development of the Licensed Compound and Licensed Products in the Field in the Territory, and Athira shall use Commercially Reasonable Efforts to Develop at least one Licensed Product in the Field in the Territory. As between the Parties, Sermonix shall (itself or through its Affiliates or Ex-Territory Licensee(s)) have the sole right and responsibility, at its sole cost and expense, to conduct all Development of the Licensed Compound and Licensed Products in the Field in the Retained Territory. Each Party will, and Sermonix will cause its Ex-Territory Licensee(s) to, conduct all Development activities for which it is responsible under this Agreement in a good scientific manner, and in compliance with Applicable Law (including GLP, GCP and GMP, as applicable).
3.2Development Records. Each Party will, and Sermonix will cause its Ex-Territory Licensees to, maintain written or electronic records, in sufficient detail, in a good scientific manner, in accordance with Applicable Law (including GLP, GCP, and GMP, as applicable), and appropriate for regulatory and patent purposes, and that are complete and accurate and reflect all Development work performed and results achieved, in each case, by or on behalf of such Party (or in the case of Sermonix, its Ex-Territory Licensees). Each Party shall retain such records for at least [***] after the end of the Term or for such longer period as may be required by Applicable Law.
3.3Know-How Transfer. Promptly after the Effective Date, Sermonix will transfer and disclose to Athira copies of all Licensed Know-How existing as of the Effective Date (“Initial Know-How Transfer”). Thereafter, during the Term, Sermonix will transfer and disclose to Athira any additional Licensed Know-How (to the extent not previously provided) promptly after the generation, receipt, development, making or acquisition of Control of such Licensed Know-How by or on behalf of Sermonix (including its Ex-Territory Licensees). In addition, if at any time Athira or its Affiliates identify any Licensed Know-How that has not been transferred and disclosed by Sermonix in accordance with the preceding two sentences, Sermonix will transfer and disclose such Licensed Know-How to Athira promptly upon Athira’s request for the same. All Licensed Know-How shall be provided to Athira in electronic or other reasonable form [***]. In addition, at Athira’s written request, Sermonix shall provide Athira with reasonable technical assistance to help in the understanding and use of Licensed Know-How in connection with the Development, Commercialization or other Exploitation of Licensed Compounds and Licensed Products in the Field in the Territory, [***].
3.4Service Provider Agreements. Sermonix hereby assigns to Athira all of Sermonix’s right, title and interest in and to the agreements set forth in Exhibit 3.4 (the “Existing Service Provider Agreements”) and Athira agrees to assume Sermonix’s obligations under such agreements to the extent that such obligations arise after the Effective Date and are not due to any breach by Sermonix that occurred prior to the Effective Date. Sermonix shall use Commercially Reasonable Efforts to obtain all Third Party consents prior to the Effective Date that are necessary to assign the Existing Service Provider Agreements to Athira. At Athira’s request during the Term, Sermonix shall either (a) assign to Athira of all of Sermonix’s right, title, and interest in and to any agreement to which Sermonix is a party with a Third Party service provider that is related to the
Development, Manufacture or Commercialization of the Licensed Compound or any Licensed Product (the “Additional Service Provider Agreements” and together with the Existing Service Provider Agreements, the “Service Provider Agreements”), or (b) introduce Athira to such Third Party service provider and facilitate Athira’s entry into a separate agreement with such Third Party service provider; provided that, [***]. As between the Parties, if Athira requests during the Term that Sermonix assign any Additional Service Provider Agreement to Athira, Sermonix shall use Commercially Reasonable Efforts to obtain all Third Party consents promptly following such request that are necessary to assign such Additional Service Provider Agreement to Athira. Prior to assigning any Service Provider Agreement to Athira, Sermonix shall not (i) amend, modify, or terminate any Service Provider Agreement in a manner that would reasonably be expected to adversely affect Athira after such Service Provider Agreement is assigned to Athira or its rights thereunder or to increase any obligations of Athira in connection therewith, in each case without the prior written consent of Athira; (ii) breach any Service Provider Agreement; or (iii) knowingly take any action or fail to take any action that would reasonably be expected to permit the counterparty of such Service Provider Agreement to terminate such Service Provider Agreement or otherwise diminish the scope or other rights of Sermonix that are granted to Athira hereunder. Sermonix shall (A) immediately notify Athira in writing of any notice it receives that Sermonix has breached a Service Provider Agreement, (B) keep Athira fully apprised of Sermonix’s plan for curing such breach and the implementation of such plan and (C) at Athira’s request, permit Athira to cure such breach on Sermonix’s behalf and at Sermonix’s expense.
3.5Development Reports. Within [***] after [***], Athira shall provide Sermonix with a report summarizing [***].
Article 4
REGULATORY
4.1General. Athira shall use Commercially Reasonable Efforts to obtain and maintain Regulatory Approvals for at least one Licensed Product in the Field in the Territory. As between the Parties, Athira will have the sole right, control and decision-making authority over the conduct of all regulatory activities necessary for obtaining and maintaining Regulatory Approvals for the Licensed Products in the Field in the Territory, including control and authority for the content of all Regulatory Materials and all communications and other interactions with Regulatory Authorities relating to Licensed Products in the Field in the Territory, at its sole cost and expense.
4.2Existing Regulatory Material Transfer. As soon as practicable, and in any event within [***], after the Effective Date, unless otherwise agreed by the Parties, Sermonix will assign to Athira all rights, title and interests in and to the INDs and other Regulatory Materials for the Licensed Compound and any Licensed Product in the Territory as set forth on Exhibit 4.2, including without limitation all INDs, MAAs and other Regulatory Materials transferred by Ligand to Sermonix prior to the Effective Date (collectively, the “Transferred Existing Regulatory Materials”) and will transfer to Athira copies (in electronic format unless not available electronically) of all Regulatory Materials Controlled by Sermonix or its Affiliates as of the Effective Date that are necessary to assign to Athira or otherwise included in any Transferred Existing Regulatory Material, along with any associated files, documentation and databases. Prior to such assignment and transfer, Sermonix shall take all actions needed to bring each Transferred Existing Regulatory Material up to date and in full compliance with all applicable Laws, including
by filing all required annual reports, safety reports and clinical study reports. The Parties shall [***] to effectuate the assignments described in this Section 4.2 with any applicable Regulatory Authorities, including duly executing and delivering, or causing to be duly executed and delivered, such instruments (including the filing of such assignments, agreements and documents) as may be necessary in order to effect such assignment of the Transferred Existing Regulatory Materials from Sermonix to Athira.
4.3Regulatory Transfer and Assistance. Upon Athira’s written request, to the extent not previously transferred to Athira (including pursuant to Section 3.3 or 4.2), Sermonix shall (a) [***], provide to Athira all information and documents Controlled by Sermonix or its Affiliates that are necessary or useful for Athira’s regulatory activities relating to Licensed Products in the Field in the Territory, [***], and (b) [***], provide reasonable assistance in connection with Athira’s regulatory activities for the Licensed Products in the Field in the Territory, including assistance with the preparation and submission of MAAs and other Regulatory Materials for MAA Approvals. In addition, upon Athira’s reasonable written request, Sermonix shall participate in (or, if requested by Athira, attend as an observer) any meeting with a Regulatory Authority in the Territory relating to the Licensed Compound or any Licensed Product. [***].
4.4Regulatory Approval Holder; Regulatory Materials. Athira or its Affiliate, Sublicensee or other designee, shall apply for Regulatory Approvals of the Licensed Products in the Field in the Territory in its own name and at its own cost and expense, and Athira or such Affiliate, Sublicensee or designee shall be the named holder of such Regulatory Approvals in the Territory. All Regulatory Materials relating to the Licensed Compound or any Licensed Product with respect to the Territory shall, as between the Parties, be owned by and the sole property of Athira, and shall be held in the name of Athira or its Affiliate, Sublicensee or designee. Sermonix shall assign, and hereby does assign, all of its right, title and interest in and to such Regulatory Materials.
4.5Regulatory Authority Communications. Neither Party (nor any of its Affiliates or (sub)licensees) shall seek Regulatory Approvals or submit any Regulatory Materials for any Licensed Product outside its respective territory. In addition, except as contemplated in Section 4.3 with respect to Sermonix’s participation or attendance at Regulatory Authority meetings, neither Party (nor any of its Affiliates or (sub)licensees) shall communicate with any Regulatory Authority outside its respective territory regarding any Licensed Product, unless so ordered by such Regulatory Authority, in which case (a) if such Regulatory Authority is in the Retained Territory, Athira shall immediately notify Sermonix of such order and (b) if such Regulatory Authority is in the Territory, Sermonix shall immediately notify Athira of such order.
(a)Each Party will cooperate with the other Party with regard to the reporting, handling and exchange of safety information involving any Licensed Product in accordance with Applicable Law and regulatory requirements related to pharmacovigilance and clinical safety. In order to maintain the safety profile of the Licensed Product, the exchange of safety information will need to include data from the Retained Territory. Therefore, at Athira’s request, Sermonix will [***] agreement which will define the pharmacovigilance responsibilities [***] with respect to all Licensed Products, [***] (as may be amended, the “PV Agreement”). The PV Agreement
will describe, [***]. The PV Agreement will be promptly updated if required by changes in legal requirements. If there is a conflict between the terms and conditions of this Agreement and the PV Agreement then the terms of [***] will govern with respect to [***]. Athira will [***] under this Section 4.6.
(b)Athira will own and maintain the global safety database (“GSDB”) for Licensed Products [***]. In order for Athira to maintain a full dataset in the GSDB, safety information from worldwide cases will be required, which will require [***]. Athira will be responsible for all processing of information related to any adverse events for the Licensed Products in the Territory and Henlius will be responsible for all processing of information related to any adverse events for the Licensed Products in the Retained Territory, in each case, in accordance with Applicable Law and regulatory requirements related to pharmacovigilance and clinical safety, including any information regarding such adverse events that is received from a Third Party. Each of Athira and Henlius will [***] related to the Licensed Products. Henlius will [***] for the Retained Territory as [***].
4.7Remedial Actions. Each Party shall notify the other Party immediately, and the other Party shall promptly confirm such notice in writing, if it obtains information indicating that any Licensed Product may be subject to any a recall (whether voluntary or mandated), corrective action, or similar regulatory action by any Regulatory Authority or other Governmental Authority (a “Remedial Action”). At Athira’s request, Sermonix shall assist Athira in gathering and evaluating such information as is necessary to determine the necessity of conducting a Remedial Action in the Territory. As between the Parties, Athira will have the sole discretion and decision-making authority with respect to any matters relating to any Remedial Action in the Territory, including any decision to commence such Remedial Action and the control over such Remedial Action. As between the Parties, Sermonix (or its Affiliate or (sub)licensee) will have the sole discretion and decision-making authority with respect to any matters relating to any Remedial Action in the Retained Territory, including any decision to commence such Remedial Action and the control over such Remedial Action. [***].
Article 5
MANUFACTURE AND SUPPLY
5.1Transfer of Existing Inventory. Effective as of the Effective Date, Sermonix hereby transfers title to Sermonix’s existing inventory of drug substance and drug product for the Licensed Compound and Licensed Products to Athira (“Existing Inventory”), which is currently held by [***] at one or more of its facilities pursuant to the [***] Agreement or by [***] at one or more of its facilities pursuant to the [***] Agreement. Sermonix shall notify [***] in writing of such title transfer within [***] after the Effective Date. Athira agrees to [***]. Sermonix agrees to [***], and shall cooperate with Athira and [***], to ensure that Athira has access following the Effective Date to Sermonix’s Existing Inventory being held by [***] to support the ongoing ELAINE 3 Study under the terms of the [***] Agreement [***]. Sermonix agrees to [***], and shall cooperate with Athira and [***], to ensure that Athira has access following the Effective Date to Sermonix’s Existing Inventory being held by [***] to support the ongoing ELAINE 3 Study under the terms of the [***] Agreement [***]. Except for [***], such Existing Inventory shall be
[***]. Following the transfer of such inventory and [***], Athira shall [***] responsibility [***] for supplying drug substance and drug product for the Licensed Compound and the Licensed Products to Henlius [***] for Henlius’s conduct of the ELAINE 3 Study in the Retained Territory and Athira may retain all amounts received from Henlius with respect to such supply.
5.2Manufacturing Technology Transfer. Upon Athira’s request, [***] Sermonix shall transfer Manufacturing-related records and data and provide Athira with reasonable technical assistance to enable Athira or its designated Third Party contract manufacturer to Manufacture the Licensed Compound and Licensed Products in the Territory (the “Manufacturing Technology Transfer”). Sermonix shall provide Athira with such technical assistance [***]. After the initial Manufacturing Technology Transfer and for the duration of the Term, Sermonix shall provide or make available to Athira (or its Affiliate or designated CMO(s)) as promptly as practicable any additional Manufacturing-related records and data, to the extent that such Manufacturing-related records and data comes to Sermonix’s attention (or is reasonably requested by Athira) and has not previously been provided or made available to Athira.
5.3Manufacture by Athira. After completion of the Manufacturing Technology Transfer, unless otherwise agreed by Sermonix and Athira, Athira shall, either by itself or through its Affiliates, Sublicensees or CMOs, be solely responsible for the Manufacture and supply of all of Athira’s and its Affiliates’ and Sublicensees’ requirements for the Licensed Compound and Licensed Products for Development and Commercialization in the Field in the Territory, [***].
Article 6
COMMERCIALIZATION; MEDICAL AFFAIRS
6.1Responsibilities. After Athira (itself or through its Affiliates or Sublicensees) has obtained Regulatory Approval and [***] for a given Licensed Product in a given country in the Territory, Athira shall use Commercially Reasonable Efforts to Commercialize such Licensed Products in the Field in such country in the Territory. As between the Parties, Athira (itself or through its Affiliates or Sublicensees) will have the sole right, control and decision-making authority over the Commercialization of Licensed Products in the Field in the Territory, at its sole cost and expense, including booking all sales for the Licensed Products in the Territory.
6.2No Diversion. Each of Sermonix and Athira hereby covenants and agrees that: (a) it will not, and will ensure that its Affiliates and its Sublicensees (and, with respect to Sermonix, its Ex-Territory Licensees) will not, directly or indirectly, promote, market, distribute, import, sell or have sold any Licensed Product, including via internet or mail order, outside its respective territory; (b) with respect to any country or region outside its territory, it will not, and will ensure that its Affiliates and its Sublicensees (and, with respect to Sermonix, its Ex-Territory Licensees) will not (i) unless otherwise agreed by the Parties in writing, establish or maintain any branch, warehouse or distribution facility for any Licensed Product in such country or region, (ii) engage in any advertising or promotional activities relating to any Licensed Product that are directed primarily to customers or other purchasers or users of such Licensed Product located in such country or region, (iii) solicit orders for any Licensed Products from any prospective purchaser located in such country or region, or (iv) sell or distribute any Licensed Product to any Person in such Party’s territory who, to such Party’s knowledge, intends to sell or has in the past sold any Licensed Product in any country or region outside such Party’s territory; (c) if a Party receives any
order for any Licensed Products from a prospective purchaser reasonably believed to be located in a region or country outside its territory, then such Party will promptly refer that order to the other Party, and such Party will not accept any such orders; (d) it will not deliver or tender (or cause to be delivered or tendered) any Licensed Products into a country or region outside its territory; and (e) it will not, and will ensure that its Affiliates and its Sublicensees (or, with respect to Sermonix, its Ex-Territory Licensees) will not, knowingly restrict or impede in any manner the other Party’s exercise of its exclusive rights to Commercialize any Licensed Products in the other Party’s territory. Notwithstanding the foregoing, the terms of this Section 6.2 shall not restrict either Party’s (or their Affiliates’ or Sublicensee(s)’) Development or Manufacturing (including packaging and labeling) rights with respect to Licensed Products under this Agreement.
6.3Medical Affairs. Each Party will be solely responsible, [***], for conducting medical affairs activities with respect to Licensed Products in its respective territory, including communications with key opinion leaders, medical education, symposia, advisory boards (to the extent related to medical affairs or clinical guidance), publications, congress presentations and posters, published manuscripts, activities performed in connection with patient registries and post-approval trials, and other medical programs and communications, including educational grants, research grants (including conducting investigator-initiated studies), and charitable donations to the extent related to medical affairs and do not involve the promotion, marketing, sale or other Commercialization of Licensed Products, and each Party will conduct such medical affairs activities in accordance with Applicable Law.
Article 7
PAYMENTS AND MILESTONES
7.1Upfront Issuance. In consideration of the rights and licenses granted to Athira hereunder, Athira shall issue certain securities of Athira to Sermonix in accordance with the SPA.
7.2Assumed Liabilities. In consideration of the rights and licenses granted to Athira hereunder, on the Effective Date, Athira hereby assumes, and agrees to pay and discharge or otherwise extinguish, those liabilities of Sermonix existing as of the Effective Date that are listed on Exhibit 7.2 (collectively, the “Assumed Liabilities”); provided, however, that [***]. The Parties acknowledge and agree that the total of Assumed Liabilities is $16,815,728.00, [***] prior to the Effective Date, and in no event will Athira be obligated to pay (a) to any Third Party listed on Exhibit 7.2 more than the amount listed on Exhibit 7.2 for such Third Party, (b) any amount to any Person not listed on Exhibit 7.2 (except as otherwise agreed by Athira with respect to contracts assigned by Sermonix to Athira in accordance with Section 3.4) or (c) an aggregate of [***] after the Effective Date pursuant to this Section 7.2.
(a)Milestone Payments. In consideration of the rights and licenses granted to Athira hereunder, Athira shall pay to Sermonix each of the following one-time, non-refundable milestone payments (each, a “Milestone Payment”) for the first achievement of the corresponding events set forth below by Athira or its Affiliate prior to consummation of a Partnering Transaction for a Licensed Product in the Territory (each such event, a “Milestone Event”).
|
|
Milestone Events |
Milestone Payments (in Dollars) |
1) First Commercial Sale in the United States for the first Licensed Product |
$50,000,000 |
[***] |
[***] |
[***] |
[***] |
Total |
$100,000,000 |
(b)Achievement of Milestone Events. Each Milestone Payment shall be payable only once upon the first achievement of the corresponding Milestone Event by Athira or its Affiliate prior to consummation of a Partnering Transaction, and no amounts shall be due for subsequent or repeated achievements of any Milestone Event. The maximum of all Milestone Payments payable by Athira to Sermonix pursuant to this Section 7.3 shall not exceed $100,000,000. Athira shall give Sermonix written notice of the achievement of each Milestone Event by Athira or its Affiliate prior to consummation of a Partnering Transaction no later than [***] following such achievement of such Milestone Event. Following receipt of such notice, Sermonix shall submit an invoice to Athira for the applicable Milestone Payment payable under this Section 7.3 and Athira shall pay such Milestone Payment within [***] after Athira’s receipt of such invoice; provided that solely with respect to the Milestone Payment for Milestone Event #1 set forth above, (i) Athira may, at its election and in its sole discretion, pay such Milestone Payment in cash or by a combination of cash and issuance of equity in Athira, wherein [***]; (ii) Athira will notify Sermonix of its decision to pay in cash or a combination of cash and equity no later than [***]; and (iii) if Athira elects to pay such Milestone Payment in a combination of cash and equity, then the Parties shall negotiate and execute [***] an agreement documenting the issuance of the applicable amount of such equity by Athira to Sermonix [***] and such agreement shall include [***]. No payments will be owed pursuant to this Section 7.3 for any Milestone Event achieved after consummation of a Partnering Transaction.
(a)Royalty Rates. In consideration of the rights and licenses granted to Athira hereunder, subject to Section 7.4(b) and Section 7.4(c), Athira shall pay to Sermonix tiered royalties calculated by multiplying the applicable royalty rate set forth in the table below by the corresponding amount of incremental, aggregated Annual Net Sales of all Licensed Products sold, during the Royalty Term and prior to consummation of a Partnering Transaction, by Athira or its Affiliate in the Territory in a Calendar Year (a “Royalty Payment”).
|
|
Portion of Annual Net Sales of all Licensed Products in the Territory in a given Calendar Year (in Dollars) |
Royalty Rate |
|
[***] |
|
[***] |
|
[***] |
(b)Royalty Term. The Royalty Payments will be payable on a Licensed Product-by-Licensed Product basis and country-by-country basis in the Territory from the First Commercial Sale of such Licensed Product in such country until the latest to occur of: (i) the expiration of the last-to-expire Valid Claim in the Licensed Patents in such country that Covers such Licensed Product in such country; (ii) the expiration of the last-to-expire Regulatory Exclusivity for such Licensed Product in such country; and (iii) the tenth (10th) anniversary of the date of such First Commercial Sale (the “Royalty Term”); provided, however, that no Royalty Payments will be owed pursuant to this Section 7.4 for any Licensed Products sold by a Sublicensee after consummation of a Partnering Transaction. After the end of the Royalty Term for a given Licensed Product in a given country in the Territory, Net Sales of such Licensed Product in such country shall be excluded from the calculation of Annual Net Sales for purposes of calculating the Royalty Payment due hereunder.
(i)No Valid Claim. Subject to Section 7.4(c)(iv), on a country-by-country and Licensed Product-by-Licensed Product basis in the Territory, if during any Calendar Quarter during the Royalty Term for such Licensed Product in such country, no Valid Claim of any Licensed Patent exists that [***], then for the remainder of the Royalty Term for such Licensed Product in such country (or if sooner, until the consummation of a Partnering Transaction), the royalty rates set forth in Section 7.4(a) shall be reduced by [***] for such Licensed Product in such country.
(ii)Generic Approval. Subject to Section 7.4(c)(iv), on a country-by-country and Licensed Product-by-Licensed Product basis in the Territory, if in any [***] during the Royalty Term for such Licensed Product in such country, a Generic Product of such Licensed Product [***], then the royalty rates set forth in Section 7.4(a) shall be reduced by [***] for all [***] thereafter for the remainder of the Royalty Term for such Licensed Product (or if sooner, until the consummation of a Partnering Transaction).
(iii)Third Party Payments. Subject to Section 7.4(c)(iv), if Athira or its Affiliate determines, on a Licensed Product-by-Licensed Product and country-by-country basis in the Territory, that it is [***] to acquire rights under a Third Party’s Patents or Know-How through a license, acquisition or other agreement with such Third Party, including, where applicable, in connection with any settlement of a Third Party Infringement Claim, in order to Exploit such Licensed Product in such country, then Athira may credit [***] of [***] made by Athira or its Affiliate to such Third Party (including [***]) in a given [***] against the Royalty Payments under Section 7.4(a) with respect to such Licensed Product in such country in such [***].
(iv)Royalty Floor. With respect to any Licensed Product, country, and [***], the operation of Sections 7.4(c)(i) through 7.4(c)(iii), individually or in combination, will not reduce the Royalty Payments owed to Sermonix by more than [***] of the Royalty Payments otherwise payable to Sermonix under Section 7.4(a) with respect to such Licensed Product in such country during such Calendar Quarter. Notwithstanding the foregoing, Athira may carry forward amounts it could not deduct pursuant to Sections 7.4(c)(i) through 7.4(c)(iii) in any Calendar Quarter as a result of the foregoing floor and may apply such amounts against Royalty Payments or Partnering Transaction Consideration owed in any subsequent Calendar Quarter.
7.5Royalty Report and Payment. Within [***] after the end of each Calendar Quarter during the applicable Royalty Term prior to consummation of a Partnering Transaction, Athira will provide to Sermonix a written report (each, a “Royalty Report”) setting forth, on a Licensed Product-by-Licensed Product basis (a) [***], (b) [***] Net Sales [***], (c) [***] royalty rates [***], (d) [***], and (e) the Royalty Payments due under Section 7.4. Following receipt of each Royalty Report, Sermonix shall send Athira an invoice for the royalties payable and, within [***] after receipt of such invoice, Athira shall pay the Royalty Payments due hereunder for the [***] covered by the applicable Royalty Report. The information contained in each Royalty Report shall be the Confidential Information of Athira.
7.6Advances. As of the Effective Date, Athira has provided to Sermonix advances in an aggregate amount of $311,531.43, which shall be treated as an advance on Milestone Payments, Royalty Payments and Partnering Transaction Consideration owed under this Agreement (the “Initial Advances”). Following the Effective Date, Athira shall provide to Sermonix monthly advances, which shall be treated as an advance on Milestone Payments, Royalty Payments and Partnering Transaction Consideration owed under this Agreement (each such monthly payment, a “Monthly Advance” and all Monthly Advances together with the Initial Advances, the “Advances”). The Monthly Advance amount shall initially be $75,000. At [***], but not more than [***], the Parties shall negotiate in good faith to determine whether any adjustment should be made to the Monthly Advance amount [***]. The first Monthly Advance shall be paid on the Effective Date, pro rated for the portion of the calendar month remaining following the Effective Date, and each subsequent Monthly Advance shall be paid on [***] of each calendar month during the Term. All Advances shall be creditable against Milestone Payments payable to Sermonix under Section 7.3, Royalty Payments payable to Sermonix under Section 7.4 and Partnering Transaction Consideration under Section 7.7. Athira’s obligation to make Advances shall end on the earlier of (a) Sermonix’s first receipt of any Milestone Payment, Royalty Payment or Partnering Transaction Consideration, whether in whole or in part, after deduction of all Advances previously paid to Sermonix and after application of any offset to which Athira is entitled pursuant to Section 7.3(c) or (b) expiration or termination of this Agreement.
7.7Partnering Transaction Consideration.
(a)As further consideration for the rights and licenses granted to Athira hereunder, if Athira enters into a definitive agreement pursuant to which Athira sells or transfers to a Third Party all or substantially all of Athira’s assets relating to the Licensed Product and associated business, or grants a sublicense of a license set forth in Section 2.1 (other than [***] ) (such transaction, a “Partnering Transaction” and such agreement, a “Partnering Transaction Agreement”), Athira shall pay to Sermonix non-refundable payments equal to the percentage of Net Proceeds set forth in the table below (“Partnering Transaction Consideration”) based on [***], in each case in accordance with the following provisions of this Section 7.7. For clarity, a Partnering Transaction excludes [***], no Partnering Transaction Consideration will be due for a [***], and consummation of a Partnering Transaction shall extinguish all of Athira’s payment obligations pursuant to Sections 7.3 through Section 7.6.
(i)Consummation of the Partnering Transaction prior to [***]:
|
|
Portion of Net Proceeds in the Partnering Transaction |
Percentage of Net Proceeds |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
(ii)Consummation of the Partnering Transaction after [***] but prior to the [***]:
|
|
Portion of Net Proceeds in the Partnering Transaction |
Percentage of Net Proceeds |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
(iii)Consummation of the Partnering Transaction after [***]:
|
|
Portion of Net Proceeds in the Partnering Transaction |
Percentage of Net Proceeds |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
(b)Athira shall notify Sermonix of Athira’s entry into a Partnering Transaction Agreement within [***] after such entry. Within [***] after the end of each [***] after consummation of a Partnering Transaction, Athira will provide Sermonix with a report specifying the amount of Net Proceeds (if any) for such Calendar Quarter and the Partnering Transaction Consideration due to Sermonix on such Net Proceeds. Sermonix will submit an invoice to Athira for such Partnering Transaction Consideration within [***] of the date of such report. Athira shall pay such Partnering Transaction Consideration within [***] after Athira’s receipt of such invoice therefor from Sermonix.
7.9Rights and Obligations under Henlius Agreement. Athira shall own and receive certain payments from Henlius, each as further described in Section 2.4(a).
7.10Financial Records and Audits. Each Party shall maintain complete and accurate books and records in sufficient detail in relation to this Agreement to permit the other Party to confirm the accuracy of the amount of royalties and other payments due under this Agreement. Each Party will keep such books and records for at least [***] following the Calendar Year to which they pertain. Upon at least [***] prior written notice, each Party (the “Auditing Party”) shall have the right to inspect and audit such books and records of the other Party (the “Audited Party”) during regular business hours at such place or places where such records are customarily kept, by an independent certified public accounting firm of internationally-recognized standing (the “Auditor”) selected by the Auditing Party and reasonably acceptable to the Audited Party for the sole purpose of verifying for the Auditing Party the accuracy of the financial reports, statements or invoices furnished by the Audited Party pursuant to this Agreement or of any payments made to or charged by the Audited Party pursuant to this Agreement. Before beginning its audit, the Auditor shall execute a written agreement acceptable to Audited Party by which the Auditor agrees to keep confidential all information reviewed during the audit. Such audits may occur no more often than [***] and not more frequently than [***]. Each Auditing Party shall only be entitled to audit the Audited Party’s books and records from the [***] prior to the Calendar Year in which the audit request is made. The Auditor shall not disclose the Audited Party’s Confidential Information to the Auditing Party, except to the extent such disclosure is necessary to verify the accuracy of the financial reports, statements or invoices furnished by the Audited Party or the amount of payments made to or charged by the Audited Party under this Agreement. If the final result of the audit reveals an undisputed underpayment or overpayment, the underpaid or overpaid amount shall be settled within [***] after the Auditor’s report. The Auditing Party shall bear the full cost of such audit unless such audit reveals an overpayment to, or an underpayment by, the Audited Party that resulted from a discrepancy in the financial report, statement or invoice provided by the Audited Party for the audited period, which underpayment or overpayment was more than [***] of the amount set forth in such report, in which case the Audited Party shall reimburse the Auditing Party for the costs for such audit. The financial reports, statements or invoices furnished by the Audited Party and the payments made to or charged by the Audited Party pursuant to this Section 7.10 shall be binding and conclusive upon both Parties, and the Audited Party shall be released from any further liability or accountability with respect thereto. The Auditing Party shall treat all financial information subject to review under this Section 7.10 in accordance with the confidentiality and non-use provisions of Article 9.
7.11Currency; Exchange Rate; Manner of Payment. All payments to be made under this Agreement shall be payable in U.S. Dollars and all references to Dollars and “$” herein shall refer to U.S. Dollars. When conversion of Net Sales or any other amounts from any currency other than U.S. Dollars is required, such conversion shall be calculated using [***]. All payments owed under this Agreement shall be made by wire transfer in immediately available funds to a bank account designated in writing by the payee Party.
7.12Taxes. Each Party shall be solely responsible for the payment of all taxes imposed on its income arising directly or indirectly from the activities of the Parties under this Agreement. The Parties agree to cooperate with one another and use reasonable efforts to avoid or reduce tax withholding or similar obligations in respect of the payments made under this Agreement. The amounts payable pursuant to this Agreement shall not be reduced on account of any withholding taxes unless required by Applicable Law. To the extent Athira is required by Applicable Laws to deduct and withhold taxes on any payment to Sermonix, Athira shall pay such taxes to the proper
Governmental Authority in a timely manner and promptly transmit to Sermonix an official tax certificate or other evidence of such payment. Athira shall have the right to deduct any such tax, levy or charge required to be withheld from a payment due to Sermonix. Any taxes withheld from a payment due to Sermonix and paid to the proper Governmental Authority shall be deemed to have been paid to Sermonix for purposes of this Agreement, in full satisfaction of Athira’s obligation with respect to such amounts. Sermonix shall provide Athira any tax forms that may be reasonably necessary in order for Athira to not withhold tax or to withhold tax at a reduced rate under an applicable bilateral income tax treaty, to the extent legally able to do so. Each Party agrees to use commercially reasonable efforts to assist the other Party in claiming exemption from such deductions or withholdings under double taxation or similar agreement or treaty from time to time in force and in minimizing the amount required to be so withheld or deducted.
Article 8
INTELLECTUAL PROPERTY
(a)As between the Parties, each Party will retain all rights, title, and interests in and to all Know-How, Patents, and other intellectual property rights Controlled by such Party as of the Effective Date or that come into the Control of such Party outside of the performance of activities under this Agreement.
(b)Ownership of all Inventions will be allocated based on inventorship, as determined in accordance with the rules of inventorship under U.S. patent laws. As between the Parties, each Party will own all Inventions that are invented, discovered, generated, conceived, reduced to practice or made solely by or on behalf of it, its Affiliates, or its or its Affiliates’ employees, agents or independent contractors (“Sole Inventions”). The Parties will jointly own all Inventions that are invented, discovered, generated, conceived, reduced to practice or made jointly by a Party (including its Affiliate, or its or its Affiliate’s employees, agents or independent contractors) together with the other Party (including such other Party’s Affiliates, or its or its Affiliate’s employees, agents or independent contractors) (“Joint Inventions”). Patents claiming any (i) Sole Invention owned by Athira shall be referred to as “Athira Sole Patents”; (ii) Sole Invention owned by Sermonix shall be referred to as “Sermonix Sole Patents”, and (iii) Joint Inventions shall be referred to as “Joint Patents.” Each Party will own an undivided equal interest in the Joint Inventions and Joint Patents, without a duty of accounting or an obligation to seek consent from the other Party for the exploitation or license of the Joint Inventions or Joint Patents (subject to the licenses granted to Athira under this Agreement), and each Party hereby waives any right it may have under the laws of any jurisdiction to require any such accounting or consent.
8.2Disclosure of Inventions. Sermonix will promptly disclose to Athira all Inventions to the extent pertaining to or relating to the Licensed Compound, the Licensed Product, or any Competing Product (including any Invention relating to the Development or Manufacturing thereof), including all invention disclosure or other similar documents submitted to Sermonix by its or its Affiliates’ employees, agents, or independent contractors relating to such Inventions, and will also promptly respond to reasonable requests from Athira for additional information relating to such Inventions. Sermonix will maintain customary documentation relating to each such Invention, including all laboratory notebooks and researcher’s reports relating thereto.
8.3Assignment; Further Actions. Each Party shall (and shall cause all its Affiliates and (sub)licensees (including in the case of Sermonix, its Ex-Territory Licensees) to) cause all of its employees, agents and independent contractors who perform Development or Manufacturing activities for the Licensed Compound or any Licensed Product to be under an obligation to assign to such Party (or to its Affiliates or (sub)licensees (including in the case of Sermonix, its Ex-Territory Licensees) for further assignment to such Party) all of their rights in and to any Inventions invented, discovered, made, developed, generated, conceived or reduced to practice by such employee, agent or independent contractor in the course of performing such Development or Manufacturing activities.
(a)Athira Controlled Patents. [***], as between the Parties, Athira shall [***] Prosecute all (i) Licensed Patents, including any Joint Patents in the Territory and (ii) Retained Territory Patents, including any Joint Patents in the Retained Territory (collectively, the “Athira Controlled Patents”) [***]. Athira will (A) keep Sermonix reasonably informed of all progress with regard to the Prosecution of the Athira Controlled Patents; (B) provide to Sermonix copies of all material patent office submissions and correspondence with respect to the Athira Controlled Patents; and (C) provide to Sermonix copies of drafts of all material patent office submissions and correspondence with respect to the Athira Controlled Patents in advance of submitting such submissions and correspondence [***].
(b)Sermonix Step-in Rights. In the event that Athira intends to abandon or cease the Prosecution of any Athira Controlled Patent, Athira will provide reasonable prior written notice to Sermonix of such intention to abandon or cease such Prosecution (which notice will be given no later than [***] prior to the next deadline for any action that must be taken with respect to any such Athira Controlled Patent in the relevant patent office). In such case, at Sermonix’s request, Sermonix shall have the right, but not the obligation, to assume responsibility for Prosecution of such Athira Controlled Patent. Upon such assumption, Sermonix shall control the Prosecution of such Athira Controlled Patent subject to the same terms and conditions set forth in this Section 8.4(b), as applicable to Athira, [***].
(c)Cooperation. Each Party will cooperate with the other Party in connection with all activities relating to the Prosecution of the Licensed Patents, Joint Patents and Retained Territory Patents undertaken by such other Party pursuant to this Section 8.4, including: (i) executing all papers and instruments, or requiring its employees or contractors, to execute such papers and instruments, to enable the other Party to Prosecute the Licensed Patents, Joint Patents or Retained Territory Patents as permitted by this Section 8.4; and (ii) promptly informing the other Party of any matters coming to such Party’s attention that may materially affect the Prosecution of any Licensed Patent, Joint Patent or Retained Territory Patent. Each Party will also promptly provide to the other Party all information reasonably requested by such other Party with regard to such Party’s activities pursuant to this Section 8.4.
8.5Defense of Third Party Infringement Claims. If any Party or its Affiliates or (sub)licensees becomes the subject of a Third Party’s claim or assertion that the Exploitation of the Licensed Compound or any Licensed Product infringes one or more Patents of such Third Party (each, a “Third Party Infringement Claim”), the Party first becoming aware of the Third Party
Infringement Claim will promptly notify the other Party in writing. As between the Parties, Sermonix will have [***] to control the defense of any Third Party Infringement Claim involving alleged infringement of a Third Party Patent by Sermonix’s or its Affiliates’ or (sub)licensees’ activities and Athira will have [***]. As between the Parties, Athira will have [***] to control the defense of any Third Party Infringement Claim involving alleged infringement of a Third Party Patent by Athira’s or its Affiliates’ or Sublicensees’ activities [***], and Sermonix will have [***]. Neither Party will enter into any settlement of any Third Party Infringement Claim: (a) in a manner that would [***]; or (b) that would [***]. This Section 8.5 shall not limit or exclude either Party’s rights to indemnification pursuant to Article 11.
(a)Notice. Each Party will promptly notify the other Party in writing of (i) any known, suspected, alleged or threatened Third Party infringement of any Licensed Patent of which it becomes aware (“Infringement”) or (ii) any action or threatened action contesting the scope, validity or enforceability of, or seeking a determination of non‑infringement of, a Licensed Patent (other than a Joint Patent) in the Territory, or a Joint Patent worldwide, including a Third Party counterclaim brought in an action or proceeding regarding an Infringement of which it becomes aware.
(b)Athira Controlled Patents.
(i)[***], as between the Parties, Athira will have the first right, but not the obligation, to bring and control any other action or proceeding regarding any alleged or threatened Infringement or claim of invalidity or unenforceability of any Athira Controlled Patent, or to defend against any challenge to any of the foregoing, at its own expense and by counsel of its own choice. Athira will keep Sermonix reasonably informed of the status and progress of such action or proceeding. In addition, Athira shall provide Sermonix with drafts of all material papers to be filed with the court or patent office, as applicable, in connection with such action or proceeding to the extent permitted by Applicable Law or any protective or confidentiality order entered by such court or patent office (excluding any information that is confidential to a Third Party), and shall [***]. Sermonix shall [***].
(ii)If (A) Athira elects not to commence an action or proceeding that Athira has the first right to bring and control pursuant to Section 8.6(b)(i), or settle or otherwise secure the abatement of such Infringement, or (B) Athira fails to commence any such action or proceeding pursuant to Section 8.6(b)(i) within [***], then Sermonix may bring and control any such action or proceeding, [***]. In such event, Sermonix will keep Athira reasonably informed of the status and progress of such action or proceeding. In addition, Sermonix shall provide Athira with drafts of all material papers to be filed with the court or patent office (excluding any information that is confidential to a Third Party), as applicable, to the extent permitted by Applicable Law or any protective or confidentiality order entered by such court or patent office, and shall [***]. Athira shall [***].
(c)Cooperation. In the event a Party brings an infringement action in accordance with Section 8.6(b), the other Party will cooperate fully, at the enforcing Party’s request and expense, including, if required to bring such action, the furnishing of a power of
attorney or being named as a party. The enforcing Party will not enter into any settlement or compromise of any action under this Section 8.6: (i) in a manner that would [***]; (ii) that would [***]; or (iii) that would [***].
(d)Recoveries. Except as otherwise agreed by the Parties in connection with a cost-sharing arrangement, any recovery as a result of any action or proceeding pursuant to Section 8.6, whether by way of settlement or otherwise, will first be used [***], and any remainder of the recovery, after [***], will be [***].
8.7Patent Term Extensions. Subject to the Existing Upstream License Agreements, as between the Parties, Athira shall [***], and [***].
8.8Patent Listing. Each Party shall have the sole and exclusive right to make all patent linkage listings (or other listings similar to the Orange Book and Purple Book listings in the U.S. and any foreign equivalent) of any Licensed Patent, including any Joint Patent, with respect to any Licensed Product in its respective territory.
8.9Trademarks. Each Party will have the right to brand the Licensed Products in its territory using trademarks, logos and trade names that it determines appropriate for the Licensed Products (such trademarks, logos, and trade names, the “Product Marks”). Pursuant to the Trademark Assignment Agreement, Sermonix has assigned to Athira, effective as of the Effective Date, all right, title and interest in and to certain Product Marks in the Territory that were owned by Sermonix or its Affiliates (the “Assigned Product Marks”). Neither Party may use any trademark Controlled by the other Party or its Affiliates (including their corporate names) to brand the Licensed Products without the other Party’s prior written consent. Each Party will own all rights in the Product Marks in its territory and will register and maintain the Product Marks in its territory that it determines reasonably necessary. Neither Party will use any trademark, logo or trade name in its territory with respect to any Licensed Product that is confusingly similar to any of the other Party’s Product Mark. To the extent requested by a Party and commercially practicable, the Parties will cooperate with respect to developing and implementing a global branding strategy for the Licensed Products, subject to separate discussion and agreement. If the Parties do not agree upon a global branding strategy, then each Party will have the right to brand the Licensed Products in its territory in a manner that it determines appropriate in its sole discretion.
Article 9
CONFIDENTIALITY
9.1Confidential Information. Except as expressly provided in this Agreement, each Party agrees that, during the Term and for a period of [***] thereafter, such Party (the “Receiving Party”) will keep confidential and will not publish or otherwise disclose and will not use for any purpose, other than as expressly provided for in this Agreement, any information furnished to it by or on behalf of the other Party (the “Disclosing Party”) pursuant to this Agreement or under the Confidentiality Agreement (“Confidential Information”), whether before or after the Effective Date, and whether in written, electronic, oral, visual, graphic or any other form. The Receiving Party may use Confidential Information only to the extent required to exercise its rights or perform its obligations under this Agreement. The Receiving Party will use at least the same
standard of care as it uses to protect proprietary or confidential information of its own, but no less than reasonable care, to require that its, and its Affiliates’, employees, agents, consultants, contractors and other representatives (“Representatives”) do not publish or disclose or make any unauthorized use of the Confidential Information of Disclosing Party. The Receiving Party will promptly notify the Disclosing Party upon discovery of any unauthorized use or disclosure of the Disclosing Party’s Confidential Information by the Receiving Party, its Affiliates or their respective Representatives. Notwithstanding any provision to the contrary set forth in this Agreement, [***] will be deemed the Confidential Information of [***].
9.2Exceptions. The confidentiality and non-use obligations in Section 9.1 shall not apply to any portion of the Confidential Information that the Receiving Party can demonstrate by competent written proof:
(a)was already known by the Receiving Party, other than under an obligation of confidentiality, prior to the time of receiving such information from the Disclosing Party, as evidenced by its pre-existing written records;
(b)is, as of the Effective Date, or thereafter becomes, generally known or available to the public, other than through any act or omission of the Receiving Party or any of its Representatives in breach of this Agreement;
(c)was subsequently lawfully disclosed to the Receiving Party on a non-confidential basis by a Third Party, as a matter of right (i.e., without breaching any obligation such Third Party may have to the Disclosing Party or any other Third Party); or
(d)is independently discovered or developed by the Receiving Party, independently of the activities undertaken by the Receiving Party pursuant to this Agreement and without the use of or reference to Confidential Information furnished by the Disclosing Party, as evidenced by the Receiving Party’s contemporaneously maintained written records.
9.3Authorized Disclosure. Notwithstanding the confidentiality and non-use obligations set forth in Section 9.1, the Receiving Party may disclose Confidential Information of the Disclosing Party as expressly permitted by this Agreement, or if and to the extent such disclosure is reasonably necessary in the following instances:
(a)filing or Prosecuting Patents as permitted by this Agreement;
(c)subject to Section 9.4(a), prosecuting or defending litigation as permitted by this Agreement;
(d)subject to Section 9.4(a), complying with a valid order of a court of competent jurisdiction or other Governmental Authority, or other judicial or administrative process, or if in the reasonable opinion of the Receiving Party’s legal counsel, such disclosure is otherwise required by Applicable Law, in each case, other than disclosure of the terms of this Agreement to a Securities Regulator;
(e)subject to Section 9.4(b), disclosure of the terms of this Agreement to securities regulators (including the U.S. Securities and Exchange Commission) or any securities exchange on which the Receiving Party’s securities are or are planned to be listed or traded (each, a “Securities Regulator”), in each case as required by Applicable Law promulgated by such Securities Regulators;
9.4Confidential Treatment.
(a)Generally. In the event the Receiving Party is required to make a disclosure of the Disclosing Party’s Confidential Information pursuant to Section 9.3(c) or Section 9.3(d), the Receiving Party will, except where prohibited by Applicable Law or judicial or administrative process, (i) give reasonable advance notice to the Disclosing Party of such required disclosure, (ii) use efforts to secure confidential treatment of such information at least as diligently as the Receiving Party would use to protect its own Confidential Information, but in no event less than reasonable efforts and (iii) cooperate with any lawful efforts by the Disclosing Party, at the Disclosing Party’s request and expense, to contest such disclosure, to obtain a protective order for the Confidential Information required to be disclosed, or to secure other confidential treatment of such Confidential Information. In the event that no such protective order or other remedy is obtained, or the Disclosing Party waives compliance with certain terms of this Article 9, then the Receiving Party will furnish only that portion of Confidential Information that [***]. Disclosure by the Receiving Party of Confidential Information in accordance with Section 9.3(c) or Section 9.3(d) will not, in and of itself, cause the information so disclosed to cease to be treated as Confidential Information under this Agreement, except to the extent that, by virtue of disclosure by the Receiving Party in full compliance with this Section 9.4, such information becomes generally known or available. In any event, the Receiving Party agrees to take all reasonable action to avoid disclosure of Confidential Information hereunder.
(b)Securities Filings. In the event the Receiving Party is required to make a disclosure of the terms of this Agreement to a Securities Regulator pursuant to Section 9.3(e), such Party will, within [***] prior to any such filing, provide the other Party with a copy of this Agreement showing any provisions hereof as to which such Party proposes to request confidential treatment, will provide the other Party with an opportunity to comment on any such proposed redactions and to suggest additional redactions, and will [***]; provided that [***], and provided, further, that[***].
9.5Use of Names. Subject to Section 9.6, neither Party nor any of its Affiliates will mention or otherwise use the name, logo or other Trademark of the other Party or any of its Affiliates in any publication, press release, marketing and promotional material or other form of publicity in connection with this Agreement or activities hereunder without the prior written consent of such other Party; provided that, subject to compliance with the remainder of this Section
9.5, such consent will not be required for a Party’s use of the other Party’s name and company logo to identify such other Party as a collaborator on such first Party’s website and in public presentations. Neither Party nor any of its Affiliates will use the other Party’s or such other Party’s Affiliate’s corporate name or Trademark in such manner that the distinctiveness, reputation, and validity of any Trademarks and corporate or trade names of such other Party will be impaired. Each Party will use the other Party’s and such other Party’s Affiliate’s corporate name and Trademarks in such manner that is consistent with the standards and guidelines used by such other Party or its Affiliates.
9.6Publication of Licensed Product Information. Athira shall have the right to review and comment on any publication by Sermonix, its Affiliate or any Ex-Territory Licensee, including any public oral presentation, manuscript or abstract, of [***]. Sermonix shall provide Athira with a copy of any such manuscript at least [***] before the anticipated submission for publication and Athira will provide its comments, if any, on such manuscript as soon as practicable to Sermonix, but in no event later than [***] from the date of delivery to Athira. Sermonix shall provide Athira with a copy of any such oral presentation, abstract, or posters at least [***] before the anticipated presentation or submission for publication and Athira will make reasonable efforts to expedite review of such materials, abstracts and posters, and will provide its comments, if any, as soon as practicable to Sermonix, but in no event later than [***] from the date of delivery to Athira. Sermonix shall [***]. In addition, if such publication contains patentable subject matter, then at Athira’s request, Sermonix shall [***].
(a)The Parties have agreed to issue a joint press release, substantially in the form attached hereto as Exhibit 9.7(a), promptly upon execution of this Agreement. Except as may be expressly permitted under Section 9.3, Section 9.6 or this Section 9.7, neither Party nor its Affiliates will make any other public announcement concerning [***] without first [***]. The Party desiring to make any such public announcement will provide the other Party with a written copy of the proposed announcement [***] prior to public release to allow the other Party to comment upon such announcement prior to public release. If such public announcement is required to be made by Applicable Law, judicial order or stock exchange or quotation system rule, the Party making such public announcement will provide to the other Party a copy of the proposed public announcement in written or electronic form upon [***] for the purpose of allowing the notified Party to review and comment upon such public announcement. Under such circumstances, the releasing Party will [***]. Neither Party will be required to seek the permission of the other Party to repeat any information regarding the terms of this Agreement hereto that has already been publicly disclosed by such Party or by the other Party in accordance with the terms of this Agreement; provided that [***].
(b)Each Party may make public statements regarding [***], provided that [***].
9.8Prior Non-Disclosure Agreements. Upon execution of this Agreement, the terms of this Article 9 will supersede any prior non-disclosure, secrecy or confidentiality agreement between the Parties, including the Confidentiality Agreement. Any information disclosed under such prior agreements by Sermonix will be deemed disclosed by Sermonix under this Agreement,
and any information disclosed under such prior agreements by Athira will be deemed disclosed by Athira under this Agreement.
Article 10
REPRESENTATIONS AND WARRANTIES
10.1Representations, Warranties and Covenants of Each Party. Each Party represents and warrants to the other Party as of the Effective Date and covenants (as applicable) that:
(a)it is a company or corporation duly organized, validly existing, and in good standing under the laws of the country or jurisdiction in which it is incorporated, and has full corporate power and authority and the legal right to own and operate its property and assets and to carry on its business as it is now being conducted and as contemplated in this Agreement;
(b)it has the corporate power and authority and the legal right to enter into this Agreement and perform its obligations hereunder, it has taken all necessary corporate action on its part required to authorize the execution and delivery of the Agreement and the performance of its obligations hereunder, and this Agreement has been duly executed and delivered on behalf of such Party, and constitutes a legal, valid, and binding obligation of such Party that is enforceable against it in accordance with its terms, subject to applicable bankruptcy, insolvency, reorganization, moratorium and similar laws affecting creditors’ rights and remedies generally;
(c)it is not a party to, and will not enter into during the Term, any agreement that would prevent it from granting the rights granted to the other Party under this Agreement or performing its obligations under the Agreement; and
(d)it has not been Debarred/Excluded and no proceeding that could result in it being Debarred/Excluded is pending, and in the course of performing its obligations or exercising its rights under this Agreement, it shall comply with all Applicable Laws, including as applicable, GMP, GCP, and GLP, and it shall not employ or engage any Person who has been Debarred/Excluded or, to such Party’s knowledge, is the subject of debarment proceedings by a Regulatory Authority.
10.2Representations, Warranties and Covenants of Sermonix. Sermonix represents and warrants to Athira as of the Effective Date and covenants (as applicable) that:
(b)it has the full right, power and authority to grant the licenses to Athira under the Licensed Technology as purported to be granted under Section 2.1;
(c)it has not granted any license or other right under the Licensed Technology that is inconsistent with the license granted to Athira under Section 2.1;
(d)all Existing Licensed Patents are listed on Exhibit 1.53; and each Patent listed on Exhibit 1.53 [***]. The pending applications included in the Existing Licensed Patents are [***]. Except as specified on Exhibit 1.53, [***];
(g)there are no pending, alleged or, to its Knowledge, threatened, (i) inter partes review, post-grant review, interference, re-examination, cancellation, opposition or other protest proceedings involving the Existing Licensed Patents, (ii) actions or litigation seeking to invalidate or challenge the enforceability of any of the Existing Licensed Patents, (iii) claims or actions challenging its or any of its Affiliates’ right to use or grant licenses under the Licensed Technology, or (iv) any inventorship challenges involving the Existing Licensed Patents that are in or before any patent office (or other Governmental Authority performing similar functions);
(j)to its Knowledge, neither the practice of the Licensed Technology nor the Development of the Licensed Compound or any Licensed Product by it or its Affiliates or Ex-Territory Licensees prior to the Effective Date infringes, misappropriates or otherwise violates any intellectual property rights or proprietary rights of any Third Party;
(k)to its Knowledge, no Third Party is infringing or misappropriating any of the Licensed Technology;
(l)it and its Affiliates have conducted, and, [***], all Development of the Licensed Compound and Licensed Products, including [***], in accordance with Applicable Law, including GLP, GCP and GMP as applicable;
(n)neither it, any of its Affiliates, nor, [***], has made any untrue statement of material fact or fraudulent statement to the FDA, the EMA or any other Regulatory Authority with respect to the Development or Manufacture of the Licensed Compound or any Licensed Product, or failed to disclose a material fact required to be disclosed to the FDA, the EMA or any other Regulatory Authority with respect to the Development or Manufacture of the Licensed Compound or any Licensed Product;
(o)no claims have been asserted, or, to its Knowledge, threatened by any Person, nor to its Knowledge, are there any valid grounds for any claim of any such kind, alleging any misconduct, violation of Applicable Law or injury or harm to any Person in connection with its or any of its Affiliates’ or, to its Knowledge, Ex-Territory Licensees conduct of Manufacturing or Development of the Licensed Compound or any Licensed Product;
(t)(i) it has provided or made available to Athira complete and accurate copies of: all material Existing Regulatory Materials; study reports; briefing documents; meeting minutes; reports or notices of any Serious Adverse Events; protocols and protocol amendments for all past and ongoing Clinical Trials and other studies (as applicable); and all other material documentation, information, data and results, in each case, in its Control and pertaining to the Development, Manufacture, safety and efficacy of the Licensed Compound and Licensed Products, (ii) [***], and (iii) the information, documentation, and other materials furnished or made available to Athira prior to the Effective Date, including in connection with Athira’s due diligence, are true, complete, and correct copies of what they purport to be and do not contain any untrue statement of a material fact or omit to state any material fact necessary to make the statements or facts contained therein not misleading;
(x)there are no adverse actions, claims, suits or proceedings, and no Governmental Authority investigations or inquiries, against or owed by it or any of its Affiliates, or that are pending or, to its Knowledge, threatened, involving any Licensed Technology or any Licensed Compound or Licensed Product.
10.3Additional Covenants. Each of Athira and Sermonix hereby covenant to the other:
(a)No Bribery. It will not in the future offer, promise, pay, authorize, or give, money or anything of value, directly or indirectly, to any Government Official or Other Covered Party for the purpose, pertaining to this Agreement, of: (i) influencing any act or decision of the Government Official or Other Covered Party; (ii) inducing the Government Official or Other Covered Party to do or omit to do an act in violation of a lawful duty; (iii) securing any improper advantage; or (iv) inducing the Government Official or Other Covered Party to influence the act or decision of a government or government instrumentality, in order to obtain or retain business, or direct business to, any Person, in each case, in the performance of any activities under this Agreement;
(b)Anti-Corruption Laws Compliance. In performing under this Agreement, it and its Affiliates agree to comply with all Applicable Law, including applicable anti-corruption laws, including: the Foreign Corrupt Practices Act of 1977 and the UK Bribery Act 2010, as amended from time-to-time; the anti-corruption laws of the Territory; and all laws enacted to implement the Organization for Economic Co-operation and Development Convention on Combating Bribery of Foreign Officials in International Business Transactions.
(c)Ligand Agreements. As of the Effective Date, Sermonix and Ligand have entered into the Ligand-Sermonix Agreement, pursuant to which Ligand grants to Sermonix certain licenses and rights with respect to intellectual property rights owned or controlled by Ligand for Exploitation of the Licensed Compound and Licensed Products in the Retained
Territory, and Athira and Ligand have entered into the Ligand-Athira Agreement, pursuant to which Ligand grants to Athira certain licenses and rights with respect to intellectual property rights owned or controlled by Ligand for Exploitation of the Licensed Compound and Licensed Products in the Territory. The Parties agree that (i) the Ligand-Athira Agreement is being entered into to, among other things, facilitate direct payment of royalty and milestone obligations from Athira to Ligand, (ii) upon termination of this Agreement for any reason, [***], and (iii) upon termination of the Henlius Agreement for any reason, [***].
10.4NO OTHER WARRANTIES. EXCEPT AS EXPRESSLY STATED IN THIS AGREEMENT, NO REPRESENTATIONS OR WARRANTIES WHATSOEVER, WHETHER EXPRESS OR IMPLIED, INCLUDING WARRANTIES OF MERCHANTABILITY, FITNESS FOR A PARTICULAR PURPOSE, NON-INFRINGEMENT, OR NON-MISAPPROPRIATION OF THIRD-PARTY INTELLECTUAL PROPERTY RIGHTS, ARE MADE OR GIVEN BY OR ON BEHALF OF A PARTY. ALL REPRESENTATIONS AND WARRANTIES, WHETHER ARISING BY OPERATION OF LAW OR OTHERWISE, ARE HEREBY EXPRESSLY DISCLAIMED. Each Party acknowledges and agrees that the Licensed Products are the subject of ongoing research and development and that neither Party can assure the safety, usefulness or successful Development or Commercialization of any Licensed Product in the Field in the Territory.
Article 11
INDEMNIFICATION
11.1Indemnification by Athira. Athira shall indemnify, defend and hold harmless Sermonix, its Affiliates, and its and their respective directors, officers, employees and agents (individually and collectively, the “Sermonix Indemnitee(s)”) from and against any and all losses, liabilities, damages and expenses (including reasonable attorneys’ fees and costs) (collectively, “Losses”) to which any Sermonix Indemnitee may become subject as a result of any claim, demand, action or other proceeding by any Third Party (a “Claim”), to the extent such Claim or Losses arise out of:
(a)the Development, Manufacture or Commercialization of the Licensed Compounds or Licensed Products in the Territory by Athira or any of its Affiliates or Sublicensees;
(b)any breach of, or inaccuracy in, any representation or warranty made by Athira in this Agreement, or any breach or violation of any covenant or agreement of Athira in this Agreement; or
(c)the negligence, willful misconduct or breach of this Agreement by any Athira Indemnitee in the performance of Athira’s obligations under this Agreement;
provided that, Athira shall have no obligation to indemnify any Sermonix Indemnitees to the extent that the Losses arise out of or result from any matters for which Sermonix is obligated to indemnify Athira under Section 11.2.
11.2Indemnification by Sermonix. Sermonix shall indemnify, defend and hold harmless Athira, its Affiliates, and its and their directors, officers, employees and agents (individually and collectively, the “Athira Indemnitee(s)”) from and against any and all Losses
to which any Athira Indemnitee may become subject as a result of any Claim, to the extent such Claim or Losses arise out of:
(a)the Development, Manufacture or Commercialization of the Licensed Compounds or Licensed Products by Sermonix or any of its Affiliates or Ex-Territory Licensees, whether prior to or after the Effective Date;
(b)any breach of, or inaccuracy in, any representation or warranty made by Sermonix in this Agreement, or any breach or violation of any covenant or agreement of Sermonix in this Agreement; or
(c)the negligence, willful misconduct or breach of this Agreement by any Sermonix Indemnitee in the performance of Sermonix’s obligations under this Agreement;
provided that, Sermonix shall have no obligation to indemnify any Athira Indemnitees to the extent that the Losses arise out of or result from any matters for which Athira is obligated to indemnify Sermonix under Section 11.1.
11.3Indemnification Procedure. If either Party is seeking indemnification under Sections 11.1 or 11.2, as applicable (the “Indemnified Party”), it shall inform Athira (if the Indemnified Party is Sermonix) or Sermonix (if the Indemnified Party is Athira) (the “Indemnifying Party”) of the Claim giving rise to the obligation to indemnify pursuant to such Section 11.1 or 11.2, as applicable, within [***] after receiving notice of the Claim (it being understood and agreed, however, that the failure or delay by an Indemnified Party to give such notice of a Claim shall not affect the indemnification provided hereunder except to the extent the Indemnifying Party shall have been prejudiced as a result of such failure or delay to give notice). The Indemnifying Party shall have the right to assume the defense of any such Claim for which it is obligated to indemnify the Indemnified Party, provided that [***]. The Indemnified Party shall cooperate with the Indemnifying Party and the Indemnifying Party’s insurer as the Indemnifying Party may reasonably request, and at the Indemnifying Party’s cost and expense. The Indemnified Party shall have the right to participate, at its own expense and with counsel of its choice, in the defense of any Claim that has been assumed by the Indemnifying Party unless [***]. The Indemnified Party shall not admit liability or settle any such Claim without the prior written consent of the Indemnifying Party, [***]. If the Indemnifying Party does not assume and conduct the defense of the Claim as provided above and provided the Indemnifying Party does not dispute that the Claim is indemnifiable by the Indemnifying Party, (i) the Indemnified Party may defend against, and consent to the entry of any judgment or enter into any settlement with respect to the Claim in any manner the Indemnified Party may deem reasonably appropriate (and the Indemnified Party need not consult with, or obtain any consent from, the Indemnifying Party in connection therewith), and (ii) the Indemnified Party reserves any right it may have under this Article 11 to obtain indemnification from the Indemnified Party. No Indemnifying Party shall have the obligation to indemnify the other Party in connection with any settlement made without the Indemnifying Party’s written consent, [***]. If the Parties cannot agree as to the application of Section 11.1 or 11.2 as to any Claim, pending resolution of the Dispute pursuant to Article 13, the Parties may conduct separate defenses of such Claims, with each Party retaining the right to seek indemnification from the other Party in accordance with Section 11.1 or 11.2 upon resolution of the underlying Claim.
11.4Mitigation of Loss. Each Indemnified Party shall take and shall cause its Affiliates to take all such reasonable steps and actions as are reasonably necessary or as the Indemnifying Party may reasonably request in order to mitigate any Claims (or potential losses or damages) under this Article 11. Nothing in this Agreement shall or shall be deemed to relieve any Party of any common law or other duty to mitigate any losses incurred by it.
11.5Limitation of Liability. NEITHER PARTY HERETO SHALL BE LIABLE FOR SPECIAL, INCIDENTAL, CONSEQUENTIAL OR PUNITIVE DAMAGES ARISING OUT OF THIS AGREEMENT OR THE EXERCISE OF ITS RIGHTS OR PERFORMANCE OF ITS OBLIGATIONS HEREUNDER, REGARDLESS OF ANY NOTICE OF SUCH DAMAGES. NOTWITHSTANDING THE FOREGOING, NOTHING IN THIS SECTION 11.5 IS INTENDED TO OR SHALL LIMIT OR RESTRICT (A) ANY DAMAGES ARISING FROM OR RELATED TO [***] OR (B) THE INDEMNIFICATION RIGHTS OR OBLIGATIONS UNDER SECTION 11.1 OR 11.2.
11.6Insurance. Each Party shall procure and maintain insurance, including product liability insurance, with respect to its activities hereunder and which is consistent with normal business practices of prudent companies similarly situated at all times during which any Licensed Product is being clinically tested in human subjects or commercially distributed or sold. Each Party shall provide the other Party with evidence of such insurance upon request and shall provide the other Party with written notice at least [***] prior to the cancellation, non-renewal or material changes in such insurance. Such insurance shall not be construed to create a limit of any Party’s liability under this Agreement.
ARTICLE 12
TERM AND TERMINATION
12.1Term. The term of this Agreement shall commence on the Effective Date and, unless earlier terminated as set forth in Section 12.2, will continue in full force and effect until Athira, its Affiliates, and its Sublicensees are no longer Developing or Commercializing any Licensed Product in any country in the Territory (the “Term”). On a Licensed Product-by-Licensed Product and country-by-country basis in the Territory, upon the earlier of (a) expiration of the Royalty Term for such Licensed Product in such country or (b) payment of all Partnering Transaction Consideration pursuant to Section 7.7, the licenses granted by Sermonix to Athira under Section 2.1 with respect to such Licensed Product in such country will become fully paid-up, perpetual, irrevocable, sublicensable (through multiple tiers) and royalty-free.
(a)Termination by Athira for Convenience. Athira shall have the right to terminate this Agreement in its entirety or on a Licensed Product-by-Licensed Product or country-by-country basis at any time upon [***] prior written notice to Sermonix; provided that Athira may not exercise this right to terminate prior to Athira’s receipt of the first Phase 3 Clinical Trial topline results for any Licensed Product.
(b)Termination for Material Breach. This Agreement may be terminated by a Party at any time during the Term if the other Party is in material breach of this Agreement, upon
written notice by such Party if the other Party has not cured such material breach within [***] after written notice identifying the material breach in reasonable detail; provided that, [***]. Notwithstanding the foregoing, if the allegedly breaching Party disputes [***] the existence, materiality, or failure to cure of any breach, and [***], the other Party will not have the right to terminate this Agreement in accordance with this Section 12.2(b) unless and until it has been determined, in accordance with Article 13, that the allegedly breaching Party has materially breached this Agreement and failed to cure such breach. During the pendency of such Dispute, (A) the applicable cure period will be tolled, (B) all the terms of this Agreement will remain in effect, and (C) the Parties will continue to perform all of their respective obligations hereunder.
(c)Termination for Insolvency. Each Party has the right to terminate this Agreement in its entirety upon delivery of written notice to the other Party if such other Party incurs an Insolvency Event; provided, however, in the case of any involuntary bankruptcy proceeding, such right to terminate shall only become effective if the affected Party consents to the involuntary bankruptcy or if such proceeding is not dismissed or stayed within [***] after the filing thereof. As used herein, “Insolvency Event” means [***].
(d)Termination for Safety Reasons. Athira shall have the right to terminate this Agreement in its entirety or on a Licensed Product-by-Licensed Product or country-by-country basis at any time (i) upon [***] prior written notice to Sermonix in the event that [***] for safety reasons or (ii) [***] upon written notice to Sermonix in the event that [***] for safety reasons and [***].
12.3Effects of Termination. The following shall apply upon any termination of this Agreement (or, if this Agreement is terminated with respect to a Licensed Product or country, the following shall apply with respect to the Terminated Product or Terminated Territory), except as specified otherwise:
(a)Licenses. All licenses and other rights with respect to the Terminated Product and Terminated Territory granted by Sermonix to Athira hereunder shall terminate.
(b)Transition Activities. If this Agreement is terminated by Athira pursuant to Section 12.2(a) or by Sermonix pursuant to Section 12.2(b) (but not if this Agreement is terminated pursuant to Section 12.2(c) or by Athira pursuant to Section 12.2(b)), and Sermonix provides written notice to Athira within [***] after the effective date of termination that Sermonix wishes to continue Development or Commercialization with respect to any Terminated Product, the Parties shall [***].
(c)Sublicenses. Sermonix shall grant to each Sublicensee in the Terminated Territory, at each such Sublicensee’s written request to Sermonix within [***] after the effective date of termination with respect to such Terminated Territory, a direct license, provided that such Sublicensee (i) is not then in default of its sublicense agreement or this Agreement, (ii) agrees in writing to comply with the terms of this Agreement to the extent applicable to the rights originally sublicensed to such Sublicensee by Athira, and (iii) agrees to [***]. The scope of such direct license shall be no less than the scope of the license granted herein and sublicensed to such Sublicensee, and Sermonix shall have no obligation to perform any task for such Sublicensee other
than those tasks that Sermonix would have been obligated to perform for Athira under this Agreement if it had not been terminated.
(d)Return of Confidential Information. Upon termination or expiration of this Agreement in its entirety, each Receiving Party will, or will cause its Affiliates to, return (or destroy, as directed by the Disclosing Party) all data, files, records, and other materials containing or comprising the Disclosing Party’s Confidential Information that are in the Receiving Party’s or its Affiliates’ possession. Notwithstanding the foregoing, the Receiving Party will be permitted to retain one copy of such data, files, records, and other materials for archival and legal compliance purposes, and the Receiving Party will not be required to destroy Confidential Information that is stored automatically, such as through server backup processes.
12.4Rights in Bankruptcy.
(a)The Parties intend to take advantage of the protections of Section 365(n) (or any successor provision) of the U.S. Bankruptcy Code or any analogous provisions in any other country or jurisdiction to the maximum extent permitted by Applicable Law. All rights and licenses granted to Athira under this Agreement shall be deemed to be “intellectual property” for the purposes of Section 365(n) or any analogous provisions in any other country or jurisdiction. During the Term, Sermonix shall create and maintain current copies to the extent practicable of all such intellectual property. Athira shall retain and may fully exercise all of its rights and elections under the U.S. Bankruptcy Code or any analogous provisions in any other country or jurisdiction, including the right to obtain the intellectual property from another entity. In the event of the commencement of a bankruptcy proceeding by or against Sermonix under the U.S. Bankruptcy Code or any analogous provisions in any other country or jurisdiction, Athira shall be entitled to a complete duplicate of (or complete access to, as appropriate) all such intellectual property (including all embodiments of such intellectual property), which, if requested by Athira and not already in Athira’s possession, shall be promptly delivered to Athira (a) before this Agreement is rejected by or on behalf of Sermonix, within [***] after Athira’s written request, unless Sermonix or its trustee or receiver elects within [***] to continue to perform all of its obligations under this Agreement, or (b) after any rejection of this Agreement by or on behalf of Sermonix, if not previously delivered as provided under clause (a) above. Unless and until Sermonix rejects this Agreement, Sermonix shall perform this Agreement or provide the intellectual property (including all embodiments of such intellectual property) to Athira, and shall not interfere with the rights of Athira to such intellectual property, including the right to obtain the intellectual property from another entity. All rights of the Parties under this Section 12.4 and under Section 365(n) of the U.S. Bankruptcy Code are in addition to and not in substitution of any and all other rights, powers, and remedies that each Party may have under this Agreement, the U.S. Bankruptcy Code, and any other Applicable Law. Athira shall have the right to perform the obligations of Sermonix hereunder with respect to such intellectual property, but neither such provision nor such performance by Athira shall release Sermonix from any such obligation or liability for failing to perform it.
(b)The Parties agree that they intend the foregoing rights to extend to the maximum extent permitted by law and any provisions of applicable contracts with Third Parties, including for purposes of the U.S. Bankruptcy Code, (i) the right of access to any intellectual property (including all embodiments thereof) of Sermonix or any Third Party with whom Sermonix
contracts to perform an obligation of Sermonix under this Agreement, and, in the case of the Third Party, which is necessary for the Development, Regulatory Approval and Manufacture of the Licensed Compound or any Licensed Product and (ii) the right to contract directly with any Third Party described in (i) in this sentence to complete the contracted work.
(c)Any intellectual property provided pursuant to the provisions of this Section 12.4 shall be subject to the licenses set forth elsewhere in this Agreement.
12.5Accrued Rights; Survival. Expiration or termination of this Agreement will not relieve the Parties of any liability that accrued hereunder prior to the effective date of such expiration or termination nor preclude either Party from pursuing all rights and remedies it may have hereunder or at law or in equity with respect to any breach of this Agreement, nor prejudice either Party’s right to obtain performance of any obligation. Without limiting the foregoing, the following provisions shall survive the termination or expiration of this Agreement for any reason: Article 1, Section 2.5, Section 3.2, Article 7 (solely with respect to amounts that have accrued prior to the effective date of termination), Section 8.1, Section 8.3, Article 9, Section 10.3(c)(ii) & (iii), Section 10.4, Article 11 (excluding Section 11.6), Section 12.3, Section 12.4, this Section 12.5, Article 13 and Article 14.
12.6Termination Not Sole Remedy. Termination is not the sole remedy under this Agreement and, whether or not termination is effected and notwithstanding anything contained in this Agreement to the contrary, all other remedies shall remain available except as agreed to otherwise herein.
ARTICLE 13
DISPUTE RESOLUTION
13.1Disputes. Except for any matter for which this Agreement assigns decision-making to the Parties, including all matters within the scope of a Committee’s responsibilities, or requires the consent of one or both of the Parties (the “Excluded Matters”), the Parties agree that any claim, dispute or controversy between the Parties or any of their Affiliates arising from, relating to or in connection with this Agreement, including with respect to its formation, applicability, breach, termination, enforcement, interpretation or validity (a “Dispute”), will be resolved in accordance with this Article 13.
13.2Continuance of Rights and Obligations during Pendency of Dispute Resolution. During the pendency of any Dispute referred to dispute resolution in accordance with this Article 13, including a dispute related to the termination of this Agreement, all rights and obligations of the Parties will continue until such time as any Dispute has been resolved in accordance with the provisions of this Article 13.
13.3Escalation. Either Party, through the delivery of written notice, may refer any Dispute to the Executive Officers for attempted resolution. If the Executive Officers are unable to resolve such Dispute within [***] following the delivery of such written notice, then, upon the written request of either Party to the other Party (“Dispute Referral”), the Dispute will be subject to further resolution in accordance with the remainder of this Article 13.
(a)General. If either Party delivers a Dispute Referral under Section 13.3, then, except as set forth in Section 13.6, the Dispute will be submitted to the American Arbitration Association (“AAA”) for resolution by final and binding arbitration in accordance with this Section 13.4 under the Commercial Arbitration Rules of the AAA in effect at the time of the arbitration (the “AAA Rules”), except as modified herein. Any disputes concerning the propriety of the commencement of the arbitration or the scope or applicability of this agreement to arbitrate will be finally settled by the arbitral tribunal. The seat of arbitration will be New York, New York, and the language of the proceedings, including all communications, will be English.
(b)Process. Unless otherwise agreed by the Parties, the arbitration will be conducted by a tribunal of three independent and impartial arbitrators with at least [***]. An arbitrator shall be deemed to meet these qualifications unless a Party objects within [***] after an arbitrator is nominated. Within the time provided in the AAA Rules, each Party will nominate one arbitrator with appropriate experience, and the two Party-nominated arbitrators will nominate a third arbitrator, who will serve as the chairperson of the tribunal, within [***] of the second arbitrator’s appointment. If any of the three arbitrators are not nominated within the foregoing time periods, then the AAA will appoint the arbitrator(s). Within [***] of the commencement of arbitration, the Parties will attempt [***] to reach agreement upon, and thereafter the Parties will follow, procedures directed at assuring that the arbitration will be concluded and the award rendered within no more than [***] from the date on which the arbitration file is transmitted to the arbitral tribunal. Failing such agreement, the arbitral tribunal will design and the Parties will follow procedures directed at meeting such a time schedule.
(c)Binding Decision. The arbitral award will be final and binding on the Parties and the Parties will carry out the award without delay. Judgment on the award so rendered may be entered in any court of competent jurisdiction. The arbitral tribunal will resolve the Dispute by applying the provisions of this Agreement and the governing law set forth in Section 14.5. Notwithstanding Section 14.5 with respect to the applicable substantive law, any arbitration and this agreement to arbitrate shall be governed by the US Federal Arbitration Act, 9 U.S.C. § 1 et seq.
(d)Interim Measures. By agreeing to arbitration, the Parties do not intend to deprive any court of its jurisdiction to issue, at the request of a Party, a pre-arbitral injunction, pre-arbitral attachment or other order of interim relief to avoid irreparable harm, maintain the status quo, preserve the subject matter of the Dispute, or aid the arbitration proceedings and the enforcement of any award, including after constitution of the arbitral tribunal. Without prejudice to such provisional or interim remedies in aid of arbitration as may be available under the jurisdiction of a competent court, the arbitral tribunal has full authority to grant provisional or interim remedies and to award damages for the failure of any Party to the Dispute to respect the arbitral tribunal’s order to that effect.
(e)Waiver of Jury Trial. EACH PARTY HERETO WAIVES ANY RIGHT TO TRIAL OF ANY ISSUE BY JURY.
(f)Damages Awards. The arbitral tribunal is authorized to award any damages available under law, but is not authorized to (i) award any damages excluded under Section 11.5, or (ii) reform, modify or materially change this Agreement or any other agreements contemplated hereunder, provided, however, that the damage limitations described in clause (i) will not apply if such damages are statutorily imposed.
(g)Attorney’s Fees. Each Party will bear its own attorney’s fees, costs, and disbursements arising out of the arbitration, and will pay an equal share of the fees and costs of the AAA and the arbitrators; provided, however, that [***].
(h)Confidentiality. The existence, content, and results of an arbitration will be treated as Confidential Information of both Parties. Except as required by Applicable Law, including any disclosure or filing reasonably necessary to comply with the rules and regulations of any Securities Regulator, or as necessary for the recognition and enforcement or challenge of an arbitral award, neither a Party nor an arbitrator may disclose the existence, content or results of any arbitration hereunder without the prior written consent of the Parties. Any documents submitted to the arbitrators will be kept confidential and will not be disclosed, except that any such documents may be disclosed (i) as necessary in connection with any action to enforce or collect or challenge the award or (ii) to the extent discoverable or admissible in any action arising out of or in connection with this Agreement. No award or procedural order made in the arbitration shall be published and the Parties do not consent to the AAA publishing any form of an award or of an order issued by the arbitral tribunal.
13.5Injunctive Relief. Notwithstanding the terms of and procedures set forth in Section 13.4, any applications, motions or orders to show cause seeking temporary restraining orders, preliminary injunctions or other similar preliminary or temporary legal or equitable relief (“Injunctive Relief”) concerning a Dispute (including Disputes arising out of a potential or actual breach of the confidentiality and non-use provisions in Article 9) may immediately be brought in any court of competent jurisdiction. Alternatively, a party seeking Injunctive Relief may immediately, without invocation or exhaustion of the procedures set forth in Section 13.4, seek Injunctive Relief in accordance with the AAA emergency arbitration procedures then in effect and applying the substantive law specified in Section 14.5. In either event, once the Injunctive Relief proceedings have been conducted and a decision rendered thereon by the court or arbitral forum, the Parties will, if the Dispute is not finally resolved by the Injunctive Relief, proceed to resolve the Dispute in accordance with the terms of Section 13.4.
13.6Non-Arbitral Disputes. Notwithstanding anything to the contrary in Section 13.4, in the event of a Dispute with respect to the validity, scope, enforceability, infringement or ownership of any Patent or other intellectual property rights, and such Dispute is not resolved in accordance with Section 13.2, such Dispute shall not be submitted to arbitration in accordance with Section 13.4, unless otherwise agreed by the Parties in writing, and instead either Party may initiate litigation in a court of competent jurisdiction in any country in which such rights or remedies apply.
ARTICLE 14
MISCELLANEOUS
14.1Force Majeure. Neither Party shall be held liable to the other Party nor be deemed to have defaulted under or breached this Agreement for failure or delay in performing any obligation under this Agreement (except payment of money obligations), to the extent that such failure or delay is caused by or results from any event beyond such Party’s reasonable control including acts of God, fire, flood, explosion, earthquake, pandemic, disease, or other natural forces, war, civil unrest, acts of terrorism, accident, destruction or other casualty, any lack or failure of transportation facilities, any lack or failure of supply of raw materials outside of such Party’s reasonable control, or any other event similar to those enumerated above. The affected Party shall notify the other Party of such force majeure circumstances as soon as reasonably practical, and shall promptly undertake all reasonable efforts necessary to cure or mitigate such force majeure circumstances, provided that, if the failure to perform due to such force majeure circumstance continues for a period of [***] or more, the Parties will [***]. All delivery dates under this Agreement that have been affected by force majeure shall be tolled for the duration of such force majeure. In no event shall a Party be required to prevent or settle any labor disturbance or dispute.
(a)Except as express permitted herein, this Agreement may not be assigned or otherwise transferred, nor may any right or obligation hereunder be assigned or transferred, by a Party without the prior written consent of the other Party. Any attempted assignment not in accordance with this Section 14.2 shall be null and void and of no legal effect. Any permitted assignee shall assume all assigned obligations of its assignor under this Agreement. The terms and conditions of this Agreement shall be binding upon, and shall inure to the benefit of, the Parties and their respective successors and permitted assigns.
(b)Notwithstanding the foregoing, a Party may, without consent of the other Party, assign this Agreement and its rights and obligations hereunder in whole or in part to an Affiliate of such Party, or in whole to its successor-in-interest in connection with the sale of all or substantially all of its stock, including a merger, acquisition or similar transaction in connection with the foregoing.
14.3Severability. If any one or more of the provisions contained in this Agreement is held invalid, illegal or unenforceable in any respect, the validity, legality and enforceability of the remaining provisions contained herein shall not in any way be affected or impaired thereby, unless the absence of the invalidated provision(s) adversely affects the substantive rights of the Parties. The Parties shall in such an instance use their best efforts to replace the invalid, illegal or unenforceable provision(s) with valid, legal and enforceable provision(s) which, insofar as practical, implement the purposes of this Agreement.
14.4Notices. All notices which are required or permitted hereunder shall be in writing and sufficient if delivered personally, sent by internationally recognized overnight courier, sent by registered or certified mail, postage prepaid, return receipt requested, or sent by email, to the address set forth below:
If to Sermonix, notices must be addressed to:
|
|
[***] |
|
[***] |
|
[***] |
|
Attn: |
[***] |
Email: |
[***] |
With a copy to (which will not constitute notice for purposes of this Agreement):
|
|
[***] |
|
[***] |
|
[***] |
|
Attn: |
[***] |
Email: |
[***] |
If to Athira:
|
|
[***] |
|
[***] |
|
[***] |
|
Attn: |
[***] |
Email: |
[***] |
With a copy to (which will not constitute notice for purposes of this Agreement):
|
|
[***] |
|
[***] |
|
[***] |
|
Attn: |
[***] |
Email: |
[***] |
or to such other address as the Party to whom notice is to be given may have furnished to the other Party in writing in accordance herewith. Any such notice shall be deemed to have been given: (a) when delivered if personally delivered on a Business Day (or [***], if delivered on a non-Business Day); (b) on the [***] after dispatch if sent by internationally recognized overnight courier; (c) on the [***] following the date of mailing if sent by mail, or (d) upon [***], if sent by email.
14.5Governing Law. This Agreement shall be governed by and construed in accordance with the laws of the State of New York, without giving effect to any choice of law principles that would require the application of the laws of a different jurisdiction. The application of the U.N. Convention on Contracts for the International Sale of Goods is excluded.
14.6Entire Agreement; Amendments. This Agreement, together with the Exhibits attached hereto, contains the entire understanding of the Parties with respect to the subject matter hereof. All express or implied agreements and understandings, either oral or written, with regard to the subject matter hereof (including the licenses granted hereunder) are superseded by the terms of this Agreement. Neither Party is relying on any representation, promise or warranty not expressly set forth in this Agreement. This Agreement may be amended, or any term hereof modified, only by a written instrument duly executed by authorized representatives of all Parties hereto.
14.7Independent Contractors. It is expressly agreed that Sermonix and Athira shall be independent contractors and that the relationship between Sermonix and Athira shall not constitute a partnership, joint venture or agency. Neither Sermonix nor Athira shall have the authority to make any statements, representations or commitments of any kind, or to take any action, which shall be binding on the other, without the prior written consent of the other.
14.8Performance by Affiliates. Notwithstanding anything to the contrary set forth herein, each Party will have the right to perform any or all of its obligations and exercise any or all of its rights under this Agreement through any Affiliate. Each Party hereby guarantees the performance by its Affiliates of such Party’s obligations under this Agreement and will cause its Affiliates to comply with the provisions of this Agreement in connection with such performance.
14.9Waiver. The waiver by a Party of any right hereunder, or the failure of the other Party to perform, or a breach by the other Party, shall not be deemed a waiver of any other right hereunder or of any other breach or failure by such other Party whether of a similar nature or otherwise.
14.10Cumulative Remedies. Except as otherwise expressly provided in this Agreement, no remedy referred to in this Agreement is intended to be exclusive, but each shall be cumulative and in addition to any other remedy referred to in this Agreement or otherwise available under law.
14.11Business Day Requirements. In the event that any notice or other action or omission is required to be taken by a Party under this Agreement on a day that is not a Business Day then such notice or other action or omission shall be deemed to be required to be taken on the next occurring Business Day.
14.12English Language. This Agreement is in the English language only, which language shall be controlling in all respects, and all versions hereof in any other language shall be for accommodation only and shall not be binding upon the Parties. All communications and notices to be made or given pursuant to this Agreement, and any dispute proceeding related to or arising hereunder, shall be in the English language. If there is a discrepancy between any translation of this Agreement and this Agreement, this Agreement (i.e., the English version) shall prevail.
14.13Further Actions. Each Party agrees to execute, acknowledge and deliver such further instruments, and to do all such other acts, as necessary or appropriate in order to carry out the purposes and intent of this Agreement.
14.14Headings. The captions to the Sections and Articles hereof are not a part of this Agreement, but are merely for convenience to assist in locating and reading the several Sections and Articles hereof.
14.15Waiver of Rule of Construction. Each Party has had the opportunity to consult with counsel in connection with the review, drafting and negotiation of this Agreement. Accordingly, the rule of construction that any ambiguity in this Agreement shall be construed against the drafting Party shall not apply.
14.16Interpretation. Except where the context expressly requires otherwise, (a) the use of any gender herein shall be deemed to encompass references to either or both genders, and the use of the singular shall be deemed to include the plural (and vice versa), (b) the words “include”, “includes” and “including” shall be deemed to be followed by the phrase “without limitation”, (c) the word “will” shall be construed to have the same meaning and effect as the word “shall”, (d) any definition of or reference to any agreement, instrument or other document herein shall be construed as referring to such agreement, instrument or other document as from time to time amended, supplemented or otherwise modified (subject to any restrictions on such amendments, supplements or modifications set forth herein), (e) any reference herein to any person shall be construed to include the person’s successors and assigns, (f) the words “herein”, “hereof” and “hereunder”, and words of similar import, shall be construed to refer to this Agreement in its entirety and not to any particular provision hereof, (g) all references herein to Sections, Articles, or Exhibits shall be construed to refer to Sections, Articles or Exhibits of this Agreement, and references to this Agreement include all Exhibits hereto, (h) the word “notice” means notice in writing (whether or not specifically stated) and shall include notices, consents, approvals and other written communications contemplated under this Agreement, (i) provisions that require that a Party, the Parties or any committee hereunder “agree”, “consent” or “approve” or the like shall require that such agreement, consent or approval be specific and in writing, whether by written agreement, letter, approved minutes or otherwise (but excluding e-mail and instant messaging), (j) references to any specific law, rule or regulation, or Section, section or other division thereof, shall be deemed to include the then-current amendments thereto or any replacement or successor law, rule or regulation thereof, and (k) the term “or” shall be interpreted in the inclusive sense commonly associated with the term “and/or.”
14.17Counterparts. This Agreement may be executed in one or more counterparts, all of which taken together will be regarded as one and the same instrument. Each Party may execute this Agreement in Adobe™ Portable Document Format (PDF) sent by electronic mail or other electronic means. PDF or other electronic signatures of authorized signatories of the Parties will be deemed to be original signatures, will be valid and binding upon the Parties, and, upon delivery, will constitute due execution of this Agreement.
{Signature Page Follows}
In Witness Whereof, the Parties intending to be bound have caused this License Agreement to be executed by their duly authorized representatives as of the Effective Date.
|
|
|
|
|
Sermonix Pharmaceuticals, Inc. |
|
Athira Pharma, Inc. |
By: |
|
|
By: |
|
|
|
|
|
|
Name: |
|
|
Name: |
|
|
|
|
|
|
Title: |
|
|
Title: |
|
[Signature Page to License Agreement]
List of Exhibits
|
|
|
Exhibit 1.31 |
|
Existing Upstream License Agreements |
Exhibit 1.51 |
|
Licensed Compound |
Exhibit 1.53: |
|
Existing Licensed Patents |
Exhibit 1.77 |
|
Retained Territory |
Exhibit 3.4: |
|
Service Provider Agreements |
Exhibit 4.2: |
|
Existing Regulatory Materials |
Exhibit 7.2: |
|
Assumed Liabilities |
Exhibit 9.7(a) |
|
Joint Press Release |
Exhibit 1.31
Existing Upstream License Agreement
[***]
Exhibit 1.51
Licensed Compound
Chemical Structure of Lasofoxifene Tartrate
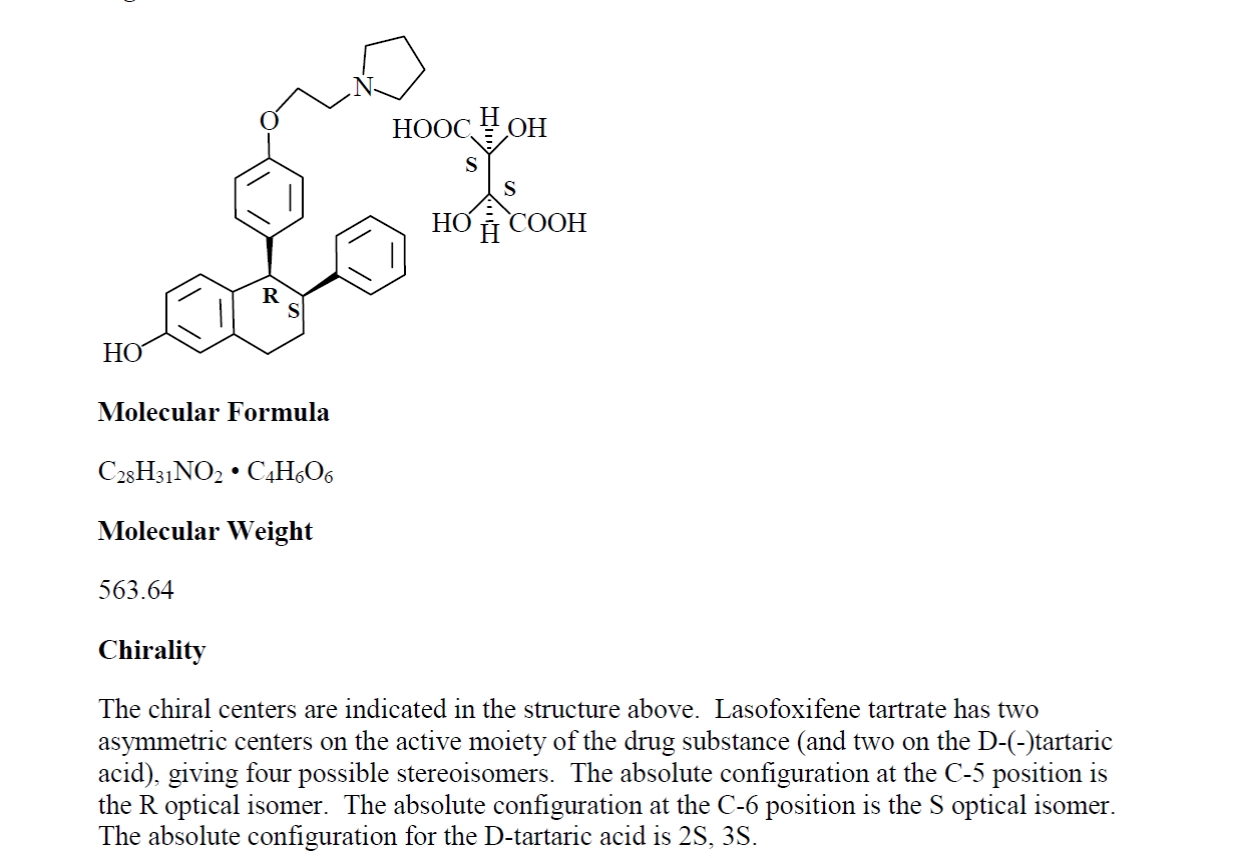
Exhibit 1.53
Licensed Patents
[***]
Exhibit 1.77
Retained Territory
[***]
Exhibit 3.4
Service Provider Agreements
[***]
Exhibit 4.2
Existing Regulatory Materials
[***]
Exhibit 7.2
Assumed Liabilities
[***]
Exhibit 9.7(a)
Joint Press Release
Athira Pharma Announces Exclusive License to Lasofoxifene Phase 3 Development Program for Metastatic Breast Cancer Candidate and a Financing for up to $236 Million
Phase 3 Development Program of Novel Selective Estrogen Receptor Modulator (SERM) Represents Potential Multi-Billion Dollar Opportunity as Treatment Option for Patients with ESR1-Mutations
$90 Million Upfront With Up to an Additional $146 Million Upon Exercise of Warrants
Financing Co-Led by Commodore Capital, Perceptive Advisors and TCGX Supports Development of Lasofoxifene through Phase 3 Clinical Topline Data Readout and Regulatory Milestones
Conference Call Today at 8:30 am Eastern Time
BOTHELL, Wash., December 18, 2025 – Athira Pharma, Inc. (NASDAQ: ATHA), a clinical-stage biopharmaceutical company dedicated to the development of novel therapeutics for high unmet medical needs, today announced that it has entered into an agreement to acquire the rights for the development and commercialization of lasofoxifene, a promising clinical asset in a potentially registrational Phase 3 trial. The ongoing Phase 3 ELAINE-3 clinical trial (NCT05696626) is greater than 50% enrolled with data expected in mid-2027.
Athira has acquired an exclusive global license (excluding Asia and certain countries in the Middle East) from Sermonix Pharmaceuticals, Inc. for rights to develop and commercialize lasofoxifene, a selective estrogen receptor modulator (SERM) for the potential treatment of metastatic breast cancer.
In conjunction with this transaction, Athira also announced an upfront financing of $90 million in private placement financing of common stock and warrants, with the warrants providing, if exercised, up to an additional $146 million to support development of the new program through key clinical and regulatory milestones. The financing was co-led by Commodore Capital, Perceptive Advisors, and TCGX, with participation from ADAR1, Blackstone Multi-Asset Investing, Kalehua Capital, Ligand Pharmaceuticals, New Enterprise Associates (NEA), Spruce Street Capital, and 9vc. The Company anticipates the upfront financing will support lasofoxifene development through its topline data readout and key regulatory milestones, with sufficient capital for runway into 2028.
“Today marks a defining moment for our company. This agreement for the rights to the Phase 3 lasofoxifene program for metastatic breast cancer is a significant step in building a pipeline with the potential to change lives and create enduring value,” said Mark Litton, Ph.D., President and Chief Executive Officer of Athira. “This program provides a near-term opportunity to generate pivotal data necessary for the approval of lasofoxifene and to establish it as the new standard of
care to treat ESR1-mutant breast cancer in patients who have progressed on aromatase inhibitors and prior CDK4/6 inhibitors. Supported by compelling Phase 2 clinical data and backed by blue-chip investors, we have a clear development strategy and are committed to advancing therapies that matter to patients while delivering value for our shareholders.”
“In the ELAINE-2 clinical trial, lasofoxifene demonstrated the potential to provide meaningful combination efficacy with 13 months of progression-free survival in heavily pre-treated second- and third-line ESR1-mutated metastatic breast cancer patients. We believe lasofoxifene has the potential to be the preferred endocrine therapy for metastatic breast cancer patients given its tissue-selective SERM profile may allow for the preservation of estrogen function in non-breast tissue, which provides tolerability and potential bone protection and quality of life benefits,” noted David Portman, M.D., Chief Executive Officer of Sermonix. “With the Phase 3 trial now more than 50% enrolled, we look forward to delivering pivotal data in mid-2027 and advancing toward a regulatory submission.”
“We are excited to announce this financing to help accelerate the development of lasofoxifene," said Cariad Chester, Managing Partner at TCGX. "With its differentiated profile, lasofoxifene has the potential to become the endocrine therapy of choice for the approximately 40% of breast cancer patients who develop ESR1 mutations and have progressed on aromatase inhibitors and prior CDK4/6 inhibitors. The accomplished team at Athira is committed to efficiently executing the ongoing potentially pivotal trial of lasofoxifene, and we believe, if approved, it may become the treatment of choice for oncologists and patients who are battling this challenging disease.”
“The scientific and clinical data supporting lasofoxifene are compelling, and we are confident in Athira’s leadership to drive the Company’s next chapter with clarity, urgency and excellence. We’re proud to support this evolution and excited by the opportunity to deliver meaningful impact for patients and shareholders alike," stated Joseph Edelman, Founder and CEO of Perceptive Advisors.
Details of the Transaction
Sermonix License Agreement
In connection with the entry into the Sermonix license agreement, Athira will issue to Sermonix, as partial consideration, a pre-funded warrant to purchase approximately 5.5 million shares of common stock, with an exercise price of $0.001 per share. Athira will also be obligated to make certain payments to Sermonix of up to $100.0 million if Athira achieves certain commercialization or annual net sales milestones with respect to licensed products with respect to lasofoxifene, including royalty payments on the net sales of any licensed products in any licensed territory, ranging from sub-single digit to low-single digit royalties depending on pre-specified net sales.
Financing
On December 18, 2025, Athira entered into a securities purchase agreement with certain investors, pursuant to which Athira has agreed to issue and sell approximately 5.4 million shares of common stock, pre-funded warrants to purchase approximately 8.8 million shares of common stock and accompanying warrants to purchase approximately 23.0 million shares of common
stock and/or pre-funded warrants (representing 162.5% of the aggregate shares and shares underlying the pre-funded warrants), with an exercise price of $6.35 per share (the “Series A common warrants”), and accompanying warrants to purchase approximately 21.3 million shares of common stock and/or pre-funded warrants (representing 150% of the aggregate shares and shares underlying the pre-funded warrants), with an exercise price of $7.62 per share (the “Series B common warrants”) (the “Private Placement”). The common stock, including the accompanying Series A common warrants and Series B common warrants, will be sold at a price of $6.35 per share, and the pre-funded warrants, including the accompanying Series A common warrants and Series B common warrants, will be sold at the price per underlying pre-funded warrant share of the common stock less the exercise price of $0.001 per share.
The Private Placement is expected to close on or about December 23, 2025, subject to the satisfaction of customary closing conditions. Additional details regarding the Private Placement and the other transactions discussed herein will be included in a Current Report on Form 8-K to be filed by Athira with the Securities and Exchange Commission (“SEC”).
Athira intends to use the net proceeds from the Private Placement to fund the development of lasofoxifene for the potential treatment of treatment-resistant metastatic breast cancer, and other clinical assets in its pipeline, such as ATH-1105 as a treatment for ALS, and for working capital and general corporate purposes.
Cantor is acting as exclusive financial advisor to Athira in connection with the license transaction and sole placement agent in connection with the Private Placement.
The securities being sold in the private placements discussed herein have not been registered under the Securities Act of 1933, as amended (the “Securities Act”), or state securities laws and may not be offered or sold in the United States absent registration with the SEC or an applicable exemption from applicable registration requirements. Athira has agreed to file registration statements with the SEC covering the resale of the shares of common stock issuable in connection with the private placements and upon exercise of the pre-funded warrants and warrants.
This press release shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction.
Conference Call Details
Athira management will host a conference call and webcast today, December 18, 2025, at 8:30 AM Eastern Time to discuss these transactions and answer questions. A live webcast of the conference call can be accessed at https://edge.media-server.com/mmc/p/zwdrpi3i and in the “Events & Presentations” section of Athira’s website at www.athira.com. A recording of the webcast will be archived on the company’s website for approximately 90 days.
About Metastatic Breast Cancer
Metastatic breast cancer (MBC) occurs when cancer spreads from the breast to other parts of the body—such as bones, lungs, liver or brain. While approximately 66% of breast cancers are diagnosed at a localized stage, a notable proportion either present as metastatic at diagnosis or progress to that stage over time.
In the United States, there are approximately 4.65 million new cases of female breast cancer, with approximately 260,000 (5.6%) diagnosed as distant (metastatic) stage at initial diagnosis. The metastatic breast cancer treatment market represents a sizable and rapidly expanding global opportunity with a global market of $17.1 billion in 2021, expected to expand to $41.7 billion by 2030, with a CAGR of around 10.4%.
These projections reflect a market rich with innovation—from chemotherapy and hormone therapies to biologics, targeted agents and emerging personalized medicine. Growth is driven by the persistent incidence of metastatic disease, regulatory and clinical advances and evolving treatment landscapes.
About Lasofoxifene
Lasofoxifene is a novel, nonsteroidal selective estrogen receptor modulator (SERM) with a unique binding profile, designed to confer potent activity against both wild-type and mutant estrogen receptors, including the clinically significant ESR1 mutations commonly associated with resistance to endocrine therapy in metastatic breast cancer. Two Phase 2 studies—ELAINE‑1 and ELAINE‑2—have demonstrated its potential to address a critical unmet need in this patient population.
ELAINE‑1, a randomized trial comparing lasofoxifene to fulvestrant, showed improved outcomes for lasofoxifene, including longer median progression-free survival (5.6 vs. 3.7 months), higher objective response rates (13.3% vs. 2.9%), and a durable complete response lasting more than 2.5 years. Patients also reported quality-of-life benefits and the treatment was well tolerated.
ELAINE‑2, an open-label study evaluating lasofoxifene in combination with abemaciclib, demonstrated clinical benefits in heavily pretreated patients, with a median progression-free survival of approximately 13 months, an objective response rate of 56%, and a clinical benefit rate of 65.5%. The combination was generally well tolerated, with most adverse events being low grade.
Lasofoxifene is being advanced in a Phase 3 clinical trial as a targeted therapy for estrogen receptor-positive (ER+), HER2-negative, ESR1-mutated metastatic breast cancer, a population with limited treatment options following progression on aromatase inhibitors and CDK4/6 inhibitors. The ongoing ELAINE-3 trial (NCT05696626) is evaluating lasofoxifene in combination with the CDK4/6 inhibitor, abemaciclib, and is aiming to establish a new standard of care for this genetically defined patient group.
About ATH-1105
ATH-1105 is Athira’s novel, orally available, brain-penetrant, next-generation small molecule drug candidate designed to positively modulate the neurotrophic HGF system for potential treatment of neurodegenerative diseases, including amyotrophic lateral sclerosis (ALS), Alzheimer’s disease and Parkinson’s disease. ATH-1105 is currently in clinical development for the potential treatment of ALS.
In May 2025, Athira presented data from the first-in-human Phase 1 clinical trial (NCT06432647) of ATH-1105 at the 4th Annual ALS Drug Development Summit highlighting ATH-1105 has demonstrated consistent and robust beneficial effects in preclinical models of ALS and showed a favorable safety profile and was well tolerated in both single and multiple ascending dose studies in healthy volunteers. In addition, ATH-1105 showed dose proportional pharmacokinetics and central nervous system penetration in this first-in-human study.
The first-in-human Phase 1 (NCT06432647) double-blind, placebo-controlled clinical trial enrolled 80 healthy volunteers to evaluate single and multiple oral ascending doses of ATH-1105. Athira plans to initiate a Phase 2 clinical trial of ATH-1105 in ALS patients in early 2026.
About Athira
Athira Pharma, Inc., headquartered in the Seattle, Washington area, is a clinical-stage biopharmaceutical company dedicated to the development of novel therapeutics for high unmet medical needs, including amyotrophic lateral sclerosis (ALS) and treatment-resistant metastatic breast cancer, with the goal of improving patients’ lives. Our lead drug candidates, lasofoxifene and ATH-1105, are novel, small molecule therapies with the potential to address devastating diseases where current treatment options are limited or ineffective. With a strong commitment to scientific excellence and patient-centered innovation, we are dedicated to developing meaningful new therapies for those who need them most.
For more information, visit www.athira.com. You can also follow Athira on Facebook, LinkedIn, X (formerly known as Twitter) and Instagram.
Forward-Looking Statements
This communication contains “forward-looking statements” within the meaning of Section 27A of the Securities Act, Section 21E of the Securities Exchange Act of 1934 and the Private Securities Litigation Reform Act of 1995. These forward-looking statements are not based on historical fact and include statements regarding: the beneficial characteristics, safety and efficacy of Athira’s drug candidates; the anticipated consummation of the transactions described herein; Athira’s ability to obtain funding for its operations, including funding necessary to develop and commercialize its drug candidates and funding necessary for the payment of future milestone and other payments that may be earned by its collaboration partners; the success, cost and timing of Athira’s development activities, nonclinical studies and clinical trials; the potential of any subsequent clinical trials to show the beneficial characteristics, safety and efficacy of ATH-1105; the potential of Athira to complete the Phase 3 ELAINE-3 clinical trial for lasofoxifene and any subsequent clinical trials to show the clinical benefits of lasofoxifene; the potential learnings from preclinical studies and other nonclinical data and their ability to inform and improve future clinical development plans; the rate and degree of market acceptance of Athira’s drug candidates; the size and growth potential of the markets for Athira’s drug candidates, if approved for commercial use, and Athira’s ability to serve those markets; anticipated milestone timelines, such as the timing of data releases, and Athira’s ability to meet such timelines; Athira’s ability to obtain and maintain orphan drug designations for any product candidates for which it may seek such designation; the potential for lasofoxifene to be a new standard of care in the genetically defined patient group; Athira’s ability to obtain and maintain regulatory approval of its drug
candidates in the United States and other jurisdictions and the timing thereof, and any related restrictions, limitations or warnings in the label of any approved drug candidate; and Athira’s expected rebranding efforts. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as “may,” “will,” “should,” “on track,” “would,” “expect,” “plan,” “believe,” “intend,” “pursue,” “continue,” “suggest,” “potential,” “target” and similar expressions. Any forward-looking statements are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements. These risks and uncertainties include, but are not limited to, the risk that the conditions to the closing of any or all of the transactions are not satisfied; uncertainties as to the timing of the consummation of the proposed transactions and the ability of each of the parties thereto to consummate them; unexpected costs, charges or expenses resulting from the transactions; potential adverse reactions or changes to business relationships resulting from the announcement or completion of any or all of the proposed transactions; risks associated with the possible failure to realize certain anticipated benefits of any or all of the proposed transactions, including with respect to future financial and operating results; the data from preclinical and clinical trials may not support the safety, efficacy and tolerability of Athira’s drug candidates; development of drug candidates may cease or be delayed; regulatory authorities could object to protocols, amendments and other submissions; future potential regulatory milestones for drug candidates, including those related to current and planned clinical studies, may be insufficient to support regulatory submissions or approval; whether Athira’s trials are sufficiently powered to meet the planned endpoints; Athira may not be able to recruit sufficient patients for its clinical trials; the outcome of legal proceedings that may in the future be instituted against Athira, its directors and officers; possible negative interactions of Athira's drug candidates with other treatments; FDA regulatory delays and uncertainty and new policies, including executive orders, changes in the leadership of federal agencies such as the FDA and SEC, staff layoffs, budget cuts to agency programs and research and changes in drug pricing controls; Athira’s assumptions regarding its financial condition and the sufficiency of its cash, cash equivalents and investments to fund its planned operations may be incorrect; adverse conditions in the general domestic and global economic markets, including as a result of tariffs; the impact of competition; the impact of drug candidate development and clinical activities on operating expenses; the impact of new or changing laws and regulations; as well as the other risks detailed in Athira’s filings with the SEC from time to time. These forward-looking statements speak only as of the date hereof and Athira undertakes no obligation to update forward-looking statements. Athira may not actually achieve the plans, intentions or expectations disclosed in its forward-looking statements, and you should not place undue reliance on the forward-looking statements.
Investor & Media Contact:
Julie Rathbun
Athira Pharma
Julie.rathbun@athira.com
206-769-9219
