| 1 Envudeucitinib (ESK-001) in Moderate-to-Severe Plaque Psoriasis: 24-Week Results From the Randomized, Double-Blind, Active Comparator- and Placebo-Controlled, Phase 3 ONWARD 1 and 2 Studies Andrew Blauvelt1, Howard Sofen2, April Armstrong3, Benjamin Ehst4-6 , Jennifer Soung7, Maryam Shayesteh Alam8, David Rodriguez9, Jolanta Weglowska10 , Domenico Vitarella11, Grace Ma11, Elisa Muscianisi11 1Blauvelt Consulting, LLC, Annapolis, MD, USA; 2Department of Medicine/Dermatology, David Geffen School of Medicine at UCLA, Los Angeles, CA, USA; 3Division of Dermatology, David Geffen School of Medicine at UCLA, Los Angeles, CA, USA; 4Oregon Medical Research Center, Portland, OR, USA; 5Broadway Medical Clinic, Portland OR, USA; 6Oregon Health & Science University, Portland, OR, USA; 7Southern California Dermatology Inc., Santa Ana, CA, USA; 8SimcoDerm Medical and Surgical Dermatology Centre, Barrie, ON, Canada; 9International Dermatology Research, Inc., Miami, FL, USA; 10Department of Dermatology, Research and Development Center, Regional Specialist Hospital, Wrocław, Poland; 11Alumis Inc., South San Francisco, CA, USA. Exhibit 99.2 |
| 2 Disclosures 〉 Presenting author: AB has served as a speaker for and received honoraria from Almirall, Eli Lilly, LEO Pharma, Sanofi, and UCB; has served as a scientific adviser for and received honoraria from AbbVie, Almirall, Alumis Inc., Amgen, AnaptysBio, Apogee Therapeutics, Arcutis Biotherapeutics, Eli Lilly, Incyte, Janssen, LEO Pharma, Novartis, Oruka Therapeutics, Pfizer, Regeneron Pharmaceuticals, Sanofi, Sun Pharma, Takeda, and UCB; and owns stock in Lipidio Pharma and Oruka Therapeutics 〉 Coauthors (relevant to study): HS has nothing to report. AA has served as a research investigator, scientific adviser, or speaker for Alumis Inc. BE has received fees/honoraria/royalties as an advisory board member, contributor, and/or consultant for Alumis Inc., and received institutional funding as an investigator for Alumis Inc. JS has served as an investigator for Alumis Inc. MSA and DR have nothing relevant to disclose. JW has served as an investigator for Alumis Inc. DV, GM, and EM are employees and shareholders of Alumis Inc. 〉 All authors met the ICMJE authorship criteria and had full access to relevant data 〉 The ONWARD program is currently ongoing, and these studies were sponsored by Alumis Inc. 〉 Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency ICMJE, International Committee of Medical Journal Editors. |
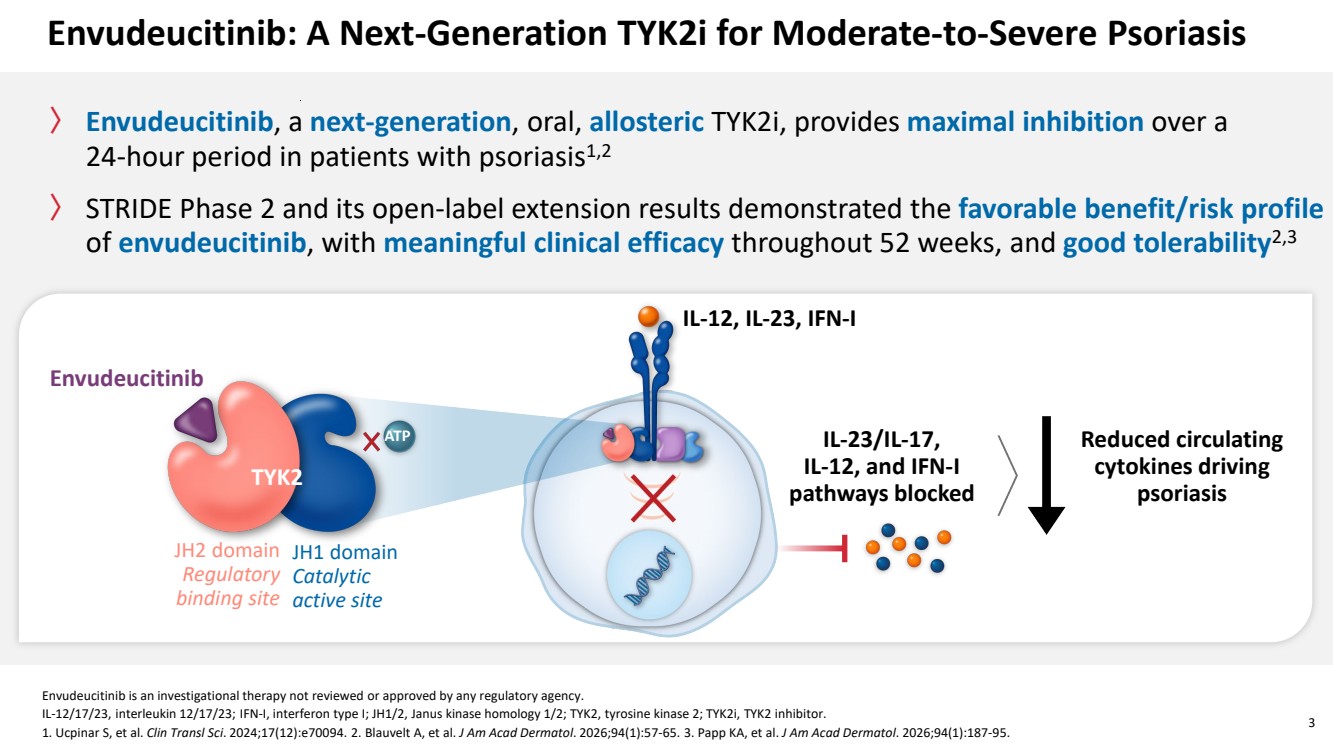
| Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency. IL-12/17/23, interleukin 12/17/23; IFN-I, interferon type I; JH1/2, Janus kinase homology 1/2; TYK2, tyrosine kinase 2; TYK2i, TYK2 inhibitor. 1. Ucpinar S, et al. Clin Transl Sci. 2024;17(12):e70094. 2. Blauvelt A, et al. J Am Acad Dermatol. 2026;94(1):57-65. 3. Papp KA, et al. J Am Acad Dermatol. 2026;94(1):187-95. 3 Envudeucitinib: A Next-Generation TYK2i for Moderate-to-Severe Psoriasis 〉 Envudeucitinib, a next-generation, oral, allosteric TYK2i, provides maximal inhibition over a 24-hour period in patients with psoriasis1,2 〉 STRIDE Phase 2 and its open-label extension results demonstrated the favorable benefit/risk profile of envudeucitinib, with meaningful clinical efficacy throughout 52 weeks, and good tolerability2,3 Envudeucitinib TYK2 ATP JH2 domain Regulatory binding site JH1 domain Catalytic active site IL-12, IL-23, IFN-I IL-23/IL-17, IL-12, and IFN-I pathways blocked Reduced circulating cytokines driving psoriasis |
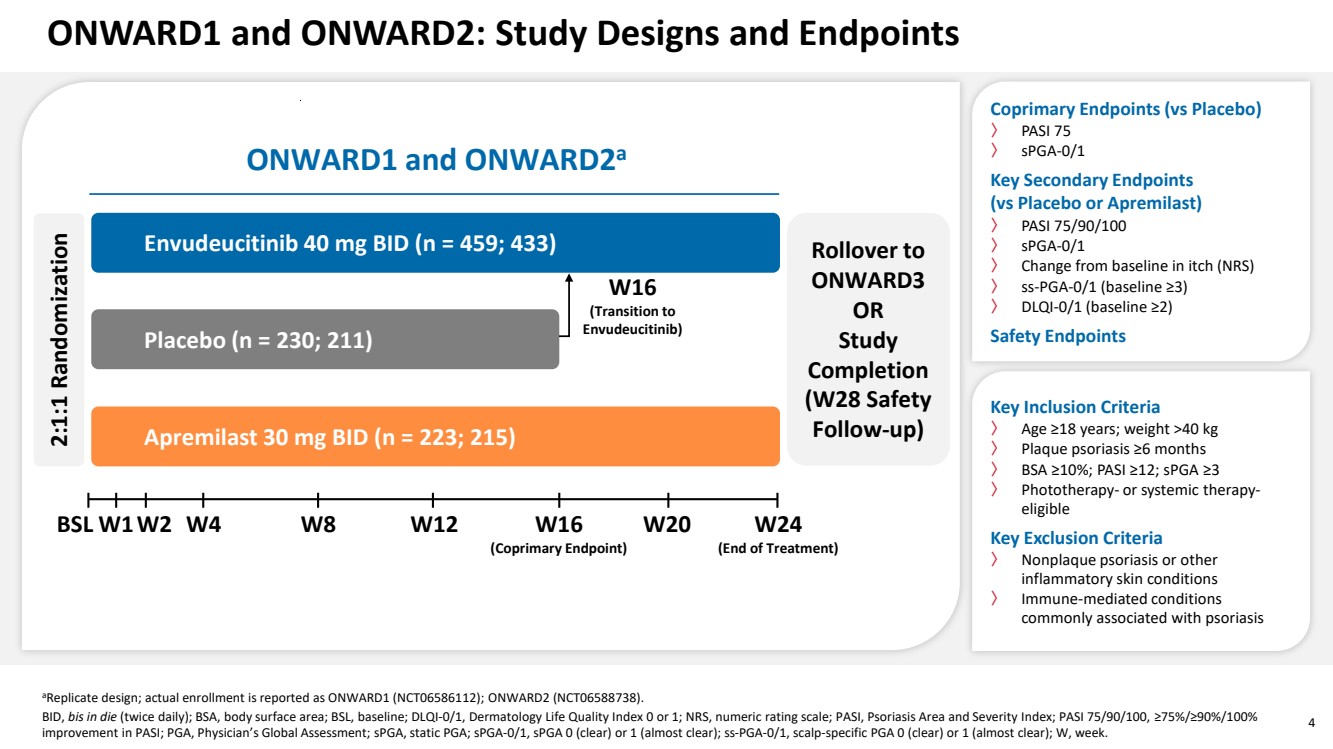
| 4 ONWARD1 and ONWARD2: Study Designs and Endpoints aReplicate design; actual enrollment is reported as ONWARD1 (NCT06586112); ONWARD2 (NCT06588738). BID, bis in die (twice daily); BSA, body surface area; BSL, baseline; DLQI-0/1, Dermatology Life Quality Index 0 or 1; NRS, numeric rating scale; PASI, Psoriasis Area and Severity Index; PASI 75/90/100, ≥75%/≥90%/100% improvement in PASI; PGA, Physician’s Global Assessment; sPGA, static PGA; sPGA-0/1, sPGA 0 (clear) or 1 (almost clear); ss-PGA-0/1, scalp-specific PGA 0 (clear) or 1 (almost clear); W, week. BSL W24 (End of Treatment) W16 (Coprimary Endpoint) Placebo (n = 230; 211) Envudeucitinib 40 mg BID (n = 459; 433) 2:1:1 Randomization Apremilast 30 mg BID (n = 223; 215) Rollover to ONWARD3 OR Study Completion (W28 Safety Follow-up) W16 (Transition to Envudeucitinib) W2 W4 W8 W12 W20 ONWARD1 and ONWARD2a W1 Coprimary Endpoints (vs Placebo) 〉 PASI 75 〉 sPGA-0/1 Key Secondary Endpoints (vs Placebo or Apremilast) 〉 PASI 75/90/100 〉 sPGA-0/1 〉 Change from baseline in itch (NRS) 〉 ss-PGA-0/1 (baseline ≥3) 〉 DLQI-0/1 (baseline ≥2) Safety Endpoints Key Inclusion Criteria 〉 Age ≥18 years; weight >40 kg 〉 Plaque psoriasis ≥6 months 〉 BSA ≥10%; PASI ≥12; sPGA ≥3 〉 Phototherapy- or systemic therapy-eligible Key Exclusion Criteria 〉 Nonplaque psoriasis or other inflammatory skin conditions 〉 Immune-mediated conditions commonly associated with psoriasis |
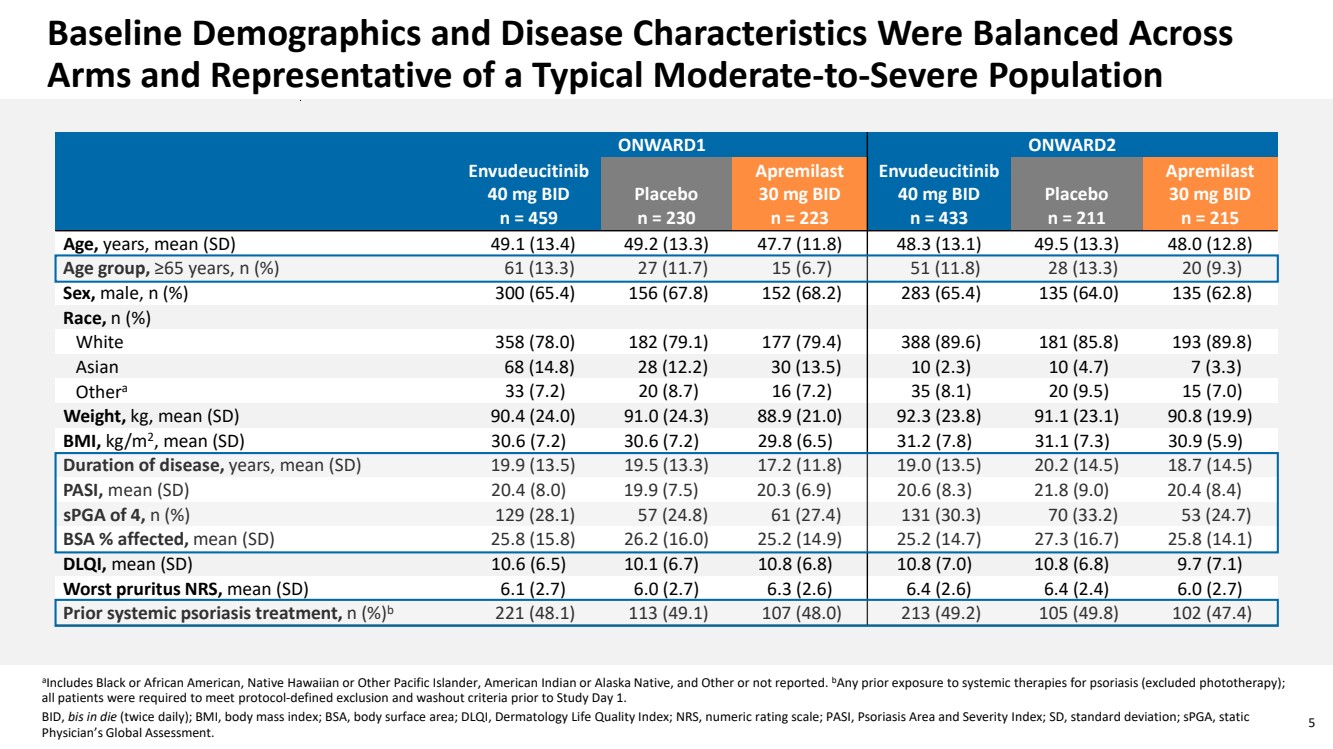
| 5 Baseline Demographics and Disease Characteristics Were Balanced Across Arms and Representative of a Typical Moderate-to-Severe Population aIncludes Black or African American, Native Hawaiian or Other Pacific Islander, American Indian or Alaska Native, and Other or not reported. bAny prior exposure to systemic therapies for psoriasis (excluded phototherapy); all patients were required to meet protocol-defined exclusion and washout criteria prior to Study Day 1. BID, bis in die (twice daily); BMI, body mass index; BSA, body surface area; DLQI, Dermatology Life Quality Index; NRS, numeric rating scale; PASI, Psoriasis Area and Severity Index; SD, standard deviation; sPGA, static Physician’s Global Assessment. ONWARD1 ONWARD2 Envudeucitinib 40 mg BID n = 459 Placebo n = 230 Apremilast 30 mg BID n = 223 Envudeucitinib 40 mg BID n = 433 Placebo n = 211 Apremilast 30 mg BID n = 215 Age, years, mean (SD) 49.1 (13.4) 49.2 (13.3) 47.7 (11.8) 48.3 (13.1) 49.5 (13.3) 48.0 (12.8) Age group, ≥65 years, n (%) 61 (13.3) 27 (11.7) 15 (6.7) 51 (11.8) 28 (13.3) 20 (9.3) Sex, male, n (%) 300 (65.4) 156 (67.8) 152 (68.2) 283 (65.4) 135 (64.0) 135 (62.8) Race, n (%) White 358 (78.0) 182 (79.1) 177 (79.4) 388 (89.6) 181 (85.8) 193 (89.8) Asian 68 (14.8) 28 (12.2) 30 (13.5) 10 (2.3) 10 (4.7) 7 (3.3) Othera 33 (7.2) 20 (8.7) 16 (7.2) 35 (8.1) 20 (9.5) 15 (7.0) Weight, kg, mean (SD) 90.4 (24.0) 91.0 (24.3) 88.9 (21.0) 92.3 (23.8) 91.1 (23.1) 90.8 (19.9) BMI, kg/m2, mean (SD) 30.6 (7.2) 30.6 (7.2) 29.8 (6.5) 31.2 (7.8) 31.1 (7.3) 30.9 (5.9) Duration of disease, years, mean (SD) 19.9 (13.5) 19.5 (13.3) 17.2 (11.8) 19.0 (13.5) 20.2 (14.5) 18.7 (14.5) PASI, mean (SD) 20.4 (8.0) 19.9 (7.5) 20.3 (6.9) 20.6 (8.3) 21.8 (9.0) 20.4 (8.4) sPGA of 4, n (%) 129 (28.1) 57 (24.8) 61 (27.4) 131 (30.3) 70 (33.2) 53 (24.7) BSA % affected, mean (SD) 25.8 (15.8) 26.2 (16.0) 25.2 (14.9) 25.2 (14.7) 27.3 (16.7) 25.8 (14.1) DLQI, mean (SD) 10.6 (6.5) 10.1 (6.7) 10.8 (6.8) 10.8 (7.0) 10.8 (6.8) 9.7 (7.1) Worst pruritus NRS, mean (SD) 6.1 (2.7) 6.0 (2.7) 6.3 (2.6) 6.4 (2.6) 6.4 (2.4) 6.0 (2.7) Prior systemic psoriasis treatment, n (%)b 221 (48.1) 113 (49.1) 107 (48.0) 213 (49.2) 105 (49.8) 102 (47.4) |
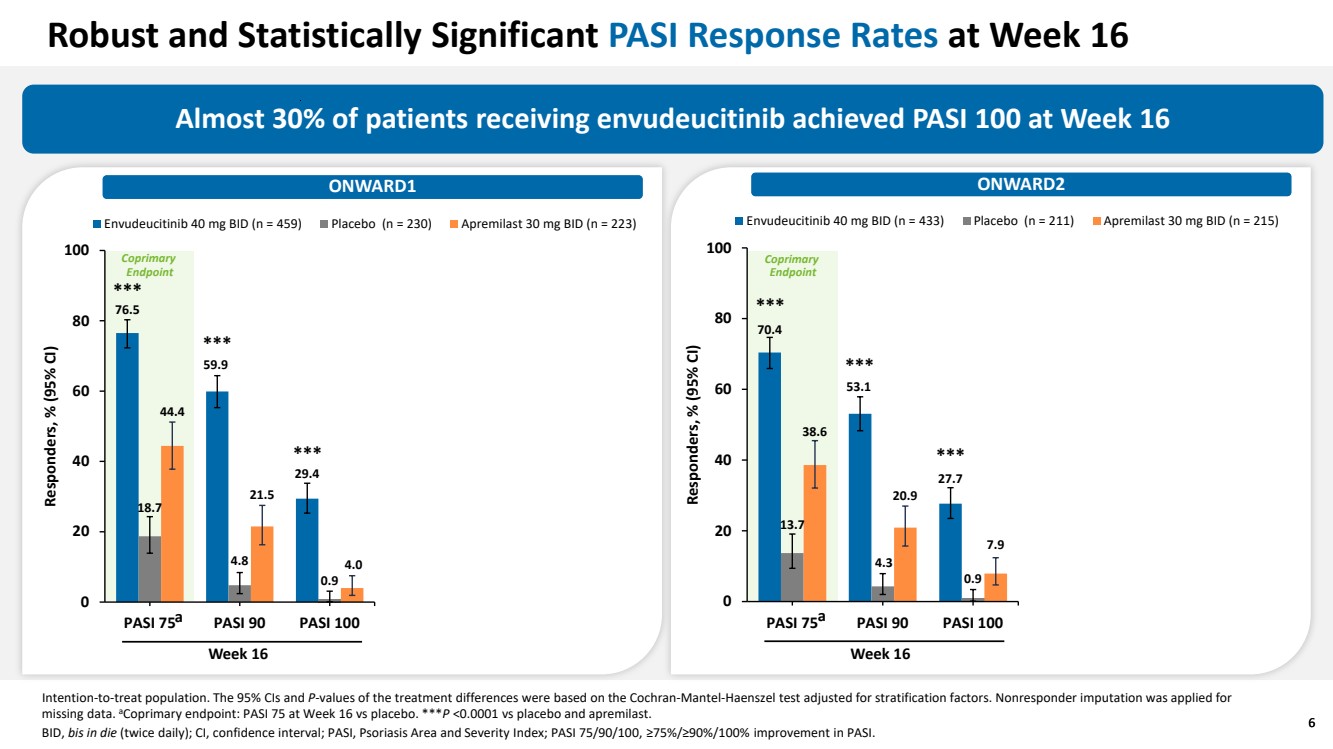
| 6 Robust and Statistically Significant PASI Response Rates at Week 16 Almost 30% of patients receiving envudeucitinib achieved PASI 100 at Week 16 ONWARD1 ONWARD2 Intention-to-treat population. The 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. aCoprimary endpoint: PASI 75 at Week 16 vs placebo. ***P <0.0001 vs placebo and apremilast. BID, bis in die (twice daily); CI, confidence interval; PASI, Psoriasis Area and Severity Index; PASI 75/90/100, ≥75%/≥90%/100% improvement in PASI. 6 ONWARD2 70.4 53.1 27.7 13.7 4.3 0.9 38.6 20.9 7.9 0 20 40 60 80 100 PASI 75 PASI 90 PASI 100 Responders, % (95% CI) Envudeucitinib 40 mg BID (n = 433) Placebo (n = 211) Apremilast 30 mg BID (n = 215) Week 16 *** *** *** ONWARD1 Week 16 *** *** *** a a 76.5 59.9 29.4 18.7 4.8 0.9 44.4 21.5 4.0 0 20 40 60 80 100 PASI 75 PASI 90 PASI 100 Responders, % (95% CI) Envudeucitinib 40 mg BID (n = 459) Placebo (n = 230) Apremilast 30 mg BID (n = 223) Coprimary Endpoint Coprimary Endpoint |
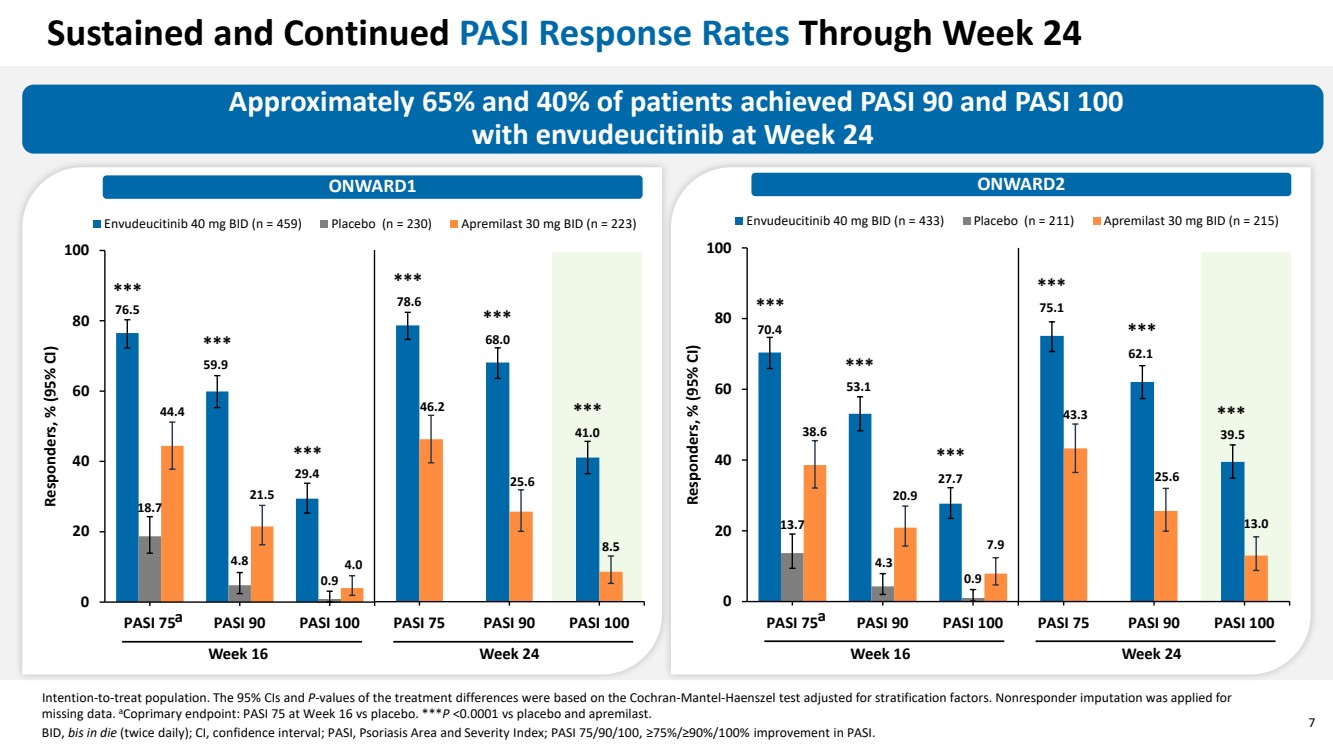
| 7 Approximately 65% and 40% of patients achieved PASI 90 and PASI 100 with envudeucitinib at Week 24 ONWARD2 Intention-to-treat population. The 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. aCoprimary endpoint: PASI 75 at Week 16 vs placebo. ***P <0.0001 vs placebo and apremilast. BID, bis in die (twice daily); CI, confidence interval; PASI, Psoriasis Area and Severity Index; PASI 75/90/100, ≥75%/≥90%/100% improvement in PASI. 70.4 53.1 27.7 13.7 4.3 0.9 38.6 20.9 7.9 0 20 40 60 80 100 PASI 75 PASI 90 PASI 100 Responders, % (95% CI) Envudeucitinib 40 mg BID (n = 433) Placebo (n = 211) Apremilast 30 mg BID (n = 215) Week 16 Week 24 Sustained and Continued PASI Response Rates Through Week 24 *** *** *** *** *** *** ONWARD1 76.5 59.9 29.4 18.7 4.8 0.9 44.4 21.5 4.0 0 20 40 60 80 100 PASI 75 PASI 90 PASI 100 Responders, % (95% CI) Envudeucitinib 40 mg BID (n = 459) Placebo (n = 230) Apremilast 30 mg BID (n = 223) Week 16 Week 24 *** *** *** *** *** 78.6 68.0 41.0 46.2 25.6 8.5 PASI 75 PASI 90 PASI 100 75.1 62.1 39.5 43.3 25.6 13.0 PASI 75 PASI 90 PASI 100 a a *** |
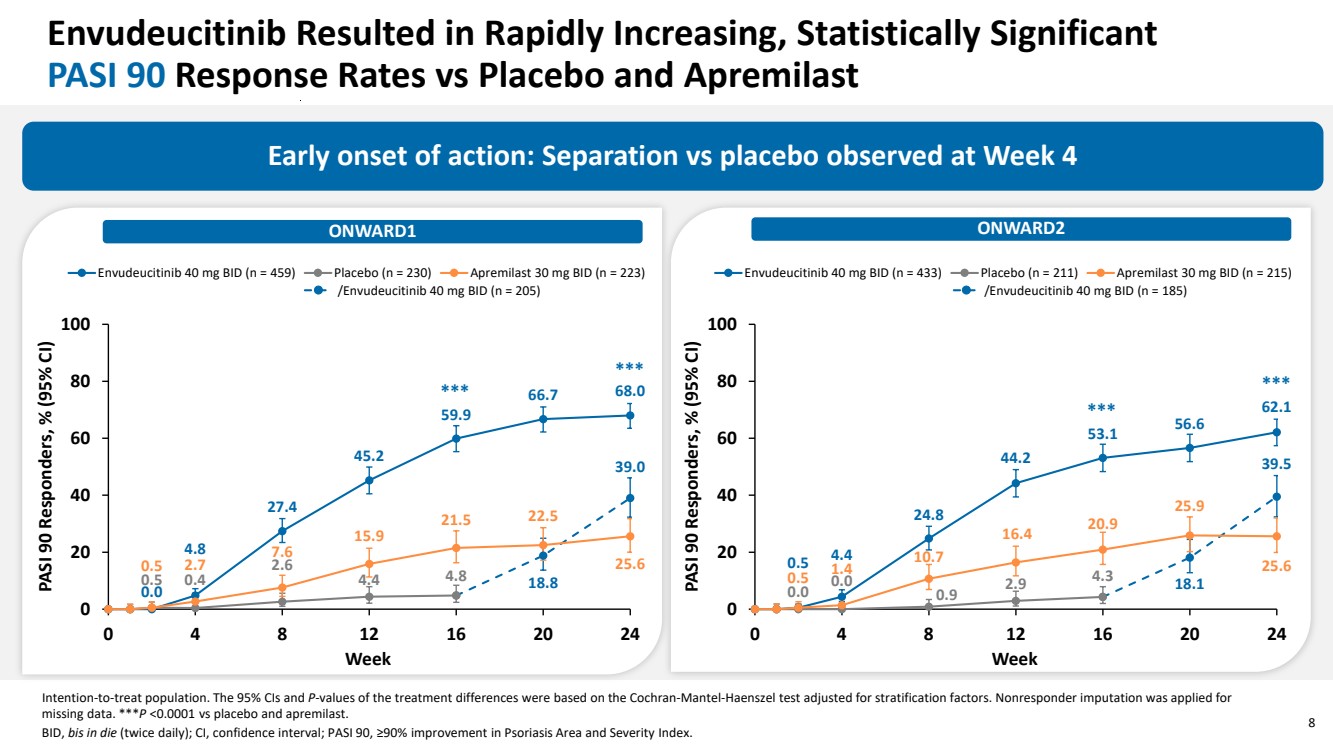
| 8 Envudeucitinib Resulted in Rapidly Increasing, Statistically Significant PASI 90 Response Rates vs Placebo and Apremilast Early onset of action: Separation vs placebo observed at Week 4 Intention-to-treat population. The 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. ***P <0.0001 vs placebo and apremilast. BID, bis in die (twice daily); CI, confidence interval; PASI 90, ≥90% improvement in Psoriasis Area and Severity Index. 0.0 4.8 27.4 45.2 59.9 66.7 68.0 0.5 0.4 2.6 4.4 4.8 18.8 39.0 0.5 2.7 7.6 15.9 21.5 22.5 25.6 0 20 40 60 80 100 0 4 8 12 16 20 24 PASI 90 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 459) Placebo (n = 230) Apremilast 30 mg BID (n = 223) /Envudeucitinib 40 mg BID (n = 205) ONWARD1 ONWARD2 *** *** 0.5 4.4 24.8 44.2 53.1 56.6 62.1 0.0 0.0 0.9 2.9 4.3 18.1 39.5 0.5 1.4 10.7 16.4 20.9 25.9 25.6 0 20 40 60 80 100 0 4 8 12 16 20 24 PASI 90 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 433) Placebo (n = 211) Apremilast 30 mg BID (n = 215) /Envudeucitinib 40 mg BID (n = 185) *** *** |
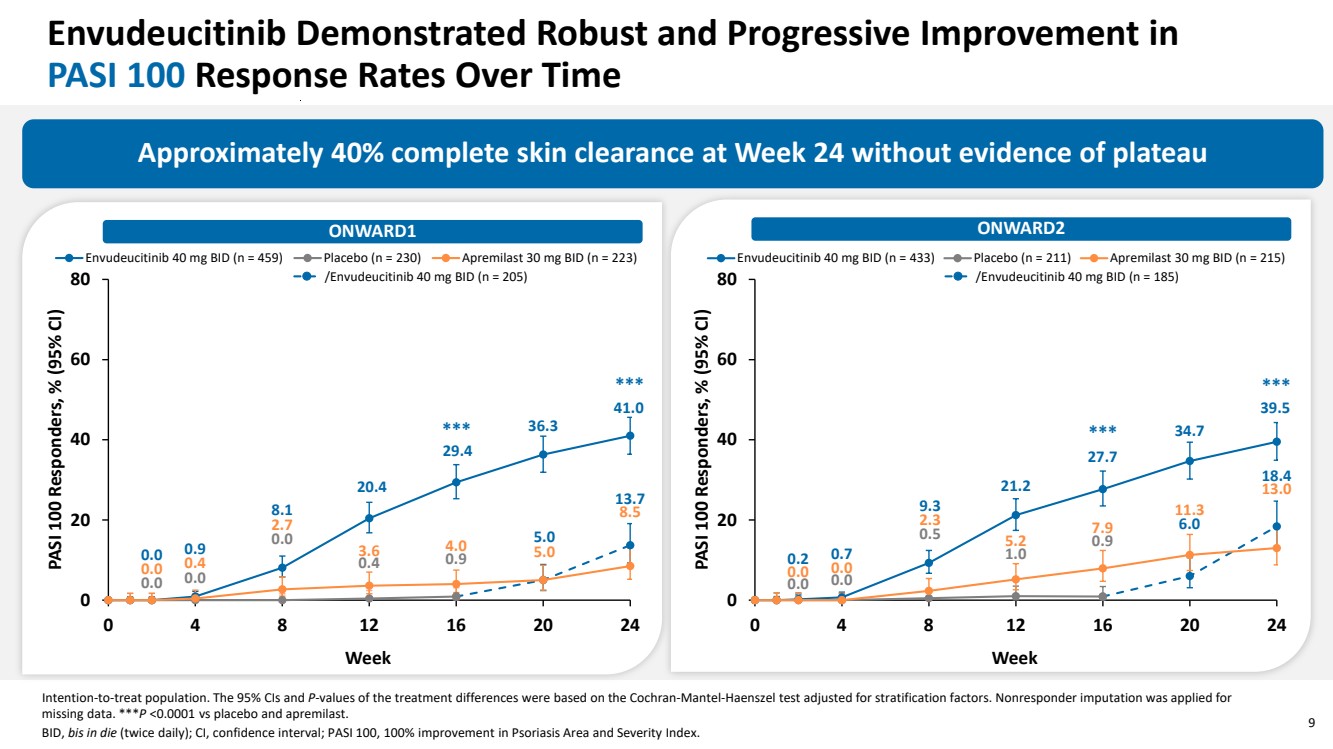
| 9 Envudeucitinib Demonstrated Robust and Progressive Improvement in PASI 100 Response Rates Over Time /Envudeucitinib 40 mg BID (n = 205) Approximately 40% complete skin clearance at Week 24 without evidence of plateau Intention-to-treat population. The 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. ***P <0.0001 vs placebo and apremilast. BID, bis in die (twice daily); CI, confidence interval; PASI 100, 100% improvement in Psoriasis Area and Severity Index. *** *** 0.2 0.7 9.3 21.2 27.7 34.7 39.5 0.0 0.0 0.5 1.0 0.9 6.0 18.4 0.0 0.0 2.3 5.2 7.9 11.3 13.0 0 20 40 60 80 0 4 8 12 16 20 24 PASI 100 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 433) Placebo (n = 211) Apremilast 30 mg BID (n = 215) /Envudeucitinib 40 mg BID (n = 185) *** *** ONWARD1 ONWARD2 0.0 0.9 8.1 20.4 29.4 36.3 41.0 0.0 0.0 0.0 0.4 0.9 5.0 13.7 0.0 0.4 2.7 3.6 4.0 5.0 8.5 0 20 40 60 80 0 4 8 12 16 20 24 PASI 100 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 459) Placebo (n = 230) Apremilast 30 mg BID (n = 223) |
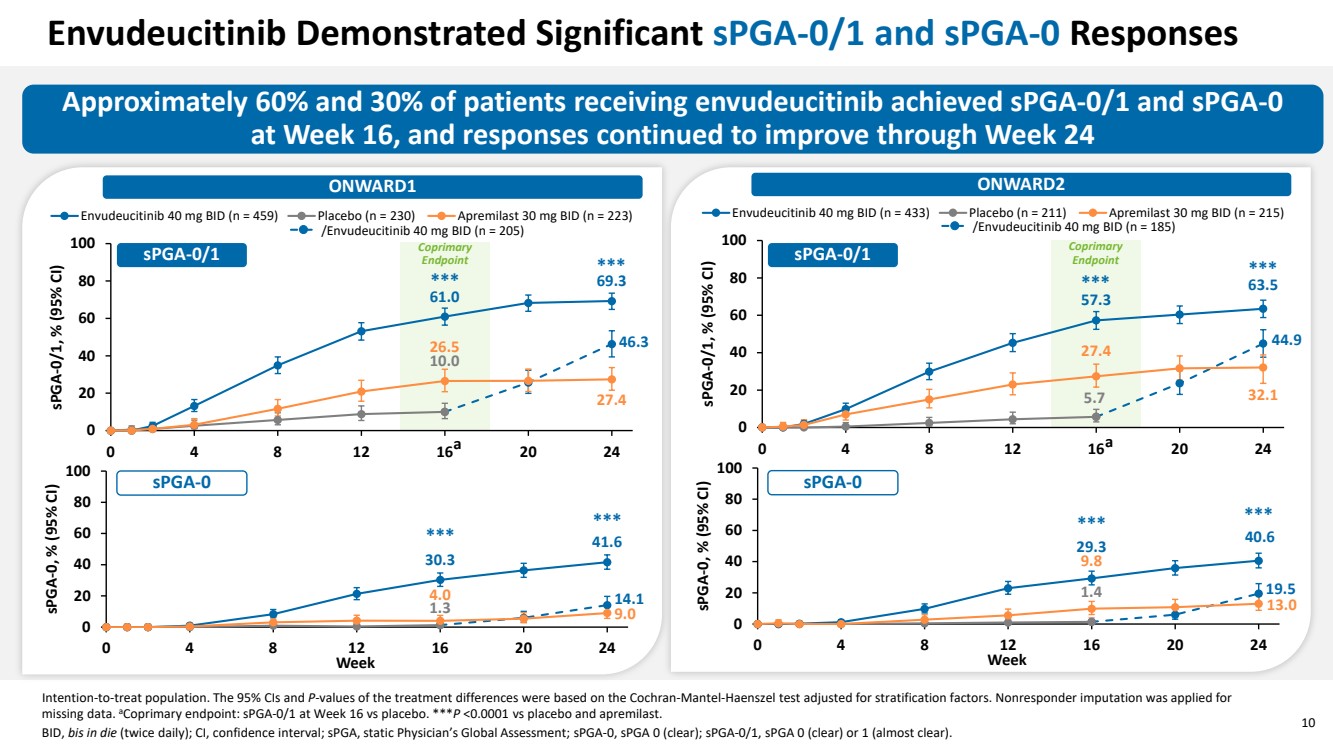
| 10 Envudeucitinib Demonstrated Significant sPGA-0/1 and sPGA-0 Responses Approximately 60% and 30% of patients receiving envudeucitinib achieved sPGA-0/1 and sPGA-0 at Week 16, and responses continued to improve through Week 24 ONWARD1 ONWARD2 Intention-to-treat population. The 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. aCoprimary endpoint: sPGA-0/1 at Week 16 vs placebo. ***P <0.0001 vs placebo and apremilast. BID, bis in die (twice daily); CI, confidence interval; sPGA, static Physician’s Global Assessment; sPGA-0, sPGA 0 (clear); sPGA-0/1, sPGA 0 (clear) or 1 (almost clear). 61.0 69.3 10.0 46.3 26.5 27.4 0 20 40 60 80 100 0 4 8 12 16 20 24 sPGA-0/1, % (95% CI) Envudeucitinib 40 mg BID (n = 459) Placebo (n = 230) Apremilast 30 mg BID (n = 223) 1.3 30.3 41.6 4.0 14.1 9.0 0 20 40 60 80 100 0 4 8 12 16 20 24 sPGA-0, % (95% CI) Week *** *** *** *** 57.3 63.5 5.7 44.9 27.4 32.1 0 20 40 60 80 100 0 4 8 12 16 20 24 sPGA-0/1, % (95% CI) Envudeucitinib 40 mg BID (n = 433) Placebo (n = 211) Apremilast 30 mg BID (n = 215) 1.4 29.3 40.6 19.5 9.8 13.0 0 20 40 60 80 100 0 4 8 12 16 20 24 sPGA-0, % (95% CI) Week *** *** *** *** /Envudeucitinib 40 mg BID (n = 205) /Envudeucitinib 40 mg BID (n = 185) a a sPGA-0/1 sPGA-0/1 sPGA-0 sPGA-0 Coprimary Endpoint Coprimary Endpoint |
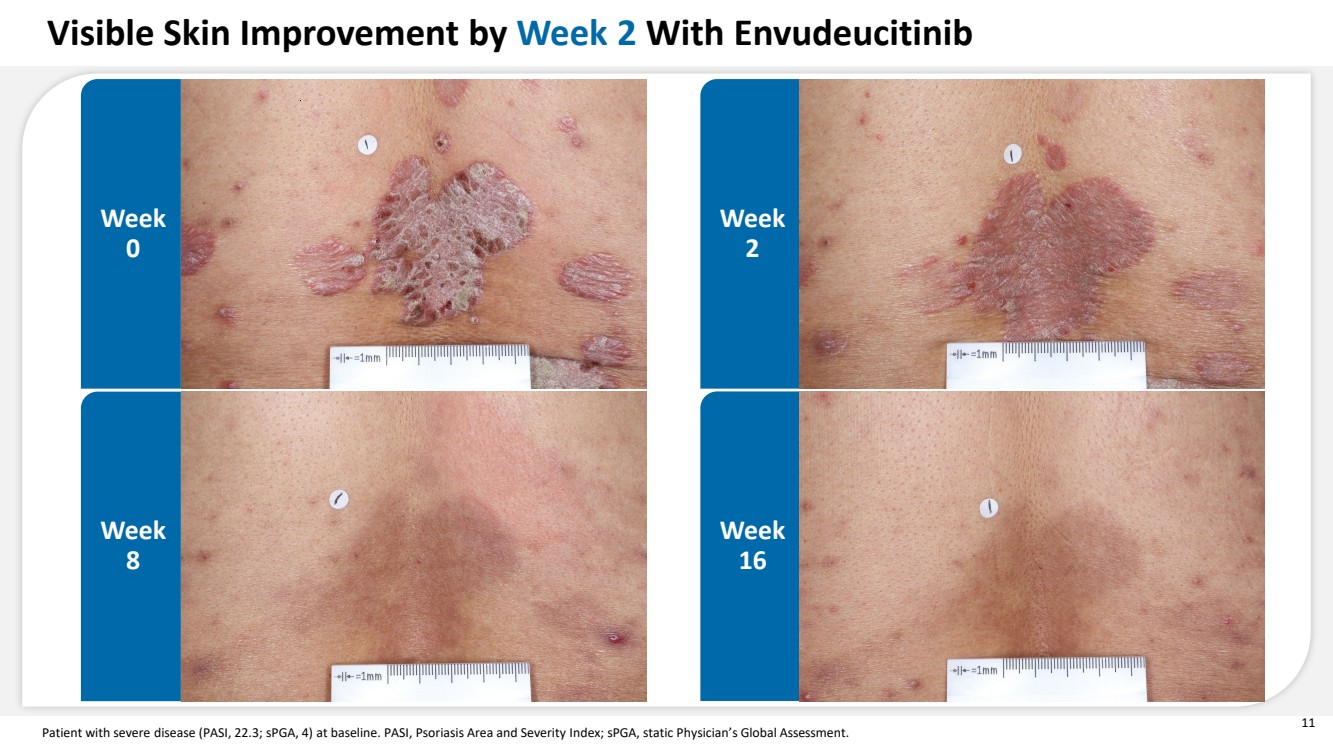
| 11 Patient with severe disease (PASI, 22.3; sPGA, 4) at baseline. PASI, Psoriasis Area and Severity Index; sPGA, static Physician’s Global Assessment. Week 0 Week 2 Week 8 Week 16 Visible Skin Improvement by Week 2 With Envudeucitinib |
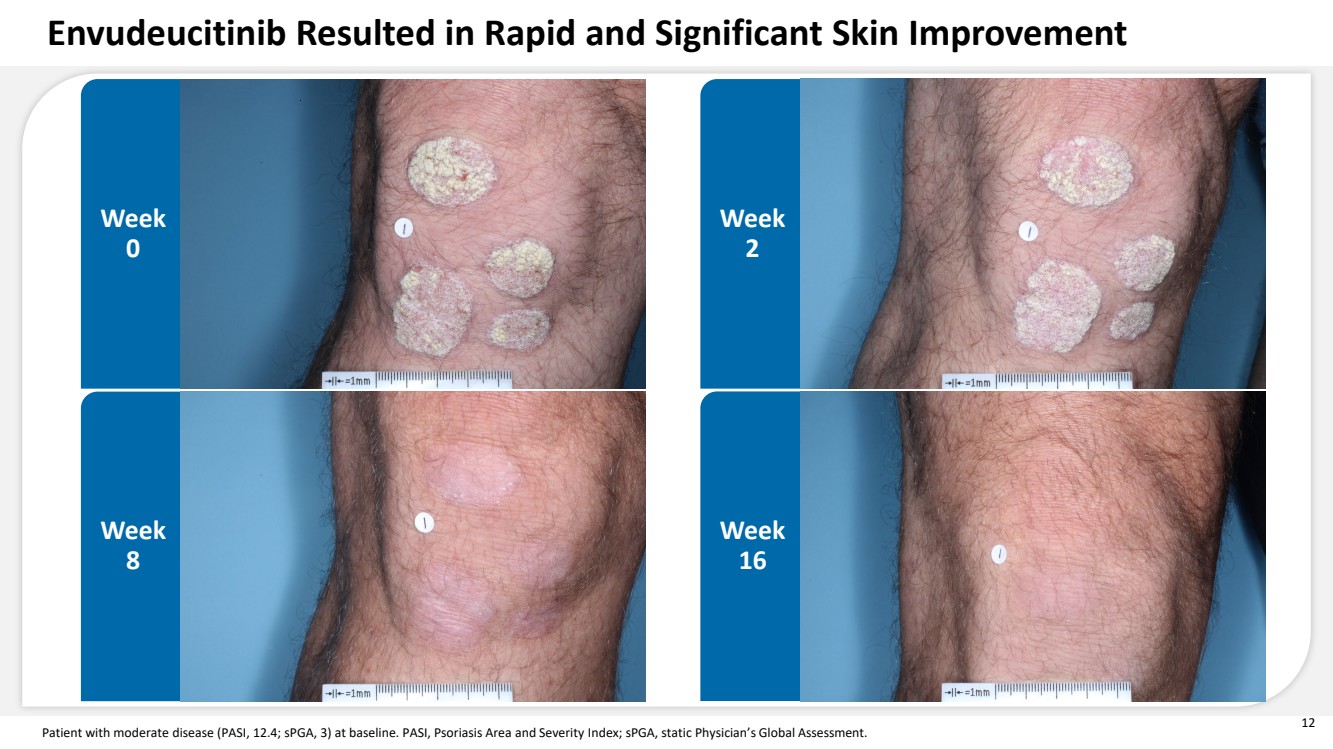
| 12 Patient with moderate disease (PASI, 12.4; sPGA, 3) at baseline. PASI, Psoriasis Area and Severity Index; sPGA, static Physician’s Global Assessment. Week 0 Week 2 Week 8 Week 16 Envudeucitinib Resulted in Rapid and Significant Skin Improvement |
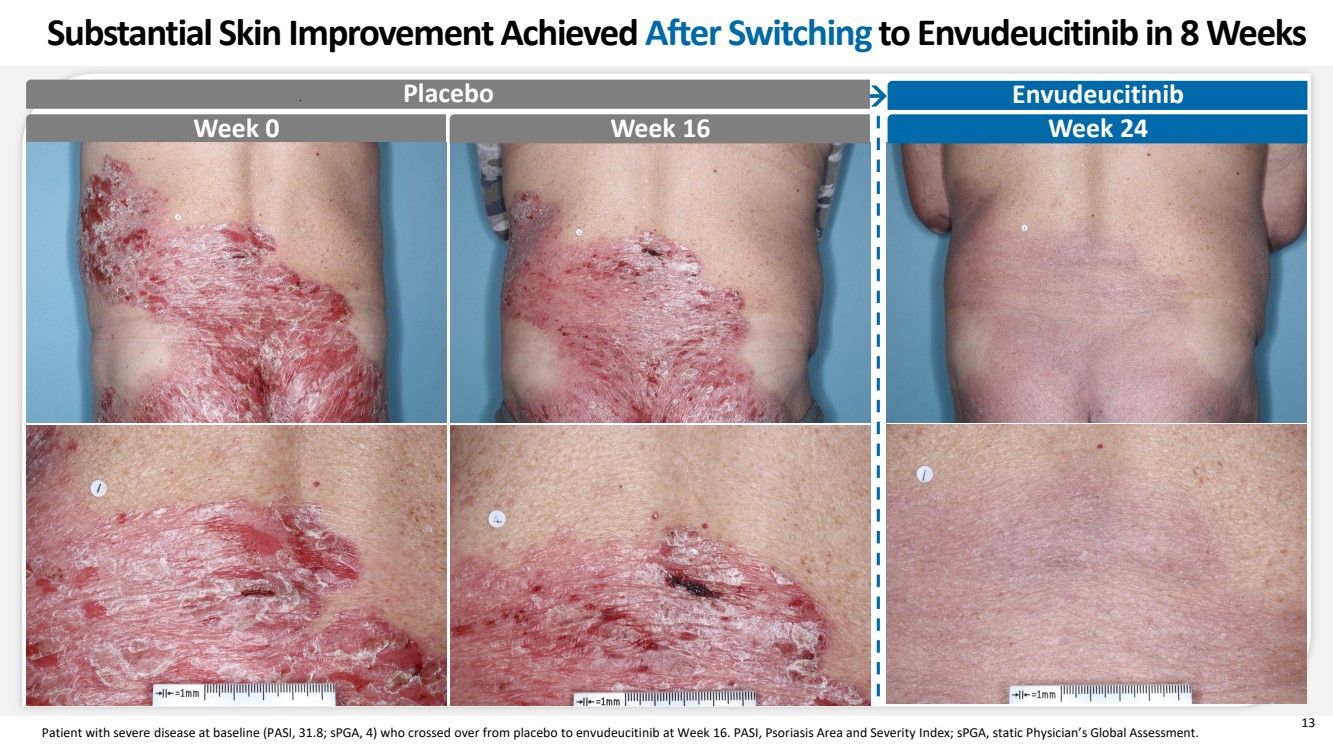
| 13 Patient with severe disease at baseline (PASI, 31.8; sPGA, 4) who crossed over from placebo to envudeucitinib at Week 16. PASI, Psoriasis Area and Severity Index; sPGA, static Physician’s Global Assessment. Week 0 Week 16 Week 24 Substantial Skin Improvement Achieved After Switching to Envudeucitinib in 8 Weeks Placebo Envudeucitinib |
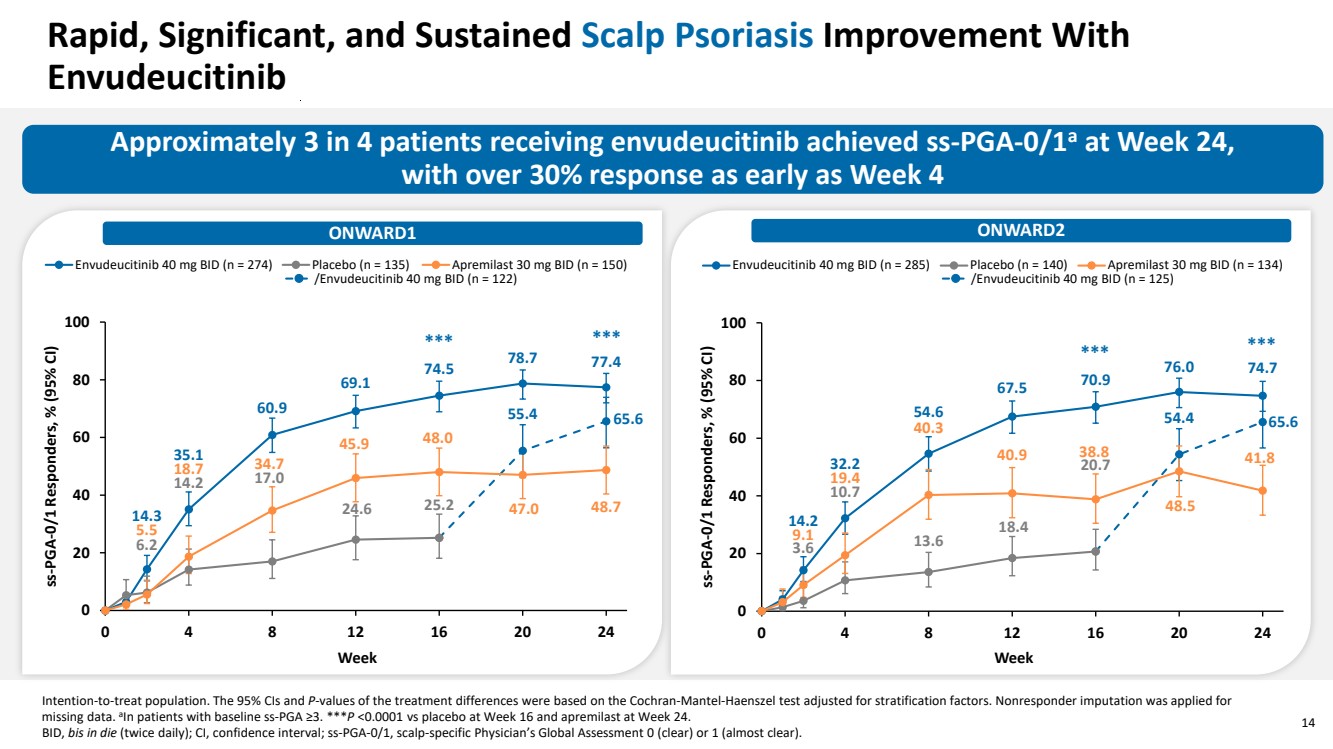
| 14 Rapid, Significant, and Sustained Scalp Psoriasis Improvement With Envudeucitinib Approximately 3 in 4 patients receiving envudeucitinib achieved ss-PGA-0/1a at Week 24, with over 30% response as early as Week 4 Intention-to-treat population. The 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. aIn patients with baseline ss-PGA ≥3. ***P <0.0001 vs placebo at Week 16 and apremilast at Week 24. BID, bis in die (twice daily); CI, confidence interval; ss-PGA-0/1, scalp-specific Physician’s Global Assessment 0 (clear) or 1 (almost clear). 14.3 35.1 60.9 69.1 74.5 78.7 77.4 6.2 14.2 17.0 24.6 25.2 55.4 65.6 5.5 18.7 34.7 45.9 48.0 47.0 48.7 0 20 40 60 80 100 0 4 8 12 16 20 24 ss-PGA-0/1 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 274) Placebo (n = 135) Apremilast 30 mg BID (n = 150) /Envudeucitinib 40 mg BID (n = 122) 14.2 32.2 54.6 67.5 70.9 76.0 74.7 3.6 10.7 13.6 18.4 20.7 54.4 65.6 9.1 19.4 40.3 40.9 38.8 48.5 41.8 0 20 40 60 80 100 0 4 8 12 16 20 24 ss-PGA-0/1 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 285) Placebo (n = 140) Apremilast 30 mg BID (n = 134) ONWARD1 ONWARD2 /Envudeucitinib 40 mg BID (n = 125) *** *** *** *** |
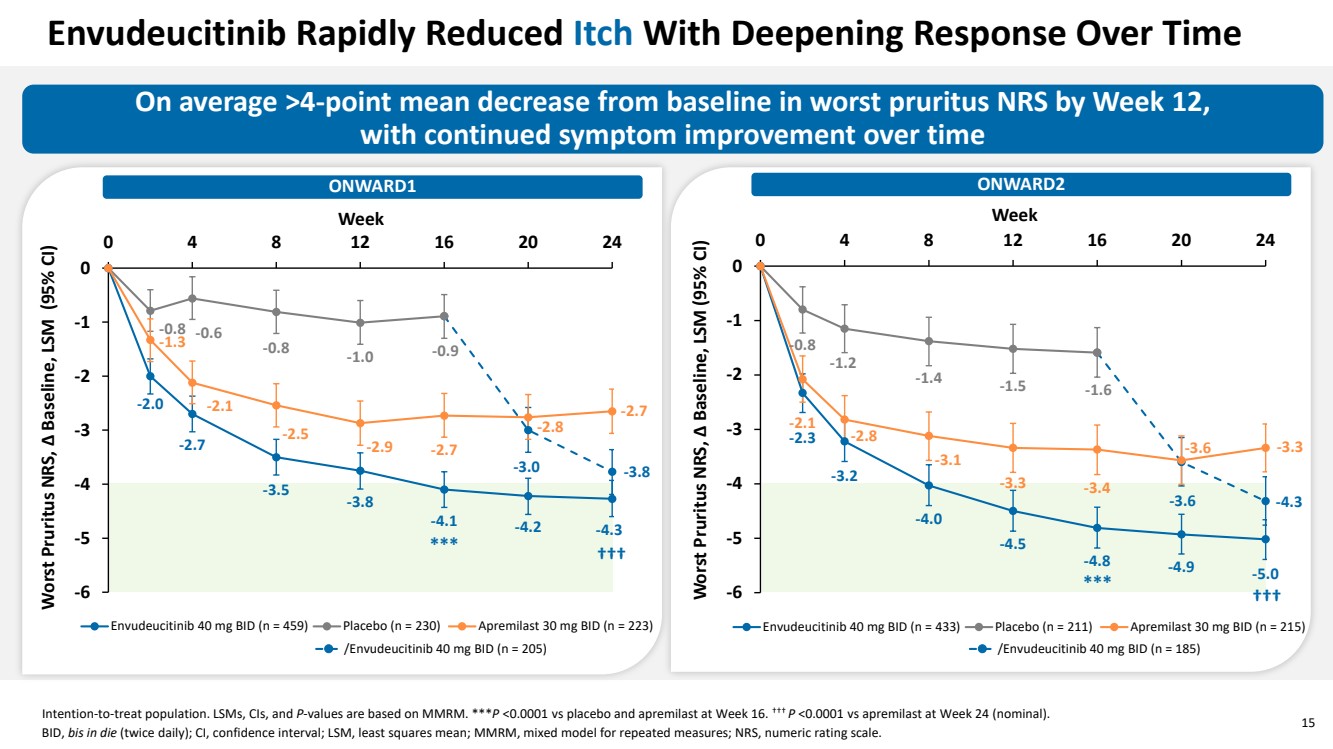
| 15 Envudeucitinib Rapidly Reduced Itch With Deepening Response Over Time -2.0 -2.7 -3.5 -3.8 -4.1 -4.2 -4.3 -0.8 -0.6 -0.8 -1.0 -0.9 -3.0 -3.8 -1.3 -2.1 -2.5 -2.9 -2.7 -2.8 -2.7 -6 -5 -4 -3 -2 -1 0 0 4 8 12 16 20 24 Worst Pruritus NRS, Δ Baseline, LSM (95% CI) Week Envudeucitinib 40 mg BID (n = 459) Placebo (n = 230) Apremilast 30 mg BID (n = 223) /Envudeucitinib 40 mg BID (n = 205) On average ˃4-point mean decrease from baseline in worst pruritus NRS by Week 12, with continued symptom improvement over time ONWARD1 ONWARD2 Intention-to-treat population. LSMs, CIs, and P-values are based on MMRM. ***P <0.0001 vs placebo and apremilast at Week 16. ††† P <0.0001 vs apremilast at Week 24 (nominal). BID, bis in die (twice daily); CI, confidence interval; LSM, least squares mean; MMRM, mixed model for repeated measures; NRS, numeric rating scale. -2.3 -3.2 -4.0 -4.5 -4.8 -4.9 -5.0 -0.8 -1.2 -1.4 -1.5 -1.6 -3.6 -4.3 -2.1 -2.8 -3.1 -3.3 -3.4 -3.6 -3.3 -6 -5 -4 -3 -2 -1 0 0 4 8 12 16 20 24 Worst Pruritus NRS, Δ Baseline, LSM (95% CI) Week Envudeucitinib 40 mg BID (n = 433) Placebo (n = 211) Apremilast 30 mg BID (n = 215) /Envudeucitinib 40 mg BID (n = 185) *** *** ††† ††† |
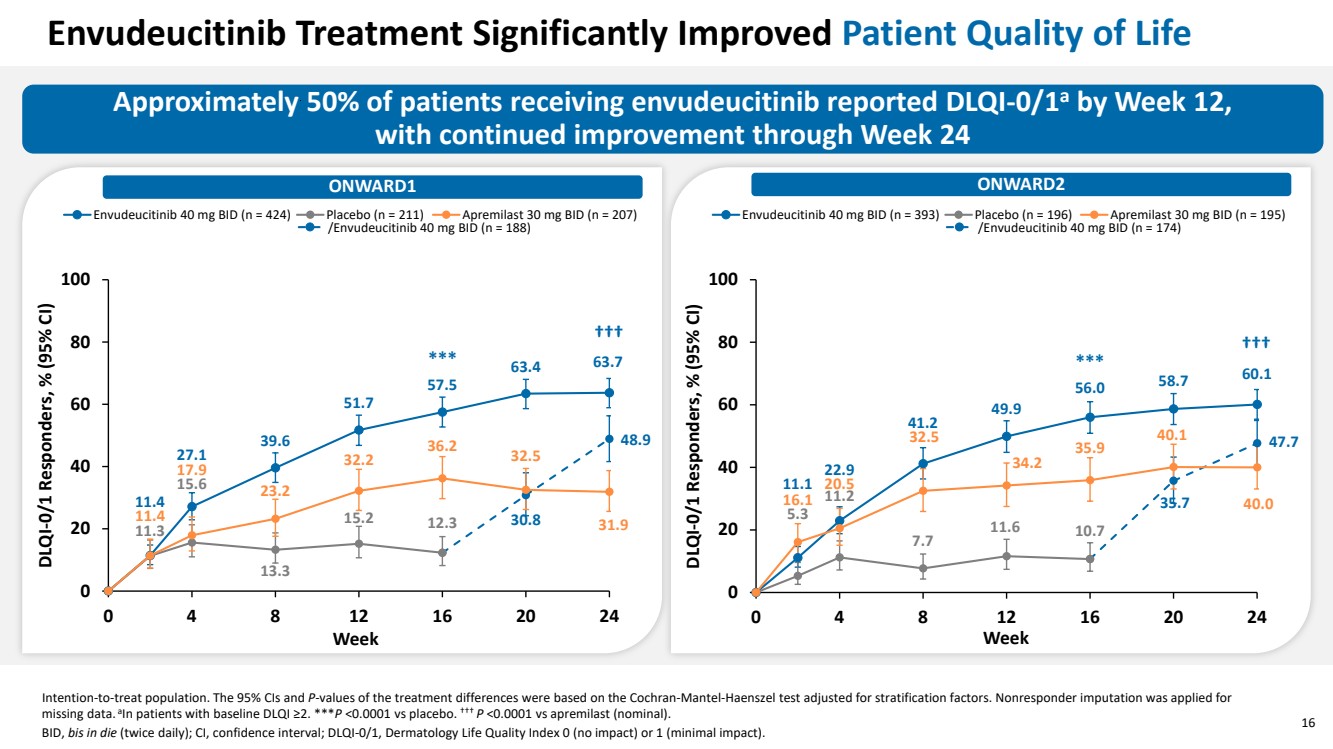
| 16 Approximately 50% of patients receiving envudeucitinib reported DLQI-0/1a by Week 12, with continued improvement through Week 24 ONWARD1 ONWARD2 Intention-to-treat population. The 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. aIn patients with baseline DLQI ≥2. ***P <0.0001 vs placebo. ††† P <0.0001 vs apremilast (nominal). BID, bis in die (twice daily); CI, confidence interval; DLQI-0/1, Dermatology Life Quality Index 0 (no impact) or 1 (minimal impact). Envudeucitinib Treatment Significantly Improved Patient Quality of Life 11.4 27.1 39.6 51.7 57.5 63.4 63.7 11.3 15.6 13.3 15.2 12.3 30.8 48.9 11.4 17.9 23.2 32.2 36.2 32.5 31.9 0 20 40 60 80 100 0 4 8 12 16 20 24 DLQI-0/1 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 424) Placebo (n = 211) Apremilast 30 mg BID (n = 207) /Envudeucitinib 40 mg BID (n = 188) 11.1 22.9 41.2 49.9 56.0 58.7 60.1 5.3 11.2 7.7 11.6 10.7 35.7 47.7 16.1 20.5 32.5 34.2 35.9 40.1 40.0 0 20 40 60 80 100 0 4 8 12 16 20 24 DLQI-0/1 Responders, % (95% CI) Week Envudeucitinib 40 mg BID (n = 393) Placebo (n = 196) Apremilast 30 mg BID (n = 195) /Envudeucitinib 40 mg BID (n = 174) *** ††† *** ††† |
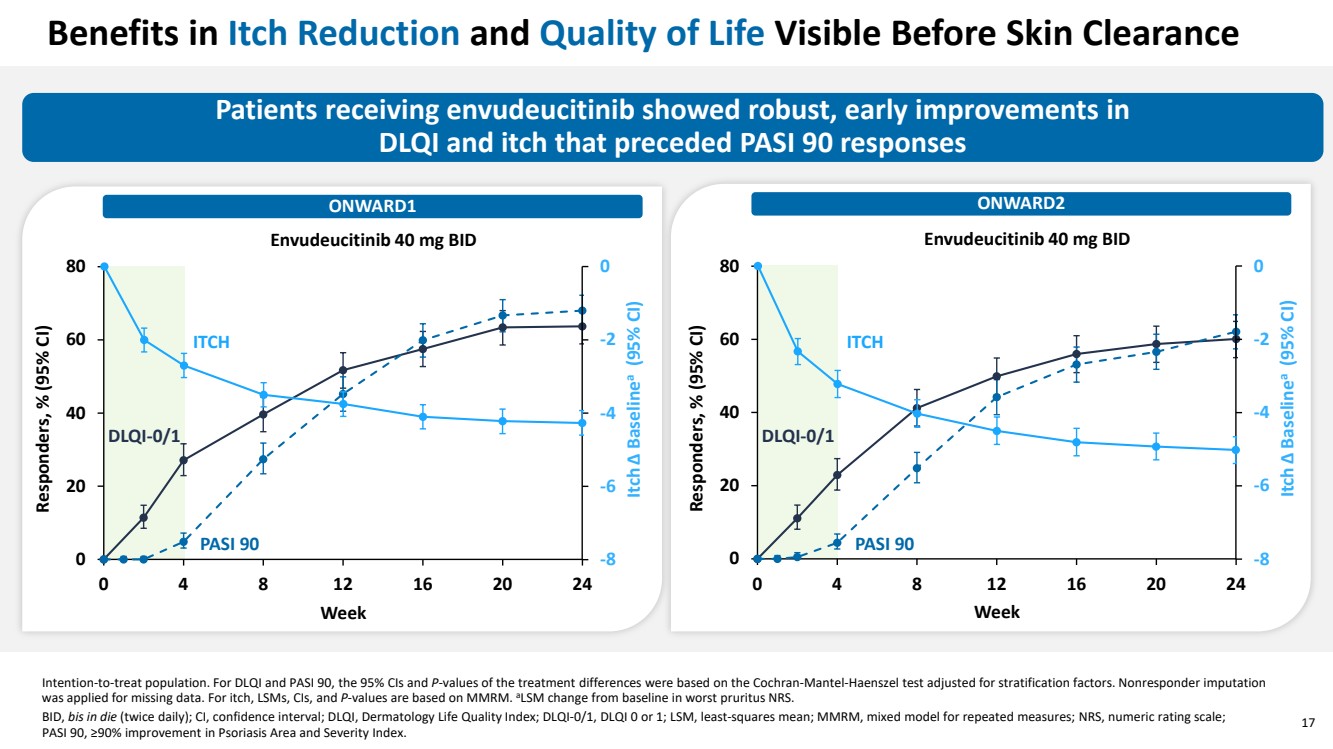
| 17 Benefits in Itch Reduction and Quality of Life Visible Before Skin Clearance Patients receiving envudeucitinib showed robust, early improvements in DLQI and itch that preceded PASI 90 responses ONWARD1 ONWARD2 Intention-to-treat population. For DLQI and PASI 90, the 95% CIs and P-values of the treatment differences were based on the Cochran-Mantel-Haenszel test adjusted for stratification factors. Nonresponder imputation was applied for missing data. For itch, LSMs, CIs, and P-values are based on MMRM. aLSM change from baseline in worst pruritus NRS. BID, bis in die (twice daily); CI, confidence interval; DLQI, Dermatology Life Quality Index; DLQI-0/1, DLQI 0 or 1; LSM, least-squares mean; MMRM, mixed model for repeated measures; NRS, numeric rating scale; PASI 90, ≥90% improvement in Psoriasis Area and Severity Index. Week 0 20 40 60 80 0 4 8 12 16 20 24 Responders, % (95% CI) Envudeucitinib 40 mg BID -8 -6 -4 -2 0 Itch Δ Baselinea (95% CI) DLQI-0/1 PASI 90 ITCH Week 0 20 40 60 80 0 4 8 12 16 20 24 Responders, % (95% CI) Envudeucitinib 40 mg BID -8 -6 -4 -2 0 Itch Δ Baselinea (95% CI) DLQI-0/1 PASI 90 ITCH |
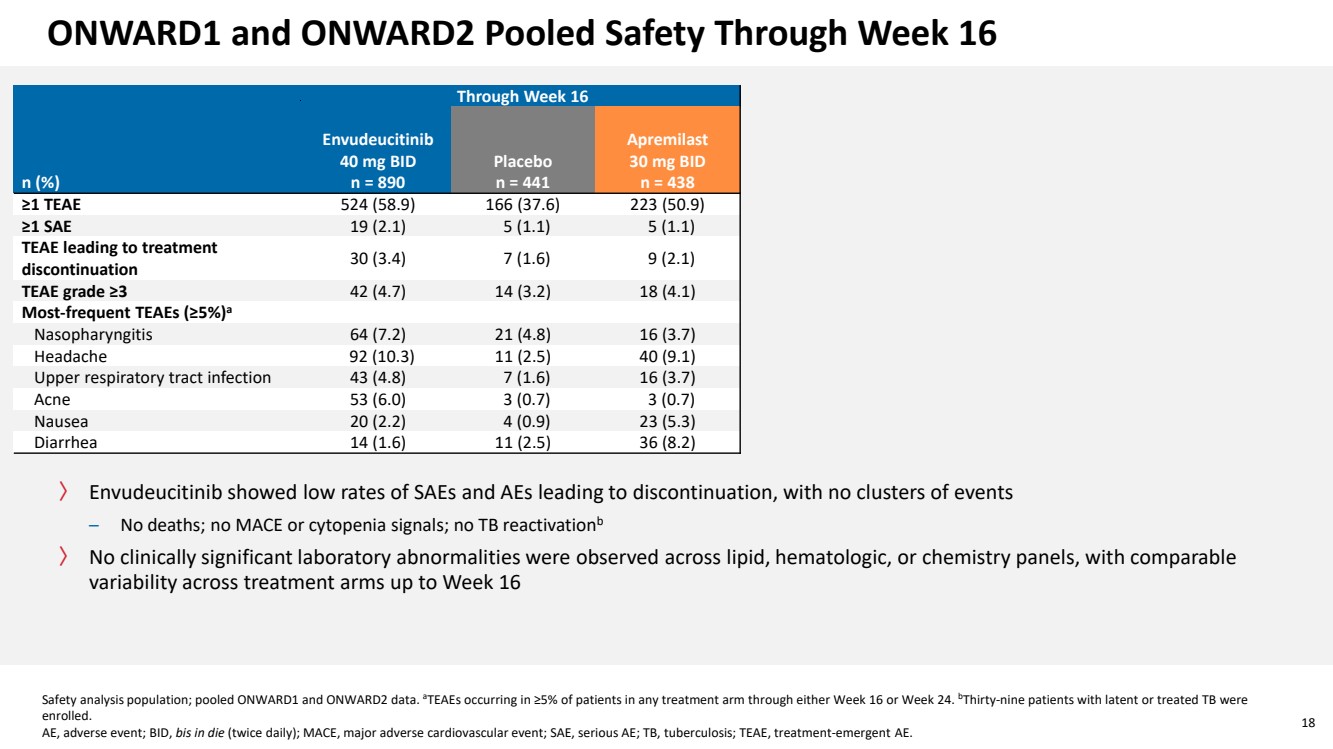
| 18 ONWARD1 and ONWARD2 Pooled Safety Through Week 16 〉 Envudeucitinib showed low rates of SAEs and AEs leading to discontinuation, with no clusters of events – No deaths; no MACE or cytopenia signals; no TB reactivationb 〉 No clinically significant laboratory abnormalities were observed across lipid, hematologic, or chemistry panels, with comparable variability across treatment arms up to Week 16 Safety analysis population; pooled ONWARD1 and ONWARD2 data. aTEAEs occurring in ≥5% of patients in any treatment arm through either Week 16 or Week 24. bThirty-nine patients with latent or treated TB were enrolled. AE, adverse event; BID, bis in die (twice daily); MACE, major adverse cardiovascular event; SAE, serious AE; TB, tuberculosis; TEAE, treatment-emergent AE. Through Week 16 n (%) Envudeucitinib 40 mg BID n = 890 Placebo n = 441 Apremilast 30 mg BID n = 438 ≥1 TEAE 524 (58.9) 166 (37.6) 223 (50.9) ≥1 SAE 19 (2.1) 5 (1.1) 5 (1.1) TEAE leading to treatment discontinuation 30 (3.4) 7 (1.6) 9 (2.1) TEAE grade ≥3 42 (4.7) 14 (3.2) 18 (4.1) Most-frequent TEAEs (≥5%)a Nasopharyngitis 64 (7.2) 21 (4.8) 16 (3.7) Headache 92 (10.3) 11 (2.5) 40 (9.1) Upper respiratory tract infection 43 (4.8) 7 (1.6) 16 (3.7) Acne 53 (6.0) 3 (0.7) 3 (0.7) Nausea 20 (2.2) 4 (0.9) 23 (5.3) Diarrhea 14 (1.6) 11 (2.5) 36 (8.2) |
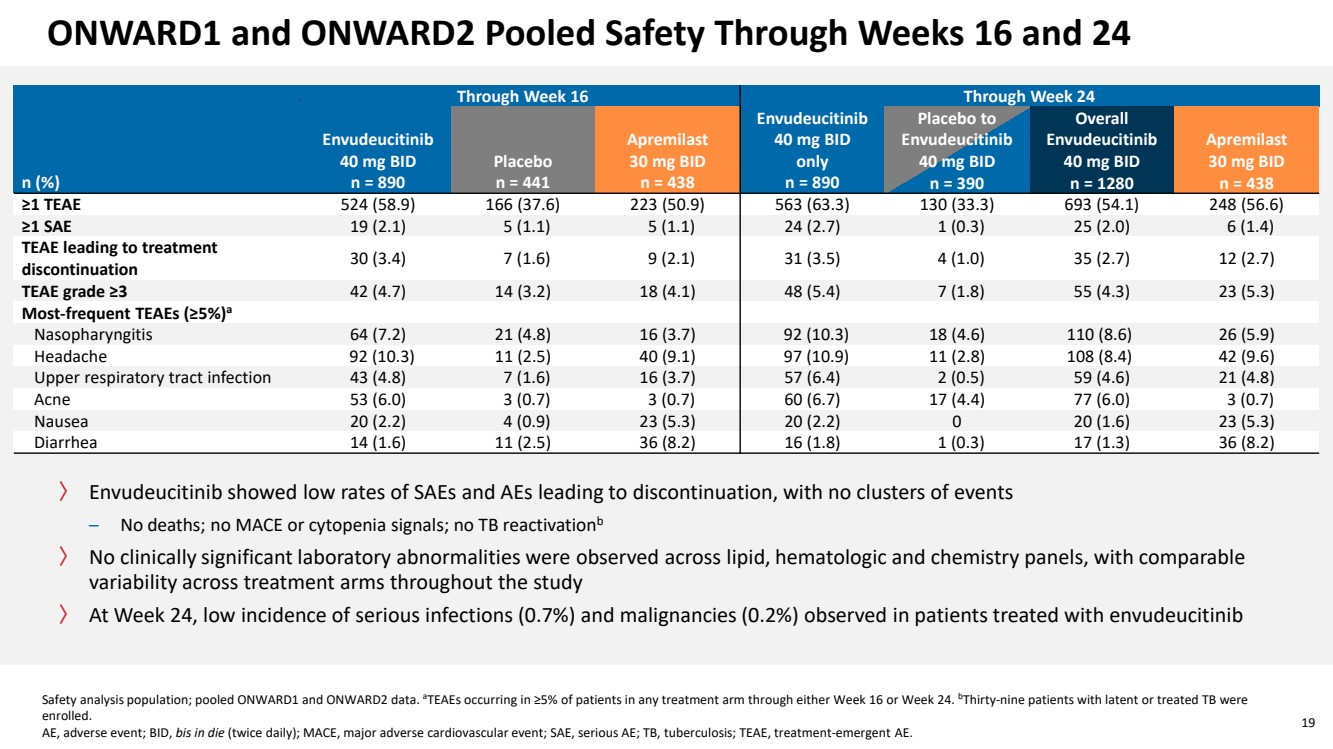
| 19 ONWARD1 and ONWARD2 Pooled Safety Through Weeks 16 and 24 Through Week 16 Through Week 24 n (%) Envudeucitinib 40 mg BID n = 890 Placebo n = 441 Apremilast 30 mg BID n = 438 Envudeucitinib 40 mg BID only n = 890 Placebo to Envudeucitinib 40 mg BID n = 390 Overall Envudeucitinib 40 mg BID n = 1280 Apremilast 30 mg BID n = 438 ≥1 TEAE 524 (58.9) 166 (37.6) 223 (50.9) 563 (63.3) 130 (33.3) 693 (54.1) 248 (56.6) ≥1 SAE 19 (2.1) 5 (1.1) 5 (1.1) 24 (2.7) 1 (0.3) 25 (2.0) 6 (1.4) TEAE leading to treatment discontinuation 30 (3.4) 7 (1.6) 9 (2.1) 31 (3.5) 4 (1.0) 35 (2.7) 12 (2.7) TEAE grade ≥3 42 (4.7) 14 (3.2) 18 (4.1) 48 (5.4) 7 (1.8) 55 (4.3) 23 (5.3) Most-frequent TEAEs (≥5%)a Nasopharyngitis 64 (7.2) 21 (4.8) 16 (3.7) 92 (10.3) 18 (4.6) 110 (8.6) 26 (5.9) Headache 92 (10.3) 11 (2.5) 40 (9.1) 97 (10.9) 11 (2.8) 108 (8.4) 42 (9.6) Upper respiratory tract infection 43 (4.8) 7 (1.6) 16 (3.7) 57 (6.4) 2 (0.5) 59 (4.6) 21 (4.8) Acne 53 (6.0) 3 (0.7) 3 (0.7) 60 (6.7) 17 (4.4) 77 (6.0) 3 (0.7) Nausea 20 (2.2) 4 (0.9) 23 (5.3) 20 (2.2) 0 20 (1.6) 23 (5.3) Diarrhea 14 (1.6) 11 (2.5) 36 (8.2) 16 (1.8) 1 (0.3) 17 (1.3) 36 (8.2) Safety analysis population; pooled ONWARD1 and ONWARD2 data. aTEAEs occurring in ≥5% of patients in any treatment arm through either Week 16 or Week 24. bThirty-nine patients with latent or treated TB were enrolled. AE, adverse event; BID, bis in die (twice daily); MACE, major adverse cardiovascular event; SAE, serious AE; TB, tuberculosis; TEAE, treatment-emergent AE. 〉 Envudeucitinib showed low rates of SAEs and AEs leading to discontinuation, with no clusters of events – No deaths; no MACE or cytopenia signals; no TB reactivationb 〉 No clinically significant laboratory abnormalities were observed across lipid, hematologic and chemistry panels, with comparable variability across treatment arms throughout the study 〉 At Week 24, low incidence of serious infections (0.7%) and malignancies (0.2%) observed in patients treated with envudeucitinib |
| 20 Envudeucitinib, a next-generation TYK2i, delivered early and progressively deepening skin clearance, with meaningful improvements in patient-reported outcomes in ONWARD1 and 2 〉All primary and secondary efficacy endpoints met, with approximately 65% and 40% of patients receiving envudeucitinib achieving PASI 90 and PASI 100 at Week 24 〉Rapid and significant improvement in moderate-to-severe scalp psoriasis, itch, and quality of life 〉Envudeucitinib treatment was generally well tolerated, with no new signals and a safety profile consistent with the previous long-term Phase 2 studies 〉One year, Phase 3 long-term data will be available in the second half of 2026 〉Once-daily formulation and pediatric plan under development Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency. PASI 90/100, ≥90%/100% improvement in Psoriasis Area and Severity Index; TYK2i, tyrosine kinase 2 inhibitor. |
| 21 Acknowledgments 〉 The Authors thank the patients and their families for their participation and the study sites and teams for their facilitation of the ONWARD clinical studies 〉 The Authors thank Komal Bawa, PharmD, of Alumis Inc., for assistance with data publication and editorial review. Writing and editorial support were provided by Sylvia Stankov, PhD, of Red Nucleus, and were funded by Alumis Inc. |