| March 2026 Zervimesine: a Once-daily Oral Therapeutic Advancing Toward Phase 3 |
| 2 FORWARD-LOOKING STATEMENTS This presentation contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. All statements contained in this presentation, other than statements of historical facts or statements that relate to present facts or current conditions, including but not limited to, product candidates, including zervimesine, also known as zervimesine, and any expected or implied benefits or results, including that initial clinical results observed with respect to zervimesine will be replicated in later trials, and our clinical development plans, including statements regarding our clinical studies of zervimesine and our regulatory plans, expectations regarding potential patient populations, expectations regarding our patent portfolio, and our expected cash runway, are forward-looking statements. These statements, including statements related to the timing and expected results of our clinical trials, involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed or implied by the forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,” “should,” “expect,” “plan,” “aim,” “seek,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “forecast,” “potential” or “continue” or the negative of these terms or other similar expressions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition, and results of operations. These forward-looking statements speak only as of the date of this presentation and are subject to a number of risks, uncertainties and assumptions, some of which cannot be predicted or quantified and some of which are beyond our control. Factors that may cause actual results to differ materially from current expectations include, but are not limited to: our ability to successfully advance our current and future product candidates through development activities, preclinical studies and clinical trials and costs related thereto; uncertainties inherent in the results of preliminary data, preclinical studies and earlier-stage clinical trials being predictive of the results of early or later-stage clinical trials; the timing, scope and likelihood of regulatory filings and approvals, including regulatory approval of our product candidates; competition, our ability to secure new (and retain existing) grant funding, our ability to grow and manage growth, maintain relationships with suppliers and retain our management and key employees; changes in applicable laws or regulations; the possibility that the we may be adversely affected by other economic, business or competitive factors, including ongoing economic uncertainty; our estimates of expenses and profitability; the evolution of the markets in which we compete; our ability to implement our strategic initiatives and continue to innovate our existing products; our ability to defend our intellectual property; impacts of global political changes and global economic conditions on our business, supply chain and labor force; our ability to maintain the listing of our common stock on the Nasdaq Global Market; and the risks and uncertainties described more fully in the “Risk Factors” section of our annual and quarterly reports filed with the Securities & Exchange Commission that are available on www.sec.gov. These risks are not exhaustive, and we face both known and unknown risks. You should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in a dynamic industry and economy. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that we may face. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. TRADEMARKS This presentation may contain trademarks, service marks, trade names and copyrights of other companies, which are the property of their respective owners. Solely for convenience, some of the trademarks, service marks, trade names and copyrights referred to in this presentation may be listed without the TM, SM © or ® symbols, but we will assert, to the fullest extent under applicable law, the rights of the applicable owners, if any, to these trademarks, service marks, trade names and copyrights. MARKET & INDUSTRY DATA Projections, estimates, industry data and information contained in this presentation, including the size of and growth in key end markets, are based on information from third-party sources and management estimates. Although we believe that these third party-sources are reliable, we cannot guarantee the accuracy or completeness of these sources. Our management’s estimates are derived from third-party sources, publicly available information, our knowledge of our industry and assumptions based on such information and knowledge. Our management’s estimates have not been verified by any independent source. All of the projections, estimates, market data and industry information used in this presentation involve a number of assumptions and limitations, and you are cautioned not to give undue weight to such information. In addition, projections, estimates and assumptions relating to us and our industry's future performance are necessarily subject to a high degree of uncertainty and risk due to a variety of factors, including, but not limited to, those described above, that could cause future performance to differ materially from our expressed projections, estimates and assumptions or those provided by third parties. Forward-looking Statements |
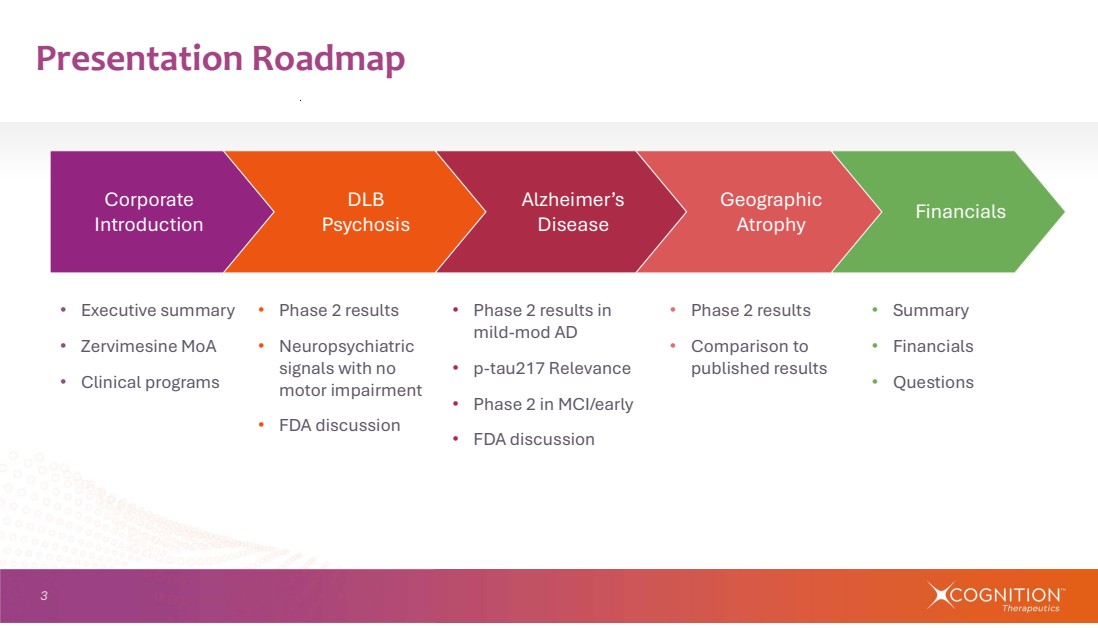
| 3 • Executive summary • Zervimesine MoA • Clinical programs • Phase 2 results • Comparison to published results Presentation Roadmap Financials Geographic Atrophy • Phase 2 results • Neuropsychiatric signals with no motor impairment • FDA discussion • Phase 2 results in mild-mod AD • p-tau217 Relevance • Phase 2 in MCI/early • FDA discussion Alzheimer’s Disease DLB Psychosis Corporate Introduction • Summary • Financials • Questions |
| 4 Over $170M in grants from NIH & others substantially funded development through Phase 2 • Potential first-to-market for DLB psychosis - Majority of DLB patients experience psychosis - Psychotic symptoms result in higher HC costs and institutionalization - No drugs approved; off label use limited by side effects • Zervimesine treatment stabilized psychotic symptoms in Phase 2 - DLB psychotic symptoms slowed by 102% vs placebo (NPI-4) - Disease-modifying MoA expected to result in sustained treatment effect • Well tolerated safety profile in over 450 people treated to date - Modest side effect profile for use in aging population • Oral QD administration - No required imaging surveillance - ARIA unexpected based on MoA • Robust intellectual property through 2040 with PTE ARIA: amyloid-related imaging abnormalities; MOA: mechanism of action; PTE: patent term extension |
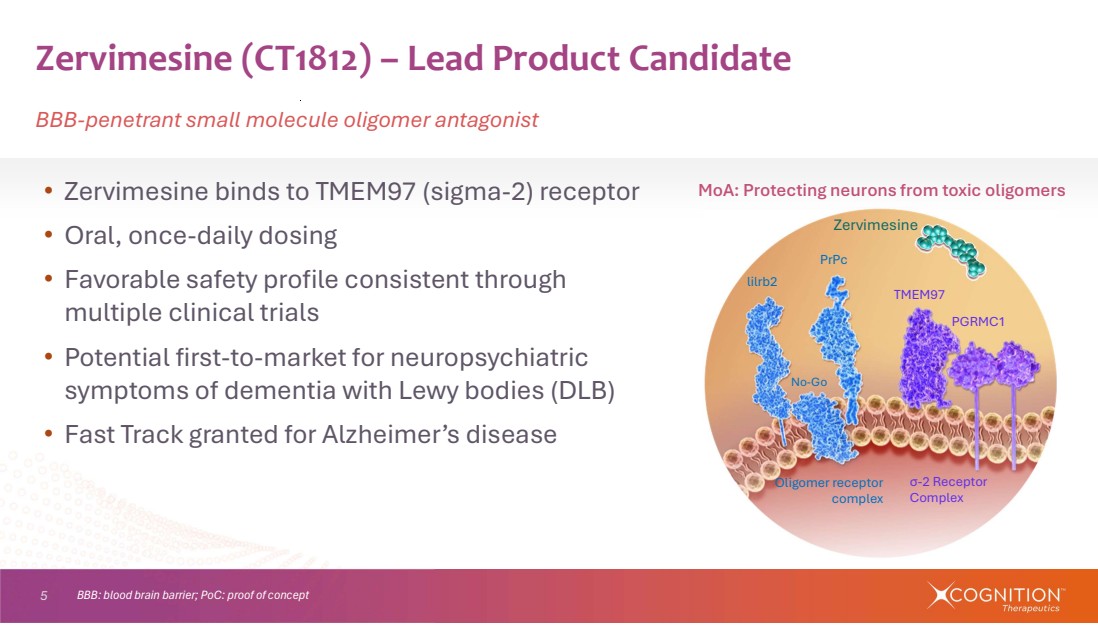
| 5 • Zervimesine binds to TMEM97 (sigma-2) receptor • Oral, once-daily dosing • Favorable safety profile consistent through multiple clinical trials • Potential first-to-market for neuropsychiatric symptoms of dementia with Lewy bodies (DLB) • Fast Track granted for Alzheimer’s disease Zervimesine (CT1812) – Lead Product Candidate BBB-penetrant small molecule oligomer antagonist lilrb2 PrPc No-Go TMEM97 PGRMC1 σ-2 Receptor Complex Oligomer receptor complex Zervimesine BBB: blood brain barrier; PoC: proof of concept MoA: Protecting neurons from toxic oligomers |
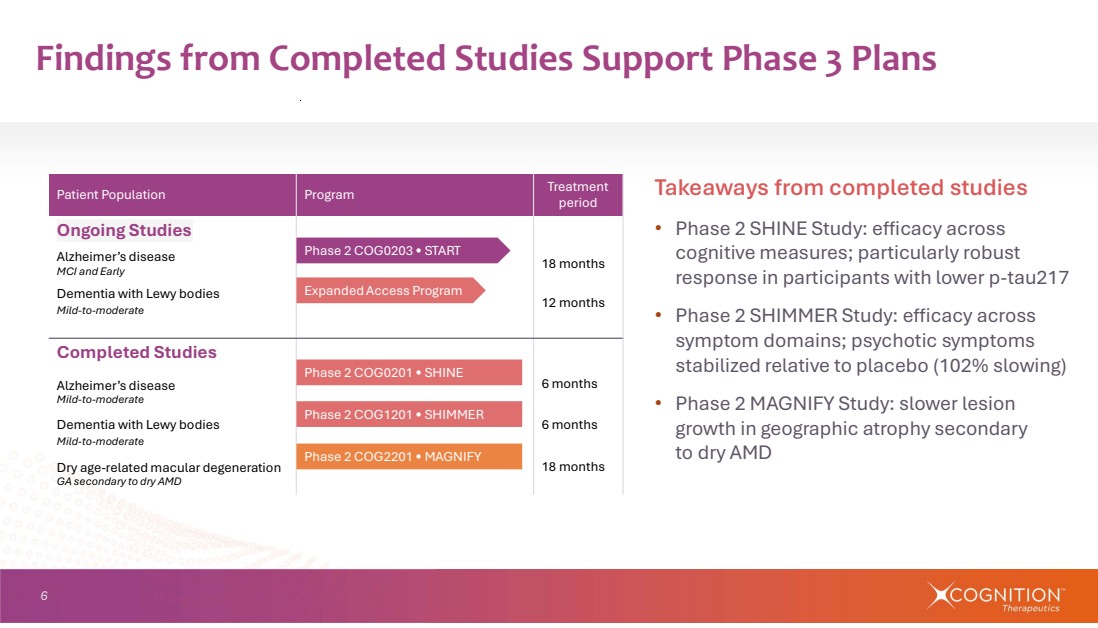
| 6 Findings from Completed Studies Support Phase 3 Plans Treatment period Patient Population Program Ongoing Studies 18 months Alzheimer’s disease MCI and Early 12 months Dementia with Lewy bodies Mild-to-moderate Completed Studies Alzheimer’s disease 6 months Mild-to-moderate Dementia with Lewy bodies 6 months Mild-to-moderate Dry age-related macular degeneration 18 months GA secondary to dry AMD Takeaways from completed studies • Phase 2 SHINE Study: efficacy across cognitive measures; particularly robust response in participants with lower p-tau217 • Phase 2 SHIMMER Study: efficacy across symptom domains; psychotic symptoms stabilized relative to placebo (102% slowing) • Phase 2 MAGNIFY Study: slower lesion growth in geographic atrophy secondary to dry AMD Phase 2 COG0203 • START Phase 2 COG2201 • MAGNIFY Phase 2 COG1201 • SHIMMER Phase 2 COG0201 • SHINE Expanded Access Program |
| 7 Completed: AD End-of-Phase 2 Meeting • Aligned with FDA on following: - Disease stage: mild-to-moderate AD - Enrichment: p-tau217 at screening ≤ 1.0pg/mL - Treatment period: 6 months - Randomization: 1:1 zervimesine (100mg) vs pbo - Endpoints: composite cognitive and functional - Open-label extension to follow • EMA proposes longer trial Completed: Type C Meeting for DLB • Focused on clinical outcomes that encompass DLB’s complex symptomatology • Plan to advance zervimesine for DLB psychosis - Based on strong signals in behavioral and psychiatric symptoms: hallucinations, delusions, anxiety and agitation/aggression • Process to move to FDA’s Division of Psychiatry underway with meeting(s) anticipated in mid-2026 Prioritizing DLB Psychosis Following Regulatory Feedback Completed FDA meetings complete with feedback on AD and DLB clinical programs |
| Addressing unmet need of majority of DLB patients Dementia with Lewy Bodies (DLB) Psychosis |
| 9 Four Core Domains + Functional Impairment Drive DLB Burden DLB is “a multifactorial disease with a buffet of symptoms” James E. Galvin, MD, MPH, Univ Miami Miller School of Medicine Diminished ability to bathe, shop, prepare meals Difficulty standing, maintaining balance Fluctuations Impaired memory and problem solving Hallucinations, anxiety, delusions Patient symptoms • 2nd most common cause of dementia after Alzheimer’s disease • Correct diagnosis often requires multiple specialist visits over 18 months • Faster decline than Alzheimer’s • More common in men Behavior Cognition Fluctuations Movement Function |
| 10 Complex Symptomology Requires Array of Assessments All assessments are standardized and have been used in clinical trials and drug approvals Assessment tools measure a variety of symptoms ADCS-Activities of Daily Living (ADL) MDS-Unified PD Rating Scale Part III (UPDRS III) Clinician Assessment of Fluctuation (CAF) Cognitive Drug Research (CDR) System Neuropsychiatric Inventory (NPI) Assessment tools Montreal Cognitive Assessment (MoCA) Care Partner’s NPI of Distress Behavior Cognition Fluctuations Movement Function |
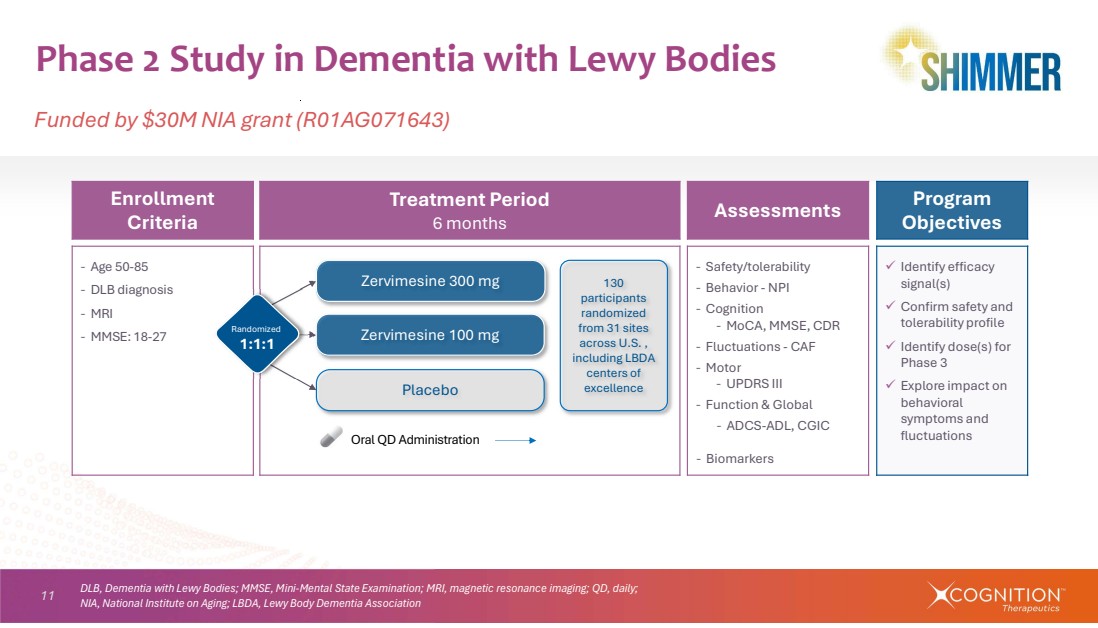
| 11 Phase 2 Study in Dementia with Lewy Bodies Funded by $30M NIA grant (R01AG071643) Program Objectives Assessments Treatment Period 6 months Enrollment Criteria Identify efficacy signal(s) Confirm safety and tolerability profile Identify dose(s) for Phase 3 Explore impact on behavioral symptoms and fluctuations ‐ Safety/tolerability ‐ Behavior - NPI ‐ Cognition ‐ MoCA, MMSE, CDR ‐ Fluctuations - CAF ‐ Motor ‐ UPDRS III ‐ Function & Global ‐ ADCS-ADL, CGIC ‐ Biomarkers ‐ Age 50-85 ‐ DLB diagnosis ‐ MRI ‐ MMSE: 18-27 Placebo Zervimesine 100 mg Zervimesine 300 mg Oral QD Administration Randomized 1:1:1 130 participants randomized from 31 sites across U.S. , including LBDA centers of excellence DLB, Dementia with Lewy Bodies; MMSE, Mini-Mental State Examination; MRI, magnetic resonance imaging; QD, daily; NIA, National Institute on Aging; LBDA, Lewy Body Dementia Association |
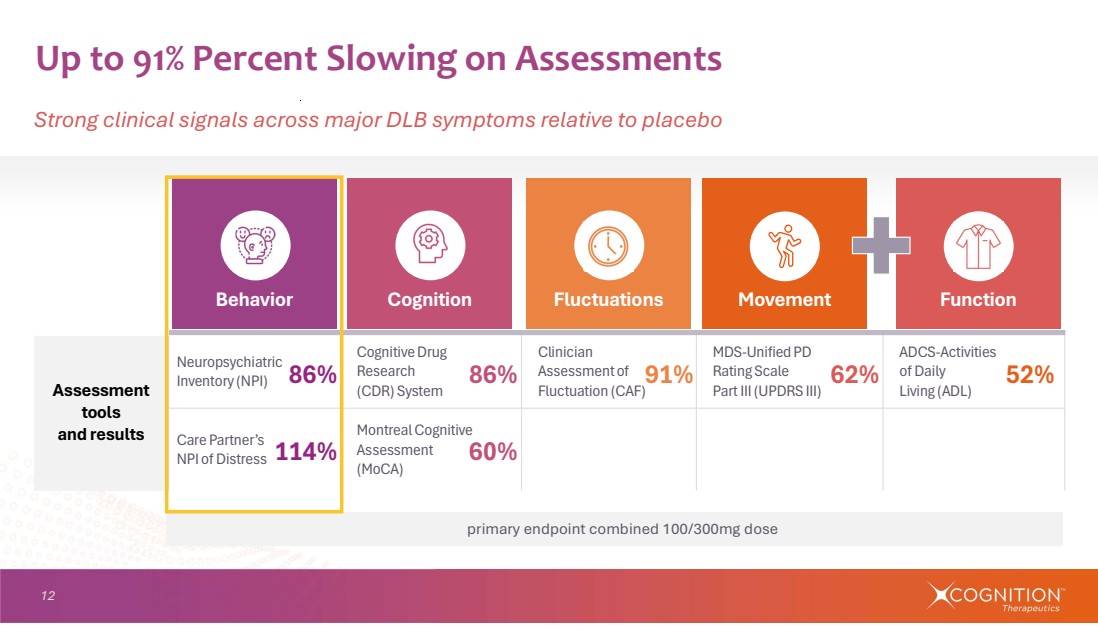
| 12 Up to 91% Percent Slowing on Assessments Strong clinical signals across major DLB symptoms relative to placebo 86% 114% 86% 91% 52% 60% 62% ADCS-Activities of Daily Living (ADL) MDS-Unified PD Rating Scale Part III (UPDRS III) Clinician Assessment of Fluctuation (CAF) Cognitive Drug Research (CDR) System Neuropsychiatric Inventory (NPI) Assessment tools and results Montreal Cognitive Assessment (MoCA) Care Partner’s NPI of Distress primary endpoint combined 100/300mg dose Behavior Cognition Fluctuations Movement Function |
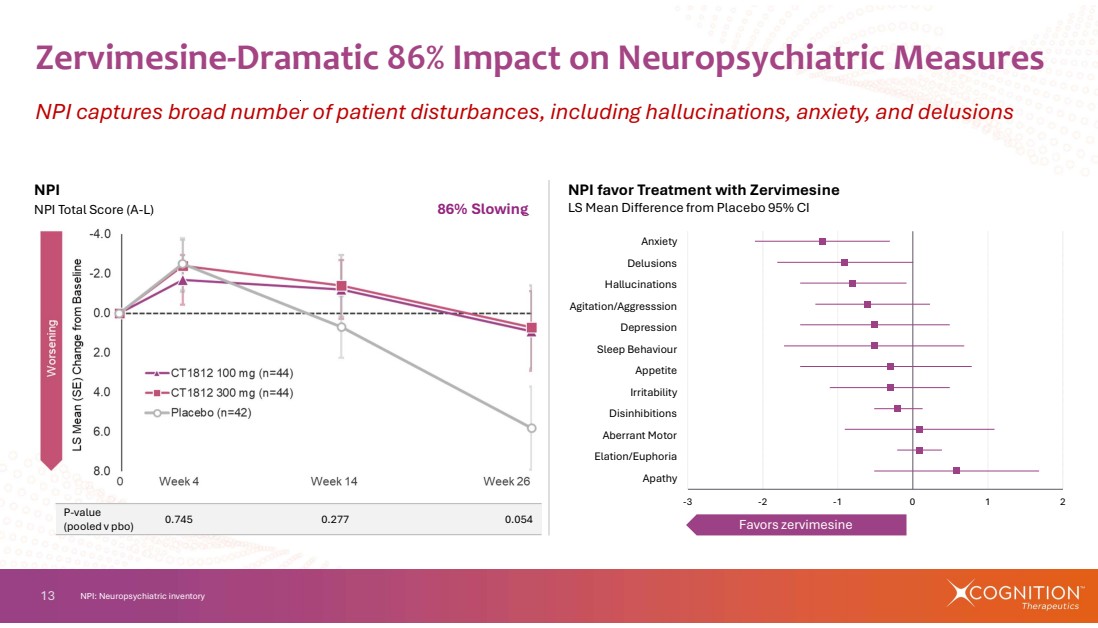
| 13 Zervimesine-Dramatic 86% Impact on Neuropsychiatric Measures NPI captures broad number of patient disturbances, including hallucinations, anxiety, and delusions NPI: Neuropsychiatric inventory NPI favor Treatment with Zervimesine LS Mean Difference from Placebo 95% CI Favors zervimesine 0.745 0.277 0.054 P-value (pooled v pbo) NPI NPI Total Score (A-L) 86% Slowing -3 -2 -1 0 1 2 Apathy Elation/Euphoria Aberrant Motor Disinhibitions Irritability Appetite Sleep Behaviour Depression Agitation/Aggresssion Hallucinations Delusions Anxiety |
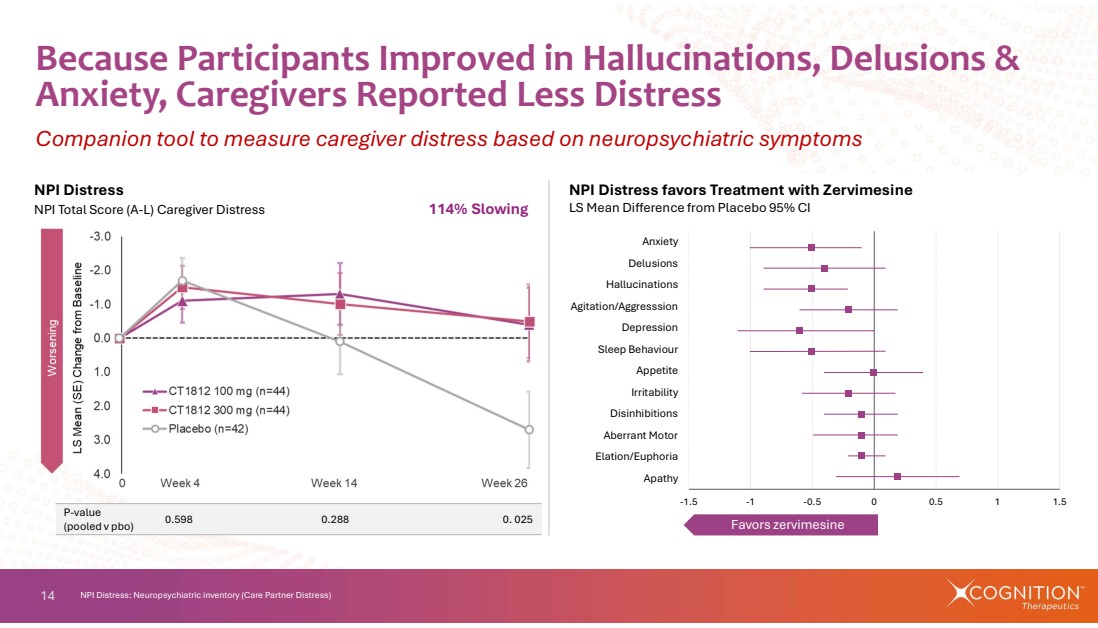
| 14 -1.5 -1 -0.5 0 0.5 1 1.5 Apathy Elation/Euphoria Aberrant Motor Disinhibitions Irritability Appetite Sleep Behaviour Depression Agitation/Aggresssion Hallucinations Delusions Anxiety Favors zervimesine Because Participants Improved in Hallucinations, Delusions & Anxiety, Caregivers Reported Less Distress Companion tool to measure caregiver distress based on neuropsychiatric symptoms NPI Distress: Neuropsychiatric inventory (Care Partner Distress) NPI Distress favors Treatment with Zervimesine LS Mean Difference from Placebo 95% CI NPI Distress NPI Total Score (A-L) Caregiver Distress 114% Slowing 0.598 0.288 0. 025 P-value (pooled v pbo) |
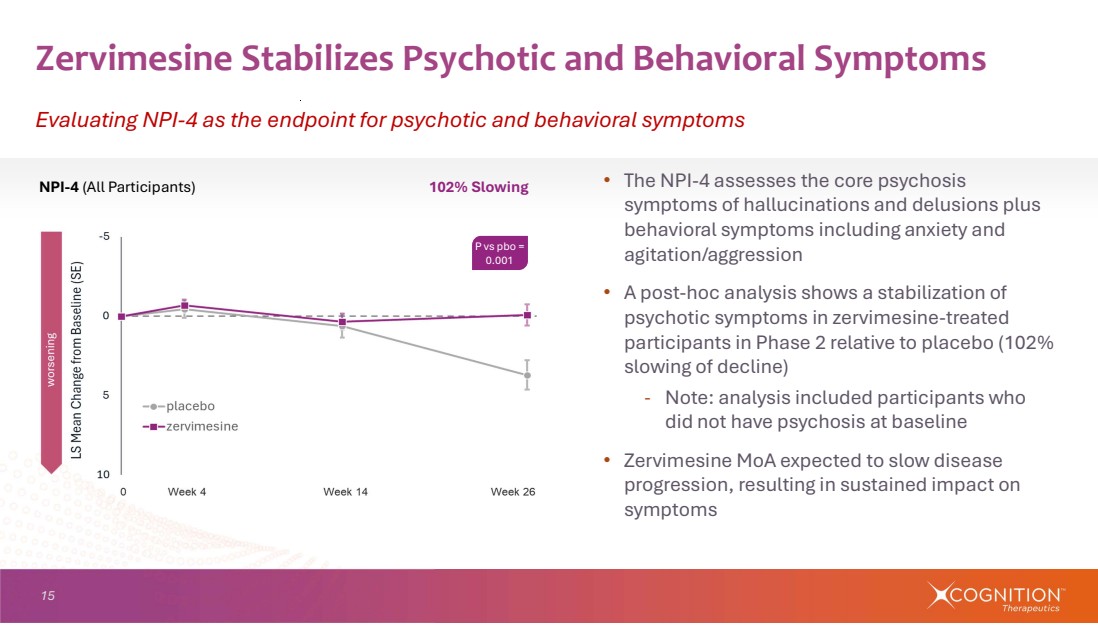
| 15 • The NPI-4 assesses the core psychosis symptoms of hallucinations and delusions plus behavioral symptoms including anxiety and agitation/aggression • A post-hoc analysis shows a stabilization of psychotic symptoms in zervimesine-treated participants in Phase 2 relative to placebo (102% slowing of decline) - Note: analysis included participants who did not have psychosis at baseline • Zervimesine MoA expected to slow disease progression, resulting in sustained impact on symptoms Zervimesine Stabilizes Psychotic and Behavioral Symptoms Evaluating NPI-4 as the endpoint for psychotic and behavioral symptoms NPI-4 (All Participants) 102% Slowing P vs pbo = 0.001 |
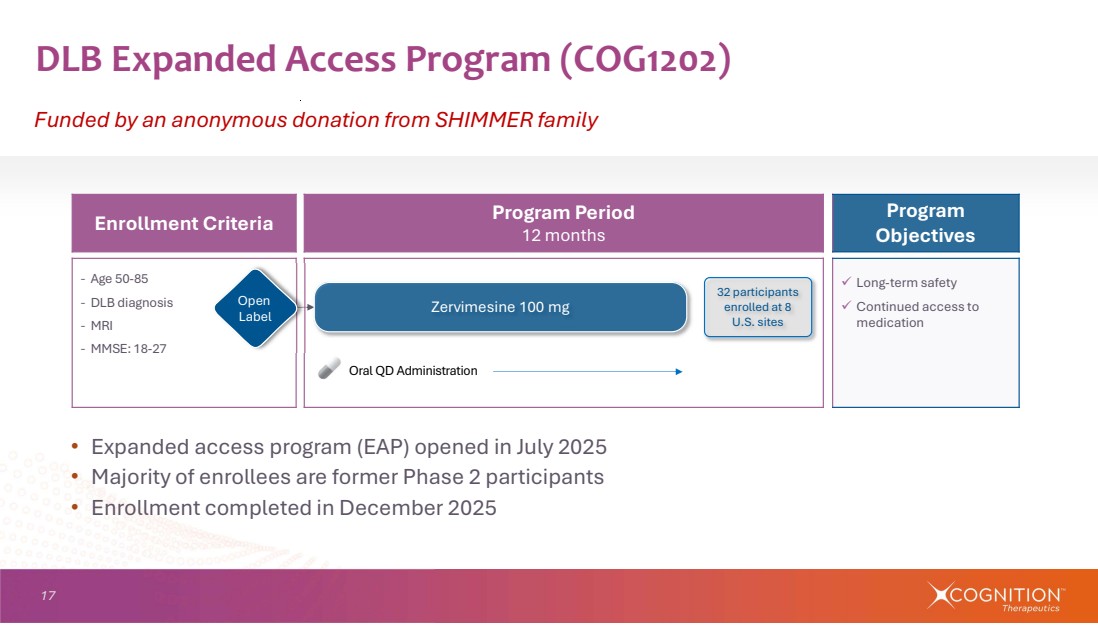
| 17 DLB Expanded Access Program (COG1202) Funded by an anonymous donation from SHIMMER family Program Objectives Program Period 12 months Enrollment Criteria Long-term safety Continued access to medication ‐ Age 50-85 ‐ DLB diagnosis ‐ MRI ‐ MMSE: 18-27 Zervimesine 100 mg Oral QD Administration Open Label 32 participants enrolled at 8 U.S. sites • Expanded access program (EAP) opened in July 2025 • Majority of enrollees are former Phase 2 participants • Enrollment completed in December 2025 |
| 18 Zervimesine Making an Impact for DLB Patients Participant from Phase 2 and EAP comments on his experience with zervimesine in SHIMMER In the mornings he would stand there and forget what to do. Now he gets out of bed and knows exactly what he needs to do. Our lives are absolutely more enriched.” For years, Susan would order for me at restaurants because I couldn't put sentences together. Now, I can order on my own. I make complete sentences, my speech is different, and my actions are different. It's amazing.” SHIMMER and EAP participant at Banner Sun Health Care partner Excerpted from Sep 2025 press release “Banner Health offers hope for Lewy body dementia.” https://tinyurl.com/2a2xmok9 |
| 19 • Meeting mid-May 2026; minutes June 2026 • Finalized registrational plan to follow • Randomization 1:1 zervimesine (100mg) : placebo • Endpoints are validated and have previously supported FDA approvals • Open-label extension planned following randomized treatment period for all participants Regulatory Next Steps New IND required to transition to FDA Division of Psychiatry |
| 95% slowing of cognitive decline in lower-p-tau217 'SHINE' participants Alzheimer’s Disease |
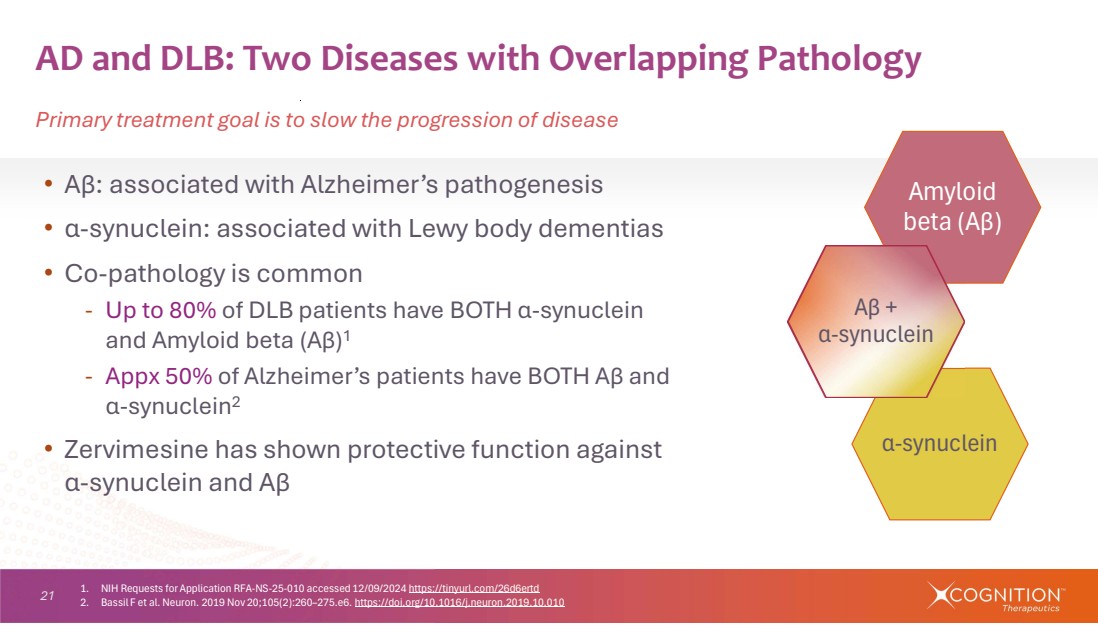
| 21 α-synuclein Amyloid beta (Aβ) • Aβ: associated with Alzheimer’s pathogenesis • α-synuclein: associated with Lewy body dementias • Co-pathology is common - Up to 80% of DLB patients have BOTH α-synuclein and Amyloid beta (Aβ)1 - Appx 50% of Alzheimer’s patients have BOTH Aβ and α-synuclein2 • Zervimesine has shown protective function against α-synuclein and Aβ AD and DLB: Two Diseases with Overlapping Pathology Primary treatment goal is to slow the progression of disease Aβ + α-synuclein 1. NIH Requests for Application RFA-NS-25-010 accessed 12/09/2024 https://tinyurl.com/26d6ertd 2. Bassil F et al. Neuron. 2019 Nov 20;105(2):260–275.e6. https://doi.org/10.1016/j.neuron.2019.10.010 |
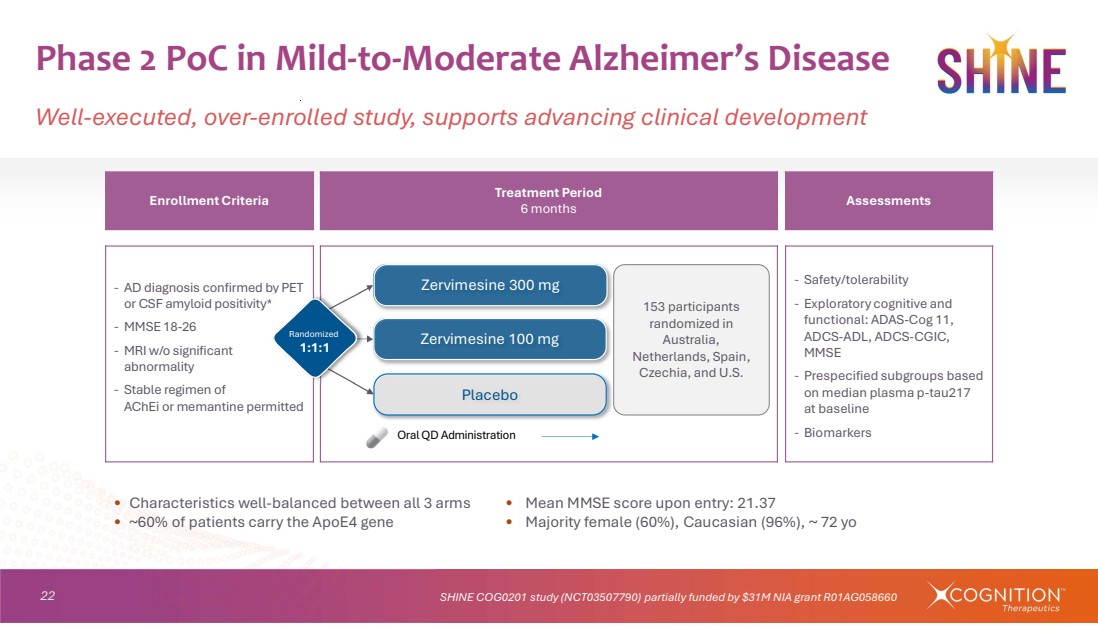
| 22 • Characteristics well-balanced between all 3 arms • Mean MMSE score upon entry: 21.37 • ~60% of patients carry the ApoE4 gene • Majority female (60%), Caucasian (96%), ~ 72 yo Phase 2 PoC in Mild-to-Moderate Alzheimer’s Disease Well-executed, over-enrolled study, supports advancing clinical development SHINE COG0201 study (NCT03507790) partially funded by $31M NIA grant R01AG058660 Assessments Treatment Period 6 months Enrollment Criteria ‐ Safety/tolerability ‐ Exploratory cognitive and functional: ADAS-Cog 11, ADCS-ADL, ADCS-CGIC, MMSE ‐ Prespecified subgroups based on median plasma p-tau217 at baseline ‐ Biomarkers ‐ AD diagnosis confirmed by PET or CSF amyloid positivity* ‐ MMSE 18-26 ‐ MRI w/o significant abnormality ‐ Stable regimen of AChEi or memantine permitted Placebo Zervimesine 100 mg Zervimesine 300 mg 153 participants randomized in Australia, Netherlands, Spain, Czechia, and U.S. Oral QD Administration Randomized 1:1:1 |
| 23 ADAS-Cog 11 ADAS-Cog 13 MMSE ADCS-ADL ADCS-CGIC 95% 103% 108% 68% 61% median p-tau217 Up to 108% Slowing on Assessments in Lower p-Tau Population Cognition Function Endpoints measured 100/300mg zervimesine pooled vs placebo Response of 100 and 300 mg dose groups are similar |
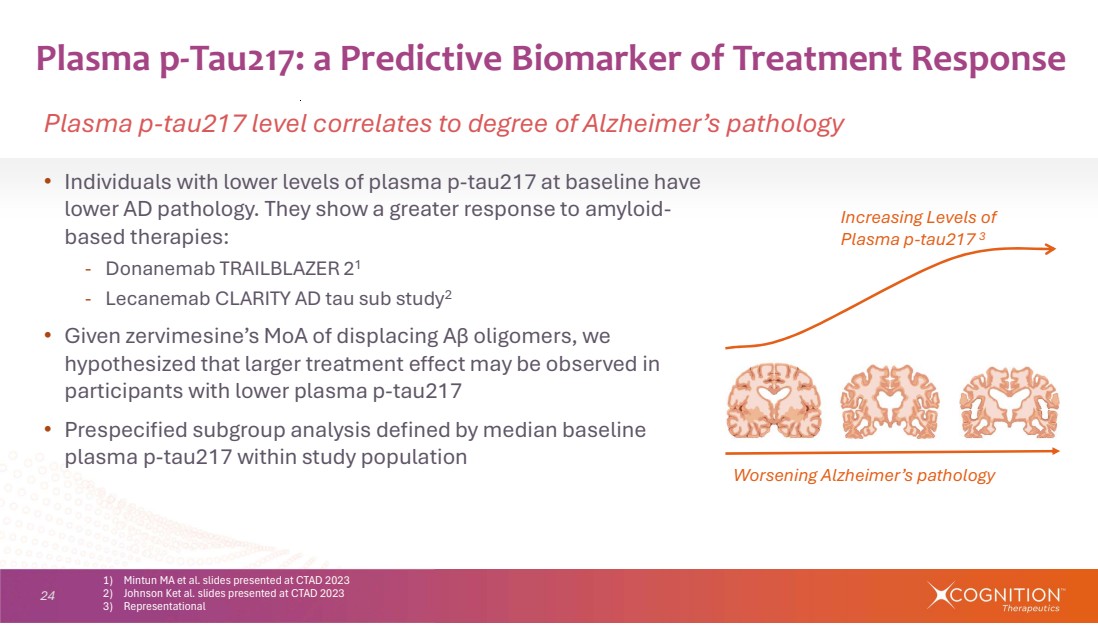
| 24 • Individuals with lower levels of plasma p-tau217 at baseline have lower AD pathology. They show a greater response to amyloid-based therapies: - Donanemab TRAILBLAZER 21 - Lecanemab CLARITY AD tau sub study2 • Given zervimesine’s MoA of displacing Aβ oligomers, we hypothesized that larger treatment effect may be observed in participants with lower plasma p-tau217 • Prespecified subgroup analysis defined by median baseline plasma p-tau217 within study population Plasma p-Tau217: a Predictive Biomarker of Treatment Response Plasma p-tau217 level correlates to degree of Alzheimer’s pathology 1) Mintun MA et al. slides presented at CTAD 2023 2) Johnson Ket al. slides presented at CTAD 2023 3) Representational Increasing Levels of Plasma p-tau217 3 Worsening Alzheimer’s pathology |
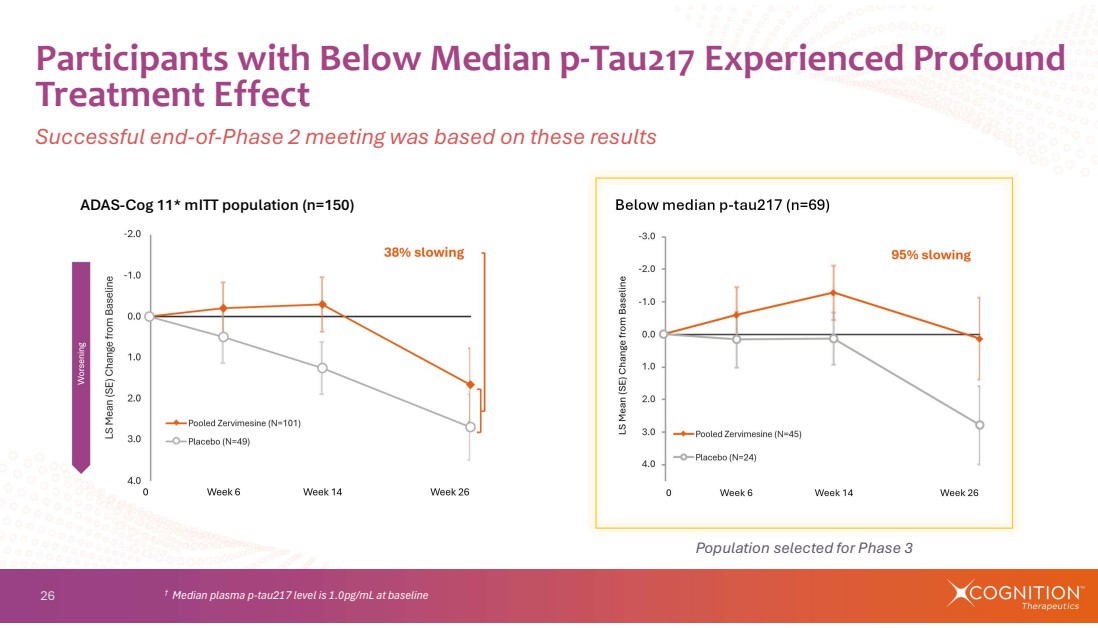
| 26 Participants with Below Median p-Tau217 Experienced Profound Treatment Effect Successful end-of-Phase 2 meeting was based on these results † Median plasma p-tau217 level is 1.0pg/mL at baseline ADAS-Cog 11* mITT population (n=150) Below median p-tau217 (n=69) Population selected for Phase 3 |
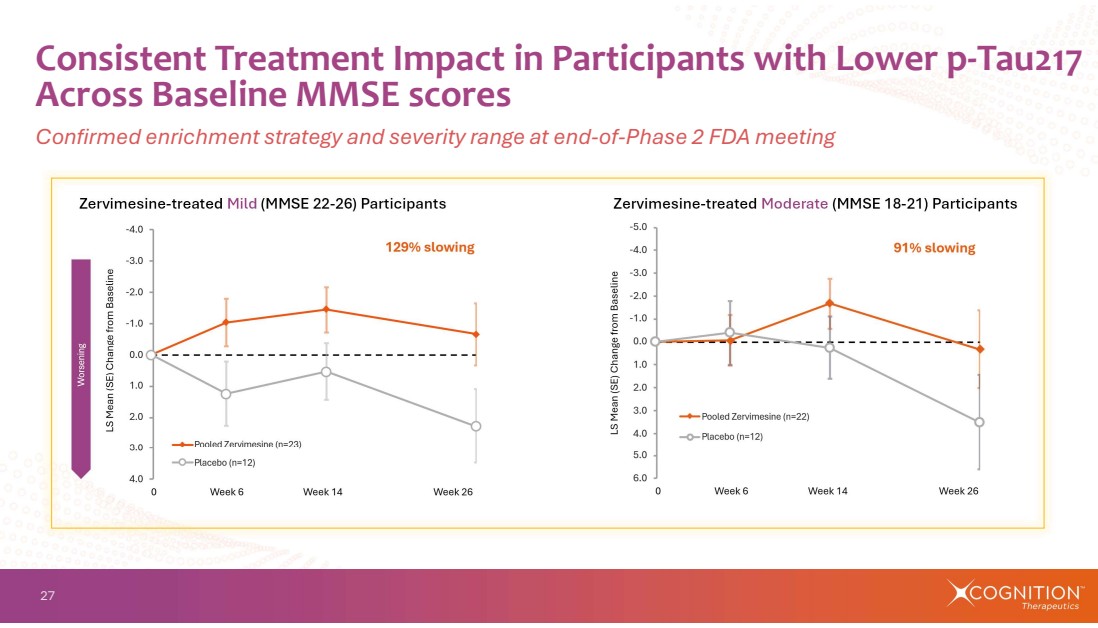
| 27 Consistent Treatment Impact in Participants with Lower p-Tau217 Across Baseline MMSE scores Confirmed enrichment strategy and severity range at end-of-Phase 2 FDA meeting Zervimesine-treated Mild (MMSE 22-26) Participants Zervimesine-treated Moderate (MMSE 18-21) Participants |
| 28 • Average age was ~75 years • Adverse events (AEs) were well balanced between treatment and placebo arms - Serious AEs occurred at comparable or higher rates in placebo- compared to zervimesine-treated arms - Three participants in 100mg dose group had LFT elevations greater than 3x ULN (6.8%) • Elevated liver enzymes normalized after cessation of drug • Discontinuations due to AEs are similar between 100mg dose groups (5.8%) and placebo groups (5%) Consistent Safety Profile Across Completed Phase 2 Studies Well tolerated in 271 participants with AD, DLB, dry AMD |
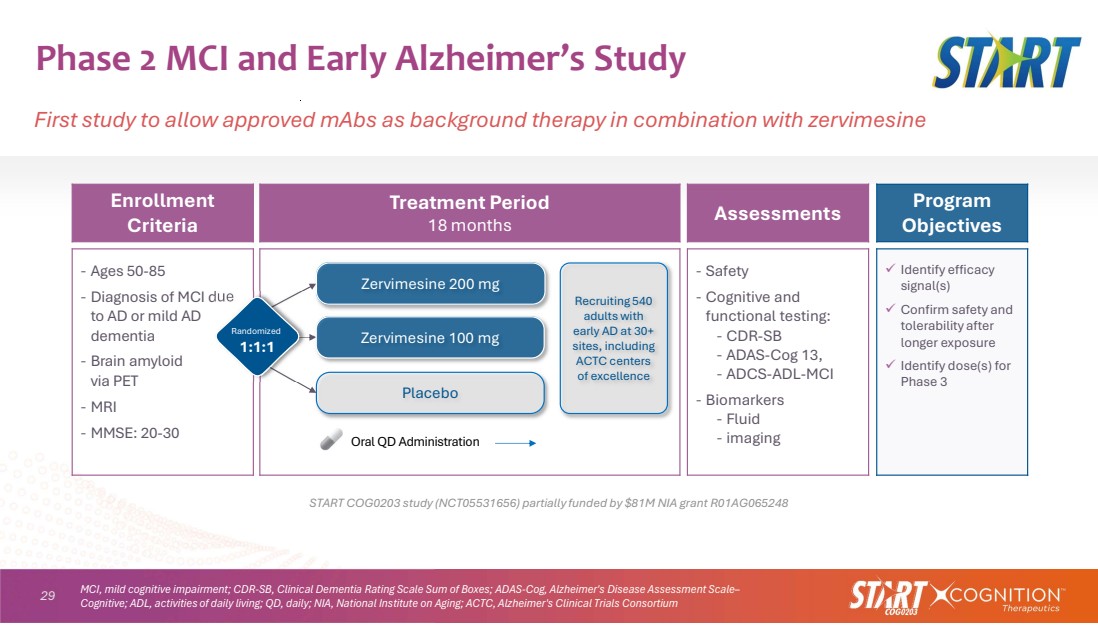
| 29 Phase 2 MCI and Early Alzheimer’s Study First study to allow approved mAbs as background therapy in combination with zervimesine START COG0203 study (NCT05531656) partially funded by $81M NIA grant R01AG065248 Program Objectives Assessments Treatment Period 18 months Enrollment Criteria Identify efficacy signal(s) Confirm safety and tolerability after longer exposure Identify dose(s) for Phase 3 ‐ Safety ‐ Cognitive and functional testing: ‐ CDR-SB ‐ ADAS-Cog 13, ‐ ADCS-ADL-MCI ‐ Biomarkers ‐ Fluid ‐ imaging ‐ Ages 50-85 ‐ Diagnosis of MCI due to AD or mild AD dementia ‐ Brain amyloid via PET ‐ MRI ‐ MMSE: 20-30 Placebo Zervimesine 100 mg Zervimesine 200 mg Oral QD Administration Randomized 1:1:1 Recruiting 540 adults with early AD at 30+ sites, including ACTC centers of excellence MCI, mild cognitive impairment; CDR-SB, Clinical Dementia Rating Scale Sum of Boxes; ADAS-Cog, Alzheimer's Disease Assessment Scale– Cognitive; ADL, activities of daily living; QD, daily; NIA, National Institute on Aging; ACTC, Alzheimer's Clinical Trials Consortium |
| 30 • Enrollment complete (n=545) - First participant enrolled February 2024 - Last participant enrolled in December 2025 • Cognitive and functional testing: CDR-SB, ADAS-Cog 13, ADCS-ADL-MCI • Trial duration: 18 months • Topline results anticipated 2H 2027 • 15-20% of participants enrolled on lecanemab or donanemab • Sub-studies: MRI, biomarkers, PET START Study Completed Enrollment 4Q 2025 Groundbreaking ability to test combination with mABs |
| 31 3 Major Diseases Addressed with Once-Daily Oral Pill Collective Phase 2 results support advancing zervimesine (CT1812) to registrational studies Dementia with Lewy Bodies Marked slowing of progression across multiple domains Geographic Atrophy Slowing of GA growth rate and area Alzheimer’s Disease Slowing of progression; robust response in lower tau cohort |
| 32 Current Financial Position Cash runway through 2Q 2027 Cash, cash equivalents, and restricted cash equivalents $ 37 M Grant funding for zervimesine studies Preclinical through Phase 2 ~$171 M Approximate funding used ($135 M) Remaining grant funding $36 M As of December 31, 2025 |
| Thank You Lisa Ricciardi President & CEO info@cogrx.com |