
Exhibit 99.2

The Quality Longevity Company Powered by a Prediction Markets Treasury

Forward - looking statements This presentation contains forward - looking statements within the meaning of Section 27A of the Securities Act of 1933, as amende d, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward - looking statements may be identified by words such as “expects,” “plans,” “proje cts,” “will,” “may,” “anticipates,” “believes,” “should,” “would,” “could,” “intends,” “estimates,” “suggests,” “target,” “has the potential to,” “goal,” and oth er words of similar meaning, including statements relating to the anticipated benefits of the digital asset treasury strategy of Enlivex Ltd. (the “Company”); the assets to be held by the Company; the expected future market, price, trading activity, and liquidity of the RAIN token; the impact of expanded exchange listings and increased toke n l iquidity on market participation and accessibility; the potential effects of digital asset liquidity on the liquidity of the Company’s ordinary shares; macroecono mic , political, and regulatory conditions surrounding digital assets; the Company’s plans for value creation and strategic positioning; market size and growth opportun iti es; regulatory conditions; competitive position; technological and market trends; future financial condition and performance; expected clinical trial results; marke t o pportunities for the results of current clinical studies and preclinical experiments; and the effectiveness of, and market opportunities for, Allocetra programs. Each forward - looking statement contained in this presentation is subject to risks and uncertainties that could cause actual resu lts to differ materially from those expressed or implied by such statement. Applicable risks and uncertainties include, among others, the risk of failure to real ize the anticipated benefits of the Company’s digital asset treasury strategy; changes in business, market, financial, political, and regulatory conditions; risks relating to the Company’s operations and business, including the highly volatile nature of the price, trading volume, and liquidity of RAIN and other cryptocurrencies; risks associated w ith digital asset exchange listings, trading venues, and market infrastructure; the risk that the price and liquidity of the Company’s ordinary shares may be correlated with the pri ce or liquidity of the digital assets it holds; risks related to increased competition in the industries in which the Company operates; risks relating to significant legal, com mercial, regulatory, and technical uncertainty regarding digital assets generally; risks relating to the treatment of crypto assets for U.S. and foreign tax purposes; and t hos e risks and uncertainties identified in the Company’s filings with the Securities and Exchange Commission, including the Company’s most recent Annual Report on Form 20 - F an d filings on Form 6 - K. The forward - looking statements in this presentation speak only as of the date of this document, and the Company undertakes no obligation to update or revise any of these statements, except as required by applicable law. This presentation has been prepared by the Company and is made for informational purposes only to familiarize yourself with t he Company . This presentation does not purport to be all inclusive or to contain all of the information the recipient may require in connection with an investigatio n o f the Company . Nothing contained in this presentation is, or should be construed as, a recommendation, promise or representation by the Company or any officer, direct or, employee, agent, an affiliate, representative or advisor of the Company. The Recipient should not construe the contents of this presentation as legal, tax, acc ounting or investment advice or a recommendation. This presentation is neither an offer to sell or purchase, nor a solicitation of an offer to sell, buy or sub scr ibe for any securities in any jurisdiction. 02

Enlivex : The World's First Healthspan - Wealthspan Company 03 ● Advanced Clinical - stage Allocetra immunotherapy currently targeting age - related osteoarthritis diseases ● Addresses quality - of - life decline in aging populations: mobility, independence Primary Engine Thesis: Healthspan extension becomes a multi - trillion - dollar category as the world ages Quality Longevity Through Potential Medical Breakthroughs ● Largest institutional holder of RAIN, the governance token for the leading decentralized prediction market ● Public equity vehicle for deflationary, revenue - backed tokenomics with buybacks - and - burns Secondary Engine Thesis: Prediction markets eclipse forecasting, capturing trillions in volume, and replace speculation. Prediction Markets Treasury

Executive Summary 04 The Capital Structure Innovation: Enlivex is pioneering a hybrid institutional model, utilizing a digital asset treasury designed to fuel breakthrough quality longevity therapeutics. The Quality Longevity Therapeutics: Allocetra targets primarily age - related knee - osteoarthritis, designed to rellief pain , increase mobility and independence, leading to higher quality of longevity years. The Economic Accelerator: Through a $212M private placement, Enlivex’s strategy evolved with a prediction market treasury in November 2025 using the RAIN token to potentially capture exponential sector growth. Deflationary Tokenomics: Direct upside participation in prediction markets via a protocol that systematically buys back and burns supply based on trading volume, which is beneficial to large token holders . Asymmetric Convexity: Exclusive option to acquire up to $898 million in RAIN tokens at $0.0033, expiration date Dec 31, 2027, providing ENLV shareholders with - 400% “option” coverage with an anti - dilutive effect. The Dual Engine Architecture

How Capital Velocity Potentially Accelerates Longevity Breakthroughs 05 The Traditional Model: Clinical development funding through frequent equity raises that progressively dilute shareholders. The Enlivex Architecture: Wealthspan Velocity: Prediction markets treasury generates non - dilutive capital through RAIN's deflationary mechanics and active yield. Healthspan Durability: Capital funds Allocetra development, advancing therapies that extend quality of life globally. The Cycle Compounds: Treasury growth funds clinical advancement. Potential clinical breakthroughs expand market opportunity and shareholder value.

Prediction Markets: How Distributed Intelligence Prices the Future 07 A prediction market is a trading platform where contract value is determined entirely by the outcome of future events. Participants buy and sell outcome - specific positions, putting real capital behind their convictions. The result: a real - time probability engine. One that aggregates information from tens of thousands of independent actors simultaneously, potentially outperforming polls, single analysts, and centralized forecasting models. Academic research has consistently found that prediction markets produce more accurate probability estimates than expert consensus, because they incorporate information asymmetries and enforce accountability through financial risk. These markets are now processing political outcomes, macroeconomic events, sports, and everything in between.

The Institutional Transition Is Already Underway 08 $2BN investment ($8BN Valuation) by Intercontinental Exchange (ICE), the parent company of New York Stock Exchange (NYSE) in Polymarket Kalshi's $1BN financing ($22BN Valuation) with Coatue Management , following previous financing round led by Paradigm, Andreessen Horowitz and Sequoia Source : ICE Announces Strategic Investment in Polymarket , Oct 7 2025, Bloomberg Mar 19, 2026
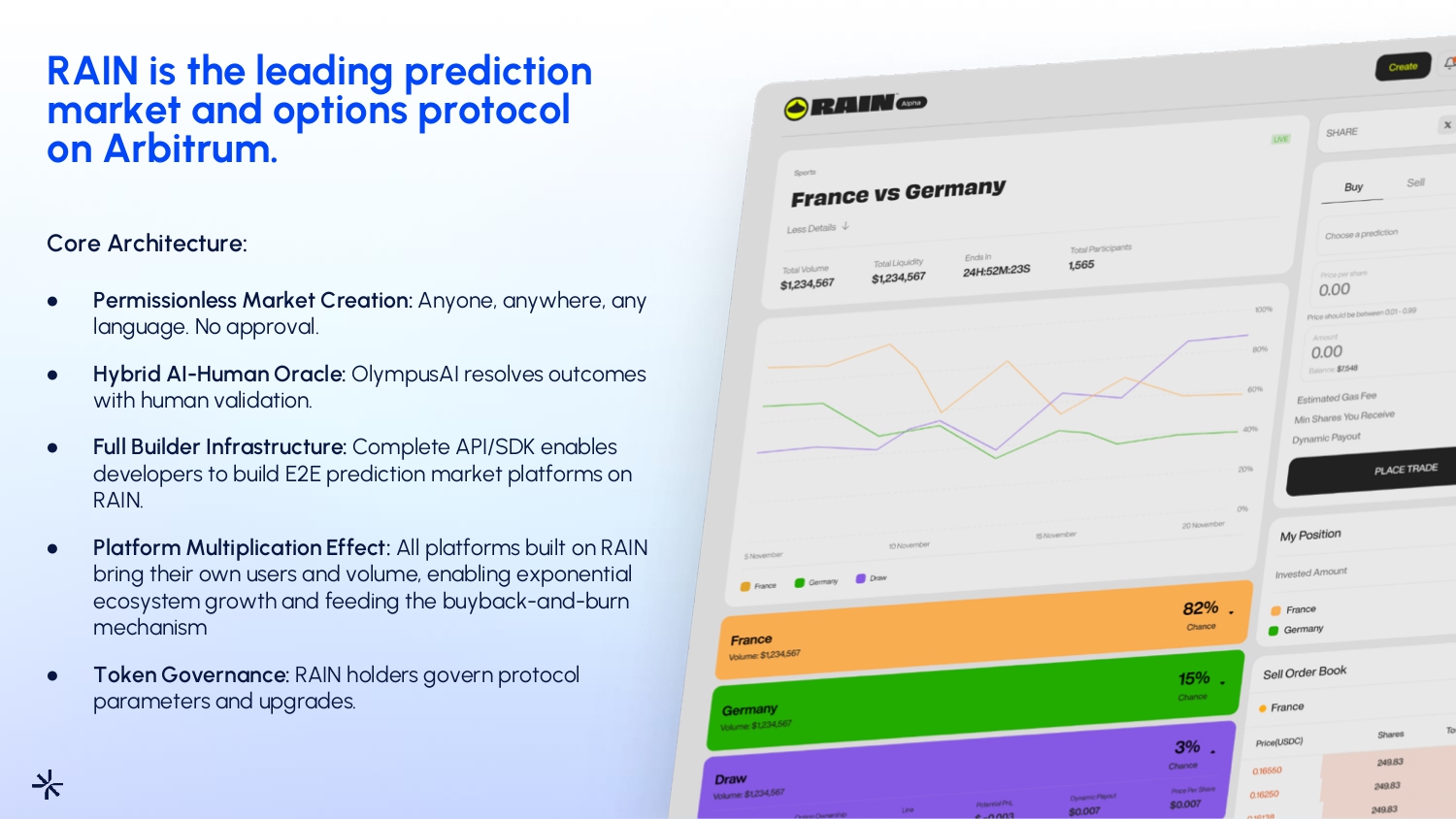
RAIN is the leading prediction market and options protocol on Arbitrum . 08 Core Architecture: ● Permissionless Market Creation: Anyone, anywhere, any language. No approval. ● Hybrid AI - Human Oracle: OlympusAI resolves outcomes with human validation. ● Full Builder Infrastructure: Complete API/SDK enables developers to build E2E prediction market platforms on RAIN. ● Platform Multiplication Effect: All platforms built on RAIN bring their own users and volume, enabling exponential ecosystem growth and feeding the buyback - and - burn mechanism ● Token Governance: RAIN holders govern protocol parameters and upgrades.
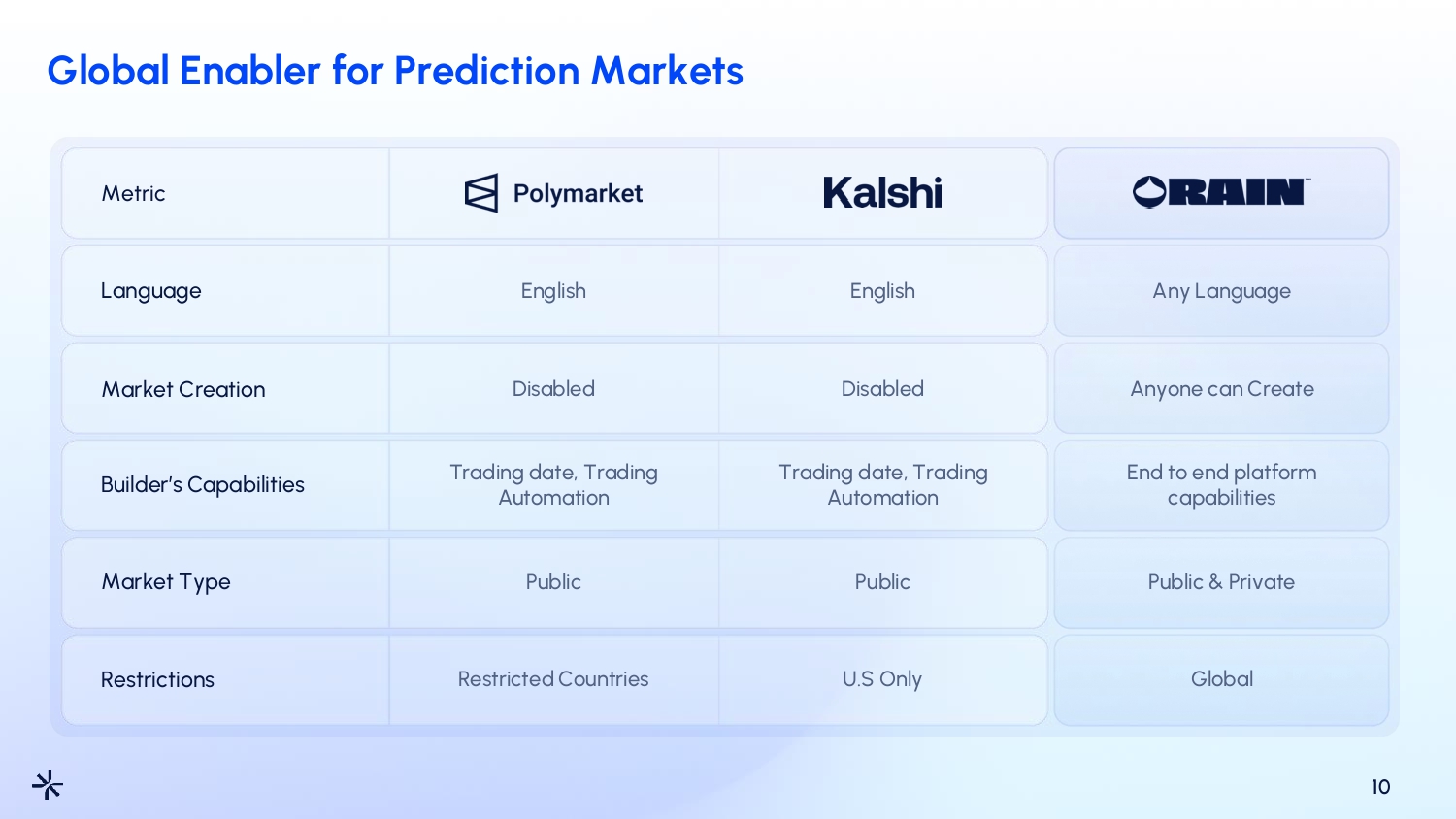
Global Enabler for Prediction Markets 10 Metric Language Market Creation Builder’s Capabilities Market Type Restrictions English Disabled Trading date, Trading Automation Public Restricted Countries English Disabled Trading date, Trading Automation Public U.S Only Any Language Anyone can Create End to end platform capabilities Public & Private Global

The 5% Protocol Fee and the Mechanistic Deflationary Engine 2.5% — Participation Rewards 11 Distributed to market participants to ensure deep, self - sustaining liquidity: ● 1.2% to Market Creators ● 1.2% to Liquidity Providers ● 0.1% to Resolvers 2.5% — Buyback & Burn The protocol automatically acquires RAIN from the open market and permanently removes it from circulation and reducing supply

RAIN Buyback & Burn Forecast 12 $2.5B in Cumulative Supply Reduction by 2030 Prediction Markets Forecast: ● $1 trillion trading volume by 2030 1 ● RAIN market shares estimated to be 5% 2 Period 2026 – 2029 (Aggregate) 2030 (Annual) Through 2030 (Total) Buyback & Burn Volume $1.25 Billion $1.25 Billion $2.25 Billion 1 Industry analysis by Eilers and Krejcik Gaming (EKG), December 2025 2 Rain Foundation forecast

Rain Ecosystem Expansion Expected Roadmap Provided By The Rain Foundation 13 Ecosystem Expansion Developer Infrastructure Network Growth Builders program Platform partnerships SDK / API release Shared liquidity protocol Mainnet production Ecosystem integrations Milestone Timeline Q1 2026 Q1 2026 Q2 2026 Q2 2026 Q2 2026 Q3 2026 Description

Expanding market accessibility and liquidity depth for RAIN 14 Current Listings: ● Reduced slippage for institutional - scale trades ● Enhanced price discovery through deeper order books ● 24/7 global trading depth enabling algorithmic strategies Structural Impact: Target: 1 top - 5 exchange listing in H1 2026

Scaling Rain Protocol’s global presence 15 Objective: ● 100+ KOL Activations across crypto - native, finance, and technology channels ● Ambassador Program deployment ● International Hackathon Series to accelerate developer platform construction on RAIN infrastructure ● Major Conference Presence Campaign Architecture: Timeline: Q2 2026 The Rain Foundation is executing a global campaign to drive protocol adoption, platform volume, and institutional awareness.

Leveraging the economic accelerator 16 Enlivex is currently the largest corporate holder of RAIN. Enlivex instructed its RAIN asset manager, at its discretion, to utilize yield - enhancement activities, which potentially could result in outperformance of the spot token. ● Full Protocol Staking: Capturing baseline governance and validation rewards. ● Restaking Upon Availability: Compounding yield through emerging restaking protocols. ● Selective DeFi Deployment: Tactical allocation to high - quality liquidity pools. Potential active yield - enhancement activities performed by t he asset manager:

Portfolio Update 16 As part of its 2025 year - end assessment, the Company currently estimates that the value of the RAIN portfolio (short and long - term digital assets) as of December 31, 2025, was approximately $607 million, representing an estimated gain from appreciation in portfolio valuation (income on digital assets) of approximately $363 million in 2025. In addition, the Company holds an option to purchase additional RAIN tokens from the RAIN Foundation. The Company currently estimates that this option had a value of approximately $1.7 billion as of December 31, 2025, representing a change in fair value of of the option of $1.26 billion in 2025. These figures are only preliminary estimates, and are calculated on a pre - tax basis. The Company’s tax rate is 23%. Our final results could differ from these estimates upon completion of our financial closing procedures due to final adjustments and developments that may arise between now and the time our financial statements for the year ended December 31, 2025 are issued. For example, during the course of the preparation of our financial statements and related notes, additional items that would require adjustments to the foregoing preliminary estimated financial information may be identified. These estimates should not be viewed as a substitute for full audited financial statements prepared in accordance with GAAP, which will be filed with Enlivex's Annual Report on Form 20 - F for the year ended December 31, 2025. The updated unaudited mark - to - market treasury metrics are publicly available on the Company’s website: https://enlivex.com/dashboard/

Quality Longevity Therapeutics 17
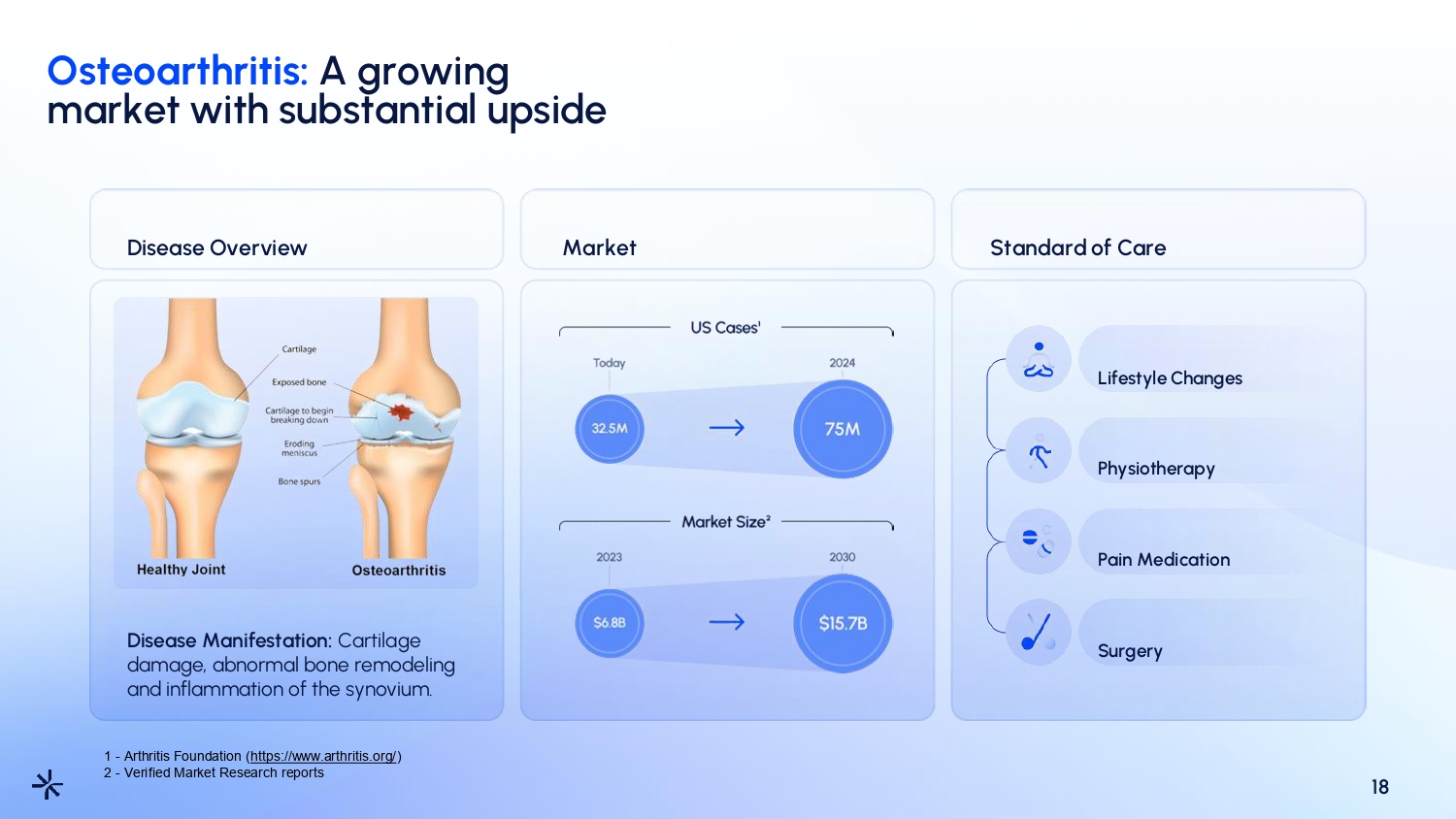
Osteoarthritis: A growing market with substantial upside 18 Disease Overview Market Standard of Care Disease Manifestation: Cartilage damage, abnormal bone remodeling and inflammation of the synovium. Lifestyle Changes Physiotherapy Pain Medication Surgery 1 - Arthritis Foundation ( https://www.arthritis.org/ ) 2 - Verified Market Research reports

KNEE OSTEOARTHRITIS: Increased risk of mortality and reduced quality of longevity 19 The most comprehensive and recent meta - analysis (2024), covering over one million participants, found that knee OA patients had a 21% higher risk of all - cause mortality compared to those without knee OA 1 . Knee OA increases mortality through mediating pathways: ● Reduced physical activity and walking disability: Reduced mobility leads to cardiovascular deconditioning, weight gain, and metabolic deterioration. ● NSAID use: Chronic NSAID use for OA pain carries its own cardiovascular and gastrointestinal risks. ● Comorbidities: Diabetes, obesity, and cardiovascular disease - common in OA patients are major independent predictors of death. ● Social isolation was independently associated with increased mortality risk among individuals with arthritis 2 . 1 Risk of all - cause mortality in patients with knee osteoarthritis: A systematic review and meta - analysis of cohort studies, Osteoarthr Cartil Open. 2024 Nov 8 2 Association Between Osteoarthritis and Social Isolation: Data from the EPOSA Study, J Am Geriatr Soc. 2019 Sep 17
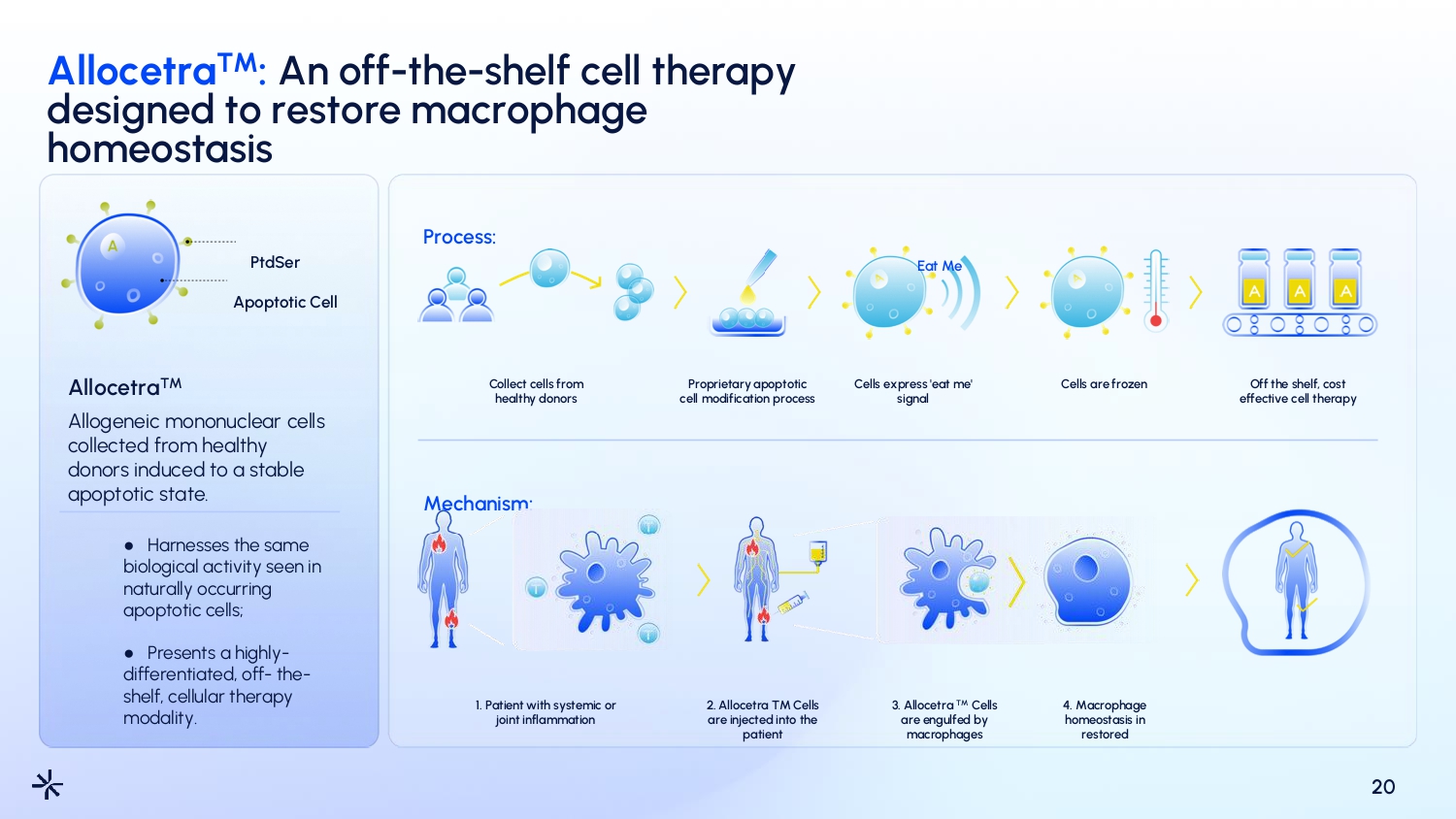
Allocetra TM : An off - the - shelf cell therapy designed to restore macrophage homeostasis 20 PtdSer Apoptotic Cell Allocetra TM Allogeneic mononuclear cells collected from healthy donors induced to a stable apoptotic state. ● Harnesses the same biological activity seen in naturally occurring apoptotic cells; ● Presents a highly - differentiated, off - the - shelf, cellular therapy modality. Process: Collect cells from healthy donors Mechanism: Proprietary apoptotic cell modification process Cells express 'eat me' signal Eat Me Cells are frozen Off the shelf, cost effective cell therapy 1. Patient with systemic or joint inflammation 2. Allocetra TM Cells are injected into the patient 3. Allocetra TM Cells are engulfed by macrophages 4. Macrophage homeostasis in restored

ENX - CL - 05 - 001: PHASE I/lla 2 - stage trial design - randomized, double - blind, placebo - controlled, multi - country study 21 Patient Criteria ● Patients with symptomatic moderate to severe knee OA who have failed to respond to conventional OA therapy; ● Age 45 - 80 years; ● Kellgren - Lawrence (K - L) Grade 2 or 3. ClinicalTrials.gov Registration: NCT06233474 NRS = numerical rating scale. WOMAC = Standard knee questionnaire evaluating pain, stiffness & physical function Phase I: Dose escalation & safety 15 patients Independent safety committee→ no negative safety signal, highest dose selected for Phase lla Endpoints 134 patients 3 injections (in total) of Allocetra TM or Placebo, each injection 2 weeks from the previous injection Safety and tolerability. Primary Change in pain and function assessments (NRS, WOMAC) Secondary Efficacy: 3 - month, 6 - month Safety: 12 - month follow - up Timepoints Efficacy objectives ● Reduction in pain, increase in function and reduction in stiffness ● Numerical grading based on the patients' assessment using a questionnaire ● The validated questionnaire is named WOMAC ● Aligned with FDA's accepted Phase III endpoints and timepoints Phase IIa : Randomized, double - blind, placebo - controlled
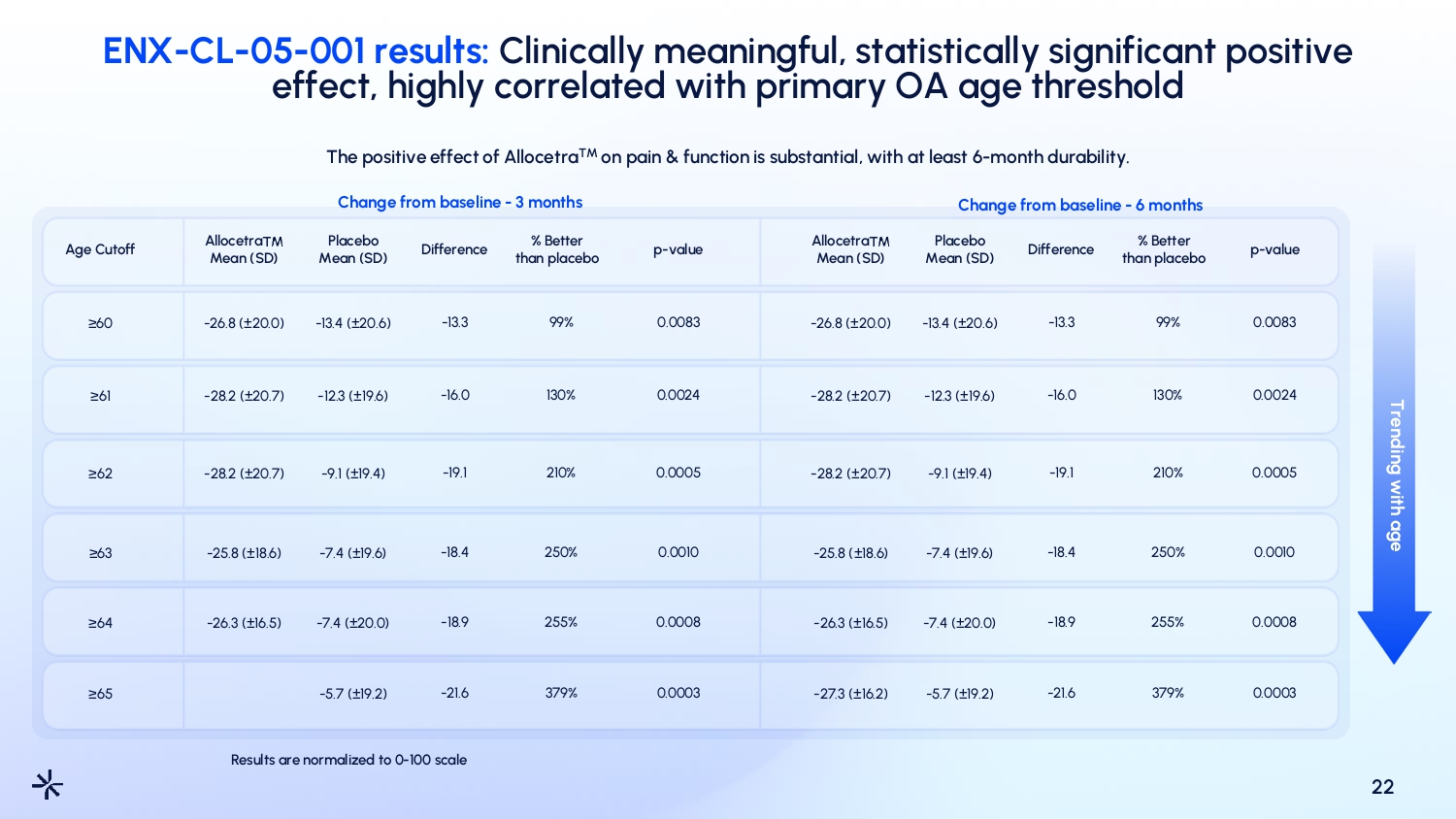
ENX - CL - 05 - 001 results: Clinically meaningful, statistically significant positive effect, highly correlated with primary OA age threshold 22 The positive effect of Allocetra TM on pain & function is substantial, with at least 6 - month durability. Change from baseline - 3 months Change from baseline - 6 months Age Cutoff ≥60 ≥61 ≥62 ≥63 ≥64 ≥65 Allocetra Mean (SD) - 26.8 ( ± 20.0) - 28.2 ( ± 20.7) - 28.2 ( ± 20.7) - 25.8 ( ± 18.6) - 26.3 ( ± 16.5) Results are normalized to 0 - 100 scale Placebo Mean (SD) - 13.4 ( ± 20.6) - 12.3 ( ± 19.6) - 9.1 ( ± 19.4) - 7.4 ( ± 19.6) - 7.4 ( ± 20.0) - 5.7 ( ± 19.2) Difference - 13.3 - 16.0 - 19.1 - 18.4 - 18.9 - 21.6 % Better than placebo 99% 130% 210% 250% 255% 379% p - value 0.0083 0.0024 0.0005 0.0010 0.0008 0.0003 Allocetra Mean (SD) - 26.8 ( ± 20.0) - 28.2 ( ± 20.7) - 28.2 ( ± 20.7) - 25.8 ( ± 18.6) - 26.3 ( ± 16.5) - 27.3 ( ± 16.2) Placebo Mean (SD) - 13.4 ( ± 20.6) - 12.3 ( ± 19.6) - 9.1 ( ± 19.4) - 7.4 ( ± 19.6) - 7.4 ( ± 20.0) - 5.7 ( ± 19.2) Difference - 13.3 - 16.0 - 19.1 - 18.4 - 18.9 - 21.6 % Better than placebo 99% 130% 210% 250% 255% 379% p - value 0.0083 0.0024 0.0005 0.0010 0.0008 0.0003 Trending with age

The Quality Longevity Opportunity 23 Management team with a track record of creating shareholder value and getting drug products through marketing approvals globally in multi - billion dollar market segments. Cost - effective, novel therapeutic modality with strong IP protection. Targeted at high and low grade inflammation in multi - billion dollar segments with poor treatment alternatives. Platform for multiple indications. Allocetra TM can be infused systemically or locally to treat various diseases. Simple, scalable, and cost - effective manufacturing process resulting in an off - the - shelf cell therapy. Favorable safety profile demonstrated across 200+ patients. Clinical data supportive of proposed MOA. Expected to commence a global Phase 2b trial in knee OA with first patient dosed mid 2026 and topline data expected at Q2 and Q3 2027 supporting a pivotal Phase 3 study and potential partnership. Clinically meaningful and statistically significant results in age - related knee osteoarthritis supporting late - stage development.

Enlivex Quality Longevity Team 24 Dr. Oren Hershkovitz, CEO of Enlivex since 2019, is a biotech leader with 15+ years’ experience advancing Phase I – III programs and leading global development partnerships. Oren Hershkovitz, Ph.D. Chief Executive Officer Prof. Dror Mevorach, co - founder of Enlivex, is a leading immunology expert, former Hadassah Chairman of Medicine, and author of 140+ papers on apoptotic cell research. Prof. Dror Mevorach, M.D. Co - Founder & Scientific Adviser Einat Galamidi joined Enlivex in 2022 to lead clinical programs, bringing 20+ years’ drug development experience and pivotal Phase 3 leadership that secured FDA approval for Omisirge®. Einat Galamidi, M.D. VP Medical Dr. Veronique Amor - Baroukh, Senior Director of Operations at Enlivex since 2015, leads CMC, quality and clinical supply, advancing Allocetra’s GMP and formulation development. Veronique Amor - Baroukh, Ph.D. Senior Director of Operations Shai Novik is Co - Founder and Executive Chairman of Enlivex (Nasdaq: ENLV), leading immunotherapy innovation. He founded PROLOR, sold it for $560M, and secured major Pfizer deals. Shai Novik Executive Chairman Shachar Shlosberger, CPA, has served as Enlivex CFO since 2016, bringing 12+ years’ biotech finance experience, including leadership roles at PROLOR Biotech. Shachar Shlosberger CFO Mrs. Tavor holds an MSc. in Quality Engineering Biotechnology Systems and has over 20 years of experience in regulatory and quality affairs in the pharmaceutical and biotech industry. Iris Tavor Senior Director of Quality & Regulatory Affairs Dr. Ankri leads Pre - Clinical and Clinical Pharmacology at Enlivex. She has extensive immunotherapy research experience and holds a Ph.D. in Cancer Immunotherapy from Bar - Ilan University, Israel. Chen Ankri, Ph.D. Director of Pre - Clinical & Clinical Pharmacology Mrs. Arad joined Enlivex in 2021 as Director of Human Resources and has over 15 years of HR experience in the biotech industry, including 10 years at FutuRx. She holds a B.Ed. and HR certifications from Bar - Ilan University. Sigal Arad Director of Human Resources

Enlivex treasury team 25 Matteo focuses on engaging sovereign wealth funds and governments to develop national crypto treasury strategies, including the adoption of the Enlivex RAIN strategy. He brings decades of political leadership, international relations expertise, and high - level diplomatic access. Matteo Renzi Board Member, Enlivex Former Prime Minister of Italy Former CEO of the Israeli ministry of transport, has held senior roles as BD consultant, at Yandex/Nebius, JVP. His expertise spans strategic investment, technology growth, and high - level public sector management. Ofer Malka Strategy advisor, Enlivex Former Global Investigator in Binance’s Sanctions Department. advises on compliance, sanctions, and regulatory strategy. Amit Levin Advisor Entrepreneur, Supervisory Board Member & Investor. Former Federal Minister of the Republic of Austria. Elli Köstinger Advisor CEO of GEMS and formerly with eToro, brings extensive expertise in investment strategy, capital markets, and the blockchain industry. Isaac Joshua Senior Investment Advisor, Enlivex

Where prediction markets fund longevity breakthroughs. shachar@enlivex.com www.enlivex.com