| APEX Part A 52-week readout March 23, 2026 |
| 2 © Apogee Therapeutics, Inc. Disclaimers and Forward-looking statements Other than statements of historical facts, all statements included in this presentation are forward-looking statements, including statements about our plans for our current and future product candidates and programs; the anticipated timing of initiation of our clinical trials, including the Phase 3 trial of zumilokibart (APG777) in AD; the expected timing of results from our clinical trials, including the initial readout from Part B of our Phase 2 trial of zumilokibart in AD and the initial readout from our Phase 1b trial of APG279 in AD; planned clinical trial designs; our plans for current and future clinical trials and expansion indications; the potential launch timing of our product candidates, including zumilokibart in AD; the potential clinical benefit, treatment outcomes, dosing regimen, and safety of zumilokibart and our other product candidates, including combination therapies, and any other potential programs; our expected timing for future pipeline updates; our potential path to regulatory approval; our expectations regarding the time period over which our capital resources will be sufficient to fund our anticipated operations, our cash runway, our planned business strategies; and estimates of market size. In some cases, you can identify forward-looking statements by terms such as “anticipate,” “believe,” “can,” “could,” “design,” “estimate,” “expect,” “intend,” “likely,” “may,” “might,” “plan,” “potential,” “predict,” “suggest,” “target,” “will,” “would,” or the negative of these terms, and similar expressions intended to identify forward-looking statements. The forward-looking statements are based on our beliefs, assumptions and expectations of future performance, taking into account the information currently available to us. These statements are only predictions based upon our current expectations and projections about future events. The data included in this presentation may be subject to change following the availability of additional data or following a more comprehensive review of the data. Forward-looking statements are subject to known and unknown risks, uncertainties and other factors that may cause our actual results, level of activity, performance or achievements to be materially different from those expressed or implied by such forward-looking statements, including those risks described in “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our Annual Report on Form 10-K for the year ended December 31, 2025, filed with the U.S. Securities and Exchange Commission (the SEC) on March 2, 2026 and subsequent disclosure documents we have filed and may file with the SEC. Although we have attempted to identify important factors that could cause actual results to differ materially from those contained in forward-looking statements, there may be other factors that cause results not to be as anticipated, estimated or intended. We claim the protection of the Safe Harbor contained in the Private Securities Litigation Reform Act of 1995 for forward-looking statements. This presentation concerns drug candidates that are under clinical investigation, and which have not yet been approved by the U.S. Food and Drug Administration. These are currently limited by federal law to investigational use, and no representation is made as to their safety or effectiveness for the purposes for which they are being investigated. The assumptions used in the preparation of this presentation, although considered reasonable by us at the time of preparation, may prove to be incorrect. You are cautioned that the information is based on assumptions as to many factors and that actual results may vary from the results projected and such variations may be material. Accordingly, you should not place undue reliance on any forward-looking statements contained herein or rely on them as predictions of future events. All forward-looking statements in this presentation apply only as of the date made and are expressly qualified by the cautionary statements included in this presentation. We do not undertake to update any forward-looking statements, except in accordance with applicable securities laws. This presentation also uses estimates and other statistical data made by independent parties and us relating to the data and analysis about our industry. The data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. The trademarks, trade names and service marks appearing in this presentation are the property of their respective owners. Certain information contained in this presentation relate to or are based on studies, publications and other data obtained from third-party sources as well as our own internal estimates and research. While we believe these third-party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. This presentation contains data based on cross-study comparisons and not based on any head-to-head clinical trials. Cross-study comparisons are inherently limited and may suggest misleading similarities and differences. The values shown in cross-study comparisons are directional and may not be directly comparable. |
| 3 © Apogee Therapeutics, Inc. Agenda Introduction Michael Henderson, MD Chief Executive Officer Carl Dambkowski, MD Chief Medical Officer Kristine Nograles, MD SVP, Head of Clinical Development & Medical Affairs APEX Phase 2 Part A 52-Week Results Zumilokibart Development Program Building a Leading I&I Company Analyst Q&A Michael Henderson, MD Chief Executive Officer Michael Henderson, MD, CEO Carl Dambkowski, MD, CMO Jane Pritchett Henderson, CFO Jeff Hartness, CCO Invited KOL: Ruth Ann Vleugels, MD, MPH, MBA Unmet Need in Atopic Dermatitis Invited KOL: Ruth Ann Vleugels, MD, MPH, MBA Mass General Brigham, Harvard Medical School |
| Introduction Michael Henderson, MD Chief Executive Officer |
| 5 © Apogee Therapeutics, Inc. Building a leading I&I company to address Type 2 inflammatory conditions Atopic dermatitis (AD) is growing rapidly and could be the largest I&I market • AD market is projected to reach $50B+ • New entrants with limited differentiation are quickly becoming blockbusters © Apogee Therapeutics, Inc. 5 Zumilokibart is a potentially best-in-class antibody targeting IL-13, the primary driver of AD and other Type 2 inflammatory diseases • Zumilokibart’s prior 16-week induction data showed rapid reduction in itch and lesions • 52-week results now demonstrate zumilokibart maintains those responses and could be the first every 6-month dosed drug in dermatology, if approved • Both every 3- and 6-month dosing led to deepening across all endpoints, in contrast to DUPIXENT NOTE: Future $50B AD market size based on EvaluatePharma and company projections. Actual market size may differ materially.Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. |
| 6 © Apogee Therapeutics, Inc. APEX PART A Data support planned zumilokibart Phase 3 initiation in 2H 2026 with every 3- and every 6-month dosing regimens Durable maintenance of response at 52 weeks Maintenance of EASI-75: • 75% every 3-month dosing • 85% every 6-month dosing Maintenance of IGA 0/1: • 86% every 3-month dosing • 78% every 6-month dosing Deepening responses through Week 52 • Deepening of response seen across all lesional and itch endpoints tested for both every 3- and 6-month dosing • Up to 36 percentage points of deepening on the most stringent endpoints (IGA 0/1, EASI-90, EASI-100) Well-tolerated safety profile consistent with class • Both maintenance regimens well-tolerated • Safety profile generally in line with class APEX 52-week data demonstrated a potentially best-in-class profile with both every 3-month and every 6-month dosing NOTE: Safety data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. IGA = Investigator Global Assessment. EASI = Eczema Area and Severity Index. Phase 3 plans subject to regulatory outcomes. Zumilokibart assessed Validated Investigator Global Assessment (vIGA) during APEX Part A. |
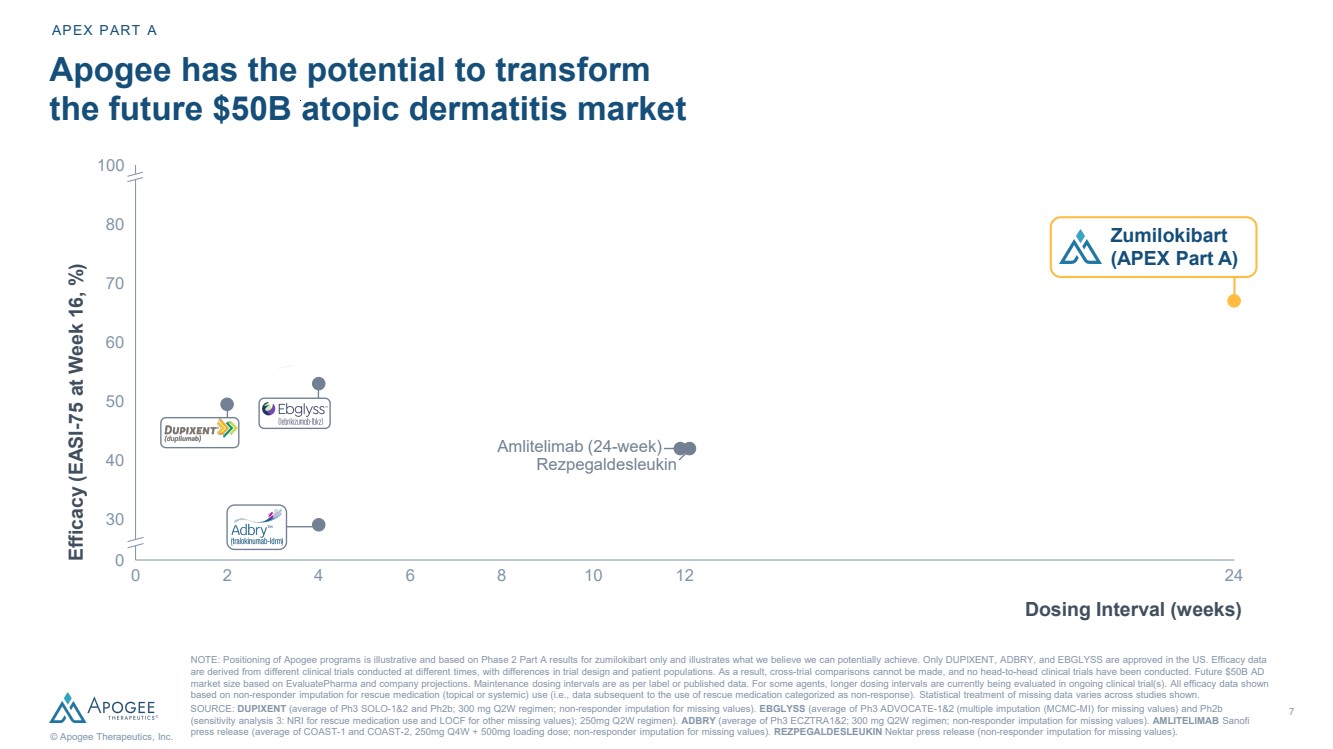
| 7 © Apogee Therapeutics, Inc. 0 2 4 6 8 10 12 24 0 30 40 50 60 70 80 90 Amlitelimab (24-week) Apogee has the potential to transform the future $50B atopic dermatitis market Efficacy (EASI-75 at Week 16, %) APEX PART A NOTE: Positioning of Apogee programs is illustrative and based on Phase 2 Part A results for zumilokibart only and illustrates what we believe we can potentially achieve. Only DUPIXENT, ADBRY, and EBGLYSS are approved in the US. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Future $50B AD market size based on EvaluatePharma and company projections. Maintenance dosing intervals are as per label or published data. For some agents, longer dosing intervals are currently being evaluated in ongoing clinical trial(s). All efficacy data shown based on non-responder imputation for rescue medication (topical or systemic) use (i.e., data subsequent to the use of rescue medication categorized as non-response). Statistical treatment of missing data varies across studies shown. SOURCE: DUPIXENT (average of Ph3 SOLO-1&2 and Ph2b; 300 mg Q2W regimen; non-responder imputation for missing values). EBGLYSS (average of Ph3 ADVOCATE-1&2 (multiple imputation (MCMC-MI) for missing values) and Ph2b (sensitivity analysis 3: NRI for rescue medication use and LOCF for other missing values); 250mg Q2W regimen). ADBRY (average of Ph3 ECZTRA1&2; 300 mg Q2W regimen; non-responder imputation for missing values). AMLITELIMAB Sanofi press release (average of COAST-1 and COAST-2, 250mg Q4W + 500mg loading dose; non-responder imputation for missing values). REZPEGALDESLEUKIN Nektar press release (non-responder imputation for missing values). 100 Zumilokibart (APEX Part A) Dosing Interval (weeks) Rezpegaldesleukin |
| APEX Part A 52-week results Carl Dambkowski, MD Chief Medical Officer |
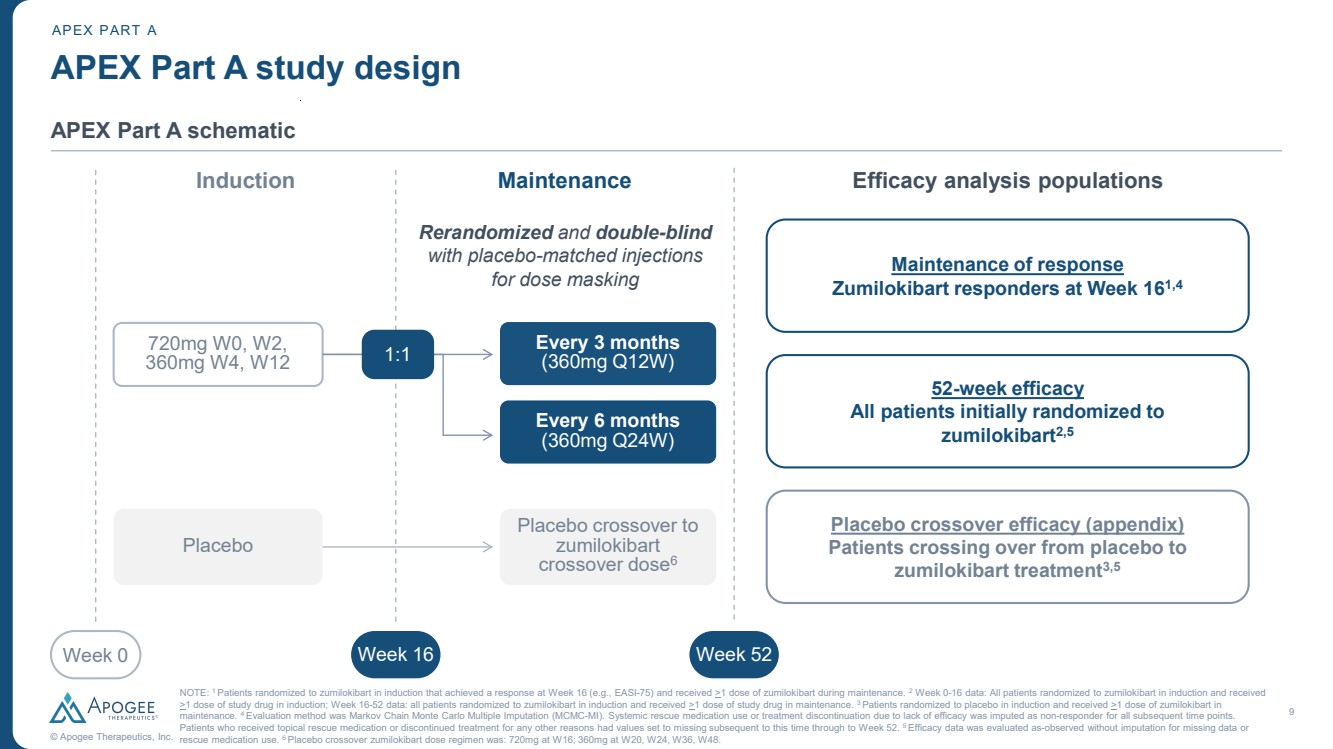
| 9 © Apogee Therapeutics, Inc. APEX Part A study design APEX Part A schematic APEX PART A Induction Maintenance Week 0 Week 16 Week 52 720mg W0, W2, 360mg W4, W12 Every 3 months (360mg Q12W) Every 6 months (360mg Q24W) 1:1 Placebo Placebo crossover to zumilokibart crossover dose6 Efficacy analysis populations Rerandomized and double-blind with placebo-matched injections for dose masking Maintenance of response Zumilokibart responders at Week 161,4 52-week efficacy All patients initially randomized to zumilokibart2,5 NOTE: 1 Patients randomized to zumilokibart in induction that achieved a response at Week 16 (e.g., EASI-75) and received >1 dose of zumilokibart during maintenance. 2 Week 0-16 data: All patients randomized to zumilokibart in induction and received >1 dose of study drug in induction; Week 16-52 data: all patients randomized to zumilokibart in induction and received >1 dose of study drug in maintenance. 3 Patients randomized to placebo in induction and received >1 dose of zumilokibart in maintenance. 4 Evaluation method was Markov Chain Monte Carlo Multiple Imputation (MCMC-MI). Systemic rescue medication use or treatment discontinuation due to lack of efficacy was imputed as non-responder for all subsequent time points. Patients who received topical rescue medication or discontinued treatment for any other reasons had values set to missing subsequent to this time through to Week 52. 5 Efficacy data was evaluated as-observed without imputation for missing data or rescue medication use. 6 Placebo crossover zumilokibart dose regimen was: 720mg at W16; 360mg at W20, W24, W36, W48. Placebo crossover efficacy (appendix) Patients crossing over from placebo to zumilokibart treatment3,5 |
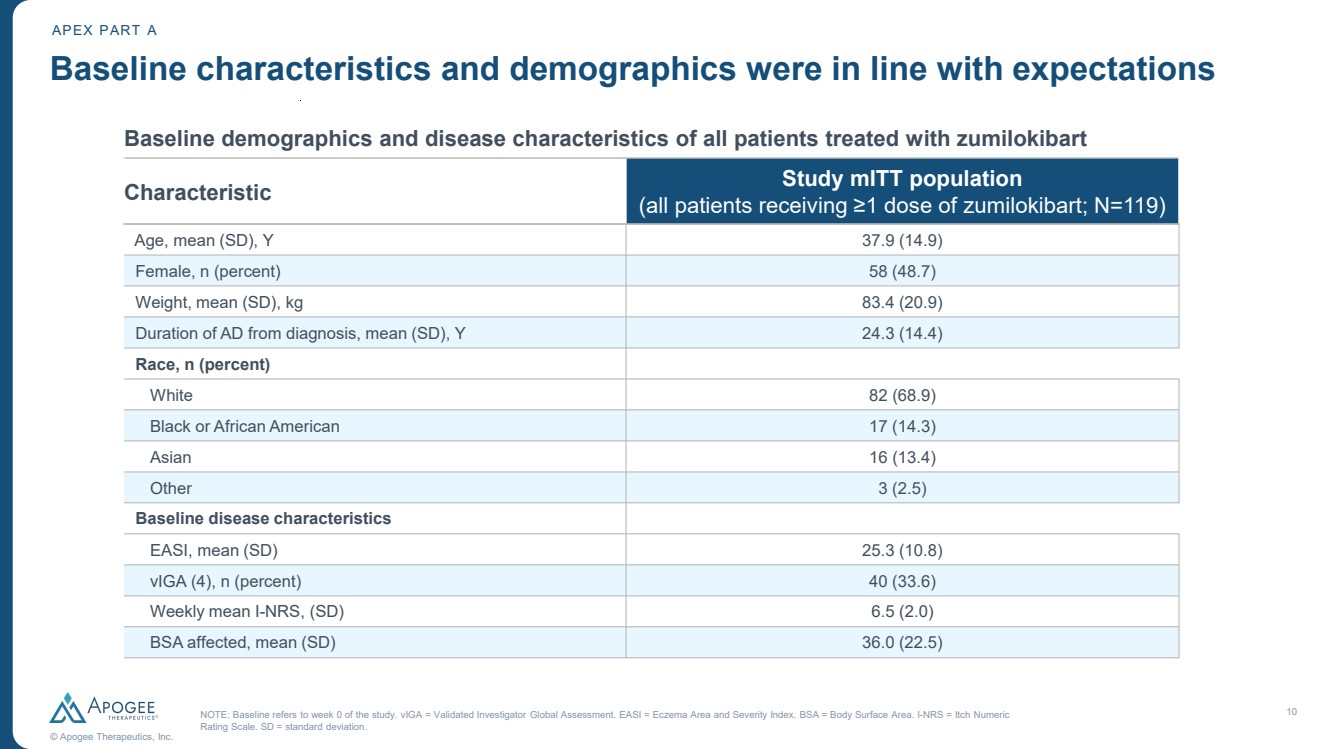
| 10 © Apogee Therapeutics, Inc. Baseline characteristics and demographics were in line with expectations APEX PART A Baseline demographics and disease characteristics of all patients treated with zumilokibart Characteristic Study mITT population (all patients receiving ≥1 dose of zumilokibart; N=119) NOTE: Baseline refers to week 0 of the study. vIGA = Validated Investigator Global Assessment. EASI = Eczema Area and Severity Index. BSA = Body Surface Area. I-NRS = Itch Numeric Rating Scale. SD = standard deviation. Age, mean (SD), Y 37.9 (14.9) Female, n (percent) 58 (48.7) Weight, mean (SD), kg 83.4 (20.9) Duration of AD from diagnosis, mean (SD), Y 24.3 (14.4) Race, n (percent) White 82 (68.9) Black or African American 17 (14.3) Asian 16 (13.4) Other 3 (2.5) Baseline disease characteristics EASI, mean (SD) 25.3 (10.8) vIGA (4), n (percent) 40 (33.6) Weekly mean I-NRS, (SD) 6.5 (2.0) BSA affected, mean (SD) 36.0 (22.5) |
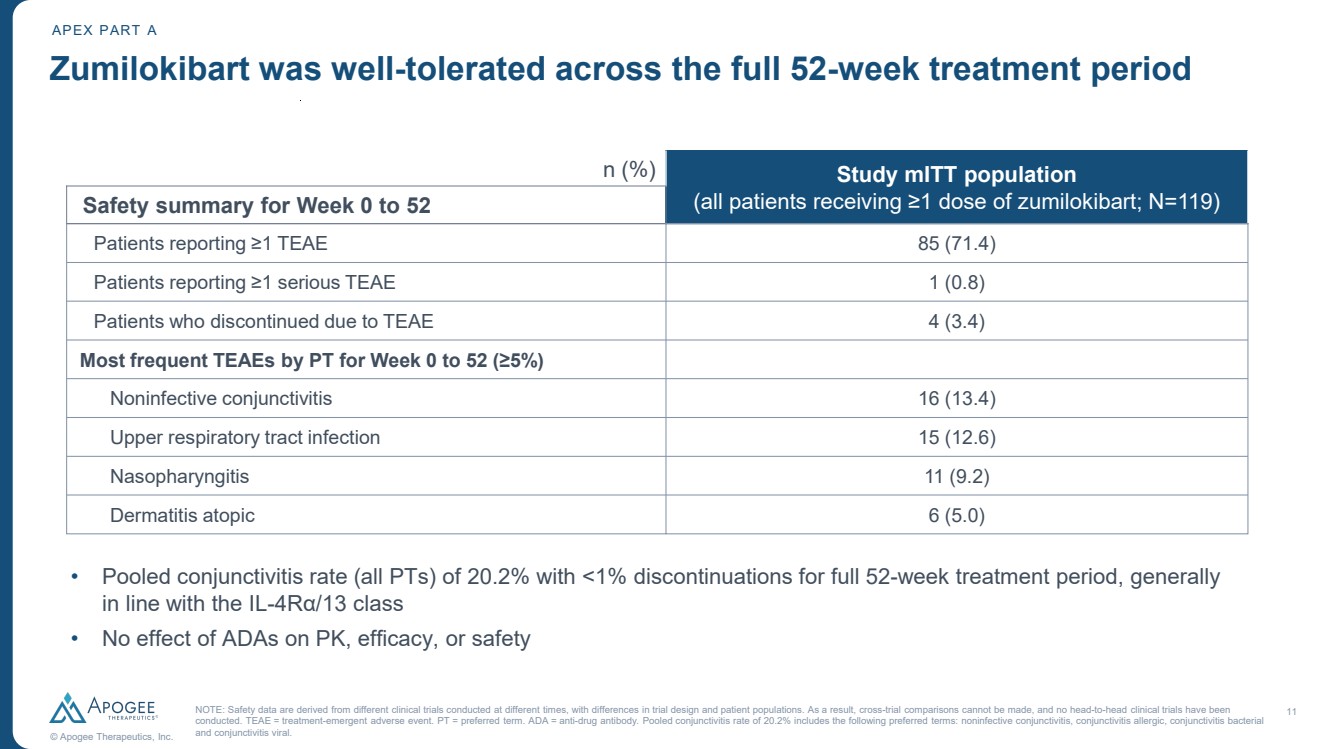
| 11 © Apogee Therapeutics, Inc. APEX PART A Zumilokibart was well-tolerated across the full 52-week treatment period NOTE: Safety data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. TEAE = treatment-emergent adverse event. PT = preferred term. ADA = anti-drug antibody. Pooled conjunctivitis rate of 20.2% includes the following preferred terms: noninfective conjunctivitis, conjunctivitis allergic, conjunctivitis bacterial and conjunctivitis viral. • Pooled conjunctivitis rate (all PTs) of 20.2% with <1% discontinuations for full 52-week treatment period, generally in line with the IL-4Rα/13 class • No effect of ADAs on PK, efficacy, or safety n (%) Study mITT population Safety summary for Week 0 to 52 (all patients receiving ≥1 dose of zumilokibart; N=119) Patients reporting ≥1 TEAE 85 (71.4) Patients reporting ≥1 serious TEAE 1 (0.8) Patients who discontinued due to TEAE 4 (3.4) Most frequent TEAEs by PT for Week 0 to 52 (≥5%) Noninfective conjunctivitis 16 (13.4) Upper respiratory tract infection 15 (12.6) Nasopharyngitis 11 (9.2) Dermatitis atopic 6 (5.0) |
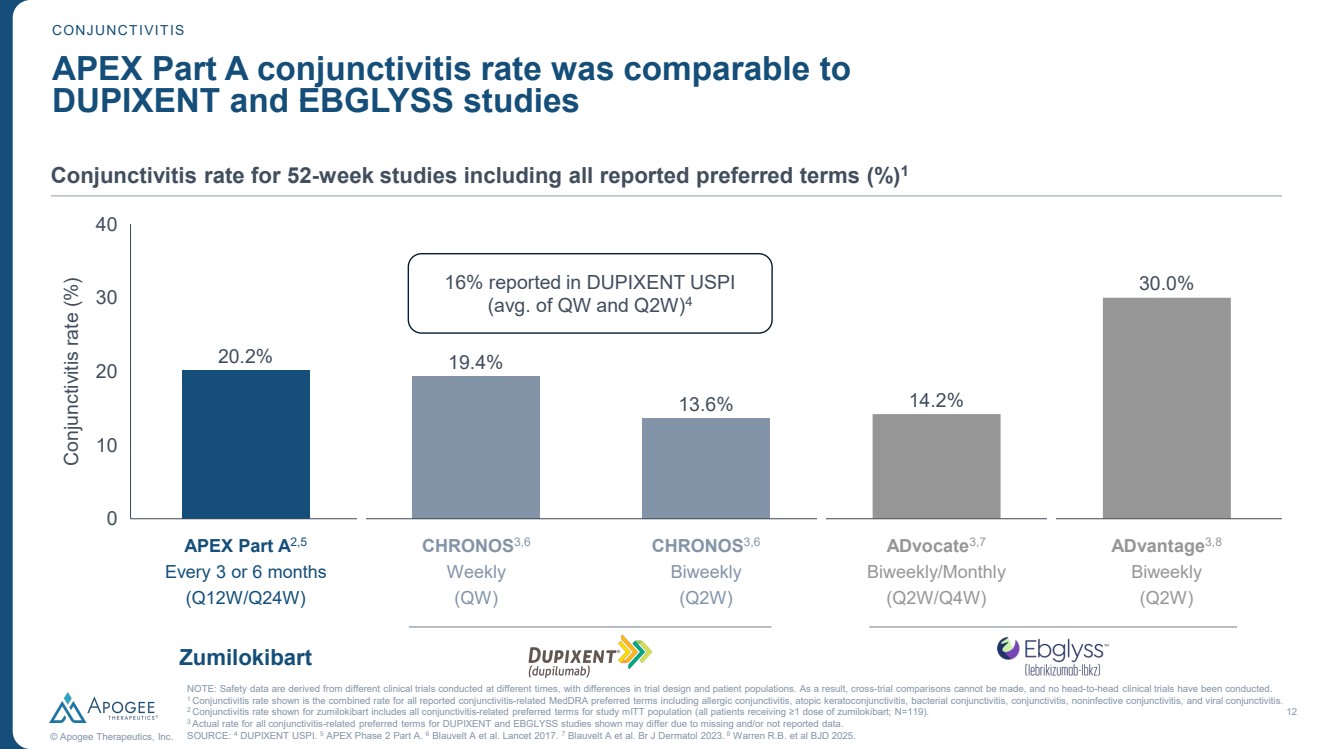
| 12 © Apogee Therapeutics, Inc. NOTE: Safety data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. 1 Conjunctivitis rate shown is the combined rate for all reported conjunctivitis-related MedDRA preferred terms including allergic conjunctivitis, atopic keratoconjunctivitis, bacterial conjunctivitis, conjunctivitis, noninfective conjunctivitis, and viral conjunctivitis. 2 Conjunctivitis rate shown for zumilokibart includes all conjunctivitis-related preferred terms for study mITT population (all patients receiving ≥1 dose of zumilokibart; N=119). 3 Actual rate for all conjunctivitis-related preferred terms for DUPIXENT and EBGLYSS studies shown may differ due to missing and/or not reported data. SOURCE: 4 DUPIXENT USPI. 5 APEX Phase 2 Part A. 6 Blauvelt A et al. Lancet 2017. 7 Blauvelt A et al. Br J Dermatol 2023. 8 Warren R.B. et al BJD 2025. CONJUNCTIVITIS APEX Part A conjunctivitis rate was comparable to DUPIXENT and EBGLYSS studies 0 10 20 30 40 Conjunctivitis rate (%) APEX Zumi2,5 CHRONOS QW3,6 CHRONOS Q2W3,6 ADvocate Lebri3,7,8 ADvantage Lebri3,9 20.2% 19.4% 13.6% 14.2% 16% reported in DUPIXENT USPI 30.0% (avg. of QW and Q2W)4 Conjunctivitis rate for 52-week studies including all reported preferred terms (%)1 APEX Part A2,5 Every 3 or 6 months (Q12W/Q24W) Zumilokibart CHRONOS3,6 Weekly (QW) CHRONOS3,6 Biweekly (Q2W) ADvocate3,7 Biweekly/Monthly (Q2W/Q4W) ADvantage3,8 Biweekly (Q2W) |
| Zumilokibart achieved durable maintenance of Week 16 responses |
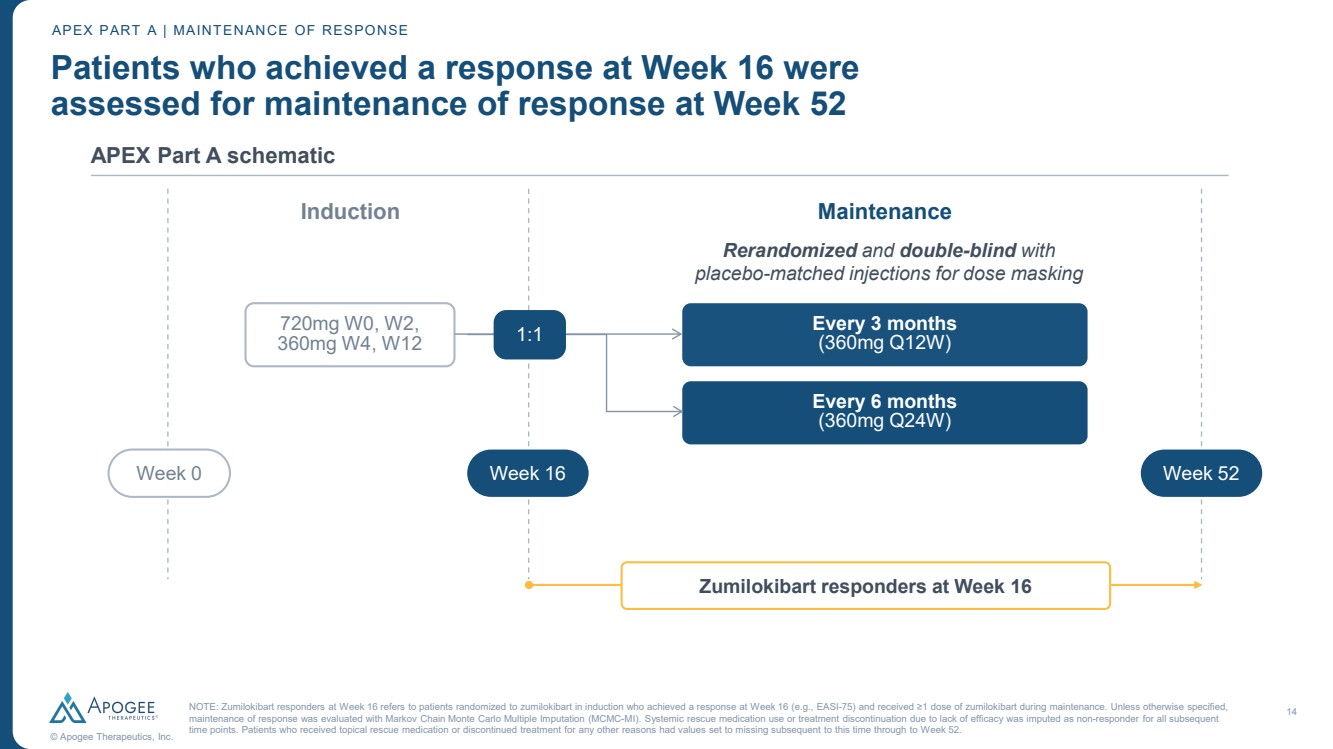
| 14 © Apogee Therapeutics, Inc. Patients who achieved a response at Week 16 were assessed for maintenance of response at Week 52 APEX Part A schematic APEX PART A | MAINTENANCE OF RESPONSE 720mg W0, W2, 360mg W4, W12 Induction Every 3 months (360mg Q12W) Every 6 months (360mg Q24W) Maintenance Week 0 Week 16 Week 52 Zumilokibart responders at Week 16 1:1 Rerandomized and double-blind with placebo-matched injections for dose masking NOTE: Zumilokibart responders at Week 16 refers to patients randomized to zumilokibart in induction who achieved a response at Week 16 (e.g., EASI-75) and received ≥1 dose of zumilokibart during maintenance. Unless otherwise specified, maintenance of response was evaluated with Markov Chain Monte Carlo Multiple Imputation (MCMC-MI). Systemic rescue medication use or treatment discontinuation due to lack of efficacy was imputed as non-responder for all subsequent time points. Patients who received topical rescue medication or discontinued treatment for any other reasons had values set to missing subsequent to this time through to Week 52. |
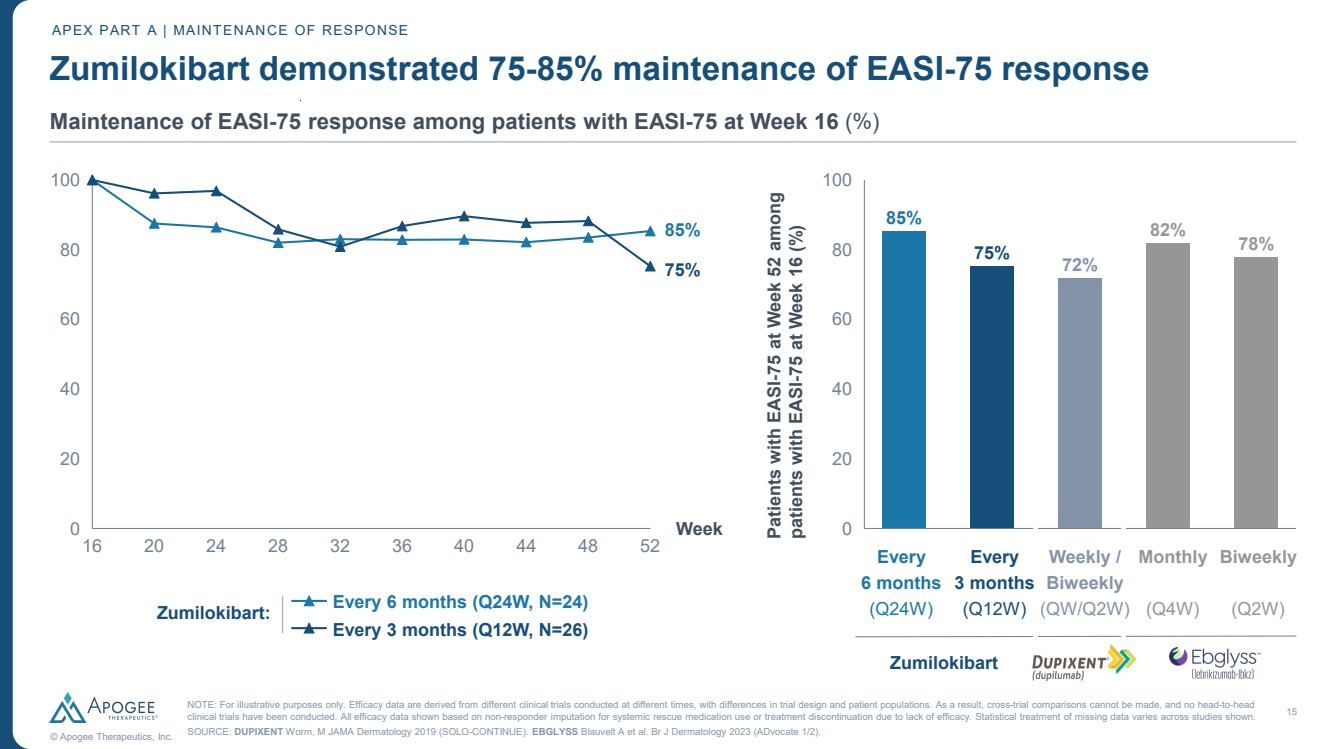
| 15 © Apogee Therapeutics, Inc. Zumilokibart demonstrated 75-85% maintenance of EASI-75 response APEX PART A | MAINTENANCE OF RESPONSE Maintenance of EASI-75 response among patients with EASI-75 at Week 16 (%) 16 20 24 28 32 36 40 44 48 52 0 20 40 60 80 100 Week Every 6 months (Q24W, N=24) Every 3 months (Q12W, N=26) Zumilokibart: 0 20 40 60 80 100 Patients with EASI-75 at Week 52 among patients with EASI-75 at Week 16 (%) 85% 75% 72% 82% 78% Every 6 months (Q24W) Zumilokibart Monthly (Q4W) Biweekly (Q2W) Weekly / Biweekly (QW/Q2W) NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. All efficacy data shown based on non-responder imputation for systemic rescue medication use or treatment discontinuation due to lack of efficacy. Statistical treatment of missing data varies across studies shown. SOURCE: DUPIXENT Worm, M JAMA Dermatology 2019 (SOLO-CONTINUE). EBGLYSS Blauvelt A et al. Br J Dermatology 2023 (ADvocate 1/2). 75% 85% Every 3 months (Q12W) |
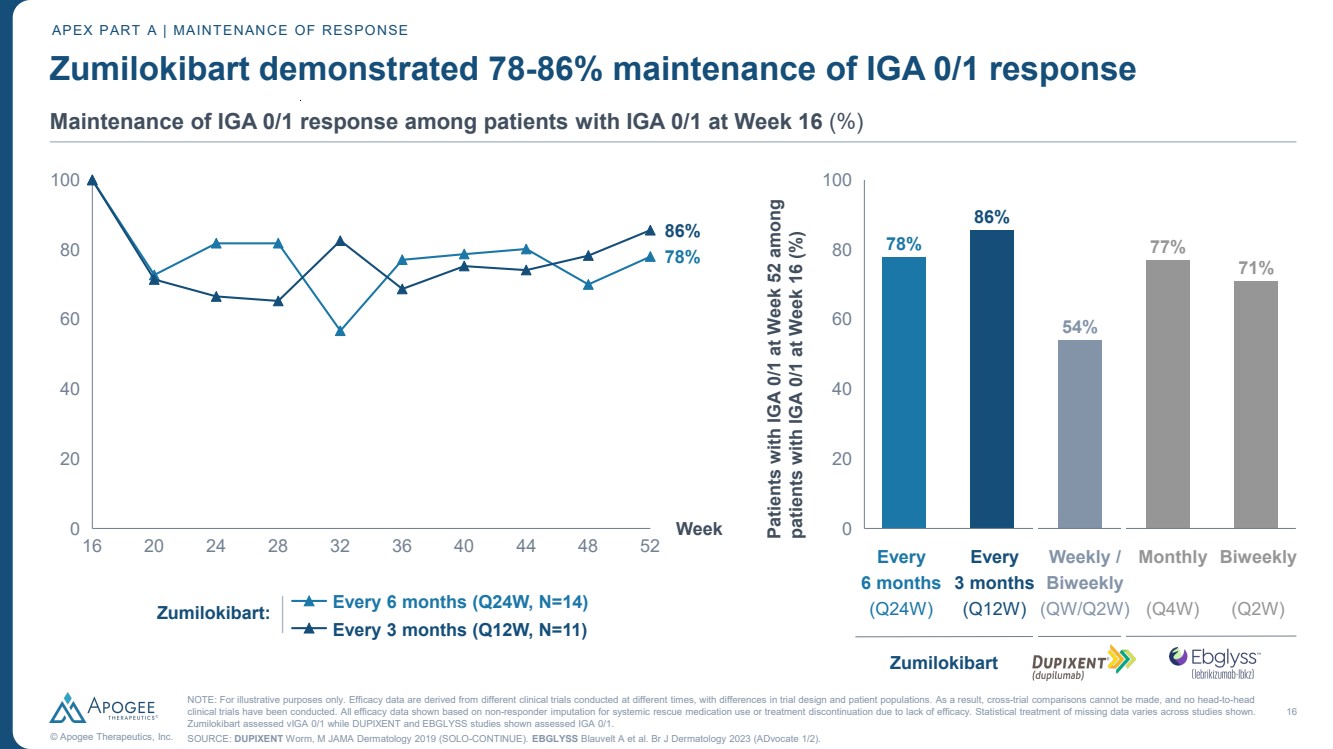
| 16 © Apogee Therapeutics, Inc. Zumilokibart demonstrated 78-86% maintenance of IGA 0/1 response APEX PART A | MAINTENANCE OF RESPONSE NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. All efficacy data shown based on non-responder imputation for systemic rescue medication use or treatment discontinuation due to lack of efficacy. Statistical treatment of missing data varies across studies shown. Zumilokibart assessed vIGA 0/1 while DUPIXENT and EBGLYSS studies shown assessed IGA 0/1. SOURCE: DUPIXENT Worm, M JAMA Dermatology 2019 (SOLO-CONTINUE). EBGLYSS Blauvelt A et al. Br J Dermatology 2023 (ADvocate 1/2). Maintenance of IGA 0/1 response among patients with IGA 0/1 at Week 16 (%) 16 20 24 28 32 36 40 44 48 52 0 20 40 60 80 100 Week Every 6 months (Q24W, N=14) Every 3 months (Q12W, N=11) Zumilokibart: 0 20 40 60 80 100 Patients with IGA 0/1 at Week 52 among patients with IGA 0/1 at Week 16 (%) 78% 86% 54% 77% 71% Every 6 months (Q24W) Zumilokibart Monthly (Q4W) Biweekly (Q2W) Weekly / Biweekly (QW/Q2W) Every 3 months (Q12W) 86% 78% |
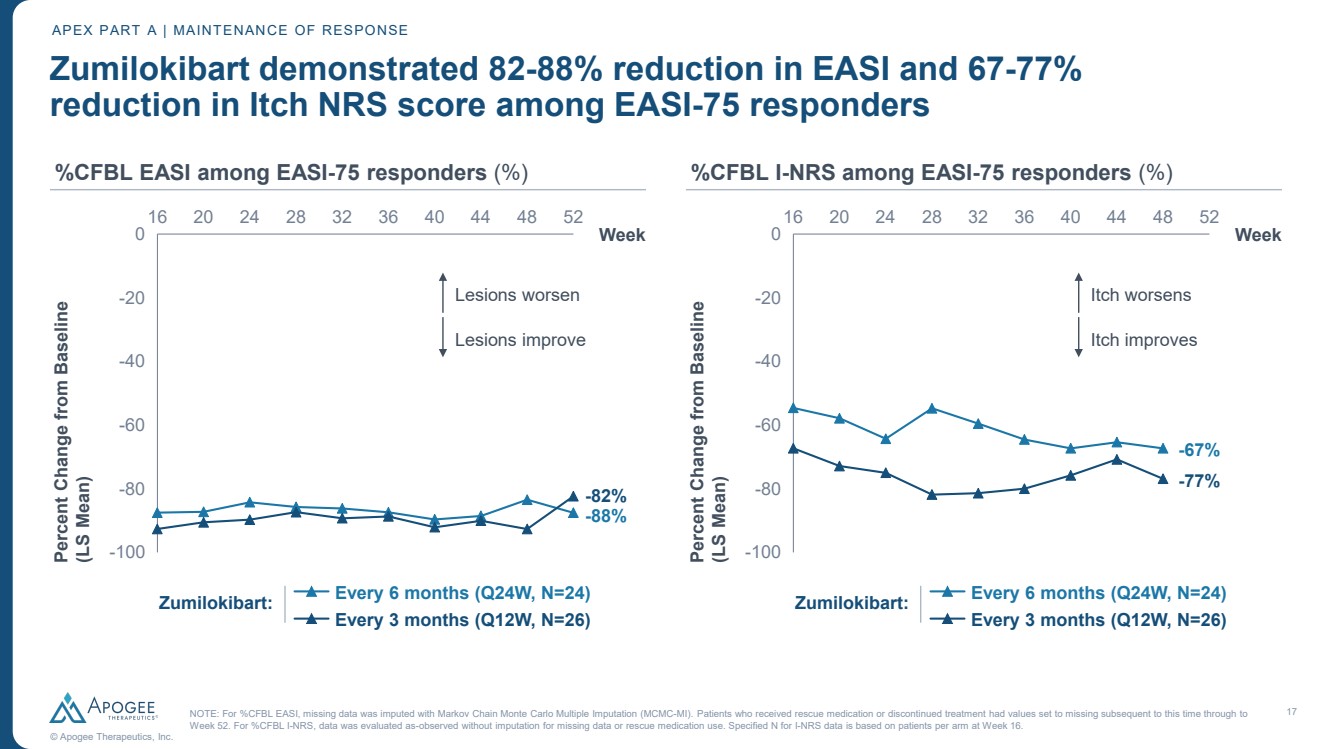
| 17 © Apogee Therapeutics, Inc. Zumilokibart demonstrated 82-88% reduction in EASI and 67-77% reduction in Itch NRS score among EASI-75 responders APEX PART A | MAINTENANCE OF RESPONSE %CFBL EASI among EASI-75 responders (%) NOTE: For %CFBL EASI, missing data was imputed with Markov Chain Monte Carlo Multiple Imputation (MCMC-MI). Patients who received rescue medication or discontinued treatment had values set to missing subsequent to this time through to Week 52. For %CFBL I-NRS, data was evaluated as-observed without imputation for missing data or rescue medication use. Specified N for I-NRS data is based on patients per arm at Week 16. 16 20 24 28 32 36 40 44 48 52 -100 -80 -60 -40 -20 0 Week Percent Change from Baseline (LS Mean) Every 6 months (Q24W, N=24) Every 3 months (Q12W, N=26) %CFBL I-NRS among EASI-75 responders (%) -77% -67% Itch worsens Itch improves Zumilokibart: 16 20 24 28 32 36 40 44 48 52 -100 -80 -60 -40 -20 0 Week Percent Change from Baseline (LS Mean) Every 6 months (Q24W, N=24) Every 3 months (Q12W, N=26) -82% -88% Lesions worsen Lesions improve Zumilokibart: |
| Zumilokibart led to deepening responses over 52 weeks |
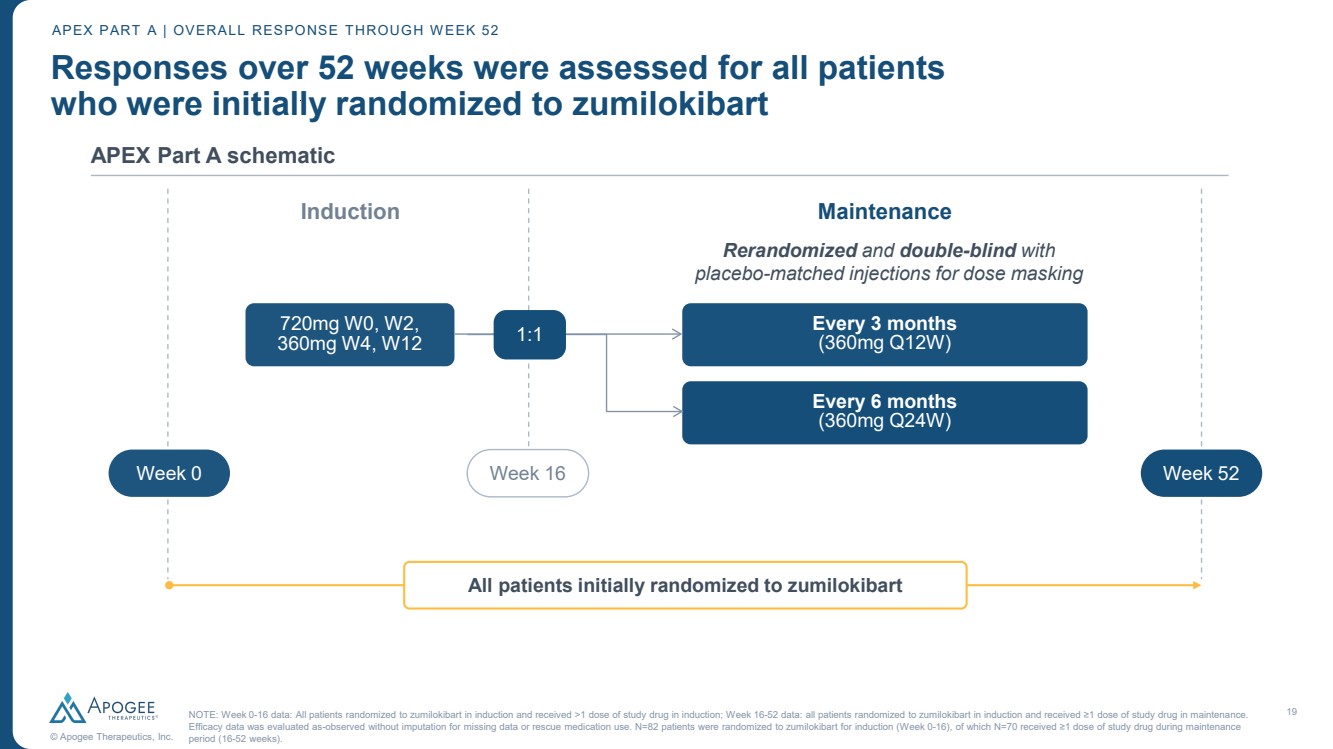
| 19 © Apogee Therapeutics, Inc. Responses over 52 weeks were assessed for all patients who were initially randomized to zumilokibart APEX Part A schematic APEX PART A | OVERALL RESPONSE THROUGH WEEK 52 720mg W0, W2, 360mg W4, W12 Induction Every 3 months (360mg Q12W) Every 6 months (360mg Q24W) Maintenance Week 0 Week 16 Week 52 1:1 Rerandomized and double-blind with placebo-matched injections for dose masking All patients initially randomized to zumilokibart NOTE: Week 0-16 data: All patients randomized to zumilokibart in induction and received >1 dose of study drug in induction; Week 16-52 data: all patients randomized to zumilokibart in induction and received ≥1 dose of study drug in maintenance. Efficacy data was evaluated as-observed without imputation for missing data or rescue medication use. N=82 patients were randomized to zumilokibart for induction (Week 0-16), of which N=70 received ≥1 dose of study drug during maintenance period (16-52 weeks). |
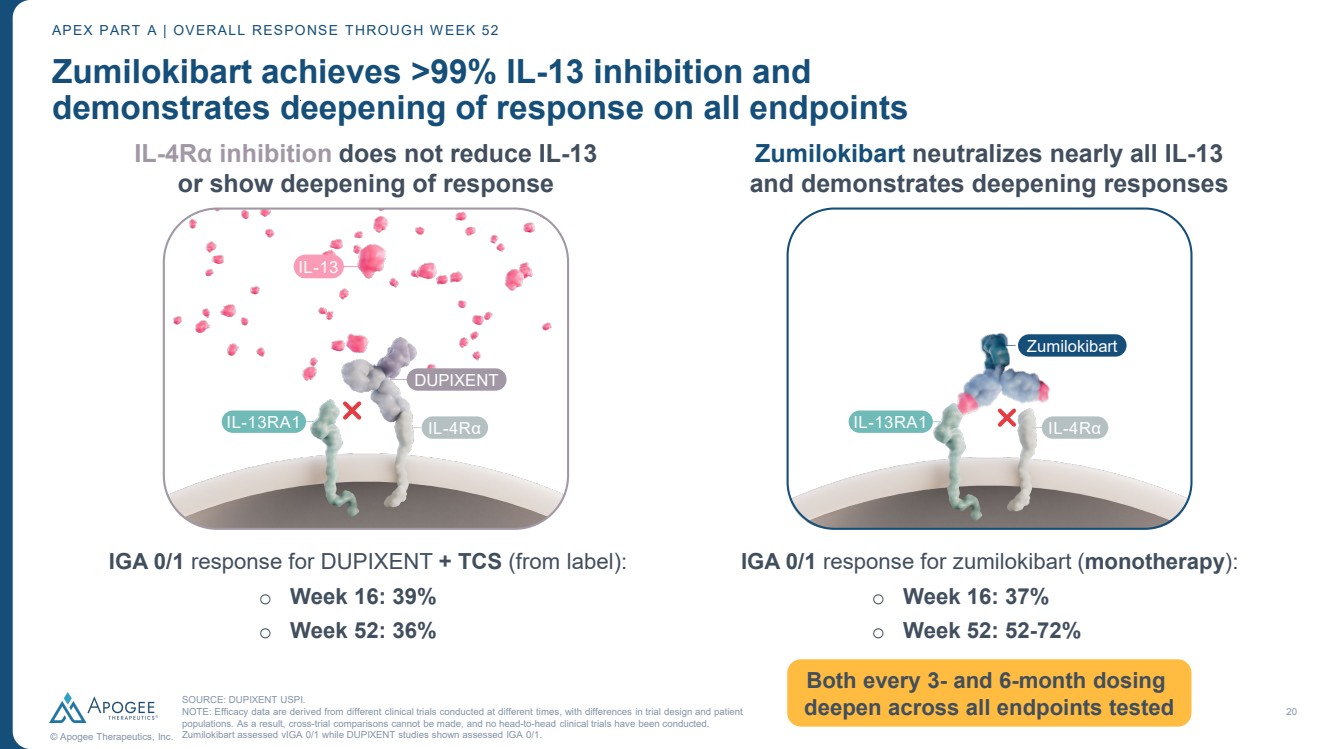
| 20 © Apogee Therapeutics, Inc. Zumilokibart achieves >99% IL-13 inhibition and demonstrates deepening of response on all endpoints SOURCE: DUPIXENT USPI. NOTE: Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Zumilokibart assessed vIGA 0/1 while DUPIXENT studies shown assessed IGA 0/1. APEX PART A | OVERALL RESPONSE THROUGH WEEK 52 IGA 0/1 response for DUPIXENT + TCS (from label): o Week 16: 39% o Week 52: 36% IGA 0/1 response for zumilokibart (monotherapy): o Week 16: 37% o Week 52: 52-72% IL-13 IL-13RA1 IL-4Rα DUPIXENT Zumilokibart IL-13RA1 IL-4Rα Zumilokibart neutralizes nearly all IL-13 and demonstrates deepening responses IL-4Rα inhibition does not reduce IL-13 or show deepening of response Both every 3- and 6-month dosing deepen across all endpoints tested |
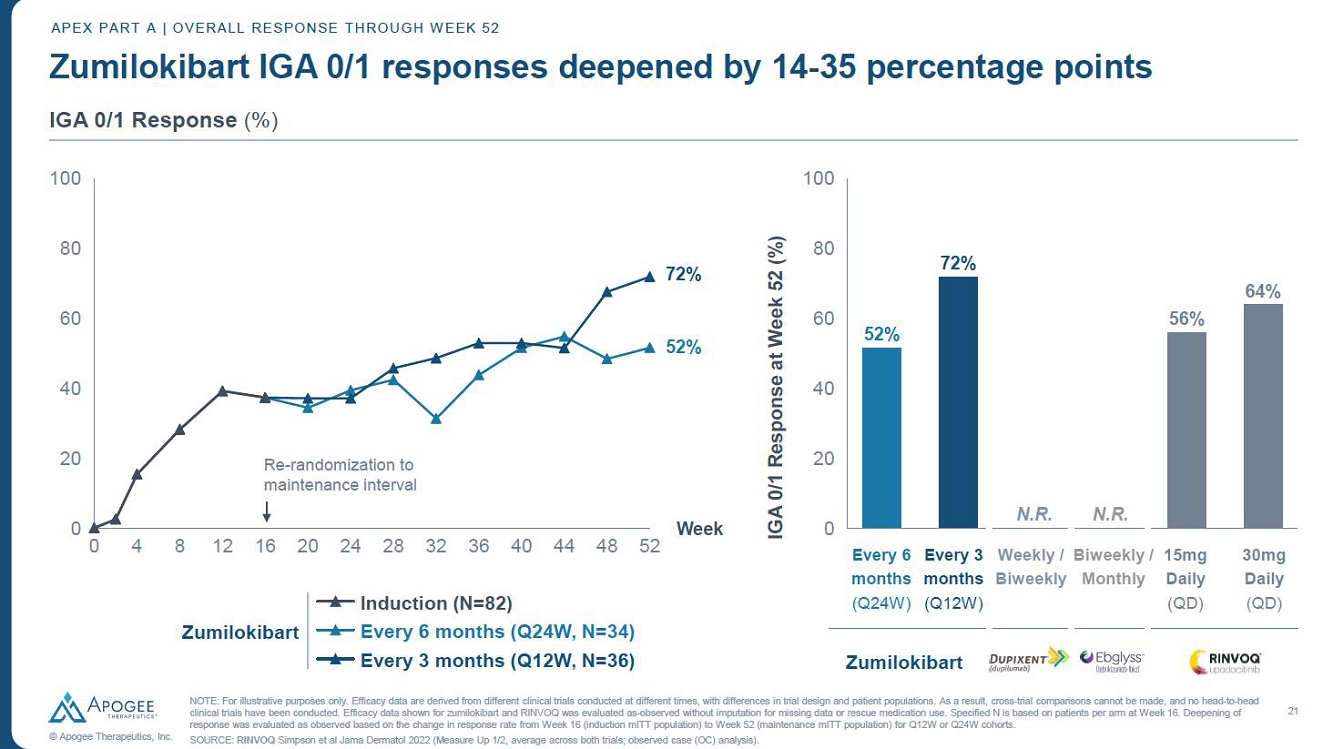
| 21 © Apogee Therapeutics, Inc. Zumilokibart IGA 0/1 responses deepened by 14-35 percentage points APEX PART A | OVERALL RESPONSE THROUGH WEEK 52 IGA 0/1 with a Reduction of ≥2 Points from Baseline (%) 0 4 8 12 16 20 24 28 32 36 40 44 48 52 0 20 40 60 80 100 Week Induction (N=82) Every 6 months (Q24W, N=34) Every 3 months (Q12W, N=36) 0 20 40 60 80 100 IGA 0/1 Response at Week 52 (%) 52% 72% N.R. N.R. 56% 64% 72% 52% Zumilokibart Re-randomization to maintenance interval Every 6 months (Q24W) Every 3 months (Q12W) Zumilokibart 30mg Daily (QD) 15mg Daily (QD) Biweekly / Monthly Weekly / Biweekly NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Efficacy data shown for zumilokibart and RINVOQ was evaluated as-observed without imputation for missing data or rescue medication use. Specified N is based on patients per arm at Week 16. Deepening of response was evaluated as observed based on the change in response rate from Week 16 (induction mITT population) to Week 52 (maintenance mITT population) for Q12W or Q24W cohorts. SOURCE: RINVOQ Simpson et al Jama Dermatol 2022 (Measure Up 1/2, average across both trials; observed case (OC) analysis). |
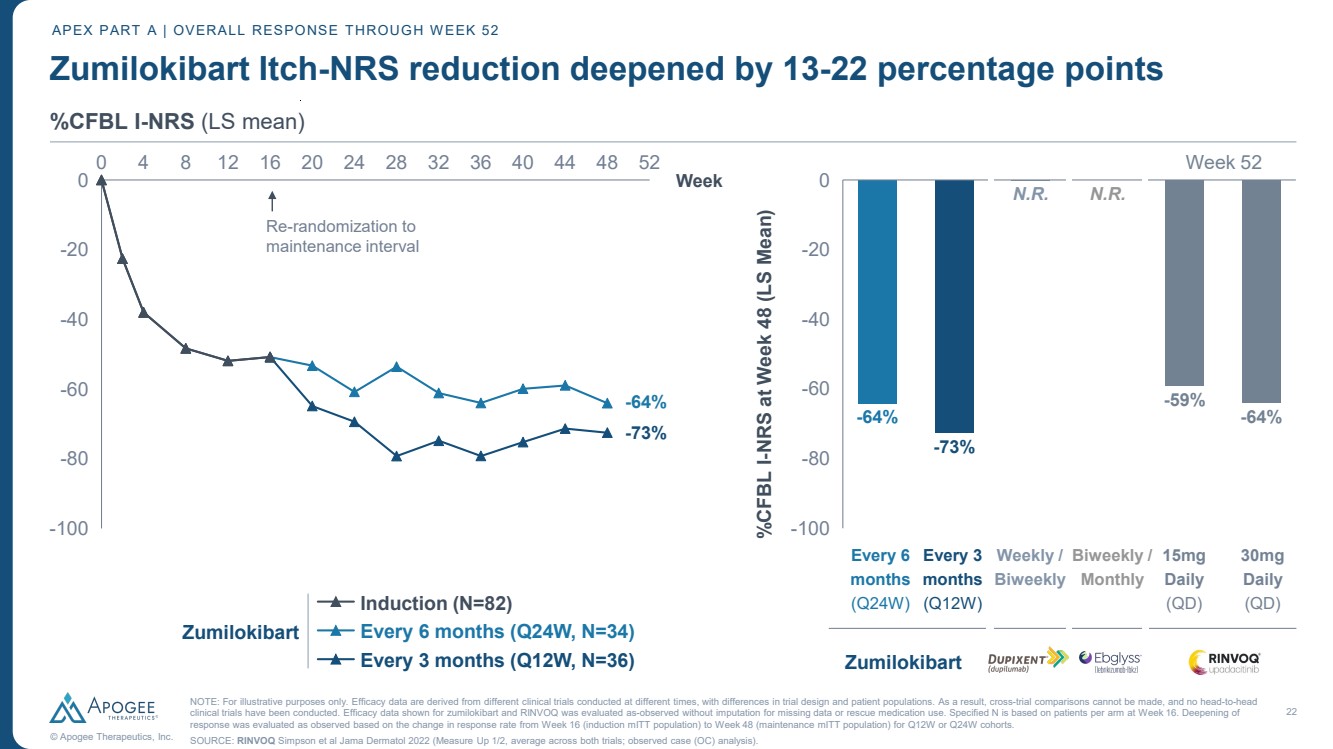
| 22 © Apogee Therapeutics, Inc. Zumilokibart Itch-NRS reduction deepened by 13-22 percentage points APEX PART A | OVERALL RESPONSE THROUGH WEEK 52 %CFBL I-NRS (LS mean) 0 4 8 12 16 20 24 28 32 36 40 44 48 52 0 -80 -60 -40 -20 -100 Week Induction (N=82) Every 6 months (Q24W, N=34) Every 3 months (Q12W, N=36) Zumilokibart -100 -80 -60 -40 -20 0 %CFBL I-NRS at Week 48 (LS Mean) -64% -73% N.R. N.R. -59% -64% Zumilokibart Week 52 Every 6 months (Q24W) Every 3 months (Q12W) 30mg Daily (QD) 15mg Daily (QD) Biweekly / Monthly Weekly / Biweekly -73% -64% Re-randomization to maintenance interval NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Efficacy data shown for zumilokibart and RINVOQ was evaluated as-observed without imputation for missing data or rescue medication use. Specified N is based on patients per arm at Week 16. Deepening of response was evaluated as observed based on the change in response rate from Week 16 (induction mITT population) to Week 48 (maintenance mITT population) for Q12W or Q24W cohorts. SOURCE: RINVOQ Simpson et al Jama Dermatol 2022 (Measure Up 1/2, average across both trials; observed case (OC) analysis). |
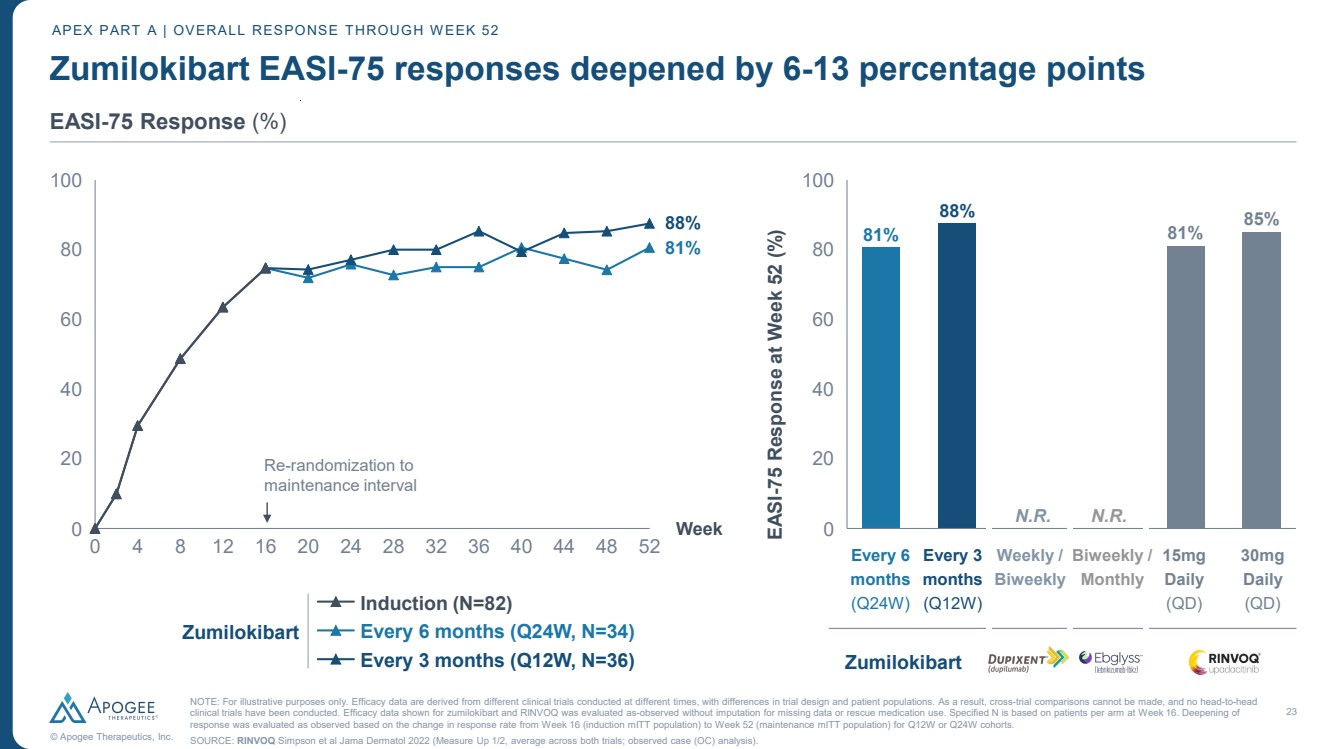
| 23 © Apogee Therapeutics, Inc. Zumilokibart EASI-75 responses deepened by 6-13 percentage points APEX PART A | OVERALL RESPONSE THROUGH WEEK 52 EASI-75 Response (%) 0 4 8 12 16 20 24 28 32 36 40 44 48 52 0 20 40 60 80 100 Week Induction (N=82) Every 6 months (Q24W, N=34) Every 3 months (Q12W, N=36) 0 20 40 60 80 100 EASI-75 Response at Week 52 (%) 81% 88% N.R. N.R. 81% 88% 85% 81% Zumilokibart Re-randomization to maintenance interval Every 6 months (Q24W) Every 3 months (Q12W) Zumilokibart 30mg Daily (QD) 15mg Daily (QD) Biweekly / Monthly Weekly / Biweekly NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Efficacy data shown for zumilokibart and RINVOQ was evaluated as-observed without imputation for missing data or rescue medication use. Specified N is based on patients per arm at Week 16. Deepening of response was evaluated as observed based on the change in response rate from Week 16 (induction mITT population) to Week 52 (maintenance mITT population) for Q12W or Q24W cohorts. SOURCE: RINVOQ Simpson et al Jama Dermatol 2022 (Measure Up 1/2, average across both trials; observed case (OC) analysis). |
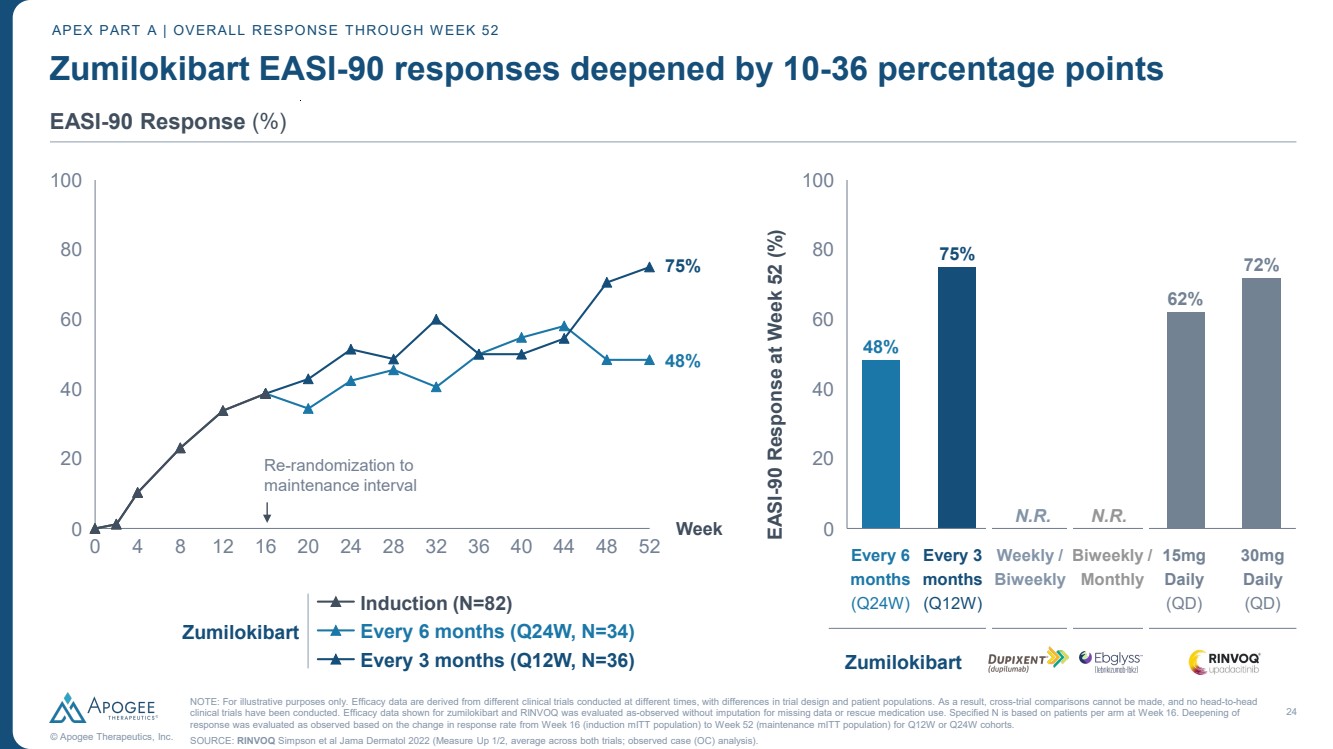
| 24 © Apogee Therapeutics, Inc. Zumilokibart EASI-90 responses deepened by 10-36 percentage points APEX PART A | OVERALL RESPONSE THROUGH WEEK 52 EASI-90 Response (%) 0 4 8 12 16 20 24 28 32 36 40 44 48 52 0 20 40 60 80 100 Week Induction (N=82) Every 6 months (Q24W, N=34) Every 3 months (Q12W, N=36) 0 20 40 60 80 100 EASI-90 Response at Week 52 (%) 48% 75% N.R. N.R. 62% 75% 72% 48% Zumilokibart Re-randomization to maintenance interval Every 6 months (Q24W) Every 3 months (Q12W) Zumilokibart 30mg Daily (QD) 15mg Daily (QD) Biweekly / Monthly Weekly / Biweekly NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Efficacy data shown for zumilokibart and RINVOQ was evaluated as-observed without imputation for missing data or rescue medication use. Specified N is based on patients per arm at Week 16. Deepening of response was evaluated as observed based on the change in response rate from Week 16 (induction mITT population) to Week 52 (maintenance mITT population) for Q12W or Q24W cohorts. SOURCE: RINVOQ Simpson et al Jama Dermatol 2022 (Measure Up 1/2, average across both trials; observed case (OC) analysis). |
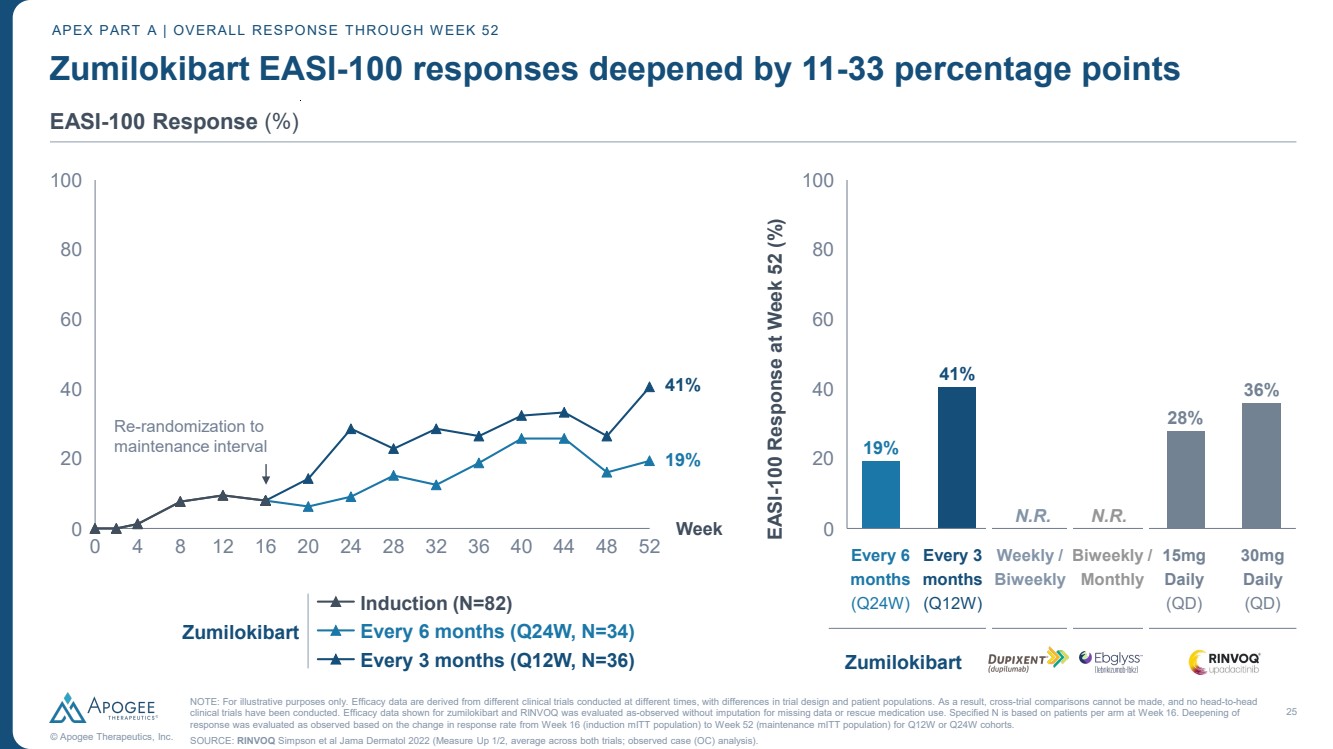
| 25 © Apogee Therapeutics, Inc. Zumilokibart EASI-100 responses deepened by 11-33 percentage points APEX PART A | OVERALL RESPONSE THROUGH WEEK 52 EASI-100 Response (%) 0 4 8 12 16 20 24 28 32 36 40 44 48 52 0 20 40 60 80 100 Week Induction (N=82) Every 6 months (Q24W, N=34) Every 3 months (Q12W, N=36) 0 20 40 60 80 100 EASI-100 Response at Week 52 (%) 19% 41% N.R. N.R. 28% 41% 36% 19% Zumilokibart Re-randomization to maintenance interval Every 6 months (Q24W) Every 3 months (Q12W) Zumilokibart 30mg Daily (QD) 15mg Daily (QD) Biweekly / Monthly Weekly / Biweekly NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Efficacy data shown for zumilokibart and RINVOQ was evaluated as-observed without imputation for missing data or rescue medication use. Specified N is based on patients per arm at Week 16. Deepening of response was evaluated as observed based on the change in response rate from Week 16 (induction mITT population) to Week 52 (maintenance mITT population) for Q12W or Q24W cohorts. SOURCE: RINVOQ Simpson et al Jama Dermatol 2022 (Measure Up 1/2, average across both trials; observed case (OC) analysis). |
| Unmet Need in Atopic Dermatitis Ruth Ann Vleugels, MD, MPH, MBA Heidi and Scott C. Schuster Distinguished Chair in Dermatology Director, Atopic Dermatitis Program Brigham & Women’s Hospital Department of Dermatology Professor of Dermatology, Harvard Medical School |
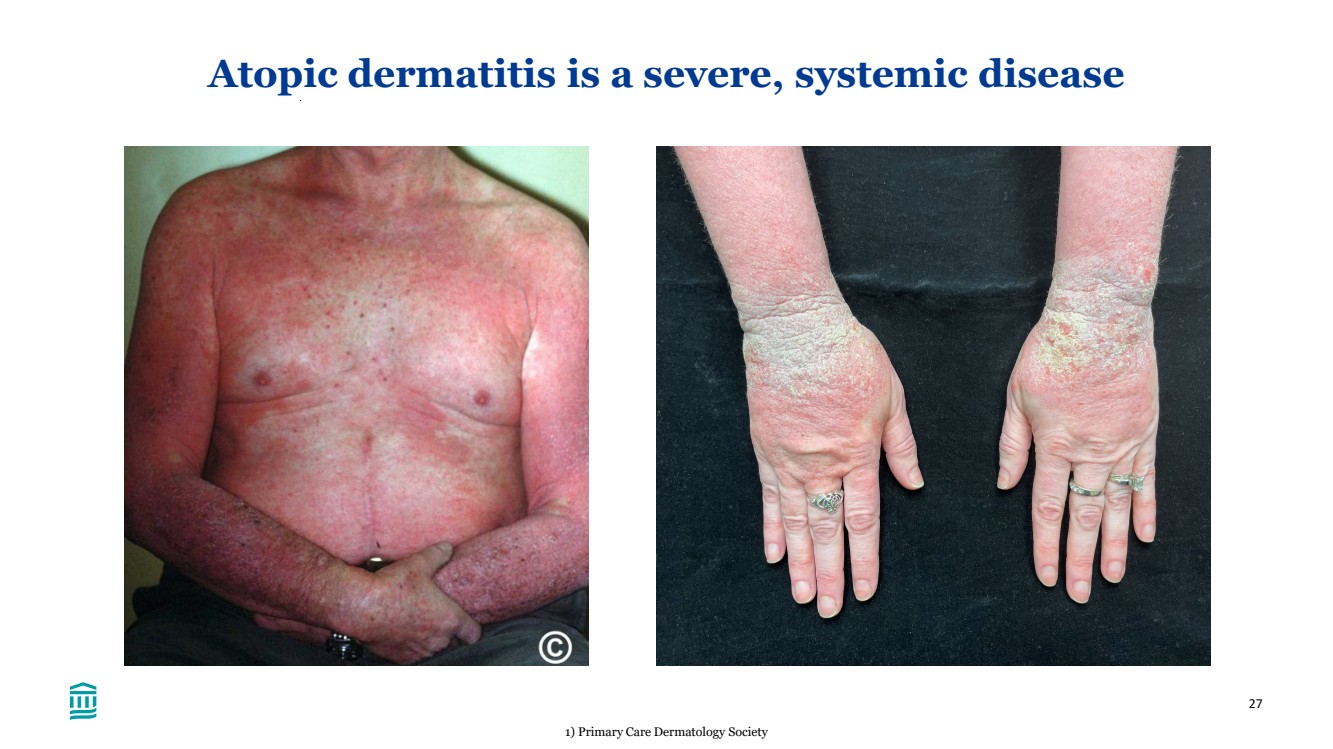
| 27 Atopic dermatitis is a severe, systemic disease 1) Primary Care Dermatology Society |
| 28 Atopic dermatitis has a profound impact on patient quality of life Children Adults Healthcare system 1) Bridgman et al., 2018. Ann Allergy Asthma Immunol. 2) Irish Skin Foundation. Loss of sleep Absence from school Growth restriction Depression Work sick leave Reduced physical activity Hospitalizations Annual cost burden Specialist consultations |
| 29 Approval of new therapies has improved AD care Pre-2017 2017 2020 2023 2026 Dupilumab (IL-4Ra) Year of AD approval Tralokinumab (IL-13) Lebrikizumab (IL-13) Nemolizumab (IL-31) Topicals Biologics Oral JAK inhibitors Hydrocortisone & other TCS Roflumilast (PDE4) Ruxolitinib (JAK) Upadacitinib Abrocitinib 1) Symons et al., Journal of Drugs in Dermatology 2025 |
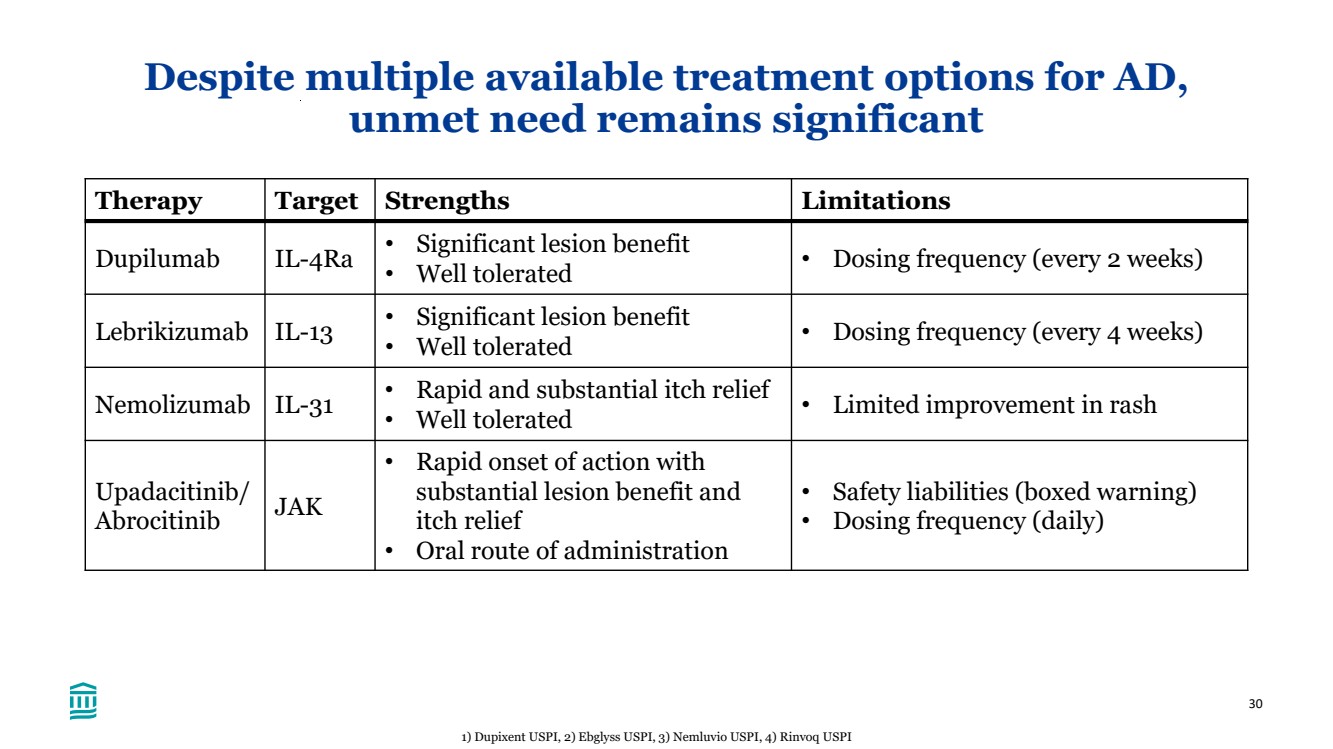
| 30 Despite multiple available treatment options for AD, unmet need remains significant Therapy Target Strengths Limitations Dupilumab IL-4Ra • Significant lesion benefit • Well tolerated • Dosing frequency (every 2 weeks) Lebrikizumab IL-13 • Significant lesion benefit • Well tolerated • Dosing frequency (every 4 weeks) Nemolizumab IL-31 • Rapid and substantial itch relief • Well tolerated • Limited improvement in rash Upadacitinib/ Abrocitinib JAK • Rapid onset of action with substantial lesion benefit and itch relief • Oral route of administration • Safety liabilities (boxed warning) • Dosing frequency (daily) 1) Dupixent USPI, 2) Ebglyss USPI, 3) Nemluvio USPI, 4) Rinvoq USPI |
| 31 Zumilokibart could address several unmet needs in AD1 • Although newer therapies have greatly improved the lives of patients with AD, substantial unmet need still exists for therapies that are both safe and effective as well as have the ability to reduce injection burden • Zumilokibart demonstrated strong clinical response at week 16 and responses were maintained with every 3-month and every 6-month dosing • Deep responses on skin and itch were achieved in a substantial proportion of patients by week 52, including on EASI-100, which represents completely clear skin • Zumilokibart was well-tolerated across both dosing regimens with a safety profile generally in line with the IL-4/13 class • Zumilokibart has the potential to improve the lives of patients with AD by providing long-term disease control with only 2-4 maintenance injections per year 1) Safety and efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. |
| Zumilokibart Development Program Kristine Nograles, MD SVP, Clinical Development & Medical Affairs |
| 33 © Apogee Therapeutics, Inc. Zumilokibart Part B trial design Mod-Sev AD: EASI ≥16, vIGA ≥3, BSA ≥10%, up to 20% biologic experienced Induction Maintenance Enrollment complete W16 Primary Endpoint: EASI-75 N = 347 1:1:1:1 NOTE: Phase 3 and launch plans subject to clinical and regulatory outcomes. EASI = Eczema Area and Severity Index. vIGA = validated Investigator Global Assessment for Atopic Dermatitis. BSA = body surface area. LTE = long term extension. Modeled exposure vs. EBGLYSS Placebo Every 3 months (Q12W) High dose ~90-100% greater Every 6 months (Q24W) Mid dose (Part A dose) ~30-40% greater Low dose ~40-50% less LTE or 52-week follow-up period W52 Endpoint Week 16 readout expected in Q2 2026 to support Phase 3 dose selection Phase 3 studies expected to initiate in 2H 2026 Zumilokibart launch planned in 2029 ZUMILOKIBART Part B has completed enrollment; planned Q2 2026 readout potentially enables Phase 3 initiation in 2026 |
| 34 © Apogee Therapeutics, Inc. Beyond zumilokibart in AD, ASPIRE and ELEVATE dose-ranging trials could enable Phase 3 for multiple blockbuster indication expansions ZUMILOKIBART • Allergic Rhinitis (perennial) • COPD • CRSwNP Respiratory ASPIRE Asthma Phase 2 Plans TBA this year • BP • CSU • Cold Inducible Urticaria • Prurigo Nodularis Dermatology APEX AD Part B 16-week Q2 2026 expected readout • EGID (non-EoE) Gastroenterology ELEVATE EoE Phase 2 Plans TBA this year Dose Optimized dose in each TA could enable straight to Phase 3 approach for add’l expansions -ranging trial Expansion potential NOTE: 1Cendakimab positive Phase 3 in EoE (Dellon SE et al NEJM Evid 2025). Phase 3 and launch plans subject to clinical and regulatory outcomes. BP =Bullous Pemphigoid. CSU = Chronic Spontaneous Urticaria. COPD = Chronic Obstructive Pulmonary Disease. CRSwNP = Chronic Rhinosinusitis with Nasal Polyps. EGID = Eosinophilic Gastrointestinal Disorders. TA PoC achieved Positive Phase 2 Part A data for zumilokibart in AD Positive Phase 1b data for zumilokibart in asthma Positive Phase 3 data for IL-13 inhibition in EoE1 |
| Building a Leading I&I Company Michael Henderson, MD Chief Executive Officer |
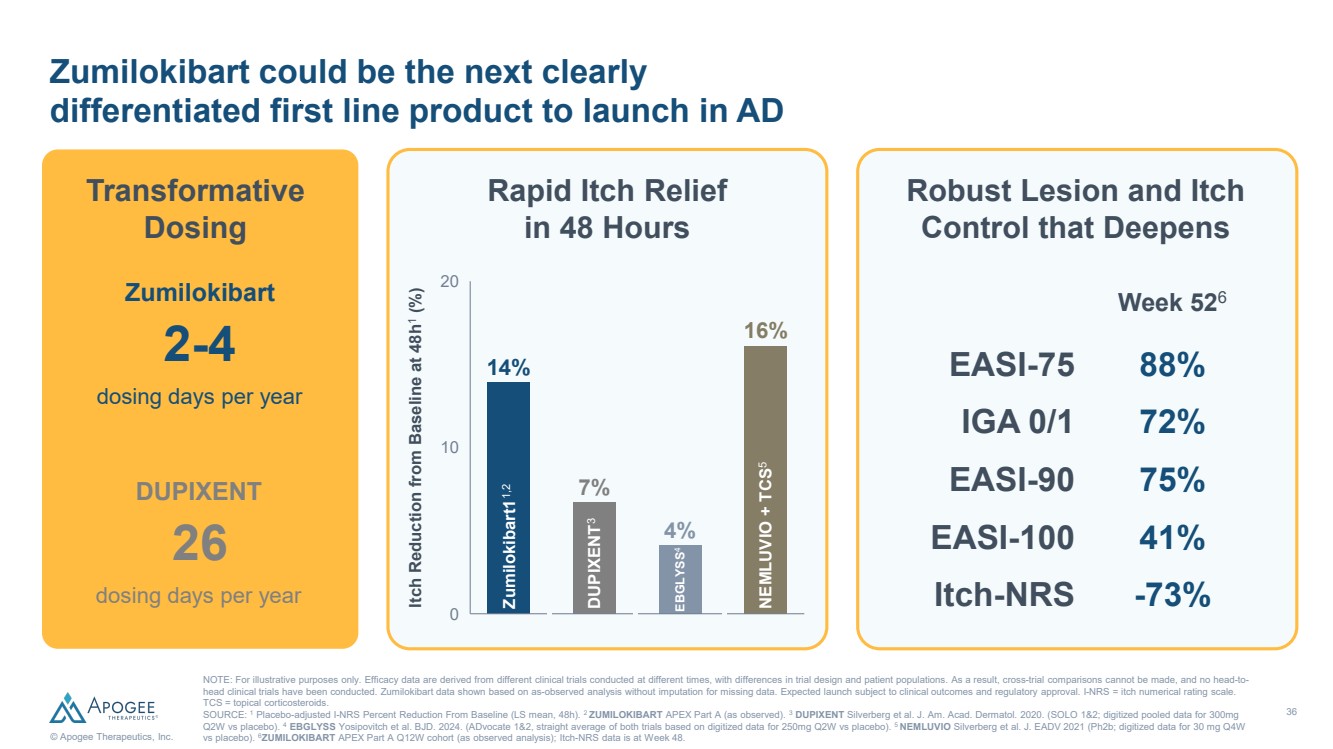
| 36 © Apogee Therapeutics, Inc. Zumilokibart could be the next clearly differentiated first line product to launch in AD Robust Lesion and Itch Control that Deepens Rapid Itch Relief in 48 Hours NOTE: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Zumilokibart data shown based on as-observed analysis without imputation for missing data. Expected launch subject to clinical outcomes and regulatory approval. I-NRS = itch numerical rating scale. TCS = topical corticosteroids. SOURCE: 1 Placebo-adjusted I-NRS Percent Reduction From Baseline (LS mean, 48h). 2 ZUMILOKIBART APEX Part A (as observed). 3 DUPIXENT Silverberg et al. J. Am. Acad. Dermatol. 2020. (SOLO 1&2; digitized pooled data for 300mg Q2W vs placebo). 4 EBGLYSS Yosipovitch et al. BJD. 2024. (ADvocate 1&2, straight average of both trials based on digitized data for 250mg Q2W vs placebo). 5 NEMLUVIO Silverberg et al. J. EADV 2021 (Ph2b; digitized data for 30 mg Q4W vs placebo). 6ZUMILOKIBART APEX Part A Q12W cohort (as observed analysis); Itch-NRS data is at Week 48. Week 526 EASI-75 88% IGA 0/1 72% EASI-90 75% EASI-100 41% Itch-NRS -73% Transformative Dosing 2-4 Zumilokibart dosing days per year 26 DUPIXENT dosing days per year 0 10 20 Itch Reduction from Baseline at 48h1 (%) 14% 7% 4% 16% Zumilokibart11,2 DUPIXENT3 EBGLYSS4 NEMLUVIO + TCS5 |
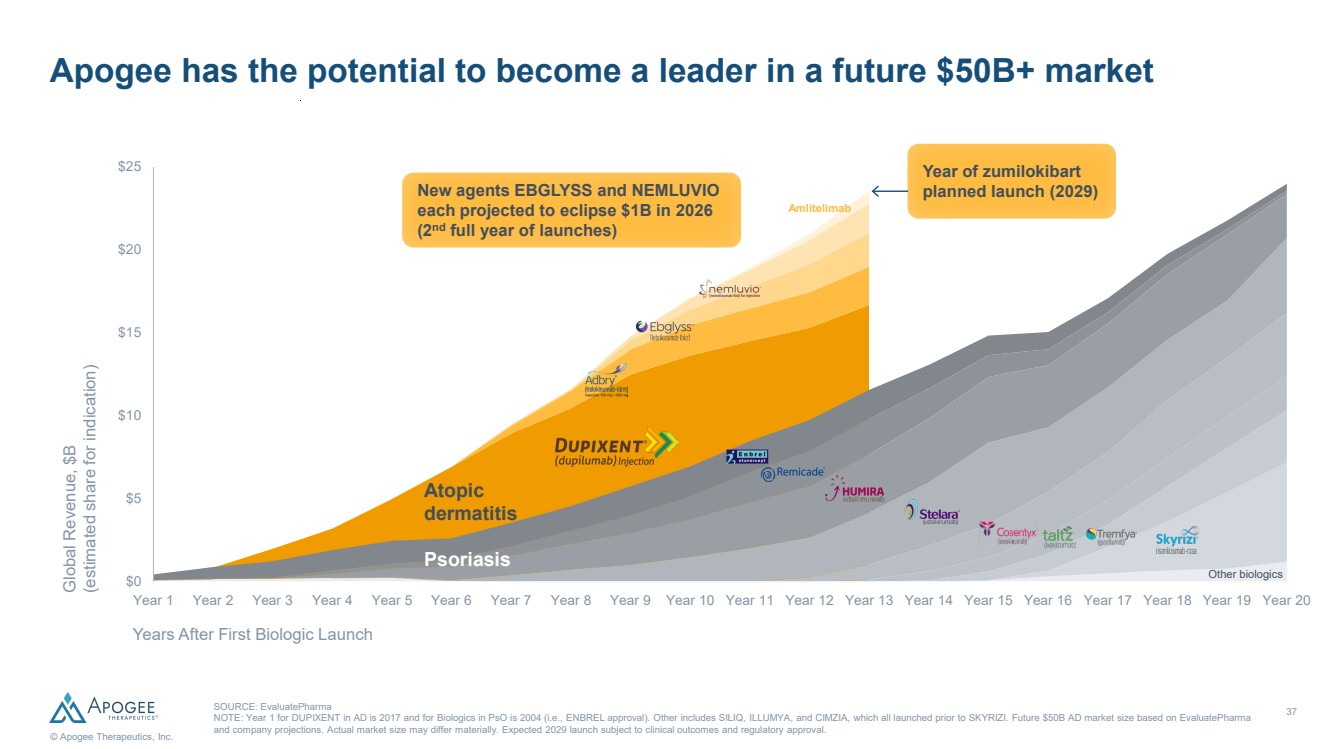
| 37 © Apogee Therapeutics, Inc. $0 $5 $10 $15 $20 $25 Global Revenue, $B (estimated share for indication) Year 1 Year 2 Year 3 Year 4 Year 5 Year 6 Year 7 Year 8 Year 9 Year 10 Year 11 Year 12 Year 13 Year 14 Year 15 Year 16 Year 17 Year 18 Year 19 Year 20 Years After First Biologic Launch SOURCE: EvaluatePharma NOTE: Year 1 for DUPIXENT in AD is 2017 and for Biologics in PsO is 2004 (i.e., ENBREL approval). Other includes SILIQ, ILLUMYA, and CIMZIA, which all launched prior to SKYRIZI. Future $50B AD market size based on EvaluatePharma and company projections. Actual market size may differ materially. Expected 2029 launch subject to clinical outcomes and regulatory approval. $0 $5 $10 $15 $20 $25 Psoriasis Atopic dermatitis Other biologics Year of zumilokibart planned launch (2029) Amlitelimab Apogee has the potential to become a leader in a future $50B+ market New agents EBGLYSS and NEMLUVIO each projected to eclipse $1B in 2026 (2nd full year of launches) |
| 2026 is becoming a transformational year for Apogee Apogee poised for sustained leadership in AD starting with potential zumilokibart launch in 2029 NOTE: 1 Cash, cash equivalents and marketable securities were $902.9 million as of December 31, 2025. 2 APG279 is a combination of zumilokibart and APG990. APG279 is co-administered in the proof-of-concept Phase 1b trial; coformulation planned for future clinical studies and commercialization. Future $50B AD market size based on EvaluatePharma and company projections. Actual market size may differ materially. AD Phase 3 planned initiation is subject to positive Part B clinical results and regulatory alignment. Expected 2029 launch subject to clinical outcomes and regulatory approval. Well-capitalized to deliver key milestones with $903M in cash1 and runway into 2H 2028 © Apogee Therapeutics, Inc. 38 Demonstrated potential best-in-class dosing for zumilokibart in a potential future $50B+ AD market • Q1 2026: APEX Phase 2 Part A 52-week positive data • Q2 2026: APEX Phase 2 Part B 16-week expected readout • 2H 2026: AD Phase 3 planned initiation Optimizing Phase 3 dose to advance zumilokibart into late-stage development • 2H 2026: AD Ph1b expected readout (vs DUPIXENT; fully enrolled) Serial innovation in atopic dermatitis with first-in-class APG279 combination2 Expanding zumilokibart beyond atopic dermatitis • Q1 2026: Asthma Phase 1b positive data • 2H 2026: Expect to announce clinical trial plans in asthma and EoE |
| Apogee /ˈapəjē/ noun The highest point in the development of something; a climax or culmination |
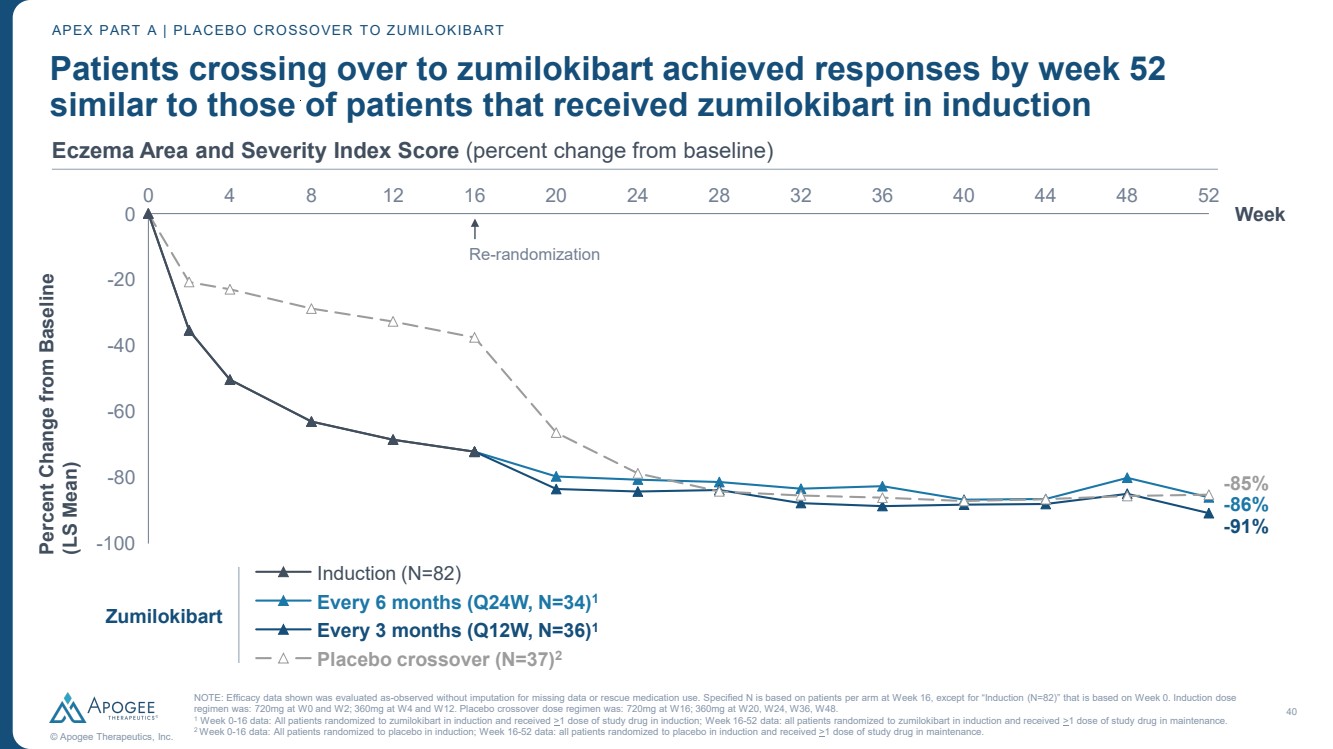
| 40 © Apogee Therapeutics, Inc. Induction (N=82) Every 6 months (Q24W, N=34)1 Every 3 months (Q12W, N=36)1 Placebo crossover (N=37)2 -91% -86% 0 4 8 12 16 20 24 28 32 36 40 44 48 52 -100 -80 -60 -40 -20 0 Week Percent Change from Baseline (LS Mean) Re-randomization Patients crossing over to zumilokibart achieved responses by week 52 similar to those of patients that received zumilokibart in induction NOTE: Efficacy data shown was evaluated as-observed without imputation for missing data or rescue medication use. Specified N is based on patients per arm at Week 16, except for “Induction (N=82)” that is based on Week 0. Induction dose regimen was: 720mg at W0 and W2; 360mg at W4 and W12. Placebo crossover dose regimen was: 720mg at W16; 360mg at W20, W24, W36, W48. 1 Week 0-16 data: All patients randomized to zumilokibart in induction and received >1 dose of study drug in induction; Week 16-52 data: all patients randomized to zumilokibart in induction and received >1 dose of study drug in maintenance. 2 Week 0-16 data: All patients randomized to placebo in induction; Week 16-52 data: all patients randomized to placebo in induction and received >1 dose of study drug in maintenance. APEX PART A | PLACEBO CROSSOVER TO ZUMILOKIBART Zumilokibart -85% Eczema Area and Severity Index Score (percent change from baseline) |