
Exhibit 99.2

Removing Barriers to Effective Neuro - Drug Delivery Corporate Presentation 202 6 NT HI Phase 1/2 Safety and Efficacy of NEO212 in Patients With Astrocytoma IDH - mutant, Glioblastoma IDH - wildtype or Brain Metastasis (NCT06047379)

Important Cautions Regarding Forward Looking Statements © NeOnc Technologies Holdings, Inc. 2022 - 2025. All Rights Reserved. Proprietary. 2 This document does not constitute or form part of an invitation or recommendation to subscribe for or purchase any securities. The distribution may be restricted by law in certain jurisdictions and persons into whose possession this document comes should inform themselves about, and observe, any such restriction. Any failure to comply with these restrictions may constitute a violation of the laws of any such jurisdiction. NeOnc Technologies Holdings, Inc. (“NeOnc”, “Company,” “our” or “ours”) shall not have any responsibility for any such violations. Any decision to purchase or subscribe for securities in any offering must be made solely on the basis of the information issued in connection with such offering. This document was prepared exclusively for the benefit and internal use of investors to evaluate the feasibility of a possible transaction or transactions and does not carry any right of publication or disclosure to any other party. This document is incomplete without reference to and should be viewed solely in conjunction with, the oral briefing provided by the Company. This presentation may not be used for any other purpose without the prior written consent of the Company. In preparing this document, we have relied upon and assumed, without independent verification, the accuracy and completeness of all information available from public sources or which was provided to us or otherwise reviewed by us. We do not represent that such information is accurate or complete, and it should not be relied on as such. Any opinions expressed herein reflect our judgment at this date, all of which are accordingly subject to change. We have based the forward - looking statements on our current expectations and projections about future events. These forward - looking statements are subject to known and unknown risks, uncertainties, and assumptions about us and our affiliate companies with respect to specific factors identified in this presentation and the Company’s filings with the U.S. Securities Exchange Commission, and which may cause our actual results, levels of activity, performance or achievements expressed or implied by such forward - looking statements differ materially from those currently anticipated in such statements. This presentation has been prepared by the Company for informational purposes only and not for any other purpose. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction. THIS PRESENTATION AND ANY OTHER INFORMATION THAT MAY BE FURNISHED BY US INCLUDES OR MAY INCLUDE CERTAIN STATEMENTS, ESTIMATES AND FORWARD - LOOKING PROJECTIONS OF THE COMPANY WITH RESPECT TO THE ANTICIPATED FUTURE PERFORMANCE OF THE COMPANY. SUCH STATEMENTS, ESTIMATES AND FORWARD - LOOKING PROJECTIONS REFLECT VARIOUS ASSUMPTIONS OF MANAGEMENT THAT MAY OR MAY NOT PROVE TO BE CORRECT AND INVOLVE VARIOUS RISKS AND UNCERTAINTIES.

Senior Management Team Board of Directors Scientific Advisory Board
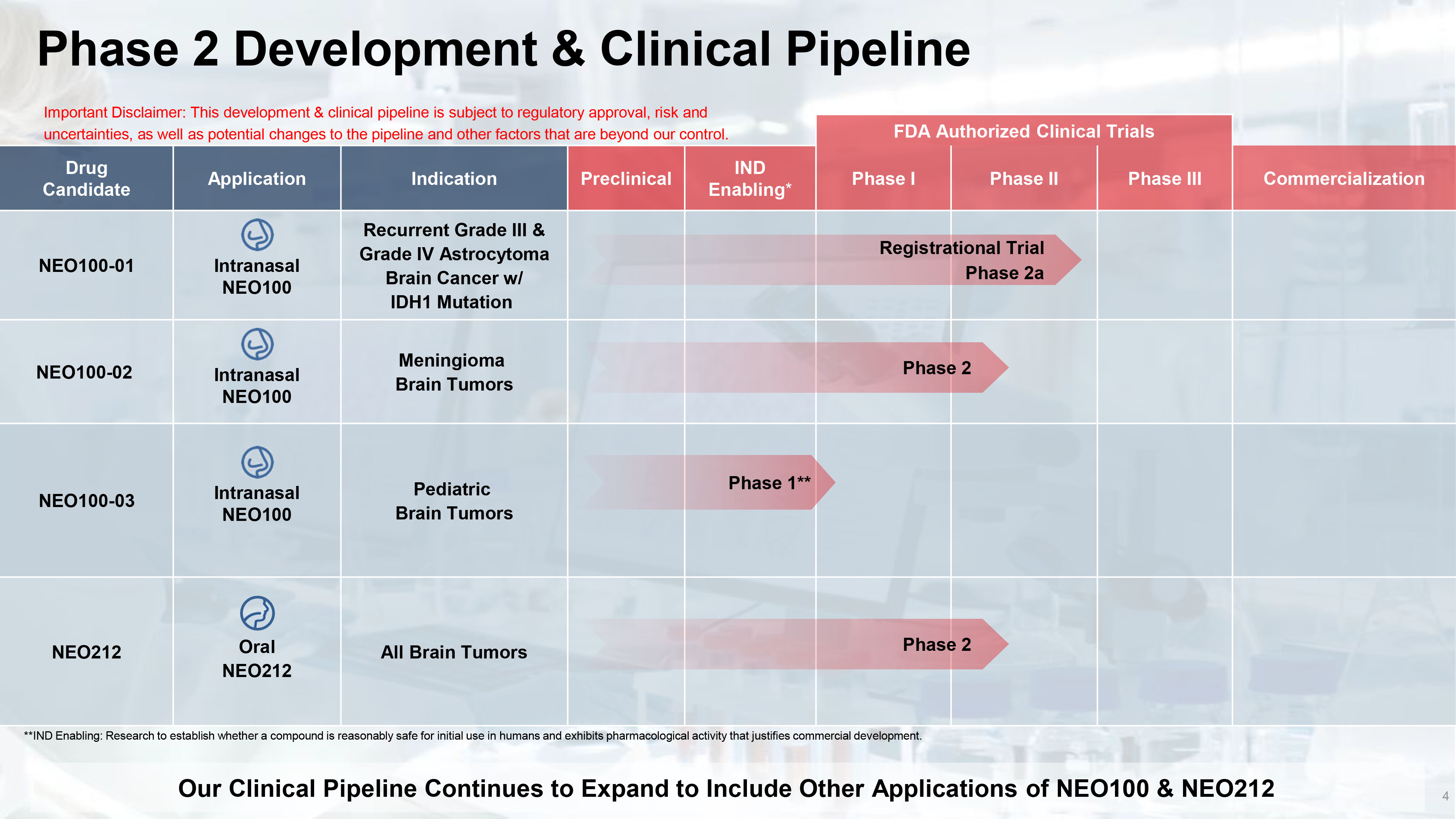
Phase 2 Development & Clinical Pipeline 4 FDA Authorized Clinical Trials Commercialization Phase III Phase II Phase I IND Enabling * Preclinical Indication Application Drug Candidate Recurrent Grade III & Grade IV Astrocytoma Brain Cancer w/ IDH1 Mutation Intranasal NEO100 NEO100 - 01 Meningioma Brain Tumors Intranasal NEO100 NEO100 - 02 Pediatric Brain Tumors Intranasal NEO100 NEO100 - 03 All Brain Tumors Oral NEO212 NEO212 Phase 2 Phase 1** Our Clinical Pipeline Continues to Expand to Include Other Applications of NEO100 & NEO212 Important Disclaimer: This development & clinical pipeline is subject to regulatory approval, risk and uncertainties, as well as potential changes to the pipeline and other factors that are beyond our control. Phase 2 Registrational Trial Phase 2a **IND Enabling: Research to establish whether a compound is reasonably safe for initial use in humans and exhibits pharmacolo gic al activity that justifies commercial development.
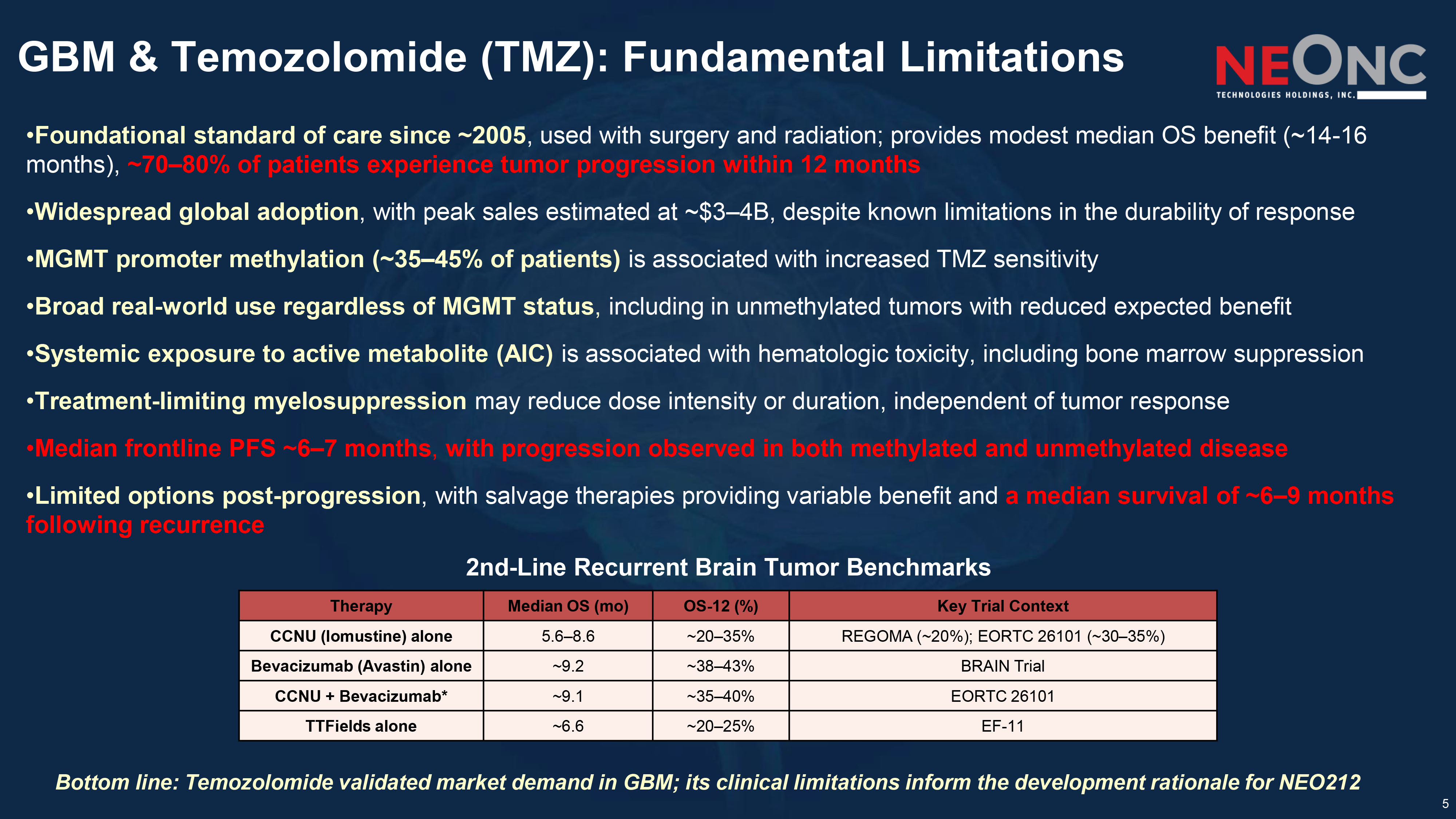
5 GBM & Temozolomide (TMZ): Fundamental Limitations Bottom line: Temozolomide validated market demand in GBM; its clinical limitations inform the development rationale for NEO212 2nd - Line Recurrent Brain Tumor Benchmarks • Foundational standard of care since ~2005 , used with surgery and radiation; provides modest median OS benefit (~14 - 16 months), ~70 – 80% of patients experience tumor progression within 12 months • Widespread global adoption , with peak sales estimated at ~$3 – 4B, despite known limitations in the durability of response • MGMT promoter methylation (~35 – 45% of patients) is associated with increased TMZ sensitivity • Broad real - world use regardless of MGMT status , including in unmethylated tumors with reduced expected benefit • Systemic exposure to active metabolite (AIC) is associated with hematologic toxicity, including bone marrow suppression • Treatment - limiting myelosuppression may reduce dose intensity or duration, independent of tumor response • Median frontline PFS ~6 – 7 months , with progression observed in both methylated and unmethylated disease • Limited options post - progression , with salvage therapies providing variable benefit and a median survival of ~6 – 9 months following recurrence Key Trial Context OS - 12 (%) Median OS ( mo ) Therapy REGOMA (~20%); EORTC 26101 (~30 – 35%) ~20 – 35% 5.6 – 8.6 CCNU ( lomustine ) alone BRAIN Trial ~38 – 43% ~9.2 Bevacizumab (Avastin) alone EORTC 26101 ~35 – 40% ~9.1 CCNU + Bevacizumab* EF - 11 ~20 – 25% ~6.6 TTFields alone

NEO212: Brain - Optimized Temozolomide Bioconjugate Designed to Enhance Brain Tumor Targeting NEO100 Temozolomide (TMZ) Carbamide Bond NEO212 Bioconjugated Molecule 6 Novel bioconjugate combining NEO 100 with temozolomide (TMZ) . Builds on TMZ, the established GBM standard of care, with the goal of improving brain delivery and durability of response . Enhanced BBB penetration targets brain tumors more effectively than TMZ Dual - mechanism : Complementary activity from NEO 100 and TMZ in one molecule . Bioconjugation optimizes PK and CNS exposure Clinically ready for studies in primary and secondary malignant brain cancers, including GBM and brain metastases . Multiple delivery routes planned, with evaluation of both oral and intranasal administration .
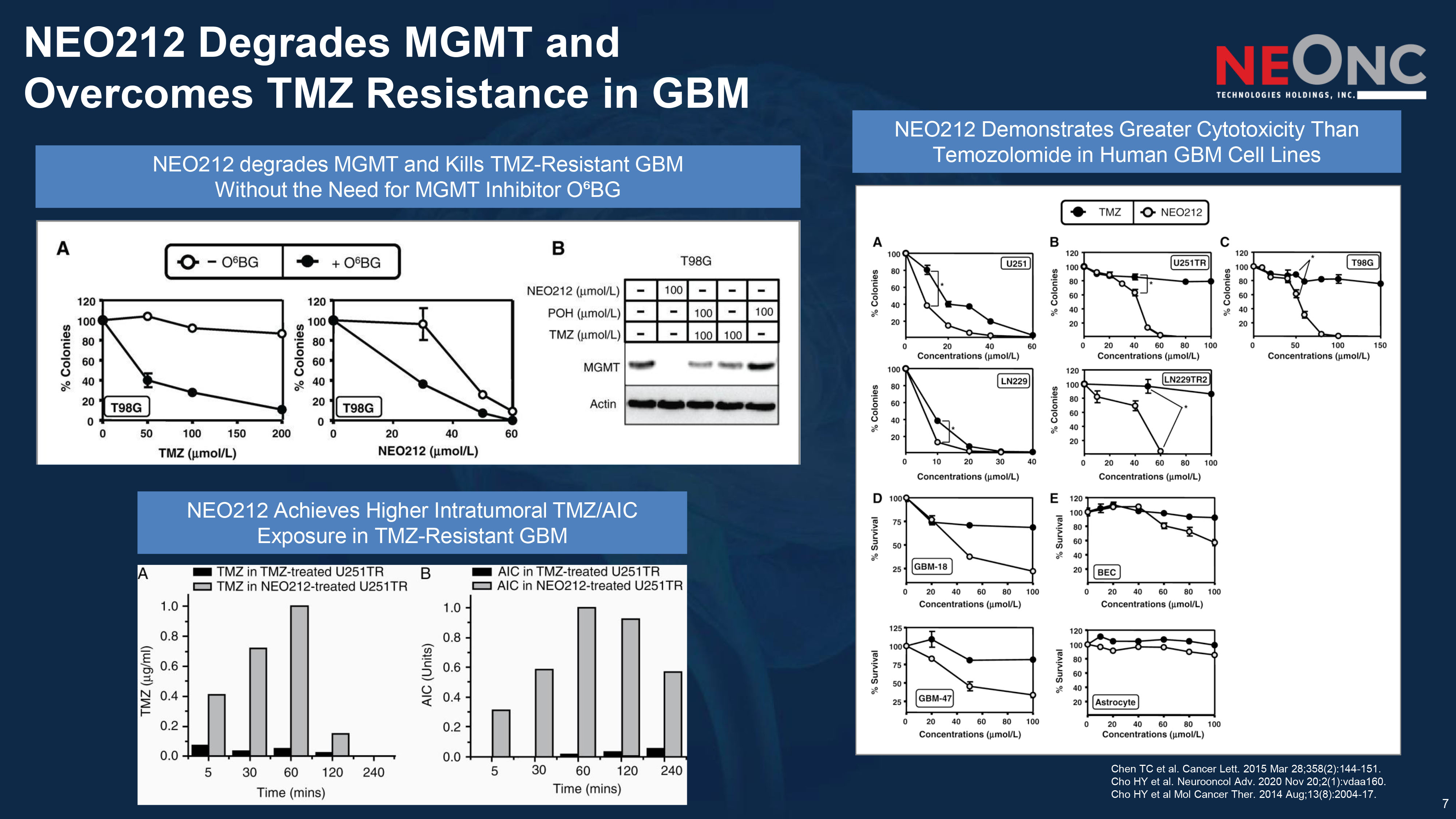
7 NEO212 Degrades MGMT and Overcomes TMZ Resistance in GBM Chen TC et al. Cancer Lett. 2015 Mar 28;358(2):144 - 151. Cho HY et al. Neurooncol Adv. 2020 Nov 20;2(1):vdaa160. Cho HY et al Mol Cancer Ther. 2014 Aug;13(8):2004 - 17. NEO212 Achieves Higher Intratumoral TMZ/AIC Exposure in TMZ - Resistant GBM NEO212 degrades MGMT and Kills TMZ - Resistant GBM Without the Need for MGMT Inhibitor O⁶BG NEO212 Demonstrates Greater Cytotoxicity Than Temozolomide in Human GBM Cell Lines
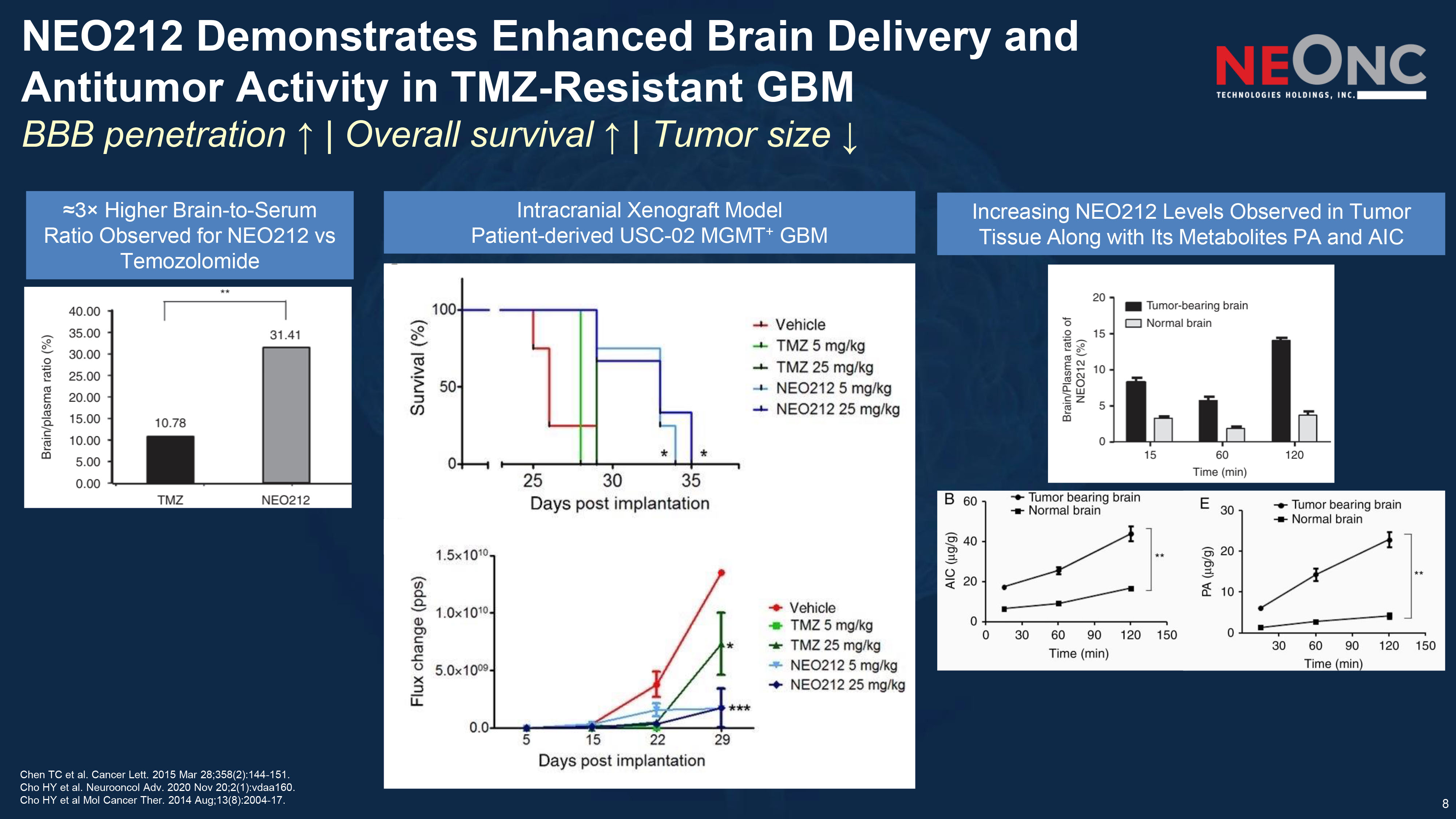
8 NEO212 Demonstrates Enhanced Brain Delivery and Antitumor Activity in TMZ - Resistant GBM BBB penetration ↑ | Overall survival ↑ | Tumor size ↓ Chen TC et al. Cancer Lett. 2015 Mar 28;358(2):144 - 151. Cho HY et al. Neurooncol Adv. 2020 Nov 20;2(1):vdaa160. Cho HY et al Mol Cancer Ther. 2014 Aug;13(8):2004 - 17. Intracranial Xenograft Model Patient - derived USC - 02 MGMT + GBM ≈3 î Higher Brain - to - Serum Ratio Observed for NEO212 vs Temozolomide Increasing NEO212 Levels Observed in Tumor Tissue Along with Its Metabolites PA and AIC
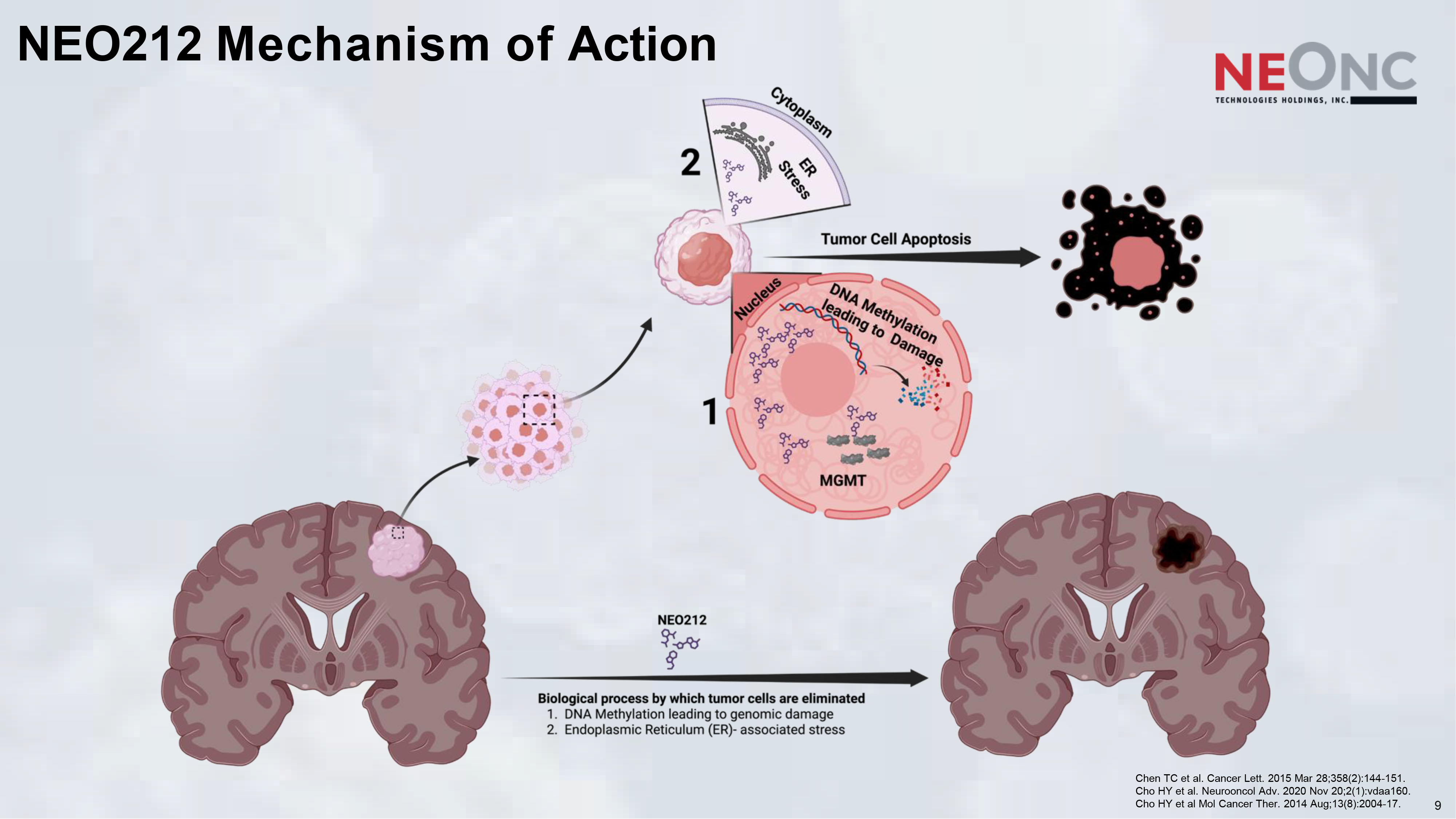
NEO212 Mechanism of Action 9 Chen TC et al. Cancer Lett. 2015 Mar 28;358(2):144 - 151. Cho HY et al. Neurooncol Adv. 2020 Nov 20;2(1):vdaa160. Cho HY et al Mol Cancer Ther. 2014 Aug;13(8):2004 - 17.

NEO212: Novel Alkylating Therapy with Preclinical Evidence of MGMT Inactivation and Brain Penetration in Clinical Development for Brain Tumors 10 Broad Scope: Primary brain tumors and brain metastases as monotherapy and in combination with SOC Differentiation vs TMZ: Up to 10 î greater preclinical efficacy , including MGMT - positive (TMZ - resistant) tumors with NEO212 - Mediated MGMT Inactivation Safety Advantage: Lower systemic toxicity and myelosuppression vs TMZ Brain Delivery: 3 î higher BBB penetration (brain: serum ratio) vs TMZ Phase I (completed): Assessing safety, tolerability, and preliminary efficacy in primary brain tumors and solid - tumor brain metastases Strategic Opportunity: Designed to replace or augment TMZ across primary and metastatic brain cancer indications

Phase 1: Safety and Efficacy of NEO212 in Patients With Astrocytoma IDH - mutant, Glioblastoma IDH - wildtype or Brain Metastasis Phase 1: (dose escalation) Primary Objectives : • Assess the safety and tolerability of increasing dose levels of orally administered NEO212 alone in patients with Astrocyto ma IDH - mutant, Glioblastoma IDH - wildtype or patients with select solid tumors with uncontrolled metastases to the brain. • Identify the maximum tolerated dose (MTD) of NEO212. • Determine the recommended Phase 2 dose (RP2D) of NEO212. Secondary Objectives: • Characterize the pharmacokinetics (PK) of NEO212. • Evaluate anti - tumor activity of NEO212 in patients with Astrocytoma IDH - mutant, Glioblastoma IDH - wildtype and patients with select solid tumors with uncontrolled metastases to the brain. Total Subjects MGMT Status Tumor Type 3 N=1, Not applicable Squamous NSCLC - to - Brain Metastasis Cohort 1 (170mg/day) N=2 Methylated GBM IDH1 - wildtype 3 N=2 Methylated, N=1 Unmethylated GBM IDH1 - wildtype Cohort 2 (220mg/day) 3 N=1, Not applicable Esthesioneuroblastoma - to - Brain Metastasis Cohort 3 (400mg/day) N=1, Not applicable Breast - to - Brain Metastasis N=1, Methylated GBM IDH1 - wildtype 3 N=1 Equivocal1, N=1 Unmethylated, N=1 Methylated GBM IDH1 - wildtype Cohort 4 (610mg/day) 2 N=2 Methylated GBM IDH1 - wildtype Cohort 5 (810mg/day) NEO212 Phase 1 – Patient Distribution by Cohort Subjects received NEO212 orally once daily on Days 1 through 5 of each 28 - day treatment cycle.
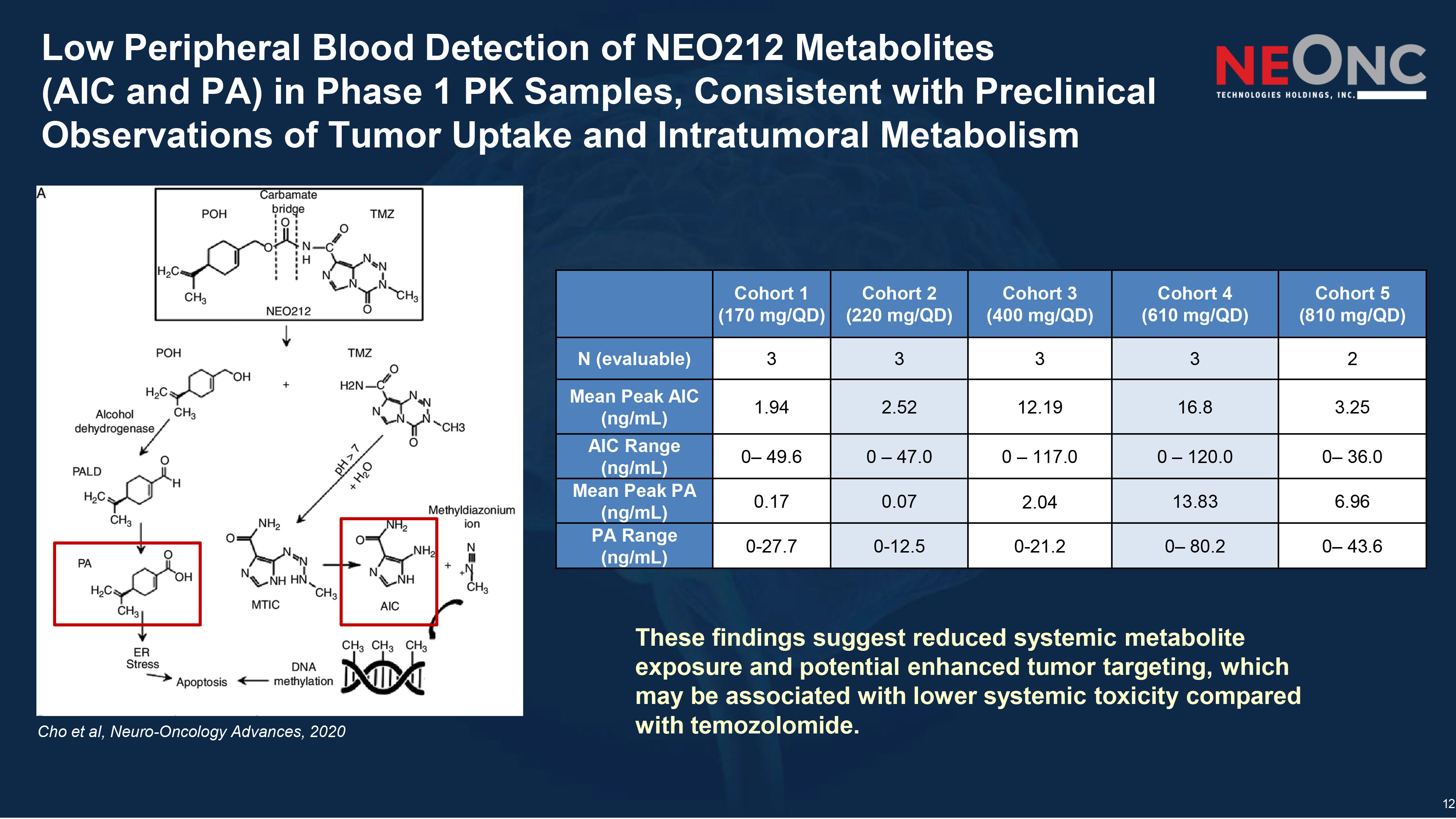
12 Low Peripheral Blood Detection of NEO212 Metabolites (AIC and PA) in Phase 1 PK Samples, Consistent with Preclinical Observations of Tumor Uptake and Intratumoral Metabolism Cohort 5 (810 mg/QD) Cohort 4 (610 mg/QD) Cohort 3 (400 mg/QD) Cohort 2 (220 mg/QD) Cohort 1 (170 mg/QD) 2 3 3 3 3 N (evaluable) 3.25 16.8 12.19 2.52 1.94 Mean Peak AIC (ng/mL) 0 – 36.0 0 – 120.0 0 – 117.0 0 – 47.0 0 – 49.6 AIC Range (ng/mL) 6.96 13.83 2.04 0.07 0.17 Mean Peak PA (ng/mL) 0 – 43.6 0 – 80.2 0 - 21.2 0 - 12.5 0 - 27.7 PA Range (ng/mL) Cho et al, Neuro - Oncology Advances, 2020 These findings suggest reduced systemic metabolite exposure and potential enhanced tumor targeting, which may be associated with lower systemic toxicity compared with temozolomide.
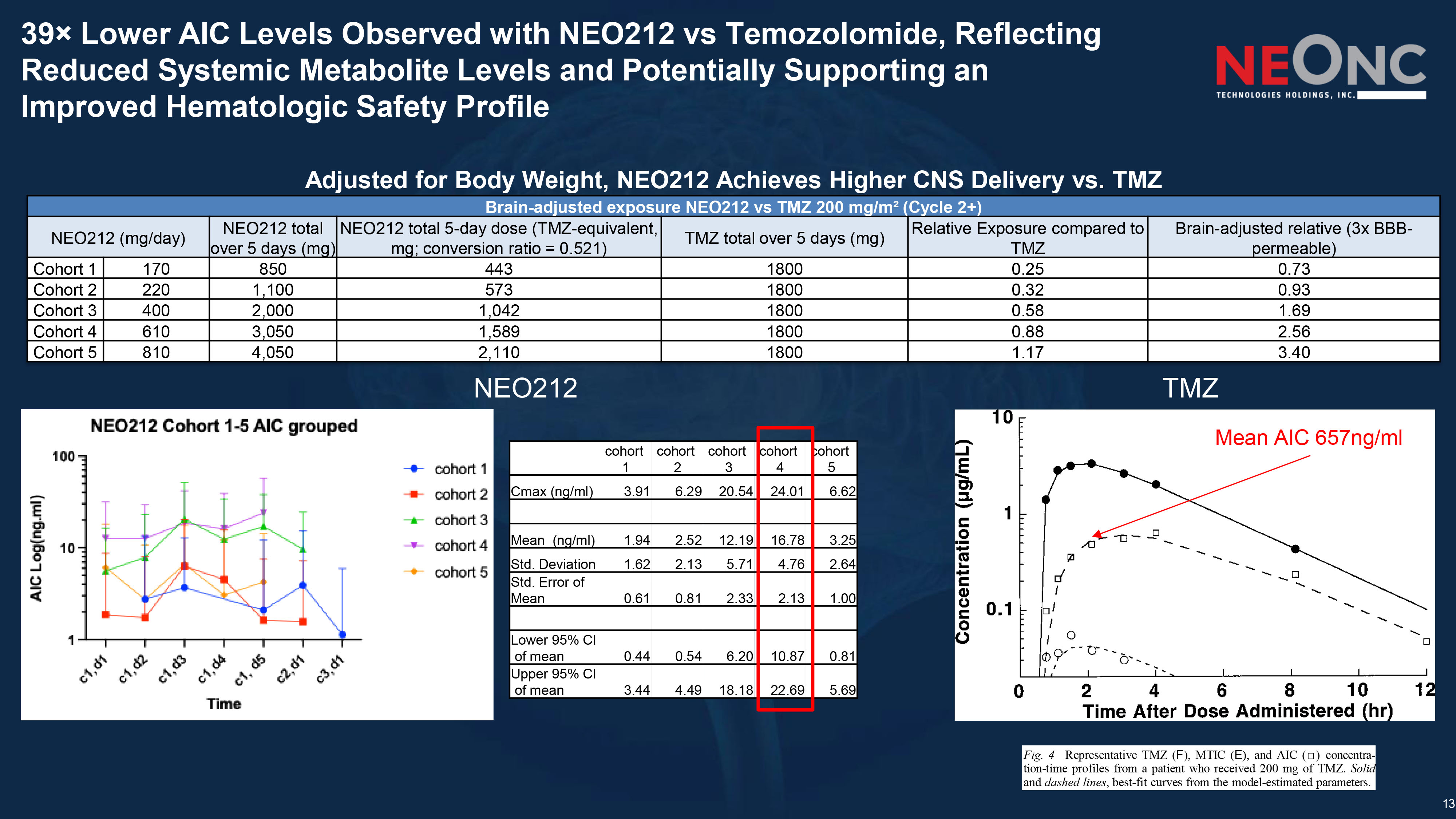
13 39 î Lower AIC Levels Observed with NEO212 vs Temozolomide, Reflecting Reduced Systemic Metabolite Levels and Potentially Supporting an Improved Hematologic Safety Profile Fig . 4 Representative TMZ ( F ), MTIC ( E ), and AIC ( Ƒ ) concentra - tion - time profiles from a patient who received 200 mg of TMZ . Solid and dashed lines , best - fit curves from the model - estimated parameters . NEO212 TMZ Brain - adjusted exposure NEO212 vs TMZ 200 mg/m² (Cycle 2+) Brain - adjusted relative (3x BBB - permeable) Relative Exposure compared to TMZ TMZ total over 5 days (mg) NEO212 total 5 - day dose (TMZ - equivalent, mg; conversion ratio = 0.521) NEO212 total over 5 days (mg) NEO212 (mg/day) 0.73 0.25 1800 443 850 170 Cohort 1 0.93 0.32 1800 573 1,100 220 Cohort 2 1.69 0.58 1800 1,042 2,000 400 Cohort 3 2.56 0.88 1800 1,589 3,050 610 Cohort 4 3.40 1.17 1800 2,110 4,050 810 Cohort 5 cohort 5 cohort 4 cohort 3 cohort 2 cohort 1 6.62 24.01 20.54 6.29 3.91 Cmax (ng/ml) 3.25 16.78 12.19 2.52 1.94 Mean (ng/ml) 2.64 4.76 5.71 2.13 1.62 Std. Deviation 1.00 2.13 2.33 0.81 0.61 Std. Error of Mean 0.81 10.87 6.20 0.54 0.44 Lower 95% CI of mean 5.69 22.69 18.18 4.49 3.44 Upper 95% CI of mean Mean AIC 657ng/ml Adjusted for Body Weight, NEO212 Achieves Higher CNS Delivery vs. TMZ
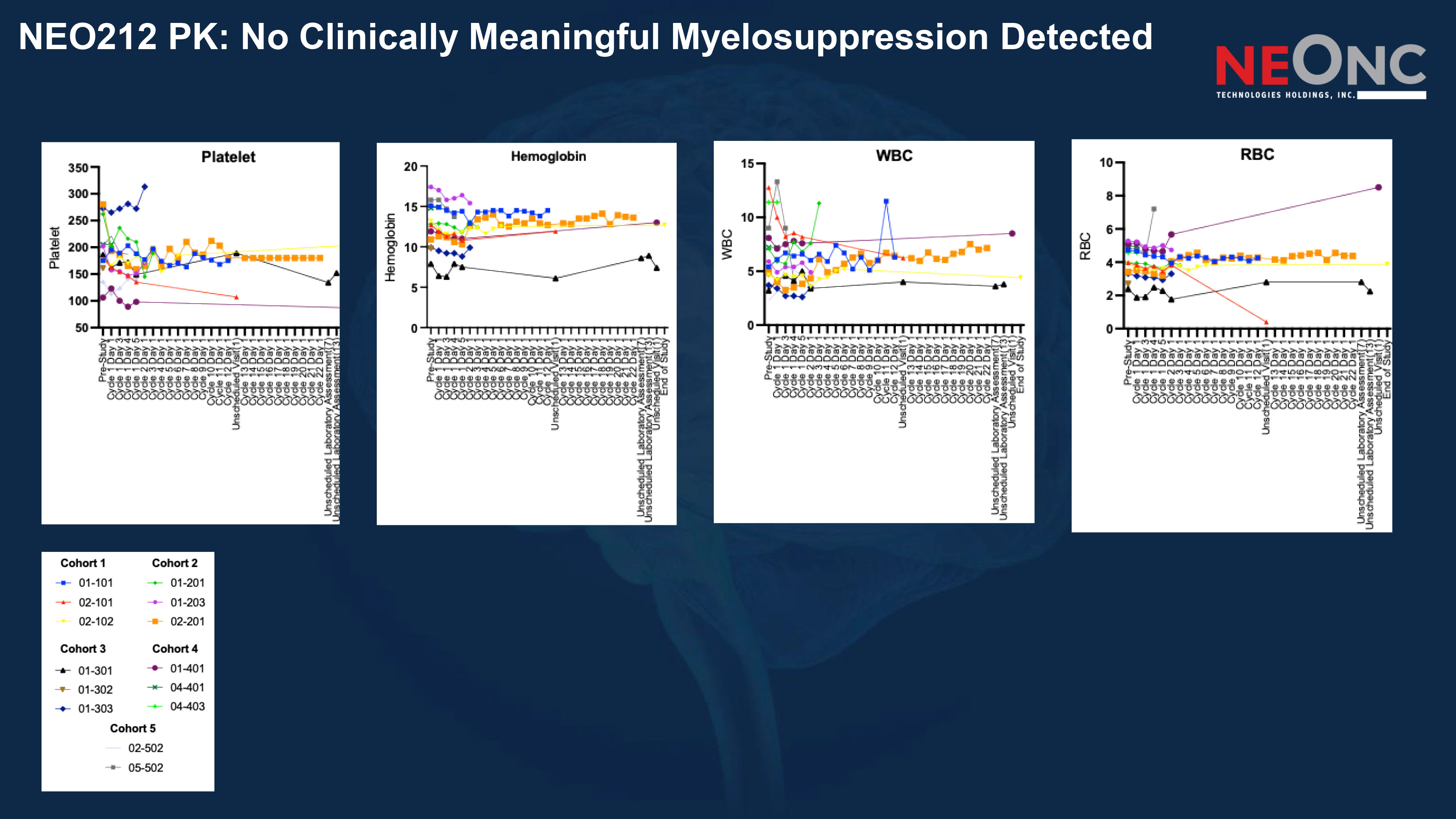
NEO212 PK: No Clinically Meaningful Myelosuppression Detected
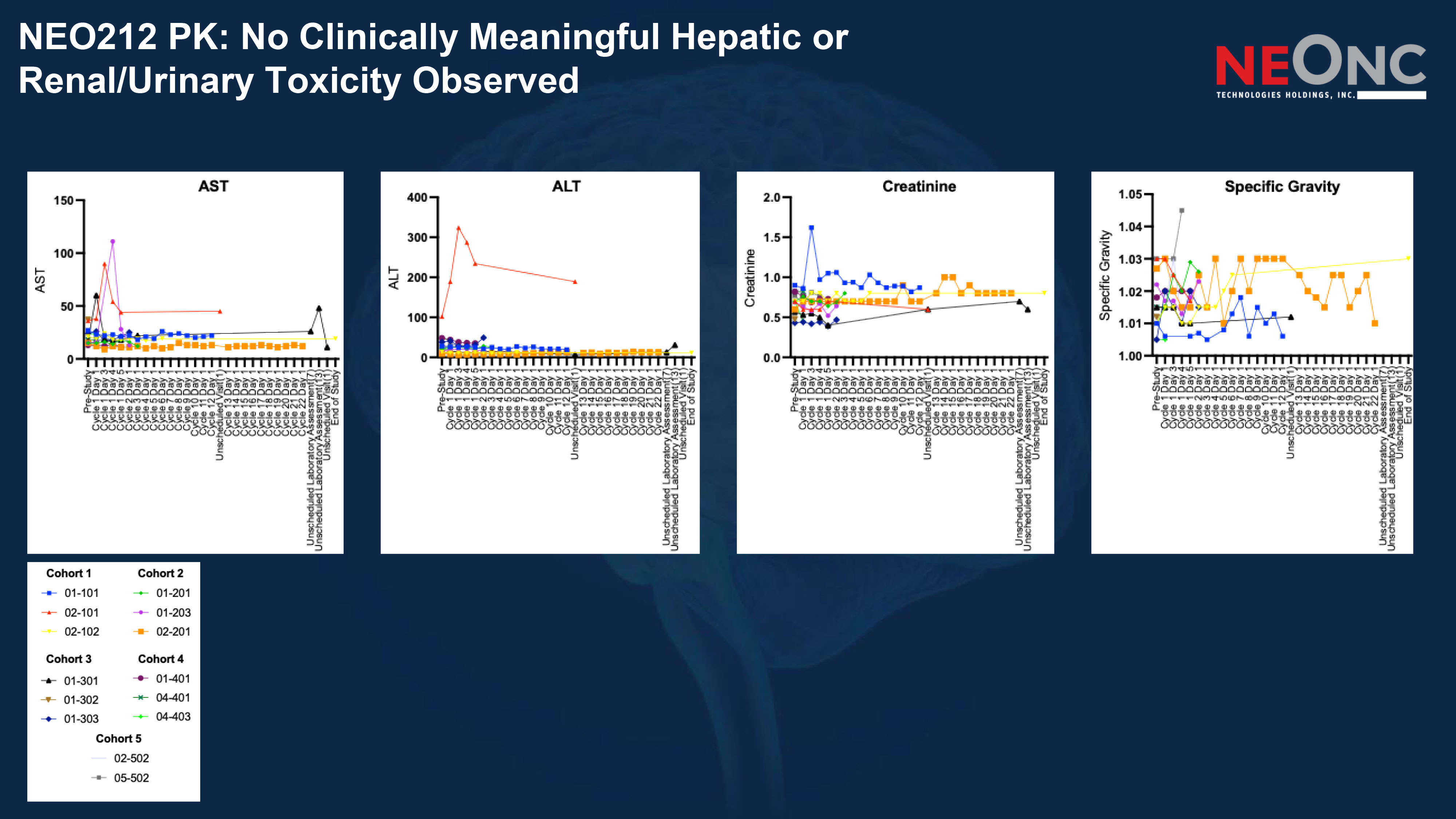
NEO212 PK: No Clinically Meaningful Hepatic or Renal/Urinary Toxicity Observed
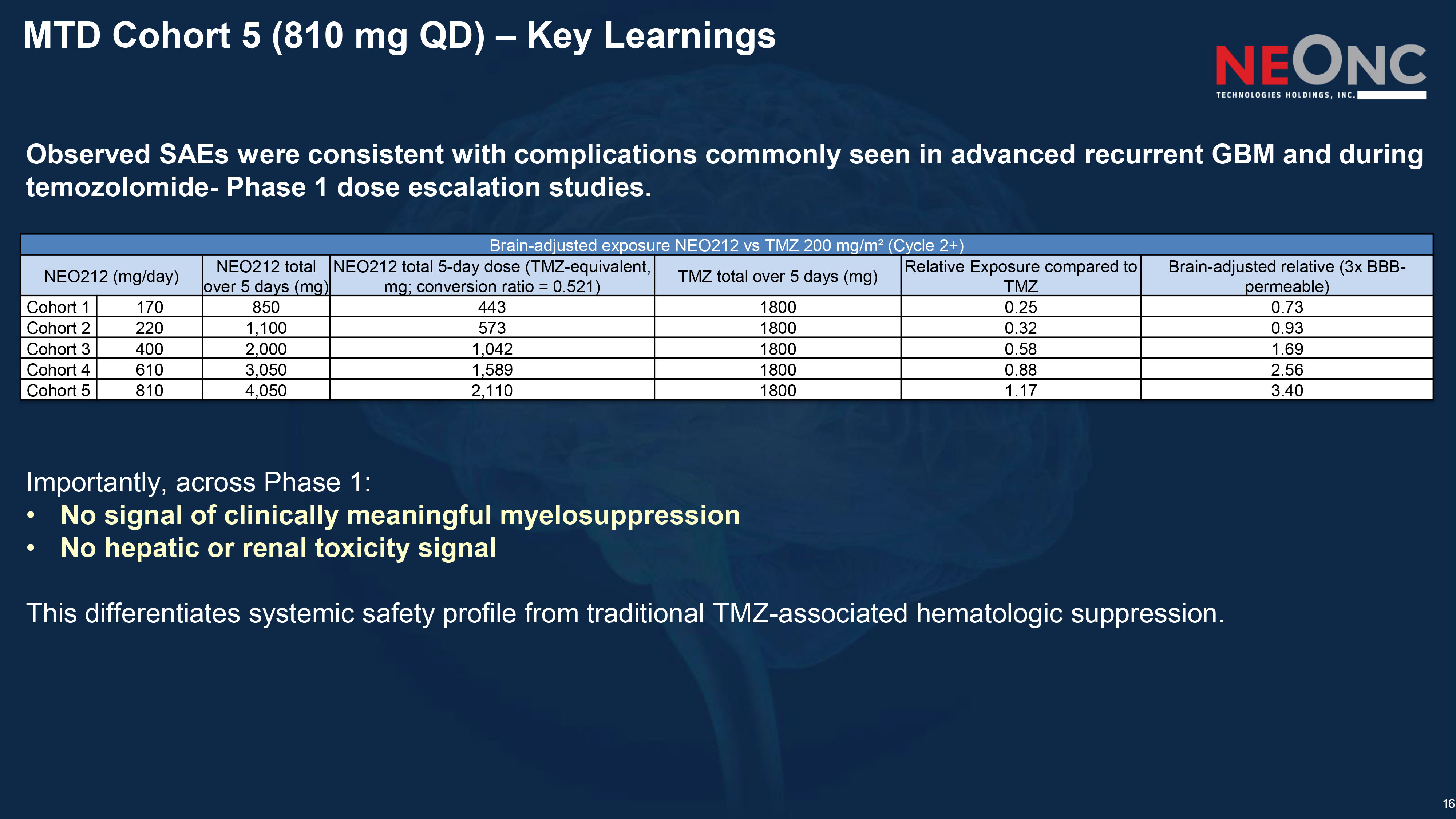
16 MTD Cohort 5 (810 mg QD) – Key Learnings Observed SAEs were consistent with complications commonly seen in advanced recurrent GBM and during temozolomide - Phase 1 dose escalation studies. Importantly, across Phase 1: • No signal of clinically meaningful myelosuppression • No hepatic or renal toxicity signal This differentiates systemic safety profile from traditional TMZ - associated hematologic suppression. Brain - adjusted exposure NEO212 vs TMZ 200 mg/m² (Cycle 2+) Brain - adjusted relative (3x BBB - permeable) Relative Exposure compared to TMZ TMZ total over 5 days (mg) NEO212 total 5 - day dose (TMZ - equivalent, mg; conversion ratio = 0.521) NEO212 total over 5 days (mg) NEO212 (mg/day) 0.73 0.25 1800 443 850 170 Cohort 1 0.93 0.32 1800 573 1,100 220 Cohort 2 1.69 0.58 1800 1,042 2,000 400 Cohort 3 2.56 0.88 1800 1,589 3,050 610 Cohort 4 3.40 1.17 1800 2,110 4,050 810 Cohort 5

17 NEO212 measurable anti - tumor activity in Phase 1
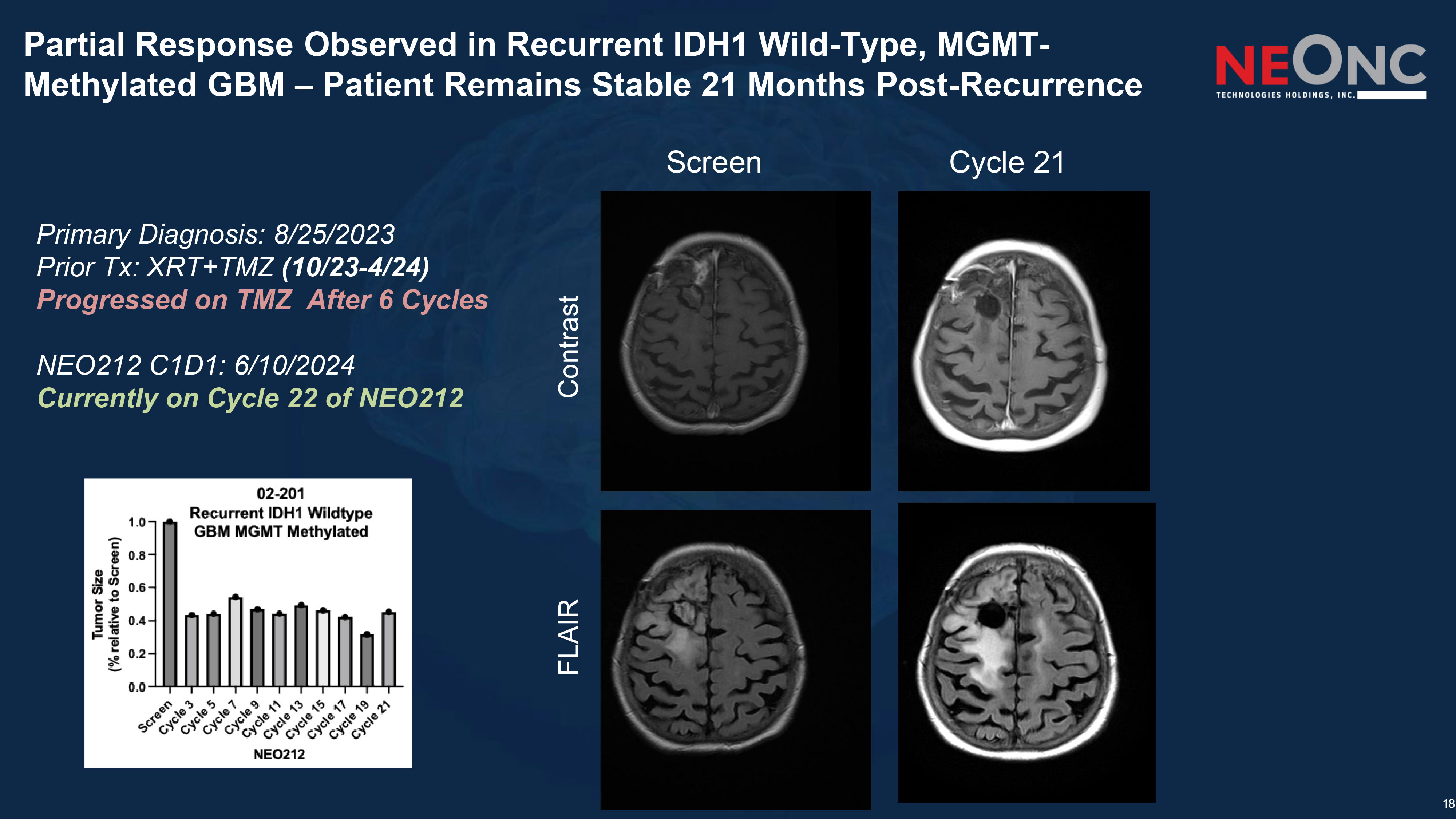
18 Partial Response Observed in Recurrent IDH1 Wild - Type, MGMT - Methylated GBM – Patient Remains Stable 21 Months Post - Recurrence Screen Cycle 21 Contrast FLAIR Primary Diagnosis: 8/25/2023 Prior Tx: XRT+TMZ (10/23 - 4/24) Progressed on TMZ After 6 Cycles NEO212 C1D1: 6/10/2024 Currently on Cycle 22 of NEO212
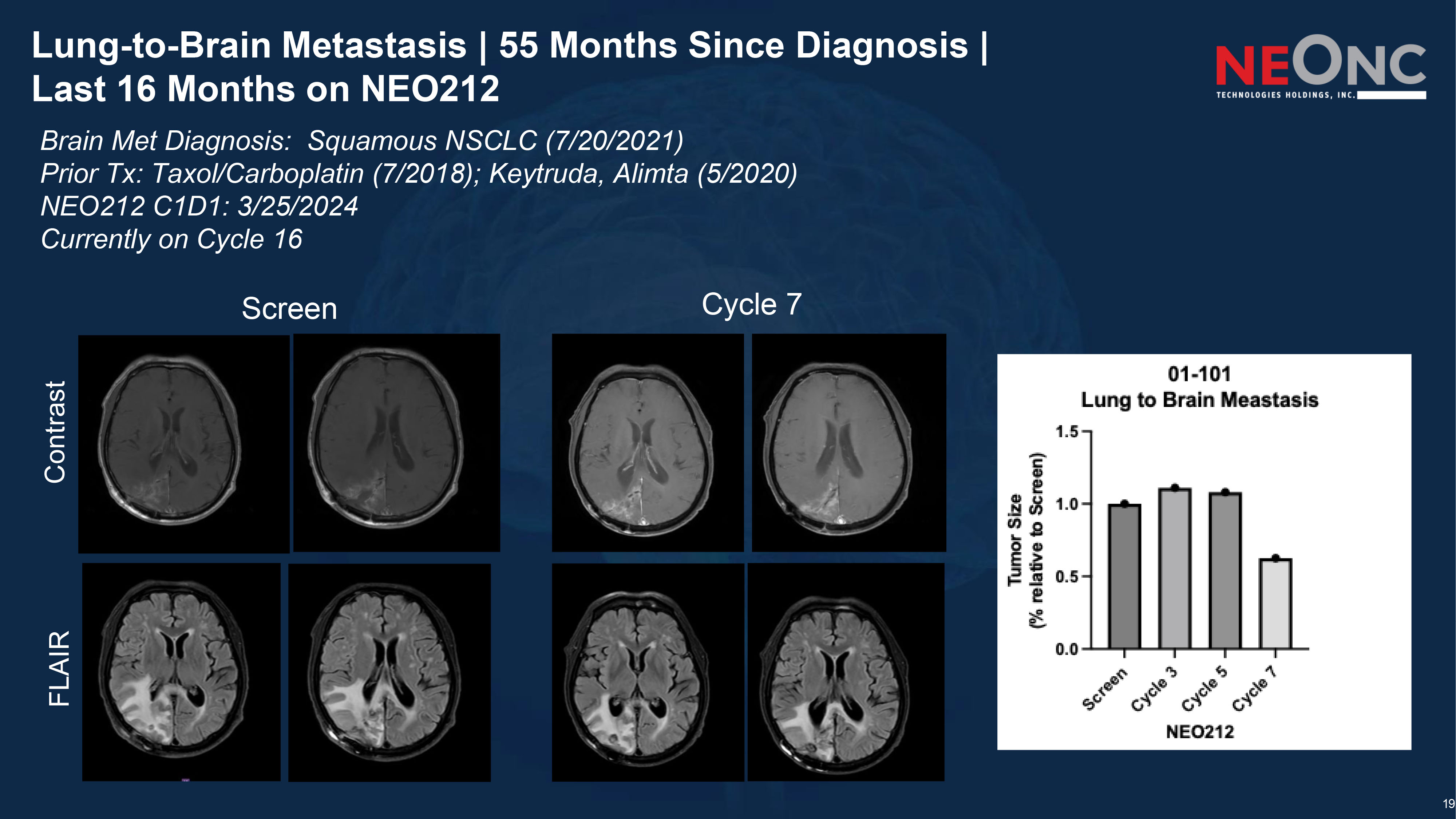
19 Lung - to - Brain Metastasis | 55 Months Since Diagnosis | Last 16 Months on NEO212 Screen Cycle 7 Contrast FLAIR Brain Met Diagnosis: Squamous NSCLC (7/20/2021) Prior Tx: Taxol/Carboplatin (7/2018); Keytruda, Alimta (5/2020) NEO212 C1D1: 3/25/2024 Currently on Cycle 16

20 NEO212 — Next Potential Steps & Accelerated Regulatory Approval Phase I Readout: Initial safety, PK, and preliminary efficacy data Accelerated Approval Strategy: Advancement into Two Phase II Clinical Trials •Trial 1: Randomized evaluation of single - agent NEO212 in recurrent GBM versus standard - of - care therapy •Trial 2: Randomized evaluation of NEO212 in combination with standard - of - care therapy versus standard - of - care therapy alone in patients with brain metastases Unmet Need (Brain Mets): >30% of adult cancer patients develop brain metastases with no durable systemic standard of care TAM — Primary GBM: ~$3.0 - 4.0B estimated market opportunity TAM — Brain Metastases: Approximately 10x the size of the primary GBM market

NEO212: Phase 1 Clinical Data Informing an Accelerated Phase 2 Development Strategy in TMZ - Resistant Glioblastoma and Brain Metastases 1. Safety Foundation Established • Phase 1 demonstrated favorable systemic tolerability Why It Matters: Temozolomide’s dose ceiling is bone marrow suppression. NEO212 may enable sustained alkylator therapy without the traditional hematologic limitation. 2. Early Signals of Clinical Activity in Recurrent GBM • Durable disease control observed in heavily pretreated patients • Biological rationale supported by activity in TMZ - resistant, MGMT - high preclinical models • Designed to enhance brain delivery while preserving cytotoxic potency Why It Matters: ~70 – 80% of GBM patients progress within 12 months of TMZ. NEO212 is positioned to retain activity where TMZ fails. 3. Phase 2 Targets the Highest - Value Unmet Segment • Focus on MGMT - unmethylated and TMZ - refractory patients • Includes patients who discontinued TMZ due to hematologic toxicity • Randomized vs SOC for clean efficacy signal • ORR primary endpoint built for Accelerated Approval Why It Matters: Phase 2 directly tests NEO212 in the population most underserved by current standard therapy — resistance - driven and toxicity - limited patients.

THE FUTURE OF DRUG DELIVERY Company Contact NeOnc Technologies Holdings, Inc. 23975 Park Sorrento , Suite 205 Calabasas, CA 91302 Tel +1 (818) 470 - NTHI Info@neonc.com www.neonc.com Investor Relations Hayden IR 2369 Lexington Avenue , Second Floor New York, New York 10017 Brett Maas Managing Partner brett@haydenir.com James Carbonara Partner James@haydenir.com 646 - 755 - 7412 Contact Us 22